Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
A multicystic dysplastic kidney (MCDK) is the most common form of cystic renal dysplasia. With high-grade obstruction or atresia of the upper urinary tract (renal pelvis and/or ureter) during early renogenesis, disordered parenchymal development results in an MCDK. Two types of MCDK exist: the pelvoinfundibular type (common) and the hydronephrotic type (uncommon). MCDK can occur in half of a duplex kidney (usually the upper moiety) or in renal fusion anomalies, such as a horseshoe kidney and crossed fused renal ectopia. MCDK is sporadic in most cases, although familial cases have been described. Vesicoureteral reflux (VUR) (both ipsilateral [into an atretic ureter] and contralateral) and contralateral ureteropelvic junction (UPJ) obstruction are associated with this anomaly, and Müllerian anomalies may coexist in girls. Long-term sequelae (e.g., infection and hypertension) are rare, because most affected kidneys either partially or completely involute on their own over time.
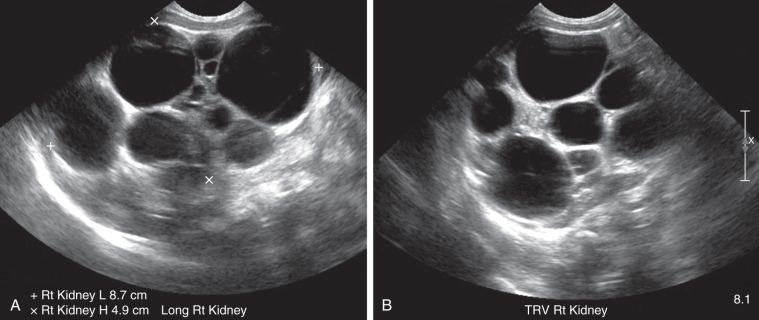
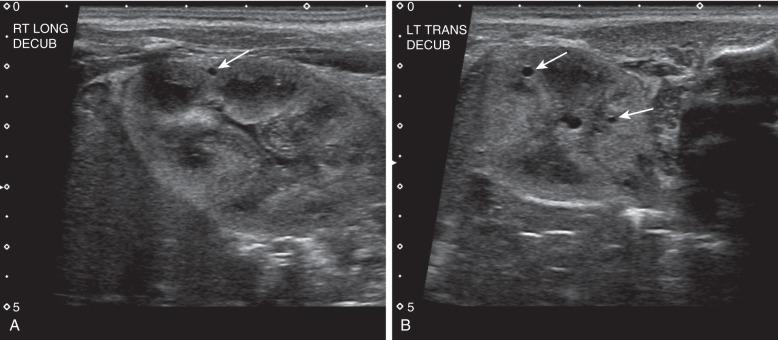
On ultrasound, computed tomography (CT), and magnetic resonance imaging (MRI), a classic MCDK is characterized by multiple cysts of various sizes (ranging from 1 mm to several cm) that do not communicate, with no identifiable normal renal parenchyma ( Fig. 113.1 ). The size of the affected kidney may be small, normal, or enlarged. Compensatory hypertrophy of the contralateral kidney often is present. The hydronephrotic form has cysts that communicate, mimicking pelvicaliectasis, but no normal functioning renal tissue. The diagnosis of both pelvoinfundibular and hydronephrotic MCDK can be confirmed by diuretic renal scintigraphy ( e-Fig. 113.2 ) or magnetic resonance urography (MRU) ( e-Fig. 113.3 ). A voiding cystourethrogram (VCUG) may reveal VUR into a blind-ending ureter on the side of the MCDK.
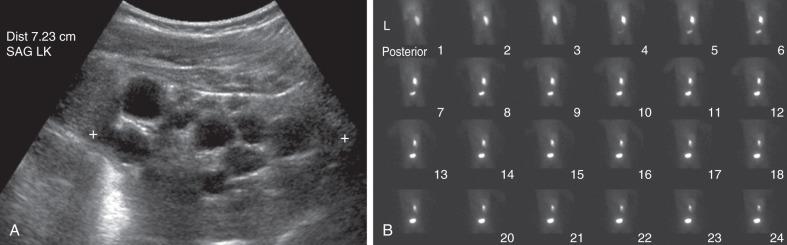
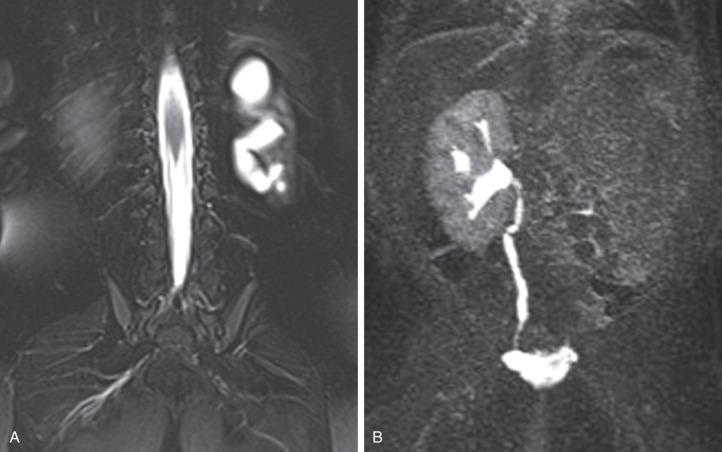
Follow-up ultrasound typically demonstrates involution of the cysts over time, eventually resulting in remnant dysplastic renal tissue that may be difficult to visualize ( e-Fig. 113.4 ). Resection of an MCDK is rarely indicated and is usually reserved for cases in which there is increasing kidney size rather than involution. In the past, routine nephrectomies were performed due to a fear of malignant degeneration. Current evidence discounts this possibility, and, if no other complication exists, surgery is not indicated and limited radiologic follow-up may be appropriate.
Autosomal dominant polycystic kidney disease (ADPKD), the most common form of inherited renal cystic disease, is now recognized to be a ciliopathy (disorder of the primary cilia) and is seen in about 1 in 800 live births. Two types of ADPKD exist, each with clearly defined chromosomal mutations. The more severe form (PKD1) accounts for about 85% to 90% of cases, whereas the milder phenotype (PKD2) presents later in life, results in less morbidity, and is less common. Cysts arise from both the renal cortex and the medulla.
Systemic hypertension, hematuria, and slowly progressive chronic kidney disease are the typical clinical manifestations. ADPKD is very rarely detected prenatally because fewer than 5% of nephrons are cystic before birth. Diagnosis of ADPKD early in life portends a poor prognosis; 43% of affected persons die in the first year of life, hypertension develops in 67%, and some affected children progress rapidly to end-stage renal disease. Because in some instances it can be difficult to delineate between autosomal recessive and autosomal dominant forms of polycystic kidney disease, screening of siblings and parents may be helpful. ADPKD can coexist with tuberous sclerosis as a “contiguous gene syndrome,” and the recognition of this specific syndromic form has implications for management.
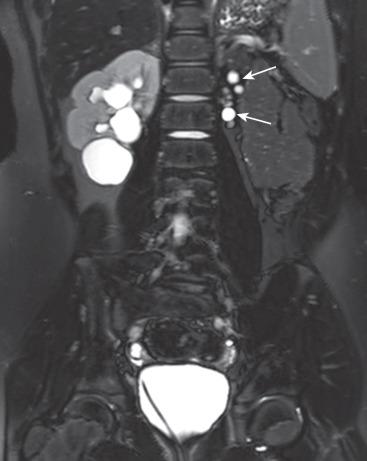
The diagnosis of ADPKD usually is made on the basis of ultrasound ( Fig. 113.5 ), although CT or MRI also can suggest it. While the kidneys may be normal during the first decade of life, they also may contain one or more simple cysts ( Fig. 113.6 ). The kidneys may be abnormally enlarged and echogenic, mimicking autosomal recessive polycystic kidney disease (ARPKD) in early life Although it is typically a bilateral process, imaging findings may be asymmetric, and in young children they can manifest as a unilateral abnormality. Microscopic cysts, as well as larger cysts that can be detected by cross-sectional imaging techniques, can involve the liver and less frequently other organs. Nephrolithiasis may be seen at ultrasound or CT. The remainder of the urinary tract is usually normal.
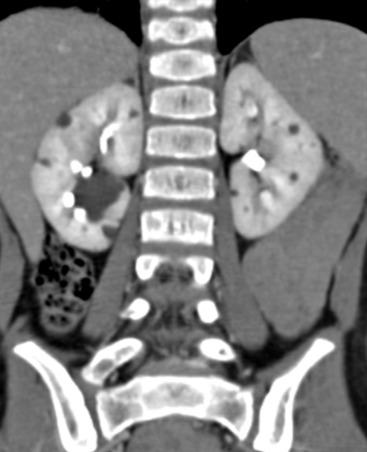
Although renal function is generally maintained during childhood, ADPKD is responsible for the need for chronic dialysis in 10% to 12% of adult dialysis patients. Hypertension should be managed medically to prevent long-term complications. While 12% to 15% of patients with ADPKD have an intracranial aneurysm, screening magnetic resonance (MR) angiography usually is not performed in childhood because aneurysm rupture is rare in this period.
ARPKD is a rare disorder that is seen in about 1 : 20,000 live births and that is also recognized to be a ciliopathy. Genetic studies have localized the inheritance of ARPKD to chromosome 6p. The PKHD1 gene is expressed in fetal and adult kidneys and livers, the two major sites affected by the disease. Approximately 1 in 70 persons is a carrier for this condition. Histologically, the kidneys have a spongelike appearance, replaced by myriad mostly minute 1- to 2-mm cysts of relatively uniform size, representing dilated and elongated collecting ducts. These cysts radiate from the hilum toward the surface of the kidney without a clear demarcation between the medulla and cortex; fibrosis is present in the renal interstitium.
ARPKD has been subdivided into categories based on clinical presentation. The perinatal and neonatal forms, which have the poorest prognosis, are associated with severe renal dysfunction, and affected patients may die of the disease early in life. While the degrees of renal and hepatic abnormalities historically have been considered to be inversely proportional, a recent study suggests that the severity of renal and liver involvement are independent. Liver involvement may be the cause of clinical presentation in a minority of affected children due to ductal plate malformations (e.g., congenital hepatic fibrosis and intrahepatic bile duct ectasia, or Caroli syndrome) and associated complications, such as portal hypertension and cholangitis.
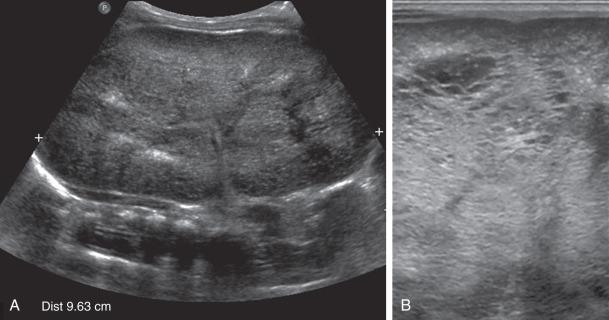
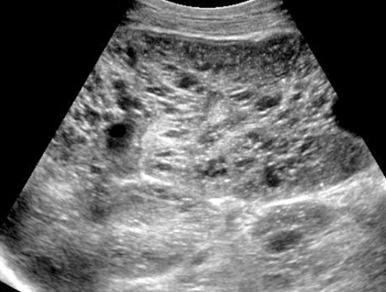
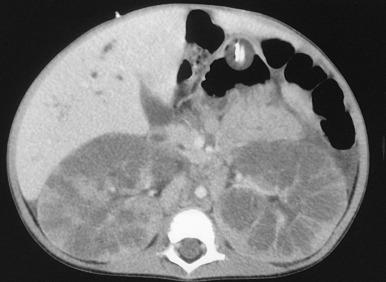
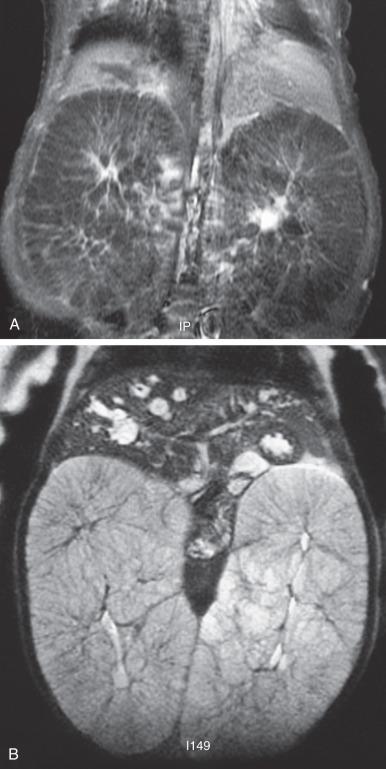
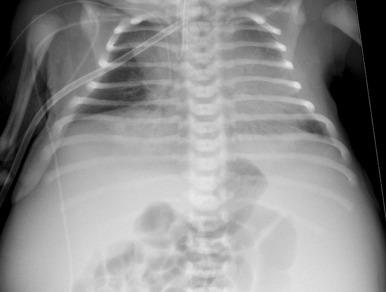
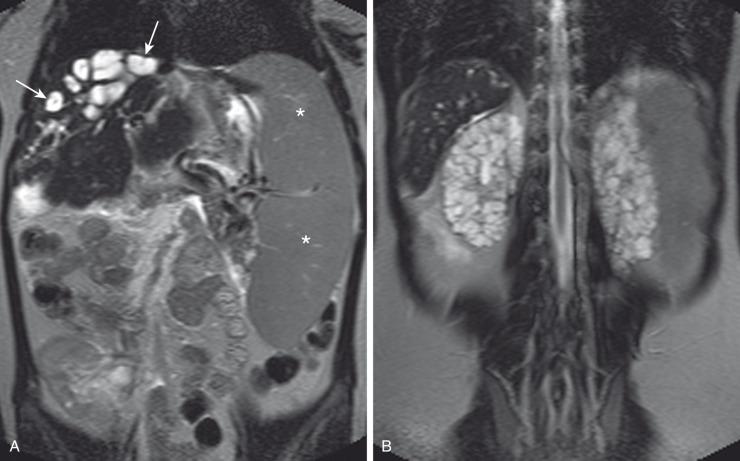
Abdominal radiography in the neonatal period commonly reveals bilateral abdominal or flank masses with centralized gas-filled bowel loops. The lungs may appear small because of a combination of pulmonary hypoplasia and mass effect from marked nephromegaly ( e-Fig. 113.7 ). On ultrasound, affected neonatal kidneys tend to be smoothly enlarged (frequently more than four standard deviations above the mean expected length for age), are hyperechoic as a result of the multiple acoustic interfaces at the walls of the dilated ducts, and have poor corticomedullary differentiation ( Fig. 113.8 ). On occasion, discrete cysts (solitary or multiple) of variable size (most commonly <1 cm) may be seen ( Fig. 113.9 ). A sonolucent rim may be seen at the periphery of the kidney as a result of cortical compression and relative sparing. Contrast-enhanced CT ( Fig. 113.10 ), while rarely performed, can show delayed opacification of the renal parenchyma with a prolonged and striated nephrogram. When studied with MRI, the dilated tubules in affected kidneys can appear linear and hyperintense on T2-weighted imaging, radiating from the medulla toward the cortex giving rise to a “spoke wheel” appearance ( Fig. 113.11 ). Ultrasound and MRI are most often used to evaluate for liver involvement, typically showing altered liver echogenicity and signal intensity due to congenital hepatic fibrosis, dilated intrahepatic bile ducts (sometimes appearing cystic and showing the central dot sign), and splenomegaly ( e-Fig. 113.12 ).
Eighty-six percent of patients who survive beyond 1 month of age are alive at 1 year, and 67% are alive at 15 years. Bilateral nephrectomy and renal replacement therapy may be required in neonates or infants when renal enlargement is massive and causes symptomatic mass effect or pulmonary compromise. In infants who have initially adequate (although not normal) renal function, the disease may not be detected until later in the first decade. Systemic renovascular hypertension may occur and require medical management. Many patients with ARPKD eventually will require renal replacement therapy, either dialysis or renal transplantation; combined liver-kidney transplantation may be performed in some children.
Other uncommon genetic disorders (syndromes) are associated with renal cysts ( Box 113.1 ). Recent studies suggest that several of these conditions are also due to genetic mutations that affect primary cilia or centromere function (sometimes referred to as nephronopthisis ), with a variety of genes implicated. Depending on the specific mutation, clinical presentation, associated anomalies, and age of presentation with regard to renal disease may vary.
Tuberous sclerosis
von Hippel-Lindau disease
Trisomies 13 and 18
Medullary cystic kidney disease
Other ciliary disorders/nephronophthisis
Asphyxiating thoracic dystrophy (Jeune syndrome)
Bardet-Biedl syndrome
Meckel-Gruber syndrome
Joubert syndrome
Oral-facial-digital syndrome type 1
Short rib polydactyly
Zellweger syndrome (cerebrohepatorenal syndrome)
A calyceal diverticulum represents a focal outpouching of the renal collecting system that is located within the renal parenchyma and that contains urine. This condition has an incidence of 3.3 to 4.5 : 1000 in children, based on excretory urography. Calyceal diverticula are likely developmental in origin; they are lined with urothelium and surrounded by muscularis mucosa. Although these structures most commonly are an incidental finding, they can be associated with pain as well as infection and calculus formation as a result of urinary stasis.
Abdominal radiographs may be normal or may demonstrate one or more calculi (or milk of calcium) within a calyceal diverticulum. On ultrasound, such diverticula may mimic a solitary renal cyst, appearing round, thin-walled, and anechoic. They can vary in size ranging from a few millimeters to several centimeters (with a mean size around 11 mm), and they most commonly occur within the upper pole of the kidney. Calyceal diverticula can rarely be bilateral and/or multiple. Upon excretory phase imaging (including MRI and CT), calyceal diverticula may fill with contrast material ( Fig. 113.13 ). A thin connection, or neck, to an adjacent calyceal fornix may or may not be identified.
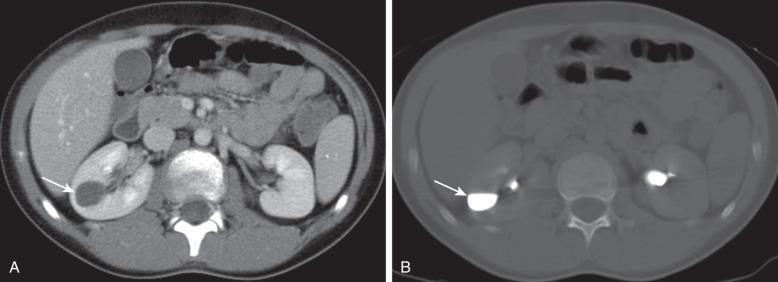
A symptomatic calyceal diverticulum may require endoscopic or laparoscopic ablation. Although both lithotripsy of calculi within the diverticulum and percutaneous ablation of the cavity have been described in adults, such techniques have not been applied widely to children.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here