Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Double-outlet ventricle is no more than an abnormal ventriculoarterial connection. Therefore the feature may occur with each atrial arrangement, any atrioventricular connection, and all possible variations of ventricular morphology. The morphologic arrangement can then be further complicated by associated defects, some common and others rare. The clinical picture is as inconstant as the anatomic permutations and associations suggest. This chapter reviews the various combinations in which the outflow tracts arise from the same ventricle but in the setting of concordant atrioventricular connections. When both arterial trunks are found in the setting of discordant or mixed atrioventricular connections, the abnormal arrangements at either the atrioventricular or venoatrial junctions dominate the picture. Double-outlet ventricle can also be found in the setting of the functionally univentricular heart.
Abernethy of St. Bartholomew's Hospital in London described the first known example of double-outlet right ventricle: “Both ventricles, the left by means of an opening in the upper part of the septum ventriculorum, projected their blood into the aorta.” The term double-outlet right ventricle , however, did not appear until 1957, although Fallot described the origin of the aorta as arising exclusively from the right ventricle when he was assessing the pathologic findings in hearts obtained from patients presenting with la maladie bleue. Prior to 1957, hearts with both arterial trunks arising from the right ventricle were usually considered to represent partial transposition. This was because only the aorta was deemed to be placed across the ventricular septum. Transposition was considered complete when both arterial roots were placed across the septum to rise from morphologically inappropriate ventricles. It is now generally accepted that the discordant ventriculoarterial connections are the essence of transposition (see Chapter 37 ). Transposition, therefore, is now considered to be mutually exclusive from double-outlet right ventricle. The relationship between double-outlet right ventricle and tetralogy of Fallot has also been contentious. This is perhaps surprising since, as already indicated, the aorta was observed to arise exclusively from the right ventricle in one of the original hearts examined by Fallot himself. In many examples with the phenotypic morphology of tetralogy (see Chapter 35 ), the greater part of the overriding aortic root, when considered relative to its short axis, is supported predominantly by right ventricular structures. Patients having such hearts are included in this chapter. The arguments regarding the relationship between tetralogy and double-outlet ventricles had centered on whether it was necessary to find bilaterally complete muscular infundibula to diagnose double-outlet right ventricle. As emphasized in Chapter 1 , structures should be defined in their own right and not on the basis of another feature, which is variable. Application of this principle, known as the morphologic method, means that it is inappropriate to define double-outlet ventricle, a specific form of ventriculoarterial connection, on the basis of infundibular morphology. The latter is but one of the many complicating features seen when both arterial trunks arise in larger part from the same ventricle. Our recent investigation of a large series of hearts having both arterial trunks exclusively supported by the right ventricle revealed that two-thirds lacked bilaterally complete infundibula. Double-outlet from the right ventricle, however, accounts for only part of the overall group of hearts to be described in this chapter. The other group, namely involving a double-outlet from the morphologically left ventricle, is very much rarer. At one point this finding was considered an embryologic impossibility. However, description of a heart with both arterial trunks arising from the left ventricle in the setting of an intact ventricular septum proved its existence beyond doubt. However, cases still tend to be described in isolation rather than in large series either from the pathologic or clinical viewpoint.
Double-outlet right ventricle forms a rare spectrum of congenital cardiac anomalies that represents approximately 1% of all cardiac defects. Its reported prevalence varies widely and ranges between 3 and 24 in 100,000 live births. Variability in the reported prevalence exists due to controversies in the definition of double-outlet right ventricle and the resulting inconsistent classification. Despite the rare occurrence of the malformation, its clinical implications are significant, as its morphologic spectrum demands complex decision making in both medical and surgical therapy. Among the subtypes proposed by Lev et al., subaortic interventricular communications are most common, followed by subpulmonary, noncommitted, and doubly committed communications ( Table 39.1 ). Even rarer is the malformation where the arterial trunks are supported to a greater degree by the left ventricle, so-called double-outlet left ventricle. Double-outlet left ventricle is estimated to occur in fewer than 1 in 100,000 live births; however, exact data are lacking. Genetic anomalies associated with double-outlet ventricles have been identified in approximately 40% of patients. Trisomy 13 and 18, as well as 22q11.2 deletion syndrome, have been the most common chromosomal abnormalities.
| All | Brown et al. | Bradley et al. | Li et al. | Villemain et al. | |
|---|---|---|---|---|---|
| N (%) | N (%) | N (%) | N (%) | N (%) | |
| Number of patients | 1272 (100.0) | 124 (100.0) | 393 (100.0) | 380 (100.0) | 433 (100.0) |
| Subaortic | 432 (48.2) a | 57 (46.0) | 156 (46.6) | 219 (57.6) | 271 (62.6) |
| Subpulmonary | 331 (26.0) | 39 (31.5) | 76 (22.7) | 77 (20.3) | 139 (32.1) |
| Doubly committed | 38 (4.2) a | 6 (4.8) | 15 (4.5) | 17 (4.5) | – b |
| Noncommitted | 200 (15.7) | 22 (17.7) | 88 (26.3) | 67 (17.6) | 23 (5.3) |
| Pulmonary obstruction | 672 (52.8) | 65 (52.4) | 257 (65.4) | 166 (43.7) | 184 (42.5) |
| Era | 1980–2000 | 1980–2000 | 2005–2012 | 1992–2013 |
a Excluding patients from Villemain et al.
b In this study, the authors did not differentiate between interventricular communications that were subaortic or doubly committed, but referred to them as subaortic only. Therefore, the number given for subaortic interventricular communications combines interventricular communications that authors of other papers have separated into subaortic and doubly committed interventricular communications.
Since double-outlet ventricle is only one variety of ventriculoarterial connection, it follows that multiple phenotypes are appropriately described within this grouping. It helps, therefore, to consider first the categorization of the various malformations and only then proceeding to provide details of specific morphology. We concentrate in this regard on the more common and more important lesions to be found within the groups.
Analysis, be it in the clinical or pathologic situation, follows the sequential segmental approach (see Chapter 1 ). The first step is to ascertain the atrial arrangement and venous connections. The atrioventricular connections must then be established with certainty. At the ventricular level, note must be taken of the interrelationships of the arterial trunks, the specific infundibular morphology, and the nature and severity of any obstructive problems in the subaortic and subpulmonary outflow tracts. The size, site, and morphology of the interventricular communication, however, are the most important features. This channel provides the outflow tract for the other ventricle when both arterial trunks arise from the same ventricle ( Fig. 39.1 ).
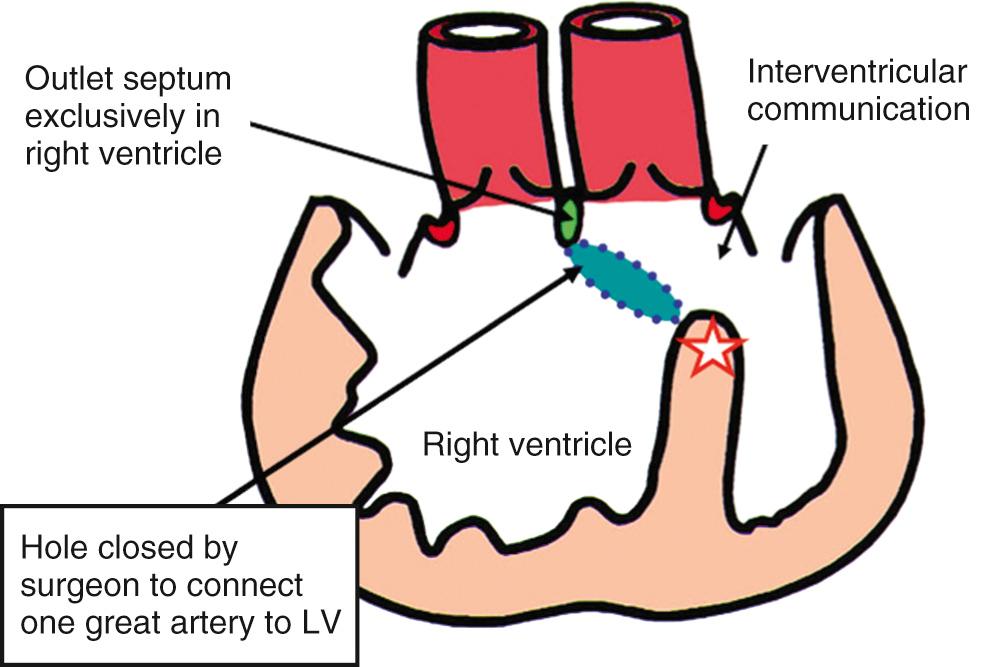
The relationship of the interventricular communication with the subarterial ventricular outlets has provided the basis for most categorizations. In these previous categorizations, however, the channel has usually been described as a ventricular septal defect . The borders of the channel between the ventricles that exists in the setting of double-outlet right ventricle, however, are markedly different from those that are found when the ventriculoarterial connections are concordant or discordant. In tetralogy of Fallot, for example, the cranial border of the channel between the ventricles, which is the area usually described as the ventricular septal defect, is the outlet septum. When both arterial trunks arise from the right ventricle or when one arterial trunk is overriding with the other arising from the right ventricle, the outlet septum of necessity will itself be a right ventricular structure. Contrariwise, should both arterial trunks arise from the left ventricle, the outlet septum will be a left ventricular structure. It follows that when both arterial trunks arise predominantly from the right ventricle, the outlet septum will form the cranial border of the area of surgical ventricular septation. The cranial margin of the channel between the ventricles, in contrast, is the inner heart curvature, also known as the ventriculoinfundibular fold. This is the area of putative ventricular septation, representing the locus around which the surgeon will place a patch to tunnel the channel into the aortic root; it is described as the ventricular septal defect in the setting of tetralogy of Fallot ( Fig. 39.2 ).
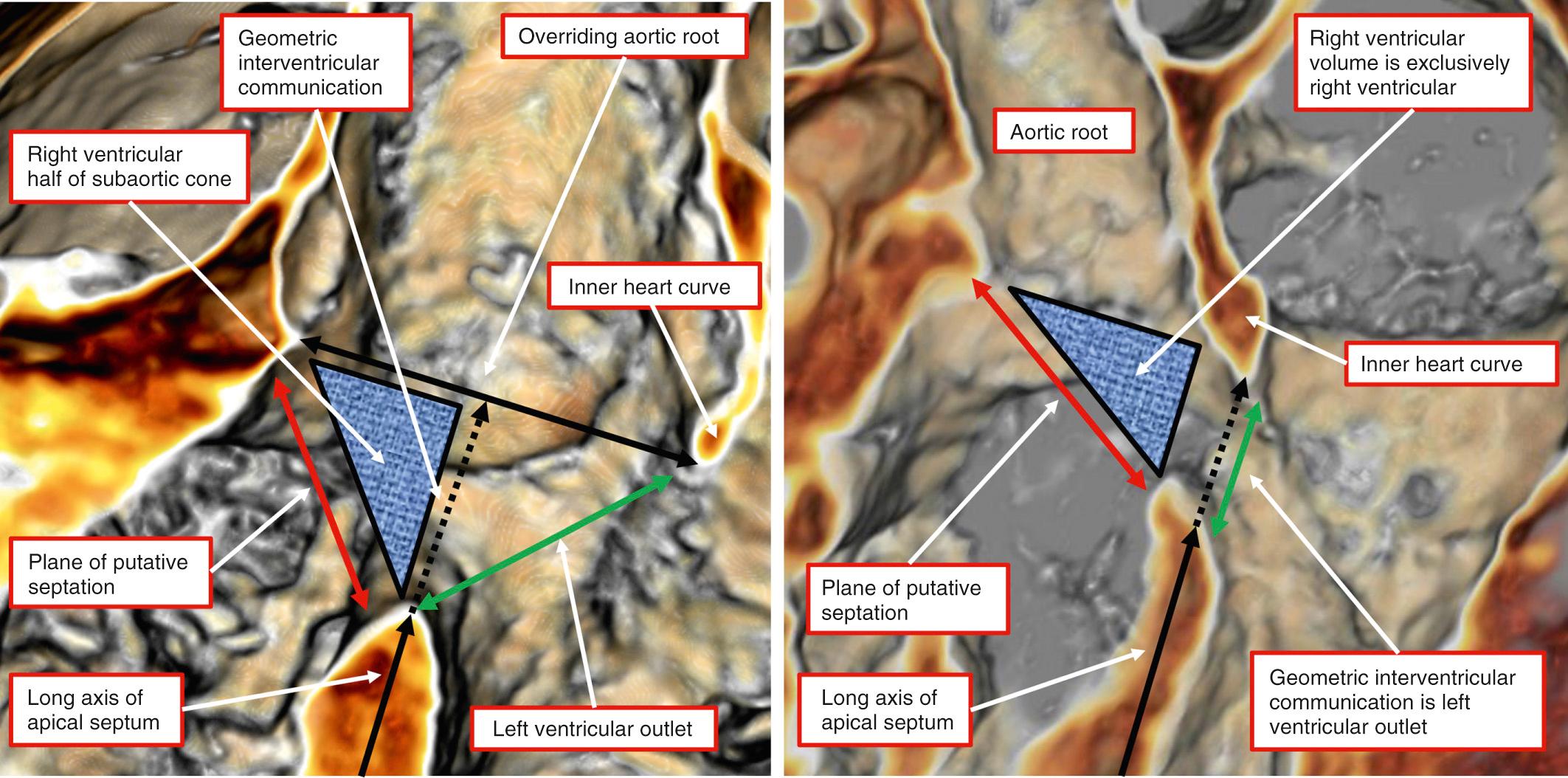
In light of the earlier discussions, it is questionable whether the channel found between the ventricles when both arterial trunks arise from the right ventricle can justifiably be called a ventricular septal defect. The channel can never be closed surgically, unless an alternative channel is constructed to provide the outlet from the ventricle not directly supporting an arterial trunk. Throughout this chapter, we describe the channel as the interventricular communication rather than a ventricular septal defect, although we recognize that most will continue to describe it in the latter fashion. More importantly, because of the marked phenotypic variability coupled with the additional variation within recognized phenotypes, the categorization of each case should be individualized. In comparable fashion, the medical and surgical management must be tailored to cater for the particular problems of the individual patient. Certain anatomic combinations, nonetheless, do occur with sufficient frequency to merit specific discussion.
The more common variants, in approximate order of frequency, are as follows:
Those with the interventricular communication in subaortic position, with the aorta spiraling from right to left relative to the pulmonary trunk but in combination with pulmonary stenosis. This is the Fallot variant.
Those with the interventricular communication in subpulmonary position, with the aorta to the right of the pulmonary trunk and parallel to, it. This is the Taussig-Bing variant.
Those with the interventricular communication in subaortic position, with the aorta spiraling from right to left relative to the pulmonary trunk but in the absence of pulmonary stenosis.
Less common variants are the following:
Those with the interventricular communication uncommitted, or noncommitted, to either subarterial outlet and with the aorta to the right of the pulmonary trunk, with either spiraling or parallel arterial trunks.
Those with the interventricular communication in doubly committed position, with the aorta to the right of the pulmonary trunk and spiraling arterial trunks.
Those with the interventricular communication in subaortic position but with the aorta to the left of the pulmonary trunk with parallel arterial trunks.
Those with the usual atrial arrangement and discordant atrioventricular connections, usually with the aorta parallel to and to the left of the pulmonary trunk. Because of the significance of the discordant atrioventricular connections, this variant is discussed in Chapter 38 .
Those with mirror–imaged atrial arrangement and any of the above variations.
Those with isomeric atrial appendages and, hence, mixed and biventricular atrioventricular connections. These variants are discussed in Chapter 26 .
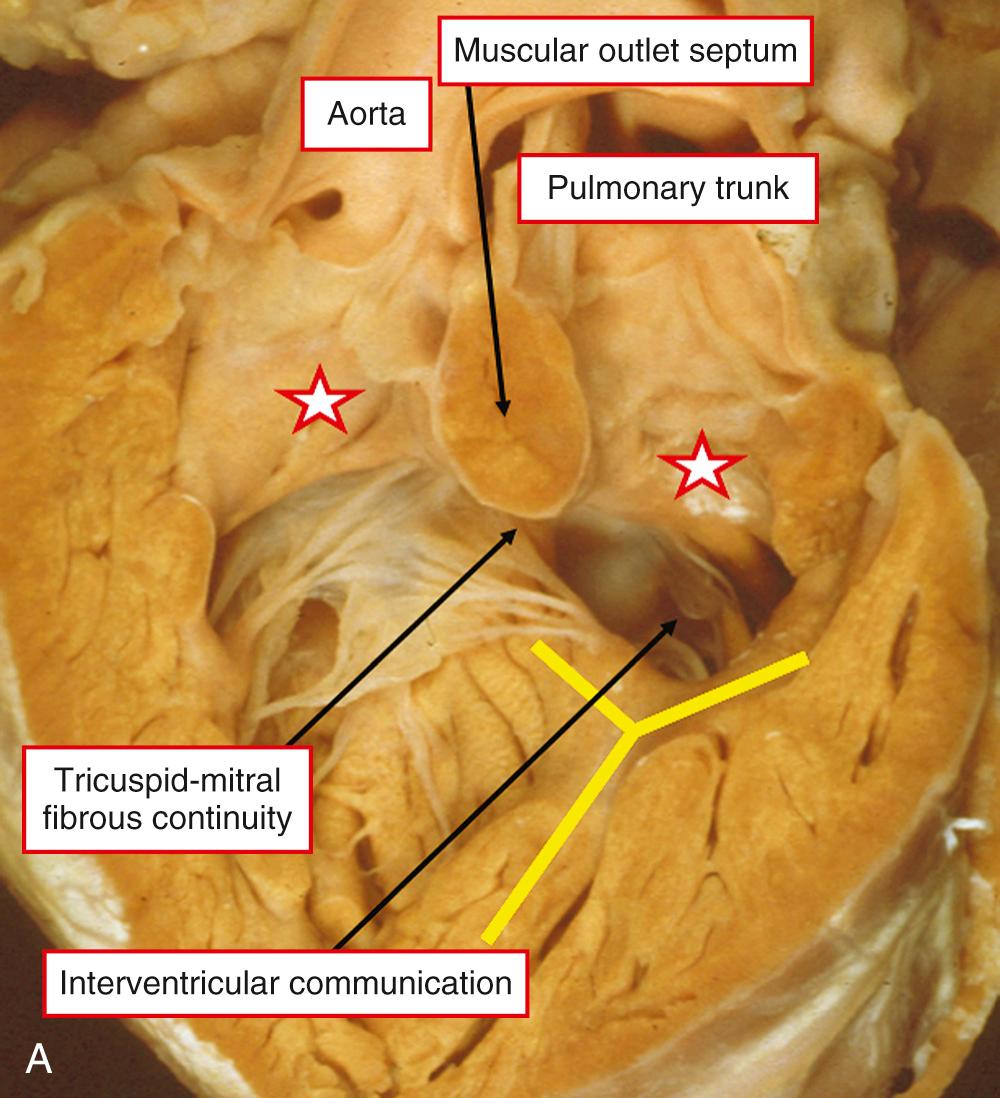
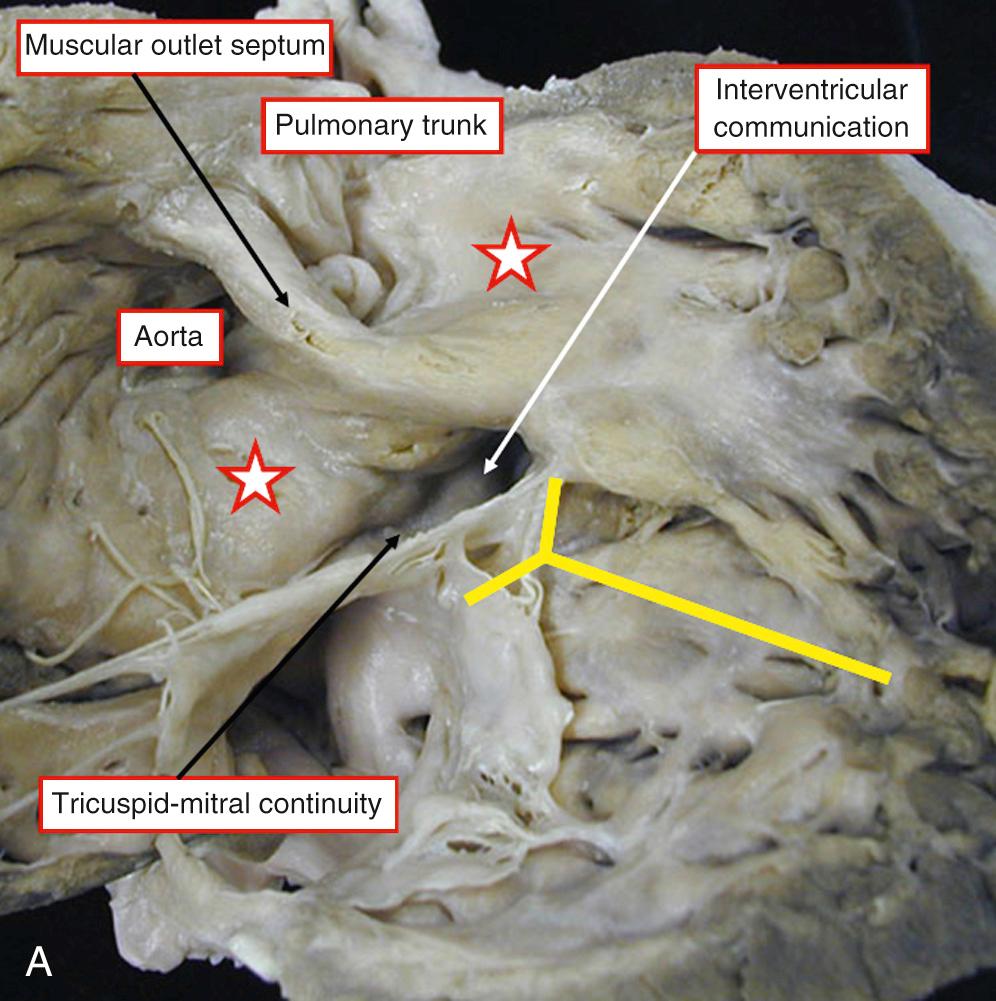
There is usually a complete muscular infundibulum supporting each arterial valve ( Fig. 39.3A ), although fibrous continuity between the aortic and atrioventricular valves is found in many cases (see Fig. 39.3B ). The subpulmonary outflow tract is narrowed, typically with hypoplasia of the infundibulum. Often the valve is additionally involved. The obstructive lesions are similar to those seen in tetralogy of Fallot. The interventricular communication, cradled within the limbs of the septomarginal trabeculation, is perimembranous in most cases (see Fig. 39.3 ), but can have a muscular posteroinferior rim.
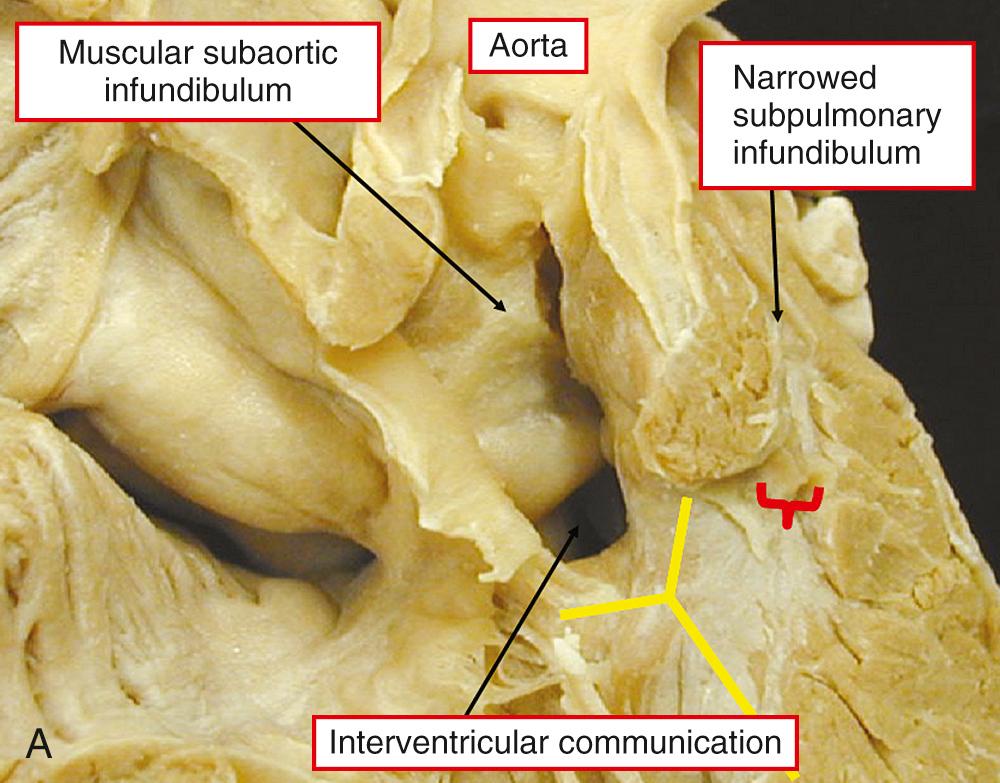
The specific morphology of the interventricular communication has major surgical significance. When there is a muscular posteroinferior rim, the atrioventricular conduction axis is not directly related to the margins of the defect and is less vulnerable at operation. In many cases the aortic valve retains part of its connection within the left ventricle. As long as most of the aorta is supported by the right ventricle, it is appropriate to diagnose the ventriculoarterial connection as being double-outlet, remembering that commitment is assessed on the basis of the short axis of the overriding arterial root ( Fig. 39.4 ). Important associated lesions may include a restrictive interventricular communication, which in essence represents obstruction of the left ventricular outlet. Mitral stenosis can also be found.
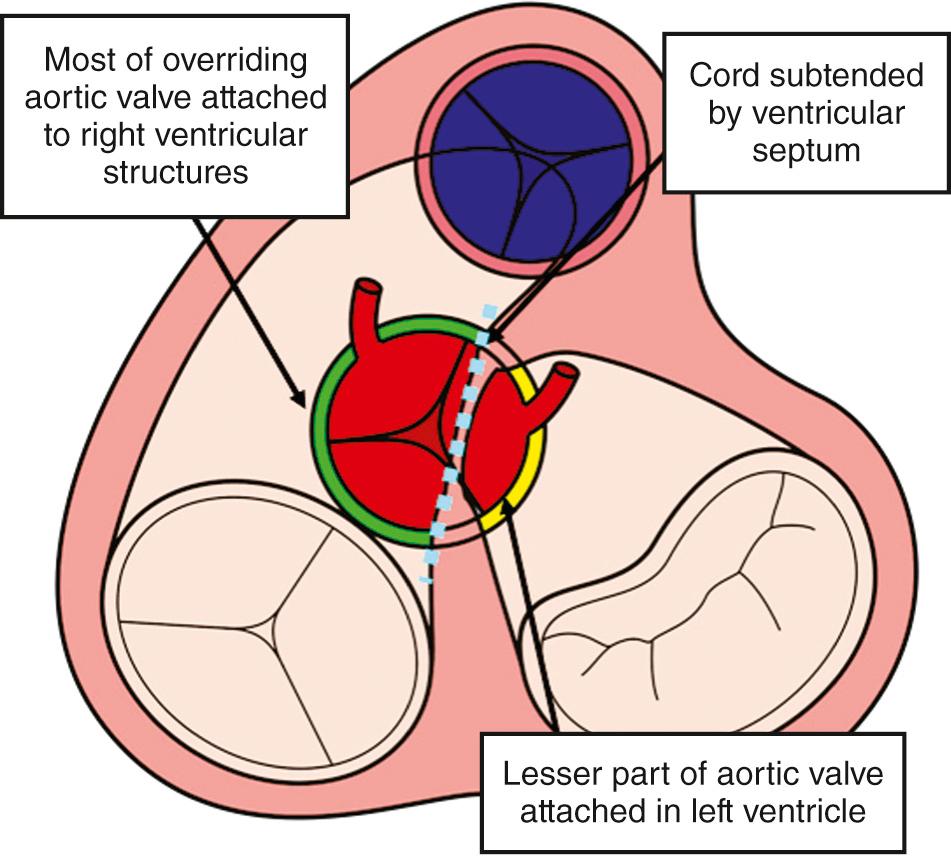
These hearts are usually described as the Taussig-Bing malformation. In the initial heart thus described, both arterial valves were supported by complete muscular infundibula. The term is now used to describe the spectrum of overriding of the pulmonary trunk in the setting of parallel arterial trunks, with the ends of the spectrum being either double-outlet right ventricle ( Fig. 39.5 ) or discordant ventriculoarterial connections (see Chapter 37 ). The interventricular communication, again opening to the right ventricle between the limbs of the septomarginal trabeculation, does so beneath the pulmonary trunk, with the muscular outlet septum attached to the ventriculoinfundibular fold. A muscular posteroinferior rim, formed by union of trabeculation and fold, often separates the rim of the defect itself from the membranous septum (see Fig. 39.5A ). As explained earlier, when present, the rim protects the atrioventricular conduction axis. In most cases, nonetheless, the defect extends posteriorly, becoming perimembranous (see Fig. 39.5B ).
This malformation is similar to the lesion having a subaortic defect and pulmonary stenosis (compare Figs. 39.3 and 39.6 ). There can be bilateral infundibula (see Fig. 39.6A ) or fibrous continuity between the leaflets of the aortic and mitral valves (see Fig. 39.6B ). When bilateral infundibula are found, the arterial valves are usually side by side. In the setting of fibrous continuity between the leaflets of the aortic and mitral valves, the aortic valve is typically posterior and to the right of the pulmonary valve. The interventricular communication is usually perimembranous (see Fig. 39.6A ), although it may have a muscular posteroinferior margin (see Fig. 39.6B ). When there is continuity between the leaflets of the aortic and mitral valves, the aortic root usually retains part of its connection with the left ventricle. Although the interventricular communication is often large, it can be restrictive. As emphasized earlier, such a feature produces obstruction to the outflow from the left ventricle.
On occasion, there can be multiple interventricular communications. Abnormalities of the atrioventricular valves are found in up to one-quarter of the hearts seen in autopsied series and include mitral stenosis and straddling mitral valve. Obstruction to one or other of the arterial outlets occurs frequently. Pulmonary stenosis, when present, is similar to that seen in tetralogy of Fallot (see Fig. 39.3A ) but is infrequent in the setting of the Taussig-Bing heart. Obstruction of the systemic outlet is much more frequent in the latter setting, with severe coarctation or interruption of the aortic arch found in a significant proportion of such cases. Other associated malformations include juxtaposition of the atrial appendages, totally anomalous pulmonary venous connection, and twisted atrioventricular connections. Variations in coronary arterial anatomy are particularly important in the setting of the Taussig-Bing hearts, since these are now corrected surgically using the arterial switch procedure. The most common arterial variation is 1L2RCx, with the left coronary being rather long. Interatrial communications within the oval fossa are also frequent.
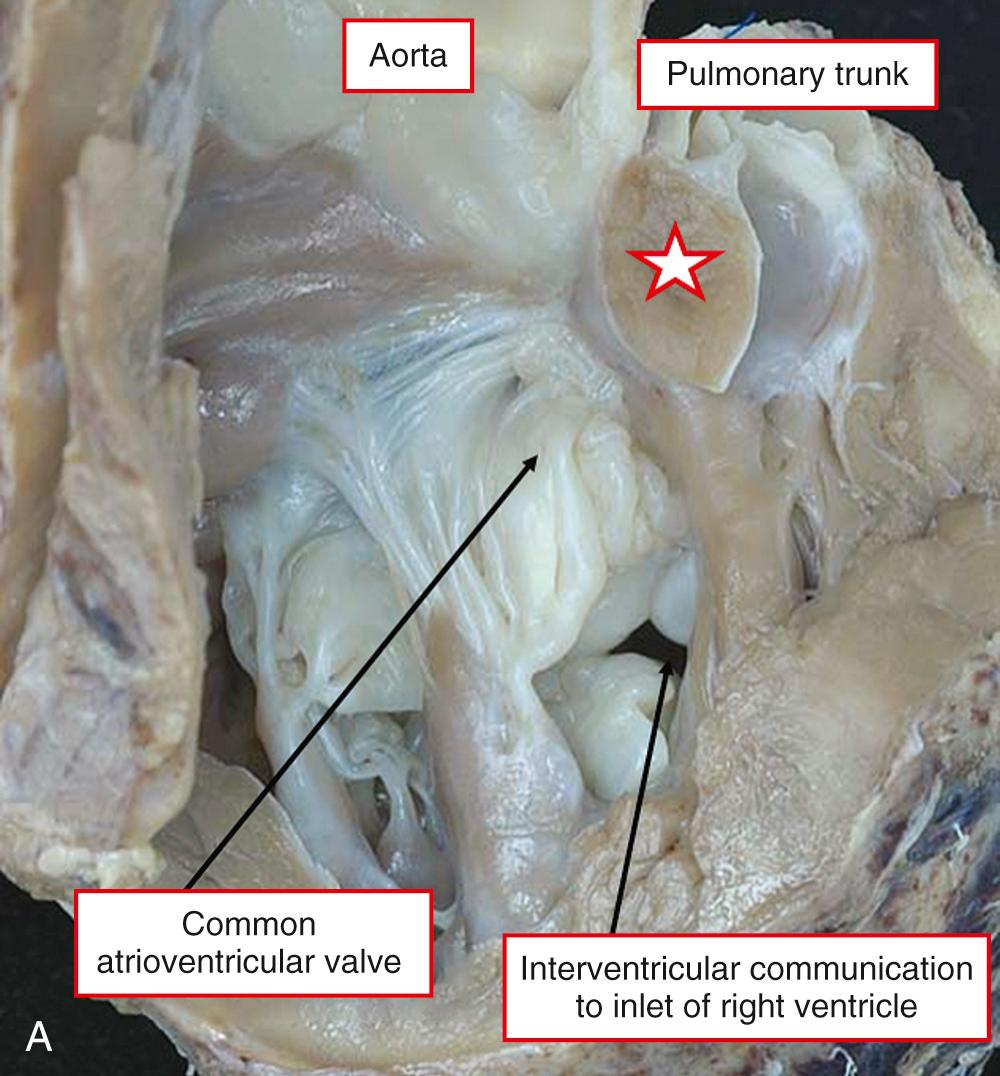
In a minority of patients, the interventricular communication is not directly committed to either arterial outlet. Such defects are usually the ventricular component of an atrioventricular septal defect, typically with a common atrioventricular valve ( Fig. 39.7A ). More rarely, the noncommitted communication is an isolated muscular defect that opens to the inlet or apical part of the right ventricle (see Fig. 39.7B ). Although the ventricular component of an atrioventricular septal defect may seem noncommitted to the anatomist, the surgeon is frequently able to create a long tunnel to one or other outflow tract (see Fig. 39.7A ). The relationships of the defect to the arterial outlets and to other structures within the right ventricle, therefore, require careful analysis. A key feature determining surgical noncommitment will be the presence of leaflet tissue derived from the tricuspid valve interposed between the defect and the arterial outlets (see Fig. 39.7B ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here