Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
When Siegal reexamined the original second century Greek text of Galen, he pointed out that Galen was familiar with many aspects of the fetal circulation even though he did not realize that blood circulated. Galen understood that fetal blood was aerated in the placenta and that blood was diverted away from the liver by a short vessel connecting the portal to the inferior caval vein. He also knew that blood passed through the oval foramen to bypass the right ventricle and reach the left side of the heart directly. He realized that some blood still entered the right ventricle and pulmonary trunk, from whence it was shunted into the aorta through a special fetal channel, thereby bypassing the lungs. The quotation at the beginning of this chapter, taken from Siegal, provides a clear indication of Galen's understanding of the dramatic readjustment of the circulation at birth.
Nevertheless, Botallo is the name we usually associate the persistently patent arterial duct. In fact, Botallo described postnatal patency of the oval foramen! It was by a series of misinterpretations and careless translations that his name became quite unjustifiably attached to the arterial duct ; the attribution of the discovery should more accurately be called an independent rediscovery. William Harvey, who was a pupil of Fabrizi d'Acquapendente in Padova for 2 years, synthesized previous detailed descriptions of fetal cardiac anatomy from his mentor in his own writings. His genius resided in proposing the concept of active circulation of the blood. He was well aware of the large size of the arterial duct prior to birth and the fact that blood flowed through it from right to left during fetal life. Harvey was incorrect only in his belief that flow of blood to the lungs was completely lacking in the fetus. Subsequently, Highmore, a friend of Harvey, described closure of both the oval foramen and the arterial duct as occurring with the onset of respiration, believing the arterial duct to collapse as a consequence of blood being diverted to the lungs. Since then, several ingenious theories have been put forward to explain closure of the duct, all being based on postmortem appearances and all invoking mechanical factors. Virchow first suggested that the closure results from contraction of its mural smooth muscle, and Gerard introduced the concept of two-stage closure, in which functional constriction is followed by anatomic obliteration. Thanks to Huggett, we came to understand the role of oxygen in causing functional closure by muscular contraction.
As early as 1907, in an address to the Philadelphia Academy of Surgery, John Munro had suggested surgical ligation of the persistently patent arterial duct. After an interval of 31 years, the first attempt at surgical closure was made in a 22-year-old woman with bacterial endocarditis. Unfortunately, although the patient survived the surgery, she died a few days later from complications of the infection. As a result, Robert Gross of Boston first successfully ligated a patent duct in a 7-year-old child with intractable heart failure. He thereby introduced an amazing era of progress in the surgery of congenital malformations of the heart. No historical review of the arterial duct, however brief, would be complete without recognizing the importance of Gibson's exquisite description of the murmur that typifies its persistence, notwithstanding the fashion in which Gibson's initial account has subsequently been misquoted.
From time to time, authors have argued that the terms “patent” or “persistent” are redundancies that should be avoided when describing the arterial duct. In our view, this oversimplifies the situation, and both terms retain their value. Persistence implies that the duct is present after the time of its expected closure and therefore distinguishes a pathologic from a physiologic state. The concept of patency remains useful in the perinatal period, especially in the premature infant in whom the term can be used to signify a duct that is functionally open, as opposed to one that is functionally closed but retains the potential to reopen.
Approximately two-thirds of the fetal cardiac output originates from the right ventricle, with only 5% to 10% passing through the lungs. As such, the majority of right ventricular output passes through the arterial duct into the descending aorta, and its presence is essential for normal fetal development, permitting right ventricular output to be diverted away from the high-resistance pulmonary circulation. Premature construction or closure may lead to right heart failure, resulting in fetal hydrops.
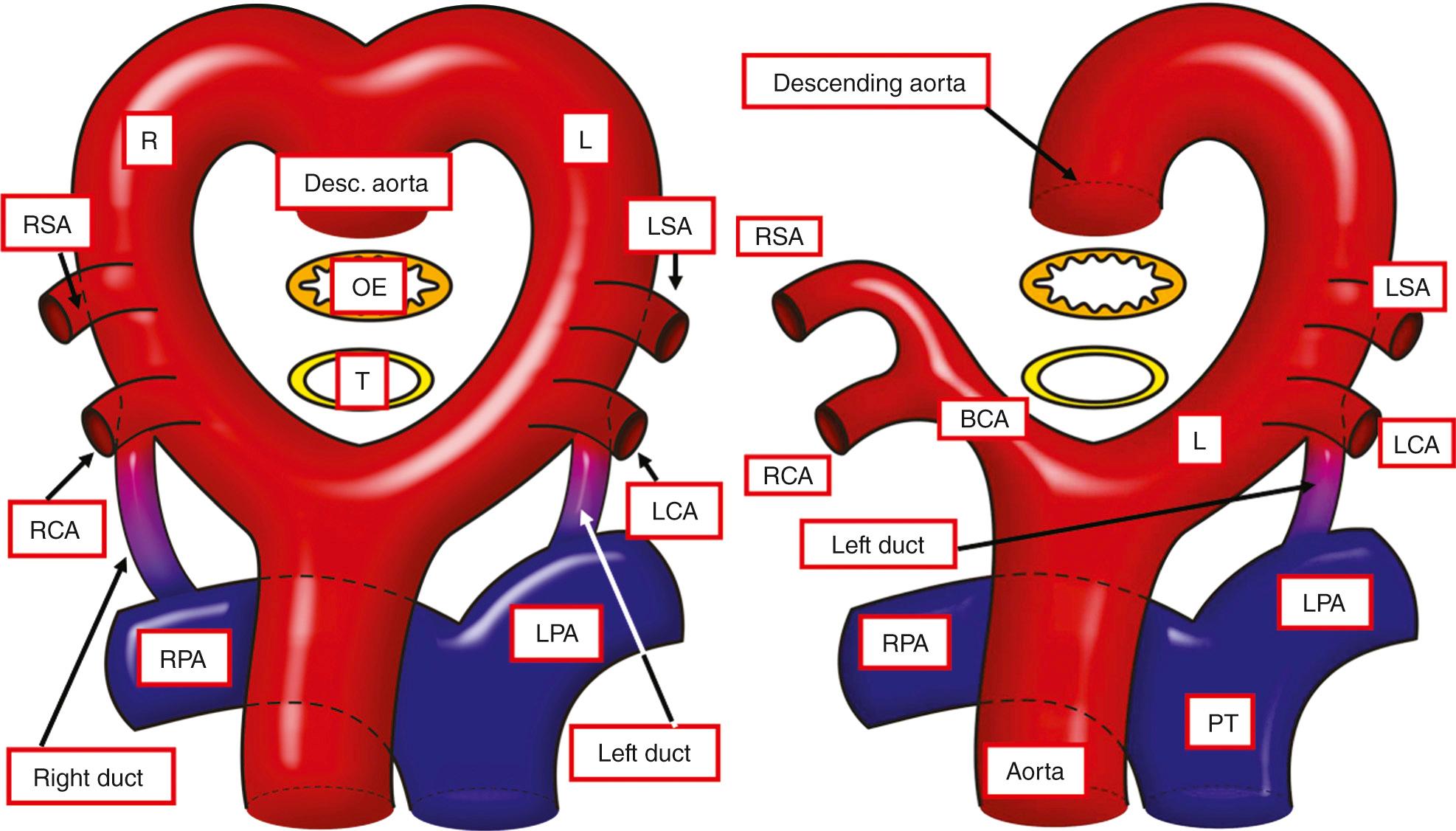
During early fetal development, five arterial arches link the aortic sac with the paired dorsal aortas, although all arches are never present simultaneously. The arches are numbered 1 to 6. However, to the best of our knowledge, there is but a single example identified of a fifth arch artery that occupies a discrete segment of pharyngeal mesenchyme. It has become conventional to interpret several congenital malformations on the basis of persistence of this enigmatic entity. These examples are discussed in Chapter 48 . The initial symmetric arrangement of the pharyngeal arch arteries does provide the basis for the understanding the pattern of the hypothetical double arch proposed by Edwards to explain vascular rings and slings. This bilaterally symmetric arrangement, with paired brachiocephalic arteries and arterial ducts ( Fig. 41.1 , left ), is usually transformed to the configuration seen in postnatal life by the disappearance of some arterial segments and realignment of others (see Fig. 41.1 , right ).
Therefore the normal arterial duct develops from the dorsal portion of the left sixth arch. From their inception, the sixth arches are associated with the developing lungs, with the arteries feeding the developing lungs developing within the anterior wall of the mediastinum. The arteries take their origin of the floor of the aortic sac, which initially feeds also the bilateral arteries of the sixth arches ( Fig. 41.2 , left ).
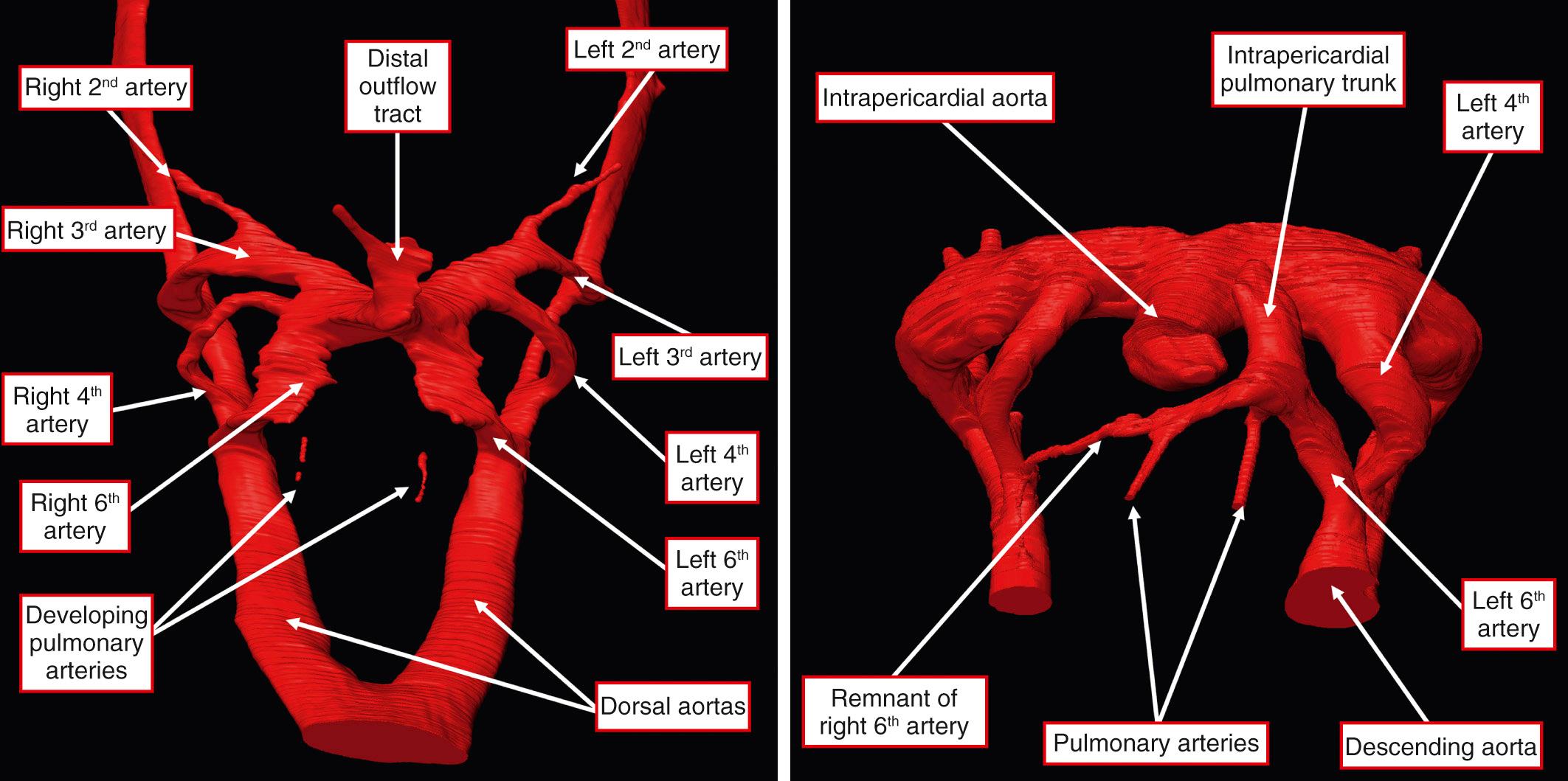
When the developing distal part of the outflow tract is divided to form the intrapericardial components of the aorta and pulmonary trunk, the sixth arches, originating from the caudal part of the aortic sac, are placed in continuity with the pulmonary channel. A significant event in the appropriate connection of the pulmonary arteries with the intrapericardial pulmonary trunk is the obliteration and disappearance of the right sixth arch. On the left side, the artery of the sixth arch persists as the arterial duct, with the pulmonary arteries left in continuity with the channel from the pulmonary trunk to the left sixth arch (see Fig. 41.2 , right ).
All developmental anomalies of the aortic arch, including those associated with an abnormally situated duct, are well explained on the basis of the hypothetical double arch system devised by Edwards, irrespective of whether the duct itself is patent or represented by the arterial ligament. The various possibilities are discussed in Chapter 47 , including those in which there is persistence of both sixth arches, producing bilateral arterial ducts. Such circumstances are rare and are always associated with intracardiac anomalies. Typically, bilaterally persistently patent arterial ducts supply discontinuous pulmonary arteries in the setting of tetralogy of Fallot with pulmonary atresia, albeit that such bilateral ducts can also be found in association with isolation of a subclavian artery.
However, the arterial duct does not always persist on one or other side. Its absence was first described as a postmortem finding in 1671, being seen in a grossly malformed infant with an extrathoracic heart and tetralogy of Fallot described by Nicolas Steno. Absence of the duct is a typical finding in the syndrome of tetralogy of Fallot with so-called absent pulmonary valve and dilated pulmonary arteries. It was thought that the absence of the duct, and hence absence of any “overflow,” explained the dilated pulmonary arteries, but the pulmonary arteries can be dilated even when the duct is present and patent. The duct is also absent in the majority of patients having a common arterial trunk. Absence of significant flow through the duct in the presence of a larger aortopulmonary connection permits the duct to disappear early in fetal life. However, in more complex varieties of common arterial trunk, such as those with so-called absence of one pulmonary artery or those patients with an associated interruption or atresia of the aortic arch, patency of the duct is essential to maintain both the systemic and pulmonary circulations (see Chapters 43 , 45 , 69 ).
A number of teratogens are known to influence the development of the duct, including rubella, alcohol, amphetamines, the anticonvulsant hydantoin, and topiramate, with the duct being most sensitive from 18 to 60 days of gestation. Absence of the duct has been induced experimentally in chick embryos by the administration of agonists of β-adrenoceptors, leading to the suggestion that the teratogenesis is mediated by cyclic adenosine monophosphate.
Patency is maintained by the relatively low fetal oxygen tension and cyclooxygenase-mediated products of arachidonic acid metabolism, primarily prostaglandin and prostacyclin, whose levels are high because of production by the placenta and decreased metabolism in the fetal lungs. Produced both locally, in ductal tissue, and circulating, these mediators cause vasodilation through interaction with prostanoid receptors.
In the fetus, and in the neonate prior to its closure, the arterial duct is a short and wide vessel of variable length. It connects the pulmonary arteries to the lesser curve of the arch of the aorta, terminating at the point of transition from the isthmus to the descending aorta, distal to the origin of the left subclavian artery ( Fig. 41.3 ).
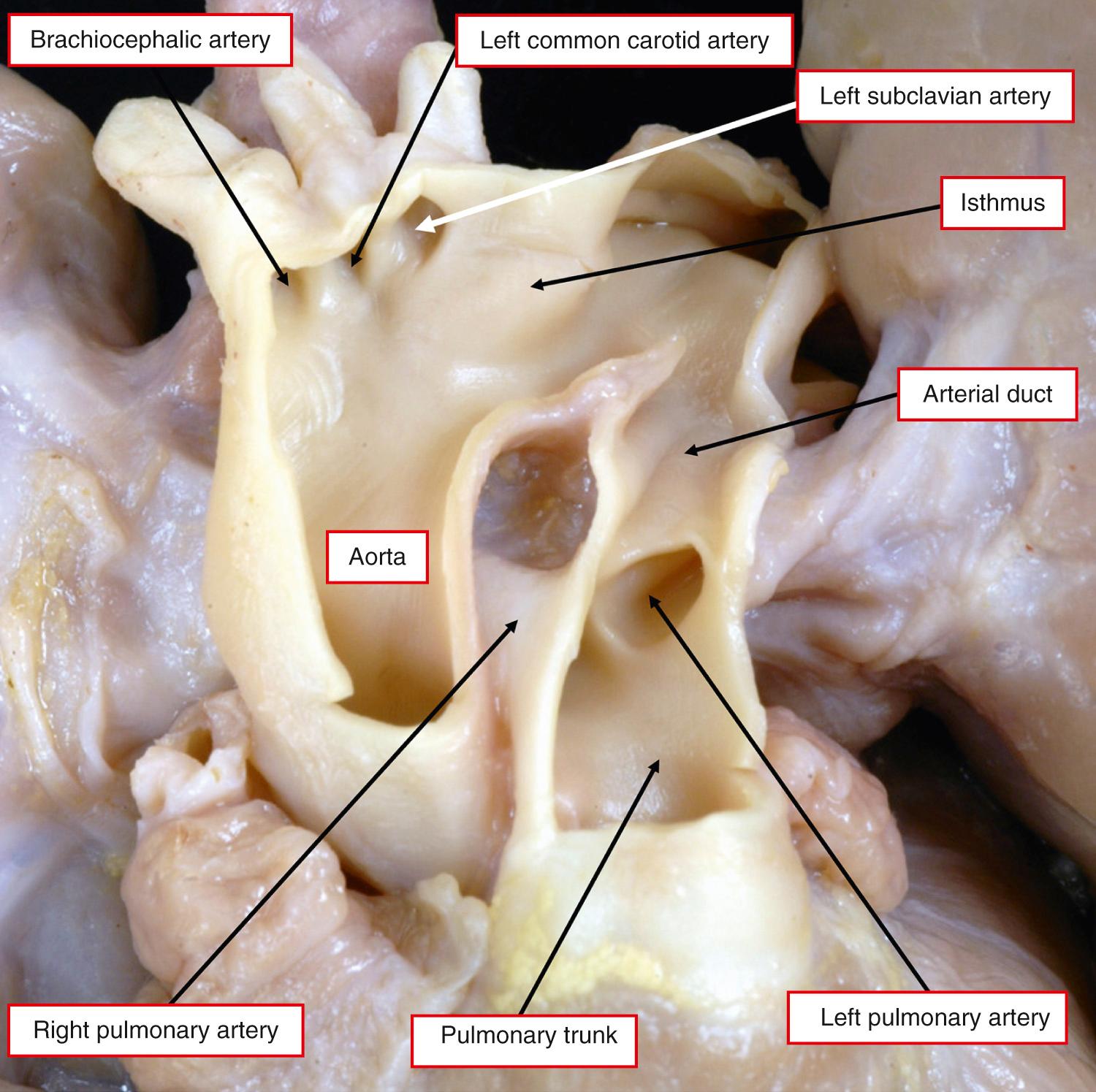
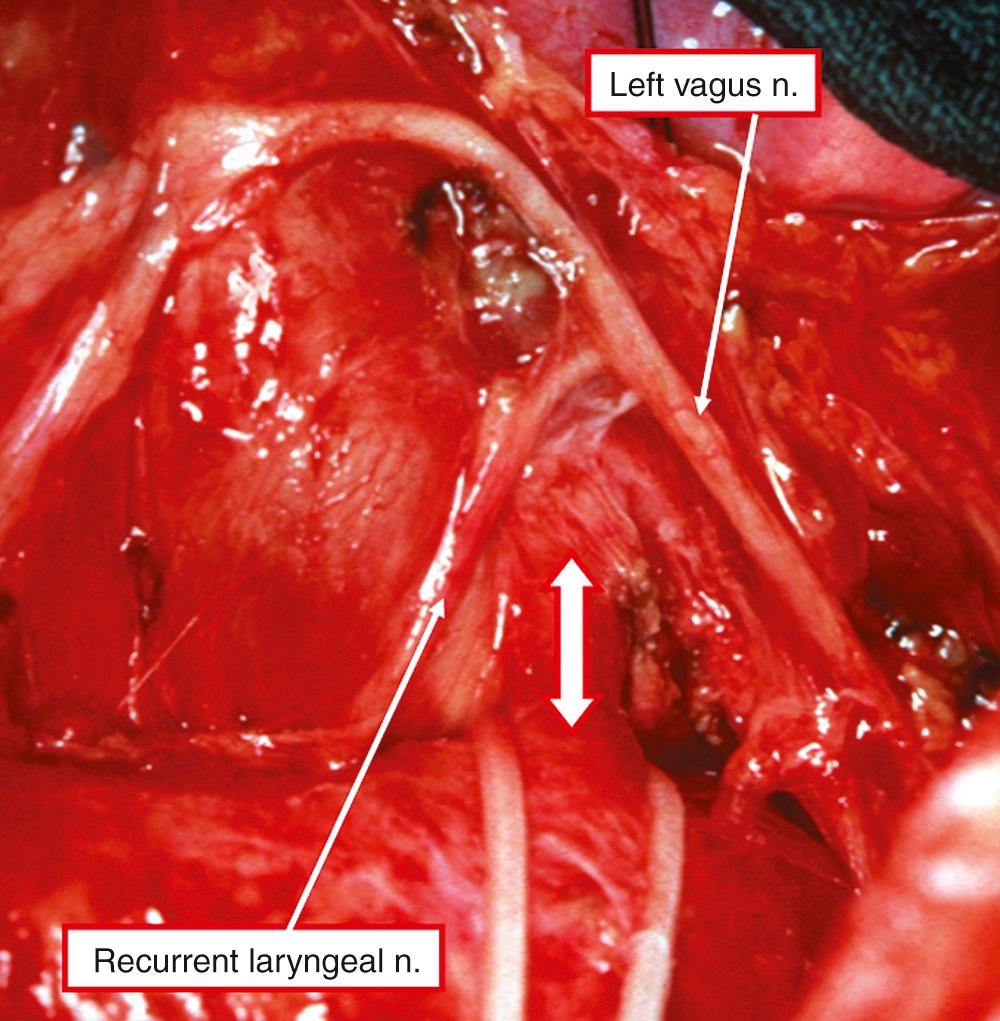
Prior to birth, the duct is very much the direct continuation of the pulmonary trunk, with the left and right pulmonary arteries taking origin as smaller branches from the trunk. Posteriorly, the duct is related posteriorly to the left main bronchus, while anteriorly it is crossed by the vagus nerve. This gives off the left recurrent laryngeal nerve, which encircles the duct before ascending behind the aortic arch into the neck ( Fig. 41.4 ).
The pulmonary arterial end of the vessel is covered by a reflection of the pericardium.
In terms of its microscopic structure, the duct is a muscular artery endowed with an intima, media, and adventitia, differing markedly from the adjacent pulmonary trunk and aorta ( Fig. 41.5 ).
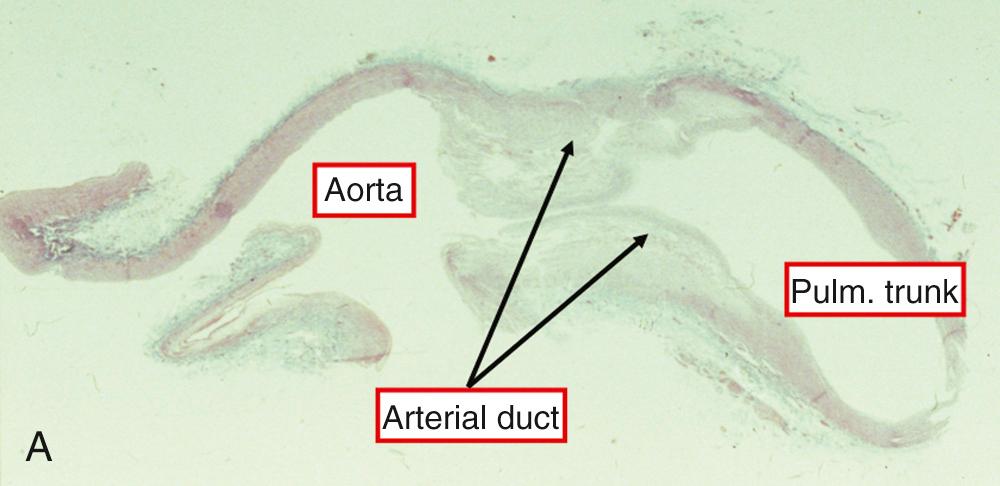
The media of the aorta is composed mainly of circumferentially arranged elastic fibers, whereas the media of the duct consists largely of spirally arranged smooth muscle cells, some with circular and others with longitudinal orientation, with an increased content of hyaluronic acid. The intimal layers are thicker than those of the adjoining vessels and contain increased amounts of mucoid substance. In the newborn, the tissues are rather loosely arranged, with a well-defined internal elastic lamina that may be single or focally duplicated, with small interruptions encountered regularly. No collagen is seen in the media by light microscopy, but abundant material that strains positively for acid mucopolysaccharides is observed between the muscle and the elastic laminas. Electron microscopy reveals fine collagen fibrils lying between adjacent lamellas of smooth muscle cells and elastin. During the second half of gestation, the smooth muscle cells show decreasing evidence of secretory activity, and increasing maturation of their contractile elements.
It is known that vessels cannot close by isolated contraction of circularly arranged muscle, so coincident shortening of the less abundant longitudinally arranged muscle fibers is critical to effective closure. The duct is innervated mostly by adrenergic fibers, supplying largely the adventitia and outer media, with cholinergic fibers being extremely sparse or totally absent. Vessels are also found in its walls that may have a role in fueling contraction at birth. Some degree of hyperemia of these vessels is common in newborn infants.
There is controversy with regard to the structure of the intimal layers during fetal life. Eccentrically placed intimal cushions, or mounds composed of smooth muscle and elastic tissue, have been described by many authors, with suggestions made that the formation of these mounds precedes normal ductal closure subsequent to birth. However, it is questionable whether the intimal cushions are produced during normal fetal maturation. When account is taken of the high flow of blood through the fetal duct, it is difficult to conceive that prominent protrusions of intima into the lumen could exist without inducing turbulent flow and a bruit. No such bruit is heard in the undisturbed ducts of fetal lambs at term. Furthermore, in many of the studies suggesting the presence of intimal cushions, the tissues have usually been subjected to one or more perturbations, such as relatively slow fixation, mechanical stimulation, or cessation of circulation with loss of intraluminal distending pressure. On this basis, the existence of intimal cushions as prenatal structures has been challenged. In a series of experiments, Hornblad and colleagues showed that, independent of the degree of closure, the lumen of the duct remained round, was without deformation, and showed no evidence of formation of mounds. Wall thickness increased at the time of closure, while the internal elastic lamina became corrugated, especially in the midportion of the vessels. A decrease in the lumen was associated with accumulation of endothelial cells within the lumen. They concluded that closure was aided by passive central displacement of endothelial and inner medial cells but that no part of the medial layer was prepared prenatally for this process. These findings endorsed earlier studies in the human, which suggested that the cushions appeared as a normal reparative reaction to distending forces during fetal life.
At birth, the vessel unequivocally constricts. The intimal thickenings, or cushions, become irregular ridges protruding into the lumen, running mainly lengthwise. By their extrusion, they exert traction on the media, causing disorganization and formation of mucoid lakes (see Fig. 41.5 ). Anatomic obliteration follows functional closure. The process begins with necrosis of the inner wall, followed by the formation of dense fibrous tissue. The lumen is progressively obliterated by a process of fibrosis, probably representing organization of mural or occlusive thrombus. Eventually, the duct becomes converted into a fibrous strand, the arterial ligament, which may become calcified. Anatomic obliteration may take several weeks to complete. Approximately two-thirds of ducts are normally obliterated by the age of 2 weeks and almost all by 1 year.
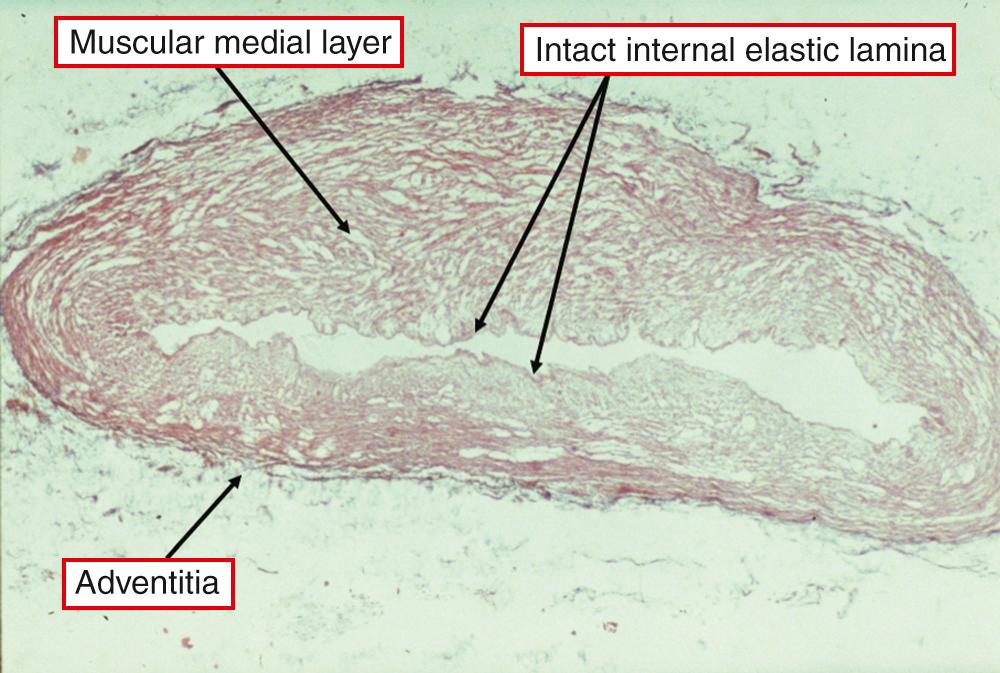
As stated earlier, in normal circumstances all ducts should be converted to an arterial ligament within the first year of life, with two-thirds closing in the initial 2 weeks. However, some ducts never close. These are the channels best described as showing persistent patency. Gittenberger-de-Groot found the internal elastic lamina to be intact in some, but not all, of the persistently patent ducts studied histologically, along with a sparsity of intimal cushions ( Fig. 41.6 ). Bakker had noted similar findings, describing them in terms of “aortification.”
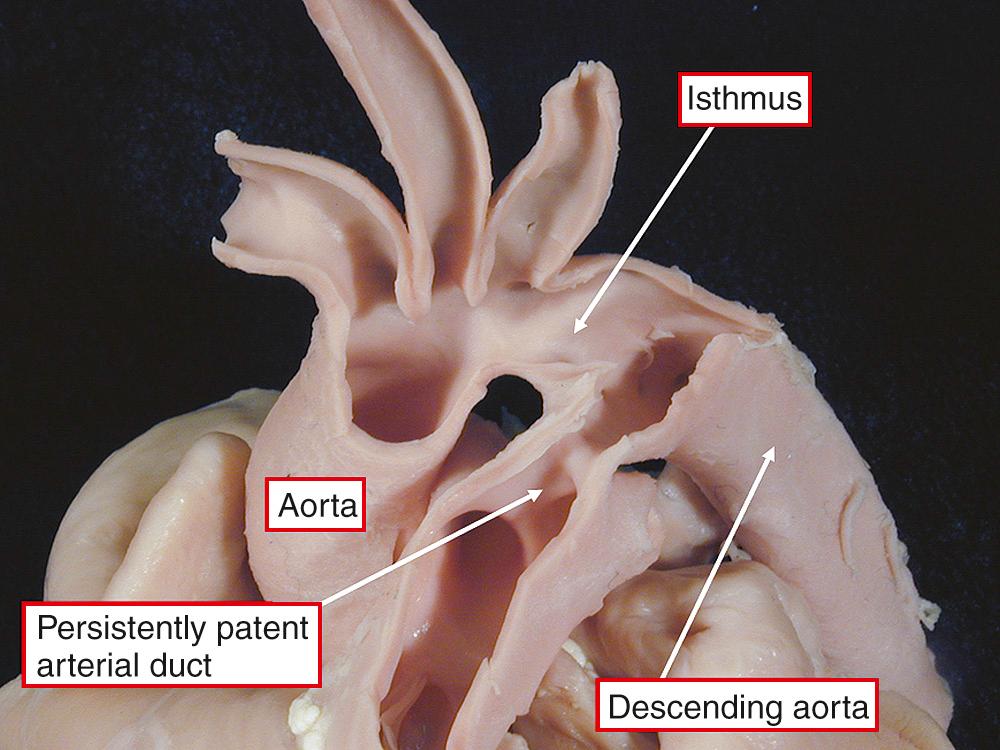
The duct, if persisting as a patent structure, joins the pulmonary arteries to the descending aorta in the fashion seen in the neonate ( Fig. 41.7 ). The channel itself can vary markedly in its width ( Fig. 41.8 ).
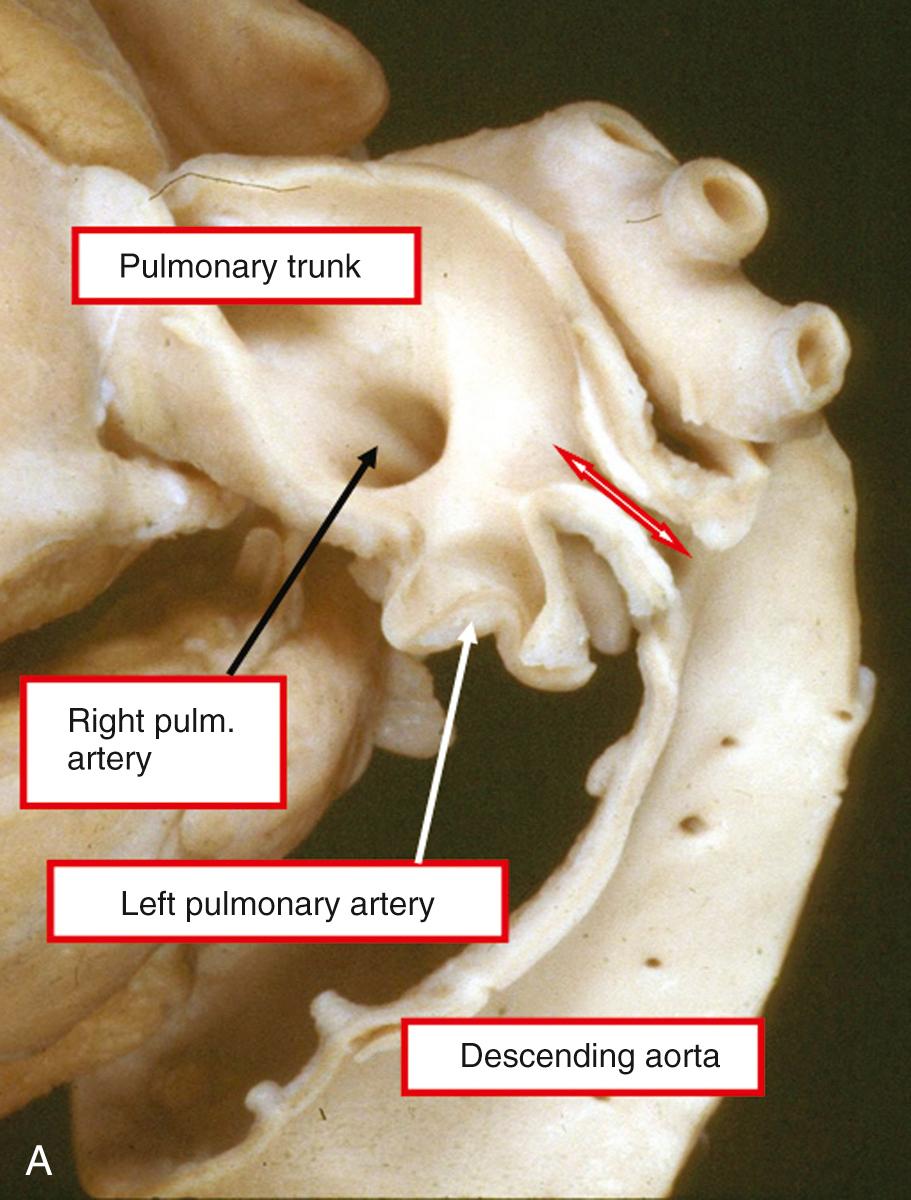
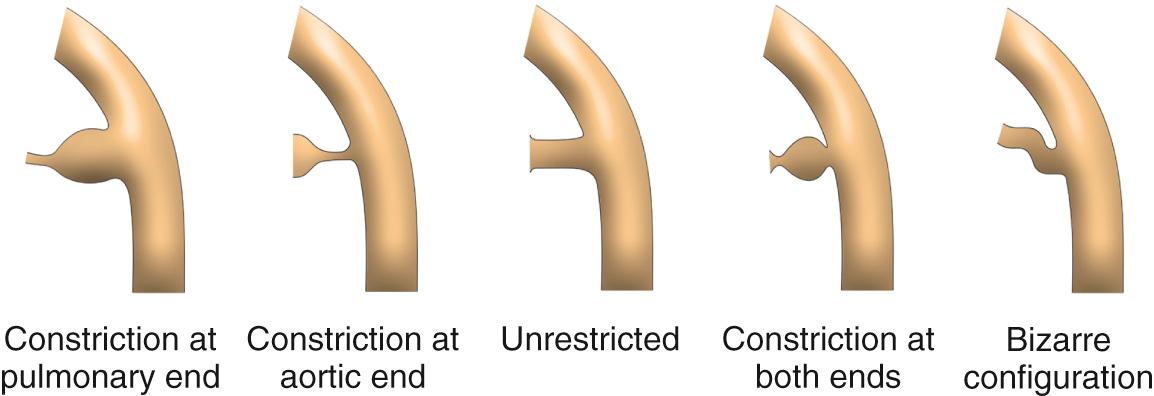
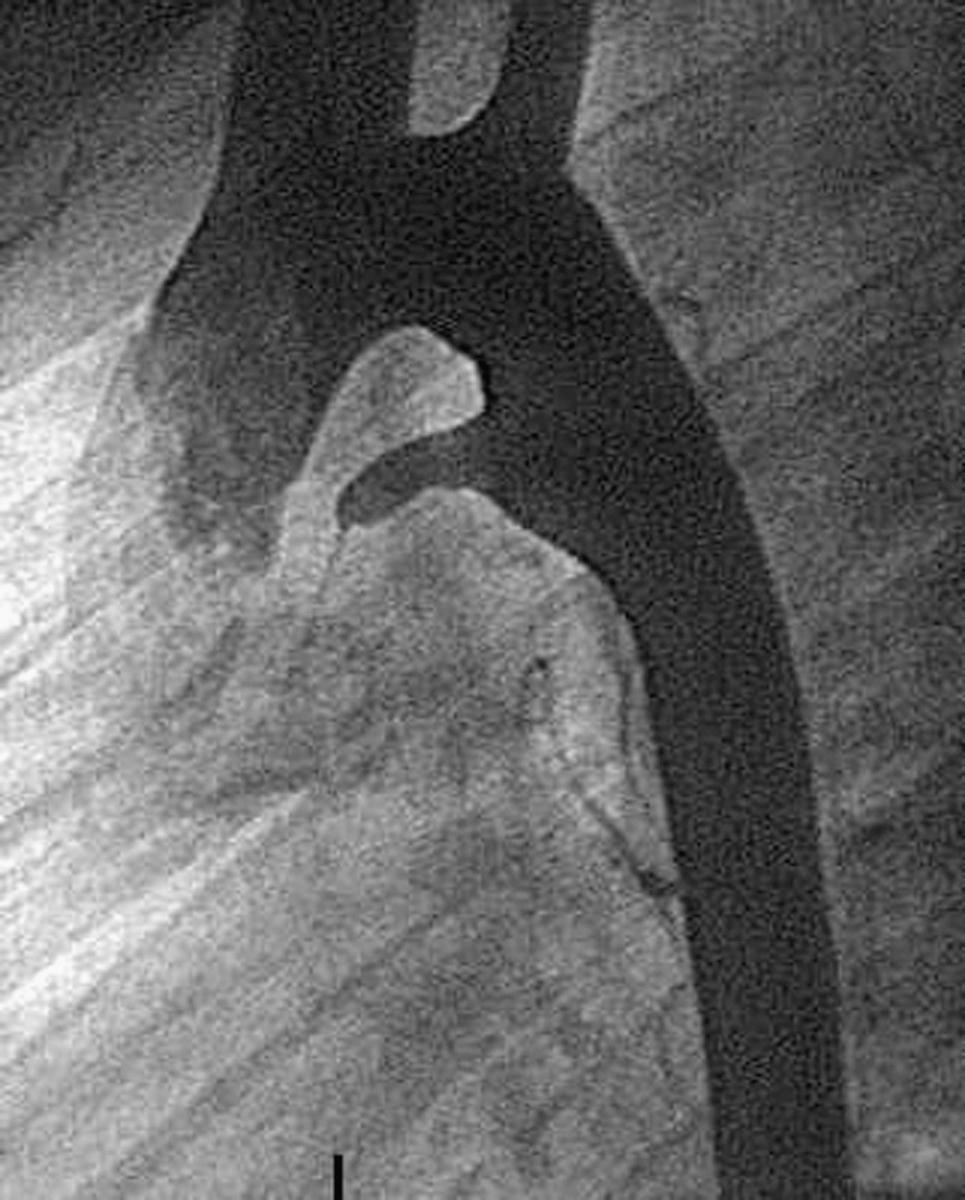
The duct can also vary considerably in its shape. Study of a large number of angiograms from patients with persistently patent ducts undergoing interventional closure at the Hospital for Sick Children, Toronto, showed that the most frequent pattern was to find a constriction at the pulmonary end of the duct. This pattern was seen in two-thirds of cases. In just less than 20%, a constriction was found at the aortic end of the duct, and in just less than 10% the lumen was unrestricted. In just less than 5%, there was a constriction at both ends, and the remaining patients showed bizarre patterns not lending themselves to classification ( Fig. 41.9 ). Unique to the premature infant, the duct is elongated and unrestrictive, with a slight cranial angulation followed by a caudal turn as it connects to the pulmonary artery.
In the past, persistently patent arterial ducts were often the nidus for infectious endocarditis, but this complication is now extremely rare in developed countries. Ducts could also become aneurysmal and elongated ( Fig. 41.10 ), but this is also now a rare finding, as is ductal rupture.
The factors responsible for persistent ductal patency beyond the first days of life are not fully understood. An increased incidence is seen in the premature neonate due to physiologic factors more related to prematurity rather than an inherent abnormality of the duct. In infants born at term, persistent patency occurs sporadically, but there is increasing evidence that genetic factors, or prenatal infection, play a role in many children. The estimated incidence lies between 1 in 2000 and 1 in 5000 live births. Persistent patency of the duct accounts for approximately 12% of all congenital cardiac malformations. The most extensive study from a relatively homogeneous population was that performed by Carlgren, when he charted the incidence of congenital cardiac disease to children born in the Swedish City of Gothenburg. Persistent ductal patency was the third most common lesion identified, representing approximately 0.04% of live births. If children with the so-called silent duct are included, found incidentally by echocardiography performed for another reason, the incidence may be as high as 1 in 500. A significantly higher incidence of ductal patency is also seen in infants born with low weight, patency being found in almost half of infants weighing less than 1750 g at birth, and up to 80% of those weighing less than 1200 g (see also Chapter 15 ).
Unlike premature infants, as noted earlier, where persistent ductal patency is more often due to developmental immaturity, in the infant born at term there is likely a structural abnormality. There is also an increased frequency in several genetic syndromes, such as those with defined chromosomal aberrations, examples being trisomy 21, 14q- and 4p- syndromes, the Rubinstein-Taybi and CHARGE syndromes, single-gene mutations such as Carpenter and Holt-Oram syndromes, and X-linked mutations such as incontinentia pigmenti. Although most cases of persistent patency are sporadic, many are also believed to be due to a multifactorial inheritance pattern, with a genetic predisposition and an environmental trigger that occurs at a vulnerable time. Females predominate in a ratio of greater than 2 to 1. The mode of inheritance appears to be autosomal recessive with incomplete penetrance. In a family having one sibling with a patent duct, there is approximately a 3% chance of a persistent duct in a subsequent offspring and a higher risk to the offspring given one affected parent, some 45 times greater than the that for the general population. The risk to further children in sibships where two children have been affected is probably in the order of 10% and increases with each affected child.
Maternal rubella during the first trimester of pregnancy, particularly in the first 4 weeks, is associated with a high incidence of a persistently patent arterial duct. The histology resembles that of a very immature duct, with an extensive subendothelial elastic lamina, thought to retard anatomic sealing. Additional environmental factors have been reported associated with persistence, such as in fetal valproate syndrome, or after thalidomide. A patent arterial duct may be more likely to be found in infants born at high altitude.
Persistent patency results in shunting of blood from one side of the circulation to the other, the volume of flow depending on the length and internal diameter of the duct and on the systemic and pulmonary vascular resistances. Because pulmonary resistance is usually much lower than systemic, the flow is from the aorta to the pulmonary trunk. Hence flow to the lungs is increased, resulting in left atrial and left ventricular volume overload. If the duct is widely patent, flow depends entirely on the ratio of resistances. Right ventricular failure may occur in the presence of a large duct with pulmonary hypertension or pulmonary edema, and an elevated left atrial pressure. In most patients, the duct is partially constricted and the major factor limiting flow. Under these conditions, pulmonary arterial pressure is normal or only mildly elevated. Symptoms and clinical findings are largely determined by the magnitude of the shunt.
Most patients are asymptomatic, the lesion recognized with detection of the characteristic murmur. Occasionally, there may be a history of prematurity or asphyxia during birth. Children with large shunts may fail to thrive, experience difficulty with feeding during infancy, and frequently suffer recurrent infections of the upper respiratory tract. Occasionally, congestive heart failure develops.
There is retarded growth in approximately one-third of children. They are acyanotic in the absence of complicating factors. The peripheral pulses are easily palpable, with a rapid upstroke and decay. There is a widened pulse pressure, with lowering of the diastolic component. Arterial pulsation in the neck may be prominent in those with large shunts. The precordial examination reveals an active cardiac impulse, with the forceful cardiac apex displaced to the left. When the shunt is small, the only abnormal finding may be the murmur. The continuous, or “machinery,” murmur of the uncomplicated persistent duct is best heard in the left infraclavicular area, although it is occasionally maximal at the third left interspace. Gibson's description of the murmur as quoted by Tynan :
It begins quite obviously after the commencement of the first sound. It is continued during the latter part of that sound and the whole of the short pause. It persists throughout the second sound and dies away gradually during the long pause. The murmur is distinctly rough and thrilling in its character. It begins, however, somewhat softly, and increases in intensity to reach its acme just about, or immediately after, the incidence of the second sound, and from that point wanes until its termination. The second sound can be heard to be loud and clanging and when carefully analyzed it is the pulmonary part of that sound which is accentuated.
Turbulent flow through the duct itself causes the murmur. Additional murmurs may be present due to increased flow across the aortic valve, producing an ejection systolic sound, and across the mitral valve, giving a diastolic murmur with loud onset. The systolic component of the continuous murmur may be transmitted into the neck, may be associated with a thrill in the second left intercostal space, or may increase in intensity during inspiration due to a fall in pulmonary vascular resistance. Many patients with loud continuous murmurs also have multiple “clanging” sounds. These are relatively localized to the pulmonary area and are most frequent in the second half of systole, corresponding to the period of peak flow within the duct. Neill and Mounsey attributed these sounds to the turbulence caused by the “head on” collision of opposed flow from the duct and the right ventricle and named them eddy sounds. These auscultatory findings are only for an uncomplicated persistent duct in a child. It should be remembered that these features may differ in infancy or be altered by the development of complications.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here