Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Colonic emergencies result from obstruction, inflammation/infection, perforation, haemorrhage or ischaemia. Herein we discuss aetiology, pathogenesis, presentation and management. Emergency compared with elective colon surgery is associated with a two- to threefold increase in mortality, hence interventions to convert an emergency into an elective operation should be considered when safe to do so. In addition to a thorough history and physical examination, patients must be resuscitated and optimised as far as practicable prior to proceeding to the operating room. Multidisciplinary approaches to patient care and thorough radiographic and colonoscopic evaluation are often extremely beneficial for planning and treating this complex population. When possible, patients should be preoperatively evaluated, educated and site-marked by stoma therapists. If facilities are inadequate, transfer to a referral centre should be considered.
Colonic obstruction can occur secondary to multiple causes ( Table 16.1 ) and can be partial or complete, intrinsic or extrinsic, and adynamic or mechanical. The most common causes are neoplasia, diverticulitis and volvulus. Treatment for obstruction is dependent on the location, cause, patient presentation and goals of care.
| Neoplasm |
| Volvulus |
| Diverticulitis |
| Pseudo-obstruction |
| Hernia |
| Stricture |
| Faecal impaction |
| Inflammatory |
| Intussusception |
| Endometriosis |
| Adhesions |
| Ischaemia |
A distal colonic obstruction in the presence of a competent ileocaecal valve hindering retrograde flow of enteric content, or colonic volvulus, will result in a closed loop obstruction. Antegrade flow of enteric content and bacterial overgrowth progressively increase intraluminal pressure, which is followed by venous occlusion, intramural hypoperfusion, arterial occlusion, thrombosis, ischaemia, necrosis and perforation. Consequently, a closed loop obstruction mandates expedited care.
Mild obstruction (stenosis) typically presents with cramping abdominal pain and constipation. Imaging may reveal an area of luminal stenosis. Endoscopic advancement through the stricture is often not possible. Colon proximal to the stricture is not dilated and surgery can be planned in an elective fashion. Severe obstruction presents with proximal bowel dilatation, pronounced abdominal distension, absolute constipation (failure to pass flatus or stool) and varying degrees of tenderness, which in combination with signs of systemic toxicity (elevated heart rate, leucocytosis) necessitate emergency surgery (ES).
Approximately 20% of patients with colon cancer will present with an obstruction, with the majority being elderly. Nearly 30% will have hepatic metastasis at the time of surgery. Obstruction secondary to colon cancer often presents with a constellation of symptoms including abdominal distension, alteration in bowel habits, weight loss, change in stool calibre and/or consistency, nausea, emesis, obstipation and absence of flatus. Symptoms usually develop slowly and typically reflect the site of obstruction. Right-sided masses typically cause obstructive symptoms (abdominal cramping, pain), whereas left-sided cancers are more commonly associated with a change in stool calibre, consistency and alteration in bowel habits.
Initial management includes bowel rest, analgesia, resuscitation and correction of laboratory abnormalities. Nasogastric tubes do not decompress the colon and are not necessary unless the patient is experiencing nausea/emesis. Plain abdominal radiographs will identify large bowel dilatation, and if the ileocaecal valve is incompetent, small intestine distension may also be noted. Computed tomographic (CT) imaging informs surgical planning and provides staging information which may be key in defining goals of care, and should be obtained unless immediate operative intervention cannot be delayed. If CT is not available, a water-soluble contrast enema will identify the level of obstruction and can be useful in distinguishing pseudo-obstruction (Ogilvie syndrome) from causes of mechanical obstruction. Endoscopy may be both diagnostic and therapeutic.
Colonic self-expandable metal stents (SEMS) may be used as a bridge to surgery for those with partial obstruction or for palliation in patients with inoperable disease. Although technical success is greater than 90%, complications include perforation (up to 13%), stent failure after technically successful positioning (up to 12%), stent migration (1–13%), re-obstruction (4–23%), pain (up to 7%) and bleeding (up to 4%). , Perforations are more common with intraluminal stents left in place for a prolonged period of time, stents crossing acute angles and in patients receiving antiangiogenic drugs such as bevacizumab. , Non-palliative stenting (‘bridge to surgery’) provides the opportunity to decompress the distended proximal colon, medically optimise patients in preparation for surgery, complete a preoperative colonoscopy, and pursue a single-stage operation using a minimally invasive approach. However, clinically silent microperforations associated with stents have been documented at the time of surgery, raising concerns regarding oncological outcomes. Small et al. at the Mayo Clinic identified five risk factors associated with complications from self-expandable colonic stents, which included male sex, complete colonic occlusion, balloon dilatation prior to stent placement, mid-stent diameter less than 22 mm, and placement by an interventionalist not familiar with pancreaticobiliary procedures. The first randomised prospective study comparing open surgery to SEMS followed by a minimally invasive resection (endolaparoscopic) for left-sided colon cancer was reported in 2009. No stent-related complications were reported, technical and clinical success for stenting was 83%, and only 38% underwent a single-stage operation in the open compared with 67% in the endolaparoscopic arm. All in the stent arm underwent ostomy reversal and six in the open group were left with a permanent stoma. Pirlet et al. in a French multicentre prospective randomised controlled trial compared the outcomes of ES to SEMS as a bridge for surgery. The primary outcome was the need for a stoma (temporary or permanent) for any reason. The secondary endpoints were mortality, morbidity and length of hospital stay. The study was closed early secondary to stent-related perforations. Among the 60 patients accrued, 17 in the open group versus 13 in the SEMS group were left with a stoma. The multi-institutional Dutch Stent-In Study Group evaluated colonic stenting as a bridge to elective surgery versus ES, with the primary outcome of mean global health status during a 6-month follow-up. The study was closed early secondary to increased morbidity in the SEMS group, and specifically six perforations in the SEMS versus none in the operative arm. They concluded that colonic stenting has no decisive clinical advantages to ES. Critics pointed out that centres not highly experienced in SEMS placement were involved in the study, which likely resulted in higher rates of perforation. Increased failures and complications have been described if stenting is performed by endoscopists who have placed less than 40 SEMS. A systematic review and meta-analysis of eight RCTs demonstrated that SEMS as a bridge to surgery, compared with ES, had lower short-term overall morbidity (33.9% in SEMS group vs 51.2% in ES group) and lower rates of temporary colostomy (33.9% in SEMS group and 51.4% in ES group). SEMS also achieved a lower permanent stoma rate (22.2% for SEMS vs 35.2% for ES) and an increased success of primary anastomosis (70% in SEMS group and 54.1% in ES group), with no difference in overall mortality within 60 days after surgery. Foo et al. performed a meta-analysis of seven randomised controlled trials evaluating oncologic outcomes and found that SEMS had a significantly higher overall recurrence rate than ES, although disease-free and overall survival at 3 years were similar. The multicentre randomised controlled ESCO trial recently compared morbidity rates after colonic stenting as a bridge to surgery versus ES in the management of left-sided malignant large-bowel obstruction and found that the two treatment strategies were equivalent. There was no difference in oncologic outcomes at a median follow-up of 36 months, and there was a significantly lower stoma rate noted in the SEMS group. Preliminary results from the UK ColoRectal Endoscopic Stenting Trial (CREST) revealed that 30-day postoperative mortality, length of hospital stay, critical care usage and quality of life were not different between the two treatment groups (SEMS and surgery). Long-term results are still pending. The European Society of Gastrointestinal Endoscopy (ESGE) recently updated their guidelines with recommendations for the use of SEMS.
![]() According to the European Society of Gastrointestinal Endoscopy (ESGE):
According to the European Society of Gastrointestinal Endoscopy (ESGE):
Colonic stenting to be reserved for patients with clinical symptoms and radiological signs of malignant large-bowel obstruction, without signs of perforation.
Stenting as a bridge to surgery to be discussed, within a shared decision-making process, as a treatment option in patients with potentially curable left-sided obstructing colon cancer as an alternative to emergency resection.
Colonic stenting as the preferred treatment for palliation of malignant colonic obstruction.
Consideration of colonic stenting for malignant obstruction of the proximal colon either as a bridge to surgery or in a palliative setting.
A time interval of approximately 2 weeks until resection when colonic stenting is performed as a bridge to elective surgery in patients with curable left-sided colon cancer.
Colonic stenting should be performed or directly supervised by an operator who can demonstrate competence in both colonoscopy and fluoroscopic techniques and who performs colonic stenting on a regular basis.
A decompressing stoma as a bridge to elective surgery is a valid option if the patient is not a candidate for colonic stenting or when stenting expertise is not available.
Resectable disease can be pursued in an oncological fashion in single or multiple stages. Choice of operative approach is multifactorial and dictated by surgeon experience, clinical condition of the patient and operative factors (tissue quality, contamination, synchronous cancer, proximal dilatation). For those not in extremis, a single-stage primary anastomosis with or without a protective diverting ostomy can be pursued. Morbidity and mortality have been shown to be equivalent between single- and multi-staged procedures in appropriate candidates. Laparoscopic approaches have typically been avoided secondary to lack of visualisation, risk of iatrogenic perforation and increased time to perform the procedure, although in the hands of skilled laparoscopic surgeons these are not absolute contraindications.
Some form of intraoperative colonic decompression is usually required to facilitate handling of the distended colon and visualisation of the intracoelomic cavity. Techniques to decompress through a laparotomy incision include needle aspiration, suction, lavage and endoscopic. However, on-table lavage of faeces has not been noted to decrease anastomotic complications.
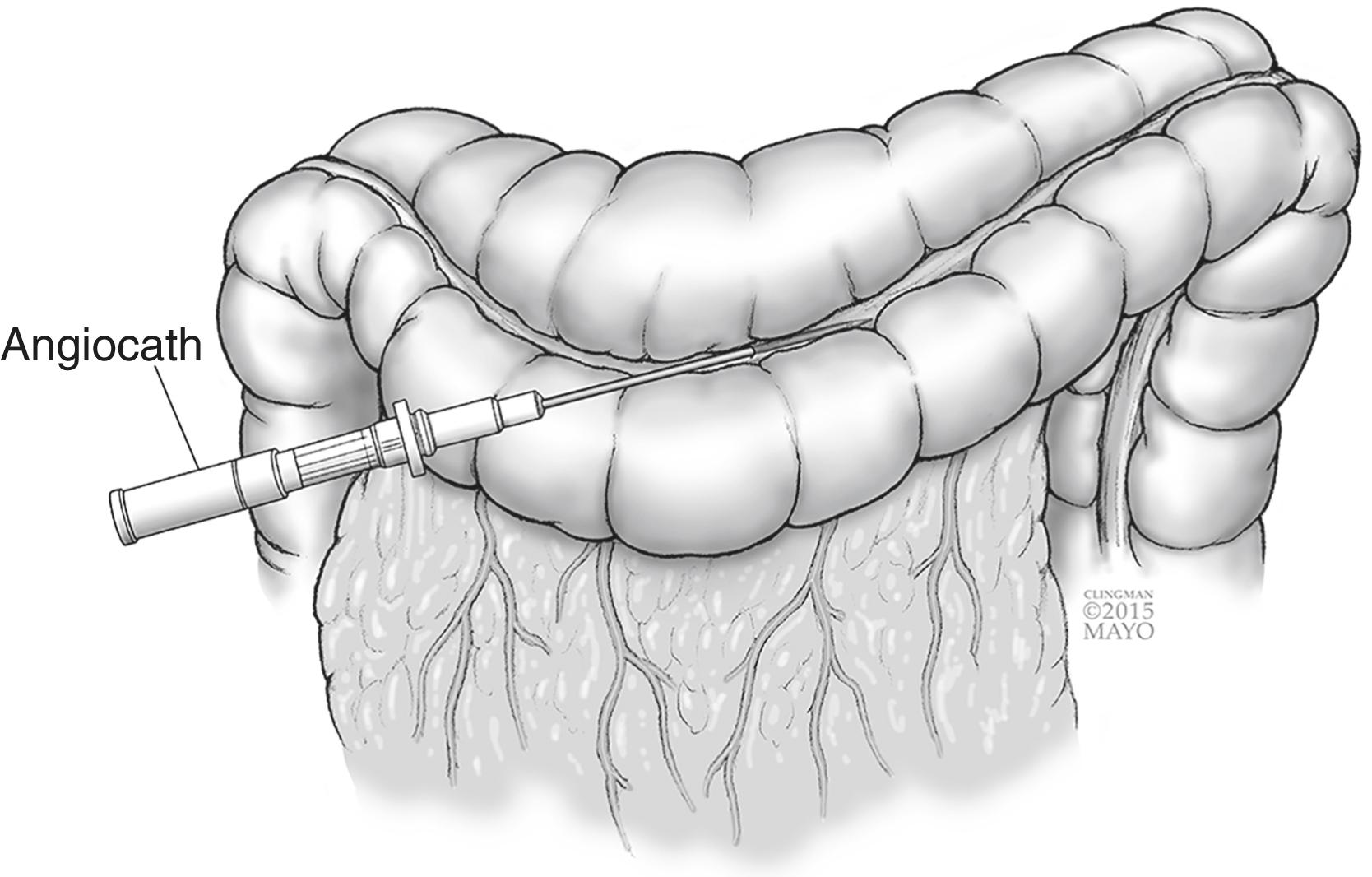
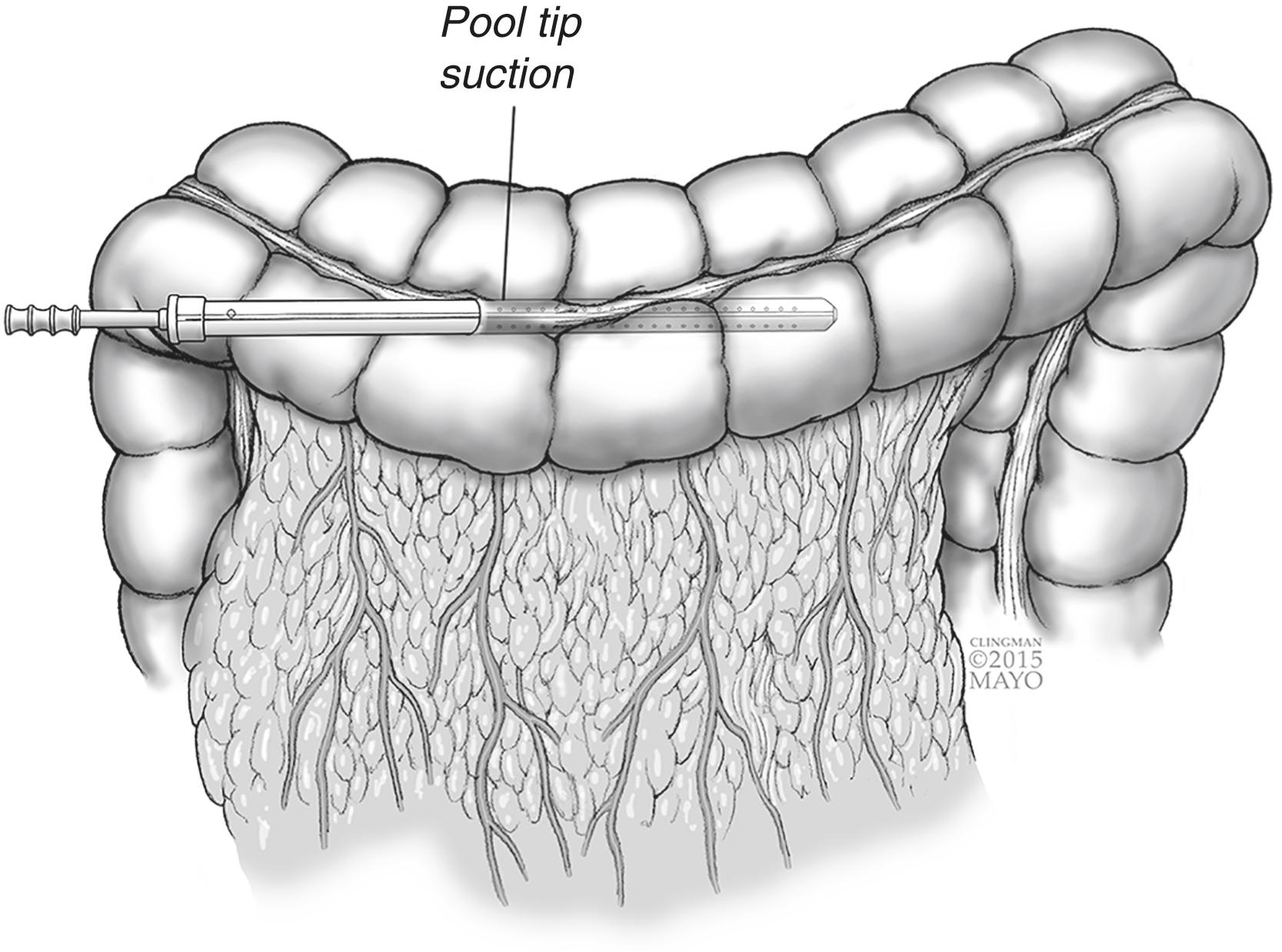
For needle decompression, a purse-string suture using a small needle is placed through the tenia of an accessible portion of dilated colon. A large angiocath (14-gauge or larger) is placed through the middle of the purse-string and the needle is removed leaving the soft flexible catheter sheath in the lumen of colon ( Fig. 16.1 ). The sheath is then connected to suction. Needle decompression works well to decompress gas, though it is too small to decompress stool and blood. If unable to decompress adequately, a pool tip suction catheter can be placed through the same defect after making a small colotomy ( Fig. 16.2 ). Once adequate decompression is obtained, the catheter can be removed and the purse-string suture secured.
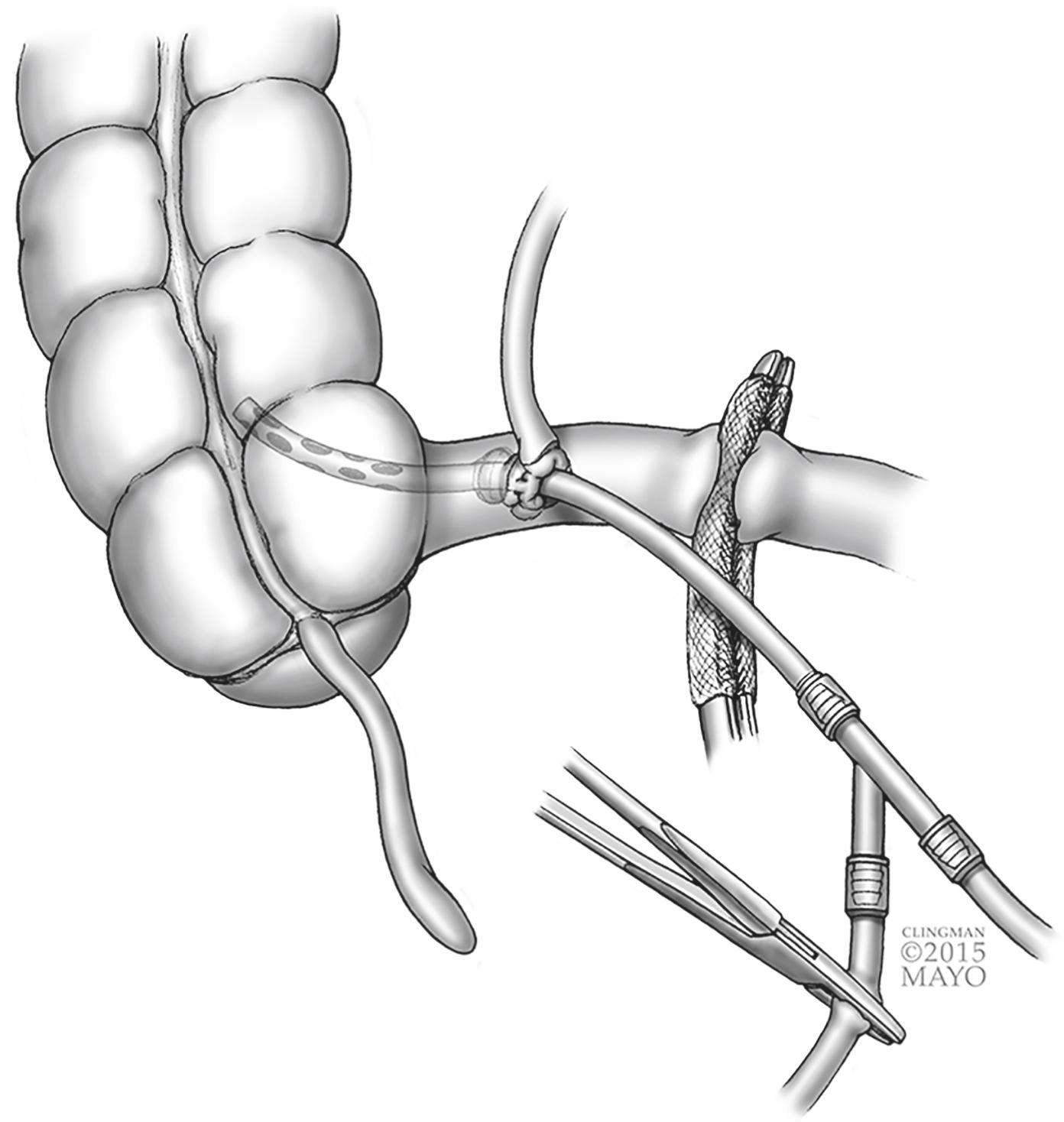
More thorough lavage can be carried out after placing a large-diameter catheter with multiple distal holes connected to a Y-connector through the ileocaecal valve into the right colon. One limb of the Y-connector is connected to irrigation and the other to suction ( Fig. 16.3 ). If the catheter becomes clogged, it can be readily irrigated. After decompression is achieved, the catheter can be removed and the purse-string secured, or pushed into the ascending colon and removed with the specimen for a right colectomy. Closed systems for retrograde colonic lavage are also available.
Evaluation for synchronous tumours, liver metastasis, local invasion and the presence of peritoneal carcinomatosis should be carried out. A curative resection, including en bloc resection of surrounding involved structures, is pursued when possible. Metastasis does not preclude resection and re-establishment of intestinal continuity. For palliation during surgery, a decompressive ostomy is the preferred approach in those with unresectable disease who are operative candidates. If creation of an ostomy is not possible, an intestinal bypass procedure can be considered. In rare instances, a loop ostomy can be performed under local anaesthesia for palliation in decompensated patients unable to tolerate general anaesthesia.
In Westernised countries, diverticular disease affects around 40% by age 60 and 60% by age 80, and the diagnosis appears to be rising. Acute diverticulitis occurs in 10–25%, and of those 20–25% experience a complicated form (perforation, abscess, stricture, fistula). Recurrent episodes of diverticulitis can result in fibrosis of the colon wall, stricturing and obstructive symptoms. Approximately 10% of colonic obstructions result from diverticular strictures.
Initial management and imaging is as noted above. For obstruction presenting with a concomitant abscess, active inflammation or phlegmon, a trial of conservative treatment including antibiotics, with or without interventional radiology-guided procedures, may allow the obstruction to resolve as inflammation improves.
Urgent/ES is rarely performed for colonic strictures secondary to diverticular disease and only in cases of complete obstruction. Partial obstruction solely related to stricturing is pursued in an elective fashion. SEMS can be used as a bridge to surgery allowing for decompression of the distended proximal colon, provide the opportunity to medically optimise patients, complete a preoperative colonoscopy and pursue a single-stage surgery. Few studies describe SEMS for diverticular strictures and most are single-institution experiences. Small et al. described the use of stents in 23 patients, of whom 16 were associated with diverticular disease. Complications included perforation (4), migration (2) and re-obstruction (2), and 87% of the time occurred 7 days after placement. An emergency procedure was converted to an elective surgery for 84%, although the authors recommended that resection should be attempted within 7 days of SEMS placement. Multiple other small studies have also reported high rates of complications; mainly perforation, migration, obstruction and no improvement. Using SEMS for diverticular strictures is unproven at this time and may not represent a valid therapeutic option unless the patient is unfit for surgery.
Operative approaches include open or minimally invasive segmental resection, with or without a protective diverting ostomy, and Hartmann’s procedure. It is important to note that in many cases it may not be possible to distinguish diverticular stricture from malignancy preoperatively, in which case resection should follow oncological principles given that malignancy is more frequent. As noted above, the choice of operation and need for ostomy is dependent on the condition of the patient, tissues and the experience of the surgeon. Procedures for decompressing the colon apply as described above.
Sigmoid volvulus is for the most part an acquired condition and encompasses 60–80% of all colonic volvulus. In Westernised countries, it is most commonly associated with the elderly, debilitated, neurological conditions, those institutionalised or taking psychiatric medications and laxative abuse. Males and females are equally affected. Although the aetiology is unknown, it is thought to be secondary to constipation and colonic lengthening. Sigmoid volvulus in younger age groups is more common in non-Westernised countries. Worldwide, sigmoid volvulus is the most common cause of bowel obstruction during pregnancy. Congenital anomalies such as Hirschsprung’s disease, gut malrotation and parasitic disease caused by Trypanosoma cruzi (Chagas disease) are also associated with sigmoid volvulus. Volvulus of the sigmoid colon can occur in either a clockwise or counter-clockwise orientation as the colon elongates and rotates on a narrow pedicled mesenteric base.
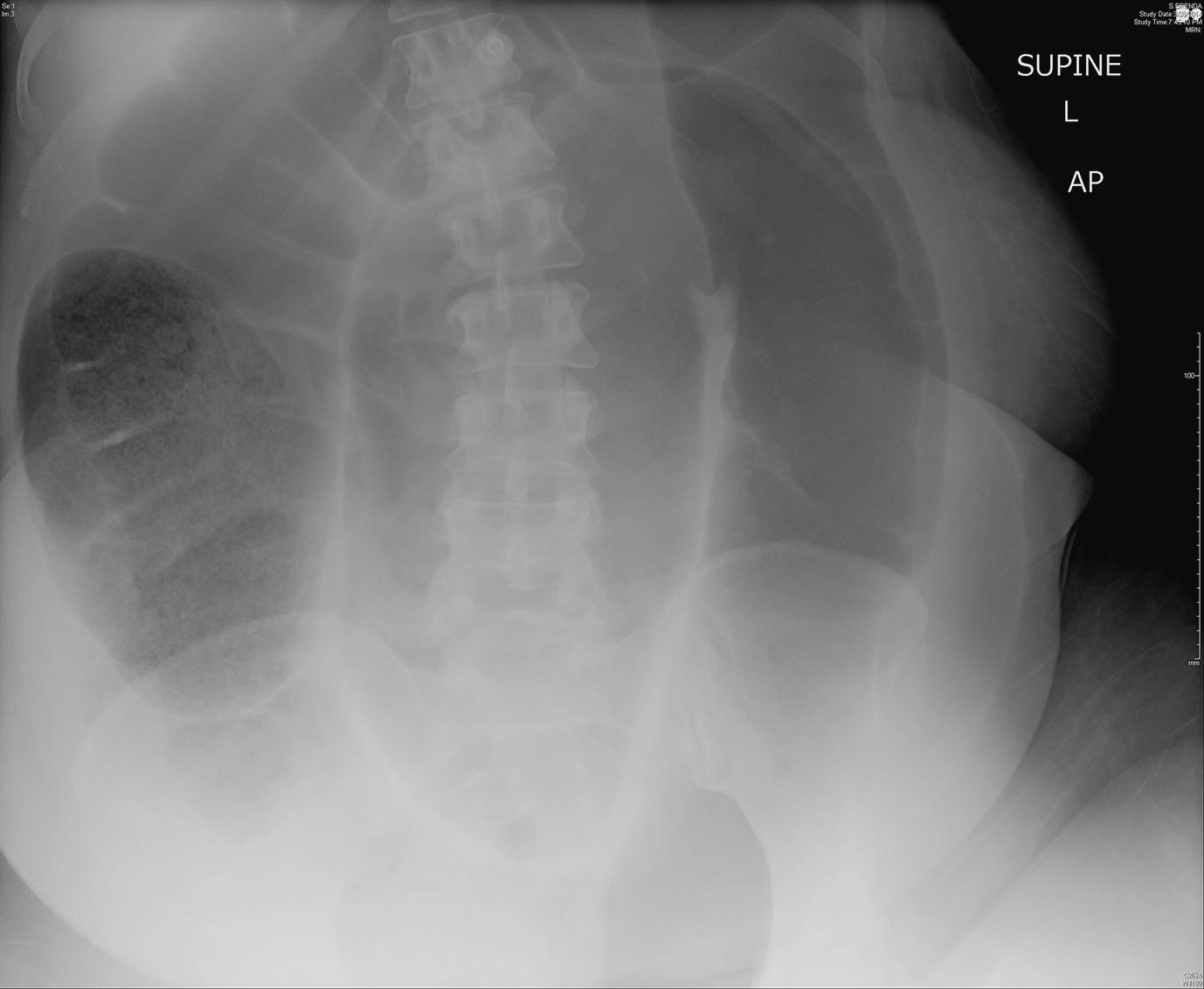
Although a few patients describe symptoms of recurrent distension followed by spontaneous detorsion resulting in explosive large-volume bowel movements, the vast majority present as an emergency with abdominal pain, distension and absolute constipation. Diagnosis can be made most of the time with an abdominal X-ray revealing a dilated colon in the shape of a ‘coffee bean’ or ‘omega loop’ with its apex pointing toward the right upper quadrant, though only present radiographically in 50–80% ( Fig. 16.4 ). A water-soluble contrast enema reveals a ‘bird’s beak’ appearance in the rectosigmoid region, which is secondary to narrowing at the level of the torsion. CT has become the imaging modality of choice and in addition to a dilated loop of colon often reveals a characteristic ‘whirl sign’ whereby the mesentery twists around the vasculature.
Fluid and electrolyte abnormalities, common in patients with sigmoid volvulus, need to be corrected. For patients without perforation or peritonitis, rigid or flexible sigmoidoscopy with minimal air insufflation is performed to detort the volvulus and decompress the proximal colon. Flexible endoscopy allows for direct visual inspection of the colonic mucosa and in those with suspicion of necrosis detorsion should not be attempted and surgery should be pursued. Detorsion alone results in recurrence in the majority of cases. A rectal tube may be placed to decrease the chance of immediate re-volvulisation while the patient is optimised for colonic resection, preferably during the same hospitalisation. Evaluating patients in the United States Veterans Affairs Medical System, Grossmann et al. noted mortality of 24% for ES versus 6% for those able to be treated in an elective fashion following endoscopic decompression. A single-centre experience of 952 patients over a 46-year period noted that morbidity and mortality for ES was 35% and 16% versus 12.5% and 0% for elective. If the patient has not had a recent colonoscopy, and if feasible, a full mechanical bowel preparation and endoscopic colonic evaluation should be pursued prior to proceeding to the operating room. In addition to higher rates of recurrence, non-operative management has been shown to be associated with higher mortality rates in those without prohibitive operative risks.
![]() A single-centre experience of 952 patients over a 46-year period noted that morbidity and mortality for emergency surgery was 35% and 16% versus 12.5% and 0% for elective. The principal strategy in the treatment for sigmoid volvulus is early non-surgical detorsion followed by elective surgery in uncomplicated patients, while emergency surgical treatment is performed for patients with bowel gangrene, perforation, or peritonitis, other difficulties with diagnosis, unsuccessful non-surgical detorsion and early recurrence.
A single-centre experience of 952 patients over a 46-year period noted that morbidity and mortality for emergency surgery was 35% and 16% versus 12.5% and 0% for elective. The principal strategy in the treatment for sigmoid volvulus is early non-surgical detorsion followed by elective surgery in uncomplicated patients, while emergency surgical treatment is performed for patients with bowel gangrene, perforation, or peritonitis, other difficulties with diagnosis, unsuccessful non-surgical detorsion and early recurrence.
ES is pursued for perforation, peritonitis, ischaemia or the inability to endoscopically reduce the volvulus. Unless prohibited at the time of surgery (haemodynamic instability, intraperitoneal contamination, etc.), a segmental resection and primary anastomosis should be pursued. In those with uncomplicated sigmoid volvulus, morbidity and mortality have not been shown to differ between primary anastomosis and Hartmann’s procedure. Faecal decompression with on-table lavage carries the added risk of spillage and contamination and has not been noted to decrease anastomotic complications. If the volvulus is ischaemic, resection should be pursued without reducing the volvulus to decrease the chance of systemically disseminating toxins. Previously described fixation and plasty procedures (sigmoidopexy, mesenteric fixation, mesosigmoidoplasty, etc.) are associated with high rates of recurrence and should be avoided.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here