Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Congenital heart defects are the most common type of birth defect in infants worldwide. The following chapter provides an overview of the anatomy, physiology, clinical presentation, diagnostic evaluation, and management strategies for a wide range of congenital heart disease encountered in neonates and infants. While the information herein is detailed, it is important to keep in mind that many different anatomic and physiologic variants are encountered in practice, often altering clinical management. The chapter is generally organized based on the predominant pathophysiology—cyanotic, obstructive, or shunting—for a given congenital heart defect. There can be considerable overlap between these physiologies, particularly in complex forms of congenital heart disease involving multiple defects. Defect physiology can also change considerably over time, particularly in the immediate postnatal transitional period around ductal closure. Prompt recognition and diagnosis of significant congenital heart defects contribute greatly to effective management and improved outcomes in affected infants.
In D-transposition of the great arteries (D-TGA), the aorta arises from the right ventricle, which receives systemic venous deoxygenated blood, and the pulmonary artery arises from the left ventricle, which receives pulmonary venous oxygenated blood. The blood flow in these patients is in parallel with the deoxygenated blood entering the aorta and recirculating to the body, while the richly oxygenated pulmonary venous return is recirculated to the lungs. In the absence of any shunting across connections between the systemic and pulmonary circulations to allow mixing of the deoxygenated and oxygenated blood, this results in systemic hypoxia and is often lethal. In D-TGA, three levels where mixing can occur are an atrial level defect (patent foramen ovale or atrial septal defect), patent ductus arteriosus (PDA), or ventricular septal defect (VSD). The best site of blood mixing has been shown to be at the atrial level through a patent foramen ovale (PFO) or atrial septal defect (ASD), with the other forms (PFO and VSD) being less reliable. It is, therefore, extremely important at birth to make sure not only that there is an atrial level communication, but also that it is adequate in size.
Associated defects have a dramatic effect on the presentation and pathophysiology of newborn infants with D-TGA. At a minimum, an atrial level defect with balanced bidirectional shunting is essential for survival. The presence of a PDA also allows for additional shunting from the aorta (deoxygenated blood) to the pulmonary artery (oxygenated blood), resulting in increased pulmonary venous return and left-to-right shunt at the atrial level. A VSD occurs in about 25% of infants with D-TGA. Its presence contributes to mixing of the oxygen-rich and desaturated blood, but as previously stated, atrial level mixing is the most reliable. Coarctation of the aorta or aortic arch interruption can also occur in association with D-TGA with VSD.
D-TGA is the most common cyanotic heart defect identified in the first week of life, and the diagnosis should be considered in any cyanotic neonate. Fetal diagnosis is common but not uniform. Even with prenatal diagnosis, however, profound hypoxia caused by a highly restrictive atrial level defect can lead to rapid deterioration and death within the first hours of life. Respiratory symptoms in this disorder are absent or limited to hyperpnea or tachypnea without dyspnea. The patient's second heart sound is loud and persistently single because of the anterior position of the aorta and the posterior position of the pulmonary artery. A holosystolic murmur, when present, suggests an associated VSD; a systolic ejection murmur is auscultated when the patient has pulmonary stenosis. In the absence of these associated defects, murmurs are generally not heard. The peripheral pulses are normal unless coarctation of the aorta is present. Persistent ductal patency or high pulmonary vascular resistance will affect the clinical findings of an associated ventricular septal defect or coarctation of the aorta.
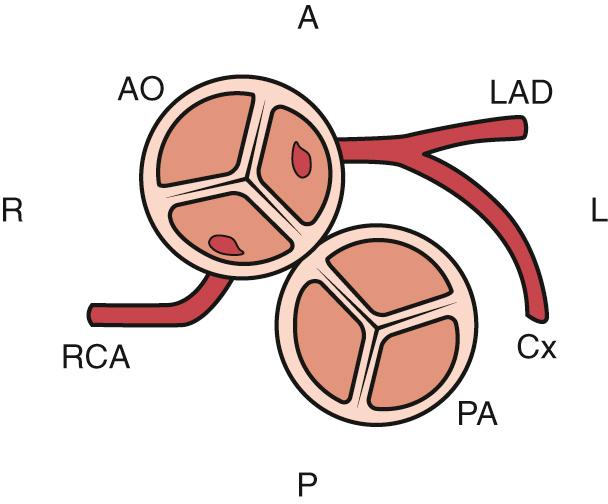
The electrocardiogram is normal or may demonstrate right ventricular hypertrophy after a few weeks of life. Similarly, the classic egg-shaped heart with increased pulmonary vascularity on the chest radiograph might not be seen in the newborn period. Echocardiography defines the associated defects and coronary artery anatomy, which is central to surgical planning. Cardiac catheterization is usually reserved for neonates requiring balloon atrial septostomy, but it is occasionally useful for clarifying the coronary anatomy or other anatomic details ( Fig. 75.1 ).
Establishing patency of the ductus arteriosus with intravenous prostaglandin E 1 infusion in newborn infants with D-TGA often improves arterial oxygenation by increasing shunting from the oxygen-rich pulmonary artery to the deoxygenated aorta. Aortic to pulmonary artery shunting at the ductal level may increase pulmonary venous return, distending the left atrium and facilitating left-to-right atrial shunting of fully oxygenated blood across the atrial level defect. If the prostaglandin E 1 infusion does not result in adequate systemic oxygenation, a balloon atrial septostomy is performed either at the bedside or in the cardiac catheterization lab with echocardiographic guidance. This enlarges the atrial level defect, allowing improved atrial mixing of the systemic and pulmonary venous return.
Historically, the surgical interventions for D-TGA were the atrial switch operations of Senning (1959) and Mustard (1964). In these procedures, the deoxygenated blood from the vena cavae is baffled to the morphologic left ventricle and thence the pulmonary artery, while the oxygenated pulmonary venous blood is directed to the morphologic right ventricle and the aorta. This results in physiologic correction. However, the Senning and Mustard procedures are associated with a number of long-term consequences, including right ventricular dysfunction, systemic venous baffle obstruction and leaks, and atrial arrhythmias.
Drs. Jatene and Yacoub pioneered the anatomic repair of D-TGA via the arterial switch operation, which requires transection of the aorta and main pulmonary artery, reconnection to establish ventriculoarterial concordance (systemic left ventricle to the aorta and pulmonic right ventricle to the main pulmonary artery), and reimplantation of the coronary arteries to the neoaortic root. Other associated intracardiac defects are generally repaired at the time of surgery. The arterial switch operation is optimally performed in the first week of life for infants with D-TGA. If there is a prolonged delay, the risk increases for left ventricular deconditioning and resultant inability to function adequately as the systemic ventricle. The follow-up data on patients who have undergone the arterial switch operation are quite favorable. Risk factors for early mortality include prematurity and right ventricular hypoplasia. Abnormal coronary anatomy (particularly intramural coronary arteries), a risk factor in earlier experiences, can now be managed effectively, although with potentially higher hospital morbidity. Long-term follow-up studies demonstrate excellent ventricular function, normal rhythm, and low incidence of obstruction at the aortic and coronary suture lines. Narrowing in the supravalvar pulmonary area and branch pulmonary arteries may require subsequent surgeries or catheter interventions. Neoaortic root dilation and aortic regurgitation are found in some children, but these rarely require further intervention. Follow-up data in children 16 years of age with D-TGA from the Boston Circulatory Arrest Study suggest that adolescents with D-TGA who have undergone the arterial switch operation are at increased neurodevelopmental risk and may benefit (along with other children born with congenital heart disease) from ongoing surveillance to identify emerging difficulties.
In 1888, Fallot described the association of a large ventricular septal defect, infundibular (subvalvar) and valvar pulmonary stenosis, right ventricular hypertrophy, and a large aorta overriding the ventricular septum—the tetralogy which now bears his name. The spectrum of pulmonary lesions in tetralogy of Fallot (TOF) can range from mild pulmonary stenosis with excellent saturations to pulmonary atresia with very low saturations. Infants with severe pulmonary stenosis or atresia may have severely hypoplastic or absent pulmonary arteries with major aortopulmonary collateral arteries serving as the primary source of pulmonary blood flow. In TOF, cyanosis is proportional to the degree of right ventricular outflow tract (RVOT) obstruction, with severe obstruction limiting pulmonary blood flow and leading to very low saturations. Shunting across the VSD is determined by an intricate balance between the systemic vascular resistance (SVR) and pulmonary vascular resistance (PVR). Acute narrowing or stenosis from the release of endogenous catecholamines, for example, increases PVR more than SVR, resulting in right-to-left shunting and worsening systemic desaturations.
A right aortic arch is found in approximately 25% of patients with TOF. Coronary artery anomalies (e.g., left anterior descending coronary artery arising from the right coronary artery) are not uncommon and must be identified prior to surgical intervention. The branch pulmonary arteries may be stenotic or discontinuous with collateral supply from the aorta. Some patients also have multiple ventricular or atrial septal defects, anomalous systemic venous drainage, or atrioventricular valve abnormalities. A rare, complex form is tetralogy of Fallot with absent pulmonary valve, which results in pulmonary valve annulus hypoplasia and severe branch pulmonary artery dilation with significant bronchial compression and airway abnormalities, including bronchomalacia.
The clinical presentation of TOF is directly related to the severity of RVOT obstruction. The intensely cyanotic newborn infant generally has severe pulmonary stenosis or atresia with markedly diminished pulmonary blood flow and right-to-left shunting across the VSD. The minimally cyanotic or acyanotic infant has a lesser degree of pulmonary stenosis with mainly left-to-right shunting across the VSD. On exam, in neonates with TOF, the primary murmur is a systolic ejection murmur along the left sternal border secondary to pulmonary stenosis/RVOT obstruction. There may also be a single second heart sound due to marked diminishment of its pulmonary component and a palpable right ventricular impulse. The VSD in TOF is generally large and unrestrictive and does not produce a significant murmur. A continuous murmur may suggest systemic-to-pulmonary collaterals as the source of pulmonary blood flow.
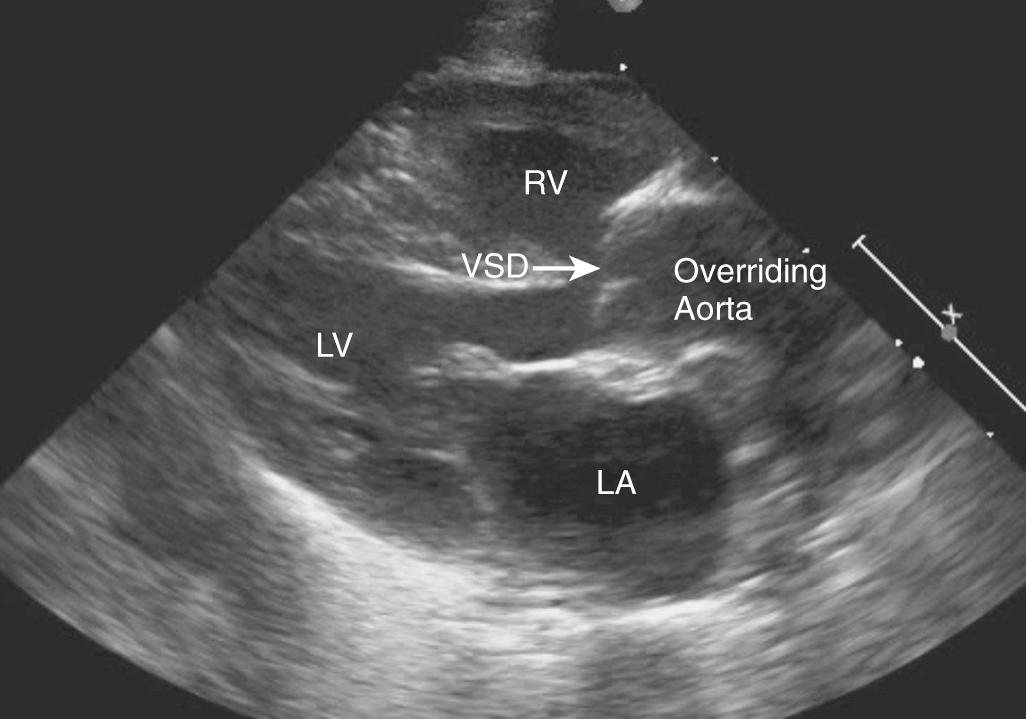
In the newborn with TOF, the classic finding on chest X-ray is a boot-shaped heart caused by right ventricular hypertrophy and consequent upturned cardiac apex. The pulmonary vascular markings should also be carefully examined and may indicate the severity of RVOT obstruction. Echocardiography is the primary diagnostic tool and illustrates the anterior malalignment VSD with RVOT obstruction, right ventricular hypertrophy, and overriding aorta ( Fig. 75.2 ). The levels and degrees of pulmonary obstruction and the size of the main and branch pulmonary arteries should be elucidated, as well as other associated abnormalities, including coronary artery anomalies, additional VSDs, or collateral vessels. Presurgical diagnostic cardiac catheterization is only indicated if the pulmonary artery anatomy and pulmonary blood flow sources cannot be defined by noninvasive imaging modalities.
Neonates with severe cyanosis in the immediate postnatal period are stabilized via intravenous prostaglandin E 1 infusion to maintain patency of the ductus arteriosus and increase pulmonary blood flow. In general, these neonates require a palliative systemic-to-pulmonary shunt (e.g., Blalock-Thomas-Taussig or central shunt) or complete repair in the newborn period to provide a reliable source of pulmonary blood flow. Balloon dilation or stenting of the stenotic pulmonary valve or RVOT has been favored in a few centers as an alternative to shunts. Rehabilitation of branch pulmonary artery stenosis by surgery or interventional catheterization is another important step at the time of primary repair or as part of a staged approach. This is of particular importance in patients with pulmonary atresia and major aortopulmonary collaterals.
Complete TOF repair consists of relief of pulmonary stenosis and RVOT obstruction (often with a transannular patch) and patch closure of the VSD. Associated intracardiac defects such as atrial level defects are also addressed. In the absence of marked pulmonary artery hypoplasia or unfavorable coronary artery anatomy, surgery can be undertaken at virtually any age, with most centers doing elective repairs when infants are between 4-6 months of age. Neonatal TOF repair is feasible for babies with normal or appropriate pulmonary artery size at birth.
Tricuspid atresia (TA) is defined by platelike tissue in place of the tricuspid valve with no direct communication between the right atrium and right ventricle. Systemic venous return from the vena cavae enters the right atrium and crosses the atrial septum with resultant complete mixing of the systemic and pulmonary venous return in the left atrium. Pulmonary blood flow is supplied by left-to-right shunting through either a PDA or VSD. Associated right ventricular hypoplasia and pulmonary artery hypoplasia are proportional to the size of the VSD and the degree of subpulmonary and pulmonary stenosis. Systemic hypoxia is proportional to the relative systemic and pulmonary blood flows. Neonates with tricuspid atresia, a small VSD, and severe pulmonary stenosis have severely limited pulmonary blood flow and are ductal dependent. Those with a large VSD and no pulmonary stenosis have high pulmonary blood flow, and when PVR falls in the postnatal period, pulmonary flow dramatically increases with resultant pulmonary overcirculation and risk for respiratory issues and feeding/growth failure.
Tricuspid atresia is not a diagnosis that occurs in isolation. All neonates with TA must have an atrial level defect for postnatal survival. Other associated lesions include VSD, transposition of the great arteries (TGA), atrioventricular septal defect, and aortic coarctation. The size of the VSD is extremely important in neonates with TA with TGA since oxygenated blood flows through the VSD into the right ventricle and then the aorta. Any restriction through the VSD in this case severely compromises systemic circulation.
The typical neonatal presentation of TA is cyanosis at or before ductal closure in an infant with a murmur. Precordial activity is normal, and the second heart sound is often single. There may be a systolic ejection murmur if there is RVOT obstruction. If the VSD is large, tachypnea, a left ventricular impulse, and a third heart sound may develop during the first few days of life as the PVR falls. Hepatic enlargement is an indicator of a restrictive atrial septal defect and elevated right atrial pressures.
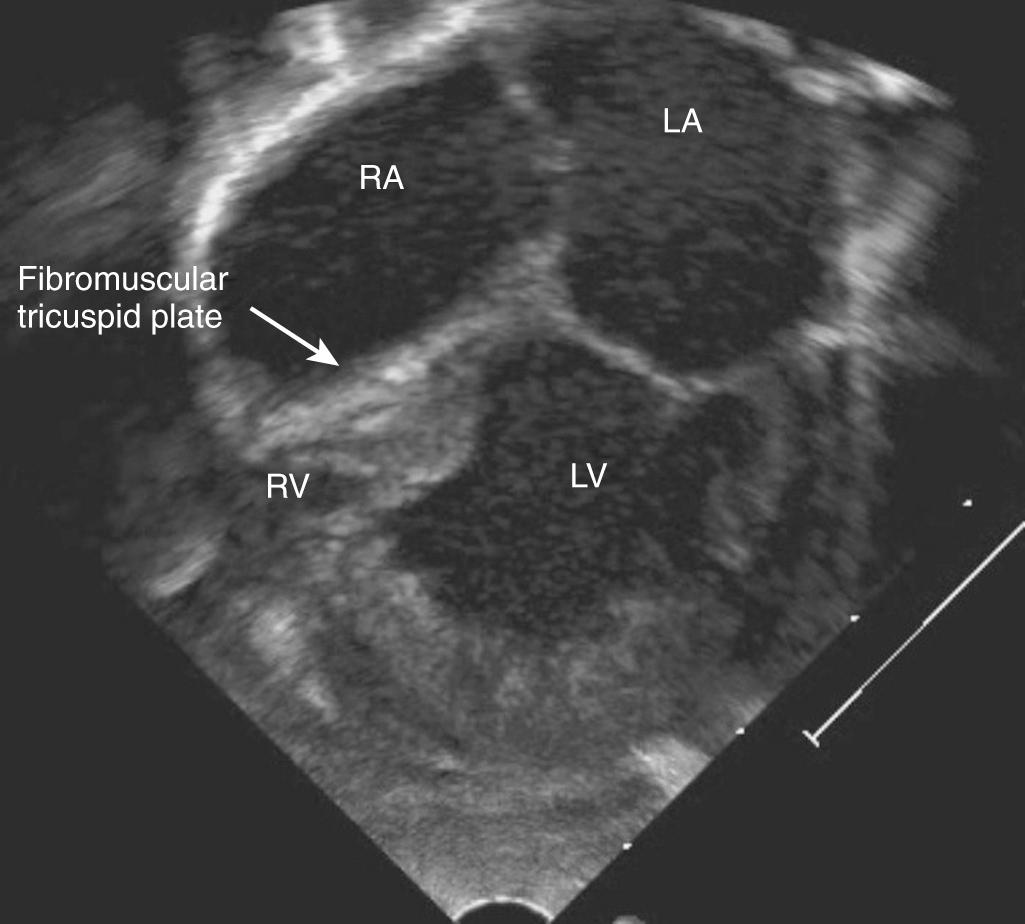
The electrocardiogram is usually diagnostic in neonates with TA, demonstrating right atrial enlargement and left axis deviation. The chest radiograph tends to be nonspecific, although severely cyanotic neonates show decreased pulmonary vascularity. The echocardiogram ( Fig. 75.3 ) clearly defines the anatomy in this disorder, particularly the RVOT, pulmonary arteries, atrial level defect, and PDA, in addition to the presence or absence of associated anomalies such as TGA or aortic coarctation. Catheterization is indicated only for balloon atrial septostomy in those with a restrictive atrial septum.
Flow across the VSD to supply the pulmonary circulation may be adequate in the first months of life as long as the atrial level defect remains nonrestrictive. It is generally possible to predict by echocardiogram when infants require a PDA to maintain adequate arterial saturations. A balloon atrial septostomy is rarely necessary unless there is significant restriction at the atrial level. If an infant is ductal-dependent or if hypoxia increases in the first month of life, a systemic-to-pulmonary shunt is necessary to provide adequate pulmonary blood flow and oxygenation. Conversion to a bidirectional cavopulmonary (Glenn) anastomosis is usually considered when an infant is between 3 and 6 months of age. Some infants with well-balanced systemic and pulmonary flows can proceed directly to an early bidirectional Glenn operation or a total cavopulmonary (Fontan) operation at about 2-3 years of age. Among children who undergo the Fontan type of operation, those with TA have excellent long-term prognosis, with a low prevalence of ventricular dysfunction, mitral regurgitation, arrhythmias, and systemic venous congestion.
The pulmonary valve in pulmonary atresia with intact ventricular septum (PA/IVS) can range from a well-formed pulmonary annulus with a plate that obstructs outflow to complete absence of the valve. All forms may lead to severe right ventricular hypertrophy with right ventricular dysfunction and varying right ventricular hypoplasia. Since the ventricular septum is intact, pulmonary blood flow in PA/IVS is ductal-dependent and can only occur via shunting at the atrial level and flow through a PDA. The tricuspid valve is rarely normal in patients with PA/IVS, ranging from severely hypoplastic and stenotic to regurgitant.
A very important prognostic feature in PA/IVS concerns the presence or absence of ventriculocoronary connections (sinusoids and fistulae) and coronary artery abnormalities. These must be well defined prior to surgical or transcatheter intervention. In the case of right ventricular (RV)–dependent coronary circulation, where coronary artery flow depends on high RV pressures for adequate myocardial perfusion, any decompression of the RV can lead to untoward consequences such as myocardial ischemia, infarction, and death.
Fetal diagnosis of PA/IVS frequently identifies risk factors for subsequent transcatheter and surgical management, including tricuspid valve and RV hypoplasia and coronary sinusoids and fistuale. Neonates with PA/IVS have severe hypoxia with PDA closure and can rapidly become unstable with tachypnea, tachycardia, hepatomegaly, and cardiorespiratory collapse. Cyanosis becomes apparent within hours of birth and is progressive. Tachypnea may be prominent. The first and second heart sounds are single, and there may be a holosystolic murmur heard best at the left lower sternal border consistent with tricuspid regurgitation. A PDA murmur may be audible, particularly following initiation of prostaglandin infusion. Unless there is severe tricuspid regurgitation or a restrictive atrial communication, the liver generally is not enlarged while ductal patency is maintained.
Chest radiography in PA/IVS often shows cardiomegaly, owing to right atrial enlargement. Echocardiography is diagnostic, demonstrating the pulmonary atresia, tricuspid valve abnormalities, RV hypoplasia severity, and coronary anomalies, including sinusoids and fistulae. Cardiac catheterization is indicated to assess coronary flow, the presence (including size and location) or absence of coronary sinusoids and fistulae, and pulmonary blood flow sources and for consideration of pulmonary valve intervention.
Intravenous prostaglandin E 1 infusion is essential for maintaining ductal patency in PA/IVS. Careful assessment of the anatomy and physiology provides a framework for transcatheter and surgical management. Balloon pulmonary valvuloplasty is feasible in infants with even a tiny orifice in the pulmonary valve, and other transcatheter techniques, including radiofrequency perforation of the valve, may be considered. If interventional catheterization is unsuccessful, surgical valvotomy is indicated unless there is clear RV-dependent coronary flow. In that circumstance, RV decompression can result in coronary hypoperfusion, so a systemic-to-pulmonary shunt is recommended as initial palliation to provide pulmonary blood flow. In infants with successful surgical or transcatheter valvotomies, RV growth is possible and biventricular circulation can be restored. Severe tricuspid and RV hypoplasia or RV-dependent coronary circulation is an indication for single ventricle palliation. Infants with RV hypoplasia should also be considered candidates for superior cavopulmonary (Glenn) anastomosis combined with closure of the atrial level defect (the so-called 1.5 ventricle repair). This results in direction of inferior vena cava flow across the tricuspid valve into the RV, promoting potential RV growth.
In Ebstein anomaly of the tricuspid valve, there is arrested delamination of the leaflets during valve development. In particular, the septal leaflet can become tethered to the interventricular septum to varying degrees with apical displacement of its annular attachment, potentially leading to significant tricuspid regurgitation. The anterior tricuspid leaflet can also become markedly enlarged and redundant (“sail-like”), causing RVOT obstruction with decreased blood flow and functional or anatomic pulmonary atresia. Secondary to apical tricuspid annular displacement into the right ventricle, a portion of the RV becomes atrialized, detrimentally affecting its contractile efficiency. Forward pulmonary blood flow is, therefore, dependent on the severity of tricuspid valve displacement and regurgitation and RV atrialization and dysfunction. If functional or anatomic pulmonary atresia occurs, an obligatory right-to-left shunt must exist at the atrial level. The degree of this atrial shunt is proportional to the severity of the tricuspid valve abnormality.
Patients with Ebstein anomaly often have other associated intracardiac lesions, including the aforementioned pulmonary atresia and atrial level defects. Conduction system abnormalities such as accessory pathways (e.g., Wolff-Parkinson-White syndrome) are found in up to 20% of patients.
Infants with severe manifestations of Ebstein anomaly may present with profound cyanosis, tachypnea, and cardiovascular collapse. Cyanosis is dependent on the extent of atrial right-to-left shunting. A low-frequency holosystolic murmur of tricuspid regurgitation is often heard on exam, and the first heart sound may be widely split.
The electrocardiogram of infants with severe Ebstein anomaly shows an RSR′ pattern across the right side of the chest, suggesting RV dilation. Chest radiography can demonstrate severe cardiomegaly caused by right atrial dilation. Echocardiography helps delineate the extent of tricuspid valve displacement, atrial level shunting, and functional or anatomic pulmonary atresia. The ratio of the right atrial plus atrialized RV size to the functional RV plus left heart (left atrium and ventricle) size has been a useful measure of severity and outcome.
Mild forms of Ebstein anomaly require no specific treatment, and these infants in general do well. The severely affected neonate with severe tricuspid valve displacement and regurgitation is often ductal dependent with minimal or no anterograde flow across the pulmonary valve. Maintaining ductal patency with prostaglandin E 1 infusion can be useful in the short term. However, ductal flow into the pulmonary artery can make anterograde flow more difficult. In severe Ebstein anomaly, measures to lower the pulmonary vascular resistance, including nitric oxide, and several attempts to wean prostaglandin E 1 might be necessary before anterograde flow across the pulmonary valve can be established. Brief support with extracorporeal membrane oxygenation to facilitate this fetal to neonatal transition has been successful in limited cases. Surgical repair or replacement of the valve is feasible in older children, but this is not useful in neonates. Either surgical exclusion of the right ventricle, with plans for a long-term, single-ventricle palliation, or transplantation might be the only alternatives for the neonate with severe Ebstein anomaly and persistent cyanosis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here