Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Abnormal uterine bleeding is most commonly due to dysfunctional anovulatory bleeding in premenopausal women and endometrial atrophy in postmenopausal women.
Approximately 90% of endometrial cancer occurs after age 50, with the most common symptom being abnormal vaginal bleeding.
Transvaginal sonography (EVS) is the initial step in the workup of postmenopausal bleeding; however, if the endometrium is not adequately visualized EVS must be followed with biopsy or additional imaging.
Saline infusion sonohysterography (SIS) is a simple, low-cost, minimally invasive procedure with a high level of diagnostic accuracy.
SIS is more sensitive than EVS for the detection of focal endometrial abnormalities and rivals hysteroscopy in the identification and characterization of focal lesions.
SIS can be used to triage patients with abnormal uterine bleeding to the appropriate biopsy technique.
Tamoxifen, a selective estrogen receptor modulator (SERM), induces endometrial proliferation, which can take the form of hyperplasia, polyp, cancer, or cystic atrophy.
Abnormal uterine bleeding accounts for up to 20% of all gynecologic visits. Once pregnancy has been excluded, the most likely cause of uterine bleeding in premenopausal women is dysfunctional anovulatory bleeding. However, as women get older, organic pathologic conditions such as endometrial polyps, submucous myomas, endometrial hyperplasia, and even frank endometrial carcinoma become more likely. The Surveillance Epidemiology and End Results (SEER) database reports that the incidence of endometrial carcinoma is 25.1/100,000 women per year. The incidence of endometrial cancer increases with age, with the median age at diagnosis being 62 years. The SEER program reports that 5.6% of new cases occur in women age 35 to 44 years, 18.4% in women age 45 to 54 years, 33.9% in women age 55 to 64 years, 23.4% in women age 65 to 74 years, and 12.6% in women age 75 to 84 years, with the remaining 6.1% at the extremes of the age spectrum. Therefore, in postmenopausal women who are not on hormonal replacement therapy, any vaginal bleeding is considered to be secondary to endometrial cancer until proved otherwise, although the incidence of malignancy in such patients ranges from 2% to 10%, depending upon risk factors.
Nonetheless, the vast majority of patients with abnormal vaginal bleeding will have either dysfunctional uterine bleeding in association with episodes of anovulation that can best be managed hormonally or expectantly with reassurance (premenopausal women), or endometrial atrophy (postmenopausal women). The utility of ultrasound to distinguish such patients from those with organic pathologic conditions in a safe, painless, and convenient manner is clear.
Endometrial curettage was first described in 1843, and for many years it was the gold standard for evaluation of the endometrium, becoming the most common hospital-based operation performed on women worldwide. However, in the mid-1900s, it was realized that the technique missed endometrial lesions in approximately 10% of cases; of these, 80% were polyps. In the 1970s, vacuum-suction curettage devices allowed endometrial sampling to be performed without anesthesia in an office setting. Subsequently, cheaper, smaller, and less painful plastic catheters with their own internal pistons to generate suction became popular (e.g., Pipelle device, Unimar, Wilton, CT). They were found to have similar efficacy but better patient acceptance. Still, the Pipelle catheter was found to sample as little as 4% of the surface area of the endometrium and, therefore, had limited ability to identify malignancy confined to a polyp or localized to less than 50% of the endometrial surface. Thus, focal endometrial abnormalities such as polyps, focal hyperplasia, or carcinoma involving small areas of the uterine cavity may go undetected with undirected endometrial sampling, whether by curettage or various types of suction aspirations.
Pelvic sonography is considered the first-line imaging modality of the uterus. Transabdominal imaging can be helpful in the setting of large leiomyomas or a globally enlarged uterus. However, the mainstay of endometrial evaluation is transvaginal sonography (EVS) with higher frequency transducers and, hence, better resolution. In premenopausal women, the examination should be performed early in the proliferative phase at about days 4 through 6 of the menstrual cycle. Postmenopausal women can be examined any time unless they are on cyclic hormone replacement, in which case imaging should be performed about 5 to 10 days after the last progestin tablet.
The endometrium should be evaluated in both the sagittal and transverse planes. Measurement of endometrial thickness is obtained in the midline sagittal plane perpendicular to the long axis of the endometrium at the site of maximal thickness. By convention, endometrial thickness is measured to include both the anterior and posterior layers (double layer thickness) ( Fig. 27-1 ). Fluid in the endometrial cavity should not be included in the measurement; rather, the single layer thickness of the anterior plus the posterior layer of the endometrium should be measured and summed. If the endometrium is not adequately visualized in its entirety, it should be reported as nonmeasurable and incompletely visualized. Poor visualization of the endometrium has been reported in 10% to 24% of cases and may be due to uterine position, distortion by leiomyomas or adenomyosis, or distortion of the endometrial-myometrial interface from endometrial carcinoma.
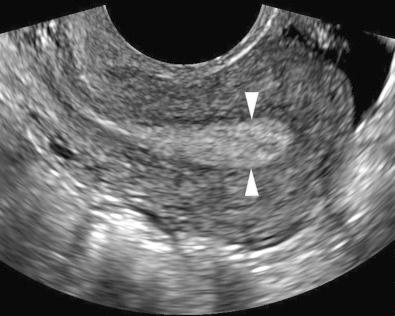
In the setting of intracavitary disease, the endometrial thickness measurement should include the lesion. A three-dimensional (3D) coronal view of the uterus has been found to be a useful adjunct to two-dimensional (2D) sonography in patients with suspected endometrial abnormalities ( Fig. 27-2 ). One study reported that additional information was obtained from the 3D coronal reconstruction in 39% of patients with an endometrial thickness of 5 mm or more. The presence and location of leiomyomas were more confidently diagnosed, in particular those in a submucosal location. Intracavitary fluid is quantified by measuring its maximal thickness in the sagittal plane.
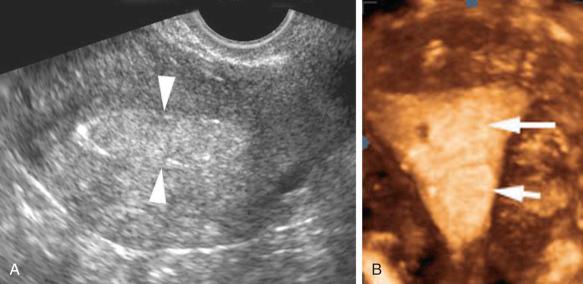
Sonohysterography, hysterosonography, EVS with fluid contrast augmentation, saline-infused sonography, and saline infusion sonohysterography (SIS) are the many appellations used to describe the instillation of sterile saline into the endometrial cavity during EVS to better evaluate the endometrium and fallopian tubes. Echogenic contrast agents such as air bubbles may be used, especially when evaluating for patency of the fallopian tubes. An examination performed with particular attention to the fallopian tubes is known as sonosalpingography. SIS is a simple, low cost, minimally invasive procedure with a high level of diagnostic accuracy. Specific advantages over hysterosalpingography are the lack of ionizing radiation and the ability to more accurately characterize masses in the endometrial cavity.
The SIS examination should be scheduled close to days 4 through 7 of the menstrual cycle in premenopausal women to decrease the possibility of false positive results because of the common normal irregularity and increased thickness of the secretory phase endometrium, as well as to avoid the possibility of performing the examination in a pregnant patient. However, in a patient with dysfunctional uterine bleeding it may not be possible to optimally time the examination. In these patients, a course of medroxyprogesterone acetate 10 mg daily for 10 days may be considered with the procedure timed to the withdrawal bleeding. The SIS patient should be advised to take a nonsteroidal anti-inflammatory drug (NSAID) approximately 30 minutes prior to her procedure to reduce discomfort. If this has not been done, an NSAID can be administered and the examination delayed by 30 minutes. The benefits of premedication outweigh the short delay. Prophylactic antibiotics may be prescribed if pelvic inflammatory disease is suspected or if the patient requires systemic prophylaxis for bacterial endocarditis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here