Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
![]() Additional content is available online at Elsevier eBooks for Practicing Clinicians
Additional content is available online at Elsevier eBooks for Practicing Clinicians
Acute heart failure (AHF) is among the most common causes for hospitalization in patients older than 65 years in the developed world. Increasingly, the spectrum of worsening HF is recognized to encompass not just patients requiring acute hospitalization, but also worsening of HF in outpatient settings and in patients already hospitalized. The prevalence of HF is projected to continue to increase over time due to a convergence of several epidemiologic trends: the aging of the population, given the age-related incidence of HF; the reduction in hypertension-related mortality and the greatly improved survival after myocardial infarction (MI), resulting in more patients living with chronic heart failure (see Chapter 48 ); and the availability of effective therapy for prevention of sudden death (see Chapter 58, Chapter 70 ) ( Fig. 49.1 ). AHF is increasingly recognized as a distinct disorder with unique epidemiology, pathophysiology, treatments, and outcomes.
A variety of overlapping terms have been used to characterize AHF in the literature, including “acute heart failure syndromes” (AHFS), “acute decompensated heart failure” (ADHF), “acute decompensation of chronic heart failure” (ADCHF), “worsening heart failure” (WHF), and “hospitalization for heart failure” (HHF). Although none of these is universally accepted, we will use the terminology “acute heart failure” in this chapter for simplicity. Broadly speaking, AHF can be defined as the new onset or recurrence of symptoms and signs of HF requiring urgent or emergent therapy and resulting in unscheduled care or hospitalization. An important source of confusion with this proposed definition is the word “acute”—although this suggests a sudden onset of symptoms, many patients may have a more sub-acute course, with gradual worsening of symptoms that ultimately reaches a level of severity sufficient to seek unscheduled medical care.
In the United States, HF is the primary diagnosis for more than 1 million hospitalized patients annually, and a secondary diagnosis for an additional 3 million hospitalizations. Similar numbers of hospitalizations are reported in Europe. The direct and indirect costs associated with HF approach 40 billion US dollars per year in the United States, and the majority of these expenditures are related to the costs of hospitalizations. , As noted earlier, the overall prevalence of chronic heart failure continues to grow. However, recent data suggest that the age-adjusted rate of HHF has begun to decrease, at least for primary heart failure events. To what extent these changes are related to more effective treatments of chronic heart failure or alternatively, changes in care to create alternative care pathways for avoiding hospitalization is unknown. Changes in medical care (especially in the United States) have led to increased efforts to manage milder forms of WHF without hospitalization, utilizing outpatient diuretic clinics and observation units, although available data suggest that even these milder forms of decompensation are still associated with adverse prognosis. , Despite these potentially encouraging trends, AHF will be a major clinical and economic problem for health care systems for the foreseeable future. An important development in the understanding of the epidemiology, clinical characteristics, and outcomes of patients with AHF has been the development of large, relatively unselected registries of AHF which provide a “real-world” perspective on the epidemiology and outcomes of AHF worldwide ( Table 49.1 ).
| Adhere ( n = 187,565) | Optimize-HF ( n = 48,612) | Perna et al. ( n = 2974) | EHFS II ( n = 3580) | Attend ( n = 4841) | Damasceno ( n = 1006) | Report-HF ( n = 18,102) | |
|---|---|---|---|---|---|---|---|
| Region | US | US | Argentina | Europe | Japan | Africa | Global |
| Age (years) | 75 | 73 | 68 | 70 | 73 | 52 | 67 |
| Male (%) | 48 | 48 | 59 | 61 | 58 | 49 | 61 |
| Preserved EF (%) | 53 | 51 | 26 | 52 | 47 | 25 | 45 |
| Prior HF (%) | 76 | 88 | 50 | 63 | 36 | – | 57 |
| Medical History | |||||||
| Coronary artery disease | 57% | 50% | 54% | N/A | 48% | ||
| Myocardial infarction | 30% | N/A | 22% | N/A | N/A | ||
| Hypertension | 74% | 71% | 66% | 62% | 69% | 56% | 64% |
| Atrial fibrillation or flutter | 31% | 31∗% | 27% | 39% | 40% | 18% | 31% |
| Chronic kidney disease | 30% | 20% | 10% | 17% | N/A | 8% | 20% |
| Diabetes | 44% | 42% | 23% | 33% | 34% | 11% | 37% |
| COPD/Asthma | 31% | 34% | 15% | 19% | 12% | N/A | N/A |
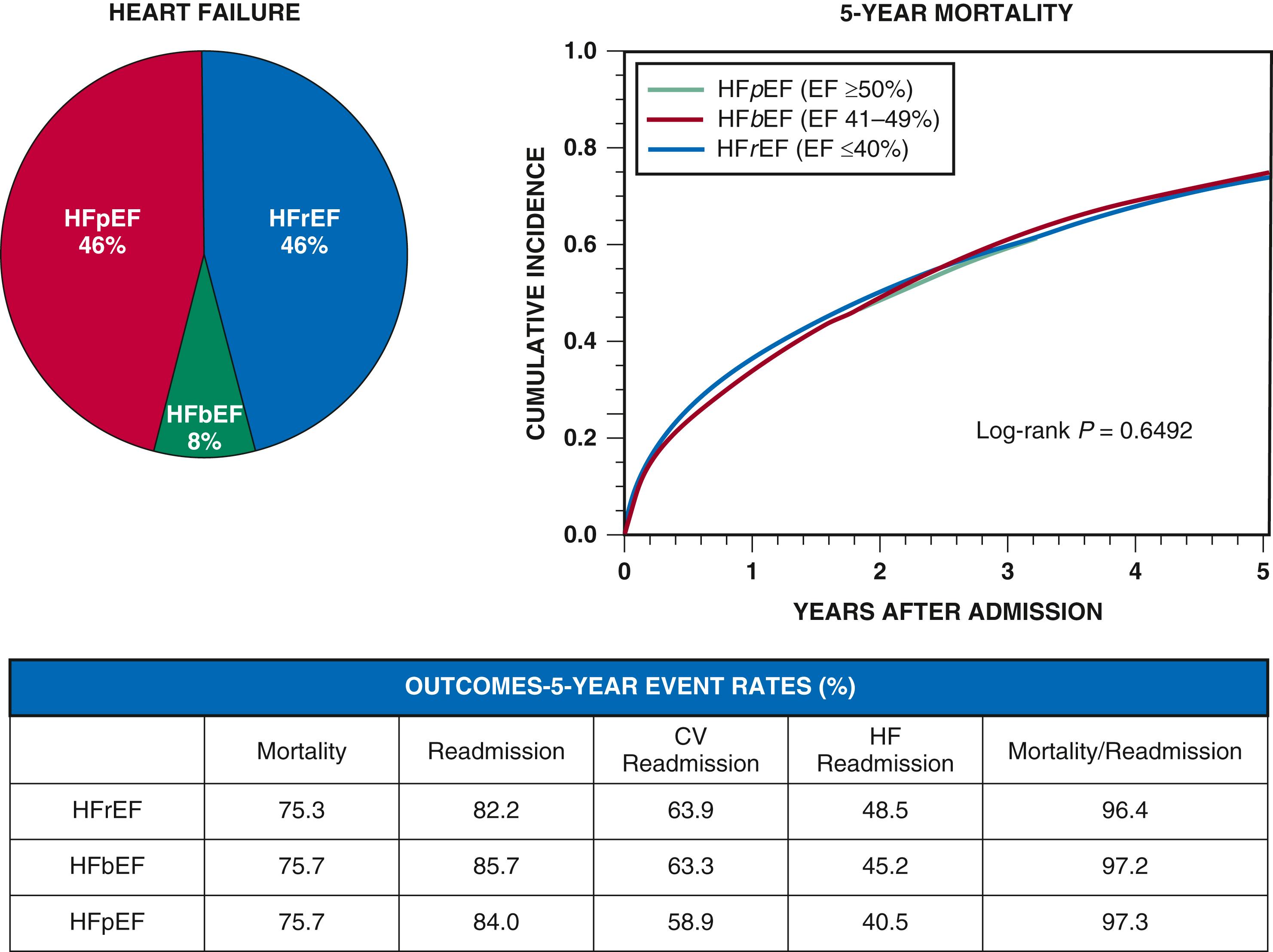
On the basis of available registry data, 40% to 50% of patients hospitalized have heart failure with preserved ejection fraction (HFpEF). Important epidemiologic differences exist between heart failure with reduced ejection fraction (HFrEF) and HFpEF (see Chapter 48 ). The in-hospital mortality of patients with HFpEF appears to be lower compared with that of patients with HFrEF, but post-discharge rehospitalization rates and long-term mortality after hospitalization are similarly high for both groups. Patients with AHF and HFpEF are more likely to be rehospitalized for and to die from non-CV causes than patients with AHF and HFrEF, reflecting their more advanced age and greater burden of comorbidity. More recently, the concept of heart failure with “mid-range ejection fraction” or “mildly reduced ejection fraction,” that is, HF-mrEF, has been proposed as an additional refinement of the standard HFrEF vs HFpEF dichotomy, but specific data on AHF outcomes in this group are limited ( Fig. 49.2 ).
There are significant differences in the epidemiology of AHF based on age, race, and gender. AHF disproportionally affects older people, with a mean age of 75 years in large registries. AHF affects men and women almost equally, but there are important differences by gender. In the ADHERE registry women admitted for AHF were older than men (74 vs. 70 years), and more frequently had preserved systolic function (51% vs. 28%). Differences in ethnic groups have been studied most extensively in the United States and have focused primarily on differences between African American and white patients. In the Organized Program to Initiate Lifesaving Treatment in Hospitalized Patients with Heart Failure (OPTIMIZE-HF) registry, African American patients admitted with AHF were younger (64 vs. 75 years), more likely to have left ventricular (LV) systolic dysfunction (57% vs. 51%) with a lower mean EF (35% vs. 40%), hypertensive cause for heart failure (39% vs. 19%), renal dysfunction, and diabetes compared to the non–African American group. Lower crude mortality rates have been reported for African Americans compared to non–African American patients, but when adjustments are made for these differences in comorbidities and age, mortality rates are similar.
Concomitant diseases are very common in patients admitted with AHF, reflective of the older population. These comorbidities not only represent diseases that are risk factors for the development of heart failure, but also can complicate diagnosis and management. Hypertension is the most prevalent concurrent condition, present in approximately two-thirds of the patients (see also Chapter 26 ), whereas coronary artery disease (CAD) is present in about half and dyslipidemia in over one-third (see Chapters 27 and 40 ). , Other conditions that are the result of the vascular injury produced by these diseases, such as stroke, peripheral vascular disease, and chronic kidney disease are also very common in patients with AHF. Diabetes mellitus is present in over 40% of US patients, most likely related to increasing incidence of obesity, and ranges from 27% to 38% in Europe. The interaction between heart failure status and diabetes has been a subject of substantial interest, given the evolving data that some classes of anti-diabetic drugs, specifically the sodium-glucose co-transporter-2 (SGLT-2) inhibitors and glucagon-like peptide-1 (GLP-1) agonists, have a favorable impact on heart failure outcomes (see Chapter 50 ). Atrial fibrillation can both precipitate AHF and complicate its management.
Although the majority of data continues to emerge from North America and from Europe, AHF is increasingly recognized as a global issue, and important differences between regions of the world have emerged in terms of epidemiology, therapy, and outcomes. Although there are a variety of country- or region-specific registries (see Table 49.1 ), to date most available data highlighting these differences have come from large global outcome trials. Although these studies can provide important insight into regional differences, they suffer from inherent selection bias of clinical trials and may not be truly representative of the general population. The recently reported REPORT-HF registry provides a more contemporary assessment of AHF globally (see Table 49.1 ).
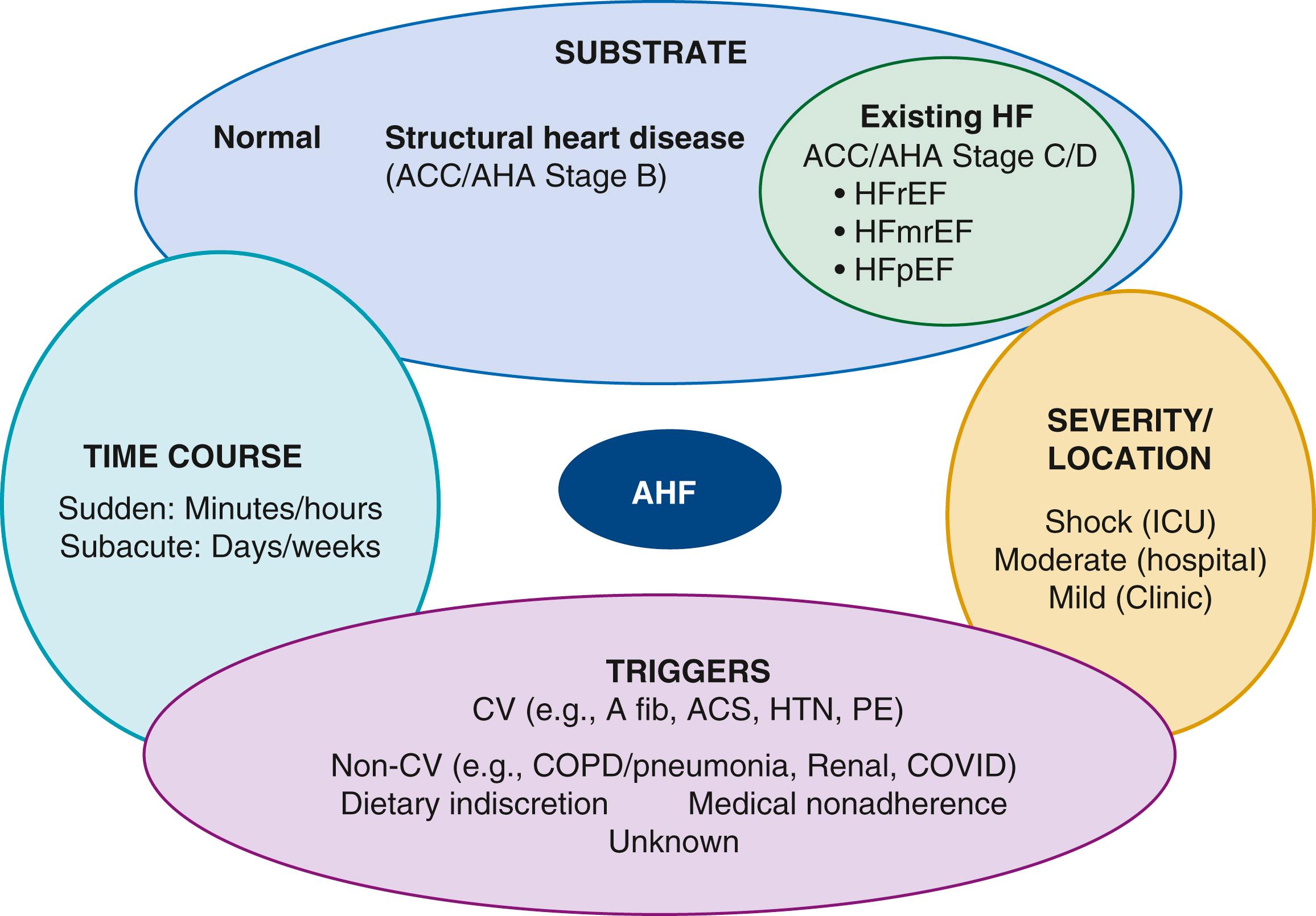
The pathophysiology of AHF is complex and highly variable, with many overlapping pathogenic mechanisms that may be operative to a greater or lesser degree. This fundamental heterogeneity complicates the attempt to create a unified conceptual model. A useful framework for understanding of the pathophysiology of AHF is to consider it as the result of the interaction of underlying substrate, initiating mechanisms or triggers, and amplifying mechanisms, all of which contribute to a common set of clinical signs and symptoms (primarily related to congestion, end-organ dysfunction, or both) that define the clinical picture of AHF ( Fig. 49.3 ). In this context, substrate refers to underlying cardiac structure and function. The underlying substrate may be one of normal ventricular function, for example patients without a prior history of HF who develop AHF because of sudden changes in ventricular function from an acute insult such as MI or acute myocarditis (see Chapter 55 ). Alternatively, some patients may have no prior history of HF but abnormal substrate (e.g., stage B patients with asymptomatic LV dysfunction) with a first presentation of heart failure (de novo heart failure). Finally, most patients with AHF have a substrate of chronic compensated HF who then decompensate and present with AHF (sometimes termed decompensated chronic heart failure [DCHF]).
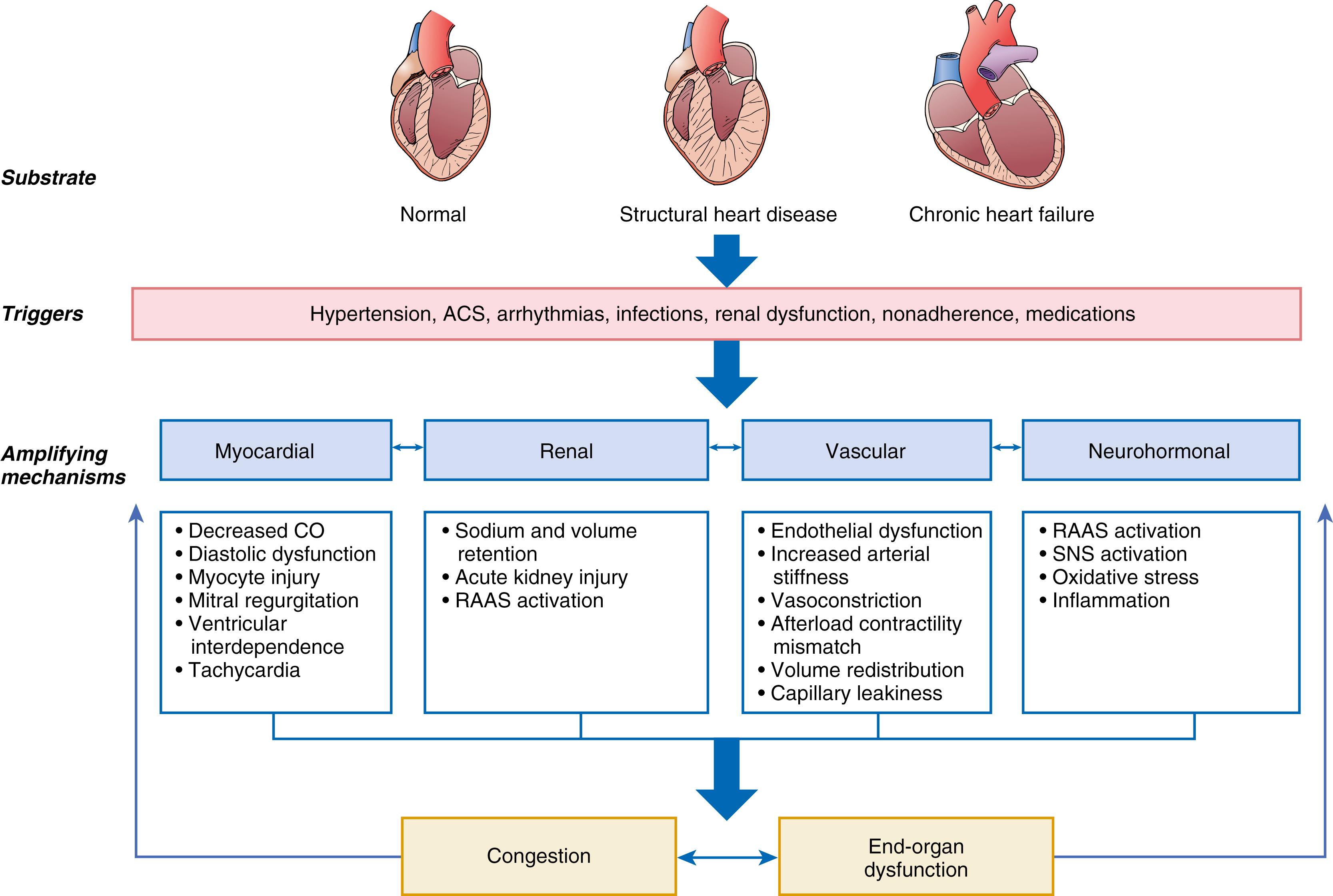
Initiating mechanisms vary according to and interact with the underlying substrate and may be cardiac or extra-cardiac. For patients with normal substrate (normal myocardium), a substantial insult to cardiac performance (e.g., acute myocarditis) is generally required to lead to the clinical presentation of AHF. For patients with abnormal substrate at baseline (asymptomatic LV dysfunction), smaller perturbations (e.g., poorly controlled hypertension, atrial fibrillation, or ischemia) may precipitate an AHF episode. For patients with a substrate of compensated or stable chronic HF, medical or dietary nonadherence, drugs such as nonsteroidal antiinflammatory agents or thiazolidinediones, and infectious processes are all common triggers for decompensation.
Regardless of the substrate or initiating factors, a variety of “amplifying mechanisms” perpetuate and contribute to the episode of decompensation. These include neurohormonal and inflammatory activation, ongoing myocardial injury with progressive myocardial dysfunction, worsening renal function, and interactions with the peripheral vasculature, all of which may contribute to the propagation and worsening of the AHF episode.
Systemic or pulmonary congestion often due to a high ventricular diastolic pressure dominates the clinical presentation of most patients hospitalized for AHF. Congestion can be seen as a final common pathway producing clinical symptoms leading to hospitalization. An oversimplified view of AHF pathophysiology is that gradual increases in intravascular volume lead to symptoms of congestion and clinical presentation, and normalization of volume status with diuretic therapy results in restoration of homeostasis. Although some data suggest that increases in body weight often precede decompensation and hospitalization for HF, careful studies using implantable hemodynamic monitors suggest that increases in invasively measured LV filling pressures can occur without substantial changes in body weight. These observations have led to increasing interest in the concept of volume redistribution and the dynamic role of the vasculature as a contributing mechanism to decompensation in heart failure (discussed in more detail in “Vascular Mechanisms” section below).
One important concept is the distinction between “clinical congestion” and “hemodynamic congestion.” Although patients present with signs and symptoms of systemic congestion such as dyspnea, rales, elevated jugular venous pressure (JVP), and edema, this state is often preceded by “hemodynamic congestion,” defined as high ventricular diastolic pressures without overt clinical signs. Similarly, clinical congestion may resolve with treatment but hemodynamic congestion may persist, leading to a high risk of rehospitalization. It has been postulated that hemodynamic congestion may contribute to the progression of HF because it may result in increased wall stress as well as in renin-angiotensin-aldosterone system (RAAS) and sympathetic nervous system (SNS) activation. This may trigger a variety of molecular responses in the myocardium, including myocyte loss and increased fibrosis. The natriuretic peptides (see Chapter 47 ), which are the intrinsic counter-regulatory hormone in heart failure, may have abnormal processing that leads to diminished biologic activity in patients with advanced heart failure. In addition, elevated diastolic filling pressures may decrease coronary perfusion pressure, resulting in sub-endocardial ischemia that may further exacerbate cardiac dysfunction. Increased LV filling pressures can also lead to acute changes in ventricular architecture (more spherical shape), contributing to worsening mitral regurgitation. These mechanisms also play an important role in pathologic remodeling of the ventricle, a chronic process that may be accelerated by each episode of decompensation. Consistent with this paradigm is the well-established clinical observation that each hospitalization for AHF heralds a substantial worsening of the long-term prognosis, an effect that appears additive with recurrent hospitalizations. Data from studies with implantable hemodynamic monitors have confirmed that chronically elevated filling pressures (i.e., hemodynamic congestion) are associated with increased risk of future events. With the recognition of congestion as the most common aspect of AHF presentation, there has been a formal attempt to better assess and quantitate congestion in heart failure.
Although a variety of extra-cardiac factors play important roles in AHF, impairments of cardiac function (systolic, diastolic, or both) remain central to our understanding of this disorder (see also Chapter 46 ). Changes in systolic function and decreased arterial filling can initiate a cascade of effects that are adaptive in the short term but maladaptive when elevated chronically, including stimulation of the SNS and RAAS. Activation of these neurohormonal axes leads to vasoconstriction, sodium and water retention, volume redistribution from other vascular beds, increases in diastolic filling pressures, and clinical symptoms. In patients with underlying ischemic heart disease, initial defects in systolic function may initiate a vicious cycle of decreasing coronary perfusion, increased myocardial wall stress, and progressively worsening cardiac performance. Increased LV filling pressures and changes in LV geometry can worsen functional mitral regurgitation, further decreasing cardiac output.
Importantly, abnormalities in diastolic function are present in heart failure patients regardless of EF. The impairment of the diastolic phase may be related to passive stiffness, abnormal active relaxation of the left ventricle, or both. Hypertension, tachycardia, and myocardial ischemia (even in the absence of CAD) can further impair diastolic filling. All of these mechanisms contribute to higher LV end-diastolic pressures, which are reflected back to the pulmonary capillary circulation. Diastolic dysfunction alone may be insufficient to lead to AHF, but it serves as the substrate on which other precipitating factors (such as atrial fibrillation, CAD, or hypertension) lead to decompensation. One underappreciated aspect of myocardial function in AHF relates to the interdependence of the left and right ventricles. Because of the constraints of the pericardial space, distention of either ventricle due to increased filling pressures can result in direct impingement of diastolic filling of the other ventricle. This may be particularly operative in clinical scenarios leading to abrupt failure of the right ventricle (such as pulmonary embolism or right ventricular (RV) infarction), resulting in diminished filling of the left ventricle and arterial hypotension.
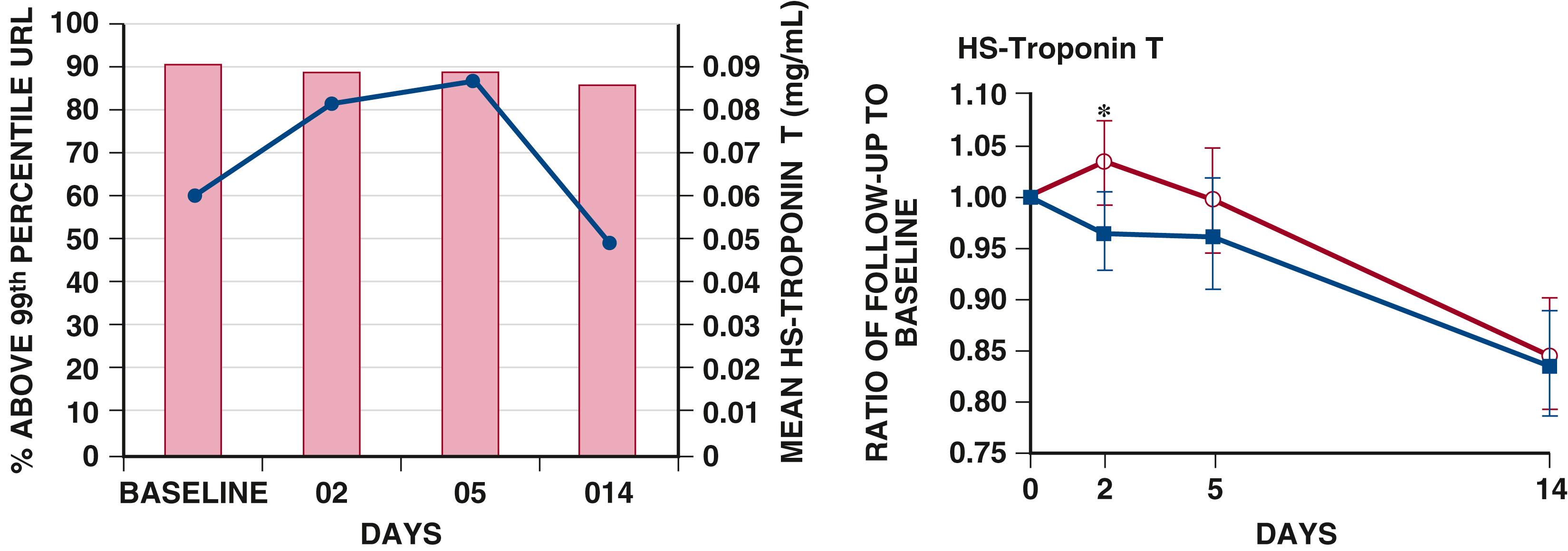
The availability of increasingly sensitive assays for circulating cardiac troponins has led to evolution of our understanding of the role of myocardial injury in AHF pathophysiology. Data from both registries and clinical trial populations indicate that circulating cardiac troponins are elevated in a large proportion of patients with AHF, even in the absence of clinically overt myocardial ischemia. , In a representative analysis of data from the RELAX-AHF study using a highly sensitive assay, 90% of patients enrolled had a troponin T level above the 99th percentile upper reference limit at baseline, and troponin elevation was associated with post-discharge outcomes out to 180 days ( Fig. 49.4 ).
The precise mechanisms mediating myocardial injury in AHF are poorly defined, but increased myocardial wall stress, decreased coronary perfusion pressure, increased myocardial oxygen demand, endothelial dysfunction, activation of the neurohormonal and inflammatory pathways, platelet activation, and altered calcium handling may all contribute to myocyte injury even in the absence of epicardial CAD. Specific therapeutic interventions that may increase myocardial oxygen demand (such as positive inotropic agents) or decrease coronary artery perfusion pressure (such as some vasodilators) may exacerbate myocardial injury and further contribute to the cycle of decompensation. Whether avoidance of myocardial injury is a specific target for therapy in AHF remains a subject of active investigation.
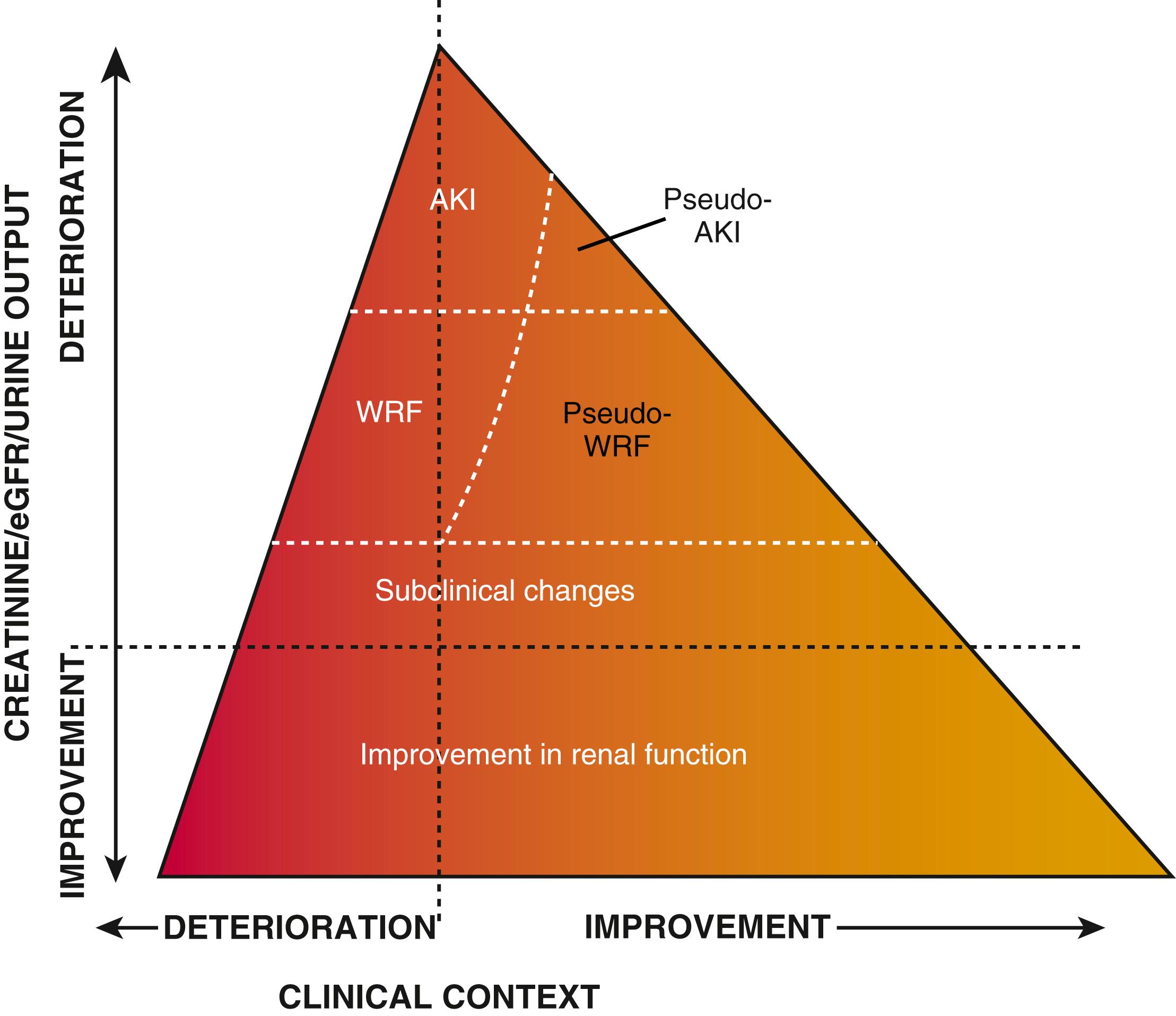
The kidney plays two fundamental roles relative to the pathophysiology of HF: it modulates loading conditions of the heart by controlling intravascular volume and is responsible for neurohormonal outputs (i.e., the RAAS system). Abnormalities of renal function are extremely common in patients with AHF and may be underestimated by creatinine alone. Baseline chronic kidney disease is an established risk factor for poor outcomes in AHF (see Risk Stratification section later), but our understanding of the implications of changes in renal function during AHF treatment has continued to evolve. The term “cardio-renal syndrome” has been increasingly used to describe pathologic interactions between the cardiac and renal axes in the setting of heart failure. Although specific definitions and nomenclature have varied, in the context of AHF the cardio-renal syndrome describes the clinical situation of worsening measures of renal function in the setting of persistent congestion. This clinical scenario has been associated with poor outcomes in a variety of observational studies. Multiple studies have investigated the pathophysiology and risk factors for this phenomenon, which is related to an intricate interplay of patient characteristics (age), comorbidities (baseline renal function as assessed by glomerular filtration rate [GFR], diabetes mellitus, hypertension), neurohormonal activation (especially of RAAS and SNS), and hemodynamic factors (central venous congestion, and less frequently arterial underfilling with renal hypoperfusion), as well as other factors such as activation of inflammatory cascades and oxidative stress. Although often assumed to be related to low cardiac output and renal blood flow, careful hemodynamic studies have repeatedly confirmed that the strongest predictor of worsening renal function in heart failure patients relates to elevated central venous pressure (CVP), which is reflected back to the renal veins and leads directly to changes in GFR. Importantly, recent data have emphasized the importance of evaluating changes in renal function in the context of the overall clinical picture. Worsening renal function in the setting of ongoing clinical improvement is generally reflective of successful decongestion and does not portend a poor prognosis ( Fig. 49.5 ). Although there has been substantial interest in the utility of newer biomarkers to identify episodes of frank kidney injury prior to changes in markers of renal function, the clinical utility of these markers remains uncertain. A detailed classification system for understanding the interplay between cardiac performance and renal function has been proposed and provides a framework for understanding the complex pathophysiology underlying the cardio-renal syndrome.
While abnormalities in cardiac function are central to the pathogenesis of AHF, there is increasing appreciation for the importance of the peripheral vasculature in this disorder. Abnormalities of endothelial function related to nitric oxide dependent regulation of vascular tone are well described in heart failure. Arterial stiffness, which is related to but distinct from blood pressure, increases cardiac loading conditions and is associated with incident heart failure and worse outcomes. Peripheral vasoconstriction in the setting of AHF redistributes blood centrally, increasing pulmonary venous congestion and edema. As noted earlier, elevated CVP reduces renal function, resulting in greater fluid retention, which further elevates venous pressures. Peripheral arterial vasoconstriction increases afterload, LV filling pressures, and post-capillary pulmonary venous pressures, resulting in worsening of pulmonary edema and dyspnea. This increased afterload causes greater ventricular wall stress and increased myocardial ischemia and cardiac arrhythmias. Abnormal vascular compliance also predisposes these patients to marked blood pressure liability with relatively minor changes in intravascular volume, causing precipitous increases in afterload and ultimately in LV filling pressures resulting in pulmonary congestion. The effects of this vascular abnormality are amplified by LV diastolic dysfunction.
The clinical observation that vasodilator treatment can improve dyspnea in many acutely hypertensive patients without significant diuresis has led to the concept that afterload-contractility mismatch can lead to increased diastolic filling pressures in the setting of minimal total body volume changes. Similarly, the recognition of the large capacitance of the venous (in particular the splanchnic circulation) system has led to increased interest in volume shifts from the “venous reservoir” into the effective circulatory volume as a potentially important and under-recognized mechanism in AHF. These shifts can be mediated by SNS activation, and this has been proposed as a potential explanation between the apparent disconnect between changes in filling pressures and changes in body weight during chronic hemodynamic monitoring. Whether fluid shifts involving this venous reservoir can be modulated therapeutically is a subject of active investigation.
Although elevations of circulating neurohormones are well-documented in patients with AHF, the precise role of neurohormonal activation in the pathophysiology of AHF remains to be fully delineated. Increased plasma concentrations of norepinephrine, plasma renin activity, aldosterone, and endothelin-1 (ET-1) have all been reported in patients with AHF—all of these axes are associated with vasoconstriction and volume retention, which could contribute to myocardial ischemia and congestion, thus exacerbating cardiac decompensation. Inflammatory activation and oxidative stress may also play a role. Pro-inflammatory cytokines such as tumor necrosis factor-alpha and interleukin-6 are elevated in patients with AHF and have direct negative inotropic effects on the myocardium as well as increasing capillary permeability and inducing endothelial dysfunction. , In addition to direct effects, this activation stimulates the release of other factors, such as the potent pro-coagulant tissue factor and ET-1, which can lead to further myocardial suppression, disruption of the pulmonary alveolar capillary barrier, and increased platelet aggregation and coagulation (potentially worsening ischemia).
The initial evaluation of the patient with AHF focuses on the following critical aspects: (1) establishing a definitive diagnosis of AHF as rapidly and efficiently as possible; (2) emergent treatment for potentially life-threatening conditions (e.g., shock, respiratory failure); (3) identifying and addressing any relevant clinical triggers or other condition requiring specific treatment (e.g., acute coronary syndrome [ACS], acute pulmonary embolism, etc.); (4) risk stratification in order to triage patient to appropriate level of care (e.g., intensive care unit [ICU], telemetry unit, observation unit); and (5) defining the clinical profile of the patient (based on blood pressure, volume status, and renal function) in order to rapidly implement the most appropriate therapy. A proposed flow diagram for the initial evaluation of patients with suspected AHF is shown in Figure 49.6 .
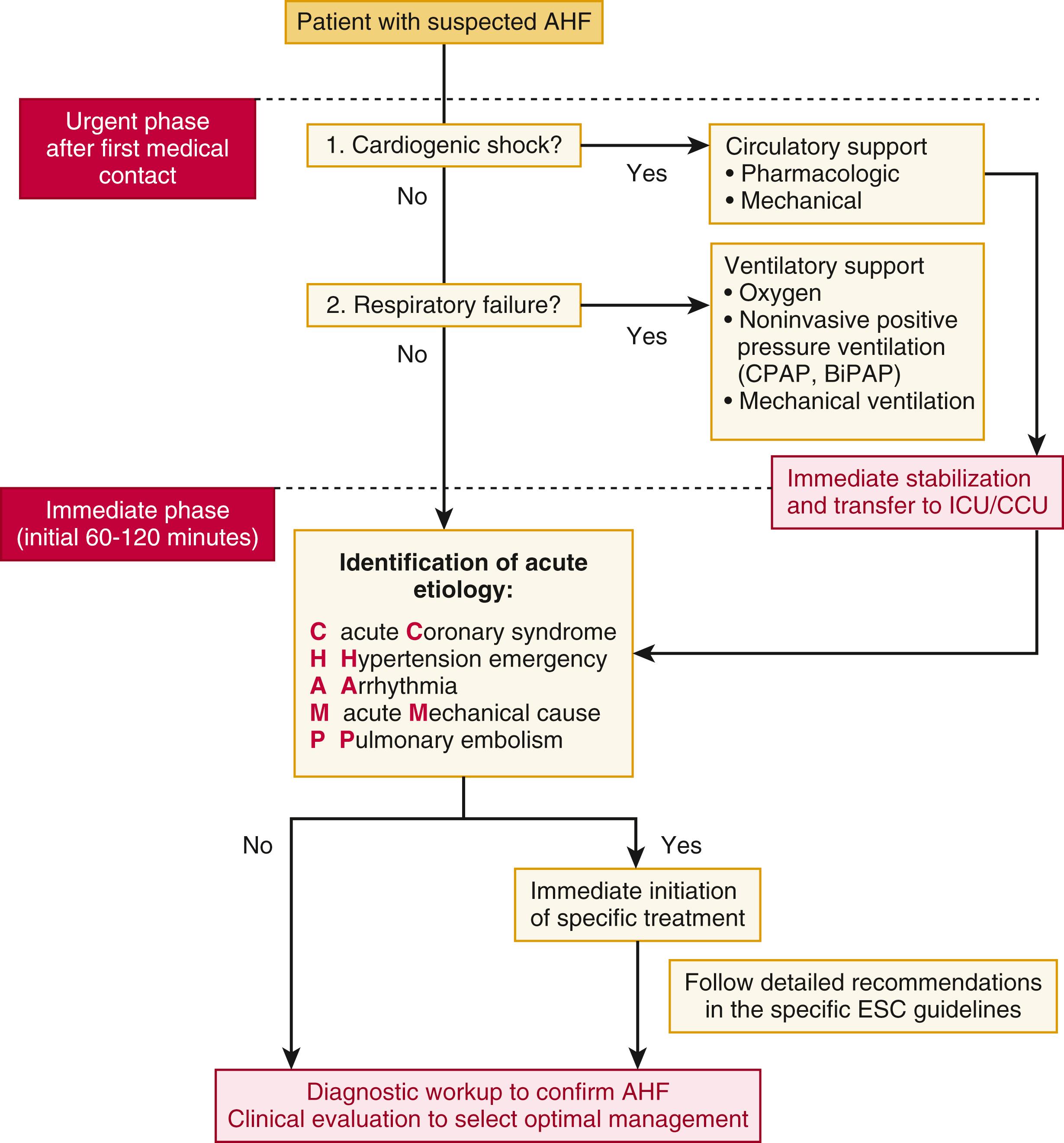
The inherent heterogeneity of AHF makes the development of a comprehensive classification scheme difficult, and no single classification system has garnered universal acceptance. There are several relevant domains to consider in classifying patients with AHF ( Fig. 49.7 ). These include underlying substrate (i.e., whether there is a prior history of structural heart disease or a background of chronic HF), severity (from mild symptoms to cardiogenic shock), acuity (gradual onset vs. sudden/acute onset), and triggers (which may be readily apparent or unknown). Each of these concepts is discussed briefly below.
New-onset or de novo heart failure makes up about 20% of hospitalizations for AHF. These patients may have no prior history of cardiovascular disease or risk factors (e.g., acute myocarditis), but more commonly, they have a background of risk factors for HF (stage A heart failure according to the American College of Cardiology/American Heart Association [ACC/AHA] guidelines) or preexisting structural heart disease (stage B heart failure according to the ACC/AHA guidelines) (see also Chapter 48, Chapter 50 ). Many of these patients with de novo heart failure develop AHF in the setting of ACS. The majority of AHF patients have a history of preexisting chronic heart failure. These patients often have a less dramatic clinical presentation because the chronic nature of the disorder has allowed for recruitment of compensatory mechanisms and remodeling (e.g., increased pulmonary lymphatic capacity). Additionally, these patients are typically already being treated with neurohormonal antagonists and loop diuretics, such that neurohormonal activation may be less profound but diuretic resistance may be more common. In patients with AHF with a background of chronic HF, these patients can be further sub-classified by ejection fraction (i.e., HFrEF, HFmrEF, HFpEF) or by cause (i.e., ischemic, nonischemic, etc.) although these factors less often impact acute management of the AHF episode.
Patients with AHF may range from modestly decompensated patients who require intensification of oral diuretics in the outpatient setting to patients with frank cardiogenic shock. Severity of presentation may be disconnected from severity of background HF. Patients with mild hypertensive heart disease may present in profound respiratory distress requiring intubation, whereas patients with very advanced chronic heart failure may present with more subtle symptoms such as fatigue and early satiety. The most severe presentations of cardiogenic shock have signs and symptoms of organ hypoperfusion despite adequate preload. Systolic blood pressure (SBP) is often (although not always) decreased, and evidence of frank or impending end-organ dysfunction (renal, hepatic, CNS) is common. Cardiogenic shock is relatively uncommon (4% of AHFS presentations in EuroHeart Failure Survey II [EHFS II]) in broad community registries but more common in tertiary care settings.
The time course of worsening symptoms is a key component of the history for many forms of acute cardiovascular disease and AHF is no exception. Patients may develop symptoms very suddenly (over minutes) or very gradually (over weeks or longer). As noted above, the acuity of symptoms may not be aligned with the severity of heart failure and the long-term prognosis but has clear implications for the immediacy of therapy needed for stabilization. The fact that many patients may have slowly developing symptoms over days to weeks presents the possibility that early intervention with intensified therapy may prevent some hospitalizations.
AHF may be triggered by very clear precipitants or alternatively the reason for decompensation may be obscure. In the OPTIMIZE-HF registry, 61% of enrolled subjects had an identifiable clinical precipitant, with pulmonary processes (15%), myocardial ischemia (15%), and arrhythmias (14%) being the most common. More than one precipitant was identified in a substantial minority of the study population. Of the identified triggers, worsening renal function was associated with the highest in-hospital mortality (8%), whereas nonadherence to diet or medication or uncontrolled hypertension had a much better prognosis (<2% in-hospital mortality for each). Infection with the novel coronavirus Sars-CoV-2 is a rapidly evolving trigger of AHF that is discussed in detail elsewhere (see Chapter 94 ). In patients with a background of HFpEF, acute hypertension is a common trigger for decompensation, and may overlap with the syndrome of hypertensive emergency. Acute hypertension may be triggered by a high sympathetic tone related to dyspnea and accompanying anxiety (reactive hypertension) or acute hypertension with accompanying changes in afterload may be a trigger for decompensation. Both of these mechanisms may be operative in a given patient, and cause and effect relationships may be difficult to discern. Frank pulmonary edema with evident rales and florid congestion on chest x-ray is much more common in this group of patients than in those with more gradual onset of symptoms, likely related to difference in LV compliance, acuity of pressure changes, and pulmonary lymphatic capacity. Although often strikingly ill at the time of initial presentation with hypoxemia and the possible need for noninvasive ventilation (NIV) or even intubation, this group tends to respond well to therapy and have lower in-hospital mortality.
The most common reasons for patients to seek medical care for AHF are symptoms related to congestion. A list of the most common presenting symptoms is provided in Table 49.2 . Dyspnea is the most common symptom and is present in over 90% of patients presenting with AHF. The duration and time course of symptom onset can vary markedly as noted above. The sensation of dyspnea is a complex phenomenon that is influenced by multiple physiologic, psychological, and social factors, and can vary dramatically between patients. Patients may also present with symptoms related to systemic venous congestion, including peripheral edema, weight gain, early satiety, and increasing abdominal girth. Importantly, atypical symptoms can predominate, especially in older patients, where fatigue, depression, altered mental status, and sleep disruptions may be the primary complaints. Bendopnea, or the sensation of dyspnea on bending over, is a commonly reported symptom that has recently been validated experimentally.
| Symptoms | Signs |
|---|---|
| Predominantly related to volume overload | |
| Dyspnea (exertional, paroxysmal nocturnal dyspnea, orthopnea, or at rest); cough; wheezing | Rales, pleural effusion |
| Foot and leg discomfort | Peripheral edema (legs, sacral) |
| Abdominal discomfort/bloating; early satiety or anorexia | Ascites/increased abdominal girth; right upper quadrant pain or discomfort; hepatomegaly/splenomegaly; scleral icterus |
| Increased weight | |
| Elevated jugular venous pressure, abdominojugular reflux | |
| Increasing S 3 , accentuated P 2 | |
| Predominantly related to Hypoperfusion | |
| Fatigue | Cool extremities |
| Altered mental status, daytime drowsiness, confusion, or difficulty concentrating | Pallor, dusky skin discoloration, Hypotension |
| Dizziness, pre-syncope, or syncope | Pulse pressure (narrow)/proportional pulse pressure (low) |
| Pulsus alternans | |
| Other signs and symptoms of AHF | |
| Depression | Orthostatic hypotension (hypovolemia) |
| Sleep disturbances | S 4 |
| Palpitations | Systolic and diastolic cardiac murmurs |
Despite advances in diagnostics technology, biomarkers, and imaging, heart failure remains a clinical diagnosis and the physical examination continues to play a fundamental role (see Chapter 13, Chapter 48 ). A useful framework in the bedside evaluation of patients with AHF is that developed by Stevenson and colleagues, which focuses on the adequacy of perfusion (“cold” vs. “warm”) and congestion at rest (“wet” vs. “dry”). While this framework does not completely encompass the heterogeneity of AHF, it does focus the evaluation on two critical aspects that will significantly influence both prognosis and choice of treatments.
Assessing blood pressure is a critical part in the evaluation of patients with AHF; hypotension is one of the strongest predictors of poor outcomes and helps to define appropriate therapeutic interventions. SBP is typically normal or elevated in patients with AHF, with almost 50% presenting with SBP greater than 140 mm Hg. The combination of underlying hypertension and the marked increase in sympathetic stimulation that accompanies AHF can result in elevations of SBP consistent with hypertensive urgencies or emergencies (12% of patients had an SBP over 180 mm Hg on admission). Patients with very low SBP are uncommon, with only 2% of patients in ADHERE presenting with an SBP less than 90 mm Hg. Although blood pressure is generally related to cardiac output and the state of organ perfusion, it is important to recognize that hypotension and hypoperfusion are not synonymous. Patients with systemic hypoperfusion may present with normal blood pressure, and similarly patients with advanced forms of heart failure may have chronically low blood pressure not associated with acute hypoperfusion. Pulse pressure (the difference between systolic and diastolic blood pressure) is a useful measure that is an indirect marker of cardiac output. A low pulse pressure is a marker of a low cardiac output and confers an increased risk in patients admitted with AHF. A high pulse pressure may alert the physician to a high output state including the possibility of unrecognized thyrotoxicosis, aortic regurgitation, or anemia.
The JVP is a barometer of systemic venous hypertension and is the single most useful physical examination finding in the assessment of patients with AHF. The accurate assessment of the JVP is highly dependent on examiner skill. The JVP reflects the right atrial pressure, which typically (although not always) is an indirect measure of LV filling pressures. JVP may not reflect LV filling pressures in isolated RV failure (e.g., from pulmonary hypertension or RV infarct), and significant tricuspid regurgitation can complicate the assessment of the JVP because the large “CV wave” of tricuspid regurgitation can lead to its overestimation.
Rales or inspiratory crackles are the most common physical examination finding and have been noted in 66% to 87% of patients admitted for AHF. However, rales are often not heard in patients with a background of chronic heart failure and pulmonary venous hypertension, due to increased lymphatic drainage, reinforcing the important clinical pearl that the absence of rales does not necessarily imply normal LV filling pressures. Cool extremities with palpable peripheral pulses suggest decreased peripheral perfusion consistent with a marginal cardiac index, marked vasoconstriction, or both. Of note, the temperature should be assessed at the lower leg as opposed to the foot, and this assessment is relative to the temperature of the examiner’s hands.
Peripheral edema is present in up to 65% of patients admitted with AHF and is less common in patients presenting with predominantly low-output heart failure or cardiogenic shock. As with rales, the presence of edema has a reasonable positive predictive value for AHF but a low sensitivity, so its absence does not exclude that diagnosis. Edema due to AHF is usually dependent, symmetric, and pitting. It is estimated that a minimum of 4 liters of extracellular fluid is accumulated to produce clinically detectable edema.
The natriuretic peptides are a family of important counter-regulatory hormones in HF with vasodilatory and other effects (see Chapter 47, Chapter 48 ). In the context of AHF, both brain natriuretic peptide (BNP) and N-terminal pro-BNP (NT-proBNP) have been shown to play an important role in the differential diagnosis of patients presenting in the emergency department with dyspnea, and are now Class I recommendations in clinical guidelines. In diagnostic testing, natriuretic peptides have greater negative predictive value (i.e., the ability to rule out heart failure as a cause of dyspnea) than positive predictive value (i.e., the ability to definitively identify a diagnosis of heart failure as the cause of dyspnea). As with all biomarker testing, false-positives (e.g., due to MI or pulmonary embolism) and false-negatives (primarily due to obesity, which results in lower natriuretic peptide levels for a given degree of heart failure) may occur. Although natriuretic peptide levels tend to be lower in patients with HFpEF than those with reduced systolic function, natriuretic peptide testing cannot reliably distinguish HFpEF from HFrEF in an individual patient. As noted previously, measurement of cardiac troponin is frequently elevated in patients presenting with AHF, and elevated levels are associated with worse in-hospital and post-discharge outcomes. Assessment of cardiac troponin in patients with AHF is now a Class I recommendation in clinical guidelines and serves to both establish prognosis as well as inform the likelihood of concurrent ACS. It is important to note that elevation of troponin in the context of a typical AHF hospitalization without clinical evidence of ACS is not synonymous with a Type II MI based on the updated fourth universal MI definition.
Assessment of renal function is a critical component in the management of patients with AHF. Estimated glomerular filtration rates (eGFRs) should be calculated because serum creatinine may underestimate the degree of renal dysfunction, especially in older adults. Blood urea nitrogen (BUN) is more directly related to the severity of AHF than creatinine, as it integrates both renal function and neurohormonal activation in AHF. A wide variety of other biomarkers, including ST2, galectin 3, and GDF15, have been evaluated in patients with AHF, but none are currently recommended for routine use in patients with AHF. In patients in whom the diagnosis of AHF is uncertain, testing to establish alternative causes (e.g., D-dimer to evaluate for pulmonary embolism or procalcitonin to evaluate for evidence of infection) may be useful.
Chest radiography is commonly performed at the time of presentation in patients with dyspnea and is a fundamental test in the evaluation of patients with suspected AHF. In the ADHERE registry, 90% of patients underwent chest radiography during hospitalization and there was evidence of congestion in over 80% of these patients. In patients with a background of chronic heart failure and/or slow onset of symptoms, evidence of congestion on chest x-ray may be subtle and frank pulmonary edema is often absent despite substantially elevated filling pressures.
The electrocardiogram (ECG) is another standard diagnostic test that is appropriate in all patients presenting with AHF (see Chapter 14 ). Careful attention for ECG changes suggestive of ischemia is of importance because troponin elevation is common in AHF regardless of cause. Arrhythmias are also a common trigger for AHF, and atrial fibrillation is present in 20% to 30%.
Utilization of echocardiography (see Chapter 16 ) is very high in patients with AHF—over 80% of patients in EHFS II had an echocardiogram performed during the index hospitalization. An echocardiogram is generally the single most useful test in evaluating the cause in the patient with AHF. Echocardiography can assess global systolic and diastolic function, regional wall motion abnormalities, valvular function, hemodynamics including estimates of filling pressures and cardiac output, and pericardial disease. The tissue Doppler ratio of peak early diastolic trans-mitral blood flow velocity (E) to the peak early diastolic mitral annular tissue velocity (E a ) (E:E a ratio) has been shown to be additive to BNP measures in diagnosing AHF patients presenting with dyspnea. An E:E a ratio of greater than 15 predicts a pulmonary capillary wedge pressure (PCWP) greater than 15 mm Hg and has been demonstrated to be accurate in the emergency room and intensive care settings.
Risk stratification can serve as important clinical tools by helping to identify those patients at both ends of the spectrum of risk; patients who are at very high risk may be observed more closely or treated more intensively, whereas patients at low risk may avoid hospitalization altogether or need less rigorous follow-up and monitoring. A variety of predictive models have been developed in AHF, which can generally be divided into two groups: those focused on in-hospital mortality, and those focused on post-discharge events (death or rehospitalization). Commonly used predictive models are summarized in Table 49.3 . Although individual models differ, several variables occur repeatedly in risk models in a variety of settings: older age, lower blood pressure, higher heart rate, higher BUN and creatinine, and hyponatremia. In settings where natriuretic peptides are available, they are also powerful predictors of long-term risk, although they may be stronger predictors at hospital discharge than on admission.
| Population | Sample Size | Endpoints | C-Index | Key Predictors | |
|---|---|---|---|---|---|
| ADHERE | US registry | 65275 | In-hospital mortality | n/a | ↑BUN, ↑Cr, ↓SBP, ↑age |
| OPTIMIZE in hospital | US registry | 48612 | In-hospital mortality | 0.75 | ↑age, ↑HR, ↓SBP, ↑Cr, ↓Na |
| OPTIMIZE post discharge | US registry | 4402 | 60–90-day outcomes | 0.72 | ↑age, ↑Cr, reactive airway dz, liver dz, ↓SBP, ↓Na, depression |
| PROTECT | Global RCT | 2015 | In-hospital outcomes | 0.67 | ↑BUN, ↑RR, ↓SBP, ↑HR, ↓albumin |
| EFFECT | Canadian population data | 4031 | 30-day and 1-year mortality | 0.80 | ↑age, SBP, ↑RR, ↑BUN, ↓Na |
| OPTIME-CHF | Global RCT | 949 | 60-day mortality | 0.77 | ↑age, ↑BUN, ↓Na, ↓Hb |
| ESCAPE | US RCT | 423 | 6-month mortality | 0.76 | ↑BNP, ↑age, ↑BUN, Na, mechanical ventilation, ↑diuretic dose |
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here