Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Acanthamoeba are a genera of free-living amebae that can cause localized infection of the central nervous system (CNS), respiratory tract, skin, eyes, and disseminated disease. Nearly 25 named species and at least 20 genotypes of Acanthamoeba have been identified worldwide. Previously, Acanthamoeba were distinguished based on morphologic features into 3 groups (I, II, III); however, molecular techniques have led to distinguishing isolates based on genotyping of the small-subunit ribosomal RNA gene (18s rRNA). , Of more than 20 genotypes identified, the T4 genotype, corresponding to the A castellanii complex, is most frequently associated with human disease. ,
The organism has two phases in its life cycle: a motile trophozoite phase and a resilient infective cyst stage ( Fig. 270.1 ). Trophozoites are 15–45 μm in diameter and have distinctive slender, spine-like projections of the plasma membrane called acanthopodia ( Fig. 270.2 ). The cysts have a double-layered wall and are 10–25 μm in diameter. Both forms have a wide distribution in nature.
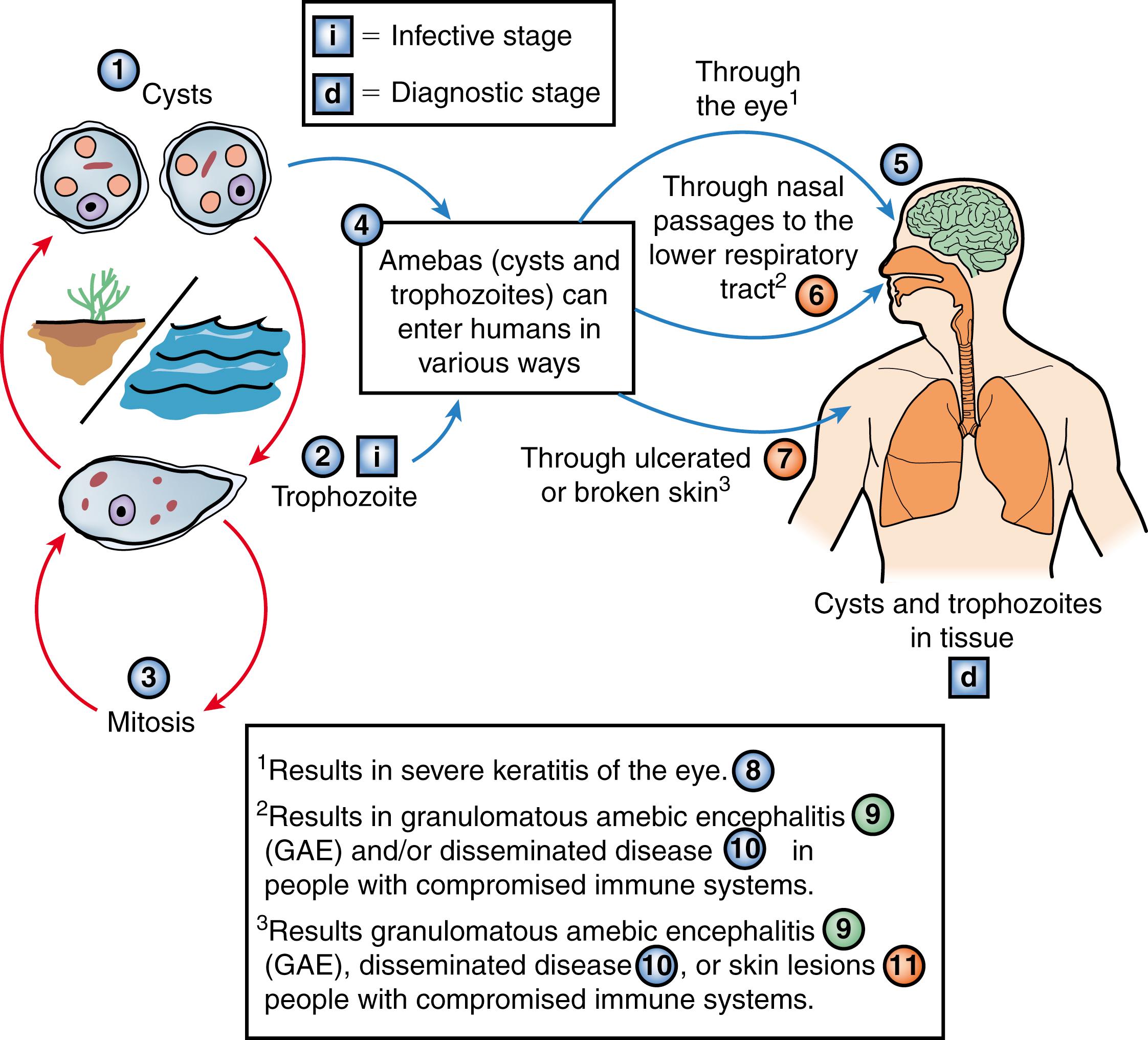
Acanthamoeba species have been isolated readily from water (including treated water; brackish water; seawater; swimming pools; sewage; domestic tap water; bottled mineral water; cooling, heating, and humidifying units; hot tubs; hospitals and dialysis units; dental irrigation units; eyewash stations; and contact lenses and associated paraphernalia), soil, dust, air, and sediments, and as contaminants in cell cultures. ,
Limited seroprevalence surveys of healthy people have demonstrated antibodies from immunoglobulin (Ig) A, IgM, and IgG classes to Acanthamoeba species, with highly variable prevalence (3%–100%) based on population, a finding indicating that asymptomatic infections likely occur.
Patients with compromised immune systems are at risk for severe manifestations of Acanthamoeba infections such as disseminated disease and granulomatous amebic encephalitis. Cases have been reported in association with diabetes mellitus, chronic liver disease, renal failure, solid organ transplantation, hematopoietic stem cell transplantation, chemotherapy, steroid use, and human immunodeficiency virus (HIV) infection. Difficulties in diagnosing amebic encephalitis limits the ability to capture the true incidence of disease. Although CNS disease with Acanthamoeba occurs predominantly in immunocompromised people, cases have been reported in both adults and children with intact immune systems. ,
Amebic keratitis most commonly occurs in immunocompetent contact lens wearers. In a multicenter review of 116 cases in the United States, 93% of cases were associated with contact lens use. In non-lens wearers, it is often associated with trauma and environmental contamination. The estimated annual incidence ranges from 1.7 to 19.5 cases per million contact lens wearers, with significant regional variation. Previous outbreaks were related to the use of nonsterile, homemade saline solution in the 1980s and the use of a commercial contact lens solution in 2007 that was later recalled. After 2007, cases of amebic keratitis decreased, but not to pre-outbreak levels. Factors for this are not well understood, but the ubiquity of contact lens use, poor lens hygiene, water contamination, and insufficient amebicidal activity of most disinfectant solutions are likely contributors.
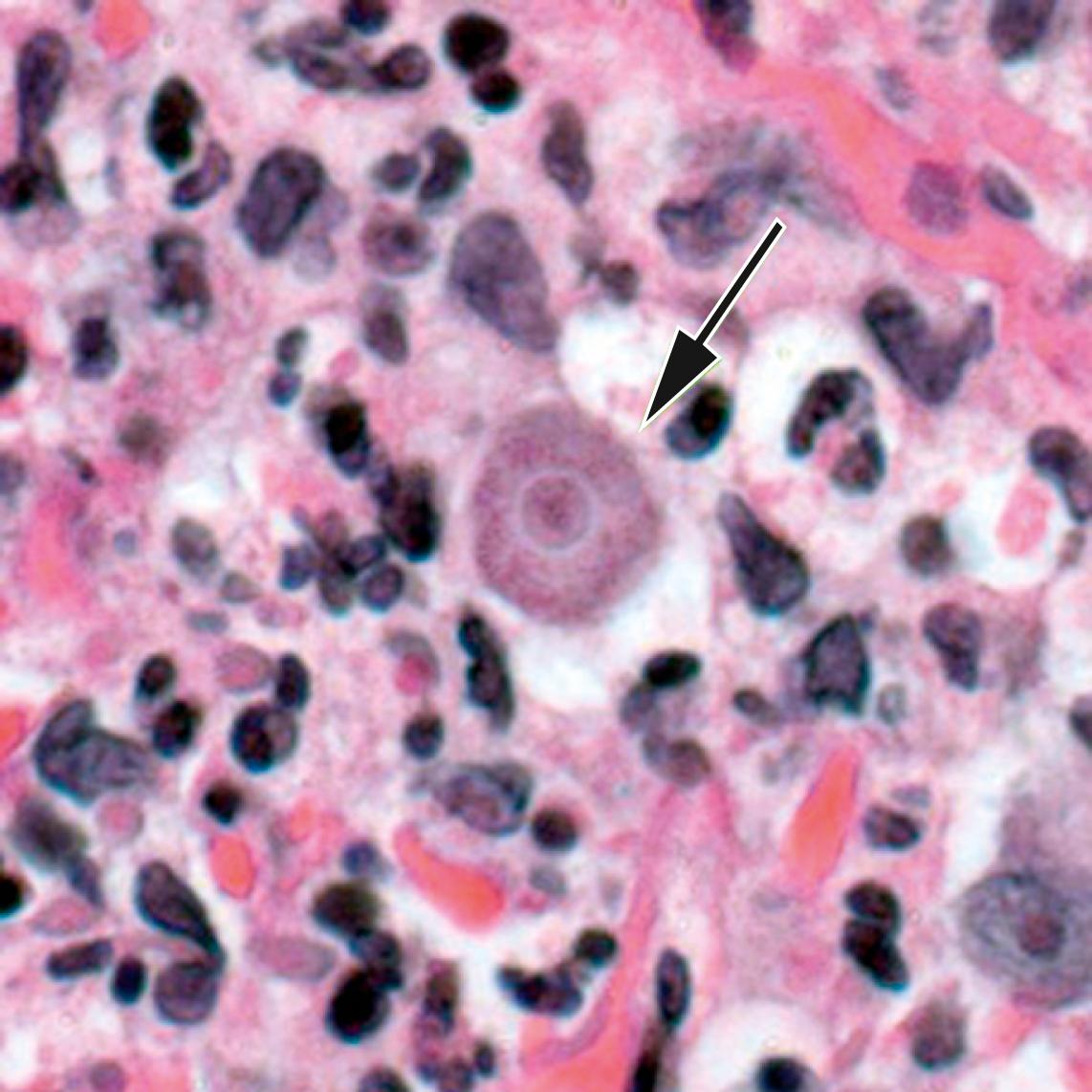
Acanthamebic encephalitis usually is termed granulomatous amebic encephalitis because of its pathologic appearance. The disease generally occurs after inoculation at other sites, thus implying that trophozoites reach the CNS by hematogenous dissemination. The route of infection is thought to be by inhalation of amebae through the nasal passages and lungs or introduction through skin lesions. Although the incubation period for Acanthamoeba infections is unknown, it can be several weeks or months. The neurologic symptoms of granulomatous amebic encephalitis have an insidious onset and include focal deficits such as hemiparesis, diplopia, cranial nerve palsies (especially of cranial nerves III and VI), and cerebellar ataxia. Non-focal neurologic manifestations include personality changes, seizures, headache, meningismus, nausea, stiff neck, lethargy, altered mental status, signs of increased intracranial pressure, and coma. Low-grade intermittent fever, nausea, and vomiting also can occur. The disease is usually fatal.
Cutaneous infections are usually reflective of disseminated disease caused by Acanthamoeba and are most common in patients with underlying immunocompromise but can occur in the absence of CNS involvement. Primary cutaneous acanthamoebiasis generally has a more favorable prognosis. The cutaneous form of the disease most commonly presents as erythematous or violaceous nodules or ulceronecrotic lesions. , Typically, early nodular lesions enlarge, suppurate, and ulcerate. Additional presentations including papules, pustules, plaques, cellulitis, and abscess have also been described, and may depend on underlying immune status. ,
Other manifestations of Acanthamoeba infection include localized infection of the sinuses, reflective of its likely portal of entry. Osteomyelitis and involvement of visceral organs also have been reported in the context of disseminated disease.
Balamuthia mandrillaris , a free-living soil ameba first identified in 1986, causes a similar spectrum of disease to Acanthamoeba (granulomatous amebic encephalitis, cutaneous, and sinus infections) and can affect both immunocompromised and immunocompetent patients. , Hispanic Americans account for a disproportionate number of cases. , Disease caused by Balamuthia can be difficult to distinguish from Acanthamoeba based on epidemiologic factors, clinical presentation, and morphology on light microscopy, but can be aided by immunofluorescence or PCR testing.
Balamuthia has been transmitted through solid organ transplantation, unlike Acanthamoeba , for which no cases of donor-derived infection have been reported to date. ,
A single case of encephalitis has been reported to be caused by another free-living ameba, Sappinia species ( diploidea/pedata ). ,
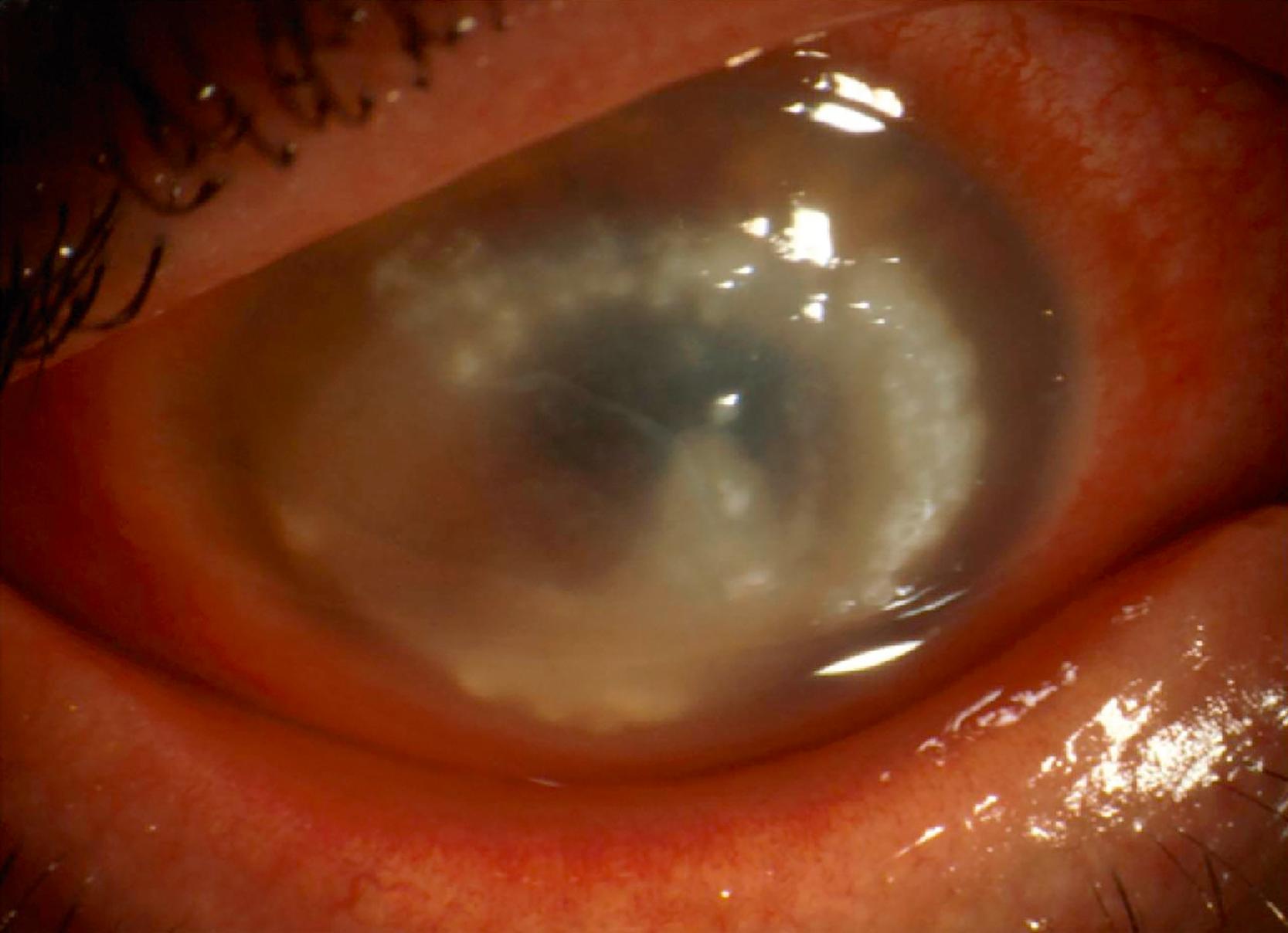
Symptoms of Acanthamoeba keratitis often begin with the sensation of a foreign body in the eye, followed by severe (usually unilateral) ocular pain, redness, photophobia, tearing, blurred vision, and eyelid edema. Clinical findings during early disease (within the first 4 weeks) are characterized as epitheliopathy, often with a dendritiform appearance. This is followed by development of stromal infiltrates and often a characteristic ring infiltrate, with perineural infiltrates highly suggestive of disease. Later disease (after 4 weeks) is characterized by progression with stromal infiltrates, ring infiltrate, ulceration, and secondary sterile anterior uveitis, sometimes with hypopyon ( Fig. 270.3 ). Limbitis can be seen in both early and late disease. Waxing and waning of symptoms and physical signs are common and often result in delayed diagnosis, and bacterial superinfection is not uncommon. , Blindness can result if disease progresses without treatment. In rare circumstances, Acanthamoeba can spread from the cornea to deeper structures in the eye, thereby causing chorioretinitis or endophthalmitis. On these occasions, the spread of Acanthamoeba has been postulated to have been facilitated by surgery. , Endophthalmitis has also been described in association with disseminated disease in immunocompromised patients. ,
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here