Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Primary immunodeficiencies are a group of disorders where the defect lies within the immune system; most of them are caused by single-gene defects.
Because of the complexity of diagnosing a primary immunodeficiency, the cooperation of the clinician, the research scientist, and the clinical pathologist is required.
A logical approach to the diagnosis of primary immunodeficiency disorders based on staged diagnostic testing or clinical picture is necessary because of the complex nature of the immune system.
Preparation for specimen collection, handling, and delivery to a specialty laboratory is required for performing complex immunologic assays.
As a result of technological advances, molecular genetic testing is increasingly employed in diagnosing primary immunodeficiency disorders and can be paramount in guiding treatment.
Primary immunodeficiency disorders (PIDDs), also known as inborn errors of immunity (IEIs), are a rare group of disorders. As per a 2007 population survey the prevalence of PIDDs in the United States is 1 in 1200 persons ( ). The prevalence of PIDD in the United States is estimated at about 38.9 to 50.5 per 100,000 persons with private insurance and 29.1 to 41.1 per 100,000 persons with public insurance between 2001 to 2007 (Kobrynski et al., 2014). The most common immunodeficiency, selective IgA deficiency, has an incidence of 1 in every 1200 healthy blood donors, with IgA levels less than 0.05 mg/dL ( ). Whereas data from European countries shows an incidence of IgA deficiency ranging from 1:163 in Spain ( ) and 1 in 396 in Finland ( ), data from other countries shows incidence of 1:651 in Iran, 1:252 in Nigeria, and 1:965 in Brazil ( ). Data from Canada and Australia estimate the incidence of primary immunodeficiency at 2.1 per 100,000 ( ) and 6.8 per 100,000 and 10.6 per 100,000, respectively ( ; ; ). Chronic granulomatous disease is found in 1 in 200,000 to 250,000 ( ). Prevalence is still unknown for some of the newer immunodeficiencies. Overall, the prevalence of PIDD can vary for each type of PIDD affecting various parts of the immune system in various ethnicities.
The immune system is an interlocking group of immune cells, proteins, and immune functions, in which any defect may result in an immunodeficiency. Immune response in humans is of two types: innate and adaptive. The natural physical barriers of the body are the first line of defense against invading microbes and include skin, mucus at exposed mucosal surfaces, and ciliated cells of the respiratory, intestinal, and genital tracts. In addition to physical barriers, immunologic functions of the body exert continual surveillance through acquired and innate modes of action. Innate responses do not require prior antigen exposure and comprise phagocytic cells (macrophages, neutrophils, and monocytes), complement, cytokines, and acute phase reactants ( ). The adaptive immunologic response is specific, requiring previous antigen exposure for sensitization, and improves with each subsequent challenge from a given antigen. These responses are the result of a proliferation of antigen-specific B and T cells, which are stimulated after cellular receptors bind to antigens. B cells produce antigen-specific antibodies to eliminate extracellular antigens. T cells assist B cells to produce antigen-specific antibodies and also kill cells that are virally infected.
Immunodeficiency disorders can be primary or secondary. Whereas PIDDs are mostly due to single gene defects that directly affect the immune system, secondary immunodeficiencies are due to defects that lie outside the immune system but secondarily affect the immune system. Secondary immunodeficiency disorders can be a result of various causes, including malnutrition, viral infections, hematologic and other types of malignancies, chemotherapy, immunosuppressive medications, and protein losing disorders. On the other hand, most of the primary immunodeficiency disorders are genetic in origin.
The list of IEIs/PIDDs ( ) is expanding rapidly. Based on the immune defect, PIDDs are classified in several categories. International Union of Immunological Societies (IUIS) updates the classification every couple of years. The latest classification for 2020 (January) gives 10 categories of PIDDs with a total of 430 disorders, with 65 newly added disorders in the past 2 years. While individual PIDDs are rare disorders, the expanding number of PIDDs makes it important to keep PIDD in the differential diagnosis.
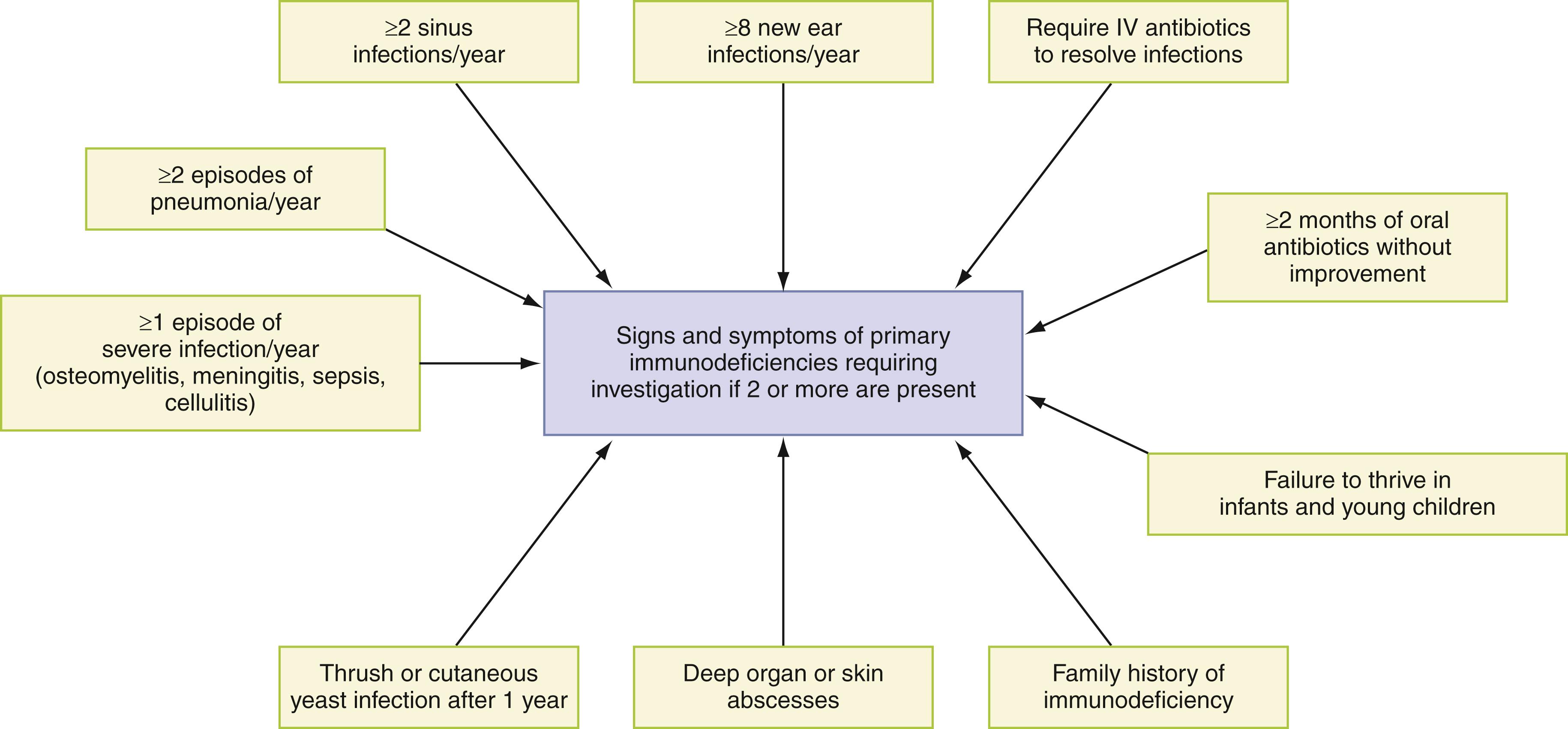
PIDDs are a group of rare disorders that can present with diverse manifestations causing severe morbidity and mortality. Infections are the most common presentation of PIDDs. Malignancy, autoimmunity, autoinflammation, and atopy can be associated with infections or be the sole manifestations of PIDDs. The manifestations can vary in terms of degree of severity, types of symptoms exhibited, or both. These conditions, when unrecognized and left untreated, can result in death from infection or noninfectious complications. Awareness of common clinical presentations can aid physicians in identifying individuals who are more likely to have a primary immune defect. Since PIDDs is a group of rare disorders, identifying children/adults with immunodeficiency presents a significant challenge to physicians. A high index of suspicion for primary immunodeficiencies is recommended for patients with recurrent infections or other features of PIDDs. Most frequently, increased susceptibility to recurrent infection is the first clinical indication of an immune defect. The type of recurrent infection provides insight into the immune defect as noted in Fig. 52.1 and helps guide the clinician in evaluating the patient. Therefore the most critical part of the medical evaluation is obtaining a complete medical history. Clinical signs and symptoms of primary immunodeficiency are presented in Fig. 52.2 . Some clues that favor possibility of immunodeficiency include frequent and prolonged infections, opportunistic infections, and failure to thrive ( ). Infections with an unusual feature such as the need for an adult to have a myringotomy, severe herpes zoster or herpes zoster appearing in a young child, or serious infection in a sterile organ should be considered a warning sign regardless of frequency. A confirmed report of an immunodeficiency disease occurring in a sibling or first-degree relative should also prompt careful clinical assessment and laboratory investigation, even without a history of severe or unusual infections. In one study of 70 patients with PIDDs, approximately 18.6% had a family history of an immunodeficiency ( ). In addition to these warning signs, the physician should be alerted by noninfectious elements in the history that are unusual, such as a history of autoimmune disease, significant lymphadenopathy, granulomas, malignancy, or intractable diarrhea or very early onset of inflammatory bowel disease in a child. These additional signs and symptoms in conjunction with recurrent infections should prompt evaluation for a possible immune defect.
It is important to consider alternative diagnoses in the evaluation of PIDDs. Various features of infections or other symptoms can provide a clue to differentiate PIDDs from other disorders that mimic PIDDs. It is also important to pay attention to the clinical circumstances and not to dwell exclusively on the number or severity of infectious episodes ( ). For example, when an infectious process involves the same anatomic site repeatedly, the cause is more likely to be structural, not failure of one of the host defenses. Examples include chronic osteomyelitis in one location, recurrent episodes of pneumonia that affect only one lobe of the lung, and recurrent infection of a surgical wound. Additionally, some PIDDs, such as chronic granulomatous disease, may present later in life than expected; however, it is still important to keep these disorders in the differential diagnosis ( ).
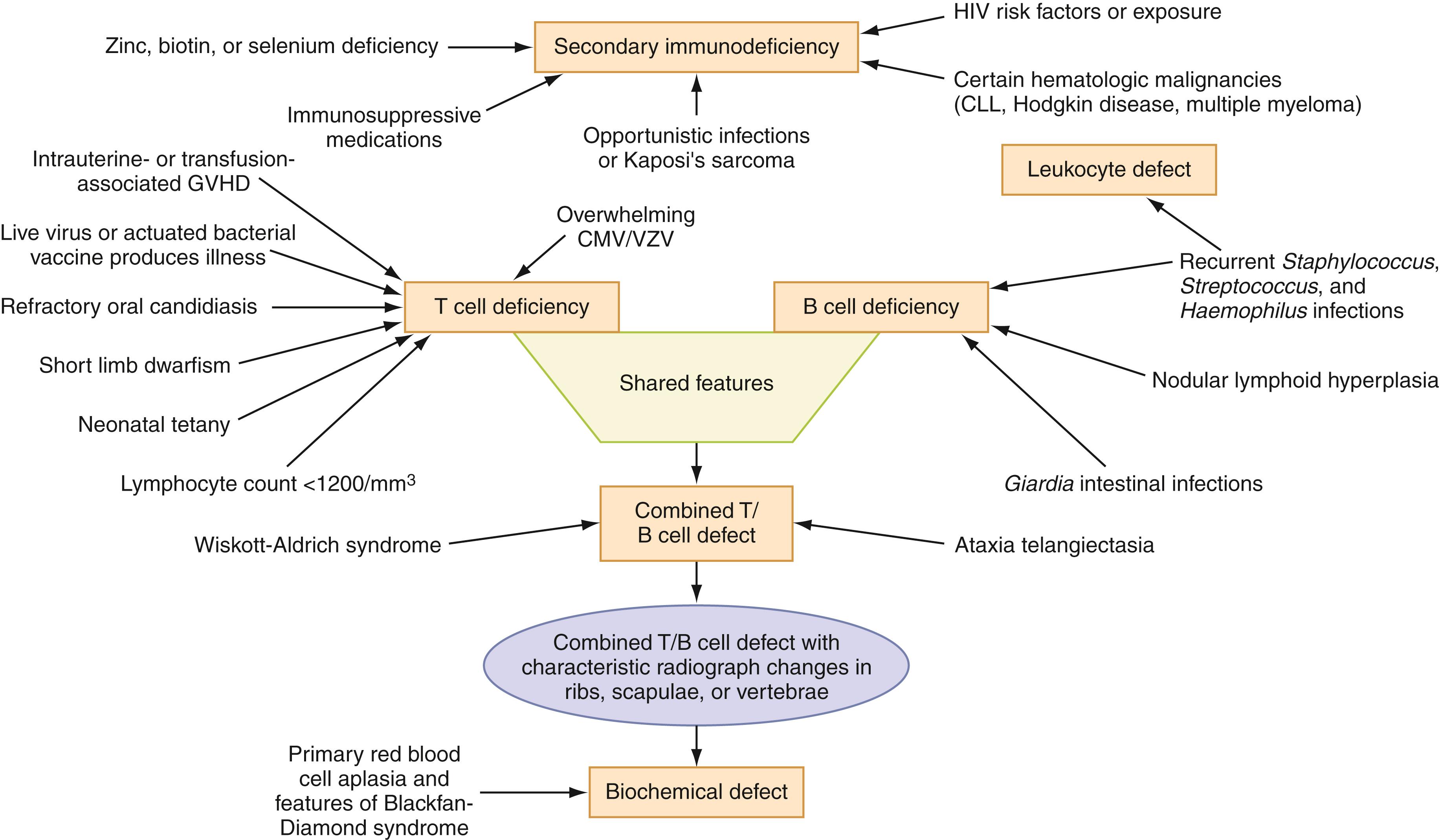
A careful history and thorough physical examination can guide the testing in patients with suspected immunodeficiency. Based on this information, it may be possible to consider specific category/categories of PIDDs during evaluation. The roles of these host defenses and their correlation with different immunodeficiency disorders are presented in Fig. 52.3 .
Based on the previous considerations, when an immune defect is suspected, evaluation of the immune system is best approached in stages, with basic screening tests performed first and more complex testing conducted as indicated. For example, if a patient presents with strong clinical symptoms suggestive of a B- or T-cell disorder but a normal lymphocyte count, further investigation by a lymphocyte enumeration panel is warranted. An overview of this staged approach is given in Fig. 52.4 .

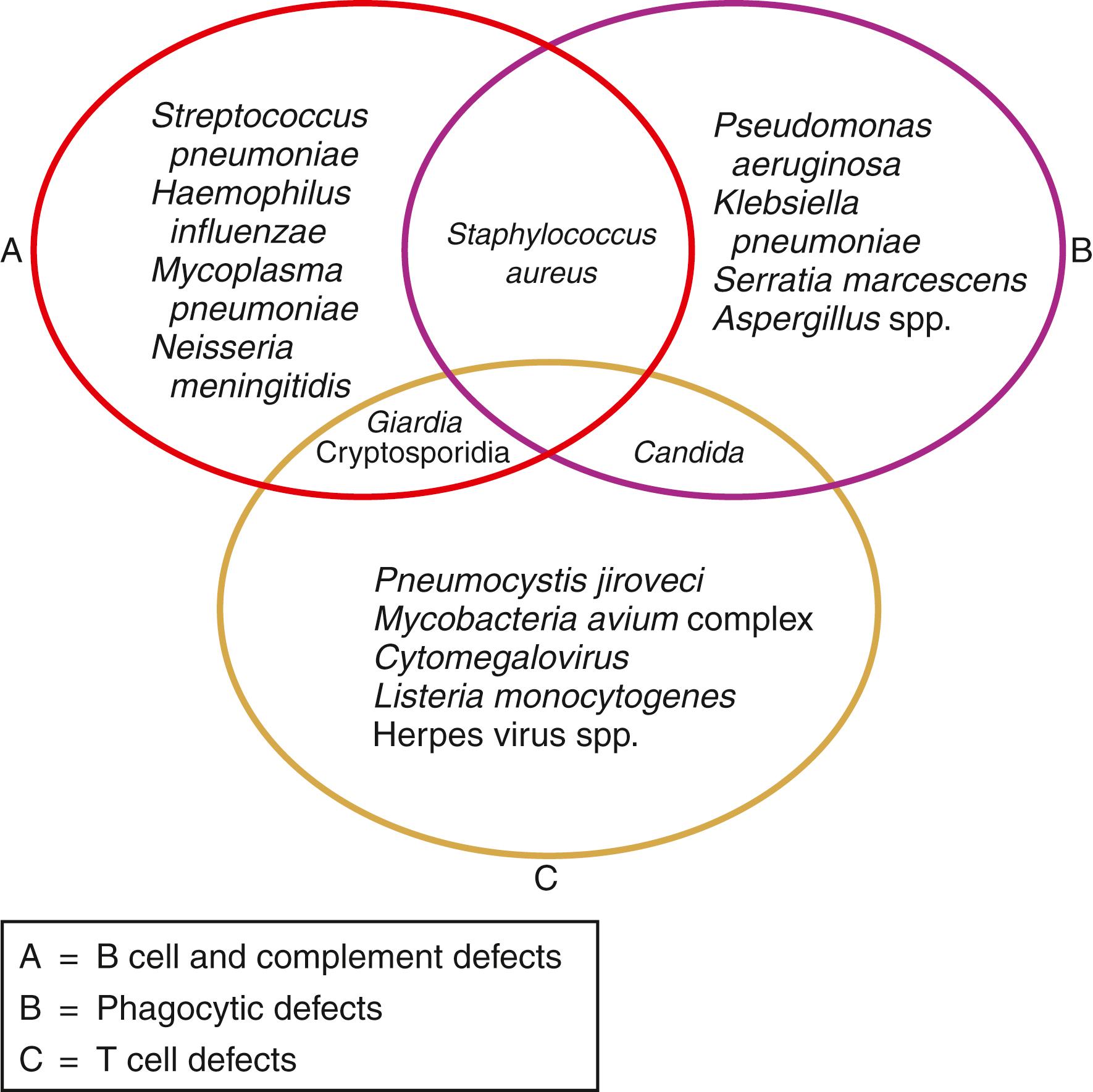
A thorough clinical history provides the primary clues in diagnosing a PIDD. Part of the first visit should include obtaining all previous medical records, including pathology reports or slides, and results of previous cultures and radiographs. The typical history includes recurrent, persistent infections or infections caused by unusual microbes. The clinician will be guided by the types of infection the patient has acquired. Patients with immunoglobulin deficiencies typically report an increased number of bacterial infections. Repeated viral, fungal, or other opportunistic infections are associated with T-cell immunodeficiencies. Patients with phagocytic defects usually report a history of infections with catalase-positive bacteria such as Staphylococcus spp. Complement deficiencies are most frequently associated with infections caused by encapsulated organisms such as Streptococcus pneumoniae or Neisseria spp. infections (see Fig. 52.1 ) ( ).
The physical examination in PIDD may reveal findings that are characteristic for that particular PIDD. Patients with PIDD may appear pale, lethargic, and chronically ill. Infants and young children affected with primary immunodeficiency disorders may present with failure to thrive, so monitoring height and weight is important. Small or absent peripheral lymph nodes, tonsils, or adenoids are indicative of X-linked agammaglobulinemia ( ). Hepatomegaly or splenomegaly may suggest presence of lymphoproliferation. In addition to respiratory infection, the infant may experience rhinitis; conjunctivitis; or hematologic, gastrointestinal, and autoimmune disorders ( ). In contrast, patients with chronic granulomatous disease (CGD) and common variable immunodeficiency present with normal or enlarged peripheral lymph nodes ( ). The genetic defect in Wiskott-Aldrich syndrome (WAS) causes immunodeficiency and thrombocytopenia, resulting in the combination of increased bruising, petechiae, and eczematous skin rashes ( ). Sparse hair and abnormalities of teeth and nails can be indications of ectodermal dysplasia seen in nuclear factor-kappa B essential modulator (NEMO) deficiency ( ). Skin rashes such as livedo reticularis, eczematous rash, ichthyosis, and erythroderma can be signs of various PIDDs.
Immune evaluation depends on several factors. Typical basic tests include complete blood count (CBC) with differential and immunoglobulins. However, based on patient’s signs and symptoms, initial tests may vary. For example, liver or perianal abscess caused by Serratia marcescens is suspicious for CGD, so the testing for CGD can be pursued as an initial test.
The first test performed to examine the immune system is the CBC. A white blood count and differential will provide information on the morphology and number of small lymphocytes (diameter <10 μm). Normal parameters vary, based on the age of the patient, and therefore must be interpreted using age-appropriate reference ranges. Absolute cell counts can provide valuable information. Low absolute neutrophil count can point to immunodeficiencies associated with neutropenia. At a minimum, regardless of age, one expects more than 1500 small lymphocytes per cubic millimeter. Because few (∼10%) of the lymphocytes are B lymphocytes, an absolute lymphocyte deficiency primarily reflects T-cell deficiency. Absolute eosinophil count can be elevated in case of Omenn syndrome, autosomal dominant or autosomal recessive hyper-IgE syndrome (HIES), along with various other primary immunodeficiencies (Navabi & Upton, 2016). Absolute monocyte count can be low in GATA2 deficiency ( ). Anemia and thrombocytopenia can be associated with various PIDDs such as autoimmune lymphoproliferative syndrome, common variable immunodeficiency, and chromosome 22q11.2 deletion syndrome. Autoimmune hemolytic anemia can be seen in Wiskott-Aldrich syndrome. Peripheral smear evaluation can provide valuable information. Large vacuoles in neutrophils are seen in Chediak-Higashi syndrome. Small platelets can be seen in Wiskott-Aldrich syndrome. Howell-Jolly bodies can suggest functional asplenia.
Because antibody deficiency diseases are more common than other immune defects, first emphasis should be placed on investigation of immunoglobulins and their production ( ; ). Quantitative immunoglobulin assays are readily available in all commercial and larger hospital laboratories and require only small volumes of serum to perform ( Chapter 20, Chapter 47 ). Because immunoglobulin values increase with age, comparison with age-matched normal ranges is necessary for correct interpretation ( ).
For the first few months of life, IgG is the major immunoglobulin found in infant serum and is maternal in origin. At approximately 3 months of age, dissipation of maternal IgG concentration, combined with a lag in production by the infant, creates a nadir in IgG levels at about 6 months of age. However, primarily IgM and secondarily IgA are synthesized by the infant; thus the absence of these two immunoglobulins may provide clues for a diagnosis of agammaglobulinemia, characterized by pan-hypogammaglobulinemia. The best understood cause of agammaglobulinemia is a defective BTK gene (known as X-linked agammaglobulinemia) causing arrest of maturing B cells and absence of mature B cells in circulation. Other autosomal recessive forms of agammaglobulinemia are now known to cause similar pan-hypogammaglobulinemia. Immunoglobulin G levels can be low due to primary immunodeficiency (antibody deficiencies such as XLA or common variable immunodeficiency or part of other combined immunodeficiencies) or secondary causes such as iatrogenic (medications such as corticosteroids, antiepileptics, B-cell–targeted therapy such as rituximab, radiation therapy), acute illness (e.g., sepsis), or protein losing conditions (protein losing enteropathy, protein losing nephropathy). While IgG level is most frequently used, IgG subclasses can be assessed (IgG1, IgG2, IgG3, IgG4) but their use is controversial and minimal. IgG3 has short half-life and can be used before 6 months of age to check if an infant can make antibodies (when the rest of the subclasses are maternal in origin).
Often, IgA is not present in significant amounts until 6 months of age; however, after 4 years of age, IgA levels that remain decreased indicate that this is a permanent defect ( ). Selective IgA-deficient patients may be asymptomatic or may have mild symptoms such as increased respiratory or gastrointestinal infection; however, patients may be at risk of developing severe or fatal anaphylactic reactions to plasma in blood products that contain IgA ( ). Such patients, once confirmed for IgA deficiency, would require plasma from other IgA-deficient individuals to avoid future anaphylactic reactions.
Hyper-IgM syndrome is characterized by normal to elevated levels of IgM with markedly low levels of IgA, IgG, and IgE ( ; ; ). Hyper-IgM syndrome is caused by defect in CD40 ligand, CD40, AID, UNG, INO80, and MSH6. In contrast, marked elevations of IgE can be a part of the hyper-IgE syndrome caused by STAT3 loss of function, DOCK8 deficiency, PGM3 defect, TGFBR, ZNF34I, ERBIN deficiency, CARD11 deficiency, interleukin-6 (IL-6) receptor deficiency or IL-6 signal transducer deficiency, and Comel Netherton syndrome ( ; ) but also are common in immunodysregulation, polyendocrinopathy, enteropathy, immunodysregulation polyendocrinopathy enteropathy X-linked syndrome (IPEX), atypical complete DiGeorge syndrome, Wiskott-Aldrich syndrome, and Omenn syndrome ( ). WAS characteristically shows marked elevation of serum IgA combined with low IgM concentrations ( ). Patients with decreased levels of IgM are often found in the context of generalized immune deficiency/dysregulation and autoimmune diseases. While very low IgM levels have been noted in chromosome 22q11.2 deletion, reduced or lacking IgM+ B cells, or impaired IgG antibody responses against pneumococcus polysaccharides, it may be part of another immunodeficiency disorder ( ).
A simple way to assess functional antibody levels in children is to check for isohemagglutinins, commonly of the IgM isotype, directed against ABO antigens that the patient lacks (e.g., assessing anti-A antibodies in a patient with the B blood type), because their presence will indicate active functional antibody production ( ). Isohemagglutinins are naturally occurring antibodies that form at some point after 2 to 3 months of age; however, testing for these antibodies is not valid for children 4 to 6 months of age because of the persistence of maternal immunoglobulin. Additionally, because of relative immaturity, or perhaps inadequate exposure, children with normal immunity may have low concentrations of isohemagglutinins until 5 to 10 years of age ( ).
Radiographs, computed tomography (CT), and magnetic resonance imaging (MRI) studies can be helpful as adjunctive tests in assessing or in monitoring complications of immunodeficiencies. Chronic interstitial pneumonia is common in infants, children, and adults with T-cell deficiencies and should be confirmed on chest radiograph. With time, many patients with primary B-cell deficiency (agammaglobulinemia, common variable immunodeficiency) may develop chronic pulmonary fibrosis or bronchiectasis ( ; ). Chest CT in a patient who has recurrent respiratory tract infection is the best means of investigating for bronchiectasis; if these changes have developed, further investigation is clearly warranted.
In the past, lateral views of the neck were used to assess pharyngeal lymphoid tissue in children. However, radiographs are not recommended for evaluation of cervical lymphoid tissue because the information they provide is more easily obtained by alternative methods. Frontal views of the chest may reveal an absence of the thymic shadow in infants, but because the thymus can shrink easily because of stress, evaluation of thymic size by this method is not reliable. Bony abnormalities, however, can alert the radiologist to an immunologic problem presenting in childhood. A subset of patients with immunodeficiencies (e.g., cartilage hair hypoplasia) involving abnormalities of T cells, or both T and B cells, have short-limbed dwarfism. These patients show characteristic lesions, usually in the metaphyseal regions of the long bones. Patients with infant-onset adenosine deaminase deficiency show readily identifiable skeletal abnormalities of ribs and hips ( ). Bony abnormalities may be present in patients with HIES; these abnormalities include diffuse osteoporosis, fractures that occur with little trauma, craniosynostosis, and midline bony defects ( ).
Immunodeficiency, especially if it has persisted for some time, will often affect the pulmonary system and its function, as previously mentioned. Therefore complete lung volumes, with forced expiratory volumes and diffusing lung capacity for carbon monoxide (DLCO), should be tested even if the chest radiograph is normal. Patients with both T- and B-cell defects are subject to pulmonary infection, often resulting in bronchospasm, restrictive or obstructive disease, or combinations of these abnormalities. Those patients who are both IgA and IgG2 deficient have been shown to have significantly abnormal pulmonary function tests, and a causal relationship may be seen between these deficiencies and deterioration of lung function ( ).
Hair microscopy can reveal pigmentation issues noted in familial hemophagocytic lymphohistiocytosis syndromes such as Chediak-Higashi syndrome, Hermansky-Pudlak syndrome, and Griscelli syndrome. Clumping of hair pigment can also be noted in cartilage hair hypoplasia.
Absence of plasma cells on biopsy of intestine can be noted in antibody deficiencies such as common variable immunodeficiency. Pulmonary alveolar proteinosis can be noted on bronchoalveolar lavage/lung biopsy in cases with GATA 2 deficiency or CSF2RB deficiency. Biopsy of any affected tissue may be helpful.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here