Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Although hematogenous spread is the most common cause of osteomyelitis in most other areas of the body, contiguous spread and direct implantation are the most common in the foot and ankle. Direct implantation can occur from puncture wounds or deep lacerations, open fractures, and surgery or injection procedures. The vast majority of osteomyelitis involving the foot and ankle in diabetic patients, however, occurs through contiguous spread from adjacent ulceration and subsequent soft tissue infection.
The worldwide prevalence of diabetes exceeds 200 million and is expected to rise to more than 300 million in the next 20 years, which is a huge economic health burden. It is estimated that 7% of the American population is afflicted with diabetes, and the prevalence of diabetes-related complications will continue to rise with the increasing longevity of this population. The lifetime risk of developing a pedal ulcer in diabetic patients is estimated at 25%, the most common risk factor for subsequent amputation. More than one million amputations are performed each year for diabetes-related foot problems, and diabetes is the most common cause of amputations in the United States. In addition to decreased quality of life after pedal amputation, there is a 50% incidence of serious complication involving the contralateral foot within 2 years, resulting in a 50% to 66% incidence of amputation within 5 years. This reflects the systemic nature of the disease but appears to also be related to shifting of weight-bearing onto the intact extremity after the initial surgery. As a result, over the past decade, there has been increasing emphasis on managing these patients earlier in their disease course with interventions, including ulcer prevention and care and revascularization procedures. As soon as infection is suspected, aggressive medical and surgical management is instituted, including débridement and partial, foot-sparing amputation intended to preserve functionality.
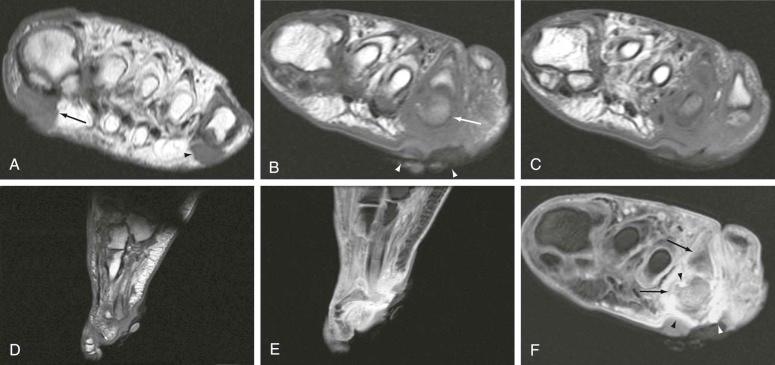
In the diabetic foot, infection typically begins as a result of a skin break, which may be due to minor trauma or breakdown of skin callus. Callus forms over areas of friction, locations of which are somewhat predictable. Callus is seen commonly adjacent to the first and fifth metatarsal heads ( Fig. 67-1A ), particularly in the setting of hallux valgus. Callus is also common adjacent to the calcaneus and the medial and lateral malleoli and over the dorsum of the toes, particularly when clawtoe deformity is present. Other deformities cause callus in atypical locations; for example, in the setting of rocker-bottom deformity, callus may be seen at the plantar aspect of the midfoot. These areas of callus and subsequent callus breakdown correspond to the most common areas of infection in the diabetic foot and ankle, and these locations should be evaluated with particular attention, especially on cross-sectional imaging studies such as MRI ( Fig. 67-1B through F ). Diabetic patients often lack pain and do not mount a systemic inflammatory response, making it difficult to differentiate a neuropathic hot, erythematous, swollen foot from an underlying soft tissue infection. However, any breach in the cutaneous integument and, more specifically, a draining ulcer in a diabetic foot should lead to a primary consideration of osteomyelitis.
Vascular disease is very common in diabetic patients (see Fig. 67-8 ). Baseline ischemia creates a setting in which cuts or other minor injuries heal slowly or not at all and calluses break down. This situation promotes formation and progression of foot ulceration. Immunopathy, also a feature of diabetes, coupled with vascular disease and diminished sensation, leads to wound infection that is typically caused by more than one organism. Progression of infected ulcers results in soft tissue abscesses, sinus tracts, septic tenosynovitis, and, eventually, septic arthritis and osteomyelitis. More advanced ischemia may cause gangrene, particularly at the digits and forefoot, referred to as dry gangrene if noninfected and wet gangrene if superinfected. Severe chronic ischemia may result in bone marrow infarcts.
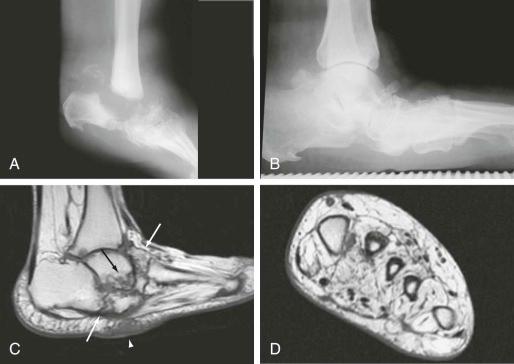
Neuropathy, a feature of diabetes, contributes to formation and progression of skin wounds and infection. Sensory neuropathy leads to a diminished perception of minor foot trauma, including cuts, ulcers, blisters, and friction-related skin breakdown, tendon and ligament injury, joint injury, and fractures. Articular injury with diminished sensation and ischemia can result in an aggressive-appearing, deforming arthropathy referred to as neuropathic osteoarthropathy, or Charcot arthropathy ( eFig. 67-1A ). Tendon dysfunction may result in articular deformity, in particular, arch collapse, causing a rocker-bottom deformity of the foot (see Fig. 60-3 and eFig. 67-1B and C ). Diabetic neuropathy affects the peripheral motor nerves, causing diffuse muscle atrophy and muscle imbalance that contribute to deformity (see Fig. 67-1D ). Foot deformity alters weight bearing, distribution of plantar pressures, and causes footwear to fit poorly, resulting in abnormal friction with subsequent callus formation and ulceration. Autonomic dysfunction also occurs, and, when combined with vascular fragility, ischemia, and reduced muscular activity, deposition of fluid in the soft tissues results. Edema within confined compartments of the foot increases intracompartmental pressures and can accentuate the ischemic cascade.
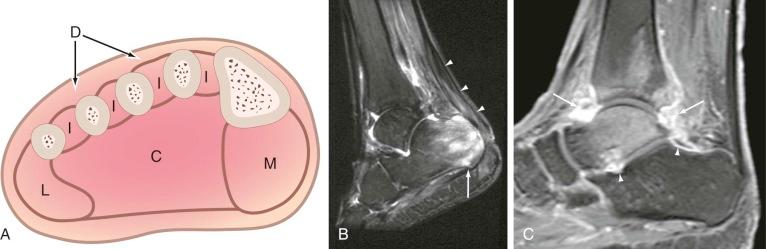
The foot is composed of distinct myofascial compartments ( eFig. 67-2A ). The plantar aspect of the foot is divided by muscle groups into medial, central, and lateral compartments, which extend from the metatarsophalangeal joints to the heel. The deep aspect of the central compartment has access to the lower calf through the long medial flexors that cross the ankle joint. If infection involves this deep fascial compartment, care must be taken to ensure that the process has not spread proximally. Interosseous compartments exist between the metatarsal bones as well. This compartmental anatomy, studied in cadavers, has been used to suggest patterns of spread of pedal infection. In the diabetic foot, however, the myofascial compartmental anatomy is not a good predictor of extent of spread. This may be due to variability of compartmental interconnections or due to ischemia and injury in diabetics causing pathologic communications. Spread of infection in diabetic patients tends to follow a centripetal pattern from the source rather than spreading proximally and preferentially along myofascial planes. However, proximal spread is a consideration that should be taken into account when interpreting imaging studies. Tendons and their sheaths are another route for proximal spread of infection, and spread through this route should be evaluated on cross-sectional imaging studies, particularly MRI (see eFig. 67-2B ). Another anatomic consideration is that, in many cases, there is communication of the tibiotalar joint and posterior facet of the subtalar joint. Involvement of one with septic arthritis often leads to rapid involvement of the other, with potential for the development of extensive osteomyelitis (see eFig. 67-2C ).
A number of factors make it difficult to propose a uniform algorithm for the diagnosis of infection in the diabetic foot. First, access to advanced imaging modalities varies geographically, as does reader expertise. Radiologist and referring clinician preference vary, often related to training. Treatment of patients with aggressive surgical management (limb-sparing surgery) versus conservative surgery (definitive amputation) or medical therapy varies, depending on severity of disease, the surgeon's training, and the patient's underlying condition. Aggressive surgical management may necessitate acquisition of a “road map” of the patient's pathologic process to achieve success, versus other treatment options that require only a positive or negative diagnosis. Venous access and extremity perfusion obviously influence the success of diagnostic examinations that require injection.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here