Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Epithelial tumors, the most common neoplasms of the ovary, encompass five distinct subtypes. Differentiation into these epithelial cell types is under the control of the same genes that determine different epithelial lineages in the female reproductive tract during embryonic development (e.g., mucinous epithelium in the endocervix, endometrioid epithelium in the endometrium, and serous or tubal epithelium in the fallopian tube). Ovarian epithelial tumors are further subclassified into benign, borderline (low malignant potential, atypical proliferating) or malignant on the basis of their histologic features. Benign, borderline, and malignant subclasses account for 60%, 5%, and 35% of all the epithelial tumors, respectively. They have been historically considered to arise from the surface epithelium of the ovary or its derivatives, but it is likely that most arise from the fallopian tube epithelium or endometriosis.
The division of the various epithelial subtypes into benign, borderline, and malignant forms is based on histologic features that correlate with biologic behavior. By definition, borderline tumors lack destructive stromal invasion but have other histologic features of malignancy (e.g., nuclear atypia, cellular stratification, and mitotic activity). For the most part, the clinical behavior of these histologically intermediate tumors is benign, with the exception of serous borderline tumors, which are frequently associated with involvement of the ovarian surface and contralateral ovary as well as extraovarian spread. They exhibit a clinical behavior intermediate between benign serous tumors and invasive serous carcinomas.
The overtly malignant epithelial tumors (carcinomas) comprise 85% of all malignant ovarian neoplasms and are the fourth most common cause of cancer death in women in the US. Approximately 1 in 70 women in industrialized countries develop ovarian cancer and 1 in 100 die of it. The incidence is lower in nonindustrialized nations, possibly due to decreased dietary fat intake. Risk factors include nulliparity, infertility, late child-bearing, and delayed menopause, whereas oral contraceptive use, breast-feeding, and tubal ligation are protective. A personal or family history of endometrial, breast, or colon cancer is also associated with increased risk, as is inherited germline mutations in the autosomal-dominant cancer susceptibility genes BRCA1 / BRCA2 or in one of the DNA base mismatch repair genes, the most frequent of which are MLH1 , MSH6 , PMS2 , and MSH2 (Lynch syndrome).
The most important factor in determining the prognosis of patients with epithelial carcinoma is tumor stage. The widely used International Federation of Gynecology and Obstetrics (FIGO) staging system, which unifies staging for ovarian, fallopian tube, and peritoneal malignancies, is shown in Table 14.1 . Stage can only be assigned after completion of a proper surgical staging procedure. Prognosis for patients with low-grade, stage IA tumors (i.e., tumor confined to one ovary, without ovarian surface involvement or extraovarian spread) is excellent, with >90% being disease free at 5 years. Unfortunately, most patients with ovarian cancer present with advanced-stage disease, and in these patients, the most important prognostic factor is whether or not the extraovarian disease is resectable; if all macroscopic disease can be surgically removed, there is >40% 5-year disease-free survival compared with <20% 5-year disease-free survival for patients with macroscopic residual disease after maximal surgical debulking. Current screening modalities for the early detection of ovarian cancer (including ultrasound and serum tumor marker assays) have been not nearly as effective as those for cervical, breast, or colon cancer. The development of an adequate screening program for early ovarian cancer is especially challenging due to the apparent absence of a long latent or in situ phase; the most common forms of epithelial carcinoma give rise to early and often extensive spread to extraovarian sites, resulting in the rapid development of nonresectable disease.
| Stage | Description |
|---|---|
| Stage I Stage IA Stage IB Stage IC |
Tumor confined to the ovaries or fallopian tubes Tumor limited to one ovary (capsule intact) or fallopian tube; no tumor on ovarian or fallopian tube surface; no malignant cells in the ascites or peritoneal washings. Growth limited to both ovaries. Otherwise like stage 1A. IC1: Intraoperative spill IC2: Capsule rupture before surgery or tumor on ovarian or fallopian tube surface IC3: Positive peritoneal washings or ascites |
| Stage II Stage IIA Stage IIB |
Tumor involves one or both ovaries or fallopian tubes with pelvic extension or primary peritoneal cancer Extension to or implant on uterus or fallopian tubes, or some combination thereof Extension to other pelvic intraperitoneal tissues |
| Stage III Stage IIIA Stage IIIB Stage IIIC |
Tumor involves one or both ovaries or fallopian tubes with involvement of the peritoneum outside the pelvis, metastasis to the retroperitoneal lymph nodes, or both. IIIA1(i): Metastasis of ≤10 mm to the retroperitoneal lymph nodes IIIA1(ii): Metastasis of >10 mm to the retroperitoneal lymph nodes IIIA2: Microscopic, extrapelvic peritoneal involvement (above the brim) with or without involvement of retroperitoneal lymph nodes Macroscopic, extrapelvic, peritoneal metastasis ≤2 cm with or without involvement of retroperitoneal lymph nodes (includes extension to capsule of liver or spleen Macroscopic, extrapelvic, peritoneal metastasis >2 cm with or without involvement of retroperitoneal lymph nodes (includes extension to capsule of liver or spleen |
| Stage IV Stage IVA Stage IVB |
Distant metastasis excluding peritoneal metastases Pleural effusion with positive cytology Distant metastasis including parenchymal metastasis to liver, spleen, or extraabdominal organs |
Ovarian epithelial carcinomas are subclassified according to cell type, and tumors of different cell types have different prognoses and show different rates of response to adjuvant chemotherapy. Assigning tumor grade is not uniform among these subtypes. Serous ovarian carcinomas are now classified as either low grade or high grade and are not otherwise further graded. Clear cell carcinoma is not graded. Endometrioid carcinoma is graded following Silverberg or FIGO systems (the latter being identical to grading of the endometrium). Most patients with epithelial carcinomas are treated by surgery, followed by adjuvant chemotherapy. The surgical management usually consists of abdominal hysterectomy, bilateral salpingo-oophorectomy, omentectomy, and removal of as much tumor as technically feasible. First-line chemotherapy typically consists of combination of platinum and taxane. Chemotherapy is not routinely given to patients with borderline tumors or low-grade serous carcinoma. The relative frequency of the main histologic subtypes of epithelial ovarian tumors, including the undifferentiated and mixed tumors, is provided in Table 14.2 .
| Histologic Type | Total (%) |
|---|---|
| Serous | 46 |
| Mucinous | 36 |
| Endometrioid | 8 |
| Clear | 3 |
| Undifferentiated | 2 |
| Mixed | 3 |
a Tumors with squamous differentiation have also historically been included among the surface epithelial stromal tumors, but pure squamous tumors are rare, as most arise in a teratoma.
Tumors with serous differentiation, representing 46% of all epithelial ovarian neoplasms, are characterized by epithelial cells resembling those of fallopian tube and encompass a group of four biologically distinct entities, including serous cystadenoma/adenofibroma, serous borderline tumor (tumor of low malignant potential, atypical serous proliferative tumor), low-grade serous carcinoma, and high-grade serous carcinoma ( Table 14.3 ). The distinction between low-grade serous carcinoma and high-grade serous carcinoma is based primarily on cytologic features and (less so) on mitotic index ( Table 14.4 ). Serous borderline tumors have the capacity for extraovarian spread, recurrence, and death, even though the tempo of disease progression is significantly more indolent when compared to low-grade and high-grade serous carcinoma.
| Low Grade | High Grade | |
|---|---|---|
| Nuclei | Uniform, round to oval nuclei | Pleomorphic nuclei (>3:1 variability) |
| Chromatin | Evenly distributed | Irregular distribution |
| Mitotic count a | ≤12 mitotic figures/10 high power fields | >12 mitotic figures/10 high power fields |
Benign serous tumors (cystadenoma, cystadenofibroma, adenofibroma, surface papilloma) comprise 50% of all serous ovarian neoplasms. They are almost always confined to the ovary.
Benign serous tumors occur over a wide age range but are most common in the reproductive age group. They are often bilateral. Women may present with abdominal enlargement, pain or vaginal bleeding, but most patients with benign tumors are asymptomatic and the tumors are discovered incidentally during routine examination or evaluation of an unrelated condition.
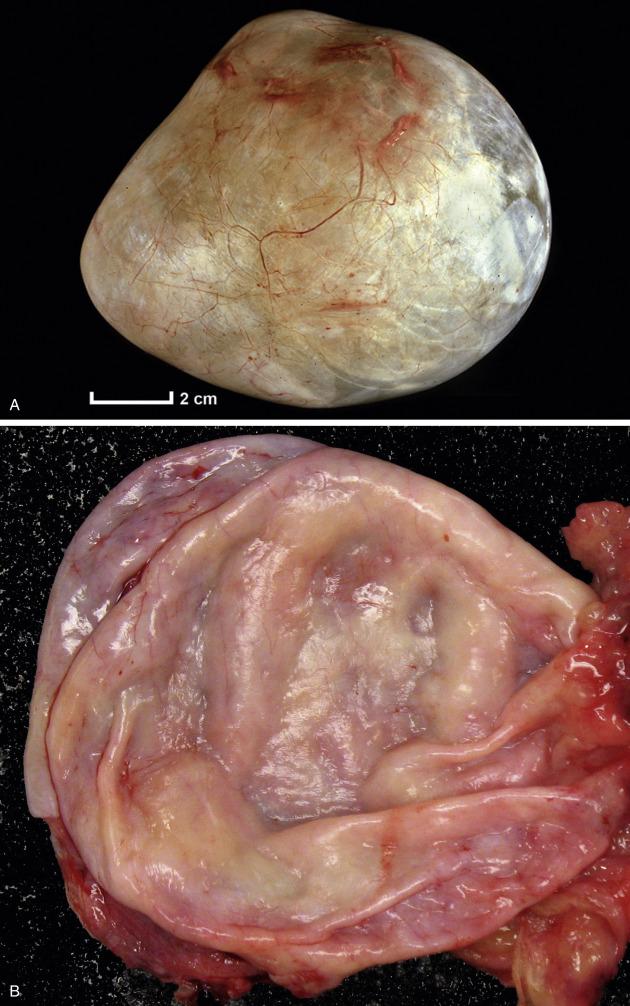
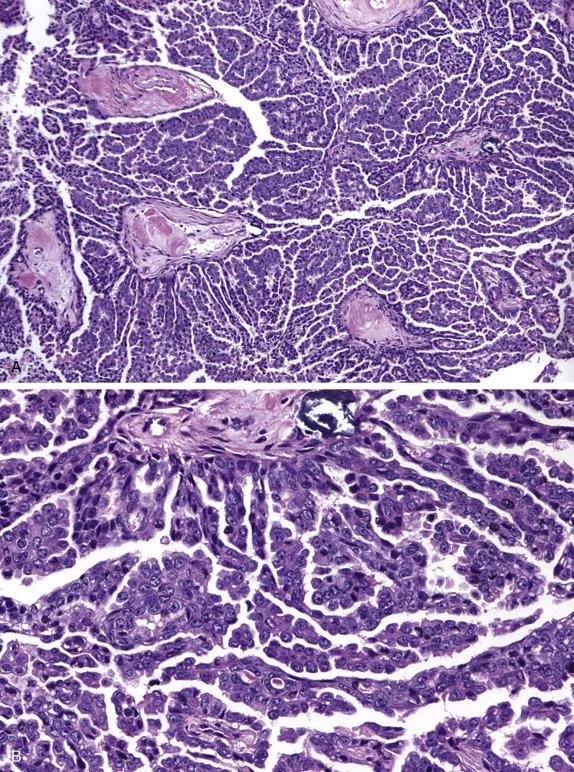
Benign serous tumors, frequently bilateral and metachronous (especially in older women), are composed of varying amounts of fibrous stroma (when predominant, serous adenofibroma) and cysts (serous cystadenoma or cystadenofibroma) and range in size from 1 to 10 (rarely up to 30) cm. The cysts are unilocular or multilocular and may contain papillary projections ( Fig. 14.1 ). On occasion, the tumor grows as an excrescence on the ovarian surface (so-called “surface papilloma”).
Benign serous tumors are lined by epithelium that closely recapitulates the ciliated and secretory cells of the fallopian tube ( Fig. 14.2 ). Varying amounts of fibromatous stroma (adenofibroma, cystadenofibroma) and cystic change (cystadenofibroma) are present. Serous surface papillomas are composed of small papillary growths lined by simple bland, serous-type epithelium growing on the surface of the ovary.
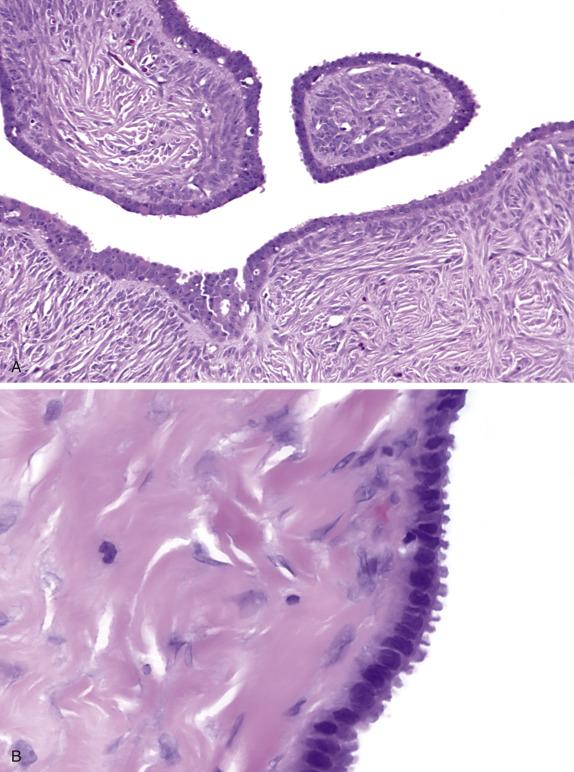
Benign serous tumors have an immunohistochemical profile that is similar to borderline and malignant serous tumors. Like many other ovarian epithelial tumors, they show a CK7-positive, CK20-negative phenotype. In addition, they express PAX8, WT1, ER, BerEP4, and CD15 (Leu-M1).
Serous cystadenomas are (rather arbitrarily) distinguished from serous epithelial inclusion cysts on the basis of size (>1 cm). Distinction from rete cysts is made on the basis of location (rete cysts are hilar), presence of prominent cilia, and absence of a smooth muscle layer and hyperplastic hilar cells. An endometriotic cyst , especially in postmenopausal women, can closely mimic a serous cystadenoma. The presence of endometrial-type stroma, at least focally, helps in this distinction. If cystic, struma ovarii may resemble a serous cystadenoma, but the constituent cysts of struma contain colloid and are immunoreactive for thyroglobulin. The chief differential diagnosis is between benign and borderline serous tumors. Borderline tumors are characterized by epithelial proliferation in the form of stratification, hierarchical papillary architecture, tufting with detached epithelial cell clusters, cytologic atypia, and occasional mitotic figures. By definition, serous tumors with focal borderline change (<10% of the tumor epithelium) in an otherwise benign tumor are designated serous cystadenoma (or cystadenofibroma) with “focal proliferation.” These have an indolent behavior. Occasionally, serous tumors can secrete material resembling mucin and thus resemble a mucinous cystadenoma , but its constituent cells do not contain intracytoplasmic mucin (PAS-diastase or mucicarmine negative).
Serous cystadenomas, adenofibromas, cystadenofibromas, and surface papillomas are clinically benign. Indeed, distinction between these categories of benign serous neoplasia does not have clinical significance. Complete excision (cystectomy or oophorectomy) is curative. To minimize surgical morbidity, laparoscopic removal is often performed. Occasionally, similar tumors in extraovarian sites accompany benign serous ovarian tumors.
Benign ovarian tumor composed of tubal-type epithelium and varying amounts of stroma
If predominantly cystic: cystadenoma
If prominent stromal component without grossly visible cysts: adenofibroma
If prominent stromal component with grossly visible cysts: cystadenofibroma
Most common benign epithelial tumor
Bilateral (20%)
No significant morbidity or mortality unless torsion, rupture, or infection
Wide age range including premenarchal to postmenopausal women, but predominantly reproductive age
Often asymptomatic, incidental finding
Benign tumors
Simple excision (cystectomy or adnexectomy) is curative
Simple, smooth-walled unilocular or multilocular cyst with varying amounts of fibromatous stroma (often <4 cm)
Solid firm areas may be present (fibromatous component)
Necrosis absent, unless complicated by torsion, infection, or infarction
Simple architecture; nonbranching papillae (if present) with rare to absent tufting
Single, orderly layer of nonstratified, cuboidal to columnar epithelium, often ciliated
Nuclear atypia minimal or absent, and rare mitoses
Surface papilloma: simple papillae growing on ovarian surface
CK7, EMA, PAX-8, ER, and WT1 positive
CK20 negative
Epithelial inclusion cyst
Rete cyst
Endometriotic cyst
Struma ovarii
Serous tumor with focal proliferation
Serous borderline tumor
Mucinous cystadenoma
Serous borderline tumors (serous tumor of low malignant potential, atypical proliferative serous tumor) comprise approximately 15% of all ovarian serous tumors and account for the vast majority of all borderline ovarian epithelial neoplasms.
These tumors are more often bilateral and larger than benign serous tumors. They occur at a slightly younger age than serous carcinoma (mean 45 vs. 60 years) and may present with disease beyond the confines of the ovary. Patients are often asymptomatic but may have abdominal enlargement or pain. Younger women often present with infertility. A significant minority of women are pregnant at the time of diagnosis.
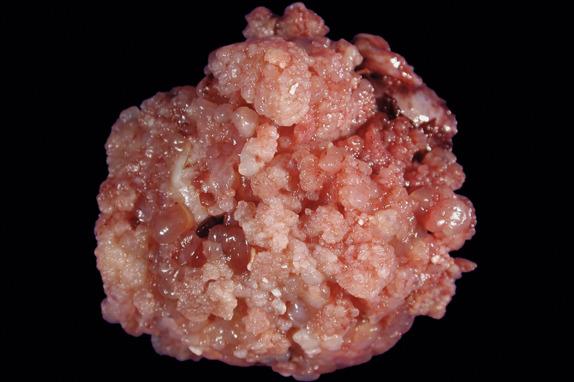
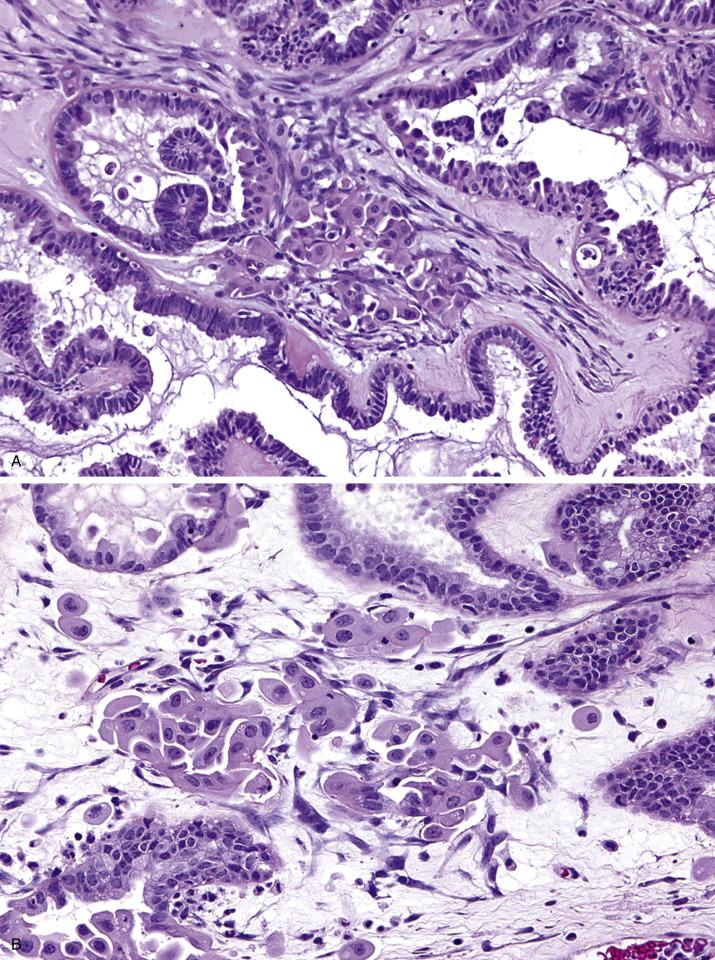
Serous borderline tumors are predominantly cystic with variable amounts of papillary epithelial projections that are often soft and delicate ( Fig. 14.3 ) or solid with surface papillary excrescences (or combination thereof).
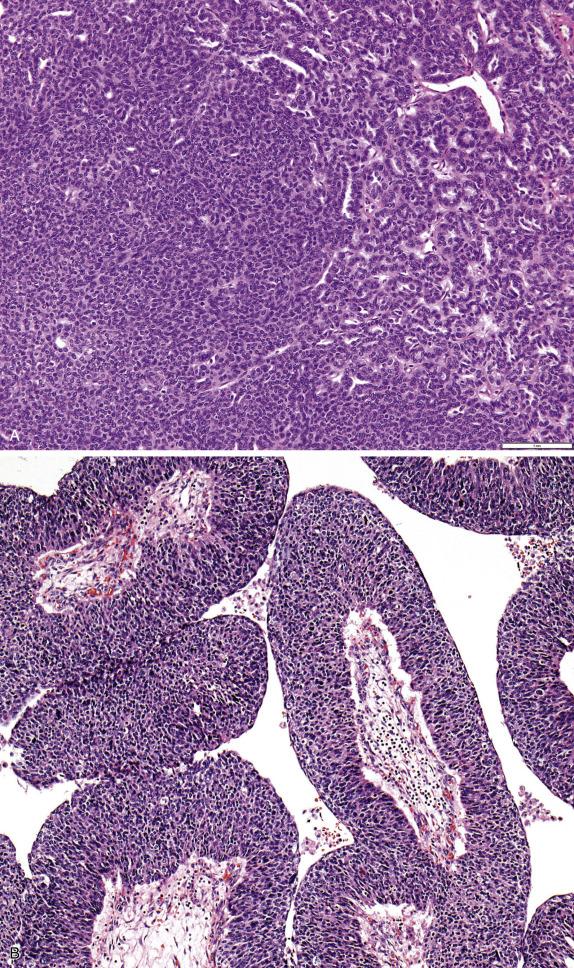
Serous borderline tumors are characterized by proliferation of the lining epithelium seen as stratification and complex hierarchical branching papillae associated with detached cellular buds. Destructive invasion of the ovarian stroma is not seen. The lining cells are cuboidal to columnar and often ciliated with interspersed eosinophilic (pink) cells, although cilia may not be conspicuous in some cases. The nuclei are uniform and mildly to moderately atypical, and mitotic rate is low. Psammoma bodies may be present but are not diagnostic ( Fig. 14.4 ).
Approximately 5%–10% of all serous borderline tumors contain foci with micropapillary architecture ( Fig. 14.5 ), defined as either nonhierarchical branching of slender, elongated papillae that are at least five times as long as they are wide or a sieve-like cribriform pattern in the surface of the papillae or within glands. When this pattern occupies a continuous area of 5 mm or more, the tumor is designated as serous borderline tumor, micropapillary variant (also referred to as “noninvasive low-grade serous carcinoma”), which is more frequently associated with bilaterality, ovarian surface involvement, and presence of extraovarian implants, a higher proportion of which are invasive.
Foci of stromal microinvasion ( Fig. 14.6 ), defined as individual eosinophilic cells, small cell clusters, or papillae (more frequently small) within the stroma, are found in 10%–15% of serous borderline tumors ( Box 14.1 ). By definition, foci are ≤5 mm in linear extent. The stroma around the epithelial cells/clusters/papillae often retracts, forming a “space.” The tumor cells in microinvasion have more abundant eosinophilic cytoplasm and acquire a round or polygonal shape. However, the nuclei have, at most, mild atypia and are similar to the intraepithelial cells. Stromal microinvasion is more often seen in pregnant women. Although it may be associated with increased long-term risk of disease recurrence, the presence of stromal microinvasion does not warrant a diagnosis of carcinoma. A less common scenario is the presence of microinvasion in the micropapillary variant of serous borderline tumor that has the appearance of a low-grade serous carcinoma but measures ≤5 mm in linear extent. It is unclear what the biologic significance of this finding is; however, some have suggested designating these foci as “microinvasive carcinoma” to separate it from microinvasion as described earlier.
∗ Individual foci of microinvasion must not exceed 5 mm in linear extent.
Individual cells or small, irregular clusters of cells with abundant eosinophilic cytoplasm within the stromal stalks of serous papillae, commonly surrounded by a tissue space or cleft. The pink cells often have enlarged nuclei with distinct nucleoli.
Small aggregates of simple and branching papillae, often composed of eosinophilic cells surrounded by a tissue space or cleft.
Micropapillae, macropapillae, or cribriform glands lined by serous borderline epithelium surrounded by a cleft. This is an uncommon form of microinvasion.
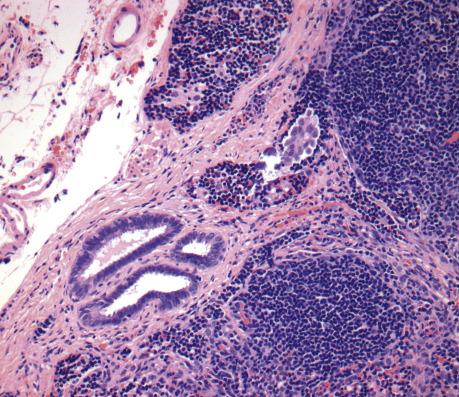
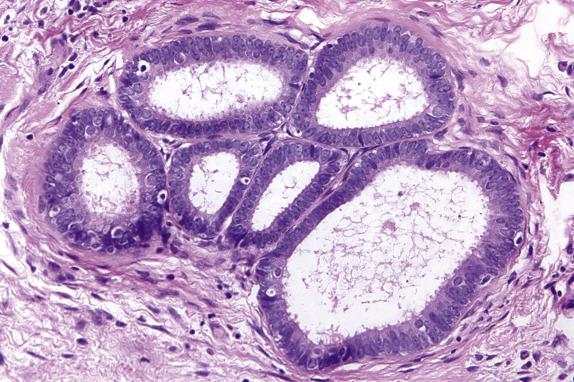
Approximately 30%–40% of serous tumors in the borderline group are associated with similar-appearing lesions in the pelvis and intraabdominal sites, including lymph nodes (high-stage disease) ( Fig. 14.7 ). These lesions, termed implants, may be microscopic or macroscopic and are subclassified into noninvasive ( Fig. 14.8A ,B) and invasive ( Figs. 14.9 and 14.10 ) types, based on the presence of destructive infiltration into underlying normal tissue ( Table 14.5 ). Noninvasive epithelial implants are exclusively composed of glands or cysts with a variable degree of papillary formation present on the peritoneal surfaces. The interface between the implant and surrounding tissue is typically smooth. Noninvasive desmoplastic-type implants may occur over the ovarian surface or between exophytic surface papillae (autoimplants), as well as on the peritoneal surfaces. They form plaques or nodules without associated invasion of underlying organs/tissues and thus are easily removed. They are characterized by prominent desmoplastic reaction and inflammation surrounding single cells or irregular nests of tumor cells, which typically represent a minor component of the lesion. In both types of noninvasive implants, the cytologic features should be similar to those seen in the primary ovarian serous borderline tumor. Invasive implants may also be associated with a desmoplastic response but typically show destructive invasion of underlying tissue in the form of irregularly shaped or confluent glands or tumor cell clusters and often the cells show cytologic features of low-grade carcinoma. Borderline tumors with invasive implants are associated with an increased risk of recurrence and a more aggressive clinical course; thus, these implants are now considered to be low-grade serous carcinoma. Since both types of implants may feature a prominent stromal connective tissue response, occasional implants may be especially difficult to subclassify; in these instances, the implants should probably be classified as indeterminate. Noninvasive implants may appear invasive if they seed the peritoneal surface between omental fat lobules or serosal infoldings, so to determine invasion, it is important to examine the overall distribution (separating fat lobules vs. infiltrating adipose tissue) and cytomorphology of the epithelium (atypia, loss of well-developed gland formation). The presence of micropapillary morphology is considered by some investigators as evidence of invasion in an implant ( Fig. 14.10A ). Rarely, extraovarian invasion by single cells with abundant eosinophilic cytoplasm may be seen ( Fig. 14.10B ). Foci of endosalpingiosis involving extraovarian peritoneum and lymph nodes are common in women with serous borderline tumors. These foci, characterized by simple glands lined by tubal-type epithelium, should be distinguished from extraovarian borderline implants, since the presence of endosalpingiosis does not warrant upstaging of an accompanying ovarian serous borderline tumor ( Fig. 14.11 ).
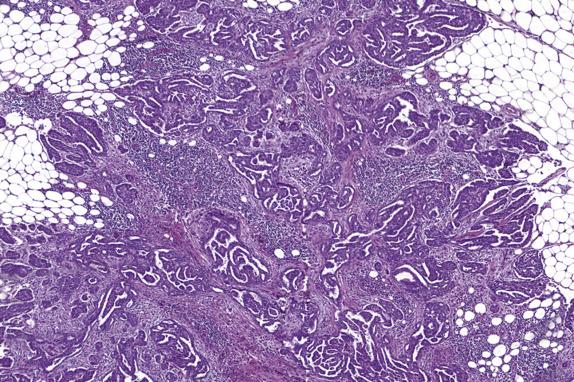
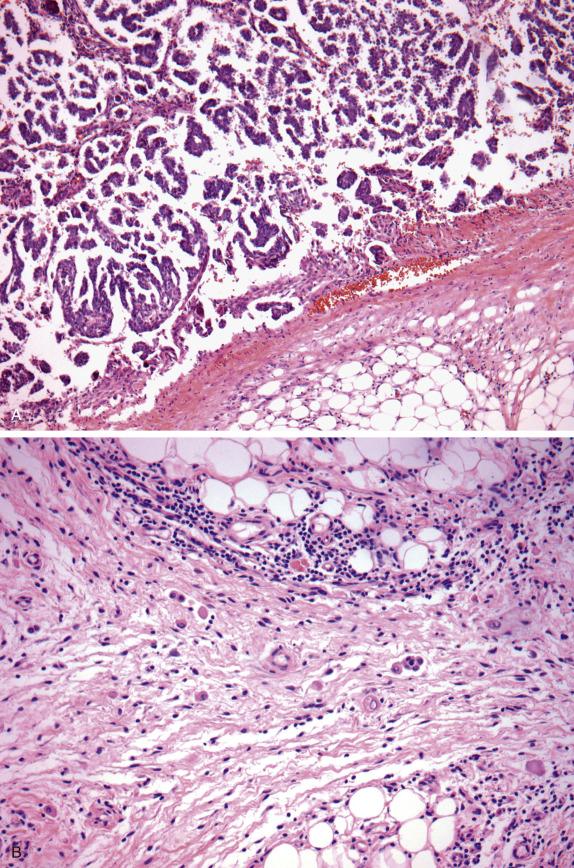
| Diagnostic Features | |
|---|---|
| Endosalpingiosis | Small, ciliated tubular glands, may have simple papillary infoldings, no atypia, smooth epithelial–stromal interface |
| Noninvasive implants Epithelial implants Desmoplastic implants |
Branching papillae, glands with complex papillary infoldings, single cell and small cell clusters, mild to moderate atypia, smooth interface with underlying/surrounding normal tissue Branching papillae, glands with complex papillary infoldings, single cell and small cell clusters, mild to moderate atypia, desmoplastic fasciitis-like stroma, smooth interface with underlying/surrounding normal tissue. |
| Invasive implants (= low-grade serous carcinoma) b | Branching papillae, glands with complex papillary and micropapillary infoldings, single cell and small cell clusters, with abundant epithelial component, moderate to carcinomatous atypia, jagged and irregular interface with underlying/surrounding normal tissue. |
a Implants should be distinguished from endosalpingiosis (which is a benign process and does not warrant upstaging of an accompanying ovarian serous borderline tumor) and are classified as invasive (low-grade serous carcinoma) or noninvasive on the basis of the interface between the implant and normal tissue. Implants with no attached normal tissue (so-called detached implants) are classified as noninvasive (either epithelial or desmoplastic) provided the atypia is moderate at most and the architecture is not overtly complex. When marked cytologic and architectural atypia is present, the implant should probably be classified as invasive (low-grade serous carcinoma) or, alternatively, as indeterminate since there is no evaluable interface. Implants in which it is difficult to determine whether the interface is smooth and expansile or focally irregular should also be classified as indeterminate.
Positive peritoneal cytologic preparations are characterized by papillary aggregates and/or tight clusters of cells with minimal nuclear atypia and can be difficult to distinguish from benign serous epithelium (endosalpingiosis), reactive mesothelial proliferations, and well-differentiated serous adenocarcinomas. Since further distinction cannot be made on cytologic ground alone, reporting of the previous findings as “serous neoplasm” is prudent. Psammoma bodies may be present but by themselves are not diagnostic. Fine-needle aspiration is only indicated for confirmation of suspected recurrent disease.
Serous borderline tumors express an immunohistochemical profile that is similar to benign and malignant serous tumors. They are usually CK7 positive and CK20 negative and additionally express PAX8, WT1, ER, BerEP4, and CD15 (Leu-M1).
The profile of mRNA expression patterns differs for serous borderline tumors and low-grade serous carcinomas, as well as for low-grade serous carcinomas and high-grade serous carcinomas. The pattern of genetic alterations also differs; e.g., the presence of point mutations in BRAF or (less commonly) KRAS is more frequently associated with borderline tumors, whereas KRAS and NRAS appear to be more frequent in low-grade serous carcinomas, and TP53 mutation and somatic or germline abnormalities of BRCA1 and/or BRCA2 are more frequently associated with high-grade serous carcinoma.
Borderline serous tumors are distinguished from benign serous tumors on the basis of epithelial proliferation. By definition, serous tumors featuring ≤10% proliferation of borderline type are designated as serous tumors with “focal proliferation.” Low-grade serous carcinoma is distinguished from serous borderline tumor by the presence of destructive stromal invasion (>5 mm). Well-differentiated papillary mesothelial tumor may mimic a surface ovarian (or extraovarian) serous borderline tumor and is distinguished by the absence of hierarchical branching of the papillae, prominent papillary connective tissue cores lined by cuboidal cells with less pseudostratification, strong calretinin staining, and lack of staining for BerEP4 and CD15. Seromucinous borderline tumors are distinguished from serous borderline tumors by the presence of both serous and mucinous endocervical-like epithelial cells, abundant neutrophils in large edematous papillae, and their more frequent association with endometriosis. Struma ovarii with cystic and papillary architecture may resemble a serous borderline tumor, but more classical areas with thyroid follicles and immunoreactivity for thyroglobulin are helpful in this differential diagnosis. Retiform Sertoli–Leydig cell tumor may simulate a serous borderline tumor; however, the papillae are typically edematous and lack branching and cell budding. Moreover, this tumor typically has distinctive features of sex cord stromal differentiation elsewhere, provided there is adequate tumor sampling.
Even in high-stage tumors, the disease tempo in women with serous borderline tumors is characteristically indolent and protracted. Prolonged periods of dormancy and even spontaneous regression may occur. However, there is a significant risk of progression to invasive low-grade serous carcinoma ( Fig. 14.12 ), which occurs in up to 7% of women, occasionally decades after initial diagnosis and is associated with increased tempo of disease and a significantly more aggressive disease course. For patients with stage I disease initially, the risk of progression is higher in those with micropapillary serous borderline tumor/noninvasive low-grade serous carcinoma (5% at 5 years, 13.9% at 20 years) compared with those with conventional serous borderline tumor (0.9% at 5 years, 3.7% at 20 years). In patients with advanced-stage disease, noninvasive implants have a risk of progression to carcinoma (hazard ratio [HR] = 7.7, 95% confidence interval [CI] = 3.9–15); invasive implants carry a significantly higher risk (HR = 42.3, 95% CI, 16–111), hence their denomination as low-grade serous carcinoma. High-grade serous carcinoma may arise during tumor progression, but this is uncommon. Stromal microinvasion and lymph node involvement do not appear to increase the risk of recurrence or progression. In a meta-analysis of studies of serous borderline tumors, disease-specific survival at 5 years was >95% for patients with low-stage (stage I) tumors and approximately 65% for patients with high-stage (stage II–IV) tumors. Since late recurrences do occur, and follow-up is limited in many of the studies analyzed, these are conservative estimates of risk of recurrence.
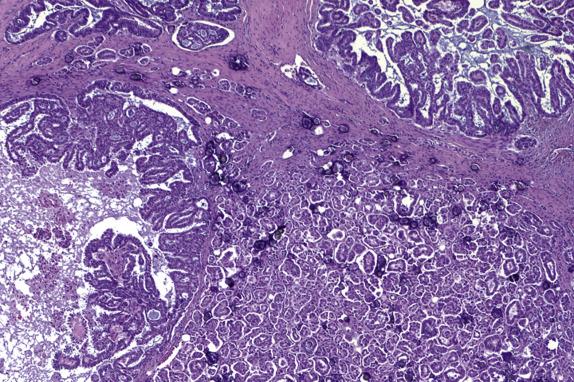
The management of serous borderline tumors is primarily surgical. Ovarian and uterine conservative surgery can be considered, especially in young, reproductive-aged women, although initial staging surgery is required to accurately determine the extent of disease and exclude the presence of invasive implants. Currently, there is no indication for adjuvant chemotherapy or radiation therapy for women with serous borderline tumors who have disease confined to the ovary or noninvasive extraovarian implants only. Patients with invasive implants (low-grade serous carcinoma) show poor response rates to traditional ovarian carcinoma chemotherapy.
Epithelial tumor composed of hierarchically branching papillae with cellular stratification and tufting, lined by tubal-type epithelium showing limited cytologic atypia
Bilateral (30%–40%)
Advanced stage (30%–40%)
Prolonged, indolent course provided extraovarian implants, when present, are noninvasive
Invasive extraovarian implants are diagosed as low-grade serous carcinoma and confer a comparatively poor prognosis
5-year disease-specific survival 90%–95% for stage I, 65% for stage II-IV patients
Most common in fourth and fifth decades (mean age 46 years)
Often asymptomatic
Abdominal enlargement or pain secondary to torsion or rupture
Elevated serum CA125
Cystectomy or adnexectomy with preservation of uterus and remaining ovarian tissue in reproductive-age women, even when high-stage disease, provided extraovarian implants are noninvasive. However, recurrence in ipsilateral or contralateral ovary common and follow-up required
In patients with stage I disease, micropapillary growth carries a higher risk of progression to invasive carcinoma
Invasive implants (low-grade serous carcinoma) (<15% of advanced-stage cases) associated with poorer prognosis
Transformation to low-grade serous carcinoma or high-grade carcinoma (<10%) associated with increased tempo of disease and poor prognosis
No proven efficacy of adjuvant chemotherapy
Large cystic or solid and cystic mass with soft papillary projections and/or surface papillary excrescences
Numerous papillae, often broad and edematous, with complex, typically hierarchical branching
Cuboidal to columnar epithelium, often ciliated with interspersed eosinophilic (pink) cells with cellular stratification and tufting
Mild to focally moderate nuclear atypia and sparse mitoses
Psammoma bodies
Micropapillary variant: elongated papillae with nonhierarchical branching (at least five times long as they are wide and at least 5 mm in continuous linear extent) or cribriform growth
Stromal microinvasion (≤5 mm in linear extent) may be present (10%)
Extraovarian peritoneal and lymph node implants may be present
CK7, EMA, WT1, PAX-8, ER, BerEP4, CA125, and CD15 positive
CK20 negative
BRAF or KRAS mutations
No association with germline or somatic BRCA1 / BRCA2 mutations
Benign serous tumor with focal proliferation
Serous carcinoma
Well-differentiated papillary mesothelial tumor
Seromucinous borderline tumor
Struma ovarii
Retiform Sertoli–Leydig cell tumor
Low-grade serous carcinomas account for 5%–10% of all serous carcinomas. They are often associated with or preceded by a serous borderline tumor.
This disease is more common in the Western hemisphere. Most women with low-grade serous carcinoma are adults and tend to present approximately one decade earlier than those with high-grade serous carcinoma. Most are advanced stage. Presenting symptoms are often vague and generally attributable to enlarged, cystic ovarian masses, but the tumors may be detected incidentally. Imaging studies reveal cystic and nodular adnexal masses. Ascites and elevated CA125 levels may be present.
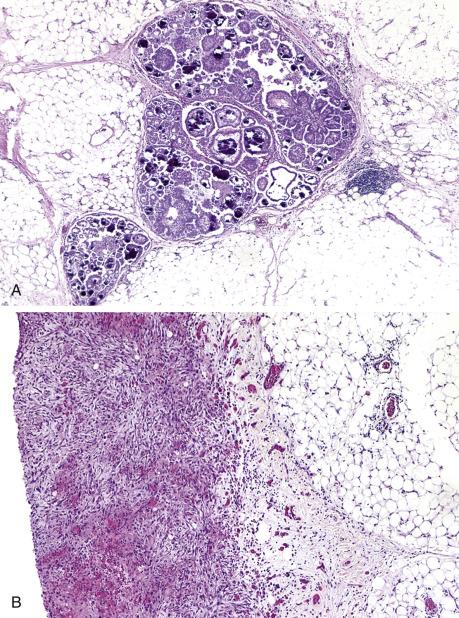
Low-grade serous carcinomas are typically cystic tumors composed of fine papillary growths ( Fig. 14.13 ). Two-thirds are bilateral, and there is often spread beyond the ovary at the time of diagnosis. Calcifications may be prominent both in the ovarian and the extraovarian lesions.
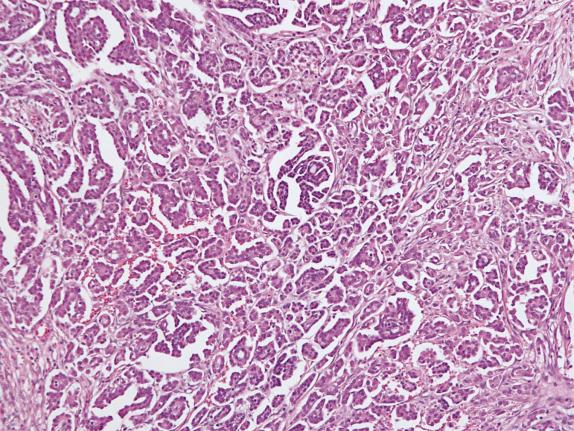
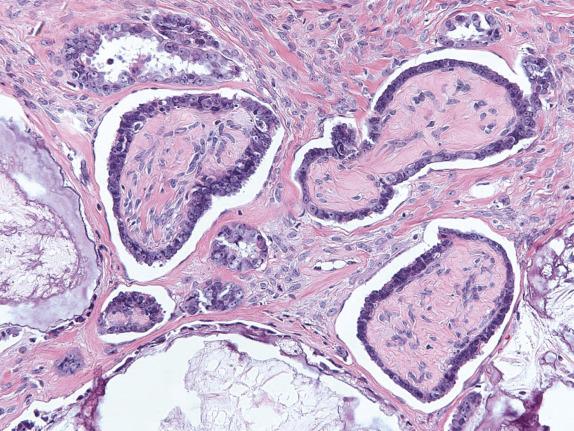
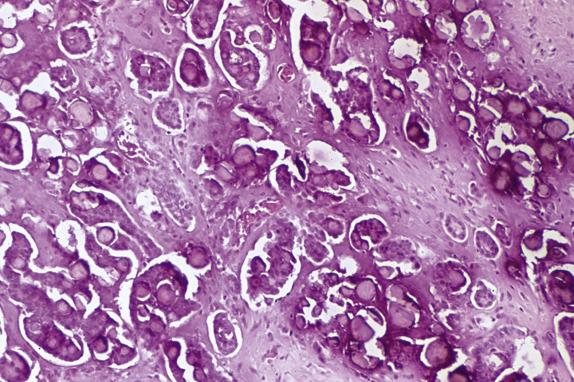
Low-grade serous carcinoma exhibits low-grade cytologic atypia with destructive invasion of the ovarian stroma. The lining cells are tall to columnar and may or may not display cilia. The nuclei are uniform, nuclear chromatin is evenly distributed, and mitotic activity is low (usually less than 12 mitotic figures per 10 high-power fields [HPFs]). Atypical mitotic figures are absent. A variety of patterns of destructive stromal invasion may be seen, including dense small aggregates of micropapillae ( Fig. 14.14 ), inverted macropapillae ( Fig. 14.15 ), and a mesothelioma-like pattern. Often, an extensive microinvasion-like pattern is seen, which may be deceiving. Psammoma bodies may be present; indeed, a variant of low-grade serous carcinoma with psammomatous calcifications representing ≥75% of the tumor volume has been formerly described under the term “psammomacarcinoma” ( Fig. 14.16 ). Approximately 80% of low-grade serous carcinomas are associated with similar-appearing lesions in the omentum and/or lymph nodes (high-stage disease), which can be microscopic or macroscopic. These need to be distinguished from endosalpingiosis and foci of epithelium with features of noninvasive implants of serous borderline tumor involving extraovarian peritoneum and lymph nodes, since these do not warrant upstaging of the accompanying ovarian low-grade serous carcinoma. Psammomacarcinoma, in particular, can present with extensive extraovarian spread and even as a predominant peritoneal mass.
Positive peritoneal cytologic preparations are virtually indistinguishable from serous borderline tumor.
Low-grade serous carcinomas express an immunohistochemical profile that is similar to other benign and malignant serous tumors. They are usually CK7 positive, CK20 negative, and express PAX8, WT1, BerEP4, ER, and CD15 (Leu-M1). Most low-grade serous carcinomas show normal p53 expression.
Compared with serous borderline tumors, low-grade serous carcinomas have a higher prevalence of KRAS and NRAS mutations and lower prevalence of BRAF mutations. Mutations in TP53 , BRCA1 , and BRCA2 are highly uncommon and likely do not play a role in the pathogenesis of low-grade serous ovarian neoplasia, unlike high-grade serous carcinoma.
Low-grade serous carcinomas are distinguished from serous borderline tumor by the presence of stromal invasion greater than 5 mm. In addition, carcinoma has a greater degree of cytologic atypia. Well-differentiated papillary mesothelial tumor may mimic a low-grade serous carcinoma and is distinguished by less hierarchical branching of the papillae, prominent papillary connective tissue cores lined by cuboidal cells with less pseudostratification, strong calretinin staining, and lack of staining for BerEP4 and CD15. Seromucinous borderline tumors are distinguished from low-grade serous carcinoma by the presence of both serous and mucinous endocervical-like epithelial cells, abundant neutrophils in large edematous papillae, absence of destructive stromal invasion, and their more frequent association with endometriosis. High-grade serous carcinoma is recognized on the basis of marked cytologic atypia with abnormal chromatin and numerous mitotic figures, including atypical division figures, as well as abnormal p53 staining. Retiform Sertoli–Leydig cell tumor lacks papillary branching and cell budding and usually has areas of conventional sex cord stromal differentiation.
Low-grade serous carcinoma is associated with a more prolonged disease course than that seen in high-grade serous carcinoma. Median survival is approximately 91 months versus 41 months for high-grade serous carcinoma. The 10-year survival for high-stage low-grade serous carcinoma is <50%. Patients with low-grade serous carcinoma do not respond well to traditional ovarian carcinoma chemotherapy, and primary surgical resection and subsequent debulking of recurrent disease are the mainstay of therapy.
Malignant epithelial ovarian tumor composed of serous (tubal-type) epithelium with low-grade cytologic atypia
Uncommon
Bilateral (60%)
10 year survival < 50%, if high stage
More common in Western hemisphere
Rare in first two decades of life, but steady increase thereafter
Pelvic or abdominal enlargement, vague pain, urinary or gastrointestinal symptoms
Ascites
Elevated serum CA125
Advanded stage (80%)
Complex cystic mass with irregular, thick cyst wall septations and solid mural nodules
Ascites
Psammomatous calcifications on X-ray
Excellent prognosis, if confined to ovary
85% 5-year and 50% 10-year survival, if advanced stage
Does not respond well to conventional platinum-based chemotherapy
Treatment mainly surgical (primary excision and subsequent debulking)
Large cystic or solid and cystic mass with soft papillary projections and/or surface papillary excrescences
Destructive stromal invasion (≥5 mm in linear extent in area) must be present as haphazard micropapillae, inverted macropapillae, or a mesothelioma-like pattern
Background serous borderline tumor often present
Uniform, round nuclei with moderate nuclear atypia and sparse mitoses (usually <12 mitotic figures per 10 high-power fields)
Psammoma bodies
Extraovarian peritoneal and lymph node disease may be present
CK7, EMA, WT1, PAX-8, ER, BerEP4, CA125, and CD15 positive
CK20 negative
Wild-type p53 expression
BRAF mutations less common than in serous borderline tumors; KRAS and NRAS mutations may be present
No association with TP53 , BRCA1 , or BRCA2 mutations
High-grade serous carcinoma
Well-differentiated papillary mesothelial tumor
Serous borderline tumor
Seromucinous borderline tumor
Retiform Sertoli–Leydig cell tumor
High-grade serous carcinomas account for more than 90% of all serous carcinomas. Although initially thought to arise in the ovary, these tumors frequently arise in the fallopian tube and spread secondarily to the ovary and peritoneum. In some cases, no fallopian tube involvement (mucosal or transmural) is identified, and assignment of primary site is based on whether there is disease in the ovaries (primary ovarian) or confined to the peritoneum (primary peritoneum). Müllerian tumors that occur in women with germline BRCA1 / BRCA2 mutations are usually high-grade serous carcinomas, which arise in the fallopian tube in most cases.
High-grade serous carcinoma is very uncommon in women younger than 20 years, but the incidence progressively increases with age, peaking in the sixth to seventh decades (mean 56 years). Most women present with advanced-stage disease. Symptoms are often vague and nonspecific and include dyspepsia, bloating, early satiety, backache or urinary symptoms. The most common finding is an adnexal, pelvic, or abdominal mass on pelvic examination or imaging studies. A subset of patients presents with sudden onset of severe abdominal pain, due to torsion of the ovarian mass. A variety of paraneoplastic syndromes have been reported, but these are rarely the presenting sign or symptom. Elevated serum CA125, although nonspecific, is present in >80% of patients.
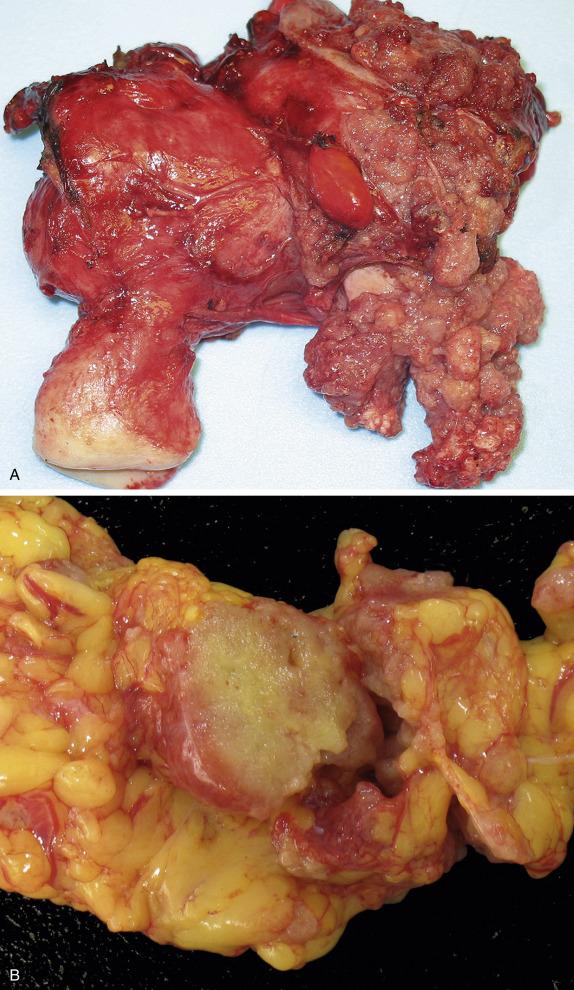
High-grade serous carcinomas are large tumors (usually >10 cm). The fallopian tube may be adherent to the mass or may be partially or completely embedded in it. The cut surface shows solid and cystic areas, both composed of friable tan tissue with hemorrhage and necrosis ( Fig. 14.17 ). Cystic spaces contain excrescences of soft and friable tumoral tissue. Surface involvement and bilateral ovarian disease is common.
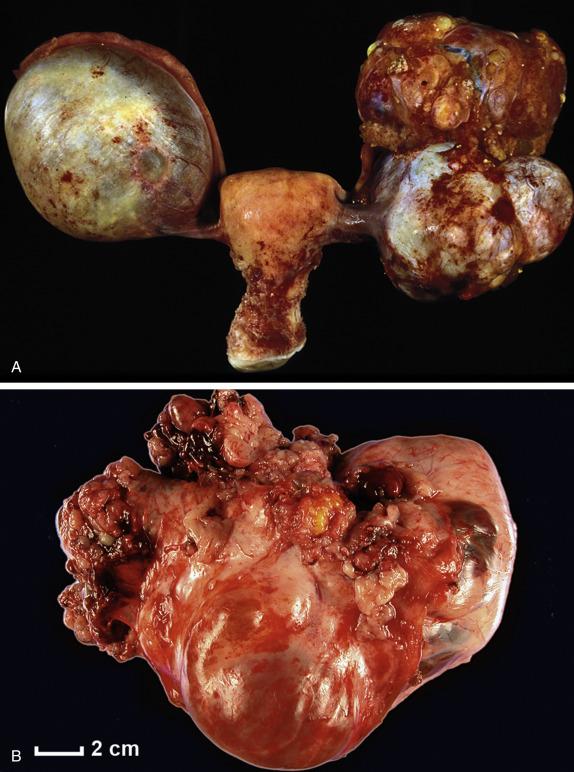
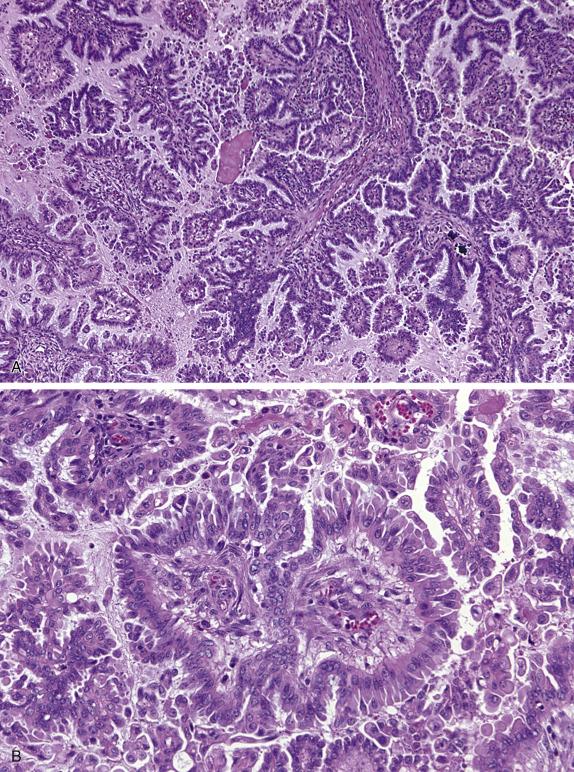
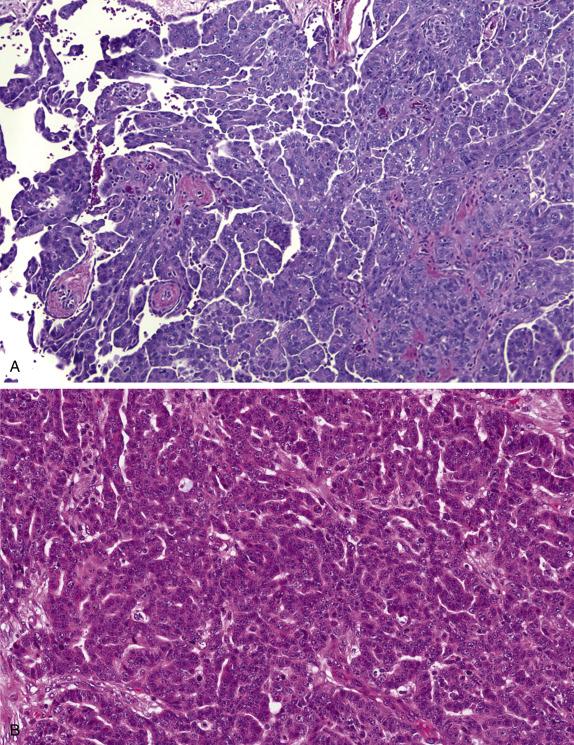
In their classic form, high-grade serous carcinomas are characterized by complex branching papillae and glands lined by a population of pleomorphic cells with loss of polarity. The cells grow in tufts and micropapillae forming narrow, slit-like spaces within the lumen. The interface with the stroma shows destructive stromal invasion, which is recognized by confluent growth of malignant cells or irregularly arranged glands, papillae, and/or nests of tumor cells with desmoplasia ( Fig. 14.18 ). Cytologically, the cells display pronounced nuclear atypia noticeable at low-power magnification, which includes bizarre cells showing large and pleomorphic round nuclei with high nuclear-to-cytoplasmic ratio, coarse to open chromatin and prominent nucleoli (many times with a cherry red appearance). Mitotic activity is usually brisk (>12 mitoses per 10 HPFs) and abnormal figures are common. Some tumors show alternative architectural patterns, including solid, endometrioid, and transitional (known as “SET”), either predominantly or exclusively ( Fig. 14.19 ). These patterns appear to be more common in BRCA mutation carriers. Although deceiving and suggestive of other types of malignancy, they display the same severe nuclear atypia and mitotic activity seen in classic forms of this tumor. Tumor cell necrosis is common and can be extensive. Psammoma bodies may be seen.
The evaluation of pathologic response to neoadjuvant chemotherapy in patients with tubo-ovarian and peritoneal high-grade serous carcinoma has prognostic significance as it correlates with progression-free survival and response to first-line platinum therapy. To this end, the Chemotherapy Response Score (CRS) system was developed to assess and document response. The system is simple and has good interobserver reproducibility especially after online training. The CRS has three categories:
CRS score 1: No or minimal tumor response
Mainly viable tumor
No or minimal regression-associated fibro-inflammatory changes, limited to a few foci
CRS score 2: Appreciable tumor response with residual tumor
Multifocal or diffuse fibro-inflammatory regressive changes with tumor in sheets, streaks or nodules
Extensive regression-associated fibro-inflammatory changes with multifocal residual tumor, which is regularly distributed and easily identifiable
CRS score 3: Complete or near-complete response
Mainly regression associated fibro-inflammatory changes
Absent or minimal tumor, the latter defined as very few, irregularly scattered individual tumor cells or cell groups or nodules up to 2 mm
Importantly, response in the omentum appears to have better correlation with patient outcome, in contrast to response assessed in the ovary, and the omentum is the site of choice for chemotherapy response scoring. Reporting the CRS in the ovary, if available, is also encouraged.
Cytologic examination of ascitic fluid or abdominal/pelvic peritoneal washings is part of the formal staging of ovarian epithelial carcinoma. Positive peritoneal cytologic preparations (seen in up to 95% of cases) are characterized by loose papillary aggregates and/or tight clusters of cells with cytoplasmic vacuolization and hyperchromatic and/or pleomorphic nuclei ( Fig. 14.18D ). Fine-needle aspiration is indicated only for the diagnosis of an inoperable cancer or confirmation of suspected recurrent disease.
Serous carcinomas are positive for CK7, PAX8, WT1, Ber-EP4, and CD15 (Leu-M1) but negative for CK20. In addition, high-grade serous carcinomas usually exhibit abnormal p53 expression, defined as strong, diffuse nuclear staining in ≥80% of tumor cells or complete absence of expression in tumor cells (null pattern). A third, and much less frequent, abnormal p53 pattern is cytoplasmic staining. There must be at least some p53 expression in adjacent normal cells in order to accurately interpret the null pattern of abnormal p53 expression. About 4% of tumors have wild type p53 staining pattern; these have TP53 mutations that do not translate into abnormal protein expression. ER expression is present in most tumors, although intensity and distribution vary.
Marked genomic instability, with complex karyotypic abnormalities and aneuploidy, is the norm.
High-grade serous carcinoma usually exhibits TP53 mutations and somatic or germline abnormalities of BRCA1 and/or BRCA2 or related genes. Detection of BRCA1/2 mutations may be useful to instate Poly ADP-ribose polymerase (PARP) inhibitor therapy, which is indicated in mutation carriers.
Serous carcinoma should be distinguished from endometrioid carcinoma , particularly if the latter is high grade or has significant solid growth. It is important to note that most tumors with high-grade nuclear morphology are indeed high-grade serous carcinomas, often with SET morphology. Endometrioid carcinoma features more elongated villous papillae, glands with more rounded luminal contours and preserved nuclear polarity. It is important to look for “confirmatory” endometrioid features, which include background of endometriosis or endometrioid adenofibroma, and secretory, squamous, or squamous morular metaplasia. Finally, endometrioid carcinomas are negative for WT1, in contrast to serous carcinomas. Clear cell carcinoma is frequently papillary but is distinguished from serous carcinoma by the small round appearance of the papillae with more prominent hyalinized cores and less epithelial stratification of the lining cells, which may show hobnailing or clear cytoplasm. Clear cell carcinomas with solid growth are more difficult but in general display a less severe degree of nuclear atypia than high-grade serous carcinoma, and the mitotic count rarely exceeds 12 mitoses per 10 HPFs. Positivity for Napsin-A, normal p53 staining and negative ER are in keeping with clear cell carcinoma. When a serous carcinoma involves the ovary and the endometrium , available evidence suggests that these tumors are clonal (i.e., not independent primaries) and primary in the endometrium in most, if not all, cases, even if there is little or no myometrial invasion by the uterine tumor. Metastatic breast carcinoma is recognized on the basis of the clinical history, strong positivity for GATA3 and GCDFP-15, and absent WT1 staining. Lastly, tumors formerly known as transitional cell carcinoma of the ovary are now regarded as high-grade serous carcinomas with transitional-like growth (part of the SET morphologic spectrum). The rare instance of a malignant Brenner tumor needs consideration; these tumors should have areas of benign or borderline Brenner tumor.
Median 5 year survival rates range from 15 to 55% with stage at presentation and amount of residual disease after debulking as the most important prognostic factors. Platinum-based chemotherapy is the first line of therapy, either after primary debulking or in the neoadjuvant setting. Regarding the latter, interval debulking is usually performed, and the degree of response to chemotherapy can be assessed using the CRS. Although most high-grade serous carcinomas initially respond to chemotherapy, responses are not durable and most patients progress towards widespread dissemination involving abdomen, lung, liver, and pleura. PARP inhibitors are now part of second-line systemic treatment for patients with BRCA1/2 mutations (either germline or somatic) and other forms of homologous recombination deficiency. Good response to neoadjuvant chemotherapy is also an emerging indication for maintenance therapy with PARP inhibitors.
Malignant epithelial tumor with serous (tubal-type) phenotype and high-grade cytomorphology
Most common histologic subtype of surface epithelial ovarian carcinoma
Bilateral (60%)
Most patients present with advanced stage (>80%), with transabdominal spread to involve peritoneum (with omental caking) and abdominal lymph nodes
Lungs, liver, and pleura common sites of distant metastases
Median 5 year survival rates range from 15 to 55%
More common in western hemisphere
Rare in first two decades, but steady increase thereafter (mean age 56 years)
Pelvic or abdominal enlargement, vague pain, urinary or gastrointestinal symptoms
Ascites
Elevated serum CA125
Amount of residual disease and stage are the most important prognostic factors
Primary surgical debulking and staging preferred
Chemotherapy, either adjuvant or neoadjuvant (followed by interval debulking), the mainstay of therapy
PARP inhibitors increasingly entering practice in addition to platinum and taxane chemotherapy
Despite therapy, vast majority eventually die of the disease.
These neoplasms, also known as “mixed Müllerian tumors,” are listed as a separate tumor type in the World Health Organization (WHO) classification. Recent insights, however, suggest that a malignant category (seromucinous carcinoma) does not exist, as these tumors can be reclassified as endometrioid (majority), serous, or mucinous based on morphology, immunophenotype, and molecular features. Benign and borderline seromucinous tumors are discussed here, although they are rare and their biology and relationship to other tumor types remain to be fully elucidated.
Solid and cystic mass with necrosis and hemorrhage
Surface excrescences and adhesions
Classic architecture: complex papillae and glands with slit-like spaces, cellular stratification, and loss of polarity
SET architecture: Solid, endometrioid, and transitional
Pleomorphic cells with high N:C ratios, large nuclei with coarse chromatin and prominent nucleoli
Mitotic activity >12 mitoses/10 HPFs
Psammoma bodies
CRS applied to cases resected after neoadjuvant therapy
PAX8, CK7, EMA, WT1, BerEP4, CA125, and CD15 positive
ER variably positive
Abnormal p53 expression
Calretinin and CK20 negative
TP53 mutations
Germline or somatic BRCA1 / BRCA2 mutations
Low-grade serous carcinoma
Endometrioid adenocarcinoma
Clear cell carcinoma
Metastatic serous carcinoma of endometrium
Metastatic breast carcinoma
Benign seromucinous tumors (cystadenoma, adenofibroma) are uncommon. They are almost always confined to the ovary.
Seromucinous tumors occur over a wide age range but are most common in the reproductive age group. They may be bilateral and are frequently associated with endometriosis. Women may present with abdominal enlargement, pain, or vaginal bleeding, but most benign tumors are asymptomatic and discovered incidentally.
Benign seromucinous tumors are composed of varying amounts of fibromatous stroma (when predominant, serous adenofibroma) and cysts (which are typically less prominent).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here