Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
For the anatomist and pathologist, the terms “univentricular” and “single” have proven to be among the most controversial words used to describe a congenitally malformed heart. Since the mid-1990s, light has begun to emerge at the end of the tunnel for those seeking a logical framework for the use of these words. In no small way, this reflects the recognition by clinicians that it is frequently not possible surgically to create a biventricular circulation in a patient having two well-formed ventricles. In these circumstances, conversion to the Fontan circulation permits both the systemic and pulmonary circulations to be supported by only one of the ventricles. In other circumstances, as when there is a double-outlet ventricle with remote interventricular communication, it is the combined ventricular mass that ejects to the systemic circulation. These facts underscore the grouping of the markedly heterogeneous anatomic entities discussed in this chapter. The arrangement in the setting of double-outlet ventricles is discussed in Chapter 39 . The anatomy involved when both circulations are supported by only one ventricle becomes easier to understand in consideration of the extensive controversies in the earlier literature, which has offered numerous illogical definitions of “single ventricles” or “univentricular hearts.” These definitions reflected the interpretation of ventricular morphology in patients having double-inlet ventricles as opposed to atrioventricular valvar atresia. It was, of course, patients having tricuspid atresia who were first converted to the Fontan circulation. The majority of patients now treated in this fashion, however, have hypoplastic left heart syndrome. Such patients have unequivocally biventricular hearts, as do many of those with pulmonary atresia and an intact ventricular septum, who are treated in comparable fashion. Here we address the basic morphology of all these lesions, along with those found in other patients with obviously biventricular morphologic arrangements. We begin, however, by explaining why the topic has proved so controversial for both morphologists and clinicians, while accepting that such considerations are now largely of historical interest.
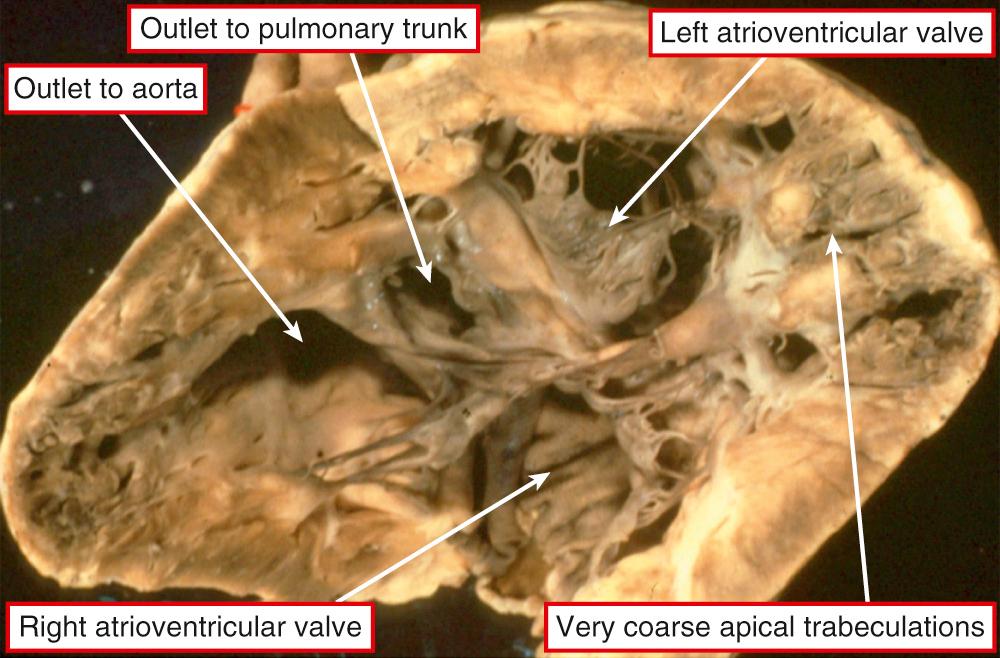
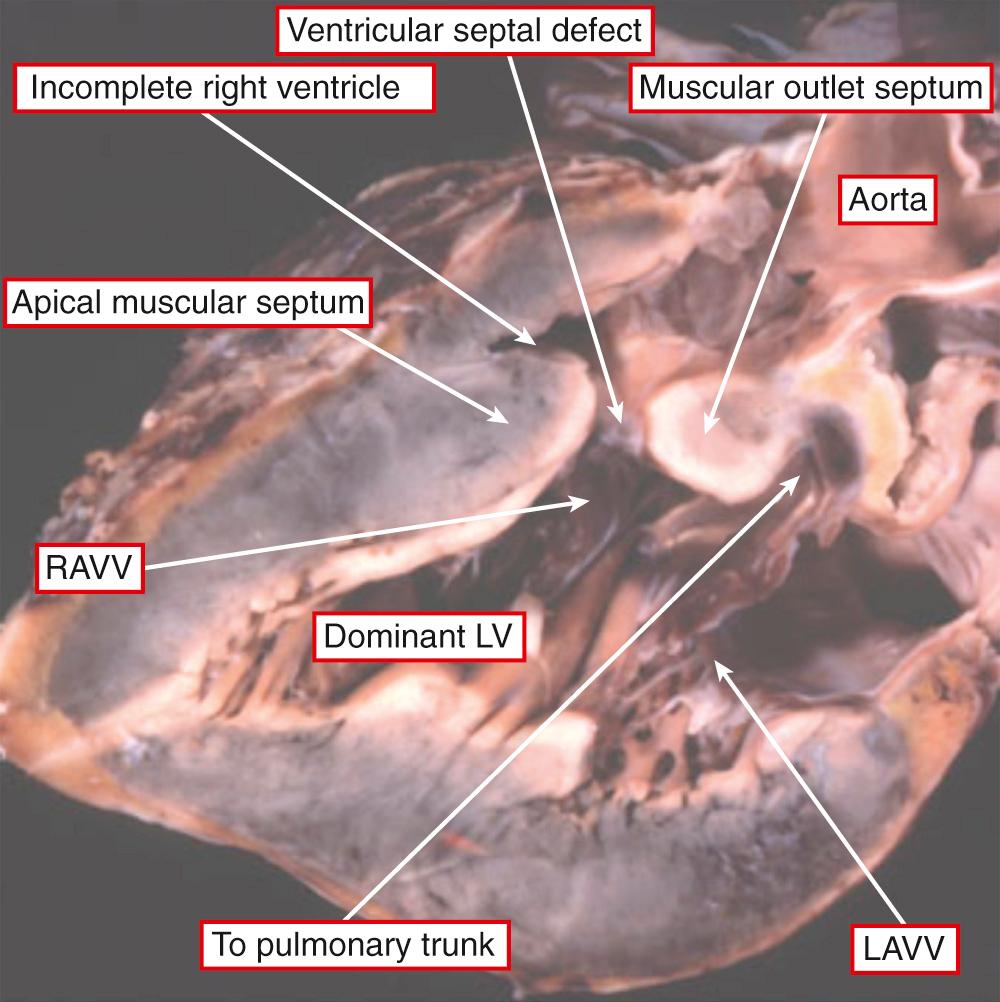
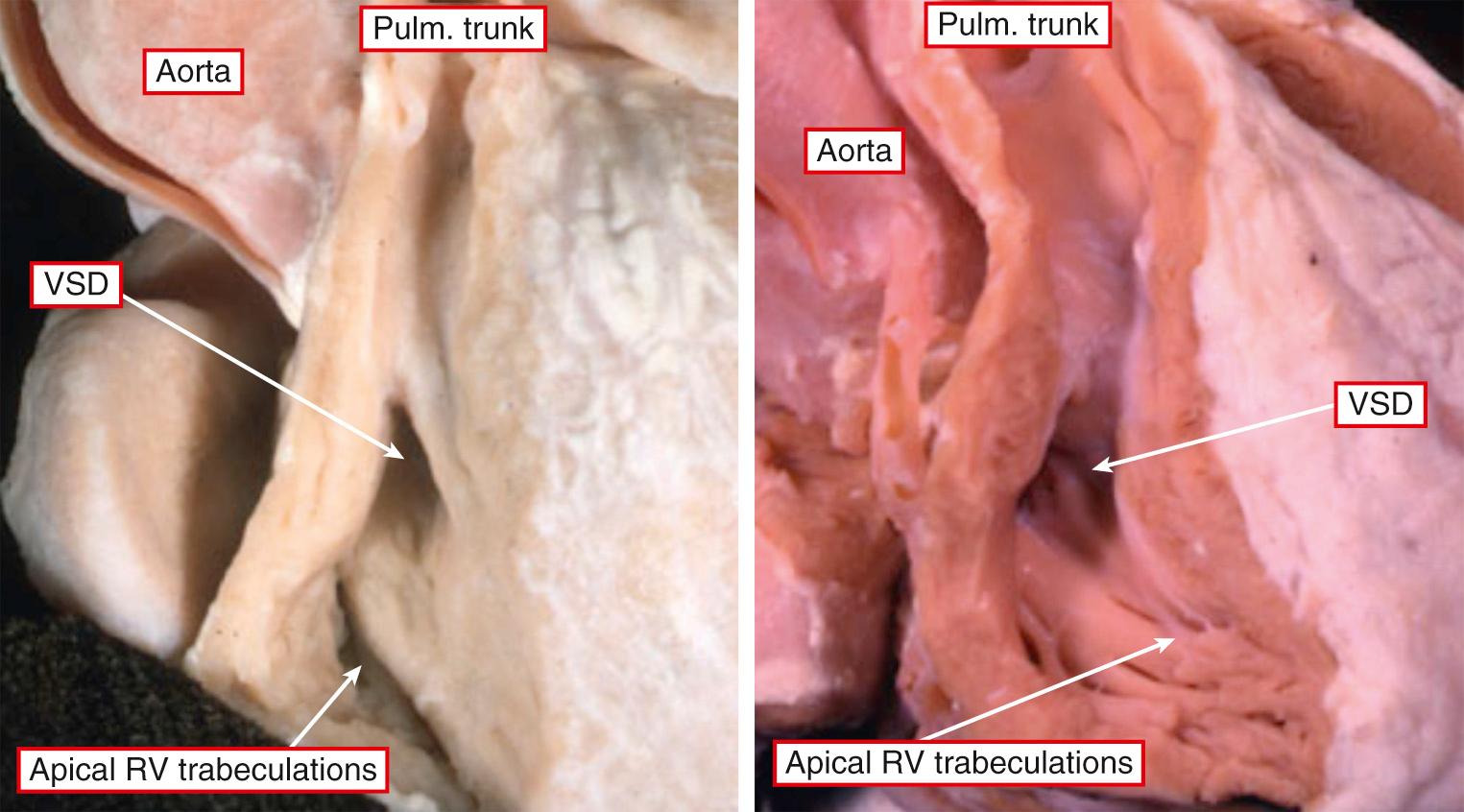
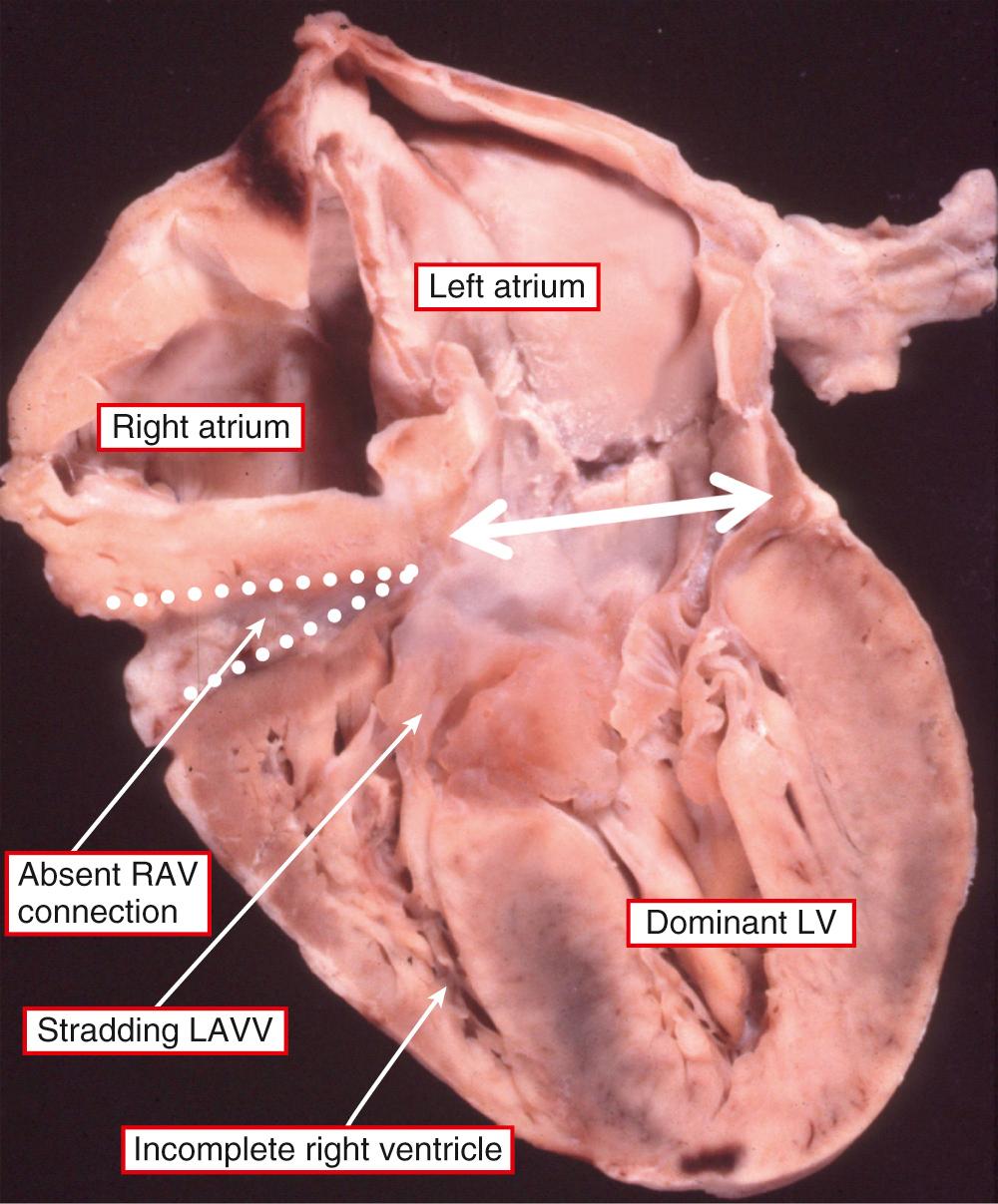
It is very unusual to find hearts with a solitary chamber within the ventricular mass. When found, they exhibit a double inlet to a ventricle that has particularly coarse apical trabeculations and in which the only septal structure present is the muscular outlet chamber ( Fig. 69.1 ). Such hearts have not only a univentricular arrangement of the ventricular mass but also a univentricular arrangement of the atrioventricular junctions. This is because, in the setting of all variants of double-inlet ventricle, the atrial chambers connect to only one ventricle irrespective of whether or not the ventricular mass is itself univentricular. This fact is significant because, subsequent to a study that appeared in 1964, it came to be accepted that the criterion for the definition of a “single ventricle” was the presence of a double-inlet atrioventricular connection. This was accepted despite the fact that those promoting this terminology recognized that, in the majority of the patients having double-inlet ventricle, a second chamber was present within the ventricular mass ( Fig. 69.2 ). At the time, such hearts were considered to be “univentricular” because the second chamber was interpreted as representing an “infundibular outlet chamber,” and hence not a “true” ventricle. There was a problem with this approach, since at that time the hearts with atrioventricular valvar atresia were deemed to be biventricular, with the small chamber in patients having tricuspid atresia accepted as being a right ventricle. When comparisons are made between the morphology of the small chamber in hearts with tricuspid atresia and those with double-inlet left ventricle—provided that the comparison is made in the setting of hearts having the same ventriculoarterial connections—the chambers are virtually indistinguishable ( Fig. 69.3 ). Such similarity is no more than should be expected, since in both tricuspid atresia and double-inlet left ventricle the larger ventricle is of left ventricular morphology. The smaller chamber is an incomplete right ventricle lacking its inlet component. In double-inlet left ventricle, this occurs because both atrioventricular junctions are supported by the dominant ventricle (see Fig. 69.2 ). In classic tricuspid atresia, in contrast, the right ventricle lacks its inlet because there is congenital absence of the right atrioventricular connection ( Fig. 69.4 ). In both settings, as in the hearts with an indeterminate ventricle, it is the atrioventricular connections that are univentricular. With one small exception, therefore, it follows that all patients with congenitally malformed hearts can be placed into one of two groups. The first group is made up of those with biventricular atrioventricular connections, specifically concordant, discordant, and mixed atrioventricular connections (see Chapter 2 ). In the second group, the atrioventricular junctions are supported by only one ventricle, either because of double-inlet ventricle or because one of the junctions is completely absent. There is a very small third group in which there is absence of one of the atrioventricular connections but with straddling and overriding of the solitary atrioventricular valve. This combination produces a uniatrial but biventricular connection ( Fig. 69.5 ). In all the patients having univentricular atrioventricular connections, apart from the very small minority having solitary and indeterminate ventricles, a second ventricle is to be found within the ventricular mass ( Fig. 69.6 ). In the presence of this second chamber, it follows that the hearts themselves are not “univentricular.” When found, the second ventricle is incomplete, lacking at the least its inlet and often also lacking any direct outlet other than the ventricular septal defect. On rare occasions, it may be possible surgically to septate the dominant ventricle in the setting of double inlet and hence to restore biventricular circulations. Patients with univentricular atrioventricular connections currently are almost always treated by conversion to the Fontan circulation, as are many patients with biventricular atrioventricular connections. In such circumstances, all the hearts will become functionally univentricular subsequent to creation of the Fontan arrangement. The addition of the word “functionally,” therefore, provides the logic for the grouping of the various entities to be described in this chapter.
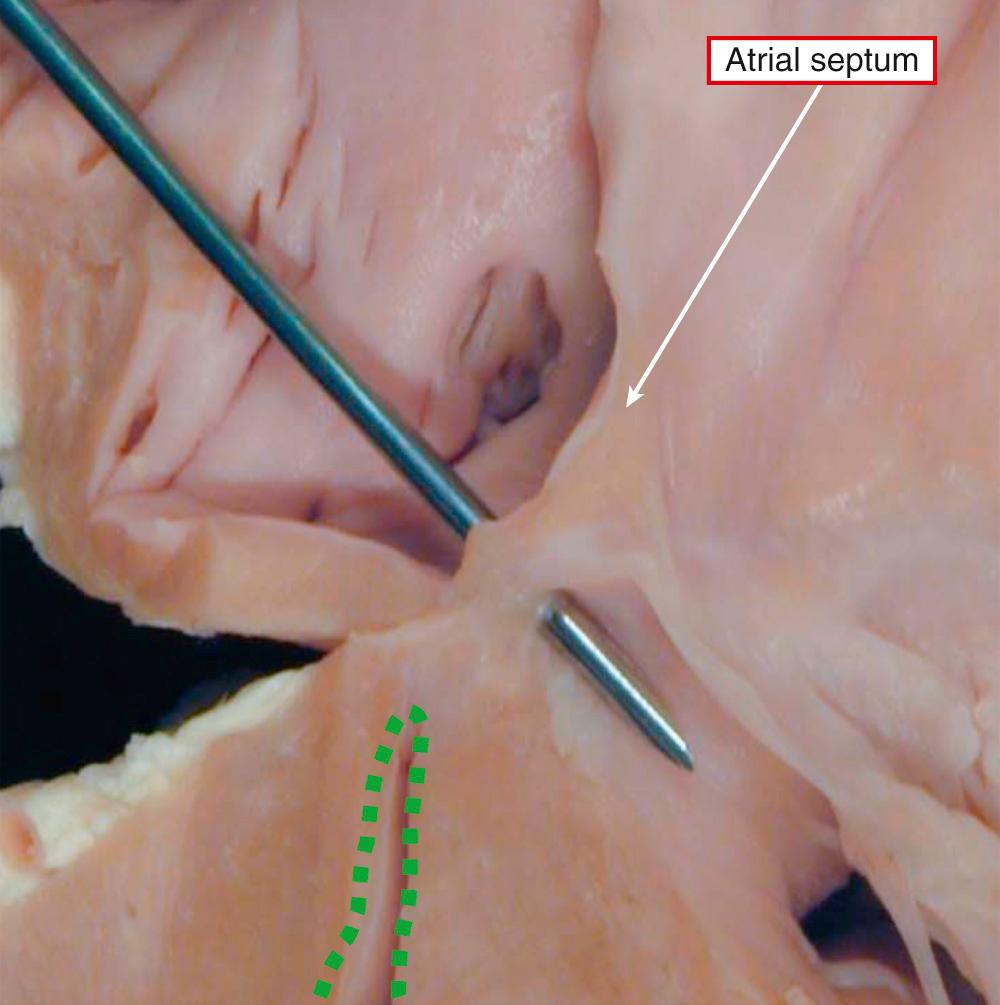

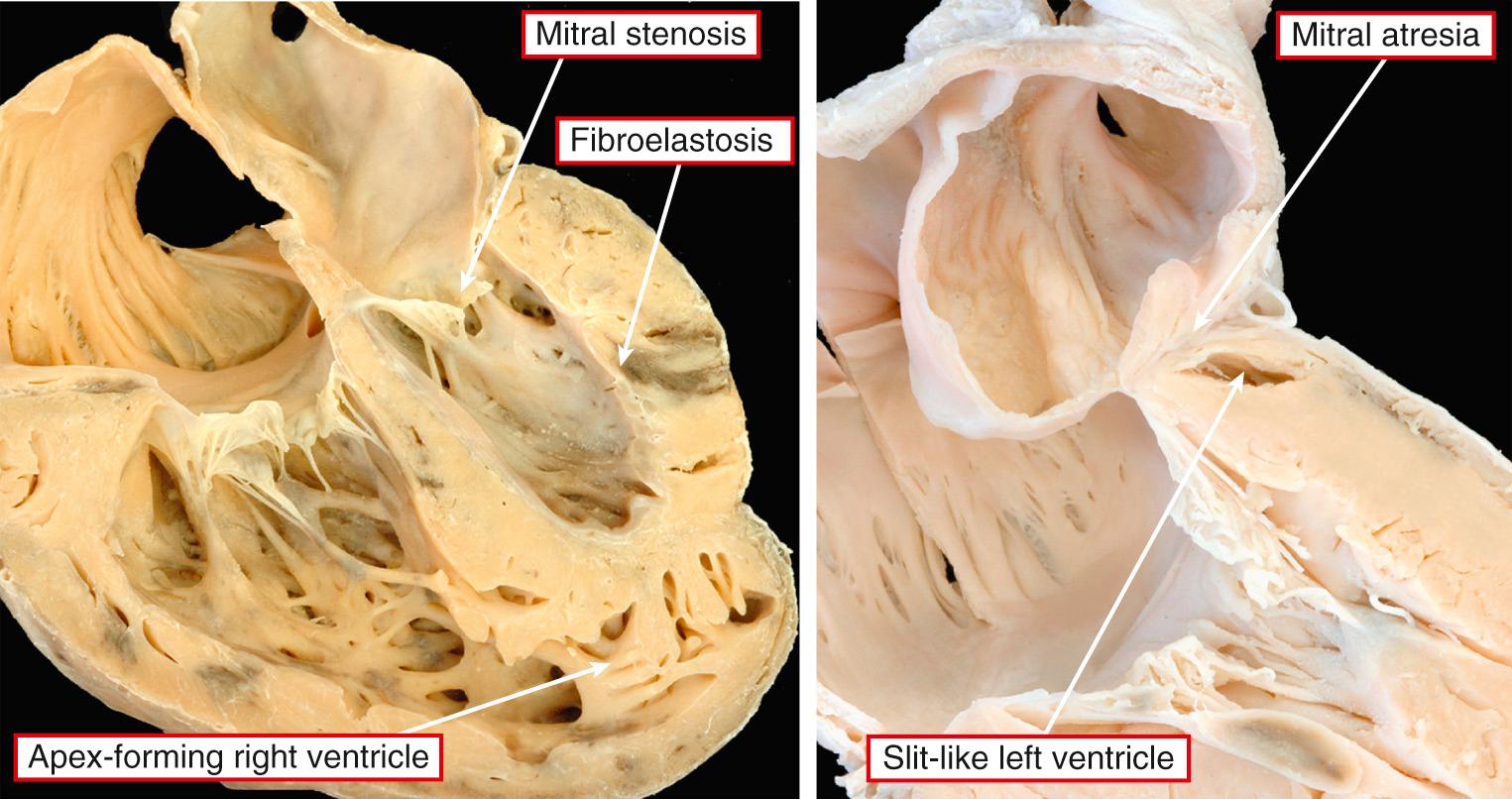
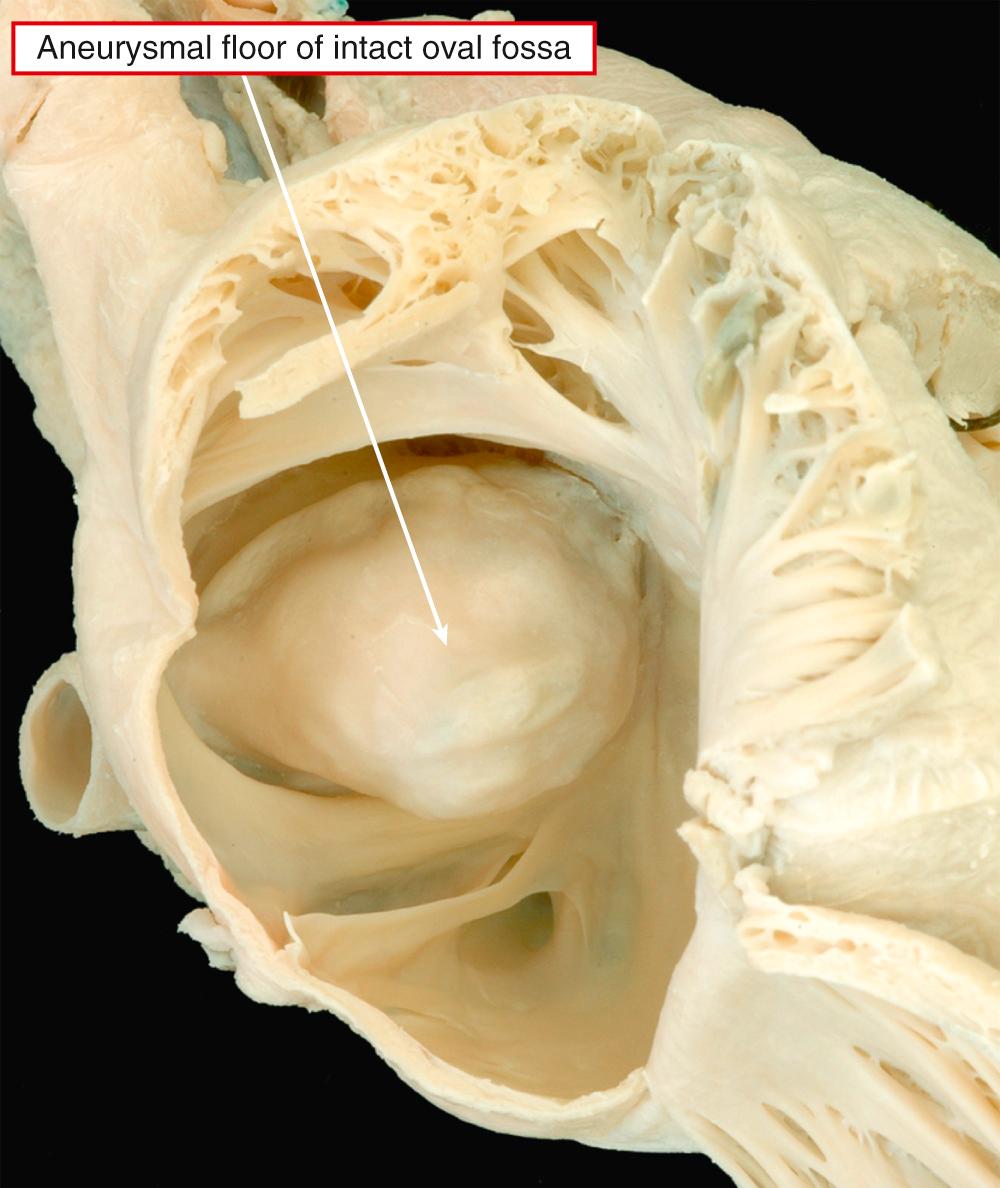
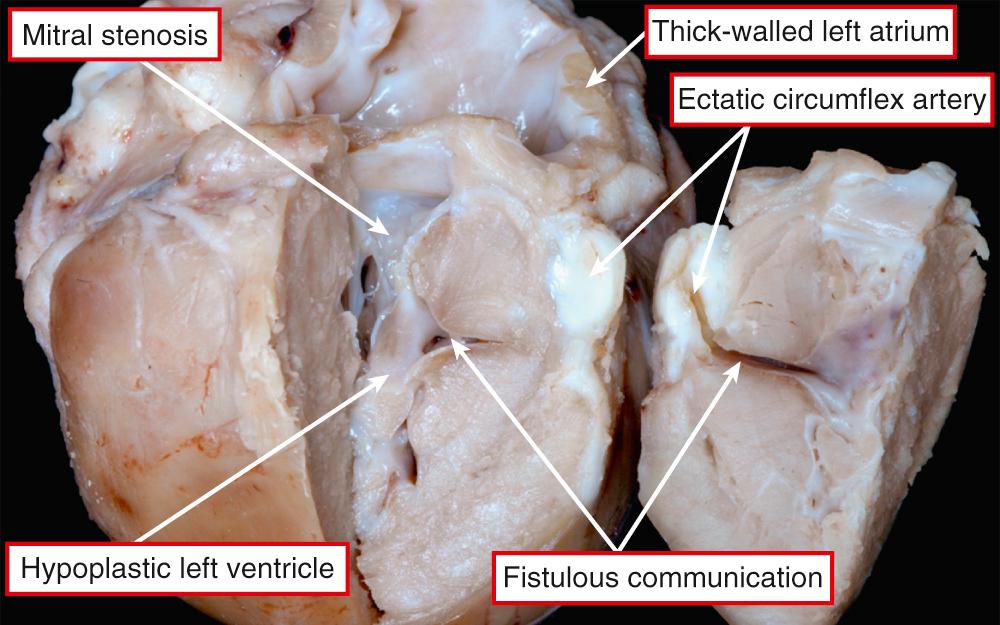
Rather than being a single anatomic entity, the hypoplastic left heart syndrome groups together entities with characteristic physiologic and clinical properties. The earliest grouping was made by Lev, but he described the syndrome as “hypoplasia of the aortic tract.” Hypoplasia of the left heart is not always the same as aortic atresia or hypoplasia. On rare occasions, the aortic lesions can be found in the setting of a left ventricle of normal size, particularly when the aorta takes its origin from the morphologically right ventricle. It was Noonan and Nadas, as far as we can establish, who first used the term “hypoplastic left heart syndrome.” Although the combinations of anatomic lesions that constitute the syndrome are variable, the features when seen together are unmistakable. Thus the syndrome includes the majority of cases of aortic atresia and many of those with critical aortic stenosis. The mitral valve can similarly be stenotic or atretic. In all instances, however, a key feature is the integrity of the ventricular septum. In most of the cases described in the initial investigation of Noonan and Nadas, aortic coarctation was the major anatomic feature, although the left ventricle was deemed to be hypoplastic. It is questionable whether patients with such findings would today be included as having hypoplastic left heart syndrome. Rare examples are found in which the mitral and aortic valves are of normal dimensions relative to the coexisting hypoplasia of the left ventricle. Biventricular repair is possible in such cases, following simple relief of any obstruction within the aortic arch and descending aorta. This shows that major difficulties remain in providing an all-encompassing anatomic definition for hypoplasia of the left heart. It is more realistic, therefore, to analyze the unifying hemodynamic features. In physiologic and clinical terms, hypoplasia of the left heart can be defined as the situation where the systemic circulation is dependent on the morphologically right ventricle in the setting of atresia or severe hypoplasia of the aortic valve. Such hypoplasia of the left heart is usually found in the setting of usual atrial arrangement with concordant ventriculoarterial connections. There can be concordant atrioventricular connections ( Fig. 69.7 , left ) or mitral atresia due to absence of the left atrioventricular connection ( Fig. 69.7 , right ). In those with mitral atresia, the left ventricular cavity is often no more than a slit in the posteroinferior wall of the ventricular mass. It is almost impossible to determine on morphologic grounds when the left ventricle is to be considered hypoplastic in patients with aortic valvar stenosis rather than atresia. When there is obvious fibroelastosis in the small ventricle and the right ventricle is apex-forming, patients with such hearts certainly fulfill the anatomic criterions for the syndrome ( Fig. 69.8 ). The left ventricular walls are hypertrophied when there is mitral stenosis rather than atresia (see Fig. 69.7 , left ). Indeed, the mass of the left ventricle may rarely be increased rather than decreased despite the cavitary hypoplasia. The left atrium is usually small, although there can be hypertrophy of the left atrial walls. Some patients can have an enlarged atrium, particularly the appendage. The oval foramen is usually patent, but in up to one-tenth of patients, the atrial septum will be intact. It can be aneurysmal, with protrusion into the cavity of the right atrium ( Fig. 69.9 ).
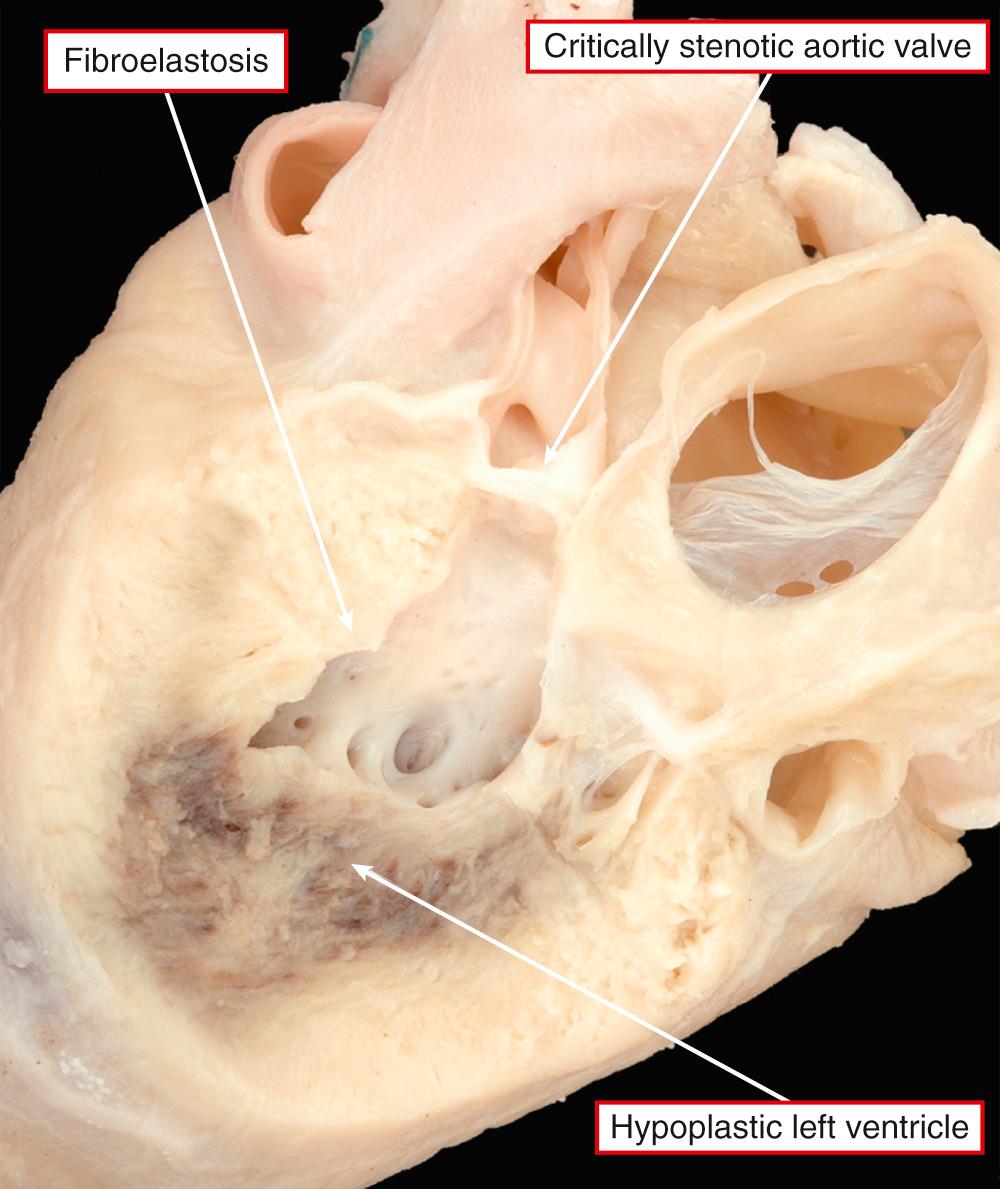
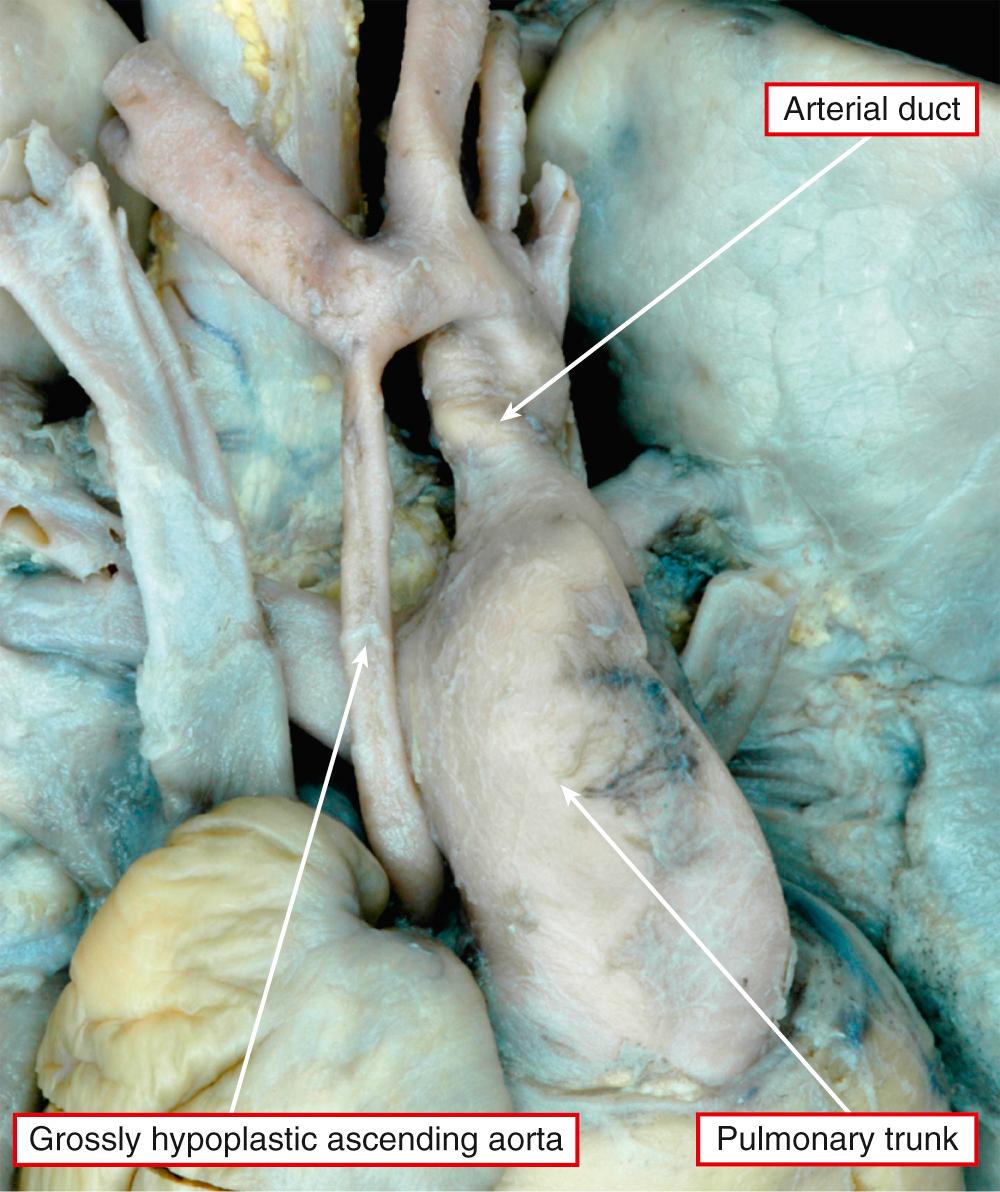
In some of these patients with an intact oval fossa, the pulmonary venous drainage can be abnormal because of the presence of a so-called levoatrial cardinal vein or because of fenestration of the walls that usually separate the left atrium from the cavity of the coronary sinus. This provides an overflow for the left atrial return. More usually, there is no such overflow when the atrial septum is intact. The resulting increase in left atrial pressure produces increased left atrial hypertrophy, along with changes in the lungs, including arterialization of the pulmonary veins and lymphangiectasia, with a “cobblestone” appearance of the pulmonary surfaces seen at autopsy. These are bad prognostic features. In most instances, as stated, the oval foramen is patent and does not obstruct flow from left to right. The primary atrial septum is frequently deviated into the left atrium. Left ventricular endocardial fibroelastosis is seen only when the mitral valve is perforate. Aortic atresia, when present, can result from an imperforate valve. More usually, there is no evidence of persisting leaflets at the ventriculoarterial junction, with fibromuscular tissue interposing between the ventricular cavity and the blind-ending aortic root. The aortic root itself is usually markedly hypoplastic, with the ascending aorta serving only as a conduit to feed the coronary arteries ( Fig. 69.10 ). Further distally, aortic coarctation is common, occurring in over four-fifths of patients. The obstructive shelf is typically in a preductal location but can be found paraductally. Ductal tissue is not only incorporated into the stenosing shelf but also may extend proximally and distally, an important point for those undertaking surgical palliation of these patients. Abnormalities of the coronary arteries, such as ventriculocoronary fistulas and abnormal tortuosity, are more common in the subgroup of patients with mitral stenosis and aortic atresia ( Fig. 69.11 ). Such coronary arterial fistulas, if present, are not necessarily associated with abnormalities of the right ventricular myocardium but can produce an increased risk of mortality at the first stage of a Norwood reconstruction. The conduction tissues are in their expected location, albeit with miniaturization of the left bundle branch in keeping with the size of the left ventricle.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here