Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Clinical evaluation of possible mesenteric ischemia begins with an appropriate index of suspicion for the diagnosis followed by a careful history and physical examination. The major categories of mesenteric ischemia include chronic mesenteric ischemia (CMI), acute occlusive mesenteric ischemia (AMI), nonocclusive mesenteric ischemia (NOMI), and mesenteric venous thrombosis (MVT) (also see Chapter 25 ). Spontaneous mesenteric artery dissections (SMADs) are infrequent causes of abdominal pain that are becoming increasingly recognized due to the routine use of modern cross-sectional imaging. Although the various etiologies differ in their underlying pathologies and the clinical settings in which they occur, there may be significant overlap in their clinical presentation. The most crucial point is to understand the variety of clinical settings in which intestinal ischemia can occur and to include mesenteric ischemia in the differential diagnosis of patients presenting with abdominal pain. The goal is to achieve a rapid and efficient diagnosis prior to the onset of bowel infarction and resulting sepsis.
The most common cause of CMI is atherosclerosis, and CMI therefore predominantly affects an older patient population, but with a slight female predominance. Most patients with atherosclerotic mesenteric vascular disease are asymptomatic and at low risk for bowel infarction due to only mild to moderate stenosis of the mesenteric arteries and a robust collateral network that may compensate for reduced flow through one mesenteric artery. On the other hand, patients who have symptomatic CMI generally have high-grade stenosis or occlusion of both the celiac and superior mesenteric arteries (SMAs). Though uncommon, single-vessel mesenteric artery disease can result in symptomatic CMI in the presence of insufficient collateral reserve. The inferior mesenteric artery (IMA) may not be a source of collateral flow in most cases due to proximal atherosclerotic disease, but occasionally can be a vital collateral preventing ischemic catastrophe.
The classic presentation of CMI is postprandial pain described as colicky or a dull intense ache that generally begins 15 to 30 minutes after eating and lasts up to 4 hours. There are no signs of peritonitis, and the degree of pain may reflect the volume of the ingested meal. Diarrhea, nausea, and vomiting may be associated with the pain. Early in the course of CMI, some meals may be ingested without pain, so symptoms may be mistakenly attributed to other potential causes such as cholelithiasis, peptic ulcer disease, or malignancy. Patients often undergo extensive evaluation with endoscopy, computed tomography (CT), barium studies, and abdominal ultrasonography prior to reaching a diagnosis of CMI. As the disease progresses, patients experience pain with each meal and may develop a fear of food, termed sitophobia. Weight loss, one of the hallmarks of CMI, results from limited nutritional intake, not malabsorption. Patients with CMI generally have normal testing for gastrointestinal malabsorption.
CMI is a clinical diagnosis and patients should have suggestive clinical symptoms. Confirmatory studies to identify mesenteric artery lesions associated with CMI include duplex ultrasonography, computed tomography angiography (CTA), digital subtraction mesenteric angiography (DSA) and, less commonly, magnetic resonance angiography (MRA).
Duplex ultrasonography can serve as a valuable noninvasive screening test for mesenteric artery stenosis and for follow-up in patients with mesenteric artery reconstructions. Duplex ultrasound examination of the mesenteric arteries can be technically difficult and should be performed by vascular technologists with extensive experience in abdominal ultrasound techniques ( Fig. 26.1 ).
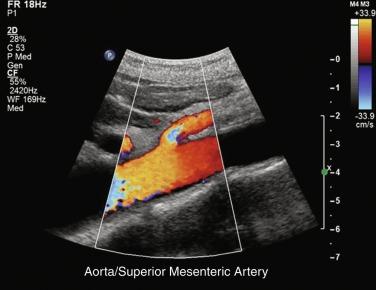
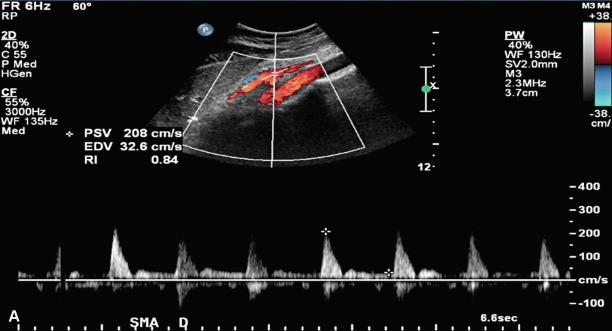
Duplex ultrasound can detect hemodynamically significant stenoses in splanchnic vessels. In 1986, investigators at the University of Washington found that flow velocities in stenotic SMAs and celiac arteries were increased when compared with normal SMAs and celiac arteries. Quantitative criteria for splanchnic artery stenosis were first developed and validated at Oregon Health and Science University. In a blinded prospective study of 100 patients who underwent mesenteric artery duplex scanning and lateral aortography, a peak systolic velocity (PSV) in the SMA of 275 cm/s or more indicated 70% or greater stenosis, and a PSV of 200 cm/s or higher in the celiac artery indicated a 70% or greater stenosis ( Fig. 26.2 ), both with high sensitivity and specificity ( Table 26.1 ). Criteria were developed to detect ≥ 70% stenosis, as symptomatic mesenteric ischemia is unusual with lesser degrees of mesenteric artery stenosis.
| > 70% stenosis | Sensitivity | Specificity | PPV | NPV | Accuracy | |
|---|---|---|---|---|---|---|
| SMA | PSV ≥ 275 cm/s | 92% | 96% | 80% | 99% | 96% |
| CA | PSV ≥ 200 cm/s | 87% | 80% | 63% | 94% | 82% |
Although the initial diagnosis of significant visceral artery stenosis can be made using duplex ultrasound, cross-sectional imaging, or in some cases, DSA, is crucial for preprocedure planning.
Multidetector CTA is a noninvasive modality with high sensitivity for detection of stenosis of the mesenteric arteries. In most cases, CTA has replaced other modalities as the imaging study of choice for evaluation of CMI because it can accurately identify significant stenosis in the celiac artery and SMA, identify significant visceral collaterals, and exclude other potential intraabdominal processes. There is not a set guideline to define significant stenosis on CTA. Some studies have used 50% luminal narrowing as a cutoff for significant stenosis, and others have delineated 70% luminal narrowing as significant stenosis in keeping with the Doppler criterion for significant stenosis.
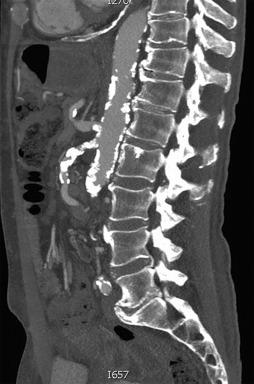
Together with noncontrast images, CTA, in many cases, offers enough anatomic detail to plan open and endovascular procedures. Preprocedure CTA is used to evaluate the angle of origin of the mesenteric vessels in relation to the aortic axis, the presence of calcium and thrombus as well as the length of the stenotic lesion, and the presence of important collaterals or unusual anatomy in proximity to the target lesion ( Fig. 26.3 ). Limitations include contrast-related nephropathy, hypersensitivity reaction, and ionizing radiation exposure.
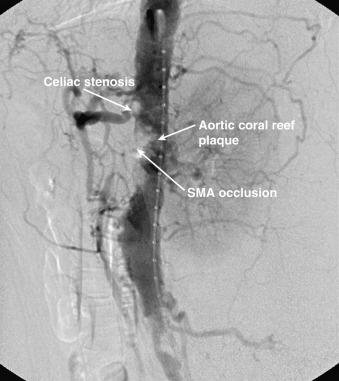
Mesenteric angiography has traditionally been the gold standard for diagnosis of hemodynamically significant mesenteric artery stenosis. Lateral and anteroposterior views of the aorta are required for full evaluation of the severity of visceral stenosis, occlusion of a mesenteric artery, and the extent of collateral development ( Fig. 26.4 ). With the improved image quality of CTA and MRA, mesenteric angiography is currently reserved primarily for treatment if the patient, based on axial imaging, is felt to be a candidate for endovascular intervention or is used in cases when noninvasive imaging is inconclusive.
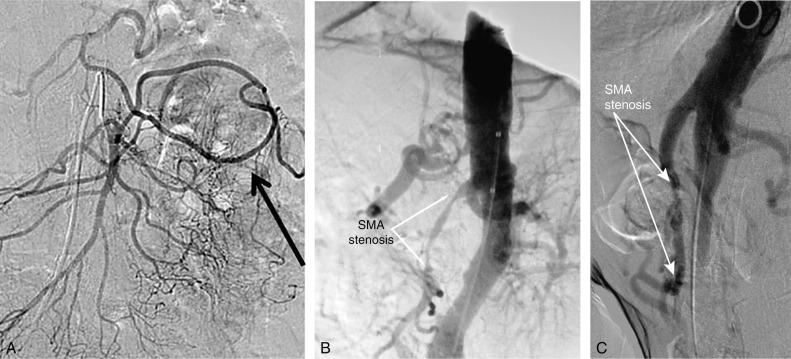
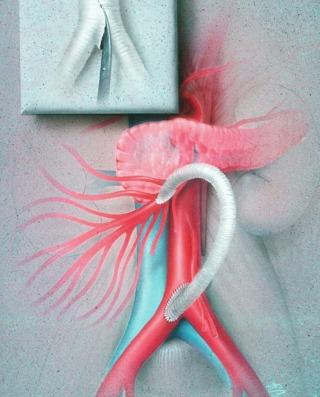
Traditionally, the preferred treatment of CMI was open surgical bypass. Operative repair, however, is associated with significant morbidity and mortality in most series. Given the expected morbidity in this patient population with significant weight loss, advanced age, malnutrition, and low albumin levels, all of which are predictors of increased morbidity and mortality after any major surgery, endovascular intervention has now surpassed open bypass as the most frequently utilized treatment for CMI. Endovascular treatment has high technical and early clinical success rates, with decreased morbidity and mortality compared to surgical intervention. It is, however, associated with lower long-term patency and a greater likelihood of the need for repeat interventions. There are no randomized controlled trials comparing treatment modalities for CMI. Therefore, treatment decisions must be based on large case series in which a variety of procedures have been used. Although endovascular intervention is the first-line treatment for most patients with CMI, certain patients remain more appropriate for surgery, including those with concomitant aneurysmal disease in need of repair, and those with flush occlusions of the culprit mesenteric vessel(s) ( Fig. 26.5 ).
Revascularization is indicated for symptomatic intestinal ischemia. Revascularization for asymptomatic high-grade SMA obstruction is recommended only in patients undergoing otherwise indicated aortic surgery for aneurysmal or occlusive disease. In this group of patients, acute intestinal ischemia following aortic surgery has been well documented, and SMA reconstruction prior to or at the time of aortic repair seems prudent.
The distal infrarenal aorta as an origin for an SMA bypass graft has advantages and disadvantages. The exposure is familiar, and risks of dissection and clamping are less than with more proximal aortic exposures. In addition, the procedure can be readily combined with other intraabdominal vascular procedures. The primary disadvantage is that the infrarenal aorta and iliac arteries are frequently calcified, increasing the technical difficulty of the proximal anastomosis.
Prosthetic grafts are used most often in cases of mesenteric revascularization. Exceptions are cases complicated by bowel necrosis. For these patients, vein grafts are preferred to minimize the possibility of graft infection. Special attention must be paid to graft configuration to avoid graft kinking when the graft is placed in a retrograde configuration. A preference for the origin of the graft is the area of the junction of the aorta and right common iliac artery (CIA), although any suitable site on the infrarenal aorta or either CIA is satisfactory. A single limb is cut from a bifurcated graft in the manner described by Wylie and colleagues, which provides a “flange” for sewing and prevents anastomotic stricture ( Fig. 26.6 ). The ligament of Treitz is dissected. The proximal (inflow) anastomosis is completed first. The graft is then arranged first cephalad, then turning anteriorly and inferiorly a full 180 degrees to terminate in an antegrade anastomosis to the anterior wall of the SMA, just beyond the inferior border of the pancreas. The graft is excluded from the peritoneal cavity by closing the mesenteric peritoneum, approximating the ligament of Treitz, and closing the posterior parietal peritoneum.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here