Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Physical pain is a leading cause of healthcare spending, work-related disability, and morbidity in the United States ( ). According to a 2008 Medical Expenditure Panel Survey, the direct and indirect costs of chronic pain were found to range from $560 to $635 billion annually ( ). From a neurosurgical perspective, acute and chronic pain are specific indications for surgical intervention for a variety of disorders. Intractable pain conditions, such as failed back surgery syndrome or trigeminal neuralgia, are currently assessed through the use of subjective, self-reported, standardized rating scales ( ). Neuromodulation therapies are initially very expensive, and although they can be useful for research purposes, these scales do not provide an objective assessment of pain severity. The development of advanced functional magnetic resonance imaging (fMRI) techniques, such as blood oxygenation level dependent (BOLD) imaging, arterial spin labeling (ASL), and resting-state functional connectivity MRI (rs-fMRI) offers researchers the prospect of developing objective diagnostic instruments for acute and chronic pain. This chapter reviews recent advancements in functional imaging, and the diagnostic and therapeutic opportunities they present for the management of pain syndromes. In addition, advanced neuroimaging techniques can assist clinicians and researchers in better understanding the effect of analgesics and placebos in the treatment of physical pain.
The advent of fMRI in the 1990s allowed clinicians to examine changes in brain activity after a stereotyped task ( ). The first imaging methods used intravenous gadolinium contrast or the BOLD technique, which detects levels of deoxyhemoglobin in stimulated brain ( ). When subjected to a simple mechanical or visual task, areas of eloquent cortex become activated and subsequently utilize oxygen to generate energy. As a result, local levels of deoxyhemoglobin increase in concentration, and these depots of deoxyhemoglobin have inherent paramagnetic properties that can be detected with BOLD imaging ( ). Since then there have been significant advancements in the imaging capabilities of fMRI, including the ability to examine brain activity at rest with rs-fMRI ( ). When compared to BOLD imaging, which depicts brain activity during stereotyped tasks, rs-fMRI demonstrates brain activity without an applied stimulus, or at rest. Although intraoperative, direct electrical stimulation remains the gold standard for mapping functional cortex, these new fMRI technologies allow better preoperative planning and intraoperative guidance.
Fomberstein et al. described several anatomic regions in the brain that have been implicated in pain perception ( ). These areas, termed the “pain matrix,” include the primary and secondary somatosensory cortex, anterior cingulate cortex, and insula ( ). Abnormal functional connectivity between these areas has been shown to be highly correlated with several chronic pain syndromes ( ). The authors found that functional connectivity between the prefrontal cortex, insula, and anterior cingulate cortex could be used as a marker for chronic pain that correlates highly with poor treatment response to opioid therapy ( ) ( Fig. 46.1 ) . The authors noted several interesting patterns of activation, displayed on fMRI in several chronic pain states. For example, patients with chronic low back pain (CLBP) had increased activation of the medial prefrontal and anterior cingulate cortex when compared to normal controls. After 2 weeks of treatment, connectivity of the dorsal median prefrontal cortex and insula was seen in the treatment-refractory patient group ( ). In addition, higher connectivity between the dorsal median prefrontal cortex and insula was directly correlated with decreased responsiveness to placebo treatment in this study ( ). These results support the long-held belief that chronic pain syndromes are a multifaceted entity involving more than just noxious stimulus of peripheral or autonomic nerves ( ).
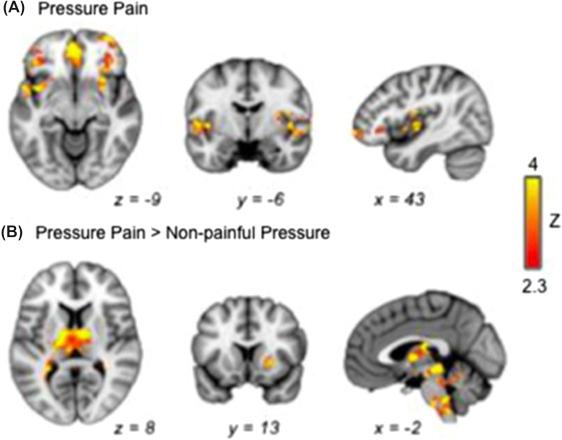
As most clinicians are well aware, patients with complex chronic pain syndromes can be quite challenging to manage and often require multidisciplinary care. These results suggest that fMRI techniques could be used to predict treatment response to analgesia, thus creating a framework to build patient-specific analgesic regimens that are supported by connectivity. Connectivity could be used to predict opioid response and probability of treatment success/failure, set thresholds for treatment duration, and monitor treatment progression. Unlike conventional MRI, fMRI can be used to obtain objective information about subjective clinical data, such as pain. With this in mind, there have been more recent efforts to tailor fMRI sequences to detecting areas that become active after a noxious stimulus, in hope that this data can be used to “diagnose” pain.
In 2013 Wager et al. were the first to develop an fMRI-based neurologic signature that could detect physiological pain. In response to a noxious stimulus, Wager’s pain signature demonstrated increased activity in the primary and secondary somatosensory cortices that was over 90% sensitive and specific for the experimental noxious stimulus ( ). The study investigators hypothesized that the signature could be applied to clinical situations when patients are unable to communicate pain or clinicians suspect incorrect self-reporting of pain.
In another study conducted by Loggia et al. in 2014, fMRI was used to characterize pain physiology and circuit pathways in 13 patients with fibromyalgia. The investigators found that these patients repeatedly experienced an exaggerated response to a painful stimulus when compared to normal volunteers. In addition, fibromyalgia patients demonstrated reduced reward/punishment signaling, thought to be due to decreased activity in the periaqueductal gray and ventral segmental areas of the midbrain. They hypothesized that suppression of activity in these regions may explain why fibromyalgia patients do not respond well to traditional pain therapies ( ). In fact, the treatment of fibromyalgia often requires a multidisciplinary clinical approach and the judicious use of neuroleptic, antidepressant, and occasionally antiepileptic mediations ( ). The results of these studies emphasize the difficulty in treating chronic pain disorders with opioid medications alone, and may suggest that a multimodality approach is needed.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here