Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Cystic lesions of the pancreas include malignant and benign processes and may or may not cause clinical symptoms. The risk of malignancy in symptomatic cyst can be high, whereas asymptomatic cysts can be benign, malignant, or premalignant. Pancreatic cystic neoplasms account for 10% to 15% of pancreatic cysts, and less than 1% of primary pancreatic malignancies.
Currently, CT and magnetic resonance imaging (MRI) are used in the assessment of pancreatic cystic lesions. CT is widely available, requires less scan time, and is the most accessible. MRI is able to identify communication of the cystic lesion with the main pancreatic duct, although, with currently available thin-section imaging, CT has a similar capacity. 2-[ 18 F] fluoro-2-deoxy-D-glucose-positron emission tomography (PET)/CT has been used to differentiate malignant from benign cystic lesions based on metabolic activity.
Cross-sectional imaging can characterize a cystic lesion based on the imaging findings. Currently, the available imaging modalities for detecting and diagnosing cystic lesions are contrast-enhanced CT (CECT), ultrasound (US), MRI, and PET/CT.
CECT and MRI are not operator-dependent, and can visualize the entire pancreas without difficulty. Endoscopic US (EUS) is operator- and patient-dependent, and EUS-guided fine needle aspiration (EUS-FNA) plays a complementary role in accomplishing this difficult task by providing additional real-time assessment and cystic content for further analysis. In combination with a Doppler device, this approach allows tissue sampling by FNA, avoiding the major vessels. PET/CT is an evolving modality for detecting malignancy in cystic pancreatic lesions. It has been shown to detect invasive cancer in cystic lesions; however, it cannot identify carcinoma in situ . This modality can help detect distant metastases in the setting of invasive cancer. Pancreatic cystic lesions may be identified by transabdominal US. However, this approach is limited in patients with a large body habitus, and overlying bowel gas may obscure lesions. Calcifications within the lesion are better seen on CECT; septations may or may not be seen, depending on the size of the lesion and the thickness of the septations. MRI can better identify septations and hemorrhagic contents of the cystic lesions; however, detection of mucin is equivocal. EUS can be used to evaluate the cyst, and concomitant cytological analysis of cyst fluid can be obtained by EUS-FNA. The pancreatic cyst fluid analysis should include staining for mucin, and measurement of the levels of amylase, lipase, and various tumor markers such as carcinoembryonic antigen (CEA) in the cyst fluid.
Cystic lesions of the pancreas can be divided into two categories: primary cystic lesions, which include pseudocysts, serous cystadenomas, various mucin-containing cysts (mucinous nonneoplastic cysts [MNCs], mucinous cystic neoplasm [MCNs], mucinous cystadenocarcinomas, and intraductal papillary mucinous neoplasms), pseudopapillary tumors of the pancreas and lymphoepithelial cysts, and secondary cystic lesions, which comprise various solid neoplasms undergoing cystic changes (ductal adenocarcinoma with cystic features and cystic neuroendocrine tumors). In this chapter we will discuss primary cystic lesions of the pancreas.
Pseudocysts make up the majority of all cystic lesions of the pancreas, whereas the rest are cystic tumors and true cysts. Hydatid cysts of the pancreas are rare, and should be considered as a differential diagnosis of a cystic pancreatic lesion in countries where hydatid disease is endemic. Pancreatic primary cystic tumors fall into one of three major groups: serous tumors (including serous cystadenoma and cystadenocarcinoma), mucinous tumors (including MCNs, mucinous cystadenocarcinomas, intraductal papillary adenomas, and intraductal papillary adenocarcinoma), and solid pseudopapillary tumors (SPTs). The majority of cystic pancreatic tumors are asymptomatic and slow-growing. When patients are symptomatic, the symptoms are due to mass effect, and are usually vague and poorly localized. Intraductal papillary mucinous neoplasms (IPMNs) may present with pain that can mimic chronic pancreatitis. On cross-sectional imaging, they can be characterized as simple (containing no internal nodularity) or complex (containing internal septations or mural nodules). They can also be classified as unilocular or multilocular, depending on the number of locules present.
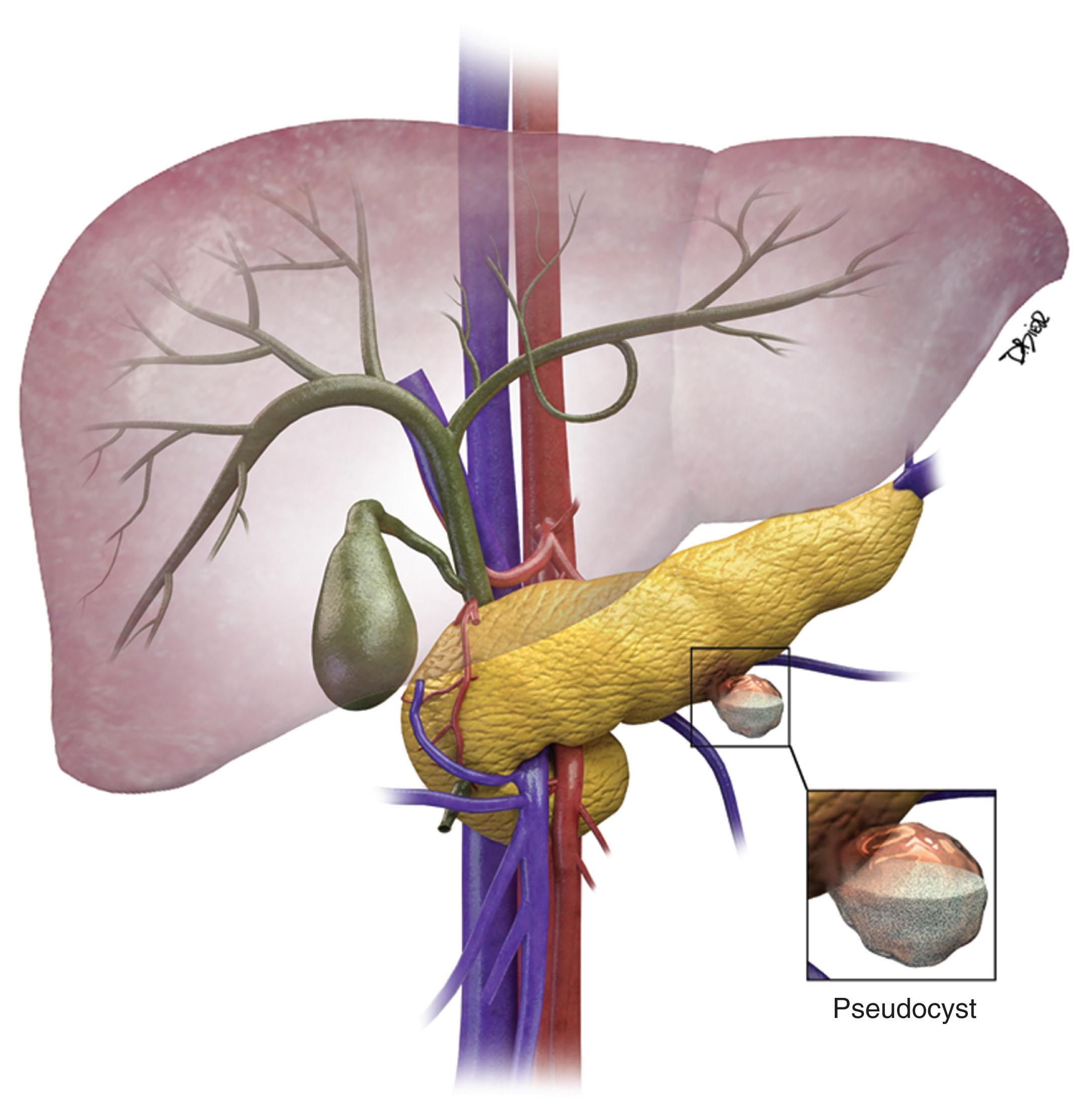
The majority of cystic lesions found in the pancreas are pseudocysts, which comprise 70% of all cystic lesions. They lack an epithelial lining and have a fibrous capsule. Pancreatic pseudocysts occur after an episode of pancreatitis and are caused by leakage of pancreatic enzymes, such as amylase and lipase, which cause fat necrosis and hemorrhage because of erosion of adjacent vessels.
Patients with pancreatitis may present with nausea, vomiting, abdominal pain, or sepsis because of infection or obstructive jaundice. Because there is leakage of pancreatic enzymes, fistulation into nearby structures, such as the common bile duct, esophagus, and pleura, can occur. Pancreatic pseudocysts seldom demonstrate internal hemorrhage, but this finding is associated with increased mortality. Pseudocysts can erode adjacent vessels and cause pseudoaneurysms, which may rupture, leading to hemorrhage. The most common arteries involved are the splenic, gastroduodenal, and superior pancreaticoduodenal arteries. Pseudocysts are commonly seen in males, are usually are seen in alcoholics, and can be associated abdominal trauma.
On cross-sectional imaging ( Figs. 13.1 and 13.2 ), pseudocysts usually appear as unilocular lesions without internal septations or mural nodules. They do communicate with the main pancreatic duct. These cysts on imaging have an irregular wall in the early stages, but tend to evolve and become well-marginated. On CECT, pseudocysts look like a fluid collection, which may have an imperceptible or a thick wall that may or may not enhance in the delayed phase images, as it is fibrous. Pseudocysts may have a high signal on T1-weighted (T1W) MRI attributed to blood products. On magnetic resonance cholangiopancreatography (MRCP), they may show communication with the main pancreatic duct. On US, they may appear as thin-walled, unilocular, anechoic masses with increased through transmission. Complicated cysts may have low-level internal echoes with a fluid-debris level, or can have internal septations. If calcification is present within the cyst wall, this may obscure the cyst. Cysts do not have enhancing nodules; if nodules are present, one must consider a mucinous cystic neoplasm in the differential. One clue that can be seen on CT or MRI is peripancreatic stranding, which is considered the hallmark of pancreatitis, and, when associated with a cystic lesion, is suggestive of a pseudocyst.
Treatment is usually conservative if the patient is asymptomatic. These cysts may be drained via a transperitoneal, retroperitoneal, transgastric, or transduodenal route if they are larger than 4 to 5 cm or if the patient is symptomatic. Sometimes cystogastrostomy or cystojejunostomy can be performed.
Depending on the suspicion, these tumors can be followed over a period of time, because a pseudocyst will resolve, and a cystic neoplasm may persist without change or growth. Although pseudocysts have no known malignant potential, many cystic neoplasms of the pancreas may mimic pseudocysts. The differential diagnosis of the cyst is mucinous cystic neoplasm, and if communication to the pancreatic duct is present one may consider an IPMN. To clinch the diagnosis of a pseudocyst, age, gender, and history of pancreatitis can help.
Pseudocysts are a sequela of pancreatitis, and are not lined by epithelium.
There is a known association with alcohol abuse.
On endoscopic ultrasound-guided fine needle aspiration the cyst fluid has high amylase and low carcinoembryonic antigen levels.
True cystic lesions of the pancreas are common, are lined by epithelium, and can be seen in patients with von Hippel-Lindau (vHL) disease, cystic fibrosis, or polycystic kidney disease. These cysts have a similar imaging appearance to the simple cysts seen in the liver and the kidneys.
Most simple cysts are found incidentally.
On CT simply cysts have Hounsfield unit (HU) values consistent with fluid. On MRI, they have a high signal on T2-weighted (T2W) imaging and a low signal on T1W imaging and do not show enhancement. They do not have mural nodules. On US, they are anechoic with posterior acoustic enhancement. The differential of these cysts includes MCN because there is no distinguishing radiological feature between the two. When these cysts are larger than 3 cm, EUS can be performed, and a biopsy can be obtained to exclude a mucinous neoplasm. These cysts are not usually resected.
These cysts require no treatment unless they are symptomatic.
Simple cysts are associated with von Hippel-Lindau disease, cystic fibrosis, and polycystic kidney disease.
They have a thin, imperceptible wall on computed tomography and appear as cysts on ultrasound.
On endoscopic ultrasound-guided fine needle aspiration they have low carcinoembryonic antigen and low amylase.
MNCs are common cystic tumors of the pancreas. It is speculated that they are caused by a developmental defect of the pancreas resulting in a focal cystic transformation of the duct system.
MNCs are lined by tall columnar epithelium that secretes mucin. Their maximum diameter ranges from 3 to 12 cm, they may be either unilocular or multilocular, and they may be filled with turbid or bloody fluid. These tumors do not have an ovarian stroma, which is a characteristic of MCNs. These cysts do not communicate with the main pancreatic duct, do not show cellularity, do not have papillary projections as seen in IPMNs, and are believed to be benign.
Clinical symptoms may include epigastric discomfort and pain. Obstructive jaundice can be caused by compression of the common bile duct by the cystic lesion when it occurs in the pancreatic head.
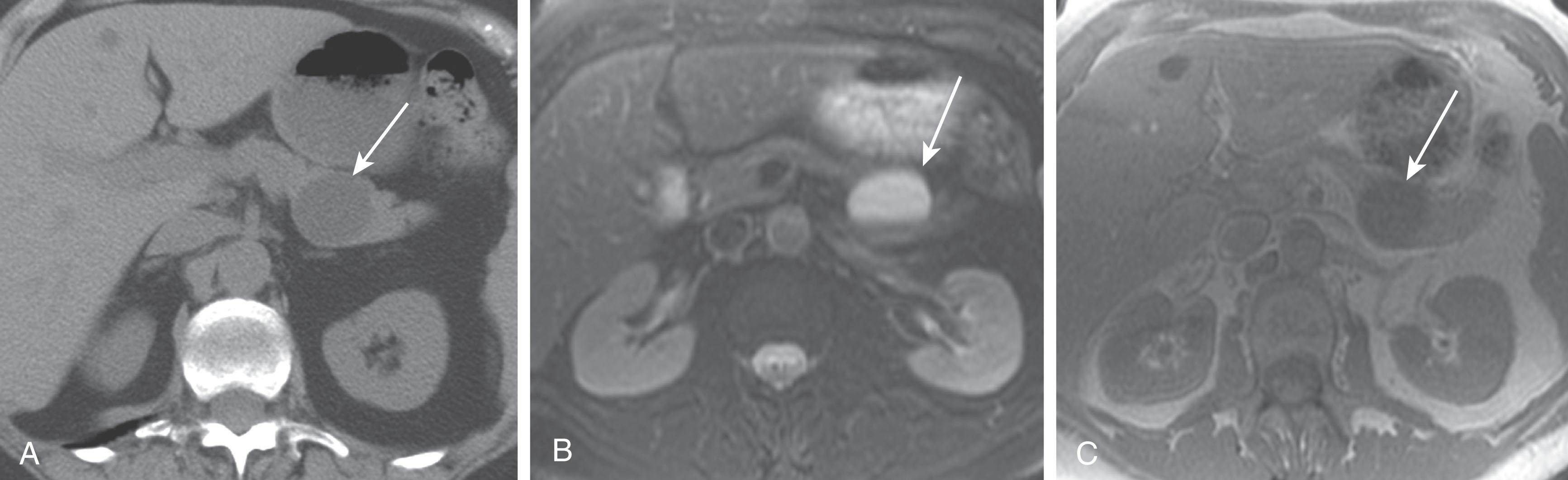
On imaging, MNCs ( Fig. 13.3 ) are usually small and unilocular, with thin septa. On MRI they have a signal intensity consistent with simple fluid, and on CECT their HU is less than 20. On imaging, MNCs cannot be easily differentiated from MCNs, especially if the mucinous cyst is large or has a thick wall. One of the important distinguishing factors is that MCNs are commonly seen in females, whereas MNCs have no sex predilection. If these cysts are larger than 2 cm, then EUS-FNA can be performed, and the cyst contents can be differentiated from those of other MCNs by immunohistochemical staining ( Table 13.1 ).
| SCA | MCN | IPMN | SPT | PSEUDO | |
|---|---|---|---|---|---|
| Prevalent age | Elderly women | Middle age | Elderly men | Young | Variable |
| Sex | Female > male | Female | Male > female | Female | Male > female |
| Alcohol abuse | No | No | No | No | Yes |
| Pancreatitis | No | No | Not uncommon | No | Yes |
| Location | Evenly | Body/tail | Head and tail | Evenly | Evenly |
| Malignant potential | Rare | Moderate/high | Low/high | Low | None |
Usually MNCs do not require treatment unless they become symptomatic because of internal hemorrhage or cause biliary obstruction, which is rare.
If an MNC occurs in the head of the pancreas, it can cause ductal obstruction, and should be followed at different time intervals depending on its size.
These tumors secrete mucin but do not have the ovarian stroma.
They do not communicate with the main pancreatic duct, but may cause biliary obstruction when in a critical location.
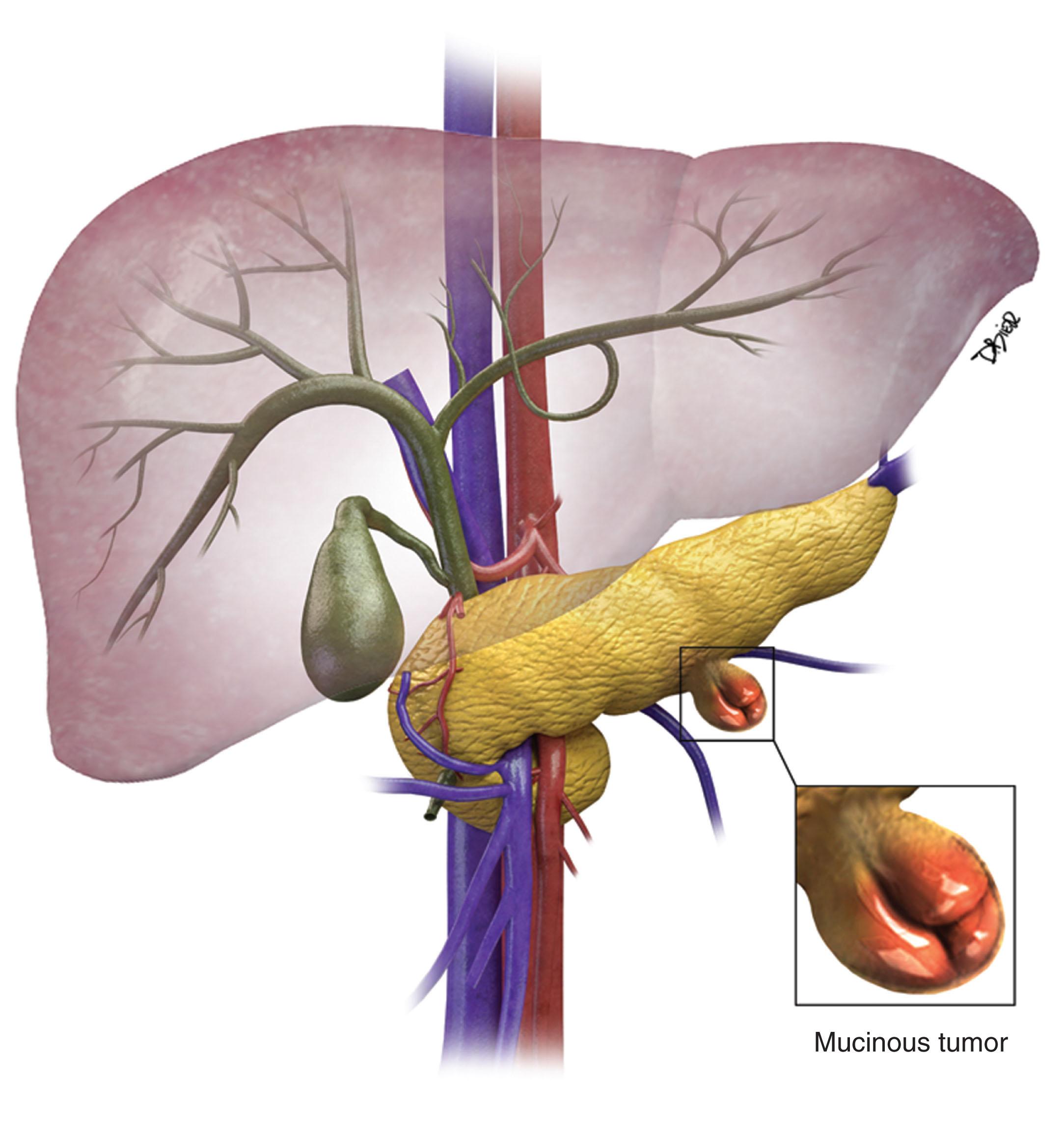
MCNs account for approximately 10% of pancreatic cystic neoplasms.
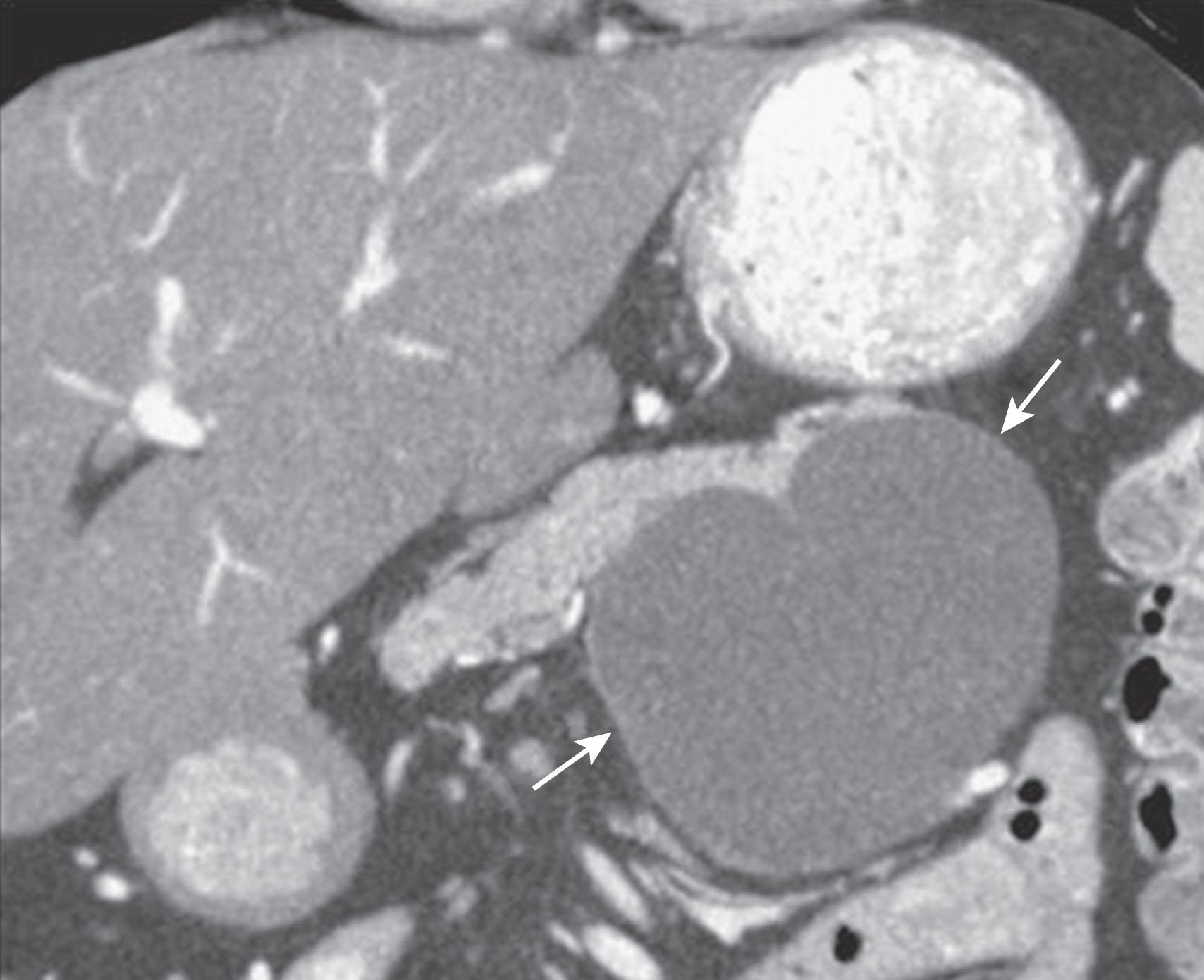
MCNs ( Fig. 13.4 ) are generally found in the body and tail of the pancreas; they are usually solitary, and their size ranges from 6 to 35 cm. They are lined with an inner layer of cells that secrete mucin and an outer layer of cells that resemble the ovarian stroma. They have several locules and a thick wall. These tumors do not communicate with the main pancreatic duct unless they erode into the duct, causing a fistula. They have fewer than six locules, and the size of the locules is generally greater than 2 cm. They may contain watery fluid or hemorrhagic, necrotic, or mucinous material. These tumors usually have intramural nodules, which histologically may range from high-grade dysplasia to invasive carcinoma. Pancreatic MCNs resemble ovarian MCNs, are seen almost exclusively in women of reproductive age (>95%; mean age 45 years), and are typically present in the body or tail of the pancreas. The malignancy rate in MCNs with ovarian stroma is reported to be as low as 10% to 17%. Curvilinear calcifications may be noted in the periphery of the tumor and/or capsule in 10% to 25% of cases.
Patients with MCNs may present with vague abdominal discomfort and have symptoms such as weight loss and anorexia.
On cross-sectional imaging, these tumors may appear as a single large cyst or as a large cyst with multiple locules, a thick outer wall, thick septations, and enhancing intramural nodules. Sometimes calcification of the outer wall can be seen, and the outer wall of the cyst may enhance on the delayed phase, a finding that correlates with the observation of a fibrotic capsule on histopathology. On MRI, the fluid within the cyst may have a low signal on T1W imaging and a high signal on T2W imaging; the increased signal on T1W imaging is owing to presence of mucin, but unfortunately this is an unreliable test for definitively diagnosing MCN. The presence of enhancing nodules should raise suspicion for malignancy.
On US, MCNs ( Fig. 13.5 ) can appear as well-demarcated, anechoic, thick-walled cystic masses and may have echogenic thick septa, internal nodularity, or mural calcification. Differential diagnosis of these lesions includes pseudocyst; these can be differentiated on serial follow-up imaging, in which the pseudocyst may decrease in size, whereas the mucinous neoplasm usually persists.
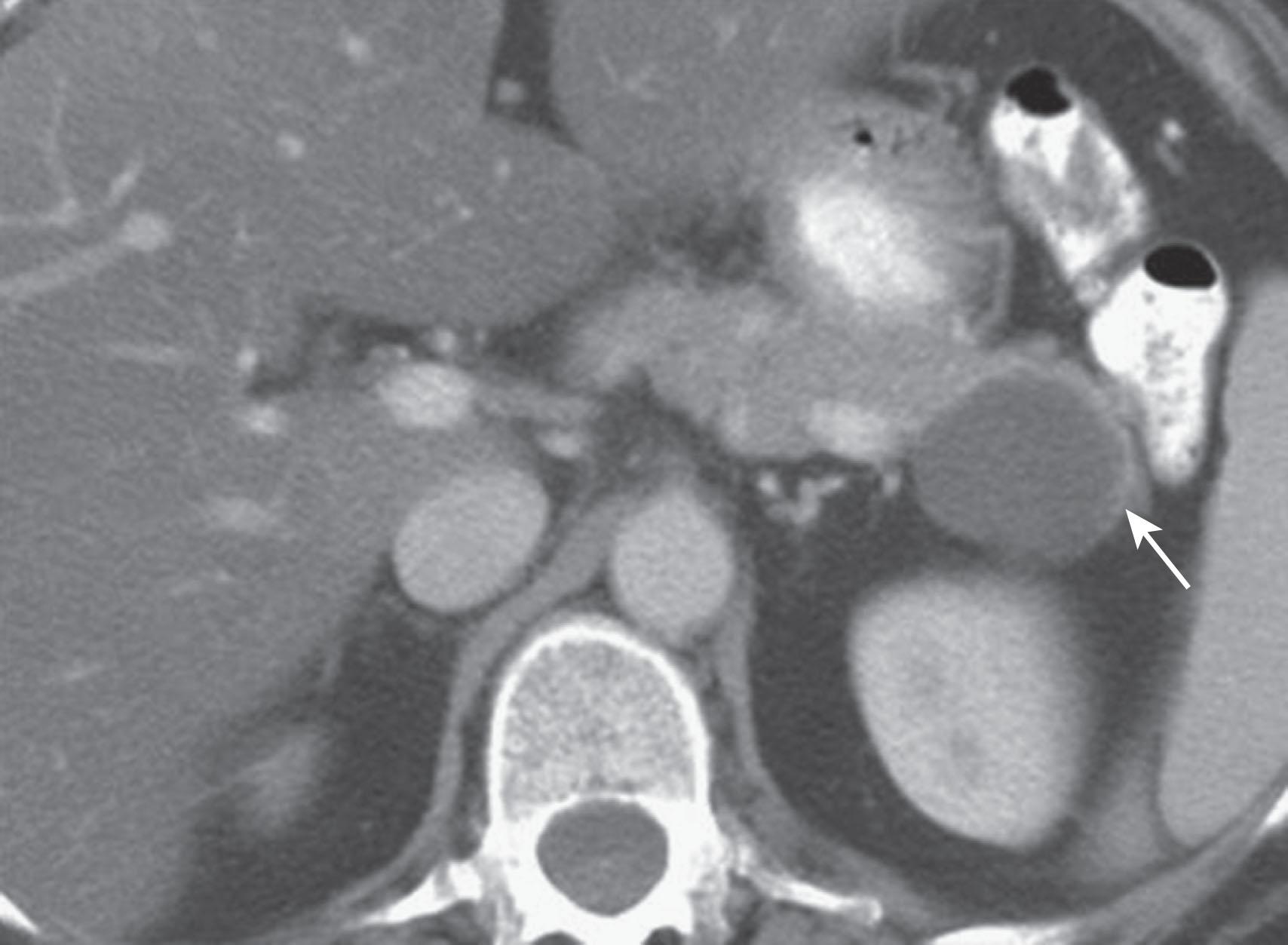
These tumors are resected, as cystadenoma and carcinoma cannot be differentiated by imaging, and these tumors have malignant potential.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here