Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Many types of autoantibodies to intracellular antigens are present in the various systemic autoimmune rheumatic diseases. Currently it is considered important not only to detect the presence and quantity of the intracellular autoantibody in the patient but also to identify its antigenic specificity.
Past studies have shown that distinct diagnostic autoantibody profiles are observed in many of the systemic autoimmune rheumatic diseases. Some of the diseases are characterized by the presence or absence of a specific antibody or by differences in the quantitative level or titer of the autoantibody.
Much progress has been made in improvement of the sensitivity, specificity, and quality control of the many laboratory assays used for the detection of autoantibodies to intracellular antigens.
Indirect immunofluorescence microscopy using human cells, like HEp-2 cells, allows the sensitive detection of serum autoantibodies that react very specifically with various cellular proteins and nucleic acids. Antinuclear antibody (ANA) automation is now increasingly employed in immunology laboratories, although identifying only a limited number of patterns.
Widely used tests for screening of intracellular autoantibodies are immunofluorescence microscopy, immunoenzyme, and multianalyte solid phase immunoassays such as LIA and ALBIA.
Many systemic autoimmune rheumatic diseases (SARDs) are characterized by the presence of one or more autoantibodies that may be directed against components of the surface, cytoplasm, nuclear envelope, nucleus, or cell cycle–specific components of the cell. Such autoantibodies, collectively known as anticellular antibodies (previously antinuclear antibodies [ANA]) ( , 2016), are a hallmark of SARDs ( ; ; ). Many of the SARDs have a distinctive autoantibody profile with diagnostic and prognostic implications. Moreover, the biochemical and biological functions of many of the target antigens involved in deoxyribonucleic acid (DNA) replication, cell division, splicing of messenger ribonucleic acid (mRNA) precursors, RNA processing, protein quality control, and other fine biologic processes were elucidated by using sera from SARD patients ( ; ). Classification of the various SARDs has been difficult because of a lack of firm etiologic basis for most of the diseases ( ) but recently are being reclassified based on autoantibodies and other molecular markers ( ; ; ; ). Our abridged classification of SARDs and related disorders is shown in Box 53.1 .
Systemic lupus erythematosus (SLE)
Drug-induced lupus erythematosus
Sjögren syndrome (primary and secondary)
Systemic sclerosis (scleroderma)
Diffuse
Limited/CREST syndrome
Sine scleroderma
Rheumatoid arthritis (RA)
Juvenile inflammatory arthritis
Still disease (juvenile and adult)
Idiopathic inflammatory myopathies
Dermatomyositis (adult and juvenile; amyopathic form)
Antisynthetase syndrome
Necrotizing myopathies
Inclusion body myositis
Overlap syndromes
Mixed connective tissue disease (MCTD)
RA and SLE (lupus)
SLE and scleroderma (lupoderma)
Scleroderma and dermatomyositis (sclerodermatomyositis)
Systemic vasculitis
Takayasu arteritis
Giant cell arteritis and polymyalgia rheumatica
Granulomatosis with polyangiitis (Wegener granulomatosis)
Eosinophilic granulomatosis with polyangiitis (Churg-Strauss syndrome)
Polyarteritis nodosa
Other syndromes
Undifferentiated connective tissue disease
Behçet disease
Autoimmune syndrome induced by adjuvants (ASIA) a
a Data from ; ; .
CREST, Calcinosis cutis, Raynaud phenomenon, esophageal dysmotility, sclerodactyly, telangiectasias.
Systemic lupus erythematosus (SLE) is the prototype SARD and has the following significant features ( ; ; ; ):
It is a nonorgan-specific autoimmune disease in which the tissue injury is mediated primarily by immune complexes and direct cell-mediated cytotoxicity.
It is a multisystem disease that affects persons of all ages and both sexes, although it is most prevalent in women during childbearing years.
The disease demonstrates a hyperactive immune system with multiple abnormalities, displaying genetic associations with aberrant immunoregulatory signaling pathways, such as the interferon pathway and toll-like receptors ( ).
The typical patient has an average of three different circulating autoantibodies present simultaneously. The prevalence of autoantibodies varies over a wide range, and more than 180 different autoantibody specificities have been identified to date ( ).
In 1971 the American College of Rheumatology (ACR; previously the American Rheumatism Association [ARA]), published preliminary criteria for the classification of systemic lupus erythematosus (SLE) ( ; ). Patients were considered to have SLE if four of the criteria were met sequentially or simultaneously during any interval of observation. In 1982 the Subcommittee for SLE Criteria of the ARA published revised criteria that incorporated new immunologic knowledge and improved the disease classification of SLE ( ). The 1982 revised criteria for classification of SLE included 11 categories, adding (1) abnormal titer of antinuclear antibody by immunofluorescence or an equivalent assay, and (2) antibody to native DNA and/or Sm antigen. In contrast to the 1971 criteria, the 1982 criteria removed Raynaud phenomenon and alopecia because of their lack of sensitivity and specificity.
When the 1982 ARA Criteria for Classification of SLE were compared with the 1971 criteria, there was definite improvement in sensitivity and specificity. The 1982 criteria showed 83% sensitivity and 93% specificity, when evaluated with known SLE and control patients ( ). In 1997, an update of the criteria was suggested, in which the LE cell test was deleted and anticardiolipin antibodies were added, changes emphasized in the “Guidelines for Referral and Management of SLE in Adults” by the ACR Ad Hoc Committee on SLE . The Systemic Lupus International Collaborating Clinics (SLICC) group validated and updated the ACR 1982 SLE criteria ( ; ; ), with resultant improved sensitivity (97% by the updated 2012 SLICC criteria vs. 83% by the 1982 ACR criteria) but lower specificity (84% by SLICC vs. 96% by ACR). The proposal includes at least one clinical and one immunologic criterion satisfied, or biopsy-proven lupus nephritis together with positive ANA or anti-DNA autoantibodies ( Box 53.2 ). More recently, the European League Against Rheumatism (EULAR) and the American College of Rheumatology proposed a new set of SLE criteria where the ANA IFA, or its equivalent, is a mandatory inclusion requirement for SLE classification ( ). The new criteria had a sensitivity of 96.1% and specificity of 93.4% in a validation cohort of 1001 patients, representing improvement in relation to the ACR 1982/1997 and SLICC 2012 criteria.
a The proposed classification is based on 11 criteria. The Systemic Lupus International Collaborating Clinics (SLICC) classification rule is as follows: Classify a patient as having SLE if he or she satisfies four of the clinical and immunologic criteria used in the SLICC classification criteria, including at least one clinical criterion and one immunologic criterion, OR if he or she has biopsy-proven nephritis compatible with SLE in the presence of ANAs or anti-dsDNA antibodies ( ; ; ). ANA positivity is a mandatory criteria in the recent EULAR/ACR classification criteria ( ).
Acute cutaneous disease
Includes malar rash, photosensitivity, and subacute cutaneous lupus
Chronic cutaneous disease
Includes discoid rash, panniculitis
Oral or nasal ulcers
Nonscarring alopecia (in the absence of other causes)
Synovitis involving two or more joints
Serositis (pleuritis or pericarditis with rub)
Renal disorder (persistent proteinuria, cellular casts)
Neurologic disorder (seizures, psychosis, peripheral/cranial neuropathy)
Hemolytic anemia
Leukopenia <4000/mm 3 OR lymphopenia <1000/mm 3 at least once
Thrombocytopenia <100,000/mm 3 in the absence of offending drug
ANA+ (anticellular antibodies in HEp-2 cells)
Anti-DNA: antibody to native DNA in abnormal titer
Anti-Sm: presence of antibody to Sm nuclear antigen
Antiphospholipid antibodies (anticardiolipin or anti-β 2 GP I antibodies IgA, IgG, or IgM; or a positive test result for lupus anticoagulant, or a false-positive test for syphilis
Low serum complement levels (C3, C4, or CH50)
Direct Coombs+ in the absence of hemolytic anemia
There are many diseases and syndromes that share certain clinical features with SLE but have different etiologies and pathogeneses. Diseases in this category include vasculitis, cryoglobulinemia, relapsing polychondritis, lymphoproliferative diseases, rheumatic fever, glomerulonephritis, syphilis, lupoid hepatitis, drug-induced lupus, and occult malignancy ( ; ; ). There is a very broad category of patients who demonstrate fewer than four of the 1982 ARA/ACR classification criteria for SLE ( ; ; ; ) but are considered to have “lupus-like” illnesses. These patients have been classified as follows: (1) undifferentiated rheumatic disease; (2) nonrheumatic disease; (3) overlap syndrome; and (4) incomplete, latent, or incipient lupus. If these patients are followed, some are stable with mild symptoms—and up to 75% never fulfill a diagnostic criterion for specific systemic rheumatic diseases ( ) and remain labeled as “undifferentiated connective tissue disease” (UCTD) ( ). A few eventually developed definite SLE, while others evolved into other SARD.
The presence of a broad spectrum of autoantibodies is characteristic of SLE, such as antibodies to dsDNA, chromatin or nucleosomes, Sm antigen, U1nRNP, SS-A/Ro60, SS-B/La, C1q, ribosomal RNP, phospholipids and related proteins, and several other nonhistone protein or nonhistone protein-RNA complexes ( ; ; ; ).
Anti-dsDNA, anti-Sm, anti-C1q, and antiribosomal P are generally regarded as specific for SLE, but the prevalence of these autoantibodies varies widely depending on demographic variations, cohort composition (inception vs. cross sectional), and diagnostic assays used to detect the autoantibodies. Autoantibodies to dsDNA and chromatin are detectable in up to 90% of individuals with active disease ( ), and antibodies to C1q tend to segregate with SLE patients having glomerulonephritis and active disease ( ; ) ( Table 53.1 ).
| Antigen | Molecular Structure | Prevalence |
|---|---|---|
| Native DNA | Double-strand DNA | 55–90% |
| Denatured DNA | Single-strand DNA | 50–90% |
| Histones | H1, H2A, H2B, H3, H4 | 50–70% |
| Nucleosome (chromatin) | DNA-histones complex | 50–90% |
| Sm | Small ribonucleoproteins 29 (B’), 28 (B), 16 (D), and 13(E) kDa, complexed with U1, U2, and U4-U6 snRNAs; spliceosome component | 15–30% |
| Nuclear RNP (U1nRNP) | Proteins 70, 33(A), and 22 (C) kDa, complexed with U1 snRNA; spliceosome component | 5–40% |
| SS-A/Ro | Proteins 60 and 52 kDa, complexed with Y1-Y5 RNAs | 24–60% |
| SS-B/La | Phosphoproteins 48 kDa, complexed with nascent Y1 RNA Pol transcripts | 9–35% |
| Ku | Proteins 86 and 66 kDa, DNA-binding proteins | 1–39% |
| hnRNP protein A1 | Nuclear protein 34 kDa | 31–37% |
| PCNA | Protein 36 kDa; auxiliary protein of DNA polymerase | 3% |
| Ribosomal RNP | Phosphoproteins 38, 16, and 15 kDa associated with ribosomes | 10–20% |
| Hsp-90 | Heat-shock protein 90 kDa | 5–50% |
| Golgi complex | Golgins, giantin | ∼1% |
| HMG-17 | Chromatin-associated proteins, 9 to 17 kDa | 34–70% |
| β 2 glycoprotein I | Binds to anionic phospholipids, cardiolipin | 25% |
Anti-dsDNA antibodies are rather specific for SLE and are observed at a frequency of 75% to 90% in SLE patients with active disease ( ; ). Although there have been many reports of antibodies to dsDNA in diseases other than SLE, it is possible that the reactive antibodies to DNA in the other diseases were actually anti–single-stranded (ss)DNA antibodies because the dsDNA antibody tests often use dsDNA preparations contaminated with denatured or ssDNA. Antibodies to dsDNA appear to play a role in the pathogenesis of SLE. In studies of SLE patients, antibody to dsDNA is followed by the appearance of circulating DNA antigen, a sequence of events that results in the formation of immune complexes. Such DNA–anti-DNA immune complexes, mostly containing complement-activating IgG3, have a tropism for basement membranes and are deposited in the kidney glomeruli. This initiates kidney damage through inflammatory pathways that culminate with complement activation and cell lysis ( ; ; ).
Earlier methods for detection of dsDNA antibodies included insensitive precipitation methods, complement fixation, and passive hemagglutination. Current methods used are indirect immunofluorescence (IFA) on Crithidia luciliae substrates, enzyme-linked immunosorbent assay (ELISA), and addressable laser bead immunoassays ( ). These can detect anti-dsDNA in 45% to 90% of active untreated SLE patients. Transient increases in anti-DNA antibodies were described in RA patients treated with anti-TNF therapy ( ; ; ). Occasional clinical lupus-like cases may occur in this situation ( ; ).
Precipitating antibodies to the Sm antigen are considered highly specific markers for SLE ( ; ; ). Antibodies to both Sm and nuclear ribonucleoproteins (nRNP) are found in SLE patients. used molecular biology tools to show that Sm and nRNP antigens were macromolecular particles comprised of small nuclear RNAs complexed with a family of well-defined small ribonuclear proteins (snRNP). The particle bound by anti-nRNP is composed of an RNA component designated U1 (U for uridine-rich), complexed to at least seven proteins varying in molecular mass from 12 to 68 kD ( ), found at a frequency of 5% to 40% in active SLE ( ).
The Sm antigens consist of several proteins, notated as B/B’ (27/28 kD), D1/D2/D3 (14 kD), and E (12 kD) ( ), which are components of the Sm core proteins organized as a seven-member ring structure. Purified anti-B/B’ antibodies cross-react with the D protein and vice versa. Thus there are at least two epitopes on the B/B’ protein recognized by anti-Sm sera. No unique clinical features are apparent in SLE patients with anti-Sm antibodies ( ), although some authors reported that these autoantibodies are associated with renal disease or disorders of the central nervous system ( ; ; ; ). Patients who have antibodies to only U1nRNP have a low frequency of antibodies to dsDNA and a low frequency of clinically apparent renal disease ( ). Anti-Sm and anti-U1nRNP antibodies were historically detected by immunodiffusion, passive hemagglutination, or counterimmunoelectrophoresis. However, the abovementioned methods do not accurately distinguish between antibodies directed against different snRNP. The reactivities with individual RNAs and polypeptides can be best demonstrated by RNA immunoprecipitation (IP) and immunoblotting techniques, respectively. Currently, the most widely used laboratory tests for the detection of anti-Sm and anti-nRNP antibodies are ELISA, dot blot or line immunoassays, and addressable laser bead immunoassays. These tests can differentiate between Sm and nRNP antibodies but do not define the specific antibody epitopes present in patients’ sera.
SLE patients can have antibodies to SS-A/Ro60 alone or they may have both anti-SS-A/Ro60 and anti-SS-B/La. Having anti-SS-A/Ro60 alone was strongly associated with human leukocyte antigen (HLA) DR2 and with being young (<22 years of age at onset). The presence of both anti-SS-A/Ro60 and anti-SS-B/La in SLE was associated with HLA-DR3 and seen in older patients (>50 years of age at disease onset) ( ). A study of 55 patients with SLE showed that patients with anti-SS-A/Ro60 alone had much more serious renal disease ( ). SLE patients with only anti-SS-A/Ro60 also had a higher incidence of concomitant anti-DNA antibodies than those SLE patients with both the anti-SS-A/Ro60 and anti- SS-B/La antibodies ( ). Anti-SS-A/Ro60 autoantibodies have been closely associated with the appearance of nephritis, vasculitis, lymphadenopathy, photosensitivity, and leukopenia in SLE patients. Anti-SS-B/La antibodies, like anti-SS-A/Ro60 antibodies, are antibodies noteworthy for their strong association with Sjögren syndrome, occurring in more than two-thirds of patients with this disorder. The SS-B/La antigen is a cellular protein bound to small cytoplasmic RNA species, forming a small RNP macromolecular complex that may function in processing of RNA polymerase III transcripts ( ).
Attention has been drawn to the differentiation between antibodies to SSA/Ro60 and anti-Ro52/TRIM21 ( ). There are different clinical associations for each system, while both anti-SSA/Ro60 and anti-Ro52/TRIM21 are associated with primary Sjögren syndrome and SLE, and anti-Ro52/TRIM21 alone is also associated with scleroderma ( ; ) and myositis ( ; ). Masked reactivities in up to 20% of tested sera were reported when both antigens were present in commercial ELISAs, pointing to the fact that each autoantibody should be tested separately.
Elevated levels of anti-SS-A/Ro60 autoantibodies are related to several clinical autoimmune disorders, including (1) subacute cutaneous lupus erythematosus; (2) neonatal lupus syndrome with congenital heart block and/or cutaneous lesions; (3) homozygous C2 and C4 deficiency with SLE-like disease; (4) primary Sjögren syndrome vasculitis, rheumatoid factor positivity, and severe systemic symptoms; (5) ANA-negative SLE patients; (6) SLE with interstitial pneumonitis; and (7) serologic features of antiphospholipid syndrome ( ; ).
The Ku-antigen system consists of a pair of proteins called p70/p80 with DNA helicase activity ( ; ; ). These proteins have a high affinity for DNA and are known to be DNA-binding proteins, interacting covalently with the blunt ends of native DNA or DNA damaged by ionizing radiation ( ). Anti-Ku antibodies were reported in systemic sclerosis and myositis overlap with interstitial lung disease. A multinational cohort of 2140 scleroderma patients found isolated anti-Ku antibodies by line immunoassay in only 1.1% of individuals, with higher frequency of interstitial lung disease ( ). Mahler et al. found anti-Ku antibodies by chemiluminescence using full-length recombinant human Ku in 10% of SLE, 4% of systemic sclerosis, and 4% of autoimmune myositis patients ( ). Overlap syndromes were common in those anti-Ku positive individuals: myositis and SLE, scleroderma and SLE, or even mixed connective tissue disease.
The discovery of autoantibodies to ribosomal proteins (anti-Rib-P) dates back more than 50 years when antibodies to ribosomes were identified in SLE sera ( ). Since then, anti-Rib-P antibodies have been the subject of extensive study and became known as a highly specific biomarker for the diagnosis of SLE. Some studies reported an association with neuropsychiatric SLE (NPSLE) ( ; ). The primary targets of anti-Rib-P autoantibodies are three ribosomal phosphoproteins of 38, 19, and 17 kDa (P0, P1, P2) ( ). Evidence from in vitro studies and murine models suggests that anti-Rib-P may have a pathogenic role in lupus nephritis and NPSLE. Despite a wealth of evidence, in comparison to other SLE autoantibodies such as anti-Sm and anti-dsDNA, anti-Rib-P has not been included in classification criteria for SLE. A significant challenge is the variability of assays used to detect anti-Rib-P, including the antigens and diagnostic platforms employed ( ). This may account for the marked discrepancies in frequencies (10–47%) and association with clinical and demographic features reported in SLE cohorts. Although a characteristic pattern by immunofluorescence on HEp-2 cells has been attributed to anti-Rip-P antibodies ( www.anapatterns.org ), it is not advisable to rely solely on that finding ( ). Some sera are ANA negative so that novel solid phase commercially available assays, mostly depending on the C22 peptide, are being employed ( ).
Antibodies to proliferating cell nuclear antigen (PCNA) proteins are detected in up to 3% of patients with active SLE but show no distinctive clinical associations ( ; ; ). The PCNA complex has been characterized as cell-cycle related, and PCNA antibodies have been useful probes in the study of DNA replication, cell proliferation, and blast transformation. There are questions about the specificity of anti-PCNA autoantibodies for SLE ( ).
The antiphospholipid antibody syndrome is characterized by the presence of circulating antibodies to phospholipids and phospholipid-related proteins (anticardiolipin IgG/IgM, anti-β 2 glycoprotein I IgG/IgM, and lupus anticoagulant) and clinical features of arterial and venous thrombosis, thrombocytopenia, hemolytic anemia, miscarriages, central nervous system disease, and several other systemic symptoms ( ). Antiphospholipid antibodies are found in up to 60% of patients with SLE but also in other disorders such as infectious diseases ( ; ). This is a very heterogeneous family of antibodies, functionally and immunochemically ( ). Cardiolipin (anionic phospholipid) has been widely used for the detection of antiphospholipid antibodies. Antibodies to anionic phospholipids may be IgG or IgM, whereas antibodies to zwitterionic phospholipids are more frequently IgM.
Antibodies to phospholipids are identified in SLE patients in three ways ( ; ): (1) serologically false-positive test for syphilis by a positive Venereal Disease Research Laboratory (VDRL) test; (2) the lupus anticoagulant assay, which is a prolongation of the kaolin partial thromboplastin time (KPTT), which is not corrected by normal plasma; and (3) cardiolipin immunoassay with the use of cardiolipin and/or β 2 glycoprotein I and domain I of β 2 glycoprotein I.
Harris and colleagues ( ; ; , ) convened international workshops to improve the precision and accuracy of antiphospholipid antibody immunoassays. Reference sera were prepared and standard units (GPL and MPL) were defined. One GPL (MPL) unit is defined as the cardiolipin-binding activity of 1 ug/mL of an affinity-purified standard IgG (IgM) sample ( ). However, suggested that an IgA-specific assay for antiphospholipid antibody is important in assessing the antiphospholipid syndrome in patients with SLE ( ; ; ; ). A higher prevalence of the IgA isotype was reported as a feature in black patients.
Newly developed assays for domain I of β 2 glycoprotein I autoantibodies increase the specificity of these assays, since it was described that the β 2 glycoprotein is a specific target autoantigen in antiphospholipid antibody syndrome ( ). Furthermore, IgA antibodies targeting β 2 glycoprotein I, or antibodies recognizing a complex of β 2 GP I/oxLDL (oxidized low density lipoprotein), seem to be pathogenic in atherosclerotic vascular disease ( ; ; ). Domain 4 of the molecule seems to be the target for proatherosclerotic anti-β 2 GP I autoantibodies, and domain 1 was suggested to be involved in the antiphospholipid syndrome as demonstrated in deletion mutants studies ( ; ; ). Additional antiphospholipids have been identified in recent years, in particular antiprothrombin and antiphosphatidylserine/prothrombin complex antibodies, both associated with increased risk of thrombosis ( ; ).
A comparatively benign form of lupus may present as “discoid” (i.e., coin or disc shaped) cutaneous lesions without symptoms or findings of systemic disease. This disorder is called chronic discoid lupus erythematosus (CDLE) ( ). The skin lesions of CDLE are as follows: (1) persistent localized erythema, (2) adherent scales, (3) follicular plugging, (4) telangiectasis, and (5) atrophy ( ). Subacute cutaneous lupus erythematosus (SCLE), another skin condition, consists of papulosquamous or nonscarring lesions with a major association to SS-A/Ro60 and Ro52 autoantibodies ( ). Also, additional variants of CDLE, such as lupus panniculitis (also called lupus profundus) and urticarial lupus, have been described ( ; ).
Unfortunately CDLE is a cutaneous autoimmune disorder that lacks any unifying diagnostic serologic biomarker. It is a mild form of lupus erythematosus that uncommonly progresses to SLE. Otherwise there is considerable overlap between CDLE and SLE, since up to 15% of patients with SLE have cutaneous discoid lesions. About 6% to 12% of patients with SLE had discoid lupus for a varying number of years before the onset of systemic disease ( ). Antinuclear antibodies are commonly found, with estimates of prevalence in CDLE from 6% to 50%. The sex ratio of discoid lupus (two females/one male) is much less biased toward females than the systemic form. Figure 53.1 depicts some clinical aspects of cutaneous disease in SLE.
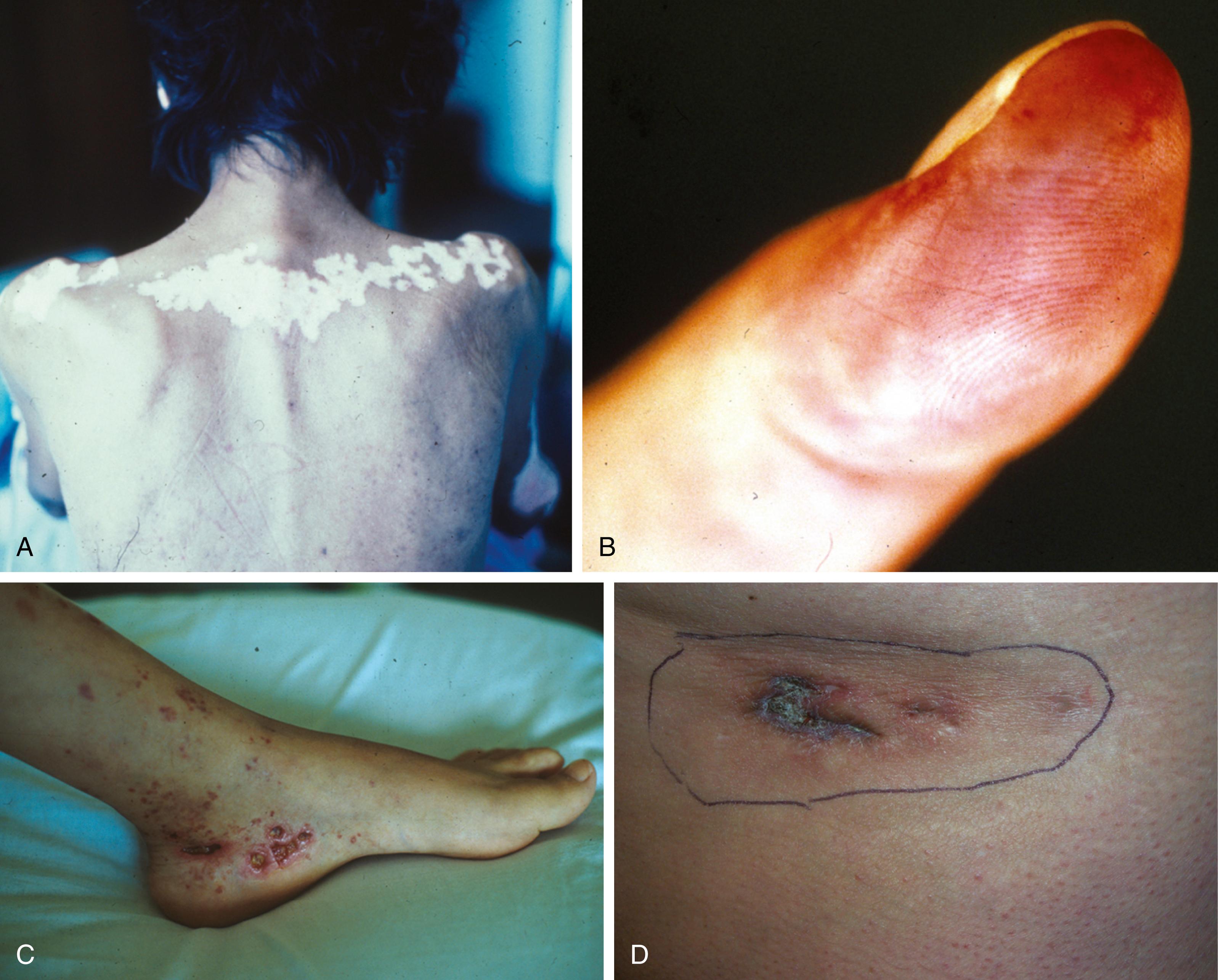
Characteristic of drug-induced lupus associated with procainamide, hydralazine, quinidine, isoniazid, and a long list of other drugs and xenobiotics is the presence of histone and/or chromatin autoantibodies ( ; ). Histones are cationic proteins containing high molar ratios of positively charged amino acids (lysine and arginine), and they are bound to nuclear DNA in eukaryotic cells. The subunit of the histone-DNA complex is called a nucleosome, which has two molecules of each of the “core” histones (H2A, H2B,H3, and H4) and one molecule of H1, along with DNA of about 200 base pairs in length ( ).
In the studies of some drug-induced lupus, the liver enzyme acetyltransferase appears to play a major function ( ). Acetyltransferase is an enzyme that acetylates some drugs such as hydralazine and procainamide, playing an important role in detoxification and excretion of the drug. Patients with low levels of acetyltranferase were more prone to develop ANA and clinical symptoms than patients who were treated with hydralazine and had phenotypically high levels of acetyltransferase. Patients with high levels of enzyme and who were rapid acetylators were not protected from the development of ANA. These patients, however, took a longer and larger cumulative dose of hydralazine before developing disease. These findings concerning acetyltransferase phenotypes have been confirmed in patients treated with procainamide ( ). Fifty percent of all patients treated with procainamide developed ANAs after 1 year of treatment ( ). The slow acetylators developed ANAs more rapidly than the rapid acetylators ( ). All patients on prolonged procainamide treatment developed a positive ANA response irrespective of acetylator phenotype. Some of the drugs (procainamide, hydralazine, quinidine) are no longer widely used and hence the drug-induced lupus syndrome related to these medications has become quite rare.
Drug-induced lupus is typically associated with antibodies to ssDNA, chromatin, and histones, but in SLE, by comparison, in addition to antibodies to histone and chromatin, antibodies to dsDNA, Sm, U1nRNP, and SSA/Ro60 antigens are commonly present. Procainamide was used to treat patients with cardiac arrhythmias, and most patients eventually developed antihistone antibodies, but only 10% to 20% of procainamide-treated patients developed symptomatic autoimmune disease. Patients with symptomatic disease developed a unique type of IgG antihistone antibody, which, rather than reacting with individual histones, shows specific reactivity with the histone H2A–H2B dimer complex. Thus the IgG anti–(H2A-H2B) is a useful diagnostic marker, with high sensitivity and specificity for symptomatic disease, in contrast to the benign form of procainamide-induced autoimmunity, with IgM antibodies to the individual histone.
Since antihistone antibodies are found in many other conditions, more specific antibodies like antinucleosome (sometimes called antichromatin) autoantibodies have largely replaced assays for antihistones. Synonyms for antinucleosome antibodies are antichromatin, antideoxyribonucleoprotein (DNP), and anti-(H2A-H2B-DNA). Antinucleosome antibodies are found in up to 75% of all patients with SLE, in up to 100% of drug-induced lupus patients, and in 20% to 50% of patients with autoimmune hepatitis type I (lupoid hepatitis). In SLE, antinucleosome antibodies correlate better with kidney disease than anti-dsDNA ( ; ; ; ; ; ; ; ; ; ; ), although emphasis for strong association with active lupus nephritis is being pushed toward the presence of antibodies to the first component of the classical complement cascade: anti-C1q autoantibodies ( ). Nevertheless, these autoantibodies have a relatively high specificity for SLE and a clear association of antinucleosome antibodies with lupus disease activity and renal disease in many publications ( ; ; ).
An autoantibody that seems to be protective against SARDs is represented by the dense fine speckled (DFS) nuclear pattern, which can be initially detected by indirect immunofluorescence on HEp-2 cells ( , ; ; ; ) and then confirmed by a solid phase assay based on the DFS70/LEDGF (lens epithelium-derived growth factor) protein ( ). In fact, the presence of high titer anti-DFS70 antibodies without other SARD-related autoantibodies is considered useful to aid in the exclusion of ANA-associated rheumatic diseases ( ), and this has also been demonstrated recently by a report confirming that mono-specific anti-DFS70 antibodies might be helpful to discriminate individuals with and without ANA-associated rheumatic diseases ( ).
Sjögren syndrome (SjS) is a chronic inflammatory exocrinopathy, an autoimmune disease marked by dryness of the eyes, mouth, and other mucous membranes ( ). The disease may evolve from exocrine gland involvement to a systemic disorder, as well as to B-cell lymphoproliferative disease. It is much more frequently found in women than men, with an increasing prevalence throughout adult life. Often associated with SjS are other autoimmune diseases, such as RA, SLE, primary biliary cholangitis, and systemic sclerosis ( Box 53.3 ). The affected salivary or lacrimal glands are infiltrated with aggregates of lymphocytes. Extraglandular manifestations include lymphadenopathy, cutaneous vasculitis, interstitial pneumonitis, and peripheral neuropathy.
Rheumatoid arthritis
Systemic lupus erythematosus
Polydermatomyositis
Mixed connective tissue disease
Primary biliary cholangitis
Necrotizing systemic vasculitis
Autoimmune thyroiditis
Chronic active hepatitis
Mixed cryoglobulinemia
Hypergammaglobulinemic purpura
Systemic sclerosis
Ataxia and other nervous system diseases
Renal tubular acidosis
The disease has been classified as (1) primary SjS, which is not associated with another autoimmune disease, with isolated sicca complex (keratoconjunctivitis sicca and xerostomia) as its hallmark feature; and (2) secondary SjS, in which SARDs or other autoimmune disorders are present. Autoantibodies in SjS are primarily directed to SS-A/Ro and SS-B/La antigens ( ), but patients with other autoantibody specificities such as anti-Golgi complex, GW/processing body, or anti-NuMA autoantibodies have been reported ( ). The anti-SS-A/Ro and anti-SS-B/La are present in SLE but in lower prevalences than in SjS. Clinical and laboratory characteristics of the disease are seen in Fig. 53.2 .
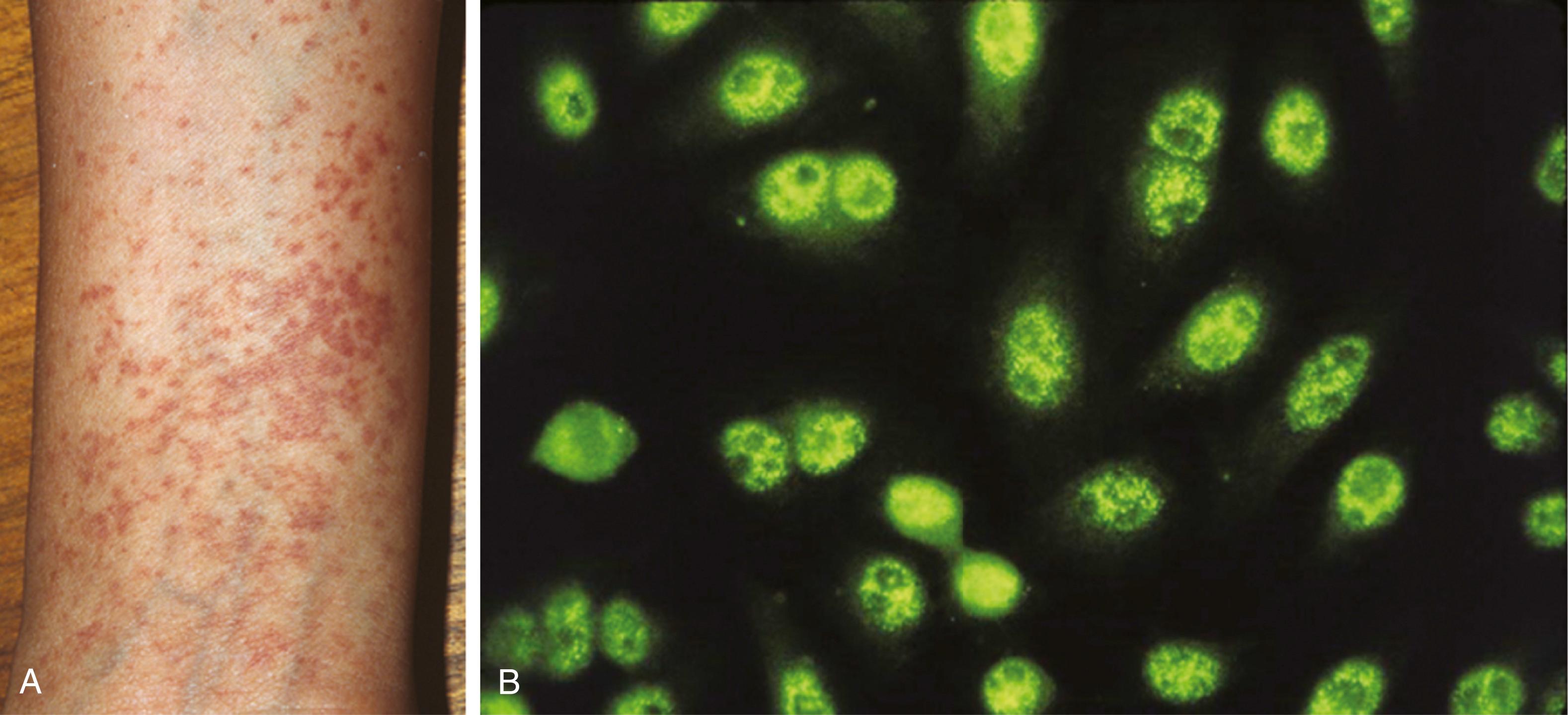
Anti-SS-A/Ro and anti-SS-B/La are the most clinically important and best characterized autoantibodies in SjS ( ). The SS-A/Ro autoantigen was first identified as a specific precipitin in the Ouchterlony double-immunodiffusion assay and later shown to be the 60kDa protein (Ro60) existing as ribonucleoprotein complexes with four small hY (human cytoplasmic) RNA molecules ( ; ; ). In ANA test performed in clinical laboratories, anti-SS-B/La and anti-SS-A/Ro are often reported as showing nuclear fine speckled pattern. The cellular function for Y RNAs remains unknown but the Ro60 protein is currently postulated to play roles in small RNA regulation and the enhancement of cell survival following exposure to ultraviolet irradiation. Anti-SS-B/La antibodies were first defined by immunodiffusion in association with SS-A/Ro precipitins. SS-B/La is a distinct 48 kDa protein and associates with a variety of small RNAs derived from RNA polymerase III, including Ro hY RNAs, pre-5S RNA, pre-tRNAs, as well as many viral RNAs ( ). The SS-B/La protein has been described to have multiple functions, including as a transcription termination factor for RNA polymerase and a factor in mediating the correct ribosome translational start sites ( ). Anti-SS-B/La antibodies are almost invariably accompanied by anti-SS-A/Ro antibodies, reflecting the physical association of these molecules in SS-A/Ro and SS-B/La ribonucleoprotein particle complex, but anti-SS-A/Ro antibodies frequently occur in the absence of anti-SS-B/La antibodies.
Many anti-Ro60 positive sera also react with a structurally unrelated 52kDa protein (Ro52, also known as TRIM21) ( ; ; ). There has been report of association of Ro60 and Ro52 via direct protein-protein interaction; however, the interaction may be weak or transient and was not observed by other investigators. Ro52/TRIM21 is an E3 ubiquitin ligase. Thus it is possible that Ro60 and/or SS-B/La are substrates for Ro52-mediated ubiquitinylation and somehow they are stabilized during certain disease states and the complexes contribute to the formation of autoantibodies to these components.
The reported prevalence of anti-SS-A/Ro60 and SS-B/La antibodies depends on the method and sensitivity of detection. Overall, anti-SS-A/Ro60 antibodies occur in approximately 60% to 75% of patients with primary SjS and also observed in secondary SS ( Table 53.2 ). Anti-SS-B/La antibodies have been reported to occur in up to 50% of primary SS patients. reported that among 321 SjS patients, anti-SS-B/La was strongly correlated to organ involvement and cytopenias and thus could serve as a prognostic marker in primary SjS.
| Autoantibodies | Prevalence in pSjS (%) | |
|---|---|---|
| Anti-SS-A/Ro | 60–75 | |
| Anti-SS-B/La | 25–50 | |
| Anti-M3 muscarinic receptor | 63 | |
| Anti-α-fodrin | 40–70 | |
| Rheumatoid factor | 70 | |
| NA14 | 11–14 | |
By comparison, anti-Ro52/TRIM21 is seen in many SARDs, especially systemic sclerosis where it was reported to be the second most common autoantibody and tended to be associated with interstitial lung disease ( ; ). Anti-Ro52/TRIM21 autoantibodies are also comparatively common in autoimmune inflammatory myopathies, especially in antisynthetase syndrome ( ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here