Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Cystic fibrosis (CF) is an autosomal recessive disease that affects approximately 30,000 children and adults in the United States and 70,000 worldwide. CF involves multiple organ systems, and presents with varied clinical symptoms. Although pulmonary disease is the major cause of morbidity and mortality among patients with CF, 85% to 90% of individuals have exocrine pancreatic insufficiency and gastrointestinal symptoms. The majority of patients are diagnosed prenatally or soon after birth in the United States because of newborn screening programs, which are now available in every state. Failure to thrive, chronic diarrhea, and recurrent respiratory symptoms used to be the typical symptoms associated with diagnosing CF in infancy. These signs and symptoms are now rare at presentation because most infants are diagnosed through newborn screening before symptoms develop. A positive newborn screening test requires a follow-up sweat test, with diagnosis of CF confirmed by an elevated sweat chloride, or the identification of two disease-causing abnormal mutations of the CF gene (cystic fibrosis transmembrane conductance regulator, CFTR ). Early diagnosis is associated with better growth and improved outcomes. The current average life expectancy is approximately 42 years, but is significantly higher in patients with sufficient pancreatic function and milder disease phenotypes.
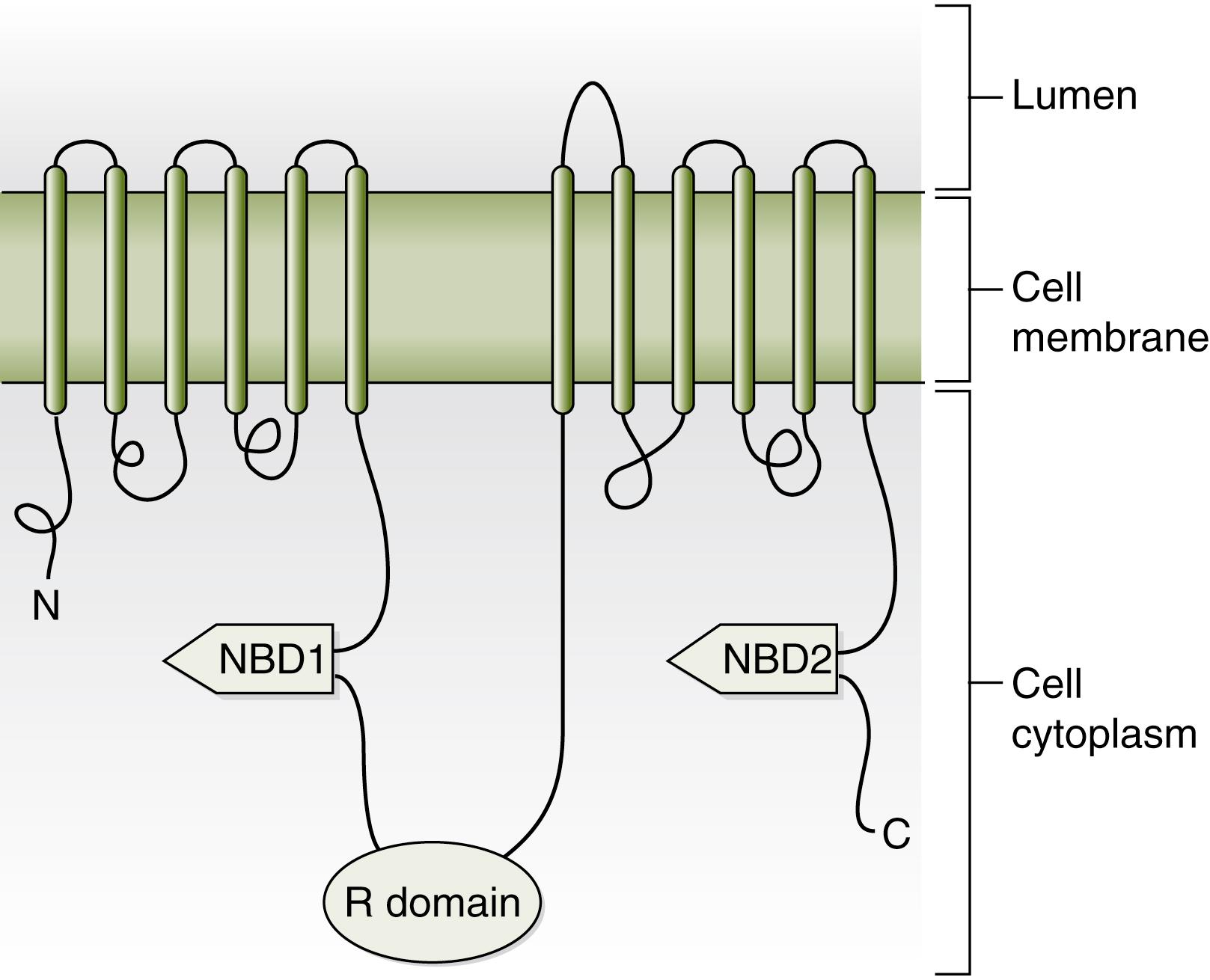
CF is an autosomal recessive disorder caused by mutations on both alleles of a 250,000 base-pair gene on the long arm of chromosome 7 (7q31 region). This gene, the CFTR , codes for a single-chain polypeptide of 1480 amino acids called the CFTR protein. CFTR is a cyclic adenosine monophosphate (cAMP)-regulated chloride channel, which also regulates other ion channels. It is a member of a family of proteins called ATP-binding cassette (ABC) proteins, which are involved with ATP hydrolysis-mediated solute transport. CFTR contains two membrane-spanning domains (MSD1 and MSD2), two nucleotide-binding domains (NBD1 and NBD2), and a central intracellular regulatory domain, or R region, with multiple phosphorylation sites ( Fig. 81.1 ). , The MSDs form the channel pore, and ATP hydrolysis by NBDs controls gating through the pore. Phosphorylation of the R domain determines channel activity.
CFTR mutations can be divided into five major classifications (some use six classifications), reflecting the location of the protein-processing defect ( Fig. 81.2 ). Class I mutations have little or no protein synthesis because of premature termination signals. Class II mutations are characterized by defective processing or trafficking of CFTR protein. F508del, a class II mutation—and the most common CF mutation—has a three-nucleotide deletion of phenylalanine, which prevents normal glycosylation. The partially glycosylated protein is retained in the endoplasmic reticulum, where it is degraded, instead of being transported and inserted into the apical cellular membrane. In class III mutations, proteins are processed properly but lack adequate function, due to impaired ATP binding and hydrolysis, which limits the energy-dependent channel from opening, or “gating.” G551D is an example of a class III mutation. Class IV mutations are characterized by alterations of conductance and have reduced cAMP-regulated currents in the chloride channel. Class V mutations affect splicing of messenger RNA (mRNA), producing extra or skipped exons, which affects stability of the protein on the cell membrane. (Class VI mutations have accelerated protein turnover at the cell surface and may also affect the regulatory function of CFTR on other ion channels.) Individual mutations can have characteristics of more than one class.
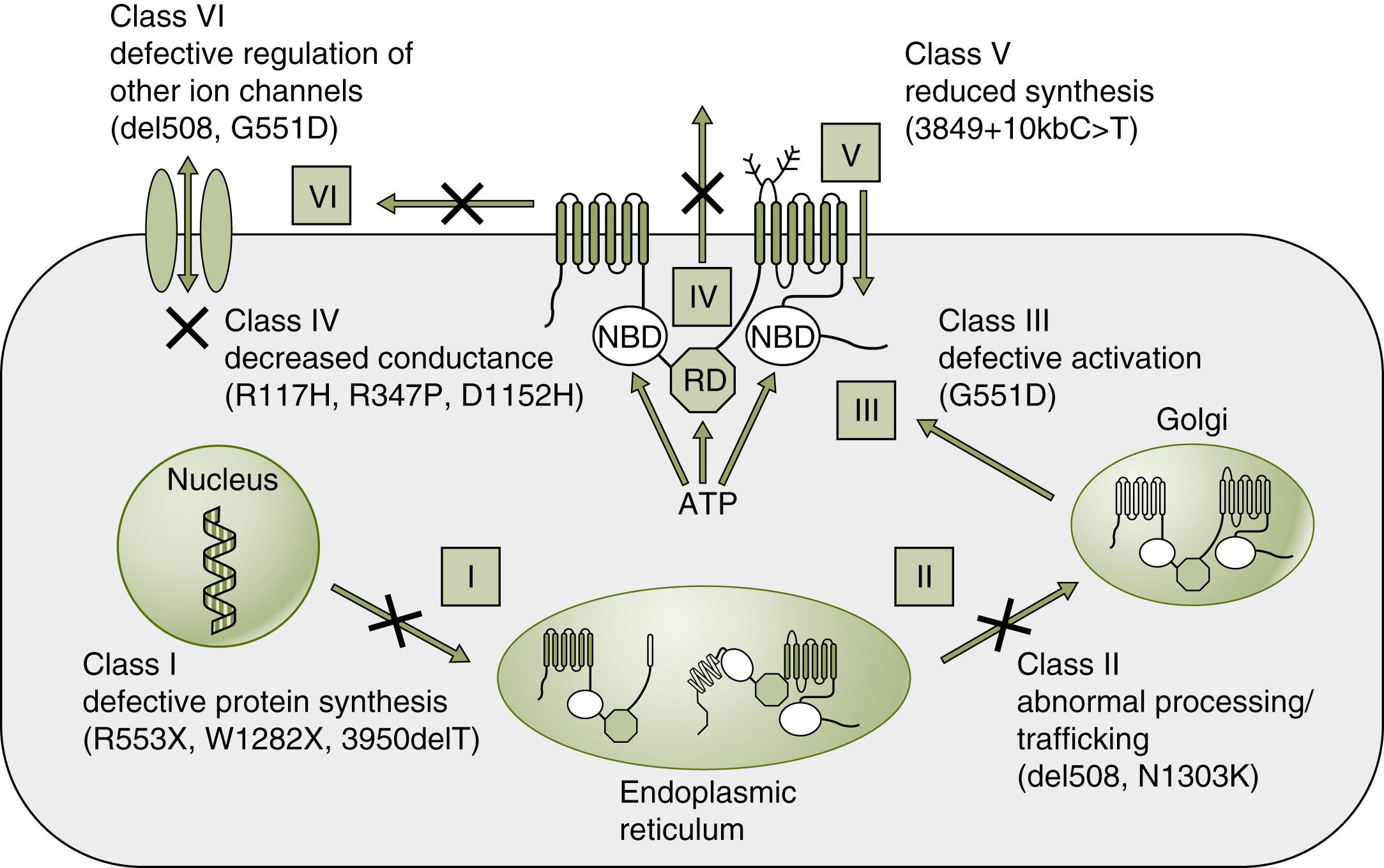
Class I, II, and III mutations are associated with more severe pulmonary disease and pancreatic insufficiency, and class IV and V mutations are associated with mild disease or no symptoms of CF. Polymorphisms, especially the truncated polythymidine tract called 5T, can affect CFTR function, either when found on two alleles or in combination with a deleterious mutation on the opposite allele. 5T defects are usually associated with congenital bilateral absence of the vas deferens (CBAVD), either without other clinical symptoms of CF or with mild CF symptoms.
Currently, more than 1900 mutations of CFTR have been identified ( https://www.cftr2.org/ ). F508del is the most common mutation of CFTR , found in more than 80% of chromosomes in American whites with CF. There are ethnic and regional variations of CFTR mutations. F508del is found in approximately 45% of the chromosomes of African Americans, approximately 30% of Ashkenazi Jews, and in less than 5% of Native Americans. The CFTR protein has variable expression and probably functions differently in the epithelium of different organs. There is clinical variability among patients with the same CFTR genotype, suggesting that environmental and hereditary factors can modify the disease phenotype. Evidence is accumulating that phenotypic heterogeneity is influenced by genes at one or more unlinked loci in the human genome, which act as modifier genes of CFTR.
Until recently, treatments for CF were focused on ameliorating clinical symptoms associated with the disease. Now there are mutation-specific therapies, known as CFTR modulators, that target specific variants to improve the function of CFTR, thereby treating the underlying defect in CF ( Table 81.1 ). , These therapies can be separated into the following categories: potentiators that promote the open configuration of CFTR and improve gating in the channel, correctors that increase the trafficking of variant CFTR to the plasma membrane, stabilizers that increase the duration of time that CFTR stays at the plasma membrane, amplifiers that increase the quantity of variant CFTR available for modulation by other therapies, and readthrough agents that suppress premature stop codons and allow for production of full-length CFTR protein. Therapeutic agents from the potentiator and corrector categories have been approved for clinical use, and others are in clinical trials. Ivacaftor is a potentiator agent that is approved for individuals with at least one gating mutation and for those who carry a residual function mutation. , Lumacaftor is a CFTR corrector that is approved as a combination drug with ivacaftor for individuals older than 6 years of age who are homozygous for the F508del mutation. Tezacaftor is a newer corrector that is currently approved, in combination with ivacaftor, for individuals older than 12 years of age who are homozygous for the F508del mutation. The ultimate goal in CF therapeutics is to develop CFTR modulators that will be effective for all mutation classes. As more therapeutic agents are approved, the practice of theratyping (grouping mutations functionally and biochemically by their response to various modulators) may be more commonly used to guide a personalized approach to therapy.
| Class | Impact on CFTR Protein | Common Mutation | Approved Therapies | Past and Developing Therapies |
|---|---|---|---|---|
| I | Lack of protein synthesis (reduced quantity) | Trp1282X | Ataluren—not effective in Phase 3 trials | |
| II | Abnormal processing and misfolding of protein, reduced quantity at cell surface | Phe508del | Lumacaftor/Ivacaftor (age 6 and up) Tezacaftor/Ivacaftor (age 12 and up) |
VX-445 and VX-659—correctors in Phase 2 and 3 trials |
| III | Conductance defect, decreased opening and reduced gating function | Gly551Asp | Ivacaftor | QBW251—potentiator in Phase 2 trials |
| IV | Created in insufficient quantity | 3849 + 10kb C->G | PTI-428—amplifier in Phase 2 trials | |
| V | Rapid turnover and insufficient quantity | Cys1400X | PTI-428—amplifier in Phase 2 trials |
In the pancreas, CFTR is found predominantly in the cell membrane of centroacinar and intralobular duct epithelium, which is responsible for secreting fluid with a high concentration of sodium bicarbonate. Normally, chloride excreted into the lumen is exchanged for bicarbonate, with sodium and water following. There is evidence that CFTR functions as a bicarbonate channel and functions as a Cl − /HCO 3 − exchanger in pancreatic duct cells. In CF, the abnormal CFTR limits this exchange and limits apical trafficking of zymogen, resulting in a more viscous sodium bicarbonate-depleted fluid. Loss of CFTR function limits fluid secretion in the pancreas, resulting in a more viscous, acidic fluid in the pancreatic ducts. The lower pH prematurely activates trypsin and other zymogens that cannot be flushed from the pancreas, resulting in recurrent injury, progressive fibrosis, and, in some cases, chronic pancreatitis.
There are progressive histologic findings in the CF pancreas, correlating to the severity and age of the patient. Initially, focal intraluminal eosinophilic infiltration with pancreatic duct dilation is present. There is progression of acinar atrophy and fibrosis with ductal ectasia. Late in the disease, there is total loss of acinar tissue and ductal obliteration. Islets of Langerhans are diminished but not totally destroyed. The preserved islets have decreased glucagon-secreting α cells, insulin-secreting β cells, and pancreatic polypeptide-secreting cells. Autopsy studies show that pancreatic nesidioblastosis (hyperinsulinemic hypoglycemia related to abnormal Β cells) is common in children with CF but not in adolescents and adults. With age, there is increased loss of β cells, increasing the prevalence of diabetes, which is rare in young children with CF, possibly because of children’s ability to form new islets.
Approximately 10% to 15% of patients with CF retain enough residual pancreatic secretion for normal digestion and are termed pancreatic sufficient . The CFTR genotype–phenotype relationship has its strongest correlation with pancreatic function. Mutations that are associated with pancreatic sufficiency are considered “mild” mutations. Patients with two mild mutations, or one mild and one severe mutation, are typically pancreatic sufficient. Patients who remain pancreatic sufficient have less enzyme secretion and lower concentrations of bicarbonate in pancreatic fluid compared with normals. Their growth is usually normal or close to normal, and therefore, as a group they were diagnosed with CF later than patients with pancreatic insufficiency, until routine CF newborn screening was instituted. In general, pancreatic-sufficient patients have less severe disease and lower sweat chloride levels, and their life expectancy is substantially longer than that of pancreatic-insufficient patients; however, they are much more likely to develop chronic or recurrent pancreatitis.
“Severe mutations” identify genotypes specifically associated with pancreatic insufficiency and do not necessarily correlate with severity of lung function or disease. Mutations associated with pancreatic insufficiency are typically class I–III defects, in which CFTR function is more severely affected. Clinical signs of pancreatic insufficiency develop when less than 10% of normal pancreatic enzyme activity is present in the duodenum.
CF is the most common cause of pancreatic exocrine failure in children. Approximately 85% to 90% of patients with CF are pancreatic insufficient. At birth, approximately 65% of infants with CF are pancreatic insufficient. Of the remaining pancreatic-sufficient infants, approximately 15% to 20% will have progressive loss of pancreatic function by 3 years of age. In pancreatic insufficient individuals, the calorie and protein losses in the stool prevent normal weight gain, growth, and development and can be associated with hypoalbuminemia, edema, and normochromic, normocytic anemia.
Although most infants and children with CF have pancreatic insufficiency, pancreatic function testing ( Table 81.2 ) should be obtained before beginning pancreatic enzyme replacement therapy. The most accurate method for measuring pancreatic exocrine function is through direct stimulation of pancreatic secretions. The pancreas is stimulated with exogenous hormones, secretin/cholecystokinin, or endogenous nutrients that stimulate pancreatic secretions, and the secretions are collected through a tube placed at the ligament of Treitz and analyzed for pH, bicarbonate, chymotrypsin, trypsin, lipase, colipase, amylase, and carboxypeptidase output. Although this is the most accurate test for evaluating pancreatic function, because of the invasive nature and expense, it is seldom used in clinical practice. In addition, measurements of the fluid may be variable. Secretin-stimulated diffusion-weighted magnetic resonance imaging (SS-DW-MRI) discriminates patients with mild, moderate, and severe exocrine insufficiency but is not frequently used as it is expensive and not widely available. Indirect analysis of pancreatic function is less accurate but more practical clinically. A 72-hour quantitative fecal fat collection evaluates the amount of fat ingested compared to the amount excreted. The coefficient of fat absorption is calculated as (grams of fat in stool)/(grams of fat in diet). Fat absorption less than 15% is normal in children younger than 3 months; in older children and adults, less than 7% is normal. The 72-hour stool fecal fat collection is a noninvasive test but is difficult to perform and is not specific for exocrine pancreatic insufficiency.
| A. Examples of Direct Tests of Pancreatic Function | ||
|---|---|---|
| Test | Advantages | Disadvantages |
| Lundh test: Stimulation with a standardized meal followed by analysis of duodenal fluid for enzyme content | Physiologic, relatively inexpensive | Highly variable, depends on endogenous hormone release |
| Secretin test: Stimulation with human synthetic secretin followed by analysis of duodenal fluid for bicarbonate concentration | May predict mass of functional pancreatic tissue | Measurements may be inaccurate due to incomplete recovery of duodenal fluid; invasive |
| Cholecystokinin (CCK) test: Stimulation with CCK receptor agonist followed by analysis of duodenal fluid for enzyme concentration | May be more sensitive for mild exocrine dysfunction | May be contaminated with gastric fluid; invasive |
| Secretin-CCK test: Stimulation with both hormones followed by analysis of duodenal fluid for enzyme and bicarbonate concentrations | May improve sensitivity for mild exocrine dysfunction | Highly variable; concentrations of enzymes may be unreliable; invasive |
| B. Examples of Indirect Tests of Pancreatic Function | ||
|---|---|---|
| Test | Advantages | Disadvantages |
| Serum trypsinogen | Inexpensive, sensitive when <20 ng/mL | Less sensitive and specific when 20–29 ng/mL |
| Fecal fat: Calculate coefficient of fat absorption = (grams of fat in stool)/(grams of fat in diet). Normal is <7% | 72 h quantitative collection is more accurate than spot fecal fat | Difficult to perform, not specific for EPI |
| Fecal chymotrypsin | Spot collection correlates well with prolonged collection | Less sensitive for mild-to-moderate EPI |
| Fecal elastase | Correlates with elastase in pancreatic fluid and stool, highly sensitive and specific | Watery diarrhea may dilute specimen and produce false positives |
| Secretin-enhanced MRI/MRCP | Noninvasive, correlates with direct testing | Expensive, not widely available |
Detection of fecal enzymes is an easier test for determining pancreatic function and does not require prolonged stool collections. Fecal elastase measurements have a high level of sensitivity and specificity for pancreatic insufficiency. , Fecal enzyme levels can be influenced by supplemental pancreatic enzyme replacement, with the exception of fecal elastase, which is not found in pancreatic enzyme supplements. Fecal elastase concentration is currently the simplest and most reliable indirect method of accessing pancreatic exocrine function. Fecal chymotrypsin is less sensitive than elastase for mild-to-moderate exocrine pancreatic insufficiency.
Immunoreactive trypsin (IRT) is a pancreatic enzyme precursor. The serum IRT level is elevated in infants with CF due to pancreatic damage that occurs in utero, but the level returns to normal or below normal by 8 weeks of age. It then falls sharply in the first few years of life in pancreatic-insufficient individuals and is typically subnormal at 7 years. In children younger than the age of 7 years, IRT fails to distinguish pancreatic-insufficient from pancreatic-sufficient patients. IRT is not as sensitive or specific as the fecal enzyme studies but because its levels are elevated in CF newborns, it is used as the basis for most CF newborn-screening programs.
Pancreatic insufficiency in CF is treated with oral exogenous pancreatic enzyme-replacement therapy derived from processed porcine pancreas. The enzymes are enclosed in microspheres inside gelatin capsules to preserve efficacy. The capsules protect the enzymes from gastric acid. Then, the microspheres’ coating dissolves in the alkaline duodenum at a pH above 5.5, allowing enzyme activation to occur in the proximal small bowel. Most enzyme preparations consist of delayed-release enteric-coated capsules, which should be swallowed whole or opened and the contents sprinkled on soft, room temperature, acidic food such as pureed fruit. In infants, mixing preparations with the smallest microspheres in a small amount of fruit or applying them to the tongue just before offering the bottle or breast works well. The regular release non–enteric-coated tablet preparation should be swallowed whole with sufficient liquid; none of the preparations should be crushed or chewed. Recently, a cartridge containing immobilized lipase was developed for use with continuous enteral feedings.
Bioengineered enzymes are being developed, which will likely replace porcine-derived products in the near future. They are likely to improve fat and protein absorption, with fewer pills and fewer side effects, compared to current products.
Pancreatic enzyme replacement therapy is initiated once pancreatic insufficiency has been diagnosed. The goals of therapy are to promote weight gain, growth, and development and to limit gastrointestinal symptoms. There is a wide individual variation in response to supplemental enzyme dosing. The lower range of the recommended dosage should be started and adjusted upward based on stool pattern and weight gain. In infants, dosing of enzymes can be based on food intake, but for the older child, weight-based dosing in comparison to fat/protein intake is better. The Cystic Fibrosis Foundation guidelines for pancreatic enzyme dosing (see Table 81.2 should be used when starting or adjusting therapy. To decrease the risk of fibrosing colonopathy, enzyme doses should be less than 2500 lipase units/kg per meal, less than 10,000 lipase units/kg of body weight per day, or less than 4000 lipase units/g fat per day. ,
With appropriate enzyme replacement, fat absorption improves, and abdominal complaints decrease for most patients. Once adequate dosing of pancreatic enzyme replacement is established, a rapid improvement in the degree of steatorrhea should be seen, with decreased stool frequency, reduced abdominal pain and bloating, and increased appetite with improved weight gain. Because the absorption and digestion of fats is complex and requires pancreatic enzymes, bile salts, and a normal intestinal mucosa and milieu, some patients continue to be symptomatic with abnormal fat absorption, despite taking in appropriate amounts of enzymes. Many of these patients benefit from the addition of H2-receptor antagonists or proton pump inhibitors, which optimize the intraluminal action of the supplemental enzymes by reducing the acidity of the gastric environment. , Other common causes of diminished response to enzyme replacement include nonadherence, lack of synchronized delivery with meals, and decreased enzyme potency as a result of either exposure to heat or use past the expiration date ( Box 81.1 ). There is a tendency for clinicians to continue to increase the enzyme dose when a patient complains of increased abdominal symptoms; however, patients receiving high-dose enzymes and acid suppression therapy who continue to have persistent abdominal symptoms require a fecal fat absorption study and evaluation for other causes of steatorrhea or abdominal pain, including celiac disease, Crohn disease, lactose intolerance, parasitic disease, and bacterial overgrowth ( Box 81.2 ).
Nonadherence to prescribed dosing
Expired enzymes
Inactivated enzymes due to prolonged exposure to heat
High acidic small intestinal environment
Prolonged exposure of microspheres to alkaline foods
Taking enzymes after eating
“Grazing”
Generic enzymes with lower potency
Chewed or crushed microspheres
Inadequate dose of enzymes reaching the small intestine
Glycine-conjugated bile acids that limit micelle formation
Hepatobiliary disease with portal hypertension
Cholestatic liver disease
Celiac disease
Giardiasis
Short gut syndrome
Bacterial overgrowth of small intestine
Recurrent abdominal pain
Inflammatory bowel disease
Irritable bowel syndrome
Lactose intolerance
Infectious enteritis
Esophagitis
Gastroesophageal reflux
Fibrosing colonopathy
Eating disorders
Digestive tract cancers
Fibrosing colonopathy is a potentially significant complication of pancreatic enzyme replacement therapy and is associated with prolonged use of greater than 6000 units of lipase per kg per meal. , Symptoms include abdominal pain, vomiting, bloody or persistent diarrhea, and poor weight gain or weight loss. Thickening of the bowel wall can be seen by ultrasonography and predates stricture formation. Barium enema can show focal or generalized narrowing, with the ascending colon most frequently affected, although total colonic involvement has been reported. Many patients show clinical improvement with decreasing supplemental enzymes and parenteral nutrition, but some require surgical resection of the narrowed section of colon. The pathophysiology of the colonic damage in fibrosing colonopathy is not clear but is associated with high-dose supplemental pancreatic enzymes, even though there are reports of it occurring before initiation of supplemental enzymes in CF. The association of high-dose enzyme use with fibrosing colonopathy led to a commercial recall of very high-potency enzymes and, since the recall, there has been a sharply decreased incidence of fibrosing colonopathy. Because of this association with high doses of enzymes, the Cystic Fibrosis Foundation recommends enzyme doses be less than 2500 lipase units per kg per meal, or less than 4000 lipase units per gram of fat per day. ,
Allergic reactions and hyperuricemia were more common with older, powdered preparations of enzymes that are no longer available. Some patients may develop oral and skin reactions from contact with active enzymes, so it is recommended to rinse the mouth after taking enzymes. Porcine virus transmission is a theoretical risk associated with porcine-derived products, although it has never been described clinically.
Meconium ileus (MI) is the earliest clinical manifestation of CF. Infants with CF are at risk for delayed passage of meconium, intestinal plugging with meconium, and MI. In CF, meconium has an increased concentration of albumin and decreased water and mineral concentrations, resulting in a thicker, more viscous texture. , Increased mucus production from goblet cell secretions, combined with gelatinous meconium, contributes to inspissation of meconium, resulting in partial or total intestinal obstruction. MI occurs almost exclusively in patients with pancreatic insufficiency, with an occurrence between 10% and 18% of this cohort. Modifier genes found on chromosome 19 determine susceptibility to MI but do not influence the likelihood of intestinal obstruction later in life. , When an infant with CF develops MI, the occurrence rate significantly increases in subsequent siblings with CF. Almost all infants with MI have CF, but there are reports of rare exceptions. , Neonatal intestinal obstruction can be diagnosed with prenatal ultrasonography. A hyperechoic pattern with dilated bowel, with or without ascites, has a high specificity for CF.
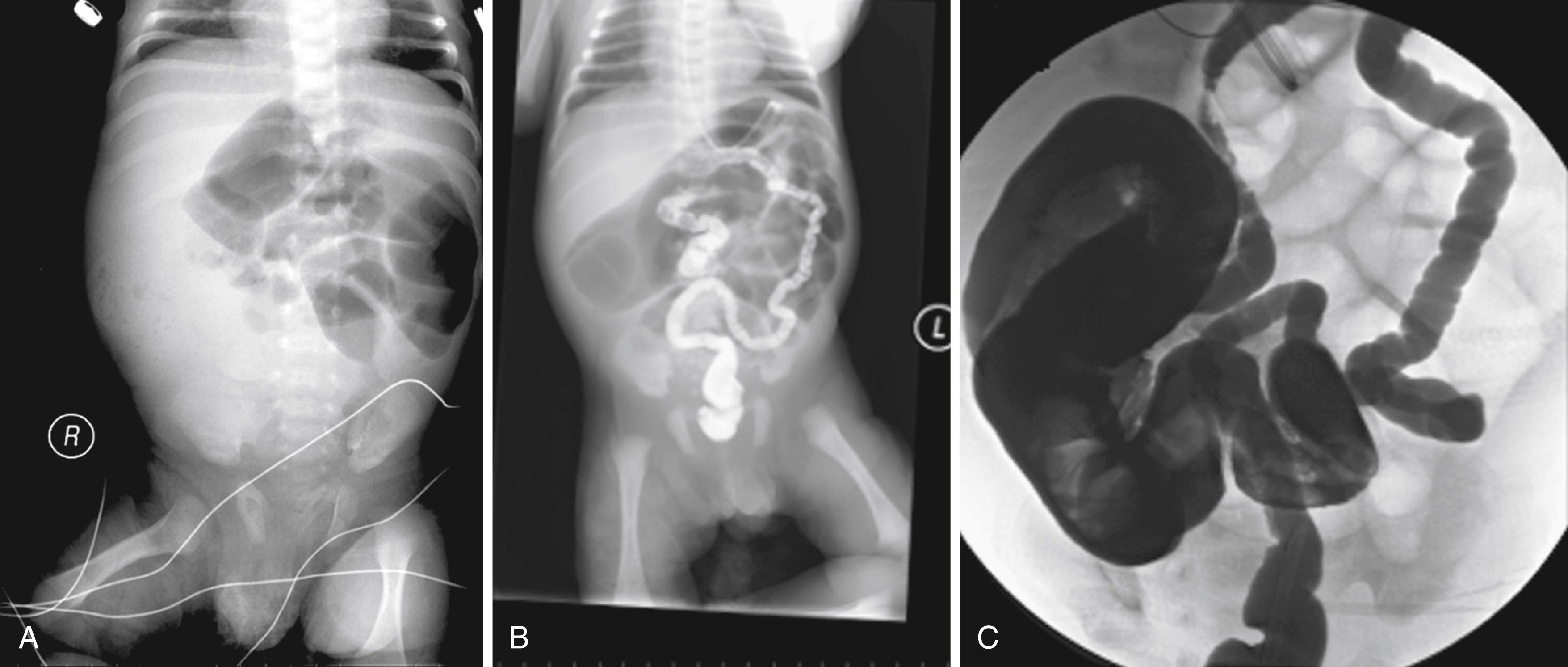
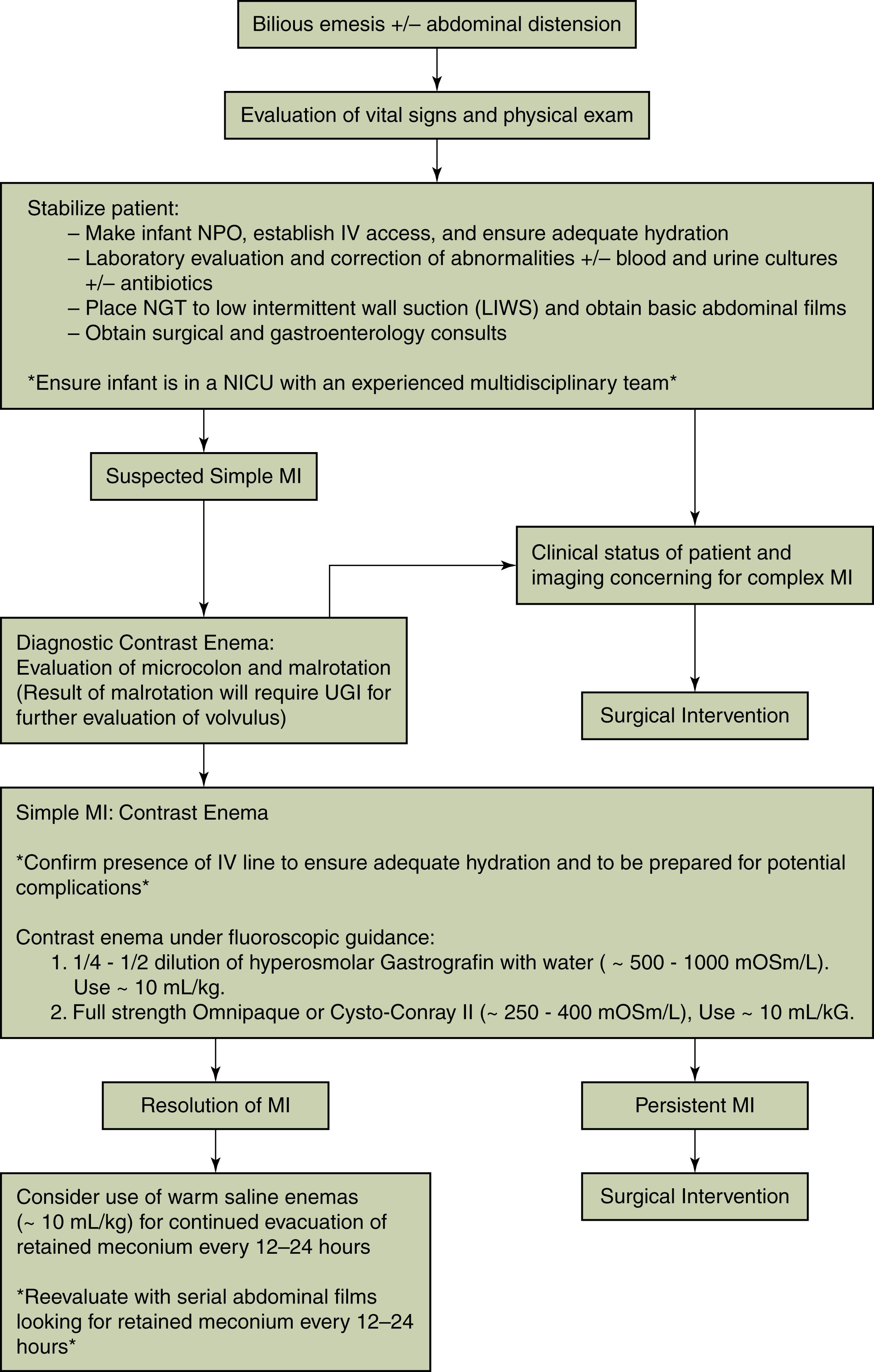
Clinical symptoms of intestinal obstruction from MI are present at birth, or soon after birth, with abdominal distension, vomiting, and absence of passage of meconium in the first 48 hours. Abdominal radiographs show distended bowel loops, usually without air-fluid levels, with a bubbly ground-glass density at the terminal ileum ( Fig. 81.3A ). Contrast enema is diagnostic, showing an unused microcolon and filling defects from inspissated meconium in the distal small bowel (see Fig. 81.3B and C ). Hyperosmolar contrast enema successfully relieves the obstruction in 50% to 90% of uncomplicated cases. , Approximately 50% of cases of MI are complicated by intestinal perforation, peritonitis, necrosis, volvulus, meconium cysts, or intestinal atresia and require laparotomy. Intraoperative bowel infusion with acetylcysteine, or hyperosmolar contrast, decreases the need for surgical resection. , If intraoperative infusions are unsuccessful or there are complications, resection of the affected bowel with primary anastomosis or side-by-side enterostomy is performed ( Fig. 81.4 ). , Current surgical and nutritional modalities have significantly decreased the formerly high mortality rate in infants with MI; however, MI in CF is associated with a higher mortality rate when diagnosed late or when skilled care is delayed. Children with CF who are born with MI have similar nutritional outcomes but go on to have more clinically significant pulmonary disease and a shorter life expectancy , than those without neonatal MI.
The abnormal meconium in CF causes other abnormalities besides MI. Meconium plug syndrome (MPS) is seen in infants with CF as well as those with Hirschsprung disease, hypotonia, and prematurity. The presenting signs and symptoms of MPS are similar to those of MI, but there is distal obstruction at the level of the colon rather than the ileum. A contrast enema shows a normal-caliber colon with filling defects from inspissated meconium and is usually diagnostic and therapeutic. Surgical intervention is usually not indicated.
Distal intestinal obstruction syndrome (DIOS), once called MI equivalent, occurs at all ages but is more common in adolescents and adults. It is unique to CF and is seen almost exclusively in patients with pancreatic insufficiency but has been reported in pancreatic-sufficient patients. Patients with prior MI may be at increased risk for DIOS, and DIOS may be a risk factor for developing liver disease. The exact incidence varies considerably and has been reported between 4% and 40%. , DIOS is believed to result from a combination of retained stool, abnormal intestinal secretions, and abnormal intestinal motility leading to impaction of stool in the terminal ileum, cecum, and proximal colon. Precipitating causes of DIOS are frequently not identified, but noncompliance with supplemental pancreatic enzymes, dehydration, change in diet, and use of opiates and anticholinergics have all been implicated. Symptoms include crampy lower abdominal pain, abdominal distension, vomiting, and anorexia. A palpable mass in the lower right quadrant can be felt on physical examination. During episodes of DIOS, normal stool patterns may continue, but progression to complete obstruction, intussusception, or volvulus can occur. Abdominal radiography shows retained stool in the ileum and cecum, which can appear as “bubbly” granular opacities. Air-fluid levels can be seen. DIOS can be classified as complete if there is evidence of complete intestinal obstruction on radiography and/or bilious vomiting, a fecal mass in the ileocecum, and abdominal pain and/or distension, or incomplete if there is no evidence of obstruction. Since the introduction of enteric-coated microsphere preparations of pancreatic enzymes, the frequency of DIOS has decreased.
In the absence of complete bowel obstruction, the preferred treatment is correction of fluid and electrolyte abnormalities followed by gastrointestinal lavage with a balanced isotonic solution containing polyethylene glycol administered orally or through a nasogastric tube. , The endpoint of therapy is to relieve the partial obstruction, which is determined by passage of stool, resolution of pain, and resolution of the palpable lower right quadrant mass. Follow-up radiography may be helpful in documenting resolution. Therapeutic enemas using water-soluble contrast containing N -acetylcysteine under radiologic control, which at one time were the standard treatment, are now rarely used because of the success of antegrade intestinal lavage. Once the acute episode of DIOS has been managed, dose optimization or improved compliance with pancreatic enzymes, increasing fluid intake, and regular administration of osmotic laxatives including polyethylene glycol help to prevent recurrence.
The most common cause of rectal prolapse in children is severe constipation; however, rectal prolapse can be a presenting sign of CF, and sweat testing should therefore be considered. Rectal prolapse occurs in up to 20% of patients with CF, usually between 6 months and 2 years of age. Rectal prolapse is uncommon in patients diagnosed with CF and treated with pancreatic enzymes before 3 months of age. Constipation, malnutrition, chronic cough, and pelvic muscle weakness increase the risk of rectal prolapse. Because infants are treated earlier as a result of newborn screening, rectal prolapse may become even less common in the future.
Rectal prolapse initially starts as intussusception of the rectum and then progresses until tissue prolapses from the anus. The prolapse can involve just the mucosa (mucosal prolapse) or all layers of the rectum—a complete prolapse or procidentia. Rectal prolapse is frequently initially noticed by the parents and is typically recurrent in CF. Usually, the prolapsed tissue can easily be reduced and does not require medical intervention but, rarely, surgical intervention is required. Recurrence of rectal prolapse is minimized by improved nutrition and decreased malabsorption with appropriate use of pancreatic enzyme supplementation.
Approximately 10% to 20% of pancreatic-sufficient patients with CF develop pancreatitis in their lifetime. Pancreatitis in CF occurs most often in older pancreatic-sufficient patients but has been described in pancreatic-insufficient children and infants and can be the presenting symptom of CF. , Pancreatitis can occur in patients with severe CFTR mutations, before the complete loss of pancreatic function. The progression from pancreatic sufficiency to pancreatic insufficiency due to recurrent pancreatitis may be more common in patients with the c. 350G>A , a class IV mutation of CFTR . , The exact pathophysiology of pancreatitis in patients with CF is not clear, but some preservation of acinar tissue function is required. The relationship between the degree of pancreatic ductal obstruction and the amount of preserved pancreatic acinar tissue influences the individual risk of CF pancreatitis.
The CFTR protein is found at the apical cell membrane of the ductal epithelial cells and maintains normal pancreatic secretions by controlling the movement of chloride, bicarbonate, and water into the pancreatic duct, which alkalizes and dilutes the fluid as it flows through the pancreas. Abnormal CFTR causes low acinar luminal pH, which inhibits endocytosis of secretary granule proteins and reduces the solubility of the concentrated fluid. Therefore, patients with CF have pancreatic secretions that are highly concentrated and proteinaceous. Physical obstruction of the pancreatic duct develops from the concentrated pancreatic secretions, proteinaceous plugs, or alteration of acinar function. Pancreatic proteolytic enzymes are subsequently activated, leading to pancreatic autodigestion. , Early destruction of functional acinar tissue leading to acinar atrophy presumably protects pancreatic-insufficient patients from pancreatic autodigestion and pancreatitis. The natural history of pancreatitis in CF is characterized by the onset of acute pancreatitis that recurs and over time can develop into chronic pancreatitis.
CFTR genotypes associated with mild phenotype effects in other organs have a greater risk of causing pancreatitis than genotypes associated with moderate-to-severe disease-causing phenotypes. CFTR compound heterozygote genotypes, containing one severe mutation and one mild-variable mutation of CFTR , are at greatest risk for CF-related pancreatitis. The 5T polymorphism on intron 8, which causes inefficient splicing of exon 10, producing shortened copies of CFTR , is frequently found in patients with CF and pancreatitis. The pancreatic secretory trypsin inhibitor (PSTI) gene is a Kazal type 1 serine protease inhibitor (SPINK1). PSTI is the first line of protection that inactivates some trypsin activity if trypsinogen is accidentally converted to trypsin in the acinar cells. The cationic trypsinogen, serine protease 1 gene (PRSS1) encodes cationic trypsinogen. CF with genetic mutations of PRSS1 is strongly associated with chronic pancreatitis and reoccurring acute pancreatitis, whereas defects in PST1/SPINK1 are associated with pancreatitis to a lesser degree. Patients may also be at increased risk of progression from acute to chronic pancreatitis, with abnormalities of both genes. The risk for recurrent pancreatitis is increased in patients with CF and pancreas divisum.
Patients with pancreatitis develop severe or progressive abdominal pain, with vomiting and epigastric tenderness. Serum and urinary amylase and serum lipase levels are elevated. Imaging studies may be normal, but narrowing of the pancreatic duct on pancreatography, peripancreatic edema on abdominal computed tomography, and increased pancreatic echogenicity on ultrasonography have been described. , Acute episodes of pancreatitis can be precipitated by alcohol ingestion, tetracycline, or fatty meals, but most episodes appear to occur spontaneously. Recurrent acute episodes are typical, but pseudocyst and systemic complications of CF-related pancreatitis are unusual. Because patients with recurrent acute episodes may eventually develop pancreatic insufficiency, pancreatic function should be monitored by fecal elastase every 1 to 2 years. Treatment for CF-related pancreatitis consists of intravenous fluids, nutritional support, and analgesics. Supplemental pancreatic enzymes may provide a negative feedback to the pancreas by limiting endogenous enzyme secretions, thereby decreasing pancreatic autodigestion and pain. In patients with frequent attacks, supplemental enzymes may prevent or increase the interval between episodes.
Gastroesophageal reflux disease (GERD) is more frequent in patients with CF than in the non-CF population. The major mechanism of GERD is increased transient relaxation of the lower esophageal sphincter. Other factors that increase the risk and severity of GERD include decreased gastric and intestinal pH, increased abdominal pressure from cough and lung hyperinflation, medications including β-adrenergic agents, and the high-fat diet that is common in CF. Supine positioning during chest physiotherapy has also been implicated. , In addition to frequent abdominal pain, GERD can exacerbate lung disease through aspiration and bronchospasm, worsening lung function and chest x-ray findings. , The concentration of bile acids in the sputum is higher in patients with CF than in non-CF individuals, and bile acid concentration correlates with lung function suggesting that duodenogastroesophageal reflux may substantially contribute to pulmonary symptoms. Macrolides may be useful to promote gastric emptying and decrease the bacterial load in the refluxate. If there is concern that GERD is contributing to pulmonary symptoms, it is recommended to medically manage GERD with acid suppression and lifestyle modification; however, if pulmonary symptoms do not improve, it is likely that they were not caused by GERD and a surgical antireflux procedure is likely not indicated. Long-standing GERD may predispose to Barrett metaplasia and esophageal adenocarcinoma.
Gastrointestinal dysmotility in CF is affected by antibiotic use, intestinal inflammation, dysbiosis, diet, glycemic control, and dehydration. Gastric emptying studies are highly variable in this population. Small bowel transit is often delayed. Malnutrition and slow motility are important risk factors for small bowel bacterial overgrowth, which may lead to enterocyte damage, malabsorption, and malnutrition. Clinical symptoms of small bowel bacterial overgrowth include bloating, flatulence, abdominal pain, diarrhea, dyspepsia, and weight loss. The utility of hydrogen breath tests is limited in CF, as patients may not be able to sufficiently exhale, and results are affected by increased production of methane and slow intestinal transit. Empiric treatment with rifaximin, metronidazole, amoxicillin-clavulanic acid, and percutaneous endoscopic gastrostomy (PEG)-based laxatives is widely practiced.
Abdominal pain is a frequent complaint in patients with CF and contributes substantially to their quality of life. A thorough history, focusing on characteristics of the pain such as localization, quality, intensity, chronicity, pancreatic function and associated symptoms, as well as a physical examination and judicious use of laboratory testing and imaging, allows for accurate and timely diagnosis. In addition to the obvious causes of abdominal pain associated with conditions described earlier, causes that occur in non-CF individuals also need to be considered. Intussusception occurs in 1% of patients with CF and is caused by inspissated secretions, enlarged lymphoid follicles, or a distended appendix creating the pathologic lead point. Ultrasound has a high sensitivity and specificity, with a typical finding resembling a coiled spring. Treatment is with fluid resuscitation, decompression with a nasogastric tube when required, and reduction under radiologic guidance with an air or contrast enema. There are reports of chronic intussusception in CF. ,
The diameter of the appendix is enlarged in CF because of luminal mucus distension; therefore, diagnosing acute appendicitis radiographically can be challenging. Acute appendicitis occurs less frequently in CF patients than in the general population, but the diagnosis is often delayed, leading to increased incidence of appendiceal abscesses and perforation. , Because imaging studies can be misleading in the diagnosis of acute appendicitis, clinical signs of increasing or persistent local tenderness in the right iliac fossa remain an important clinical clue for early diagnosis. There is an increased prevalence of celiac disease in CF patients, and an association with inflammatory bowel disease has been reported, but not confirmed, in large studies.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here