Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
This chapter will:
Identify the mechanisms of renal damage induced by nonsteroidal antiinflammatory drugs (NSAIDs) and coxibs.
Describe the incidence and epidemiology of NSAID-induced acute kidney injury.
Describe the risk factors for NSAID-induced acute vasomotor renal injury and NSAID-induced nephrotoxicity.
Nonsteroidal antiinflammatory drugs (NSAIDs) are prescribed widely in clinical practice and are available without a prescription. NSAIDs, including the more selective inhibitors of cyclooxygenase 2 (coxibs), are potentially nephrotoxic and can have significant side effects, including salt and water retention, acute tubular necrosis, acute interstitial nephritis, proteinuria, hyperkalemia, and various degrees of renal damage evolving into chronic kidney disease (CKD). Although the potential for these side effects is relatively low, the widespread use of these drugs makes the incidence of complications frequent. Among the various clinical complications, the effects on the kidney are probably the most common and severe. It has been reported that 37% of drug-associated acute kidney injury (AKI) is associated with the use of NSAIDs, and NSAID-induced AKI accounts for 7% of overall cases of AKI.
There is an increased risk of developing renal damage in patients who regularly consume NSAIDs. This risk increases with advanced age and other comorbidities. For instance, in a recent Cochrane review of ASA, steroids, or NSAIDs for Alzheimer disease, Jaturapatporn et al. identified 1745 subjects included in randomized trials of NSAIDs and the relative risk of increased serum creatinine in the traditional NSAID group was increased 4.30-fold (95% confidence interval [CI], 0.54 to 34.30), while hypertension increased 3.36-fold (95% CI, 0.84 to 13.34). Age-related changes in renal function predispose to nephrotoxicity, especially when there is a decreased glomerular filtration rate (GFR), decreased renal blood flow, and increased renal vascular resistance, dehydration, or impaired liver function. In addition, with age and comorbidity the pharmacokinetics of NSAIDs may be affected. In all these conditions, the risk for renal dysfunction is enhanced further by the presence of sepsis, multiple organ dysfunction, and critical illness in general. In a recent analysis of a large general practice database in the United Kingdom, current users of NSAIDs faced a relative risk of AKI of 3.2 (95% CI, 1.8 to 5.8), compared with nonusers. This risk increased with comorbid illness, and in particular it rose dramatically to 11.6 (95% CI, 4.2 to 32.2), when concomitant diuretics were being taken. In the critically ill patient, it is clear that a thorough medical history and pharmacologic history is paramount, especially in relation to the consumption of prescription or nonprescription NSAIDs.
AKI is a well-described complication of NSAID use. There are two main mechanisms of renal damage induced by NSAIDs: one is hemodynamically mediated, and the other is derived from a direct toxicity of the drugs on the renal parenchyma.
AKI can result from an alteration of intrarenal microcirculation (and, consequently, of glomerular filtration) caused by inhibition or blockage of endogenous prostanoids, particularly in those conditions in which kidney microperfusion and function are highly dependent on their augmented serum concentration. NSAID-induced AKI is not common, but it may occur with increased incidence in patients at risk. Risk factors for this disorder include volume and salt depletion, advanced age, renal insufficiency, liver dysfunction, diabetes, and congestive heart failure. In these patients, side effects involving the kidney can occur with classic nonselective NSAIDs and with coxibs. The explanation for the increased incidence of AKI in volume-depleted patients is based on the fact that under euvolemic conditions, basal levels of prostaglandins are not particularly high; therefore intrarenal circulation is not dependent on these substances for maintenance of GFR. In turn, the inhibition of prostaglandin synthesis by NSAIDs very seldom affects renal circulation and function. On the contrary, in volume-depleted and hemodynamically unstable patients, prostaglandin synthesis is enhanced, significantly mitigating the vasoconstriction induced by angiotensin II and the effects of vasopressin on the collecting tubular epithelium. The consequence of inhibiting endogenous prostaglandin synthesis with NSAIDs is removal of this counterbalancing force, which leads to increased vascular tone with significant antidiuresis and antinatriuresis. A complete overview of the arachidonic acid metabolic pathways and the roles of the various metabolites in renal function is presented in Fig. 218.1 .
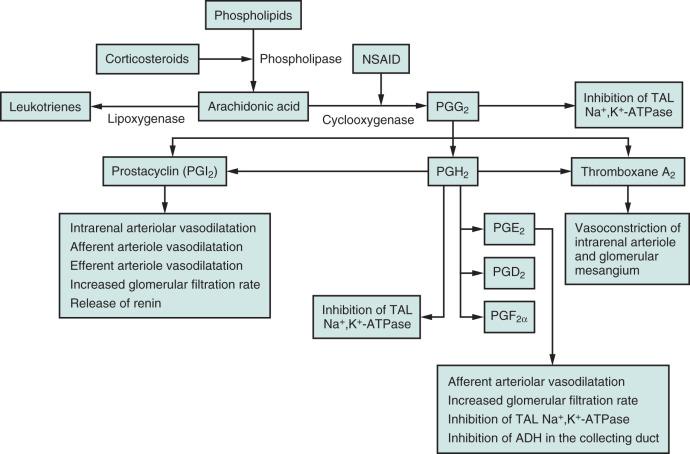
Kidneys produce a mixture of autacoids with vasodilating action (prostaglandin E 2 [PGE 2 ], PGI 2 ) and vasoconstricting action (PGF 2α , thromboxane A 2 ). Such substances contribute to the regulation of intrarenal blood flow, tubular sodium handling, tubular water transport, and renin release. In Fig. 218.1 , the actions of these metabolites of arachidonic acid are described. A delicate feedback system leads to hemodynamically mediated activation of prostaglandin secretion in response to the vasoconstriction induced by angiotensin II, endothelin, catecholamines, leukotrienes, and vasopressin. Furthermore, in patients with underlying ischemic or inflammatory renal injury (e.g., sepsis), the consumption of NSAIDs may produce a catastrophic event mediated not only by a blockage of vasodilatory prostaglandin production but also by a nonenzymatic stimulation of vasoconstrictor metabolites in a setting in which free oxygen radical production is enhanced.
Interstitial nephritis can occur in association with NSAIDs because of allergic reaction, direct cellular toxicity, alteration of metabolic pathways, and possibly obstruction. These complex and multifactorial mechanisms also may combine with hemodynamic perturbations. In some cases, the damage becomes extensive, leading to papillary necrosis. The use of NSAIDs often is associated with worsening of mild to moderate CKD, with significant reduction of GFR and evolution of CKD toward more advanced stages. Direct toxicity to the tubules is manifested by loss of polarity, loss of tight junctions, loss of cell substrate adhesion, exfoliation of viable cells from the tubular basement membrane, and aberrant renal cell-cell adhesion. Further damage may lead to altered gene expression, cellular dedifferentiation, and lethal injury such as necrosis or apoptosis. Tubular necrosis is characterized by severe depletion of cellular stores of adenosine triphosphate, reduced activity of membrane transport pumps, cell swelling, increase in intracellular free calcium, activation of phospholipases and proteases, glycine depletion, and plasma and subcellular membrane injury.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here