Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Follicular cyst is a cystic dilatation of the hair follicle epithelium.
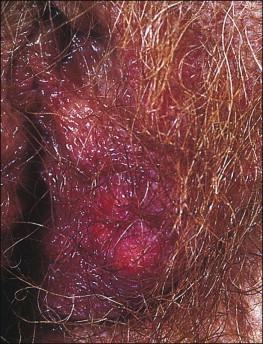
Follicular cyst presents as a solitary, creamy-white or yellowish lesion on the labium majus ( Figure 5.1 ). It is generally asymptomatic, but rupture may induce inflammation with enlargement, tenderness, erythema, and induration. Follicular cysts usually occur spontaneously and after age 30. Onset at an early age or occurrence in great numbers should prompt consideration of Gardner syndrome.
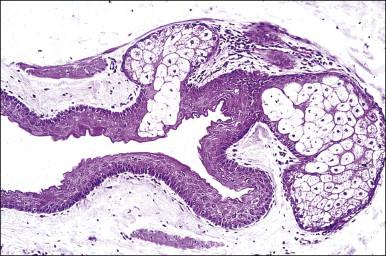
Most vulvar follicular cysts represent dilatations of the most distal portion of the follicle, the infundibulum, hence they have thin, flat, squamous epithelium lacking rete ridges, contain a granular layer, and are filled with loose-packed keratin. Its rupture may lead to leakage of keratin, resulting in an acute and chronic inflammatory foreign-body reaction.
Widespread, multiple, thin-walled cysts of the skin, lined by squamous epithelium , and including lobules of sebaceous cells.
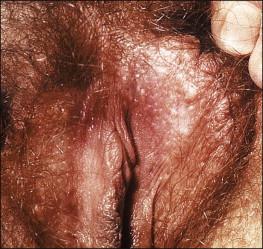
Steatocystoma multiplex is the dominantly inherited occurrence of numerous small, creamy-colored cysts exhibiting sebaceous ductal differentiation. The lesions usually appear after adolescence and may be solitary. Most common locations are the presternum, axillae, abdomen, and labia majora. Rarely, they first present as multiple vulvar cysts at an older age ( Figure 5.2 ).
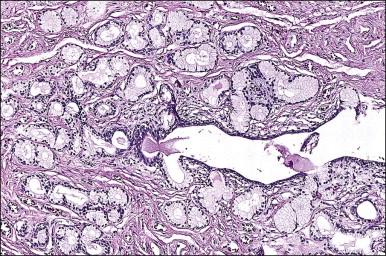
Steatocystomas are composed of an intricately folded, thin layer of stratified squamous epithelium lined internally by a corrugated, thin, compact, and strongly eosinophilic cuticle, lacking a granular layer. There may be associated small sebaceous glands ( Figure 5.3 ). Although this finding raises the differential diagnosis of dermoid cyst, the latter is extremely rare in the vulva and shows miniaturized hair follicles.
Bartholin cyst is the cystic dilatation of a major vestibular (Bartholin) gland or its duct.
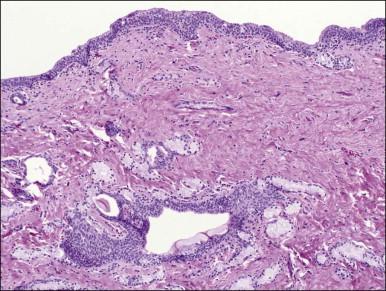
Bartholin cyst is the most common form of vulvar cyst, and is the presenting complaint for 2% of women during their annual gynecologic visit. The Bartholin glands ( Figure 5.4 ) are located behind the labia minora and their ducts open into the posterior lateral vestibules, just anterior to the hymeneal tegmentum. Bartholin cysts result from blockage of the drainage duct and resultant retention of secretions, perhaps following infection. Bartholin cysts are most common in the reproductive years. If large, they may partially obstruct the introitus. Bartholin cysts usually present as smooth-domed nodules, generally 1–10 cm in diameter. If brown or blue, they may be mistaken clinically for melanocytic nevi. The cysts contain mucoid fluid, which stains with mucicarmine, periodic acid–Schiff (PAS; with and without diastase), and Alcian blue at pH 2.5. They may recur after incision and drainage, and may require surgical excision, particularly in postmenopausal women, to rule out the possibility of associated carcinoma. Bartholin abscess is an acute process usually caused by Neisseria gonorrheal infection. Excision, drainage, and antibiotics are the treatments of choice.
Bartholin cysts show a lining of transitional epithelium, with frequent focal squamous metaplasia ( Figure 5.5 ). Smaller mucus-filled cysts may show some remnant of the original mucus-secreting glandular epithelium, but it may be flattened or cuboidal. Normal remnants of mucus glands may be presented adjacent to the cyst. Microscopically, the Bartholin duct abscess shows abundant neutrophilic infiltrate within the stroma surrounding the duct.
Mucinous cyst is the cystic dilatation of a minor vestibular gland or its duct.
True mucinous cysts most commonly occur in the vulvar vestibule, including the medial labium minus and near the Bartholin glands. Onset is typically between puberty and the fourth decade, usually in parous women or those exposed to oral contraceptives. Mucinous cysts are 2 mm to 3 cm in diameter and are usually solitary. They may cause pain or urinary complaints. Excision is curative. They presumably are due to obstruction.
These cysts are lined almost entirely by columnar or cuboidal mucus-secreting epithelium like that of endocervical glands without peripheral muscle fibers or myoepithelial cells. Foci of squamous metaplasia are sometimes present; they are considered to be of urogenital sinus origin.
An occasional cyst may be lined partially by ciliated columnar epithelium ( Figure 5.6 ). Usually this occurs as a focal change within a mucinous cyst or a Bartholin cyst; it is likely a nonspecific metaplastic change.
Paraurethral gland cyst is the cystic dilatation of the paraurethral (Skene) gland or its duct.
The paired paraurethral glands (the female homolog of the male prostate gland) are located on either side of the urethral meatus. Ductal occlusion, probably a consequence of infection (e.g., gonococcal), leads to formation of a retention cyst, generally less than 2 cm in size, located in the upper lateral introitus. Paraurethral gland cysts affect from neonates to premenopausal women, with an incidence of 1 per 2000–7000 women. Patients may be asymptomatic or complain of urinary obstruction or dyspareunia. Surgical excision should be done after medical therapy for any underlying infection.
Paraurethral gland cysts are probably derived from the duct rather than the acini, and are lined by transitional or stratified squamous epithelium upon a basement membrane, with only rare luminal cells containing intracytoplasmic mucin. Rarely, they contain calculi.
Mesonephric-like cysts are lined by cuboidal or low columnar epithelium and encased by a small amount of smooth muscle.
These cysts occur in the lateral walls of the vulva as superficial, single, domed, blue or red cysts with clear, watery contents. They resemble mesonephric ducts but their embryologic basis is unclear.
Cyst of the canal of Nuck is a cystic remnant of the processus vaginalis peritonei.
These cysts are most common in the inguinal canal, where they must be distinguished from hernias. They also occur in the mons pubis and in the superior, outer region of the labium majus. Ultrasonography or magnetic resonance imaging may be helpful for definitive clinical diagnosis. They are homologous to hydroceles in males. The processus vaginalis peritonei (canal of Nuck) is a rudimentary sac of peritoneal mesothelium carried down by the round ligament as it passes through the inguinal canal and inserts into the labium majus. Failure of this structure to obliterate normally during fetal development leads to blockage and cystic dilatation. While these cysts are usually solitary, more than one may arise if obstruction occurs at multiple sites. Generally asymptomatic, they may become tender to pressure, and may then require a procedure similar to herniorrhaphy.
In their pristine state, these are thin-walled and lined by flattened mesothelium. By the time they present clinically, repeated external trauma has usually induced fibrosis of their walls, reduced or destroyed their mesothelial lining, and caused hemosiderin deposition from old haemorrhage.
Extramammary Paget disease is a form of in situ adenocarcinoma of the squamous mucosa. For a number of these cases, recent evidence suggests an origin from Toker cells, which are found in the breast with clear cytoplasm that reacts for cytokeratin 7 (CK7) and are thought to derive from the ostia of the mammary-like glands found in the vulva, perineum, and perianal skin.
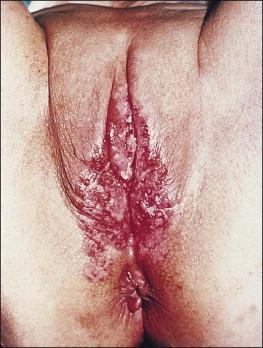
The vulva is the most common site of extramammary Paget disease, accounting for about 5% of all vulvar neoplasms. It usually presents in older women as a moist, red, eroded or eczematous-appearing plaque. One or both sides of the vulva can be involved, often with spread to the perianal skin ( Figure 5.7 ). The plaques are usually irritated and sore, and may be clinically misinterpreted as an inflammatory skin disease (e.g., intertrigo, fungal infections, immunobullous disorders, Hailey–Hailey disease). The diagnosis requires biopsy confirmation.
Treatment may be difficult because the disease usually extends well beyond the clinically apparent margins. Moreover, even surgical margin status is not particularly helpful in predicting recurrence, as about a third of patients experience recurrence regardless of whether the margins after initial surgery are positive or negative. The extent of the operation (wide local excision, simple vulvectomy, or modified radical vulvectomy) during the initial treatment also poorly correlates with disease recurrence. Mohs micrographic surgery, topical chemotherapy, and photodynamic therapy may play a role in selected cases. HER-2/neu expression has been demonstrated by some Paget disease of the vulva; thus, some patients may benefit from trastuzumab (Herceptin), a recombinant monoclonal antibody against HER-2/neu.
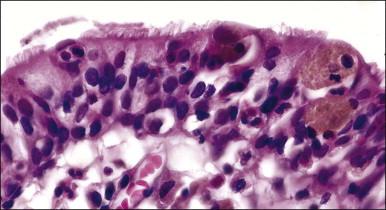
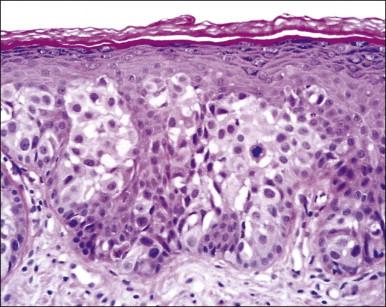
Histologically, the epidermis contains pale-staining cells that are larger than adjacent keratinocytes, arranged singly or in small to large nests ( Figure 5.8 ). When numerous, Paget cells may replace much of the epidermis, which appears swollen and thickened ( Figure 5.9 ). When Paget cells are single or few in number, they appear to be mainly above the basal layer or individually (pagetoid migration) into the upper epidermal layers. Cells appear not to be connected with the basement membrane and thus are different from melanoma in situ (see later; Figure 5.10A ). Paget cells may even be found growing down hair follicles as well as eccrine glands ( Figures 5.11 and 5.12 ). Occasionally, similar cells may be seen in the underlying dermis ( Figure 5.13 ), indicating invasion. While up to 50% of patients are reported to show invasion in some series, this is a distinctly unusual happening in our experience. Likewise, we find that metastases to lymph nodes are exceedingly rare.
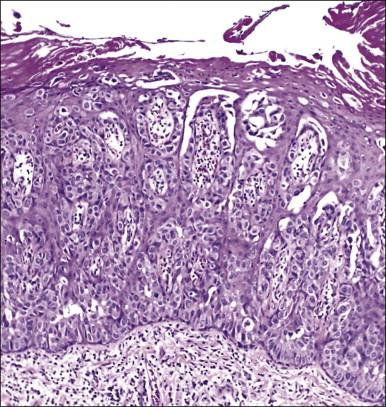
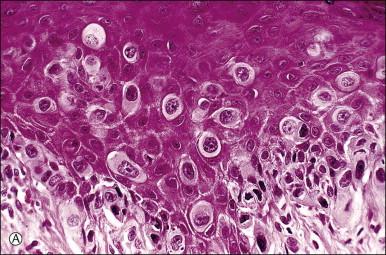
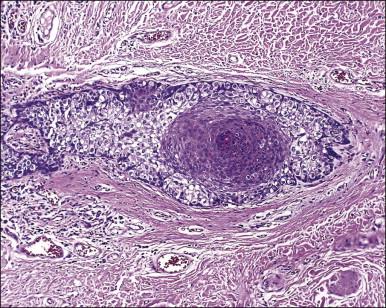
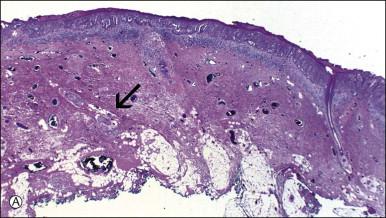
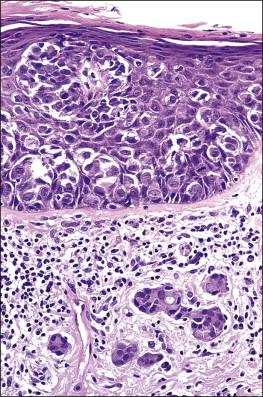
The pale cytoplasm of the Paget cell is usually finely granular, and the nuclei are central and round to oval. Mitotic figures may be found. On routine staining, Paget disease may be confused with melanoma in situ . However, the Paget cells at the basal layer are usually discrete and characteristically compress and displace the normal keratinocytes, whereas melanoma cells generally form a continuous proliferation close to the basement membrane.
Paget cells usually contain intracytoplasmic mucin (neutral and acidic), as highlighted by PAS-diastase, Alcian blue, colloidal iron, and mucicarmine stains. In problematic cases, immunohistochemistry may be useful ( Table 5.1 ), with Paget cells selectively expressing cytokeratins CAM 5.2, CK7 ( Figure 5.10B ), MUC5AC, carcinoembryonic antigen (CEA) ( Figure 5.10C ), epithelial membrane antigen (EMA), and gross cystic disease fluid protein-15 (BRST-2). About one-fifth of Paget cases are reactive for CK20. Since S-100 protein is also sometimes expressed, such cases may require study for more specific melanocytic markers (MART-1/Melan-A, HMB45, etc.). A possible pitfall is those cases in which the tumor cells contain melanin pigment since the interspersed melanocytes may show prominent dendrites and thus may be interpreted as melanoma cells. Androgen receptors can be detected in some cases.
| Diagnosis | Keratin Cocktail | CK7 | CK20 | S-100 | MART-1/HMB45 |
|---|---|---|---|---|---|
| Paget disease | + | + | −/+ * | − ** | − |
| Squamous cell carcinoma | + | + | − | − | − |
| Melanoma | − | − | − | + | + |
* CK20 is usually negative in extramammary Paget disease. However, it is positive in cases of carcinoma of the genitourinary or gastrointestinal tract.
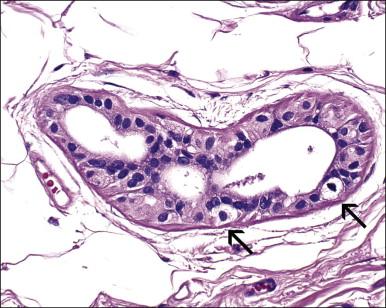
The origin of extramammary Paget disease is controversial. Some cases represent epidermotropic adenocarcinomas, but, unlike mammary cases (in which an underlying ductal carcinoma is almost invariably present), an underlying carcinoma is only rarely detected. Other postulated origins include a pluripotential stem cell within the epidermis or in situ malignant transformation of cells in the cutaneous sweat ducts as they insert into the epidermis. The most recent works point to the Toker cell ( Figure 5.14 ), which in the breast is an intraepidermal clear cell with bland nuclear features that is reactive for CK7 but not CK20. Cytogenetic findings suggest that at least some cases of Paget disease arise multicentrically within the epidermis from pluripotent stem cells, and have a molecular basis differing from other vulvar carcinomas.
Regarding the expression of mucin core proteins, vulvar Paget disease may arise from ectopic MUC5AC-positive cells originating from Bartholin or some other unidentified glands, while the unique expression of MUC2 in perianal Paget disease indicates that its origin from colorectal mucosa differs from that in the vulva.
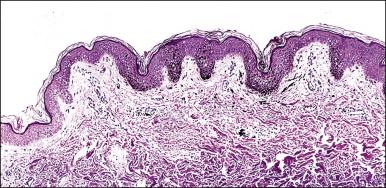
A lentigo is a circumscribed macule of increased pigmentation, generally 5 mm or less in diameter, and persisting even in the absence of sun exposure.
Lentigines are common on the labia majora or minora. They are often deeply pigmented but usually have uniform color, sharp circumscription, and regular borders. Lentigines proper are not precursors of melanoma.
The epidermis of a lentigo has elongated rete ridges and increased melanin deposition in the keratinocytes ( Figure 5.15 ). The number of melanocytes should be approximately normal. Furthermore, the melanocytes lack cytologic atypia.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here