Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Urine cytology as a method for diagnosing bladder carcinoma was introduced in 1945 by Papanicolaou and Marshall. Urothelial cells are present in all urine specimens and exfoliate readily from tumors of the urothelial lining. Urine cytology is therefore an important primary method of diagnosing urothelial tumors, and it is used as an adjunct in combination with cytoscopy and biopsy.
Cytologic examination of urine is performed in screening programs of asymptomatic but high-risk patients, in the diagnostic evaluation of symptomatic patients, and in the follow-up and monitoring of patients with known and treated disease. The clinical history, including any prior surgical procedures, treatment modalities, and comparison with previous biopsy or cytologic specimens, is essential for a cytopathologic evaluation. This may permit the diagnosis of recurrent well-differentiated tumors that could not be identified otherwise, and prevents overdiagnosis of atypias that may be associated with previous treatment.
Screening programs for bladder cancer by cytologic examination have been undertaken and reported sparingly. Such programs were conducted for individuals considered at high risk, such as industrial workers exposed to aromatic amines or cadmium, phenacetin or opium abusers, and bilharzial populations. Holmquist reported results of screening of 9870 outpatients by routine urinalysis wet preparations. Of these, 148 (1.5%) were considered abnormal. Follow-up studies revealed evidence of cancer in 12 of these patients, a rate of 1.2 per 1000. Toluidine blue and phase-contrast microscopy have been advocated for rapid screening of a clinic or hospital population. This then must be supplemented by examination of stained slides if abnormal findings are identified.
Based on the known high sensitivity of urine cytology for non-papillary carcinoma in situ, which is the usual precursor of invasive bladder cancer, screening programs of carefully chosen high-risk populations should yield significant results. This is indeed the case, and the positive yield in screening programs for high-risk groups ranges from 10 to 70 per 1000 persons. The yield is only 2 per 1000 individuals in a bilharzial population and 10 per 1000 in phenacetin users but still exceeds that in a routinely screened clinic or hospital population. Screening programs clearly lead to detection of bladder cancer at a stage earlier than that in patients who present with symptoms in unscreened groups, provided that the screening procedure is applied frequently enough. Nevertheless, existing evidence does not establish that treatment based on early detection of bladder cancer in screened populations increases the overall length of survival.
The most common presenting symptom in patients with bladder cancer is gross hematuria. Patients with gross or microscopic hematuria must be further evaluated until the source of the bleeding has been securely established. This may require an intravenous pyelogram, computed tomography, or magnetic resonance imaging study because of the possibility of renal carcinoma. Cytologic examination of the urine is indicated in these cases, and, depending on the findings, it may be followed by bladder washings for flow cytometry, cystoscopy, and bladder biopsies. Cytologic examination of voided urine is a simple, non-invasive procedure with high sensitivity for papillary and non-papillary urothelial carcinoma in situ and should be performed in all symptomatic patients. If combined with DNA ploidy measurements and immunohistochemical studies, the sensitivity exceeds 80% even for low-grade papillary tumors.
Urine cytology is of particular value in the follow-up of patients with known and treated urothelial tumors. Low-grade papillary tumors, whether they are primary or recurrent, cannot be readily diagnosed cytologically but high-grade bladder tumors and all carcinoma in situ can be diagnosed with a high degree of accuracy. These are the significant bladder cancers that frequently recur or may be associated with low-grade papillary tumors. Cystoscopy and biopsy are the appropriate procedures for visible tumors, but cytology has the distinct advantage of sampling the entire urothelial mucosa. This permits detection of clinically occult and in situ urothelial tumors. Most patients with superficial non-papillary bladder carcinoma develop clinically apparent recurrent carcinoma; yet only 40% have positive urine cytology results immediately after transurethral resection and before clinically apparent and documented recurrent bladder cancer. This may be because of inflammation and regenerative epithelial atypia in the immediate postoperative period, which may mask minimal cytologic evidence of carcinoma. If recurrence follows positive postoperative cytologic findings, it can be assumed to arise from residual neoplastic epithelium. Recurrence after negative postoperative cytologic findings suggests transformation within an unstable urothelium and is often multicentric. Many of the currently available markers for carcinoma such as bladder tumor antigen, NMP22, fibrin degradation products, telomerase, fluorescence in situ hybridization (FISH), and flow cytometry appear to have an advantage over urine cytology in terms of sensitivity. However, most markers tend to be less specific than cytology, yielding more false-positive results, according to Bassi and colleagues. The simplicity, convenience, and relative accuracy of cytologic study of urine make it the first-line diagnostic technique.
Voided urine is the specimen of choice for all screening programs and for diagnostic studies of male patients because of the ease of collection and satisfactory results. Catheterized urine is the preferred specimen from female patients. Hydration of patients, collection of the second voiding in the morning, and collection of three successive morning specimens have been recommended by some investigators, but because of the good cellularity that can be uniformly obtained by filtration or cytocentrifugation preparations as shown in Fig. 15-1 and the significant exfoliation from all non-papillary tumors of the urothelium, examination of a single urine specimen is sufficient.
Prompt fixation is important and can be best accomplished by collection of 50–100 mL of urine in an equal amount of 50% alcohol. Ethyl alcohol is preferable, but isopropyl or denatured alcohol is acceptable. If the specimen is to be processed by the Saccomanno blending technique, 2% polyethylene glycol (Carbowax) must be added. Urothelial cells remain well preserved for processing for several days, regardless of whether the urine is collected after hydration, during the morning hours, or later during the day. Urine osmolality does not significantly influence cell preservation, but a low pH is desirable and ingestion of 1 g of vitamin C at bedtime before examination has been recommended, although not practical for screening purposes.
Bladder washings with normal saline or Ringer's solution performed at cystoscopy, if indicated in combination with biopsies or resections, produce a highly cellular specimen that contains more cell clusters and more large, superficial cells than are seen in voided urine. This procedure is recommended whenever cystoscopy is performed and is the specimen that should be used for flow cytometric studies. The washings should also be collected in equal amounts of alcohol for fixation.
If lesions in this region are suspected, urine aspirates, washings, and brushings can be obtained by retrograde catheterization. These specimens contain larger numbers of superficial and often multinucleated urothelial cells with greater variation in their appearance than bladder urine and must be interpreted conservatively. Cell blocks may be prepared when tissue fragments are present.
Direct smears may be prepared after centrifugation of 50 mL of urine for 10 min at 1200 rpm. Albuminized or charged slides are recommended for better attachment of cells in direct smears.
Specimens may be processed using a filtration or a cytocentrifugation protocol. The slides are then stained by the Papanicolaou method. (See Chapter 33 for details.)
Cell blocks may be prepared when visible tissue fragments are present. (Cytopreparatory techniques for urine specimens are described in detail in Chapter 33 .)
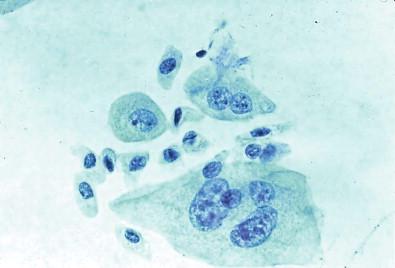
Urinary tract cytology is almost exclusively concerned with evaluation of the urothelium that lines the urinary bladder and other excretory passages – namely, the renal pelves, ureters, and portions of the urethra ( Fig. 15-2 ). The epithelium is adaptable to the changing volume and permits the storage and discharge of the urine. This urothelium is highly specialized and uniform but does include epithelial buds, subepithelial often centrally cystic nests (the Brunn's nests), and areas of squamous metaplasia or squamous mucosa. The urothelial cells have unique ultrastructural features including surface folds believed to provide additional plasma membrane during bladder expansion and robust junctional complexes that provide a watertight seal. The predominant urothelium in the calices is two or three cells thick, in the ureters is four or five, and in the empty bladder is lined by six to eight layers of urothelial cells. Three cell types are recognizable by light microscopy. These are the large, often binucleated or multinucleated superficial umbrella cells covering the cells that form the lower portions of the epithelium-like umbrellas. The underlying intermediate pyramidal cells are smaller and somewhat elongated. A layer of even smaller cuboidal cells with little cytoplasm is located adjacent to the basement membrane. The three cell types can be distinguished by flow cytometry and by different patterns of lectin binding. These cells can also be well defined by morphologic examination of urine specimens. The superficial umbrella cells are characterized by a shape maintained by a rigid surface membrane ( Fig. 15-3 ). They have an acidophilic cytoplasm. Many are binucleated and some (approximately 3%) are multinucleated ( Figs 15-4 , 15-5 ). The nuclei are large and are likely to undergo reactive change. The intermediate and basal cells are similar and have well-developed desmosomal connections and a plasma membrane that is folded in the empty bladder. It is capable of unfolding when the bladder becomes dilated. The arrangement of these cells is regular, with their long axis perpendicular to the basal membrane. Their cytoplasm contains glycogen, often resulting in a clear appearance during processing. The superficial cells also contain glycogen and sometimes mucus.
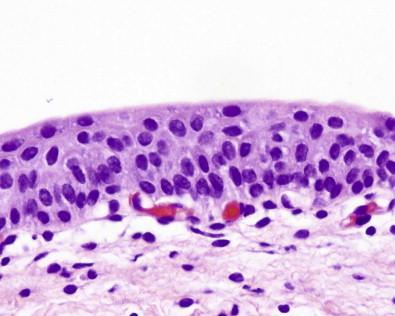
The prostatic segment of the male urethra is lined by urothelium, but the short membranous and long cavernous portions of the urethra are lined by stratified or pseudostratified columnar epithelium. The epithelium near the meatus is squamous. Many recesses, namely the lacunae of Morgagni, communicate with deeper branching tubules and the glands of Littré. The epithelium of the female urethra near the bladder is also urothelium and in the remaining parts usually squamous, often with interspersed areas of pseudostratified columnar epithelium. Invaginations lined by mucus-producing cells similar to those found in the glands of Littré may be present.
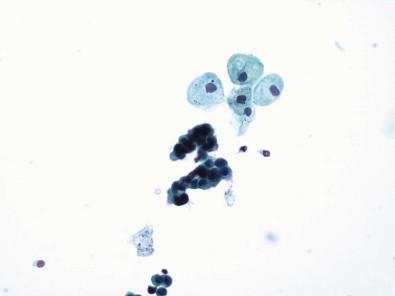
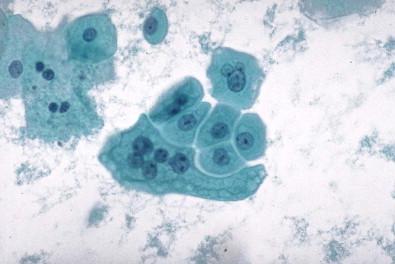
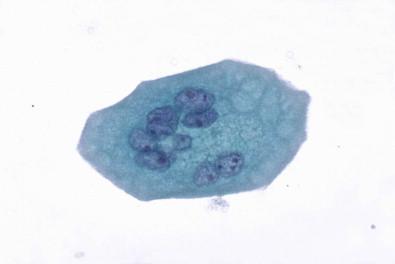
Urothelial cells are present in all urine specimens ( Fig. 15-1 ). In voided urine, they occur singly or in the form of loosely cohesive clusters or sheets. They vary considerably in size ( Figs 15-3–15-5 ), with a cell diameter from 9 to 40 µm and a cell area size of 75–1400 µm 2 with a mean of 340 µm 2 . The cytoplasm is opaque, granular, or vacuolated ( Fig. 15-6 ). Renal tubular epithelial cells are rarely found in voided or catheterized urine, except in cases of renal transplant rejection or renal parenchymal disease ( Fig. 15-7 ). The renal tubular epithelial cells have round nuclei and usually a granular cytoplasm. Many urothelial cells are elongated, and columnar cells may be present ( Fig. 15-8 ). The nuclei are usually round, but they may have an irregular outline or may appear pyknotic. Large single nucleus or binucleated superficial umbrella cells are particularly common in bladder washings and in ureteral catheterization specimens. The nuclei average 7.5 µm in diameter and the area vary from 10 to 90 µm 2 , with a mean of 36 µm 2 . The nucleoplasm is usually bland or vesicular, and the chromatin pattern of most nuclei is finely granular or has a salt-and-pepper appearance ( Fig. 15-9 ). Small nucleoli are rarely present. The described three cell types of the urothelium – namely, the large, often multinucleated superficial umbrella cells, the intermediate pyramidal cells, and the cuboidal cells adjacent to the basement membrane – can be identified in single cell suspensions prepared from tissues and in voided and catheterized urine specimens. Eldidi and Patten reported that the basal cells have a mean cytoplasmic area of 82 µm 2 , a mean nuclear area of 24 µm 2 , and a relative nuclear area of 29 µm 2 . They are predominantly oval, with basophilic, dense cytoplasm and single nuclei. The pyramidal cells are larger, with a mean cell area of 229 µm 2 and a mean nuclear area of 40 µm 2 . They are also predominantly oval with a basophilic cytoplasm and well-defined cell borders. The nuclei are generally single and oval. The superficial umbrella cells are considerably larger, with a mean area of 500 µm 2 and a mean nuclear area of 64 µm 2 . The relative nuclear area is 13%. These cells are less basophilic. Binucleation occurs in 19%, and 3% are multinucleated. Most nuclei are oval. Nucleoli are more prominent than in the smaller cells. Pyramidal or intermediate cells predominate in voided urine, but 42% of cells in washings are superficial. They are particularly prominent in ureteral catheterization specimens. Cell aggregates and cytoplasmic molding are also more common in catheterized rather than voided specimens. The large multinucleated cells prominent in ureteral or renal pelvic washings may contain as many as 20 or 30 centrally crowded nuclei ( Fig. 15-10 ).
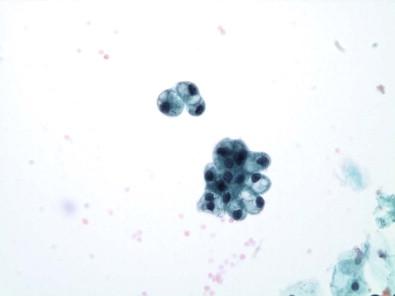
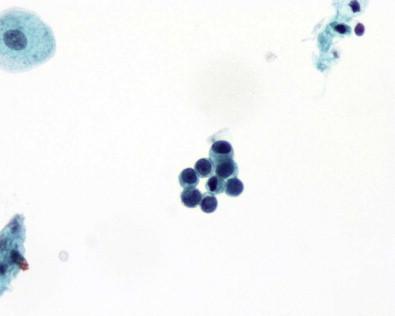
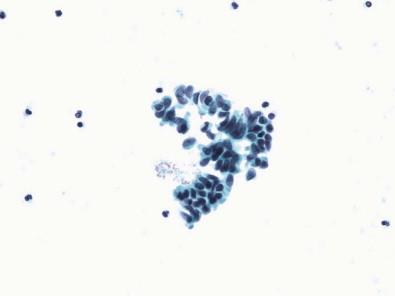
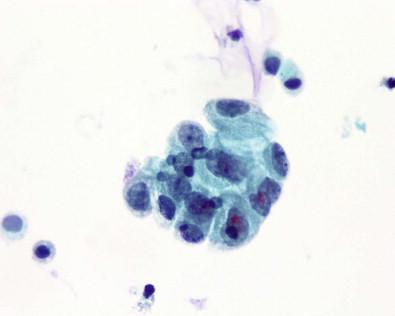
Squamous cells of intermediate and less commonly of superficial type are frequently present, more often in women ( Fig. 15-11 ). They exfoliate from portions of the urethra lined by squamous epithelium, from areas of squamous metaplasia in the urinary bladder, or from zones of squamous epithelium that may be found in the trigone of women. Some squamous cells may represent vaginal contamination. The squamous cells present do reflect the effect of estrogen. Squamous metaplastic cells are often regular and well differentiated. Exfoliated squamous cells cannot be distinguished from cells that exfoliate from areas where squamous epithelium is normally found. The Brunn's nests are buds or sprouts of urothelium that grow from the surface into the underlying lamina propria and may become centrally cavitated or cystic. The cells lining the cystic spaces may remain flattened or low cuboidal and resemble urothelium, but others become columnar and mucinous. These changes are considered to be reactive and metaplastic. When the morphologic appearance is characterized by the presence of mucin-secreting colonic-type epithelium, they are referred to as cystitis glandularis or as intestinal (goblet cells) or glandular metaplasia. Many of the columnar cells often seen in urine are cells from the intermediate zone of the urothelium, but they may exfoliate from metaplastic columnar or glandular epithelium, from the urethra, and in males, after instrumentation or prostatic massage, from the prostate. Spontaneous exfoliation of prostatic columnar cells or seminal vesicle cells that may contain cytoplasmic pigment is rarely observed. The lacunae of Morgagni and glands of Littré are other possible sites of origin. Most columnar cells are non-ciliated, but, rarely, ciliated cells have been described in voided urine. They may represent cells from the seminal vesicles, vasa deferentia, or the epididymis, structures lined by columnar cells with stereocilia, or they may be the result of an unusual differentiation. The morphologic appearance of columnar cells depends on their site of origin. If benign, the nuclei are oval, uniform, or bland or have a finely granular chromatin pattern ( Fig. 15-8 ).
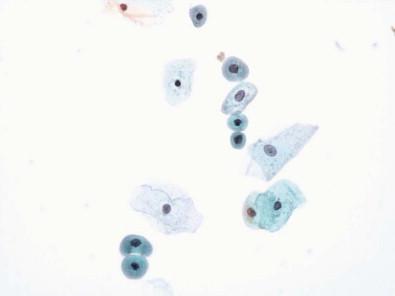
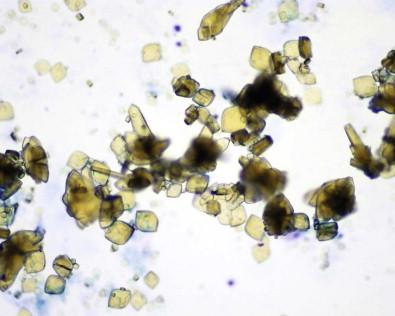
Small numbers of polymorphonuclear leukocytes are often present in normal urine. Histiocytes are found in inflammatory conditions but may be present in normal urine in small numbers. They have indistinct cell borders, a foamy cytoplasm, and eccentric, lobed, or bean-shaped nuclei. They are often difficult to differentiate from degenerated epithelial cells. Spermatozoa, crystals ( Fig. 15-12 ), Trichomonas , and lubricant ( Fig. 15-13 ) may also be seen.
Degenerative changes of exfoliated cells suspended in urine, in the bladder, or after voiding do occur and may interfere with interpretation, but extensive quantitative studies did not demonstrate a relationship between either cell size or degree of degenerative change and the osmolality.
Urothelial cells single, loosely cohesive clusters
Variably sized cells
Opaque, granular, or vacuolated cytoplasm
Squamous cells
Metaplastic cells
White blood cells
Red blood cells
Other (spermatozoa, Trichomonas , crystals, etc.).
Degenerative changes of exfoliated cells suspended in urine, in the bladder, or after voiding do occur and may interfere with interpretation. The most important step to eliminate degenerative changes is immediate fixation of the collected urine with an equal amount of 50% alcohol. If this prompt fixation is then followed by well-controlled cytopreparation, with smears or filtration or cytocentrifugation-based preparations degenerative changes do not interfere with interpretation. Reactive changes may be difficult to differentiate from neoplastic ones, as described later, but the most significant problem in interpretation is the distinction of the large umbrella cells from tumor cells. The morphology of these cells must be known. Their prominence in ureteral catheterization specimens is particularly striking. This misinterpretation may lead to nephrectomies of non-neoplastic kidneys.
Patency of the urachus at the bladder dome may lead to a urachal diverticulum, cyst, or to the presence of columnar epithelium in this area. This may be a source of exfoliated columnar cells in the urine, and adenocarcinomas are known to develop from this tissue. Exstrophic bladders may also be lined by columnar epithelium that may become the site of adenocarcinomas. Cytology has a limited role in the evaluation of this abnormality.
Endometriosis is the presence of endometrial glands accompanied by endometrial stroma in abnormal locations. It commonly involves the ovaries and pelvic peritoneum and less commonly the colon, appendix, vagina, and umbilicus. Even less common than at those sites, endometriosis of the urinary tract has been reported, and the bladder is the most common site of involvement. The condition usually occurs in women of childbearing age. The symptoms include hematuria, which may be cyclic, dysuria, and suprapubic pain. If the surface epithelium is involved and the urothelium is penetrated, endometrial stromal and glandular cells may be shed into the urine. The cytologic presentation of these endometrial cells in urine is very similar to that seen in vaginal smears. The presence of small cells with a high nucleocytoplasmic ratio in a urinary specimen raises the possibility of poorly differentiated urothelial carcinoma or lymphoma, but the marked uniformity of the cells and their occurrence in exodus-type clusters should suggest the possibility of endometriosis of the bladder.
Inflammation of the lower urinary tract is commonly caused by bacteria, usually as a complication of an obstructive process such as prostatic hyperplasia or carcinoma, strictures, or calculi. Positive urine cultures most frequently contain Escherichia coli (32%), Streptococcus group D (10%), Proteus mirabilis (9%), Pseudomonas aeruginosa (9%), and Klebsiella pneumoniae (6%) organisms. Tuberculosis of the bladder is rare and usually associated with and secondary to tuberculosis of the kidneys. It appears as granulomatous cystitis with inflammatory cells, elongate epithelioid histiocytes and multinucleated giant cells. Granulomatous cystitis is more commonly observed in patients with BCG treatment for urothelial carcinoma or patients with immunosuppression such as in acquired immunodeficiency syndrome (AIDS).
Regardless of the etiology, cystitis is cytologically characterized by the presence of polymorphonuclear leukocytes, necrotic debris, histiocytes, degenerative changes in urothelial cells, urothelial hyperplasia, and often reactive urothelial atypia ( Fig. 15-14 ). Slightly greater irregularity of the cell outlines and increased percentage of cells that contain generally small nucleoli with occasional coarsely granular nucleoplasm are noted. Red blood cells are invariably present. The urothelial cells are increased in number and are often obscured by the inflammatory cell population. The nuclei tend to be moderately enlarged, to an average nuclear diameter of 8–10 µm. They often contain clear zones or chromatin “bars” and may contain nucleoli. The cellular outline is usually hazy. Degenerated cells may be large and may have a clear or vacuolated cytoplasm that contains collections of polymorphonuclear leukocytes. Sizeable clusters of partially necrotic cells may be present. The coarsely granular chromatin pattern of malignant urothelial cells or macronucleoli is not seen. A very dense, cellular inflammatory exudate associated with hyperplastic or degenerating urothelial cells or partially necrotic cell clusters without the nuclear characteristic of malignancy or severe dysplasia is of infectious or inflammatory origin. Even extensively ulcerated and necrotic bladder urothelial carcinomas exfoliate unequivocally malignant cells and a greater amount of necrotic material and relatively fewer leukocytes than in cystitis.
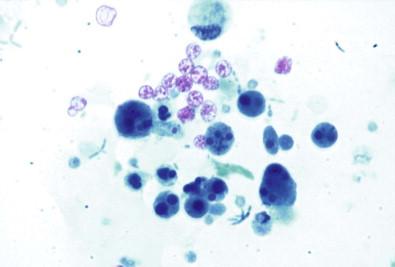
Polymorphonuclear leukocytes
Necrotic debris
Histiocytes
Degenerative changes
Reactive urothelial atypia.
Fungi such as Candida albicans appear as yeast or pseudohyphae, and are usually present in urines of pregnant women, diabetics, or those with impaired immunity ( Fig 15.15 ). Other types of fungi are rare.
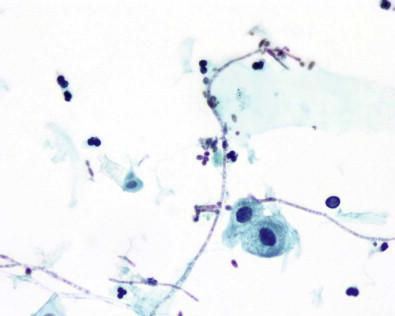
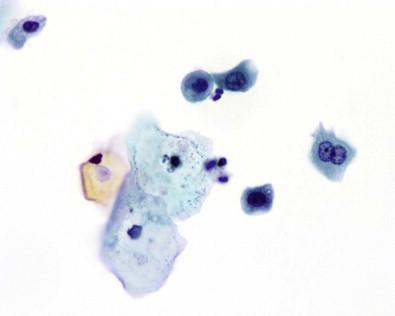
A number of virus infections cause morphologically characteristic changes in renal cells and in the urothelium. Cytomegalovirus (CMV), a member of the herpes virus group, is spread by intrauterine or perinatal transmission to the fetus by an infected mother, sexually, or through blood products. Young children and immunosuppressed patients (cancer, AIDS or post-transplants) are at risk. The renal tubular epithelium is frequently involved, and the disease is seen in renal transplant recipients. The affected cells are enlarged and contain a large basophilic or eosinophilic single nuclear cytomegalic inclusion surrounded by clear halo. The inclusion is sharply demarcated from the nuclear membrane. Remnants of chromatin are condensed peripherally ( Fig. 15-16 ).
Herpes virus isolation from human bladder urine has been described by Masukawa and coworkers. The viral cytopathic changes are the same as in other body sites. Urothelial cells are multinucleated, with smudgy chromatin, nuclear molding, or eosinophilic nuclear inclusion bodies surrounded by halos.
Papova viruses are divided into two groups: papilloma viruses and polyoma viruses. Polyoma virus is of particular importance in urine cytology because active infection is often localized to the brain and urinary tract. Urinary excretion of viruses in pregnant women has been reported, showing that the fetus is at risk of infection. Like CMV, the virus also infects or is reactivated if the host's immunity is impaired by immunosuppressive drugs such as those given to renal allograft recipients or by AIDS or cancer. Polyoma virus is emerging as a major cause of allograft dysfunction and loss. The BK-virus (BKV) genotype in urine and serum is significantly related to a high frequency and high number of virus-infected cells, also known as decoy cells. Decoy cells can be misinterpreted as carcinoma in situ, however the chromatin pattern is opaque or glassy compared to the hyperchromatic dense chromatin of carcinoma in situ. More frequently, the BKV is found in patients with other types of urinary tract infection, hematuria, or prostatic hypertrophy, reportedly in 0.3–1.2% of urine specimens in these cases. There are usually no sequelae of the infection. The virus is readily recognized morphologically in cells exfoliated in the urinary tract, and the principal differential diagnosis is with CMV. The inclusion-bearing cells are even larger than those infected with CMV and contain dense, basophilic, homogeneous nuclear inclusion bodies ( Fig. 15-17 ). Usually, only a rim of chromatin is seen at the edges of the inclusion, beneath thickened nuclear membranes. In contrast to CMV-infected cells, in which the virus inclusions are surrounded by a clear zone, the basophilic homogeneous inclusion in a polyoma virus-infected cell often completely fills the enlarged nucleus. If a clear halo remains, it is sharply distinguished from the thickened nuclear membrane. Electron-microscopic examination shows the virus particles to be unencapsulated, in contrast to the encapsulation observed in CMV. Immunoperoxidase procedures with simian virus 40 antiserum have been successfully used to confirm infection by polyomavirus.
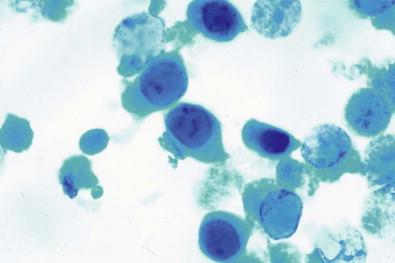
The human papilloma virus is responsible for condylomatous lesions in the lower urinary tract, but these lesions are generally confined to the urethra and the mucocutaneous regions of the meatus and are readily accessible for biopsy and excision. Koilocytosis and other morphologic changes characteristic of this condition are described elsewhere.
Decoy cell in polyoma virus (BKV)
Cytomegalic single nuclear inclusion in CMV
Multinuclear inclusions with molding and margination in herpes
Koilocytes in papilloma virus.
Malacoplakia is a granulomatous disease of the bladder or upper urinary tract. Multiple soft, nodular, often umbilicated yellowish plaques appear in the mucosa and submucosa, frequently in the region of the trigone. They may be mistaken for cancer clinically and are associated with immunodeficiency states. Several cases have been encountered in transplant recipients.
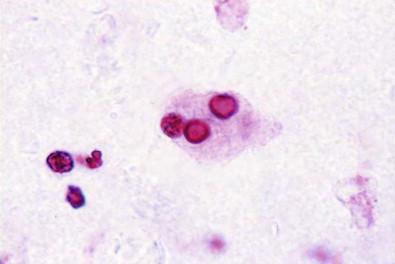
Histologically, the plaques are found to consist of epithelioid histiocytes with a granular, acidophilic (eosinophilic) cytoplasm. Some of the cells contain homogeneous or concentrically layered intracytoplasmic inclusions known as Michaelis–Gutmann bodies or calcospherites. The inclusions are basophilic and periodic acid–Schiff positive and stain for iron and calcium. They may represent the end result of bacterial degradation and degenerative changes, and the process is regarded by some as a defect in the host macrophage response to bacterial infection, usually Gram-negative bacilli. The urine contains histiocytes of this type, often with multiple granules in a foamy cytoplasm ( Fig. 15-18 ). Some may be found to contain the calcospherites that average 8 µm in diameter. The condition is rare, and finding diagnostic cells in urine cytology is exceptional.
Disintegration of the cytoplasm, pyknosis of the nuclei, and karyolysis of the nuclei of scattered urothelial cells may be seen in urine from healthy individuals as well as in urine containing malignant cells. Cells with an attached tag of partially preserved cytoplasm were initially described by Papanicolaou and are sometimes called “comet” or “decoy” cells. They may have some of the characteristics of malignancy, and it is therefore important that they be recognized for what they are. For the most part, they contain round or oval nuclei that have a coarse chromatin network or are opaque and hyperchromatic. Occasional cells are binucleate. The smooth nuclear outline, evidence of cytoplasmic degeneration, and the usually small number of these abnormal cells in an often hypocellular urine specimen should prevent a false-positive diagnosis. It should be noted that they are rare in immediately fixed urine specimens.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here