Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Vascular dementia (VaD), as conventionally defined, is one of the most frequent forms of dementia. Furthermore, when vascular contributions to other types of dementias are included, it is arguably the most common form of dementia.
Criteria for the diagnosis of VaD have evolved to include a broader phenotype, and memory impairment is not required for a diagnosis of VaD.
Vascular mild cognitive impairment (VaMCI) is a more recently recognized entity that includes mild cognitive deficits, which do not significantly impact function and hence cannot be categorized as dementia, and where the etiology is thought to be predominantly vascular rather than neurodegenerative.
Newer, more sensitive imaging techniques, including high-Tesla (3 and 7 T) magnetic resonance imaging, diffusion tensor imaging and susceptibility sequences, and blood-brain barrier (BBB) disruption measured by gadolinium-enhanced MRI, have increased the prevalence and range of subtle cerebrovascular damage that can be detected in persons with suspected VaMCI and VaD.
A number of potentially modifiable risk factors for VaD have been identified, and addressing these may help to prevent the onset of clinical symptoms or slow the pace of decline in persons with VaMCI and VaD.
Current projections suggest that the United States will have 72 million people older than 65 years of age by 2030, a greater than 10-fold increase in a century. In this older population, having one or more vascular risk factors is the norm rather than the exception. Thus, in the community-based Framingham Heart Study (FHS) sample, the lifetime risk for development of hypertension is more than 90%. Not surprisingly, the burden of age-related neurologic illness has increased in parallel with lengthening life expectancy. A 2006 analysis of data from the FHS sample predicted that one in three people currently aged 65 and free of stroke and dementia would experience one or both of these conditions during their lifetimes. It is generally acknowledged that exposure to vascular risk factors adversely affects cognitive function, although there is uncertainty about the extent and the mechanisms. Observational studies show that vascular risk factors measured in midlife, but not those measured only in late life, predict later cognitive impairment and dementia. The Cardiovascular Health Study (CHS) identifies rates for Alzheimer disease (AD) and vascular dementia (VaD) to be 19.2 and 14.6 per 1000 person-years among whites at age 80, respectively. However, the concept of what constitutes “vascular dementia” remains in evolution. There has been growing emphasis on identifying persons with early cognitive impairment due to vascular pathology, because these individuals are at maximal risk for development of VaD and might benefit the most from preventive measures.
The evolution of the concept of VaD has been elegantly summarized by Roman. At the start of the 7th century, Saint Isadore, the Archbishop of Seville, used the term dementia (from the Latin root “demens,” meaning out of one’s mind) to describe a slowly progressive dullness or dotage. In 1672, Thomas Willis described the fairly sudden development of “dullness of mind and forgetfulness” accompanying a “half palsie” (hemiplegia) in his book De Anima Brutorum . AD was described in 1904 but was considered a rare presenile form of dementia. In his 1910 Lehrbuch der Psychiatrie, Kraeplin, who has been called the Linnaeus of psychiatry, separated a presenile form of dementia from senile dementias and concluded that most senile dementia was arteriosclerotic insanity, which later became attributed to “progressive hardening of the arteries leading to slow ischemic neuronal loss.” This remained a widely accepted viewpoint until 1970, when Blessed and Tomlinson established through careful autopsy studies that the pathology underlying most cases of dementia was, in fact, the plaques and tangles associated with AD. The definition of VaD became restricted to “multi-infarct” dementia; it was argued that vascular pathology resulted in dementia largely through the occurrence of multiple small or large cerebral infarcts. In the next 20 years, careful clinical studies did establish that single infarcts could result in dementia if they were strategically located, and computerized tomography (CT) scans showed that among persons with a similar number and location of strokes, concomitant cortical atrophy increased the risk of symptomatic dementia. With the emergence of brain magnetic resonance imaging (MRI), covert brain infarcts (CBIs) and white matter hyperintensities (WMHs) were found to be widespread and to be associated with an increased risk of cognitive impairment. With novel MRI imaging sequences and high- and ultra-high-strength MRI scans, the presence of cerebral microbleeds (CMBs), as well as subtle changes in white matter integrity and presence of microscopic infarcts can also be detected. , In recent decades the concept of mild cognitive impairment (MCI) as a prodromal stage of AD has emerged, and, in parallel, a broader concept of vascular cognitive impairment (VCI) has solidified that includes all states of cognitive impairment associated with cerebrovascular disease (CVD), including VaD, AD with cerebrovascular disorder, and MCI secondary to vascular pathology (VaMCI), which was previously also called VCI with no dementia (VCIND). , ,
Since the 1970s, several sets of diagnostic criteria have been formulated in attempts to standardize the definition of VaD. Such criteria are essential for use as diagnostic tools in clinical practice, to compare prevalence and incidence in different population samples, to uncover risk factors, and to recruit homogeneous cohorts for drug trials. These criteria range from the entirely clinical Hachinski Ischemic Score (HIS) to the strict National Institute for Neurological Diseases and Stroke–Association Internationale pour la Recherche et l’Enseignement en Neurosciences (NINDS-AIREN) criteria, formulated largely for use in research settings. They include the recent, inclusive, American Heart Association and American Stroke Association (AHA-ASA) VCI criteria from 2011, the more epidemiologically applicable Diagnostic and Statistical Manual (DSM-III, IIIR, IV) criteria, the International Classification of Disease 10th revision (ICD-10) criteria, and the California Alzheimer’s Disease Diagnostic and Treatment Centers (ADDTC) criteria. In 2014 a further refinement of diagnostic criteria for vascular cognitive disorders were proposed by the International Society for Vascular Behavioral and Cognitive Disorders (VASCOG); however, the bewildering plurality of diagnostic criteria do share a few core characteristics. Finally, the international Vascular Impairment of Cognition Classification Consensus Study (VICCCS) has recently been established to synthesize the conceptual framework and harmonize diagnostic criteria for VCI and VaD. ,
The HIS does not classify dementia but seeks to predict the dominant underlying pathology. It is based entirely on the history of vascular risk factors and on the clinical signs; in an abbreviated version, a score of 7 or more out of a possible 12 points is considered diagnostic of VaD, whereas a score less than 4 is thought to exclude VaD. The NINDS-AIREN and ADDTC criteria additionally consider CT or MRI data. Each broadly requires clinical and/or imaging documentation of strokes or vascular brain injury (e.g., extensive WMH), a temporal profile linking the vascular damage and the dementia (e.g., abrupt onset or onset within 3 months after stroke, a stuttering or stepwise progression) and focal neurologic signs if thought to be of vascular etiology, and assigns a degree of certainty to the diagnosis: “probable” or “possible.” When each of the different criteria are applied to a single sample, a varying, only partially overlapping set of subjects are categorized as having VaD. When pathology is used as a “gold standard,” the sensitivities of the different clinical criteria have been noted to vary from 0.2 to 0.7 (possible VaD as defined by the ADDTC being the most sensitive criterion) and the specificities from 0.78 to 0.93 (probable VaD by the NINDS-AIREN criteria being the most specific). Thus most of these criteria emphasize specificity over sensitivity; less than 50% of all persons with moderately severe vascular pathology at autopsy are diagnosed during life as having VaD.
The criteria proposed in the AHA-ASA 2011 statement suggested that the term VCI be used for all cognitive disorders associated with any evidence of CVD and cerebrovascular brain dysfunction or injury, regardless of coexisting pathologies. Several of the criteria for VaD, including the AHA-ASA criteria, emphasize that a memory deficit is not required to diagnose VaD, only the presence of a cognitive deficit in one or more domains. Mild cognitive impairment of vascular origin (VaMCI) was also addressed, stipulating a decline in cognitive function in at least one cognitive domain, with normal or minimally affected activities of daily living. Like MCI, VaMCI includes four subtypes: amnestic, amnestic plus other domains, nonamnestic single domain, and nonamnestic multiple domain. The term “unstable vascular MCI” was also introduced in this statement to describe patients who shift from an impaired to an unimpaired cognitive state. One limitation of these criteria is the lack of established sensitivity and specificity. The 2014 VASCOG criteria identify a continuum of vascular cognitive disorders, categorized into mild vascular cognitive disorder and major vascular cognitive disorder, following the DSM-V lead of renaming MCI and dementia as “minor” and “major” neurocognitive disorders, and detail neuroimaging, clinical, and/or pathologic criteria that may be considered supportive of these diagnoses. The various criteria for VaD have been summarized and compared in prior publications. , , Table 18.1 compares the VASCOG, AHA-ASA, and NINDS-AIREN VaD criteria.
| Criterion | VICCCS-2 (2017) | VASCOG (2014) | AHA-ASA (2011) | NINDS-AIREN (1993) |
|---|---|---|---|---|
| Diagnosis of dementia | Clinically significant deficits in at least 1 domain and severe disruption to ADL/IADL | Decline in ≥1 cognitive domain that interferes with independence | ≥2 cognitive domains that affect daily living a | Impairment of memory and decline in ≥2 cognitive domains that interfere with daily living a |
| Cognitive domains assessed | Executive function, attention, memory; language, visuospatial function | Attention and processing speed; frontal-executive function; learning and memory; language; visuoconstructional-perceptual; praxis-gnosis-body schema; social cognition | Executive/attention; memory; language: visuospatial functions | Orientation, attention, language, visuospatial functions, executive functions, motor control, praxis |
| Memory deficit required | No | No | No | Yes |
| Diagnostic categories | Poststroke dementia, subcortical ischemic vascular dementia, mixed dementia, multi-infarct dementia (probable, possible based on MRI available) | Major vascular cognitive disorder or VaD | Probable, possible VaD | Definite (histopathologically verified), probable, possible VaD |
| Temporal relationship between vascular event and cognitive deficits | Yes, for post-stroke dementia (within 6 months) | Yes, OR frontal-executive, processing and attention decline WITH (1) early gait disturbance, (2) early urinary frequency, (3) personality/mood changes | Yes (probable); No (possible) | Yes (probable); No (possible) |
| Radiologic evidence of vascular disease | Yes | Yes (probable); No (possible) | Yes (probable); No (possible) | |
| Coexistent neurodegenerative diseases allowed | No | No (probable); Yes (possible) | No | |
| Exclusions | Alternative neurologic diagnosis; other medical disorders | Delirium; active drug/alcohol abuse b | Disturbance of consciousness, delirium; psychosis, severe aphasia, major sensorimotor impairment |
a Must be independent of motor/sensory sequelae of the vascular event.
Most recently, the VICCCS published two statements related to the conceptualization (VICCCS-1) and diagnosis (VICCCS-2) of VCI. In VICCCS-1, experts in the field, identified by a review of published manuscripts related to VCI or VaD, were surveyed for their opinions about the issues in defining VCI as well as categorization and classification of VCI. The majority of those surveyed cited O’Brien et al. as the preferred conceptual framework from which a revised framework of VCI may be developed. Ultimately, VICCCS-1 resulted in conceptualization of VCI into mild or major (i.e., VaD). Under the VaD category, four different subtypes were defined: poststroke dementia, subcortical ischemic VaD, multi-infarct (cortical) dementia, and mixed dementias. Using the VICCCS-1 conceptualization, VICCCS-2 aimed to further operationalize the diagnosis of VCI and its subtypes. The aforementioned AHA-ASA and NINDS-CSN statements were used as references for these revised guidelines. , VICCCS-2 also discusses neuroimaging markers deemed appropriate for evaluation of VCI.
The risk of new-onset dementia in patients who have had stroke is approximately twice the risk in age- and sex-matched controls and averages approximately 10% after the first stroke, , depending on the location, volume of damaged brain tissue, clinical severity, and presence of early poststroke complications (seizure, delirium, hypoxia, hypotension), as well as on prestroke cognitive status and concomitant imaging abnormalities such as covert or subclinical infarcts (CBIs), CMB, WMH, and medial temporal lobe atrophy. , , A 2009 review identified older age, less education, prestroke cognitive impairment, diabetes, and atrial fibrillation as factors that increased the risk, but the single strongest predictor of cognitive decline after an initial stroke was the occurrence of a second stroke. In persons with recurrent stroke, the risk of dementia rose to approximately 30%, regardless of the number and severity of vascular risk factors. Although the occurrence of VaD in persons who have suffered large infarcts makes intuitive sense, VaD may also follow small infarcts. Among 1636 participants with lacunar strokes in the Small Subcortical Strokes (SPS3) trial, 47% were classified as having VaMCI with a diverse mix of subtypes: approximately one-third amnestic, one-third amnestic multidomain, and one-third nonamnestic. The most frequently noted deficits were in the domains of episodic memory, language and executive function (verbal fluency), and motor speed and dexterity. The Determinants of Dementia After Stroke (DEDEMAS) Study, an international multicenter observational prospective study aimed at identifying and characterizing the determinants of cognitive impairment after stroke, has been ongoing since 2013.
The concept of VCI outlined in 2006 by the NINDS in collaboration with the Canadian Stroke Network (CSN), in a statement advising harmonization standards for data collection, and further elaborated upon in the 2011 AHA-ASA VCI statement, is broad. It includes, but is not restricted to, dementia following stroke. The term “vascular cognitive impairment” may be used to describe any syndrome of cognitive and behavioral impairment that is thought to be due to vascular factors affecting the brain. Thus it could range from the subtle cognitive deficits seen in persons with diabetes when compared with their nondiabetic peers, through the multifocal cognitive deficits that may accompany the presence of multiple brain infarcts or hemorrhages to clinical VaD. The last requires the presence of cognitive or behavioral problems severe enough to affect social or occupational functioning (i.e., amounting to “dementia”), the documentation of disease affecting the blood vessels or blood flow to part or all of the brain, and clinical or radiologic evidence of structural damage to the brain due to these vascular factors per the NINDS-CSN collaboration. There have also been efforts to define diagnostic clinical criteria for subcortical VaD syndromes, conditions that have been described as Binswanger syndrome: extensive WMHs and cognitive slowing in the absence of clinical strokes or CT/MRI infarcts and hemorrhages. , The criteria proposed in the 5th edition of the Diagnostic and Statistical Manual (DSM5) replace the potentially stigmatizing term “dementia” with “major neurocognitive disorder,” and the prodromal stage of MCI or VaMCI is called “minor neurocognitive disorder.” The 2011 AHA-ASA scientific statement on vascular contributions to cognitive impairment and dementia defines VCI as “a syndrome with evidence of clinical stroke or subclinical vascular brain injury and cognitive impairment affecting at least one cognitive domain.” The entire spectrum of cognitive disorders is included, with all forms of cerebral vascular brain injury, ranging from MCI to dementia. Importantly, memory impairment is not a requirement for diagnosis because unlike AD, which in most cases begins in the mesial temporal lobe structures and affects hippocampal—and thus memory—function, vascular damage is topographically heterogeneous and can involve deficits isolated to other domains. The writing group emphasized the need for continued development and refinement of cognitive batteries for VCI, in addition to identification of biomarkers and pathologic-radiologic correlation.
The various criteria for VaD and AD define “probable AD” and “probable VaD” but do not address the large number of persons with mixed AD and VaD. , , The VICCCS group has addressed this issue in their newest conceptualization of VCI. , The current consensus among clinicians and epidemiologists is to independently decide on the presence or absence of clinically probable AD and VaD without requiring that the presence of one condition excludes the other diagnosis. There are two issues at play here. First, although a clinical diagnosis of AD is accurate approximately 90% of the time in autopsy studies, , clinicians are more likely to miss a diagnosis of a mixed dementia. Second, a clinician’s estimate of vascular pathology is only as good as the imaging modality (e.g., MRI) used for the assessment. Higher-field-strength MRI scanners at 7 T are beginning to show microinfarcts that might not have been seen on older machines with less powerful magnetic fields. The degree to which microinfarctions may play a role in VCI and VaD is an active area of research. AD and VCI may share pathogenic mechanisms, as evidenced by animal work, , and the vascular risk factors associated with dementia are further discussed later in this chapter.
Recent studies have explored associations between vascular pathology and risk factors and imaging markers of neurodegenerative pathology, in an effort to elucidate the relationship between these two entities. The Harvard Brain Aging Study has examined associations between imaging markers of vascular and neurodegenerative pathologies. Among 223 nondemented older adults, the Harvard Brain Aging Study found that greater Framingham Heart Study cardiovascular disease (FHS-CVD) risk score and greater β-amyloid burden (imaged by positron emission tomography [PET]) was related to faster cognitive decline. Furthermore, they observed a significant multiplicative interaction between FHS-CVD risk score and β-amyloid burden, implying that vascular risk and neurodegenerative pathology have a synergistic relationship. Similar associations were observed when examining imaging markers of tau pathology burden, further lending support that vascular and neurodegenerative pathologies interact. However, it is still unclear whether these pathologies directly impact one another. Data from the Mayo Clinic suggest that greater white matter integrity loss is not related to amyloid burden. However, evidence from the ARIC study suggests that vascular risk factor burden in midlife is related to greater amyloid burden, implying that timing of exposure plays a role.
When a similar set of criteria are used, the age-adjusted prevalence of VCI may be lower in low-to-middle-income countries that are early in the process of demographic transition. However, it is these countries that may see the fastest increase in the prevalence of VCI. The incidence of VaD in the United States among persons older than 65 years has been estimated at 14.6 per 1000 person-years. The proportion of all cases of dementia that is vascular appears to vary by age, ethnicity, geography, and definition used. Mixed AD and VaD may be the most common explanation for cognitive impairment in the elderly. The ideal way to define the complete spectrum of CVD appears to be to prospectively follow epidemiologic cohorts exposed to vascular risk factors to define the full range of structural and cognitive changes associated with these vascular risk factors and also to determine which of these intermediate phenotypes results in clinical disease ( Fig. 18.1 ). This approach is feasible because a number of studies of cardiovascular disease have incorporated brain imaging and cognitive testing in the past two decades, and a few have additionally initiated brain banks to permit clinical imaging–pathologic correlations.
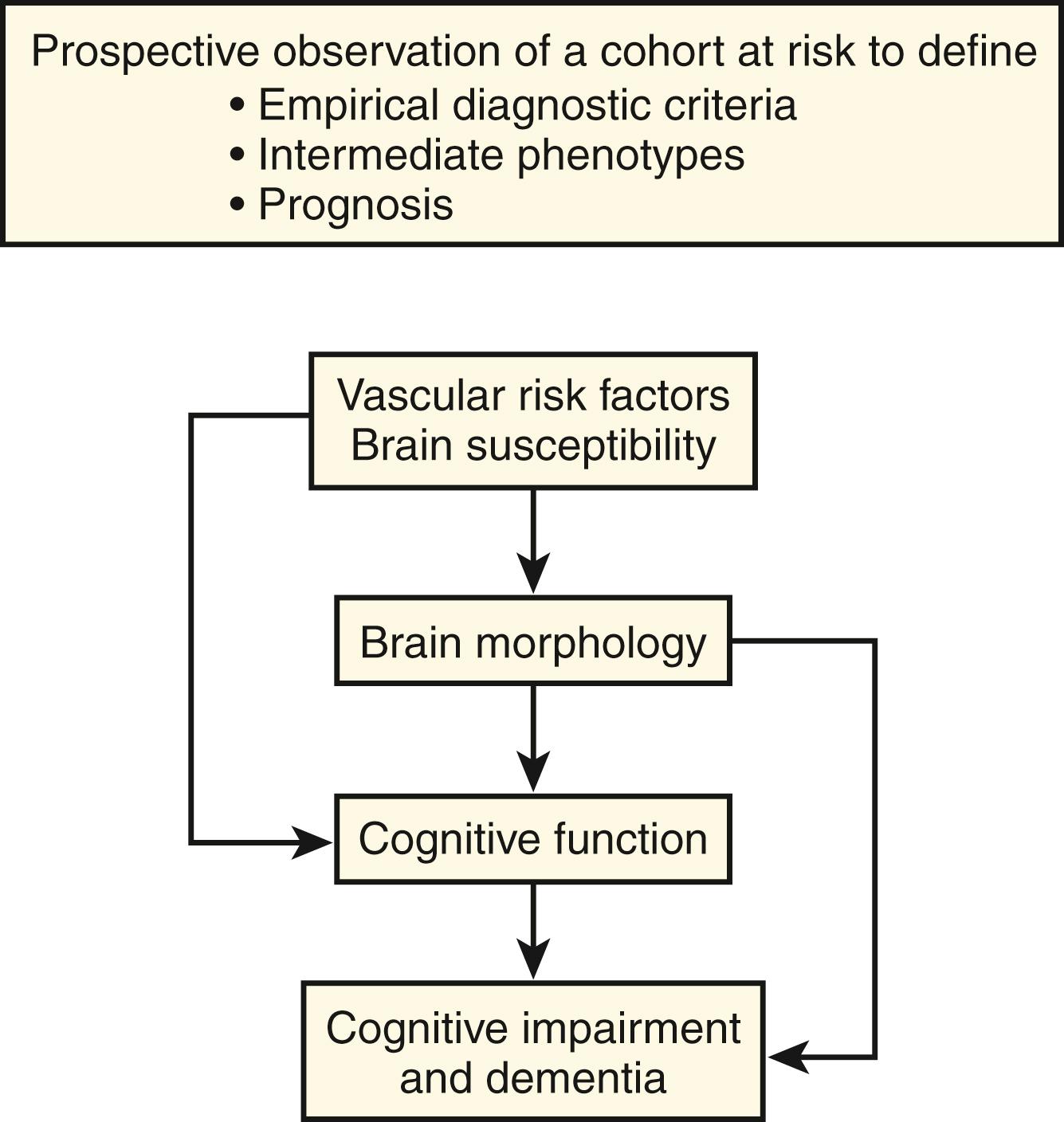
The VCI syndrome is best diagnosed and characterized by identifying the presence and quantifying the extent of (1) the cognitive deficits (and presence or absence of VaD and VaMCI) by clinical and formal neuropsychological testing and (2) the vascular brain injury through neuroimaging supplemented by cerebral and systemic vascular imaging as indicated. The term “VCI” can be used for cognitive disorders associated with varying forms of vascular brain injury: atherothrombotic, cardioembolic, hemorrhagic, and rare genetic vascular disorders. The role of other vascular pathologies, including white matter lesions and microbleeds, in VCI is unclear. Recent research points to an association between asymptomatic carotid artery stenosis, defined as stenosis without a recognized neurologic deficit, and cognitive dysfunction primarily within the domains of processing speed and learning/memory. Further studies are needed to confirm this association and potential mechanisms.
The clinical neurologic profile of VaD can be extremely varied depending on the underlying pathology, patient age and education, and presence or absence of concomitant AD pathology. However, patients with VaD are typically younger and more likely to be male. Some clinical symptoms and signs that are more frequently seen in persons with VaD than in persons with typical AD are a pattern of multifocal rather than global deficits, presence of focal neurologic signs (including gait abnormalities), an early and disproportionately severe involvement of executive dysfunction (unlike the early verbal memory loss in AD), and relatively preserved recognition (improved performance when categorical or phonemic clues are offered) in comparison with spontaneous recall. Depression and emotional lability are also more frequently seen. The frequently considered differential diagnoses for VaD include AD, frontotemporal dementia, normal-pressure hydrocephalus, dementia with Lewy bodies, and dementia associated with cerebral infections such as human immunodeficiency virus (HIV). The finding of cerebral vascular brain injury on CT or MRI can help to establish a clinical diagnosis of VaD.
Community-based data have shown that even before the occurrence of an initial stroke, persons who subsequently experience a stroke do worse on global tests of cognition such as the Mini-Mental State Examination (MMSE) than do age- and sex-matched controls. This feature may represent a subtle effect of cumulated exposure to vascular risk factors over the preceding years; indeed, a pattern of VCI can be defined that is associated with covert or subclinical brain injury and that doubles the risk of subsequent stroke. , Conversely, in a community-based series of subjects with MCI, a significant proportion has relative preservation of memory function and disproportionate involvement of attention, processing speed, and executive function domains. They are more likely to have vascular risk factors, imaging evidence of covert vascular brain injury, and a higher risk for subsequent stroke than persons with the predominantly amnestic MCI that seems to precede clinical AD; hence these subjects were frequently considered to have VaMCI. However, the situation is likely more complex with MCI due to both neurodegenerative and vascular etiologies having multiple presentations. Thus all MCI may be categorized along multiple axes as single or multiple domain, predominantly amnestic or nonamnestic, and pathologically as likely vascular or neurodegenerative. , On follow-up, any sample of persons with VaMCI includes persons who revert to normal and are deemed to have “unstable MCI” (either they recover from the effects of a vascular insult such as a stroke or their clinical picture was due to a reversible cause such as depression), others who go on to demonstrate VaD, and yet others who have typical AD. , The MCI and subtype definitions are applied both in research studies and as a useful interim category for classifying patients in clinical practice.
The neuropsychological assessment of patients with suspected VCI requires a comprehensive cognitive battery, which should include tests sensitive to executive function abnormalities, a salient feature of the condition. Thus the Montreal Cognitive Assessment (MOCA) has been recommended in place of the MMSE, also called the Folstein test, because the former is more sensitive for the detection of executive function abnormalities. The 2006 NINDS-CSN VCI harmonization guidelines described several neuropsychological test protocols of varying length (30 and 60 minutes), each of which covers the domains of frontal executive function (animal naming and phonemic fluency, digit-symbol substitution ± trail-making test), visual perception and organization (a figure-copying task such as using the Rey-Osterreith figure), a verbal learning and memory task (such as the Hopkins verbal learning test), a lexical retrieval task (such as the Boston naming test), and a questionnaire-based screen for neuropsychiatric and depressive symptoms (using the neuropsychiatric inventory [NPI] and the Center for Epidemiological Studies–Depression [CES-D] Scale). They also suggest the MOCA as a 5-minute battery for use in patients with suspected VCI. Detailed discussion of these protocols is beyond the scope of this chapter, but the tests listed have been well standardized and are referenced in the NINDS-CSN guidelines. Other batteries that cover these domains have been used in existing epidemiologic studies and in clinics. , The MOCA and trail-making test are also part of a cognitive test panel recommended by a National Human Genetics Research Institute (NHGRI) panel convened to suggest tests for use by phenotypic nonexperts conducting genome-wide association studies ( www.phenxtoolkit.org ). The major limitation of neuropsychological testing geared toward identifying executive function abnormalities, such as the MOCA, is that deficits in executive function are not specific to CVD. In a study of FHS subjects comparing new cognitive deficits that appeared in persons after stroke with cognitive deficits appearing over time in persons without stroke, persons with stroke had poorer cognitive function in multiple domains including immediate recall of verbal and visual memories, verbal learning and language, and the more traditionally expected domains of executive function and visuospatial and motor skills. The frequent coexistence of VaD and AD also makes reliance upon neuropsychological testing for diagnostic clarification difficult.
VCI may also be defined as a constellation of cognitive and functional impairments associated with the structural cerebrovascular brain injury (CVBI) noted on imaging (in vivo) or at autopsy. CVBI on imaging includes clinically symptomatic large and small artery infarcts and hemorrhages . However, clinically asymptomatic (covert) brain infarction is even more common, and the full spectrum of CVBI also includes WMHs and total and regional brain atrophy , , , (see Table 18.2 for a summary of main studies). The relationship between CVBI on brain imaging and cognitive impairment is not simple or linear and remains inadequately understood. Partly, the prevalence and extent of CVBI detected vary with the imaging protocol used. Thus subtle white matter injury in normal-appearing white matter can be detected only by diffusion tensor imaging (DTI), and some forms of CVBI, such as microinfarcts, are not detectable by conventional 1.5- or 3-T MRI imaging, although they can be detected by ultra-high-resolution 7-T brain MRI imaging. Furthermore, many changes that could be attributed to ischemia (such as generalized and lobar atrophy, hippocampal sclerosis, and, to a lesser extent, WMHs) have also been associated with AD and other neurodegenerative pathologies. Finally, in most elderly persons both vascular and nonvascular pathologies coexist, making it difficult to attribute the cognitive impairment or dementia detected to a specific etiology. Quantifying the extent of AD pathology through in vivo using newer amyloid imaging techniques with PET using the 11C-labeled Pittsburgh compound B (PiB) and other 18F-containing amyloid labeling compounds are steps in that direction. Though rare, pure subcortical VaD has been identified using 11C-PiB to rule out amyloid pathology. Studies combining these modalities (volumetric MRI and amyloid PET) are underway in clinical and epidemiologic settings.
| Cohort Study | Brief Description | SBIs | WMH | Total Brain Volume | Regional Brain Volume | Notes |
|---|---|---|---|---|---|---|
| Framingham Offspring Study | N = ∼2040, 53% women, mean age = 62, mostly non-Hispanic white | Albert et al., 2010 | Au et al., 2006 ; Stavitsky et al., 2010 ; Albert et al., 2010 | Seshadri et al., 2004 | — | Greater WMH is associated with worse cognition, including executive function and visual organization; greater brain volume is associated with better cognition. Among men, greater WMH and presence of infarcts is related to worse executive function. |
| Cardiovascular Health Study | N = ∼3660, mean age = 76 | Price et al., 1997 | Longstreth Jr. et al., 2005 | — | — | Presence of SBIs and greater WMH grade are associated with worse cognition. |
| Atherosclerosis Risk in Communities | N = ∼1846, mean age = 76, 60% women, 28% non-Hispanic black | Mosley Jr. et al., 2005 | Mosley Jr. et al., 2005 | Schneider et al., 2019 | Schneider et al., 2019 | Presence of SBIs and greater WMH grade are associated with worse cognition. Those with the greatest decline in memory had smaller medial temporal lobe, amygdala, hippocampal, and entorhinal cortex volume. Those with the greatest decline in language or executive function had smaller volumes across all brain regions. |
| Northern Manhattan Study | N = 1290, mean age = 70, 66% Hispanic/Latino | Wright et al., 2016 | Dong et al., 2015 ; Glazer et al., 2015 ; Wright et al., 2016 | Dong et al., 2015 | Dong et al., 2015 | Greater WMH is associated with worse cognition, including general cognition, learning ability, and processing speed. Smaller hippocampal volumes were associated with worse cognition across multiple domains. Smaller frontal lobe volume was associated with worse executive function. |
| 3C-Dijon | N = ∼1677, 62% women, mean age = 72 | — | Kaffashian et al., 2016 | Godin et al., 2010 | Godin et al., 2010 | Greater WMHV is associated with greater cognitive decline. Greater hippocampal volume is associated with better cognition. |
| AGES-Reykjavik | N = ∼2612, 41% men, mean age = 75 | Sigurdsson et al., 2017 | — | — | — | Incident brain infarcts are associated with greater cognitive decline. |
| Washington Heights/Inwood Columbia Aging Project | N = ∼638, mean age = 80, 29% non-Hispanic White, 36% African American, 35% Hispanic | Zahodne et al., 2015 | Zahodne et al., 2015 | Zahodne et al., 2015 | Zahodne et al., 2015 | Greater WMHV and presence of infarcts were associated with worse cognition, including language and executive function. Larger hippocampal volume was associated with better memory. |
| Rotterdam Study | N = ∼1015, mean age = 72 | Vermeer et al., 2003 ; Prins et al., 2005 | de Groot et al., 2000 ; Prins et al., 2005 | Vibha et al., 2018 ; Prins et al., 2005 | Ikram et al., 2010 ; Vibha et al., 2018 | Lower brain volume is associated with worse cognitive decline, including processing speed, and executive function. Smaller gray matter volume is associated with worse memory; smaller white matter volume is associated with worse processing speed and executive function. Greater WML volume is associated with worse general cognition, processing speed, and executive function. Presence of infarcts are associated with worse processing speed and executive function. |
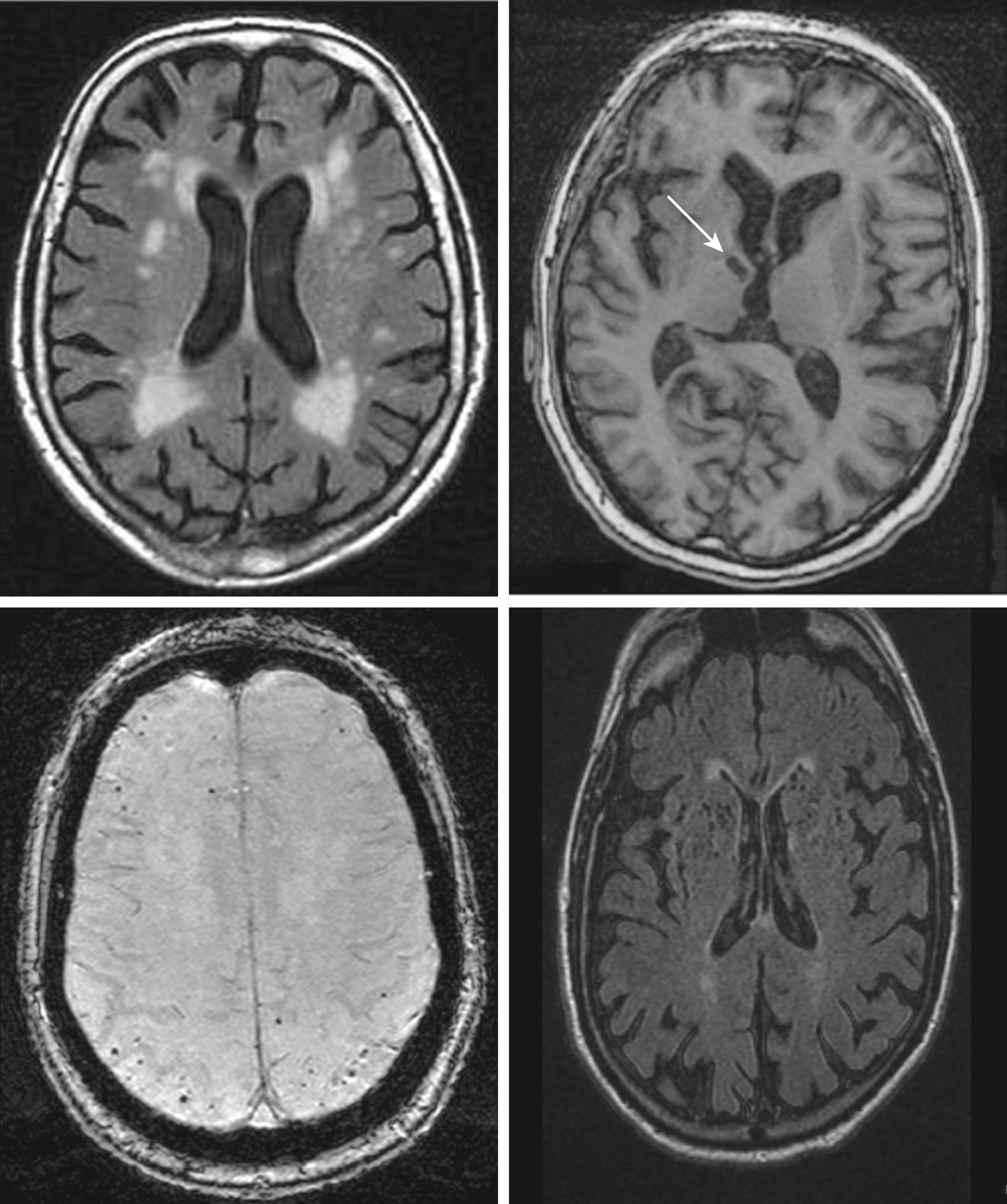
Historically, because of these caveats, neuroimaging is currently used to describe the vasculature and CVBI rather than to diagnose VCI or VaD. However, the VICCCS-2 diagnostic guidelines have recommended the use of imaging in the diagnosis of VCI and VaD, based on the “acceptable MRI measures” outlined in the NINDS-CSC guidelines. This includes the number, size and location of infarcts, and hemorrhages, extent on a quantitative or validated semiqualitative scale of WMH volume, , , and measures of total brain (or ventricular) and hippocampal volumes. Furthermore, the NINDS-CSC VCI harmonization guidelines suggest that in a research setting, a minimal imaging dataset should include an MRI (≥1 T) with three-dimensional (3D) T1-weighted, T2-weighted, fluid-attenuated inversion recovery (FLAIR), and gradient echo (GRE) sequences. Diffusion-weighted images (for acute stroke), DTI for assessing the state of the white matter tracts (abnormal DTI [lower fractional anisotropy] in normal-appearing white matter has been associated with poorer executive function and with vascular risk factors), PET for β-amyloid, and noninvasive assessment of the cerebral vasculature (carotid ultrasound and/or MR angiogram) are encouraged.
Similar to the harmonization of VCI conceptualization and diagnosis, efforts are underway to harmonize the measurement of neuroimaging markers of cerebrovascular injury for the diagnosis of VCI. The Harmonizing Brain Imaging Methods for Vascular Contributions to Neurodegeneration (HARNESS) group has provided openly available MR protocols and analysis tools for research use ( https://harness-neuroimaging.org/ ). This initiative follows the work of Wardlaw et al. regarding establishment of the Standards for Reporting Vascular Changes on Neuroimaging (STRIVE) criteria, which aimed to harmonize definitions of common cerebrovascular pathologies. Similarly, the MarkVCID initiative funded by NINDS currently aims to establish and validate both neuroimaging and serum- or fluid-based biomarkers for VCI ( https://markvcid.partners.org/ ).
Newer imaging measures of cerebrovascular injury are emerging and will inform future research and definitions of VCI, especially the measurement of blood-brain barrier (BBB) integrity imaged by gadolinium-enhanced MRI. Measures of BBB integrity have been related to cognitive dysfunction in smaller clinical samples, but unlike other measures of white matter (WM) integrity, BBB disruption has not been as widely measured in larger studies. Future work is warranted to capture BBB disruption longitudinally and examine associations with vascular and neurodegenerative pathology.
Recent statements have proposed the addition of vascular dysfunction, including BBB disruption, to the research framework for the biomarker-based definition of AD. WMHs are still the most prevalent marker of cerebrovascular injury and robustly related to cognitive decline and VCID. Future work is warranted to translate the use of multiple biomarkers into clinical practice.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here