Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
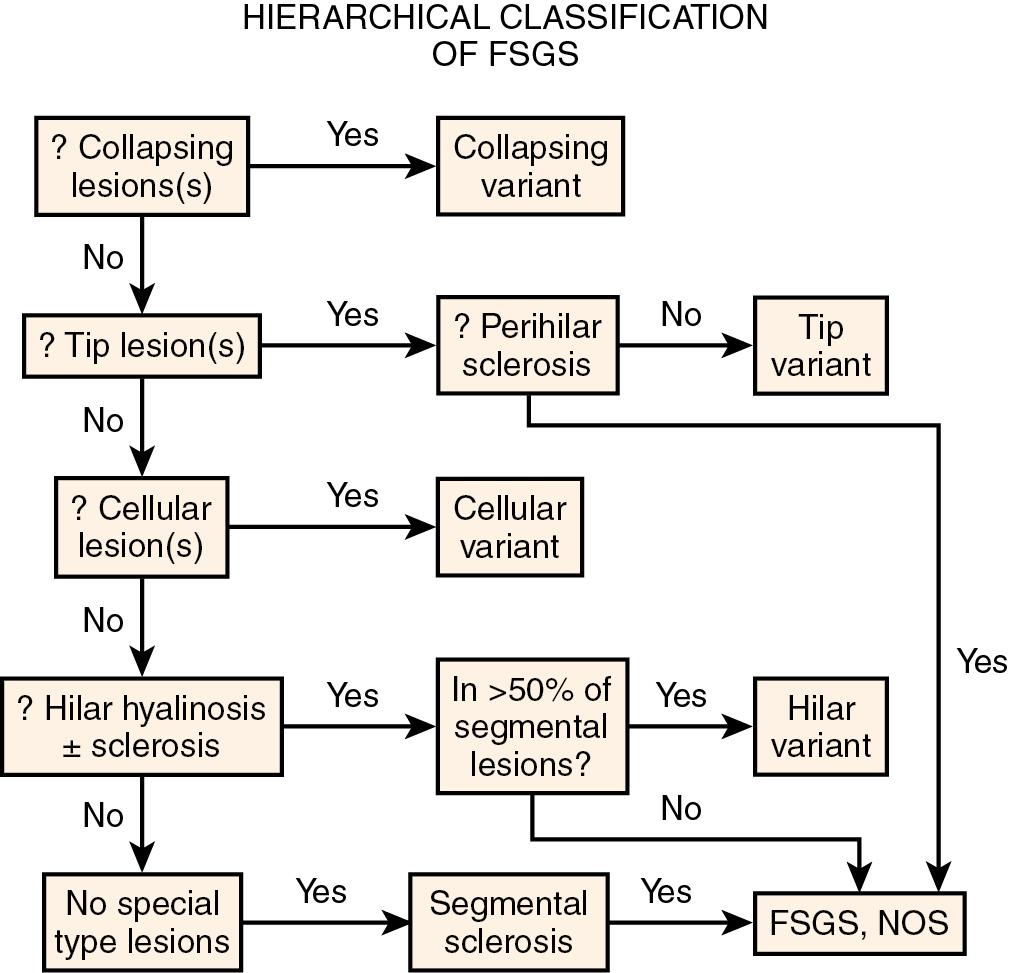
Minimal change disease (MCD) and focal segmental glomerulosclerosis (FSGS) both typically present as nephrotic syndrome and cannot be readily distinguished based solely on clinical presentation. Approximately 90% of children ages 1 to 7 years with nephrotic syndrome appear to have MCD, and the vast majority respond to steroids. Thus biopsy is only done if the child is unresponsive to steroids or has clinical features suggesting another etiology of the nephrotic syndrome. Among the minority of nephrotic children who were steroid unresponsive/dependent or had frequent relapses and therefore underwent kidney biopsy, FSGS was diagnosed in about 7%. In biopsied adolescents, FSGS is observed more commonly than MCD. In adults, MCD accounts for 10% to 15% of nephrotic syndrome. The term “FSGS” is unfortunately used both for a disease with primary injury to podocytes that appears mediated by a circulating factor and for any glomerular scarring lesion that occurs in a focal and segmental distribution. Primary podocytopathy-related FSGS has increased in incidence, and in the United States in adults has surpassed membranous nephropathy as a cause of nephrotic syndrome (18.7% incidence), especially in African Americans and Hispanics. Similar increased incidence of FSGS linked to ethnicity has also been reported in children with nephrotic syndrome. The incidence of FSGS is less in older adults. Serologic studies, including complement levels, are typically within normal limits in both MCD and FSGS. Renal biopsy is thus essential to determine the etiology of nephrotic syndrome in adults and also in children who are not steroid responders. The ultimate prognosis differs dramatically, with complete recovery the rule in MCD, contrasting with progressive renal insufficiency in FSGS. Several morphologic variants of FSGS have also been investigated for their prognostic significance. Of note, the morphologic subtypes are not specific for a primary etiology of FSGS lesions. The impact of these morphologic variants on prognosis was investigated in the National Institute of Health (NIH) FSGS Clinical Trials cohort of 138 patients with steroid-resistant primary FSGS, treated with mycophenolate mofetil and dexamethasone versus cyclosporine, with no difference in response between treatment groups. The morphologic variants of FSGS, however, were associated with differences in treatment responses, with worst outcome in those with collapsing variant and best in those with tip variant, with intermediate results for those with FSGS not otherwise specified (NOS). The Columbia working classification proposal is given in Table 3.1 and the hierarchical relationship of the variants is shown in Fig. 3.1 . Each of the subtypes will be discussed later.
| Type | Defining Feature |
|---|---|
| FSGS, not otherwise specified | Discrete segmental sclerosis |
| FSGS, perihilar variant | Perihilar sclerosis and hyalinosis |
| FSGS, cellular variant | Endocapillary hypercellularity |
| FSGS, tip variant | Sclerosis at tubular pole with adhesion at tubular lumen/neck |
| FSGS, collapsing variant (collapsing glomerulopathy) | Segmental or global collapse of tuft and visceral epithelial cell hyperplasia/hypertrophy |
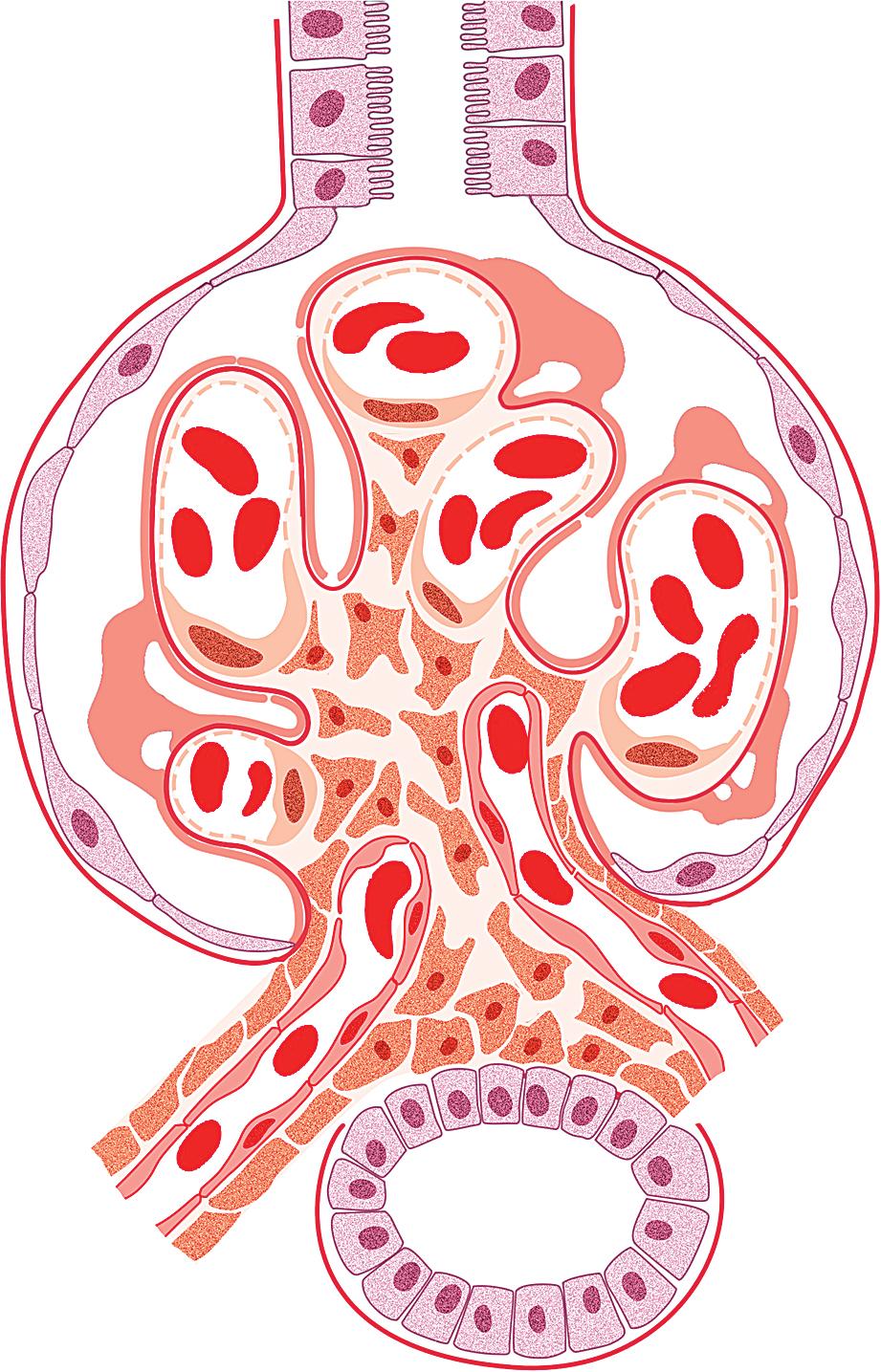
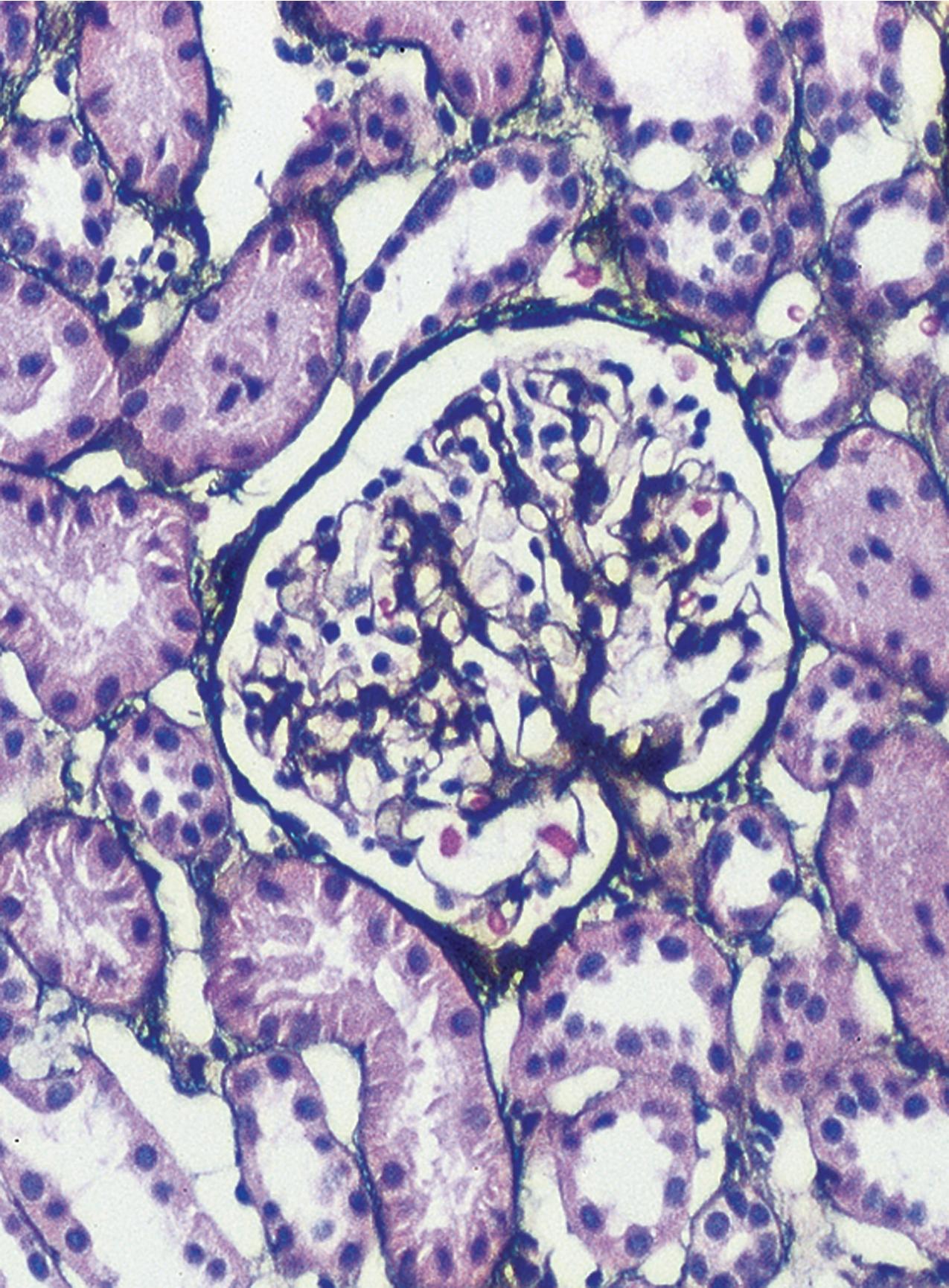
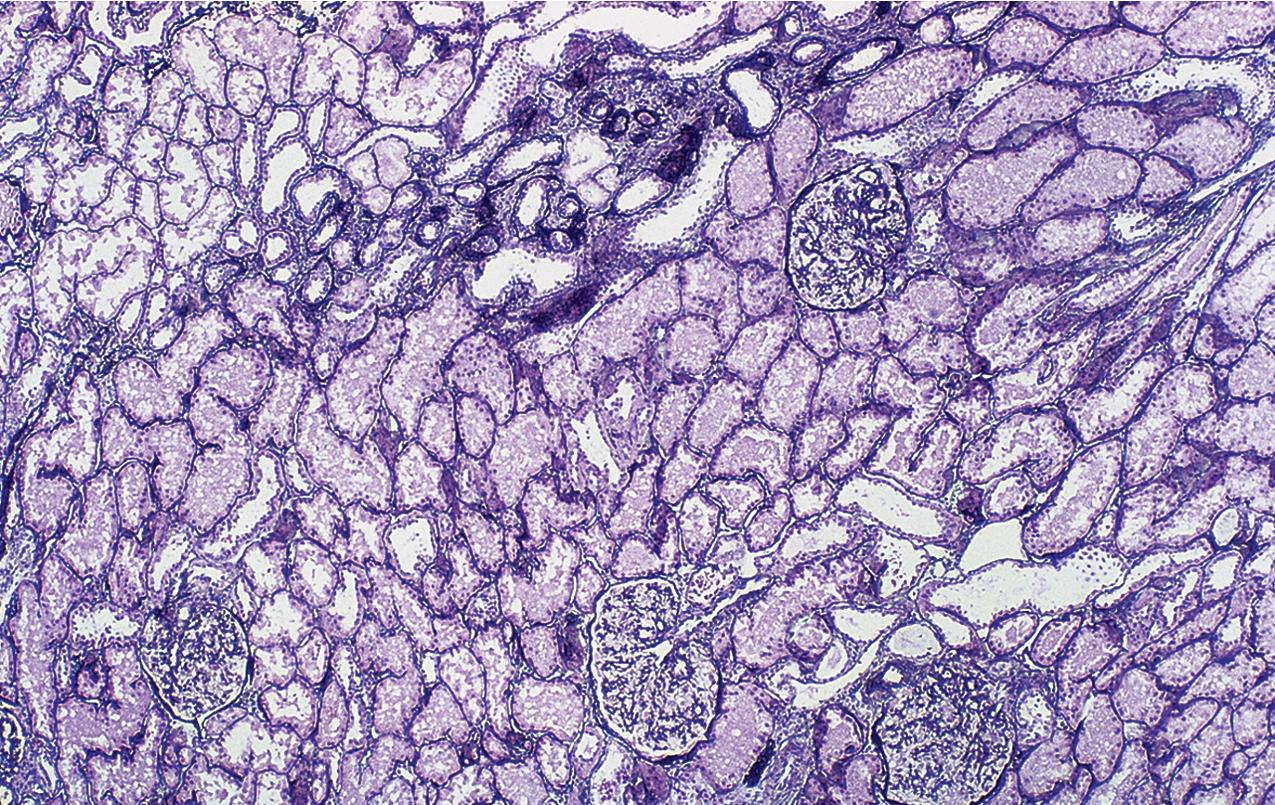
MCD is named for the apparent structurally normal glomeruli by light microscopy ( Figs. 3.2–3.3 ). There are no specific vascular or tubulointerstitial lesions in idiopathic MCD. Nevertheless, MCD may also occur in the middle-aged or older adult (10%–15% of nephrotic syndrome in adults) who has nonspecific focal areas of tubulointerstitial scarring and mild vascular lesions (arteriosclerosis, arteriolar hyaline related to hypertension, or other unrelated disease). Global glomerulosclerosis, in contrast to the segmental glomerular sclerotic lesion, is not of special diagnostic significance in considering the differential of MCD versus FSGS. Globally sclerotic glomeruli may be normally seen at any age, show an obsolescent pattern (see also “Age-Related Sclerosis,” in Chapter 7 ), and are thought to result from normal “wear and tear” and not specific disease mechanisms in most cases. Up to 10% of glomeruli may be normally globally sclerosed in people younger than 40 years of age. The extent of global sclerosis increases with aging, up to 30% by age 80 (estimated by calculating half the patient’s age minus 10).
Glomerular MCD-type lesions with associated acute interstitial nephritis (AIN), which is characterized by edema and interstitial lymphoplasmacytic infiltrate, often with eosinophils, suggest a drug-induced hypersensitivity reaction. This combined syndrome of MCD and AIN is classically because of nonsteroidal antiinflammatory drugs (NSAIDs). This condition is usually reversible with discontinuation of the drug.
Immunofluorescence (IF) studies are typically negative in MCD. The presence of immunoglobulin M (IgM) staining in otherwise apparent MCD biopsies has been a source of previous controversy, with some authors considering this a specific entity known as “IgM nephropathy” (see later).
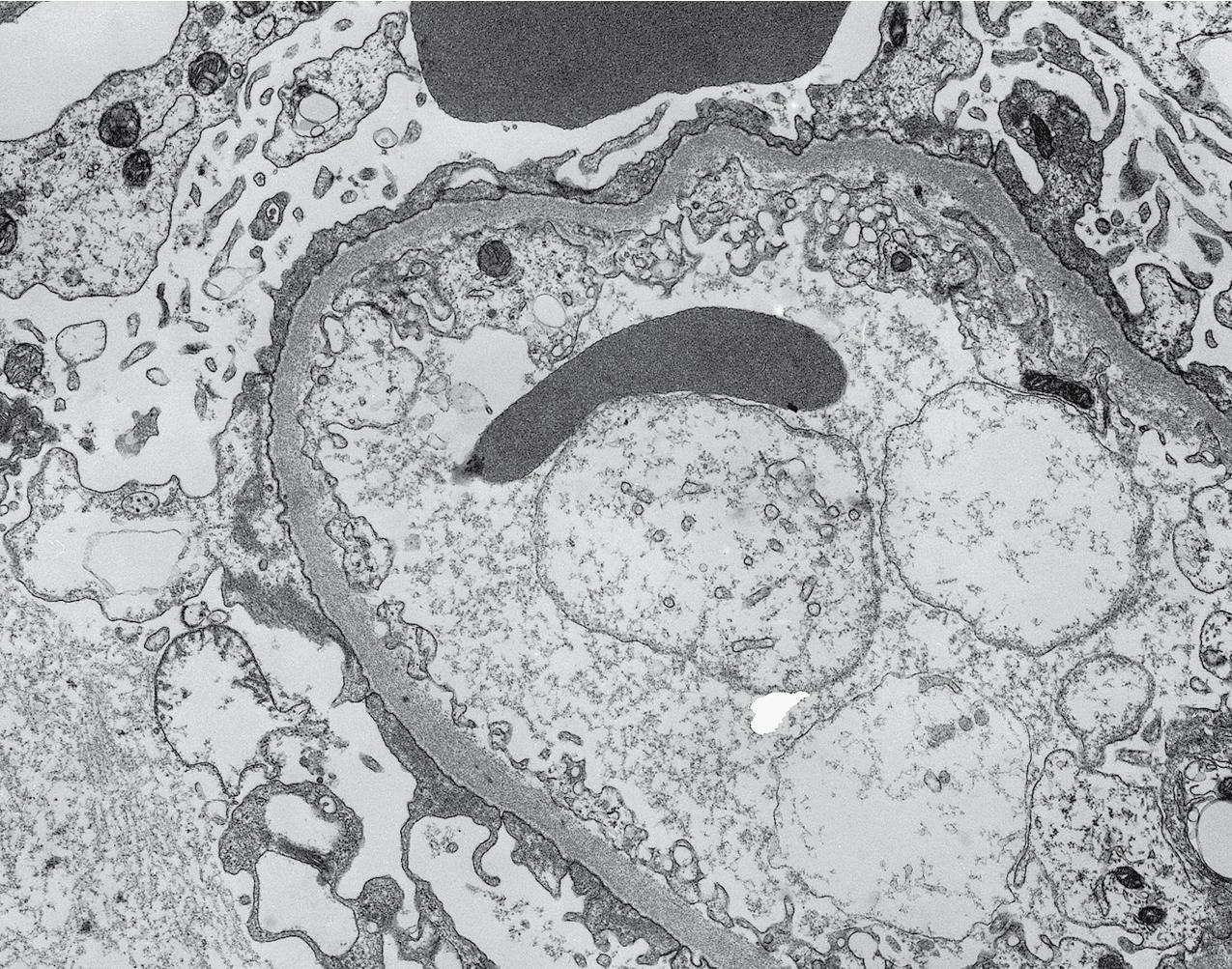
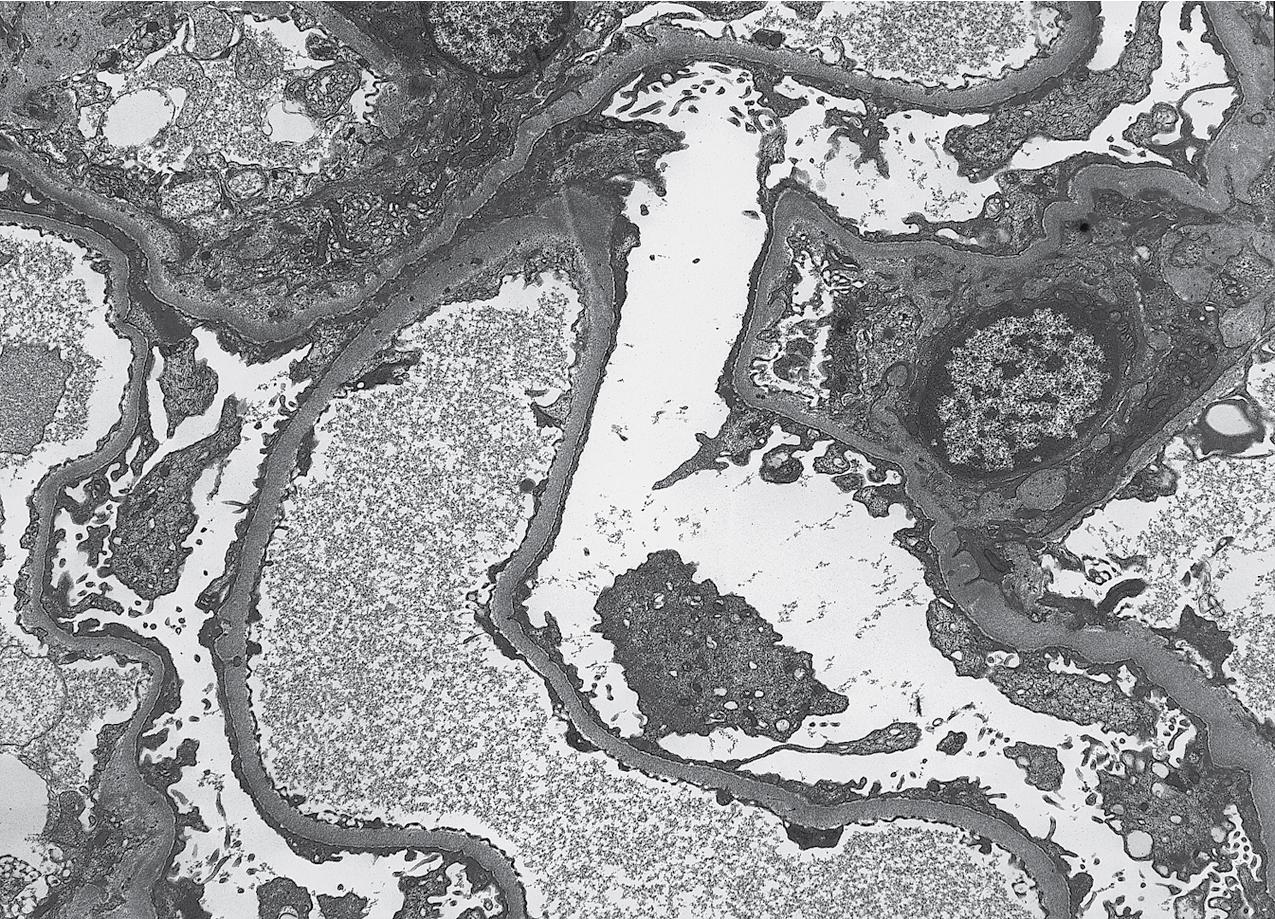
Electron microscopy (EM) shows extensive foot process effacement, vacuolization, and microvillous transformation of podocytes in MCD ( Figs. 3.4–3.5 ). Patients who have been treated with immunosuppression with partial or complete response before biopsy may show less or even no significant foot process effacement.
The pathogenesis of MCD appears related to interactions of abnormal cytokines and podocytes that only affect glomerular permeability and do not promote sclerogenic mechanisms. Dysregulated interaction of T-cells via cytotoxic T lymphocyte antigen-4 (CTLA-4) with podocyte CD80 has been postulated, but not proven, to contribute to MCD. T helper type 2 cells may release interleukin (IL)-13, which in experimental studies increased CD80 on podocytes with foot process effacement and proteinuria ensuing. MCD has been associated with drug-induced hypersensitivity reactions, and can be triggered by, for example, NSAIDs (see earlier). MCD also has been associated with Hodgkin’s lymphoma, bee stings, and other venom exposure and after viral or, rarely, other infection or atopic episodes, implicating immune dysfunction as an initiating factor. In most cases, the triggers for initial disease or relapses remain unknown. In some patients with lupus nephritis (LN) or IgA nephropathy (IgAN; see specific sections), limited mesangial deposits are associated with extensive foot process effacement and marked proteinuria, possibly representing an MCD-like injury related to the underlying immune complexes, or less likely, coincidental occurrence of two separate diseases.
D’Agati VD, Kaskel FJ, Falk RJ. Focal segmental glomerulosclerosis. N Engl J Med. 2011;365:2398–2411.
Fogo AB. Causes and pathogenesis of focal segmental glomerulosclerosis. Nat Rev Nephrol. 2015;11:76–87.
Gulati S, Sharma AP, Sharma RK. et al. Changing trends of histopathology in childhood nephrotic syndrome. Am J Kidney Dis. 1999;3:646–650.
Kaneko K, Tsuji S, Kimata T, Kitao T, Yamanouchi S, Kato S. Pathogenesis of childhood idiopathic nephrotic syndrome: A paradigm shift from T-cells to podocytes. World J Pediatr. 2015;11:21–28.
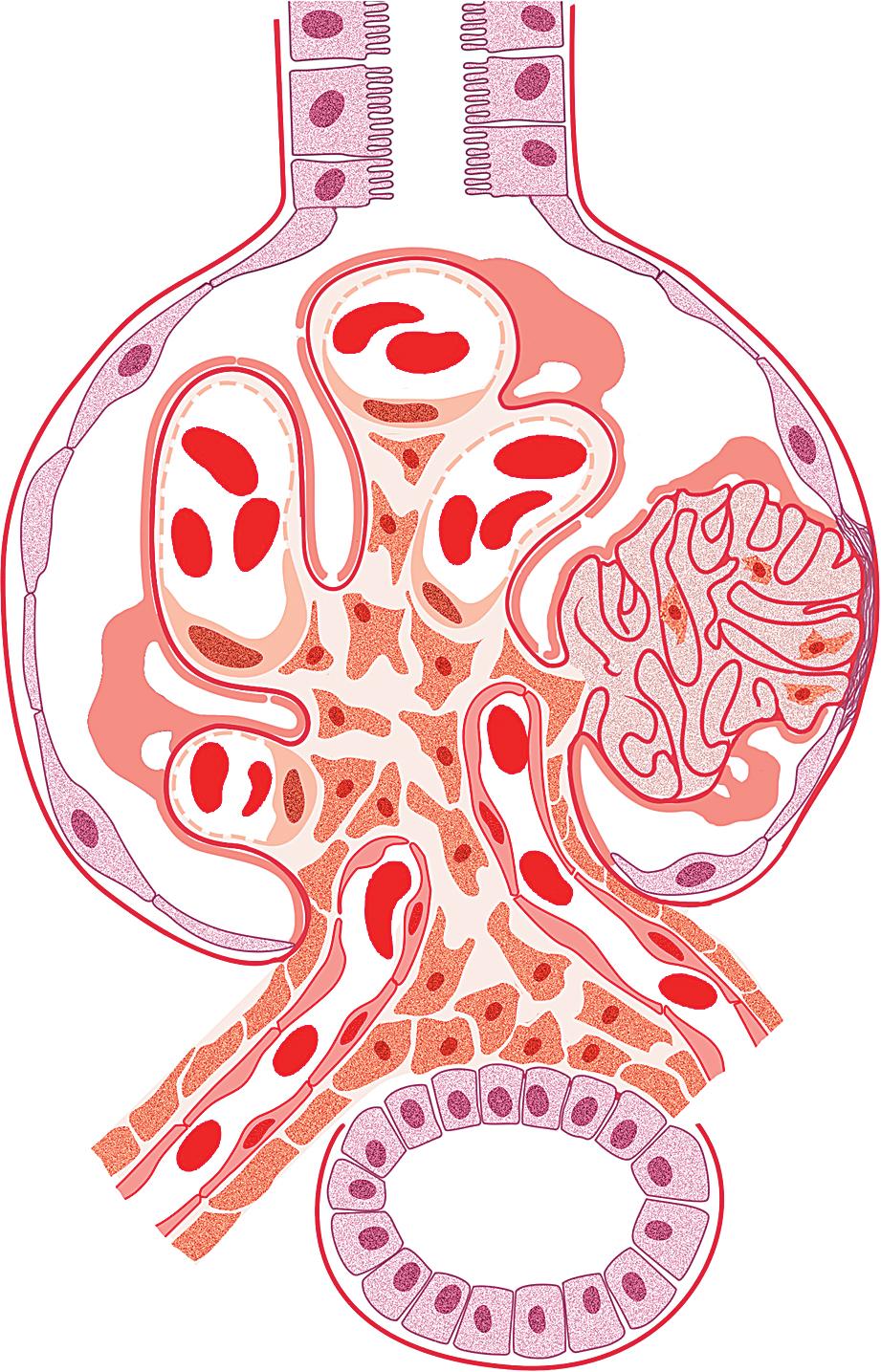
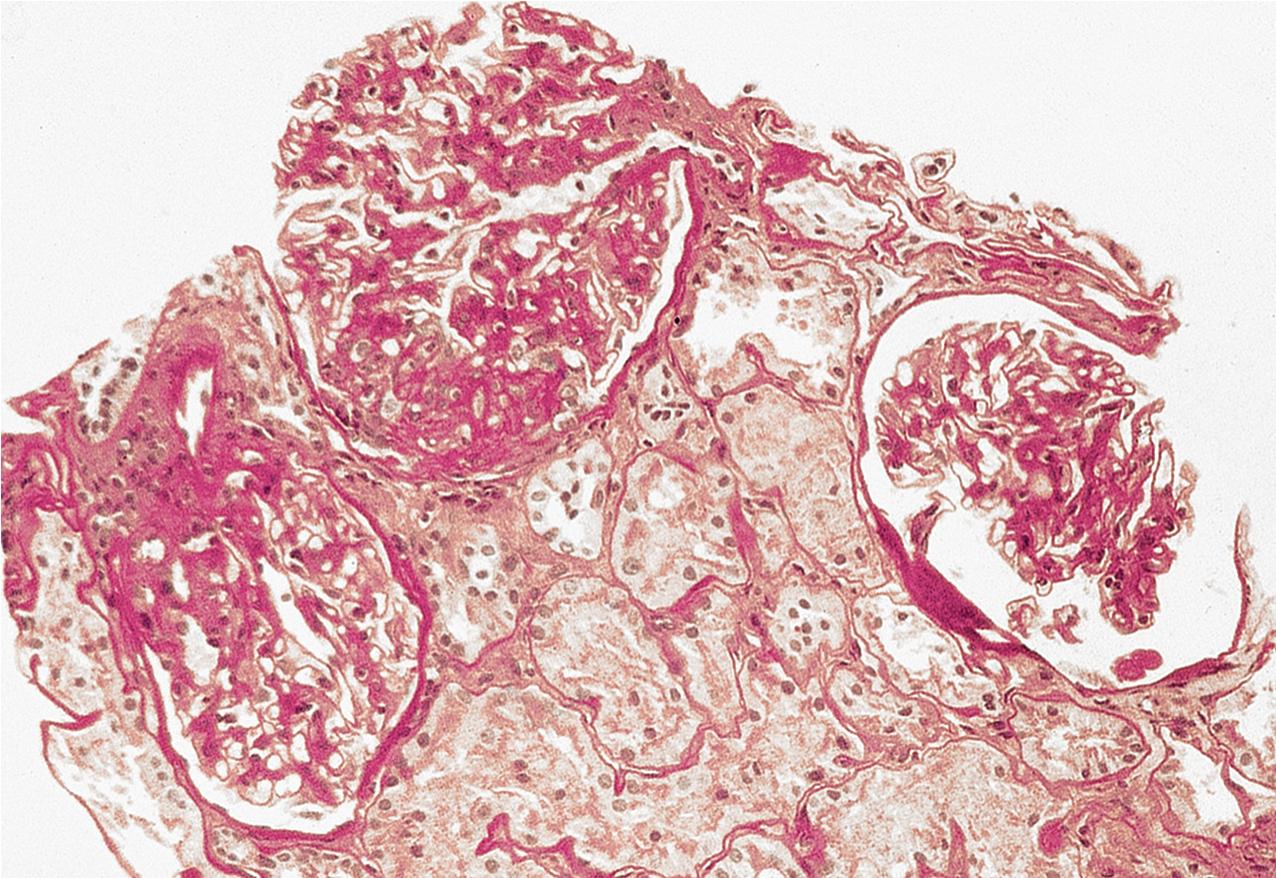
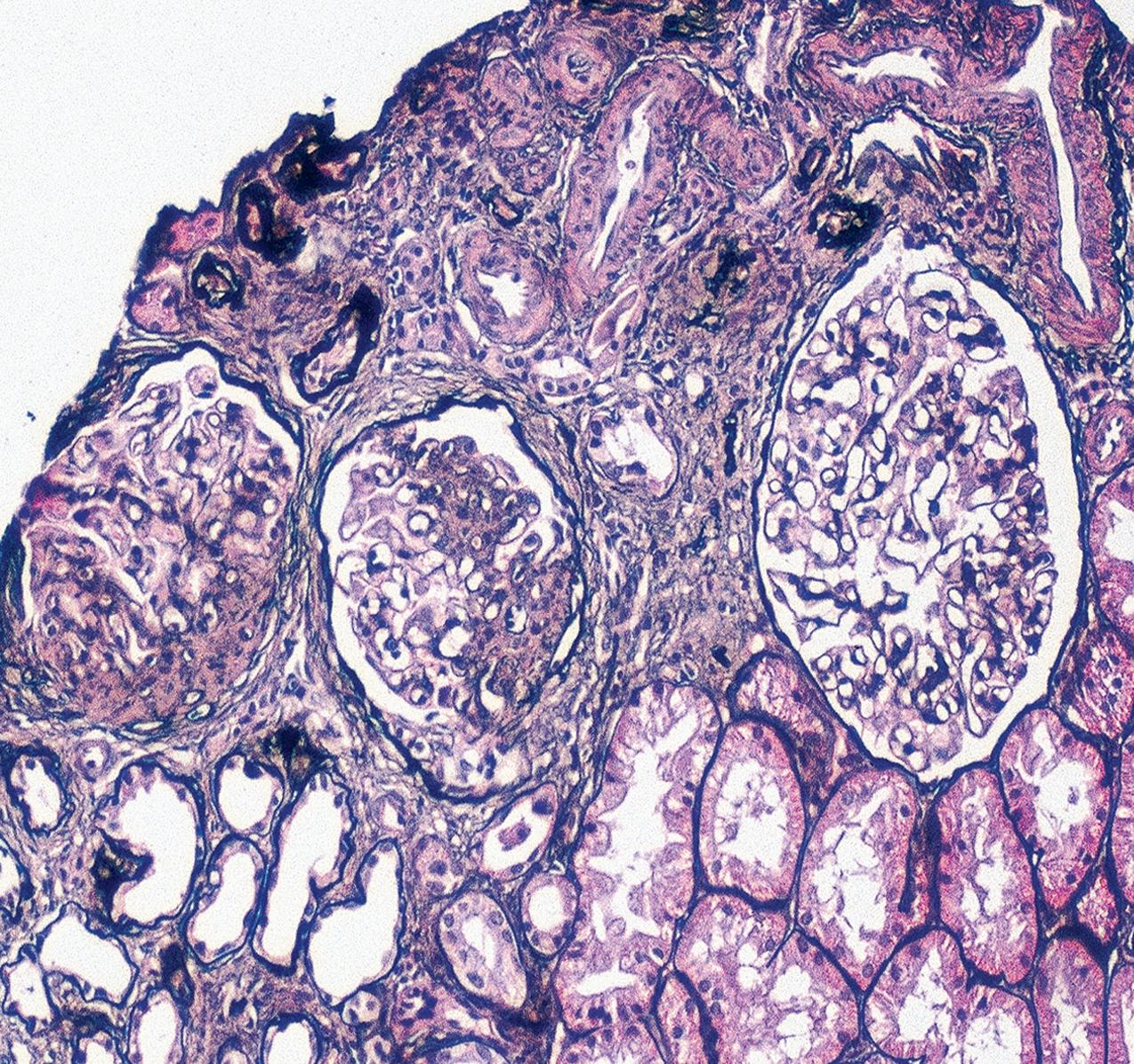
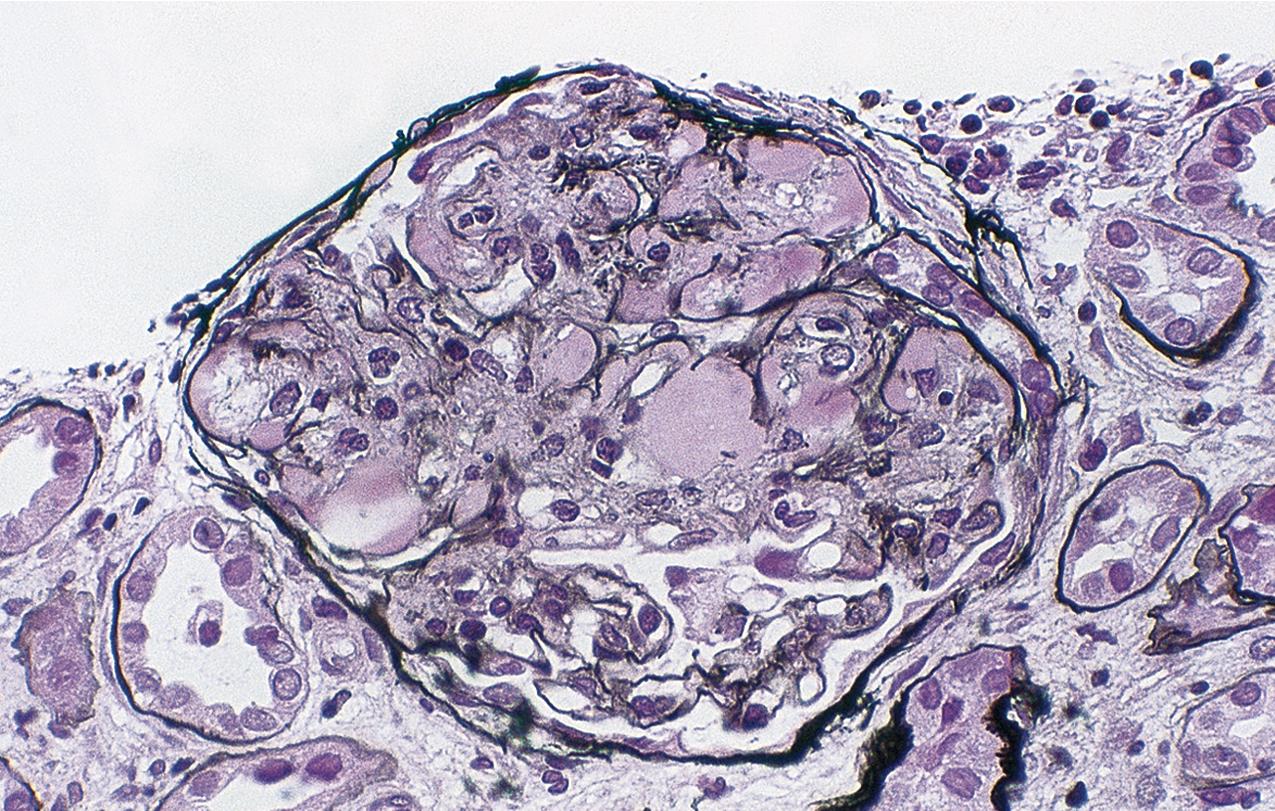
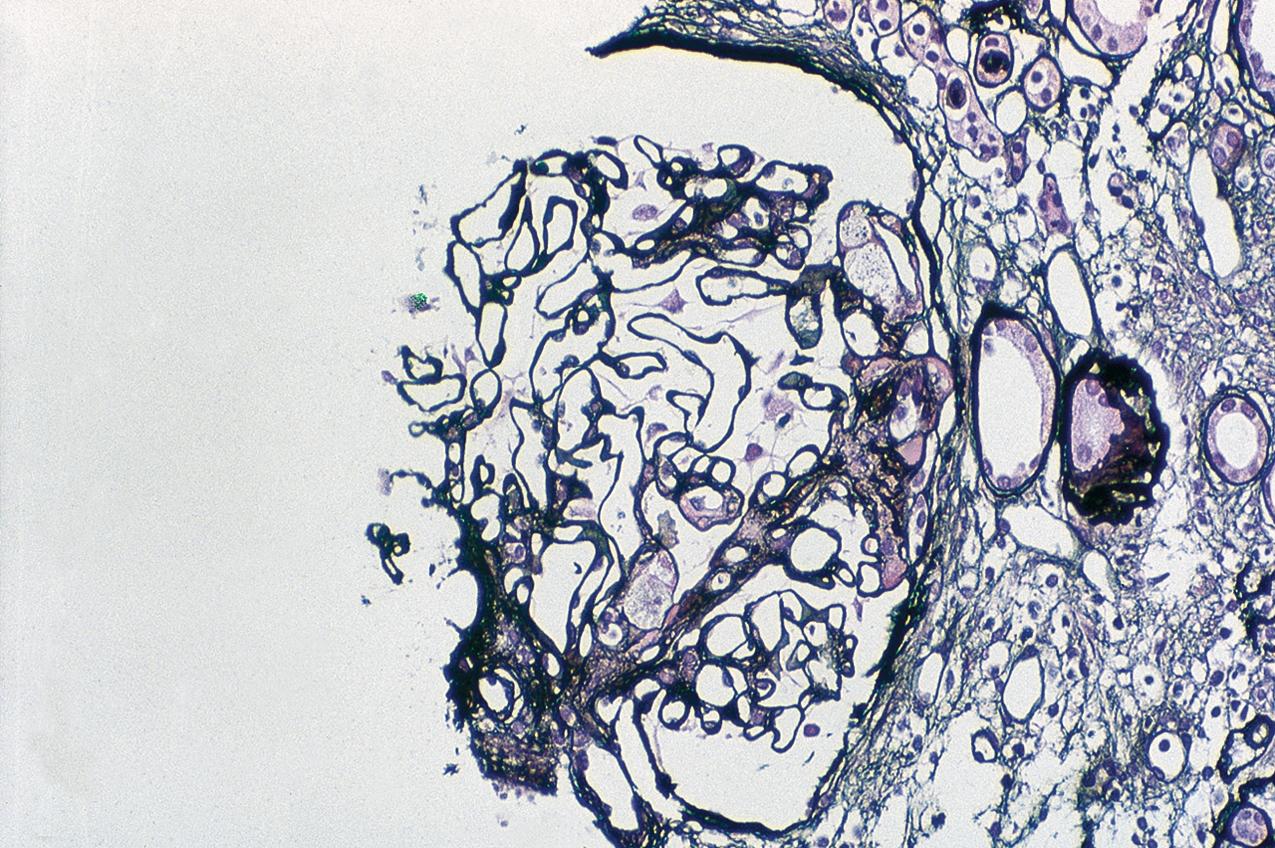
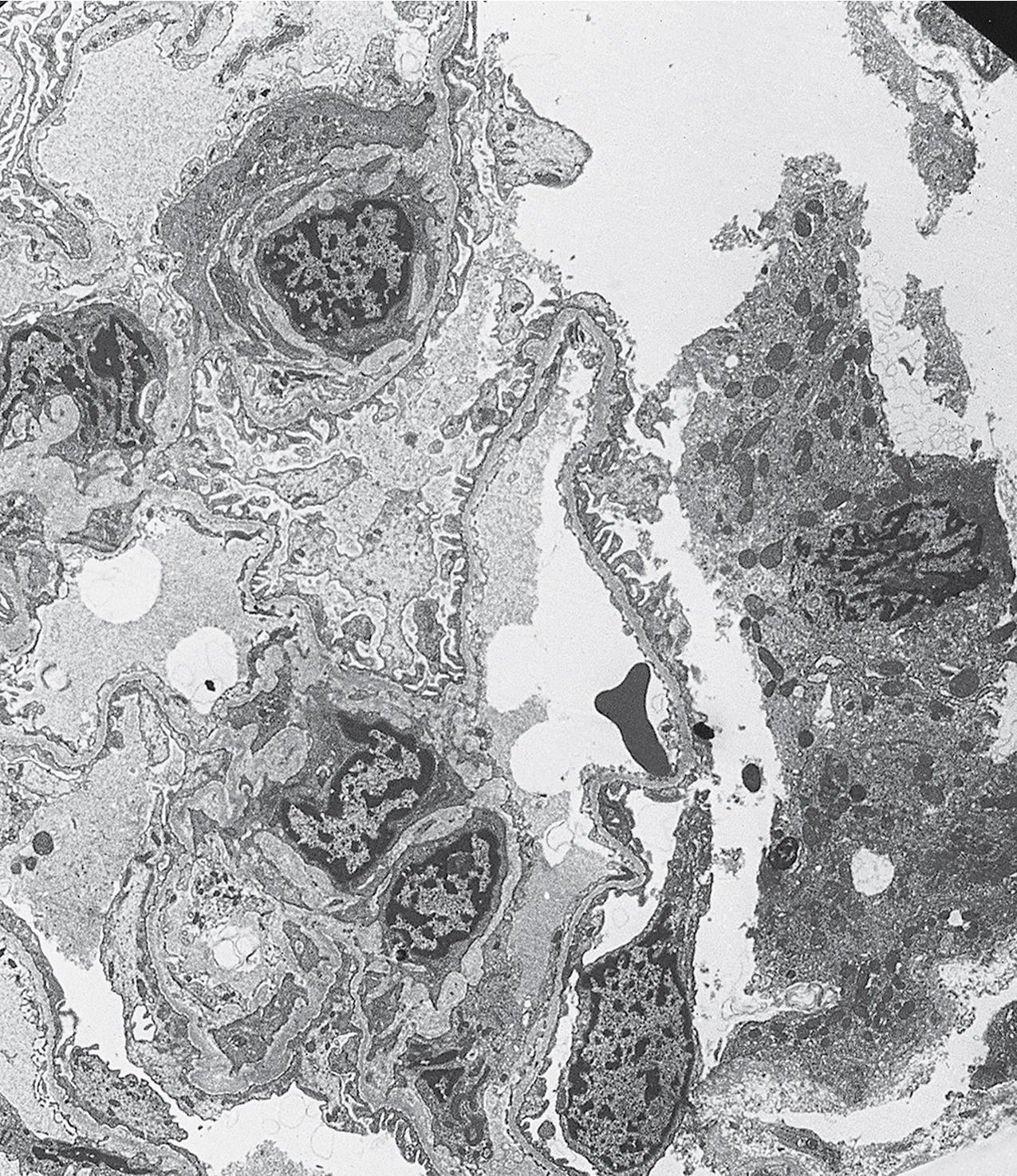
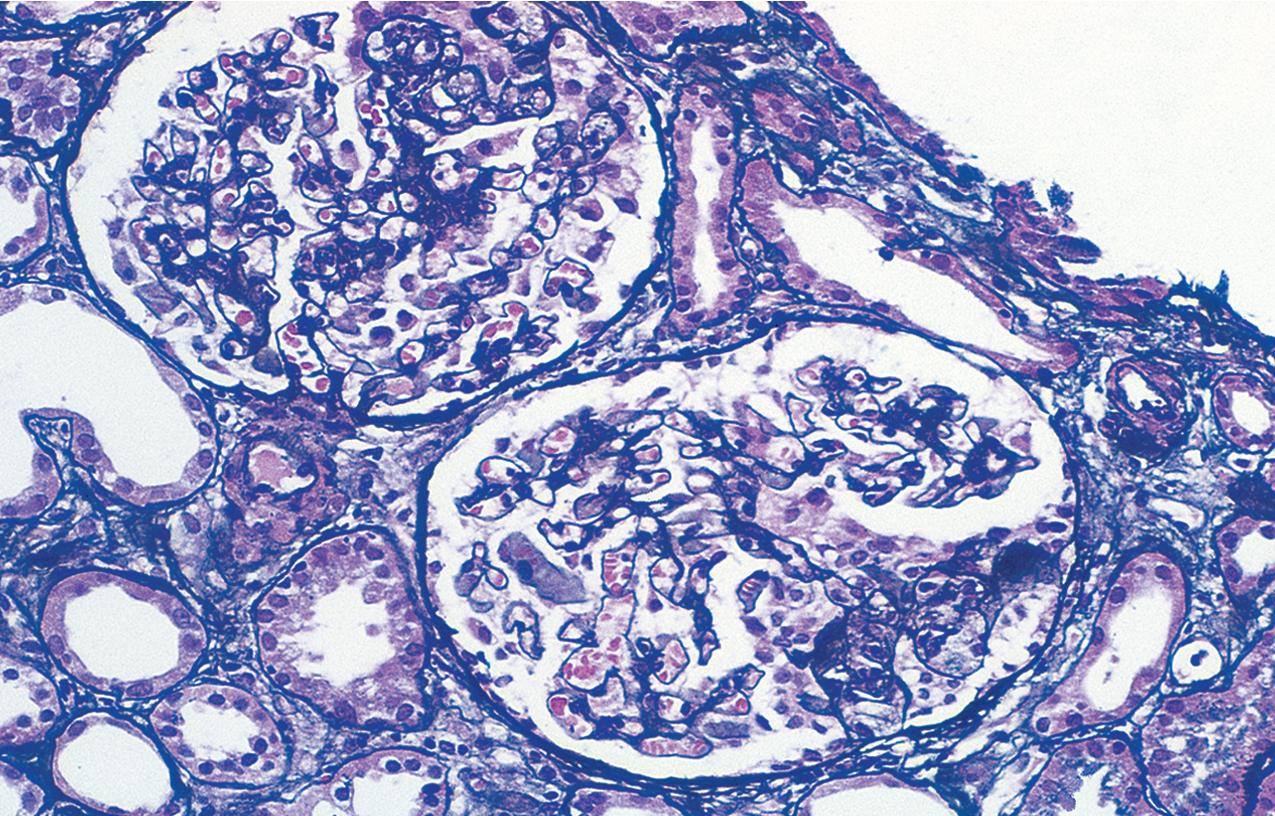
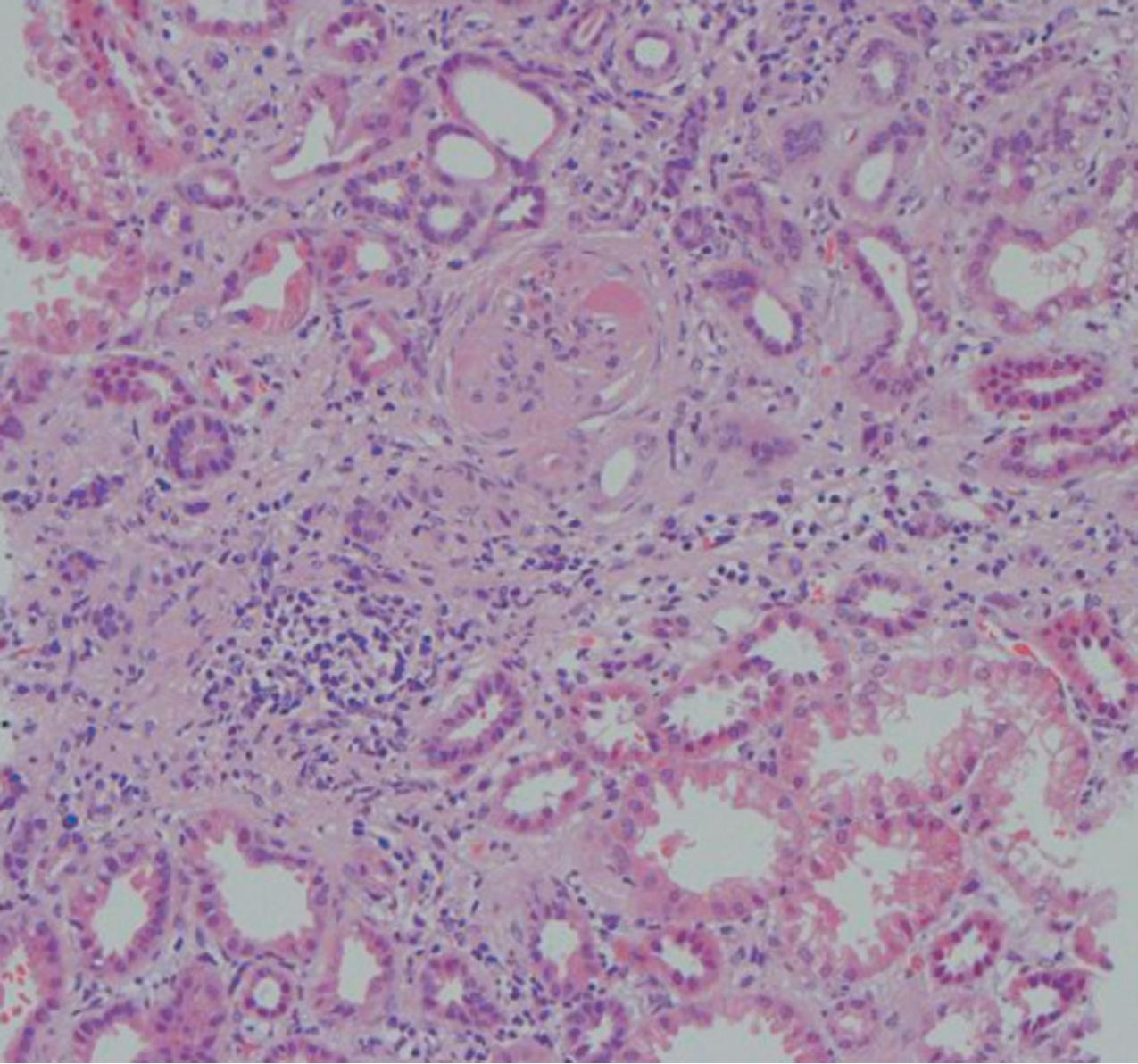
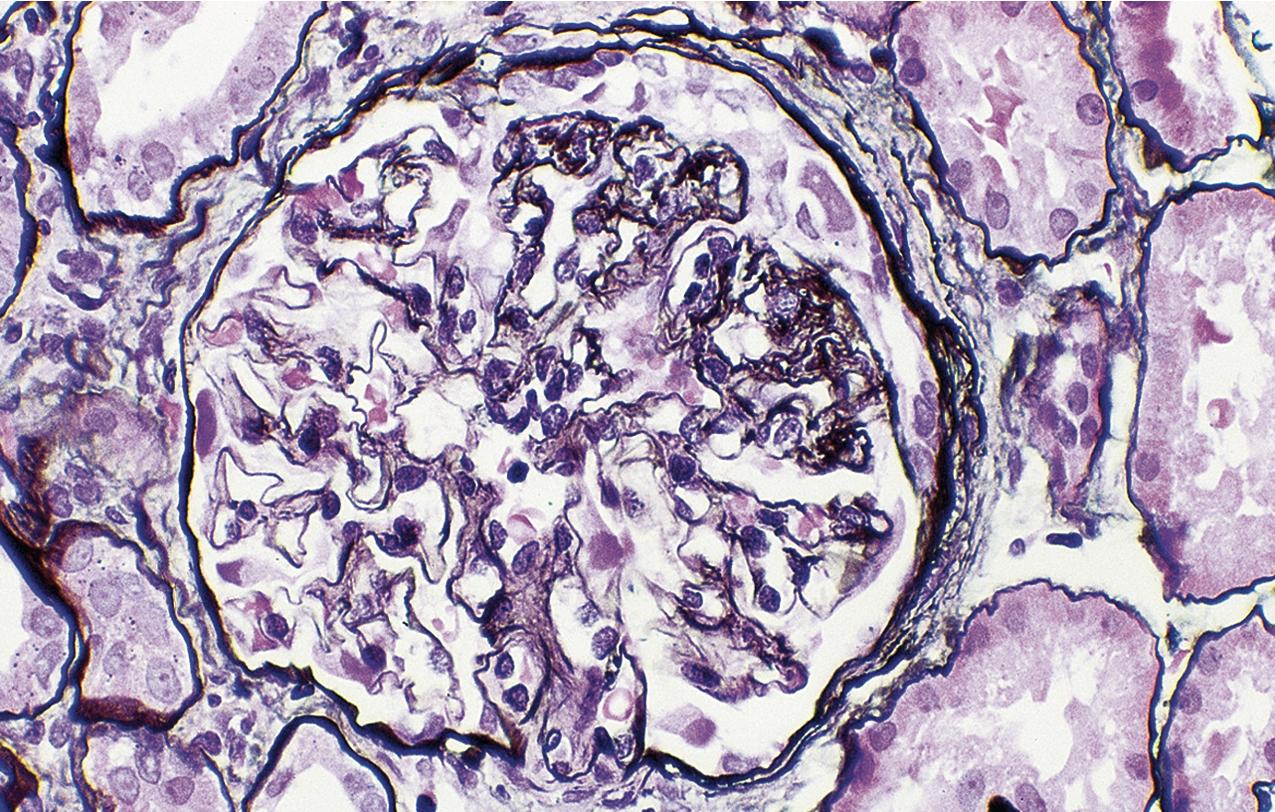
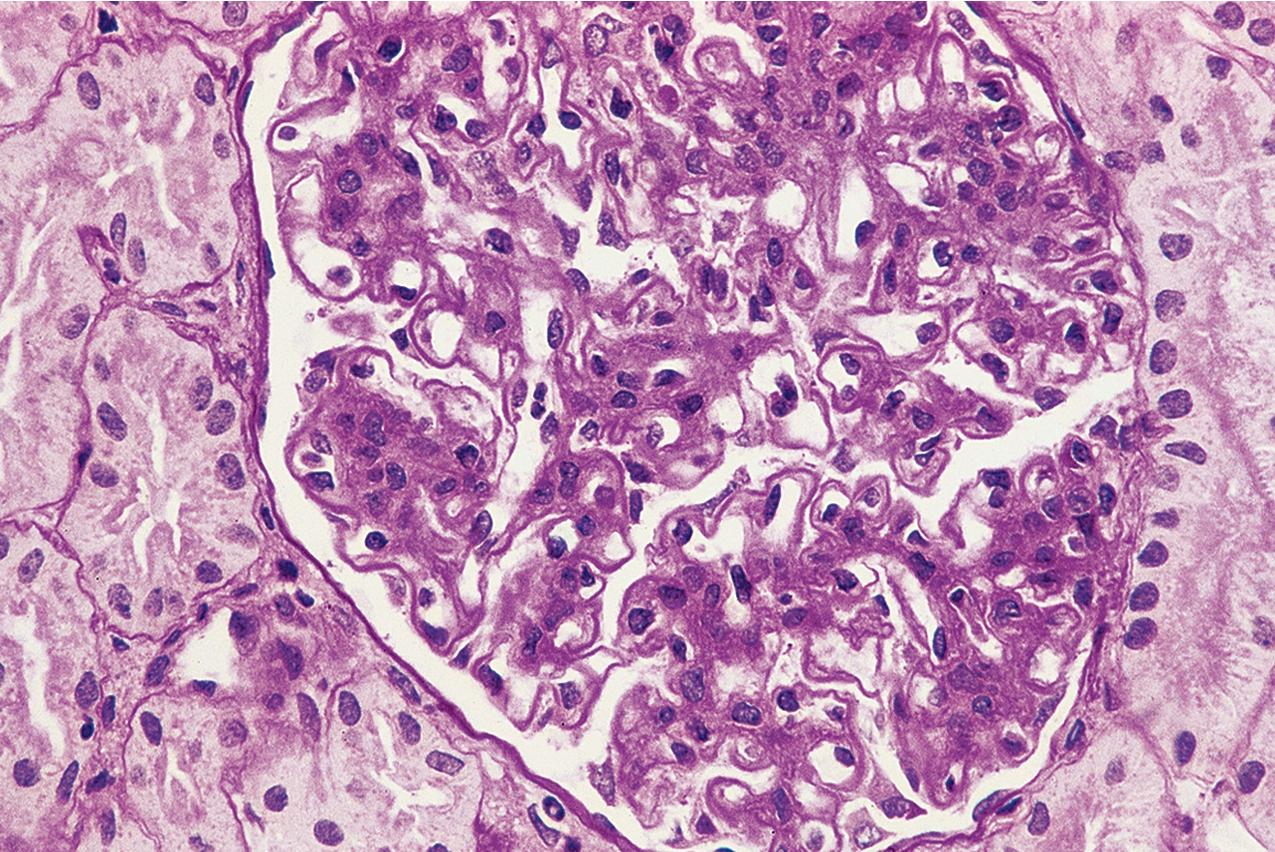
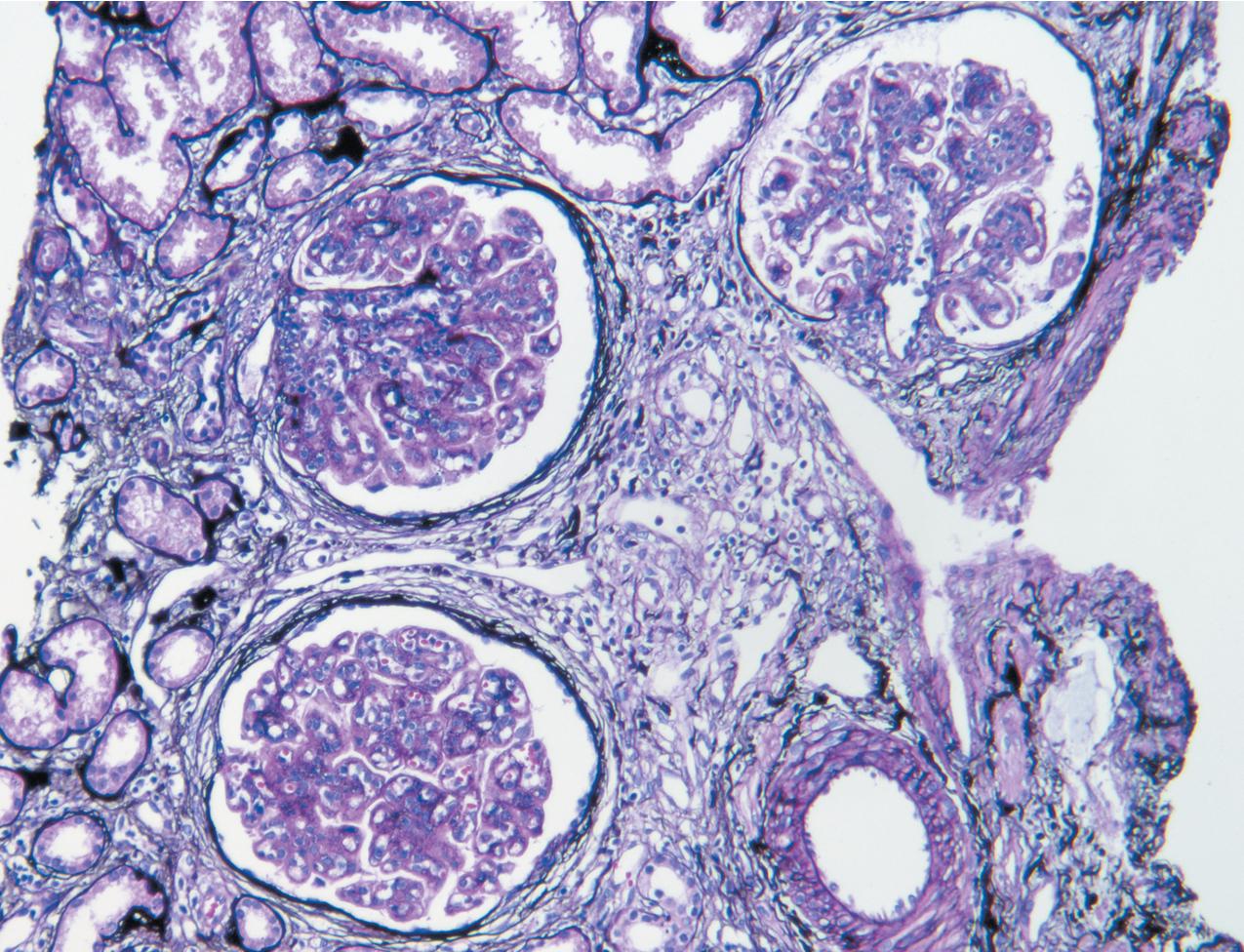
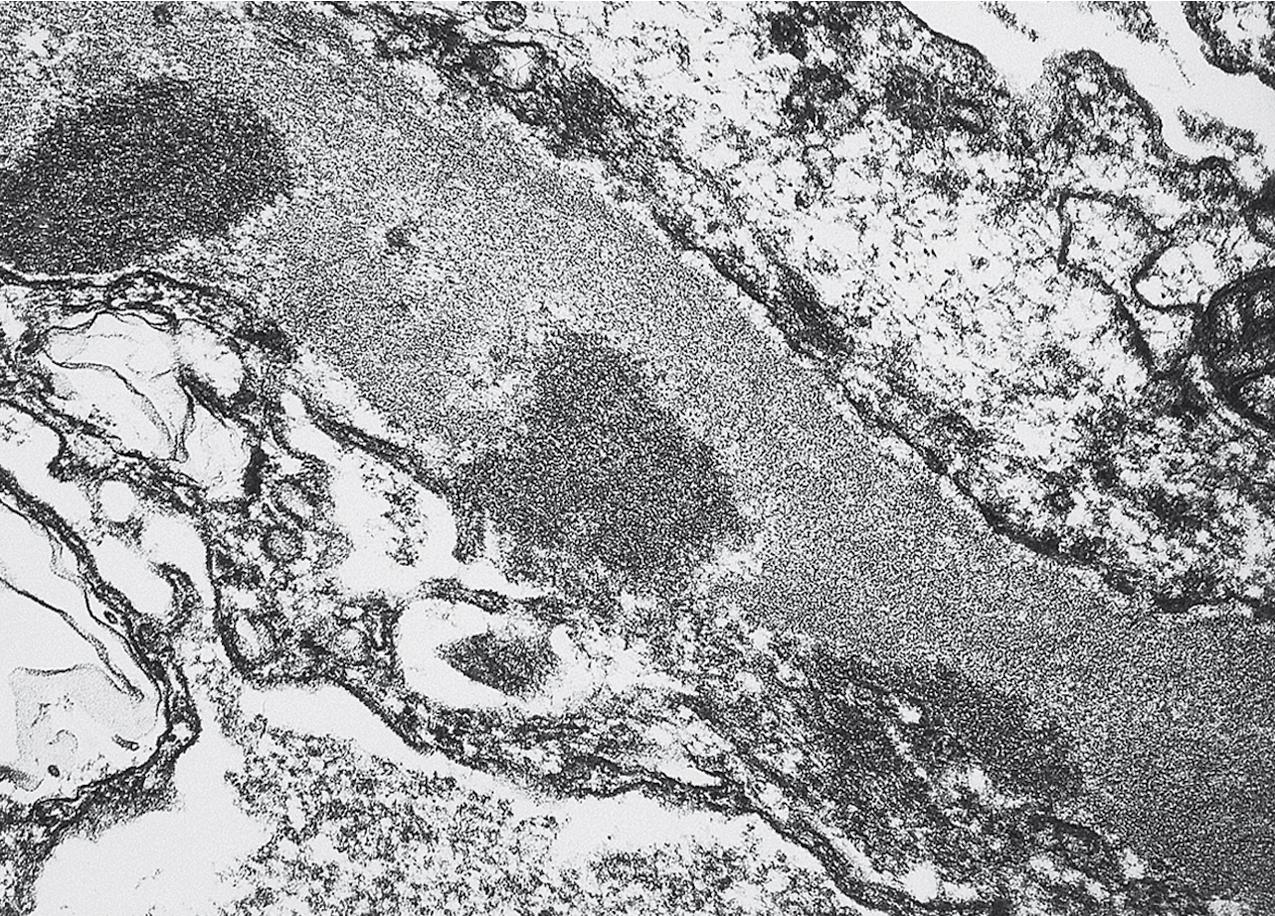
In FSGS of usual type (NOS; Table 3.1 ), sclerosis involves some, but not all, glomeruli (focal), and the sclerosis affects a portion of, but not the entire, glomerular tuft (segmental; Figs. 3.6–3.7 ). The morphologic diagnosis of FSGS is a light microscopic description of this pattern of scarring, which may occur in many settings. Differentiation of MCD (see earlier) from FSGS relies on a large enough sample to detect the sclerotic glomeruli because the detection of even a single glomerulus involved with segmental sclerosis is sufficient to invoke a diagnosis of FSGS rather than MCD. Thus it is apparent that the distinction of MCD and FSGS may be difficult, especially with the smaller samples obtained with current biopsy guns and smaller needles. A sample of only 10 glomeruli has a 35% probability of missing a focal lesion that affects 10% of the nephrons, decreasing to 12% if 20 glomeruli are sampled. The initial sclerosis is in the juxtamedullary glomeruli, and this region should be included in the sample (see Fig. 3.7 ). Conversely, sampling on one section by definition cannot identify all of the focally and segmentally distributed scars. Three-dimensional (3D) studies examining serial sections of glomeruli in cases of idiopathic FSGS have demonstrated that the process indeed is focal; that is, glomeruli without any sclerosis exist even when disease is well established ( Figs. 3.8–3.9 ).
Because of these limitations in detection of sclerotic lesions, other diagnostic features in glomeruli uninvolved by the sclerotic process have been sought to suspect FSGS even without sclerosed glomeruli. Abnormal glomerular enlargement (see later) appears to be an early indicator of the sclerotic process even before overt sclerosis can be detected. The presence of marked glomerular enlargement in a biopsy of otherwise apparent MCD would therefore rather suggest an early, incipient stage of FSGS. Dystroglycan, a component of normal glomerular basement membrane (GBM) that contributes to podocyte–matrix interaction, is generally maintained in nonsclerotic segments in FSGS and decreased in MCD (but also in collapsing type FSGS). This marker, or other emerging biomarkers from molecular and proteomic studies, although not completely sensitive or specific, may be of aid in favoring unsampled FSGS versus MCD in a biopsy with extensive foot process effacement and no defining segmental lesion. CD44 has emerged as one such marker. It is expressed in lymphocytes and in activated parietal epithelial cells. CD44 positive visceral epithelial cells were detected on the glomerular tuft in early recurrent FSGS in the transplant at the stage of foot process effacement, even before detectable sclerosing lesions, and also in early FSGS lesions in native kidneys. Activated parietal epithelial cells are postulated to migrate to the tuft, where they may in this setting contribute to sclerosis, producing the specific matrix LKIV69. Staining for LKIV69 aided in detection of very early segmental sclerotic lesions in native kidney biopsies. In contrast, in MCD, CD44 expression in this location was exceedingly rare. Diffuse mesangial hypercellularity may be a morphologic feature superimposed on changes of either MCD or FSGS, with or without IgM deposits, without defined prognostic significance (see later).
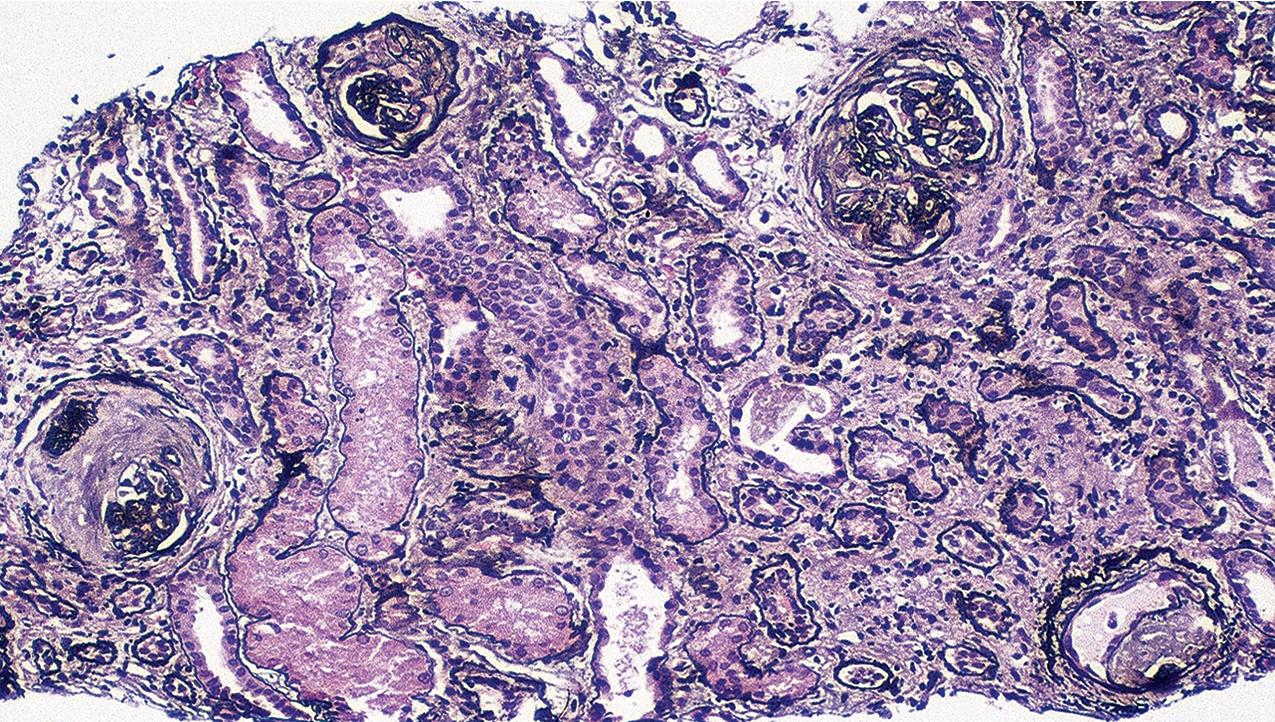
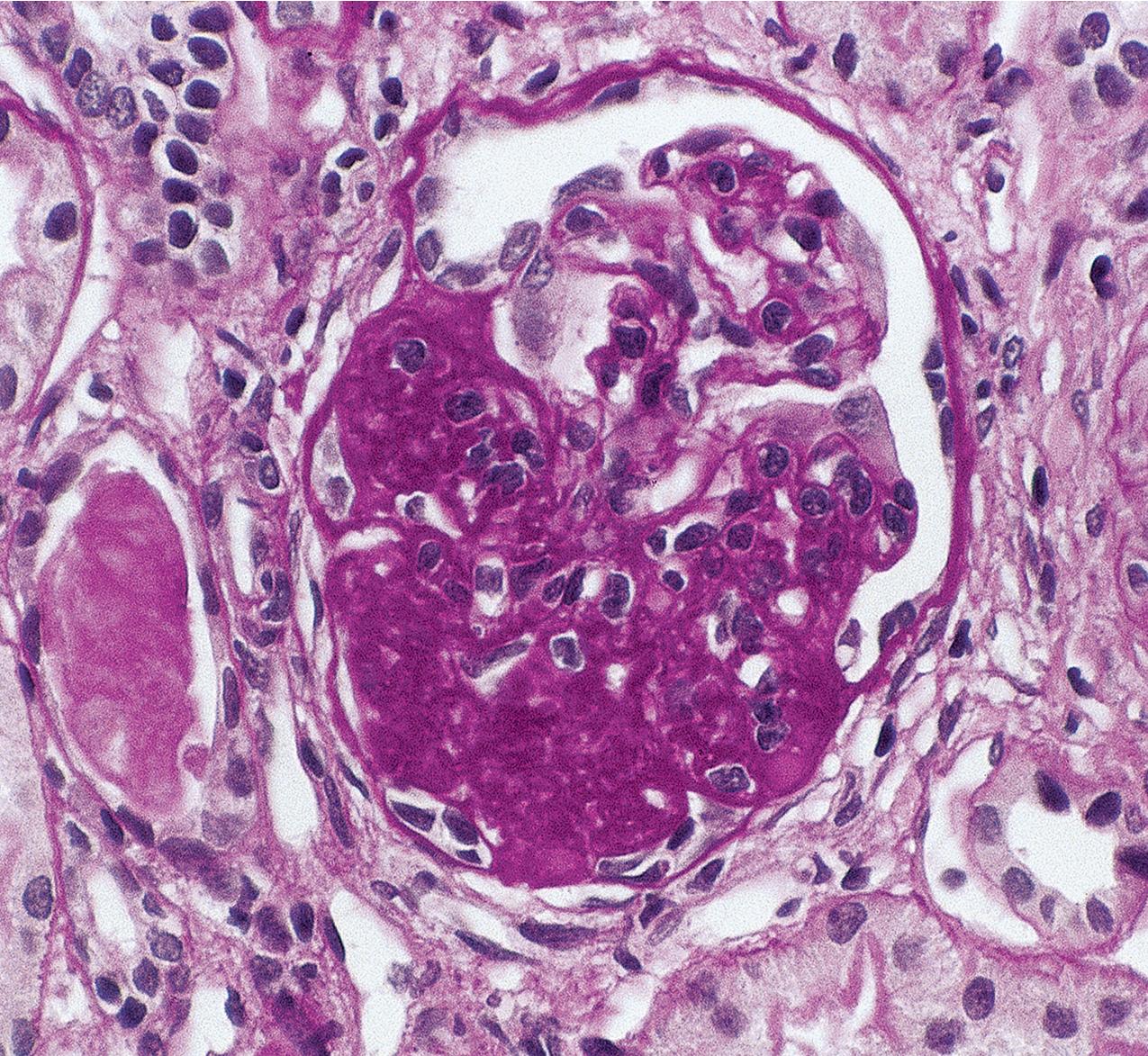
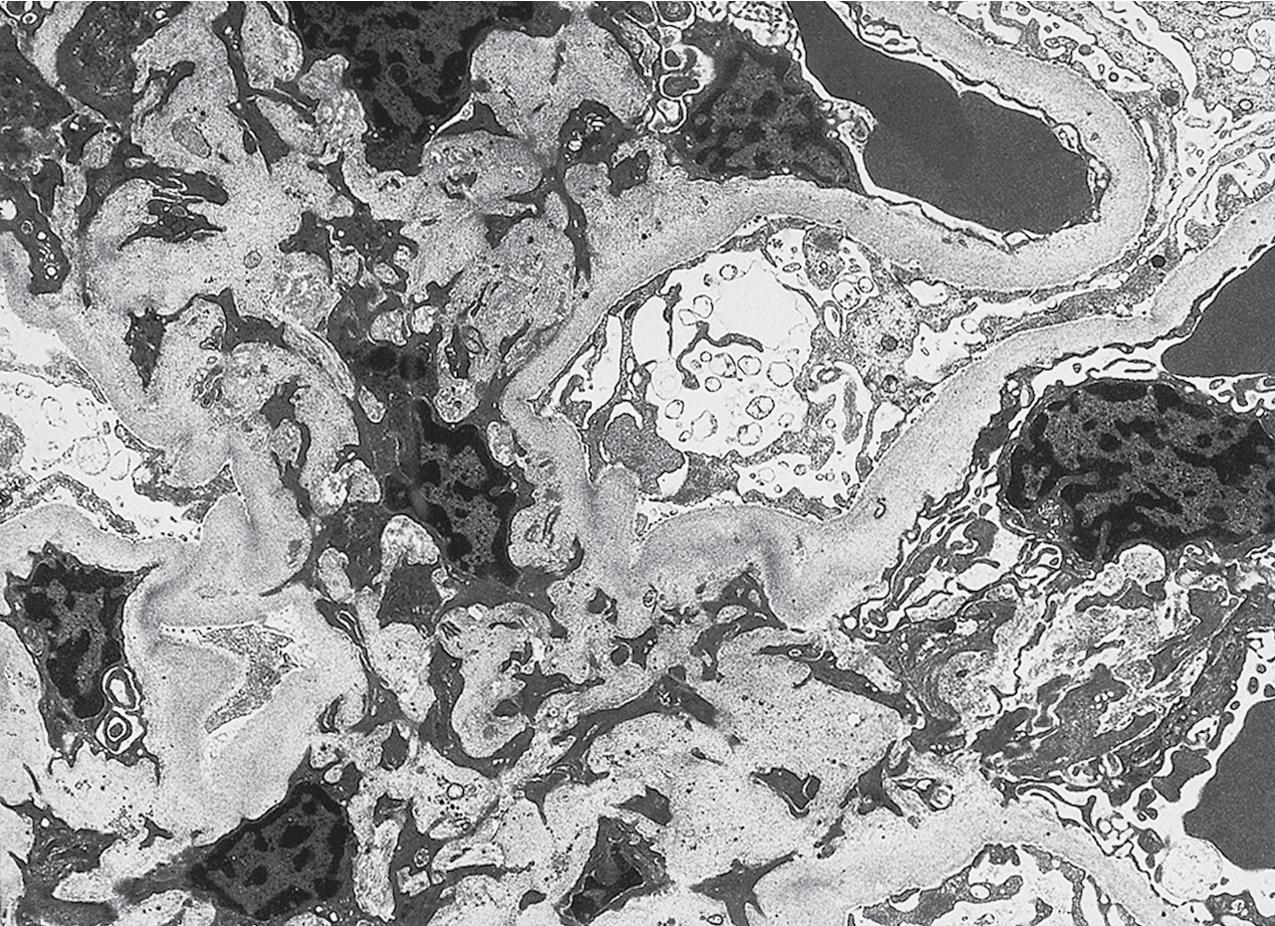
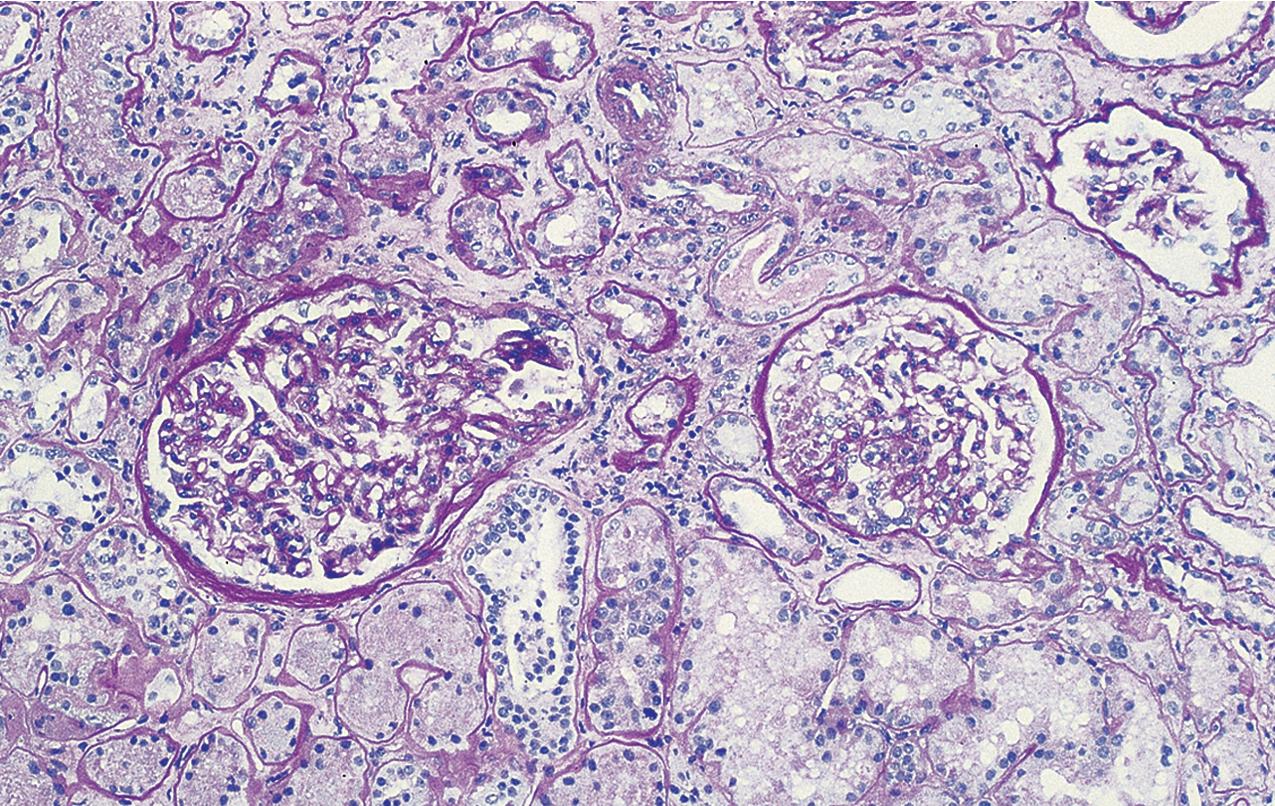
The periodic acid–Schiff (PAS)-positive acellular material in the segmental sclerotic lesions of the glomerulus may have different composition depending on the diverse pathophysiologic mechanisms discussed later. The sclerotic process is defined by glomerular capillary obliteration with increase in matrix, and varies from small, early lesions to near global sclerosis ( Figs. 3.10–3.13 ). The segmental sclerosis lesions are discrete and may be located in perihilar and/or peripheral portions of the glomerulus. There may be associated global glomerulosclerosis of obsolescent type (see “Age-Related Sclerosis,” Chapter 7 ), which has no specific diagnostic significance. Uninvolved glomeruli show no apparent lesions by light microscopy but may appear enlarged, as do glomeruli with early-stage segmental sclerosis. The glomerulosclerosis may be associated with hyalinosis, resulting from insudation of plasma proteins, producing a smooth, glassy (hyaline) appearance ( Fig. 3.14 ). This occurs particularly in the axial, vascular pole region. Of note, arteriolar hyalinosis may occur with hypertension-associated injury and should not be taken per se as evidence of a glomerular sclerotic lesion (see hilar-type FSGS, discussed later). Vascular sclerosis may be prominent late in the course of FSGS. Adhesion of the glomerular tuft to Bowman’s capsule (synechiae) is an early manifestation of sclerosis ( Fig. 3.15 ). Glomerulosclerosis, when fully established, is accompanied by tubular atrophy, interstitial fibrosis with interstitial lymphocytes, proportional to the degree of scarring in the glomeruli (see Fig. 3.11 ). Of note, in HIV-associated nephropathy (HIVAN) and collapsing glomerulopathy, tubular lesions are often microcystic and disproportionally severe (see later).
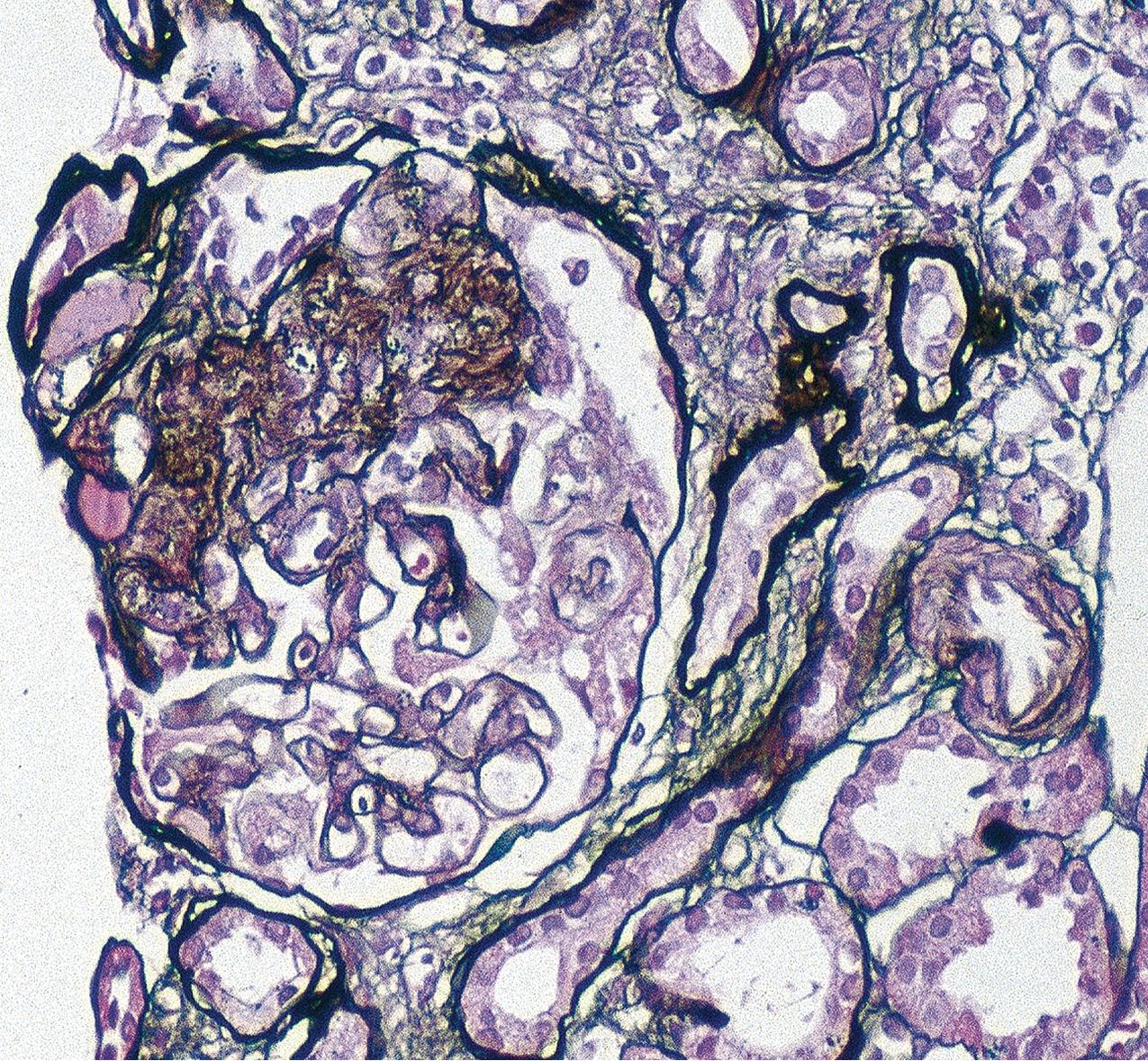
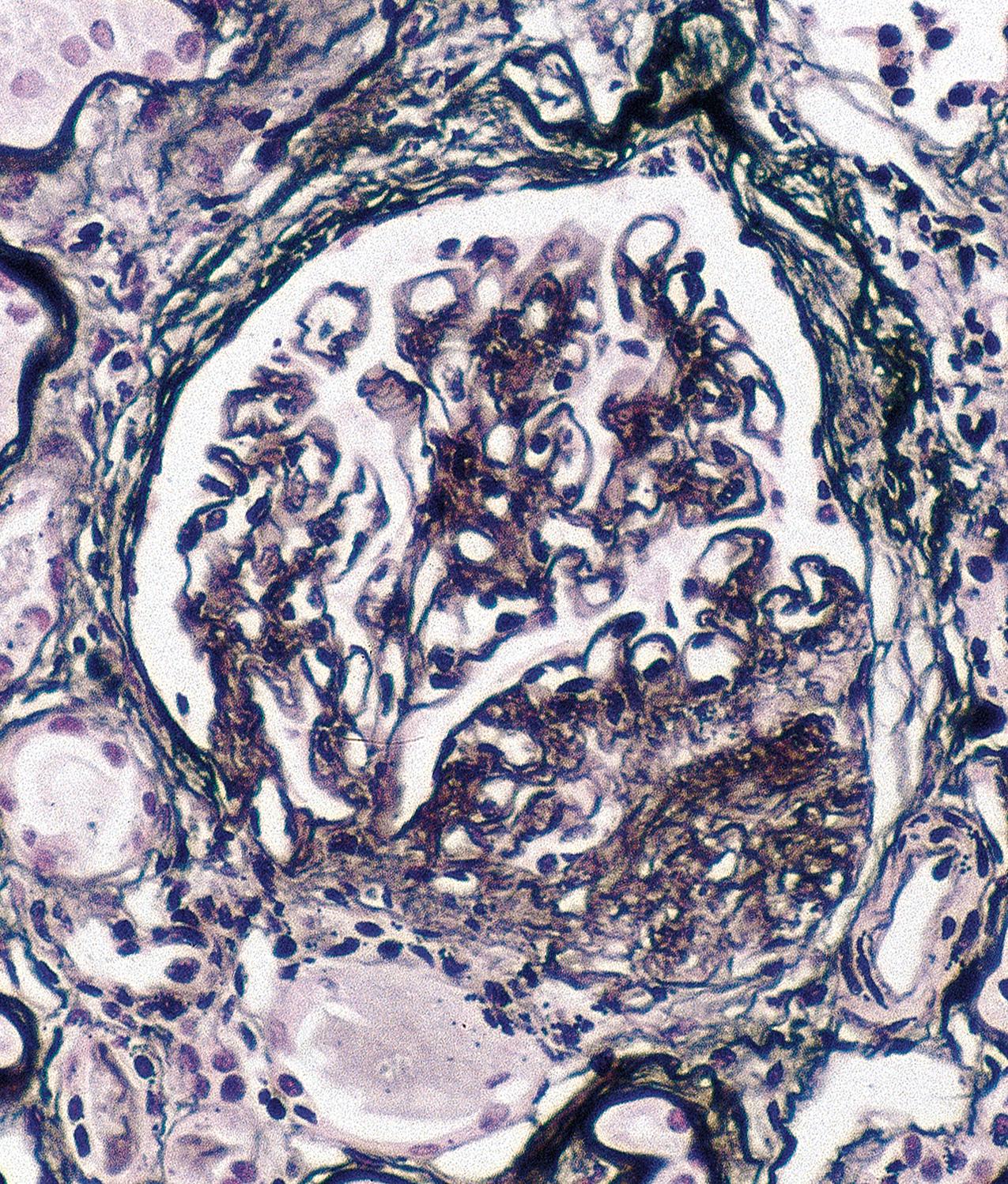
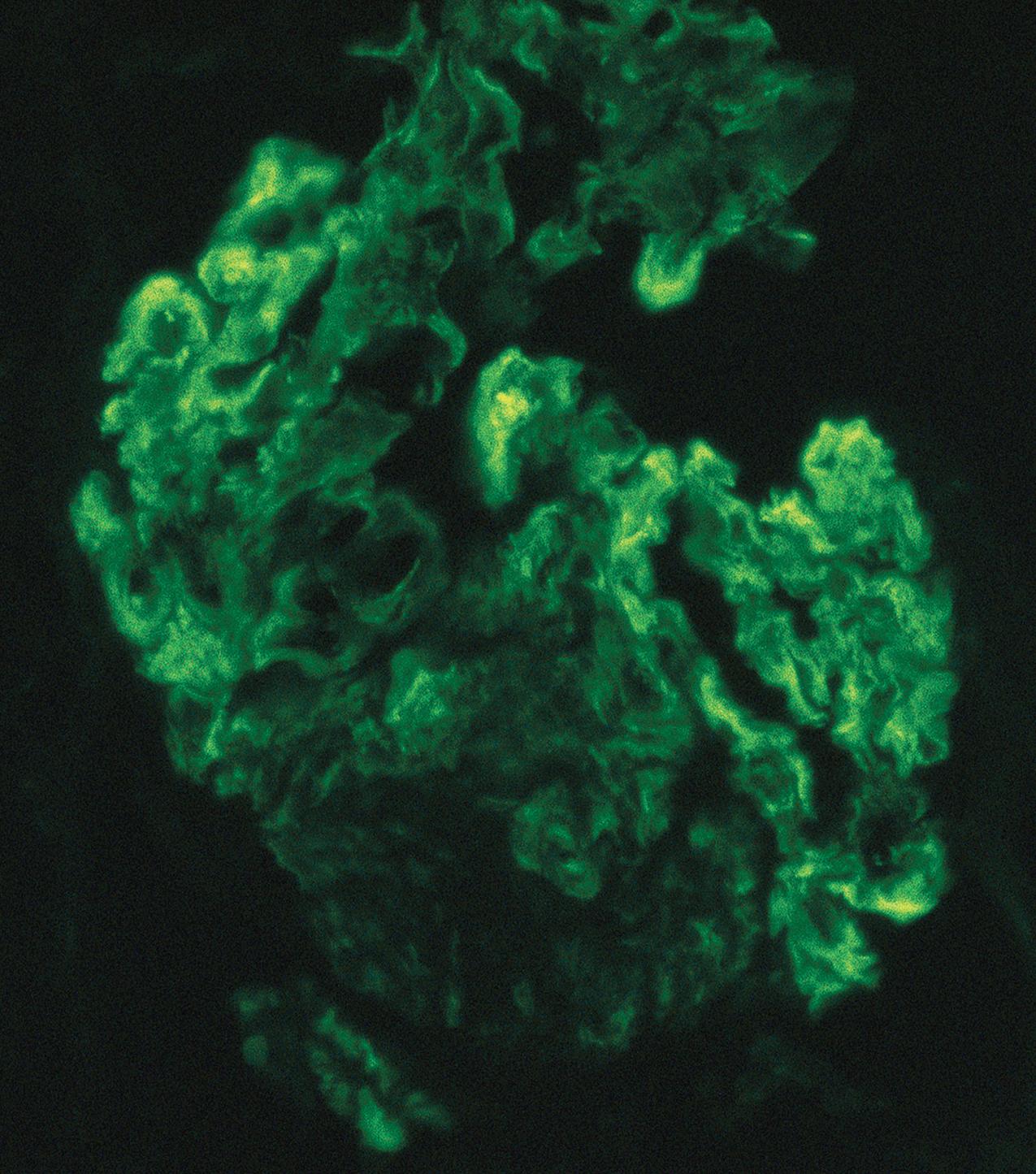
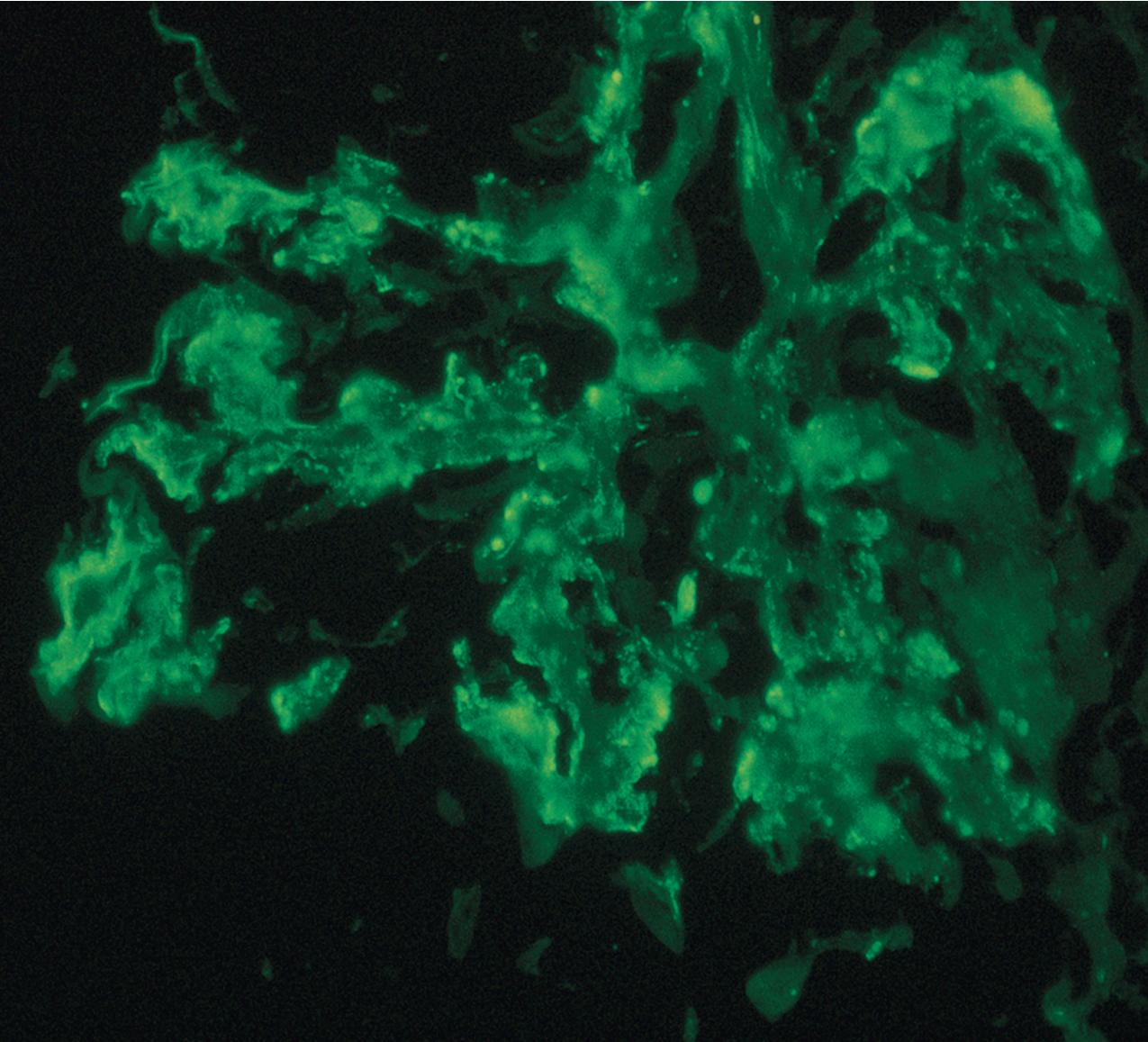
IF may show nonspecific entrapment of IgM and C3 in sclerotic areas or areas where the mesangial matrix is increased ( Fig. 3.16 ).
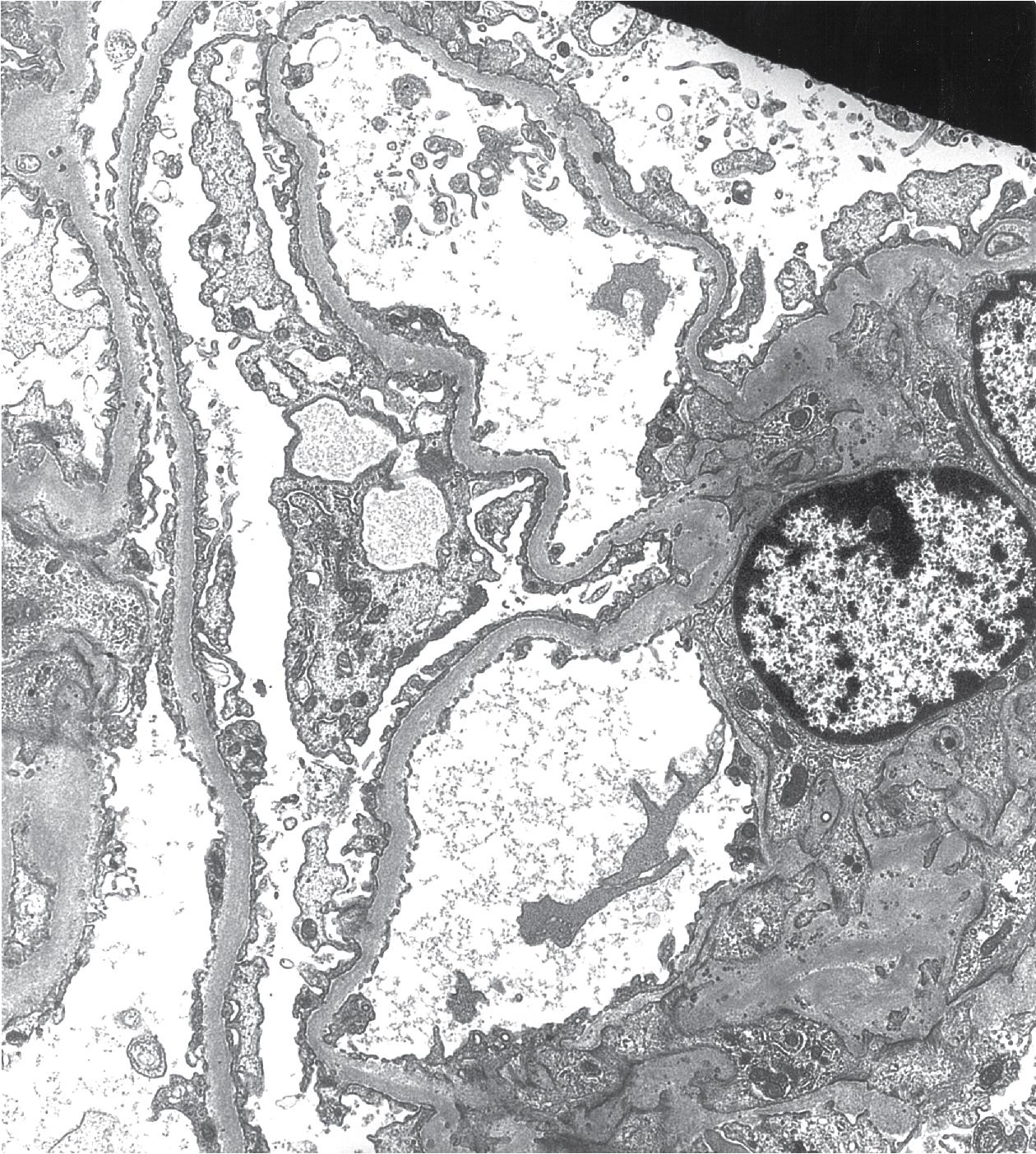
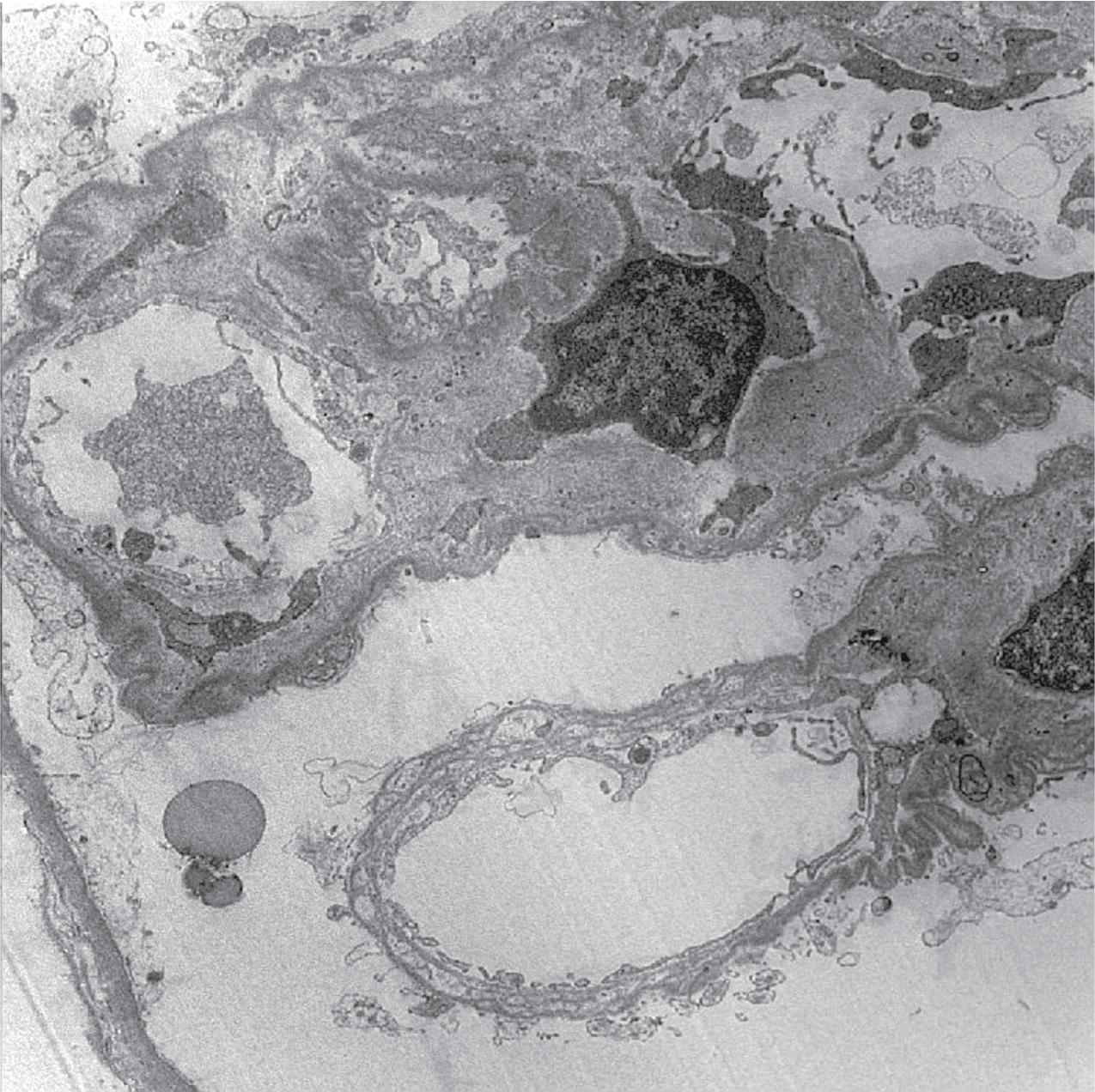
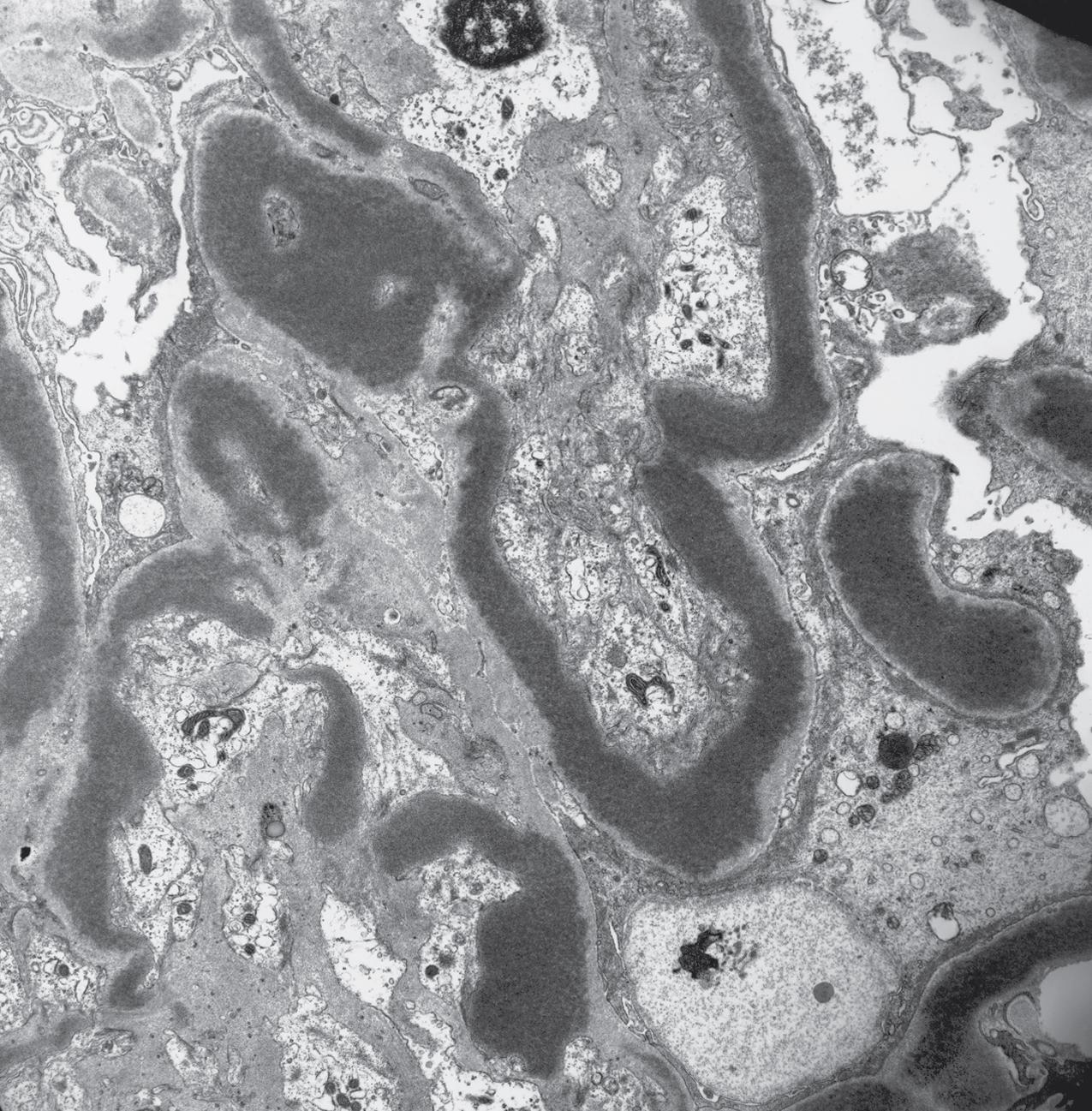
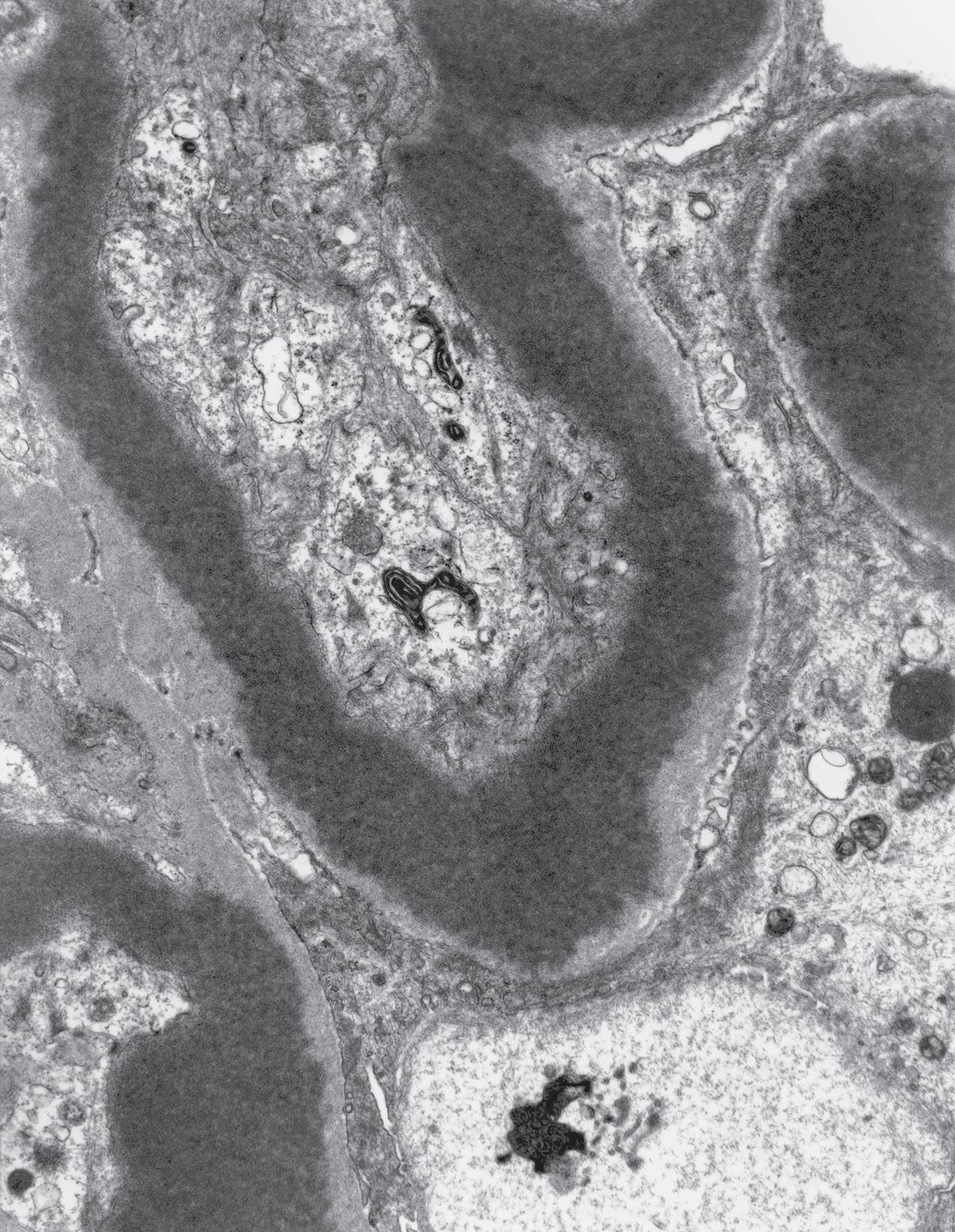
Electron microscopy shows extensive foot process effacement, even in glomeruli without a segmental sclerosing lesion ( Fig. 3.17 ). Thus extent of foot process effacement does not allow precise distinction between MCD and FSGS in individual cases. Foot process effacement tends to be more extensive in primary FSGS compared with secondary FSGS; however, the overlap between these two categories does not allow one to use this as a diagnostic feature in individual cases. Secondary FSGS lesions may show extensive foot process effacement in glomeruli affected by the segmental sclerosing process. Conversely, the absence of significant (i.e., less than 50%) foot process effacement should cast doubt on the diagnosis of untreated primary, idiopathic FSGS. There are no immune deposits in idiopathic FSGS, but mesangial matrix is increased in sclerotic areas ( Fig. 3.18 ). Areas of hyaline may be present in the sclerotic segments and appear dense by EM but should be readily recognized as hyaline, and not confused with immune complexes, by observing scattered lipid droplets and correlating with scout section light microscopic appearance ( Fig. 3.19 ). The presence of numerous reticular aggregates in endothelial cells in the setting of segmental glomerulosclerosis with collapsing features suggests possible HIVAN (see later). Reticular aggregates have also been observed in some patients with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection and collapsing glomerulopathy, likely linked to enhanced cytokine release in these patients.
So-called “primary” FSGS recurs in 30% to 40% of patients. Most recurrences occur within the first months after transplantation. Proteinuria may recur immediately after the graft is implanted, implicating circulating factor(s) in the recurrence. Foot process effacement is present at the time of recurrence of proteinuria and precedes the development of sclerosis, typically by weeks to months. Glomerular enlargement at this stage of recurrent FSGS is prominent in children who otherwise do not undergo glomerular enlargement when receiving an adult kidney. In contrast, an adult recipient of a single kidney will normally have marked renal and glomerular growth to provide adequate glomerular filtration rate (GFR). Overt sclerosis is not noted until weeks to even months after recurrence of nephrotic syndrome. Thus during this time interval in the setting of the FSGS patient with nephrotic syndrome in the transplant, foot process effacement alone, even without detectable segmental sclerosis, is evidence of recurrent FSGS. Activated parietal epithelial cells, staining for CD44, are increased on the glomerular tuft itself even in this early phase of recurrent FSGS and are postulated to have migrated from Bowman’s capsule. Recurrent FSGS often, but not invariably, shows a similar phenotype as diagnosed by the Columbia classification of FSGS to what the patient had in their native kidney.
Some investigators have felt that the common clinical presentation and similar findings in intact glomeruli indicate that MCD and FSGS are two manifestations of the same disease. Our data and those from others rather support differences even at the earliest time points. CD44 is one such marker differentially expressed in visceral epithelial cells in FSGS versus MCD (discussed previously). Parietal epithelial cell-derived matrix LKIV69 expressed on the tuft is an additional useful marker to diagnose early FSGS. Much evidence has pointed to the participation of abnormal glomerular adaptation and growth factors in the pathogenesis of glomerulosclerosis. Several studies have shown that glomerular enlargement precedes overt glomerulosclerosis, in both pediatric and adult patients who otherwise had apparent MCD initially. Patients with abnormal glomerular growth, even on initial biopsies that did not show overt sclerotic lesions, subsequently developed overt glomerulosclerosis, as documented in later biopsies. A cut-off of greater than 50% larger glomerular area than normal for age was a sensitive indicator of increased risk for progression in one series of children with nephrotic syndrome. Of note, glomeruli grow in size until approximately 18 years of age, although no new glomeruli are formed after birth, so age-matched controls must be used in the pediatric population to assess normal glomerular size.
The finding of mesangial hypercellularity (>80% of glomeruli with more than three cells per mesangial region) has been proposed to indicate a subgroup of patients with poorer prognosis and increased risk for developing FSGS. Lack of uniform application of criteria for morphologic definition of mesangial hypercellularity makes it difficult to assess the impact of this feature on prognosis. Nevertheless, several series have failed to confirm a definite clinical correlation of this morphologic variant. Thus patients with mesangial hypercellularity in renal biopsies that otherwise show apparent MCD ultimately had good prognosis despite decreased initial response to steroids. Children with FSGS and mesangial hypercellularity did not show worse prognosis than those with typical FSGS. Thus diffuse mesangial hypercellularity does not appear to impart a specific prognostic significance in either MCD or FSGS, nor does it differentiate between apparent MCD and unsampled FSGS.
IgM deposits by IF in association with mesangial hypercellularity may indicate a poorer response to steroids, and some patients have shown histologic FSGS on second biopsy after an initial biopsy showed IgM nephropathy; however, the significance of IgM deposits by IF in the setting of normal glomeruli by light microscopy has been difficult to assess. Again, series of biopsies from children with FSGS and nephrotic syndrome have failed to show a specific predictive value of the IgM staining with or without diffuse mesangial hypercellularity. If deposits are present by EM and by IF, a mesangiopathic/mesangioproliferative immune complex glomerulonephritis (GN) should be diagnosed.
In summary, the diagnosis of FSGS cannot be completely excluded when segmental sclerotic lesions are not detected, even with a biopsy of adequate size. It is therefore best to include the possibility of unsampled FSGS in biopsies from patients with nephrotic syndrome, no immune complexes, and extensive foot process effacement, especially when glomerular number is less than 25, or other morphologic findings indicative of probability of unsampled FSGS are present. These include glomerular enlargement and interstitial fibrosis (in young patients), and possibly preserved Dystroglycan staining, the parietal epithelial cell-derived matrix LKIV69, or CD44-positive epithelial cells on the glomerular tuft.
Primary FSGS is thought to result from an undefined circulating factor or factors that mediate abnormal glomerular permeability and ultimately sclerosis. Soluble urokinase plasminogen activator receptor (suPAR) and cardiotrophin-like cytokine-1 (CLC-1) have been postulated to represent such causative circulating factors. suPAR, however, is increased with decreased GFR, regardless of cause, and is increased in a variety of inflammatory conditions without FSGS. Inactivation of CLC-1 by galactose infusion has not prevented recurrent FSGS. Thus a proven causal circulating factor in FSGS has not been definitively identified. Recent studies have pointed to podocyte injury and dedifferentiation of its phenotype, with loss of podocytes, with activated parietal epithelial cell migration to the tuft, in the pathogenesis of the sclerotic lesions. The finding of CD44-positive cells, a marker of such activated parietal epithelial cells, on the glomerular tuft precedes overt sclerosing lesions in recurrent FSGS in the transplant, and CD44 or matrix LKIV69 staining on the tuft could also enhance recognition of early sclerosing lesions in native kidneys.
Expanded understanding of the molecular biology of the podocyte and identification of genes mutated in rare familial forms of FSGS (e.g., ACTN4, NPHS2, which encodes podocin, TRPC-6, PLCE1, INF-2, WT1, CD2AP, LAMB2 ), or in congenital nephrotic syndrome of Finnish type (nephrin, coded by the NPHS1 gene), have given important new insights into the mechanisms of progressive glomerulosclerosis and nephrotic syndrome. Genetic studies of familial nephrotic syndrome/FSGS show a high yield of detection of pathogenic or possibly pathogenic mutations in younger patients. Thus in children less than 1 year old with steroid-resistant FSGS, 50% had mutations detected, decreasing to 25% in children between the ages of 1 and 6, and even less in older children and adolescents (18% and 11%, respectively). With increasing age of onset of FSGS and in nonfamilial cases, the detection of causal mutations is substantially less, about 8% in adult sporadic FSGS in one series. We will only briefly discuss some of these genetic forms of FSGS. Key gene mutations causing FSGS/nephrotic syndrome include those encoding structural proteins of the podocyte cytoskeleton or slit diaphragm, those governing podocyte–GBM interaction, affecting mitochondrial function, or coenzyme Q10 function ( COQ ) . Nephrin localizes to the slit diaphragm of the podocyte and is tightly associated with CD2-associated protein (CD2AP). Nephrin functions as a zona occludens–type junction protein and, along with CD2AP, plays a crucial role in receptor patterning, cytoskeletal polarity, and signaling. Mice engineered to be deficient in CD2AP develop congenital nephrotic syndrome, similar to congenital nephrotic syndrome of Finnish type (CNF). Autosomal dominant FSGS can be caused by mutations in one of several genes, including alpha-actinin 4 (ACTN4 ; Figs. 3.20–3.21 ). This is hypothesized to cause altered actin cytoskeleton interaction, perhaps causing FSGS through a gain-of-function mechanism, contrasting with the loss-of-function mechanism implicated for disease caused by the nephrin mutation mice, with either knockout or knock-in of mutated ACTN4 that develop FSGS lesions. Thus balance of ACTN4 is crucial for the podocyte. Patients with ACTN4 mutation progress to end stage by age 30, with rare recurrence of nephrotic syndrome in the transplant, perhaps related to as-yet-undefined immune reactions to the normal, nonmutated transplant kidney. Transient receptor potential cation channel-6 (TRPC-6) is a channel molecule expressed in the podocyte, and when mutated, a gain-of-function altered calcium flux occurs. FSGS develops in adulthood with variable penetrance. Mutation in inverted formin ( INF2) is a relatively common cause of autosomal dominant FSGS, and patients present in teenage years or as adults. Podocin, another podocyte-specific gene (NPHS2), is mutated in autosomal recessive FSGS that has an early onset in childhood with rapid progression to end-stage kidney disease with frequent steroid resistance. Podocin is an integral stomatin protein family member and interacts with the CD2AP–nephrin complex, indicating that podocin could serve in the structural organization of the slit diaphragm. In contrast to the steroid resistance of the aforementioned, some patients with PLCE1 mutations may respond to steroids. Acquired disruption of some of these complexly interacting podocyte molecules has been demonstrated in experimental models and in human proteinuric diseases. Genetic variants of apolipoprotein L1 ( APOL1; G1, G2 variants vs. G0 with no increased risk) that are protective against trypanosomal disease have been linked to increased FSGS in patients of black African ethnicity. Mechanisms for renal disease susceptibility are postulated to involve increased susceptibility of the podocytes to additional second hits in patients homozygous or compound heterozygous for the risk alleles (see “ APOL1 -Associated Nephropathies,” Chapter 6 ). Thus it is possible that novel molecular and immunostaining techniques to detect abnormalities in these genes and the proteins they encode will become of diagnostic and prognostic utility. For example, some patients with mutations of the COQ genes respond to treatment with CoQ10. Mutations in collagen IV genes, of alpha 3, 4, or 5 chains, causal in autosomal and X-linked forms of Alport, have been found in families with FSGS lesions and proteinuria. Nevertheless, EM showed thin or otherwise abnormal GBM in most, and this may thus represent an unusual part of the spectrum of Alport with FSGS lesions as a major manifestation. In most cases, there currently are no specific morphologic findings recognized to distinguish the FSGS cases caused by genetic mutations from other types of FSGS (see Figs. 3.20–3.21 ), with the exception of, for example, some features suggestive of CNF, GBM abnormalities suggestive of Alport/ COL4 mutations, and mitochondrial abnormalities in patients with a mutation in mtDNA-A3243G (causing mitochondrial myopathy, encephalopathy, lactic acidosis, stroke-like episodes, MELAS [mitochondrial encephalopathy, lactic acidosis, and stroke-like episodes], and FSGS).
Extensive foot process effacement
Absence of immune complexes
Diagnostic segmental lesions
Global glomerulo sclerosis may be found normally or in any condition and does not differentiate between MCD and FSGS.
Extent of foot process effacement does not distinguish between primary FSGS and minimal change disease: Less than 50% effacement indicates the process is not likely either untreated MCD or untreated primary FSGS.
Even in the absence of diagnostic segmental lesions (see earlier), unsampled FSGS may be considered in biopsies with a small sample size.
Surrogate markers of unsampled FSGS include marked glomerulomegaly and/or interstitial fibrosis in young patients.
Subtotal (i.e., <50%) foot process effacement strongly favors secondary FSGS.
Extensive foot process effacement may, however, occasionally occur even in secondary FSGS.
Key differential features:
Arterionephrosclerosis: extensive vascular sclerosis, increased global glomerulosclerosis in solidified pattern, periglomerular fibrosis around nonsclerotic glomeruli, and increased lamina rara interna.
Chronic pyelonephritis/reflux nephropathy: sharply delineated, geographic pattern of scarring and thyroidization of tubules, periglomerular fibrosis, and occasionally increased lamina rara interna and subtotal foot process effacement.
Secondary collapsing glomerulopathy causes:
HIVAN; numerous reticular aggregates suggest HIVAN (or possibly lupus nephritis or SARS-CoV-2–associated collapsing glomerulopathy, COVAN).
Examples of other secondary causes of collapsing lesions, usually with less extensive foot process effacement: pamidronate toxicity, interferon treatment, severe ischemia (such as that seen with cyclosporin, cocaine), anabolic steroids, SLE, parvovirus. Clinical correlation is essential.
Braden GL, Mulhern JG, O’Shea MH, et al. Changing incidence of glomerular diseases in adults. Am J Kidney Dis. 2000;35:878–883.
Corwin HL, Schwartz MM, Lewis EJ. The importance of sample size in the interpretation of the renal biopsy. Am J Nephrol. 1988;8:85–89.
D’Agati V. The many masks of focal segmental glomerulosclerosis. Kidney Int. 1994;46:1223–1241.
D’Agati VD, Fogo AB, Bruijn JA, et al. Pathologic classification of focal segmental glomerulosclerosis: A working proposal. Am J Kidney Dis. 2004;43:368–382.
D’Agati VD, Kaskel FJ, Falk RJ. Focal segmental glomerulosclerosis. N Engl J Med. 2011;365:2398–2411.
Deegens JK, Dijkman HB, Borm GF, et al. Podocyte foot process effacement as a diagnostic tool in focal segmental glomerulosclerosis. Kidney Int. 2008;74:1568–1576.
Fatima H, Moeller MJ, Smeets B, et al. Parietal epithelial cell activation marker in early recurrence of FSGS in the transplant. Clin J Am Soc Nephrol. 2012;7:1852–1858.
Fogo AB. Causes and pathogenesis of focal segmental glomerulosclerosis. Nat Rev Nephrol. 2015;11:76–87.
Fogo A, Hawkins EP, Berry PL, et al. Glomerular hypertrophy in minimal change disease predicts subsequent progression to focal glomerular sclerosis. Kidney Int. 1990;38:115–123.
Garin EH, Mu W, Arthur JM, et al. Urinary CD80 is elevated in minimal change disease but not in focal segmental glomerulosclerosis. Kidney Int. 2010;78:296–302.
Gulati S, Sharma AP, Sharma RK, et al. Changing trends of histopathology in childhood nephrotic syndrome. Am J Kidney Dis. 1999;3:646–650.
Haas M, Spargo B, Coventry S. Increasing incidence of focal-segmental glomerulosclerosis among adult nephropathies: A 20-year renal biopsy study. Am J Kidney Dis. 1995;26:740–750.
Hogg R, Middleton J, Vehaskari VM. Focal segmental glomerulosclerosis—epidemiology aspects in children and adults. Pediatr Nephrol. 2007;22:183–186.
Ijpelaar DH, Farris AB, Goemaere N, et al. Fidelity and evolution of recurrent FSGS in renal allografts. J Am Soc Nephrol. 2008;19:2219–2224.
Smeets B, Stucker F, Wetzels J, et al. Detection of activated parietal epithelial cells on the glomerular tuft distinguishes early focal segmental glomerulosclerosis from minimal change disease. Am J Pathol. 2014;184:3239–3248.
Smith SM, Hoy WE, Cobb L. Low incidence of glomerulosclerosis in normal kidneys. Arch Pathol Lab Med. 1989;113:1253–1256.
Rossini M, Fogo A. Interpreting segmental glomerular sclerosis. Curr Diagn Pathol. 2004;10:1–10.
Boute N, Gribouval O, Roselli S, et al. NPHS2, encoding the glomerular protein podocin, is mutated in autosomal recessive steroid-resistant nephrotic syndrome. Nat Genet. 2000;24:349–354.
Brown EJ, Schlöndorff JS, Becker DJ, et al. Mutations in the formin gene INF2 cause focal segmental glomerulosclerosis. Nat Genet. 2010;42:72-76.
Genovese G, Friedman DJ, Ross MD, et al. Association of trypanolytic ApoL1 variants with kidney disease in African Americans. Science. 2010;329:841–845.
Hildebrandt F, Heeringa SF. Specific podocin mutations determine age of onset of nephrotic syndrome all the way into adult life. Kidney Int. 2009;75:669–771.
Kaplan JM, Kim SH, North KN, et al. Mutations in ACTN4, encoding alpha-actinin-4, cause familial focal segmental glomerulosclerosis. Nat Genet. 2000;24:251–256.
Karle SM, Uetz B, Ronner V, et al. Novel mutations in NPHS2 detected in both familial and sporadic steroid-resistant nephrotic syndrome. J Am Soc Nephrol. 2002;13:388–393.
Lovric S, Ashraf S, Tan W, Hildebrandt F. Genetic testing in steroid-resistant nephrotic syndrome: When and how? Nephrol Dial Transplant. 2016;31:1802–1813.
Ruf RG, Lichtenberger A, Karle SM, et al. Arbeitsgemeinschaft für Pädiatrische Nephrologie Study Group: Patients with mutations in NPHS2 (podocin) do not respond to standard steroid treatment of nephrotic syndrome. J Am Soc Nephrol. 2004;15:722–732.
Sadowski CE, Lovric S, Ashraf S, et al. A single-gene cause in 29.5% of cases of steroid-resistant nephrotic syndrome. J Am Soc Nephrol. 2015;26:1279–1289.
Winn MP, Conlon PJ, Lynn KL, et al. A mutation in the TRPC6 cation channel causes familial focal segmental glomerulosclerosis. Science. 2005;308:1801–1804.
Collapsing glomerulopathy generally has a poor prognosis, with marked proteinuria, rapid loss of renal function, and virtually no responsiveness to corticosteroids alone. Of note, if detected early, prognosis may be less grim. This lesion occurs in both Caucasians and African Americans, with strong African American preponderance, about 85% in a large US-based series. The incidence of this lesion varies in different geographic regions. In New York, the incidence has increased from 11% of all cases of idiopathic FSGS from 1979 to 1985 to 20% of this group from 1986 to 1989 and to 24% of idiopathic FSGS from 1990 to 1993. In a large renal biopsy practice centered in Chicago, the collapsing variant accounted for only 4.7% of FSGS biopsies.
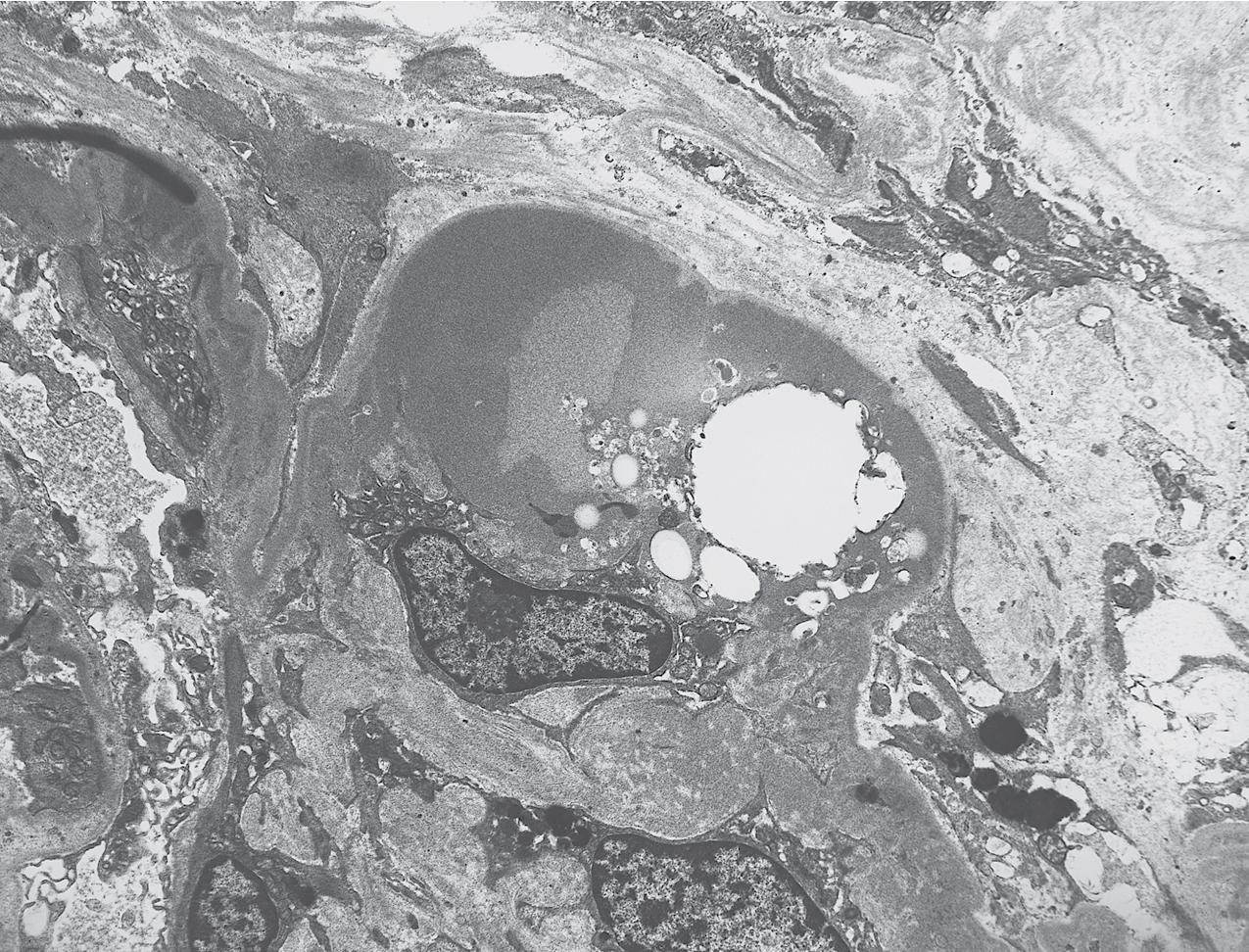
By light microscopy, there is glomerular tuft collapse (segmental or global) and overlying podocyte hyperplasia and hypertrophy ( Fig. 3.22 ). Collapsing lesions are more often global than segmental ( Figs. 3.23–3.24 ; see also Table 3.1 ). Segmental lesions may involve any portion of the glomerulus ( Fig. 3.25 ). There are frequent marked protein droplets in the hypertrophied visceral epithelial cells ( Fig. 3.26 ). Adhesions and hyalinosis are uncommon in the early stage of the lesion, as are mesangial hypercellularity and glomerulomegaly. Involvement of even a single glomerulus with this collapsing lesion is proposed to warrant classification as collapsing glomerulopathy, with its attendant poor prognosis ( Fig. 3.27 ). Other types of segmental sclerosis (see Table 3.1 ) may coexist. Differentiation of cellular or collapsing-type FSGS from usual, NOS FSGS may be difficult in some cases ( Fig. 3.28 ). Vessels do not show specific lesions. Tubules show injury disproportionate to the sclerosis with microcystic change ( Fig. 3.29 ), and there is interstitial inflammation.
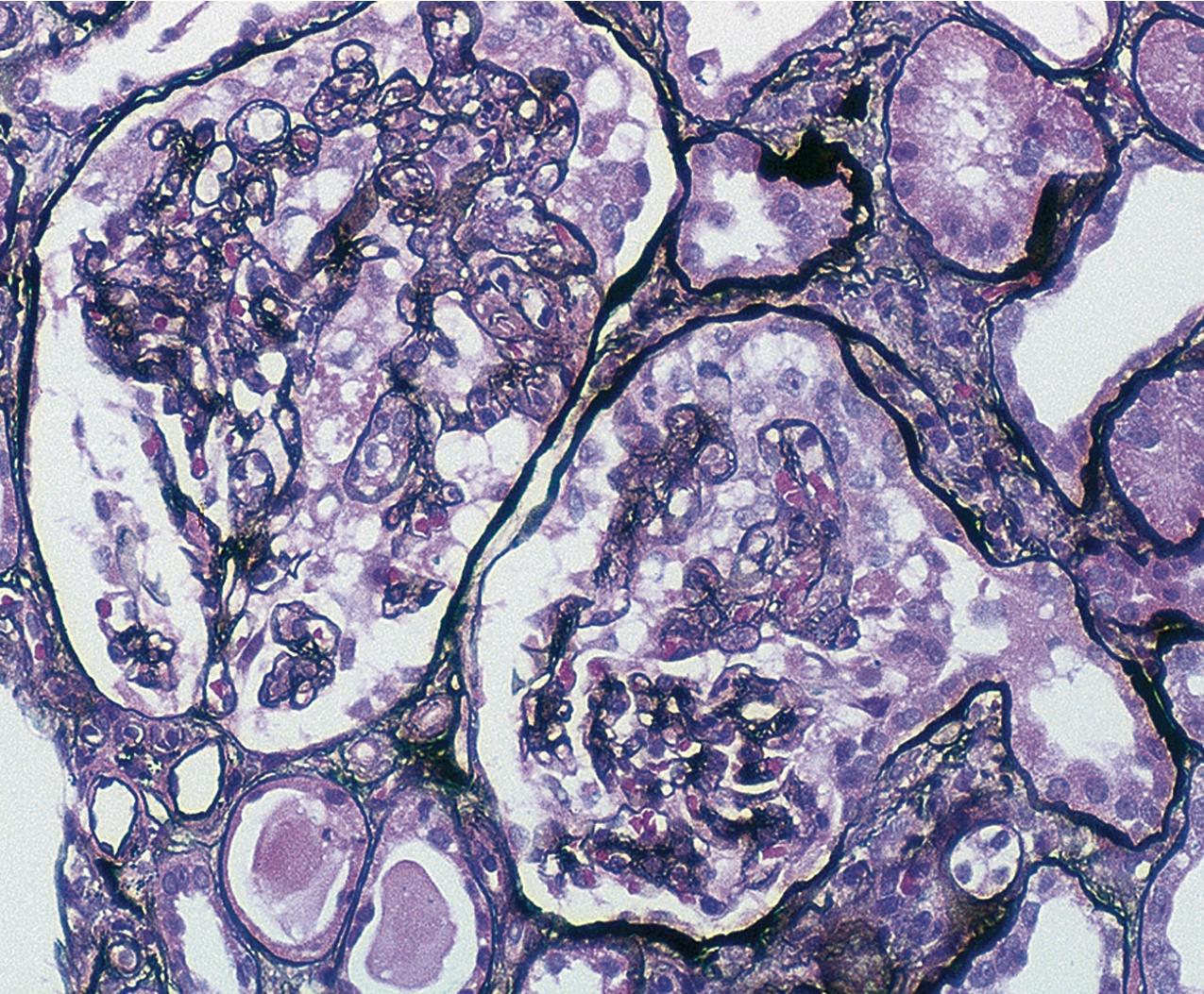
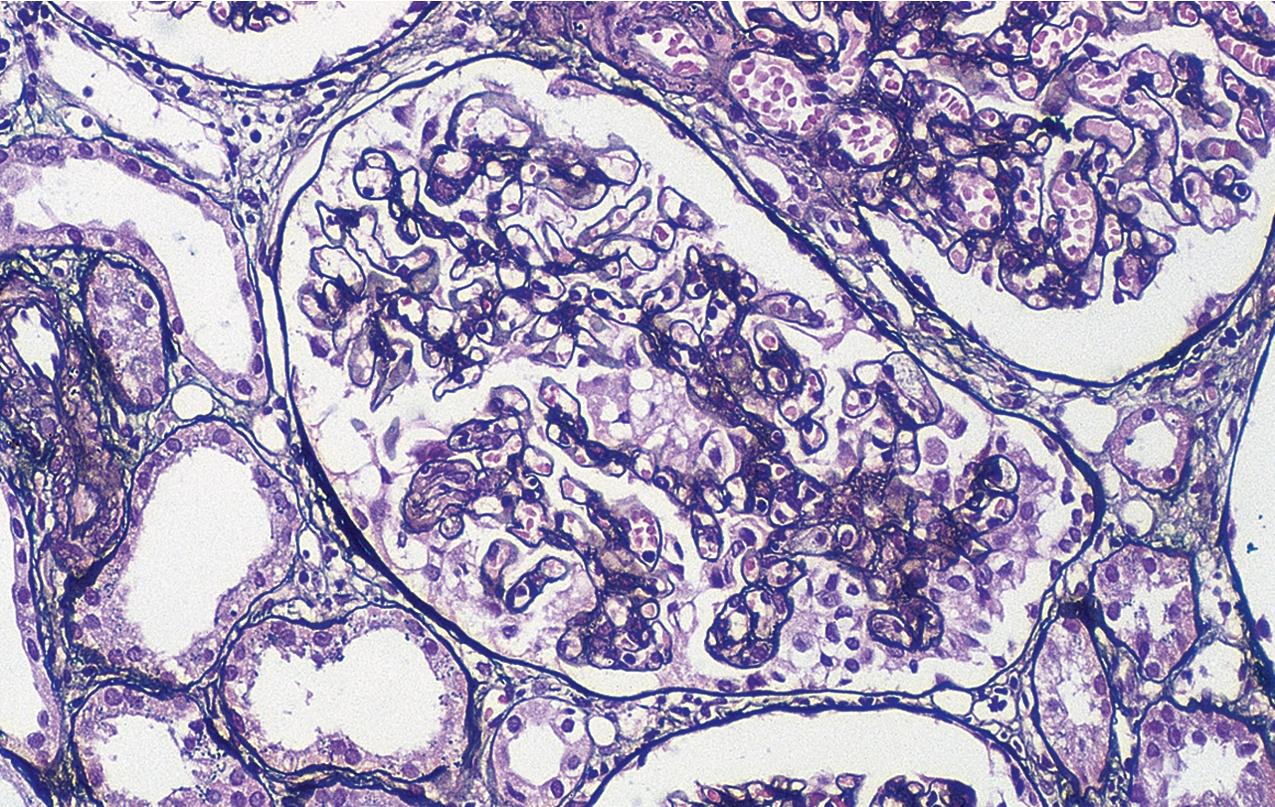
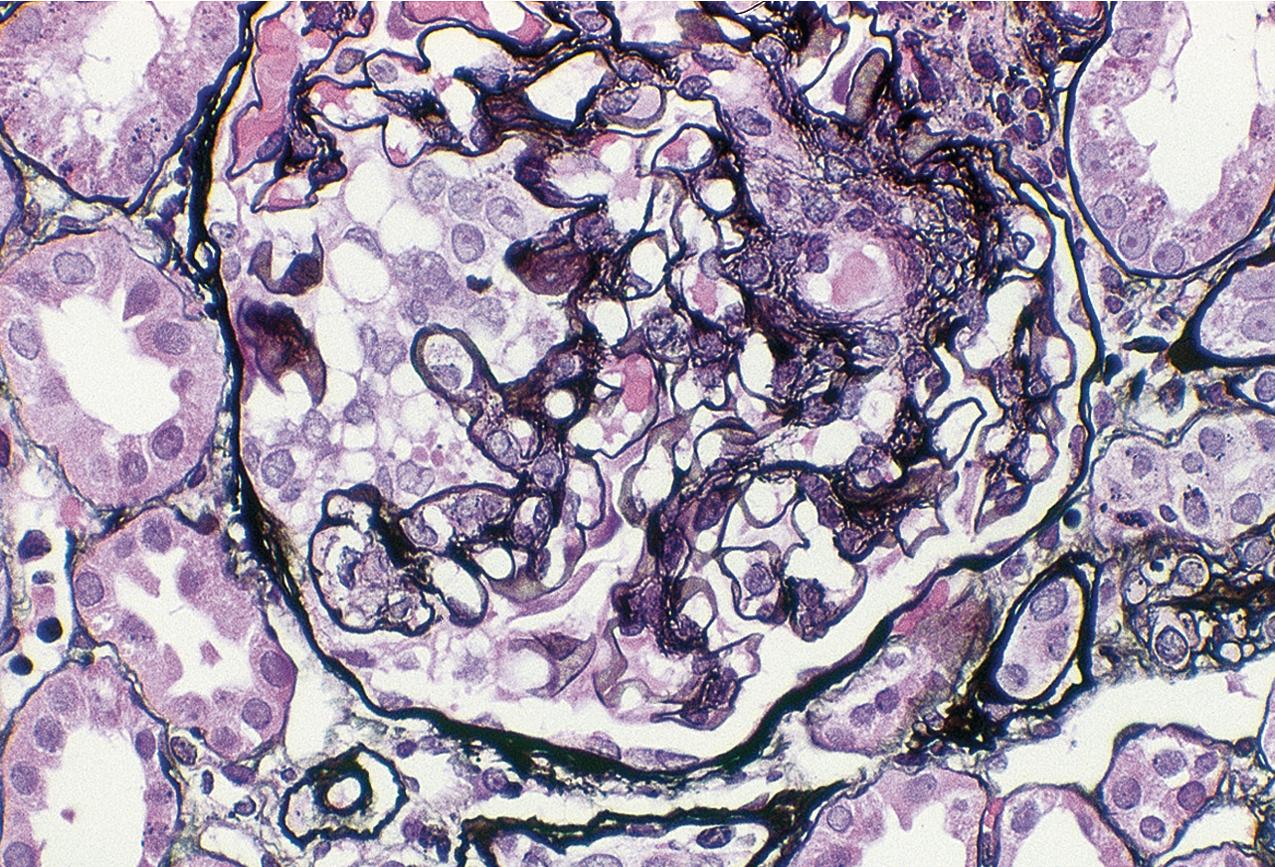
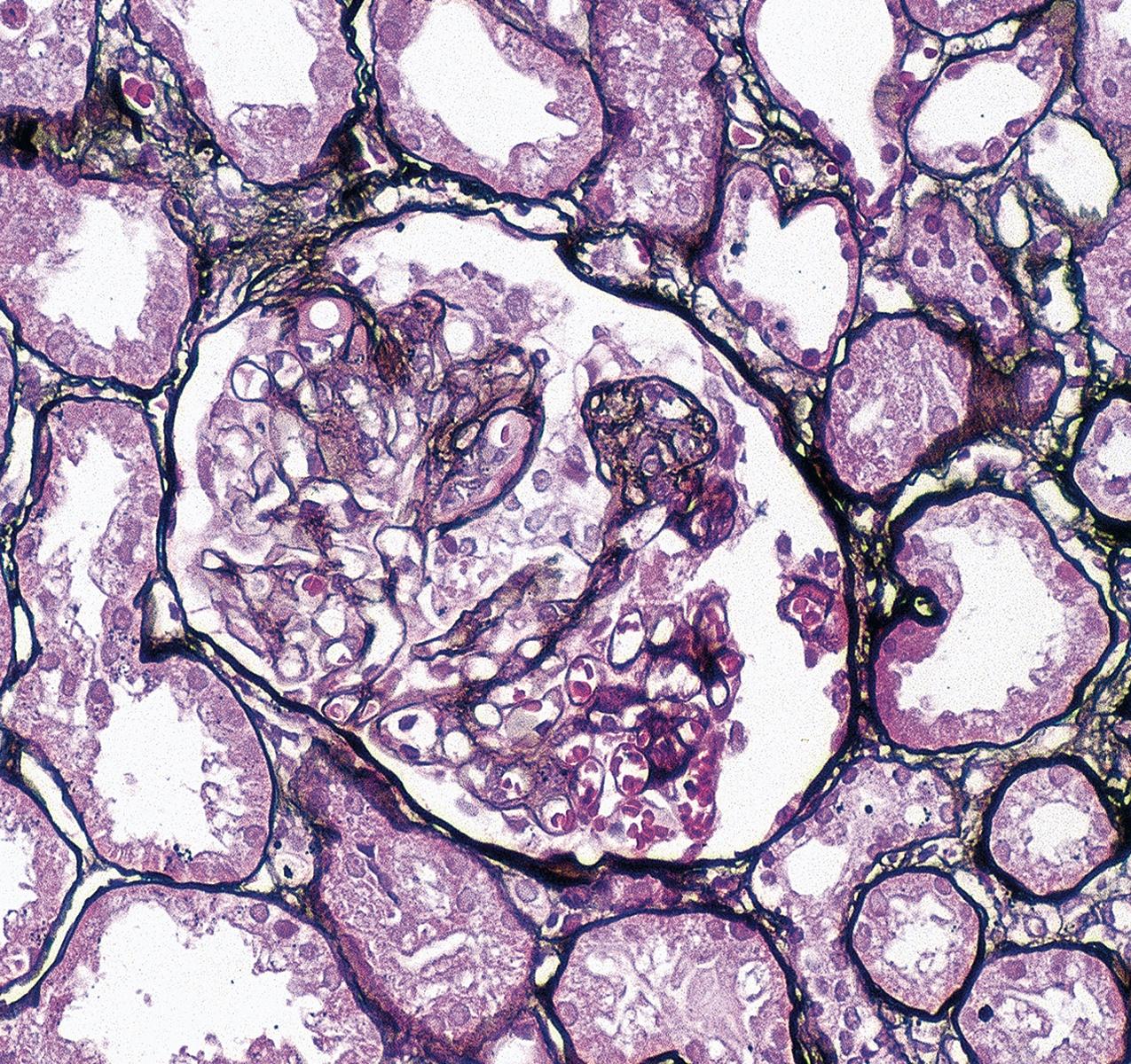
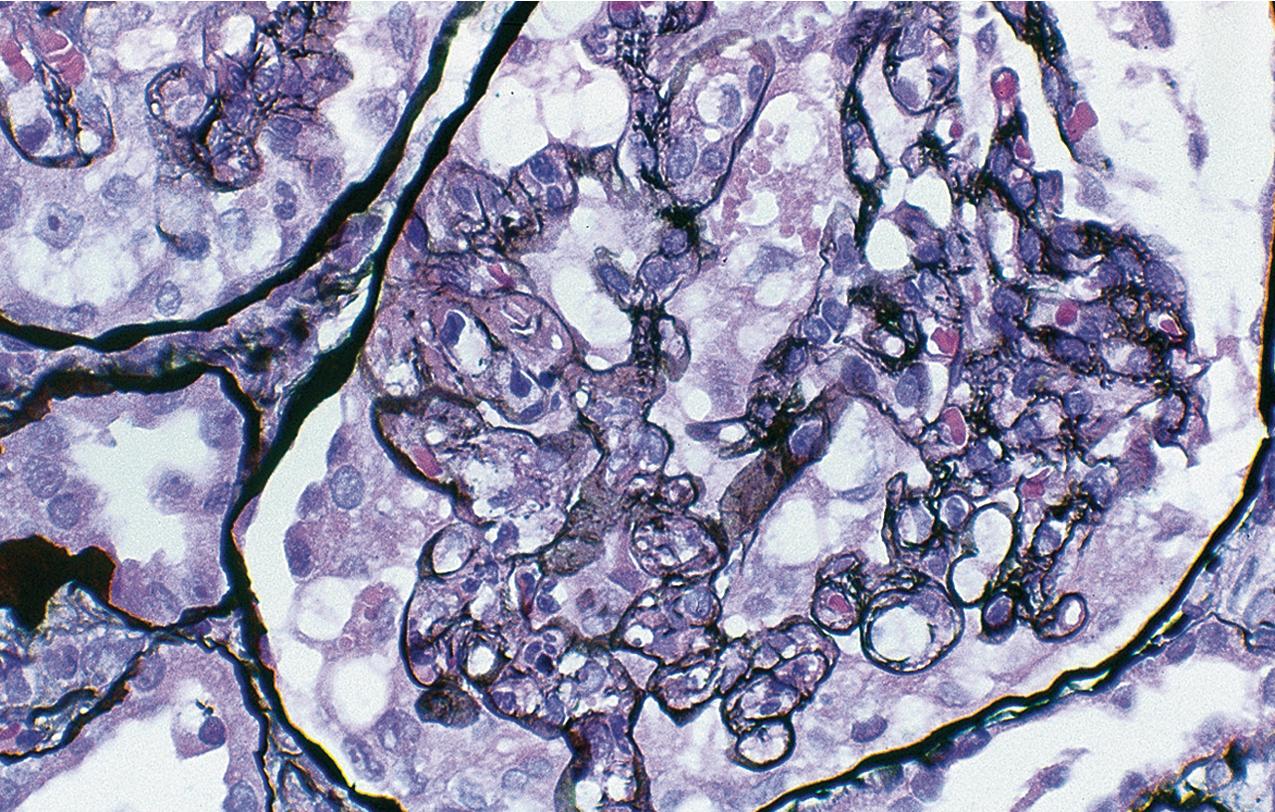
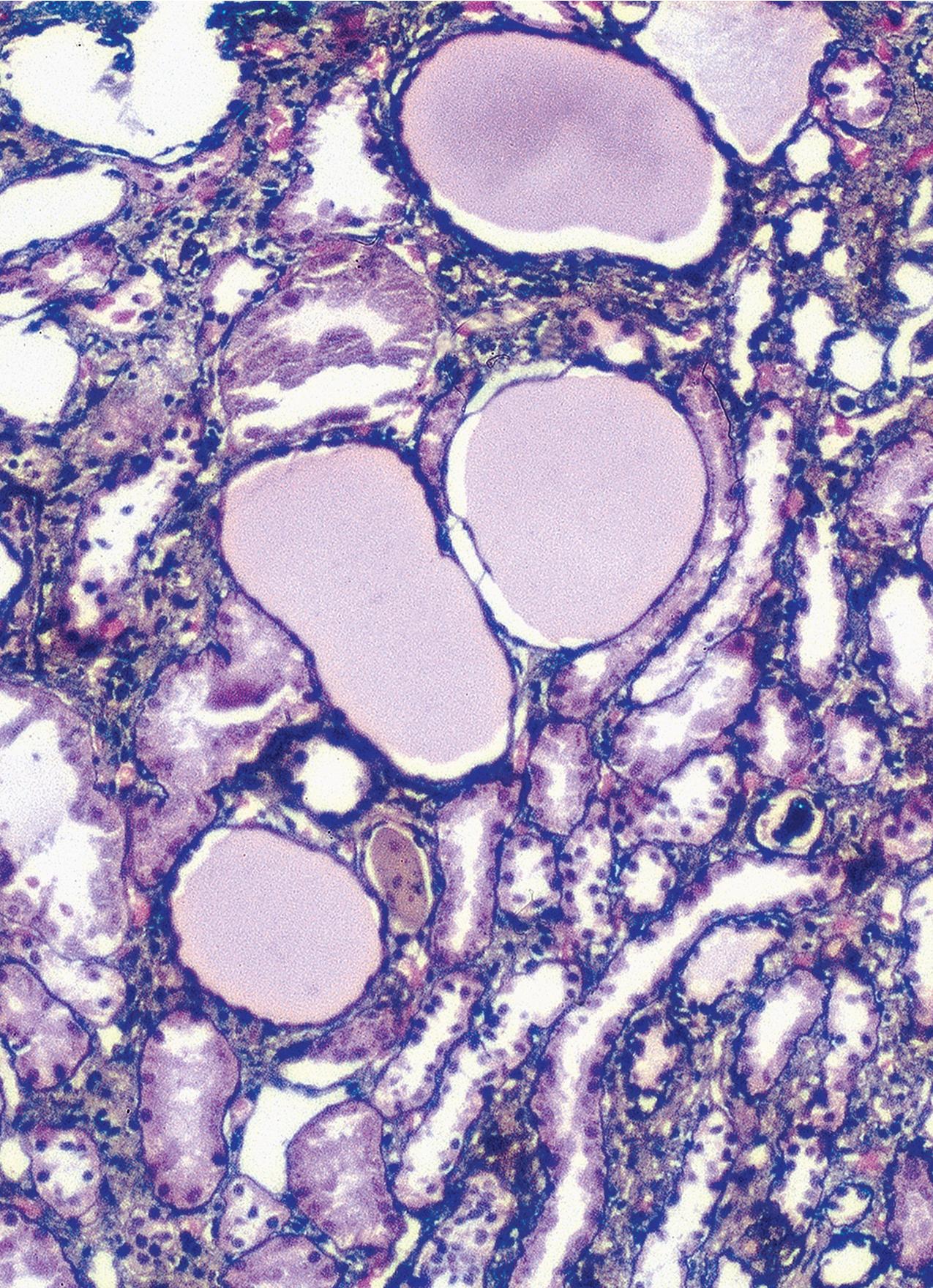
IF may show IgM and C3 in sclerotic segments. EM shows the wrinkled, collapsed GBM and overlying visceral epithelial cell hypertrophy/hyperplasia with frequent vacuoles and protein droplets. No immune complexes are present ( Fig. 3.30 ). Reticular aggregates are not present in idiopathic collapsing glomerulopathy.
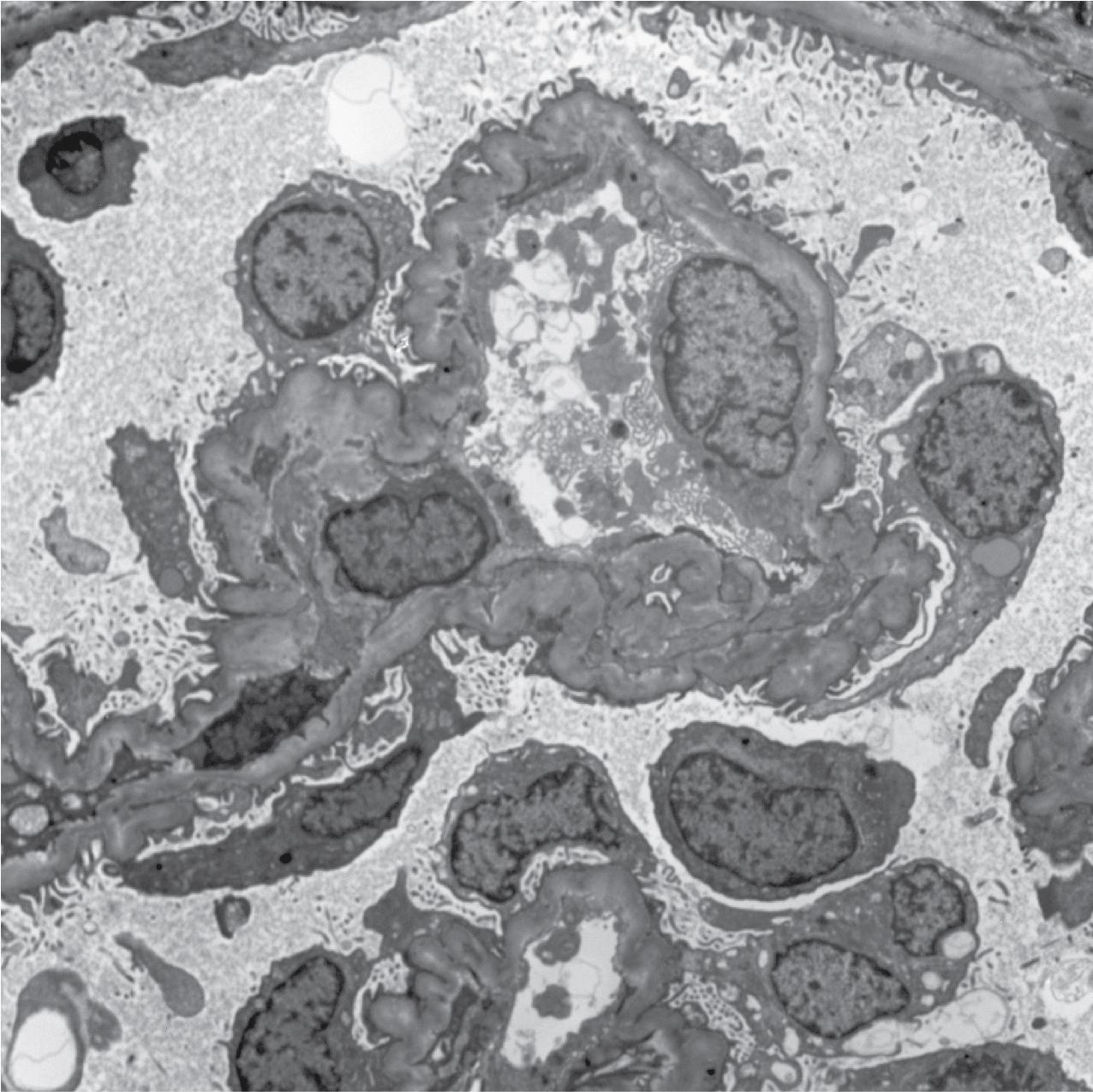
Mature podocytes do not usually proliferate because of high expression of cyclin-dependent kinase inhibitor p27kip1. In collapsing glomerulopathy and HIVAN, p27kip1 expression is lost in areas of collapse, with proliferation and dedifferentiation, with expression of parietal epithelial cell markers. These observations point to a dysregulated phenotype of these epithelial cells in the pathogenesis of these disorders. Parietal epithelial cells contribute to this hyperplasia and may also migrate along the GBM to replace injured podocytes. The etiology of collapsing glomerulopathy has not yet been defined. Increased risk is associated with presence of homozygous or compound heterozygous states of the apolipoprotein ( APOL1) risk alleles G1 or G2 rather than G0. The excess incidence of these risk alleles in African Americans is postulated to have evolved because it confers protection against a strain of trypanosomiasis. How these risk alleles of APOL1 might predispose to podocyte damage and collapsing glomerulopathy is not determined, with experimental evidence suggesting increased susceptibility of the glomerular epithelial cells to a second hit.
Possible viral agents other than HIV have also been proposed to cause collapsing glomerulopathy. Evidence of parvovirus infection was more frequent in patients with collapsing glomerulopathy compared with controls, usual-type FSGS, or HIVAN, suggesting an association. Possible linkage to cytomegalovirus has been reported. Collapsing glomerulopathy has developed in some patients with SARS-CoV-2, with respiratory manifestations and then marked proteinuria. This lesion appears to particularly occur in SARS-CoV-2 patients with APOL1 risk allele variants. Treatment with pamidronate or interferon or use of anabolic steroids has been linked to development of collapsing glomerulopathy. Some patients with LN have collapsing lesions, particularly associated with diffuse proliferative lesions of class IV International Society of Nephrology/ Renal Pathology Society (ISN/RPS) LN (see section on “Lupus Nephritis”). Severe ischemia, linked to, for example, severe hypoperfusion, cocaine use, cyclosporine treatment, or thrombotic microangiopathy of varying etiology, has been associated with collapsing lesions. Collapsing glomerular lesions in native kidneys have been noted in a zonal distribution associated with severe vascular injury. Recurrence of collapsing glomerulopathy in the transplant has been reported. De novo collapsing glomerulopathy has also been noted in the transplant, linked to calcineurin inhibitor toxicity.
Barisoni L, Kriz W, Mundel P, et al. The dysregulated podocyte phenotype: A novel concept in the pathogenesis of collapsing idiopathic focal segmental glomerulosclerosis and HIV-associated nephropathy. J Am Soc Nephrol. 1999;10:51–56.
Buob D, Decambron M, Gnemmi V, et al. Collapsing glomerulopathy is common in the setting of thrombotic microangiopathy of the native kidney. Kidney Int. 2016;90:1321–1331.
Detwiler RK, Falk RF, Hogan SL, et al. Collapsing glomerulopathy: A clinically and pathologically distinct variant of focal segmental glomerulosclerosis. Kidney Int. 1994;45:1416–1424.
Genovese G, Friedman DJ, Ross MD, et al. Association of trypanolytic APOL1 variants with kidney disease in African Americans. Science. 2010;329:841–845.
Larsen CP, Bourne TD, Wilson JD, et al. Collapsing glomerulopathy in a patient with coronavirus disease 2019 (COVID-19). Kidney Int Rep. 2020;5:935–939.
Lasagni L, Romagnani P. Glomerular epithelial stem cells: The good, the bad, and the ugly. J Am Soc Nephrol. 2010;21:1612–1619.
Laurinavicius A, Hurwitz S, Rennke HG. Collapsing glomerulopathy in HIV and non-HIV patients: A clinicopathological and follow-up study. Kidney Int. 1999;56:2203–2213.
Markowitz GS, Appel GB, Fine PL, et al. Collapsing focal segmental glomerulosclerosis following treatment with high-dose pamidronate. J Am Soc Nephrol. 2001;12:1164–1172.
Markowitz GS, Nasr SH, Stokes MB, et al. Treatment with IFN-α, -β, or -γ is associated with collapsing focal segmental glomerulosclerosis. Clin J Am Soc Nephrol. 2010;5:607–615.
Moudgil A, Nast CC, Bagga A, et al. Association of parvovirus B19 infection with idiopathic collapsing glomerulopathy. Kidney Int. 2001;59:2126–2133.
Nicholas Cossey L, Larsen CP, Liapis H. Collapsing glomerulopathy: A 30-year perspective and single, large center experience. Clin Kidney J. 2017;10:443–449.
Valeri A, Barisoni L, Appel GB, et al. Idiopathic collapsing focal segmental glomerulosclerosis: a clinicopathologic study. Kidney Int. 1996;50:1734–1746.
Wu H, Larsen CP, Hernandez-Arroyo CF, et al. AKI and collapsing glomerulopathy associated with COVID-19 and APOL1 high-risk genotype. J Am Soc Nephrol. 2020;31:1688–1695.
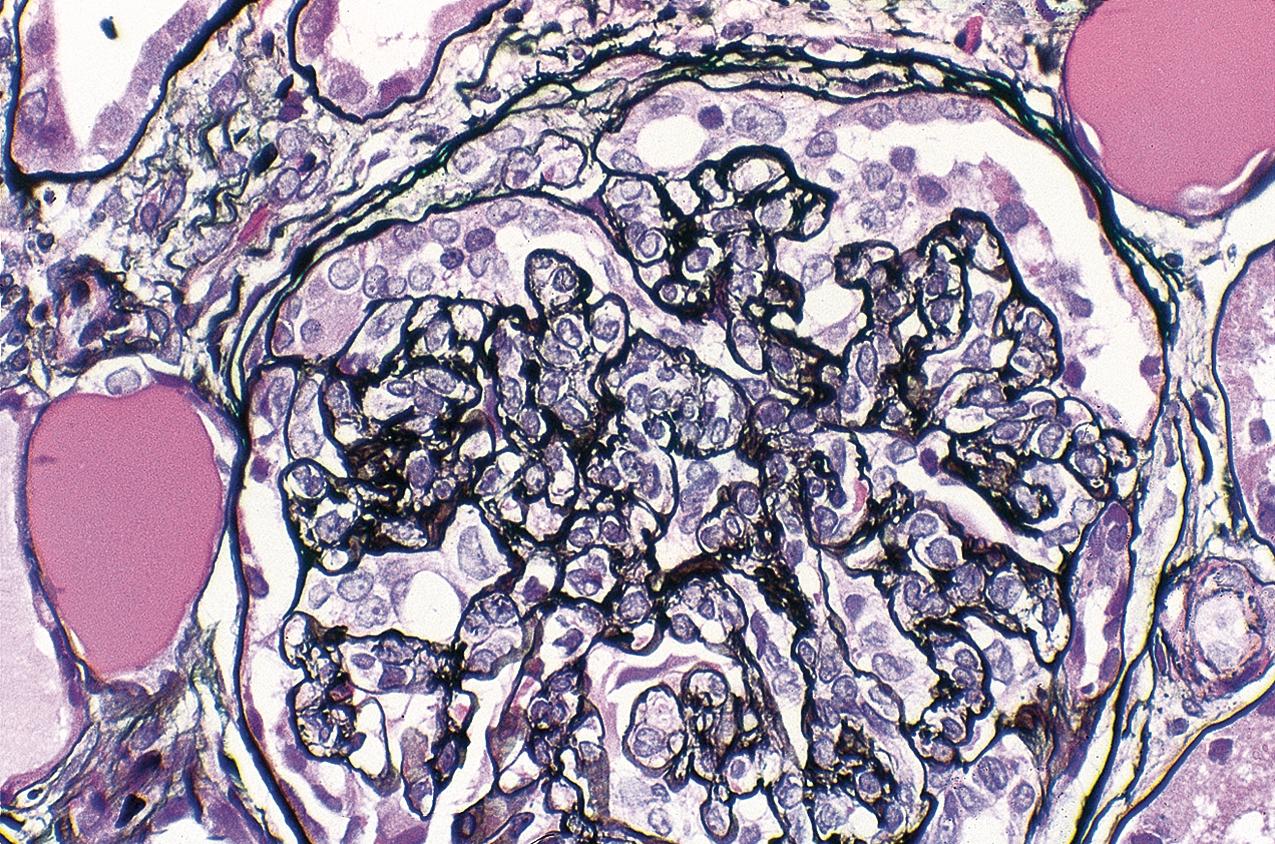
Patients with tip lesion variant of FSGS present with nephrotic syndrome. This lesion generally presents with early lesion with good prognosis similar to MCD and response to immunosuppression, with complete remission in nearly 60% and partial remission in about 14% of patients in one series. Only a minority of patients develop progressive kidney disease over long-term follow-up.
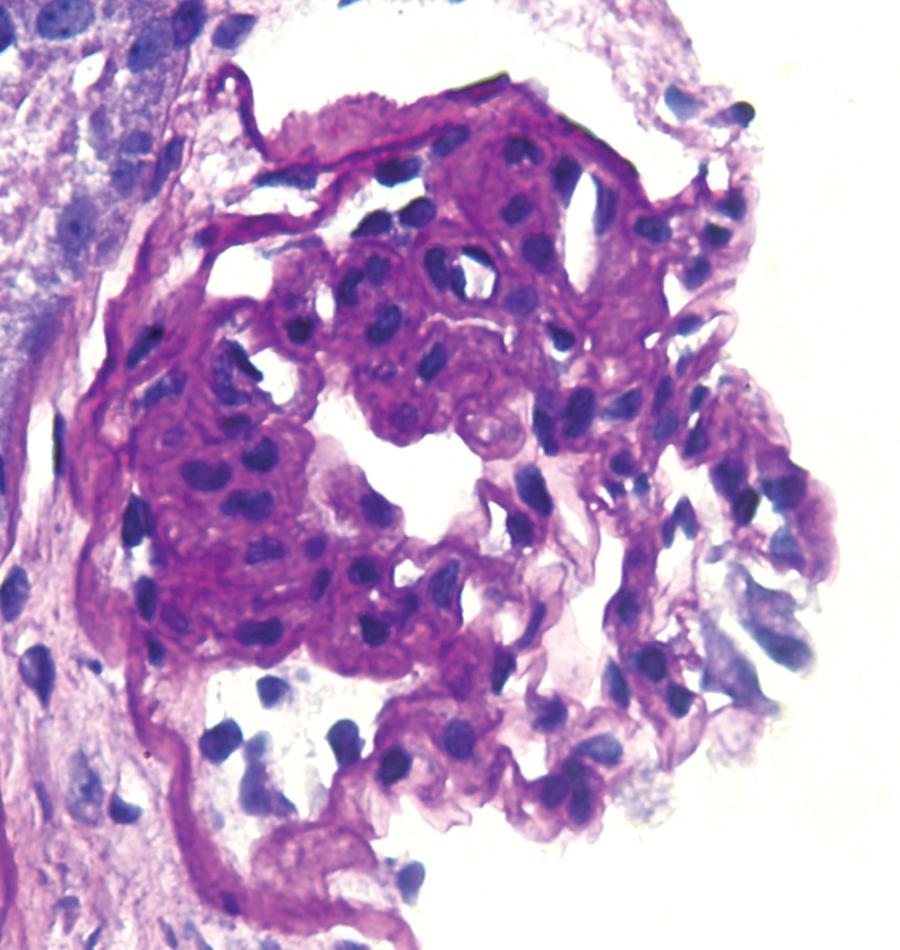
The tip lesion is defined as glomerulosclerosis involving only the tubular pole of the glomerulus ( Fig. 3.31 ). The collapsing glomerulopathy variant must be excluded to diagnose tip variant FSGS (see Table 3.1 ). It is defined as the presence of at least one segmental lesion involving the outer 25% of the glomerulus next to the proximal tubule pole with adhesion between the tuft and Bowman’s capsule at the tubule lumen or neck ( Figs. 3.32–3.33 ). Thus the proximal tubule pole must be identified to recognize and diagnose the lesion. Indeed, with additional serial sectioning, some cases of apparent cellular-type peripheral FSGS lesions showed diagnostic tip lesions (12 of 38 cases in one series), resulting in reclassification, and implying better prognosis. The segmental lesion may be characterized by either endocapillary hypercellularity (involving <50% of the tuft) or sclerosis (involving <25% of the tuft). Foam cells are common, but hyalinosis is variable. The involved area often shows podocyte hypertrophy/hyperplasia. Mesangial hypercellularity, glomerulomegaly, and arteriolar hyalinosis are variable. Other glomeruli may show usual segmental lesions (but per definition not hilar location) or cellular lesions according to the Columbia classification. So-called “pure tip lesion,” as originally defined (i.e., when it is the only segmental lesion present), may have even better prognosis. IF and EM findings are as in usual-type FSGS.
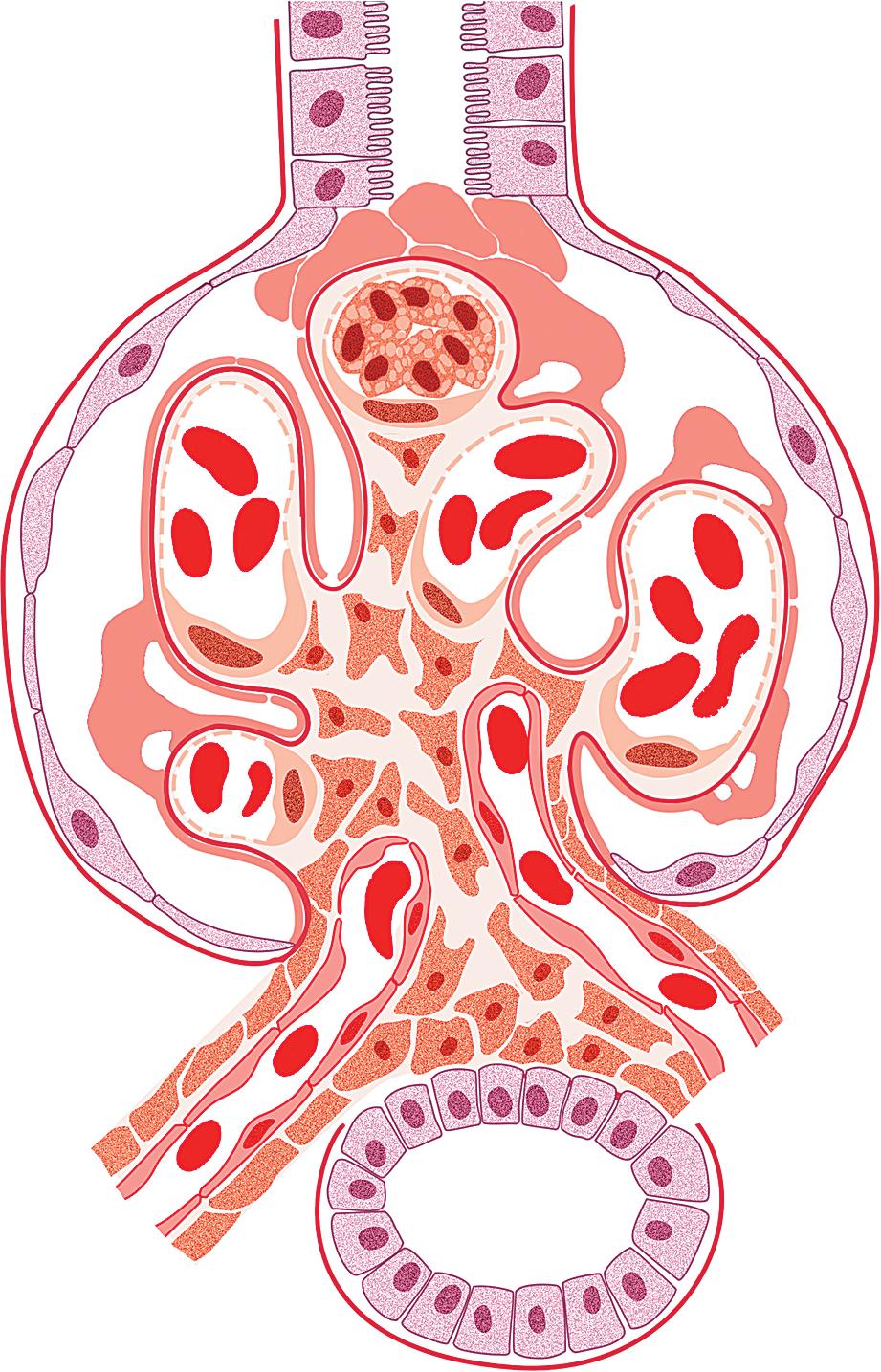
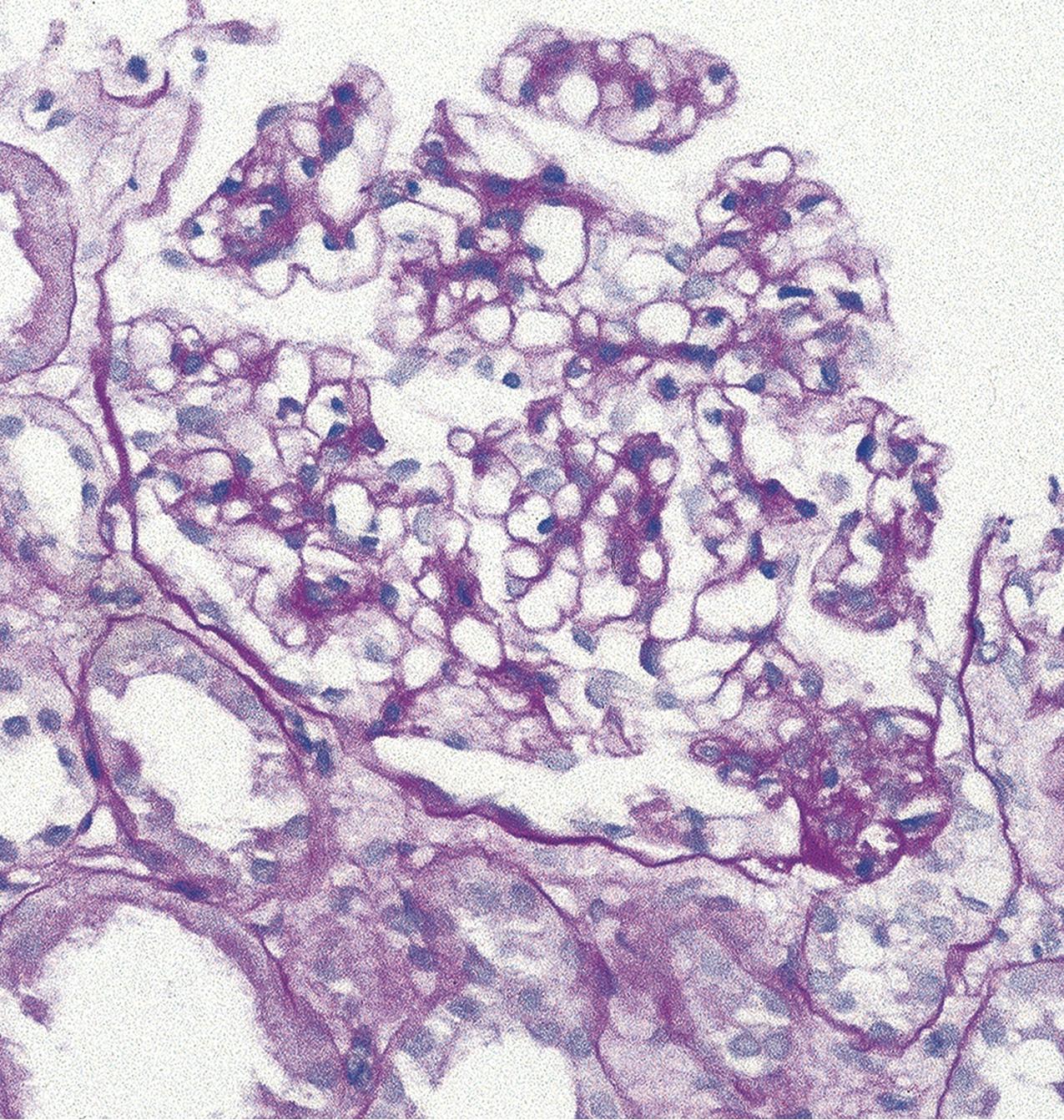
The etiology is unknown. Recent evidence points to a role of intermediate-type parietal epithelial cells in the pathogenesis of this lesion. These cells are between the proximal tubular type cells of the tubular-glomerular junction and the flat squamous cells lining most of Bowman’s capsule. With injury, they are susceptible to activation and appear to contribute to the tip lesions. Hypotheses also include increased turbulence at the tubular outflow causing podocyte injury. Tip lesions may be seen incidentally at autopsy and superimposed in other glomerular diseases and thus are not pathognomonic of idiopathic primary FSGS.
Howie AJ, Brewer DB. Further studies on the glomerular tip lesion: Early and late stages and life table analysis. J Pathol. 1985;147:245–255.
Howie AJ, Pankhurst T, Sarioglu S, et al. Evolution of nephrotic-associated focal segmental glomerulosclerosis and relation to the glomerular tip lesion. Kidney Int. 2005;67:987–1001.
Kuppe C, Leuchtle K, Wagner A, et al. Novel parietal epithelial cell subpopulations contribute to focal segmental glomerulosclerosis and glomerular tip lesions. Kidney Int. 2019;96:80–93.
Stokes MB, Markowitz GS, Lin J, et al. Glomerular tip lesion: A distinct entity within the minimal change disease/focal segmental glomerulosclerosis spectrum. Kidney Int. 2004;65:1690–1702.
Stokes MB, Valeri AM, Markowitz GS, et al. Cellular focal segmental glomerulosclerosis: Clinical and pathologic features. Kidney Int. 2006;70:1783–1792.
Thomas DB, Franceschini N, Hogan SL, et al. Clinical and pathologic characteristics of focal segmental glomerulosclerosis pathologic variants. Kidney Int. 2006;69:920–926.
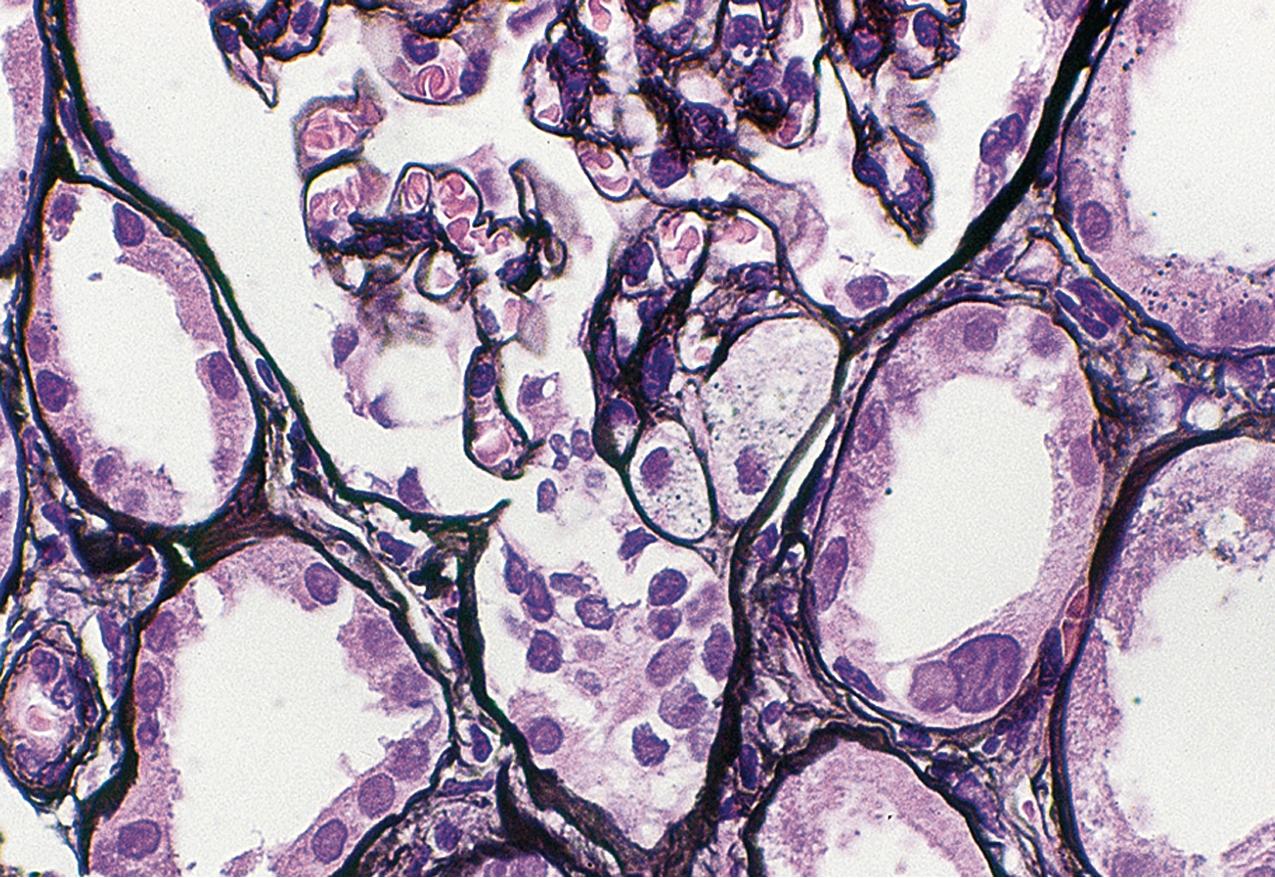
Patients with the cellular variant of FSGS present with abrupt onset of nephrotic syndrome. This lesion is the rarest of the idiopathic FSGS subtypes, and many cases may indeed rather meet criteria for tip lesion variant when extensive sectioning and analyses are performed (see later and the section on “Tip Lesion Focal Segmental Glomerulosclerosis”). To diagnose the cellular variant of FSGS, the FSGS working classification proposes that tip lesion and collapsing glomerulopathy must be excluded (see Table 3.1 ). The cellular variant of FSGS is then defined as at least one glomerulus with endocapillary hypercellularity/proliferation involving at least 25% of the tuft and occluding the lumen ( Figs. 3.34–3.35 ). The endocapillary cells typically include foam cells, macrophages, and endothelial cells. Neutrophils and lymphocytes may also be present. There may be podocyte hyperplasia/hypertrophy overlying this lesion, but unlike in collapsing glomerulopathy, this is not a required feature. These lesions may develop into progressively less cellular, more sclerotic lesions, becoming indistinguishable clinically and morphologically from classic FSGS (see Fig. 3.28 ). Thus other glomeruli in the biopsy may contain usual type of segmental or global glomerulosclerosis. IF and EM findings are as in usual-type FSGS.
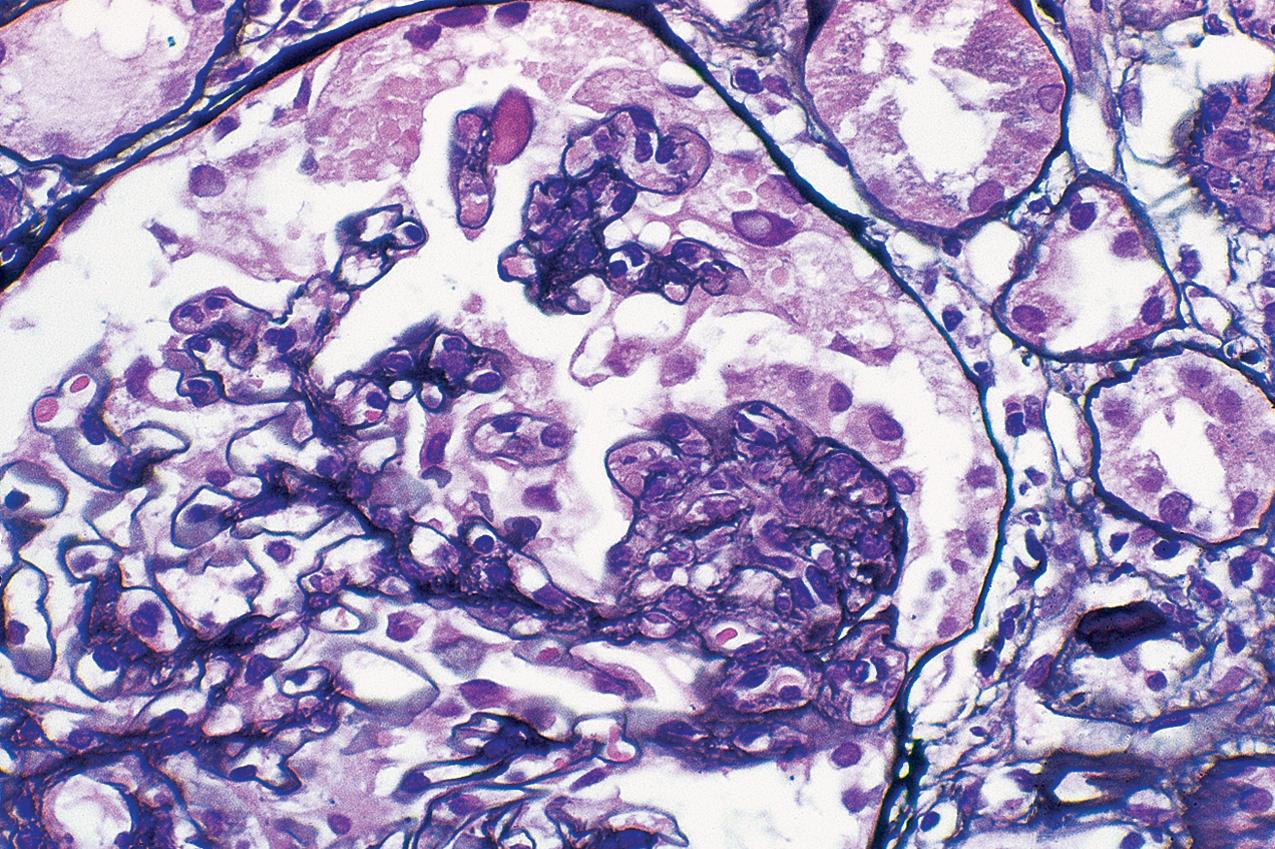
This cellular lesion may be an early abnormality seen by light microscopy when FSGS recurs in the transplant. Thus this morphologic variant is postulated to represent an early, active FSGS lesion. The cellular lesion has also been seen more commonly in children with FSGS than in adults. In some cases of apparent cellular FSGS, additional deeper sections show diagnostic tip lesions. Thus some cases with cellular lesions may rather represent tip lesion FSGS. Cellular variant FSGS overall showed intermediate prognosis compared with collapsing glomerulopathy and tip variant of FSGS.
Schwartz MM, Evans J, Bain R, et al. Focal segmental glomerulosclerosis: Prognostic implications of the cellular lesion. J Am Soc Nephrol. 1999;10:1900–1907.
Silverstein DM, Craver R. Presenting features and short-term outcome according to pathologic variant in childhood primary focal segmental glomerulosclerosis. Clin J Am Soc Nephrol. 2007;2:700–707.
Stokes MB, Valeri AM, Markowitz GS, et al. Cellular focal segmental glomerulosclerosis: Clinical and pathologic features. Kidney Int. 2006;70:1783–1792.
Patients present with proteinuria. Patients may have hypertension or other underlying conditions linked to renal scarring ( Fig. 3.36 ). To diagnose this type, cellular, tip variants of FSGS and collapsing glomerulopathy must first be excluded (see Table 3.1 ). Perihilar-type FSGS is defined by perihilar sclerosis and hyalinosis involving more than 50% of the involved glomeruli with segmental lesions. Glomerulomegaly and adhesions are common. There is often arteriolar hyalinosis, but arteriolar hyaline alone is insufficient for diagnosis ( Figs. 3.37–3.38 ). Mesangial hypercellularity is usually absent, and podocytes do not typically show hyperplasia/hypertrophy. IF and EM findings are as in usual-type FSGS.
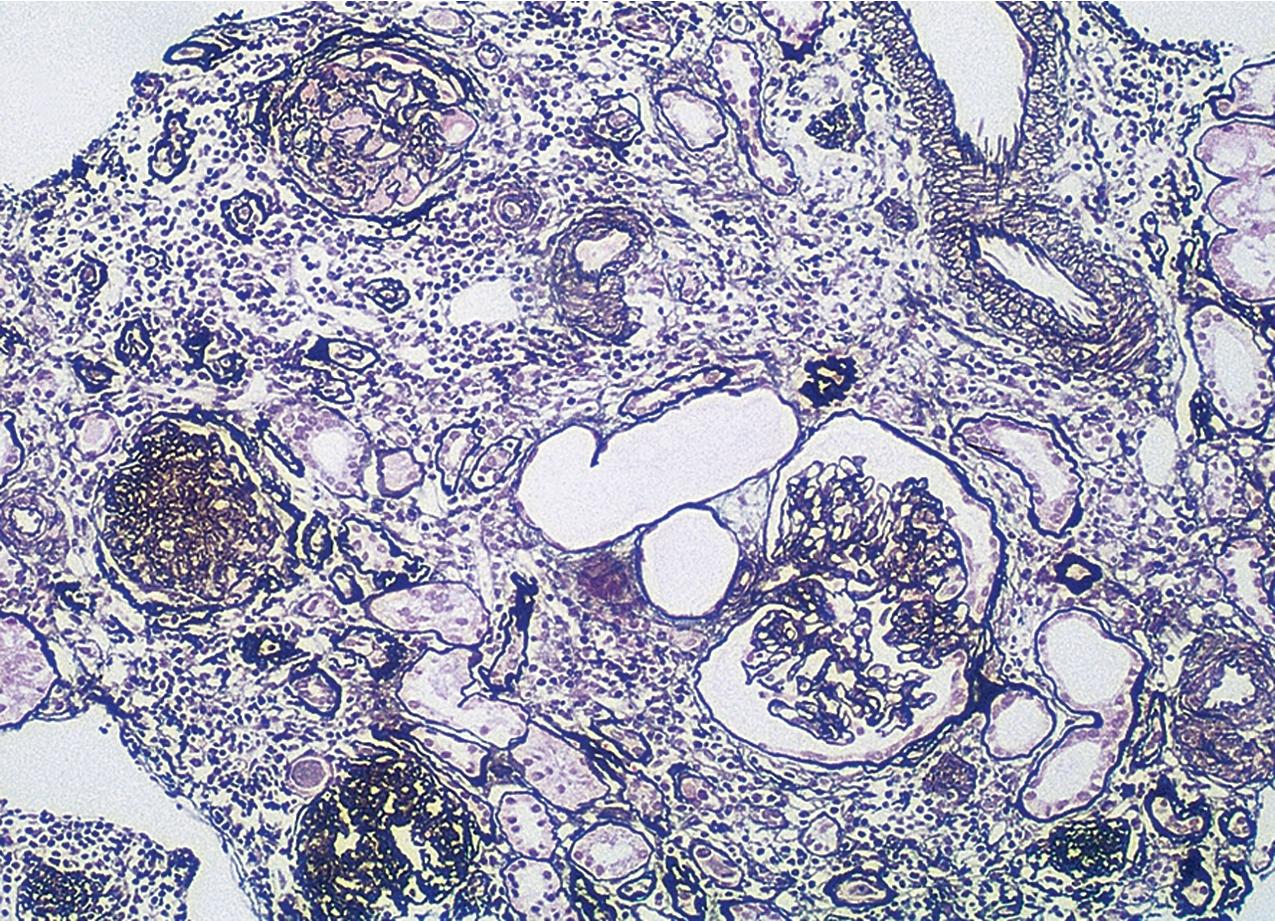
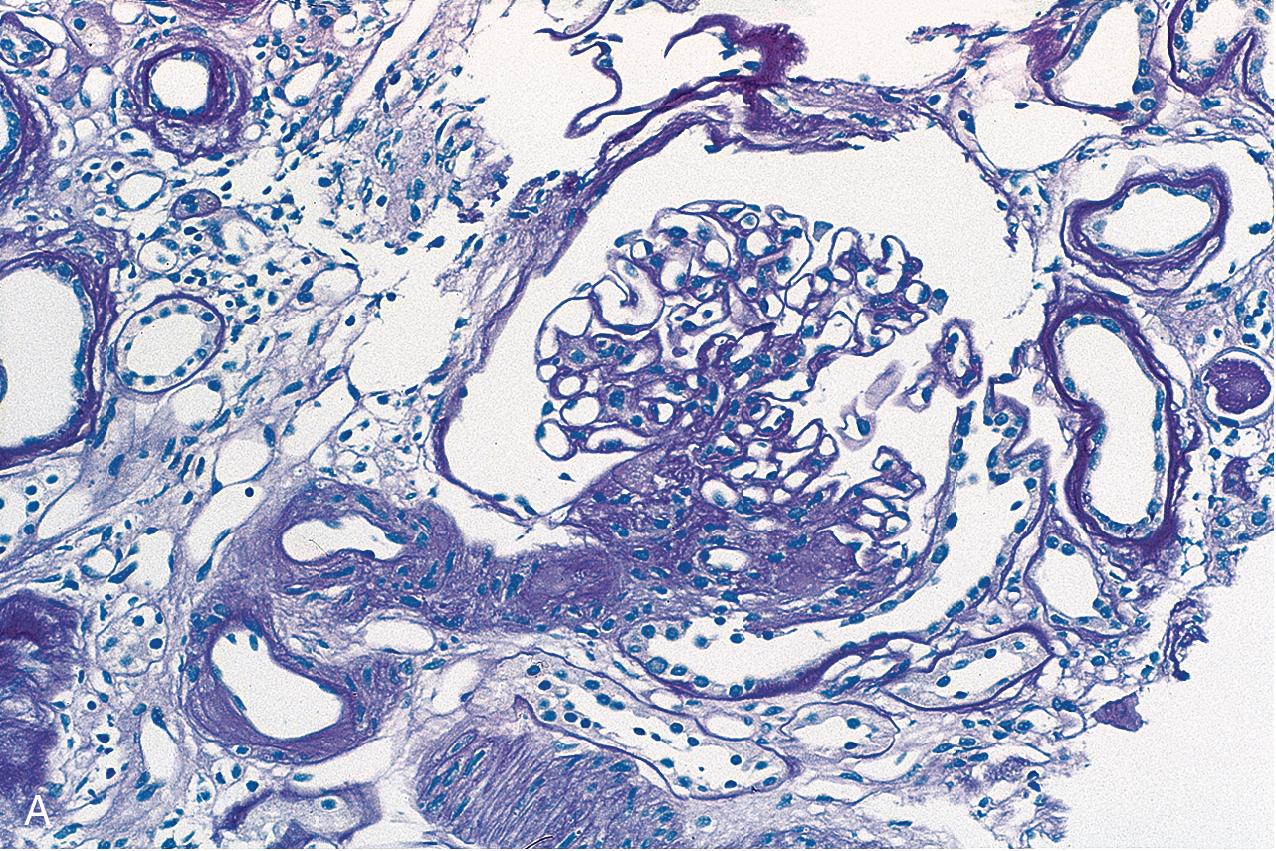
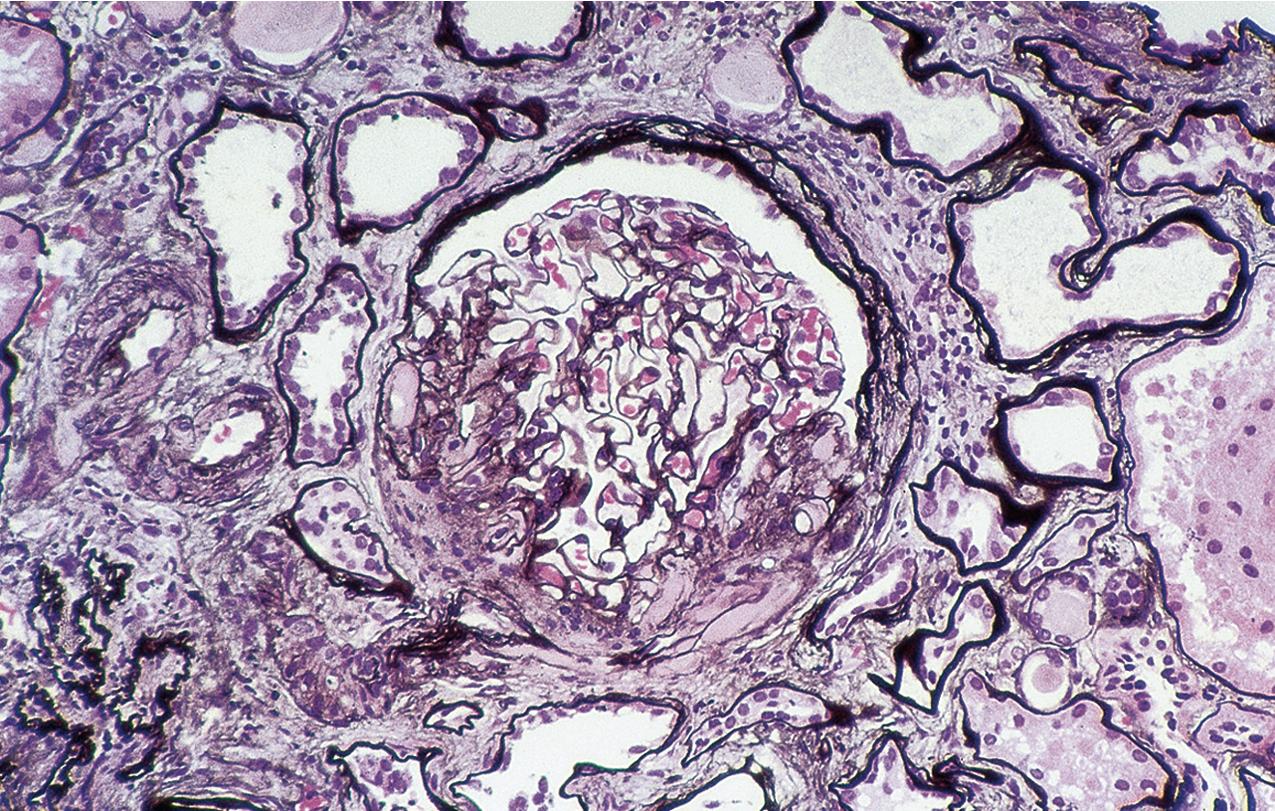
Predominantly perihilar lesions of sclerosis are proposed to represent a response to reduced renal mass. This variant may occur in idiopathic FSGS but is also common in patients with secondary forms of FSGS related to adaptive responses to reduced nephron mass and/or glomerular hemodynamic abnormalities. Many insults to the kidney may result in secondary FSGS, either as the sole manifestation of injury or superimposed on other renal disease manifestations. Lesions of segmental sclerosis in a focal pattern may be seen in association with diseases with abnormal, maladaptive responses of glomerular growth and pressures, such as in diabetes, obesity, heroin abuse, cyanotic heart disease, or sickle cell disease. Thus secondary sclerosis occurs in the chronic stage of many immune complex or proliferative diseases. In some of these settings, the morphologic appearance of sclerosis can indicate the nature of the initial insult: Obesity-associated FSGS shows mild changes related to glucose intolerance (mesangial expansion, GBM thickening), subtotal foot process effacement, and marked glomerulomegaly and may show hilar pattern sclerotic lesions. The course is more indolent than for idiopathic FSGS, with less frequent nephrotic syndrome. Anabolic steroid use has also been linked to FSGS, with some patients showing hilar lesions, whereas others show collapsing lesions. In FSGS lesions secondary to reflux nephropathy, there is frequently prominent periglomerular fibrosis and thickening of Bowman’s capsule and patchy geographic-pattern interstitial scarring, in addition to the heterogeneous glomerulosclerosis. FSGS associated with heroin use does not show pathognomonic features, although global glomerulosclerosis, epithelial cell changes, interstitial fibrosis, and tubular injury tend to be more prominent than in idiopathic cases of FSGS. In some patients, collapsing lesions are present. FSGS also can develop in association with decreased renal mass. The best example is oligomeganephronia, where the nephron number is greatly reduced, with resulting marked enlargement of the remaining glomeruli and occurrence of FSGS. Patients with unilateral renal agenesis show an apparent higher risk for FSGS than the general population. Loss of one kidney later in life does not elicit the same degree of growth response in the remaining kidney as in the young and is less likely to be associated with scarring in the contralateral kidney. Nevertheless, when one kidney and a portion of the other are lost in the adult, patients appear to have an increased risk for developing FSGS lesions. Similarly, low birth weight, with or without prematurity, has been associated with fewer nephrons, presumed to contribute to the linkage with chronic kidney disease and hypertension. FSGS lesions have been reported in some of these patients as well.
Collapsing lesion (even in only one glomerulus) indicates collapsing glomerulopathy
Tip lesion in the absence of collapsing lesion or hilar lesion indicates the tip lesion variant of FSGS.
Cellular lesion in the absence of tip and collapsing features indicates the cellular variant.
Hilar lesion, in the absence of the aforementioned, involving most of the segmentally affected glomeruli indicates hilar variant FSGS
Segmental lesions that do not fit into any of the aforementioned categories, or the standard segmental sclerosis lesion, indicate FSGS, NOS.
Herlitz LC, Markowitz GS, Farris AB, et al. Development of focal segmental glomerulosclerosis after anabolic steroid abuse. J Am Soc Nephrol. 2010;21:163–172.
Hodgin JB, Rasoulpour M, Markowitz GS, et al. Very low birth weight is a risk factor for secondary focal segmental glomerulosclerosis. Clin J Am Soc Nephrol. 2009;4:71–76.
Kambham N, Markowitz GS, Valeri AM, et al. Obesity-related glomerulopathy: An emerging epidemic. Kidney Int. 2001;59:1498–1509.
Rennke HG, Klein PS. Pathogenesis and significance of nonprimary focal and segmental glomerulosclerosis. Am J Kidney Dis. 1989;13:443–456.
CNF is an inherited autosomal recessive disease caused by mutation of the nephrin gene (NPHS1), located on chromosome 19, with incidence around 1 per 10,000 births. The disease is not exclusive to the Finnish population. Nephrotic syndrome manifests at birth or usually by age 3 months and can result in death from complications secondary to nephrotic syndrome by age 1 unless treated with renal transplantation or dialysis. End-stage kidney disease usually develops in early childhood. Microscopic hematuria is often present.
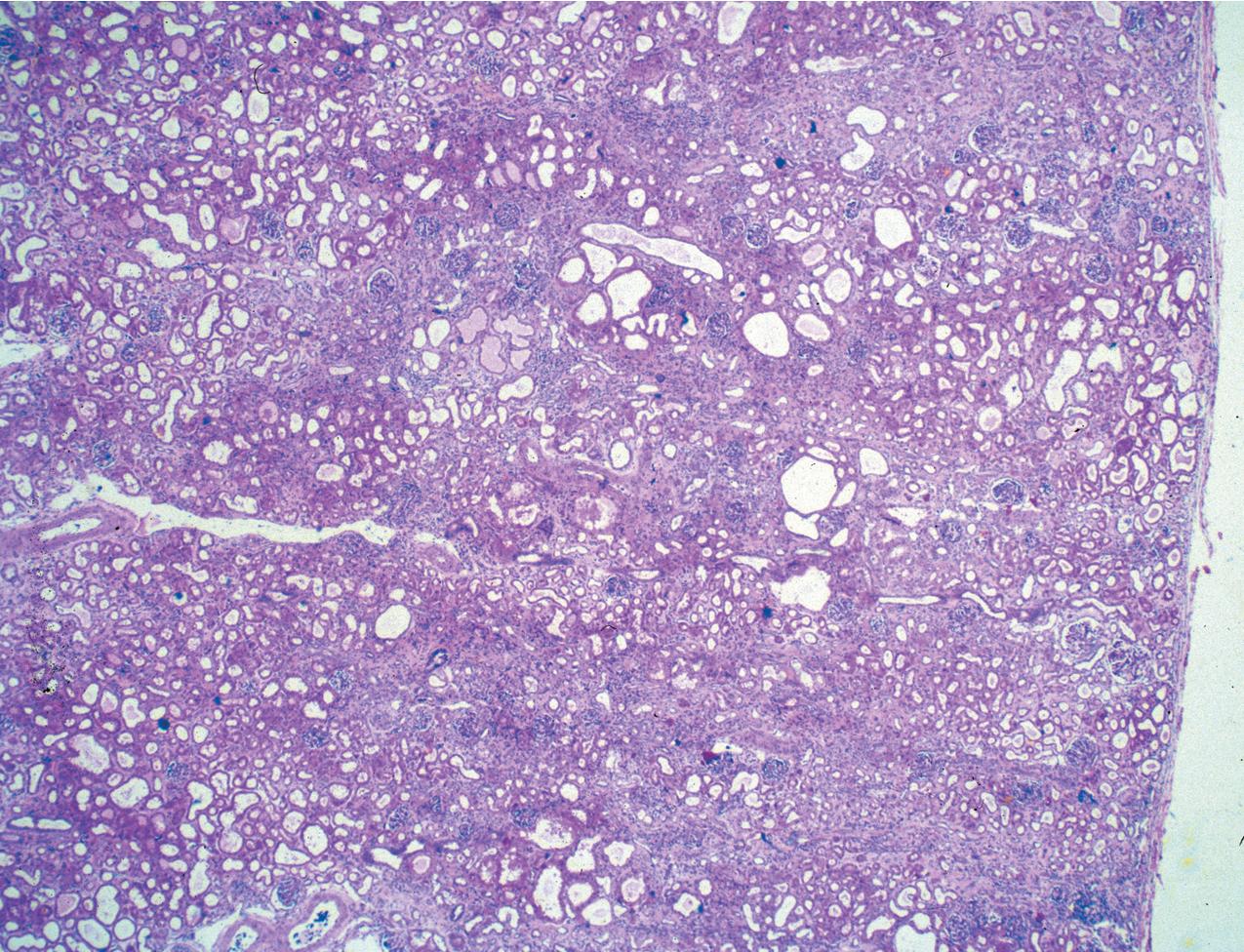
Glomeruli may be immature, more so than expected for term birth, but this may, in part, reflect the usual premature birth of affected infants. Mature glomeruli have variable mesangial cell and matrix increase and nonspecific sclerosis and occasional proliferation ( Fig. 3.39 ). Occasional crescent-type extracapillary proliferation may be present but without necrosis. Glomeruli may also be unremarkable by light microscopy. The proximal tubules are dilated ( Figs. 3.40–3.42 ). Tubules may show atrophy, and Bowman’s capsule may be dilated in some cases, although collecting ducts are not typically dilated. Of note, the typical proximal tubular lesions of dilation may be absent in early biopsies. Glomerulosclerosis with proportional tubular atrophy and interstitial fibrosis develops late in the course ( Fig. 3.43 ). Vessels show no specific lesions.
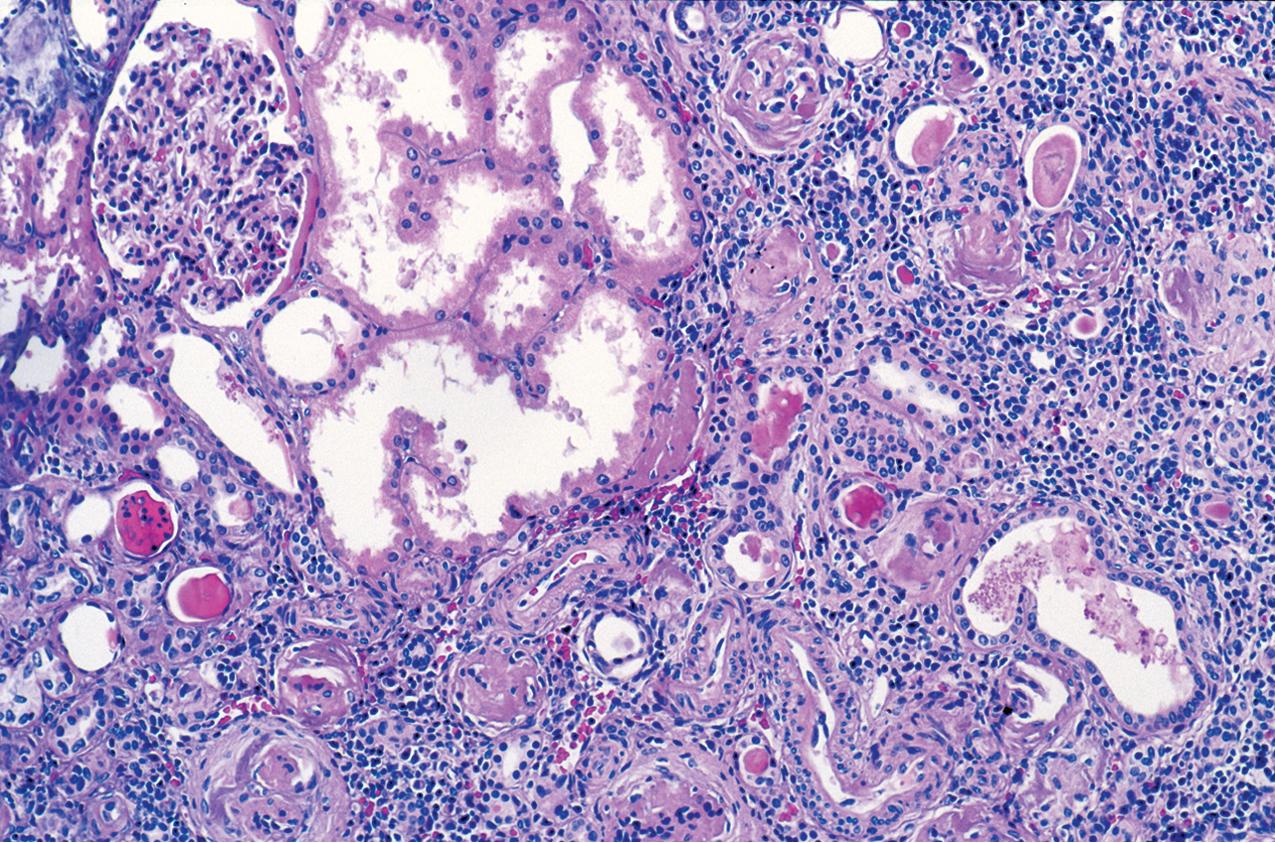
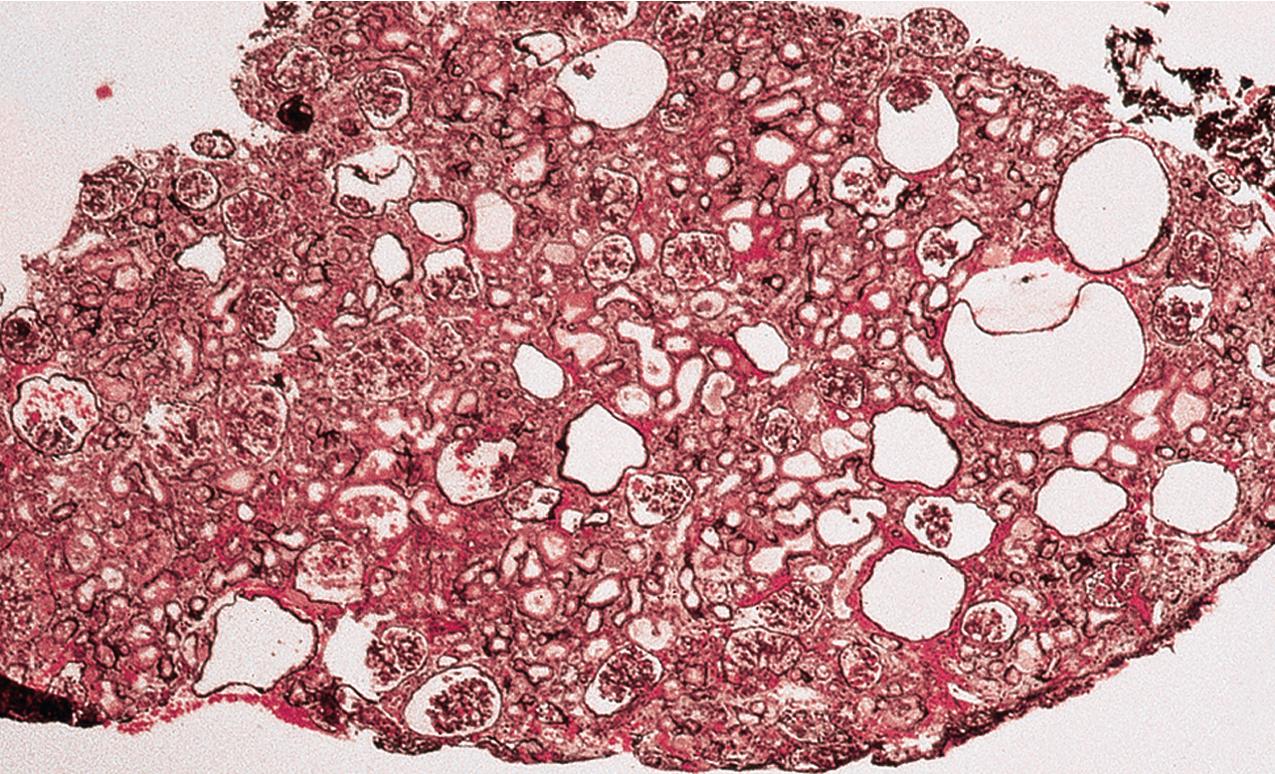
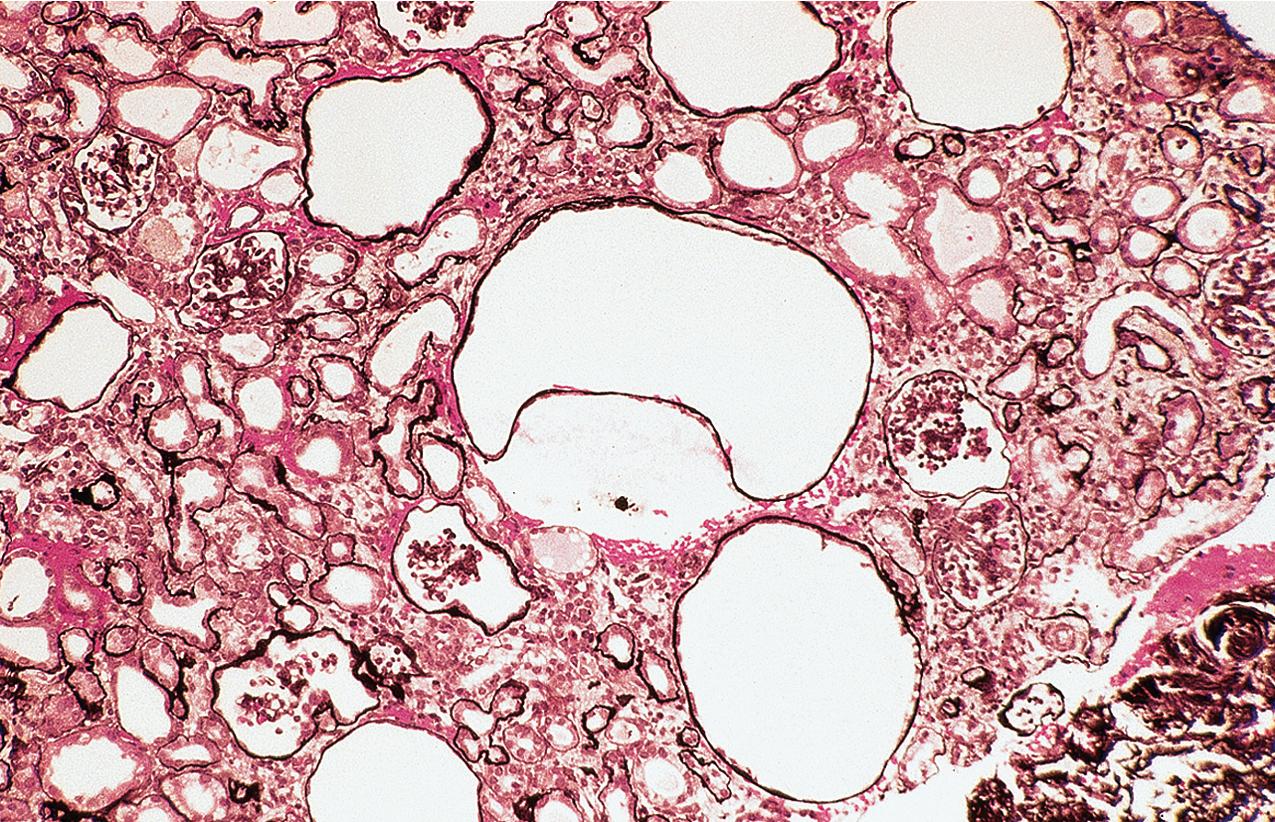
There are no deposits by IF. By EM, there is widespread effacement of foot processes and loss of slit diaphragms. The GBM may be focally attenuated.
The nephrin gene, NPHS1, is mutated in CNF. Nephrin is a prominent component of the slit diaphragm of the foot processes of the podocyte. Studies in knock-out mice reveal that intact nephrin is required for maintaining normal capillary permselectivity. Mutation of a protein tightly associated with nephrin, (CD2-associated protein, CD2AP), in mice has demonstrated that mutation of other components of the slit diaphragm or its anchoring proteins also leads to nephrotic syndrome, with clinical characteristics mirroring many of those of CNF type. Approximately one-quarter of patients transplanted develop recurrent nephrotic syndrome. Renal biopsies of the transplant performed from 3 days to 2 weeks after onset of recurrent nephrotic syndrome showed glomerular capillary endothelial cell swelling and extensive foot process effacement. These recurrences typically develop in patients with the Fin-major/Fin-major genotype, resulting in completely absent nephrin. Some of these patients had detectable anti-nephrin antibodies after transplant, supporting immune injury to the normal nephrin-bearing podocytes in the graft, although usual immune complexes are not observed. The recurrence of nephrotic syndrome may result in graft loss.
Huttunen NP, Rapola J, Wilska J, et al. Renal pathology in congenital nephrotic syndrome of Finnish type: A quantitative light microscopic study on 50 patients. Int J Pediatr Nephrol. 1980;1:10.
Patrakka J, Ruotsalainen V, Reponen P, et al. Recurrence of nephrotic syndrome in kidney grafts of patients with congenital nephrotic syndrome of the Finnish type: Role of nephrin. Transplantation. 2002;73:394–403.
Rapola J. Congenital nephrotic syndrome. Pediatr Nephrol. 1987;1:441–446.
Ruotsalainen V, Ljungberg P, Wartiovaara J, et al. Nephrin is specifically located at the slit diaphragm of glomerular podocytes. Proc Natl Acad Sci U S A. 1999;96:7962–7967.
Diffuse mesangial sclerosis may occur as an isolated lesion manifesting as nephrotic syndrome or be part of Denys–Drash syndrome. Mutations in the Wilms tumor gene (WT1), a transcription factor, occur both in isolated diffuse mesangial sclerosis and in patients with Denys–Drash syndrome. Denys–Drash syndrome is caused by heterozygous mutation in WT1. Onset is typically congenital or in the first years of life. Patients may have renal failure at presentation and typically progress to end-stage kidney disease before age 4. Clinically, patients with Denys–Drash syndrome are usually 46XY and have ambiguous external genitalia or male pseudohermaphroditism with female external genitalia with streak gonads or abnormal testes and are at risk for Wilms tumor. Occasionally, patients are 46XX with nephropathy and Wilms tumor without abnormal genitalia.
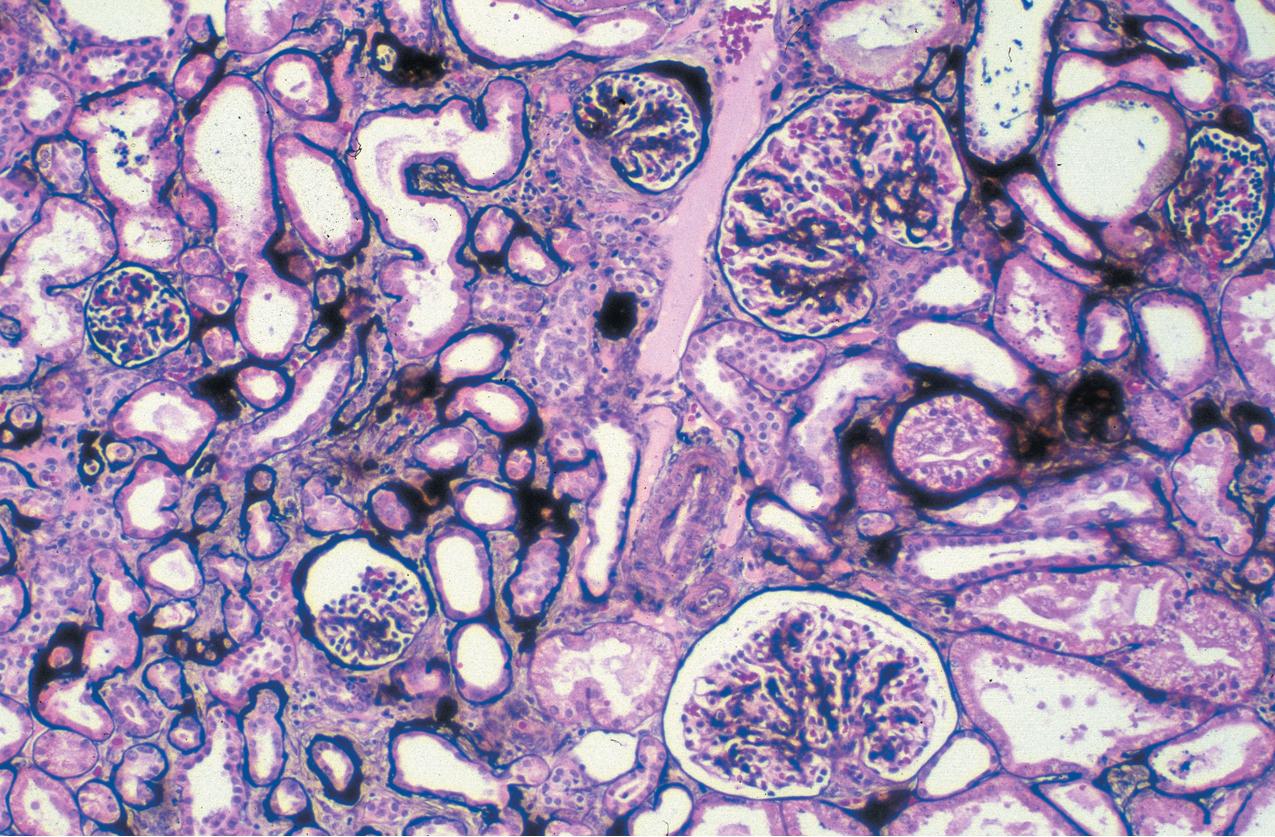
The earliest renal lesions are characterized by an increase in mesangial matrix and hypertrophic podocytes, followed by an increase in mesangial matrix that initially appears delicate and loosely woven, culminating in further mesangial increase and sclerosis with obliteration of the capillary lumens ( Figs. 3.44–3.46 ). There is no increase in mesangial cellularity, and a dense core of disorganized collagen with positive Masson trichrome staining is present in the sclerotic glomeruli. The overlying podocytes typically are hypertrophic and may appear immature, dense, and cobblestone-like. Occasional epithelial cell proliferation may be present ( Fig. 3.47 ). In contrast to idiopathic FSGS, the deeper corticomedullary glomeruli are less affected. Interstitial fibrosis and tubular atrophy are proportional to the glomerulosclerosis ( Fig. 3.48 ). Although tubules may be dilated, this is not a prominent or early feature as in CNF (see earlier).
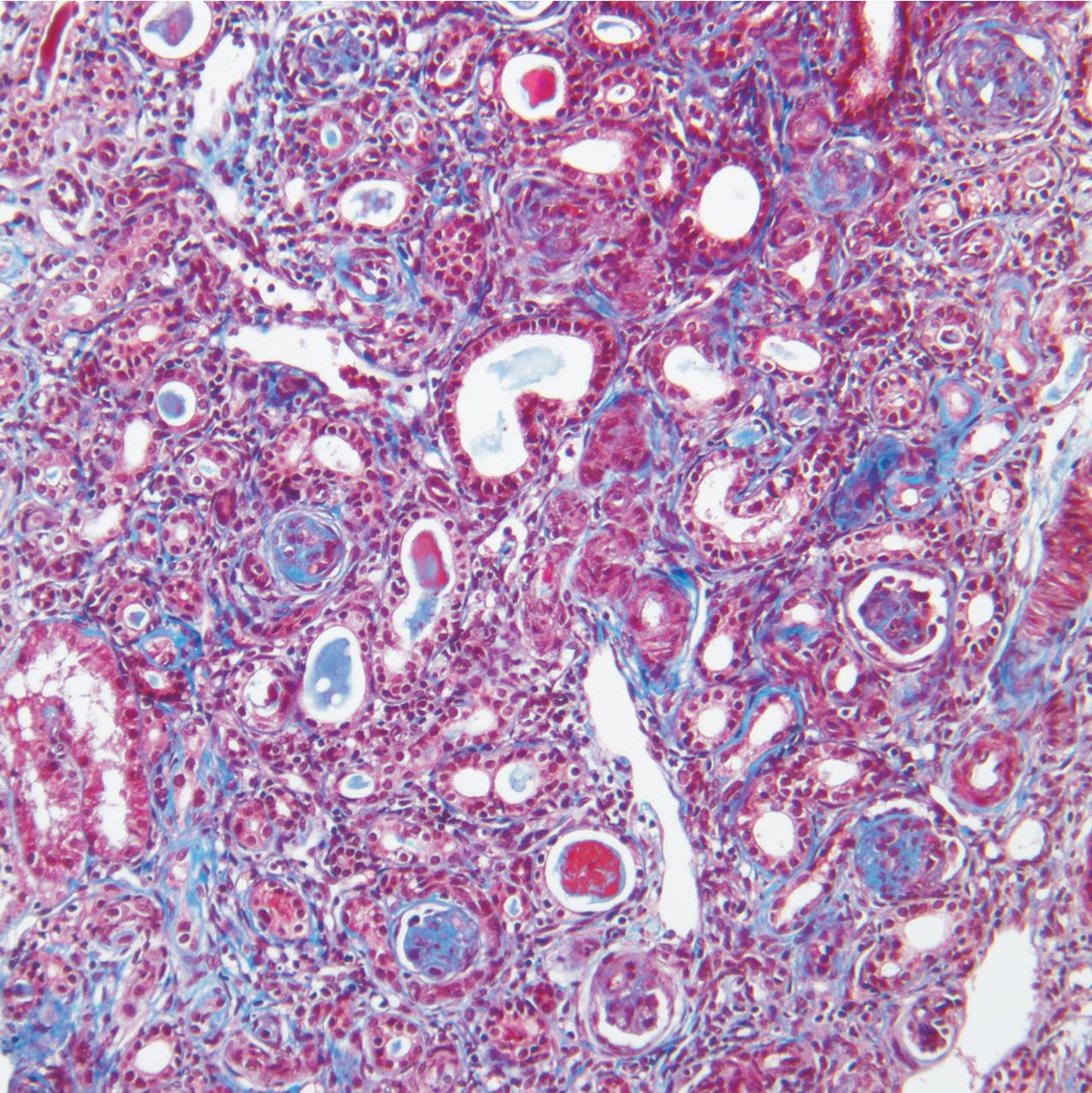
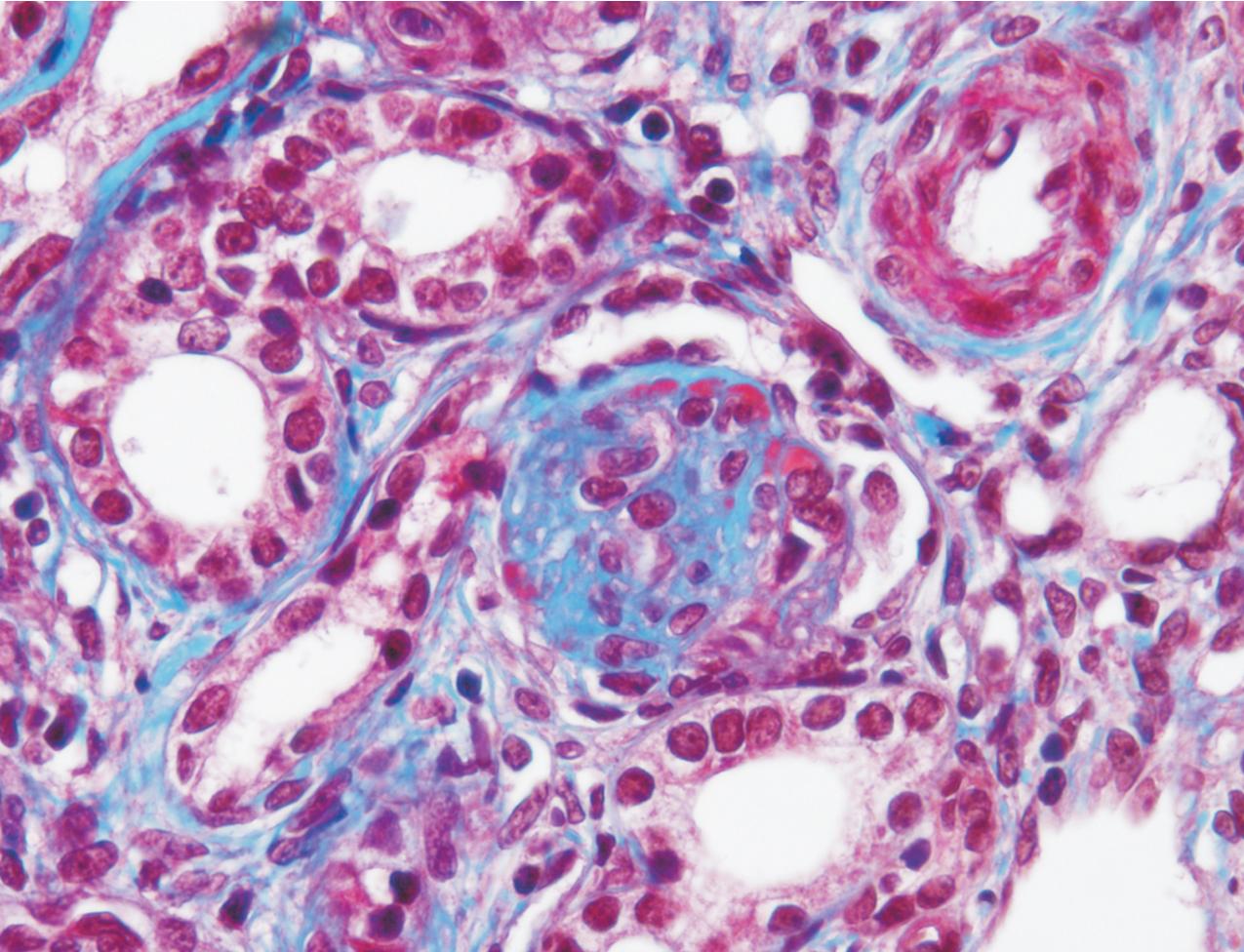
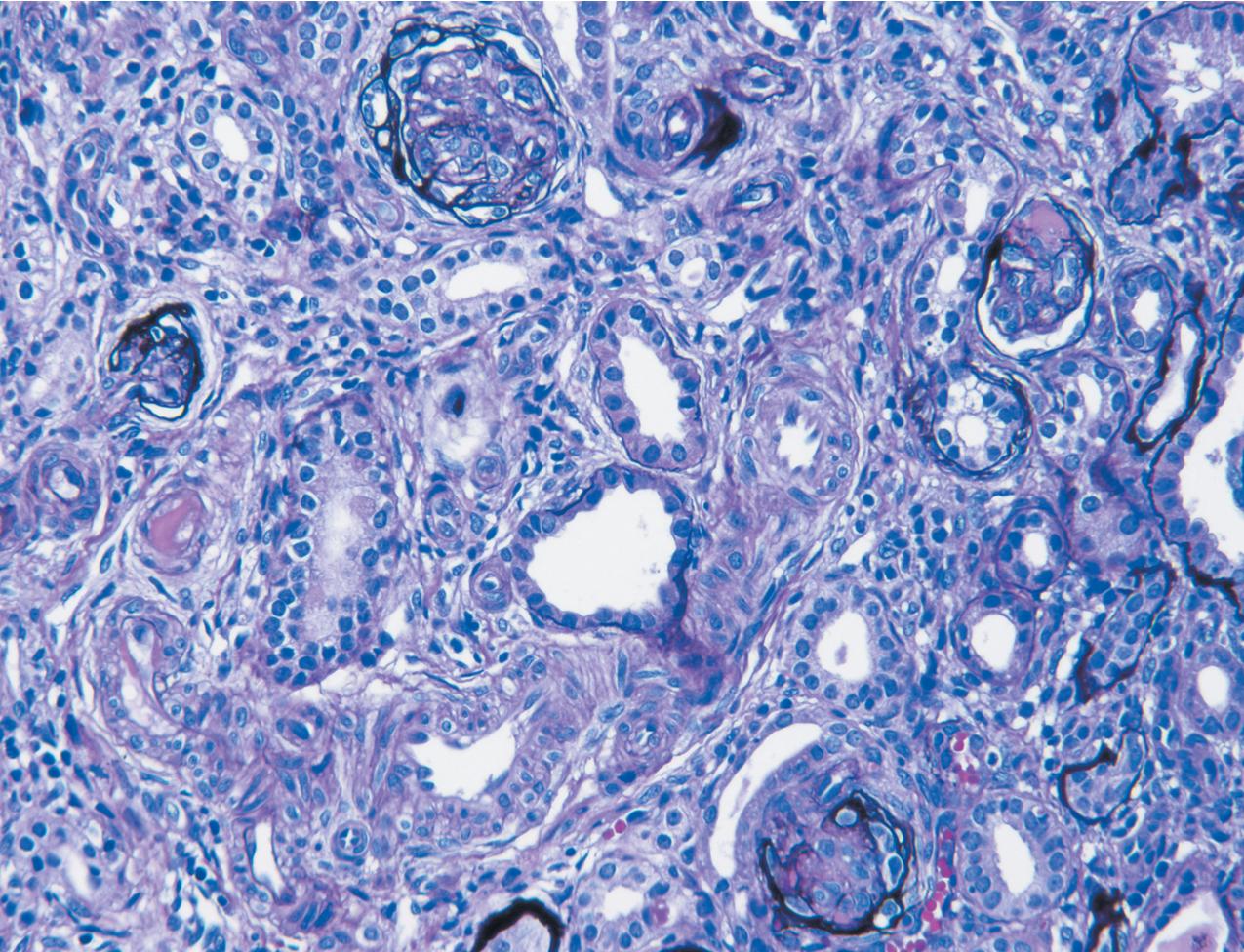
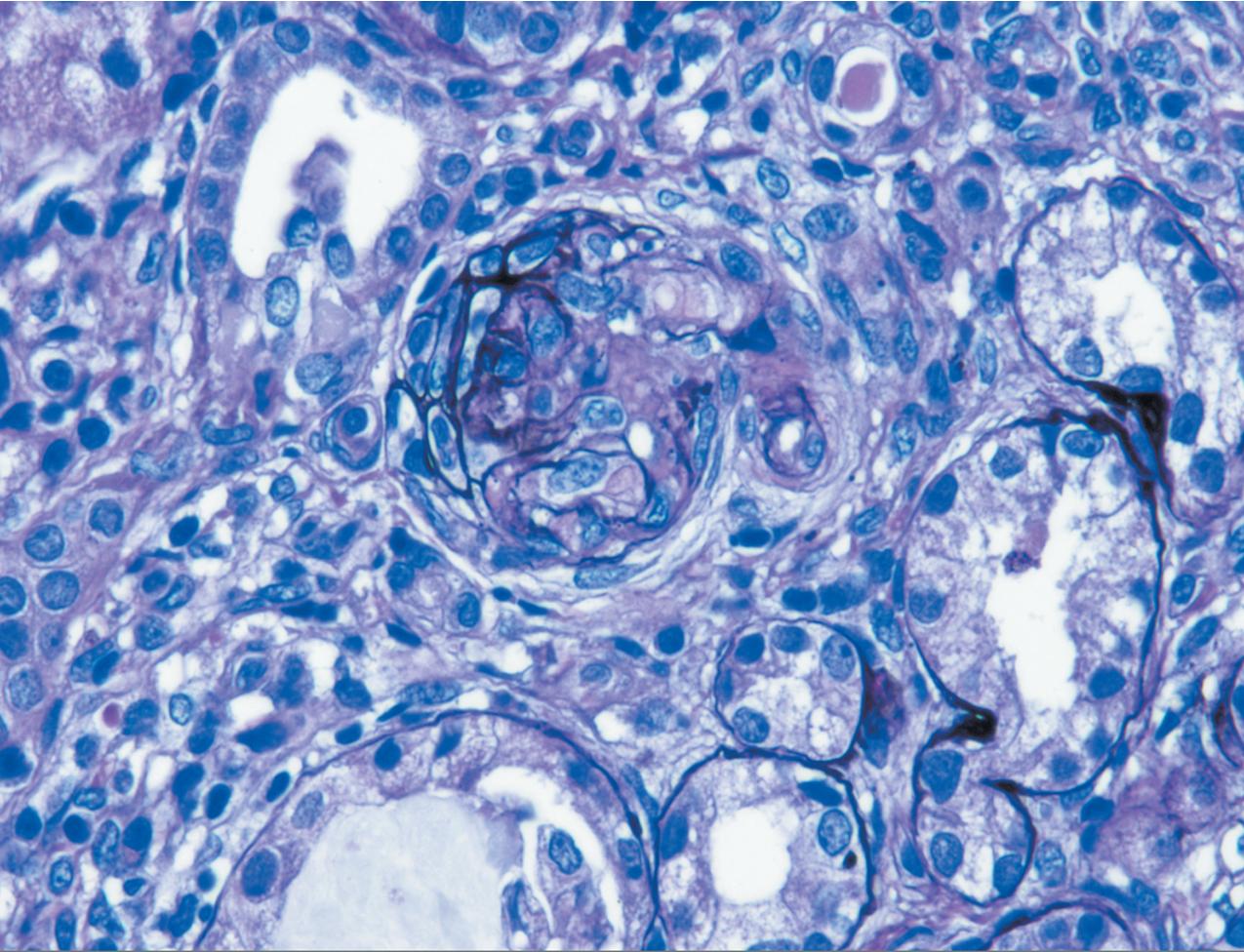
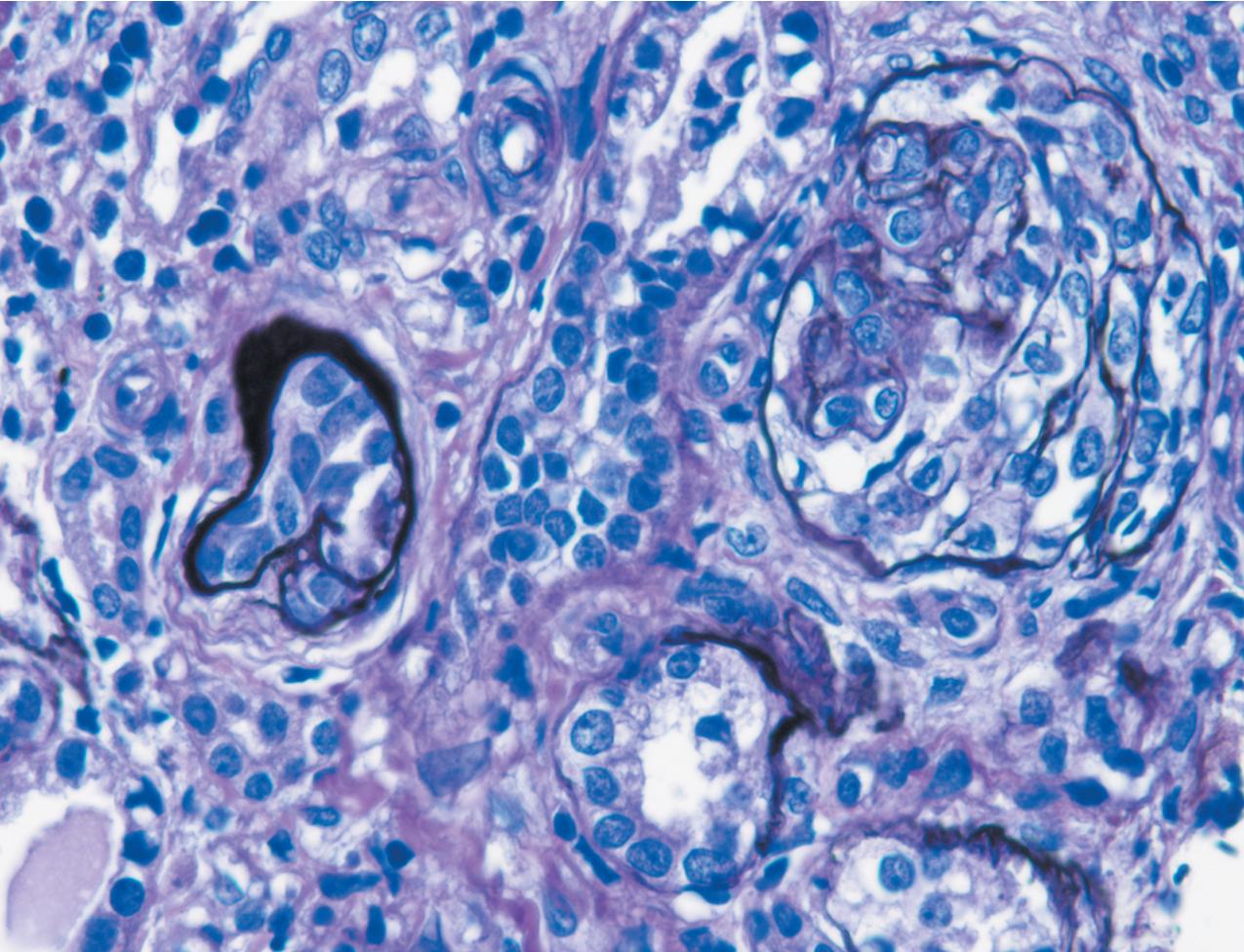
There are no immune complex deposits by IF, although variable trapping of IgM, C1q, and C3 may be present in the sclerotic mesangial areas. By EM, the GBM is somewhat thickened and foot processes are effaced. The mesangial areas show increased matrix with collagen fibrils.
Patients with Frasier syndrome also have WT1 mutations but do not have early-onset disease with diffuse mesangial sclerosis. Onset of proteinuria and renal failure is in late childhood and late adolescence. Patients show complete male pseudohermaphroditism with complete gonadal dysgenesis and thus appear externally female but do not undergo menarche at puberty. Patients have increased risk for gonadoblastoma developing in the abnormal gonads but do not develop Wilms tumor.
Renal biopsy demonstrates FSGS lesions of NOS type by light microscopy without immune deposits ( Fig. 3.49 ). By EM, some patients with Frasier syndrome may show lamellation and basket-weaving appearance that resembles Alport syndrome, although there are no mutations in type IV collagen alpha chains ( Fig. 3.50 ).
WT1 is a transcription factor that, through DNA binding, regulates numerous genes. WT1 is crucial for development of the kidney and genitalia. In the mature kidney, WT1 is expressed in podocytes and controls slit diaphragm proteins and differentiation.
Various WT1 mutations contribute to the spectrum of diffuse mesangial sclerosis, Denys–Drash syndrome, and Frasier syndrome. Denys–Drash syndrome typically is caused by heterozygous mutations in the WT1 gene, with this mutation suppressing expression of the remaining normal allele. Isolated diffuse mesangial sclerosis has also occasionally been associated with WT1 mutations. Diffuse mesangial sclerosis is rarely caused by mutations in phospholipase C epsilon but without the associated syndrome abnormalities seen in Denys–Drash syndrome.
Effects of altered WT1 on podocyte differentiation have been suggested. This gene encodes a transcription factor of the zinc finger family, with four transcripts of WT1 resulting from alternative splicing. Various mutations have been reported. When WT1 point mutations in the donor splice site in intron 9 occurs, Frasier syndrome results, with FSGS lesions.
Bilateral nephrectomies are indicated in patients with Denys–Drash syndrome when they reach end-stage kidney disease to prevent development of Wilms tumor in the kidney. Transplantation has resulted in a good outcome, without reports of recurrence in the transplant of nephrotic syndrome or glomerular lesions in patients with diffuse mesangial sclerosis or Denys–Drash syndrome or Frasier syndrome.
Barbaux S, Niaudet P, Gubler MC, et al. Donor splice-site mutations in the WT1 gene are responsible for Frasier syndrome. Nat Genet. 1997;17:467–469.
Habib R. Nephrotic syndrome in the 1st year of life. Pediatr Nephrol. 1993;7:347–353.
Salomon R, Gubler MC, Niaudet P. Genetics of the nephrotic syndrome. Curr Opin Pediatr. 2000;12:129–134.
C1q nephropathy is a descriptive term that represents a variable pattern of glomerular injury with abnormality in complement, defined by the presence of mesangial and occasional capillary wall Ig and complement deposits, with C1q IF staining intensity of at least 2+ and greater than or equal to that of other components. Systemic lupus erythematosus (SLE) and LN should be excluded. C1q nephropathy occurs primarily in children and young adults. Patients typically present with nephrotic syndrome, especially if the biopsy shows sclerosing or minimal change–type lesions by light microscopy. They may have an active urinary sediment when proliferative changes are present but do not have SLE clinically. About a third of patients with sclerosis at time of biopsy develop end-stage kidney disease. In contrast, complete remission of the nephrotic syndrome occurs in 77% of those with a minimal change–like lesion. Renal disease remains stable in just over half of those with proliferative GN at time of biopsy.
By light microscopy, there is a spectrum of possible glomerular alterations, including no histologic abnormalities, mesangial proliferation, focal or diffuse proliferative GN, or focal segmental glomerulosclerosis with or without associated mesangial proliferation ( Figs. 3.51–3.52 ).
IF microscopy shows predominant C1q, at least 2+ intensity, along with C3 and immunoglobulins ( Fig. 3.53 ), none of which by definition are more than C1q. Positivity is usually mesangial, with capillary loop extension especially in cases with hypercellular/proliferative lesions.
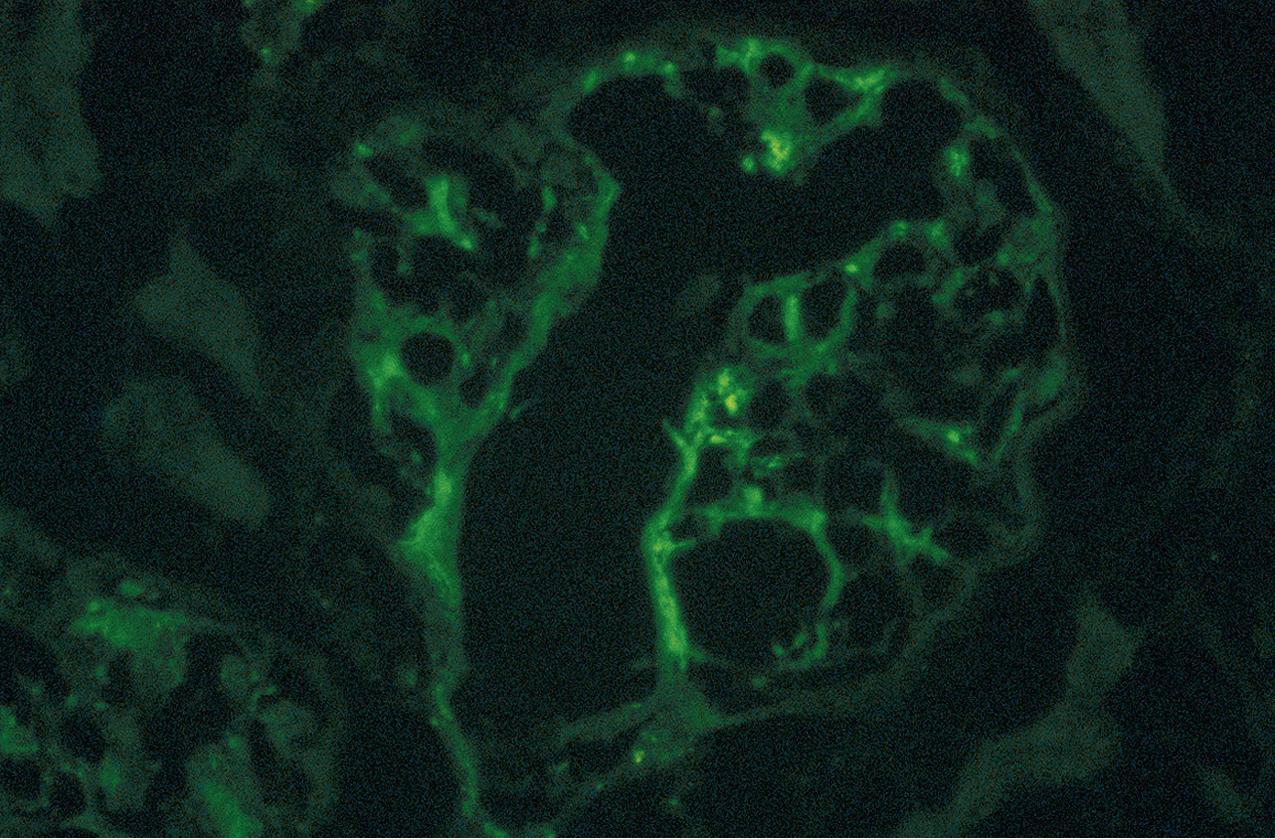
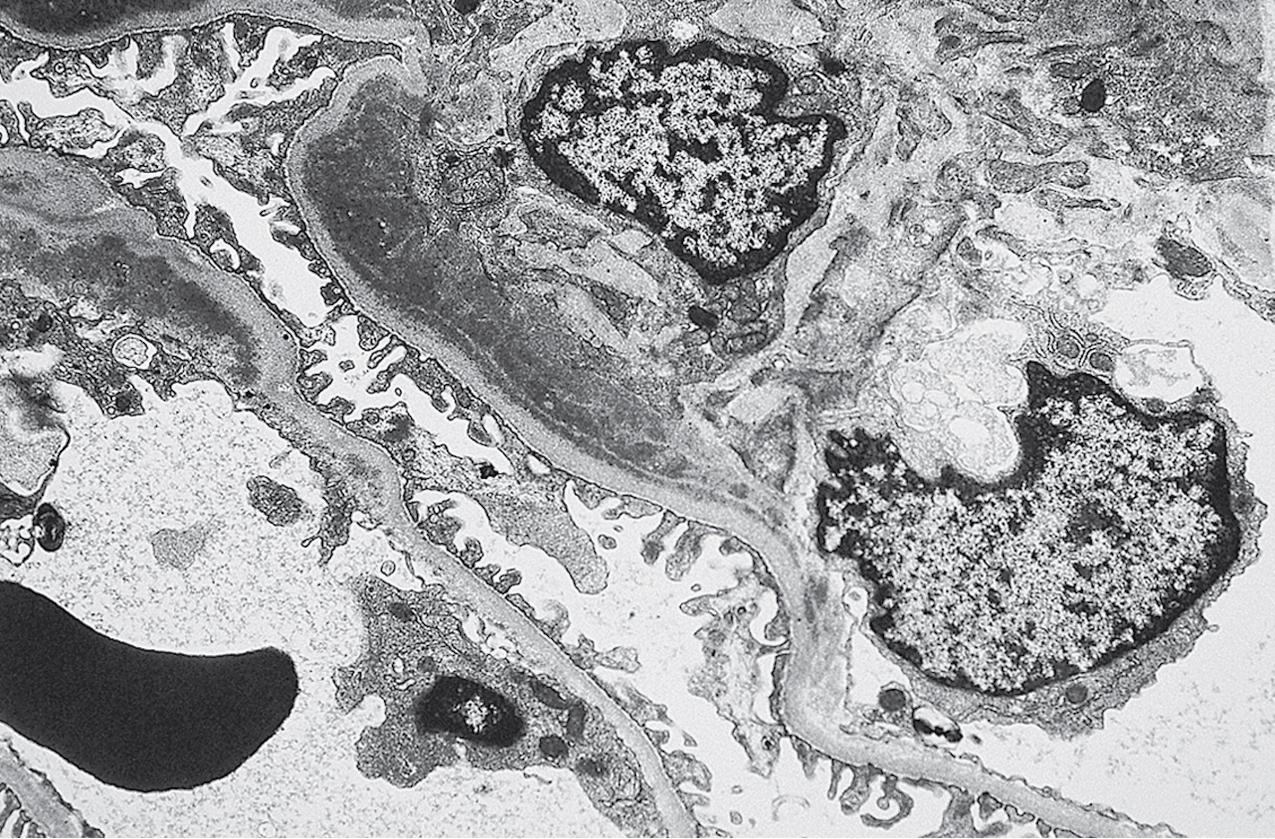
EM typically shows foot process effacement and deposits confined to the mesangium. In cases with hypercellularity/proliferation, deposits often extend to subendothelial areas. Subepithelial deposits are rare. In cases without hypercellularity/proliferation, foot process effacement is quite extensive and deposits are confined to the mesangium. Notably, reticular aggregates, a common feature in patients with LN, are absent ( Fig. 3.54 ).
The etiology and pathogenesis are unknown. In our opinion, C1q nephropathy without proliferation may be viewed as an unusual lesion related to MCD-FSGS, whereas those patients with proliferative lesions behave more like immune complex disease. The deposition of C1q suggests an abnormality of complement regulation.
Dominant (or codominant C1q, at least 2+) with corresponding deposits by electron microscopy
Variable light microscopy (minimal changes vs. segmental sclerosis vs. hypercellular)
Extensive foot process effacement
Absence of morphologic or clinical features of lupus nephritis (i.e., no reticular aggregates, lack of full-house staining with immunoglobulin G [IgG] dominant or codominant by immunofluorescence)
Jennette JC, Hipp CG. C1q nephropathy: A distinct pathologic entity usually causing nephrotic syndrome. Am J Kidney Dis. 1985;6:103–110.
Markowitz GS, Schwimmer JA, Stokes MB, et al. C1q nephropathy: A variant of focal segmental glomerulosclerosis. Kidney Int. 2003;64:1232–1240.
Vizjak A, Ferluga D, Rozic M, et al. Pathology, clinical presentations, and outcomes of C1q nephropathy. J Am Soc Nephrol. 2008;19:2237–2244.
The term membranoproliferative describes a pattern of injury with mesangial expansion because of mesangial and endocapillary hypercellularity and increased mesangial matrix and thickened capillary walls with a double contour (the so-called “tram-track” appearance). When seen with glomerular inflammation, it is called membranoproliferative GN (MPGN; see the section on MPGN). This pattern may be seen with immune complex deposition, monoclonal protein deposits, or other organized deposits (as in fibrillary GN) or be related to complement-mediated disease. A similar pattern is present in response to chronic endothelial injury with only cell interposition and no deposits or significant inflammation (chronic thrombotic microangiopathy [TMA]). MPGN pattern with immune complex-mediated injury was previously called MPGN type I. The immune complexes may be undefined in terms of the inciting antigen (“idiopathic”), related to autoimmune diseases like SLE or lupus-like illnesses, or be secondary to chronic infections. Although light microscopy may appear similar in these entities, IF findings with staining for Ig and complement and corresponding deposits by EM readily allow recognition of the immune complexes in MPGN. In contrast, C3 glomerulopathies, which include dense deposit disease (DDD) and C3 dominant GN (C3GN), show C3 staining only, or dominant C3 with scanty Ig, with deposits (either very dense, as in the aptly named dense deposit disease, or of usual density in C3GN) by EM. Of note, although the MPGN pattern is common in C3 glomerulopathies, a significant number instead have a mesangioproliferative or exudative pattern of injury. Some biopsies show intermediate features between DDD and C3GN by EM, and underlying complement abnormalities also overlap in DDD versus C3GN. The specific factors that underlie this spectrum of morphologic appearances are not yet completely understood. We will discuss these two ends of the spectrum separately because there appear to be prognostic and clinical differences between DDD and C3GN, as discussed later.
DDD is classified as part of the C3 glomerulopathies ( Fig. 3.55 ). DDD is much rarer than immune complex-mediated MPGN and accounts for about a quarter of C3 glomerulopathy cases. Patients with DDD typically present with features of nephritic/nephrotic syndrome and decreased serum levels of complements, particularly C3. Early components of the classic pathway, that is, C1q and C4, usually show normal serum levels. Patients typically present with hypertension and increased serum creatinine. Nearly all have hematuria, with nephrotic syndrome in a third of patients. Children were more likely to have reduced C3 and had less incidence of renal insufficiency than adults in one series. Some patients have partial lipodystrophy associated with DDD, and ocular drusen may also be present. Average age of patients with DDD at diagnosis is 14 years but with a large age range. In one recent series of DDD, slightly more than half of patients were adults. Overall, about 20% of DDD patients are older than 60 years, with equal incidence in males and females. On follow-up, a quarter of patients had complete response to immunosuppression with or without renin angiotensin system blockade, about half had persistent renal dysfunction, and a quarter had end-stage kidney disease. Progressive renal failure occurs in the majority of patients, with 30% to 50% reaching end-stage kidney disease within 10 years of onset. Rare cases of spontaneous improvement of the disease have been reported. More rapid progression was associated with crescents. Clinical predictors of end-stage kidney disease were older age (older than 16 years at diagnosis) and higher creatinine at biopsy. Crescents or polymorphonuclear leukocytes (PMNs) in capillary loops were associated with worse prognosis, whereas focal segmental glomerular lesions were less frequently associated with progressive renal disease. A recent large series of patients with C3GN or DDD evaluated a histology risk score, including lesions defined as active and chronic. Active lesions were defined as mesangial and/or endocapillary hypercellularity, membranoproliferative pattern, glomerular leukocyte infiltration, crescents, fibrinoid necrosis, or interstitial inflammation. Chronic lesions were defined as glomerulosclerosis, tubular atrophy, interstitial fibrosis, and arteriosclerosis and arteriolosclerosis. Higher combined overall score correlated with worse outcome. Among active lesions, only crescents correlated with worse outcome. All chronic lesions were linked to worse outcome. Recurrence in the transplant of DDD occurs in 60% to 85% of patients, with about half resulting in graft loss.
By light microscopy, mesangial proliferation is the pattern most commonly observed, followed by endocapillary hypercellularity, often with PMN infiltrate in glomeruli in early stages ( Figs. 3.56–3.57 ). There may be focal segmental necrotizing proliferative lesions with crescents. The GBMs are thickened and highly refractile and eosinophilic. The involved areas of the GBM resemble a “string of sausages.” The deposits are PAS-positive ( Figs. 3.58–3.60 ). When the dense deposits replace the GBM, silver staining may be weak or absent in segments. Thioflavin T stain and toluidine blue stain highlight the deposits (see Fig. 3.60 ). Thickening because of C3 deposits also can affect tubular basement membranes and Bowman’s capsule.
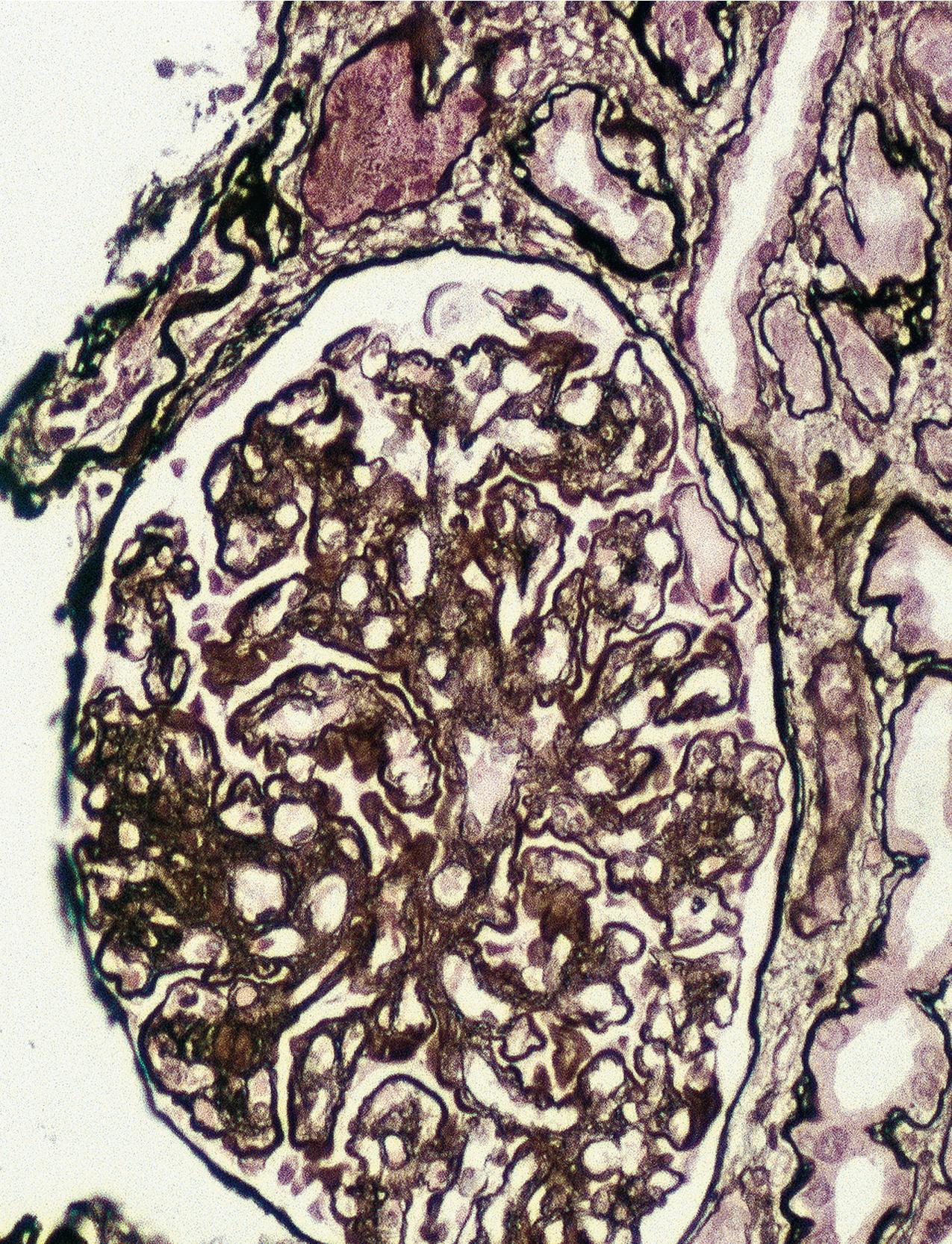
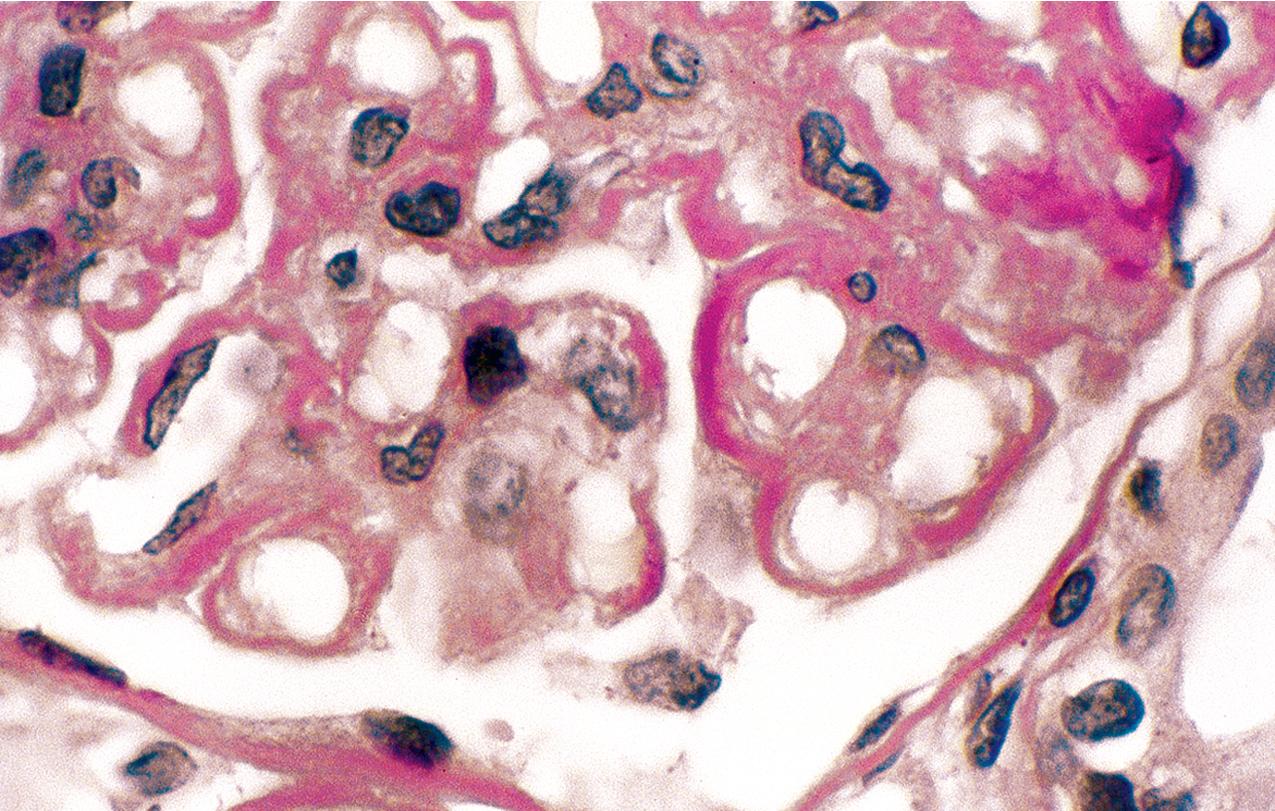
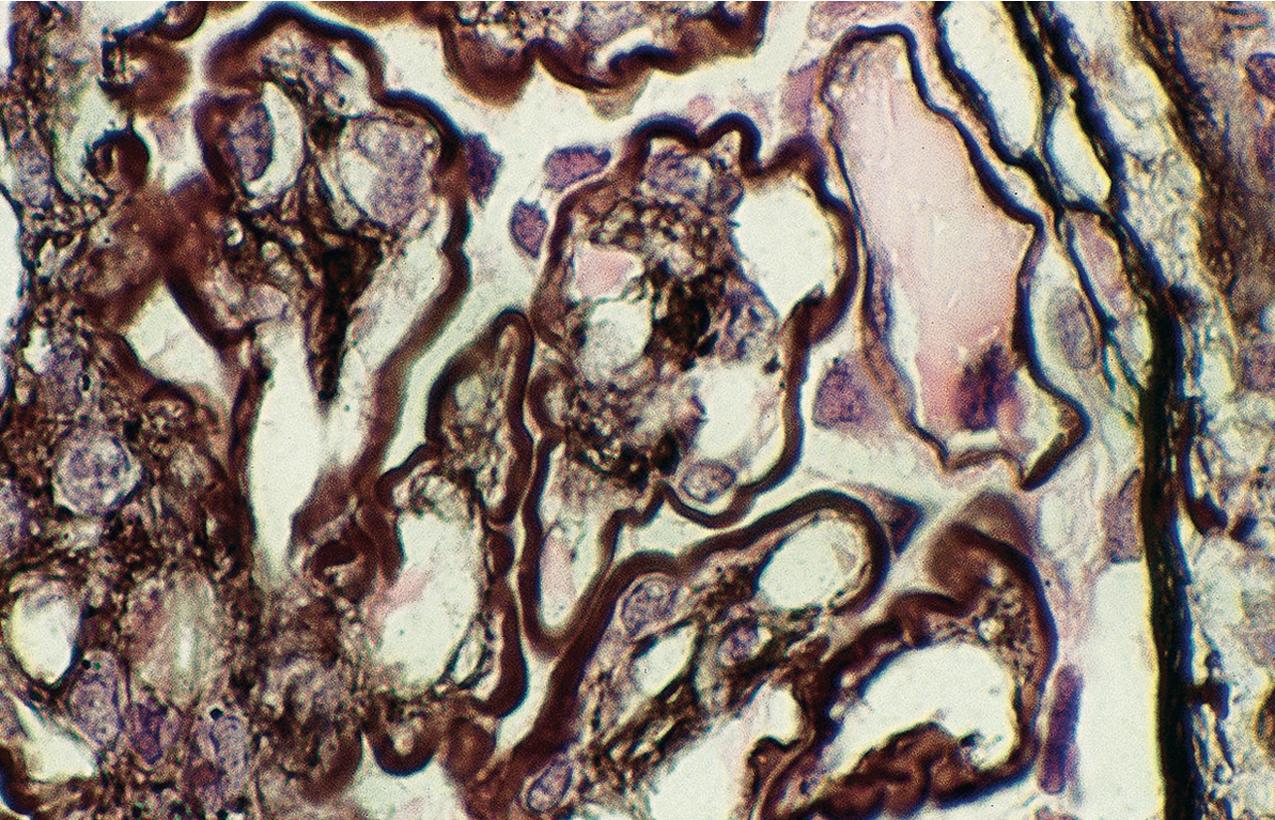
IF in DDD shows C3 staining irregularly along the capillary wall, in a smooth, granular, or discontinuous pattern ( Fig. 3.61 ). Mesangial bright staining in a distinct globular pattern within the central mesangial area is highly characteristic of DDD. Ig is usually not detected, indicating the dense deposits are not classic antigen–antibody immune complexes. Nevertheless, segmental IgM or, less often, IgG and, very rarely, IgA have been reported. A cut-off of dominant C3 at least two intensity steps greater than other immune reactants correctly captures around 80% of DDD cases as diagnosed by characteristic EM findings. Deposits may also involve Bowman’s capsule and focal tubular basement membranes.
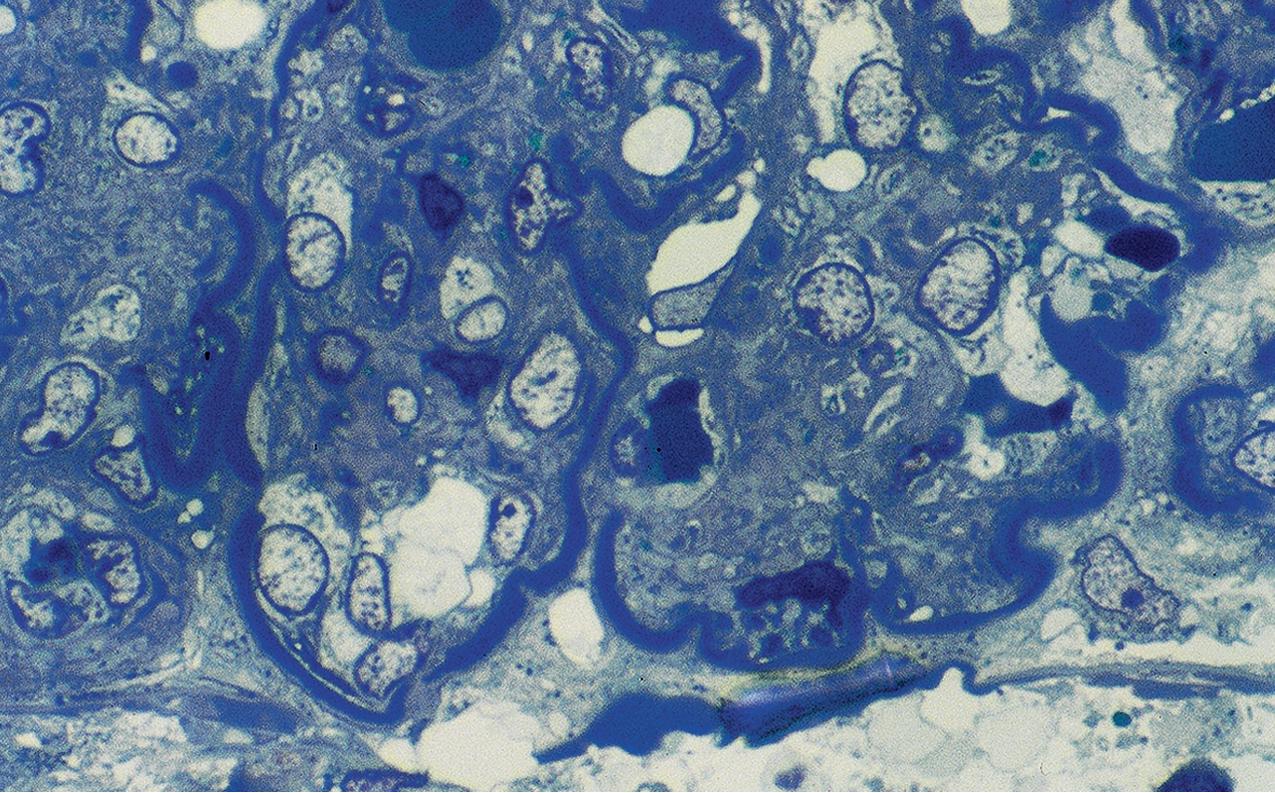
By EM, the lamina densa of the basement membrane in DDD shows a very dense transformation without discrete immune complex–type deposits ( Figs. 3.62–3.63 ). Similar dense globular deposits are often found in the mesangial areas in addition to increased matrix. Increased mesangial cellularity or cellular interposition is far less common than in immune complex MPGN. Podocytes show varying degrees of reactive changes, from vacuolization, microvillous transformation to foot process effacement. Tubular basement membranes and Bowman’s capsule may show similar densities as in the GBM.
The dense material consists of C3 degradation products, demonstrated by mass spectrometry studies. DDD patients typically have underlying abnormalities in regulation of the alternative complement pathway with uncontrolled activation of C3 convertase, with mutation and/or autoantibody against key complement regulatory molecules. DDD is thus grouped in the broader category of C3 glomerulopathy. C3 nephritic factor (C3NeF) stabilizes the C3 convertase C3bBb, resulting in alternate pathway-mediated C3 breakdown. C3NeF is detected in 80% to 85% of DDD patients. DDD sometimes occurs in association with partial lipodystrophy, a condition with loss of adipose tissue, decreased complement, and presence of C3NeF. Further, a porcine model of factor H deficiency has similarities to DDD. Factor H inactivates factor C3bBb. Inadequate factor H activity, either because of deficiency or antibody to factor H, has been observed in some patients with DDD. These associations indicate that abnormal complement regulation predisposes to DDD. Nevertheless, some patients with partial lipodystrophy and C3NeF do not have DDD, which further indicates that complement abnormalities alone are insufficient to produce the disease and that additional triggers likely tip toward unfettered complement dysregulation.
Membranoproliferative or mesangial or exudative proliferative features by light microscopy
C3 only or C3 dominant (more than two intensity steps greater than immunoglobulin staining) deposits by immunofluorescence
Dense transformation of glomerular basement membranes with round, nodular deposits in mesangium by electron microscopy
Anders D, Agricola B, Sippel M, et al. Basement membrane changes in membranoproliferative glomerulonephritis. II. Characterization of a third type by silver impregnation of ultra-thin sections. Virchows Archiv (Pathology and Anatomy). 1977;376:1–19.
Andresdottir MB, Assmann KJ, Hoitsma AJ, et al. Renal transplantation in patients with dense deposit disease: morphological characteristics of recurrent disease and clinical outcome. Nephrol Dial Transplant . 1999;14:1723–1731.
Bennett WM, Fassett RG, Walker RG, et al. Mesangiocapillary glomerulonephritis type II (dense-deposit disease): Clinical features of progressive disease. Am J Kidney Dis. 1989;13:469–476.
Berger J, Galle P. Dépots denses au sein des membranes basales du rein: étude en microscopies optique et électronique. Presse Med. 1963;71:2351-2354.
Bomback AS, Santoriello D, Avasare RS, et al. C3 glomerulonephritis and dense deposit disease share a similar disease course in a large United States cohort of patients with C3 glomerulopathy. Kidney Int. 2018;93:977–985.
Cameron JS, Turner DR, Heaton J, et al. Idiopathic mesangiocapillary glomerulonephritis. Comparison of types I and II in children and adults and long-term prognosis. Am J Med. 1983;74:175–192.
Churg J, Duffy JL, Bernstein J. Identification of dense deposit disease. Arch Pathol. 1979;103:67–72.
Cook HT, Pickering MC. Histopathology of MPGN and C3 glomerulopathies. Nat Rev Nephrol. 2015;11: 14–22.
De Vriese AS, Sethi S, Van Praet J, Nath KA, Fervenza FC. Kidney disease caused by dysregulation of the complement alternative pathway: An etiologic approach. J Am Soc Nephrol. 2015;26:2917–2929.
Habib R, Gubler MC, Loirat C, et al. Dense deposit disease: A variant of membranoproliferative glomerulonephritis. Kidney Int. 1975;7:204–215.
Hou J, Markowitz GS, Bomback AS, et al. Toward a working definition of C3 glomerulopathy by immunofluorescence. Kidney Int. 2014;85:450–456.
McEnery PT, McAdams AJ. Regression of membranoproliferative glomerulonephritis type II (dense deposit disease): Observations in six children. Am J Kidney Dis. 1988;12:138–146.
Nasr SH, Valeri AM, Appel GB, et al. Dense deposit disease: Clinicopathologic study of 32 pediatric and adult patients. Clin J Am Soc Nephrol. 2009;4:22–32.
Pickering MC, D’Agati VD, Nester CM, et al. C3 glomerulopathy: Consensus report. Kidney Int. 2013;84: 1079–1089.
Sethi S, Gamez JD, Vrana JA, et al. Glomeruli of dense deposit disease contain components of the alternative and terminal complement pathway. Kidney Int. 2009;75:952–960.
Smith RJH, Appel GB, Blom AM. C3 glomerulopathy—understanding a rare complement-driven renal disease. Nat Rev Nephrol. 2019;15:129–143.
Walker PD, Ferrario F, Joh K, et al. Dense deposit disease is not a membranoproliferative glomerulonephritis. Mod Pathol. 2007;20:605–616.
C3GN, part of the spectrum of C3 glomerulopathy, is an uncommon disorder, with an average age of onset of around 30 years, but with a wide reported range, from 7 to 70 years. Overall, C3 glomerulopathy patients are on average 21 years at diagnosis, with younger age in DDD patients than in those with C3GN. Patients typically have subnephrotic proteinuria with nephrotic syndrome present in only about 15% and most have microhematuria. Up to 40% of C3GN patients have low serum C3. C3 nephritic factor is less commonly present than in DDD (see the previous section on “Dense Deposit Disease”). At presentation, about half of patients have hypertension and slightly more than half have impaired GFR. About half of these patients maintained normal renal function, with about 25% progressing to end-stage kidney disease. Worse prognosis among C3GP patients was seen in those older than 16 years at diagnosis and with crescents. DDD patients progressed more frequently than those with C3GN. A recent study applied a histology risk score to C3 glomerulopathy cases (see “Dense Deposit Disease”), which confirmed that crescents were associated with worse outcome. Chronic lesions, including glomerulosclerosis, tubular atrophy, interstitial fibrosis, and vascular sclerosis, were also linked to poor prognosis. Recurrence in the transplant developed in about two-thirds in one small series, with graft loss within 5 years in one-third.
Light microscopic findings are variable, with half to two-thirds of patients showing a membranoproliferative pattern by light microscopy with mesangial and endocapillary hypercellularity with double contours of GBMs ( Figs. 3.64–3.65 ). About a quarter of cases show only mesangial proliferation, and about 20% show diffuse endocapillary hypercellularity without GBM double contours. A small proportion of cases have an exudative (around 12% in one series) or crescentic (about 5% in one series) appearance. Correspondingly, in a third of cases, deposits showed only a mesangial and subepithelial distribution without subendothelial deposits or mesangial proliferation.
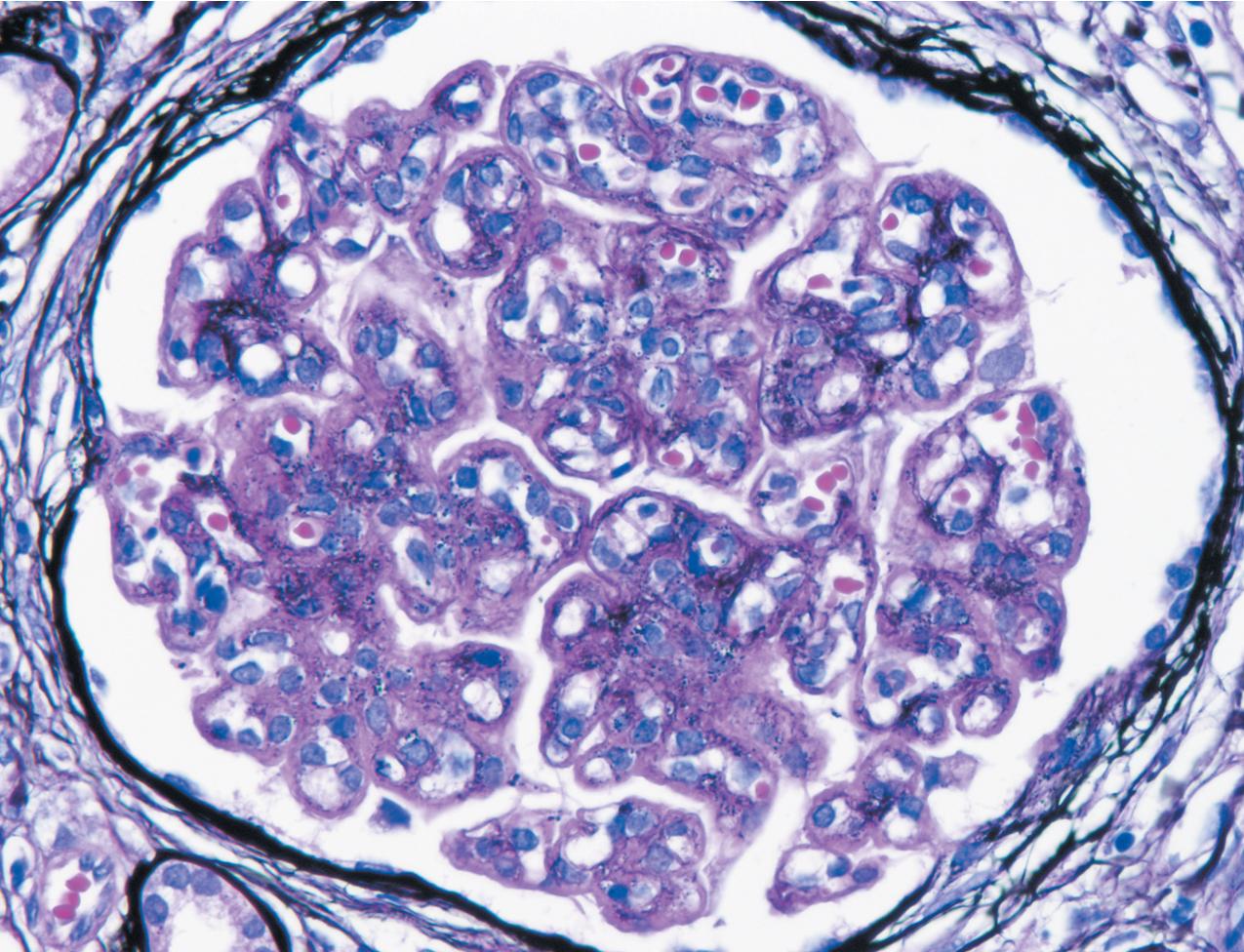
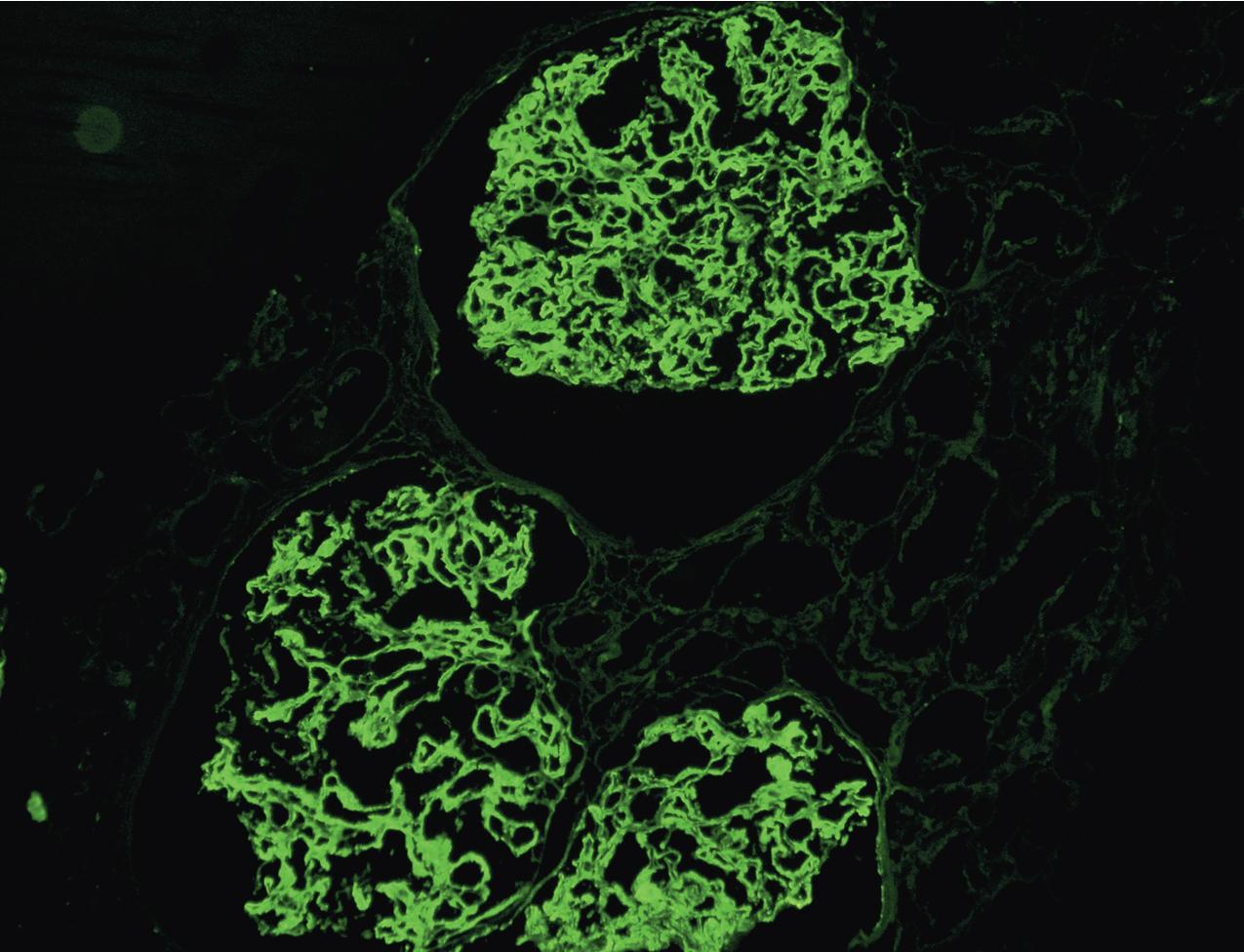
IF microscopy classically shows isolated C3 deposits without C1q or IgG ( Figs. 3.66–3.67 ). The localization of C3 deposits mirrors the light microscopic pattern, with mesangial and often additional scattered capillary loop deposits. Biopsies at different stages of disease may show varying Ig. A cut-off of C3 intensity at least two steps stronger than any other immune reactant has broadened recognition of cases of C3 glomerulopathy, and this cut-off included about 80% of DDD cases. Of note, in some cases, Ig deposits may not be detected by frozen section IF but are evident after pronase digestion on paraffin-embedded, formalin-fixed tissue, unmasking an Ig component of deposits. Thus adding pronase digestion in cases with C3 only detected in deposits by frozen section IF should be considered to avoid misdiagnosing such cases as C3GN, rather than immune complex disease. The sensitivity and best cut-off for allowable Ig deposition to diagnose C3GN is not precisely known. We recommend a sign-out of “C3 dominant GN” in cases with C3 dominance (i.e., C3 at least two intensity steps stronger than other immune reactants) and EM deposits supportive of possible C3GN. Further workup for possible complement dysregulation may then indicate whether the lesion indeed represents a C3GN.
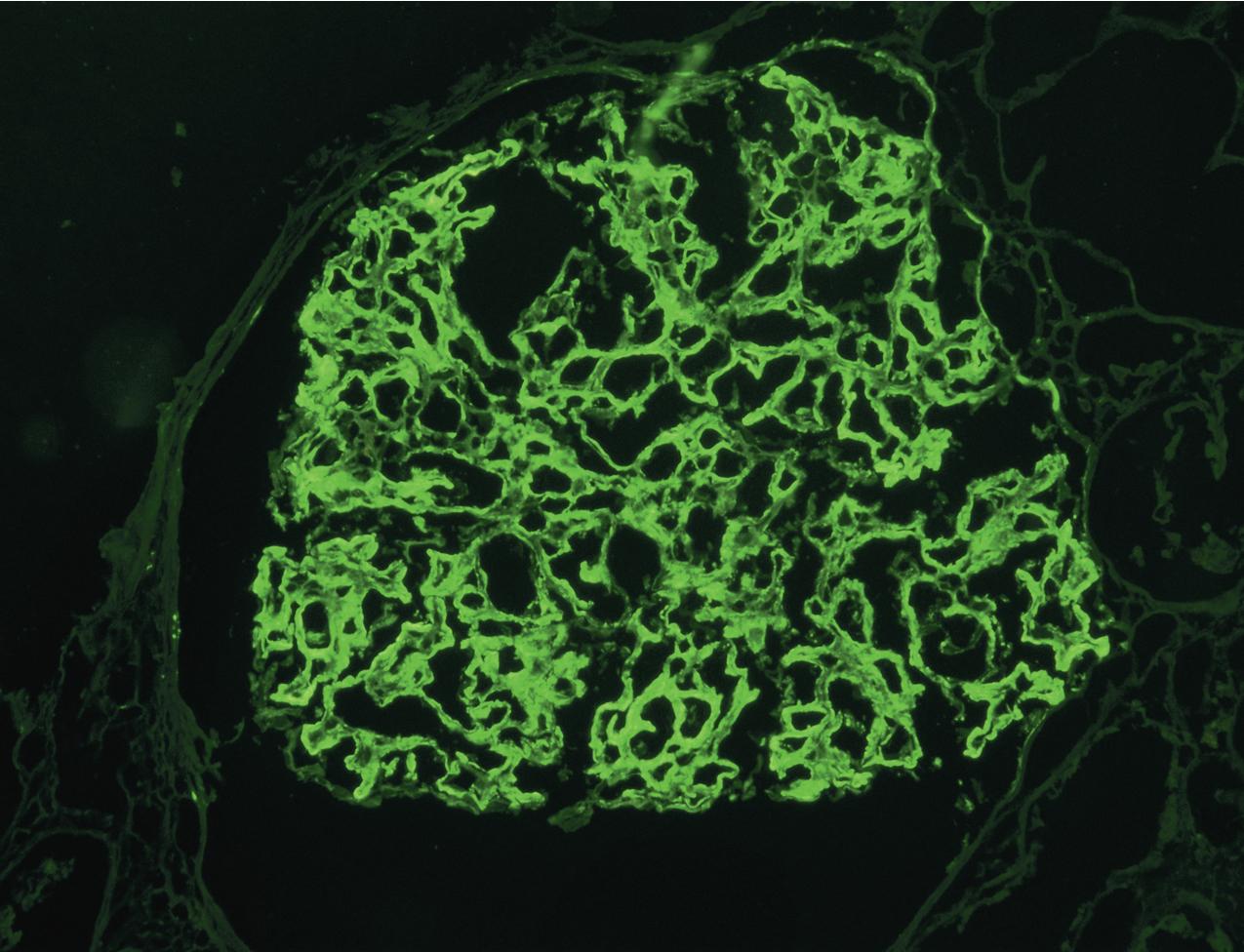
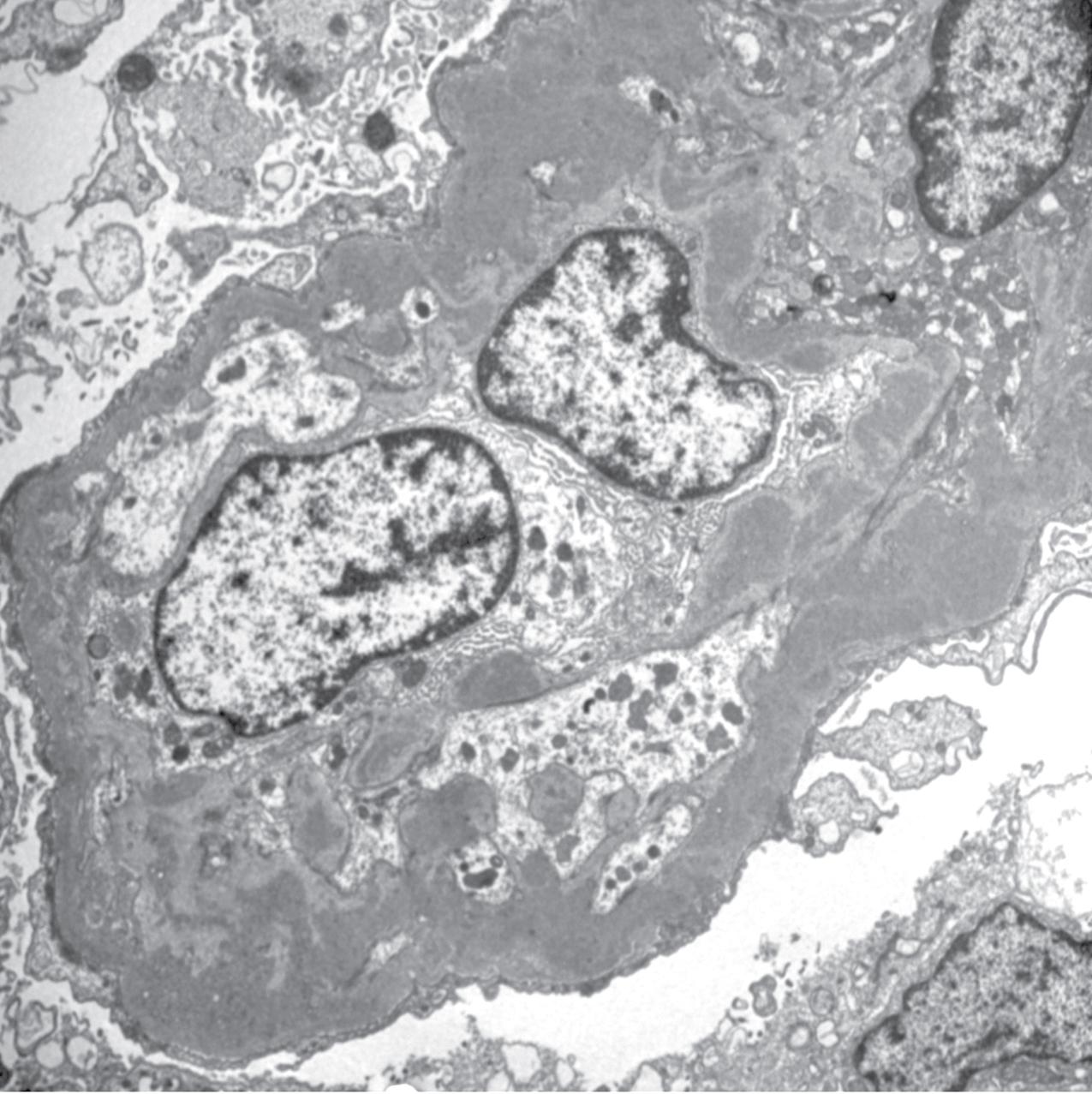
By EM, mesangial, subendothelial, and occasional subepithelial deposits, including occasional humps, can be present, without dense transformation of the GBMs ( Fig. 3.68 ). Some cases have intermediate density or very segmental areas of increased density, indicating an intermediate lesion between DDD and C3GN. Whether the density of such deposits has specific prognostic or etiologic importance is currently not known. The subendothelial deposits are varied but often show a characteristic slender elongated appearance, with density only slightly increased compared with the GBM. C3GN can include lesions previously classified as MPGN type III, with numerous subepithelial deposits in addition to subendothelial and mesangial deposits by EM ( Fig. 3.69 ). C3 nephritic factor or complement regulatory gene mutations have been found in many of these patients. Thus the pattern of injury previously called MPGN type III is now recognized as part of the spectrum of C3GN.
C3 glomerulopathy encompasses a group of disorders with isolated or dominant (two steps greater than other immune reactants) C3 deposition in glomeruli and includes DDD (see earlier) and C3GN. Abnormalities of complement regulatory proteins have been identified in many C3 glomerulopathy patients. Factor H is a key inhibitory molecule that controls constant tick over and activation of C3 in the circulation. Acquired antibodies to factor H, C3NeF (an autoantibody to C3 convertase, detected in about half of patients), genetic deficiency of factor H, or deficiency or genetic variants of other alternative complement pathway proteins have been detected in patients with C3GN. These alterations of complement-related factors include, for example, an internal duplication of CFHR5 , mutation resulting in a hybrid CFHR3-1 gene, and mutation resulting in duplicated CFHR1 . The biopsies in these patients frequently show frequent subepithelial deposits, resembling what previously was called “MPGN type III” (see previous section). CFHR5 nephropathy due to a genetic variant of CFHR5 was first diagnosed in two Cypriot families with inherited renal disease with morphologic findings of C3GN. In these kindreds, 80% of men but only 21% of women older than 50 years reached end-stage kidney disease, with reasons for this sex inequality unknown. Overall, the identified mutations and variants indicate that abnormal CFHR proteins promote unusual dimer and/or multimer formation that impacts on C3 regulation within the fluid phase in the glomerulus, resulting in abnormal C3 deposition.
In some adult patients, C3GN is related to an underlying monoclonal Ig where the monoclonal Ig triggers abnormal C3 activation, but there is no deposition of the monoclonal protein itself within the kidney. Treatment of the monoclonal gammopathy has resulted in improvement in the kidney disease in some of these patients. Conversely, persistence of the monoclonal protein was present in some patients who then had recurrent C3GN in the transplant.
Of note, some patients present after an infection and have initial clinical features and biopsy appearance typical of postinfectious/infection-related GN, with exudative hypercellular/proliferative appearance, strong C3 and lesser IgG, even with hump-type deposits by EM. A prolonged atypical clinical course has led to rebiopsy in some such patients, which showed typical C3GN. Complement dysregulation with abnormalities, including mutations and/or autoantibodies affecting key complement regulatory molecules, were then uncovered. Anti–factor B antibodies in the serum were even detected in a study of apparent typical postinfectious GN patients, indicating complement dysregulation contributes to the pathogenesis of that disease.
Dominance of C3 staining
Absent or scanty immunoglobulin deposition in glomeruli
Deposits by electron microscopy, including mesangial, elongated subendothelial, and occasionally humps (may have variable density, but by definition they are not dense as in dense deposit disease)
Dense deposit disease: Characteristic dense transformation of glomerular basement membrane and dense deposits in mesangium.
C3 glomerulonephritis (C3GN): dominant C3 (at least two intensity steps greater than other immune reactants) with no or sparse immunoglobulin deposits, exclusion of postinfectious glomerulonephritis (GN). C3GN patients often have complement regulatory protein mutation or copy number alteration (e.g., factor H or I).
CFHR5 nephropathy (familial C3GN associated with genetic variant of CFHR5 resulting in internal duplication): Isolated C3 deposits, subendothelial deposits by electron microscopy. (Genetic variants/mutations also exist for other key complement-regulatory genes and are then named accordingly.)
Infection-associated GN may ultimately evolve to a C3GN lesion. Clinical follow-up and investigation for underlying complement dysregulation may be necessary for ultimate diagnosis.
In adults with C3GN, monoclonal protein should be considered as a possible trigger for complement dysregulation and complement deposits.
Cook HT. C4d staining in the diagnosis of C3 glomerulopathy. J Am Soc Nephrol. 2015;26:2609–2611.
Fakhouri F, Frémeaux-Bacchi V, Noël LH, et al. C3 glomerulopathy: A new classification. Nat Rev Nephrol. 2010;6:494–499.
Servais A, Frémeaux-Bacchi V, Lequintrec M, et al. Primary glomerulonephritis with isolated C3 deposits: a new entity which shares common genetic risk factors with haemolytic uraemic syndrome. J Med Genet. 2007;44:193–199.
Smith RJH, Appel GB, Blom AM, et al. C3 glomerulopathy: Understanding a rare complement-driven renal disease. Nat Rev Nephrol. 2019;15:129–143.
Xiao X, Pickering MC, Smith RJ. C3 glomerulopathy: The genetic and clinical findings in dense deposit disease and C3 glomerulonephritis. Semin Thromb Hemost. 2014;40:465–471.
Membranous nephropathy was, until recently, the most common cause of nephrotic syndrome in adults in the United States but has since been surpassed by FSGS. Half of patients show microscopic hematuria but do not have active urine sediment. The peak incidence is in the fourth and fifth decades, with men affected more commonly than women (male/female ratio 2:1). Approximately one-third of patients may develop slowly progressive renal disease.
Membranous nephropathy is caused by diffuse, global subepithelial deposits ( Fig. 3.70 ). At an early time point, the only sign by light microscopy is a more rigid-appearing capillary wall without visible deposits ( Fig. 3.71 ). In favorable tangential sections, small areas of lucency seen on Jones silver stain may be detected, representing the lack of silver staining of the deposits ( Fig. 3.72 ). These so-called “pinpoint holes” are the earliest manifestation of membranous nephropathy by light microscopy. As deposits persist, the GBM matrix reaction produces small spike-like protrusions visualized by silver stain ( Figs. 3.73–3.75 ). With progressive basement membrane reaction, the matrix encircles the deposits resulting in a lace-like splitting or laddering appearance of the GBM on silver stain ( Fig. 3.76 ). The morphologic findings related to these subepithelial deposits have been divided into stages (see later).
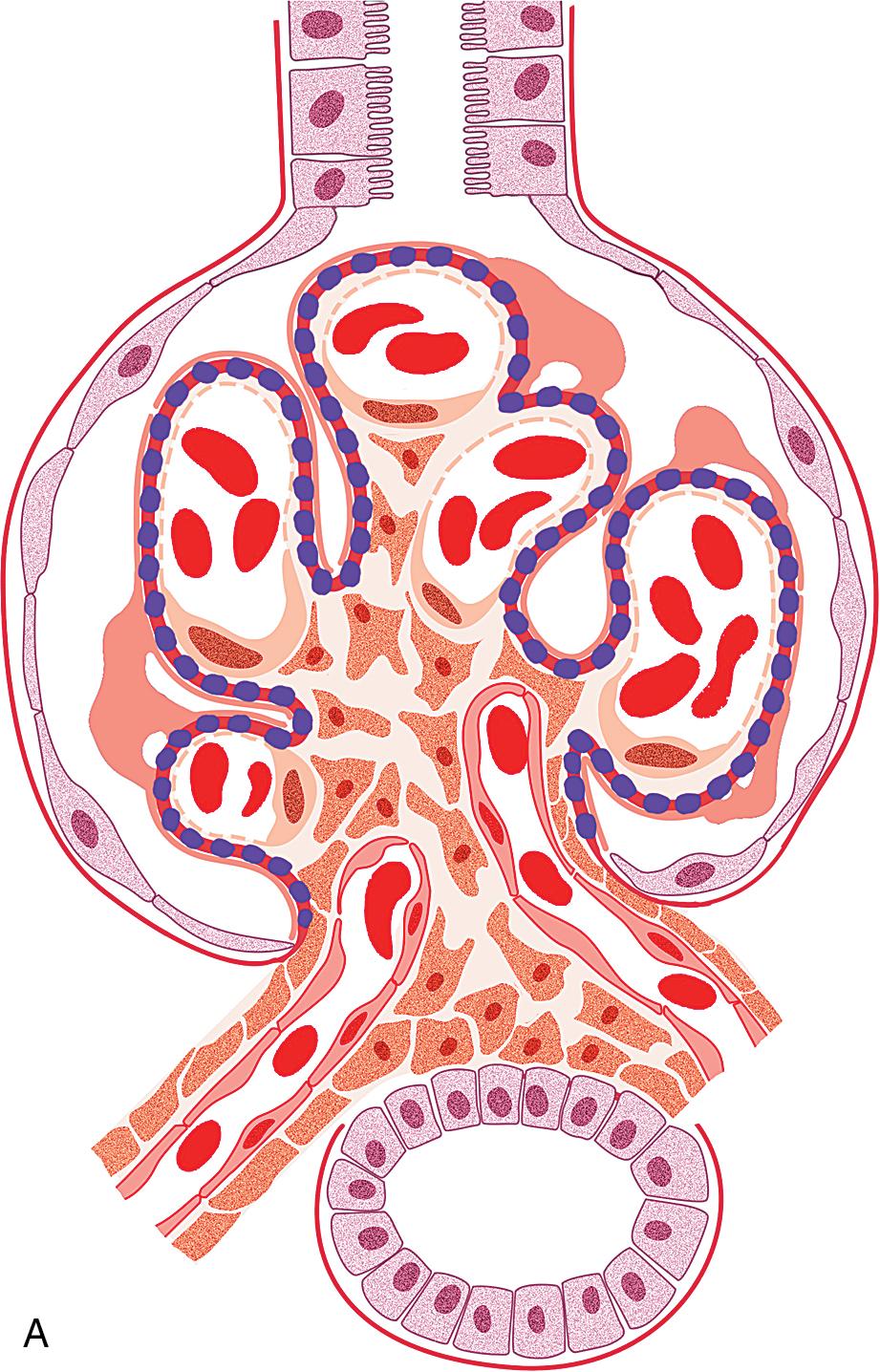
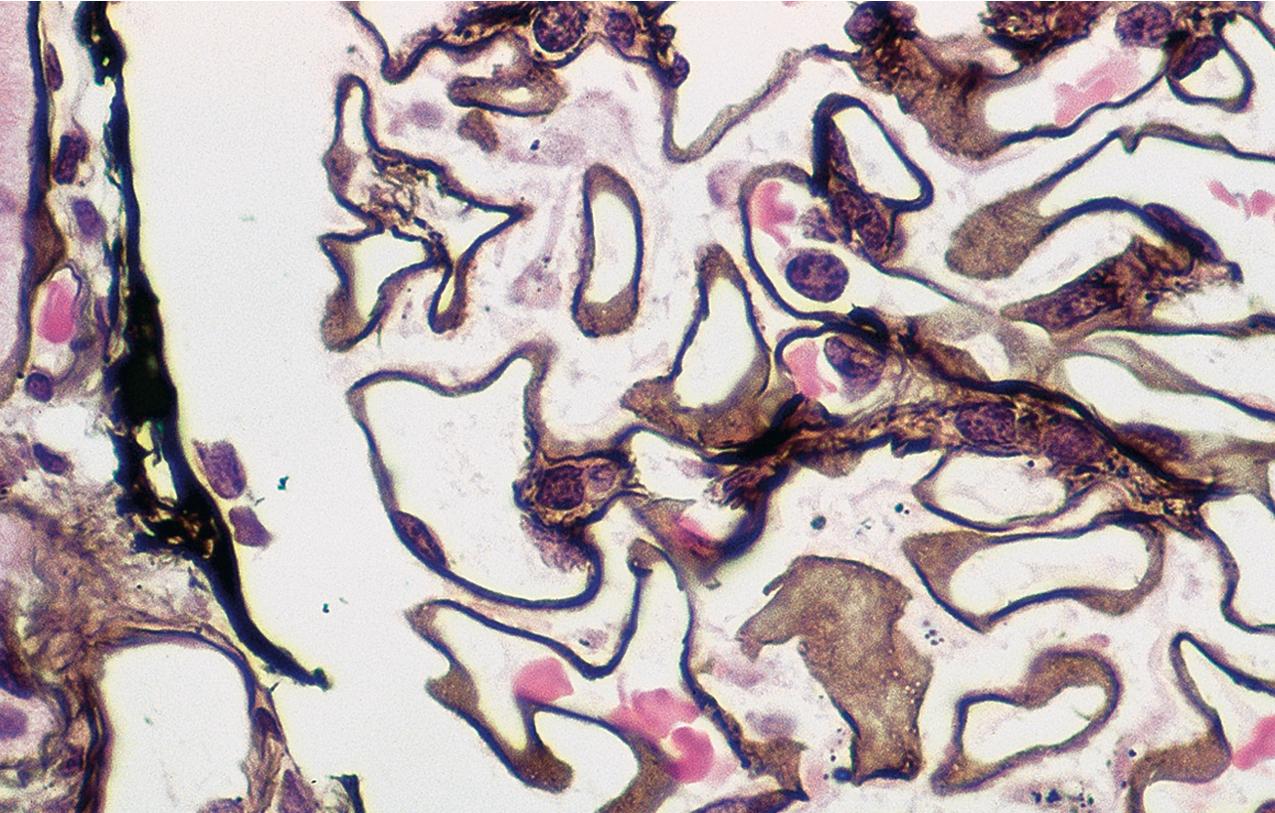
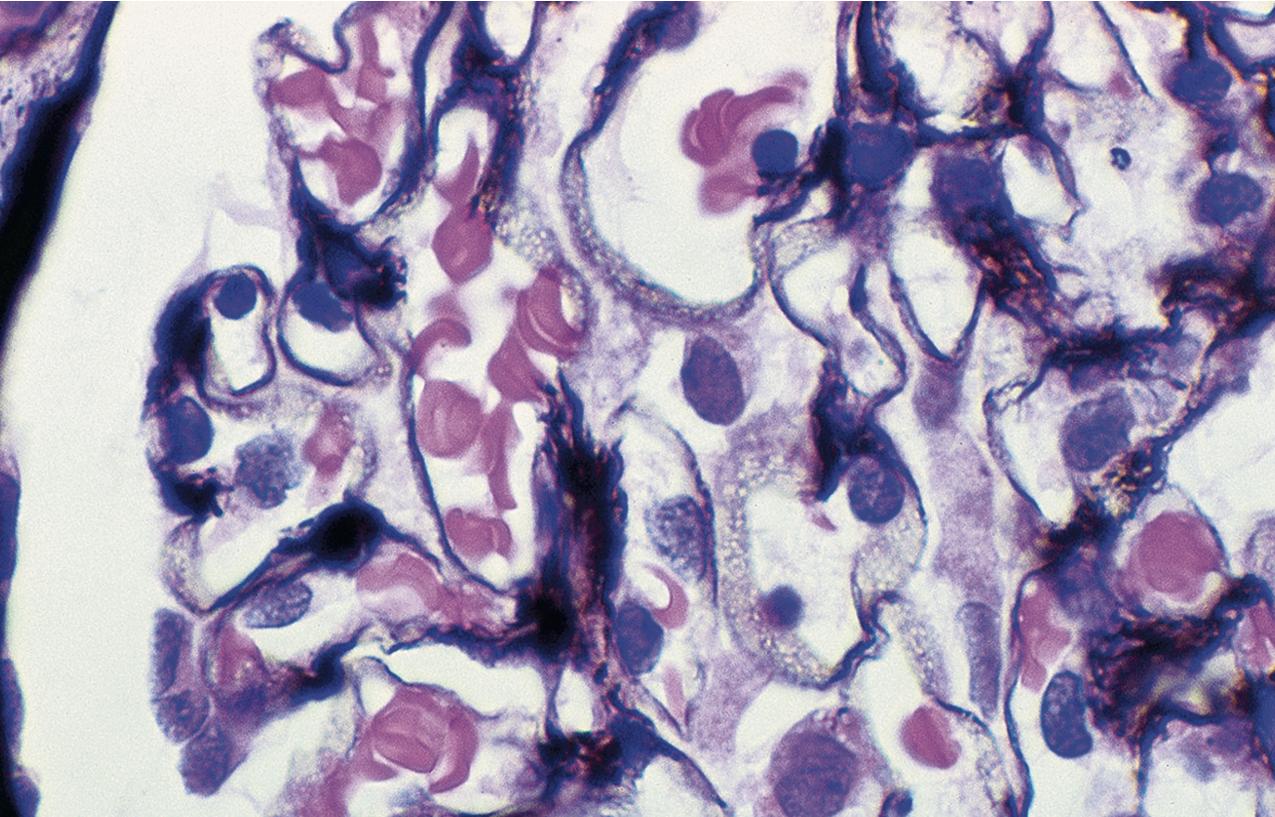
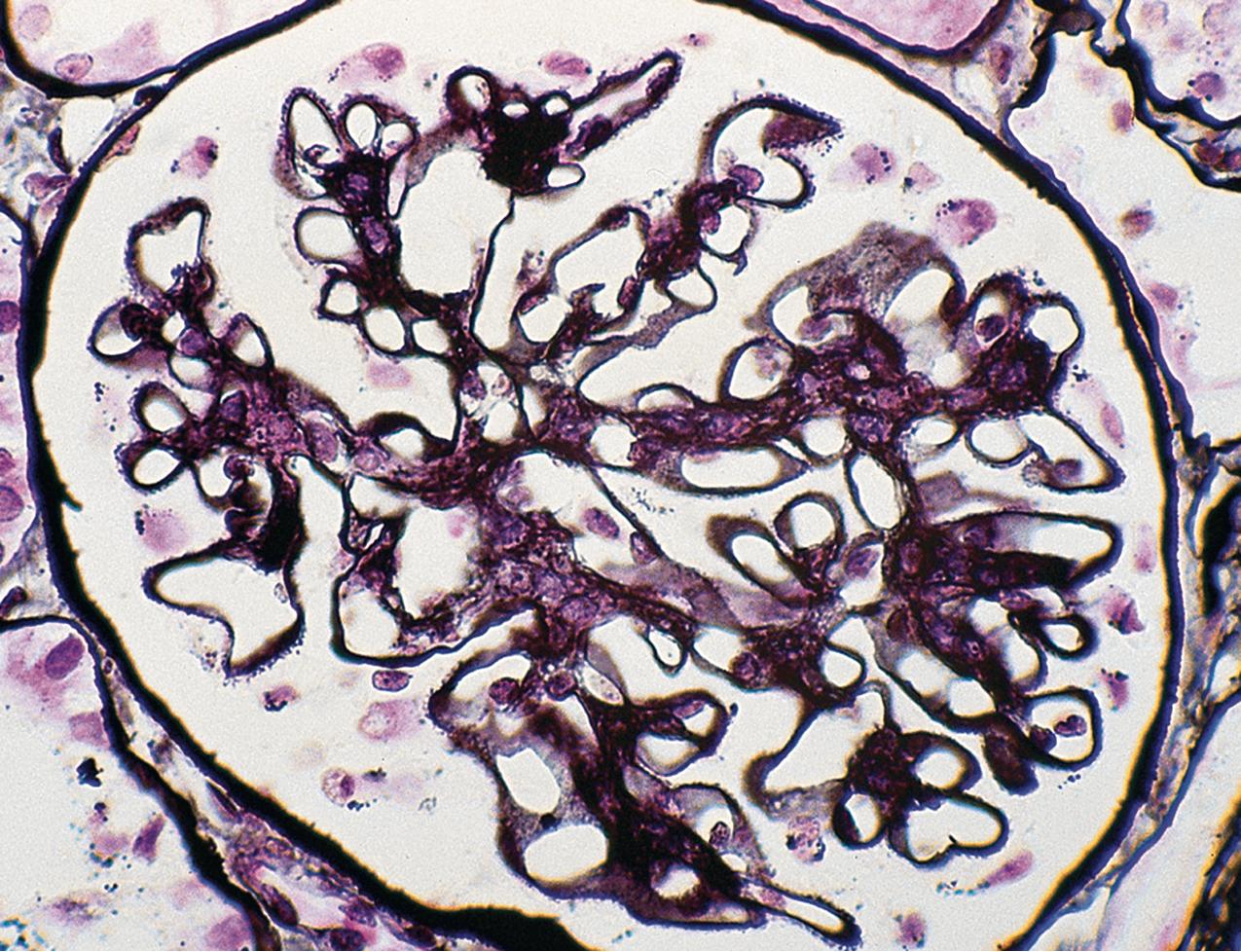
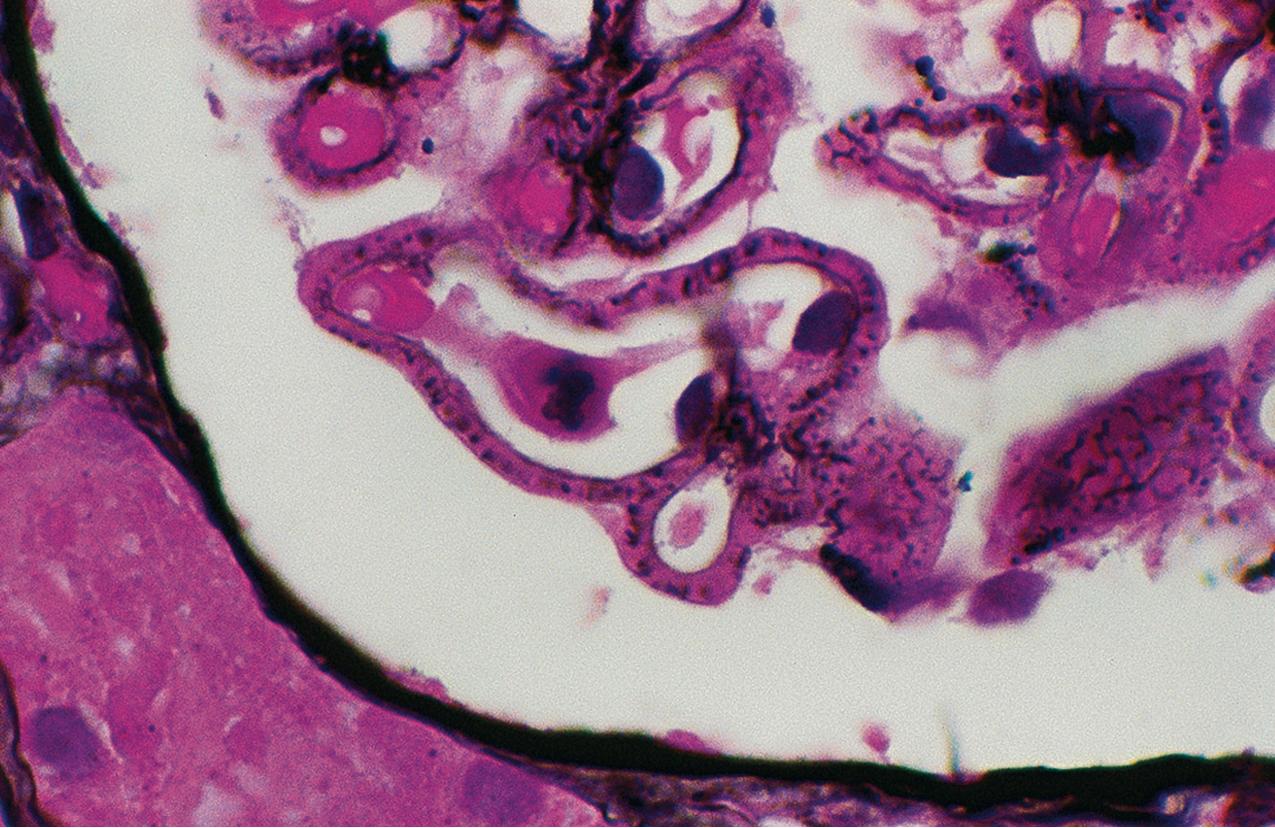
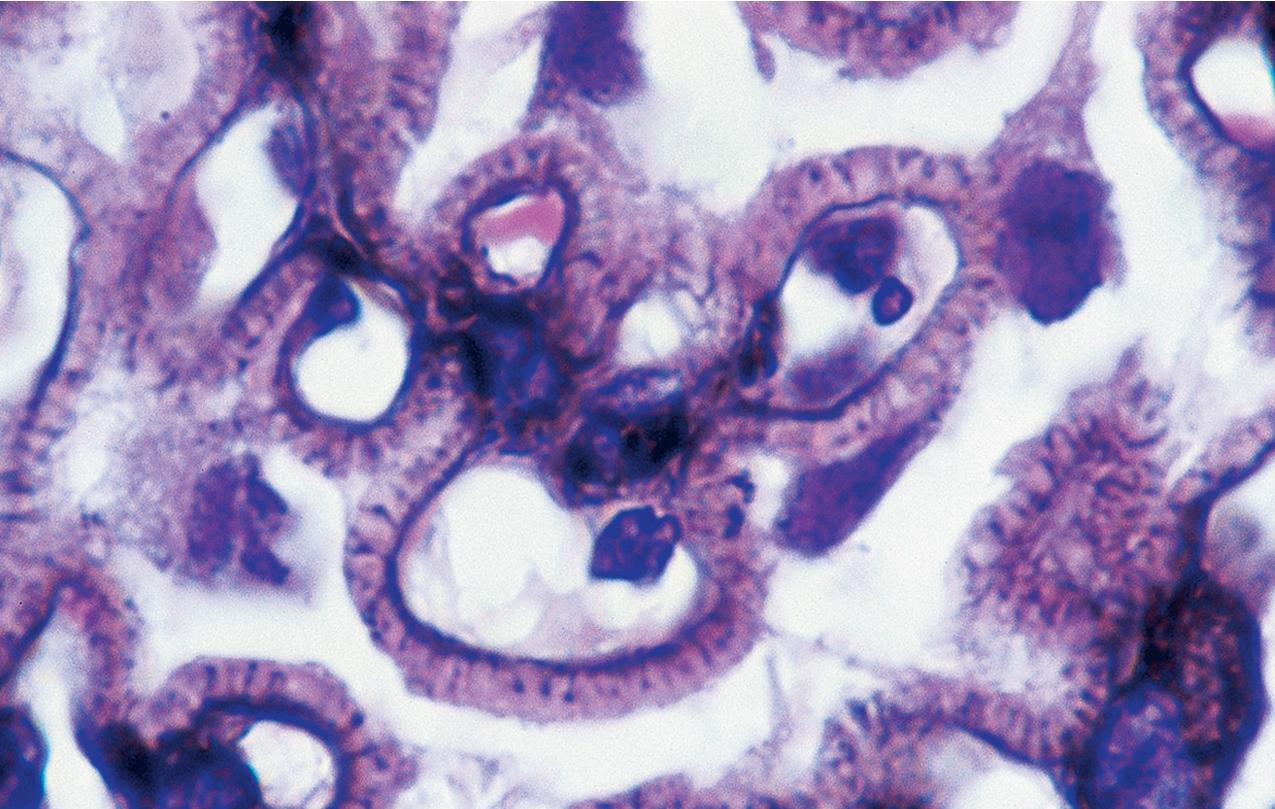
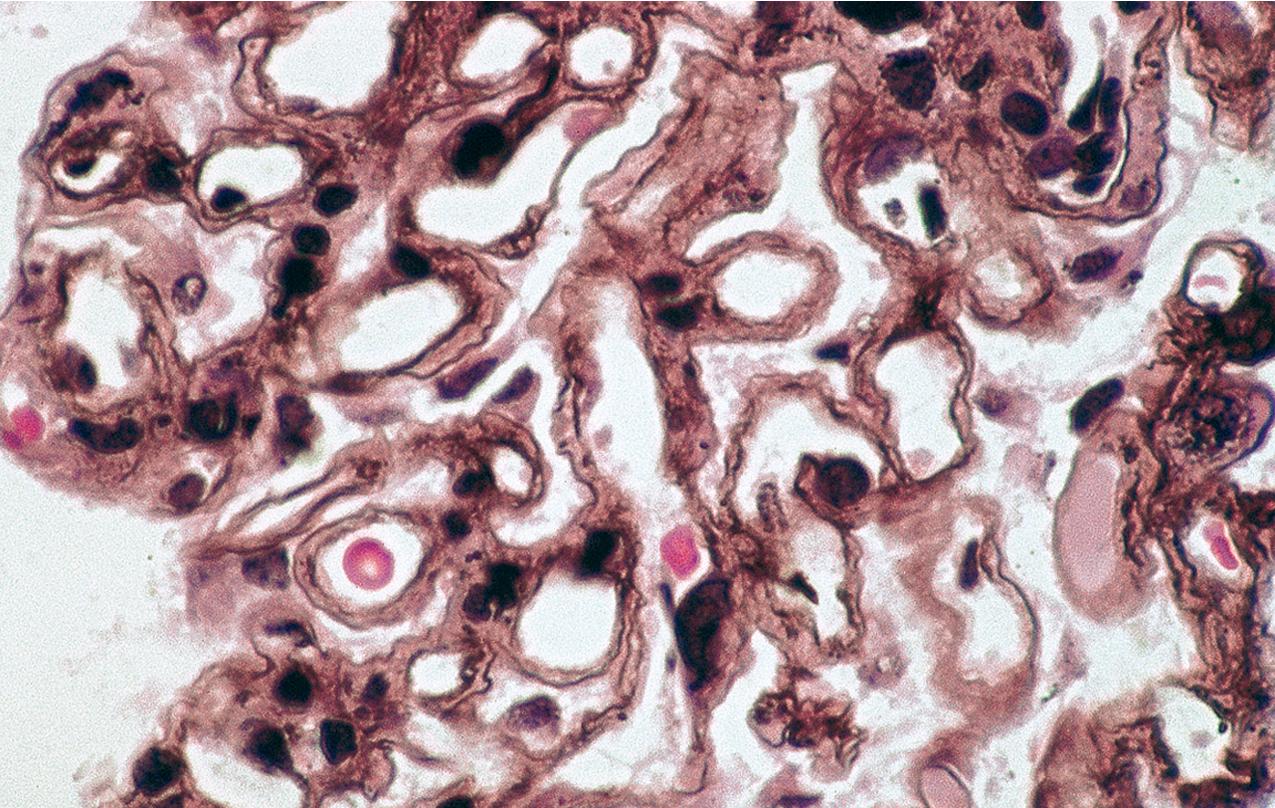
Additional lesions may be present, ranging from crescents to sclerosis. Segmental sclerosis, interstitial fibrosis, and tubular atrophy are associated with worse prognosis ( Figs. 3.77–3.78 ). Rarely, crescents may be found in cases of apparent idiopathic membranous nephropathy but are more common with lupus-associated lesions ( Fig. 3.79 ). Crescents in membranous nephropathy in patients without evidence of SLE should thus raise suspicion of a separate additional disease process, notably anti-GBM antibody–mediated GN or antineutrophil cytoplasmic antibody (ANCA)-associated GN.
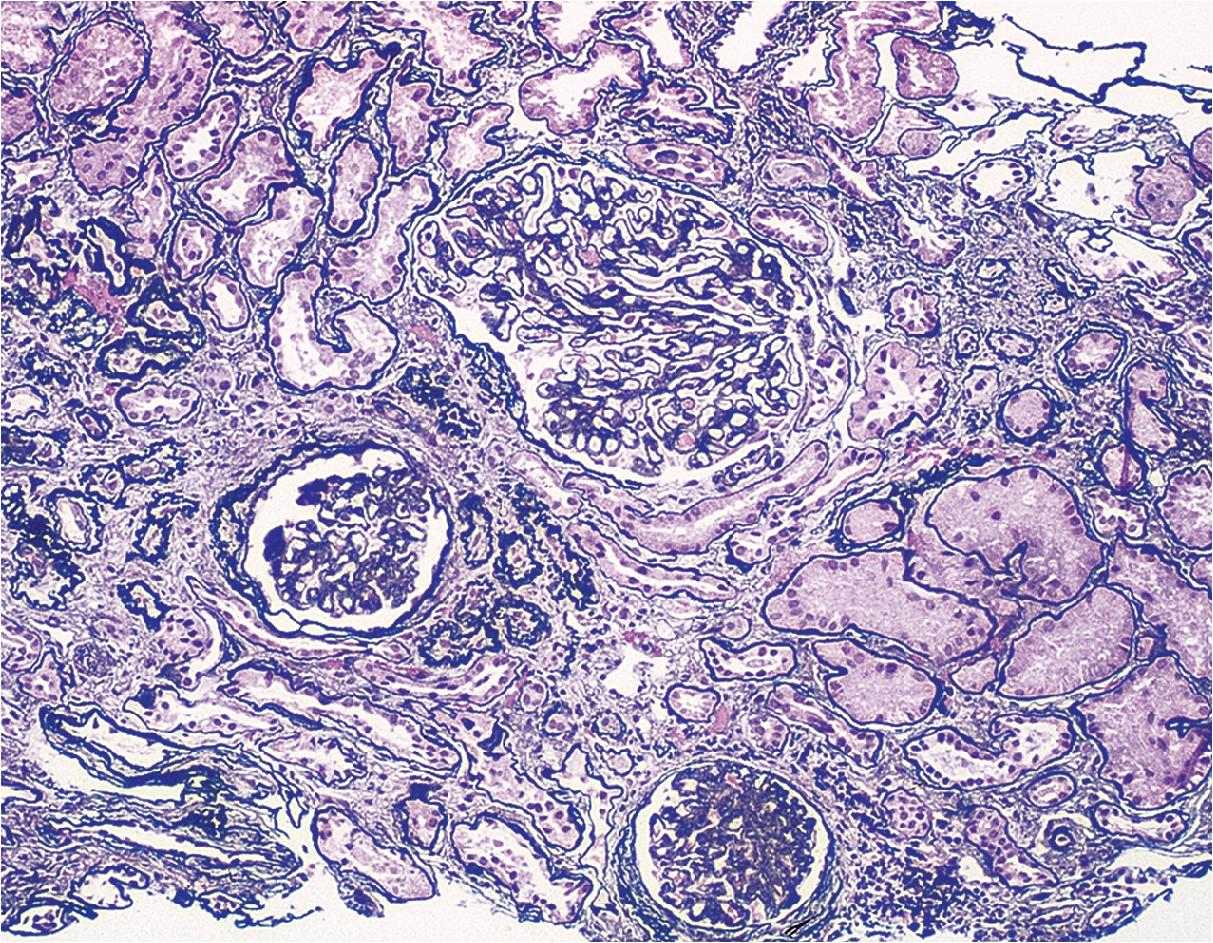
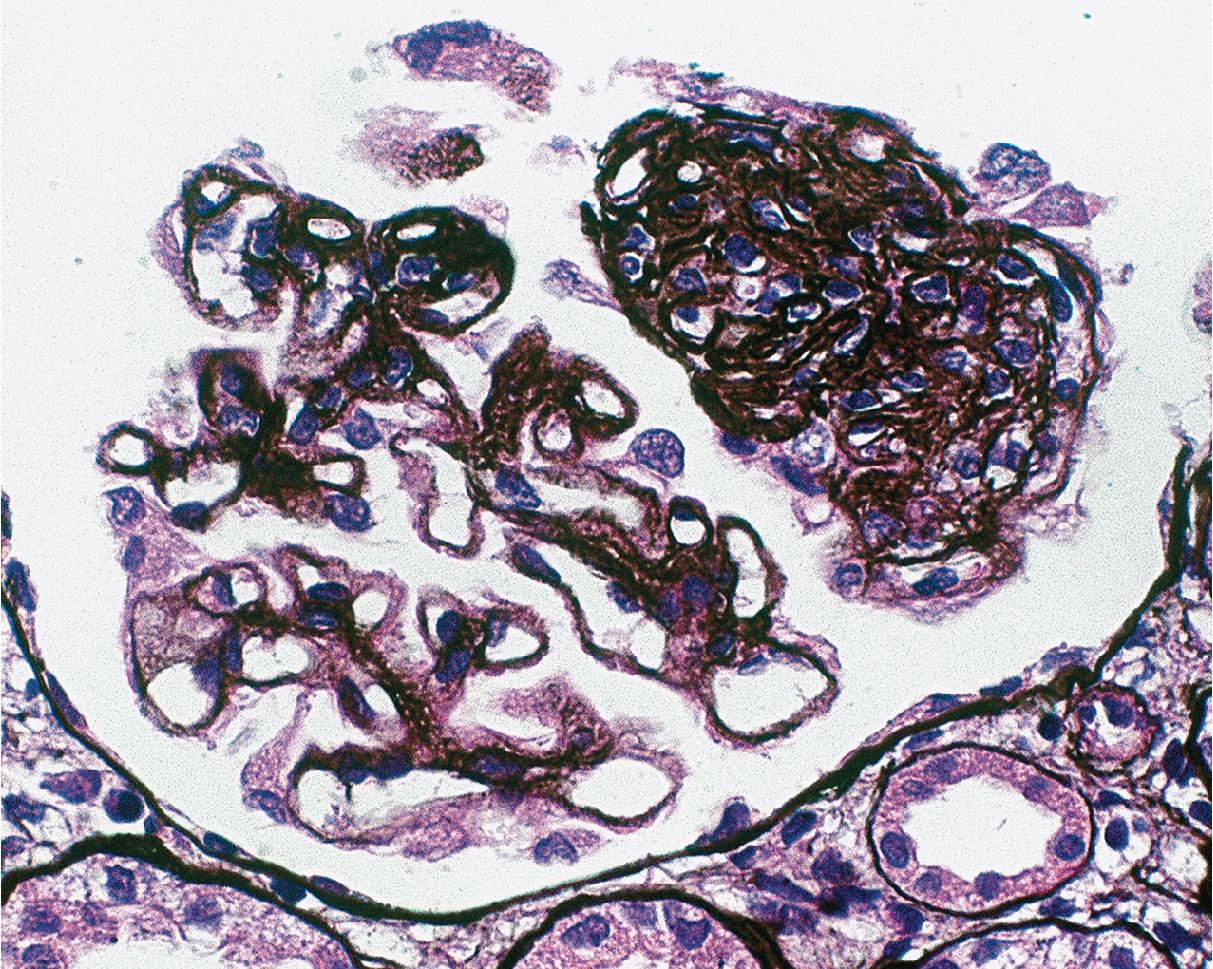
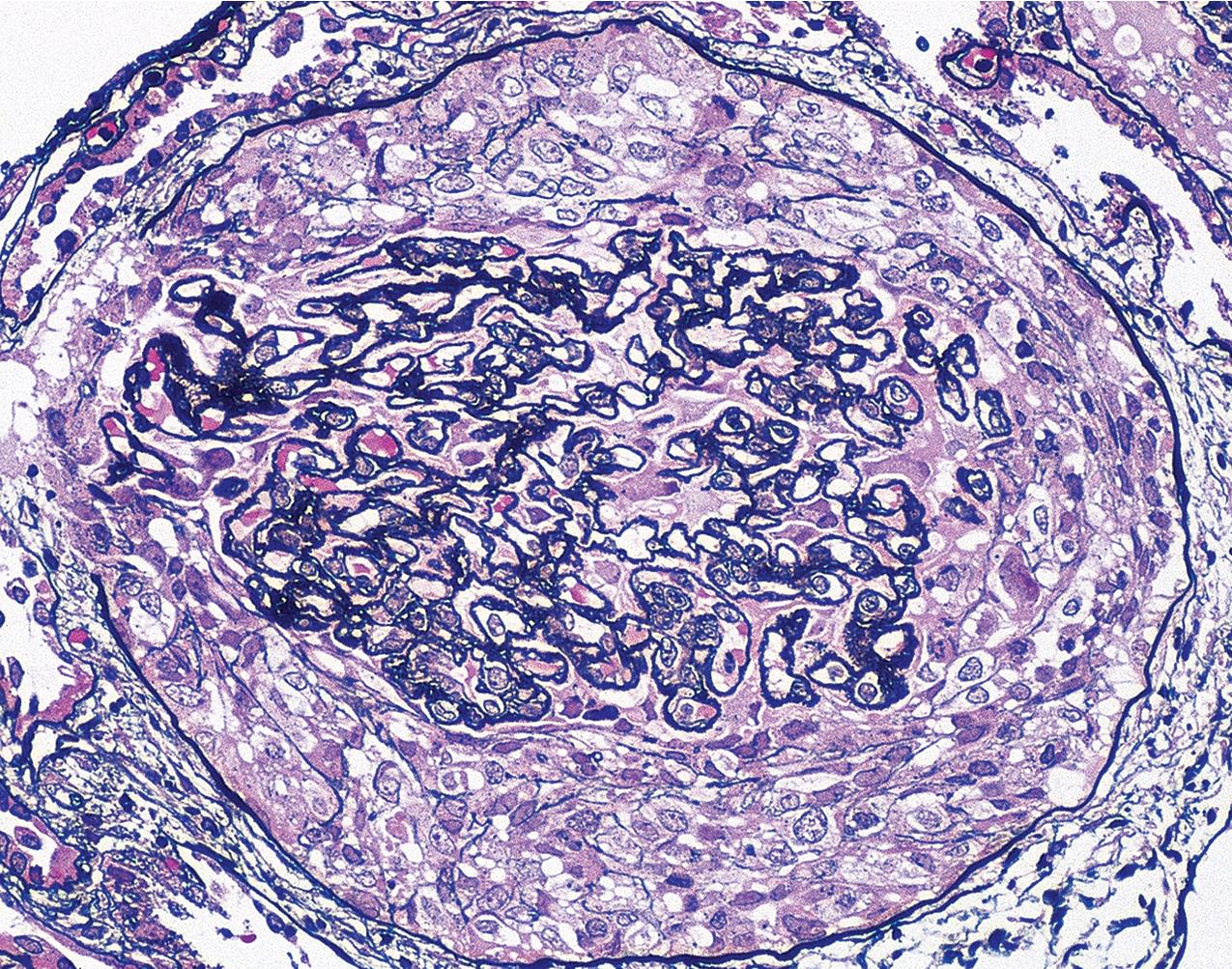
By IF, the subepithelial deposits are visualized as diffuse, global granular positivity along the capillary wall ( Figs. 3.80–3.82 ). IF microscopy is more sensitive than either light microscopy or EM for detection of deposits and is very finely granular in Stage 1 and coarsely granular with more advanced stages. Polyclonal IgG is typically the predominant Ig, and C3 is most often also present. In addition, mesangial deposits are typically present in secondary membranous glomerulopathy and are absent in most cases of idiopathic membranous nephropathy. When IgA, IgM, and C1q are also present in addition to mesangial deposits, the possibility of secondary membranous nephropathy because of SLE should be considered. Although IgG4 is dominant in idiopathic membranous nephropathy versus IgG1 in lupus membranous nephritis and IgG1 and IgG4 in membranous nephropathy associated with malignancy, there is some overlap. These IgG subtypes are not definitive in discerning primary membranous nephropathy versus malignancy-associated cases. Positive granular glomerular capillary wall staining for phospholipase A2 receptor, now recognized as the antigen in around 70% of apparent primary membranous nephropathy, indicates likelihood of a primary membranous nephropathy. Additional antigens are rapidly being identified in PLA2R negative cases of membranous nephropathy, including thrombospondin type 1 domain-containing 7A (THSD7A), exostosin-1/2, neural epidermal growth factor-like 1 protein (NELL-1) and semaphorin 3B, with varying clinical associations (see later).
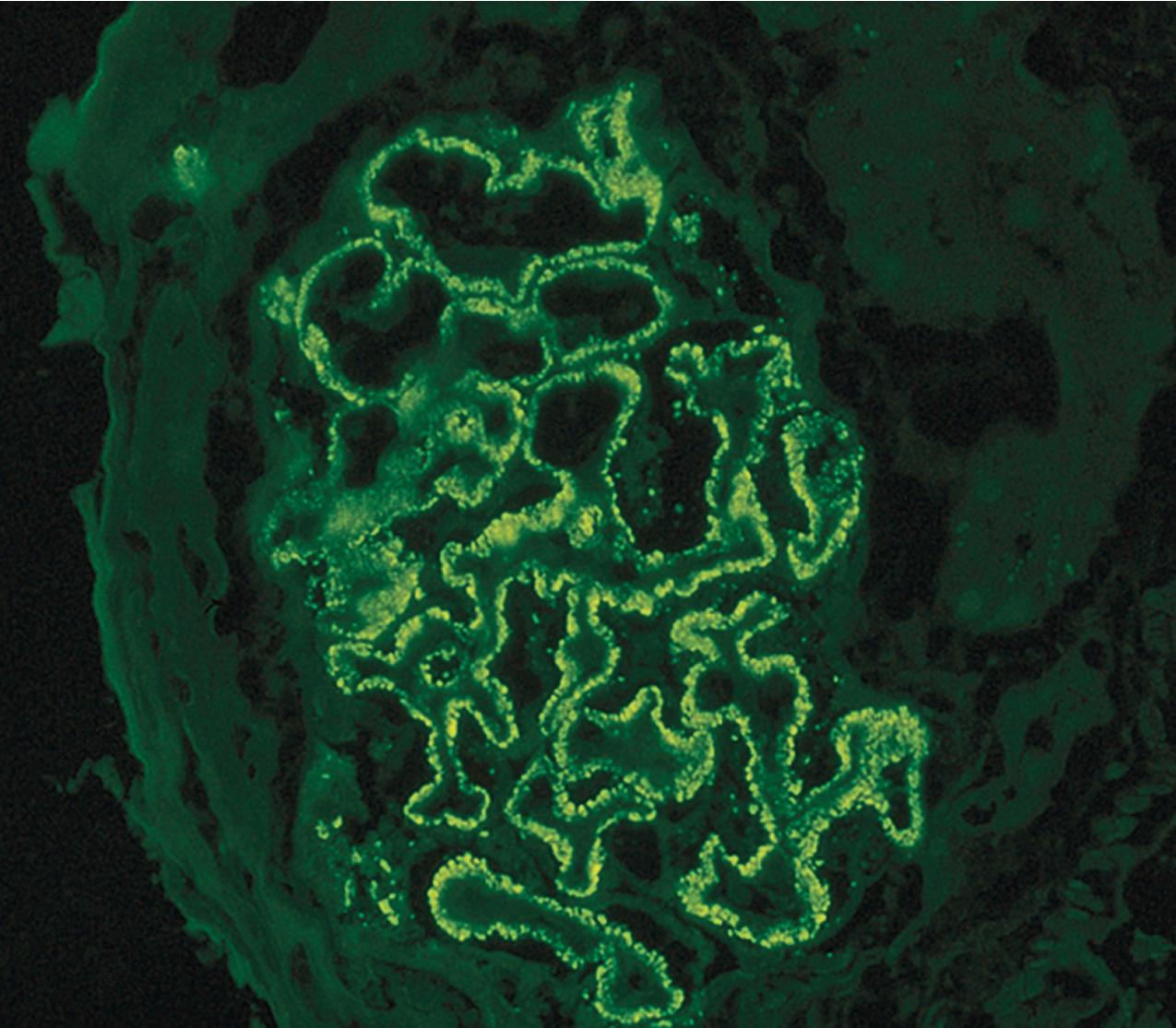
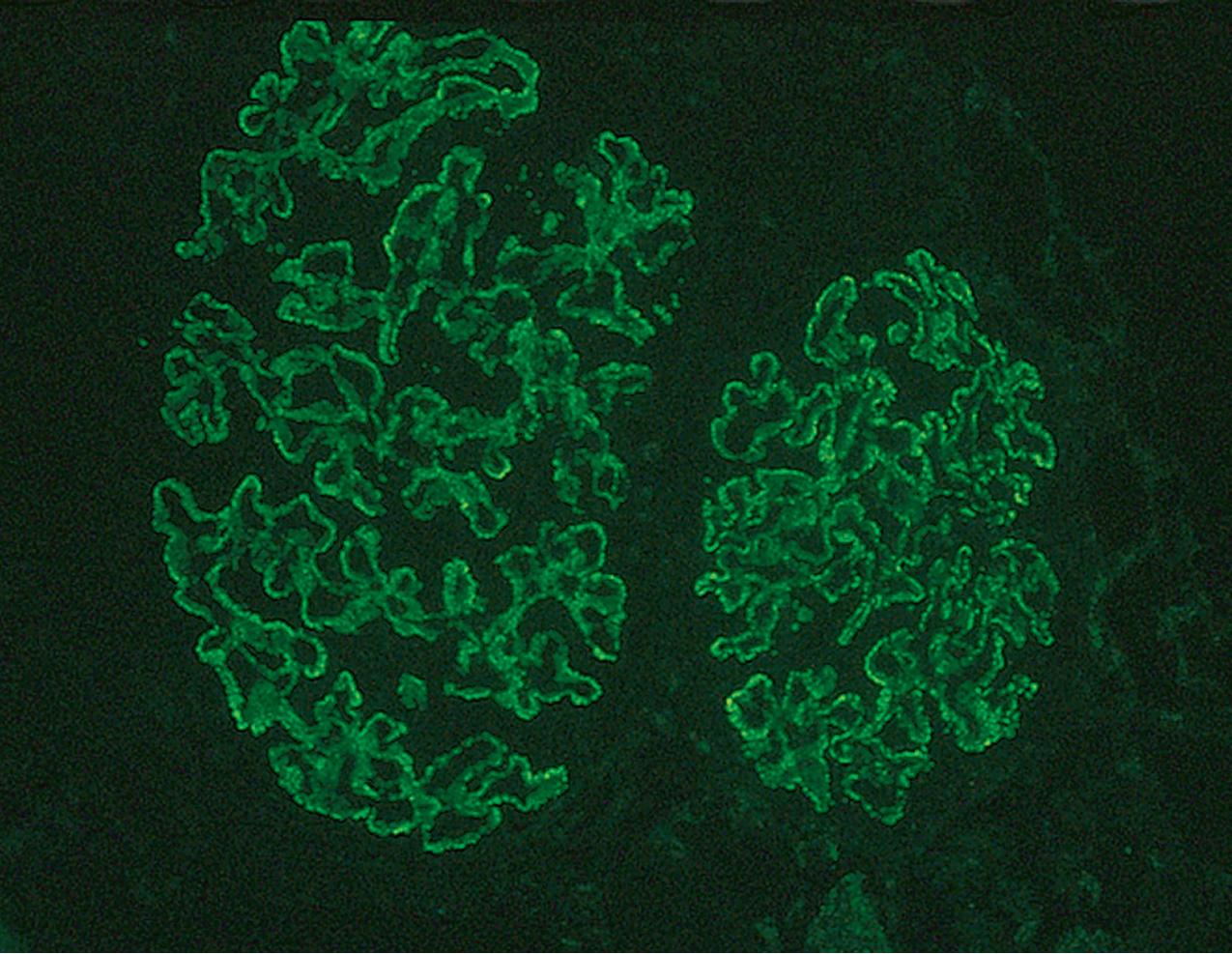
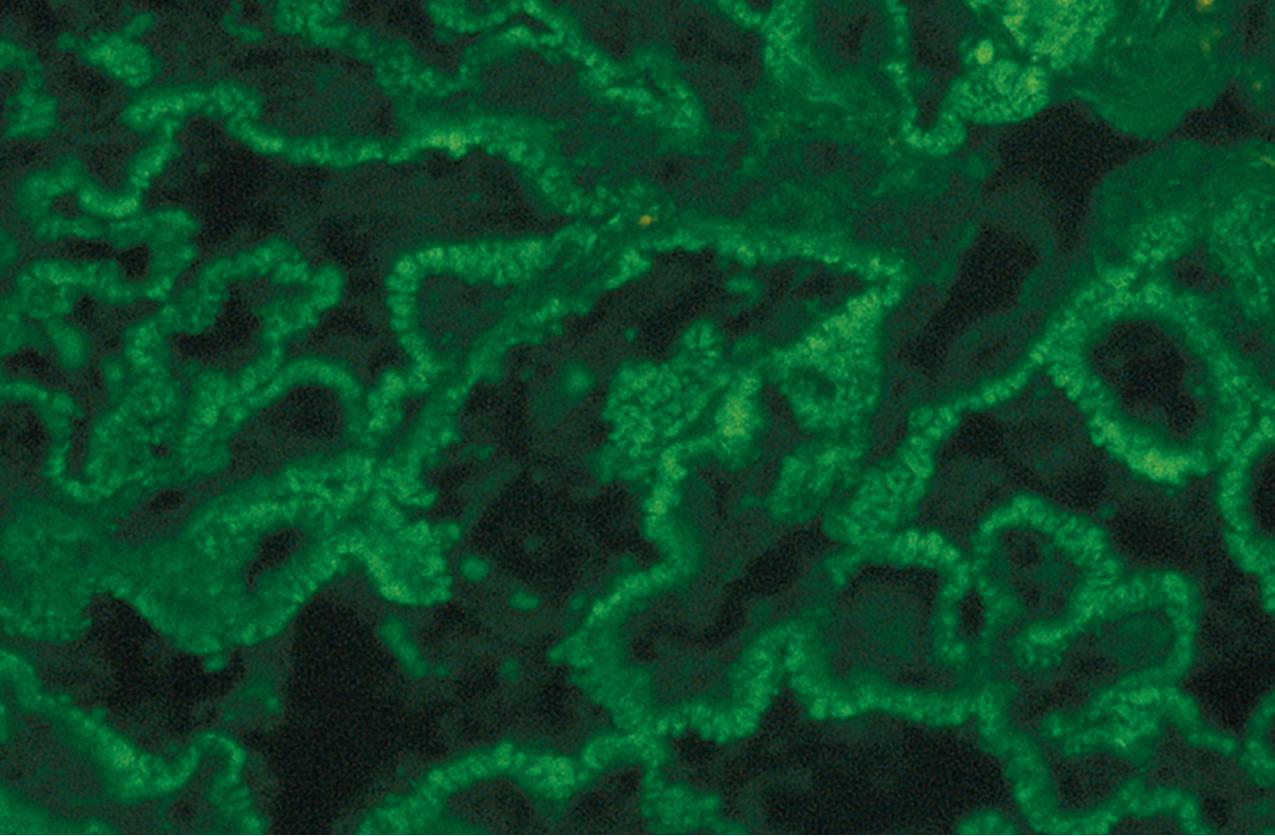
By EM, deposits corresponding to the stage of membranous glomerulopathy are visualized, with varying surrounding GBM reaction. In early stage 1 membranous nephropathy, deposits may be extremely small and inconspicuous with no surrounding GBM reaction, corresponding to the lack of spikes evident by light microscopy ( Figs. 3.83–3.84 ). In stage 2 membranous nephropathy, well-formed spike reaction is present ( Figs. 3.85–3.86 ). In stage 3 membranous nephropathy, the deposits are encircled by the GBM reaction ( Figs. 3.87–3.88 ). In stage 4 membranous nephropathy, the deposits are resorbed, leaving behind rarefied, lucent areas ( Figs. 3.89–3.90 ). Varying depths of deposits with transmembranous and deep deposits have been linked to worse prognosis.
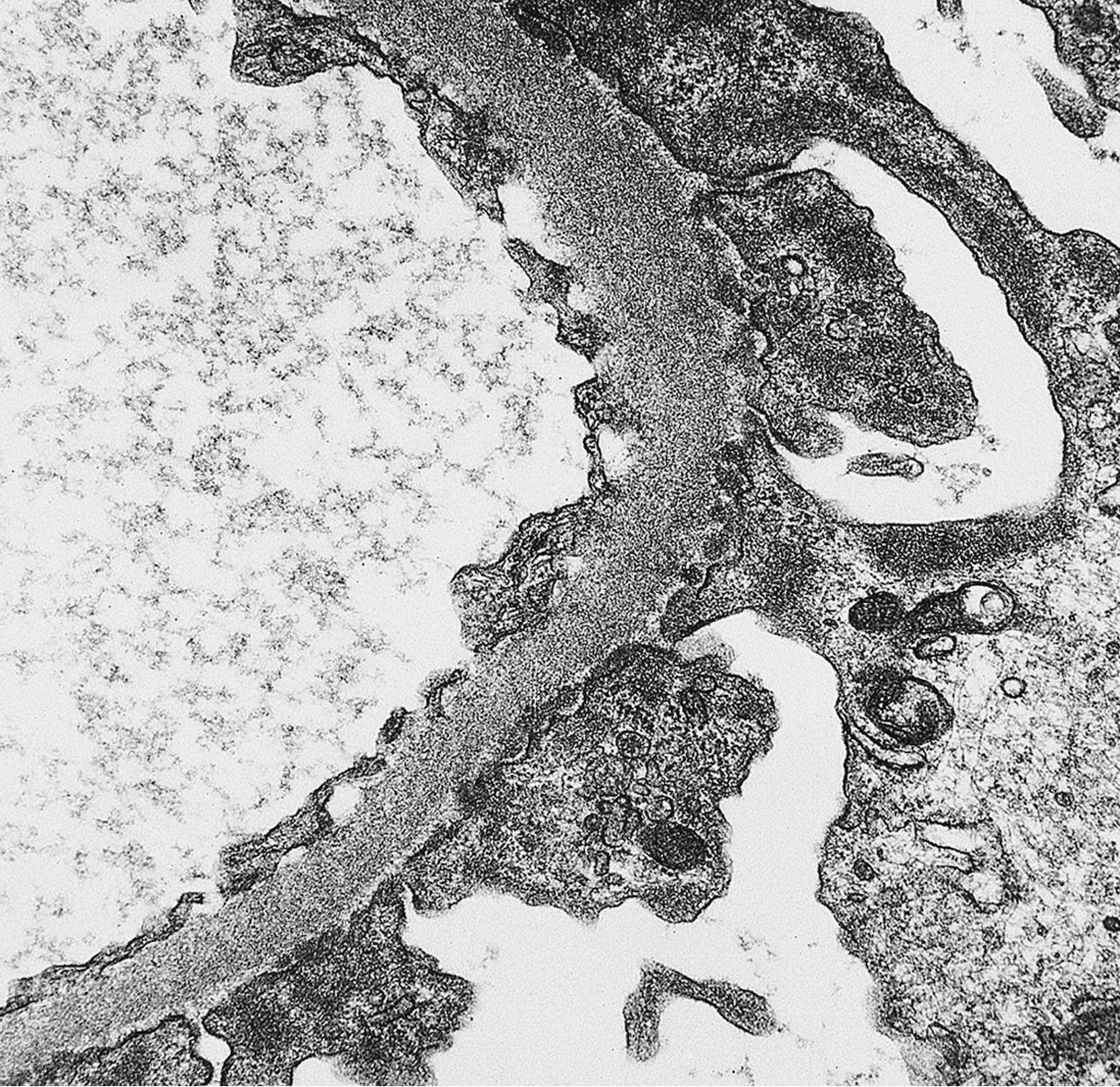
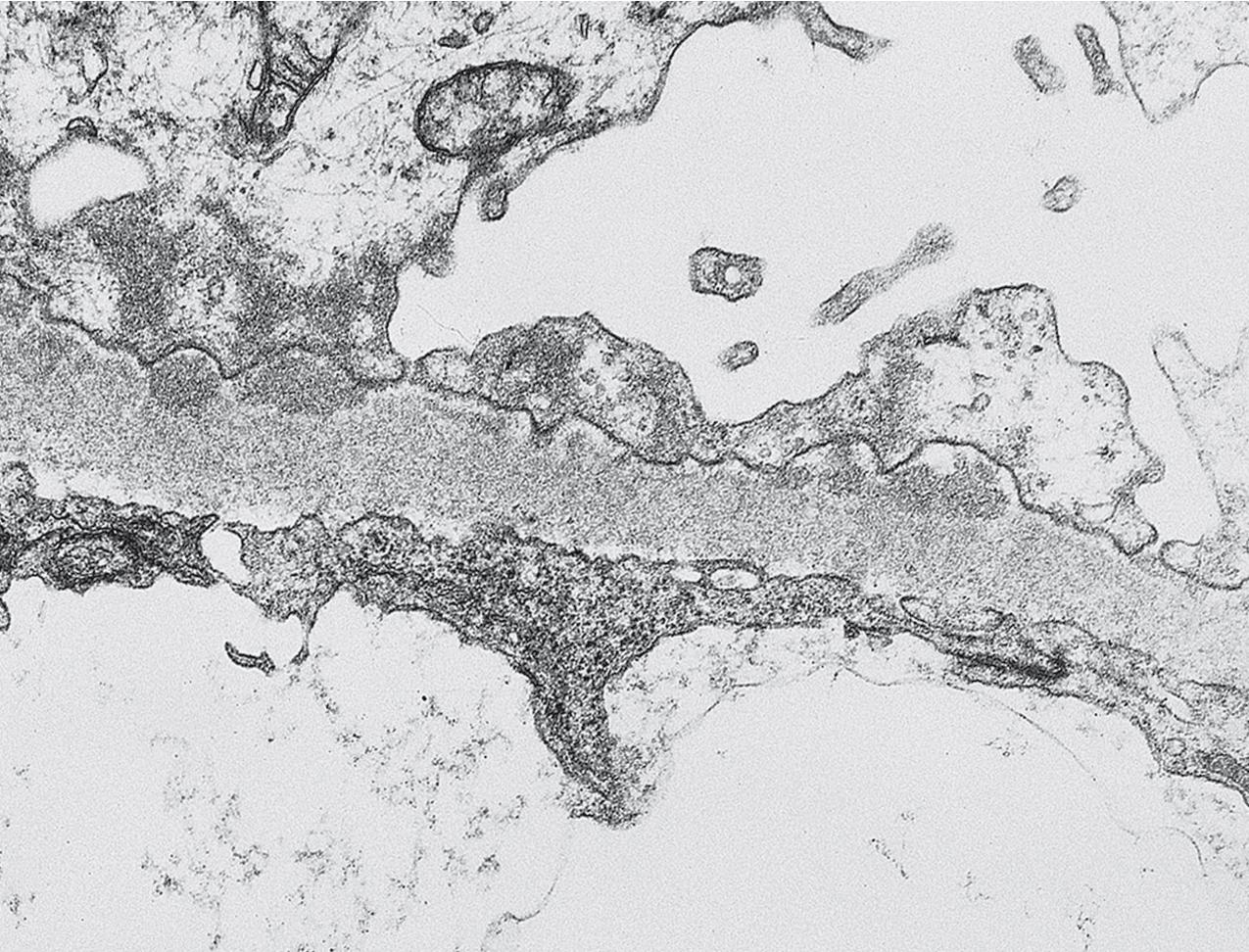
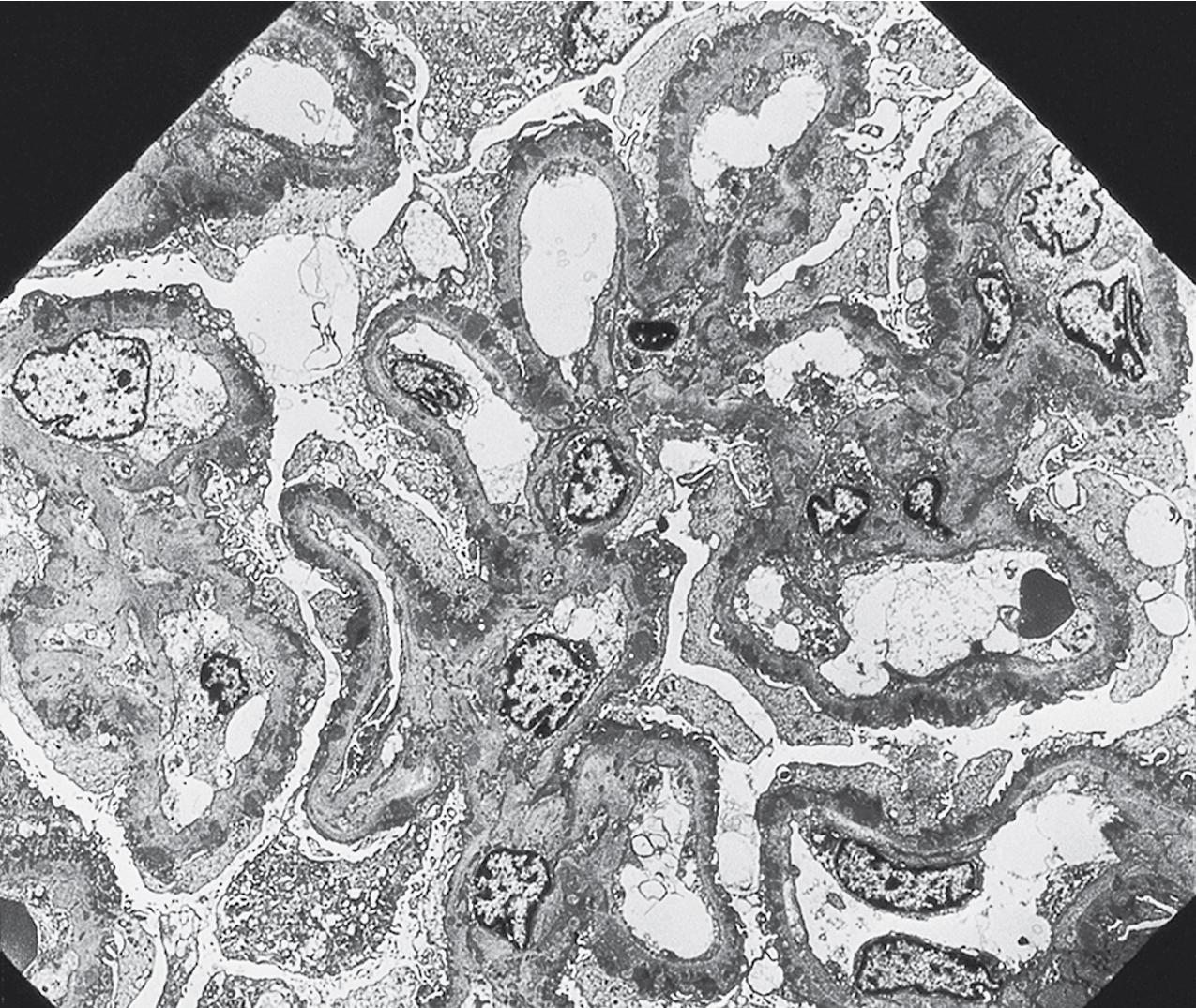
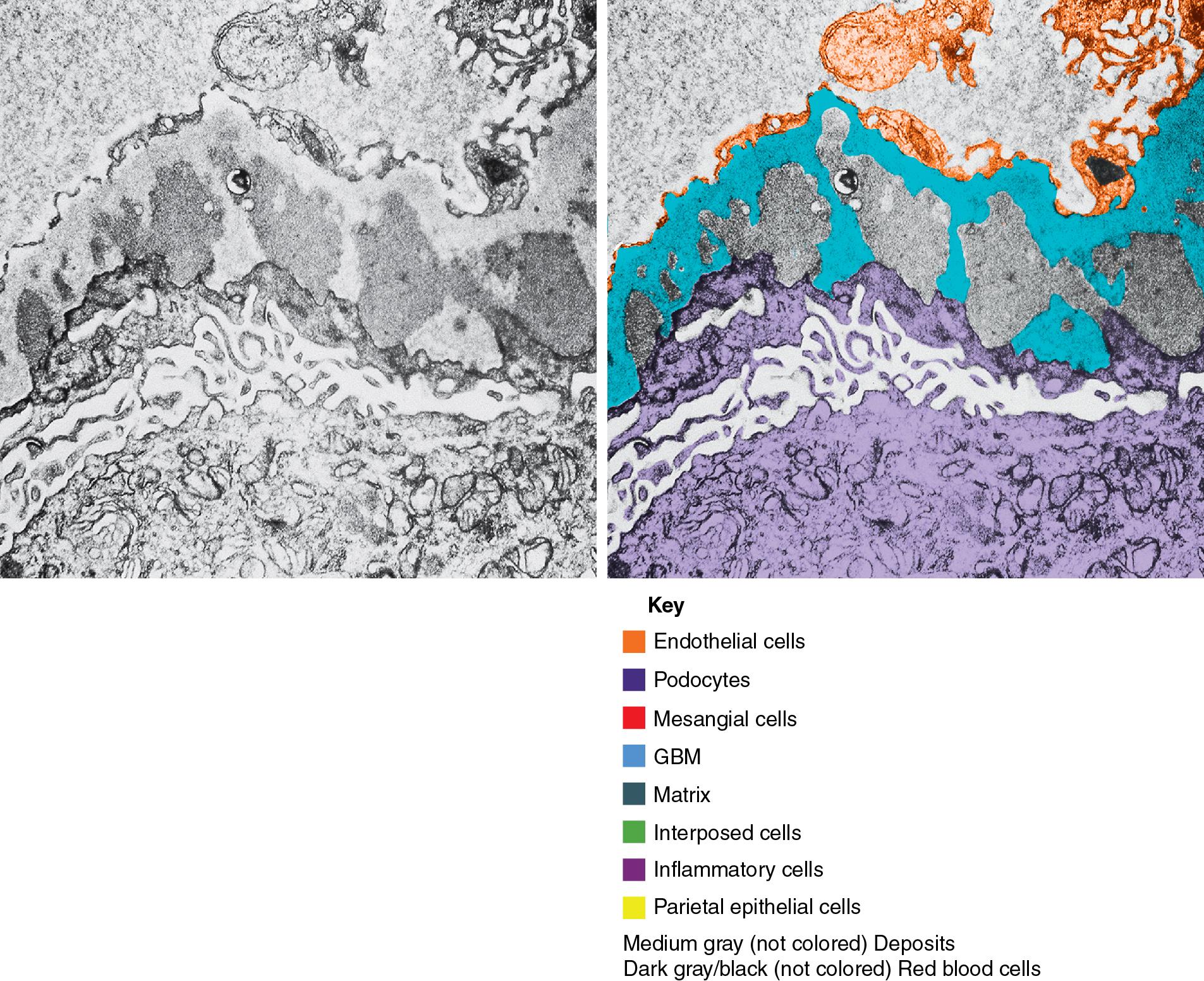
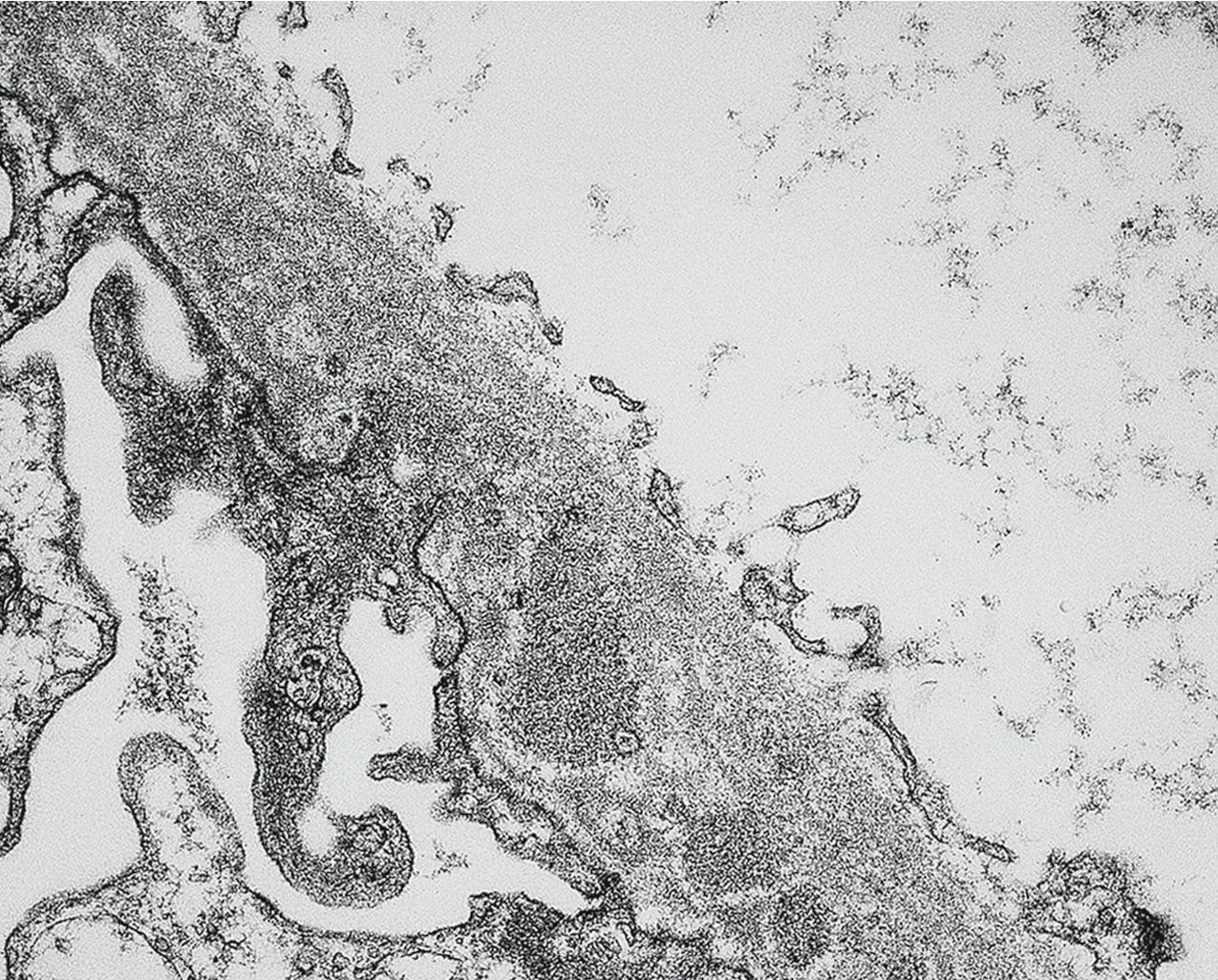
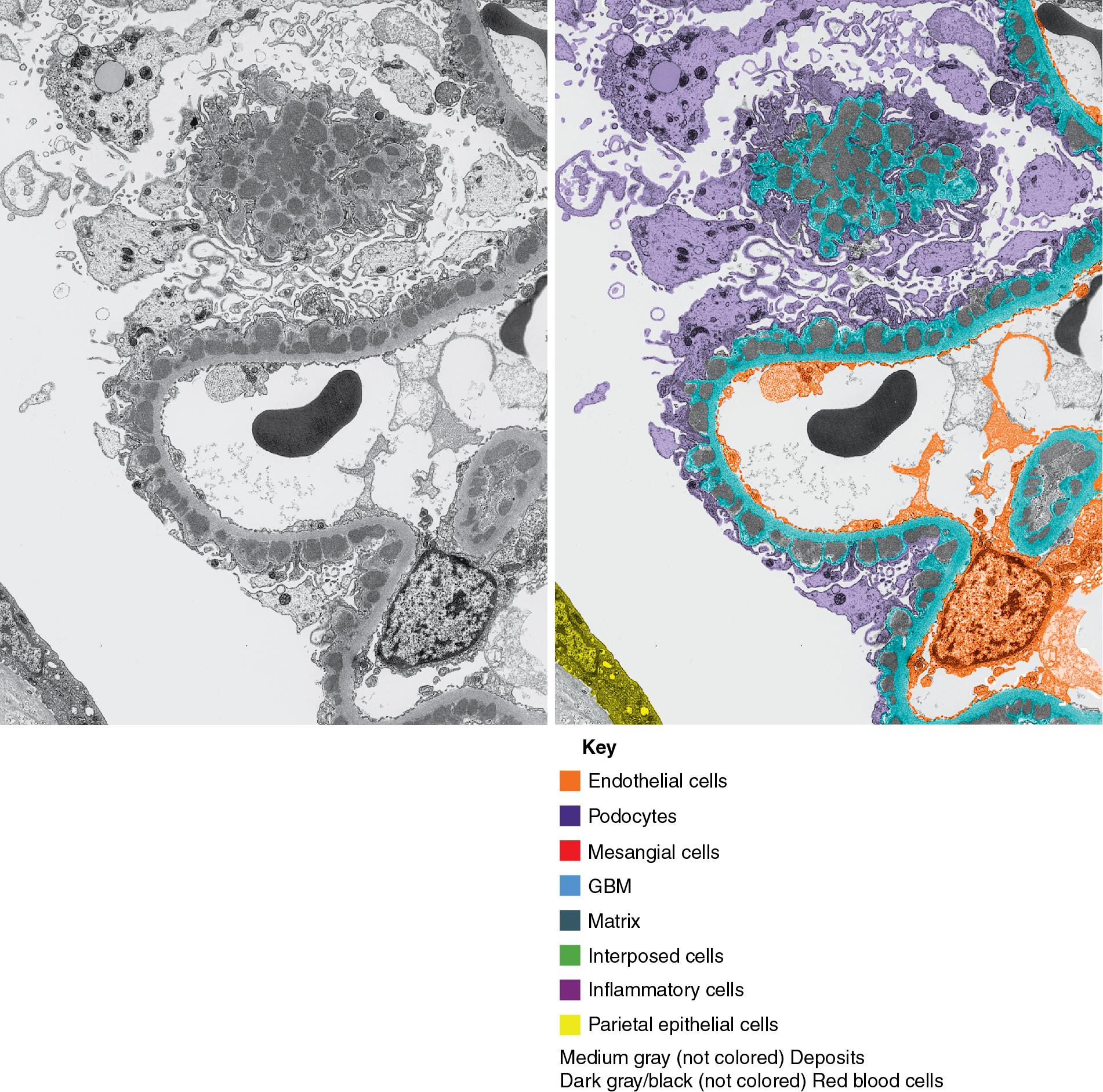
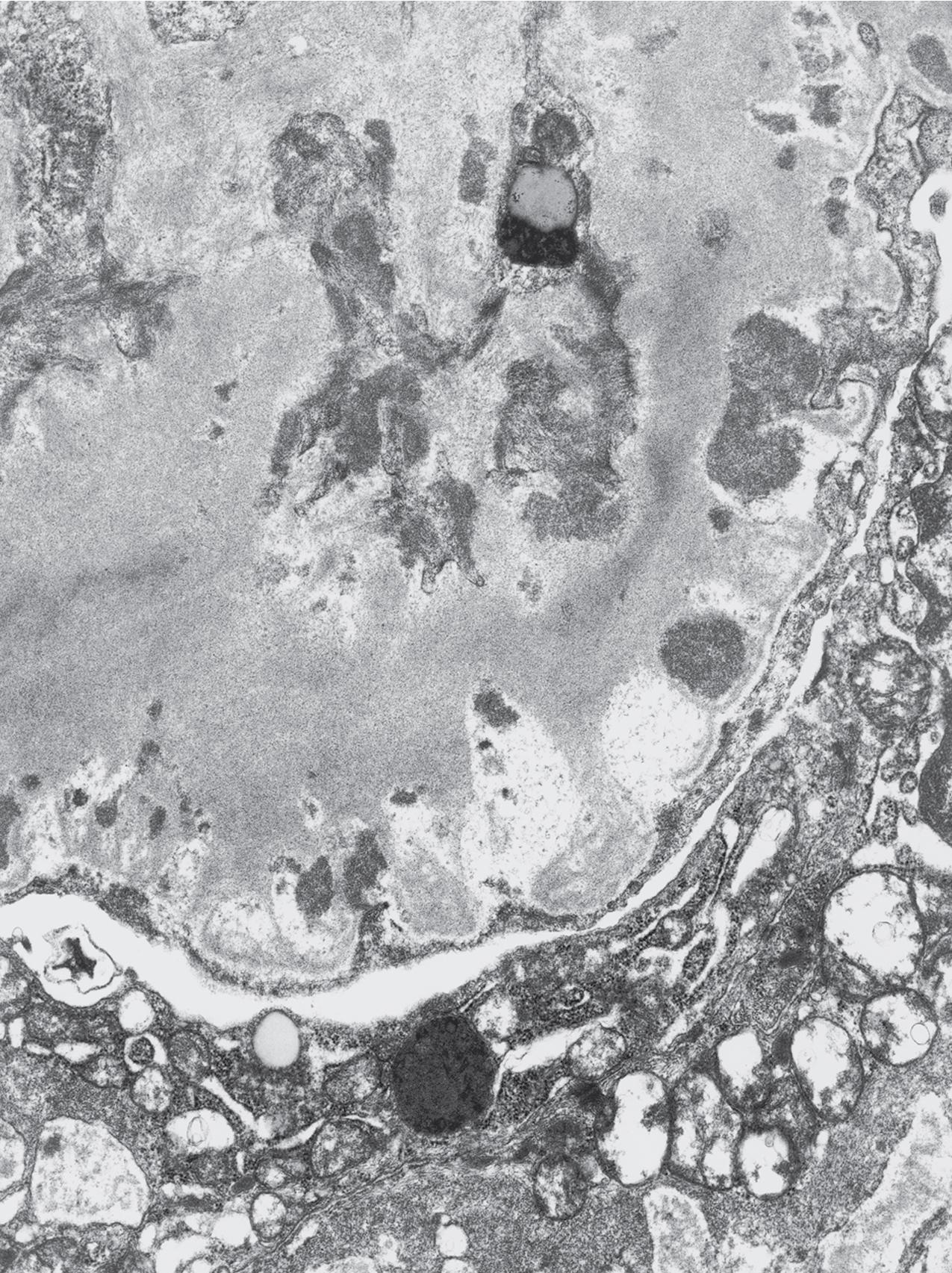
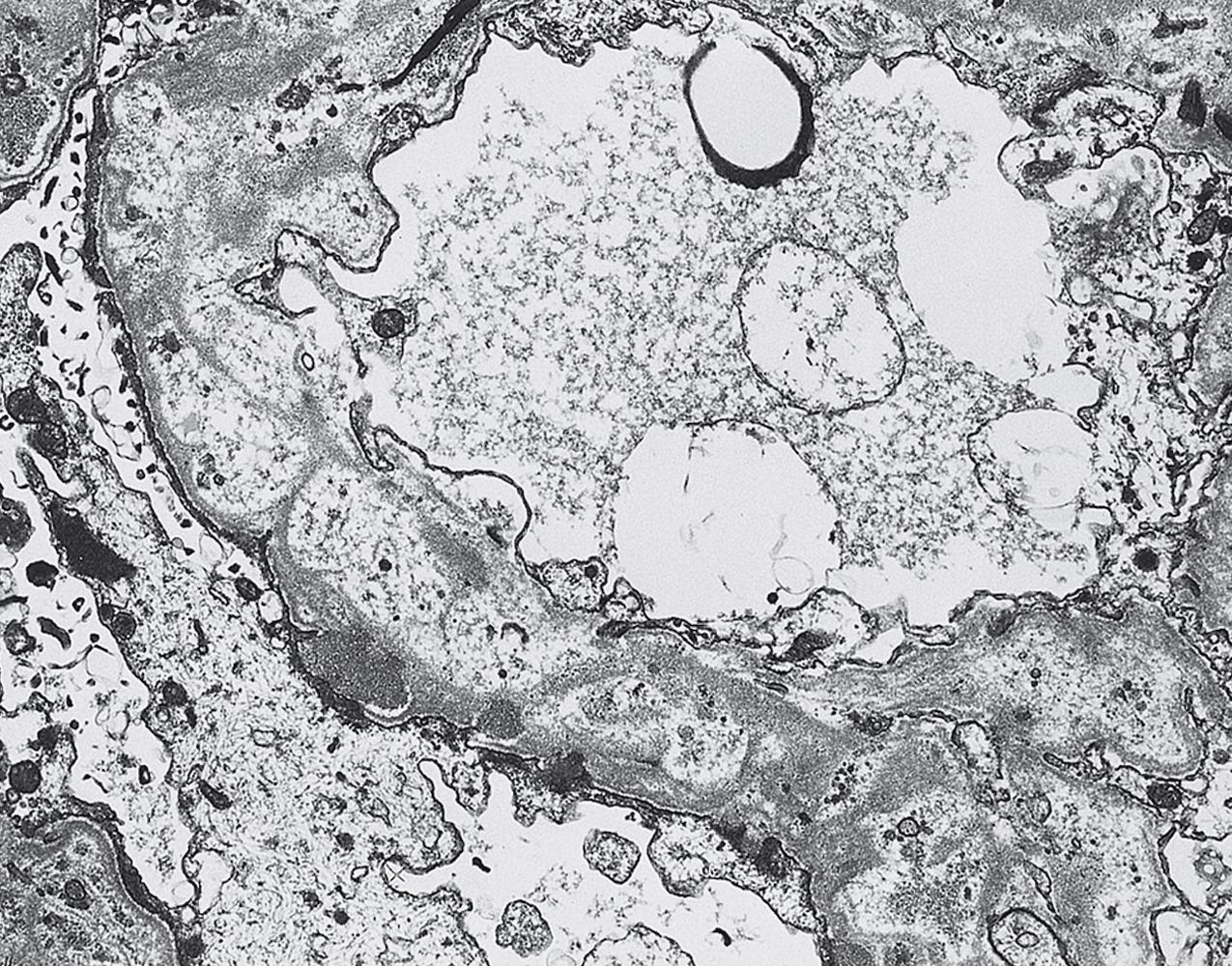
The podocytes show diffuse effacement of foot processes. When frequent mesangial deposits are present, the possibility of a secondary etiology of membranous nephropathy should be considered ( Figs. 3.91–3.92 ). If reticular aggregates are present in endothelial cell cytoplasm, the possibility of lupus-associated membranous nephropathy (ISN/RPS class V) should be considered.
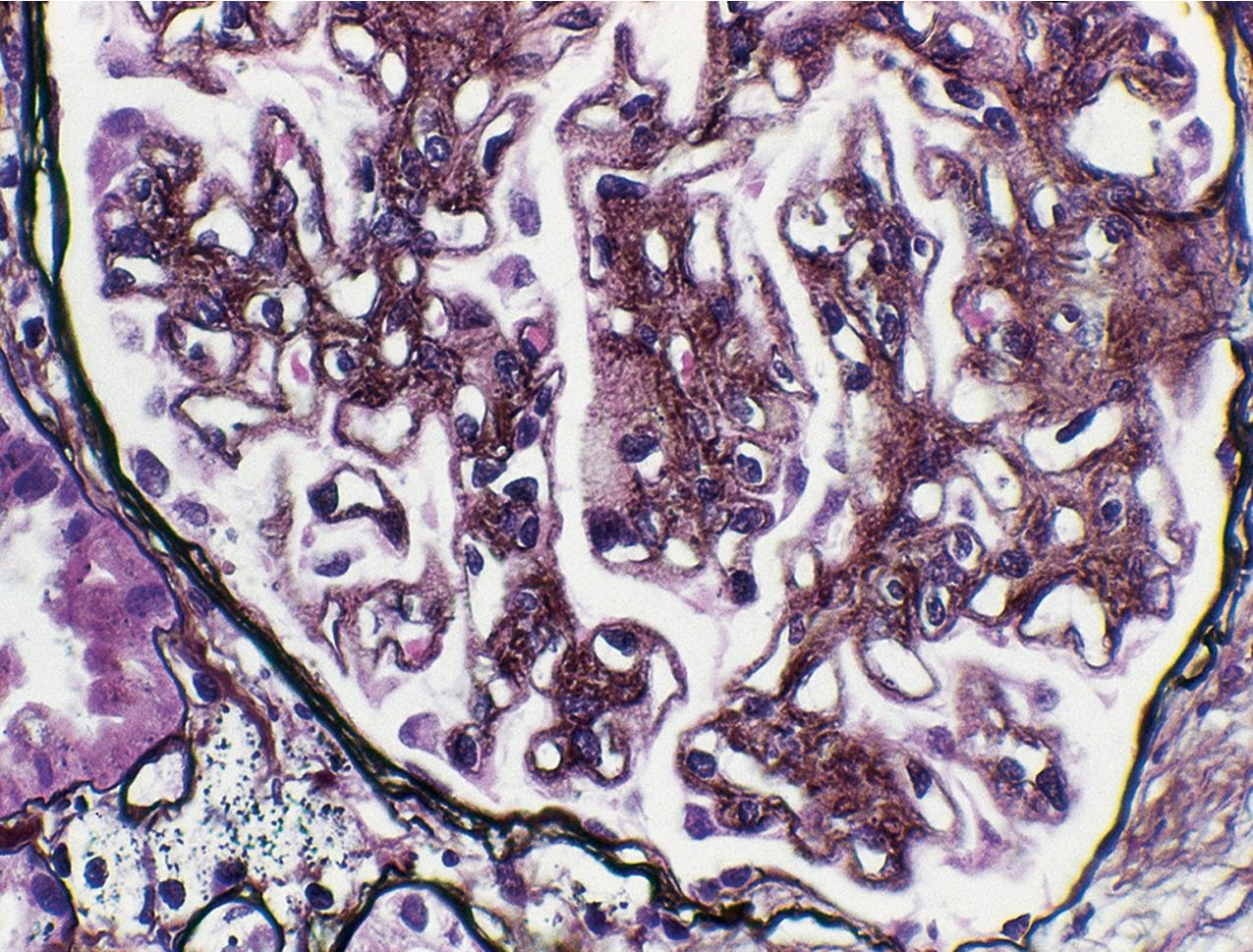
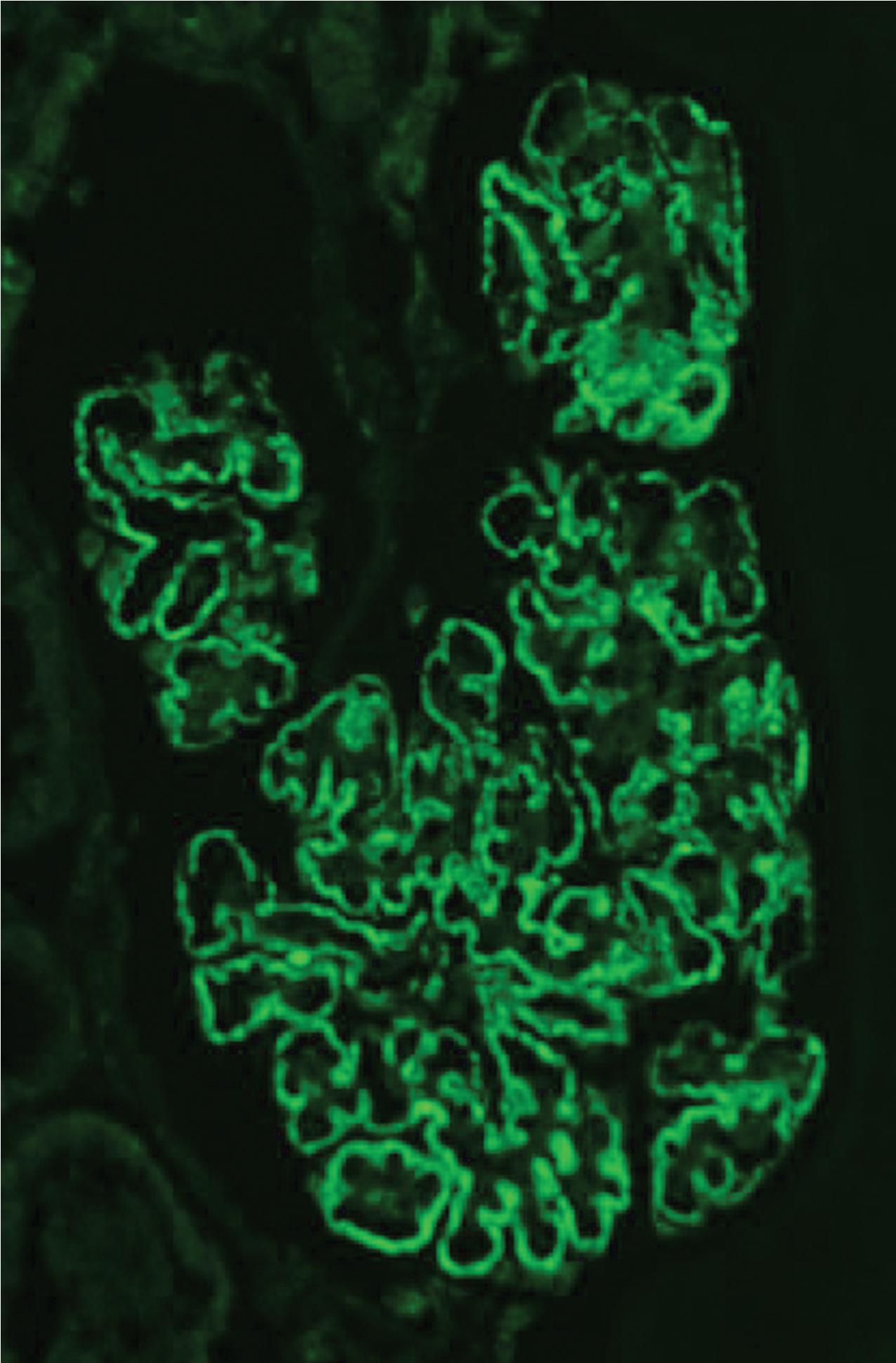
Most cases (around 70%) of previously termed “idiopathic” membranous nephropathy are caused by antibodies to the phospholipase A2 receptor (PLA2R), which is expressed on the podocyte. Circulating antibodies to PLA2R are predominantly IgG4, and subepithelial deposits are then thought to arise from autoantibody and local shedding of PLA2R, identified by IF (see Fig. 3.92 ) with in situ immune complex formation. HLA-DQA1 variants are associated with an increased risk for development of such autoantibodies. This colocalization is in contrast to secondary membranous nephropathy where other classes of IgG may be detected and PLA2R is not shed from the podocyte. Serum anti-PLA2R antibodies can be detected in about 70% of patients with membranous nephropathy that clinically appears “primary” (i.e., without evident cause), and their levels may correlate with disease activity. Decrease in serum anti-PLA2R antibody level in response to treatment typically precedes decrease in proteinuria. Of note, patients with membranous nephropathy may have anti-PLA2R staining of the deposits in the glomeruli in the kidney biopsy without detectable serum antibodies.
Ongoing research has identified novel antigens in patients with PLA2R negative membranous nephropathy, typically using mass spectrometry techniques and tissue staining. THSD7A is the antigen in a small proportion (1%–5%) of patients with membranous nephropathy. Some of these patients had cancer, with one patient with gallbladder carcinoma also showing THSD7A expression within the tumor. Neural epidermal growth factor-like 1 protein (NELL-1) is the antigen in about 5% to 10% of membranous nephropathy cases. These patients were older with IgG1 dominance. Interestingly, two different populations studied with NELL-1 positive membranous nephropathy showed variable association with cancer: none in the initial cohort but cancer was present in four of five in the validation cohort. Exostosin-1/2 is the antigen in a small subset of patients with signs of autoimmunity and also is present in some patients with diagnostic membranous LN (ISN/RPS class V) who may have better prognosis than exostosin-1/2 negative class V lupus nephritis patients. Of note, patients with membranous LN can rarely show PLA2R positivity in kidney biopsies. Semaphorin 3B was identified in the membranous deposits in a small group of PLA2R negative membranous nephropathy patients. These patients were mostly pediatric (8 of the 11), and as young as about 2 years. Additional tubular basement membrane IgG deposits were present in 4 of the 11 biopsies.
Secondary membranous nephropathy can be associated with numerous conditions including, for example, infectious agents, including bacterial, viral, or parasitic drugs or thyroid disease. Causality of the link has only been established for some, including hepatitis B, Hashimoto thyroiditis, SLE, syphilis, penicillamine, gold ( Fig. 3.93 ), mercuric chloride, and Sjögren syndrome. Patients with IgG4-related sclerosing disease, which primarily affects the tubulointerstitium, rarely can also have associated membranous nephropathy. Some patients have PLA2R-positive membranous nephropathy linked to sarcoidosis, NSAIDs, or hepatitis B. These findings indicate that exogenous injury or other antigens can cause formation of autoantibody to PLA2R. De novo membranous nephropathy in the kidney transplant, in contrast, is more likely because of antibody-mediated rejection and is largely PLA2R-negative. Cationic bovine serum albumin (from cow’s milk) was present in deposits of membranous nephropathy in some children. Malignancies, in particular some carcinomas, sarcomas, and leukemias, have been linked to membranous nephropathy, but definitive proof of causal linkage (i.e., circulating antibody–antigen immune complexes and tumor antigen within the deposits) is lacking in most instances. Interstitial fibrosis and segmental sclerosis are associated with worse prognosis.
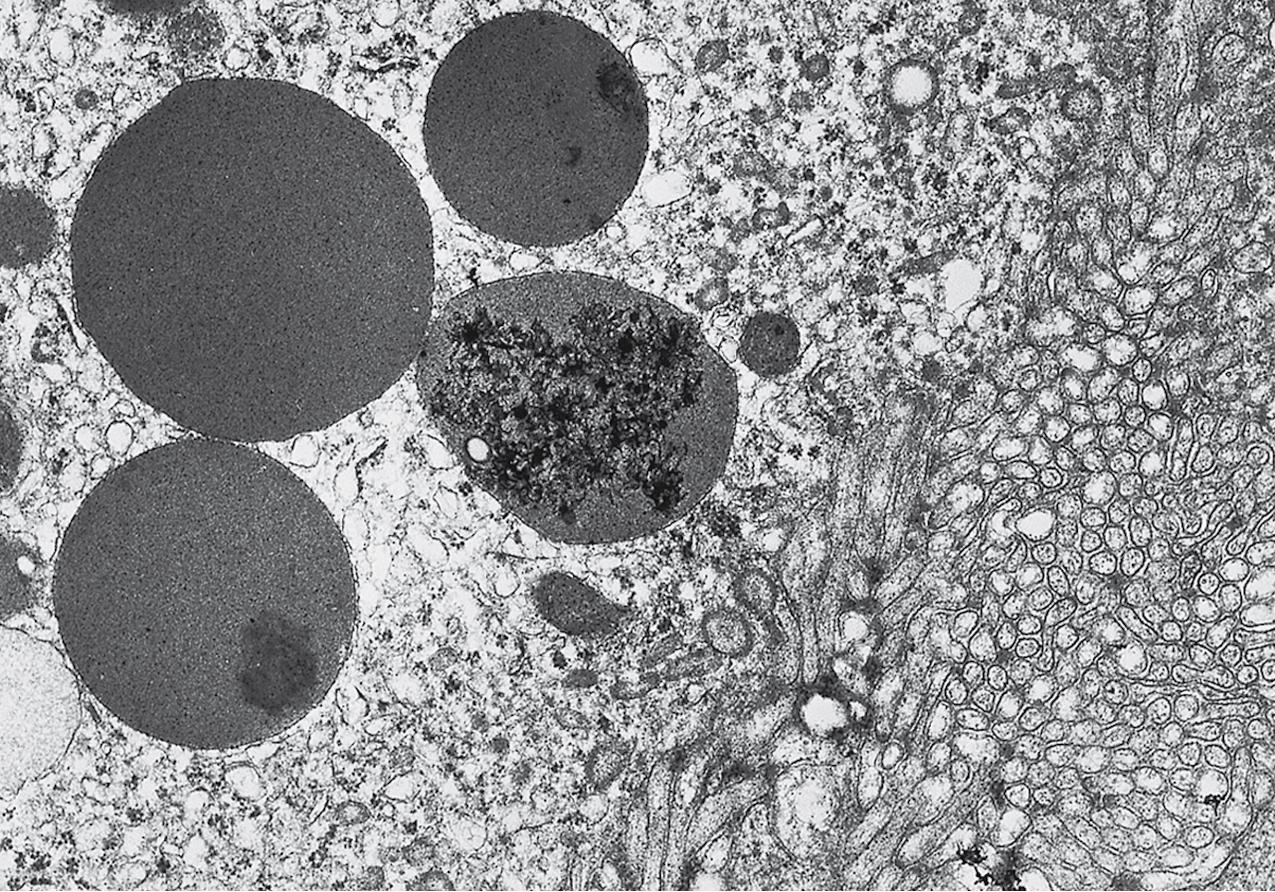
Extensive subepithelial deposits, evidenced on silver stains as “pinpoint holes” (i.e., areas of lucency because of the deposits not staining with silver) or spikes along the glomerular basement membranes by light microscopy
Granular capillary loop staining by immunofluorescence
Most cases of primary membranous nephropathy have phospholipase A2 receptor–positive deposits
Extensive subepithelial/intramembranous deposits by electron microscopy with overlying foot process effacement
A granular capillary loop pattern may occasionally be seen in fibrillary glomerulonephritis, which has typical fibrillary deposits by electron microscopy and smudgy appearance of deposits by immunofluorescence, in addition to mesangial deposits, and is DNAJB9 positive
Mesangial deposits are typically present in secondary membranous nephropathy
Mesangial deposits and the additional presence of reticular aggregates and/or full-house staining suggest membranous lupus nephritis
Ahmad SB, Appel GB. Antigens, antibodies, and membranous nephropathy: A decade of progress. Kidney Int. 2020;97:29–31.
Beck Jr LH, Bonegio RG, Lambeau G, et al. M-type phospholipase A2 receptor as target antigen in idiopathic membranous nephropathy. N Engl J Med. 2009;361:11–21.
Couser WG, Baker PJ, Adler S. Complement and the direct mediation of immune glomerular injury: A new perspective. Kidney Int. 1985;28:879–890.
Debiec H, Lefeu F, Kemper MJ, et al. Early-childhood membranous nephropathy due to cationic bovine serum albumin. N Engl J Med. 2011;364:2101–2110.
Dumoulin A, Hill GS, Montseny JJ, et al. Clinical and morphological prognostic factors in membranous nephropathy: Significance of focal segmental glomerulosclerosis. Am J Kidney Dis. 2003;41:38–48.
Ehrenreich T, Churg J. Pathology of membranous nephropathy. In: Sommers SC, ed. Pathology Annual, 3. New York: Appleton-Century-Crofts; 1968:145–154.
Fogo AB. Milk and membranous nephropathy. N Engl J Med. 2011;364:2158–2159.
Gonzalo A, Mampaso F, Barcena R, et al. Membranous nephropathy associated with hepatitis B virus infection: long-term clinical and histological outcome. Nephrol Dial Transplant. 1999;14:416–418.
Jennette JC, Iskandar SS, Dalldorf FG. Pathologic differentiation between lupus and nonlupus membranous glomerulopathy. Kidney Int. 1983;24:377–385.
Kerjaschki D. The pathogenesis of membranous glomerulonephritis: From morphology to molecules. Virchows Arch B Cell Pathol Incl Mol Pathol. 1990;58:253–271.
Sethi S, Debiec H, Madden B, et al. Semaphorin 3B-associated membranous nephropathy is a distinct type of disease predominantly present in pediatric patients. Kidney Int. 2020; 98:1253–1264.
Sethi S, Madden BJ, Debiec H, et al. Exostosin 1/exostosin 2-associated membranous nephropathy. J Am Soc Nephrol. 2019;30:1123–1136.
Tomas NM, Beck Jr LH, Meyer-Schwesinger C, et al. Thrombospondin type-1 domain-containing 7A in idiopathic membranous nephropathy. N Engl J Med. 2014;71:2277–2287.
Toth T, Takebayashi S. Idiopathic membranous glomerulonephritis: A clinicopathologic and quantitative morphometric study. Clin Nephrol. 1992;38:14–19.
Van Damme B, Tardanico R, Vanrenterghem Y, et al. Adhesions, focal sclerosis, protein crescents, and capsular lesions in membranous nephropathy. J Pathol. 1990;161:47–56.
Wakai S, Magil AB. Focal glomerulosclerosis in idiopathic membranous glomerulonephritis. Kidney Int. 1992;41:428–434.
Yoshimoto K, Yokoyama H, Wada T, et al. Pathologic findings of initial biopsies reflect the outcomes of membranous nephropathy. Kidney Int. 2004;65:148–153.
MPGN is a pattern of injury with a variety of causes. These include, for example, subendothelial and mesangial immune complex deposits, complement deposits, and some types of monoclonal protein deposits. MPGN pattern of injury may be seen in, for example, class IV diffuse LN, cryoglobulinemic GN, proliferative GN with monoclonal deposits, fibrillary GN, some cases of IgAN and C3 glomerulopathies, all with distinct IF and/or EM appearances. GBM double contours may also be seen in other nonimmune complex injuries, characterized by chronic endothelial injury with interposition of cells by EM. These include the organizing phase of thrombotic microangiopathy, radiation nephritis, chronic transplant glomerulopathy, and sickle cell disease. Although light microscopy in these cases may appear similar to other causes of the MPGN pattern previously enumerated, IF findings and EM readily allow for the recognition of the MPGN lesion related to specific deposits versus chronic endothelial injury. Rare diseases with MPGN pattern without deposits by standard IF are recognized by specific immunohistochemistry staining and/or distinct EM appearance (e.g., collagen type III glomerulopathy, fibronectin glomerulopathy).
All other causes of MPGN pattern injury beyond those with specific etiology identified as previously outlined are then called “idiopathic.” Idiopathic MPGN because of deposits is more common in children and young adults, whereas MPGN-type lesions are more commonly secondary to chronic infections in adults. MPGN because of deposits typically presents as combined nephritic/nephrotic syndrome with hypocomplementemia with decreased C3. The incidence of MPGN because of deposits appears to have decreased in children in the past decades, for unknown reasons. Children with MPGN pattern because of immune complex deposits tend to be older than children with DDD, which shows only C3 deposits (previously called MPGN type II see “Dense Deposit Disease”). The presence of the C3 nephritic factor (C3NeF) is rarer, and concurrent partial lipodystrophy is very rare in immune complex-type MPGN, compared with their relative frequency in DDD and C3GN. Patients with idiopathic MPGN often have progressive renal disease, with about 50% renal survival at 10 years. Clinical indicators of poor prognosis are hypertension, impaired renal function, and nephrotic syndrome. MPGN pattern disease because of immune complexes recurs in the transplant in about one-third of patients and may lead to graft loss, particularly if crescents are present. MPGN pattern lesions because of deposits can also occur de novo in the transplant, most often related to hepatitis C infection and cryoglobulinemia (see “Cryoglobulinemic Glomerulonephritis”).
MPGN pattern because of deposits (also called mesangiocapillary GN ) was previously divided into three types, all with similar light microscopic appearance. The entities previously called MPGN type II and III are now recognized to be C3 glomerulopathies (see “C3 Glomerulopathy”), with underlying complement dysregulation, and paucity or absence of true immune complexes (i.e., little or no Ig in deposits). The MPGN lesion is characterized by global and diffuse mesangial and endocapillary hypercellularity contributed to, in part, by proliferation of glomerular cells, with influx of inflammatory cells, with increased mesangial matrix and double contours of the GBM on silver stain (the so-called “tram-track” appearance; Fig. 3.94 ). The glomerular tuft often shows a lobular simplification ( Figs. 3.95–3.99 ). Increased mononuclear cells and occasional neutrophils may be present. The hypercellularity is typically uniform and diffuse in idiopathic MPGN because of deposits, contrasting the irregular involvement with proliferative LN. In some cases, the glomeruli may appear more solid and nodular ( Fig. 3.100 ). The capillary wall is thickened with a double contour by silver stains ( Fig. 3.101 ; see also Fig. 3.99 ). This appearance results from the presence of subendothelial deposits and so-called “circumferential cellular interposition,” whereby infiltrating mononuclear cells, occasional mesangial cells, or even portions of endothelial cells interpose themselves between the endothelium and the basement membrane, with new, inner basement membrane being laid down. A circumferential, or partial, double-contour basement membrane results. In secondary forms of MPGN, the injury may be more irregular. Crescents may occur in both idiopathic and secondary forms (see Fig. 3.100 ). Prognosis is worse with greater than 20% crescents. Lesions progress and over time show less cellularity and more pronounced matrix accumulation and sclerosis. Tubulointerstitial fibrosis and vascular sclerosis proportional to glomerular scarring are seen late in the course. More extensive tubular atrophy and interstitial fibrosis indicate worse prognosis.
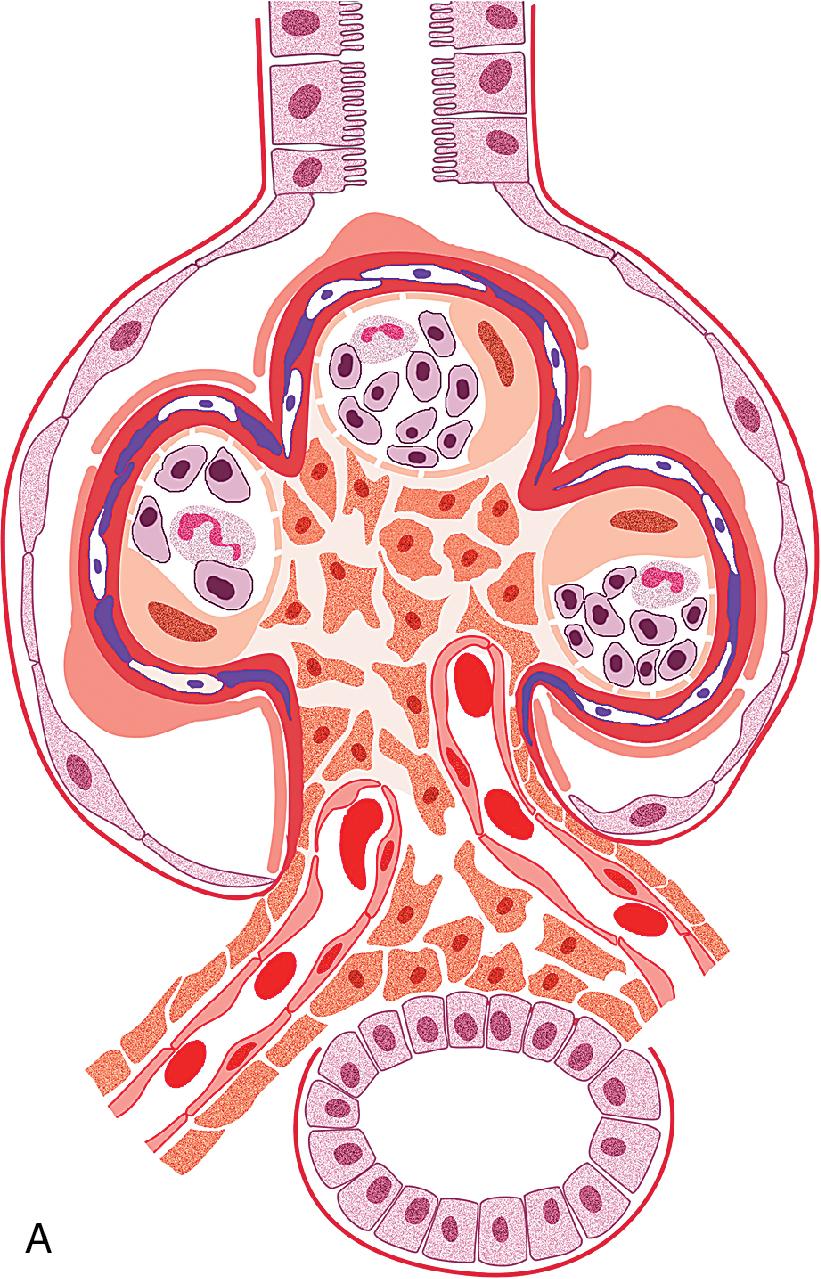
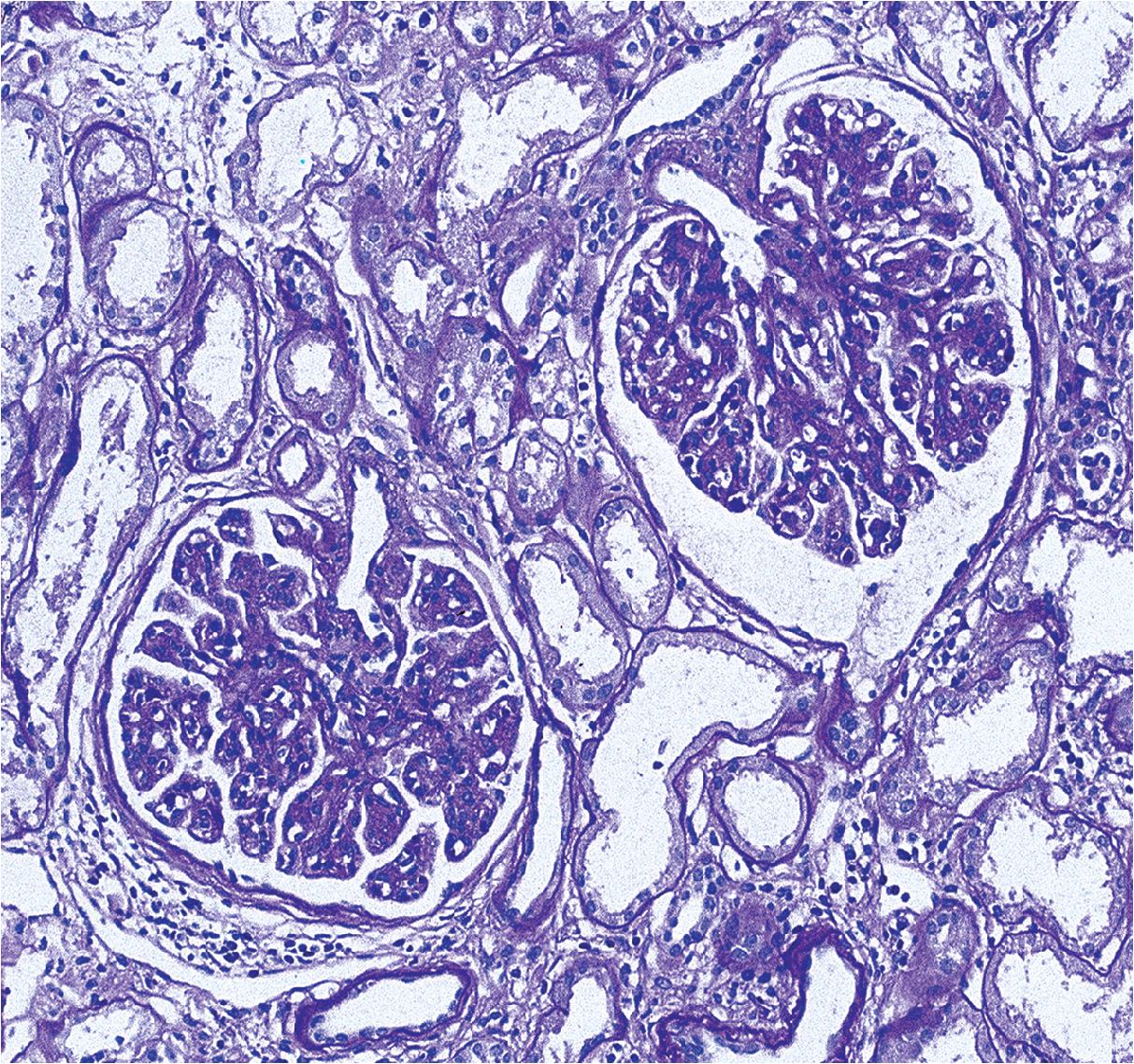
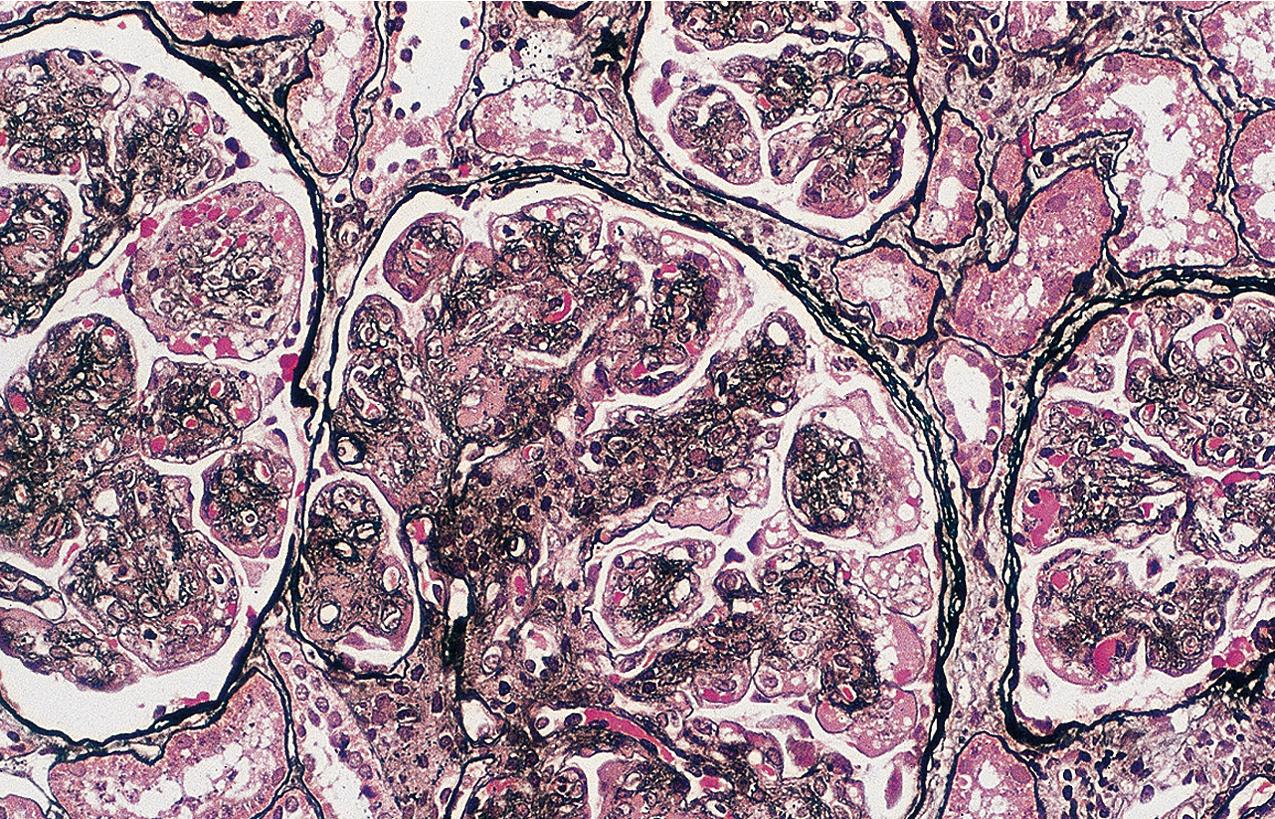
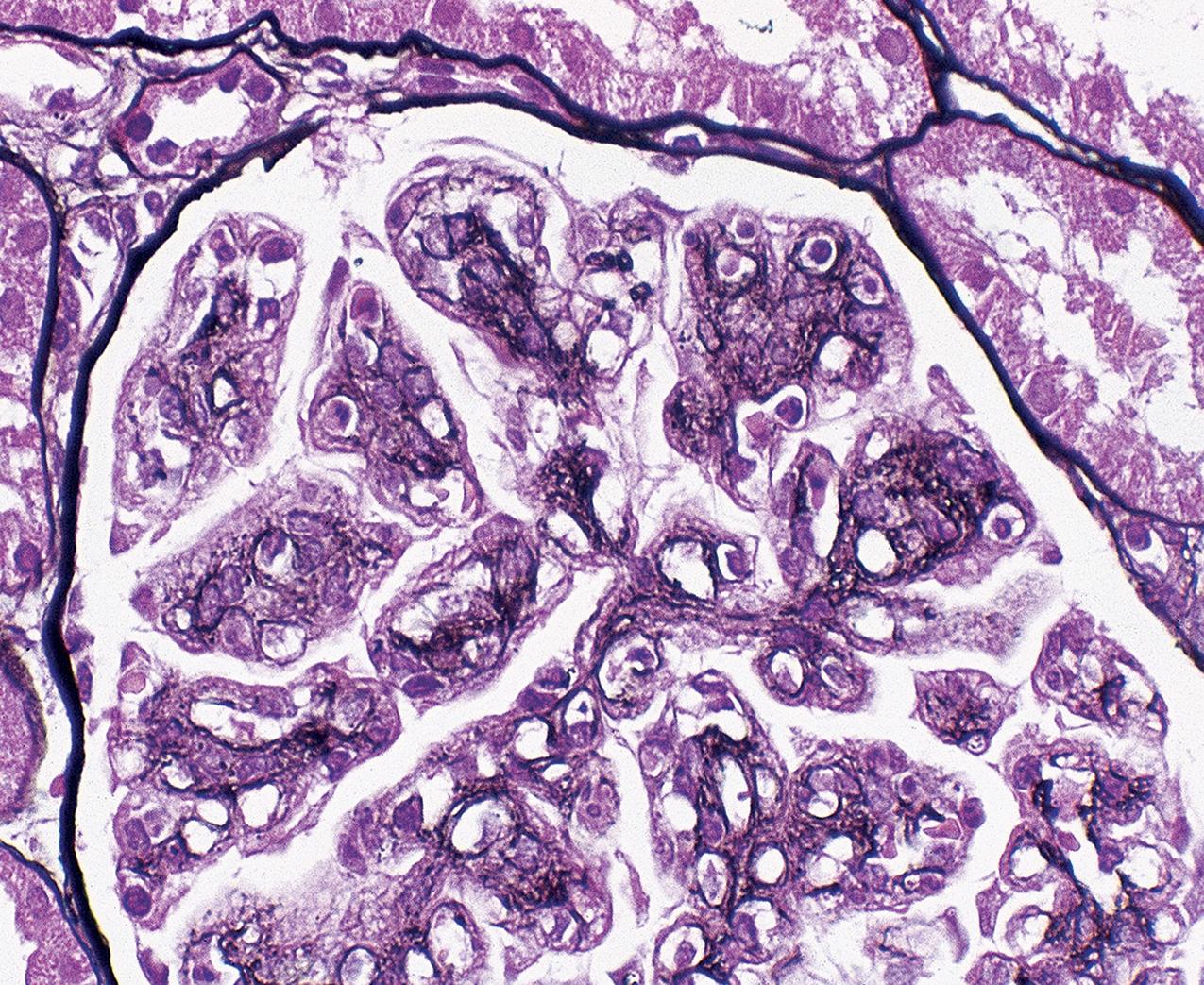
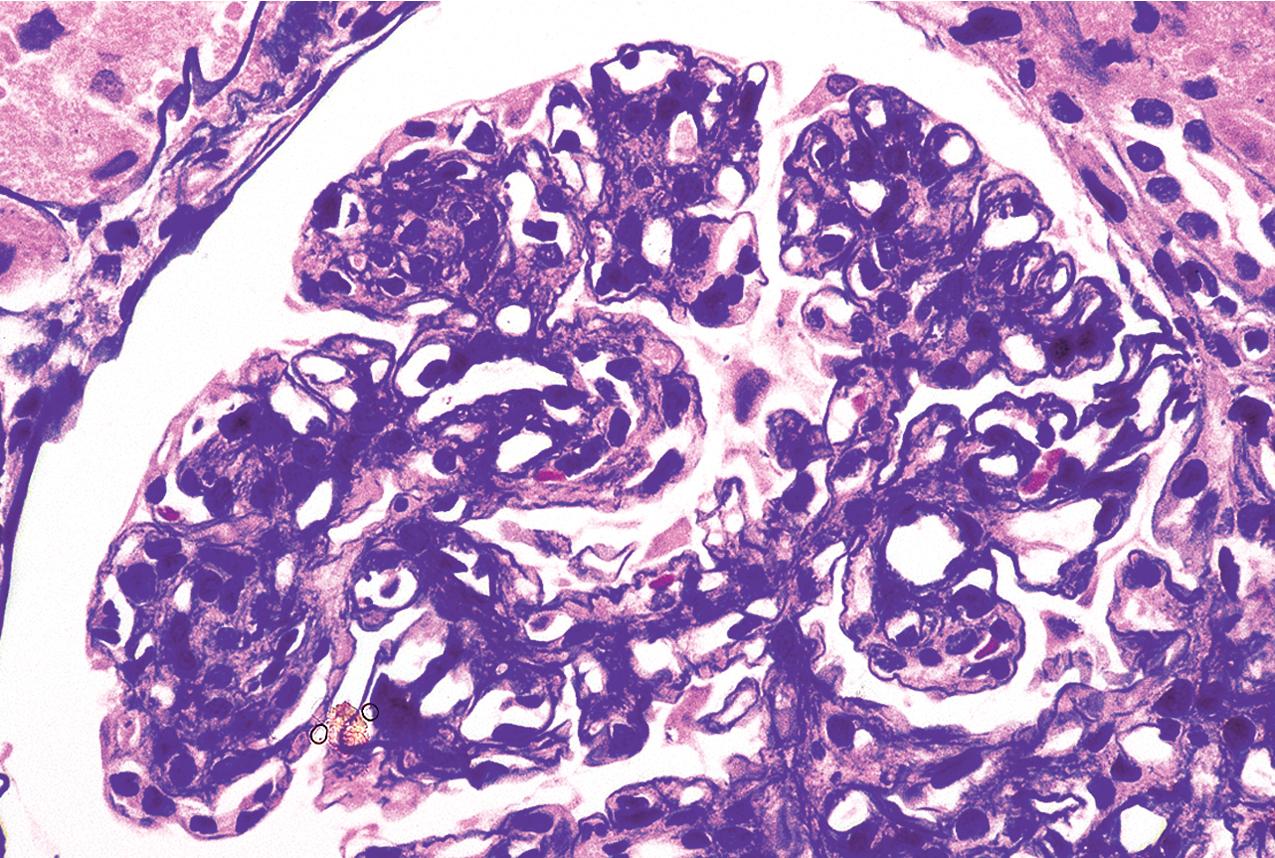
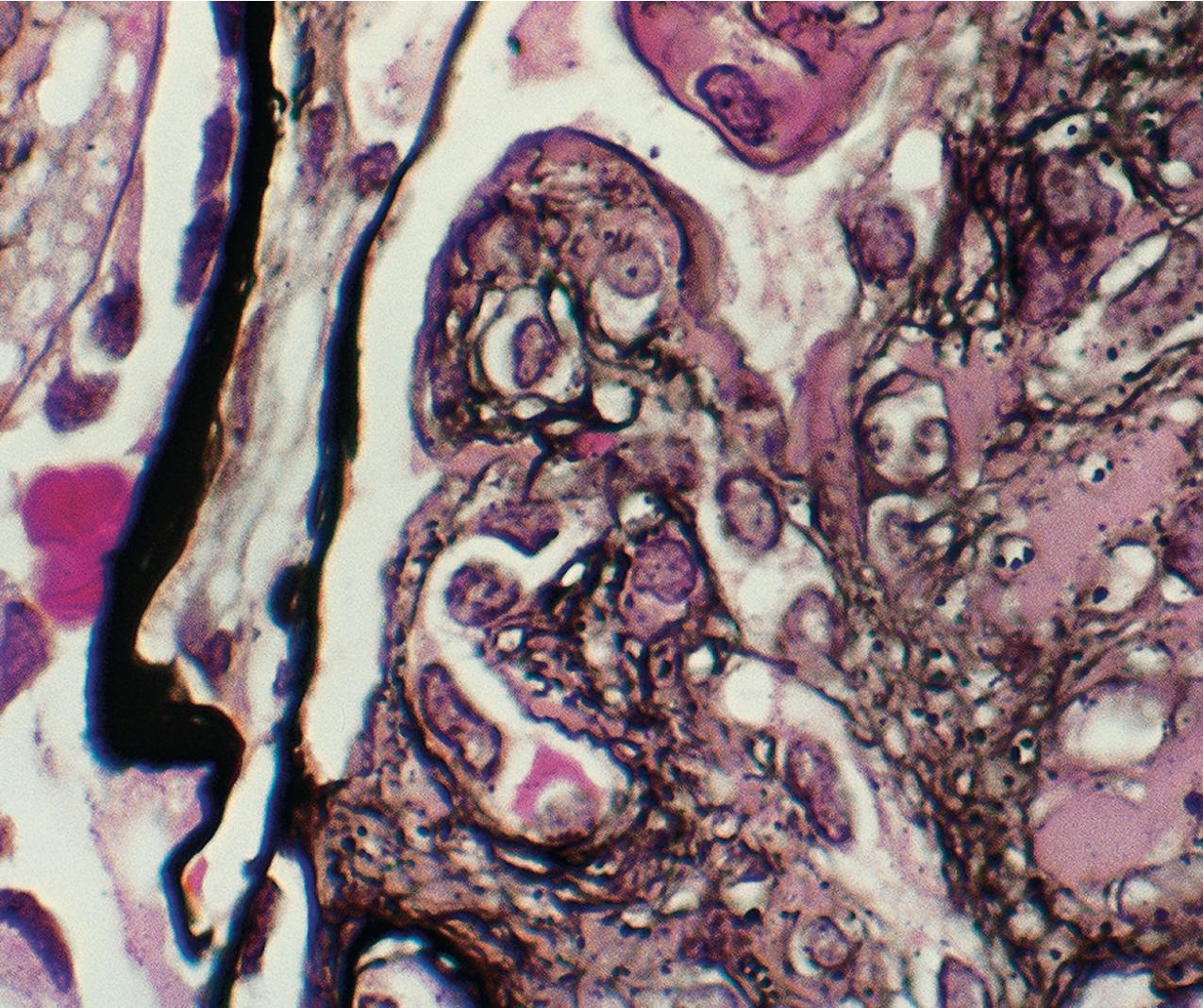
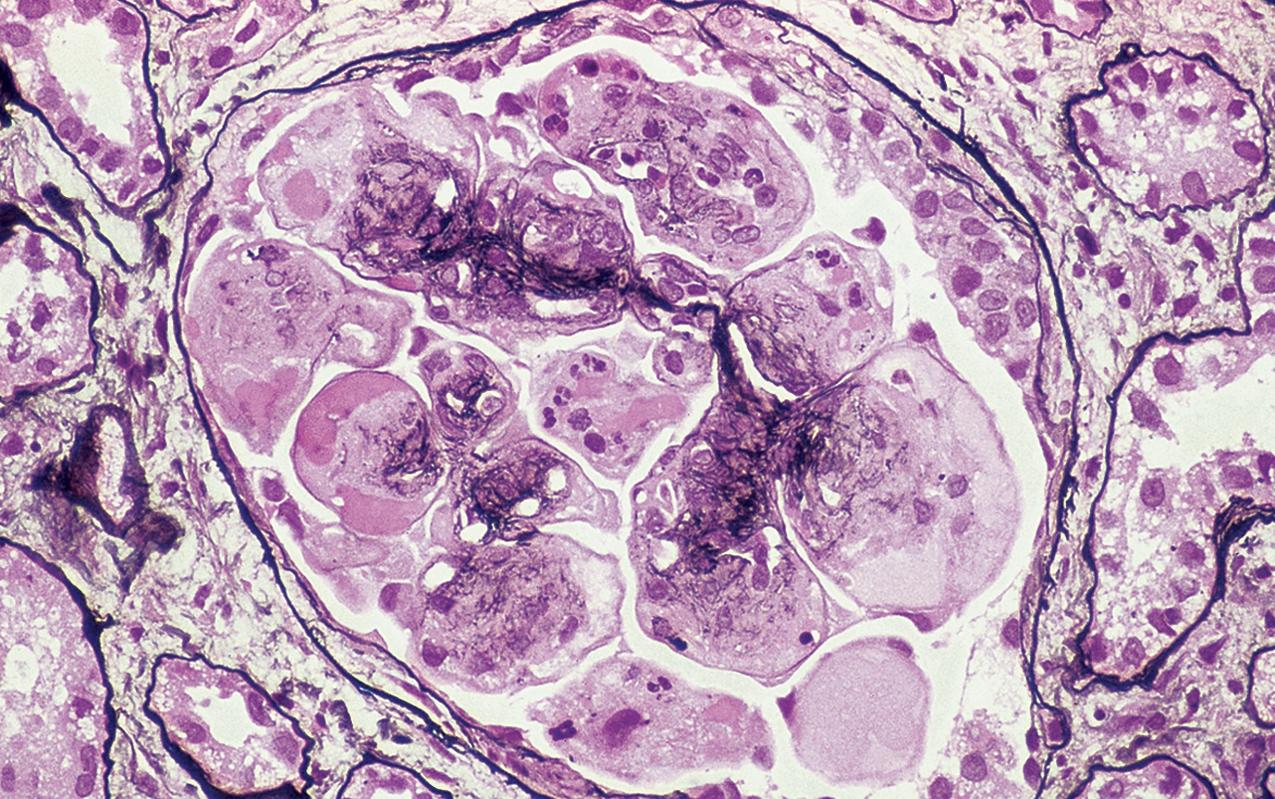
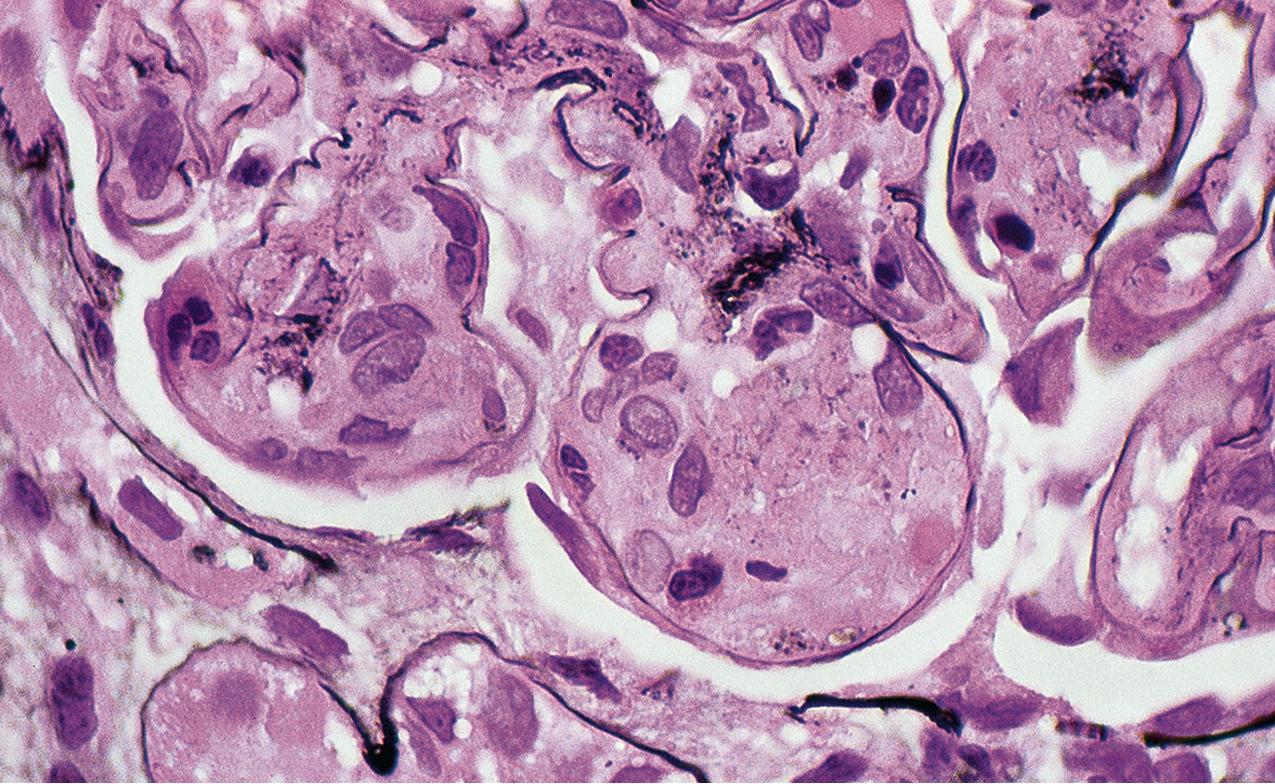
The IF findings are variable in MPGN due to deposits. Typically, IgG and IgM and C3 are present in an irregular, chunky capillary and mesangial distribution ( Figs. 3.102–3.105 ). IgA is present in only a small proportion of cases. C3 staining may be dominant, with minor Ig components, which suggests possible C3GN as the underlying cause of the lesions. The peripheral loop deposits visualized by IF typically are sausage-shaped and have a smooth outer edge because they are subendothelial and molded under the GBM (see Fig. 3.105 ).
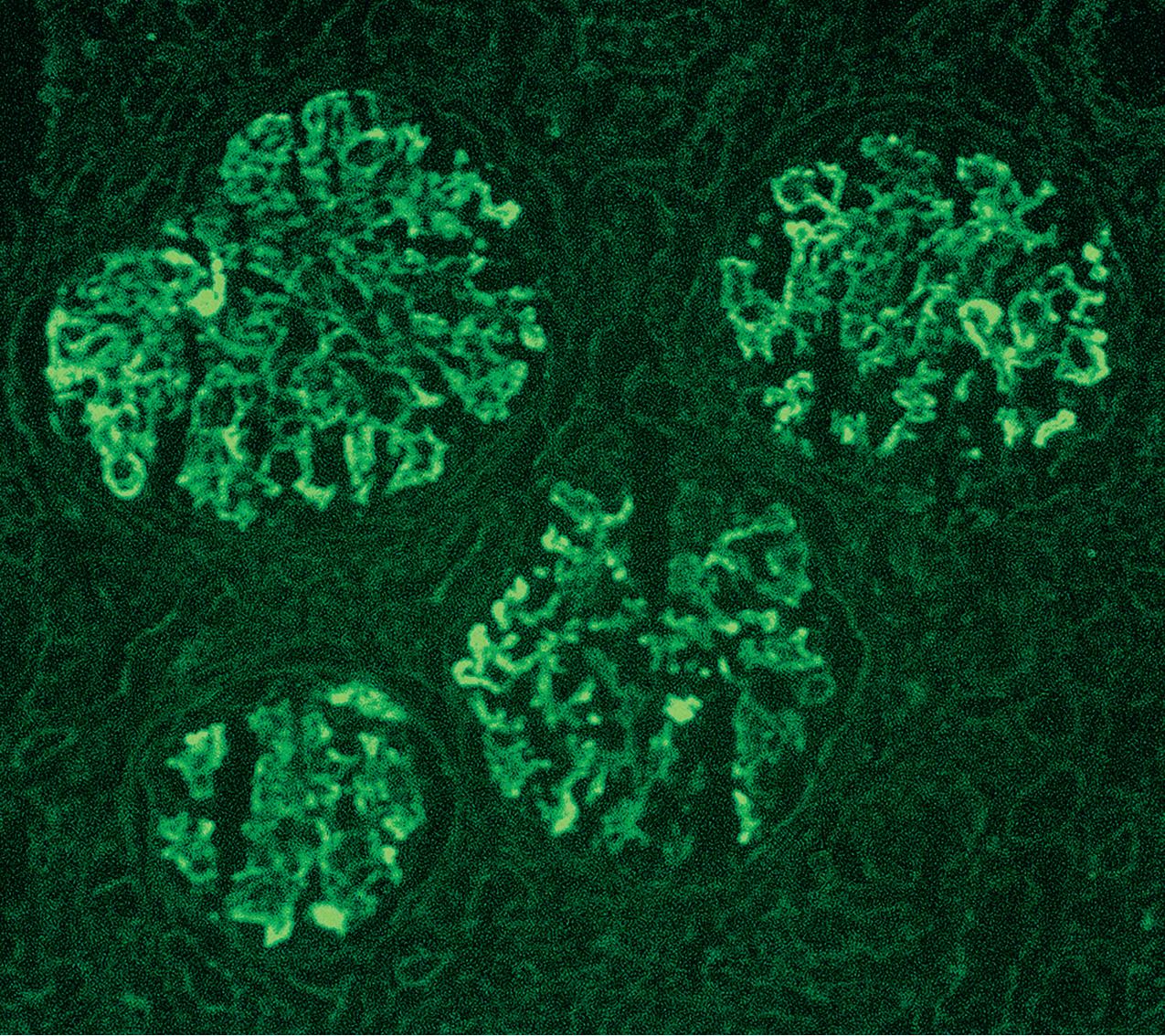
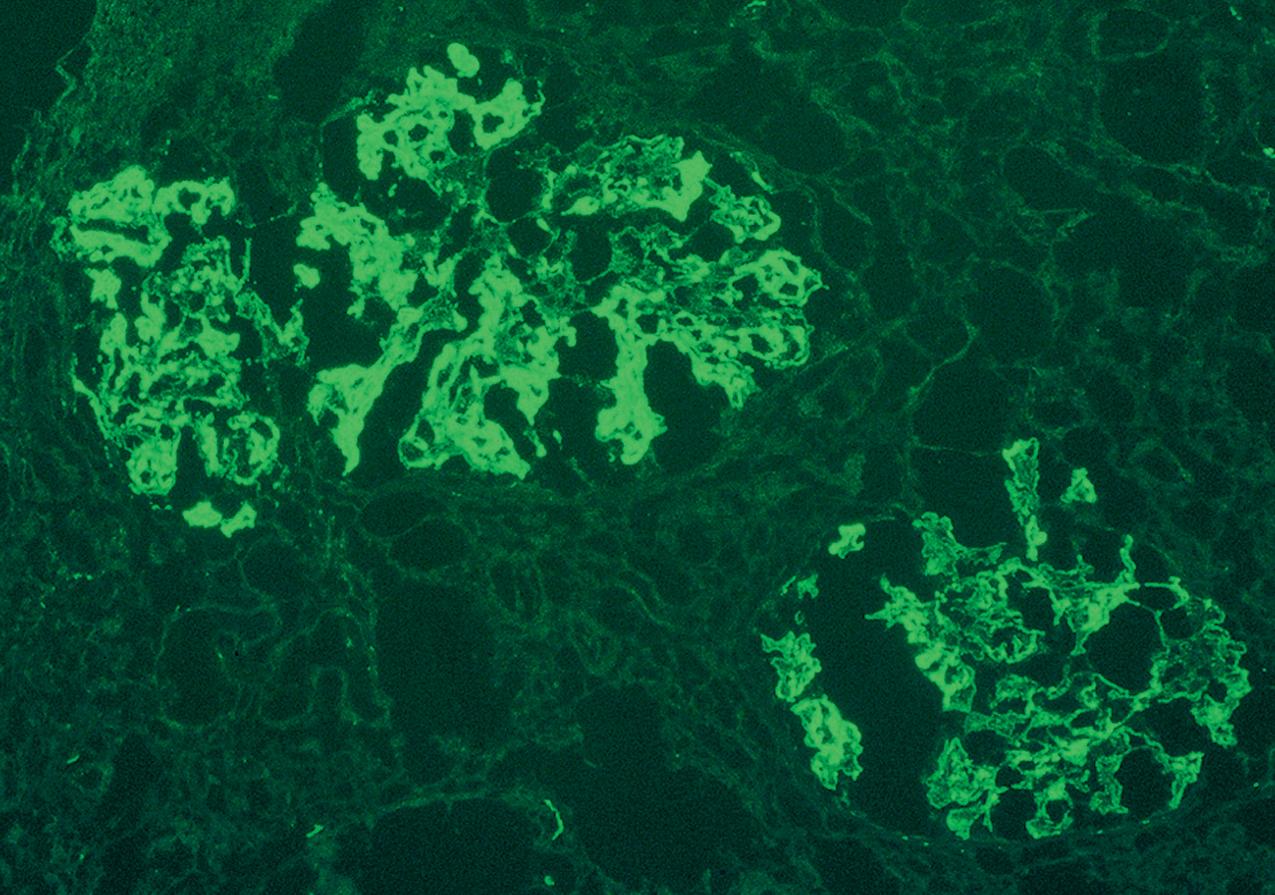
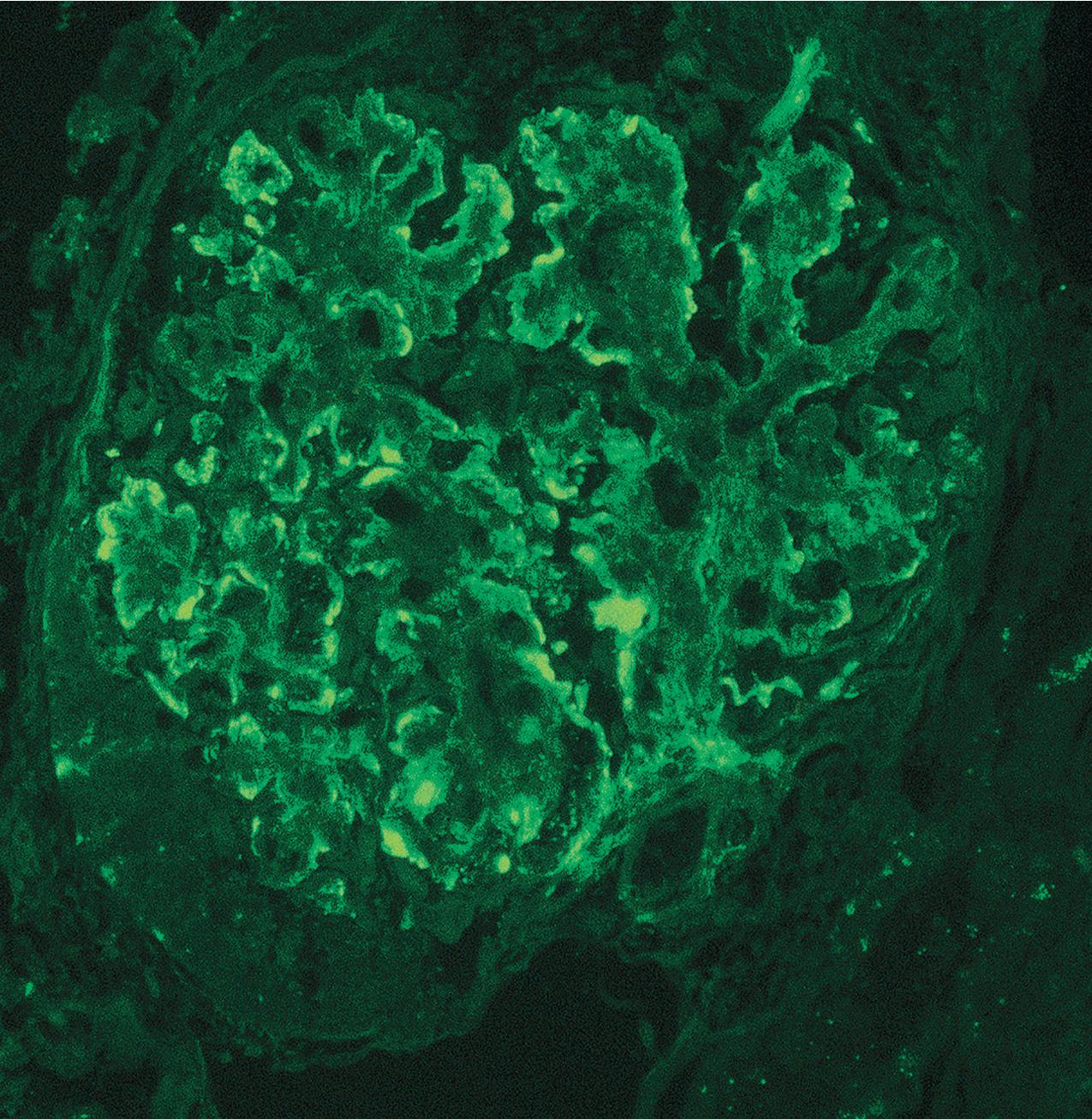
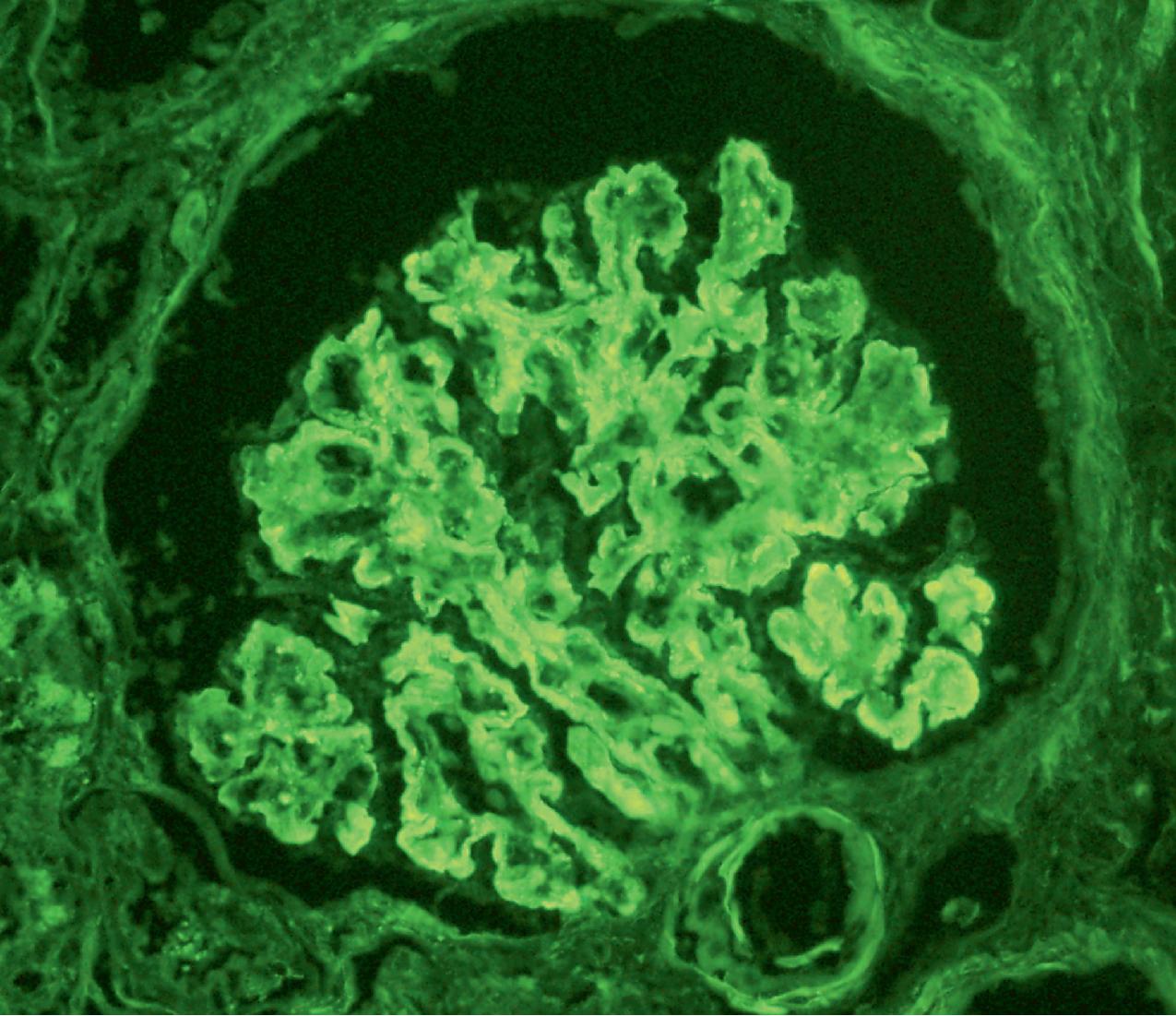
By EM, this type of MPGN shows numerous deposits in subendothelial and mesangial areas ( Figs. 3.106–3.109 ). The “subendothelial” deposits actually more commonly lie within the GBM, immediately under the original lamina densa, with new basement membrane underneath (see Fig. 3.109 ). Vague wormy or microtubular substructure suggests a possible cryoglobulin component (see Fig. 3.108 ). Cellular interposition is present. This term refers to the interposition of cytoplasmic processes of mesangial or mononuclear cells between the endothelial cell and the basement membrane ( Fig. 3.110 ). Monocyte interposition is particularly common when the MPGN lesion is related to cryoglobulinemia. New glomerular basement material is present immediately under the swollen endothelial cells, resulting in the double contours of the basement membrane seen on silver stain. The overlying podocyte foot processes are effaced.
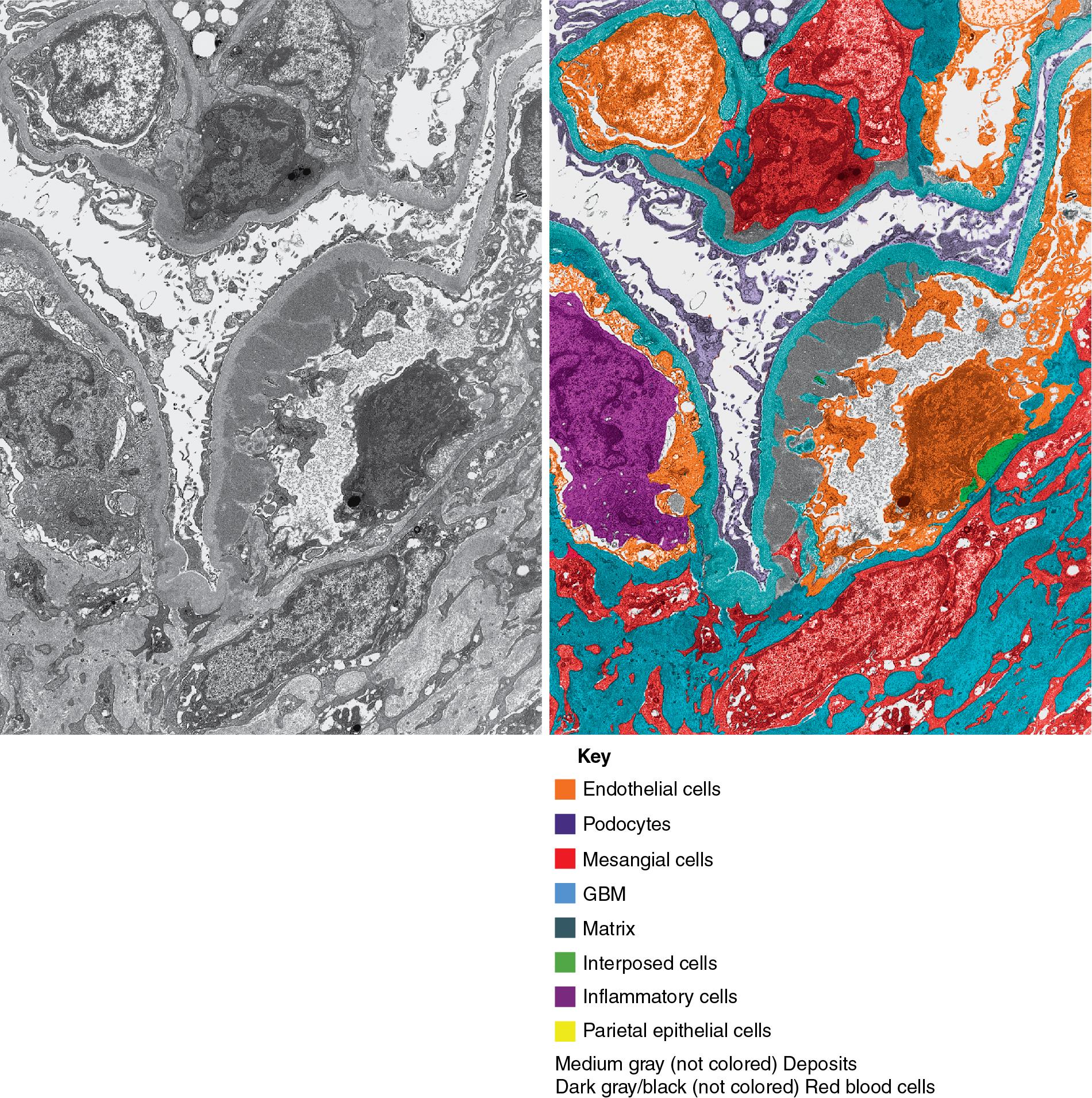
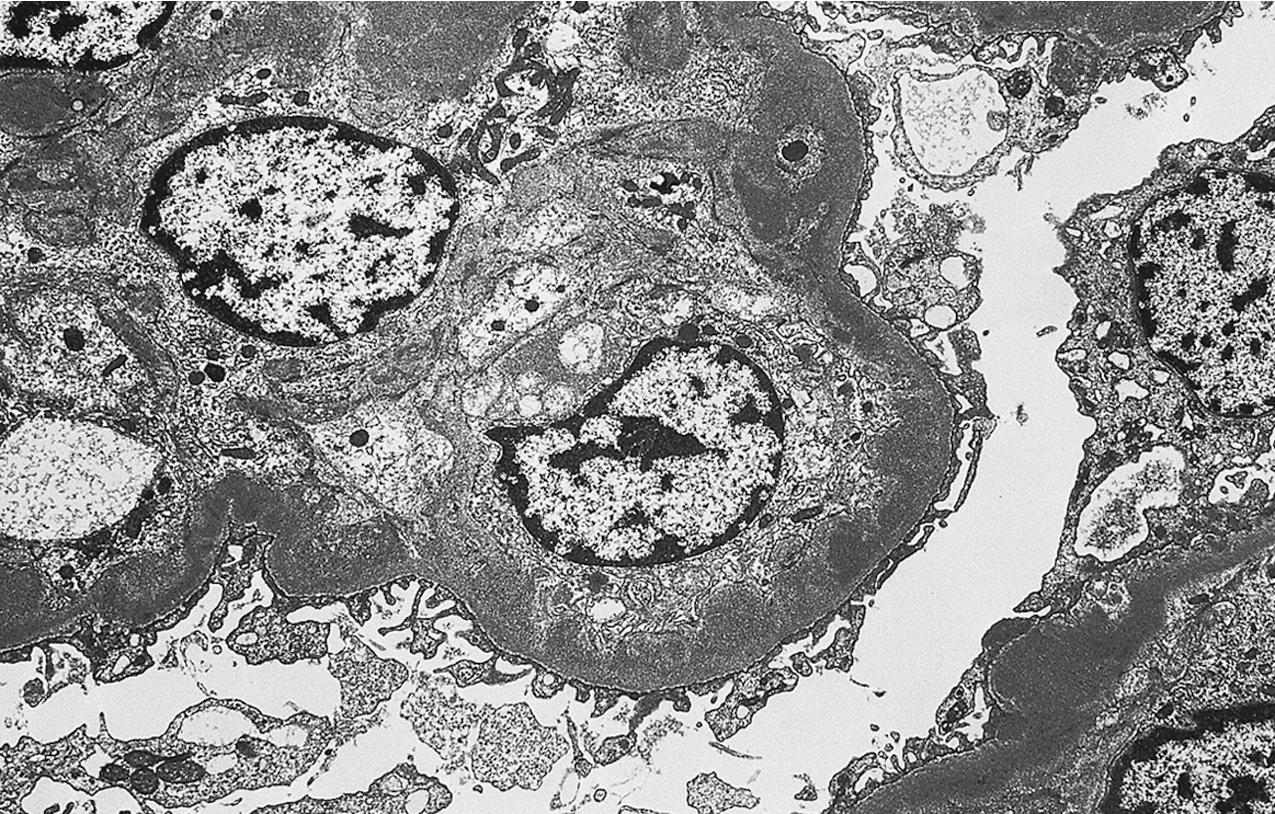
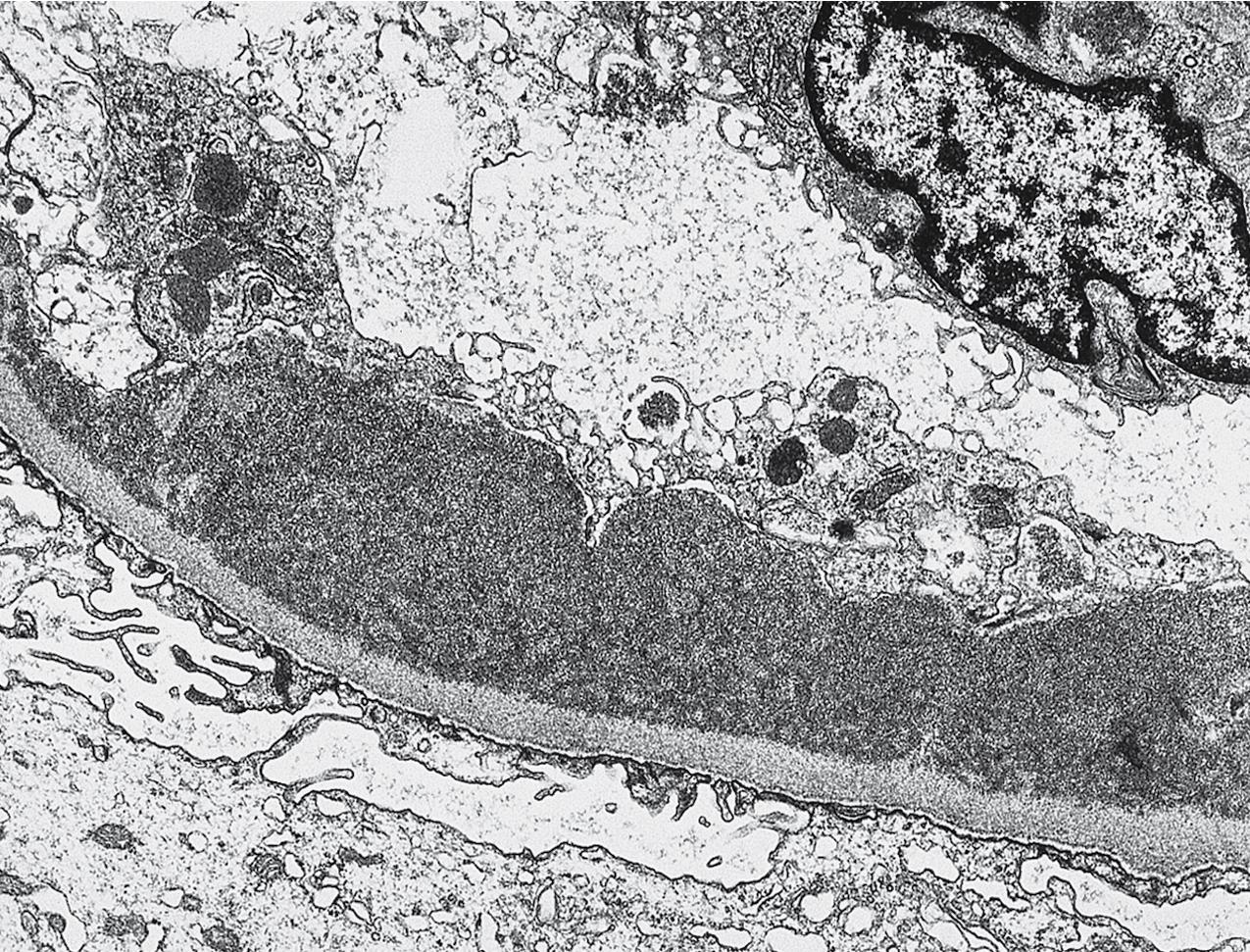
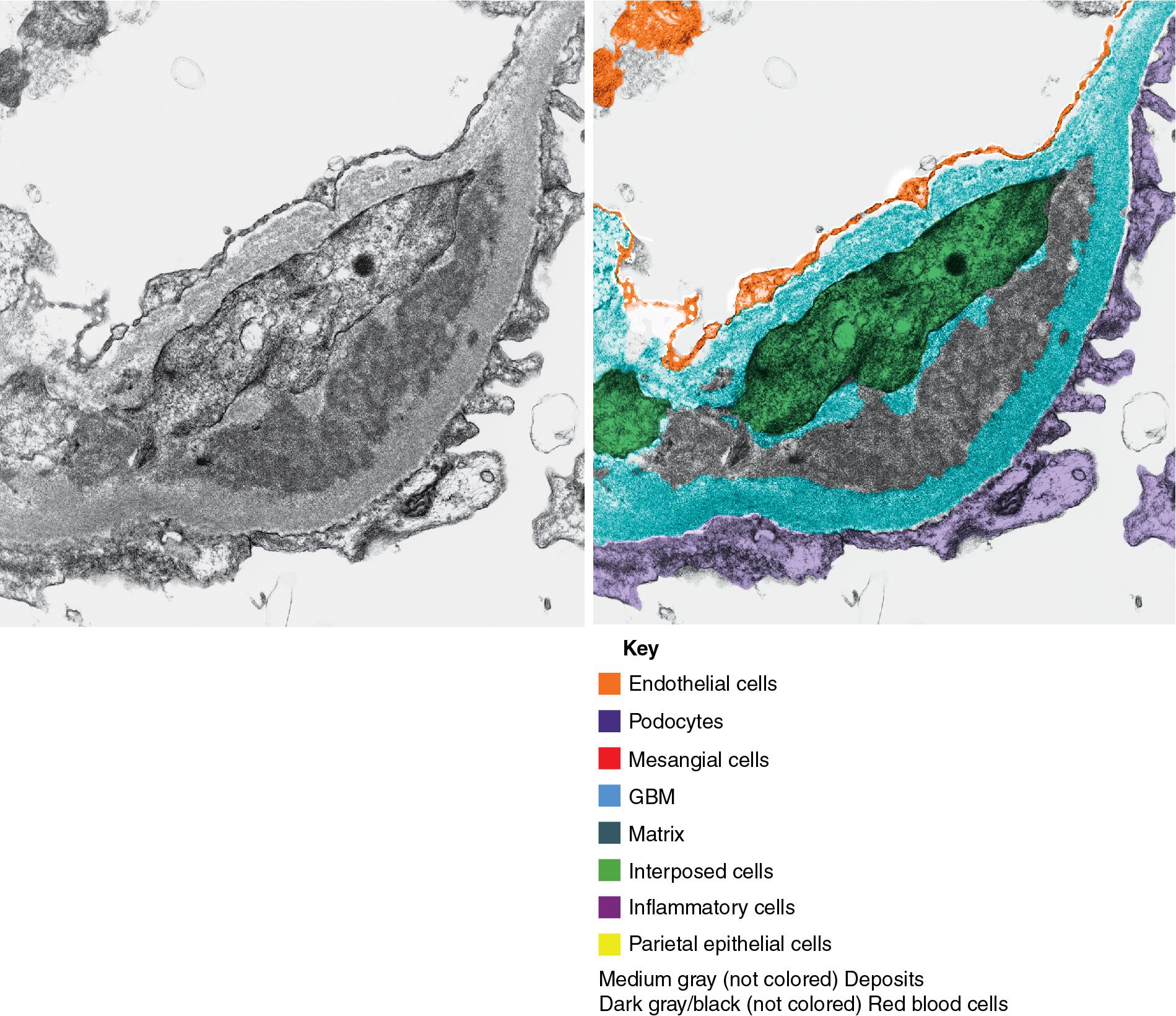
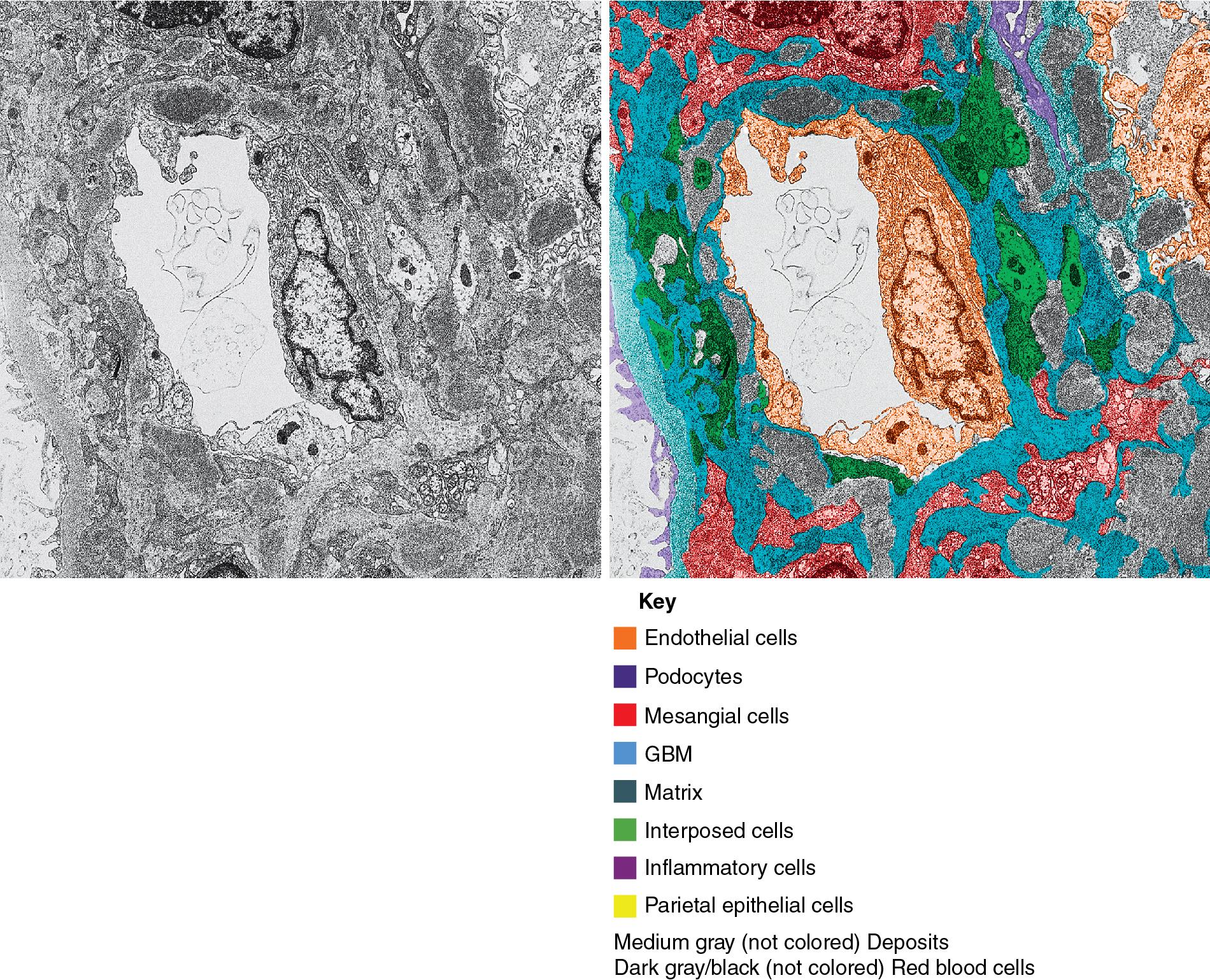
Of note, in nonimmune complex diseases with GBM double contours seen by light microscopy (e.g., transplant glomerulopathy, chronic thrombotic microangiopathy; see “Thrombotic Microangiopathy” in Chapter 4 ), EM shows that the double contour results from widening of the GBM as a result of increased lucency of the lamina rara interna and cellular interposition without immune complexes, with new basement membrane formed underneath the endothelium.
MPGN pattern lesions can occur secondary to a number of chronic infectious processes, including hepatitis B, hepatitis C, subacute bacterial endocarditis, cryoglobulin, syphilis, and so on, which are discussed separately. Morphologic features do not allow for precise classification of the underlying agent in many cases of MPGN pattern injury. MPGN-type lesions may occur as a result of inherited or acquired dysregulation of complement (see earlier, “C3 Glomerulopathies”), sometimes with partial lipodystrophy. In adults in the United States, many patients with MPGN have associated hepatitis C infection. This association has not been seen in children with MPGN. These hepatitis C–positive cases often show vague substructure of deposits, with short, curved, vaguely fibrillar deposits suggestive of mixed cryoglobulinemia (see “Cryoglobulinemic Glomerulonephritis”). Features suggestive or even diagnostic of cryoglobulin as an underlying cause of MPGN include strongly PAS-positive “cryoplugs” in capillary lumina, vasculitis, predominant IgM deposits, sometimes with clonality or clonal shift of the light chain staining, and vague substructure of deposits by EM. Morphologic clues of an underlying chronic bacterial infection causing MPGN-type lesions are the presence of hump-type subepithelial deposits (see “Postinfectious Glomerulonephritis”). Dominant C3 and scanty Ig suggest possible C3GN.
Double contour appearance of glomerular basement membrane by light microscopy
Endocapillary hypercellularity
Chunky, irregular mesangial and capillary loop staining, typical sausage-type deposits underlying basement membrane (immunoglobulin G [IgG] dominant [or IgM, suggesting cryoglobulin], with complement C3 and often C1q)
Subendothelial and mesangial deposits by electron microscopy
Double contours or so-called “tram-tracking” of glomerular basement membranes (GBMs) may occur because of, for example:
subendothelial immune complexes or
monoclonal protein deposits (proliferative glomerulonephritis with monoclonal deposits)
C3 dysregulation (C3 glomerulonephritis)
chronic endothelial injury (with less hypercellularity) or
unusual deposits with substructure (e.g., fibrillary glomerulonephritis, immunotactoid glomerulopathy, type III collagen glomerulopathy, cryoglobulinemic glomerulonephritis, other)
Notes:
The term membranoproliferative glomerulonephritis describes a pattern, and the cause of the lesions should be defined by additional IF and electron microscopy findings and immunohistochemistry as indicated.
Double contours of GBMs may occur with chronic endothelial injury, where there is no IF positivity for immune complexes. These conditions include, for example, chronic thrombotic microangiopathy, transplant glomerulopathy, radiation nephropathy.
A membranoproliferative pattern of injury may also be seen without IF positivity for Ig or complement in, for example, type III collagen glomerulopathy and fibronectin glomerulopathy.
Fibrillary glomerulonephritis often has membranoproliferative appearance with endocapillary hypercellularity/proliferation. EM demonstrates the fibrillary nature of the deposits, which are typically polyclonal IgG and stain for DNAJB/9.
Amyloid may give rise to a nodular appearance of the mesangium and also involve the capillary loops. If the amyloid is because of monoclonal light chain, IF is positive in a smudgy pattern with clonal staining with a corresponding light chain. Amyloid of all types show characteristic fibrils by EM, and Congo red stain is classically positive.
Monoclonal Ig deposition disease, including light chain deposition disease, heavy chain deposition disease, and light and heavy chain deposition disease, may show variable proliferative appearance. IF is diagnostic in defining the monoclonal component of the deposits.
Proliferative glomerulonephritis with monoclonal deposits has a single heavy chain (most often IgG, with only one IgG subclass present) and a single light chain, with hypercellular/proliferative lesions, glomerular deposits, and no substructure by EM.
Dense deposit disease most often has a mesangial proliferative or membranoproliferative appearance, typically with only C3 staining. EM is diagnostic of the dense transformation of the basement membranes.
C3 glomerulonephritis most often has a mesangial proliferative or membranoproliferative appearance, typically with only C3 staining, with no or scanty Ig. EM shows density of deposits similar to immune complex disease, or sometimes less dense and similar to that of the glomerular basement membrane, with elongated subendothelial deposits and frequent mesangial deposits, and occasional subepithelial, hump-type deposits.
Alchi B, Jayne D. Membranoproliferative glomerulonephritis. Pediatr Nephrol. 2010;25:1409–1418.
Anders D, Agricola B, Sippel M, et al. Basement membrane changes in membranoproliferative glomerulonephritis. II. Characterization of a third type by silver impregnation of ultra-thin sections. Virchows Archiv (Pathology and Anatomy). 1997;376:1–19.
Berger J, Galle P. Dépots denses au sein des membranes basales du rein: étude en microscopies optique et électronique. Presse Med. 1963;71:2351–2354.
Cameron JS, Turner DR, Heaton J, et al. Idiopathic mesangiocapillary glomerulonephritis. Comparison of types I and II in children and adults and long-term prognosis. Am J Med. 1983;74:175–192.
Cook HT, Pickering MC. Histopathology of MPGN and C3 glomerulopathies. Nat Rev Nephrol. 2015;11:14–22.
Goodship TH, Cook HT, Fakhouri F, et al. Atypical hemolytic uremic syndrome and C3 glomerulopathy: Conclusions from a “Kidney Disease: Improving Global Outcomes” (KDIGO) Controversies Conference. Kidney Int. 2017;9:539–551.
Habib R, Kleinknecht C, Gubler MC, et al. Idiopathic membranoproliferative glomerulonephritis in children: Report of 105 cases. Clin Nephrol. 1973;1:194–214.
Johnson RJ, Gretch DR, Yamabe H, et al. Membranoproliferative glomerulonephritis associated with hepatitis C virus infection. N Engl J Med. 1993;328:465–470.
Katz SM. Reduplication of the glomerular basement membrane: A study of 110 cases. Arch Pathol Lab Med. 1981;105:67–70.
Nowicki MJ, Welch TR, Ahmad N, et al. Absence of hepatitis B and C viruses in pediatric idiopathic membranoproliferative glomerulonephritis. Pediatr Nephrol. 1995;9:16–18.
Sethi S, Fervenza FC. Membranoproliferative glomerulonephritis–a new look at an old entity. N Engl J Med. 2012;366:1119–1131.
Sethi S, Haas M, Markowitz GS, et al. Mayo Clinic/Renal Pathology Society consensus report on pathologic classification, diagnosis, and reporting of GN. J Am Soc Nephrol. 2016;27:1278–1287.
Smith RJH, Appel GB, Blom AM, et al. C3 glomerulopathy–understanding a rare complement-driven renal disease. Nat Rev Nephrol. 2019;15:129–143.
Strife CF, Jackson EC, McAdams AJ. Type III membranoproliferative glomerulonephritis: Long-term clinical and morphological evaluation. Clin Nephrol. 1984;21:323–334.
Strife CF, McEnery PT, McAdams AJ, et al. Membranoproliferative glomerulonephritis with disruption of the glomerular basement membrane. Clin Nephrol. 1977;7:65–72.
Taguchi T, Bohle A. Evaluation of change with time of glomerular morphology in membranoproliferative glomerulonephritis: A serial biopsy study of 33 cases. Clin Nephrol. 1989;31:297–306
Fibrillary glomerulonephritis was first reported by Rosenmann and Eliakim as a glomerulopathy with material very similar to amyloid that did not stain with Congo red. A distinctly different morphologic form of glomerulopathy with larger, microtubular organized structures has been termed immunotactoid glomerulopathy. The classification of these lesions was previously controversial. Some investigators had chosen to use the term immunotactoid glomerulopathy to refer to this entire group of disorders with deposits with substructure. Fibrillary GN is now recognized to be an entity distinct from immunotactoid glomerulopathy. It is characterized by immunostaining for DnaJ homolog subfamily B member 9 (DNAJB9), a very rare association with hematopoietic disease and very rare clonality of deposits, in contrast to immunotactoid glomerulopathy (see later). This separation paves the way for further insights into disease-specific prognosis and pathogenesis of these two entities.
Fibrillary GN is a disease of adults, with average age of onset approximately 50 years. Patients are most often Caucasian, with a slight female predominance. This entity makes up about 1% of diagnoses among adults undergoing native kidney biopsy. Most patients present with nephrotic syndrome and frequently have associated hematuria, with two-thirds with decreased GFR at presentation. Occasional patients had concomitant infection with hepatitis C, although a causal link has not been proven. Rapidly progressive GN clinically was present in approximately one-third of patients. The prognosis is one of progression to renal loss in approximately 40% of cases over 5 years, with a median renal survival time of only 24 months from time of biopsy in one large series. Better prognosis was seen in patients younger than 40 years at presentation. No specific treatment has been described. Patients who progressed to end-stage kidney disease generally did so rapidly, reaching end stage within 10 months on average after diagnosis. Elevated serum creatinine at presentation was a clinical sign of poor prognosis. Morphologic indicators of worse prognosis included crescents and worse tubulointerstitial fibrosis. Fibrillary GN recurred in the transplant in 17% to 31% in various series, with a slower course of loss of GFR in the graft than in the native kidney.
The light microscopic appearance in fibrillary GN most frequently is that of a membranoproliferative pattern, typically lobular, with GBM double contours ( Figs. 3.111–3.113 ). Less frequently, there is a mesangial proliferative or diffuse endocapillary proliferative pattern. Some cases may even appear similar to diabetic nephropathy, with large nodular expansion of mesangial matrix (see Fig. 3.111 ). In rare cases, there are GBM spikes in a membranous pattern, reflecting GBM reaction to the fibrillary deposits in a subepithelial location. Areas of deposits stain weakly PAS- and silver-positive. Crescents, either cellular or fibrocellular, were present in one-third of cases in one series but usually involved a minority of glomeruli ( Fig. 3.114 ). Crescents more commonly are associated with diffuse endocapillary hypercellularity and were associated with worse outcome. Glomerulosclerosis is associated with more severe disease (see Fig. 3.114 ). DNAJB9 has recently been identified from mass spectrometry and immunohistochemical studies to be present in nearly all cases of fibrillary GN and is a useful marker to distinguish this entity from other disease, especially if EM is not available. Classically, Congo red stains are negative in fibrillary GN. Nevertheless, rare cases of fibrillary GN, defined by typical IF, fibrils by EM, and DNAJB9 positivity, have shown Congo red positivity. In these cases, mass spectrometry confirmed absence of amyloid signatures in the deposits. Thus caution should be used in interpreting Congo red stains in this setting, and a diagnosis based on integration of all studies should be made.
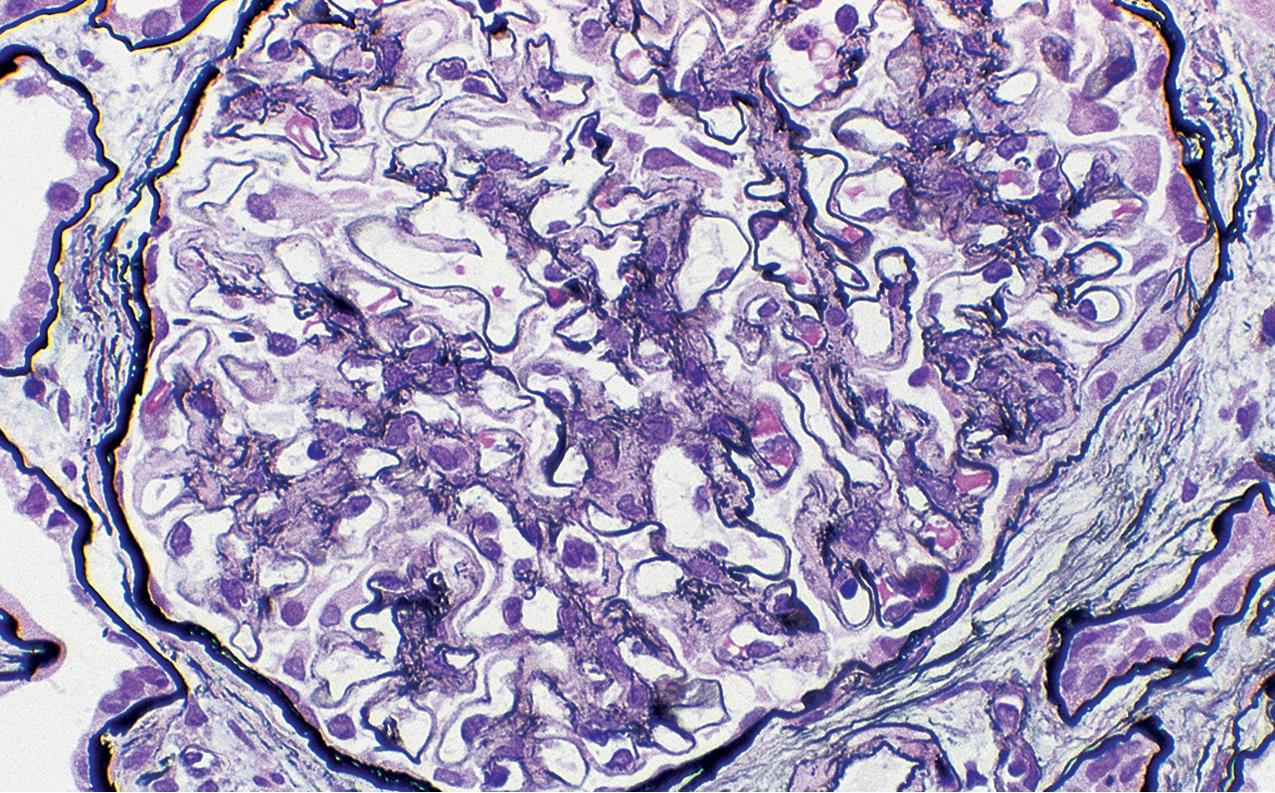
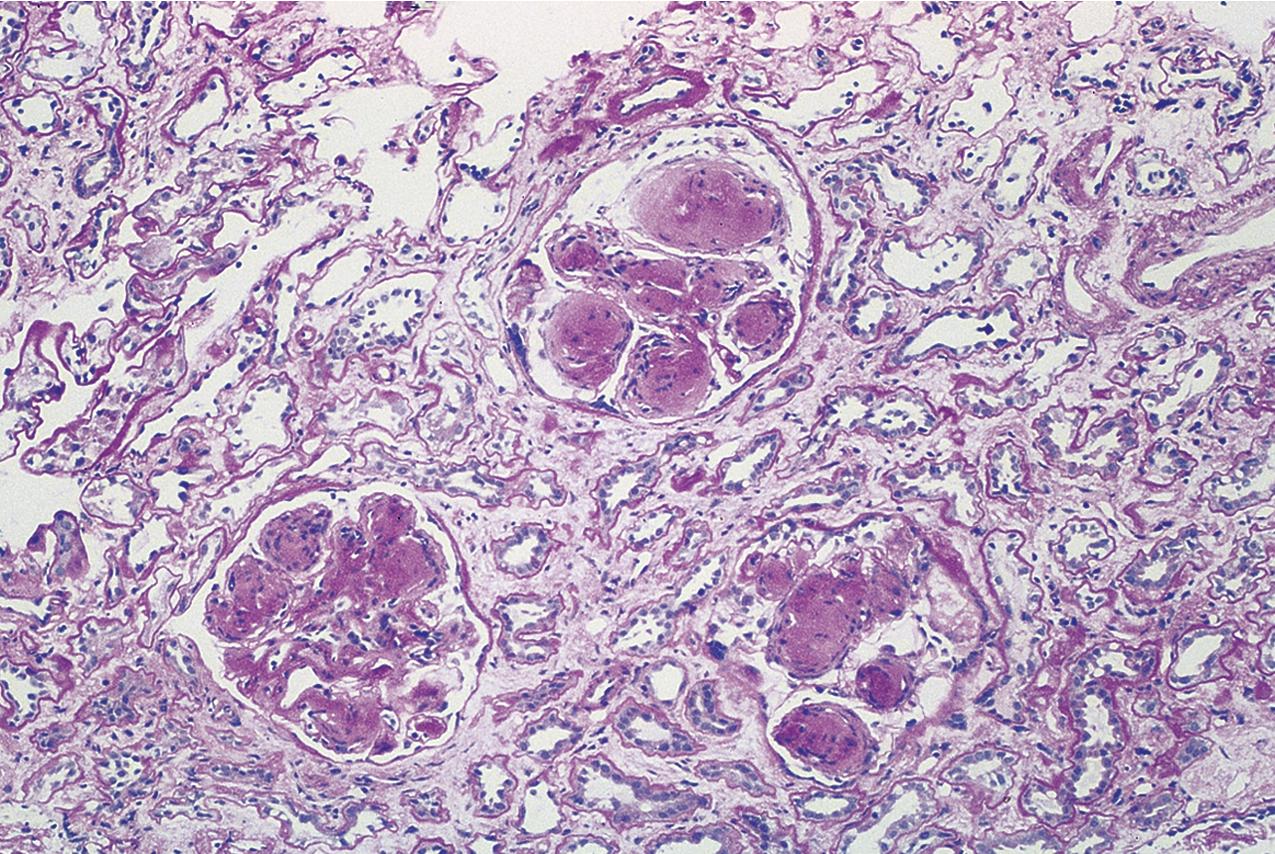
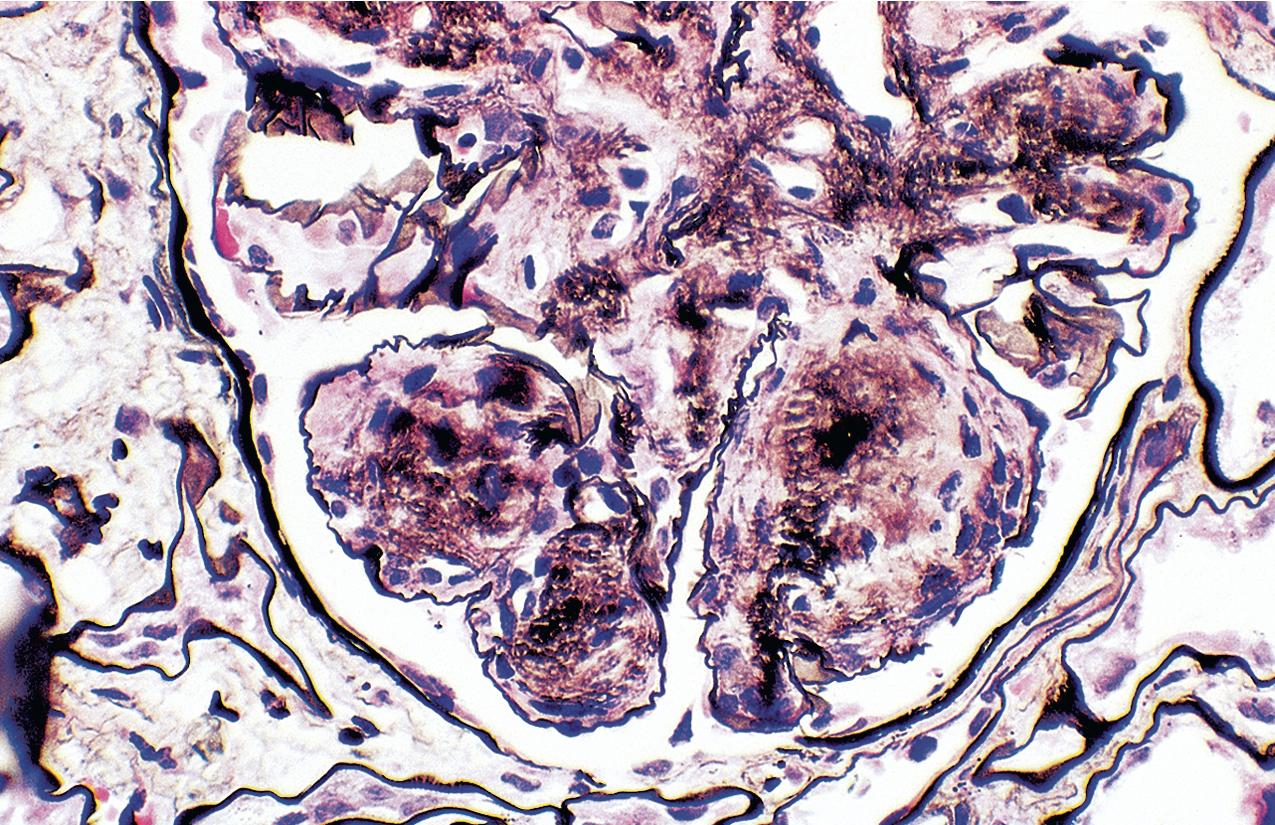
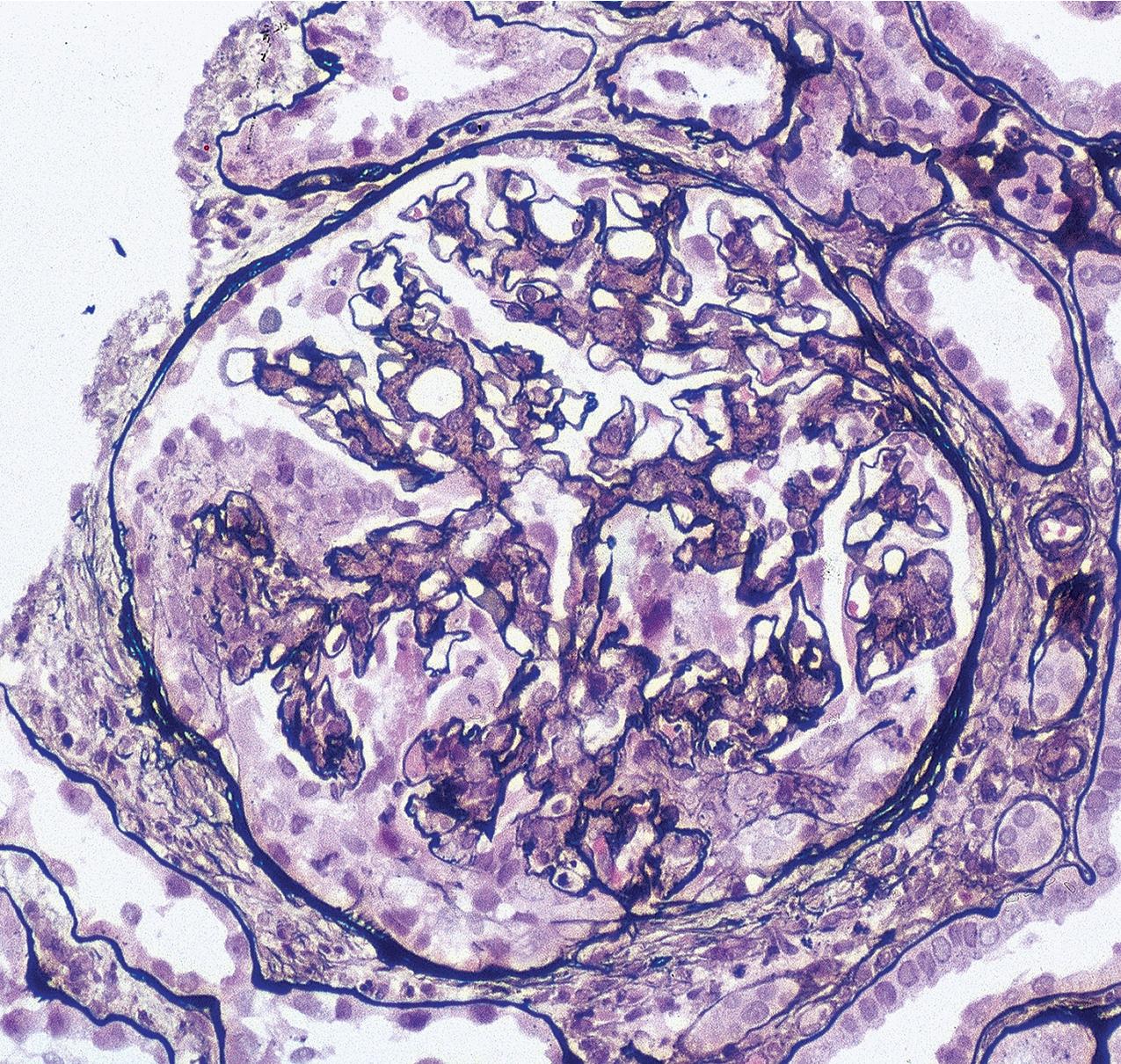
The interstitium shows interstitial fibrosis and tubular atrophy, proportional to glomerular changes. Rare cases have fibrillary deposits extending to the tubular basement membranes. Vessels do not show any specific lesions.
IF demonstrates prominent, smudgy IgG and lesser amounts of C3 in mesangial areas, and segmental, usually chunky, staining along GBMs, occasionally in a granular capillary wall membranous pattern ( Figs. 3.115–3.116 ). Polyclonal IgG4 is the dominant or exclusive subclass. Monoclonal deposits are rare. About half of cases with apparent monoclonality of deposits by standard IF studies on frozen tissue showed polyclonal deposits when further investigated by pronase digestion and staining for light chains and IgG subclasses on sections from paraffin-embedded tissue. In the rare remaining cases with monoclonal staining, detection of a circulating monoclonal protein was exceedingly rare.
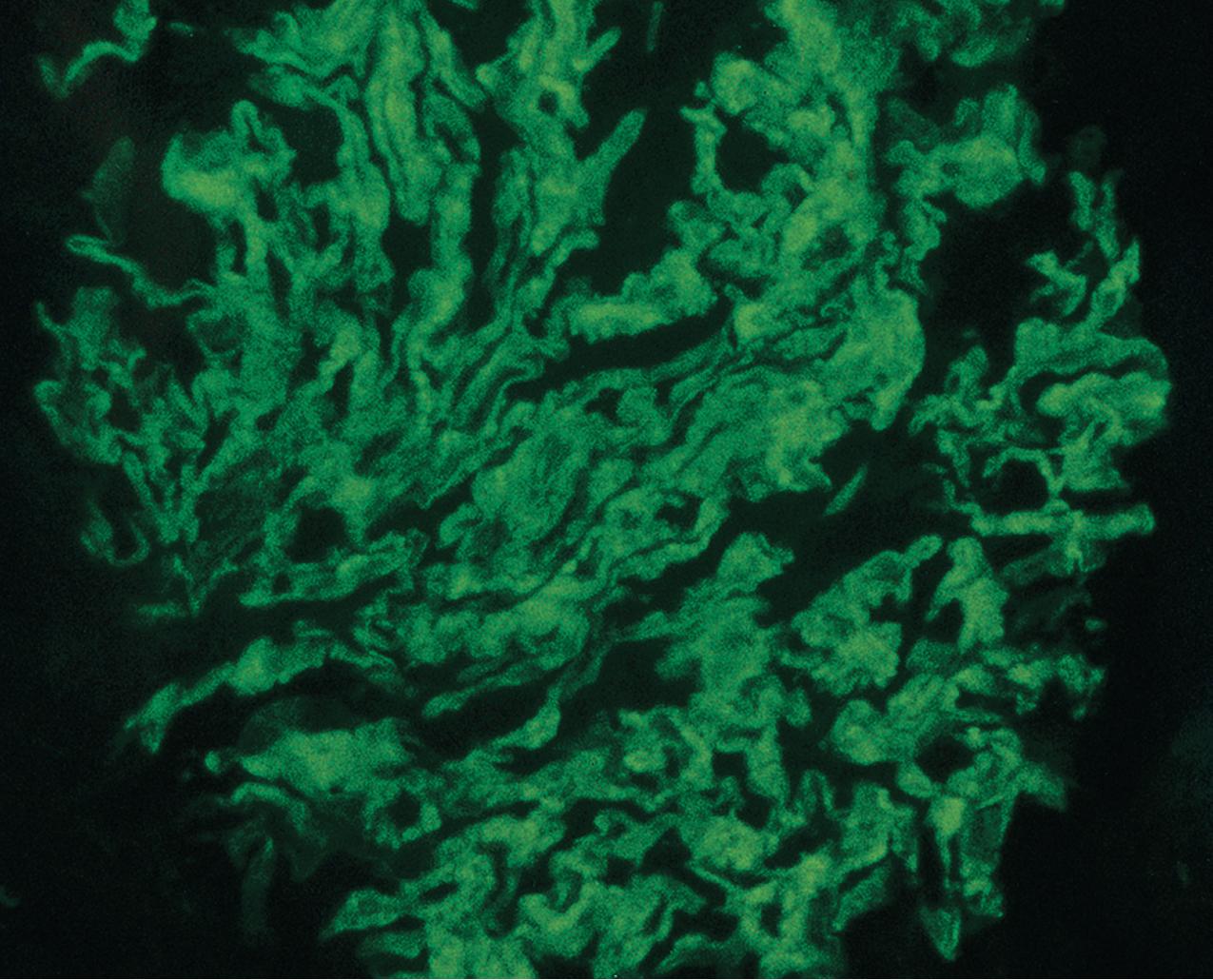
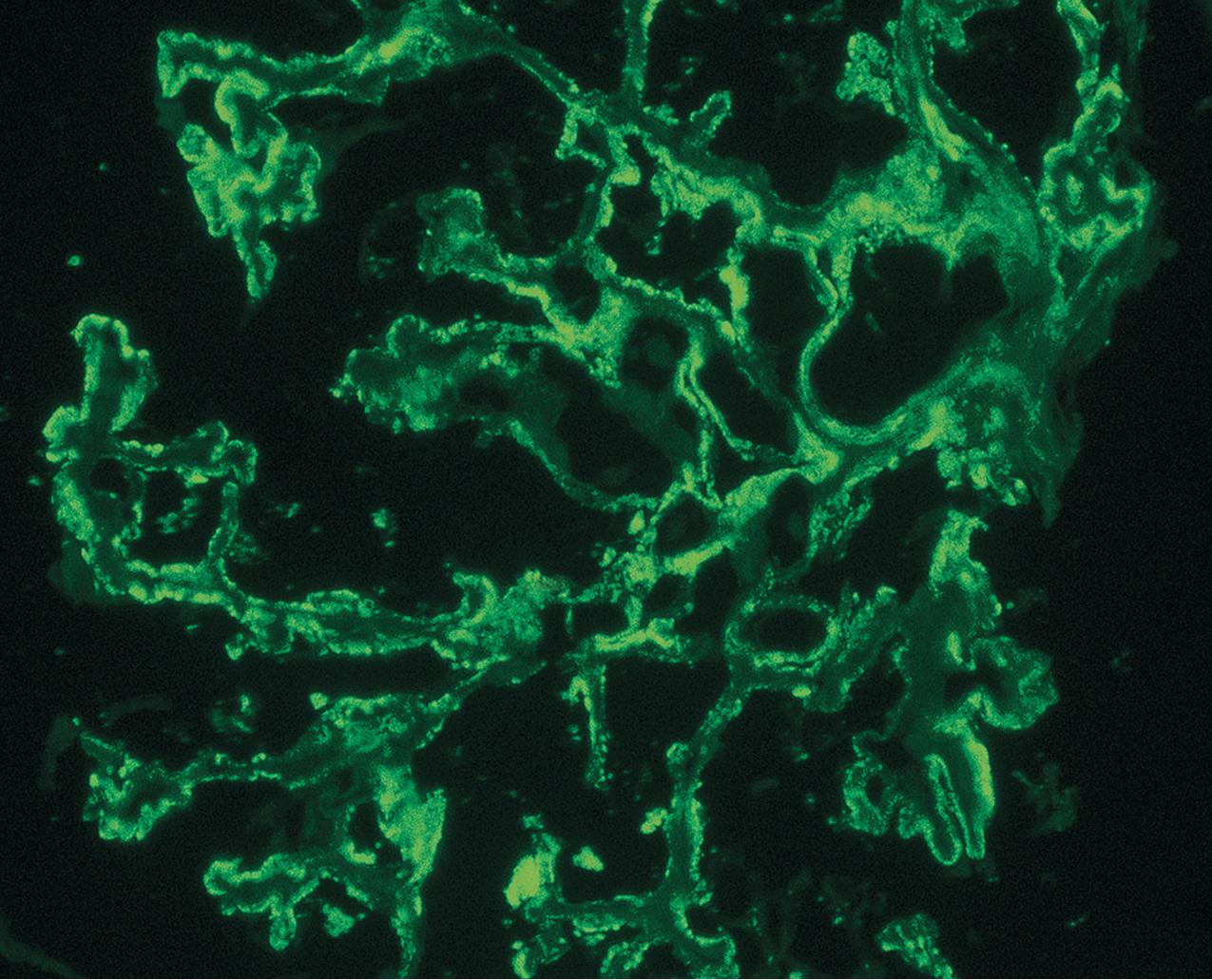
In about half of cases, weaker IgA and IgM and C1q may also be detected. Rare cases of other patterns have been reported, with predominant IgA deposits, or distinct fibrillar deposits by EM but no immunoglobulin staining. Not infrequently, the deposits are so diffuse as to provide an apparent linear staining by IF (see Fig. 3.116 ). This may, especially in cases with crescents, lead to an initial erroneous impression of possible anti-GBM antibody–mediated GN. The smudgy, predominantly mesangial staining suggests the specific diagnosis, confirmed by EM and negative Congo red stain.
The EM findings are then confirmatory of the diagnosis of fibrillary GN, showing the presence of randomly aligned fibrils that resemble amyloid fibrils but are larger ( Figs. 3.117–3.120 ). Usual fibril diameter is 12 to 22 nm, with a reported range of 13 to 39 nm in most series, contrasting with amyloid cases, which have a usual range 10 to 12 nm. In our series of fibrillary GN, however, there was some overlap with amyloid fibril size, with average fibril diameter in fibrillary GN cases of 14 nm (range 10.4–18.4 nm). DNAJB9 staining ( Fig. 3.121 ), in conjunction with Congo red–negative staining (in most cases) and typical IF staining, as described earlier, are useful as diagnostic criteria. EM may show fibrils in all glomerular compartments, including mesangium and basement membrane in intramembranous, subepithelial, and subendothelial locations. Additional dense deposits without distinct fibrillary composition have been observed in some cases (see Fig. 3.119 ). Rare tubular basement membrane fibrillary deposits may occur ( Fig. 3.122 ).
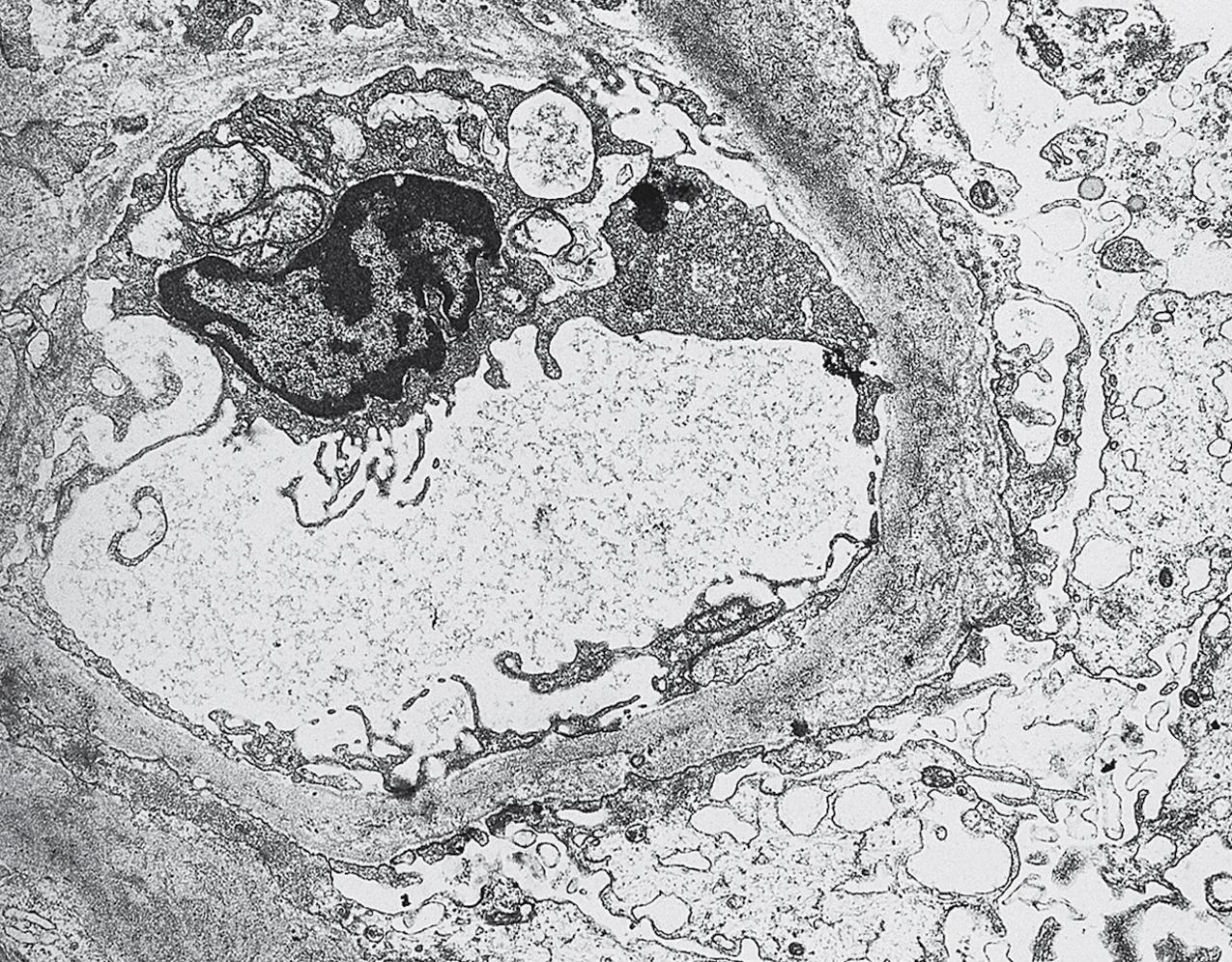
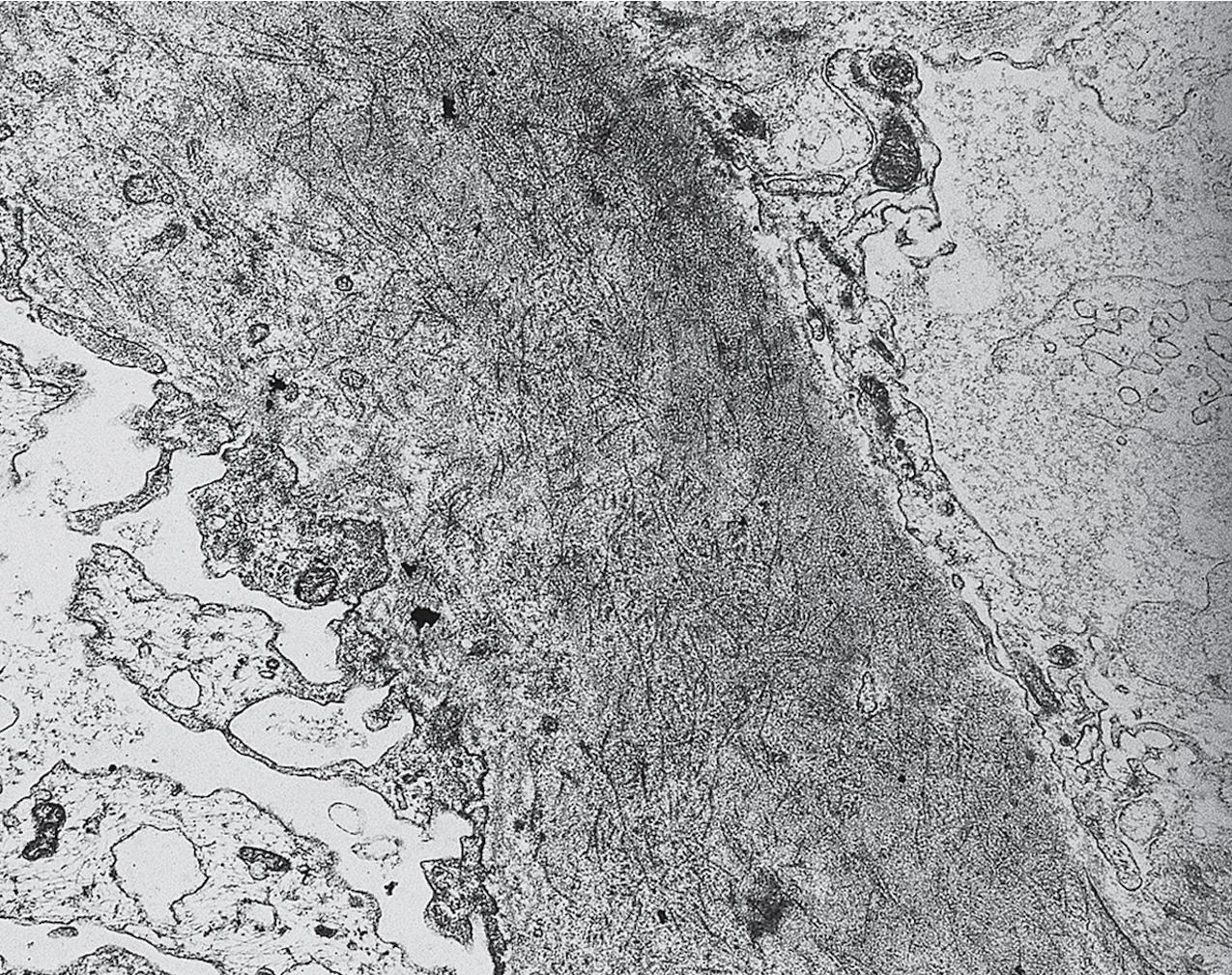
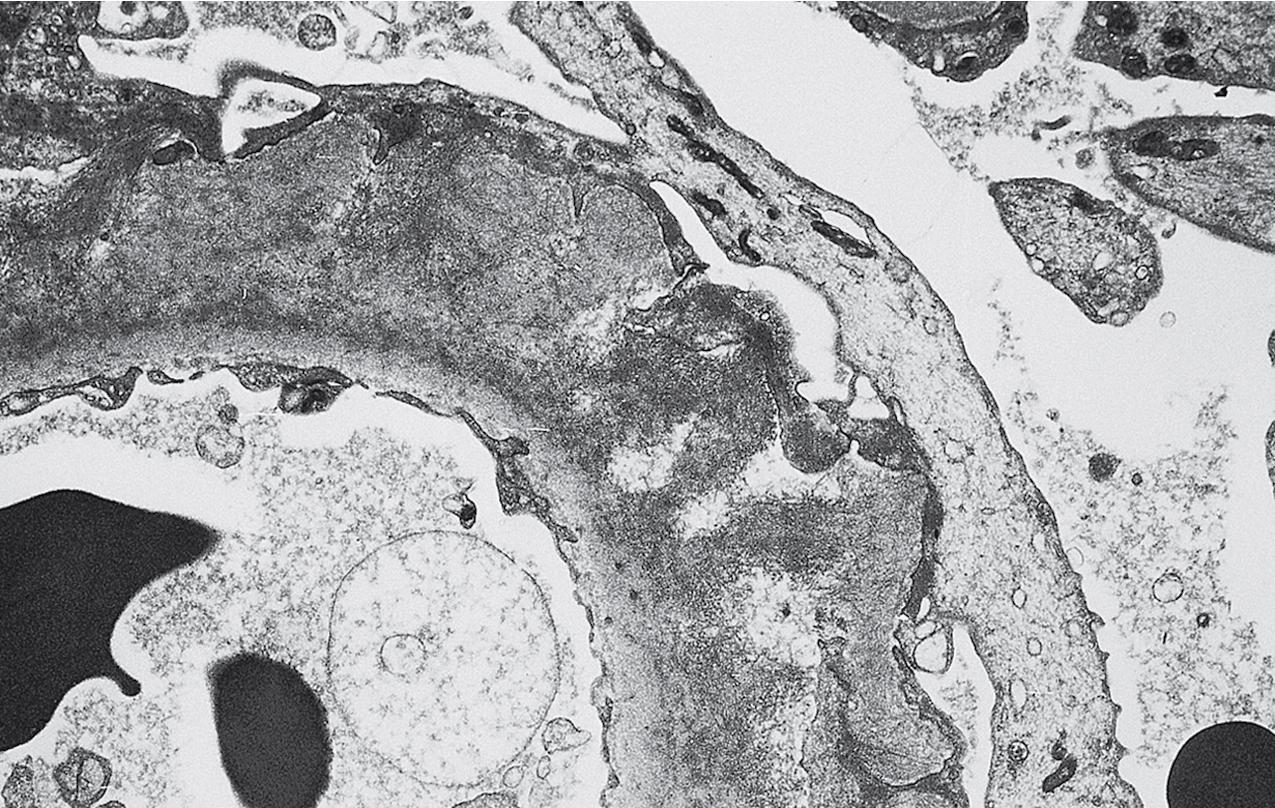
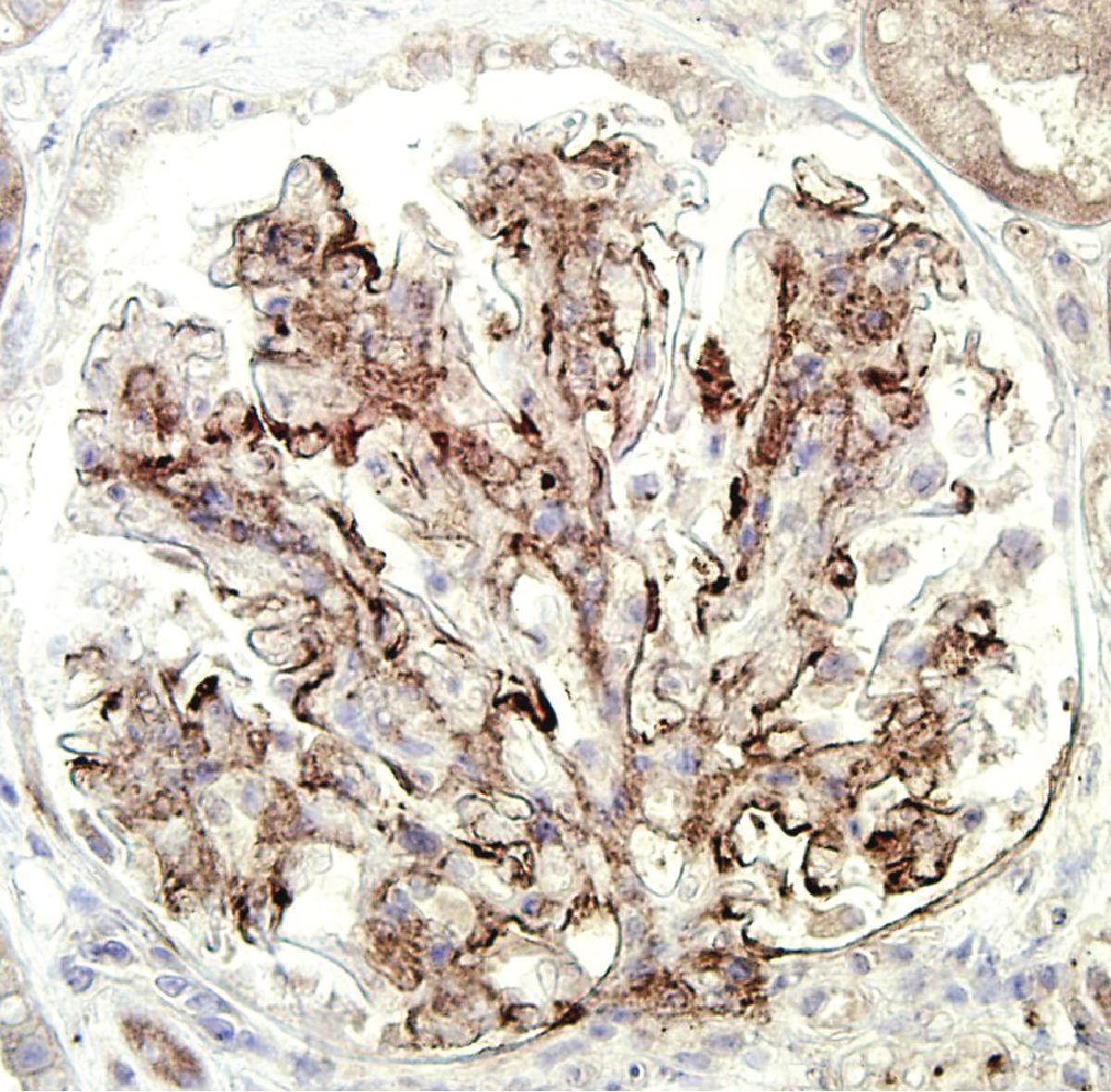
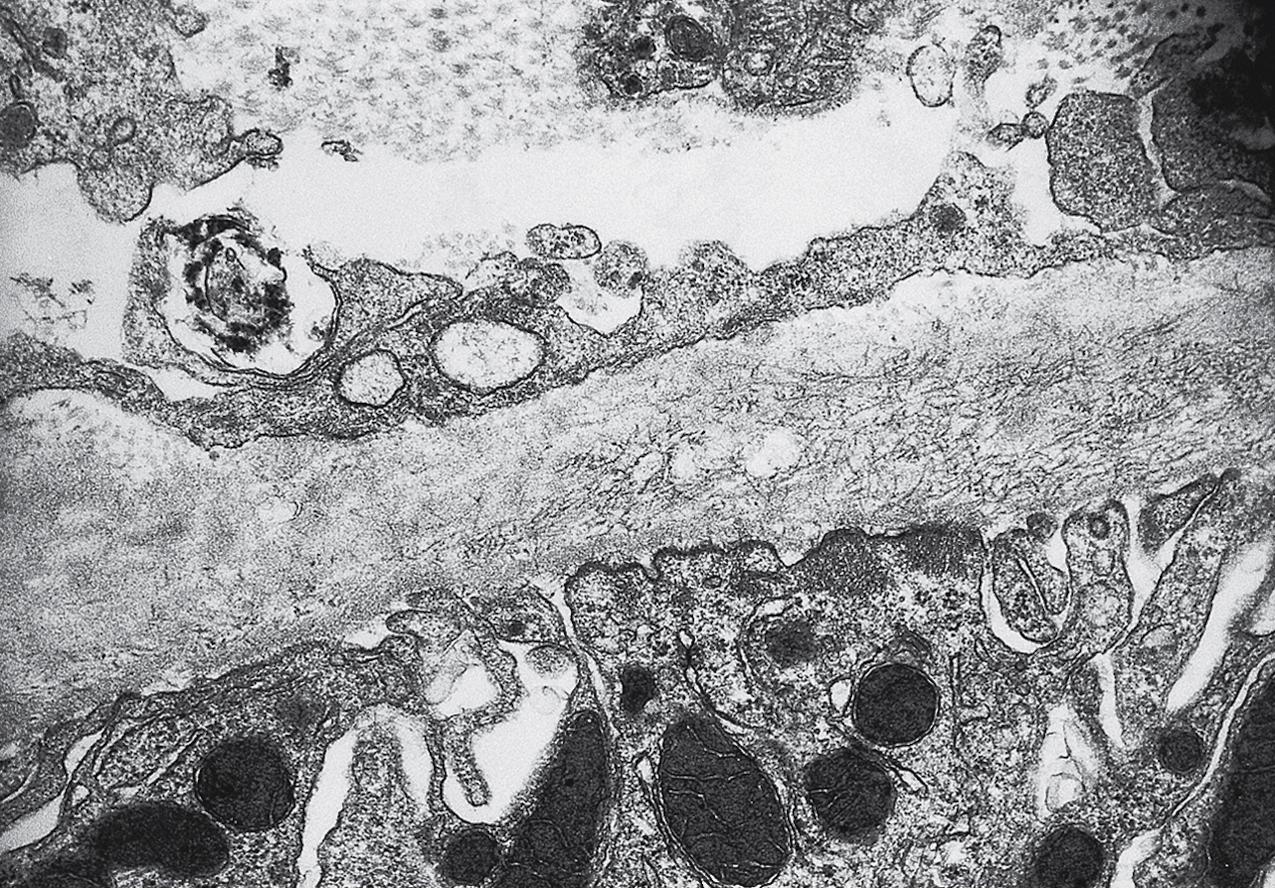
The discovery of DNAJB9 within the deposits is a useful marker of the disease. The precise etiology of fibrillary GN, however, remains unknown. DNAJB9 is a heat-shock protein that plays a role in the endoplasmic reticulum stress/unfolded response pathway. DNAJB9 binds aggregation-prone peptides. DNAJB9 transcription is not increased within glomeruli. Hypotheses for pathogenesis include, for example, DNAJB9 as an autoantigen, or DNAJB9 binding to misfolded IgG, or aberrantly synthesized DNAJB9 at extraglomerular sites and then accumulating within the glomeruli along with nonspecific IgG. Serum levels of DNAJB9 are moderately increased in fibrillary GN patients, with moderate sensitivity and high specificity compared with individuals without kidney disease or patients with other glomerular diseases. Whether these patients also have circulating antibodies specific for DNAJB9 currently is undetermined.
Occasional association with hepatitis C infection has been reported, but causality has not been proven. Importantly, fibrillary GN is not causally associated with lymphoproliferative disorders or monoclonality of deposits, in contrast to immunotactoid glomerulopathy.
Typically membranoproliferative or mesangial proliferative by light microscopy
Smudgy mesangial and capillary wall polyclonal positivity for immunoglobulin G (IgG) and C3
DNAJB9 positivity in areas of deposits
Randomly arranged fibrils by electron microscopy, classically Congo red–negative
Fibrillary glomerulonephritis
LM: proliferative, focal crescents in up to a third of cases
IHC: DNAJB9 positive
IF: IgG polyclonal, smudgy
Congo red: negative (most often)
EM: fibrils random, 12 to 24 nm (mostly 18–20 nm)
Immunotactoid glomerulopathy
LM: proliferative
IHC: DNAJB9 negative
IF: IgG, often clonal
Congo red: negative
EM: organized, parallel, microtubular (>30 nm)
Amyloid
LM: acellular, mesangial/lobular
IHC: DNAJB9 negative
IF: monoclonal light chain for AL; no light chain for non-AL
Congo red: positive
EM: fibrils random, 8 to 15 nm (mostly 10–12 nm)
Cryoglobulinemic glomerulonephritis
LM: proliferative, PAS-positive cryoplugs
IF: IgM, often clonal component
Congo red: negative
EM: microtubular (type I) or vague, short fibrillary (mixed cryoglobulin)
Alexander MP, Dasari S, Vrana JA, et al. Congophilic fibrillary glomerulonephritis: A case series. Am J Kidney Dis. 2018;72:325–336.
Alpers CE. Immunotactoid (microtubular) glomerulopathy: An entity distinct from fibrillary glomerulonephritis? Am J Kidney Dis. 1992;19:185–191.
Alpers CE, Rennke HG, Hopper JJ, et al. Fibrillary glomerulonephritis: An entity with unusual immunofluorescence features. Kidney Int. 1987;31:781–789.
Andeen NK, Yang HY, Dai DF. DnaJ homolog subfamily member 9 is a putative autoantigen in fibrillary GN. J Am Soc Nephrol. 2018;29:231–239.
Bridoux F, Hugue V, Coldefy O, et al. Fibrillary glomerulonephritis and immunotactoid (microtubular) glomerulopathy are associated with distinct immunologic features. Kidney Int. 2002;62:1764–1775.
Churg J, Venkataseshan VS. Fibrillary glomerulonephritis without immunoglobulin deposits in the kidney. Kidney Int. 1993;44:837–842.
Dasari S, Alexander MP, Vrana JA, et al. DnaJ heat shock protein family member 9 is a novel biomarker for fibrillary GN. J Am Soc Nephrol. 2018;29:51–56.
Fogo A, Quereshi N, Horn RG. Morphologic and clinical features of fibrillary glomerulonephritis versus immunotactoid glomerulopathy. Am J Kidney Dis. 1993;22:367–377.
Iskandar SS, Falk RJ, Jennette JC. Clinical and pathological features of fibrillary glomerulonephritis. Kidney Int. 1992;42:1401–1407.
Korbet SM, Schwartz MM, Rosenberg BF, et al. Immunotactoid glomerulopathy. Medicine. 1985;64:228–243.
Nasr S, Fogo AB. New developments in the diagnosis of fibrillary glomerulonephritis. Kidney Int. 2019;96:581–592.
Pronovost PH, Brady HR, Gunning ME, et al. Clinical features, predictors of disease progression and results of renal transplantation in fibrillary/immunotactoid glomerulopathy. Nephrol Dial Transplant . 1996;11:837–842.
Ray S, Rouse K, Appis A, et al. Fibrillary glomerulonephritis with hepatitis C viral infection and hypocomplementemia. Ren Fail. 2008;30:759–762.
Rosenmann E, Eliakim M. Nephrotic syndrome associated with amyloid-like glomerular deposits. Nephron. 1977;18:301–308.
Rosenstock JL, Markowitz GS. Fibrillary glomerulonephritis: An update. Kidney Int Rep. 2019;4:917–922.
Rosenstock JL, Markowitz GS, Valeri AM, et al. Fibrillary and immunotactoid glomerulonephritis: Distinct entities with different clinical and pathologic features. Kidney Int. 2003;63:1450–1461.
Said SM, Leung N, Alexander MP, et al. DNAJB9-positive monotypic fibrillary glomerulonephritis is not associated with monoclonal gammopathy in the vast majority of patients. Kidney Int. 2020;98:498–504.
Schwartz MM, Lewis EJ. The quarterly case: Nephrotic syndrome in a middle-aged man. Ultrastruct Pathol. 1980;1:575-582.
Sethi S, Theis JD, Vrana JA, et al. Laser microdissection and proteomic analysis of amyloidosis, cryoglobulinemic GN, fibrillary GN, and immunotactoid glomerulopathy. Clin J Am Soc Nephrol. 2013;8:915–921.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here