Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Urine cytology was popularized by George Papanicolaou in the 1940s as a way to detect and monitor patients with bladder cancer. By the 1960s, the cytologic, histologic, and clinical features of high-grade urothelial carcinoma were well established, and cytology became an inexpensive, quick, and reliable way to diagnose high-grade urothelial carcinoma, as well as a variety of less common tumors.
Urine cytology is an excellent test for high-grade urothelial carcinoma, but it is ineffective for low-grade tumors. A multitude of ancillary tests have been developed to improve diagnostic accuracy for the wide range of urothelial neoplasms, but their value is limited to specific clinical settings. More recently, the Paris System for Reporting Urinary Cytology was introduced to improve the practice of urine cytology by recommending standardized criteria and terminology for diagnosis and discouraging the use of cytology to identify low-grade urothelial tumors.
The utility of urine cytology in detecting, evaluating, and monitoring patients for urothelial carcinoma is not disputed. Urine cytology remains one of the most common methods for the initial diagnosis of this potentially deadly yet often manageable and even curable disease that benefits from early detection.
Patients at high risk of bladder cancer
Unexplained hematuria
Abnormal cystoscopic findings
Follow-up of patients treated for urothelial carcinoma
Although urine cytology is an excellent diagnostic test, its role in population screening is limited. Unlike cervical cytology, it is not used to screen symptom-free individuals because the benefits are outweighed by the cost. Urine cytology is used, however, to screen high-risk individuals: those with exposure to the aromatic amines used in the petrochemical industry and to aniline dyes and patients treated with cyclophosphamide.
The most common entry point for urinary cytology is hematuria, but the cost-benefit ratio of screening all patients with hematuria is high because malignancy is found in only 5% to 10% of patients with hematuria. Instead of urine cytology, the American Urological Association (AUA) recommends cystoscopy as the initial test for any patient over the age of 35 with unexplained hematuria, despite its invasiveness and higher cost, in large part because cystoscopy can determine causes of hematuria other than high-grade carcinoma (e.g., stones, anatomic abnormalities, low-grade tumors). The AUA recommends urine cytology for patients with worrisome cystoscopic findings, persistent hematuria after a negative workup, or additional risk factors for carcinoma in situ (irritative voiding symptoms, current or past tobacco use, or chemical exposures).
Urine cytology is a standard component of surveillance for recurrent or de novo urothelial carcinoma in patients with a previously diagnosed and treated urothelial cancer anywhere in the urinary tract.
There are six types of urinary specimens, each with its relative advantages ( Table 3.1 ).
| Specimen Type | Advantages | Disadvantages |
|---|---|---|
| Voided urine | Noninvasive No instrumentation artifact |
Low cellularity Vaginal contamination Poor preservation |
| Catheterized | High cellularity | Invasive Instrumentation artifact Poor preservation |
| Bladder washing | High cellularity Good cell preservation |
Invasive Instrumentation artifact |
| Upper tract washing | High cellularity Good preservation Selective sampling |
Invasive Instrumentation artifact |
| Brush cytology | Selective sampling | Invasive Air drying possible (if direct smear) |
| Ileal loop | Permits screening for recurrent bladder cancer | Low cellularity Poor preservation |
Voided urine should be obtained 3 to 4 hours after the patient has last urinated. First morning-voided urine specimens should be avoided because cells in a stagnant, low pH, and hypertonic environment undergo degenerative changes, making cytologic assessment difficult. Studies suggest that a volume of 30 mL or more of urine may have a marginally higher sensitivity than specimens with lower volumes, but this is not statistically significant.
In women, voided urine can be contaminated by vaginal cells, but in most instances this does not compromise a diagnosis. Still, to help ensure the adequacy of the sample, a midstream (“clean catch”) specimen is recommended.
Specimens obtained by catheterization have disadvantages for both the patient and cytologist. First, catheterization carries a risk of urinary tract infection. Second, urine collected from an indwelling catheter is often a pooled specimen that has been at room temperature for many hours, and cellular degeneration can be pronounced. Third, the tip of the catheter often scrapes off benign urothelial cell clusters that mimic the appearance of a low-grade papillary neoplasm.
Bladder washings are obtained through a catheter by irrigating the bladder with five to ten pulses of 50 mL of sterile normal saline, which produce a cellular suspension of freshly exfoliated epithelial cells. This specimen is collected before any biopsy sampling. The chief advantages of bladder washing over voided urine are better cellular preservation, greater cellularity, and less chance of contamination by background debris.
When an upper urinary tract malignancy is suspected, directed washings, brushings, and/or biopsies of a ureter or renal pelvis lesion are performed. Although brushings obtained by direct visualization using an endoscope were introduced in 1973, they are rarely obtained, even though their accuracy is similar to that of other cytologic methods.
The most common upper tract specimen is the directed washing with or without an upper tract biopsy. Directed washings are particularly challenging for both urologists and cytologists. The stakes are high because the operation of choice for a tumor in the upper tract is removal of the kidney, ureter, or both. Urologists often cannot visualize lesions in the upper tract as well as those in the bladder; hence, they rely on cytology at this site more than for lesions in the bladder. Although they may try to obtain a biopsy, often these biopsy specimens are small and crushed. In most cases, the significant imaging finding is a filling defect, and the differential diagnosis is tumor versus stone. Unfortunately, benign cytologic atypia produced by some stones can mimic the cytologic features of urothelial neoplasms. Finally, normal specimens from the upper tract often show diffuse nuclear enlargement, an elevated nuclear-to-cytoplasmic ratio, and very high cellularity. These entirely benign changes can suggest a tumor and result in a false-positive diagnosis. Despite these limitations, the sensitivity of the combination of ureteral washing cytology and ureteral biopsy for the detection of malignancy approaches 70% to 80%. With bilateral specimens, one can compare subtle changes between a lesion (on one side) and a presumably normal specimen (on the other side), although not all urologists sample the “normal” side. The preparation of a “cell block” (a formalin-fixed, paraffin-embedded sediment of the urine sample) can be particularly useful because small pieces of tumor are often easier to evaluate with this preparation method and may be more plentiful secondary to the urologist’s manipulation of the tumor. Cell blocks have less utility in other types of urine specimens.
At the time of cystectomy for bladder cancer, a segment of ileum is isolated and reanastomosed to the ureters (or to one ureter if a nephrectomy is also performed) to provide a conduit for urine. Urine samples from these conduits contain a large number of degenerated intestinal epithelial cells. It is important that these specimens be screened for malignant cells because patients with a history of bladder cancer have an increased risk for developing tumors of the ureters and kidneys. The cytomorphologic criteria for high-grade urothelial carcinoma are the same in this setting as for any other cytologic specimen, and the performance metrics of cytology are also similar.
Fresh specimens (less than 12 hours old) do not need fixation. If it will take a specimen 12 to 24 hours to reach the laboratory, refrigeration is recommended, and if more than 24 hours, preservation with an equal volume of 50% or 70% ethanol ± 2% carbowax is advised to avoid degeneration.
Slides can be prepared using a variety of concentration techniques, depending on the resources and preferences of the laboratory. These include sedimentation and smearing, membrane filtration, cytocentrifugation, and thinlayer methods. Slides are fixed in ethyl alcohol and stained with the Papanicolaou stain. Urine samples and bladder washings can also be prepared using the cell block technique ; the centrifuged sediment is fixed in formalin, and the slides are stained using hematoxylin and eosin.
Adequacy criteria for urine cytology have been proposed, but they are not yet fully established or widely adopted. Studies suggest that a volume of 30 mL or more of urine may have slightly higher sensitivity than specimens with lower volumes, but this is not statistically significant.
Absent or scant urothelial cells
Obscuring inflammation, blood, or lubricant
Marked degenerative changes
Even though normal urine specimens may contain no urothelial cells, a urine sample should have at least some urothelial cells for the cytologist to be sure that it is indeed urine. (An ileal conduit specimen is the exception.) In the hospital setting, for example, a laboratory might receive specimens from a patient with multiple drains, and the sample labeled “urine,” consisting of only blood and proteinaceous debris, may be from the wrong drain. Another situation is a voided urine sample from a woman that consists entirely of vaginal squamous cells. Studies suggest that specimens with less than 10 to 20 urothelial cells/10 high power fields (hpf) have a higher false-negative rate than those with higher cellularity and perhaps should be classified as a “limited specimen.” Paradoxically, specimens with more than 50 urothelial cells/10 hpf also have an elevated false-negative rate, presumably because the neoplastic cells are diluted by numerous benign urothelial cells.
In some cases, the urothelial cells are obscured by abundant acute inflammation, presumably because of an infection, blood, or use of lubricant jelly. In these settings, it is advisable to include a note stating that urothelial cells are poorly represented, and clinical correlation concerning the adequacy of the specimen is advised. In the case of abundant inflammation, further evaluation after treatment of the patient’s inflammation might be helpful.
Degenerated cells are common, especially with voided urine. If the specimen is so degenerated that an interpretation cannot be made (an uncommon circumstance), the specimen is inadequate. If only some cells are degenerated or the degenerated cells are abnormal, the specimen should not be interpreted as inadequate.
The results of urinary tract cytology are often reported using traditional diagnostic categories: “negative for malignant cells,” “atypical” (conveying a low degree of concern for malignancy), “suspicious” (conveying a strong suspicion for carcinoma), and “positive” (i.e., conclusive for malignancy). Because urine cytology is an insensitive test for low-grade urothelial neoplasms, some laboratories prefer “negative for a high-grade malignancy/carcinoma” rather than “negative for malignant cells.” The Paris System terminology is a variation that retains the simplicity of traditional terminology (see Table 3.2 ). It includes a category for low-grade urothelial carcinoma, but this has a very stringent criterion (a fibrovascular core with capillaries must be seen) and is thus used very sparingly (if at all) by any given laboratory.
| Traditional Cytology Terminology | The Paris System |
|---|---|
| Negative for malignancy | Negative for high-grade urothelial carcinoma |
| Atypical cells present | Atypical urothelial cells |
| Suspicious for malignancy | Suspicious for high-grade urothelial carcinoma |
| Positive for malignant cells | High-grade urothelial carcinoma |
| (Low-grade urothelial neoplasia) |
An atypical interpretation is generally treated as negative by the patient’s physician. The usual threshold for action on the part of the urologist is a suspicious interpretation, which warrants active investigation, such as cystoscopy. Hence, there is little practical difference between negative and atypical; the same applies to the distinction between suspicious and positive. The (perhaps surprising) lesson for the cytologist is that the most important distinction in urine cytology is arguably between atypical and suspicious.
The sensitivity of urine cytology for detecting bladder cancer is dependent on many factors, which explains the bewildering range of reported sensitivities (19% to 89%). These factors include how the sample was collected (voided vs instrumented), how many samples were obtained, how it was processed (filter, liquid-based, or cytospin preparation), and what defines a positive result (just “positive,” or “positive” plus “suspicious”).
One of the most important factors is the mix of low-grade and high-grade tumors in the population studied. It is now well accepted by urologists and cytologists alike that cytology is ineffective for the detection of low-grade tumors. Therefore, if one includes low-grade tumors, the overall sensitivity of voided urine cytology can be as low as 28%, even when suspicious and positive results are considered “positive.”
Happily, urine cytology is moderately sensitive in detecting high-grade cancers ( Table 3.3 ). For high-grade urothelial carcinoma, most studies of instrumented urine cytology show a sensitivity (combining “positive” and “suspicious” cases in the analysis) greater than 70% ; results are similar in the academic and community setting. A sensitivity below 60% for high-grade urothelial carcinoma suggests a laboratory problem (either processing or diagnostic) or an unusually difficult patient population to evaluate. The sensitivity of voided urine specimens for high-grade carcinoma is lower than for instrumented specimens.
| Number of Specimens Examined Per Patient | Number of Biopsy-Proven High-Grade Bladder Cancers | Positive Cytology (%) | Suspicious and Positive Cytology (%) | |
|---|---|---|---|---|
| Urine (voided) | ||||
| Schoonees et al. | 1 | 69 | 72 | 83 |
| Rife et al. | NS | 245 | 84 | 94 |
| Koss et al. | 3 | 103 | NS | 94 |
| Wiener et al. | NS | 46 a | NS | 89 |
| Halling et al. | 1 | 34 b | NS | 71 |
| Raab et al. | variable | 126 | 31 | 49 |
| Renshaw and Gould | NS | 162 | NS | 25 |
| Urine (instrumented) | ||||
| Zein and Milad | 1 | 150 | 77 | 86 |
| Raab et al. | variable | 40 | 50 | 65 |
| Piaton et al. | 1 | 101 | 77 | 82 |
| Renshaw and Gould | 1 | 152 | NS | 82 |
| Bladder washings | ||||
| Esposti and Zajicek | 1 | 140 | 89 | 94 |
| Loening et al. , c | 1 | 121 b | 70 | 81 |
| Curry and Wojcik | 1 | 37 | NS | 76 |
| Kim and Kim | 1 | 104 | NS | 89 d |
Sensitivity increases when more than one specimen is examined. For this reason, it is recommended that at least three specimens per patient be examined. Notably, the sensitivity of urine cytology is reduced in patients who have been treated with radiation or chemotherapy because they may have fewer neoplastic urothelial cells to shed as a result of their treatment.
The specificity of a positive urine cytology is high (range of most studies: 95% to 100%). False-positive results are uncommon but have occurred in patients with bladder stones, human polyomavirus infection, and chemotherapy. A positive cytologic result in the face of a negative biopsy may be a true-positive (termed an “anticipatory positive”) because histologic confirmation can be delayed for several years. Histologic confirmation is hampered by the subtle morphologic features of the tumor or its location outside the bladder (in the ureters, kidneys, prostate, or urethra). Until a convincing cause of a “false positive” is identified, these patients should be closely monitored.
Bladder washings have the advantage over voided urine samples in improved cellularity and cell preservation, and this is reflected in a slightly higher sensitivity (see Table 3.3 ). Nevertheless, 7% to 13% of bladder tumors detected by voided urine cytology have negative bladder washings. Washings of the ureter and pelvis have similar sensitivity (70% to 80% for high-grade lesions) but are particularly prone to false-positive results because of the marked cellularity and unusual cytologic features of these specimens.
Urothelial cells
Superficial (“umbrella”) and intermediate cells (voided urine)
Superficial, intermediate, and basal cells (catheterized urine, washings)
Squamous cells
Seminal vesicle and prostatic epithelial cells (very rare)
Degenerated intestinal epithelial cells (ileal conduit specimens)
A normal voided urine specimen is often sparsely cellular and usually contains at least some urothelial (synonym: transitional) cells ( Fig. 3.1 ). These are dispersed as isolated cells; clusters of urothelial cells are uncommon in a normal voided urine sample. Although earlier reports suggested that clusters of benign-appearing urothelial cells are associated with a higher risk of malignancy, subsequent studies failed to confirm this observation unless cytologic atypia is also present.
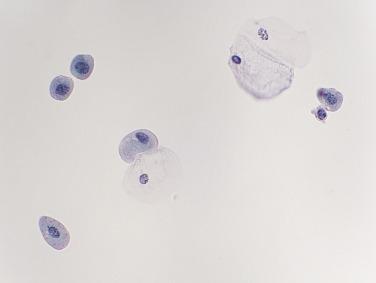
In voided specimens, most urothelial cells are intermediate in size, with a moderate amount of homogeneously granular or finely vacuolated cytoplasm, a round nucleus, and small nucleolus. In some cases they are columnar or spindled; this is a normal finding, although the reason for these shapes is not known ( Figs. 3.2 and 3.3 ). It is not uncommon for a normal urothelial cell nucleus to appear triangular or rhomboid because it is folded and has straight edges ( Fig. 3.4 ). Urothelial carcinoma cells often have angular nuclei, but theirs are usually larger, more elongated, and darker, and their chromatin is more coarsely granular than that of a normal urothelial cell nucleus.
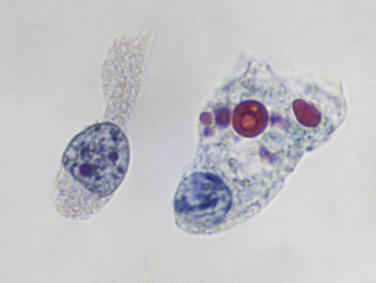
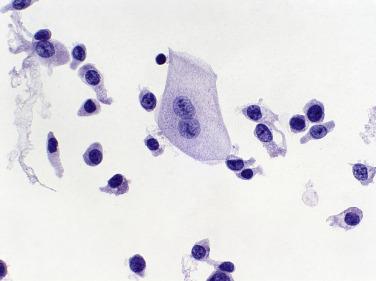
When degenerated, urothelial cells resemble histiocytes, especially because they sometimes contain round, red or green hyaline cytoplasmic inclusions called Melamed–Wolinska bodies (see Fig. 3.2 ). They are seen in almost 50% of urine specimens and are more common in voided as opposed to catheterized samples. The pathogenesis of these bodies is obscure, and they have no diagnostic value in urine, but they are useful in suggesting a urothelial origin for malignant cells in pleural effusions.
So-called umbrella cells are the superficial cells of the urinary tract. They are large and have abundant cytoplasm and large nuclei, and some have large nucleoli; binucleation and multinucleation are common (see Fig. 3.3 ). Although large and often multinucleate, umbrella cells have a very low nuclear-to-cytoplasmic ratio, relatively fine chromatin, and thin, smooth nuclear membranes, which help to distinguish them from malignant cells.
Squamous cells are common in voided urine samples; they exfoliate from foci of squamous metaplasia in the trigone of the bladder (a normal finding, especially in women). Squamous cells can also be picked up as urine passes through the urethra and urethral orifice. They can also be a contaminant from the vagina; a catheterized specimen can obtain a more pure specimen if clinically indicated.
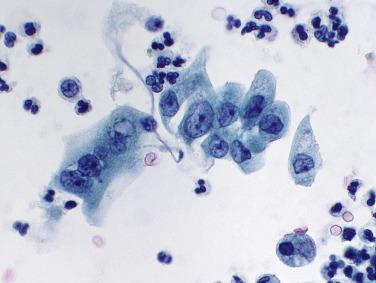
With catheterized samples, including washings and brushings, clusters of urothelial cells, some quite large, are typical and an entirely normal finding: the instrument mechanically abrades the mucosal surface, resulting in large numbers of cells in fragments. Thus the entire spectrum of basal, intermediate, and superficial (umbrella) cells is seen. Normal catheterized specimens, particularly washings and brushings, may appear worrisome for malignancy, particularly to the novice cytologist, due to the presence of intact mucosal fragments and the marked polymorphism of the cell population. Umbrella cells alone may be worrisome because of their size: they are among the largest of human epithelial cells. Even the small basal urothelial cells ( Fig. 3.5 ), because of their scant cytoplasm, are occasionally mistaken for carcinoma cells.
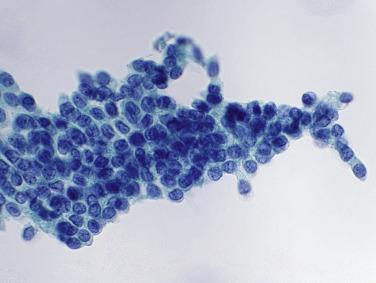
On rare occasions, seminal vesicle and prostatic epithelial cells are seen in urine samples from male patients. Seminal vesicle cells, in particular, sometimes have hyperchromatic nuclei and can be mistaken for malignant cells. The clue to their benign nature is the presence of lipofuscin, a golden-brown cytoplasmic pigment ( Fig. 3.6 ) that is normally present in some epithelial cells of the seminal vesicles and prostate.
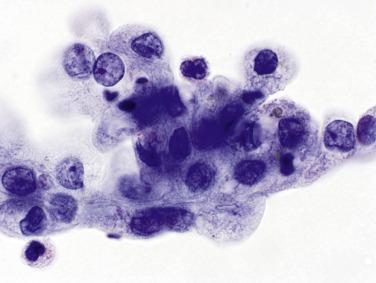
The intestinal epithelial cells in ileal loop specimens are dispersed as isolated cells and show marked degenerative changes ( Fig. 3.7A and B ), with eosinophilic intracytoplasmic inclusions like those of degenerated urothelial cells. They are commonly mistaken for macrophages by the novice.
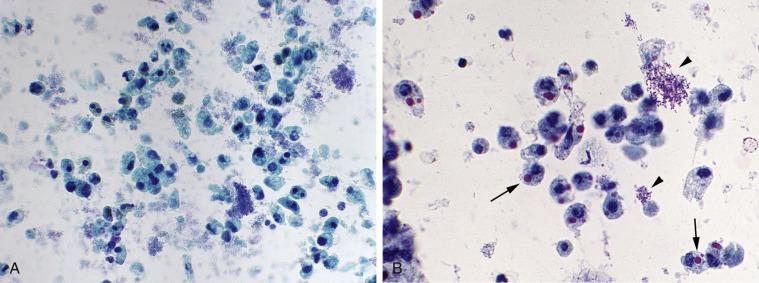
Bacteria, including malakoplakia
Fungi (especially Candida )
Herpes simplex virus
Cytomegalovirus
Trichomonas vaginalis
Polyomavirus
Papillomavirus
Crystals, red blood cells, and infections, including bacteria, viruses, and Candida , are commonly seen, but these findings are usually more thoroughly assessed by urinalysis or culture. Infections of the bladder are caused by bacteria, viruses, parasites, or fungi. In the United States, bacterial infections are most common. Cytologic preparations in cases of bacterial cystitis show a dense concentration of white blood cells (predominantly neutrophils), with plasma cells, lymphocytes, histiocytes, and numerous red blood cells. Bacteria are present, sometimes in overwhelming numbers. The inflammation can obscure the urothelial cells. The presence of bacteria in the absence of abundant neutrophils is nonspecific. This may be the result of vaginal or urethral contamination, but many patients, particularly females, have bacteria in the bladder without clinical symptoms of cystitis. Likewise, neutrophils are not necessarily indicative of a clinically significant infection.
Malakoplakia is an uncommon histiocytic inflammatory lesion of the bladder or upper respiratory tract that results from bacterial infection. Diagnosis by urine cytology is very uncommon. The cytologic hallmark is the presence of histiocytes whose abundant granular cytoplasm is filled with bacteria and bacterial fragments. The basophilic, round, lamellated bodies known as Michaelis-Gutmann bodies measure approximately 8 μm and may be intracellular within histiocytes or extracellular.
The most common fungus that infects the bladder is Candida. The organism is present as yeast forms and pseudohyphae, accompanied by a cellular, mixed inflammatory background. Urothelial cells usually show reactive changes. When Candida is present in the urine of female patients, the possibility of vaginal contamination should be considered. Vaginal contamination, rather than true infection, is likely when the background contains numerous squamous cells and bacteria and few neutrophils. As with bacteria, some patients have Candida in their bladder without symptoms of cystitis. Aspergillus can be detected in urine from patients with bladder aspergillosis.
Viral infections of the bladder include herpes simplex virus, cytomegalovirus, human polyomavirus, and human papillomavirus. Herpetic infection of the bladder is uncommon, usually seen in the immunocompromised patient. The cytopathic changes include multinucleation, a ground-glass chromatin texture, and peripheral condensation of chromatin. In some cases the cells have a large eosinophilic nuclear inclusion that can be sharply angulated. Nuclear molding is often observed. Infected cells can be greatly enlarged and bizarrely shaped, with dense, opaque cytoplasm.
Cytomegalovirus affects urinary epithelium, most commonly renal tubular cells, in immunocompromised patients. Affected cells are markedly enlarged and have both nuclear and cytoplasmic inclusions. The nuclear inclusion is solitary, darkly basophilic and surrounded by a zone of chromatin clearing. The multiple cytoplasmic inclusions are more variable in appearance: smaller, basophilic, and either finely or coarsely granular.
The parasitic protozoan Trichomonas vaginalis is responsible for one of the most common sexually transmitted diseases. It is usually associated with vaginitis, but it can cause urethritis and even prostatitis. In urine from a woman, the organisms are most likely contaminants from a vaginal infection if they are accompanied by abundant squamous cells and vaginal flora. Rarely, they cause urethritis in men and are identified in urine cytology. The organism varies in size, with an average length and width of 10 and 7 μm, respectively. The nucleus is small and oval, and the cytoplasm contains fine red granules (see Fig. 1.21A ).
BK virus, first isolated in 1971 from the urine of a renal transplant patient with initials B.K., is a human polyomavirus that is acquired in childhood and becomes latent in urothelial cells of the kidney and bladder. Immune suppression after transplantation results in BK virus reactivation in transplant recipients and clinical disease in some patients. Disease manifestations range from mild dysuria to life-threatening hemorrhagic cystitis and renal failure.
Sparse to abundant altered cells
Isolated cells, sometimes binucleated or multinucleated
Enlarged, round, eccentrically placed nucleus (tear-drop, comet-like cell shapes)
Basophilic, glassy inclusion fills entire nucleus
Thick nuclear membrane
Degenerated nuclei with a coarse, reticular chromatin pattern
Characteristic viral cytopathic changes are seen in 4% of urine samples. The infection usually has no clinical significance except in immunocompromised patients, particularly transplant patients, but infected cells appear atypical and can be confused with malignant cells. Infected urothelial cells have a large, eccentrically placed nucleus. The most characteristic alteration is a basophilic nuclear inclusion that completely replaces the nucleus and appears glassy, opaque, or cloudy ( Fig. 3.8A–D ). Nuclear membranes are markedly thickened. In children and immunosuppressed adults, the altered cells are numerous, whereas in immunocompetent adults they are usually few in number. In contrast to cytomegalovirus-infected cells, in which a halo surrounds the inclusion, the inclusion of polyomavirus affects the entire nucleus.
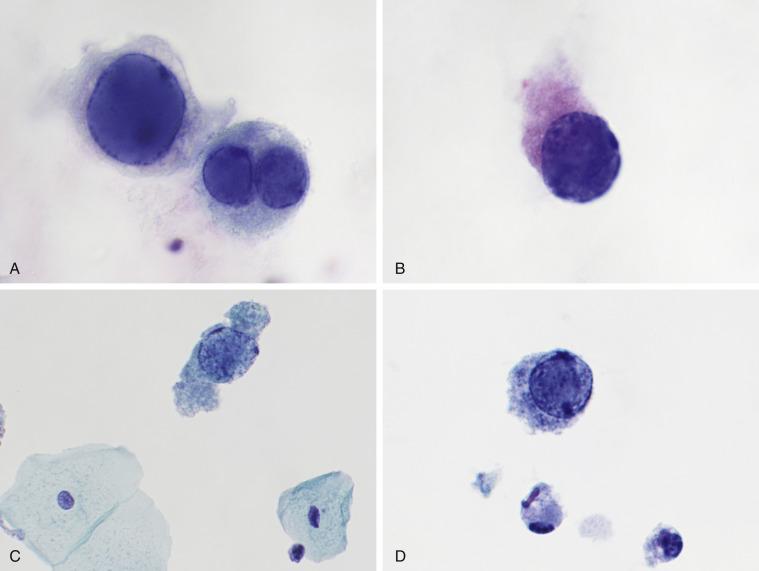
Because of their increased nuclear size and hyperchromasia, polyomavirus-infected cells can be confused with malignant cells; hence their pseudonym decoy cells . Unlike most malignant cells, however, decoy cells have a smooth and round nucleus. In contrast to tumor cells, which often cluster to form groups, polyomavirus-infected urothelial cells are found only as isolated cells. Because degenerated urothelial carcinoma cells can resemble decoy cells; however, one should interpret specimens as atypical if the morphology of the decoy cells is not classic; in such cases, a note recommending clinical correlation may be the best option. Testing for polyomavirus by immunohistochemistry or PCR is available and helpful in difficult cases. The differential diagnosis of decoy cells also includes degenerated benign urothelial cells. Polyomavirus-infected nuclei appear smudged and densely basophilic, and the chromatin is more uniform in texture than that of degenerated urothelial cells.
Human papillomavirus can infect the urinary tract, but when cytopathic changes characteristic of this virus are seen in a voided urine specimen from a woman, the cells most likely have originated from the vulva or vagina. Koilocytes in a catheterized specimen, however, indicate a condyloma in the urinary tract ( Fig. 3.9A and B ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here