Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Prior to the modern antimicrobial era, non-viral infections of the central nervous system (CNS) were almost universally fatal. The diagnosis and treatment of CNS infections have improved significantly with advancements made in spinal fluid diagnostic tests, neuroimaging, antimicrobial therapy, and neurosurgical techniques. Despite these advancements, diagnosing CNS infections can be difficult, and a delay in diagnosis and treatment can lead to increased morbidity and mortality. Thus, it is important to quickly recognize when CNS infections are high on the differential diagnosis.
When considering an infectious cause of a CNS disorder, it is important to know predisposing conditions for infection, including geographic residence, travel history, occupational and recreational activities, recent sinus or middle ear infections, chronic medical illnesses, vaccination history, and the patient’s immune status. In general, the immunocompromised will be susceptible to the same organisms as the immunocompetent but will also be susceptible to organisms that are rarely or never seen in the immunocompetent host. In addition, clinical manifestations in immunocompromised patients may be subtle or even absent as they may not generate a brisk immune response to the pathogen. Thus, the differential must always be broader and the bar lower for starting empirical treatment in these patients. The immune status of a patient is particularly important when considering fungal and parasitic diseases.
Bacteria are prokaryotic organisms that colonize all human mucosal and skin surfaces. Only rarely are bacteria pathogenic. The pathogenic bacteria that cause CNS infections can be acquired in a variety of ways, including simple colonization via oral and nasal secretions that becomes invasive (Streptococcus pneumoniae, Neisseria meningitidis) , via contaminated food (Listeria monocytogenes) , as a complication of a systemic infection, from a neurosurgical procedure, or via a contiguous focus of infection (skin, sinuses, or middle ear infections). Most pathogenic bacteria have virulence factors that partially explain why they cause disease, but various host factors (low immunoglobulin levels, impaired T-cell immunity, complement deficiencies) also play a role.
Bacterial meningitis can be divided into acute bacterial meningitis and chronic meningitis. S. pneumoniae and N. meningitidis are the most common causes of community-acquired acute bacterial meningitis. In mastoiditis-associated meningitis, sinusitis, or otitis media, anaerobes often play a role. S. pneumoniae and N. meningitidis colonize the nasopharyngeal mucosa, which provides a route of entry for these bacteria. L. monocytogenes is usually acquired via contaminated food.
Historically, children had the highest incidence of meningitis. However, with the development of an extremely effective Haemophilus influenzae type b (Hib) vaccine, adults now have the highest incidence of meningitis in developed countries. The S. pneumoniae vaccine has also decreased the incidence of meningitis in children by decreasing the incidence of otitis media ( ). Though an absolute increase in the number of cases of H. influenzae non-b and S. pneumoniae serotypes not in the vaccine (“replacement phenomena”) has been seen, this increase in absolute number is small ( ). The incidence of meningitis due to N. meningitidis has decreased with the tetravalent (serogroups A, C, W-135, and Y) meningococcal glycoconjugate vaccine, but the vaccine does not provide lasting immunity and does not include one of the major serotypes, serotype B. Vaccination against serogroup B is a separate vaccine, the MenB vaccine ( ), and has become part of the required vaccination schedule for students matriculating at most American universities. L. monocytogenes accounts for approximately 8% of acute bacterial meningitis cases and is an uncommon cause of meningitis in healthy children and adults. The most common predisposing factors for L. monocytogenes meningitis are age older than 50, diabetes, chronic illness, malignancy, and immunosuppressive therapy or an immunosuppressed state.
Subacute or chronic meningitis is caused by a more diverse group of organisms, and more typically by fungi than bacteria, with the exception of Mycobacterium tuberculosis . Given its endemicity around the world, M. tuberculosis is the leading cause of chronic bacterial meningitis worldwide. At the present time, Treponema pallidum , the causative agent of neurosyphilis, typically causes a meningitis or a meningovasculitis and less often tabes dorsalis or dementia. Other bacterial etiological agents of chronic meningitis include nontuberculous mycobacteria, Coxiella burnetii , Brucella spp., Leptospira spp., Francisella tularensis , Actinomyces spp., Ehrlichia chaffeensis , and Anaplasma phagocytophilum .
Table 79.1 lists the most common bacterial causes of acute or chronic meningitis and diagnostic tests for identifying different organisms.
| Organism | Blood | Cerebrospinal Fluid |
|---|---|---|
| Streptococcus pneumoniae | Culture | Gram stain: gram-positive diplococci in pairs Culture Meningitis/encephalitis panel |
| Listeria monocytogenes | Culture | Gram stain: gram-positive rods Culture Meningitis/encephalitis panel |
| Neisseria meningitides | Culture | Gram stain: gram-negative diplococcus Culture |
| Haemophilus influenzae type b | Culture | Gram stain: gram-negative coccobacillus Culture Meningitis/encephalitis panel |
| Mycobacterium tuberculosis | 20–30 mL for AFB stain and culture; PCR | |
| Treponema pallidum | RPR/VDRL; MHA-TPA; FTA-ABS; TPPA | VDRL (nontraumatic tap) |
| Coxiella burnetii | Acute and convalescent serologies | |
| Brucella spp. | Culture: acute and convalescent serologies | Gram stain: gram-negative coccobacillus Culture |
| Borrelia spp. | ELISA→if equivocal or +, then IgG and IgM WB (follow CDC guidelines for + WB) | Antibody index: anti- Borrelia IgG in CSF/anti- Borrelia IgG in serum to total IgG in CSF/total IgG in serum |
| Leptospira spp. | Acute and convalescent serologies (MAT only done in reference labs, ELISA and lateral flow dipstick less sensitive and specific) Culture: special media; may need to keep for 8–12 wk |
Culture: Special media, fastidious |
The classic symptoms of acute bacterial meningitis are fever, headache, meningismus, and a progressive decrease in the level of consciousness. In a study that evaluated the symptoms in 666 episodes of meningitis in adults, headache was the most common complaint (87%), followed by neck stiffness (83%), fever (77%), and altered mental status (69%). While no single symptom is particularly sensitive or specific, 95% of the patients had two of the four symptoms and only 1% had none. A petechial rash can be present, especially when N. meningitidis is the causative agent, although S. pneumoniae can produce a similar rash ( ). In immunocompromised patients, fever and meningismus may not be present.
Chronic bacterial meningitis presents more subtly than acute meningitis and is defined as symptoms of meningeal inflammation lasting longer than 4 weeks. Symptoms of chronic bacterial meningitis include insidious new headache with or without mild neck stiffness, low-grade fever, and night sweats. Often, it is not until the patient presents with a focal neurological finding, such as cranial nerve palsies, that the syndrome is recognized.
Neurosyphilis typically presents with signs and symptoms of meningitis (meningeal signs, cranial nerve palsies) or meningovasculitis (focal neurological deficits/stroke).
The diagnosis of acute bacterial meningitis depends on recognizing the clinical picture as one consistent with acute meningitis and performing a lumbar puncture (LP) to evaluate for meningeal inflammation and bacteria. Brain imaging prior to LP is recommended in patients with any of the following: altered level of consciousness, focal neurological deficits, new-onset seizures, papilledema (or other signs of increased intracranial pressure), or an immunocompromised state. The utility of imaging is twofold: (1) to evaluate for focal mass lesions and edema that put the patient at risk for uncal herniation, and (2) to find those diseases that might mimic acute bacterial meningitis but in fact are quite distinct (bacterial abscess, tumor). The imaging modality to use in such patients is computed tomography (CT), which can be done quickly and is sensitive enough to rule out lesions that predispose patients to herniation. Non-contrast imaging may show no abnormality while postcontrast images will often show diffuse meningeal enhancement. If head imaging before LP is appropriate, and acute bacterial meningitis is high in the differential, obtaining blood cultures and beginning empirical antibiotics before the patient is sent for imaging is essential. Starting empirical antibiotics quickly is critical because there is burgeoning evidence that delays in initiating antibiotic treatment for bacterial meningitis lead to increased morbidity and mortality ( ). If antibiotics are not initiated prior to imaging, it is also clear that imaging prior to LP significantly delays the time to antibiotics ( ).
The gold standard for the diagnosis of bacterial meningitis is identification of the meningeal pathogen on Gram stain and/or culture of cerebrospinal fluid (CSF). Cultures may take 48–72 hours to be positive. The organism may also be cultured from blood. Newer diagnostic techniques include polymerase chain reaction (PCR) assays; the Meningitis/Encephalitis panel has the advantage of both being sensitive and having a rapid turnaround rate ( ). The Meningitis/Encephalitis panel can detect the DNA of 14 pathogens, 6 of which are bacteria. The bacteria included in this panel are S. pneumoniae, N. meningitidis, H. influenzae, L. monocytogenes, Escherichia coli and Streptococcus agalactiae . The advantage to PCR-based diagnostics is rapid turnaround, but the disadvantages are that these tests are not routinely available, and antibiotic sensitivity data, which are essential, can only be obtained from culture. Thus, in many hospitals, Gram stain and culture remain the best tools for diagnosing bacterial meningitis.
The CSF cell count, and protein and glucose concentrations, can be helpful in differentiating bacterial meningitis from viral meningitis. In general, the classic CSF abnormalities in bacterial meningitis are pleocytosis with a predominance of neutrophils, mild to moderate increase in protein concentration, and decreased glucose concentration (or low CSF/serum glucose ratio, usually ≤0.3.) The classic CSF abnormalities in viral meningitis are a pleocytosis with a lymphocytic predominance, normal to mild elevation in protein concentration, and normal glucose concentration and CSF/serum glucose ratio.
The diagnosis of chronic infectious meningitis is much more complicated and has a broad differential whose specific etiology will often require extensive testing. LP is important in documenting meningeal inflammation, although if there are clinical signs or symptoms consistent with increased intracranial pressure, neuroimaging should be done prior to LP. To identify the cause of chronic meningitis, repeat high-volume CSF cultures or testing may be necessary. In addition, obtaining CSF from a site close to abnormalities on neuroimaging, including cervical punctures, may increase yield. These same principles hold true for accurately diagnosing carcinomatous meningitis , which can be clinically indistinguishable from chronic bacterial meningitis. Neoplastic leptomeningeal metastasis is discussed at length in Chapter 76 .
Magnetic resonance imaging (MRI) with and without contrast is the neuroimaging study of choice because of increased sensitivity compared to CT and the decreased urgency to start empirical treatment and obtain CSF for diagnostic studies. Depending on the CSF abnormalities, certain etiologies may be more or less likely. For example, a mononuclear predominance with a mildly decreased glucose concentration and increased protein concentration suggests tuberculous meningitis, while a CSF pleocytosis with a mononuclear predominance and a normal glucose concentration and either a normal or mildly elevated protein concentration is more consistent with neurosyphilis. To distinguish between these two possibilities, send serum and CSF tests for syphilis (see Table 79.1 ) and high-volume CSF acid-fast bacilli smear, culture, and PCR for M. tuberculosis .
The diagnosis of neurosyphilis is based on either a positive CSF Venereal Disease Research Laboratory (VDRL) test or a combination of a CSF pleocytosis with an elevated protein concentration in a patient with symptoms suggestive of neurosyphilis and a positive serological test (rapid plasma reagin [RPR], VDRL, fluorescent treponemal antibody–absorbed [FTA-ABS], or Treponema pallidum particle agglutination [TPPA]).
Bacterial meningitis is treated initially with empirical antibiotics, which can be narrowed once the specific organism and its antibiotic sensitivities have been determined. Choosing the appropriate empirical regimen depends on the likely organism, which is dependent upon the patient’s age and risk factors. Table 79.2 identifies which antibiotics should be used for specific patient populations, and Table 79.3 lists the recommended CNS dosing for these antibiotics. Antibiotic therapy is modified when the antimicrobial sensitivity tests results are available.
| Disease Entity | Organisms | Antibiotics |
|---|---|---|
| Acute bacterial meningitis: | ||
| Age <50 and no risk factors for Listeria | Streptococcus pneumoniae, Neisseria meningitidis | Vancomycin + ceftriaxone or cefotaxime or cefepime |
| Age >50 and/or risk factors for Listeria | As above + Listeria monocytogenes | As above + ampicillin |
| Sinusitis, mastoiditis, or otitis predisposing cause of meningitis | As above (depending on age and risk factors) + anaerobes | As above (depending on age and risk factors) + metronidazole |
| Brain abscess | Staphylococcus aureus , aerobic and anaerobic streptococci, oral and gastrointestinal flora (including Bacteroides spp.) | Vancomycin + ceftriaxone or cefotaxime or cefepime+ metronidazole |
| Nocardia | Trimethoprim-sulfamethoxazole | |
| Spinal epidural abscess (SEA) | Staphylococcal spp., Streptococcal spp., enteric gram-negative bacilli | Vancomycin + ceftriaxone or cefotaxime or cefepime |
| Chronic meningitis or SEA | Mycobacterium tuberculosis (high suspicion) | Four-drug therapy (isoniazid, rifampin, ethambutol, pyrazinamide) |
| Neurosyphilis | Treponema pallidum | Intravenous aqueous crystalline penicillin G |
| Drug | Dose (Adult, Assuming a Normal Creatinine Clearance) |
|---|---|
| Vancomycin | 40–60 mg/kg/day, divided into q 8-12 h dosing |
| Ceftriaxone | 2 g, q 12 h |
| Cefepime | 2 g, q 8 h |
| Cefotaxime | 2 g, q 4–6 h |
| Ampicillin | 2 g, q 4 h |
| Metronidazole | 500 mg, q 6 h |
| Aqueous penicillin G | 18–24 million U/day |
Early treatment with dexamethasone before or with the first dose of antibiotics has been shown to improve outcomes in patients with bacterial meningitis. In 2002, a landmark prospective double-blinded, placebo-controlled randomized study showed that adjunctive dexamethasone at the time of initiation of empirical antibiotics significantly improved the overall mortality and morbidity of those with bacterial meningitis. Subgroup analysis showed that all mortality and morbidity benefits were derived from the group that had S. pneumoniae meningitis ( ). This was followed by evidence that the nationwide implementation of dexamethasone for pneumococcal meningitis led to a decline in fatality rates ( ). Thus, the current recommendations are to initiate dexamethasone just before or with the administration of empirical antibiotics. A favorable trend for reduced rates of death and hearing loss has been demonstrated for dexamethasone therapy in patients with meningococcal meningitis ( ). Based on animal model data, there was concern that the addition of steroids to empirical antibiotics would decrease the CSF concentration of vancomycin, leading to undertreatment of penicillin-resistant S. pneumoniae meningitis, an issue not addressed by the 2002 study, as all of their isolates were penicillin-sensitive. A study in 2007 prospectively evaluated the CSF vancomycin concentration in patients with suspected S. pneumoniae meningitis who were placed on empirical antibiotics and dexamethasone. Over half of this group had penicillin-resistant S. pneumoniae , and all had CSF vancomycin concentrations at least fourfold higher than the minimum inhibitory concentration (MIC) of the cultured organism. On repeat LP, none of the patients had positive S. pneumoniae cultures, and the CSF vancomycin levels were proportional to the serum vancomycin levels ( ). Thus, it appears that concurrent administration of dexamethasone does not decrease vancomycin CSF penetration in a clinically significant manner and that in these patients, serum vancomycin levels are similarly related to the CSF concentration, as in patients without concomitant dexamethasone.
The management of chronic bacterial meningitis is highly dependent on the organism isolated. Antibiotics are important in all cases, but the appropriate antibiotic(s), length of therapy, and adjunctive use of corticosteroids are all dependent upon the etiology.
Brain abscess has become much less frequent in the post-antibiotic era where otitis media rarely goes untreated or becomes a chronic process. Though fewer abscesses occur now, contiguous infections still make up a considerable proportion of the group; while neurosurgical procedures and trauma also account for a significant proportion of cases. Other risk factors include bacterial endocarditis, diabetes, immunosuppression (alcohol, immunosuppressive drugs), and vascular abnormalities (congenital heart disease, hereditary hemorrhagic telangiectasia). The most commonly isolated bacteria are related to the source of infection and include aerobic and anaerobic streptococci, staphylococci, Bacteroides spp., Enterobacteriaceae, and anaerobic organisms. In endemic areas or in patients who have moved from endemic areas, tuberculomas and cysticercosis are also in the differential diagnosis. In immunocompromised patients, Nocardia spp. and Rhodococcus equi become possible etiologies, especially in those with a concomitant lung infection.
Most patients with brain abscess will present with the signs and symptoms of a space-occupying lesion, such as headache. They may also present with confusion, alterations in consciousness, seizure, or focal neurological deficits. Fever occurs in fewer than half of patients with brain abscess ( ) and should not be used to exclude brain abscess from the differential diagnosis. As in any space-occupying lesion, progressive nausea and vomiting can be seen as the mass expands and intracranial pressure increases. There are no signs or symptoms that definitively exclude or prove bacterial brain abscess in a patient presenting with a space-occupying lesion.
CNS imaging has highly improved the ability to diagnose bacterial brain abscesses and thus decreased mortality from 20%‑50% to less than 20% in most modern series ( ). Every patient with a suspected brain abscess needs timely contrast-enhanced brain imaging. As seen in Fig. 79.1 , the contrast imaging will often show a ring-enhancing mass. Although blood cultures are usually negative, positive cultures, which occur most often in the setting of hematogenous dissemination, can be helpful. Predisposing factors such as otitis media or sinusitis can suggest the likely etiology, but definitive diagnosis requires either a culture from another source (e.g., sputum in disseminated tuberculosis) or directly from the abscess itself. Given the improvements in neurosurgical techniques, including stereotactic aspiration, the ability to treat and definitively diagnose the etiological agent of the abscess has greatly improved. If the patient is stable from a hemodynamic and neurological standpoint, and stereotactic aspiration and cultures can be obtained quickly (within 24–48 hours of presentation), empirical antibiotics should be avoided until after abscess cultures have been obtained. The pathogen is preliminarily identified at the time of stereotactic aspiration by Gram stain, and culture is obtained for definitive identification and antimicrobial sensitivity testing.
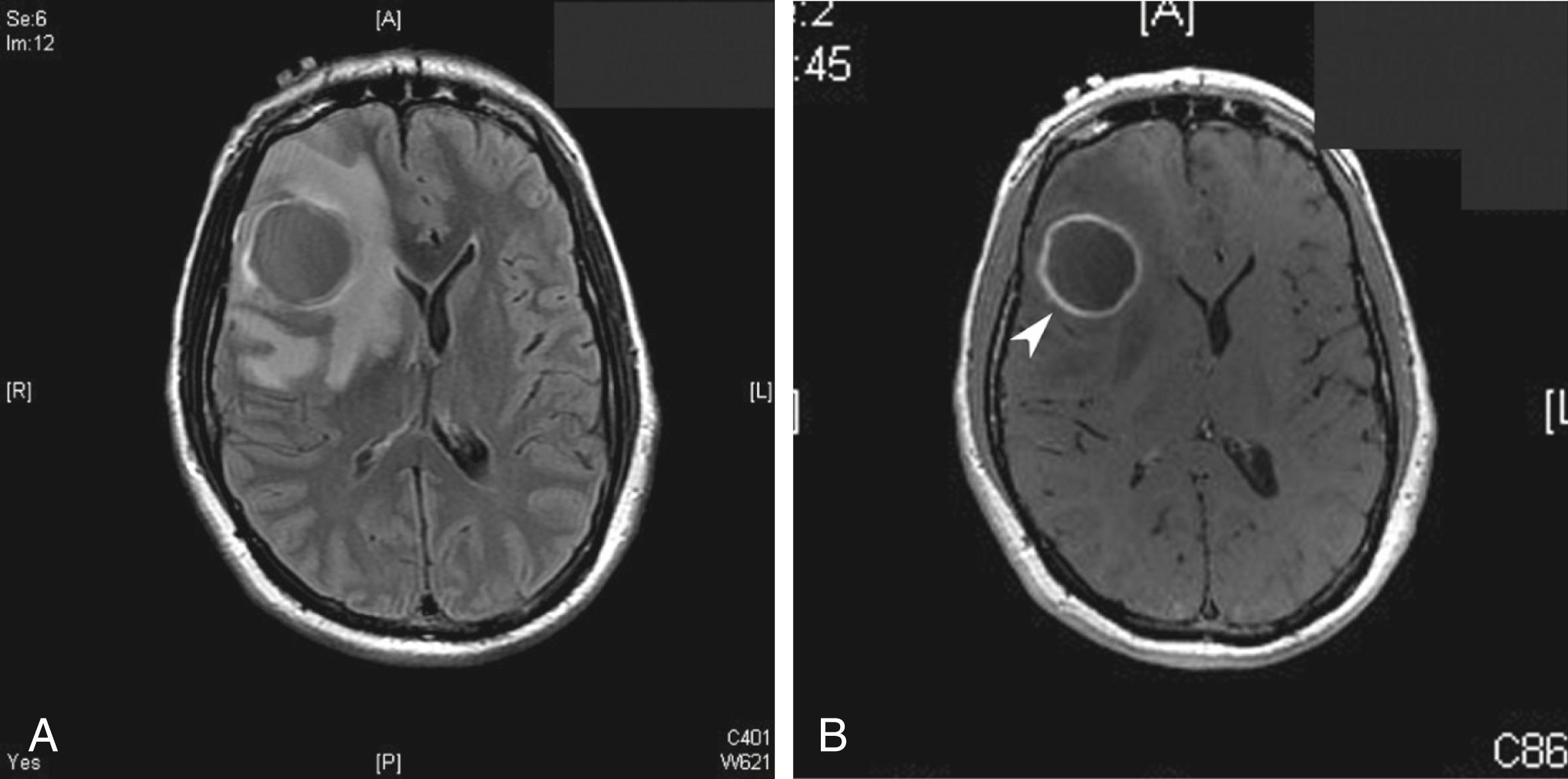
Notably, LP plays no role in the diagnosis of brain abscess. LP has the potential to lead to brain herniation if the abscess is large, culturing the causative bacteria from CSF is uncommon, and there is nothing specific about the CSF profile that will suggest or confirm bacterial brain abscess ( ).
Currently, CT-guided stereotactic aspiration of the abscess in combination with antibiotics is the standard of care. While there are reports of patients surviving with antibiotic treatment alone (typically if the abscess is very small, very deep, or there are multiple abscesses), generally both antibiotics and surgical aspiration are recommended. Aspiration offers the opportunity for both diagnosis and decreasing the size of the abscess, allowing for quicker resolution. Patients with small abscesses (<2.5 cm diameter), those who are poor surgical candidates, and those who have abscesses in deep brain structures can be treated with medical management alone, although with improved surgical technique, even some deep brain structures can be drained ( ).
The usual empirical antibiotics are those that have good CNS penetration and cover both aerobes and anaerobes. Thus, a third-generation cephalosporin in combination with metronidazole is often used. Vancomycin is added if there is concern that the organism is a Staphylococcus species. If the aspirated material or blood cultures yield an organism(s), empirical antibiotics are modified accordingly. The duration of intravenous antimicrobial therapy is usually 6–8 weeks, and some experts recommend continued oral therapy beyond this period. Imaging follow-up is recommended to assess response to treatment.
Spinal epidural abscesses (SEAs) are increasingly common due to the increasing number of diabetics, a major predisposing condition. Staphylococcus aureus is the most commonly isolated organism (∼50%–60% of the isolated organisms in any series), with Streptococcus spp., coagulase-negative staphylococci, and enteric gram-negative rods making up the remainder of the nontuberculous SEA ( ). In areas with endemic tuberculosis or in patients who have recently emigrated from endemic areas, M. tuberculosis as a cause of SEA associated with vertebral osteomyelitis should be high on the differential diagnosis.
The development of SEA can occur from direct spread (i.e., psoas abscess, vertebral osteomyelitis) or from hematogenous spread from a distant site. The cervical spine is less frequently involved than the thoracic or lumbar spine, which may be related to the extent of the epidural space and the venous drainage of the thoracic and lumbar spine ( ).
SEA is a progressive disease that usually begins with new-onset back pain or localized tenderness and progresses to radicular pain and ultimately to neurological deficits, including bowel and bladder dysfunction. The classic triad of SEA is back pain or localized tenderness, fever, and progressive neurological deficits localized to the spinal cord. Similar to bacterial meningitis, this triad is highly specific but has low sensitivity, especially early in the disease course ( ). In most studies, back pain is a presenting feature in 60%–70% of patients, and “fever” is present in 50%–60%, although it is not always clear if this is a reported or documented fever. In addition, as one study noted, patients with back pain often use anti-inflammatory medications, which have antipyretic effects, making the presence of a fever even less likely in these patients ( ).
When trying to determine whether a patient with new-onset back pain should be imaged to rule out SEA, one should assess the patient for the known risk factors for SEA. The most common risk factors are diabetes mellitus, intravenous drug use, alcohol abuse, immunocompromised state (including immunosuppressive drugs, acquired immunodeficiency syndrome (AIDS), cancer patients), spinal surgery/procedure, trauma (spinal or extraspinal), and extraspinal infections (furunculosis/cellulitis, psoas abscess, etc.). Which risk factor is most highly related to SEA depends on the study and likely reflects the different populations studied ( ). In every study there are a small number of patients without any risk factors, but in general, most patients have at least one of the previously listed risk factors ( ). In addition, while SEA can be located anywhere along the spinal axis, up to a third of patients will present with thoracic pain. As the thoracic spine is an unusual place to develop mechanical back pain, new-onset thoracic back pain should make one consider SEA sooner rather than later.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here