Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Longevity is increasing and the population of older adults is expanding . In Europe, the estimated percentage of the elderly population (aged older than 65 years) is 16.1% . In Japan, the elderly population is expected to exceed 30% in 2025 . Elderly people require dedicated therapeutic approaches because medical care for this population is different from that for younger adults . The prevalence of multiple comorbidities significantly increases with age . In addition, the “normal” for this population is different from that of the general population . Although further investigations are warranted, a recent study from Southern Italy suggested traditional cardiovascular risk factors in nonagenarians and centenarians were distinct from those in the general population; for example, high serum cholesterol was associated with a reduced mortality rate . One of the age-related physiological changes is sarcopenia, which is the reduction in muscle mass and strength. Sarcopenia plays a role in the pathogenesis of frailty and functional impairment, and it is associated with increased risk of cardiovascular disease, atherosclerosis, and mortality . It is essential for health care professionals to understand these characteristics associated with aging in order to provide integrated, comprehensive care for the elderly.
Both the incidence and prevalence of most cardiovascular diseases increase with age . Two-thirds of patients with cardiovascular disease are older than 60 years old . More than 85% of people >85 years of age have some form of cardiovascular disease . Cardiovascular disease is the leading cause of death in adults older than 75 , and atherosclerotic cardiovascular disease, particularly of the coronary arteries, is a common cause of death. Cardiac structures undergo progressive fibrosis, calcification, and degeneration, resulting in dysfunction of the involved structures. In the valves, these changes cause stenosis and/or regurgitation. Degenerative (calcific) aortic stenosis is the most common valve disease in the elderly. In the myocardium, fibrosis due to a variety of disorders causes dysfunction and arrhythmias. Fibrosis may also lead to conduction abnormalities that can be fatal or require pacemakers. Primary cardiomyopathies usually present at younger ages, but dilated and hypertrophic forms, such as Tako-tsubo cardiomyopathy, do affect the elderly. Secondary cardiomyopathy of amyloidosis is common in the very elderly and is an important cause of death.
Both structural and functional changes occur in the cardiovascular system with advancing age . There are several age-related physiological changes in cardiac function even in people without any heart diseases , resulting in progressive stiffness of the ventricle. Heart rate variability, one of the indicators of cardiac autonomic regulation, decreases with age . Systolic left ventricular function remains unchanged with age in persons without cardiovascular disease . In contrast, diastolic left ventricular function undergoes alterations. The early diastolic LV filling rate is gradually reduced with aging . Diastolic dysfunction subsequently may result in heart failure with preserved ejection fraction (HFpEF). HFpEF will be discussed later in Section “Heart failure accompanied by preserved ejection fraction.”
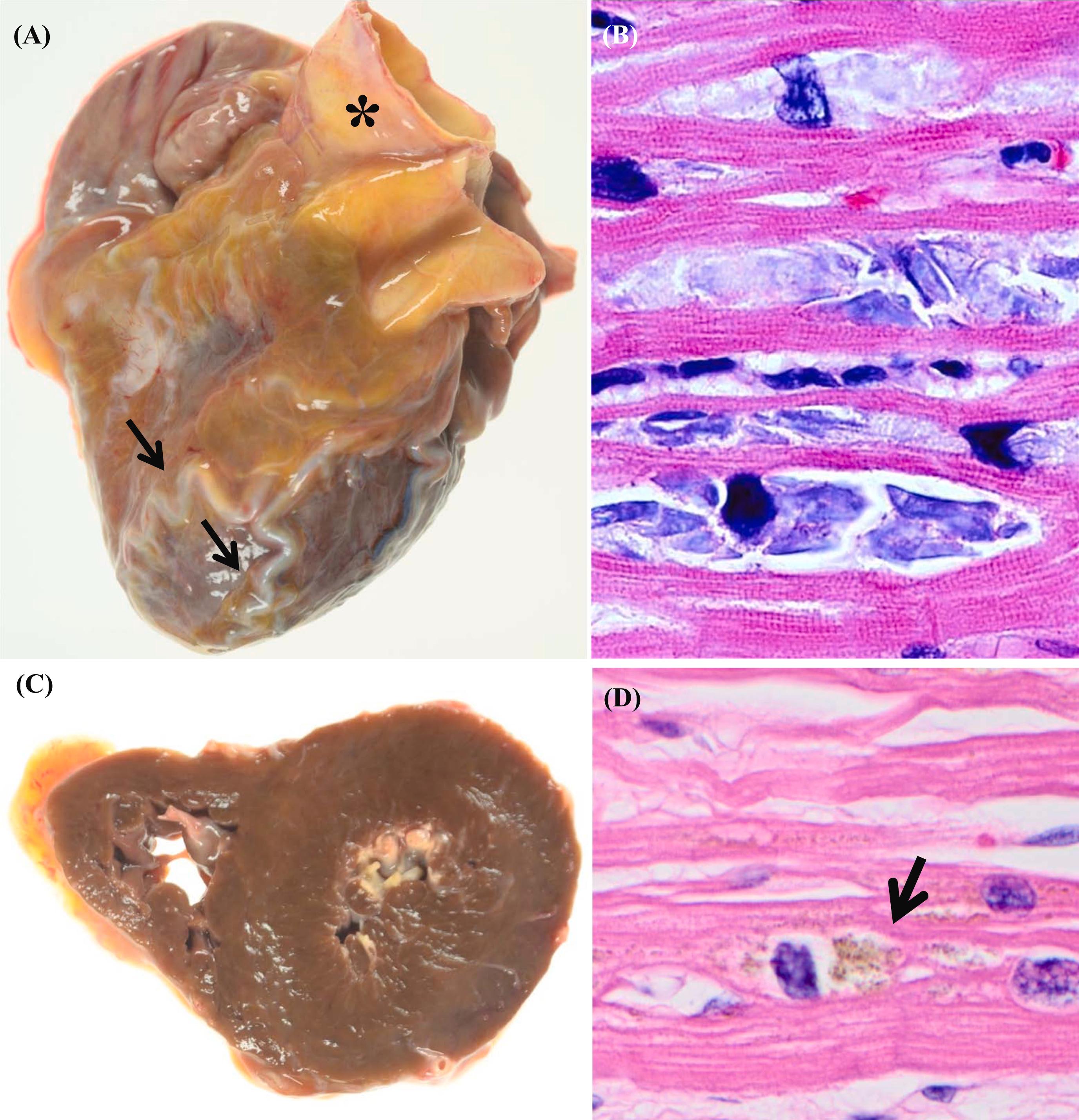
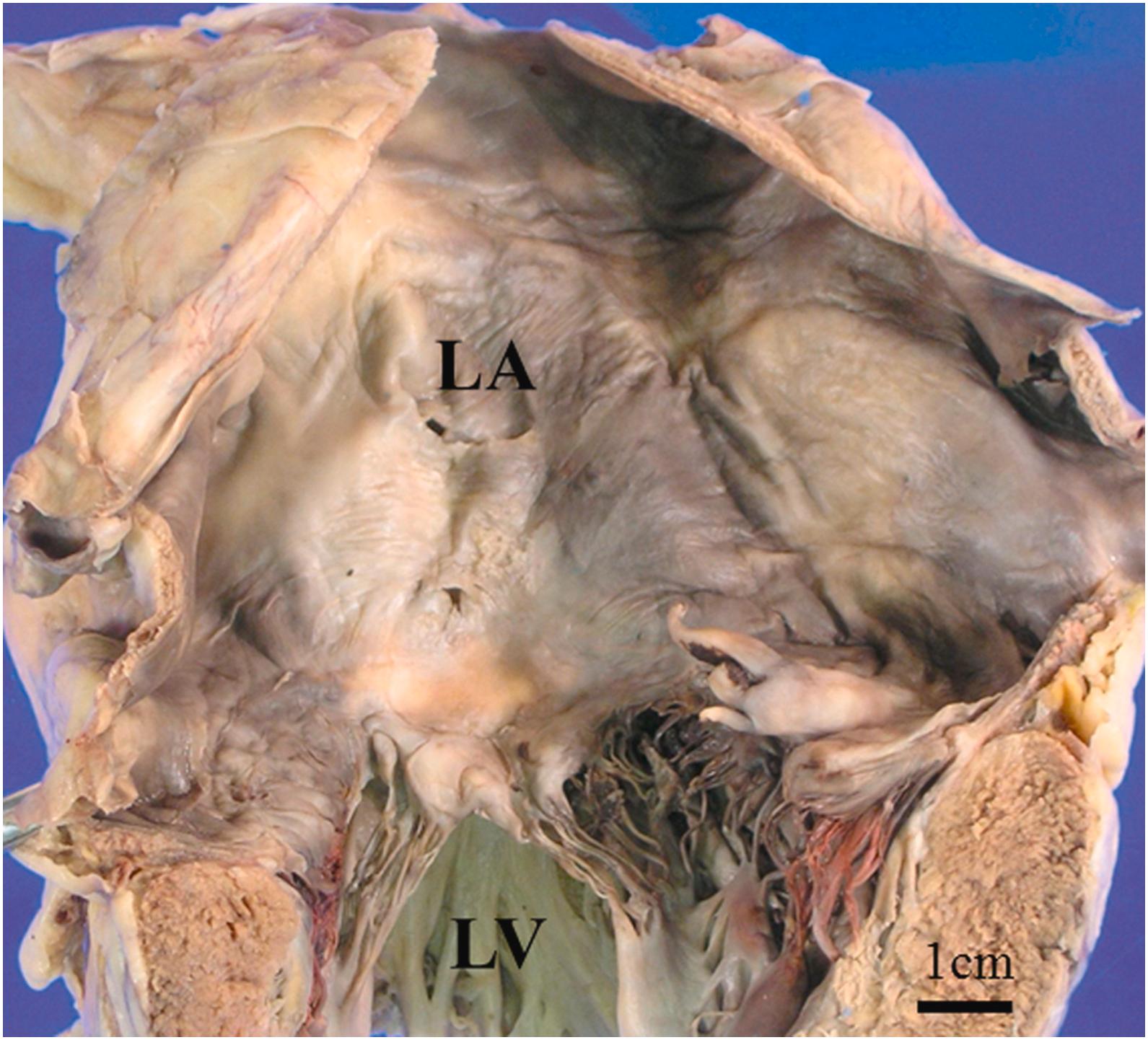
As shown in Table 3.1 , there are several common age-related gross findings in the heart. The age-related changes include brown atrophy ( Fig. 3.1 ), tortuous epicardial arteries, gelatinous changes of epicardium (serous atrophy of fat), dilated atria ( Fig. 3.2 ), smaller ventricular chambers, and dilated ascending and descending aorta .
| Atrophy | |
| Hypertrophy | |
| Increased lipofuscin (brown heart) | |
| Epicardial gelatinous changes (serous atrophy of fat) | |
| Tortuous coronary arteries | |
| Whiter, thicker endocardium | |
| Enlarged right/left atria | |
| Dilated valve annuli | |
| Septal bulge | |
| Valves | |
| Aortic valve | Cusp thickening |
| Nodules of Arantius (Noduli Arantii) | |
| Calcific deposit | |
| Lambl’s excrescences | |
| Fenestration of the cusps | |
| Mitral valve | Diffuse thickening of valve leaflets |
| Lipid deposition (anterior leaflet) | |
| Lambl’s excrescences | |
| Mucoid degeneration, hooding | |
| Mitral annular calcification | |
| Pulmonic valve | Nodules of Morgani |
| Lambl’s excrescences | |
| Fenestration of the cusps | |
The adult heart was previously considered a postmitotic organ without any capacity for regeneration. Recent reports indicate that the adult heart might be capable of cardiomyocyte turnover . The turnover of cardiomyocytes has been reported to either decrease or increase with aging. Senescence of cardiac resident stem cells has also been observed. Age-related microscopic changes include hypertrophy of cardiomyocytes, decreased number of cardiomyocytes, increased apoptosis, and increased interstitial fibrosis . In compensation for the decreased number of myocyte, myocyte size increases with age . These changes also contribute to increased passive stiffness and impaired diastolic function in the ventricles .
If there is no associated cardiovascular disease, it has been reported that total heart weight, that includes myocardium, epicardial adipose tissue, vessels, and valves, does not significantly change with aging . It is said that the heart is an exceptional organ in that the weight stays constant over life . Generally, organ weights decrease with advancing age, especially in kidney and spleen . In malnourished and cachectic elderly individuals, however, heart weight does decrease. Sawabe et al. studied the organ weights of 1615 autopsied patients that were from 60 to 99 years old . Organs with significant diseases were excluded from their study . Heart weight stayed constant over adult life in their study . Interestingly, in another autopsy study of the elderly older than 80-year old, including hearts with cardiovascular diseases, heart weights decreased with age . Certainly, the overall health status and level of activity/exercise of the individual will influence heart weight. There are several age-related changes and diseases that may increase or decrease heart weight. Normal heart weights in the elderly are summarized in Table 3.2 . Heart weights are generally greater in males than females . In the past, according to the Reginald Hudson’s Cardiovascular Pathology textbook, normal fresh heart weights in adults are 300 g on average (range 250–350 g) in male and 250 g in female (range 200–300 g) . In the study by Sawabe et al., heart weights in the Japanese elderly (age 60–99 years) were 342.0 ± 89.9 g in male and 312.3 ± 83.0 g in female . Roberts et al. considered that a heart weight, >350 g in female and >400 g in male (age 80–103 years), represented increased heart weights . However, heart weight must be normalized for body size. Since obesity is so rampant in industrialized countries, normalizing heart weight to body height is preferred . The formulas suggested by Zeek seem to give a reasonable range of heart weight based on body height (see Chapter 1: Cardiovascular Pathology: Guide to Practice and Training ).
| Age studied | Male | Female | References | ||
|---|---|---|---|---|---|
| Heart weight a | 60–99 | 342.0±89.9 g | 312.3±83.0 g | ||
| >80 | Increased if >400 g | Increased if >350 g | |||
| Valve measurement (circumference) | |||||
| Tricuspid valve | >60 | 102±10 mm | 97±9 mm | ||
| Pulmonic valve | >60 | 70±8 mm | 67±7 mm | ||
| Mitral valve | >60 | 86±8 mm | 81±11 mm | ||
| Aortic valve | >60 | 76±6 mm | 70±7 mm | ||
| Valve measurement (thickness) | |||||
| Tricuspid valve b | >60 | 0.9±0.5 mm | 0.9±0.4 mm | ||
| Pulmonic valve b | >60 | 0.5±0.3 mm | 0.4±0.3 mm | ||
| Mitral valve b | >60 | 1.3±0.5 mm | 1.3±0.6 mm | ||
| Mitral valve, closing edge of anterior leaflet c | <20 | 1.30±0.50 mm | |||
| Mitral valve, closing edge of anterior leaflet c | >=60 | 3.20±1.52 mm | |||
| Aortic valve b | >60 | 0.8±0.5 mm | 1.1±0.6 mm | ||
| Aortic valve, nodule | <20 | 0.67±0.21 mm | |||
| Aortic valve, nodule | >=60 | 1.42±0.51 mm | |||
a Heart weight should be normalized for body size, preferably body height .
b The thickeness was measured in the thickest lesion; Tricuspid valve, the thickness of the anterior tricuspid leaflet; pulmonic valve, anterior pulmonic valve; mitral valve, anterior mitral leaflet; aortic valve, noncoronary cusp.
c Closing edge of anterior leaflet, midway between middle of leaflet and posteromedial commissure.
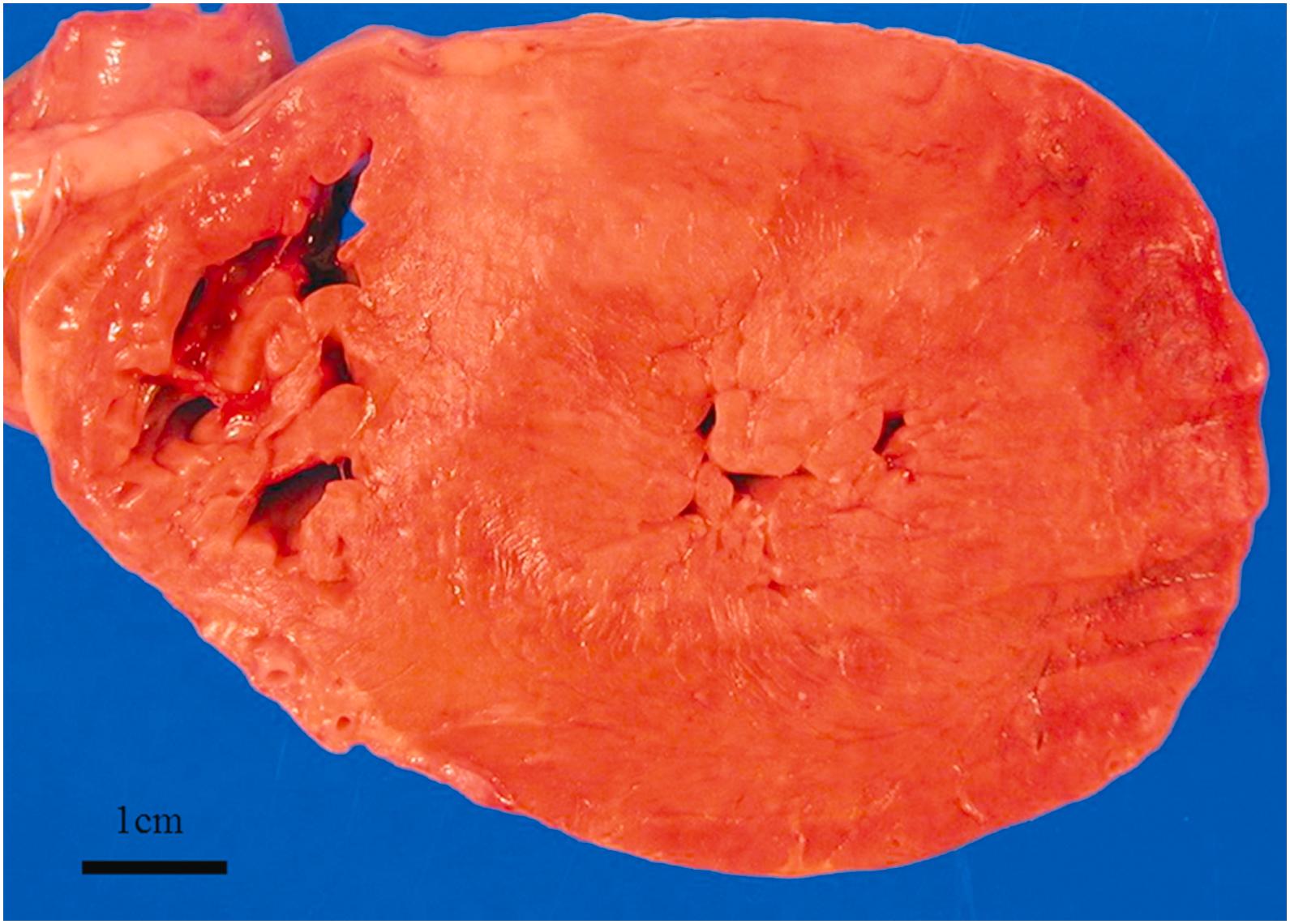
Hypertrophy and atrophy are common findings in elderly hearts. Hypertrophy is often associated with hypertension and valvular disease in the elderly ( Fig. 3.3 ). Atrophy of the heart is sometimes described as “brown atrophy.” Brown atrophy is caused by malnutrition and/or marked inactivity . Grossly, the “brown heart” is brown because of the increased deposition and concentration of lipofuscin ( Fig. 3.1 ). Lipofuscin is a brown granular material found in the cytoplasm of cardiomyocytes that consists of a collection of indigestible cellular material in lysosomes . With aging, epicardial adipose tissue significantly decreases and shows gelatinous changes, representing serous atrophy of fat. Tortuous coronary arteries are often associated with brown atrophy as the arteries need to bend as the ventricles become shorter. Microscopically, brown atrophy is characterized by atrophy of cardiomyocytes with increased lipofuscin deposition in the cytoplasm of cardiomyocytes usually in the perinuclear region . Lipofuscin increases with aging . In elderly patients with hypertension or heart failure, a greater accumulation of lipofuscin is a common finding.
The endocardium becomes whiter and thicker with age, particularly on the left side of the heart. Presumably, the endocardial thickening is due to “wear and tear” of the endothelium lining the intima. Fibroelastic hyperplasia is seen in the endocardium of the outflow surface as a result of aging . Patent foramen ovale (PFO) is a common finding in any age groups. However, the prevalence of PFO gradually decreases with increasing ages. Still, PFO is reported by some, as frequent as in 20% of individuals in their ninth decade .
Calcification is a frequent finding in the elderly heart. Cardiac calcific deposits are observed in coronary arteries, aortic valve cusps, mitral valve annulus, and left ventricular papillary muscle tips . These changes are discussed below. In one study, calcification was present in 91% of elderly hearts at autopsy . In this study, the most common site of calcific deposits was coronary arteries. More than 80% of elderly hearts had calcific deposits in atherosclerotic plaques of coronary arteries . Usually, calcium in a papillary muscle is considered as a consequence of aging, not a direct manifestation of atherosclerosis .
Basophilic degeneration of the myocardium is another microscopic age-related change in the heart ( Fig. 3.1 ) . Basophilic degeneration is characterized by a deposition of basophilic, finely granular materials in the cytoplasm of the cardiomyocytes . Basophilic degeneration is also called mucoid degeneration, mucinous degeneration, or cardiac colloid . The change is usually not seen in the first decade, but after that, it is found commonly . Histologically, the deposits are Periodic acid-Schiff positive. The cardiomyocyte with basophilic degeneration is positive for polyglucosan by immunohistochemistry, suggesting that the basophilic degeneration is a glycoprotein . The degree of basophilic degeneration also increases with aging . It is not clear if the degree of basophilic degeneration is related to that of lipofuscin . Basophilic degeneration is seen in association with hypothyroidism, but basophilic degeneration is not a specific finding for this condition.
Aging significantly affects both left ventricular and atrial function . Even in the normally aged heart, both atria tend to increase in size . Enlarged atrial chambers are common findings in the elderly hearts ( Fig. 3.2 ). The prevalence of enlarged atria increases with age . In a computed tomography (CT) study in people older than 50 years, the diameter of left atrium, left atrial appendage, and pulmonary veins significantly increased with age . Atrial fibrillation (AF), that is one of the major diseases in the elderly, also is associated with severe dilation of the atrial chambers . In a study using Doppler-echocardiography and pulsed tissue Doppler, myocardial systolic function and relaxation were decreased with age . In contrast, myocardial atrial activity and left ventricular filling pressure increased .
There are conflicting reports on changes in left ventricular volume with aging. In the Framingham Study, ventricular mass assessed by echocardiography was increased with age . However, aging also increases the prevalence of risk factors for increased left ventricular volume, such as hypertension, obesity, valvular disease, and coronary artery disease (CAD) . Dannenberg et al., from the same group of investigations as the Framingham Study, reported that left ventricular mass changed little with advancing aging, when cases with hypertension and obesity were excluded . In an autopsy study of 141 normal Japanese elderly hearts without apparent cardiovascular diseases, aged over 60 years, left ventricular volumes significantly decreased with aging . In their study, right ventricular volume and right/left atrial volumes did not change . Recent study from Japan in healthy subjects assessed left ventricular and left atrial volume ratio using three-dimensional echocardiography . Three-dimensional echocardiography could provide accurate and reliable measurements of left ventricular and atrial volumes. The authors reported that left ventricular volumes decreased with age; however, left atrial volumes increased with age .
The circumference of valve rings also changes with age . In all four cardiac valves, the annular circumferences increase . The increase is greatest in the aortic valve annulus . In addition to the increased diameter of the aortic valve, the aortic root and ascending aorta become more dilated in the elderly . The normal size of valve annuli in the elderly is listed in Table 3.2 .
“Septal Bulge” is when the base of the ventricular septum bulges prominently into the left ventricular outflow tract without left ventricular hypertrophy. Synonyms include sigmoid ventricular septum , and discrete upper septal thickening . Although asymmetric septal hypertrophy is one of the hallmarks of hypertrophic cardiomyopathy (HCM), septal bulge may also be found without the association of HCM, especially in the elderly. In a necropsy study, Goor et al. reported that there was a change in the shape of left ventricular septum with aging . In their study, the septal bulge was more common after the age of 50. The septum appeared more straight in younger adults . In the large community-based Framingham study, routine echocardiography showed that the septal bulge was more prominent in elderly individuals with elevated systolic pressure . Septal bulge has also associated with increased left ventricular systolic function, mitral annular calcification (MAC), and smaller left ventricular end-diastolic dimensions by echocardiogram . The presence of septal bulge was not associated with an increased risk of cardiovascular disease or death .
The valve cusps are uniformly translucent and thin in infants and children. As a result of aging, the valve leaflets generally become thicker, stiffer, and the annulus dilates . These changes are caused by hemodynamic factors . The degree of these changes varies between valves, and progressive aging changes are usually seen more on the left side of the heart . Degeneration of collagen fibers, calcification, and lipid deposits characterize the age-related changes in the cardiac valves.
Aortic valve cusps become white and opaque even in normal elderly hearts. Lipid deposits on the aortic surface are commonly seen. Major age-related changes in the aortic valve are an increased valve thickness by fibrosis, calcification, Lambl’s excrescences, and fenestrations ( Fig. 3.4 ).
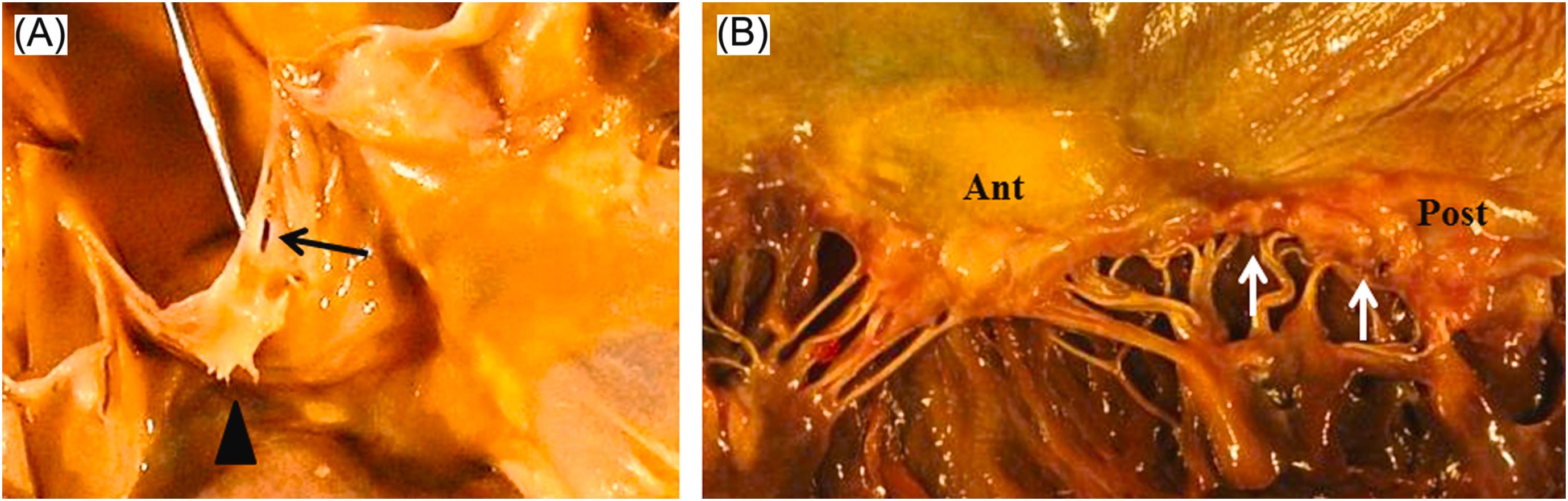
The degree of aortic valvular thickness is not related to either sex or heart weight in normal individuals . However, as we get older, the mean thickness of aortic valves generally increases . Fibrous thickening and/or calcification of the aortic valve without significant obstruction is referred to as aortic valve sclerosis (ASc). A recent meta-analysis found that the prevalence of ASc was 9% in those with a mean age of 54, and 42% in individuals with a mean age of 81 . In these studies, individuals with ASc had a 1.8%–1.9% risk per year of developing clinical aortic valve stenosis . A nodule that is sometimes found in the center of the site of coaptation, is the nodule of Arantius. Sahasakul et al. measured the thickness of aortic and mitral valves in 200 normal hearts obtained at autopsy . The thickness increased in all parts of aortic valve cusps, including closing edge, nodule of Arantius, and midportion between edge and aortic annulus . The changes in thickness became more prominent after the age of 50 . Interestingly, nodules and free edges were thicker in the noncoronary cusps compared to left- and right-coronary cusps . The thickest region was the nodule of Arantius . The nodule of Arantius normally first appeared in the noncoronary cusp . Although nodules of Arantius were seen in any aortic valve leaflets, the nodule on the noncoronary cusp was the largest . The forceful contact between valve cusps might cause local injury in the cusps, resulting in endocardial thickening. Microscopically, the nodule demonstrates a proliferation of collagen and elastic fibers in the ventricularis and fragmentation of the elastic laminae .
So-called fibroelastic hyperplasia of endocardium sometimes results in commissural adhesions in the aortic valve cusps . This hyperplasia is thought to be driven by high pressure blood flow . The prevalence of fibroelastic hyperplasia increases with age . Commissural adhesion has to be distinguished from commissural fusion that is seen in postinflammatory (rheumatic) valvular disease. In postinflammatory valvular disease, there is severe, diffuse thickening of the cusps in addition to commissural fusion.
Calcification of aortic valve cusps is a frequent finding in the elderly heart. Valvular calcification is seen in both aortic and mitral valves as a result of aging; however, the calcification is more common and appears earlier in the aortic valve than in the mitral valve . Calcification occurs first on the aortic side of the valve cusps . The free edges of the valves are usually spared from calcific deposits . The aortic valve cusps, especially the aortic side of the cusps, are chronically exposed to shear stress causing injury to the surface endothelium . In severe cases, the calcification may completely occupy the sinus of Valsalva. About 50% of elderly hearts have calcific deposits in the aortic valve cusps at autopsy . In one study, 10% of total necropsy cases had severe enough calcification to cause AS . However, not all the calcification results in AS. When calcification is prominent but not causing increased gradient across the valve, the term ASc applies. Microscopically, calcification is found first in the valve fibrosa , but eventually is full thickness, with erosion through the surface of the leaflet.
Calcification is also found in the aortic annulus. Calcification in the aortic annulus is significantly associated with atherosclerotic calcification in vascular territories, including coronary arteries, thoracic/abdominal aorta, carotid arteries, and iliac arteries . The risk factors for calcification in the aortic annulus are the same as those of atherosclerosis, and include aging, smoking, and hypertension .
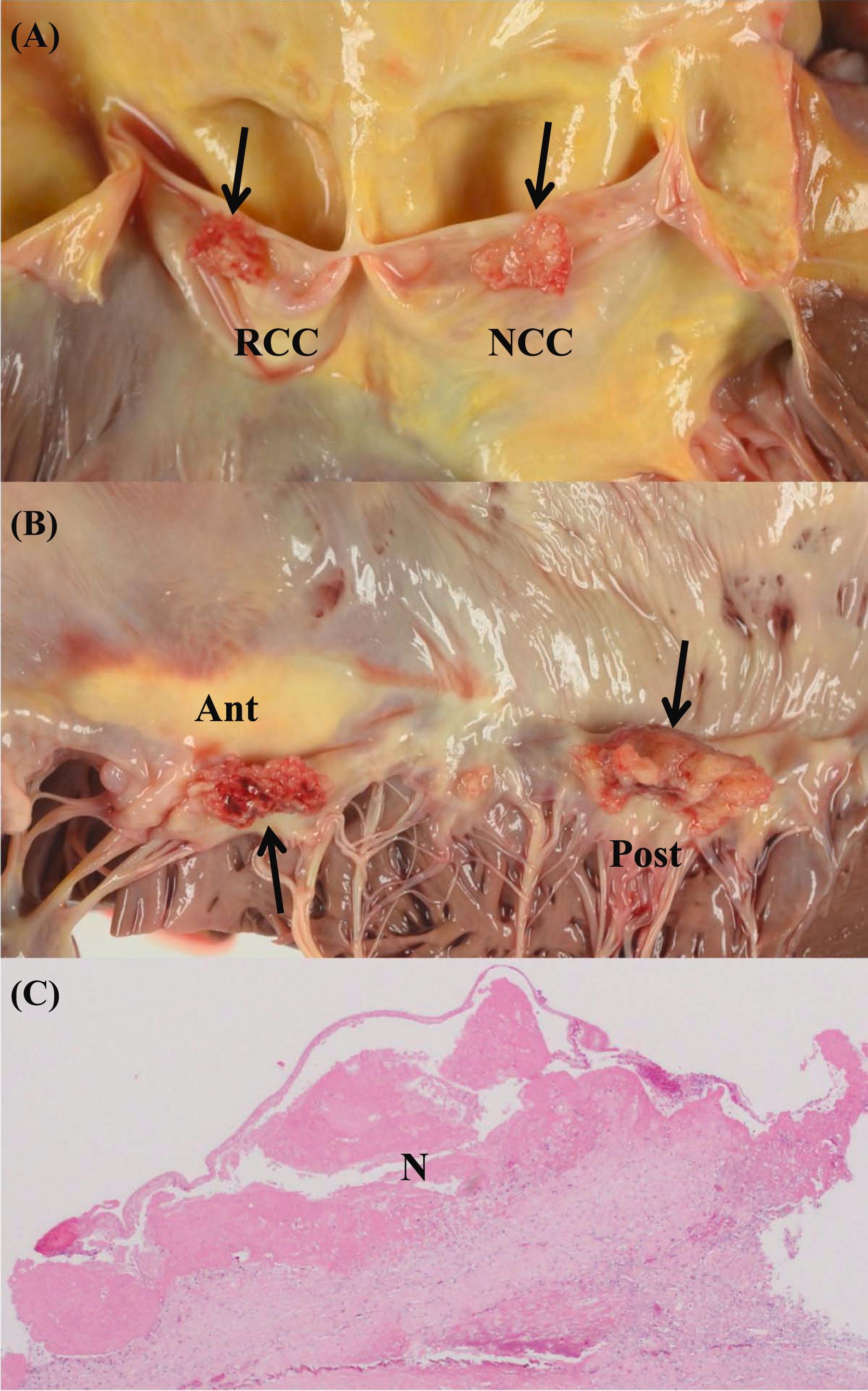
Lambl’s excrescence is a valvular strand, that is mobile, thin, thread-like/papillary, and has a white/translucent structure ( Fig. 3.4 ) . Lambl of Prague first described these excrescence in 1856 . Lambl’s excrescence is a common finding on the aortic valve, especially in the elderly heart. The prevalence increases with age . Aortic and mitral valves may both be involved. Lambl’s excrescence is rarely found on the tricuspid or pulmonic valves . Lambl’s excrescence is more commonly seen when valves are abnormally thick . The pathogenesis is unclear, but it is believed that high pressure blood flow has a role in the progression . Grossly, there are two kinds of Lambl’s excrescences; filiform or lamellar . The filiform type of Lambl’s excrescences is mainly present on the nodule of Arantius and free margin of aortic valve cusps . The filiform type is thicker in the bottom, and thinner in the tip, and sometimes there is a branching . The lamellar type has a flattened structure . The filiform type of Lambl’s excrescences in the free margin is considered to be an age-related change . Lambl’s excrescence is more common on the ventricular side of the aortic cusps. The size ranges from less than 1 mm to 10 mm. The number of Lambl’s excrescences varies between individuals . Microscopically, the central core of Lambl’s excrescences consists of elastic fibers and collagen, and is often hypocellular . The central core is covered by a single layer of endothelial cells that sometimes appear hyperplastic . The endothelial layer is continuous with the valvular endothelium . Clinically, Lambl’s excrescences may be associated with systemic embolism . However, it is unclear whether Lambl’s excrescence is actually a risk factor for embolic events .
Fenestration is a window-like defect in the valve cusps, that is found above the closure lines and adjacent to the commissures of cusps ( Fig. 3.4 ) . The incidence of fenestrations increase with aging . Fenestration is also observed in young adults. However, the frequency is less in the young. Fenestration was found in 12% of patients under 45 years old . Since fenestrations occur above the line of closure, they are of no significance.
Transcutaneous aortic valve replacement (TAVR) use has increased in recent years for elderly patients with severe degenerative aortic stenosis. TAVR will be discussed later in Section “Degenerative (calcific) aortic stenosis.”
The mean thickness of the mitral valve leaflets significantly increases with aging, but there is no relation to sex or heart weight . Some mitral valve thickening with age is considered normal ( Fig. 3.4 ) . Increase in mitral valve thickness is most prominent in the fifth and sixth decades . The mitral valve may become twice as thick in the 60s as it is in the 20–50s . The anterior leaflet is usually thicker than the posterior, especially along the line of apposition . In addition to the diffusely increased mitral valve thickness, a nodular thickening sometimes occurs on the atrial surface of anterior leaflet . The nodular thickening is usually found along the line of contact with the posterior leaflet . In one study, the nodular thickening was observed in all adults . Microscopically, there are localized plaques of collagen and elastic fibers in the atrialis layer .
Lipid deposition is also an age-related change in the normal mitral valve ( Fig. 3.4 ) . The lipid deposition is sometimes referred to as “atheromatosis” , although it is questionable if this term is appropriate. The lipid deposition is usually found in the anterior leaflet. There is a significant correlation between “atheromatosis” and aging. There is no difference between sexes . The lipid deposition in the anterior leaflet is observed in more than two-thirds of individuals after the age of 45 . In the sixth decade of life, almost all individuals have lipid deposition in their mitral valve . “Atheromatosis” does not correlate with atherosclerosis in the coronary arteries or with other aortic valve changes, hence, the term may be inappropriate . Diffuse opacity is a common finding in the mitral posterior leaflet, especially in females . The diffuse opacity histologically consists of mild fibroelastic proliferation in the atrialis.
Lambl’s excrescence is also found in the native mitral valve and less commonly even in bioprosthetic mitral valves . The morphology is similar to that of the aortic valve, and shows a mobile, thin, thread-like structure. Lambl’s excrescence is usually found on the atrial side of the mitral valve leaflets, near the closure line. Lambl’s excrescence can occur on both anterior and posterior leaflets.
Mucoid or myxomatous degeneration of mitral valve leaflets is common in the elderly. When the degeneration becomes severe and causes an excursion of one or two mitral valve leaflets above the mitral annulus during systole, it is called mitral valve prolapse (MVP) . MVP is discussed later. However, some mild myxoamtous changes may be associated with aging in the normal elderly heart. Myxomatous degeneration is more common in the posterior leaflet of the mitral valve . Myxomatous degeneration of the mitral valve is the pathologic finding associated with clinical MVP . Anatomically, prolapsed mitral valves were found in 17% of female and 13% of male at necropsy of those aged >90 years . When the mitral leaflet is thickened and opaque, and shows “ballooning,” the leaflet sometimes resembles a parachute ; a phenomenon described as hooding ( Fig. 3.4 ). With the hooding or ballooning of leaflets, the chordae tendinae become attenuated and the valve leaks . Ruptures of chordae tendineae may occur resulting sometimes in severe, acute mitral regurgitation. Microscopically, there is an increase of extracellular matrix in the spongiosa, with myxoid tissue infiltration in the fibrosa, with separation and fragmentation of collagen fibers .
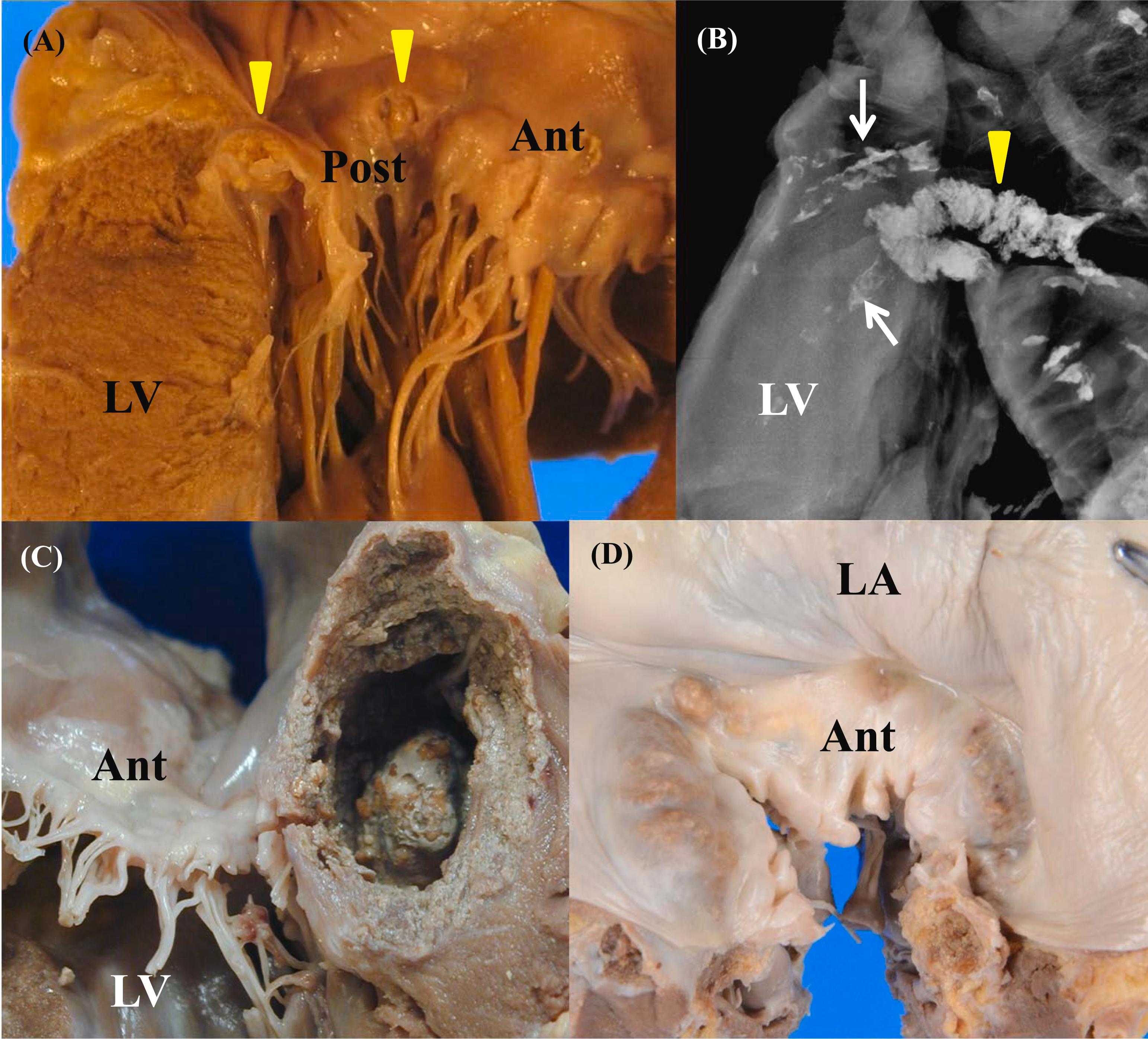
MAC is one of the common changes occurring with aging in the mitral valve ( Fig. 3.6 ). MAC is also called mitral ring calcification. Radiographically, MAC demonstrates J-, C-, U-, or O- shapes . MAC is usually located in or below the posterior mitral annulus, and between the posterior mitral leaflet and posterior left ventricular wall . The mass of calcification sometimes ulcerates through the medial commissure . Histologically, in addition to the calcification, nonspecific chronic inflammation and foreign body giant cells are sometimes observed . MAC is usually not observed in the young , unless there is connective tissue disease. The incidence of MAC significantly increases with age . In autopsy studies, MAC was found in 39% of those aged >80 years , and in 43% of those aged >90 years . MAC is more common in males than females before the sixth decade of life . After the age of 65, the occurrence of MAC becomes much higher in female . In fact the frequency of MAC is almost twice as great in the female aged older than 90 years old than in males of similar age . The complications of MAC include endocarditis and conduction abnormalities, namely heart blocks . In an echocardiographic study, MAC with caseous necrosis was associated with previous stroke . As MAC enlarges, MAC often extends into the fibrous tissue of the mitral valve leaflets, and in the fibrous continuity of mitral and aortic valves, and the myocardium . About one-third of MAC cases have very heavy calcific deposits . MAC may ulcerate and become infected, or cause arrhythmia, conduction disturbance, and even mitral stenosis . In the case of atrioventricular (AV) block due to MAC, MAC microscopically invades the mitral posterior commissure, ventricular septum, and into the bundle of His. There is a significant association between MAC and atherosclerotic calcification in the carotid arteries and thoracic/abdominal aorta . Although the calcification in the aortic annulus has been associated with atherosclerotic calcification, MAC is not associated with atherosclerotic calcification in the coronary and iliac arteries .
Transcatheter mitral valve replacement using balloon-expandable aortic transcatheter valves is a potential, less invasive treatment option for patients with high surgical risk . Accordingly, this treatment modality is of great utility in the elderly. The frequent complications include paravalvular leak, left circumflex coronary artery injury, and AV groove rupture .
The pulmonic valve sometimes remains translucent and thin even in the elderly . However, the cusps may become white with aging. With the presence of pulmonary hypertension, valve cusps shift downward into the right ventricular outflow tract, and atherosclerosis is found in the large pulmonary arteries.
Nodular thickening is sometimes found on the ventricular surface in the centers of cusp edges, called nodules of Morgani . The incidence of nodules of Morgani increases with age . Note, the same nodule in the aortic valve is called the nodule of Arantini. Histologically, these nodules show the same morphological changes; a nodular proliferation of elastic and collagen fibers . The nodule of Morgani is not as common as the nodule of Arantini .
Lambl’s excrescences and fenestration of the valve cusps are also observed in the valve. The fenestration of cusps is found in less than 10% of valves, much less often than in the aortic valve.
The tricuspid valve is usually thin and even translucent in the elderly . The tricuspid valve may show nodular thickening and myxomatous change. In the presence of a pacemaker lead, often present in elderly hearts, the tricuspid valve becomes thickened and opaque. The lead may be adherent to, or pass through, a leaflet of the tricuspid valve. Prolapse of the tricuspid valve may be found in individuals with prolapsed mitral valves. The frequency of tricuspid valve myxoid change is around 10% in those older than 90 .
Jet lesions are focal fibrous reactions in the heart or blood vessels that are the results of abnormal jet-like streams of blood . These are usually associated with structural heart disease, especially stenotic valvular diseases . Since valvular heart diseases are common in the elderly, jet lesions are often observed in the elderly heart . Grossly, the jet lesions are sites of focal fibrous thickening of the endocardium . Microscopically, jet lesions are composed primarily of collagen fibers. Some old jet lesions contain elastic fibers as well .
An aging myocardium possesses significant electrophysiological alterations. The prevalence rates of conduction disturbances, arrhythmias, and incidence of sudden cardiac death increase with age . The rate of sudden cardiac death has not significantly declined over the past 30 years . The major components of the conduction system of the heart include the sinoatrial (SA) node, AV node, His bundle, and bundle branches. Moreover, electrophysiological properties of the cardiac myocytes are regulated by the cardiac autonomic nervous system (ANS). Dysfunction of both cardiac conduction and ANS is known to play a role in the pathogenesis of sudden cardiac death . Indeed, more and more investigators are studying the relationship between the ANS and cardiovascular disease, expanding the field of “neurocardiology” (see Section “Cardiac autonomic nervous system”).
Age-related changes are also observed in the SA node. Major changes in the SA node include a decline in the number of nodal cells, increase in mature adipose cells, and fibrosis ( Fig. 3.7 ). In individuals in their 30s, the percentage of nodal cells is 20%–30% . The area of nodal cells declines, and the percentage of nodal cells becomes less than 10% in centenarians. In addition, the expression of calcium channels decreases with increased age . Fatty infiltration begins in the SA node in the third decade of life . Increases of collagen, reticular, and elastic fibers are common findings in the SA node of the elderly and these tissues may eventually replace the majority of conduction cells. Although, the increase of elastic fiber continues throughout life, collagen fiber shows a minimum increase after the fourth decade . Trichrome-EVG stain highlights the elastic and collagen fibers between conduction nodal cells. As we get older, fatty infiltration and elastic/reticular networks become more prominent .
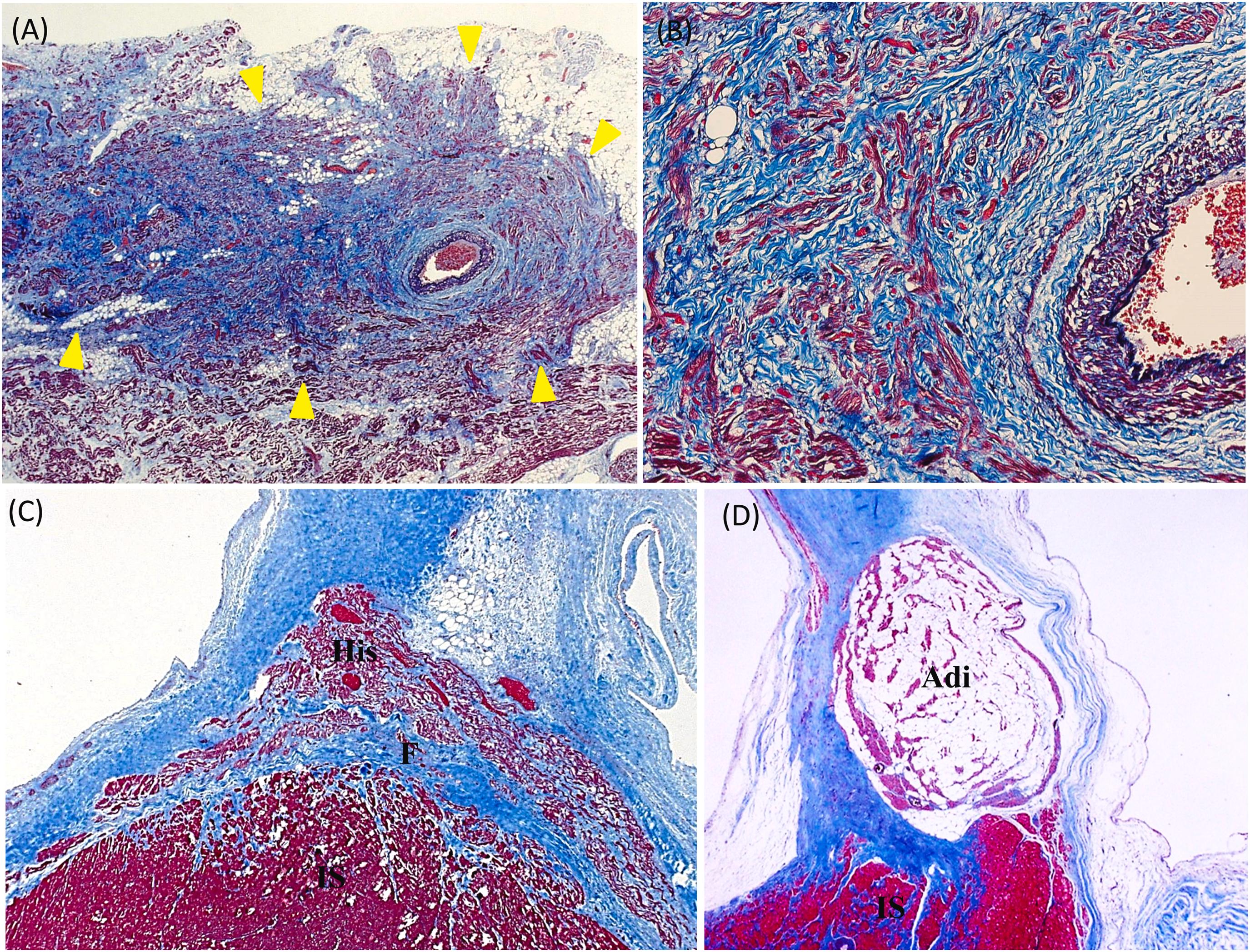
Clinically, SA node dysfunction progressively increases with age . The changes in the SA node are often associated with a decline in pacemaker function, such as sick sinus syndrome (SSS) . Pathologically, marked fibrosis and fatty infiltration in the SA node are frequently observed in patients with pacemaker placement for SSS .
The common age-related changes of fatty infiltration and increase of elastic fibers also occur in the AV node ( Fig. 3.7 ) . By the 6–7th decades, fibrosis is almost always found in the summit of the muscular ventricular septum adjacent to the central fibrous body . Calcification is sometimes seen in the central fibrous body as well . However, usually these changes in the AV node are not extensive enough to cause clinical dysfunction .
Electrophysiological properties of cardiac myocytes are regulated by the ANS . There are both extrinsic and intrinsic components of the cardiac ANS. The ANS plays an important role in maintaining normal cardiac rhythm and rate . The balance of sympathetic and parasympathetic tone regulates cardiac autonomic control . The aging myocardium exhibits a decreased β-adrenoreceptor response, β-adrenoreceptor desensitization, and a reduced β-adrenoreceptor density. Accordingly, heart rate, cardiac output, and vasodilation decreases with age . Aging of the ANS also causes the electrophysiological disturbances that lead to arrhythmias . Cardiovascular abnormalities, including hypertension, heart failure, and CAD are associated with an imbalanced cardiac ANS .
Orthostatic hypotension is a common clinical finding in older adults . Causes of orthostatic hypotension include a reduced heart rate response to upright posture, sluggish carotid baroreceptor reflex, and loss of arterial compliance . An association between orthostatic hypotension and death from cardiovascular disease was previously reported . Systolic orthostatic hypotension increases the risk of cardiovascular death in hypertensive adults with diabetes mellitus . Diastolic orthostatic hypotension measured 1 minute after standing up and systolic orthostatic hypotension measured 3 minutes after standing up predict higher vascular mortality in the home-dwelling elderly .
Atherosclerosis is the most common age-related change and disease in elastic and large muscular arteries. Almost all adults have some atherosclerosis in their arteries. Atherosclerosis is characterized by the formation of “plaques” in the artery wall, composed of a variable mixture of lipid, inflammatory cells, red blood cells, and connective tissue. Atherosclerosis is associated with decreased compliance of the artery, increased stiffness, and obstruction of luminal blood flow . In addition, atherosclerosis causes morbidity and mortality worldwide, including myocardial infarction (MI), peripheral vascular diseases, and stroke.
Aging is a strong risk factor for atherosclerosis . This relationship is typically observed in the aorta . Vascular disease, such as peripheral artery disease, carotid artery stenosis, and abdominal aortic aneurysm, are all strongly associated with advanced age . The prevalence of all vascular disease is 2% in those aged 40–50 years old. The rate gradually increases with age. One-third of patients aged 90–100 years old have at least one peripheral vascular disease . Other major risk factors for atherosclerosis include hypercholesterolemia and hypertension . However, approximately half of the patients with subclinical atherosclerosis do not have these traditional risk factors .
Initiation of atherosclerosis involves the response to multifactorial, repetitive chemical and hemodynamic injury, and endothelial cell dysfunction. One aspect of endothelial cell dysfunction is an abnormality in the production of endothelial-derived nitric oxide and resultant deleterious changes in vascular reactivity . In addition, inflammation and immunity play a significant role in the progression and complications of atherosclerosis . Both acute and chronic infections may promote atherosclerosis on a chronic ongoing or episodic basis . Circulating monocytes in the blood are recruited into the intima, and monocytes differentiate into macrophages and become form cells, which are the hallmark of early fatty streak lesions . Macrophages are the predominant component of inflammation in the intima of atherosclerotic lesions . Activated macrophages and endothelium produce multiple chemokines and growth factors, which act on smooth muscle cells to induce their proliferation and synthesis of extracellular matrix components, generating fibromuscular plaques . Cholesterol and lipoproteins are one of the key mechanisms to establish fatty lesions in arteries . Lipoproteins are oxidized, which initiates the inflammation in atherosclerotic plaques .
A single layer of endothelial cells overlies the internal elastic lamina . Tunica intima thickens in all arteries with age . Intimal thickening consists of variable layers of smooth muscle cells, and the accumulation of extracellular matrix . Smooth muscle cells migrate from the tunica media into the tunica intima, where the smooth muscle cells change their phenotype and response to growth factors, resulting in vascular remodeling .
Precursor lesions for atherosclerosis include fatty streaks and intimal thickening. Fatty streaks are most commonly found in the posterior midline of the aorta . Fatty streaks are flat or slightly elevated lesions in the intima, containing a large number of lipid-laden macrophages . The deposition of cholesterol, phospholipids, and foamy macrophages results in lipid accumulation, and a necrotic core is subsequently formed . In contrast to fatty streaks, plaques are more often on the anterior and lateral walls of the aorta . As atherosclerotic plaques develop, there is also calcification and an increase in glycosaminoglycans . Advanced atherosclerotic plaques are also associated with fibrosis, calcification, and inflammatory reactions .
Recently, clonal hematopoiesis of indeterminate potential (CHIP) was found to have a strong association with atherosclerotic cardiovascular disease . CHIP is characterized by a clonal population of hematopoietic cells bearing one or more somatic mutations in otherwise healthy adults. CHIP is associated with an increased risk of all-cause mortality , and is more commonly found in the elderly . Atherosclerosis is discussed more in detail in Chapter 7 , Vascular Pathobiology: Atherosclerosis and Large Vessel Disease.
There are age-related structural and functional changes in the aorta, including an increased diameter of large arteries, dilation of valve annuli, vascular stiffness, thickened intima, and endothelial dysfunction . Dilation is observed in the aortic root , ascending aorta , thoracic aorta, and abdominal aorta . The greatest difference in diameter occurs in the ascending aorta, and the greatest difference in aortic stiffness occurs in the abdominal aorta . Dilation and stiffness may result in increased systolic and pulse pressure, which are risk factors for developing atherosclerosis, hypertension, and stroke . Pulse wave velocity, a measure of arterial stiffness, generally increases with age even in healthy people . Fibrous intimal thickening is the most dominant in the abdominal aorta . The thickened fibrous intima is infiltrated by mesenchymal cells . With age, elastin content decreases and medial collagen increases in the aorta . The elastic fibers become fragmented. Smooth muscle cells in the media decrease in number . Intimal thickening of the large arteries is associated with luminal dilatation, reduced compliance, and increased stiffness . Dilation of the aortic annulus may result in aortic regurgitation (AR) . Other causes of aortic root dilation include syphilis, ankylosing spondylitis, and a number of connective tissue diseases.
Hypertension is one of the most common cardiovascular disease in the elderly. The prevalence of hypertension increases with age, and is 74% in people older than 80 years old . It is estimated that hypertension will affect one-third of the population by 2025 . The underlying mechanisms of hypertension include mechanical hemodynamic changes, arterial stiffness, neurohormonal and autonomic dysregulation, and the aging kidney . Although hypertension is a risk factor for atherosclerosis, hypertension itself has not always been associated with dilation of the aorta .
Age-related changes/diseases in the coronary arteries include atherosclerosis, calcification, and tortuosity of coronary arteries. Atherosclerosis in coronary arteries is often found earlier than that in cerebral arteries . Atherosclerosis and calcification are very common findings in coronary arteries among the elderly. In addition, tortuosity of coronary arteries is frequently found in the elderly heart. The tortuosity of coronary arteries increases with age . The caliber of the coronary arteries does not change with age in normal adult hearts , but coronary arteries become longer with increased age . Interestingly, the prevalence of the right coronary dominance significantly increases with advanced age . The implication is that a left dominant pattern is somehow associated with a decreased life expectancy for unknown reasons.
The presence of atherosclerosis itself does not mean that the patient has clinically significant CAD. When atherosclerosis becomes severe and causes stenosis of coronary arteries, myocardial ischemia may result. Typically, stenosis greater than 75% is said to be clinically significant. However, for a variety of factors, even greater degrees of stenosis may not diminish blood flow. In a coronary artery, intimal thickening starts to develop along the bifurcations . The intimal thickening is usually eccentric. Within the thickened intima, there are accumulations of foamy macrophages, and extracellular lipids . In an advanced atherosclerotic lesion, a fibrous cap covers a large necrotic core . Intimal calcification is often associated with advanced atherosclerosis. Calcific deposits in coronary arteries are associated with risk factors for ischemic events such as hypertension, diabetes, hypercholesterolemia, and obesity . Therefore the presence and degree of coronary artery calcification are strong and independent predictors of CAD both in younger adults and the elderly .
In the elderly, calcification is sometimes found even without significant CAD. The amount of calcification increases with age. A computer tomography study demonstrated that calcium deposits were nearly universally present in elderly individuals . The degree of calcium detected in coronary artery increases with age. There is a significant correlation between age and the prevalence of calcific deposits both in male and female . In individuals that are older than 60 years, calcification may be present in the absence of coronary risk factors . Calcification of coronary arteries is more frequently found in males . However, females show an increase of calcific deposits in coronary arteries after the age of 60 . Calcific deposits were found in 93% of male and 75% of female among those older than 70 years . An autopsy study of patients older than age 90 reported that coronary calcific deposits were present at some site in almost all the individuals .
In addition to conventional calcification in coronary arteries, there is sometimes calcification in the internal elastic lamina . With aging, a disruption and fragmentation of the internal elastic lamina and calcification increase. Internal elastic lamina calcification is usually seen in older patients, in coronary, temporal, orbital, and other arteries .
The caliber of normal coronary arteries typically does not change in normal hearts over adult life . Dodge et al. studied the change in the size of coronary arteries using arteriography in 83 patients without atherosclerotic disease . They compared the sum of cross sectional area of right, left anterior descending and left circumflex coronary arteries, and found that there was no age-related change in the size of coronary arteries when normalized to body surface area . The diameter of the coronary arteries was smaller in females than in males . Coronary artery caliber did increase linearly with heart weight . Coronary lumen diameter is greater in patients with dilated cardiomyopathy (DCM) or left ventricular hypertrophy . The volume of blood flow that is required by the myocardium may to some degree dictate the size of the coronary arteries . The presence of tortuosity does not influence the caliber of the coronary artery .
Ultrasonography is often used to assess atherosclerosis in carotid arteries, including lumen diameter, intima-media thickness ratio, and the presence/extent of atherosclerotic plaques . The carotid intima-media thickness is regarded as an indicator of atherosclerosis . The intima-media thickness in carotid arteries is known to have an association with the prevalence of cardiovascular disease . In the elderly, the carotid intima-media thickness correlates with the future incidence of MI and stroke . This association is independent of age, sex, or history of MI or stroke . The carotid intima-media thickness does not reflect the presence of carotid artery stenosis nor blood flow in the carotid artery being evaluated .
Even patients with minimal carotid atherosclerosis sometimes show CAD and MI . One consecutive autopsy study of the elderly showed that even patients with minimal carotid atherosclerosis may have severe CAD and MI . These patients often had higher serum total cholesterol and a more frequent history of diabetes mellitus and smoking .
Morphologically, fibromuscular plaques decrease and atherosclerotic plaques increase with advancing age . In older patients, carotid plaques contain a larger atherosclerotic plaque. There are less smooth muscle cells and more macrophages . Among patients with carotid stenosis, no relationship is found between age and calcification in the carotid arteries .
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here