Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The classification of abnormal, posttraumatic pain is complicated and is encompassed in the term complex regional pain syndrome (CRPS). The term reflex sympathetic dystrophy (RSD), a descriptor of posttraumatic pain, is established in the lay, medical, and legal literature despite the absence of defined pathophysiologic findings and consistent clinical symptoms and signs. RSD is a condition considered to be present in a subset of patients contained within the broader category of CRPS. The contemporary literature continues to use the terms reflex sympathetic dystrophy, causalgia, algodystrophy, and other descriptors to describe these categories of pain ( Box 53.1 ). The purpose of this chapter is to present diagnostic criteria that define clinical subtypes of CRPS and to provide a practical approach for treatment in the acute and chronic or late stages of this dynamic process. The term RSD is cited in the text when it is used in the source journal publication. The term RSD is used most often in literature published before consensus meetings clarified the taxonomy of CRPS.
Acute atrophy of bone
Algodystrophy
Algoneurodystrophy
Causalgia state/syndrome
Chronic traumatic edema
Major causalgia
Major traumatic dystrophy
Mimocausalgia
Minor causalgia
Minor traumatic dystrophy
Neurodystrophy
Neurovascular dystrophy
Osteoneurodystrophy
Pain dysfunction syndrome
Painful posttraumatic osteoporosis
Peripheral trophoneurosis
Postinfarctional sclerodactyly
Posttraumatic pain syndrome
Posttraumatic sympathetic dystrophy
Posttraumatic vasomotor abnormality
Posttraumatic vasomotor instability
Reflex nervous dystrophy
Reflex neurovascular dystrophy
Reflex sympathetic dystrophy
Shoulder-hand-finger syndrome
Shoulder-hand syndrome
Sudeck atrophy
Sympathalgia
Sympathetic algodystrophy
Sympathetic-mediated pain
Sympathetic neurovascular dystrophy
Sympathetic overdrive syndrome
Transient osteoporosis
Traumatic angiospasm
The diagnosis of CRPS is contingent on the presence of regional pain combined with autonomic dysfunction, atrophy, and functional impairment affecting musculoskeletal, neural, and vascular structures ( Fig. 53.1 ). Type 1, or “classic,” RSD is not associated with an identifiable peripheral nerve injury. Type 1 CRPS is initiated by trauma and is associated with pain that is out of proportion to the injury, swelling, color changes, skin changes, atrophy, and stiffness. Type 2 has similar signs and symptoms in addition to an identifiable peripheral nerve injury (causalgia). Type 3 includes nontraumatic causes producing extremity pain such as myofascial syndrome ( Table 53.1 ). The type 3 category of CRPS is controversial; however, the use of this category expands the concept to include all extremity pain pathologic conditions. Pain can be acute, chronic, or mixed and can be classified as nociceptive (pain that arises from actual or threatened damage to nonneural tissue and is secondary to the activation of nociceptors), neuropathic (pain initiated or caused by a primary lesion or dysfunction in the nervous system), or nocicplastic (pain that arises from altered nociception despite no clear evidence of actual or threatened tissue damage causing the activation of peripheral nociceptors or evidence for disease or lesion of the somatosensory system) in origin. In addition, pain may be sympathetically maintained or sympathetically independent. The traditional hallmark of CRPS is pain without evidence of actual or threatened tissue damage or evidence of a disease or lesion directly affecting the somatosensory system. This chapter discusses only CRPS type 1 and type 2.
| Type | Description |
|---|---|
| Type 1 | Reflex sympathetic dystrophy (pain, functional impairment, autonomic dysfunction, dystrophic changes without clinical peripheral nerve lesion/injury) |
| Type 2 | Causalgia (pain, functional impairment, autonomic dysfunction, dystrophic changes with a diagnosable peripheral nerve injury) |
| Type 3 a | Other pain dysfunction problems (e.g., myofascial pain) |
The diagnostic criteria for CRPS have evolved over the years. A consensus workshop in 1994 decided to use the term CRPS rather than RSD and developed specific diagnostic criteria codified by the International Association for the Study of Pain (IASP) ( Box 53.2 ). These ISAP criteria had high diagnostic sensitivity but poor specificity for discriminating between diagnostic groups. In 2003, another meeting was held in Budapest to improve the ISAP criteria ( Box 53.3 ). The modified diagnostic criteria were called the Budapest Criteria and were based on empirically derived criteria reported in the literature. The four distinct CRPS components included in the Budapest Criteria retained the sensitivity of the IASP criteria and enhanced the specificity of the diagnostic criteria.
a If seen without “major nerve damage,” diagnose CRPS type 1; if seen in the presence of “major nerve damage,” diagnose CRPS type 2.
The presence of an initiating noxious event or a cause of immobilization b
b Not required for diagnosis; 5% to 10% of patients will not have this.
Continuing pain, allodynia, or hyperalgesia in which the pain is disproportionate to any known inciting event
Evidence at some time of edema, changes in skin blood flow, or abnormal sudomotor activity in the region of pain (can be a sign or symptom)
This diagnosis is excluded by the existence of other conditions that would otherwise account for the degree of pain and dysfunction
Continuing pain that is disproportionate to any inciting event
Must report at least one symptom in three of the four following categories:
Sensory: report of hyperesthesia and/or allodynia
Vasomotor: reports of temperature asymmetry and/or skin color changes and/or skin color asymmetry
Sudomotor/edema: reports of edema and/or sweating changes and/or sweating asymmetry
Motor/trophic: reports of decreased range of motion and/or motor dysfunction (weaknesses, tremor, dystonia) and/or trophic changes (hair, nails, skin)
Must display at least one sign at time of evaluation in two or more of the following categories:
Sensory: evidence of hyperalgesia (to pinprick) and/or allodynia (to light touch and/or deep somatic pressure and/or joint movement)
Vasomotor: evidence of temperature asymmetry and/or skin color changes and/or asymmetry
Sudomotor/edema: evidence of edema and/or sweating changes and/or sweating asymmetry
Motor/trophic: evidence of decreased range of motion and/or motor dysfunction (weakness, tremor, dystonia) and/or trophic changes (hair, nails, skin)
There is no other diagnosis that better explains the signs and symptoms.
The distinction “major nerve damage” is confusing as a discriminator for type 2 versus type 1 CRPS. Injury to an identifiable nerve is appropriate to designate a diagnosis as type 2. Historically, causalgia was associated with an injury to a “major” mixed motor and sensory nerve (e.g., the median or sciatic nerve).
Neuropathic pain is defined as pain initiated or caused by one or more lesions or dysfunction of the peripheral nervous system or central nervous system (CNS). Nociceptive pain results from damage to tissues as a result of thermal, mechanical, chemical, or other irritants. Neuropathic pain may be associated with CRPS type 2, or it may be considered a distinct entity, depending on its source and clinical manifestations ( Table 53.2 ). Mononeuropathy, if symptoms and signs extend beyond the nerve distribution, may constitute CRPS type 2.
| Term | Definition |
|---|---|
| Pain | Unpleasant perception associated with actual or potential cellular damage |
| Analgesia | Absence of pain in response to an insult that should produce pain |
| Neuropathic pain | Pain initiated or produced by a primary lesion or lesions or dysfunction of the peripheral or central nervous system |
| Nociception | Response to an unpleasant (noxious) stimulus that produces pain in humans under normal circumstances because of thermal, mechanical, chemical, or other irritants of nonneural tissues |
| Allodynia | Pain in a specific dermatomal or autonomous distribution associated with light touch to the skin; a stimulus that is not normally painful |
| Hyperalgesia | Increased sensitivity to stimulation (includes allodynia and hyperesthesia) |
| Hyperesthesia | Increased sensitivity to stimulation (pain on response to a mild nonnoxious stimulus) |
| Sympathetic pain | Pain in the presence of or associated with overaction of sympathetic fibers; by definition, the pain is relieved by sympatholytic interventions |
| Hypoesthesia | Decreased sensitivity to stimulation |
| Hyperpathia | Abnormally painful reaction to a stimulus |
| Dysesthesia | An unpleasant abnormal sensation |
| Paresthesia | An abnormal sensation |
The diagnosis of CRPS is a clinical diagnosis based on the history and physical examination along with diagnostic testing; it is dependent on the experience and clinical judgment of the physician. Therefore criteria such as in the Budapest recommendations provide a valuable, but not an infallible, process to support the diagnosis. It is crucial to remain cognizant that CRPS is a heterogeneous process; symptoms and signs may fluctuate with time and may “resolve” with or without sequelae. Patients without pain who “no longer meet diagnostic criteria but [may be] still symptomatic” can have considerable disability and deformity associated with contractures, and so on.
Attempts to identify a pathognomonic marker for CRPS remain unsuccessful; there are no diagnostic tools, serum biomarkers, or laboratory findings that define the condition. , Early recognition along with prompt treatment of CRPS is important to minimize permanent loss of function. However, even with prompt recognition and treatment, patients with CRPS may experience permanent impairment and disability. Many patients also face work incapacity, changes in occupation, and/or psychological disorders. ,
CRPS may include a component of sympathetically maintained pain (SMP) or sympathetically independent pain (SIP). These terms recognize the dynamic nature of dystrophic responses and stress the value of reliable and consistent clinical observations and descriptions. The diagnosis of SMP is based on pain relief with sympatholytic medications or sympathetic blocks , ; however, SMP may become SIP over time. Objective and reproducible methods to assess pain, quantify trophic changes, define autonomic dysfunction, and measure functional impairment are important to provide consistent treatment regimens and reliably assess outcomes. ,
Patients with late, longstanding, or chronic CRPS may not continue to fulfill the diagnostic criteria for the process. However, they can experience residual deformity and/or debility.
Ambrose Paré, 16th century: Charles IX experienced burning pain after phlebotomy
Percivall Pott, 1771: described pain after nerve injury
Silas Weir Mitchell, 1864: classic description of causalgia in the upper extremity with a brachial artery injury and partial injury of a median nerve
Sudek, 1900: bone demineralization associated with posttraumatic pain
Leriche, 1916: posttraumatic “burning” pain
Evan, 1947 and Bonica, 1973; term reflex sympathetic dystrophy
Proceedings of the VI Congress on Pain, 1991: introduced the term complex regional pain syndrome
Budapest criteria, 2007: diagnostic criteria based on symptoms and signs with improved specificity
Normally, pain is perceived only in the presence of actual or impending cellular tissue damage; persistent pain is pathologic in the absence of continued trauma. Painful peripheral nociceptive experiences that include cellular damage produce secondary inflammation by activating and sensitizing polymodal low-threshold mechanoreceptors and nociceptor afferent neurons, which, in theory, produce ectopic chemosensitivity to α-adrenergic agonists. This information is relayed via the process of transduction through small myelinated (Aδ), large myelinated (Aβ), and small unmyelinated (C) afferent fibers to the dorsal horn of the spinal cord, where sensitization of wide–dynamic range (WDR) neurons contributes to central nociceptive discharge ( Fig. 53.2A ). Within the dorsal horn, excitatory amino acids serve as the principal neurotransmitters. Transmitters include N -glutamate, aspartate, α-amino-3-hydroxy-5-methyl-4-isoxazopropionic acid (AMPA), N -methyl- d -aspartate (NMDA), and substance P. Of these, the NMDA receptor-transmitter interaction produces long-lasting potentials, is refractory to stimulation, and is theorized to play an important role in CRPS types 1 and 2. Nociceptive input is modulated via descending pathways, and both peripheral and central factors are required for the perception of pain (see Fig. 53.2B ). Pain intensity is determined by the magnitude and extent of the initiating/ongoing event, afferent input, efferent modulation, and CNS interpretation. Conscious appreciation of nociceptive (painful) experiences is dependent on a complex interplay of afferent and efferent information modulated and balanced by physiologic adaptations. Vasomotor disturbances may result from a variety of mechanisms, including antidromic vasodilation, vasoparalytic dilation, normal somatosensory reflexes, and denervation super sensitivity. Responses to nociceptive stimuli can vary considerably among individuals. The presence of nociceptive-induced inappropriate transmitter-receptor activity can affect peripheral microcirculatory control and, thereby, result in impaired nutritive blood flow and may sensitize CNS pathways. , Autonomic nervous system involvement in the pathophysiology of CRPS is acknowledged.
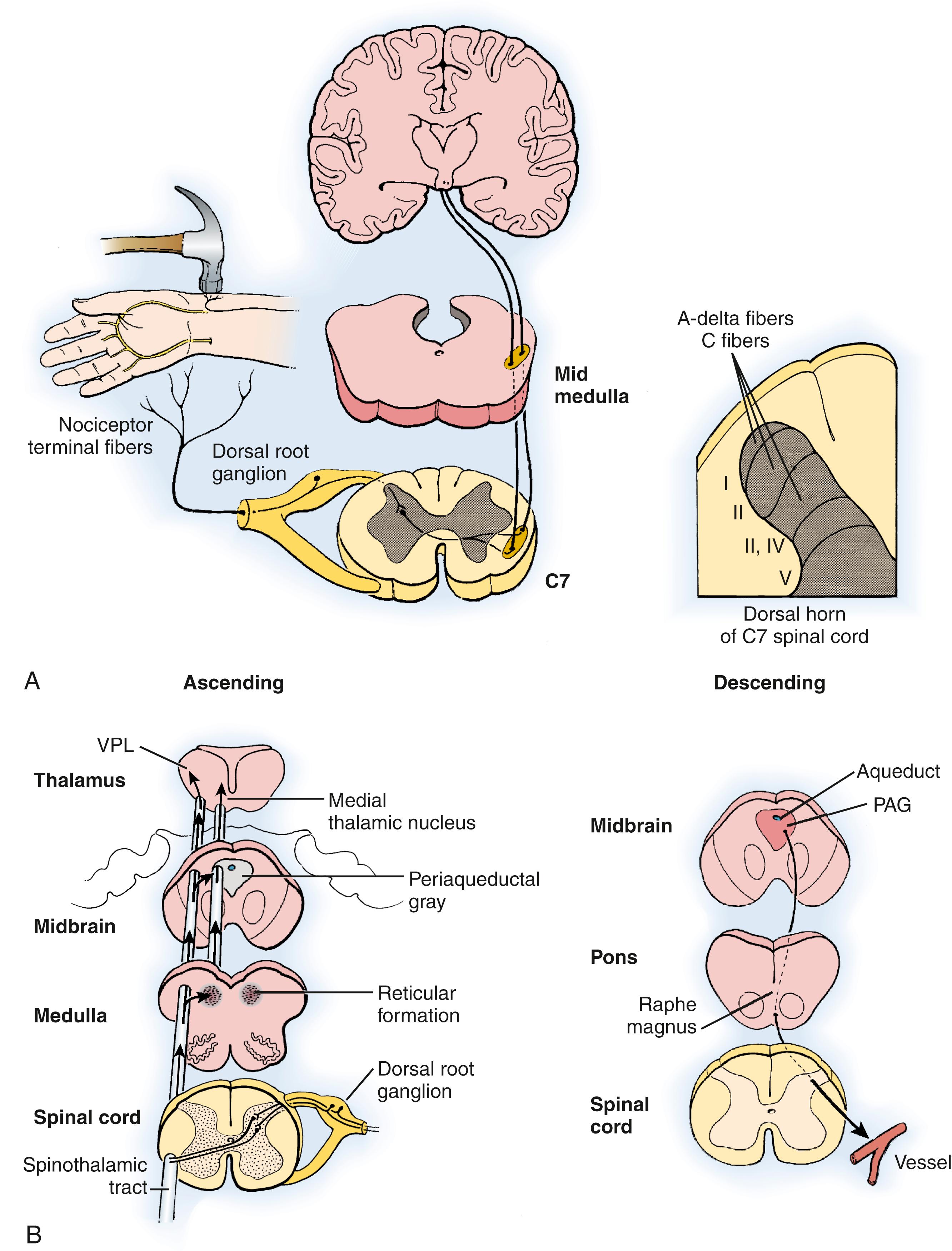
By definition, CRPS does not exist in the absence of pain. Peripheral injury stimulates endogenous inflammatory mediators via nociceptive pathways. Repetitive trauma/injury may alter protective responses by producing earlier activation through sensitization. In addition to providing central input, local nociceptors initiate the direct release of peptides and neurotransmitters, control the inflammatory process, and promote tissue repair. In addition, pain requires cognitive recognition.
A variety of nonneurogenic and neurogenic mediators participate in the transmission of information interpreted as pain. Nonneurogenic mediators include bradykinin, serotonin, histamine, acetylcholine, prostaglandins E 1 and E 2 , and leukotrienes. Neurogenic mediators, which are biologically active peptides produced by primary afferent neurons, potentiate or inhibit nociceptive information. These mediators include substance P, vasoactive intestinal peptide, calcitonin gene–related peptide, gastrin-releasing peptide, dynorphin, enkephalin, galanin, somatostatin, cholecystokinin, γ-aminobutyric acid, dopamine, and glycine.
The role of α-adrenergic receptors and local blood flow in sympathetically maintained complex regional pain states is well documented, , and relief of pain after intravenous phentolamine, a mixed α 1 - and α 2 -antagonist, is considered pathognomonic for SMP (CRPS type 1). , Abnormal regulation of adrenoreceptor function or modulation (or both) in neural and vascular structures is the major common control pathway supporting the concept that RSD (SMP) is a receptor disease. , Presynaptic and postsynaptic receptors are involved and affect nociceptive foci, blood flow, nutritional perfusion, and peripheral nerve excitability. , , The pathologic mechanisms involved in the compromise of extremity blood flow and neural control include: (1) abnormal neurotransmitter release secondary to nociceptive foci, (2) abnormal receptor distribution, and (3) alterations in receptor sensitivity (e.g., upregulation or downregulation).
The “gate theory” is an appropriate aid in the conceptualization of pain. The theory assumes that a finite amount of information can be received at the spinal cord or cortical level. The “gate” is the dorsal horn of the spinal cord. Thus painful information displaced or modified by less noxious input cannot be processed through the gate. Although aspects of this theory are as yet unproven, certain general principles relating to pain processing can be conceptualized by using this theory.
Acute pain is initiated during tissue injury or destruction, and the presence of acute pain may be beneficial or harmful. Beneficial effects include physiologic responses for maintenance of blood pressure, cardiac output, intravascular volume, and appropriate homeostasis. Acute pain warns the host of danger, prevents inappropriate motion of an injured extremity, and can diminish additional harm from repetitive injury. Persistence of pain beyond the need for protective action is unpleasant and can induce hypertension, tachycardia, coagulopathy, hyperglycemia, anxiety, fear, and chronic pain. Chronic pain that occurs in the absence of ongoing tissue destruction or that provides an inappropriate reflection of the intensity, magnitude, or duration of the tissue damage/compromise is pathologic. Although the pathophysiology of chronic pain is incompletely understood, the following processes can contribute to its establishment: persistent mechanical irritation of peripheral neural structures, incomplete regeneration of peripheral nerves, abnormal neurotransmitter activity, nutritional deprivation secondary to abnormal arteriovenous shunting, and central imprinting.
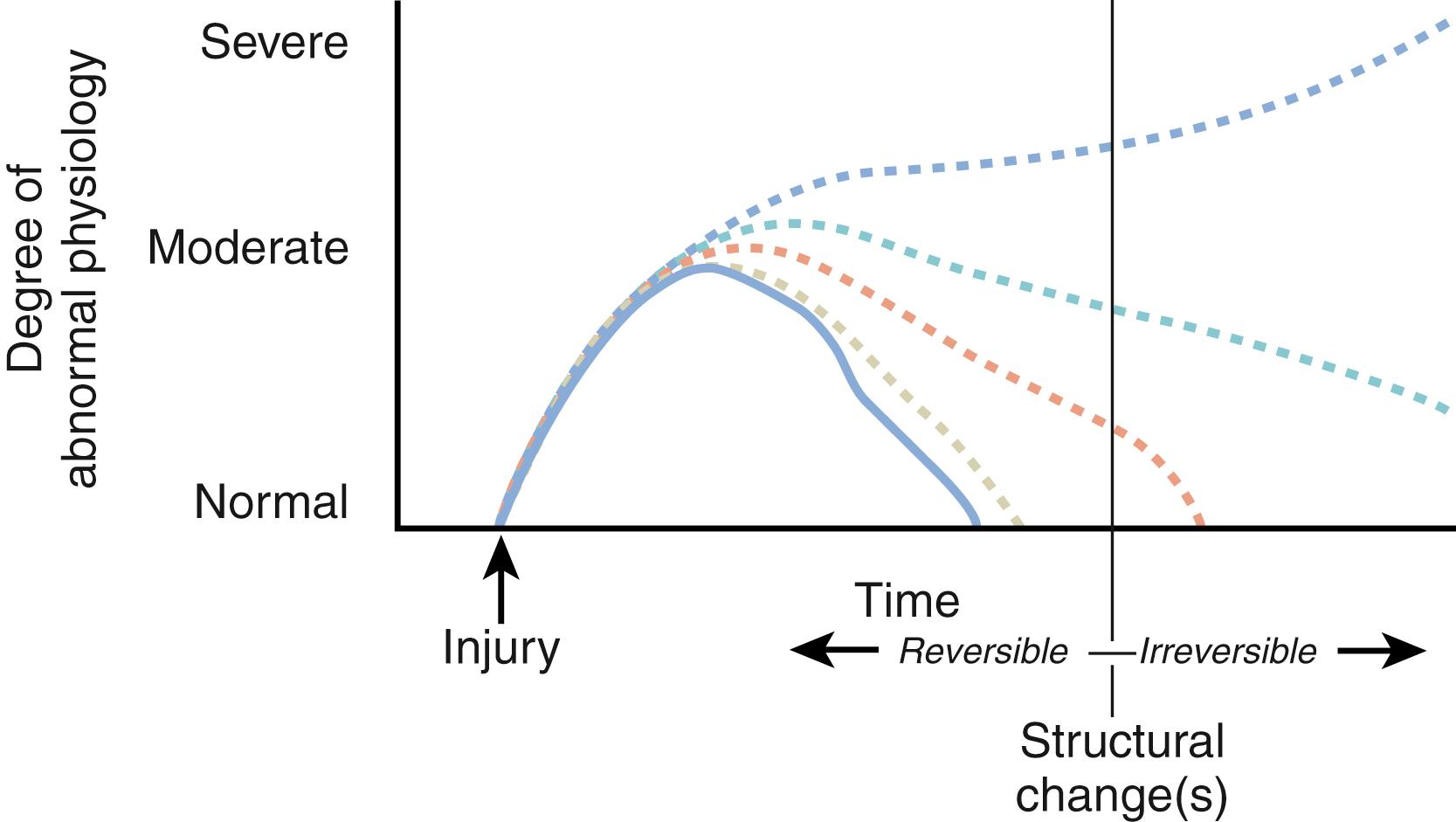
After trauma or surgery, a transient period of dystrophic extremity function is normal. However, it is abnormal for hyperpathia (heightened pain experienced by a given stimulus), allodynia (a painful response to normally nonpainful stimuli), vasomotor disturbances, and functional deficiencies to persist. If untreated, these conditions may progress to permanent compromise of the extremity. Importantly, interventions modify progression, and treatment may temporarily relieve symptoms if/when they recur. Posttraumatic alterations in extremity physiology follow a variable time course. Consequently, abnormal prolongation of these otherwise normal responses is pathologic, and, over time, irreversible changes in anatomic structures or physiologic processes may occur ( Fig. 53.3 ). When these irreversible changes occur, pain may persist, decrease, or become minimal and/or permanent deformity may occur. Therefore CRPS may be considered an abnormally severe or prolonged manifestation of a normal postinjury response. Abnormally prolonged dystrophic events may damage or compromise the arteriovenous shunt mechanism, , , , produce arthrofibrosis, , cause excessive osteopenia, alter neuroreceptor function, and result in central pain imprinting and/or any combination of these sequelae.
In 2003, the incidence of CRPS in Olmsted County, Minnesota, was 5.46 per 100,000 person-years, with four times as many female as male patients affected. From the electronic medical records of more than 600,000 patients listed in the Dutch Health Care System, the overall incidence rate of CRPS in the Netherlands was higher at 26.2 per 100,000 person-years, with females affected three times more often than males. In the Dutch population-based study, the upper extremity was more frequently involved compared with the lower extremity; fractures were identified as the most common precipitating event. , The mean age at onset was 46 years in the Minnesota study and 53 years in the Dutch study. , CRPS can also develop in children and adolescents with the majority of cases occurring in girls under the age of 12 involving the lower extremity. A favorable outcome was noted in most of pediatric CRPS patients. The risk of developing CRPS in a second anatomic location in patients with a history of CRPS is greater than the risk occurring in the general population. The incidence of cigarette smoking is higher in RSD patients, and cigarette smoking is statistically linked to RSD. Data suggesting a possible familial or genetic predisposition to CRPS is postulated but not confirmed. A predisposition to CRPS type 1 has been suggested on the involved side of hemiplegic stroke patients. There are frequent references that the majority of patients in whom RSD/CRPS was diagnosed within 1 year of injury (with treatment initiated) will improve significantly, , and patients with untreated symptoms (50% or more) lasting longer than 1 year will experience profound residual impairment. Based on these generalizations, early diagnosis and treatment portends a better but not guaranteed outcome. , The direct and indirect costs of CRPS are substantial; protracted medical care is expensive; loss of wages is major, and malpractice claims—when awarded—are greater. ,
Fracture of the distal end of the radius and ulna is one of the most common injuries associated with CRPS, and its occurrence complicates postinjury recovery in a measurable percentage of patients. The natural history of CRPS after distal radius fractures includes finger stiffness or “poor function” at 3 months after injury and correlates with residual RSD at 10 years. CRPS is more likely to occur after distal radius fractures in patients managed with tight casts or following a nociceptive or neuropathic event such as compression of the median nerve, overdistraction, instability of the distal radioulnar joint, or ulnar fracture. Dystrophic pain after surgical release of the carpal tunnel is commonly encountered in referral centers. ,
Common nerve injuries occurring during operative procedures that can precipitate CRPS include: (1) injury to the palmar cutaneous branch of the median nerve during carpal tunnel surgery, (2) damage to the superficial branch of the radial nerve during surgical approaches to the first and second dorsal compartments for tenosynovitis, and (3) trauma to the dorsal branch of the ulnar nerve during a surgical approach to the distal ulna. , Reports that CRPS can produce devastating consequences in association with simultaneous median nerve decompression at the wrist and partial palmar fasciectomy for Dupuytren contracture are not correct, and therefore these procedures can be performed safely.
CRPS is not a psychogenic condition. Extensive reviews of the existing literature do not support a psychological causation or an associated personality disorder. However, chronic pain is known to play a role in psychological well-being, with 58% of 283 consecutive patients admitted to pain centers, regardless of cause, fulfilling the criteria for personality disorders. , Dependent, passive-aggressive, and histrionic personality disorders are common in patients with chronic pain and are seen in patients with CRPS. Behavioral responses that reduce extremity use exacerbate edema and atrophy.
The differential diagnosis of CRPS includes certain psychological conditions encompassing conversion disorders and clenched fist syndromes. , A patient with SHAFT (sad, hostile, anxious, frustrated, and tenacious) syndrome is difficult to discern from a patient with CRPS without active suspicion. A history of multiple operations, absence of consistent clinical findings, a history of multiple treating physicians, use of a myriad of medications, psychiatric treatment, absence from work, disproportionate self-characterization and verbalization of symptoms, crying from pain, and a family history of disability are common factors. Misdiagnosis associated with abnormal postures is documented. Dysfunctional postures are defined, such as holding an upper extremity in a fixed posture. The typical posture of the hand of a patient with CRPS includes metacarpophalangeal (MCP) and proximal interphalangeal (PIP) joint extension. MCP and PIP joint flexion (“clenched”) is uncharacteristic of CRPS, and in such cases an alternative diagnosis is probable. Similarly, mild to moderate swelling is common; massive swelling or edema is rare and alternative explanations (e.g., factitious symptoms) in such cases should be ruled out. Lack of objective findings (e.g., trophic changes, edema, osteopenia, radiographic abnormalities, or fixed contractures) suggests that other conditions are responsible for the symptoms.
The pain associated with CRPS is observed in a variety of clinical manifestations ( Table 53.3 ). The pain may be described as burning, throbbing, pressing, cutting, searing, shooting, or aching (or any combination of these descriptors). Hyperalgesia, or the perception of pain greater than would be expected, may be primary affecting the area of injury or secondary affecting nontraumatized surrounding areas or the entire limb. Identification of the zone of primary pain can aid in the localization of a nociceptive focus; however, diagnosis of the etiologic trigger event may not be possible until successful sympatholytic management of the secondary hyperalgesia is complete. Allodynia, or perception of pain initiated by normally innocuous stimuli, is a characteristic of sympathetically maintained CRPS. Hyperpathia, or pain produced by painful stimuli that appears with a delay, outlasting the initiating stimulus and spreading beyond the normal neural distribution, is frequently encountered. It is important to differentiate spontaneous pain from stimulus-evoked pain . Difficulty sleeping because of burning pain is common and can be an early portent of progressive symptoms and signs.
| Budapest Criterion | 0, None | 1, Mild | 2, Moderate | 3, Severe | 4, Very Severe |
|---|---|---|---|---|---|
| Symptoms | |||||
| Pain | |||||
| Swelling | |||||
| Weakness | |||||
| Deformity | |||||
| Discoloration | |||||
| Cold sensitivity | |||||
| Radiculopathy | |||||
| Dystonia (movement) | |||||
| Pain | |||||
| Pain “out of proportion” | |||||
| Sensory: hyperesthesia | |||||
| Sensory: hyperpathia | |||||
| Sensory: allodynia | |||||
| Vasomotor Function | |||||
| Temperature asymmetry | |||||
| Skin color changes | |||||
| Skin color asymmetry | |||||
| Pseudomotor Function | |||||
| Edema | |||||
| Sweating changes | |||||
| Sweating asymmetry | |||||
| Other | |||||
| Motor or Trophic Abnormality | |||||
| Stiffness | |||||
| Decreased range of motion | |||||
| Trophic changes | |||||
| Weakness | |||||
| Tremor | |||||
| Dystonia | |||||
| Functional impairment | |||||
Quantifying subjective complaints of pain with the use of standardized and validated instruments or questionnaires allows an objective analysis of symptoms. , Useful instruments to evaluate symptoms and health status include the visual analog scale (VAS) for pain, the 36-item health status questionnaire (SF-36), the McGill Short Form Pain Questionnaire, and self-administered questionnaires designed to assess upper extremity symptoms/function. , The self-administered questionnaire used for assessment of the severity of symptoms and functional status for patients with carpal tunnel syndrome is applicable to dystrophic states caused by carpal tunnel surgery or injury to the median nerve at the wrist and allows the examiner to graphically follow the patient’s health-related quality of life ( Fig. 53.4 ). , , Cold intolerance, or pain on exposure to cold, can be analyzed by the McCabe Cold Sensitivity Severity Scale. The importance of standardized physiologic testing and instruments to document signs and symptoms is supported by the lack of reproducibility of isolated clinical evaluations. However, the use of published criteria and scoring systems for CRPS can be helpful. The Budapest criteria facilitate the diagnostic process.
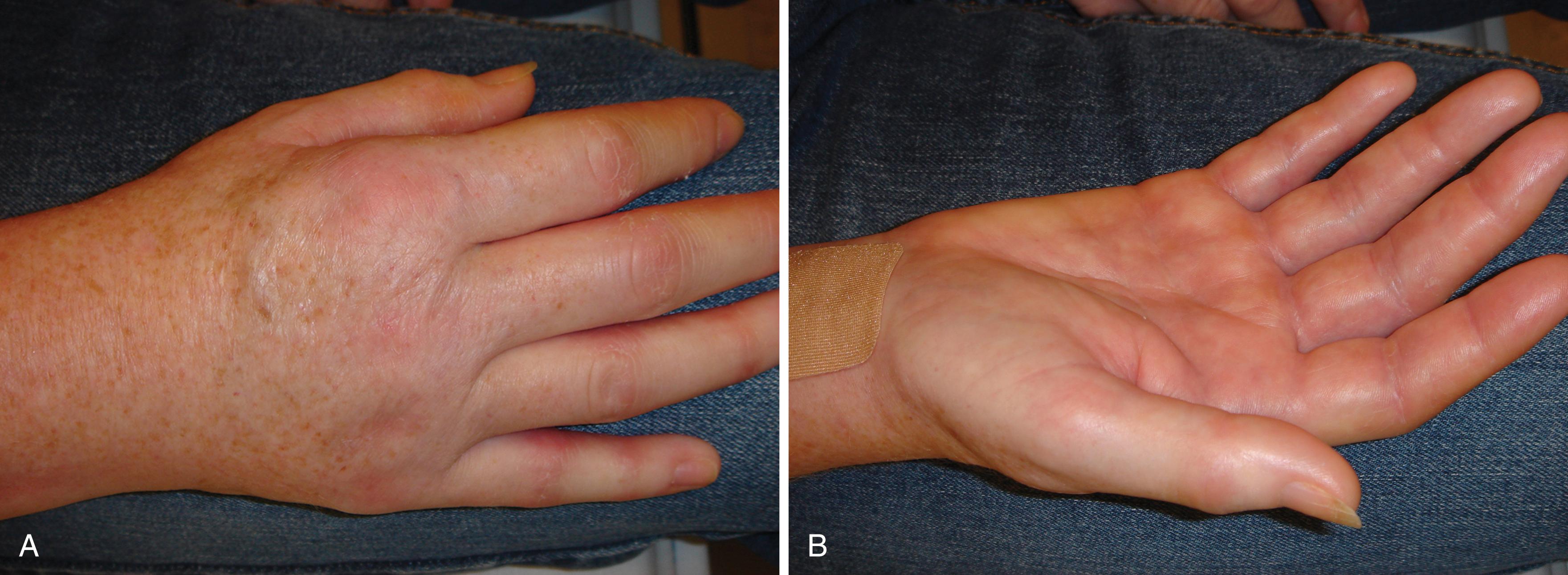
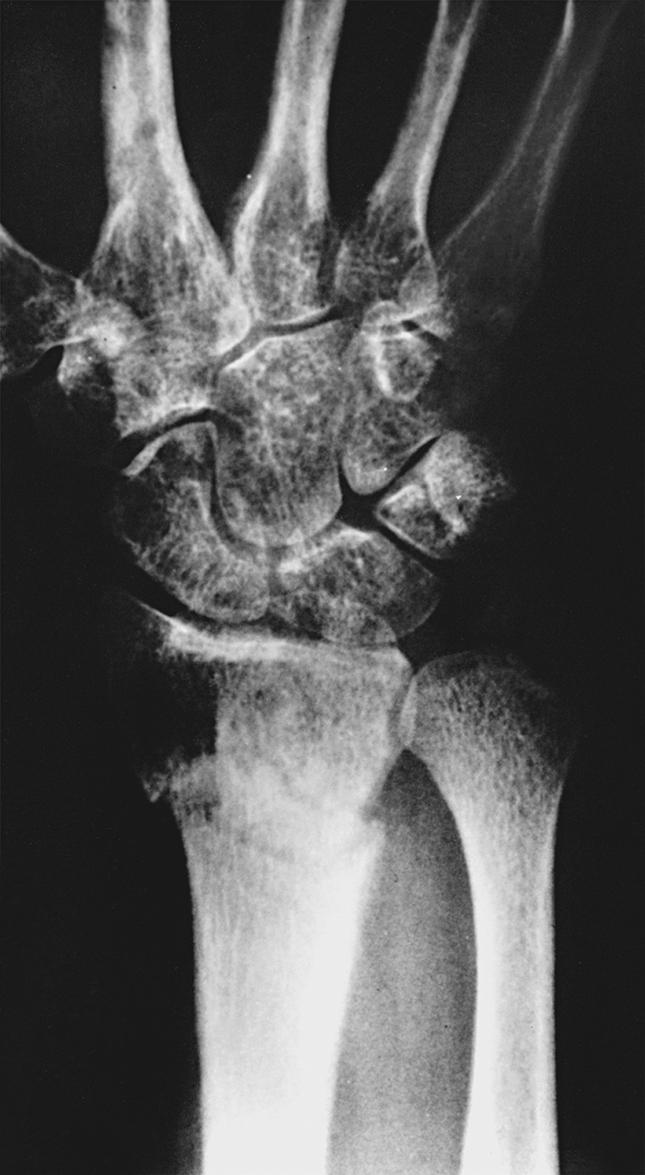
Trophic changes —stiffness; edema; osteopenia; atrophy of the hair, nails, or skin; hypertrophy of the skin (or hyperkeratosis); or any combination of these changes—may be present in patients with CRPS (see Fig. 53.1 ). Changes in the skin, hair, or nails are seen within 10 days of onset in 30% of extremities with CRPS type 1 (RSD). Despite treatment that reduces pain, cold intolerance, pain after use of the affected extremity, joint stiffness, nail and hair growth abnormalities, sensory disturbances, loss of finger extension/flexion, decreased grip strength, and shoulder stiffness are frequent and may persist. Osteopenia is common, involves both cortical and cancellous (trabecular) bone, and requires significant demineralization for visualization on plain radiographs ( Fig. 53.5 ). Objective analysis of bone demineralization requires dual-photon absorptiometry or quantitative scintigraphy. , Stiffness and atrophy of joints, muscles, and tendons may become apparent during endurance testing. , Movement disorders and dystonic posturing can also occur.
The autonomic nervous system controls microvascular perfusion and sweat gland activity. Abnormalities in autonomic and/or vasomotor control are seen during the course of CRPS in most patients with resulting loss of thermoregulatory control. , Affected extremities (hands) may be either “hot” or “cold,” and significant differences in digital temperatures occur between the affected and unaffected extremities in 80% of patients. Abnormalities in thermoregulation/vasomotion usually occur , , , , , and can vary depending on progression in the clinical stages of CRPS and can be less obvious at night in patients with chronic CRPS. Autonomic control can be evaluated by assessment of total digital blood flow, which consists of both thermoregulatory and nutritional components, and by analysis of sudomotor activity—sweating and piloerection. A detailed history will reveal symptoms of autonomic dysfunction in 80% of patients. Symptoms include abnormal sweating (excessive sweating or anhidrosis), vasomotor alterations, and heat or cold sensitivity (heat/cold intolerance). Vasomotor changes can be described as a reddish or bluish discoloration of the extremity either at rest or, more commonly, with the limb dependent. Evidence of autonomic dysfunction can be detected in 98% or more of patients by using sophisticated technology during the painful stages of CRPS. , , , Digital perfusion and its components can be analyzed by digital temperature measurements, laser Doppler fluxmetry, plethysmography, and vital capillaroscopy. , , Sweating may be analyzed by resting sweat output, the quantitative sudomotor axon reflex test (QSART), changes in galvanic skin response, or asymmetry of somatosensory-evoked potentials in the affected and contralateral extremities.
Sympathetic nervous system function, as reflected in laser Doppler fingertip blood perfusion and the vasoconstriction response to deep inspiration, varies by stage. Stage 1 patients exhibit increased blood perfusion but an unchanged vasoconstriction response, whereas stage 2 patients demonstrate decreased blood perfusion and stronger vasoconstriction.
Functional impairment may be analyzed by using standardized assessments of hand function, such as the Moberg pickup test. In addition, quantitative evaluation of extremity function before, during, and after stress with computerized equipment can detect subtle functional changes reflecting stiffness or atrophy. Thus the use of standardized tests and various computerized systems can document functional impairment that can be difficult to quantify by traditional methods ( Tables 53.3 and 53.4 ).
| Signs | 0, None | 1, Mild | 2, Moderate | 3, Severe | 4, Very Severe | |
|---|---|---|---|---|---|---|
| Clinical Signs | ||||||
| Skin | Mottled | |||||
| Cyanotic | ||||||
| Skin temperature | Cool | |||||
| Warm | ||||||
| Dry | ||||||
| Overly moist | ||||||
| Skin texture | Smooth | |||||
| Nonelastic | ||||||
| Atrophy | Pulp | |||||
| Skin | ||||||
| Joint | Stiffness | |||||
| Decreased passive range of motion | ||||||
| Nail changes | ||||||
| Hair growth | Decreased | |||||
| Increased | ||||||
| Contracture joints | ||||||
| Radiographic Signs | ||||||
| Trophic bone changes | ||||||
| Bone scan | ||||||
| Sensory | Allodynia | |||||
| Dexterity | ||||||
| Strength | Grip | |||||
| Dynamometer | ||||||
Early warning symptoms that correlate with developing CRPS include “intense” pain, difficulty sleeping, and stiffness. , Although these symptoms are nonspecific and not pathognomonic for CRPS, CRPS is less likely in the absence of these symptoms. In our experience, pain that does not respond to antiinflammatory and/or narcotic medications, burning pain, difficulty sleeping, and stiffness warrant careful evaluation for early CRPS.
The clinical characteristics of CRPS are related to altered extremity physiology. The magnitude of symptoms and signs is a reflection of the initiating event, postinjury response, inherent patient adaptability, interventional modalities, and time. The classic dystrophic progression from acute (<3 months) to dystrophic (3 to 6 months) to chronic (>6 months) occurs infrequently because of individual variability and the effects of partial treatment. Patients with CRPS who are evaluated for a painful, hot, swollen, stiff hand are easily identifiable; however, the precipitating nociceptive trigger (e.g., contusion of the superficial radial nerve) may not be identified until the SMP is managed and the dystrophic manifestations are treated. Once allodynia and hyperpathia are alleviated, careful examination can delineate an underlying disorder that may be amenable to treatment. , Ineffectively or partially treated patients with CRPS or variants of CRPS can be labeled as having “poor results” or as “noncompliant patients.” In the postoperative or posttraumatic period, unusual swelling, stiffness, pain, and restlessness can represent a dystrophic response.
The physical examination of suspected CRPS patients should include a neurologic assessment and evaluation of the cervical and thoracic spine. The presence of cervical disease (either discogenic or degenerative) may exacerbate CRPS, a form of “double-crush syndrome.” Preexisting or acquired thoracic outlet or distant compression neuropathies can represent a “triple or more” crush syndrome. Cervical spine and shoulder range of motion should be recorded, and the brachial plexus should be evaluated. The entire arm should be assessed to rule out evidence of vascular or neurologic compression within the thoracic outlet. An adhesive capsulitis, or shoulder-hand syndrome, is a frequent sequela that can be overlooked unless the shoulder is examined. Hypersensitivity, vascular adequacy, edema, sensibility, joint range of motion, motor function, grip, pinch, fibrosis, sweating, and vasomotor tone of the entire extremity must be evaluated. Grip strength is frequently abnormal and should be assessed carefully. Currently, there are no objective laboratory tests to aid in the diagnosis of CRPS.
Identification of nociceptive foci, which include peripheral nerve lesions and mechanical derangements, is important. Evaluation before and after sympatholytic intervention may delineate “trigger events” that might have initiated or propagated the dystrophic symptoms. , Nociceptor input may be associated with SMP or SIP. However, early diagnosis plus prompt surgical correction of SMP associated with peripheral nerve injury is an effective management approach.
Standardized evaluations and tests provide reproducible and objective information with regard to pain-pressure thresholds. Rubber-tipped algometers, dolorimetry, monofilaments, computer-controlled stimuli, or evaluations of thermal pain thresholds can be used to provide a quantitative measure of hyperpathia or allodynia. For example, perception of pain from a 2.83 Von Frey monofilament (normally not painful) in a specified area or dermatomal distribution defines the extent of allodynia. Success or failure of treatment can be assessed by repeated pain threshold evaluations. Monofilaments provide the most practical method for assessing and monitoring allodynia and hyperesthesia. Von Frey monofilaments are available in pocket kits, are inexpensive, and provide useful bedside sensibility and pain threshold data. Decreased pain after the use of a same-size or larger monofilament documents improvement, whereas persistent pain from the same-size or a smaller-diameter monofilament suggests ineffective management.
Regional osteopenia can be documented on plain radiographs in approximately 80% of extremities affected by CRPS. , , A substantial decrease in mineralization is necessary for changes to be visualized on standard anteroposterior and lateral radiographs (see Fig. 53.5 ). Classic Sudeck atrophy includes diffuse osteopenia with juxtacortical demineralization and subchondral erosions or cysts. Five radiographic patterns of resorption have been described: (1) irregular resorption of trabecular bone in the metaphysis creating a patchy appearance, (2) subperiosteal bone resorption, (3) intracortical bone resorption, (4) endosteal bone resorption, and (5) surface erosions in subchondral and juxtachondral bone. Although osteopenia is easily visualized in metaphyseal areas, data confirm that the density of cortical and cancellous bone is affected equally in patients with CRPS. ,
In the future, functional brain magnetic resonance imaging may be valuable in evaluating the pathophysiology associated with CRPS and in monitoring patients’ responses to interventions. Abnormalities in the size of the cortical representations of the affected hand on the primary and secondary somatosensory cortices have been reported, and “patterns of cortical reorganization in the primary somatosensory cortex seem to parallel impaired tactile discrimination.”
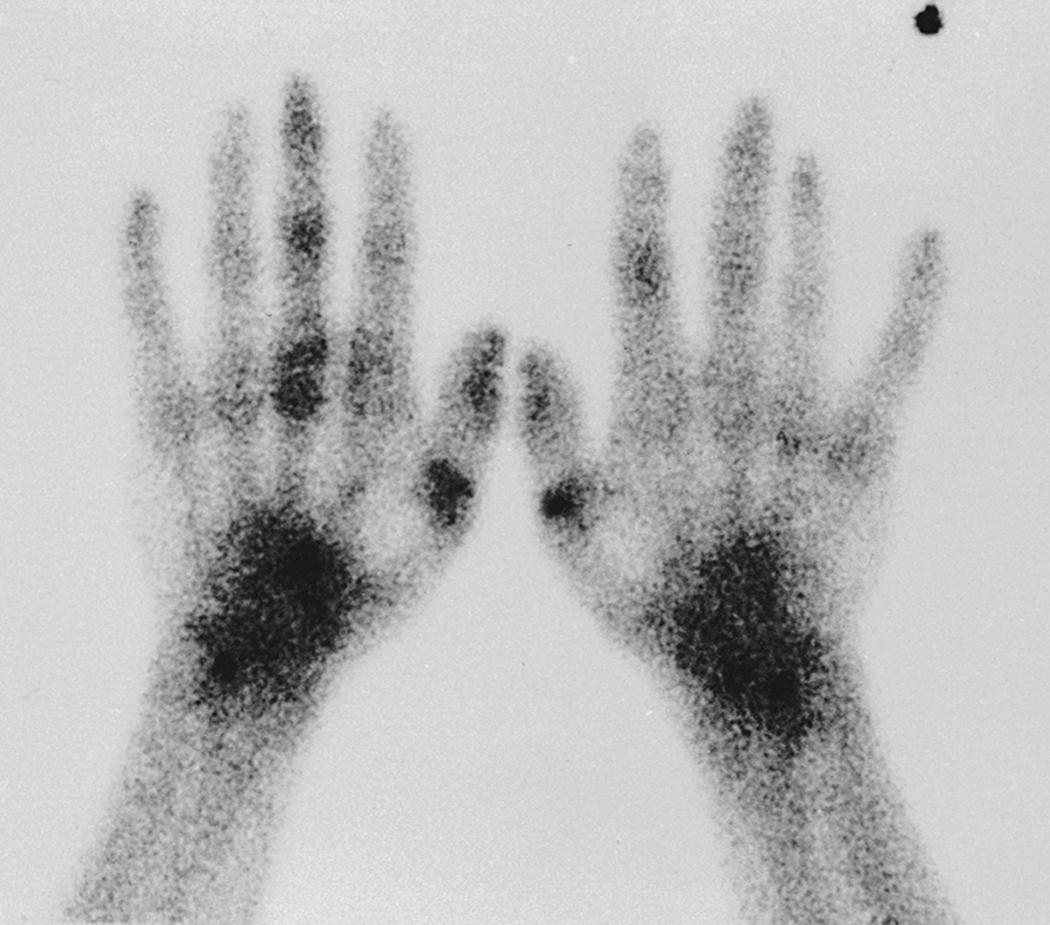
Historically, three-phase technetium 99m bone scanning (TPBS) was performed commonly and had a major role in the diagnosis of RSD. , With TPBS, the first, or “dynamic,” phase lasts 2 to 3 minutes and provides an assessment of digital perfusion. The second phase, or “blood pool image” or “tissue” phase, allows an assessment of total perfusion over a 3- to 5-minute period. The third phase, a standard bone scan, evaluates uptake of radiotracer in bony structures ( Fig. 53.6 ). In patients with CRPS, the use of a forearm or arm tourniquet or blood pressure cuff may affect accumulation of radiotracer in the hands or digits during phases I and II. Traditionally, scans have been considered “positive” if asymmetric flow is noted in phases I, II, or III. However, reports suggest that the diagnostic yield of phase III alone equals the diagnostic yield of the three-phase scan ( Fig. 53.7 ). Mackinnon and Holder have stated that a “strictly interpreted” phase III scan demonstrating “diffuse increased tracer uptake in the delayed image is diagnostic for RSD.” Although a positive phase III scan provides specific corroboration of the clinical diagnosis of CRPS, the sensitivity of scintigraphy is insufficient to provide standalone criteria, and therefore it should be used to corroborate clinical suspicion. In the majority of studies, a “positive” scan has high specificity but poor sensitivity. , , , Quantitative scintigraphy provides useful physiologic data, allows quantitative assessment of regional bone loss associated with complex regional pain, and adds useful diagnostic information. , , However, bone scan data do not correlate with symptoms, do not provide prognostic information, and are unable to aid in the prediction of which interventional approaches might be successful. ,
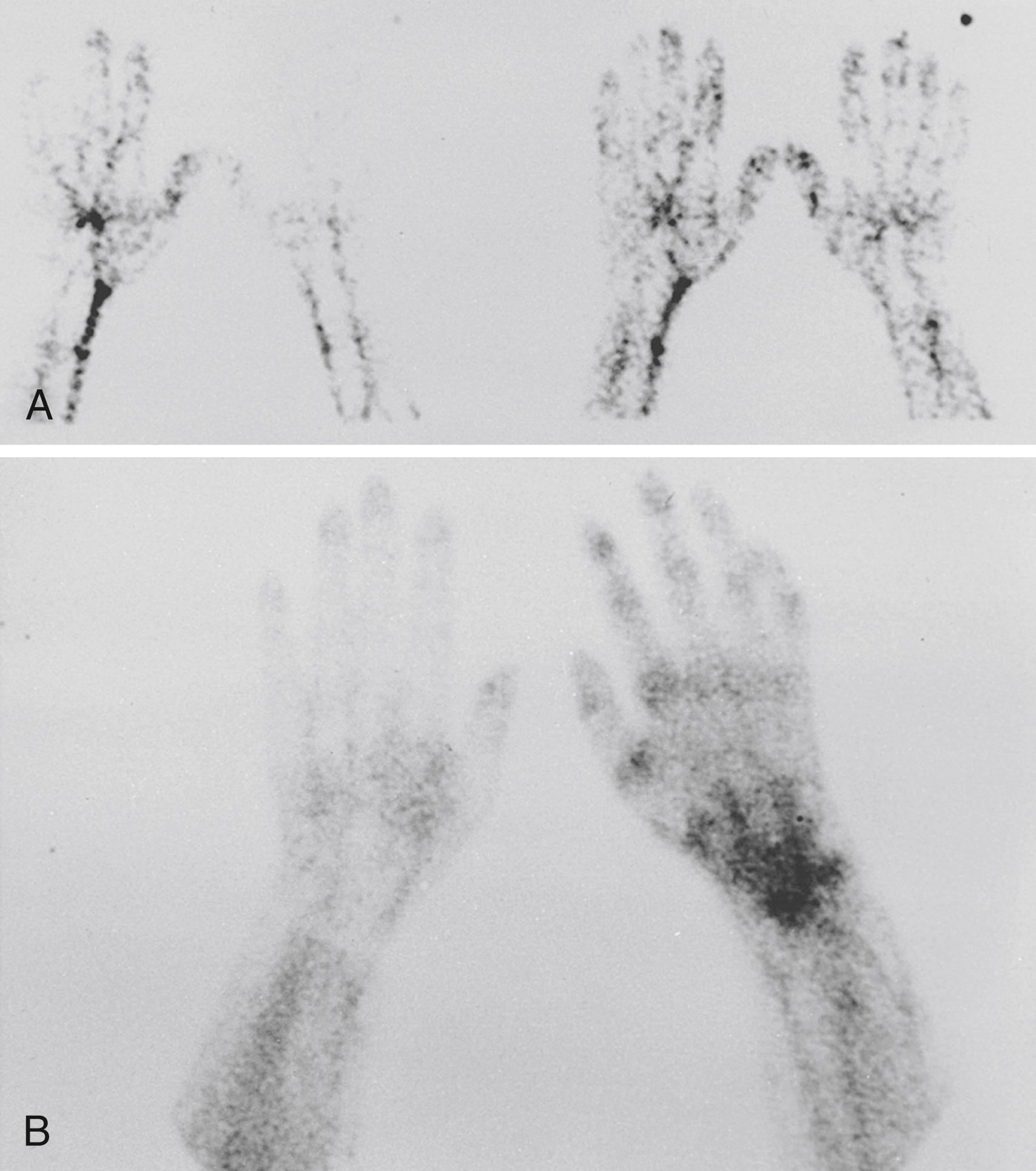
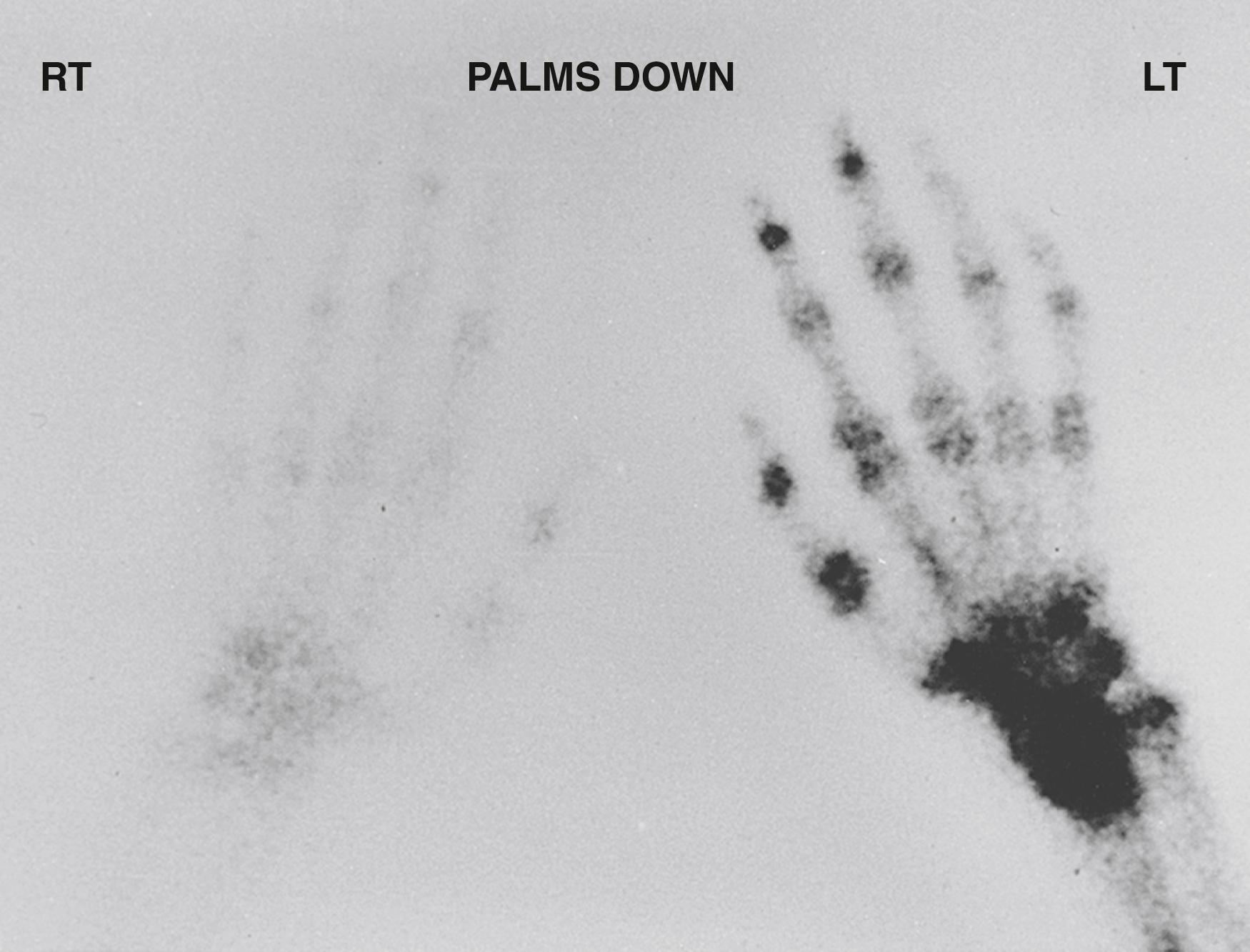
In the majority of patients, the abnormal bone turnover reflected in the TPBS is diffuse throughout the hand and wrist. The segmental distribution of RSD isolated to a digit, a single ray, or multiple rays can be evaluated by a bone scan to delineate the affected areas ( Fig. 53.8 ). Although TPBS is used frequently, there is no evidence to support (1) diagnostic accuracy in variant, partially treated, or late-stage CRPS ; (2) prognostic data that predicts outcome , ; or (3) value in determining management decisions. , ,
At this time, a positive phase III bone scan is not a prerequisite for the diagnosis of CRPS or SMP. Bone scans do not correlate with traditional staging criteria for RSD, are not part of the diagnosis of SMP or CRPS, do not predict recovery, and will not predict the potential for response to treatment. , , However, the presence of a positive phase III bone scan provides objective support for the clinical diagnosis of CRPS.
Autonomic dysfunction is associated with CRPS. Autonomic function controls sweating and microvascular perfusion. Evaluation of sympathetic control or, more specifically, the sympathetic dysfunction associated with characteristic pain from CRPS provides objective and sensitive methods to corroborate the clinical suspicion of CRPS.
Under normal conditions, total digital blood flow is composed of 80% to 95% thermoregulatory flow and 5% to 20% nutritional flow; the distribution of nutritional versus thermoregulatory flow is governed by complex factors that control arteriovenous shunting. , , Abnormal autonomic control of microvascular perfusion in patients with CRPS is reflected, in part, by ischemia secondary to inappropriate arteriovenous shunting and decreased nutritional flow. , , , , , , The importance of nutritional flow in the pathogenesis of CRPS is supported by the segmental distribution of trophic events, the rapidity of action of successful sympatholytic interventions, and the similarity of pain patterns in patients with contrasting clinical findings. Abnormal arteriovenous shunting in bone has been documented in patients with CRPS. Nutritional deprivation may exist in both a “warm, swollen” hand with high total flow and a “cold, stiff” hand with low total flow. The common underlying finding in both clinical situations is nutritional deprivation resulting from inappropriate arteriovenous shunting ( Fig. 53.9 ). Pain relief in CRPS is not dependent on peripheral vasodilation (thermoregulatory flow) after spinal cord stimulation, thus suggesting the importance of nutritional flow. In addition, there is growing evidence that physiologic subgrouping of CRPS patients, in contradistinction to staging based on timing, is important. For example, “warm” CRPS responds differently to intervention than “cold” CRPS does.
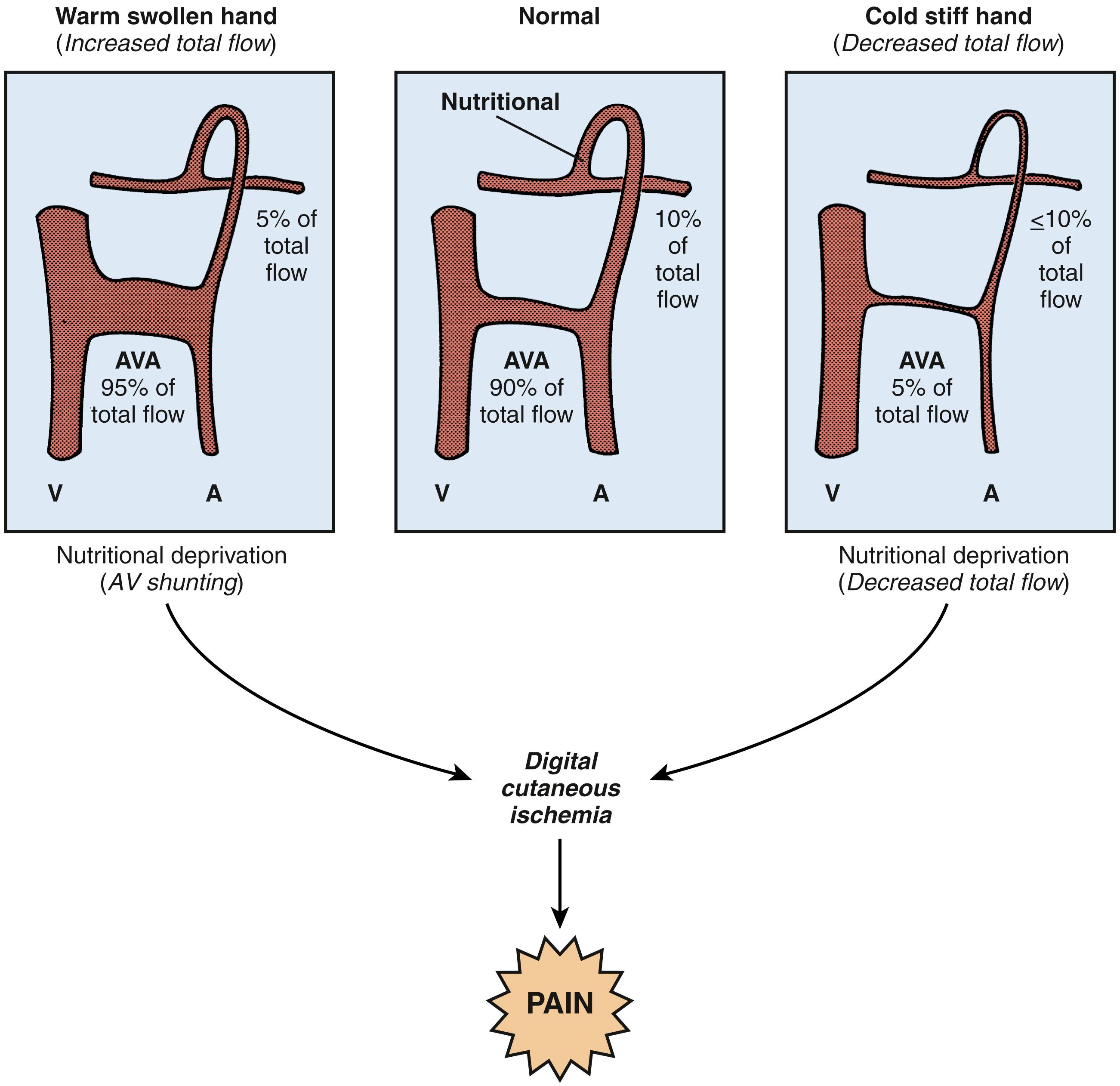
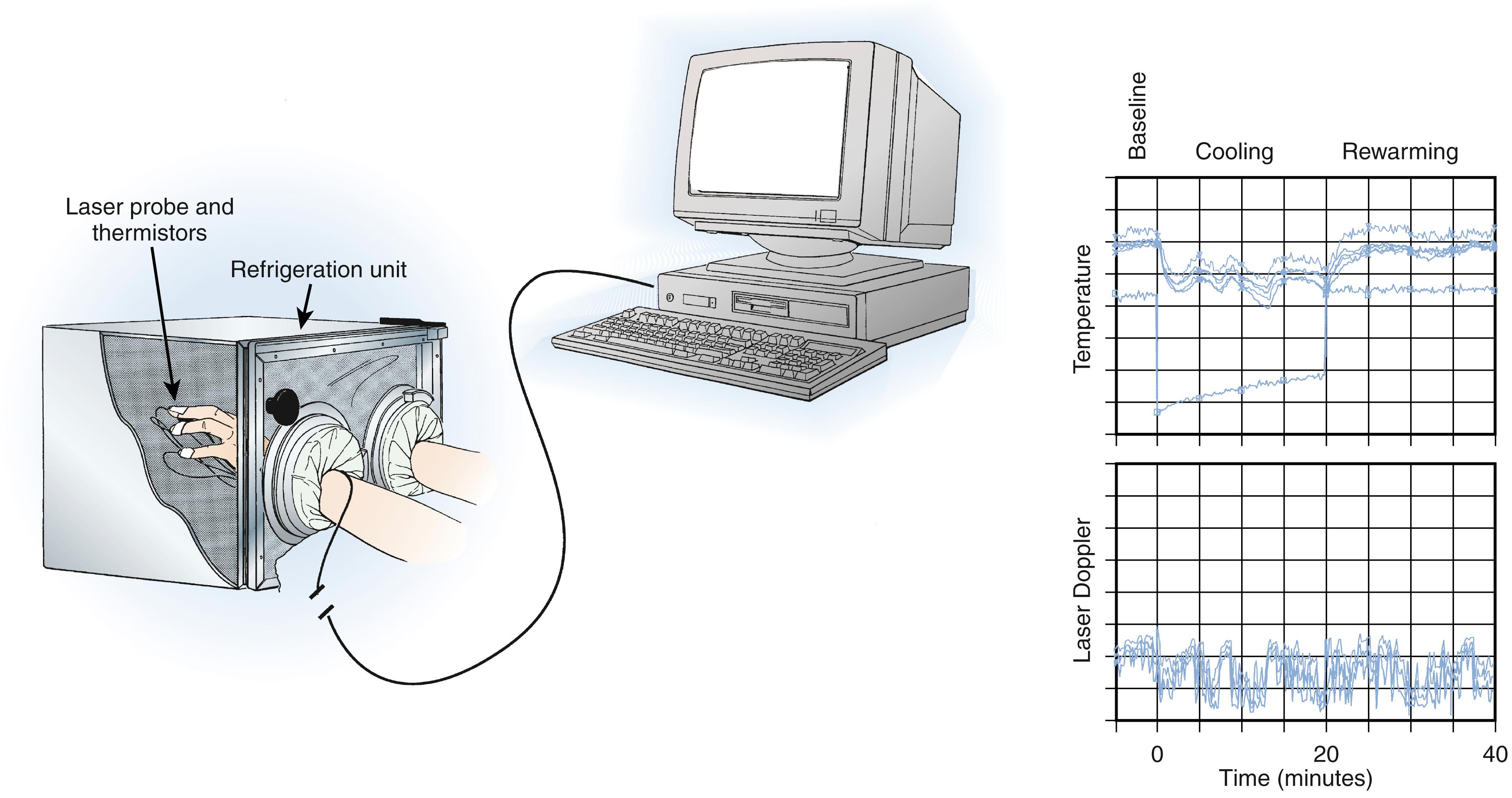
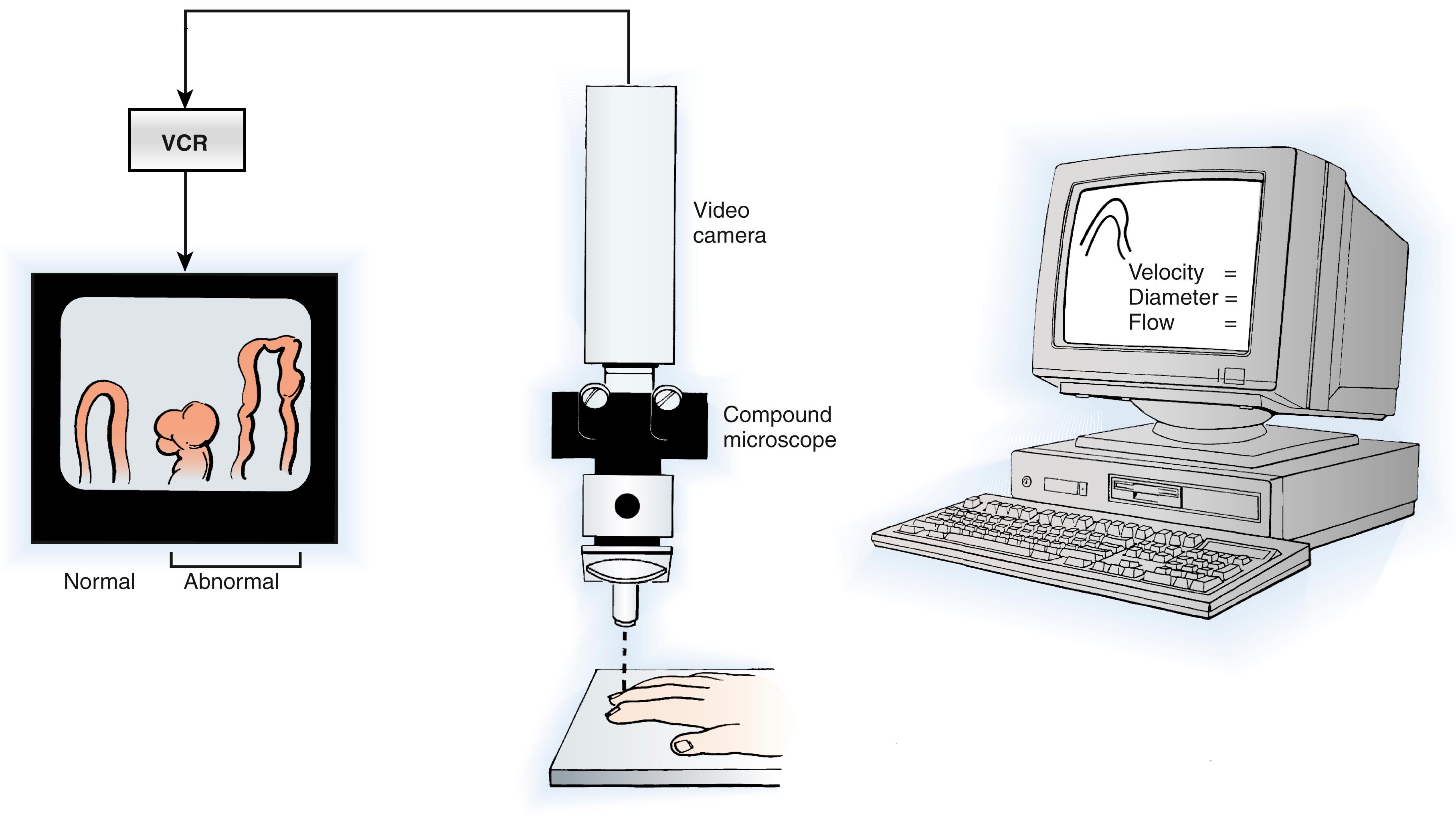
Total digital blood flow—both the thermoregulatory and nutritional components—can be analyzed by digital temperature measurements, laser Doppler fluxmetry, and vital capillaroscopy. The use of a stressor to perturb physiologic homeostasis by thermal, emotional, or ischemic methods is necessary to obtain reproducible information, to evaluate dynamic response patterns over time, and to maximize sensitivity ( Fig. 53.10 ). , One technique that combines a stressor with physiologic measurements is isolated cold stress testing. , In this procedure, total digital blood flow and microvascular perfusion can be evaluated by monitoring digital temperature and laser Doppler fluxmetry before, during, and after controlled thermal stress. The heterogeneity of digital microvascular perfusion may be analyzed by laser Doppler techniques and is reflected in average pulp temperature readings. Nutritional flow may be measured directly by vital capillaroscopy ( Fig. 53.11 ). , Analysis of temperature, laser Doppler fluxmetry, microvascular perfusion, and nutritional flow permits an objective determination of autonomic vasomotor stability and physiologic staging of autonomic sympathetic performance, defines the adequacy of nutritional flow, and provides an assessment of the effects of interventions. , , For example, the effectiveness of sympathetic blockade can be assessed by analysis of digital temperature and microvascular perfusion during the application of a physiologic stress ( Fig. 53.12 ). Laser Doppler perfusion imaging provides an assessment of microvascular perfusion over a 12- × 12-cm area and can be used to evaluate the effects of sympathetic blockade on skin blood flow.
Sudomotor activity may be assessed by cumulative unstimulated resting sweat output, QSART, galvanic skin response, combined temperature and peripheral autonomic surface potential reflex response, and analysis of sympathetic skin response evoked by activation of sympathetic unmyelinated efferent fibers. These techniques can be used to obtain quantitative measurements of sweat function, which then can be contrasted to contralateral or ipsilateral areas or normal controls (or both). When combined with microvascular perfusion indices of temperature or laser Doppler fluxmetry, these data supplement the clinical examination, document autonomic dysfunction, and provide objective evidence of the effects of intervention. ,
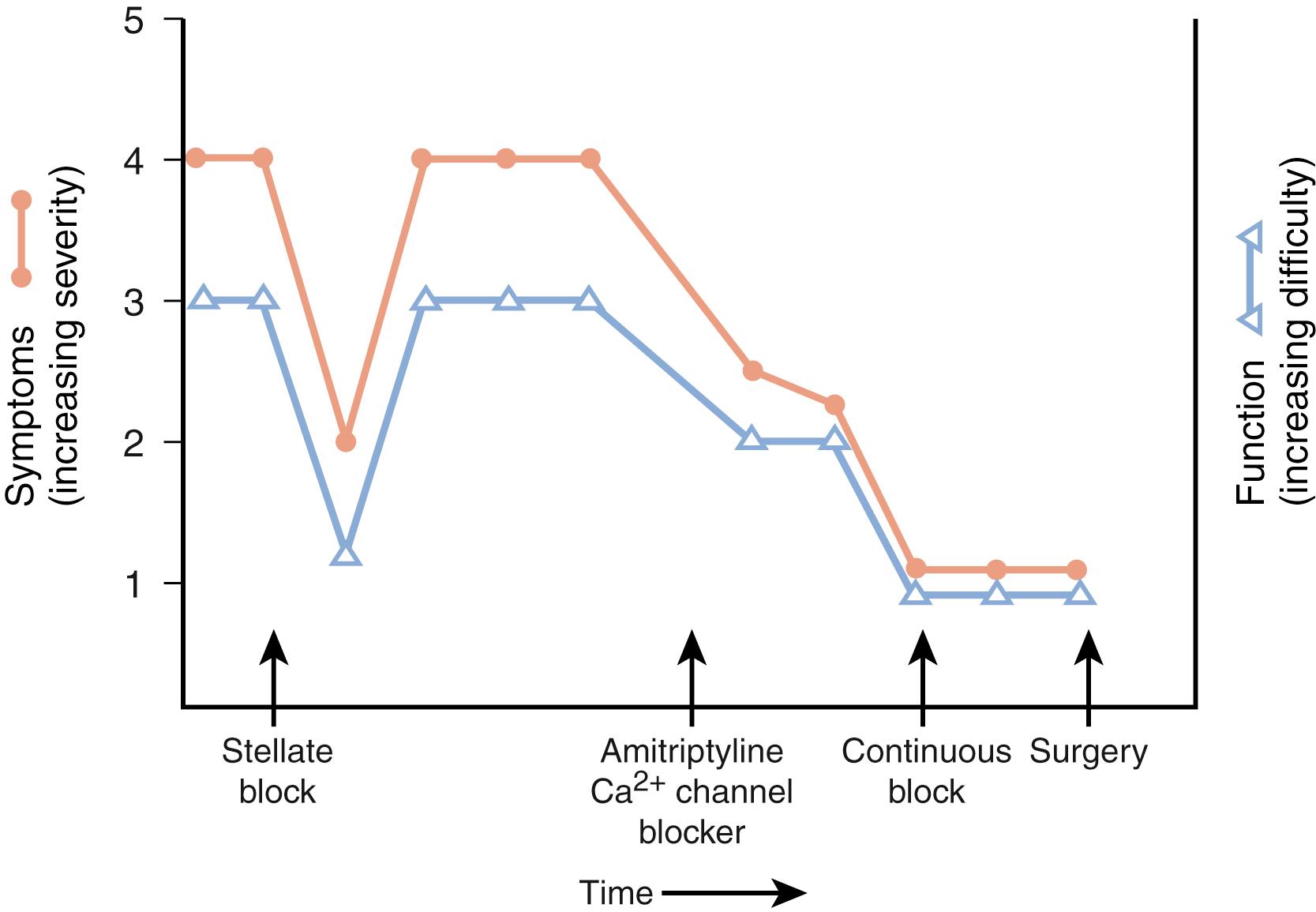
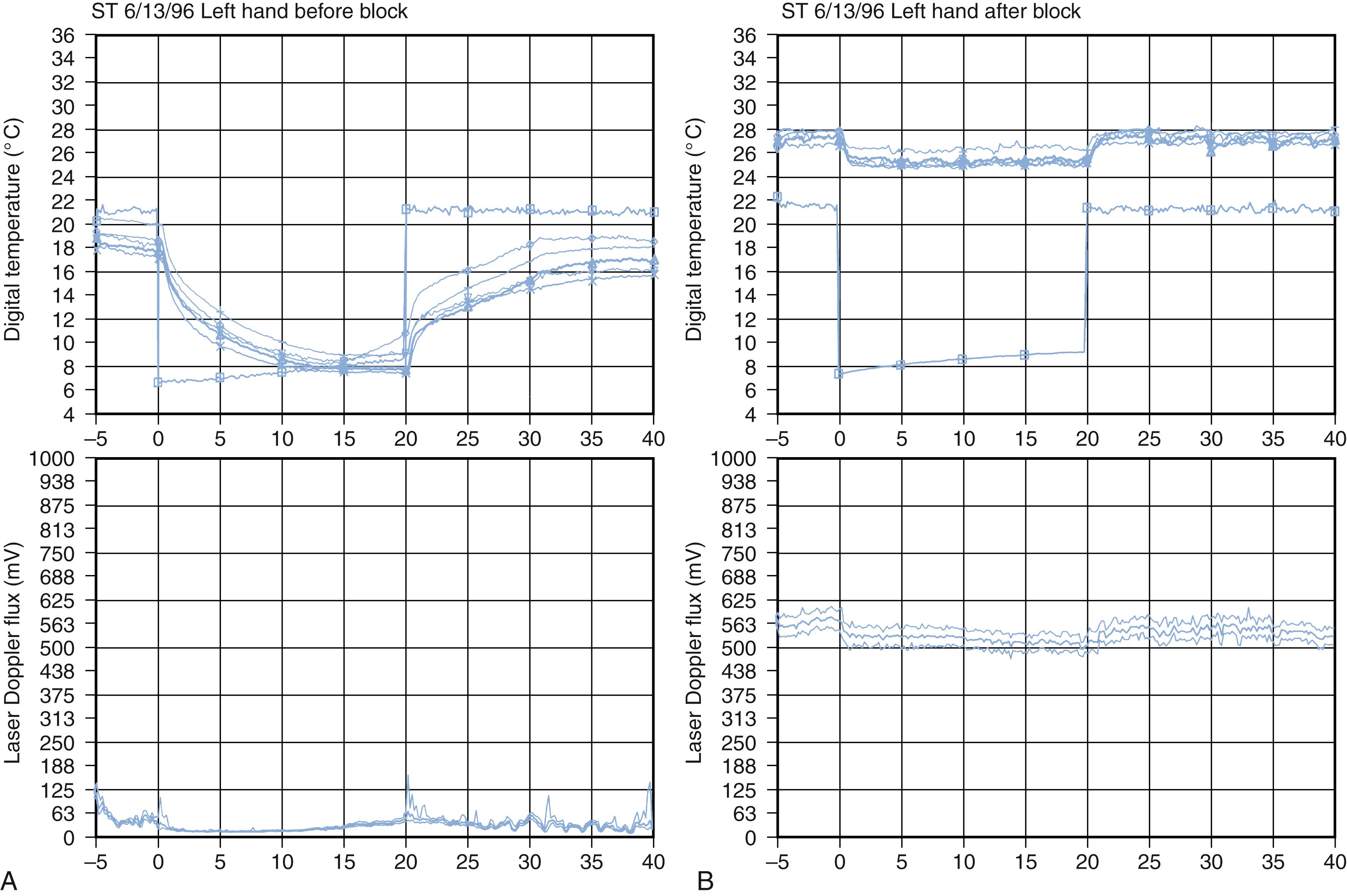
CRPS may be sympathetically maintained or sympathetically independent. Relief after sympatholytic interventions defines SMP. , Pain relief after the administration of parenteral, oral, or topical pharmacologic preparations or after neural blockade confirms the influence of the sympathetic nervous system and supports receptor-mediated abnormalities. , Pain relief after an intravenous injection of phentolamine (a mixed α 1 - and α 2 -adrenergic blocking agent) is considered presumptive evidence of SMP, or it may be considered diagnostic of SMP. Phentolamine testing is compromised unless the drug is administered in conjunction with placebo injections. , Intravenous phentolamine can produce profound transient side effects, including headache and hypotension. Unlike other pharmacologic end organ blockers (e.g., bretylium), phentolamine has no therapeutic indications and is used only as a diagnostic modality. Data indicate that adequate sympathetic blockade does not consistently correlate with the extent of pain relief and that the duration and density of the block may alter the clinical response. The role of SMP may be evaluated by nerve blocks of the stellate ganglion, epidural space, brachial plexus, or peripheral nerves. , Diagnostic blocks are usually obtained with short-acting or intermediate-acting local anesthetics (e.g., lidocaine). Stellate ganglion blocks can be performed by injections administered at the transverse process of C6 (Chassaignac tubercle) or by a medial approach at C7. Injections of the stellate ganglion or sympathetic trunk that are adequate to induce sympatholysis are dependent on variations in patient anatomy, the presence or absence of scarring in or around the sympathetic chain, drug diffusion characteristics, and the ability and skill of the injector. Therefore adequate interpretation of results requires verification of sympatholysis, which can be accomplished by the observation of indirect signs such as Horner syndrome, nasal congestion, or facial anhidrosis or by direct evidence of sympathetic nerve block such as an increase in digit temperature (≥3°C), venous engorgement of the hand veins, dry skin, changes in skin color, and characteristic laser Doppler changes. , , , Indirect “signs” of blockade are mediated by preganglionic fibers and can occur without sympathetic blockade. Consequently, it is imperative to have objective evidence of blockade before dismissing patients or declaring them to have SIP. Stellate ganglion blocks, even when administered by experienced anesthesiologists, are effective in only 70% to 75% of cases. Therefore objective determination of the adequacy/effectiveness of autonomic blockade is important. Furthermore, a patient who does not respond favorably to peripheral sympathetic blockade may achieve symptomatic relief with a continuous sympathetic block or from epidural blockade.
Thermographic techniques provide a large quantity of temperature data, allow comparison of both upper extremities, and permit rapid repeated measurements over time. However, peer-reviewed data do not support the use of thermography as a superior alternative to other conventional diagnostic modalities. Data suggest that higher temperatures indicated by thermography do not correlate with skin surface flow in RSD and that the use of temperature alone cannot distinguish RSD from “chronic upper limb pain.” Thermographic data must be strictly interpreted to be effective. Misleading color-coded asymmetry patterns can be created by inappropriate sensitivity settings; that is, settings should exceed 0.5°C to 1.0°C per color band. Repeated measurements over time from the same subject or subjects are reproducible and consistent, reflect skin blood flow, and correlate with symptoms in a variety of painful pathologic entities. The use of a stressor strengthens thermographic data, which are a reflection of total skin blood flow, , , and significantly improves the predictive and diagnostic value of telethermography.
Objective evaluation of extremity function can be accomplished by using standardized tests and computerized analysis systems. These evaluations document overall functional capacity, verify gross and fine motor skills, and quantitate strength and endurance. Repeating these measures over time provides a sensitive and reproducible methodology that is capable of delineating subtle functional deficits, documenting the effects of intervention (management), and defining the magnitude of permanent impairment. ,
The concept of “acute stage” and “late-stage” CRPS can be helpful, but many patients do not follow a consistent progression from one stage to another. The length of the acute phase of CRPS is variable and is affected by the type of injury, patient coping mechanisms, and treatment. The acute stage encompasses treatment from onset to the development of atrophy and fixed contracture. Treatment of the acute stage is designed to alleviate pain, decrease abnormal arteriovenous shunting, manage edema, and correct nociceptive and/or neuropathic abnormalities. Management of patients with late stage CRPS is divided into (1) pain palliation using chronic medications (e.g., ketamine) or peripheral nerve or centrally implanted devices (e.g., dorsal column stimulators) and (2) correction of the fixed deformity or contracture.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here