Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Descemet membrane endothelial keratoplasty (DMEK) is not suitable for all eyes.
Central thickness of Descemet stripping automated endothelial keratoplasty (DSAEK) grafts may influence the visual results.
A single- or double-pass technique can be used to reproducibly obtain DSAEK grafts of a predetermined thickness and planar profile.
Ultrathin (UT) DSAEK can be performed in patients with endothelial decompensation of any etiology.
Best spectacle-corrected visual acuity (BSCVA) ≥20/20 can be achieved in up to 49% of eyes with 20/20 visual potential undergoing UT-DSAEK.
The mean endothelial cell loss after UT-DSAEK (33.3% at 6 months) is similar to that recorded after DSAEK and DMEK.
UT-DSAEK offers the potential to achieve the visual outcomes of DMEK with the ease of handling and tissue preparation of conventional DSAEK.
To date, various authors have reported the outcomes of Descemet stripping automated endothelial keratoplasty (DSAEK), highlighting the relatively long time required for visual rehabilitation and suboptimal final level of visual acuity obtained. ,
Two new different approaches to endothelial keratoplasty (EK) have been developed with the purpose of overcoming the limits of conventional DSAEK. In 2006 Melles introduced Descemet membrane endothelial keratoplasty (DMEK), a procedure utilizing a donor graft consisting of only Descemet membrane and endothelium. In comparison with conventional DSAEK, DMEK has shown faster visual recovery, better visual outcomes, and reduced rejection rates. , However, DMEK is still performed less regularly than DSAEK, mainly because it is more technically challenging, and has an increased rate of both intraoperative and postoperative complications. , Unlike DSAEK, it is not suitable for all eyes. DMEK is not a good choice in cases with increased vitreous pressure, shallow or poorly visualized anterior chambers, or in eyes without anatomic barriers between the anterior chamber and the vitreous cavity. Finally, the learning curve of DMEK is accompanied by a high rate of tissue loss (up to 16%), a high detachment rate of up to 63%, and a failure rate of up to 8%, which is higher than that of other procedures.
Furthermore, DMEK was developed based on the assumption that the presence of a stromal interface was responsible for the suboptimal visual outcomes of DSAEK. However, although more than 50% of patients undergoing DMEK in the absence of comorbidities reach 20/20 vision, more than 40% do not, implying that there are other determining factors of final visual outcome other than the presence of a stromal interface. ,
In 2006, Holland reported in London at the annual meeting of the European Society of Cataract and Refractive Surgeons that post-DSAEK visual results with thinner grafts were better than standard DSAEK grafts, for the first time correlating postoperative vision to the morphologic characteristics of the DSAEK tissue transplanted. Since then, conflicting evidence has correlated the central thickness of DSAEK grafts with the postoperative visual performance. , However, all these studies were retrospective in nature; and, in addition; no morphologic parameters other than central graft thickness (CGT) were considered. Since then, the importance of the stromal component of a DSAEK graft has started to emerge as the key element influencing visual performance. In fact, rather than the CGT itself, aberrations deriving from irregularities in the graft shape may play a more important role in determining the final visual outcome. These irregularities in shape are more frequently encountered in thicker grafts ( Fig. 131.1A–C ). Anterior segment optical coherence tomography (AS-OCT) examination of the stromal component of DSAEK grafts is currently performed by several eye banks and represents an additional and essential step in the quality control of these grafts.
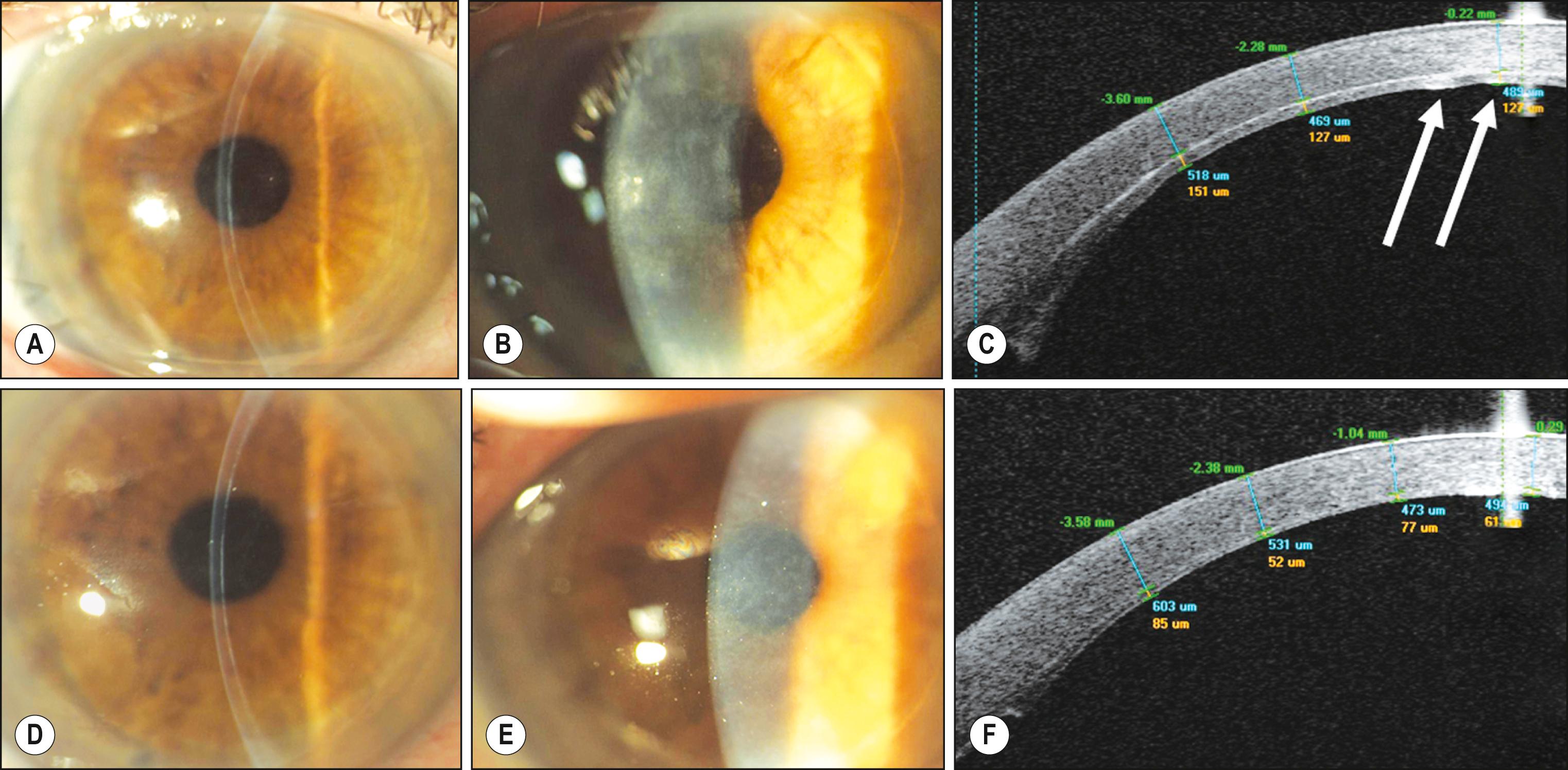
As a result, rather than eliminating the stromal interface, as is done by DMEK, a different approach was developed with the aim of optimizing the stromal component of DSAEK grafts, thus combining the technical ease of this procedure with the visual outcomes of DMEK. Busin introduced in 2009 the concept of “ultrathin” (UT)-DSAEK. This procedure is similar to DSAEK, but the graft is created with the aim of obtaining a final donor lamella of planar shape and thickness less than 131 μm (see Fig. 131.1D–F ). Since then, double and single-pass techniques have been used by surgeons and eye banks to reproducibly obtain DSAEK grafts of a predetermined thickness and planar profile, which have substantially improved the outcomes of DSAEK.
UT-DSAEK shares the same indications of conventional DSAEK. It can be performed in patients with any type of endothelial decompensation, including Fuchs endothelial dystrophy, pseudophakic or aphakic bullous keratopathy, irido-corneal-endothelial syndrome, failed penetrating keratoplasty (PK) or EK, buphthalmus, and viral endotheliitis. Like DSAEK, UT-DSAEK can be performed in phakic eyes as well as in the presence of aniridia, aphakia, extensive iris trauma, anterior chamber intraocular lenses (AC-IOL), and previous glaucoma surgery (i.e., trabeculectomy and/or aqueous shunt procedures). However, if subsequent surgery of the host cornea is anticipated, either because of its potential failure to clear (requiring sequential deep anterior lamellar keratoplasty [DALK] to remove anterior scarred recipient stroma) or because of high-degree refractive errors (requiring relaxing incisions for high astigmatism in failed PK), a thicker DSAEK graft may be preferable to guard against inadvertent penetration into the anterior chamber while performing the secondary procedure.
In addition, UT-DSAEK has been reported to be instrumental in the management of primary DMEK graft failure, allowing visual rehabilitation comparable with that of repeat DMEK while minimizing the possibility of repeat failure or other types of complications.
Endothelial dysfunction is assessed by endothelial cell (EC) density measurement in the central and peripheral cornea using specular or confocal microscopy. Corneal thickness is measured by ultrasound pachymetry or AS-OCT as an additional indicator of endothelial function.
In phakic patients, several factors influence the decision whether to perform UT-DSAEK alone or combine the procedure with phacoemulsification and IOL implantation. In patients older than 50 years with evident corneal decompensation, it is preferable to perform a combined procedure, as the retained crystalline lens will invariably develop cataract as a consequence of both surgical trauma and postoperative steroidal treatment. However, in younger patients with similar corneal conditions, the crystalline lens may be left in place since there is a fairly good chances of it remaining clear.
For patients with advanced cataract and guttae, but an otherwise clear cornea, the peripheral endothelial density is the best indicator to predict possible decompensation after cataract surgery. A density of at least 1800 cells/mm 2 in the peripheral cornea, especially in those eyes with only central guttae, usually allows phacoemulsification to be performed safely, although the central guttae themselves may limit the postoperative visual recovery to some extent. On the contrary, when the peripheral pool of ECs is low, phacoemulsification with IOL implantation should be performed in combination with UT-DSAEK.
If a flexible open loop AC-IOL or an iris enclavated AC-IOL is well positioned and of appropriate size, it may be left in place. , However, if the AC-IOL is inappropriately positioned, it may be preferable to exchange it for a posterior chamber IOL (PC-IOL) fixated into the ciliary sulcus by means of transscleral sutures.
Finally, phakic IOLs causing progressive EC loss leading to corneal decompensation may be removed at the time of combined UT-DSAEK and phacoemulsification and PC-IOL implantation, minimizing the time required for visual rehabilitation.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here