Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
This chapter will review extracranial germ cell tumors (GCTs). Please see Chapter 22 , Central nervous system tumors, for a discussion of intracranial GCTs.
Tumors of germ cell origin display a bimodal distribution, with a peak seen in young children between 0 and 4 years. During childhood, GCTs account for 3.5% of childhood malignancies. In the United States the age-adjusted incidence of GCT in children 0–14 years is 0.4 per 100,000 in males and 0.6 per 100,000 in females.
A second peak in older children and adolescents starts at 9 years of age in females and at 11 years of age in males and correlates with sex-specific onset of puberty. In patients between age 15 and 19 years, GCTs account for 13.9% of malignancies making GCT one of the most common malignancies in this age group. The incidence of GCT significantly increases to a maximum rate of 11.4 per 100,000 in adolescent males and only a slight increase to 1 per 100,000 in adolescent females.
GCTs are neoplasms that arise from early germline progenitors: the primordial germ cells of the human embryo. These primordial germ cells originate in the yolk sac endoderm of the embryo and migrate around the hindgut to the gonads. Due to their cell of origin, GCTs are biologically heterogeneous tumors resulting in a spectrum of benign and malignant diseases. The Teilum hypothesis of tumorigenesis postulates oncogenic events occurring at different stages of differentiation give rise to different histological types in GCT. Fig. 28.1 depicts the histogenesis of tumors of germ cell origin. Germinomas/seminomas arise from undifferentiated primordial cells. Embryonal carcinomas (ECs) originate from cells with early embryonic differentiation and, therefore, can differentiate into teratomas. Finally, primordial cells that have extraembryonic differentiation give rise to yolk sac tumors (YSTs) or choriocarcinomas (CCs). Importantly, the biological characteristics of these tumors correlate with the age at presentation of GCT, suggesting that GCT may comprise two related but distinct entities.
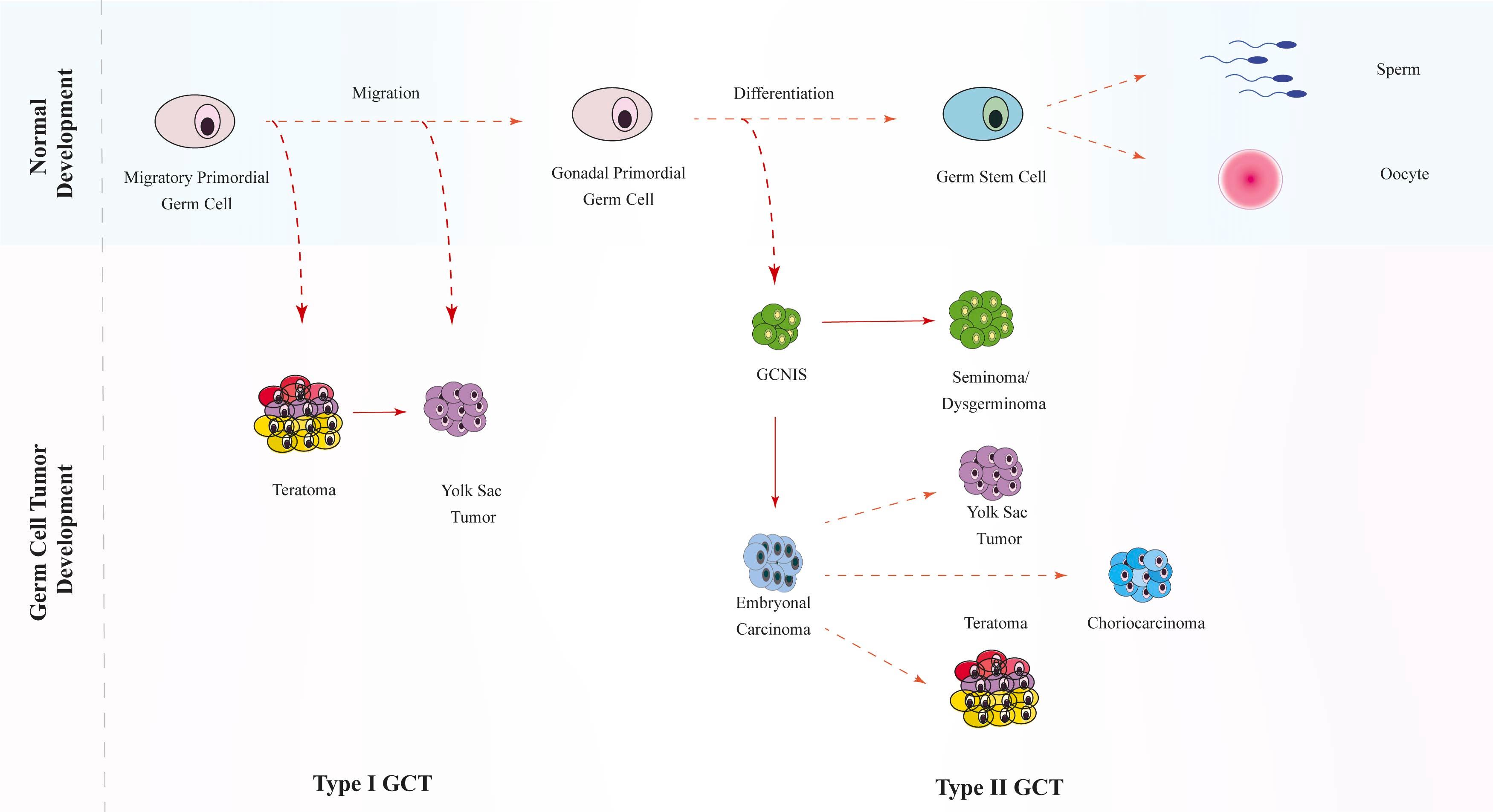
A summary of histological characteristics is presented in Table 28.1 . Histologically, GCTs are classified into two major types.
| Histologic variant | Morphological characteristics | Common sites of origin | AFP | β-hCG | PLAP |
|---|---|---|---|---|---|
| Mature teratoma |
|
|
− | − | − |
| Immature teratoma |
|
|
− | − | − |
| Germinoma |
|
|
− | − | + |
| Yolk sac tumor | Characteristics as seen microscopically:
|
|
+ | − | − |
| Embryonal carcinoma |
|
Testis (young adult) | − | ± | ± |
| Choriocarcinoma | Two types of cells are seen microscopically:
|
|
− | + | − |
Germinomatous/seminomatous GCTs : Characterized by the presence of undifferentiated germ cells histologically resembling early precursors of spermatogonia or oogonia. These tumors are termed seminoma if located in the testis, dysgerminoma if located in the ovary, or germinoma if located in extragonadal sites.
Nongerminomatous/nonseminomatous GCTs (NGGCTs) : Comprise different benign and malignant histological subtypes.
Teratomas are differentiated tumors characterized by the presence of mature cells from all three germ layers (ectoderm, endoderm, and mesoderm). In some cases, teratomas can acquire a unique neural differentiation state and termed immature teratoma .
YSTs or endodermal sinus tumors are histologically characterized by a complex endodermal morphology resembling embryonic and extraembryonic structures.
EC is an undifferentiated tumor and histologically resemble embryonic cells from the blastocyst.
CC is a rare histological subtype with characteristic trophoblastic differentiation.
Neoplasms that contain two or more malignant germ cell elements are known as mixed malignant GCTs.
Increasing evidence has emerged of the different biology driving prepubertal and postpubertal GCTs. Genomic studies have identified characteristic alterations in childhood GCT. In particular, chromosomal gains in 1q, 11q, 20q, and 22, as well as loss of 1p, 6q, and 16q are more prevalent in younger patients. In adult GCT the most frequent chromosomal aberrations are the amplification of chromosome 12p, usually as isochromosome 12 and present in 80% of adult testicular GCT. Although not exclusive of adult-like GCT, this alteration has only been identified in 29% of patients younger than 5 years with GCT.
Specific methylation patterns have helped elucidate different cells of origin in childhood and adult-type GCT. Adult-type GCTs demonstrate a widespread hypermethylation of tumor suppressor genes and secondary silencing of proapoptotic genes. The most common genes involved in this process include alterations in adenomatous polyposis coli (APC) , Ras Association Domain Family 1 isoform A (RASSF1A) , and Hypermethylated In Cancer 1(HIC1). These mechanisms may explain the higher aggressiveness of GCT in adolescents as the biology resembles adult-like disease.
Eight main miRNAs miR-371–373 and miR302a/302d are overexpressed in all malignant GCT and can be measured in serum, across age groups, histological types, and anatomical sites. Moreover, cisplatin resistance in GCT has been associated with miR-371–373 upregulation along with the downregulation of tumor suppressor miRNA miR-99a and miR-100; therefore miRNAs could be useful identifying therapeutic susceptibilities. The clinical role of these molecules was evaluated in adults with testicular GCT and demonstrated clinical usefulness in the diagnosis and recurrence monitoring. Similarly, miRNAs are currently being validated in pediatric trials.
Extracranial GCTs present in gonadal and extragonadal sites. The most common childhood extragonadal site is the sacrococcygeal region (42%) followed by mediastinum (7%) and retroperitoneum (4%). Gonadal tumors [ovaries (24%) and testes (9%)] account for one-third of patients diagnosed with GCT in childhood. Although GCTs are a diverse group histologically, distant metastatic disease is present at diagnosis in approximately 20% of patients. The pattern of spread is similar, irrespective of the primary site: lungs, liver, regional nodes, central nervous system (CNS), and bone and bone marrow.
The signs and symptoms of GCTs are dependent on the site of origin. Table 28.2 lists the clinical features of GCTs at different sites. In addition, certain histologic variants may have associated clinical findings, as listed in Table 28.3 .
| Age median (year) | Frequency (%) | Clinical features | ||||
|---|---|---|---|---|---|---|
| Tumor type | Female | Male | Female | Male | Female | Male |
| Mature/immature teratoma | 13 | 3 | 41 | 24 | Abdominal pain, abdominal mass. 10% bilateral. Gliomatosis peritonei does not affect prognosis | Nontender scrotal mass. Excellent survival with surgery alone in prepubertal males |
| Dysgerminoma/seminoma | 16 | 30 | 24 | <1 | Rapidly developed intraabdominal mass. 20% bilateral. 14–25% mixed with other germ cell elements | Virtually not seen in children, most common testicular tumor in adult males. Most patients present in their 30s |
| YST | 18 | 2 | 16 | 26 | Most common malignant histology. 75% stage I | Most common malignant histology, most pediatric tumors are pure YST. 85% stage I |
| Embryonal carcinoma | 14 | 17 | 6 | 20 | Associated with precocious puberty, amenorrhea, and hirsutism | Higher incidence of metastatic disease at presentation, patients can present with retroperitoneal mass |
| Choriocarcinoma | N/A | N/A | <1 | 16 | Rare in children, when present in infants related to maternal metastatic disease | Frequent pulmonary metastatic disease at presentation, patients can present with hemoptysis due to MetS hemorrhage |
| Gonadoblastoma | 9 | 7 | 1 | <1 | Associated with gonadal dysgenesis. Presents mostly in phenotypical females with a Y chromosome | Very rare |
| Histology | Clinical association |
|---|---|
| Teratoma | Musculoskeletal anomalies |
| Rectal stenosis | |
| Congenital heart disease | |
| Microcephaly | |
| Ovarian dysgerminoma (postpubertal) | Amenorrhea |
| Menorrhagia | |
| 46XY (male pseudohermaphrodite) | |
| Ovarian embryonal carcinoma | Precocious puberty |
| Amenorrhea | |
| Hirsutism |
The following evaluations should be carried out:
History
Physical examination
Complete blood count
Liver function tests, electrolytes, blood urea nitrogen, and creatinine
α-Fetoprotein (AFP)
Beta-subunit of human chorionic gonadotropin (β-hCG)
Lactic dehydrogenase (LDH) isoenzyme 1
Cross-sectional evaluation of primary site and regional disease
Mediastinum—chest and upper abdominal computed tomography (CT)
Ovary—ultrasound, pelvic and abdominal CT or magnetic resonance imaging (MRI)
Sacrococcygeal—pelvic and abdominal CT or MRI
Testis—ultrasound, pelvic and abdominal CT or MRI
Radiographic evaluation for distant metastases
Chest radiographs (posteroanterior and lateral)
Chest CT
Positron emission tomography scan or bone scan
Certain histologic variants of GCT secrete tumor markers that can be measured in serum and aid in the diagnosis and management of these patients. AFP is a major serum protein of the human fetus. It is produced in the embryonic liver, in the yolk sac, and, in smaller amounts, in the gastrointestinal tract. Elevated levels of AFP in an infant must be interpreted with caution and using age-adjusted values.
Serum AFP is highly elevated in YSTs and a slight to intermediate elevation seen in EC. AFP is elevated in approximately 70% of NGGCT of childhood. In adults, serum levels of AFP at diagnosis can be predictive of outcome and is part of the risk stratification criteria. Furthermore, the rate of AFP decline has been shown to be prognostic in adolescents and adults.
The β-hCG is elevated in the presence of syncytiotrophoblastic components in the tumor. High levels of β-hCG are found in CCs and slight elevations can be observed in ECs and germinomas. It is thought that 20% of germinomas have β-hCG elevation. Sudden increases of β-hCG after starting chemotherapy can be seen due to cell lysis. Additionally, it can be elevated with malignancies of the liver, pancreas, Gastrointestinal tract, breast, lung, and bladder.
Pure teratomas are not associated with AFP or β-hCG production; the elevation of either marker in association with teratoma indicates the presence of malignant germ cell elements and requires review of the histologic material.
Historically, staging classifications for GCT differed depending on the site of origin and the age of the patient. Children’s Oncology Group (COG) classification has been used for staging in the pediatric population. The International Germ Cell Consensus Classification (IGCCC) has been used to risk stratify adolescent males with testicular tumors and the International Federation of Gynecology and Obstetrics (FIGO) system has been used to stage adolescent females with ovarian tumors.
The Malignant Germ Cell International Consortium (MaGIC) was created to bring together pediatric, gynecologic, and testicular cancer specialists to promote research initiatives and provide evidence-based approaches in the management of GCTs across different age groups. In 2015 MaGIC developed a common risk stratification system by merging 25 years of clinical trial data from COG in the United States and Children’s Cancer and Leukaemia Group in the United Kingdom. The MaGIC risk stratification system incorporates the IGCCC classification and is, thus, applicable to all pediatric and adolescent patients. Table 28.4 provides an overview of different staging classification systems and the MaGIC risk stratification system.
| Testis, ovarian, extragonadal COG staging a | Stage | Testis | Ovarian | Extragonadal |
|---|---|---|---|---|
| I | Complete resection: high inguinal or high ligation scrotal orchiectomy and negative nodes | Limited to ovary (peritoneal evaluation negative), no clinical, histological, or radiological evidence of disease outside the ovary b | Complete resection at any site with negative margins, or coccygectomy for sacrococcygeal teratoma | |
| II | Transscrotal biopsy, microscopic disease in the scrotum or cord, or failure of serum markers to normalize | Microscopic residual, peritoneal evaluation negative, failure of serum tumor markers to normalize | Microscopic residual, with lymph nodes negative | |
| III | Retroperitoneal lymph node involvement, but no visceral or extraabdominal involvement | Lymph node involvement, metastatic nodule, gross residual disease, or biopsy only, contiguous visceral involvement (omentum, intestine, and bladder), peritoneal evaluation positive for malignancy | Lymph node involvement, gross residual disease, or biopsy only | |
| IV | Distant metastasis, including liver | Distant metastasis, including liver | Distant metastasis, including liver |
| FIGO ovarian staging c | Stage | FIGO | ||
|---|---|---|---|---|
| I | Tumor confined to ovary | |||
| IA | Limited to one ovary, capsule intact, no tumor on surface, negative washings | |||
| IB | Both ovaries, otherwise like IA | |||
| IC1 | Surgical spill | |||
| IC2 | Capsule rupture before surgery, or tumor on ovarian surface | |||
| IC3 | Malignant cell in ascites or peritoneal washings | |||
| II | Both ovaries or extension lower than pelvic rim or peritoneal primary | |||
| IIA | Extension and/or implant on uterus and/or fallopian tubes | |||
| IIB | Extension to other pelvic intraperitoneal structures | |||
| III | Positive RP LN and/or microscopic metastasis beyond pelvis | |||
| IIIA1 | Positive RP LN only (IIIA1i 10 mm) (IIIA1ii 10 mm) | |||
| IIIA2 | Microscopic, extrapelvic (higher than pelvic brim), peritoneal involvement with or without positive RP LN | |||
| IIIB | Macroscopic, extrapelvic peritoneal metastasis 2 cm with or without RP LN includes extension to capsule of liver/spleen | |||
| IIIC | Macroscopic, extrapelvic peritoneal metastasis 2 cm with or without RP LN includes extension to capsule of liver/spleen | |||
| IV | Distant metastasis | |||
| IVA | Pleural effusion with positive cytologic features | |||
| IVB | Metastasis to liver/spleen parenchyma and extraabdominal organs (including inguinal LN and outside abdominal cavity) | |||
| International germ cell cancer collaborative prognosis system d | Histologic feature | Good | Intermediate | Poor |
|---|---|---|---|---|
| Seminoma | The absence of nonpulmonary visceral metastasis e | The presence of nonpulmonary visceral metastasis e | Not applicable | |
| Nonseminoma | Meets all the next criteria: | Meets all the next criteria | Meets all the next criteria | |
| Gonadal or RP primary site | Gonadal or RP primary site | Mediastinal primary site | ||
| The absence of nonpulmonary visceral metastasis | The absence of nonpulmonary visceral metastasis | The presence of nonpulmonary visceral metastasis | ||
| (S0) or S1 STM | S2 STM | S3 STM | ||
| HCG<5000 IU/mL | HCG≥5000 and ≤50,000 IU/mL L | HCG>50,000 IU/mL | ||
| AFP <1000 ng/mL | AFP≥1000 and ≤10,000 ng/mL | AFP >10,000 ng/mL | ||
| LDH<1.5×ULN | LDH≥1.5×ULN and ≤10 ULN | LDH>10×ULN |
| MaGIC risk stratificat ion f | Risk group | Site | COG stage | Age (years) |
|---|---|---|---|---|
| Low risk | Testis | I | Any | |
| Ovary | I | Any | ||
| Extragonadal | I | Any | ||
| Standard risk 1 | Testis | II–IV | <11 | |
| Ovary | II–IV | <11 | ||
| Extragonadal | II–IV | <11 | ||
| Standard risk 2 | Testis (IGCCC good risk) | II–IV | >11 | |
| Ovary | II–IV | >11 | ||
| Extragonadal | II | >11 | ||
| Poor risk | Testis (IGCCC intermediate/poor risk) | II–IV | >11 | |
| Ovary | IV | >11 | ||
| Extragonadal | III–IV | >11 |
a Adapted from Olson et al. (2015) .
b The presence of gliomatosis peritonei does not result in a stage change.
e Nonpulmonary visceral metastasis refers to metastasis involving organs other than lung (lymph nodes do not count).
Due to the heterogeneity in the biology of GCTs, the therapeutic strategies are stratified to address the different biological behaviors. The management of teratomas, germinomas, and nongerminomatous GCT differs and, thus, will be presented separately.
Pathologically, teratomas can be divided into two main groups:
These tumors contain mature tissue, and the treatment is surgical excision, irrespective of the site. It is critically important that multiple histologic sections be examined to exclude the presence of focal immature tissue and malignant germ cell elements.
These tumors contain elements of immature tissue and are graded histologically ( Table 28.5 ). The treatment of choice is surgical resection. Most immature teratomas in prepubertal children do not respond to chemotherapy. However, if AFP is increased, a malignant germ cell component is present and in addition to surgery, chemotherapy should be administered.
| Grade | Description |
|---|---|
| 0 | All tissue mature; no embryonal tissue |
| 1 | Rare foci of embryonal tissue not exceeding 1 low-power field per slide |
| 2 | Moderate quantity of embryonal tissue and some atypia but not exceeding 3 lower power fields per slide |
| 3 | Large quantity of immature tissue exceeding 3 lower power fields per slide, with abundant mitoses and cellular atypia |
These tumors contain foci of frankly malignant tissue usually EC, YST, or CC. These lesions should be treated in an aggressive multimodal fashion. Chemotherapy improves local and metastatic control. The benign teratomatous portion will not respond to chemotherapy or radiation therapy and must be treated surgically. See the section on the treatment of malignant GCT.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here