Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
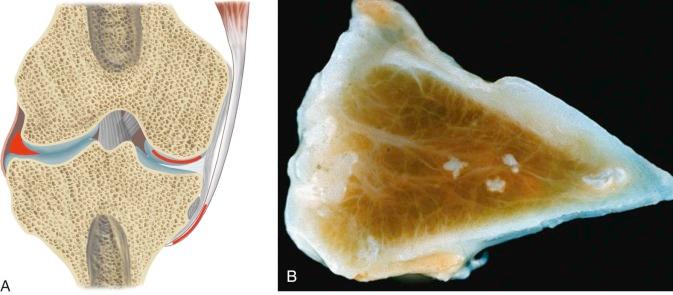
A number of crystals are associated with joint disease ( Fig. 61-1 ). The major culprits are calcium pyrophosphate dihydrate (CPPD) and hydroxyapatite (HA), a basic calcium phosphate (BCP). The focus in this chapter is on the disorders associated with crystal deposition in and around joints, except for true gout, which is discussed in Chapter 60 .
The discovery of crystals in synovial fluid that were not urate by McCarty and colleagues in the 1960s prompted a review of diseases apparently associated with crystals in joints. The same finding had been described previously in the Czech Republic as chondrocalcinosis polyarticularis (familiaris). Subsequently, two slightly different crystals were identified: calcium pyrophosphate dihydrate (CPPD), in about 3.3% of cadavers, seen radiographically as linear or punctate deposits, and dicalcium phosphate dihydrate (DCPD), which produced diffuse punctate deposits in a further 2.3%. Subsequently, a number of clinical and radiologic disorders became associated with these crystals, resulting in some confusion of terminology. These crystals will be grouped together as CPPD in the chapter.
CPPD crystals may be found in or around joints in a few circumstances, as follows:
Chondrocalcinosis . Deposits of crystals that are radiopaque may be seen pathologically, arthroscopically, or radiologically as cartilage calcification. Such calcification is not always due to CPPD (including DCPD), although it is the major cause. Chondrocalcinosis is an age-related phenomenon; about 25% of otherwise normal people age 85 and older will exhibit it. Furthermore, they may have no more joint symptoms than others without chondrocalcinosis. CPPD crystals are present as a normal constituent of hyaline cartilage and may have an important role in high-stress zones in the subarticular region. They occur at all ages, the crystal area being age related. For unknown reasons, chondrocalcinosis is particularly prevalent in those joints with both hyaline cartilage and fibrocartilage within them (e.g., the knee and wrist).
Pseudogout . Episodes of crystal shedding occur in some patients with chondrocalcinosis. Because of an apparent, although inaccurate, likeness with gout, this phenomenon became known as pseudogout. However, the clinical features of pseudogout are distinct from true gout. First, the common joints of involvement are the knee or wrist (compared with the great toe metatarsophalangeal joint). Second, the patients are often older women (compared with middle-aged men).
Pyrophosphate arthropathy . A form of osteoarthritis-like disease was described in some patients with “the pseudogout syndrome” by Martel and colleagues. Subsequently called pyrophosphate arthropathy, it became a disease in its own right. The concept of this as a distinct clinical entity has been challenged in recent years, but, in the chapter, the term pyrophosphate arthropathy will be used to describe a variant of hypertrophic osteoarthritis found in conjunction with CPPD crystals in synovial joints. The relationship between CPPD and the arthropathy is not fully clarified. Debate persists as to whether pyrophosphate arthropathy is truly an entity, distinct from osteoarthritis, or merely a hypertrophic variant of osteoarthritis in which the crystals of CPPD occur that are not necessarily the primary pathogenic cause.
The various manifestations of CPPD occur in both sexes, mainly in middle-aged and elderly patients. Chondrocalcinosis is totally asymptomatic and often an incidental finding at radiography or surgery. Classically, crystal shedding causes attacks of pseudogout. These acute or subacute self-limited attacks of arthritis involve one or more joints and last from 1 day to several weeks. They may be provoked by trauma, surgery, intraarticular injections, or systemic illness. The exact mechanism is unknown, but shedding of the superficial zones of hyaline cartilage, in which the crystals are deposited, seems to be likely. Because the distribution of involvement reflects those joints with fibrocartilage in them, the knee is the most common site.
The onset of episodes of arthropathy can be variable, and a number of clinical patterns have been suggested, including the following:
Pseudorheumatoid disease (35% to 60%) in which chronic large joint arthritis occurs with acute inflammatory episodes
Pseudo-osteoarthritis (10% to 35%) as chronic arthritis without acute exacerbations; asymptomatic joints (10% to 20%)
Pseudo-Charcot disease (0% to 2%) in which joints resemble neuropathic osteoarthropathy without the neuropathy
From the previous, it is clear that CPPD does not have a distinct clinical pattern.
Whereas chondrocalcinosis polyarticularis (familiaris) is, by definition, a hereditary disorder unassociated with other disease processes, the majority of cases of CPPD appear to be sporadic. However, CPPD deposition is increased in some metabolic disorders, particularly hyperparathyroidism and hemochromatosis, when the onset of symptoms and signs occurs at a younger age. Many other diseases have been described in association with CPPD, but no definite linkage with them has been proven. These include diseases that are themselves more common in middle-aged or older people, such as osteoarthritis, diffuse idiopathic skeletal hyperostosis, hypertension, atherosclerosis, and diabetes mellitus.
The relationship with idiopathic osteoarthritis is difficult to assess and lies at the heart of deciding whether pyrophosphate arthropathy actually exists or whether it is a subset of osteoarthritis with hypertrophic and perhaps modified features (see later discussion).
Other metabolic disorders, such as gout, have a higher frequency of CPPD crystals occurring in the same joint, and a proportion of gout patients have chondrocalcinosis. Hyperparathyroidism, especially the primary disease, has a well-documented incidence of chondrocalcinosis, which is usually asymptomatic; of interest, such patients do not develop structural changes of osteoarthritis. Many do not suffer acute pseudogout attacks. Other coexistent or precipitating conditions include hemochromatosis in which about 40% develop a joint disease resembling pyrophosphate arthropathy. Last, hypomagnesemia is associated with CPPD deposition because pyrophosphatase activity depends on the presence of magnesium ions.
The only documented condition in which a negative relationship exists with CPPD is rheumatoid arthritis. The incidence of chondrocalcinosis radiographically and CPPD in joint fluid is reduced in rheumatoid arthritis, but those few patients who do get both have erosions that are far less aggressive than usual, characterized by a patchy, asymmetric distribution, retained bone density, prominent osteophytes, and few, well-corticated erosions. This observation led to the suggestion that CPPD is a marker of a hypertrophic bone response to joint injury. Another study showed that CPPD deposition occurred more frequently in a knee that had undergone meniscectomy compared with the other, unoperated knee. Furthermore, the degree of osteoarthritis in the operated knee on the Kellgren and Lawrence scale was greater when CPPD was present. This supported the view that trauma can provoke chondrocalcinosis but also that osteophyte formation may be enhanced. A follow-up study looked at osteoarthritis of the hand in these patients after meniscectomy. It was found that the degree of osteoarthritis of the knee was worse if the patient had osteoarthritis of the hand. In other words, an underlying systemic predisposition to osteoarthritis was provoked locally by surgery to the knee and enhanced if CPPD was present.
CPPD crystals are deposited mainly in hyaline and fibrocartilage. In more advanced cases, they are found in all of the tissues of a joint as well as periarticular structures, including tendons. No abnormality is found in serum or urine, but inorganic pyrophosphate levels in synovial fluid are increased markedly during an episode of pseudogout, especially as it subsides. It is thought that the excess of inorganic pyrophosphate in cartilage results from overproduction by aging chondrocytes. The cause and/or mechanism is not known presently. The role of extracellular pyrophosphatase may be important because this enzyme may be inhibited by divalent ions, such as iron, calcium, and copper, perhaps explaining the accumulation of CPPD crystals in articular cartilage in hemochromatosis, hyperparathyroidism, and Wilson disease.
Similarly, the mechanism of crystal shedding is unknown. CPPD crystals injected subcutaneously or into joints produce an acute inflammatory response. Therefore, the pathogenesis of acute synovitis may be due simply to crystal shedding. Because the crystals lie in the subarticular region of hyaline cartilage and the radiologically visible chondrocalcinosis may disappear during an attack of pseudogout, it seems probable that the crystals are shed as the result of superficial hyaline cartilage damage. It has been shown that the crystals shed during pseudogout attacks are structurally different with a greater proportion of monoclinic, larger crystals than those found in the more chronic disease. The latter tend to be smaller, triclinic crystals.
The structural joint changes associated with CPPD crystal deposition are those of osteoarthritis, including hyaline cartilage fibrillation and erosion, subchondral sclerosis with thickened trabeculae, and multiple cysts. The cysts are larger and more numerous than conventional osteoarthritis. Collapse of cysts may underlie the reported increase in bony fragmentation and the multiple intraarticular osteochondral bodies found either loose within the joint cavity or embedded in synovium. Overall, the findings are of a hypertrophic variant of osteoarthritis with a predilection for subchondral cystic lesions.
As described earlier, it is not unusual to find pyrophosphate arthropathy in conjunction with Forestier disease or diffuse idiopathic skeletal hyperostosis. A meticulous paleopathologic study confirmed the apparent association between osteophytosis and enthesophytosis and coined the expression “bone formers.” The suggestion that CPPD marks a bone-forming predisposition has drawn much attraction, especially because centrally measured bone density is normal or high in patients with CPPD.
Because CPPD deposition results in radiographically visible calcification of certain structures, radiographs are the primary modality for their characterization. In fact, on modalities such as MRI, chondrocalcinosis can be inapparent or even misleading, occasionally simulating meniscal tear.
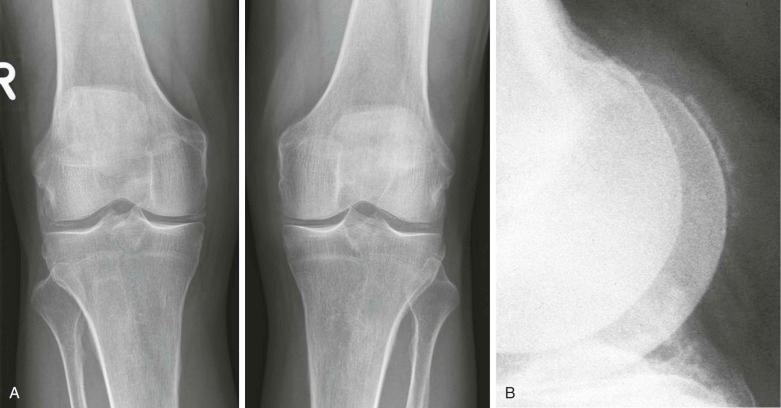
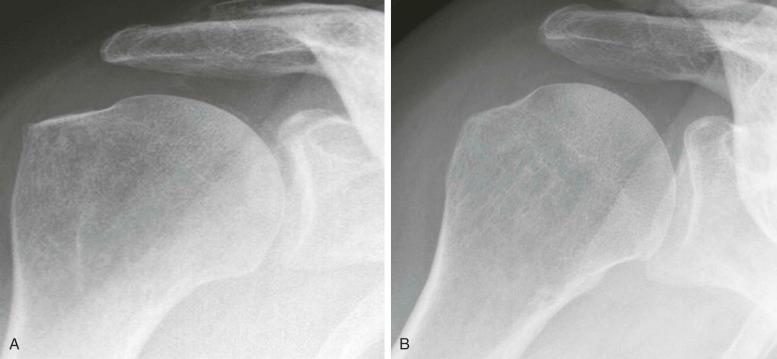
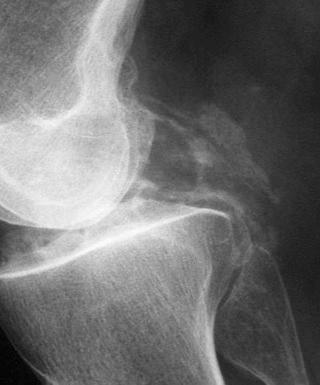
As indicated previously, articular and periarticular calcification is most commonly seen in those joints with both fibrocartilage and hyaline cartilage within them ( Fig. 61-2 ). Therefore, the knees and wrists are most commonly involved. Anteroposterior radiographs of the knees alone will detect calcification in approximately 90% of these patients. Calcification is linear or punctate in hyaline cartilage but may be granular in fibrocartilage ( Fig. 61-3 ). Fibrocartilaginous calcification is most common in the menisci of the knee and triangular cartilage of the wrist, although no joint is exempt, including the shoulder, elbow, and hip ( Fig. 61-4 ). In more severe cases, calcification may involve synovium, especially the suprapatellar pouch, capsule, or entheses around the joint ( Fig. 61-5 ). Tendon calcification typically involves the Achilles, triceps, or quadriceps. Such calcification appears thin and linear. Chondrocalcinosis on a radiograph is usually due to CPPD, but both CPPD and calcium HA can occur together.
On occasion, it is possible to demonstrate crystal shedding concurrent with a clinical episode of pseudogout ( Fig. 61-6A ). Joint aspiration reveals turbid, yellow fluid that may be misinterpreted as pus due to sepsis (see Fig. 61-6B ). Pseudogout almost always involves the knee or wrist (see eFig. 61-1 ), unlike true gout, which, in 80% of cases, involves the feet.
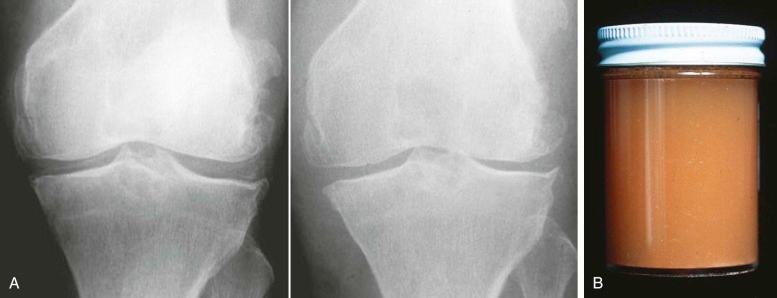
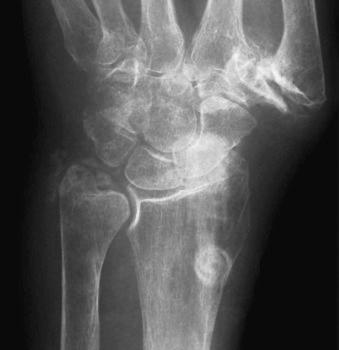
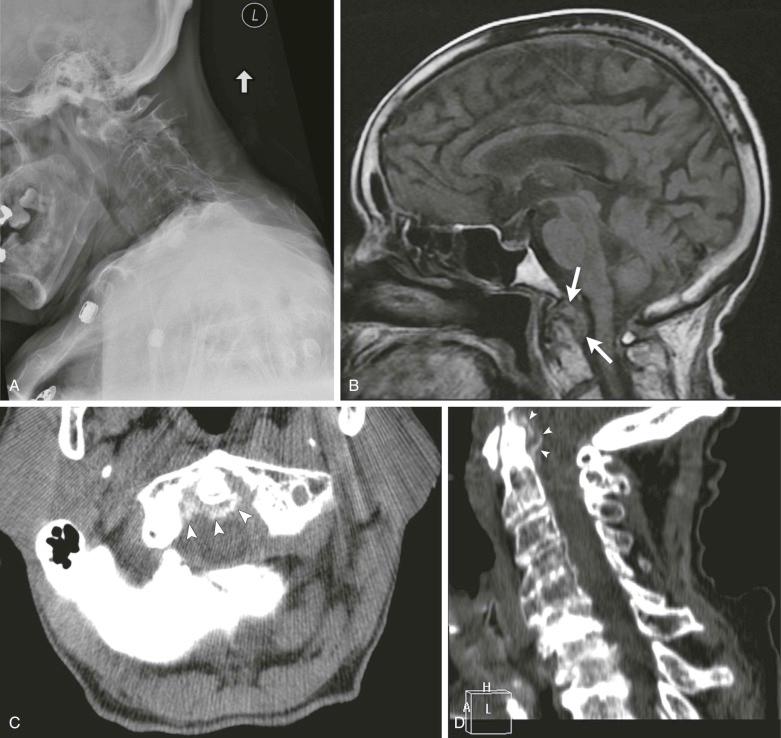
Occasionally, CPPD may present as a solitary space-occupying lesion known variously as tophaceous pseudogout or tumoral CPPD. These are most common in the digits and may present as an acute attack similar to tophaceous gout. Spinal lesions, depending on location, may present with mechanical or even constitutional symptoms ( Fig. 61-7 ) and have been recorded and associated with cord compression on rare occasions.
Not surprisingly, this most commonly involves those joints with a predilection for CPPD deposition. Therefore, the knee and the wrist are typically affected. In the original description of this form of arthritis occurring in pseudogout syndrome, five differences from conventional osteoarthritis were noted. This was subsequently reviewed and confirmed by Martel and colleagues, who again emphasized the following points:
Unusual articular distribution . Whereas knee and hip disease is typical in osteoarthritis, involvement of other sites is less commonly affected by osteoarthritis, such as the wrist and elbow. However, the wrist is frequently involved in injury in older patients, and elbow osteoarthritis is well recognized by archeologists, if not by clinicians, because it appears to be largely asymptomatic.
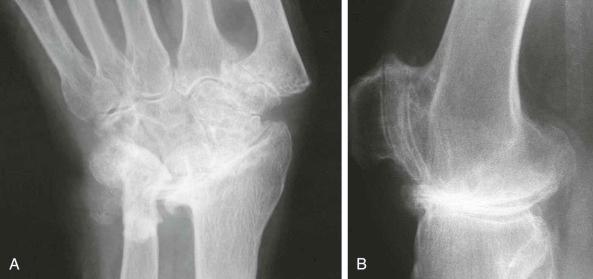
Unusual intraarticular distribution . Compared with osteoarthritis, the intraarticular distribution of pyrophosphate arthropathy may be considered unusual. Therefore, particular involvement of the scaphotrapezial joint of the wrist or the patellofemoral compartment of the knee is cited (see eFig. 61-2 ). It should be noted that although patellofemoral involvement is often implicated, this joint is much more commonly affected by patellar tracking disorders. One study confirms a higher prevalence of scaphotrapezial joint degeneration in a group of patients with chondrocalcinosis of the wrist, confirmed either radiographically or on joint aspiration, compared with conventional osteoarthritis. However, again looking at archeologic materials, 500 years ago, osteoarthritis of the patellofemoral joint was the norm, whereas tibiofemoral osteoarthritis seems to be a more recent disease.
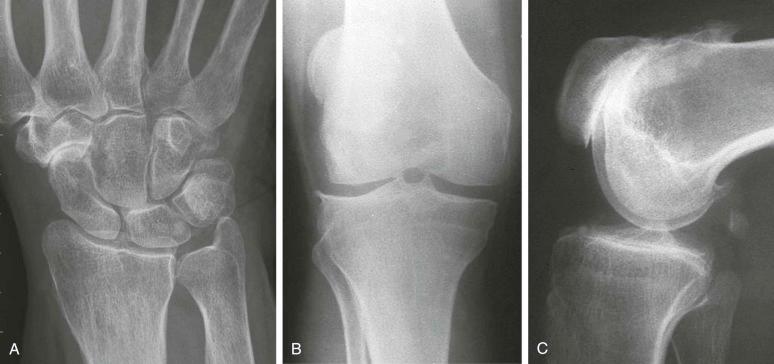
Prominent subchondral cyst formation . Lesions are often numerous, large, and well demarcated with a sclerotic border but often somewhat ill-defined. However, this is not unique to CPPD and also may be seen in hemochromatosis, Wilson disease, or even chronic bleeding states, such as hemophilia ( Fig. 61-8 ).
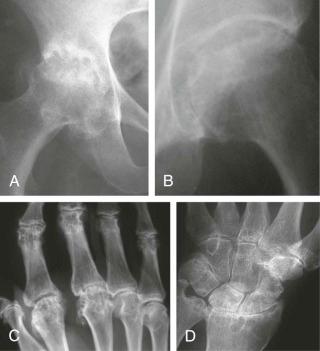
Pyrophosphate arthropathy . This may be associated with subchondral bone collapse and fragmentation, perhaps as a result of the collapse of the cysts and the release of multiple intraarticular osseous bodies.
Variable osteophyte formation . In some patients, large osteophytes are typical (see eFig. 61-3 ). In others, a polished, eburnated bony surface may be seen. However, both occur in conventional osteoarthritis and, again, are used by archeologists as the features that permit them a diagnosis of osteoarthritis. They may represent different aspects of joint failure and repair.
Synovial calcification . This is said to be common but may be a result of calcium HA as well as CPPD deposition, particularly if advanced degenerative changes were present (see section on calcium hydroxyapatite).
Subsequently, Resnick and colleagues reviewed 85 patients with probable or definite CPPD deposition, and they came to a number of conclusions, as follows:
No definite relationship was found between the presence and distribution of pyrophosphate arthropathy and the diagnostic level of confidence.
No definite relationship existed between the clinical pattern of arthritis and the presence of articular calcification or arthropathy.
Articular calcification was slightly more frequent in symptomatic compared with asymptomatic joints. No definite relationship was found between joint symptoms and the type of cartilage calcification (fibrocartilage versus hyaline cartilage).
The frequency and extent of local calcification and arthropathy did not correlate with the successful crystal recovery during joint aspiration.
Arthropathy was consistently more common, although not more severe, in joints with calcification. No relationship was found between the site of chondrocalcinosis (fibrocartilage versus hyaline cartilage) and the presence and severity of arthropathy.
Both from the spectrum of clinical presentation and the uncertainties expressed earlier, pyrophosphate arthropathy seems to be a diagnosis without finite definitions. The view that it represents a hypertrophic variant of osteoarthritis is gaining ground in which the crystals are epiphenomena rather than the cause of disease. Furthermore, CPPD may be protective of joint damage because most patients with CPPD deposition who have been followed for several years have done well clinically, with the degree of calcification and arthropathy generally remaining unchanged. In that study, after a mean of 4.6 years, symptoms had improved in 41% of joints and were unchanged in another 33%. Radiographic worsening of osteoarthritis was found in only 16%, whereas chondrocalcinosis had increased in 52%. More recently, two studies have shed doubt on the role of CPPD in the genesis of osteoarthritis changes at all. In one study, it was suggested that the relative risk of CPPD over conventional osteoarthritis was as little as 4%, and another found a strong relative risk for osteophyte formation with CPPD but not joint space narrowing, therefore the growing concept that CPPD is a marker of bone formation in joints, the entheses, and systemically.
Joint effusion and soft tissue swelling occur during episodes of pseudogout. Aspiration of joint fluid produces a turbid, yellow specimen that has a high white blood cell count and may be mistaken for septic arthritis. Chronic joint effusions are, as usual, associated with popliteal cysts.
Stress fractures in neighboring bones , especially the upper tibia, occur and are associated with angular deformities but are not unique to CPPD and are noted in conventional osteoarthritis and rheumatoid disease.
Carpal collapse may occur secondarily to radiocarpal osteoarthritis, particularly the so-called scapholunate advanced collapse wrist. This type of osteoarthritis occurs spontaneously or after trauma and is one of the most common patterns of degenerative joint disease in the wrist, with or without CPPD deposition.
Metacarpophalangeal (MCP) joint involvement has been emphasized as a feature of pyrophosphate arthropathy, especially affecting the index and middle fingers. Conventional osteoarthritis, particularly the erosive type, has a greater predilection for the interphalangeal joints, and, indeed, the MCP joints are often omitted from scoring atlases of osteoarthritis. Hemochromatosis also involves the MCP joints, often with well-marked subchondral cysts and also extending across the MCP joint row to the ring and little fingers.
Intervertebral discal calcification is thought to be an incidental finding without clinical significance, with deposits typically being in the outer fibers of the annulus fibrosus. The nucleus pulposus, generally, is not involved. CPPD may be seen, especially on CT, in other spinal tissues, including the ligamentum flavum, especially in the cervical region. Calcification has been recorded also in association with type II odontoid fractures. In all nine cases, reported erosion of the odontoid process was present as well as deposits of CPPD in the transverse ligament. It is unclear whether the CPPD contributed to the fracture, but the suggestion that the deposits might threaten the cervical cord is important.
Magnetic resonance imaging has no specific role in the diagnosis or assessment of CPPD. Indeed, a meticulous histologic study in cadavers confirmed that MRI was insensitive to the presence of CPPD deposits in the knee, even when they were widespread. The best site for visualization of the low-signal, rather granular deposits was the femoral condyles compared with other intraarticular structures. Granted that the femoral condyles have very thick hyaline cartilage, this does not seem a particularly clinically relevant finding beyond noting that CPPD may produce these changes if a radiograph is not at hand when it is being interpreted. In addition, the presence of CPPD in the meniscus has been suggested to simulate surfacing signal of a meniscal tear on MRI.
Computed tomography is highly sensitive to the presence of calcification because of chondrocalcinosis, but it can be seldom justified to diagnose it. Usually, calcification is an incidental finding or concurrent with another pathologic process, as described in cervical trauma.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here