Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Because of its anatomical and functional complexity, the liver is involved in as many primary hepatic disorders as extrahepatic or systemic problems. It is always important to think ‘out of the organ’ when dealing with tumours and tumour-like lesions of the liver ( Table 13.1 ). Two major challenges exist. First, the chronically injured liver often becomes cirrhotic, giving rise to hepatocellular nodular lesions of variable biological status, all of which need to be distinguished from hepatocellular carcinoma (HCC). Secondly, the liver is a common site for metastases from all parts of the body, and these can mimic the two most important primary liver cancers, HCC and intrahepatic cholangiocarcinoma (iCCA) with their respective variants, and vice versa. Thus, correlation with clinical and radiological findings is mandatory before rendering a definitive diagnosis.
| Epithelial tumours: hepatocellular Benign Hepatocellular adenoma (HCA) Focal nodular hyperplasia (FNH) Malignancy-associated and premalignant Large-cell change (previously known as dysplasia) Small-cell change (previously known as dysplasia) Malignant Hepatocellular carcinoma (HCC) Hepatocellular carcinoma, fibrolamellar variant Hepatoblastoma, epithelial variants Undifferentiated carcinoma Epithelial tumours: biliary Benign Bile duct adenoma Microcystic adenoma Biliary adenofibroma Premalignant lesions Biliary intraepithelial neoplasia, grade 3 (BilIN3) Intraductal papillary neoplasm Low to intermediate grade High grade Mucinous cystic neoplasm Low to intermediate grade High grade Malignant Intrahepatic cholangiocarcinoma Intraductal papillary neoplasm with associated invasive adenocarcinoma Mucinous cystic neoplasm with associated invasive adenocarcinoma |
Malignancies of mixed or uncertain origin Calcifying nested epithelial stromal tumour Carcinosarcoma Combined hepatocellular–cholangiocarcinoma Hepatoblastoma, mixed epithelial–mesenchymal Malignant rhabdoid tumour Mesenchymal tumours Benign Angiomylipoma Cavernous haemangioma Infantile haemangioma Inflammatory pseudotumour Lymphangioma Lymphangiomatosis Mesenchymal hamartoma Solitary fibrous tumour Malignant Angiosarcoma Embryonal sarcoma (undifferentiated sarcoma) Epithelioid haemangioendothelioma Kaposi sarcoma Leiomyosarcoma Rhabdomyosarcoma Synovial sarcoma Germ cell tumours Teratoma Yolk sac tumour Lymphomas Secondary tumours |
Patients with liver tumours fall into the following clinical settings:
Routine health screening uncovering abnormal liver function tests, elevated serum tumour markers or imaging abnormalities
Radiological surveillance for recurrence or metastatic disease in known cancer patients
Ultrasound screening of high-risk patients with known pre-existing liver disorders
Investigation for symptomatology referable to the liver or the primary disease
Investigation for abdominal mass or symptomatology in paediatric patients
Tissue samples come from fine-needle aspiration (FNA) biopsy, core biopsy, wedge biopsy or surgical resection, including explant (see Chapter 2 ). The necessity for preoperative tissue confirmation for classic HCC in the setting of cirrhosis is still debatable. However, one can increasingly foresee the need for small tissue sample procurement with the advent of precision or targeted molecular therapy and extended HCC recipient transplantation criteria. Also, with increasing recognition of primary hepatic carcinomas, differentiation may be difficult to classify as either hepatocellular or cholangiolar, or there is dual differentiation within the same tumour, a feature that many now refer to as ‘biphenotypic’ differentiation.
The following algorithmic approach to the cytohistological diagnosis of tumours or tumour-like lesions serves as our recommended manner of segregating certain diagnostic possibilities when confronted with a tissue sample of a ‘mass’ lesion in the liver. , This diagnostic algorithm is based on the recognition of basic patterns and cell profiles. The morphological categories are as follows:
Hepatocellular and hepatoid/epithelioid patterns, including polygonal, oncocytic, large-cell and transitional cell types
Glandular (ducts, glands and/or mucin) pattern, including biliary and papillary patterns
Mixed epithelioid-glandular (including hepatobiliary) pattern
Other predominant cell patterns: squamous, small/intermediate, clear, fat-containing, pleomorphic/giant, spindle, haematopoietic/inflammatory cell and other rare lesions
Cystic pattern
This category encompasses tumour cells that are primarily polygonal and epithelioid in appearance ( Table 13.2 ). Epithelioid cells are generally loosely cohesive cells arranged in sheets and syncytial clusters in cytology samples, and in sheets, nests, trabeculae and/or pseudoglands in histologic samples. In spite of the variability in shape and size, there is a general monomorphous appearance. Tumours in this category may also be composed of large cells, cells with oncocytic cytoplasm or cells deemed ‘transitional’ between hepatocellular and other hepatoid or epithelioid tumours (e.g. mixed hepatobiliary carcinoma, metastatic urothelial or neuroendocrine tumour [NET]).
| Patterns and cell profiles | Diagnostic considerations | ||
|---|---|---|---|
| Hepatobiliary lesions | Nonhepatobiliary lesions, primary or metastatic | ||
| Hepatocellular and epithelioid patterns Includes cell profiles: Polygonal Transitional Oncocytic Large cells |
Benign | Hepatocellular Non-neoplastic (±fatty change): Dysplastic nodule Focal fatty change Focal nodular hyperplasia Large regenerative nodule Nodular regenerative hyperplasia Neoplastic (±fatty change): Hepatocellular adenoma |
Angiomyolipoma Granular cell tumour Paraganglioma Phaeochromocytoma |
| Malignant | Hepatocellular Mature hepatocytes : Hepatoblastoma Hepatocellular carcinoma (HCC) and variants Fibrolamellar carcinoma Combined HCC-cholangiocarcinoma (CCA) Biliary Epithelioid appearance : Intrahepatic cholangiocarcinoma (iCCA) and variants |
Adenocarcinoma, poorly differentiated Adrenocortical carcinoma Angiomyolipoma, benign and malignant Angiosarcoma, epithelioid variant Epithelioid haemangioendothelioma Gastrointestinal stromal tumour (GIST), epithelioid variant Hepatoid carcinoma, extrahepatic Large-cell undifferentiated carcinoma (non-small-cell carcinoma) Leiomyosarcoma, epithelioid variant Lymphoepithelioma-like carcinoma Melanoma Neuroendocrine tumour/carcinoma Renal cell carcinoma Squamous cell carcinoma, poorly differentiated Undifferentiated carcinoma Urothelial carcinoma |
|
| Glandular (ducts, glands and/or mucin) pattern Includes biliary and papillary patterns |
Benign | Bile duct hamartoma Bile duct adenoma Biliary adenofibroma Serous cystadenoma (microcystic adenoma) Mucinous cystic neoplasm (MCN) Intraductal papillary neoplasm of bile ducts (IPNB) |
Mesenchymal hamartoma Ductular reaction, e.g. in scar of focal nodular hyperplasia or in parenchymal extinction Reactive biliary atypia, e.g. in inflammatory pseudotumour Gastrointestinal contamination during EUS-FNA |
| Malignant | iCCA and variants Combined HCC-CCA MCN with associated invasive carcinoma IPNB with associated invasive carcinoma |
Adenocarcinoma, metastatic AFP-producing adenocarcinoma Epithelioid haemangioendothelioma Papillary carcinoma, metastatic Prostatic adenocarcinoma Acinar carcinomas from pancreas and salivary glands Neuroendocrine tumour/carcinoma |
|
| Mixed epithelioid-glandular pattern | BenignMalignant | Mesenchymal hamartomaHCC (if hepatocellular component predominates) Adenocarcinoma (if glandular component predominates) Poorly differentiated carcinoma (if transitional component predominates |
|
| Combined HCC-CCA Collision tumours (HCC and iCCA) |
|||
Hepatocellular nodular lesions are characterized by benign or malignant hepatocytes forming cohesive trabeculae of variable cell thickness lined by sinusoidal endothelium. Well-differentiated hepatocellular nodular lesions must be distinguished from normal, hyperplastic, cirrhotic and steatotic liver parenchyma. Epithelioid-looking tumours of diverse origin often mimic HCCs, and vice versa. Although most lesions are of epithelial origin, some may be epithelioid variants of mesenchymal tumours. For example, hepatic angiomyolipoma is also a tumour with epithelioid and/or hepatoid features, and NETs also can have abundant cytoplasm and present as large cells. Very rare tumours such as paraganglioma and phaeochromocytoma could also fall into this category.
A glandular pattern, emanating from either ducts or acini, is the most common morphological pattern encountered in focal liver lesions ( Table 13.2 ). This consists of combinations and permutations of tubules, papillae and acini to signet ring-like cells, with variable amounts of intracellular/intraluminal-extracellular mucin (see Cystic pattern later). The benign biliary pattern is recognized on cytology by flat honeycomb sheets of uniform biliary-type cells with ‘bland’ nuclear features. In contrast, a disordered and irregular (so-called drunken) honeycomb appearance with increasing cellular pleomorphism indicates malignant transformation. Glandular lesions within or outside the liver may have similar histological and immunohistochemical (IHC) profiles, and thus a diagnosis of primary versus secondary/metastatic carcinoma can be problematic. For example, metastatic pancreaticobiliary and gastric carcinomas have similar keratin profiles to cholangiocarcinomas, and therefore these lesions can be indistinguishable from each other. In addition, extensive ductular reaction, if not perceived in the appropriate context, can be mistaken for adenocarcinoma or other biliary tumours.
This pattern primarily includes the combined or mixed hepatobiliary carcinoma and its various subtypes ( Table 13.2 ). The most recognizable is combined hepatocellular-cholangiocarcinoma (cHCC-CCA) with an intimate admixture of HCC, adenocarcinoma and/or transitional components. Nonrepresentative sampling can be expected in small tissue samples.
Most of the remaining tumours and tumour-like lesions fit into this approach if classified under cellular appearances, some of which may also indicate histogenesis ( Table 13.3 ). However, some of these categories can also occur in HCC or iCCA.
| Predominant cell patterns | Diagnostic considerations | ||
|---|---|---|---|
| Hepatobiliary lesions | Nonhepatobiliary lesions, primary or metastatic | ||
| Squamous cell | Malignant | Intrahepatic cholangiocarcinoma (iCCA) and variants | Squamous cell carcinoma (SCC), metastatic (lung, cervix, pancreaticobiliary tract) Part of adenosquamous carcinoma |
| Small/intermediate cell | Benign | Inflammatory pseudotumour Abscesses Nodular haematopoiesis |
|
| Malignant | Hepatocellular carcinoma (HCC) Hepatoblastoma, small-cell undifferentiated (SCUD) subtype iCCA with small-cell features |
Lobular carcinoma of breast Lymphoma Melanoma Merkel cell carcinoma Neuroendocrine carcinoma, high-grade small-cell type Neuroendocrine tumours Nonkeratinizing carcinoma of nasopharynx Sarcomas of small-cell types Blastomas (paediatric group) Hepatobiliary rhabdomyosarcoma Malignant extrarenal rhabdoid tumour Undifferentiated (embryonal) sarcoma of liver |
|
| Clear cell | Benign | Angiomyolipoma ParagangliomaRenal cell carcinoma Adrenocortical carcinoma Clear cell sarcomas Adenocarcinoma Liposarcoma |
|
| Malignant | HCC with clear cell features iCCA with clear cell features |
||
| Fat-containing cell | Benign | Focal fatty change Hepatocellular nodular lesions with fatty change |
Angiomyolipoma Fat ectopia Lipoma Myelolipoma |
| Malignant | HCC with fatty change | Adrenocortical carcinoma Liposarcoma Renal cell carcinoma Vacuolated (or signet ring) cells (e.g. signet ring cell adenocarcinoma, epithelioid haemangioendothelioma) |
|
| Pleomorphic and giant cell | Benign | Angiomyolipoma Granulomas, including foreign body giant cell reaction Inflammatory pseudotumour, reparative type Nodular haematopoiesis |
|
| Malignant | HCC with pleomorphic or giant tumour cells Fibrolamellar carcinoma iCCA |
Poorly differentiated SCC Poorly differentiated adenocarcinoma Metastases from giant cell carcinomas (e.g. lung, pancreas, renal cell) Pleomorphic and/or giant cell sarcomas Urothelial carcinoma Undifferentiated (embryonal) sarcoma of liver |
|
| Spindle cell | Benign | Angiomyolipoma Cavernous haemangioma Mesenchymal hamartoma (paediatric age-group) Rare mesenchymal or neural primary liver lesions: (e.g. fibroma, neurofibroma) Inflammatory pseudotumour Solitary fibrous tumour Stroma from scar of focal nodular hyperplasia Stroma from desmoplastic tumours Tunica media from arterial walls |
|
| Malignant | Hepatoblastoma, mixed epithelial and mesenchymal (MEM)-type HCC, sarcomatoid iCCA, sarcomatoid |
Angiosarcoma Gastrointestinal stromal tumour (GIST) Kaposi sarcoma Other sarcomas (e.g. fibrosarcoma, leiomyosarcoma, liposarcoma) Sarcomatoid renal cell carcinoma Follicular dendritic cell tumour Undifferentiated (embryonal) sarcoma of liver Hepatobiliary rhabdomyosarcoma Paediatric stromal tumours |
|
| Haematopoietic and inflammatory cell | Benign | Benign nodular lymphoid tissue/reactive nodular lymphoid lesion Nodular haematopoiesis Myelolipoma Abscess (e.g. pyogenic, fungal, amoebic) Eosinophilic abscess (e.g. visceral larval migrans, capillariasis, other parasitic infections) Inflammatory pseudotumour (lymphoplasmacytic, xanthomatous or fibrohistiocytic types) Granulomas (e.g. mycobacterial, fungal, other infections, extensive sarcoidosis) |
|
| Malignant | HCC with prominent inflammatory component | Non-Hodgkin lymphoma Hodgkin lymphoma Langerhans cell histiocytosis Granulomas in neoplasia (e.g. keratin-induced granulomas in SCC) Follicular dendritic cell tumour |
|
| Other rare lesions | Germ cell tumours (e.g. teratoma, yolk sac tumour) | ||
Squamous cell carcinomas are typically not primary to the liver, although they may occasionally arise in the biliary tract, oftentimes as part of adenosquamous carcinoma. These squamous tumours often undergo necrosis with suppuration that mimics abscesses and obscures tumour cells. Keratin-induced granulomas can also be seen.
Both HCC and iCCA may have small-cell features. Apart from the usual list of small round cell tumours, the pathologist must be aware of inflammatory/reactive haematological processes that also have small-cell features, although haematological malignancies tend to be diffuse when they involve the liver (see haematopoietic pattern later). Small round cell tumours are relatively common in the paediatric age group.
The clear cell pattern is typically caused by glycogen deposits or fatty change of various types that can result in clear to bubbly cytoplasm. The classic prototypes of clear cell carcinomas are renal cell and adrenocortical carcinomas, but both HCC and iCCA may also have clear cell features.
Hepatocellular nodular lesions themselves can undergo fatty change (steatotic/steatohepatitic) and should be distinguished from steatosis/steatohepatitis of the background liver. Nevertheless, it is a useful indicator of a potential lesion when a core biopsy from a well-differentiated hepatocellular nodular lesion demonstrates fatty hepatocytes in a nodular/focal pattern next to a sea of hepatocytes lacking fat; in this case, one needs to search for other architectural and cytological features to decide which side is lesional. It is also important to consider lipomatous lesions or other tumours that may contain fat cells (e.g. angiomyolipoma) and neoplastic/degenerative conditions that may contain cells mimicking intracytoplasmic ‘fat’ vacuoles.
These tumours are typically poorly differentiated malignancies with high-grade pleomorphic and giant cell features (but not spindle cells). The giant cells are much larger and with multinucleation; tumour giant cells may exhibit extraordinary malignant atypia and mitotic activity, and these types are common in the more poorly differentiated HCCs (PDHCC). Histiocytic giant cells, as well as neoplastic giant cells of carcinomas and sarcomas (including undifferentiated/embryonal sarcoma of the liver), also could fall into this category.
The spindle cell pattern consists of elongated or fusiform cells, often arranged in interlacing fascicles. Differential diagnoses range from reactive fibroblastic stroma to mesenchymal tumours. Sarcomatoid carcinomas may also have this pattern and, when primary in the liver, may be of hepatocellular or biliary origin. Rarely, hepatic angiosarcoma can present as a diffuse, non-mass-forming variant with a spindle-epithelioid vasoformative growth pattern permeating the sinusoids, which can confound the pathologist.
These tumours range from inflammatory/reactive to proliferative lesions. Intrahepatic and perihilar inflammatory masses may form as a result of distal obstructions, and such obstructions in turn may result from a malignant stricture or intraductal papillary neoplasm. Additionally, marked inflammatory reactions (both neutrophilic and lymphocytic) can be rarely seen in HCC or in tumours with extensive necrosis. A rare and unusual occurrence is granulocyte colony-stimulating factor-producing HCC which can mimic an infectious/inflammatory process, clinically and histologically.
Germ cell tumours, such as teratoma and yolk sac tumour, have characteristic features.
The pathologist should first and foremost ascertain whether the lesion is a true cyst with a specialized lining or a pseudocyst ( Table 13.4 ). It should be noted that some tumours may cavitate because of central infarction or locoregional ablational therapy. The nature of the cyst contents should be noted and triaged if clinically warranted. The demonstration of mucin is pathognomonic of a glandular neoplasm, but suppuration with or without superimposed infection can also occur even in an otherwise noninflammatory cyst.
| Cystic pattern | Diagnostic considerations | |
|---|---|---|
| With epithelial lining | Without epithelial lining | |
| Benign | Solitary bile duct cyst Fibropolycystic disease Ciliated foregut cyst Obstructive dilation of bile duct Mucinous cystic neoplasm (MCN) Intraductal papillary neoplasm of bile ducts (IPNB) |
Abscess Cavernous haemangioma, lymphangioma Cystic degeneration of benign tumours Haemorrhagic cyst Hydatid cyst Inflammatory pseudotumour with cavitation |
| Malignant | MCN with associated invasive carcinoma IPNB with associated invasive carcinoma Cystic intrahepatic cholangiocarcinoma (iCCA) Carcinomas associated with developmental cysts Cystic metastases (e.g. ovary, pancreas) |
Cystic degeneration of malignant tumours (e.g. undifferentiated embryonal sarcoma of liver) |
Hepatocellular adenoma (HCA) is a benign neoplasm that typically arises in noncirrhotic liver; reports of HCAs in cirrhotic liver almost always represent macroregenerative nodules or extremely well-differentiated HCCs (WDHCCs), except in rare cases when a specific HCA subtype is confirmed based on IHC and/or mutational analysis. There are at least four well-recognized subtypes of HCA: hepatocyte nuclear factor 1α-inactivated (HHCA), inflammatory (IHCA), β-catenin-activated (BHCA) and unclassified, each with different aetiological, histological and molecular characteristics ( Table 13.5 ).
| Lesion | Typical histological features | Key clinical associations and comments |
|---|---|---|
| HHCA | Fatty change, small- and large-droplet type No portal tracts Loss of LFABP by immunostain Packeting of hepatocytes on reticulin stain |
MODY3, obesity Almost always in women Adenomatosis |
| IHCA | Sinusoidal dilation No portal tracts Fibrovascular cores with inflammatory cells, arterial clusters and ductular reaction SAA and CRP immunostaining, diffuse |
Obesity Metabolic syndrome Women > men Adenomatosis |
| BHCA | Cytological atypia No portal tracts Architectural atypia with pseudoglands, bile plugs and focal disruption of reticulin pattern β-Catenin nuclear and diffuse GS staining |
Association with male gender and androgenic steroid use High risk of HCC |
| FNH | Central fibrovascular zone with dystrophic vessels Radiating fibrous septa into nodular periphery Cholate stasis, no normal interlobular bile ducts Ductular reaction Map-like staining with GS |
Women and men Note: Compared with FNH, FNH-like lesions arising in the setting of vascular flow abnormalities may lack GS map-like staining |
| WDHCC | Trabecular-sinusoidal pattern (cords ≥3 cells thick); may have pseudoglands Minimal cytological atypia No portal tracts Stromal invasion Loss of reticulin, multifocal or diffuse Diffuse GS/nuclear β-catenin, positive GPC3 favours HCC |
Most HCC arise in cirrhosis (80–85%) Immunostains GPC3 and AFP are often negative |
The association of HCA with oral contraceptive (OC) use in young women is well established, , but the incidence of OC-related HCAs has decreased in the last few decades with the introduction of lower oestrogen-dose OC preparations. HCA can also increase in size during pregnancy, when oestrogen levels are high. This association supports that sex hormones play a role in tumourigenesis by promoting cell proliferation but are not the initiators of hepatocellular neoplasms. A higher risk of HCA is also associated with noncontraceptive oestrogens and anabolic-androgenic steroids. ,
HCA has been reported in association with several hereditary disorders, and multiple adenomas can be seen in these patients. HCA can occur in type Ia glycogen storage diseases (GSDs) and less often in types III and IV. . Patients with these disorders may develop multiple adenomas. Maturity-onset diabetes of the young, type 3 (MODY3), a form of autosomal dominant familial diabetes mellitus with germline mutations involving TCF1 gene, can be associated with HCAs (see subtypes later) and multiple adenomas as well. Other, less common associations are familial adenomatous polyposis, McCune–Albright syndrome, Fanconi anaemia, clomiphene use and Klinefelter syndrome.
More recently, a significant increase in HCA has been related to rising levels of obesity and metabolic syndrome. In addition, some specific forms of HCA associated with obesity now appear to be increasing in incidence, especially the inflammatory and the β-catenin-activated subtype. These obesity-related lesions present in a slightly older age group in women, but also in men. Moreover, it is proposed that overweight/obesity could represent a major risk of malignant transformation of HCA, possibly via the IL-6 pathway. Hepatocellular nodules resembling HCA can also arise in the setting of chronic vascular disorders such as Budd–Chiari syndrome.
HCA typically occurs in women in the reproductive age group (15–45 years). The mean age was 30 years in older series, whereas the mean age has been 37–41 years in more recent series, , reflecting changes in aetiology and risk factors for HCA. HCA occasionally occurs outside the reproductive age group, but these are still uncommon. In the earlier series, patients usually came to medical attention with symptoms of abdominal pain, mass or haemorrhage. In more recent series, however, the majority of tumours were discovered incidentally on imaging, and about 20% of patients present with acute abdominal pain caused by haemorrhage into the tumour or into the peritoneal cavity, which may be life threatening.
HCAs demonstrate variable echogenicity on ultrasound (US) and cannot readily be distinguished from other lesions. On computed tomography (CT), HCAs are iso- or hypoattenuating and often heterogeneous with hyperattenuating areas corresponding to recent bleeding. On multiphasic CT, there is arterial enhancement and a persistent enhancement in the delayed phase. On magnetic resonance imaging (MRI), HCAs are typically hyper- or isointense relative to adjacent liver on T1-weighted images and slightly hyperintense on T2-weighted images. , The presence of fat and telangiectasia leads to distinctive appearances on contrast-enhanced US and MRI and may be able to determine HCA subtypes with an accuracy approaching 85%. ,
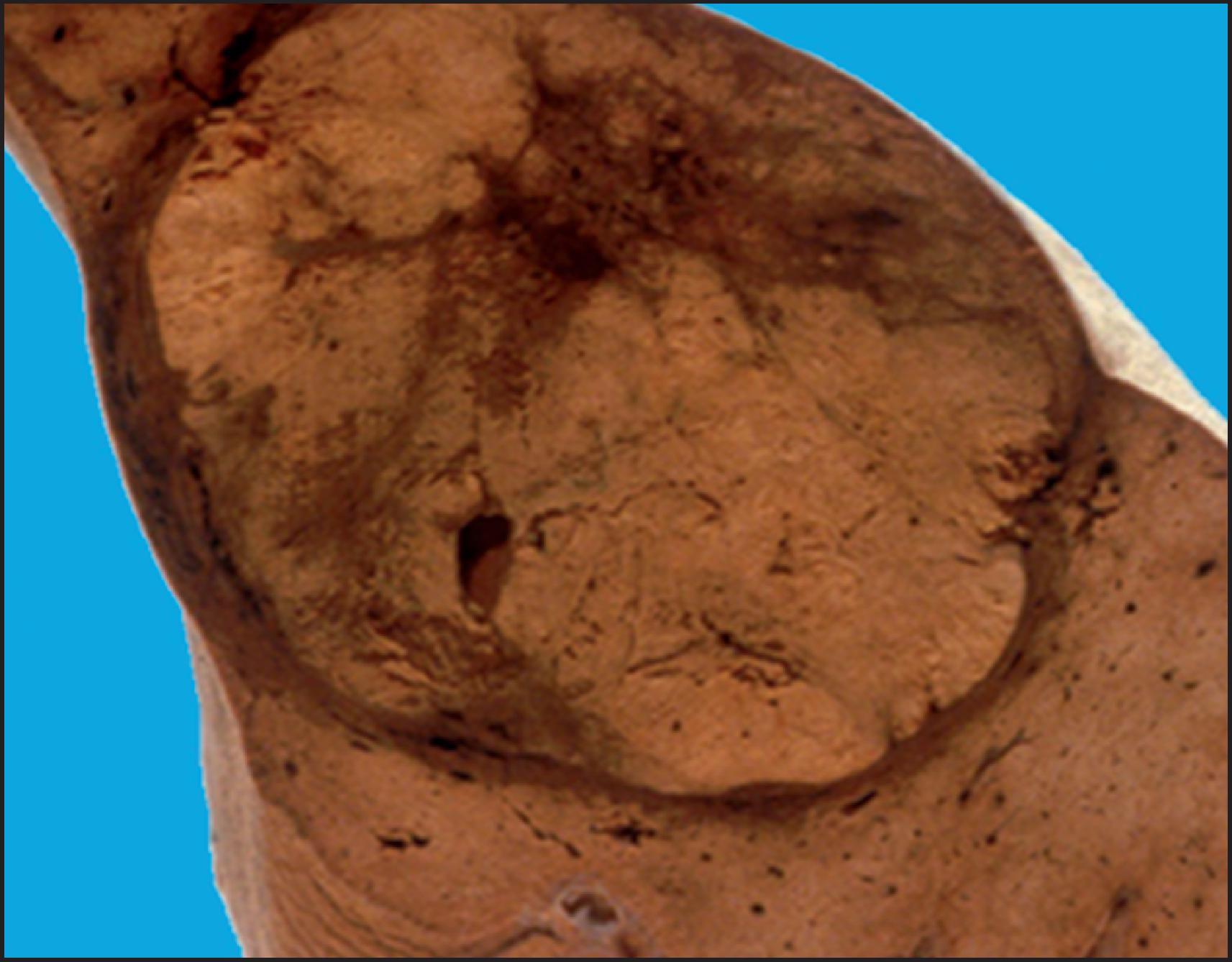
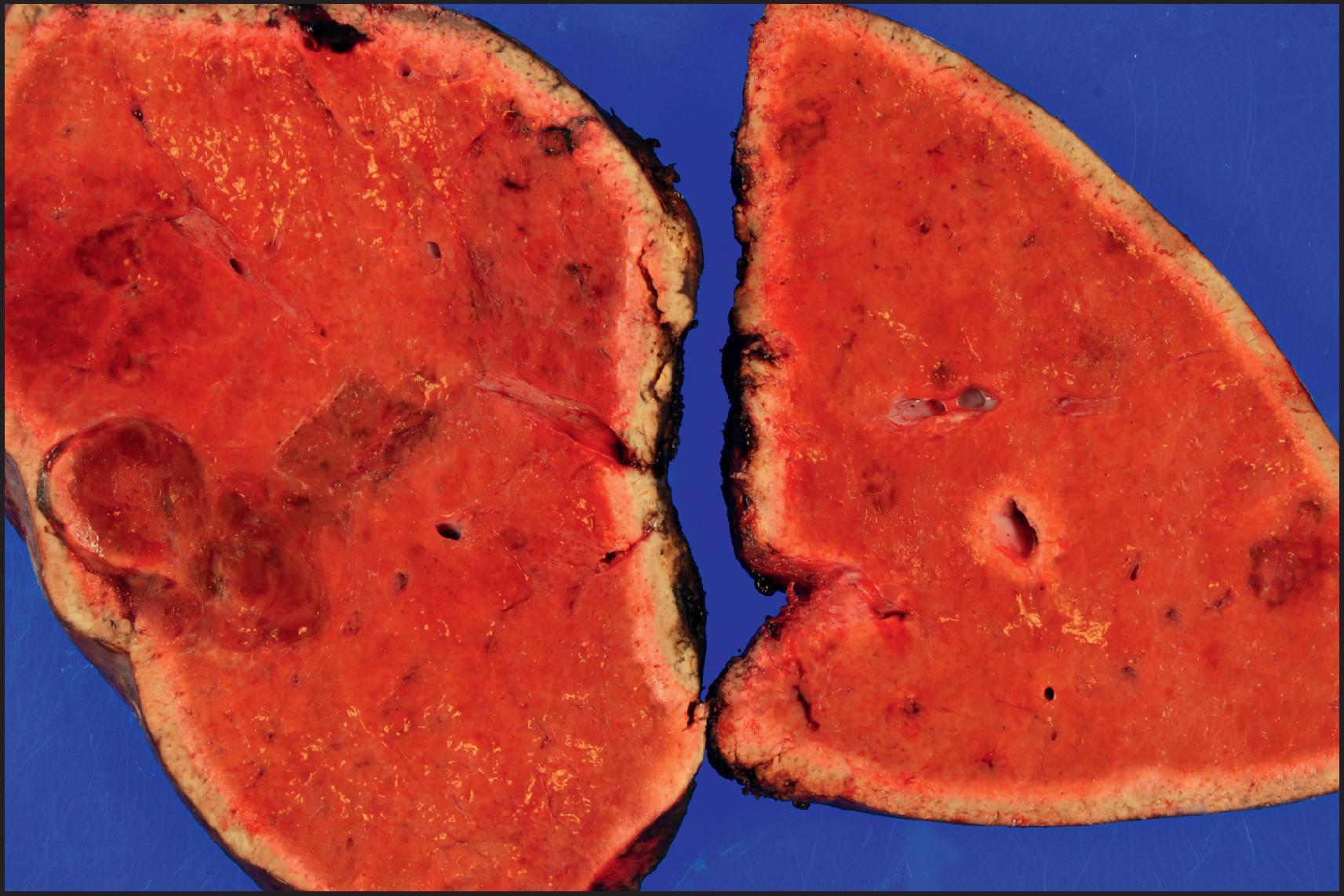
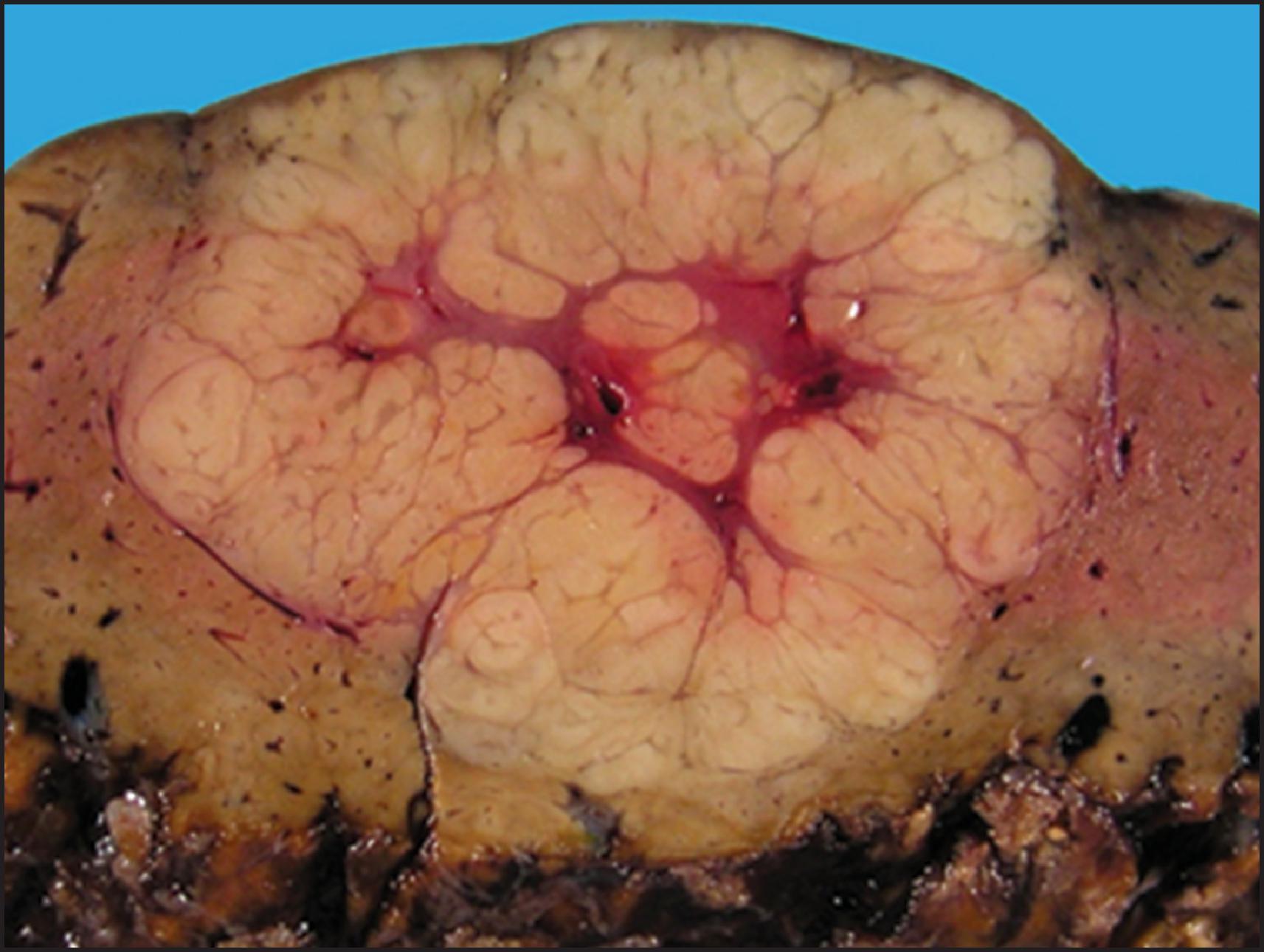
By definition, HCA is a benign neoplasm composed of hepatocytes and almost always arises in noncirrhotic liver. Multiple tumours may be present, and the term ‘hepatic adenomatosis’ has been used with ≥10 HCAs (see later). The tumours often bulge from the surface of the liver and occasionally are pedunculated. The majority of tumours are 5–15 cm but can measure up to 30 cm in diameter. The colour varies from yellow or tan to brown ( Fig. 13.1 ), and there may be green areas of bile production as well as areas of necrosis or haemorrhage. Irregular areas of fibrosis may be present. HCAs are often well circumscribed, but some cases can have ill-defined outlines and can be difficult to differentiate from the surrounding liver ( Fig. 13.2 ).
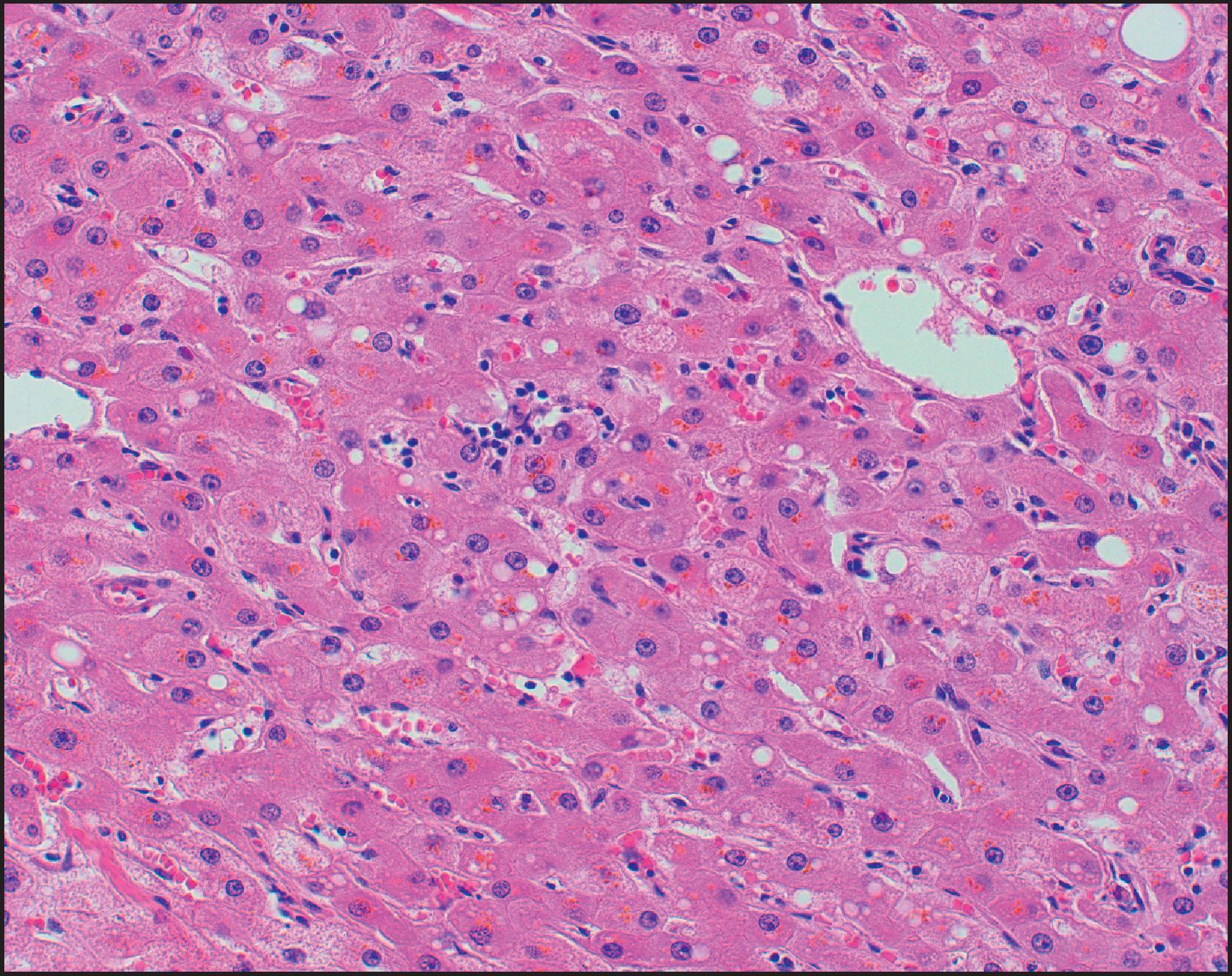
Microscopically, HCA is typically composed of hepatocytes arranged in thin cell plates (typically one or two cells thick) without cytological atypia ( Fig. 13.3 ). Tumour cells are typically uniform in size and shape, but some variation can be seen, which may result from regenerative and atrophic/ischaemic changes within the tumour. The reticulin framework is intact, outlining the cell plates in a pattern similar to that seen in normal liver without loss or fragmentation ( Fig. 13.4 ). In certain HCA subtypes, however, particularly the HHCA, there may often be near to complete encircling of small groups of tumour cells by reticulin fibres (‘packeting’). The nuclei of tumour cells are typically uniform and regular; the nuclear/cytoplasmic ratio is low; and mitoses are almost never seen (see Fig. 13.3 ). Nucleoli are seldom prominent. Occasional tumours, especially in patients with long exposure to contraceptive steroids, may have a few pleomorphic nuclei resembling large-cell change in cirrhosis.
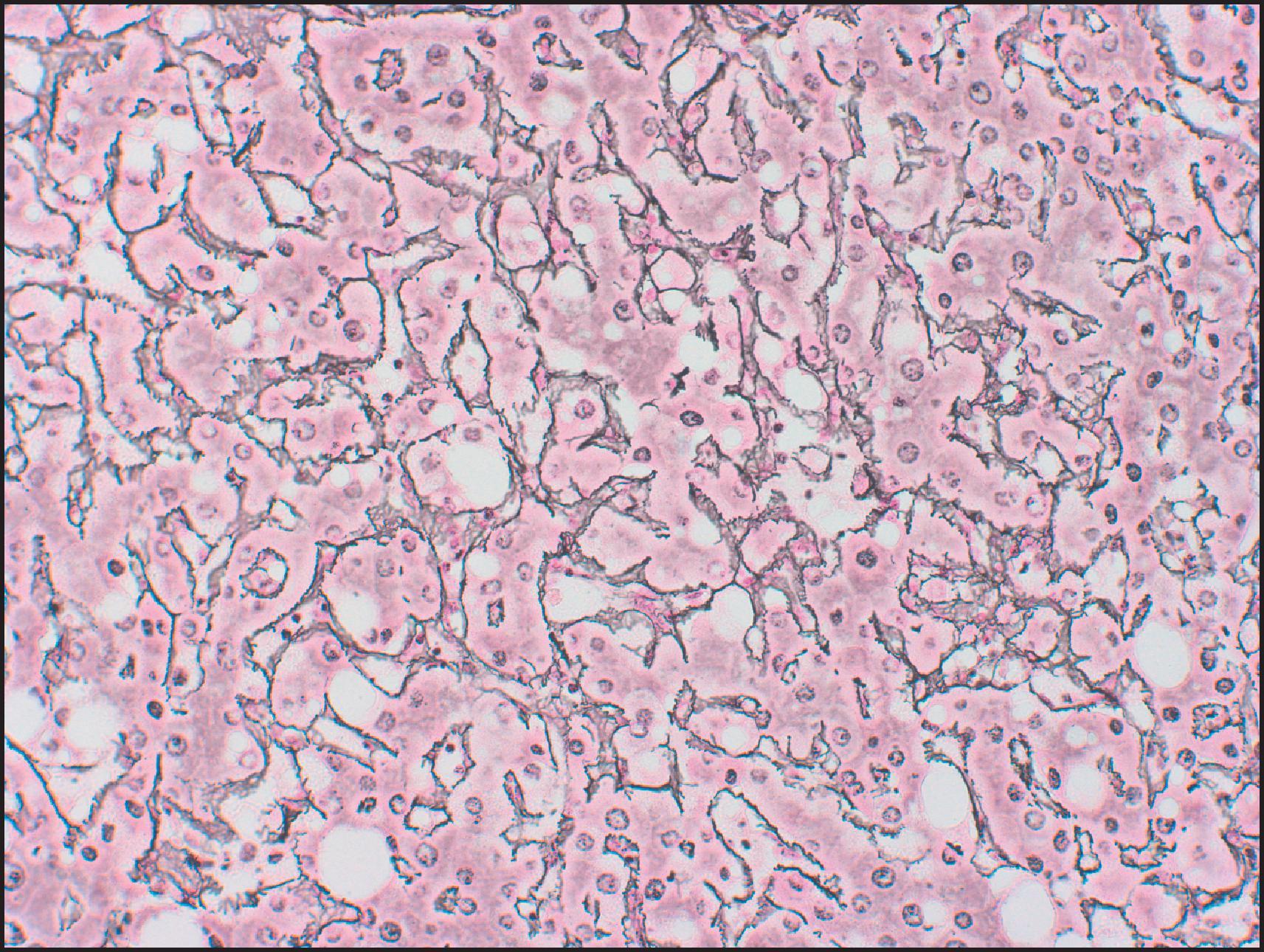
Features that are variably present include fat, glycogen production, cytoplasmic lipofuscin granules, Dubin–Johnson pigment (pigmented liver cell adenoma) and, rarely, Mallory–Denk bodies (MDBs). The fat may be abundant, simulating fatty liver, particularly in some HHCA and IHCA subtypes. The sinusoids can be compressed, leading to a sheet-like appearance. Sometimes the sinusoids are dilated and can be mistaken for peliosis hepatis. Thin-walled vascular channels and small arteries are scattered throughout the tumours, whereas large arteries are seen at the periphery. The arteries are unpaired (lacking corresponding bile ducts), because interlobular bile ducts are not found in HCA. However, bile ductules and progenitor cells may be present, particularly in the IHCA subtype. Kupffer cells are present but usually inconspicuous; stellate cells are occasionally seen. Haematopoietic elements may be noted in the sinusoids, and rare cases with noncaseating granulomas have been described. Areas of fresh haemorrhage and haemosiderin-laden macrophages from past haemorrhage may be seen, along with recent or previous infarcts. Immunohistochemically, the endothelial cells lining the plates can stain positively to variable degrees for CD34, similar to that seen in HCC. Unlike HCC, IHC for oncofetal proteins such as glypican 3 (GPC3) and alpha fetoprotein (AFP) is negative.
Cytologically, FNA aspirates are hypercellular, comprising small cohesive groups of almost normal-looking hepatocytes with transgressing endothelium in the absence of bile duct epithelium ( Fig. 13.5 ). The neoplastic hepatocytes may display fat vacuoles, pale glycogen-laden cytoplasm or pigments. They have relatively small, uniform central round nuclei with normal nuclear/cytoplasmic ratio (≤1:3 by comparison of nuclear and cell diameters). Cell blocks may not prove sufficient for IHC subtyping. An indeterminate cytodiagnosis may be proffered in the absence of appropriate clinical and radiological information. A potential pitfall is missing or discounting ghost outlines of lesional neoplastic hepatocytes in aspirates of infarcted adenomas. Clues such as vascular aberrations and reticulin abnormalities may still be discernible within the infarcted tissue if cell block material is available for further analysis.
![Figure 13.5, Hepatocellular adenoma; aspiration biopsy. Small, cohesive groups of neoplastic cells resemble normal hepatocytes, with ample granular cytoplasm, uniform central round nuclei, small nucleolus and normal nuclear/cytoplasmic ratio. (May–Grünwald Giemsa stain [MGG] stain.) Figure 13.5, Hepatocellular adenoma; aspiration biopsy. Small, cohesive groups of neoplastic cells resemble normal hepatocytes, with ample granular cytoplasm, uniform central round nuclei, small nucleolus and normal nuclear/cytoplasmic ratio. (May–Grünwald Giemsa stain [MGG] stain.)](https://storage.googleapis.com/dl.dentistrykey.com/clinical/TumoursandTumourLikeLesions/4_3s20B9780702082283000132.jpg)
Identification of distinctive genomic changes and their correlation with clinical and histological features led to the recognition of subtypes of HCA , ( Table 13.5 ), which formed the basis of the 2019 classification adopted by the World Health Organization (WHO).
Biallelic inactivation of TCF1 gene that codes for hepatocyte nuclear factor 1α (HNF1α) was observed in about 40% of sporadic HCAs in early studies of adenoma subtypes. The gene defect was found to be somatic in 90% but germline in 10%. HHCAs are overwhelmingly more common in women (>90%) and approximately one-third occur in women older than 50 years. These tumours can be associated with OC use as well as MODY3 and are often characterized by prominent intralesional steatosis ( Fig. 13.6 ). Both large and small droplets can be present, but in many cases, small-droplet fat can predominate and may be overlooked as an important diagnostic finding. Minor architectural irregularities such as small acinar structures can be seen ( Fig. 13.6 ). Inflammation and cytological atypia are typically absent, but the reticulin framework pattern often has a ‘packeted’ appearance in which reticulin fibres can partially encircle small clusters of hepatocytes ( Fig. 13.7 ). Prominent sinusoidal dilatation leading to a peliotic appearance (‘myxoid change’) can be focally present and can be extensive in rare cases. HNF1α positively regulates the FABP1 gene, which codes for liver fatty acid-binding protein (LFABP). LFABP is expressed in the cytoplasm in normal hepatocytes, as demonstrated by IHC, but is low or absent in HNF1α-inactivated adenoma (HHCA) ( Fig. 13.8 ). This is one of the subtypes that may be more frequently associated with adenomatosis, so the liver parenchyma should be examined for the presence of small fatty nodules (also LFABP negative), which represent microadenomas ( Fig. 13.9 ). The estimated risk for malignant transformation is <2%.
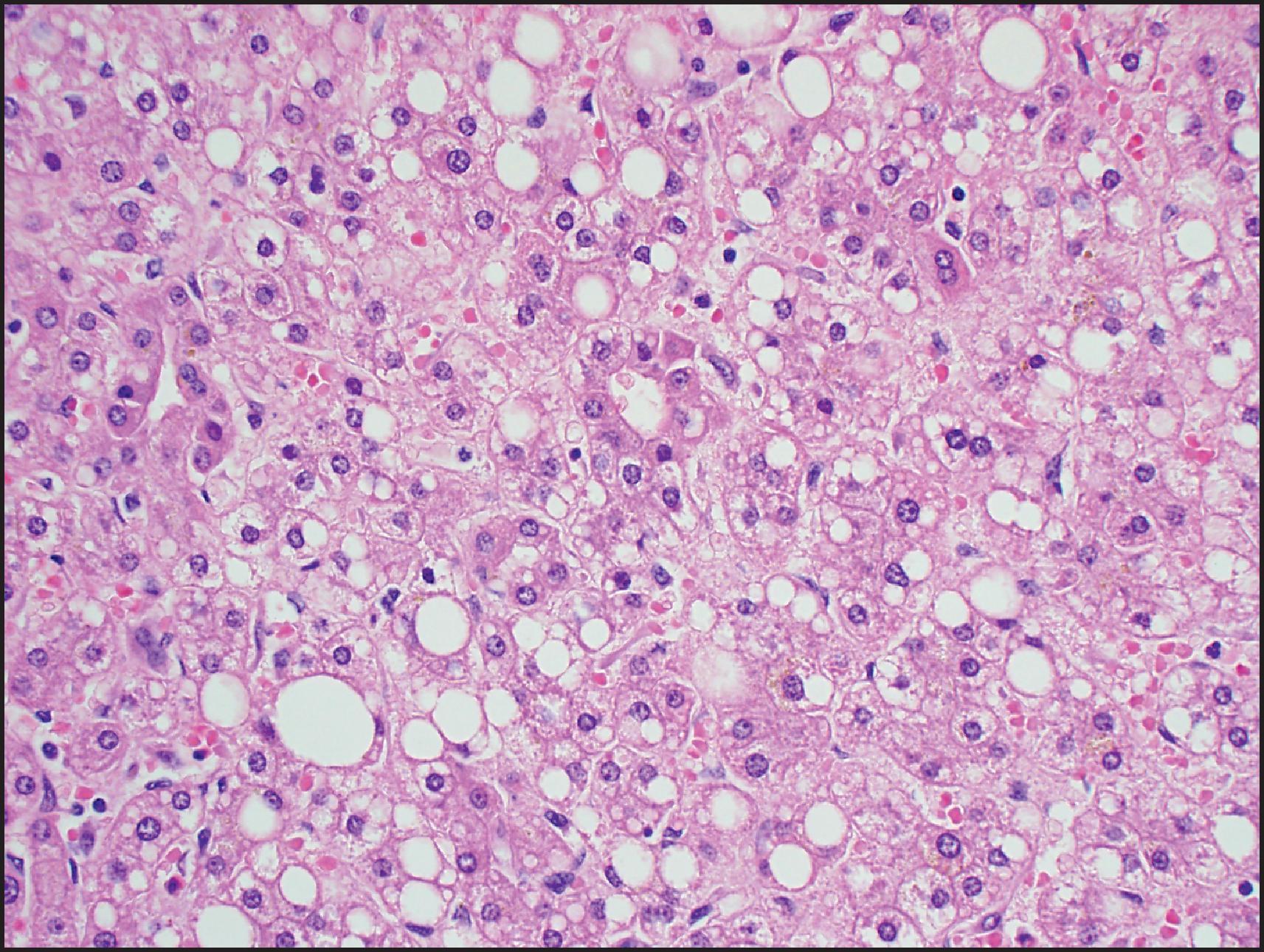
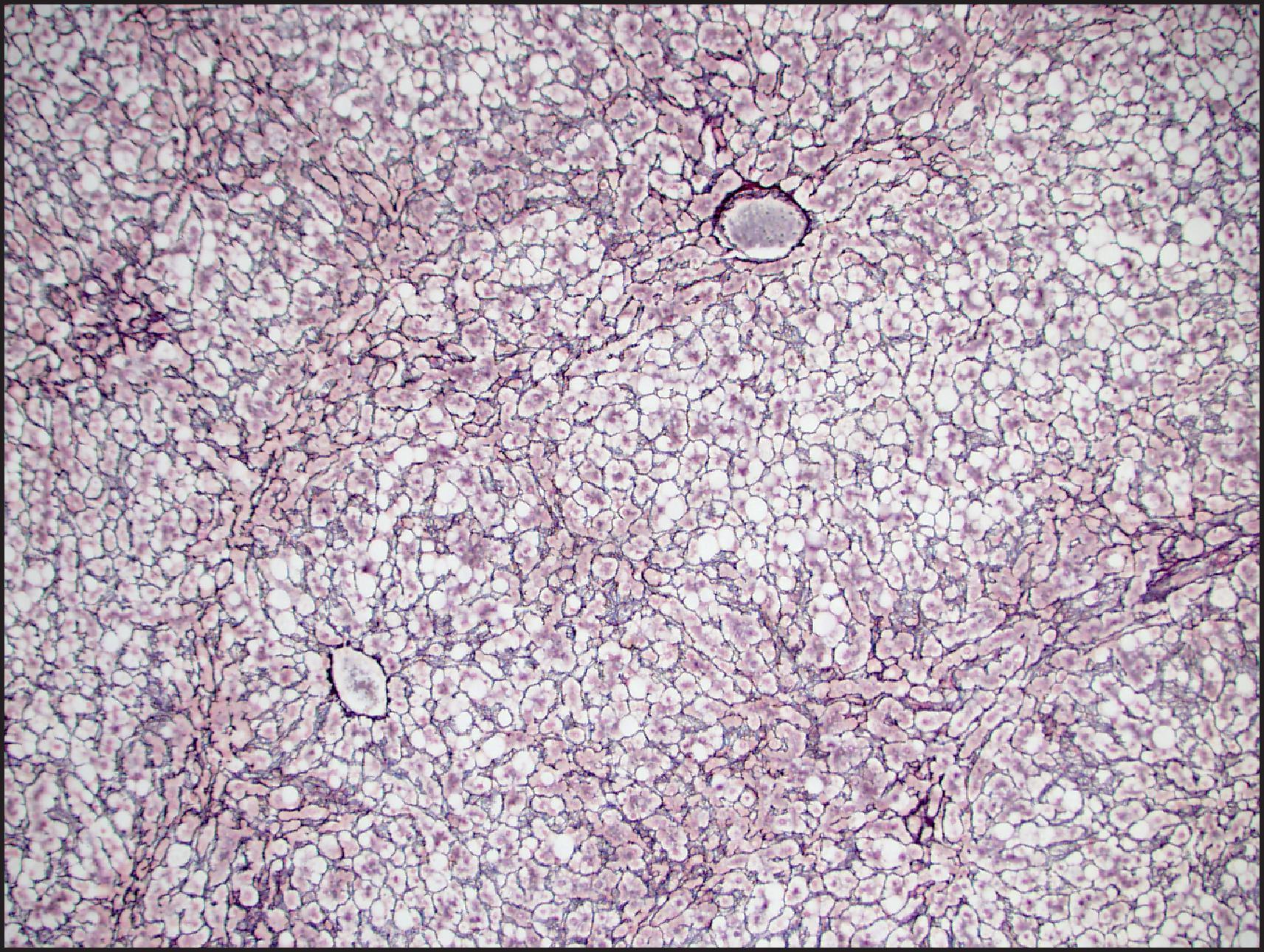
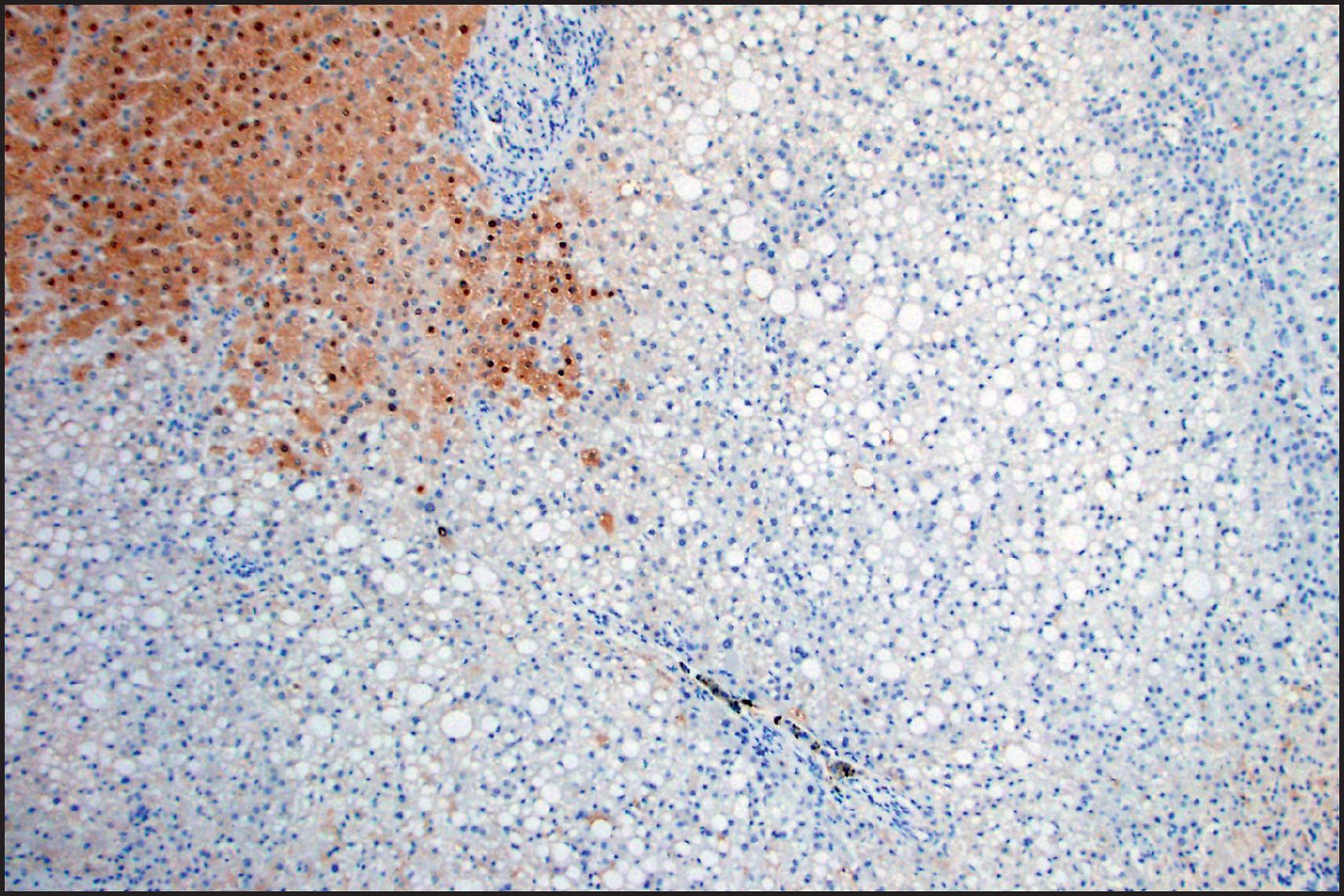

Steatosis is not specific for HHCA and can be observed in other HCA subtypes (particularly IHCA) as well as in focal nodular hyperplasia (FNH) and HCC. HHCAs lack steatosis in approximately 10% of cases. Loss of LFABP is useful to establish the diagnosis, but the IHC staining has to be carefully titrated because staining in normal hepatocytes can be weak. HCC arising in HHCA is rare; the mechanisms of malignant transformation are not known. Diffuse glutamine synthetase (GS) staining can be seen in such cases, but β-catenin mutations are typically absent, and the mechanism of diffuse GS is not known. Since LFABP loss can be observed in HCC, it should be used for histological subtyping after the diagnosis of HCA has been established based on cytological and architectural findings. Based on anecdotal cases, it appears that LFABP loss can be seen in moderately and poorly differentiated cases but may not be associated with HNF1A mutation. Absence of steatosis, prominent pseudoacini, extensive peliotic/myxoid change and marked accumulation of lipofuscin pigment are considered high-risk findings, and a diagnosis of HCC can be more reliably established when these findings are accompanied by multifocal reticulin loss. ,
The IHCA subtype accounts for 40–50% of HCAs and is more common in women, although it also can occur in men with appropriate risk factors. IHCA is often associated with obesity and metabolic syndrome; steatosis and steatohepatitis can be present in the non-neoplastic liver. These tumours are characterized by activation of the interleukin (IL)-6 signalling pathway (IL6/JAK/STAT3 pathway) resulting from somatic gain-of-function mutations in the IL6ST gene that encodes the signalling co-receptor gp130. These mutations are observed in 60% of IHCAs, leading to the activation of signal transducer and activator of transcription 3 (STAT3) in the absence of ligand. Less often, somatic mutations in other genes involved in the pathway, such as Fyn-related kinase ( FRK ), STAT3 , GNAS and Janus kinase 1 ( JAK1 ), have been implicated in IHCA, all of which promote the constitutive activation of STAT3.
The histological hallmarks of IHCA are sinusoidal dilation, ductular reaction, haemorrhage and inflammation ( Figs. 13.10 and 13.11 ). Arterioles, often in clusters and surrounded by a small amount of fibrous tissue with or without ductular reaction at the interface, can mimic portal tracts, but the arterial zones lack interlobular bile ducts (‘pseudo-portal tracts’) ( Fig. 13.11 ). Ductular reaction is seen in almost half the cases. Fibrous septa may be present, and therefore, in combination with ductular reaction, the findings can mimic FNH, although a nodular architecture typical of FNH is seen in <10% of cases. However, because of the similarities to FNH, many of these cases were previously termed ‘telangiectatic FNH’. Based on imaging characteristics, clonality and protein-clustering studies, it is now recognized that most of these lesions are IHCA. The distinction cannot be made by the presence of sinusoidal dilation alone; this feature can be present in a minority of FNH. C-reactive protein (CRP) and serum amyloid-associated (SAA) protein, both acute-phase reactants, are overexpressed in the neoplastic hepatocytes, so IHC staining for these markers helps to confirm the diagnosis ( Fig. 13.12 ).
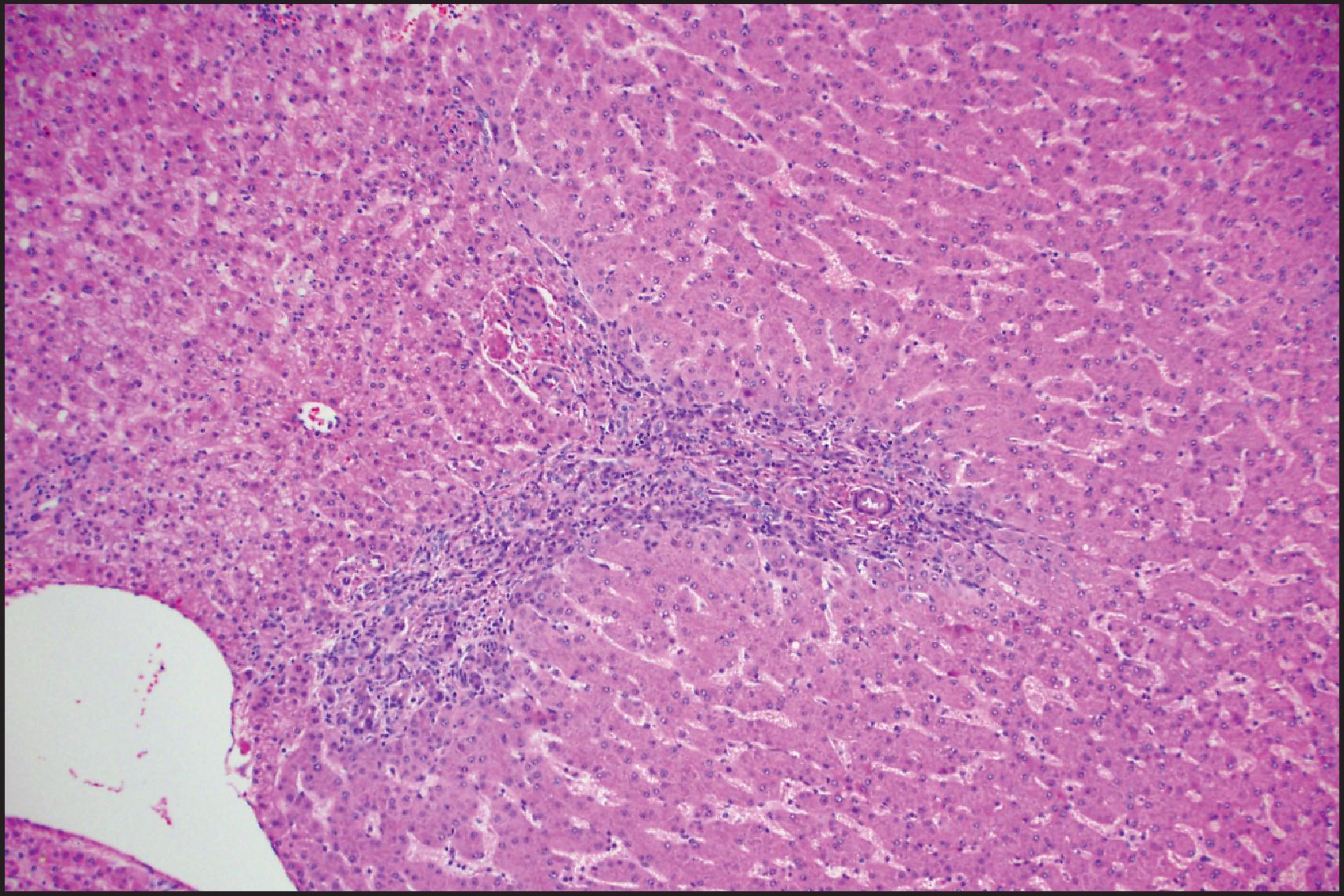
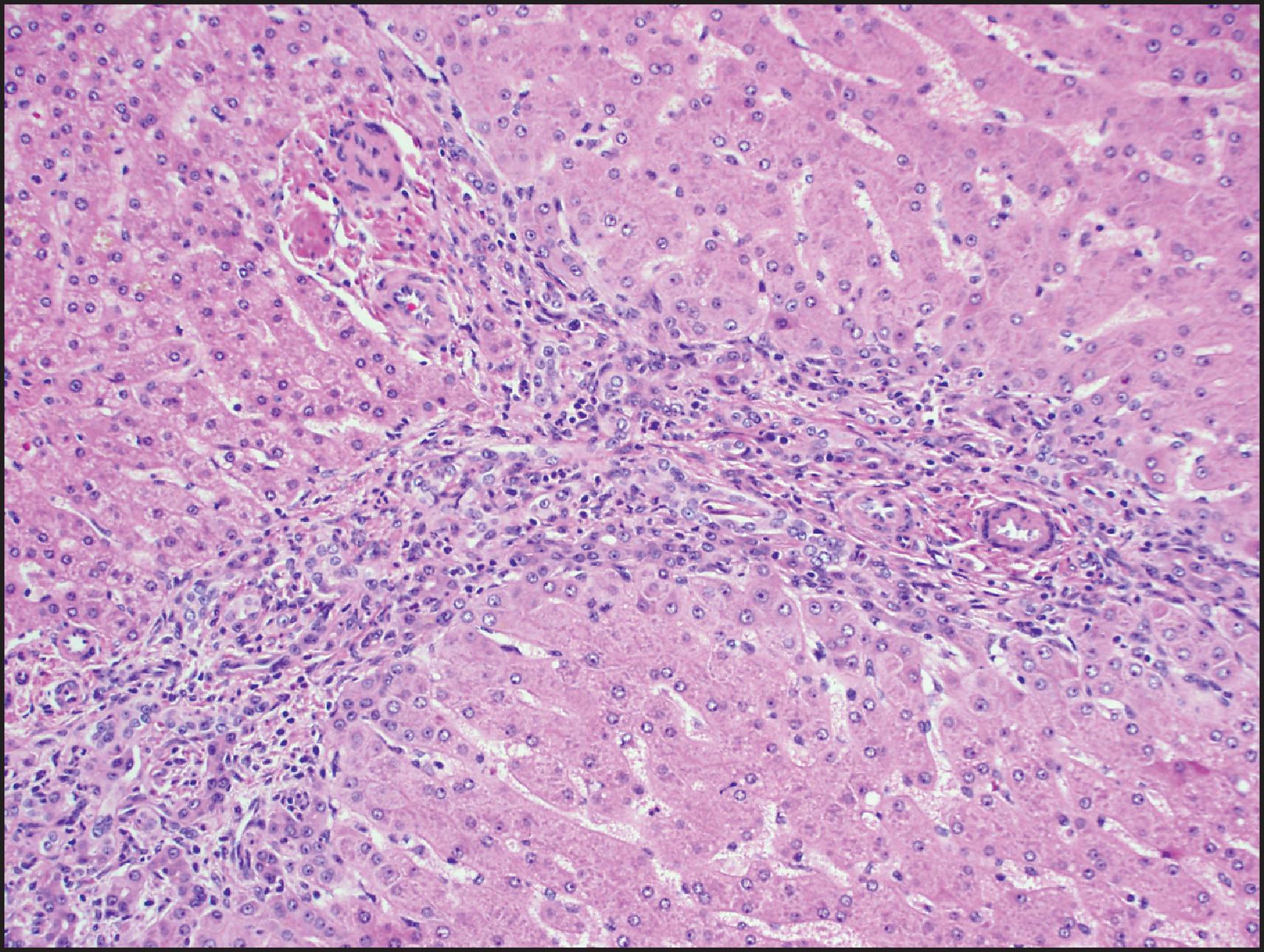
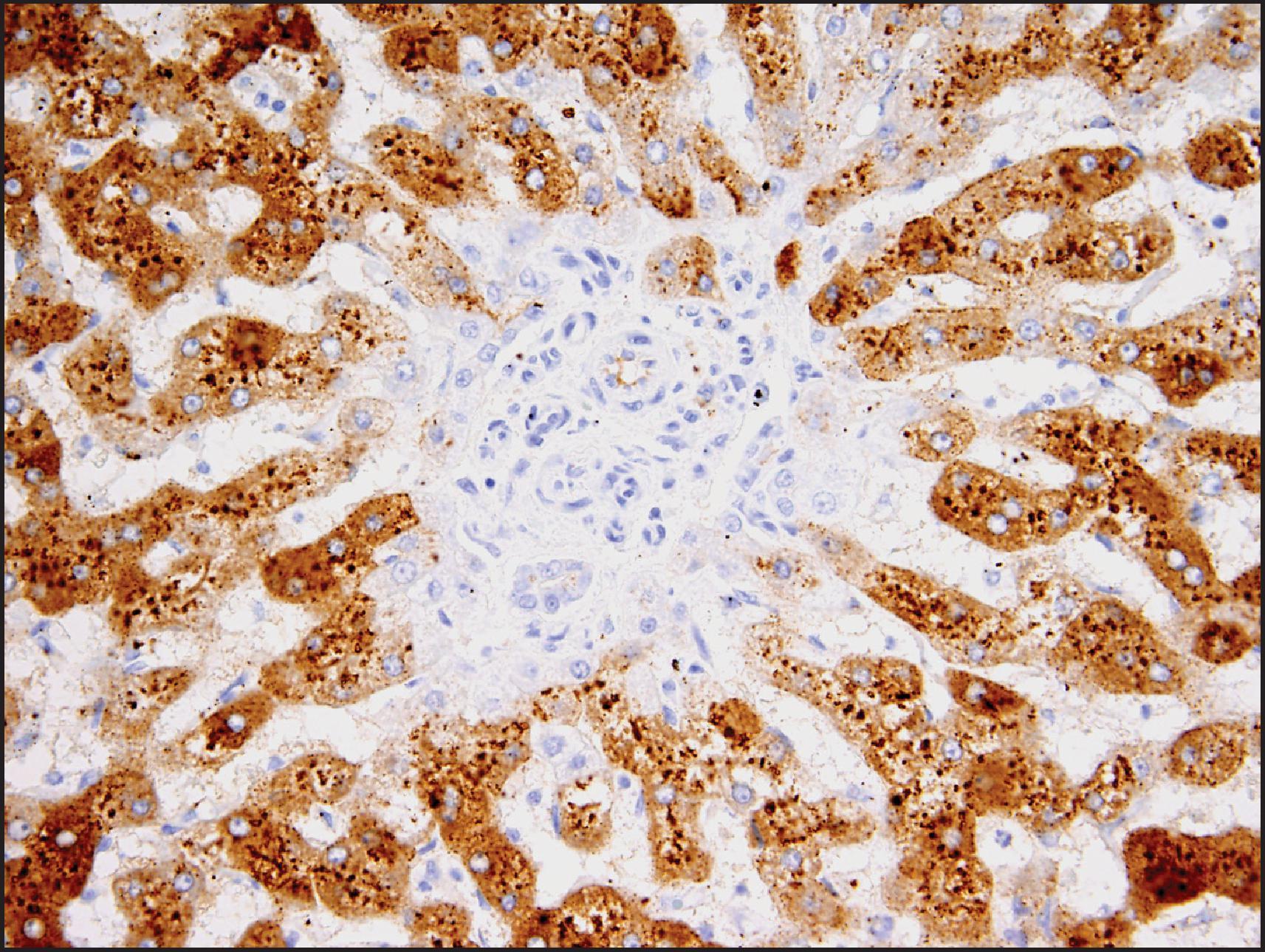
Sinusoidal dilation and/or inflammation may not be seen in up to 10% of IHCAs. , Both SAA and CRP typically stain these lesions in a diffuse manner, although SAA can be negative or minimal in 10% of cases with otherwise typical morphology, while CRP will be diffusely positive in an estimated >80% of tumour cells in essentially all these lesions. Thus, CRP has higher sensitivity, and completely negative results are rare. , Since both SAA and CRP can be positive in the adjacent non-neoplastic liver, careful correlation with morphology and CD34 staining is necessary to avoid interpreting this as a positive result in the tumour, especially in limited needle biopsies. It is important to note that activation of β-catenin is seen in approximately 10% of IHCAs, resulting in diffuse staining with GS with or without nuclear β-catenin staining; these tumours are thought to have a higher risk of progression to HCC. , A combination of gp130 alterations and β-catenin mutation is present in these cases.
The BHCA subtype occurs more often in men than do the other subtypes and often harbours atypical morphological features, such as cytological atypia, small-cell change and pseudoglandular/acinar architecture. , , Multifocal reticulin loss may occur ( Fig. 13.13 ), raising concerns for transition to HCC. Association with concurrent or subsequent HCC may occur in almost half the cases. These tumours are characterized by mutations in the exon 3 of the CTTNB1 (β-catenin) gene, which is a critical component in the Wnt signalling pathway. In normal cells, β-catenin is located in the membranous region and can be demonstrated by IHC ( Fig. 13.14 ). Cytoplasmic β-catenin binds to a protein complex that targets it for proteasomal degradation. Deletions or point mutations in the exon 3 region interfere with the binding of β-catenin to the protein complex, which in turn interferes with the cytoplasmic degradation of the protein and leads to its nuclear translocation. Nuclear β-catenin activates the T-cell factor/lymphoid enhancer factor (TCF/LEF) family of proteins, leading to expression of a variety of Wnt target genes that play an important role in cell proliferation. IHC demonstration of nuclear β-catenin can help to identify tumours with β-catenin-activation ( Fig. 13.14 ). Activated β-catenin leads to transcriptional upregulation of GLUL gene, which codes for GS, and diffuse GS staining can be demonstrated by IHC as evidence of β-catenin activation ( Fig. 13.15 ).
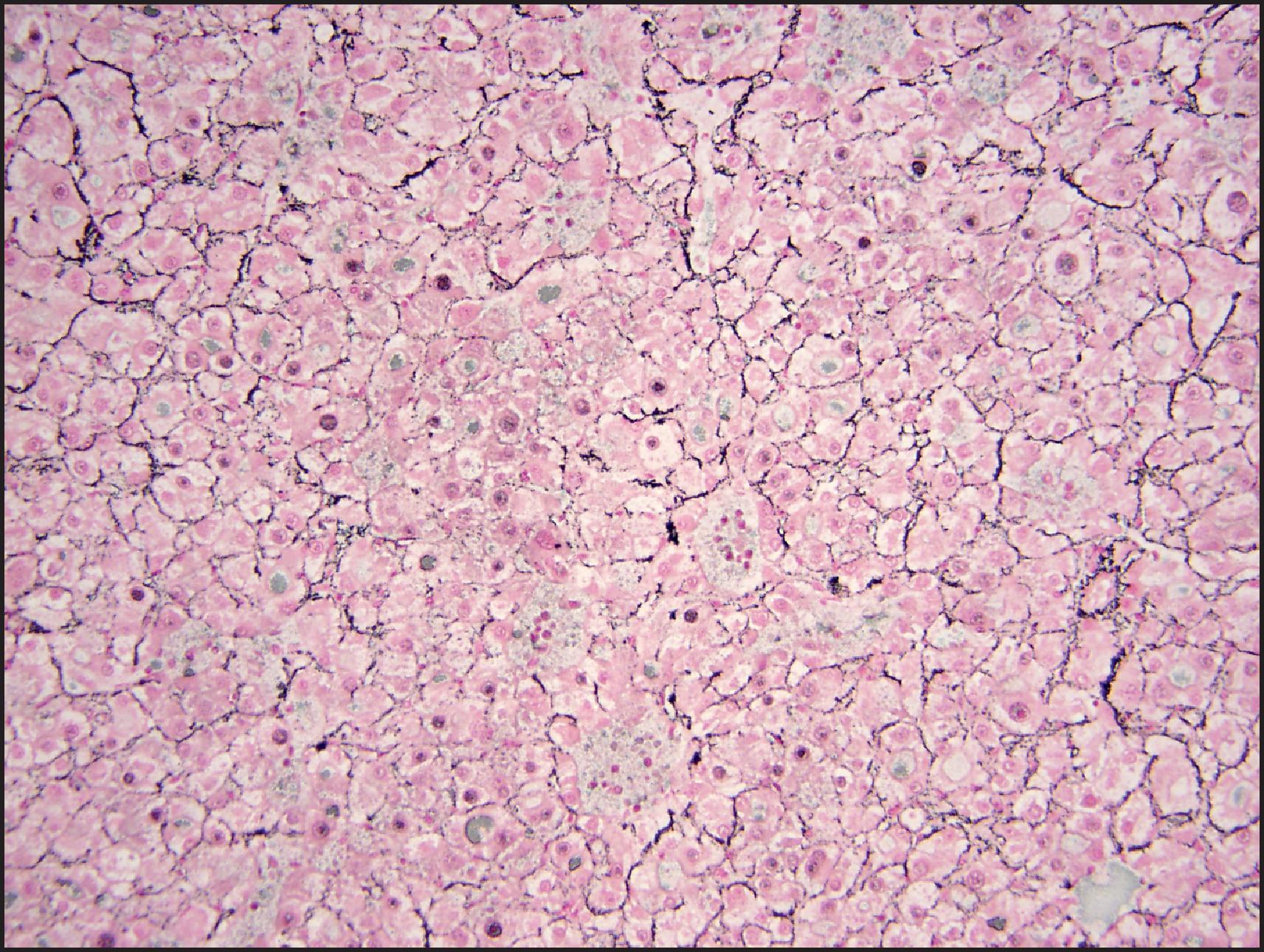
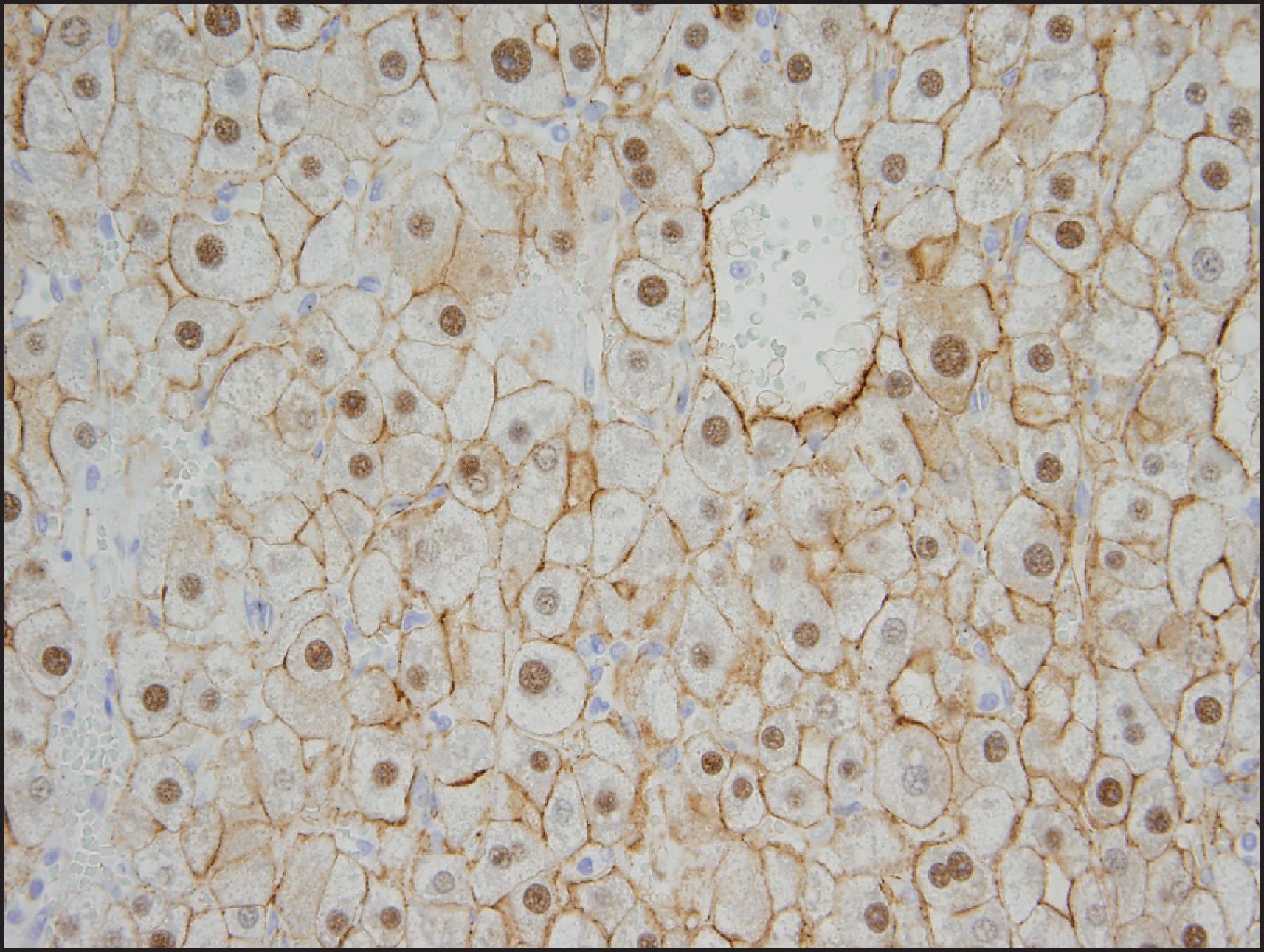
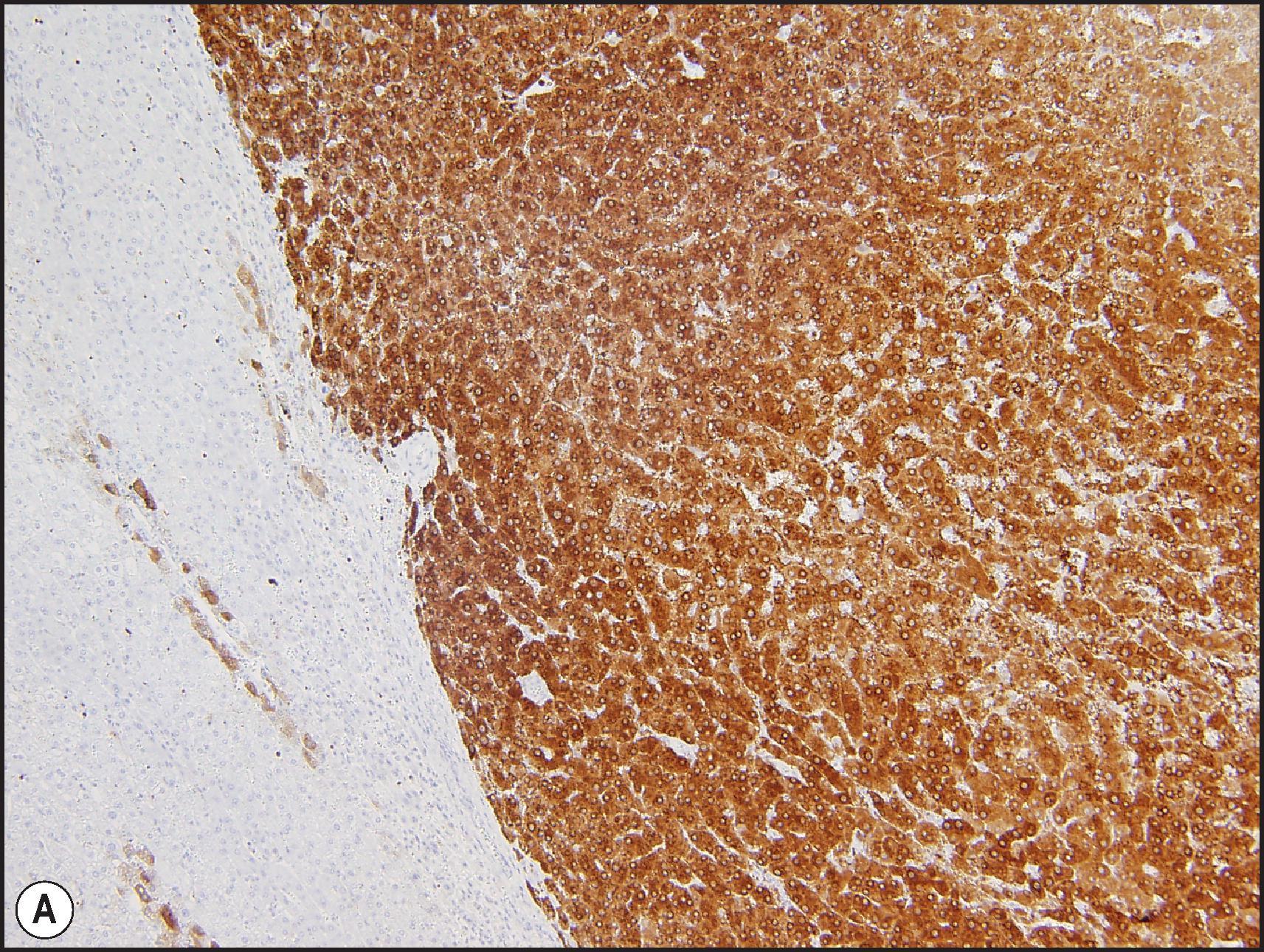
In view of the atypical histological findings, frequent reticulin loss, high risk of HCC and rare instance of metastases in these BHCAs, it may be better to regard these tumours as extremely well-differentiated variants of HCC rather than adenomas. , , Cytogenetic abnormalities such as gains of 1q and 8q are typical of HCC and are often seen in adenoma-like tumours with β-catenin activation, further supporting their classification as HCC. In some cases, adenoma-like tumours with β-catenin activation do not show features that are sufficient for definite diagnosis of HCC. It has been suggested that these tumours should be characterized as ‘atypical hepatocellular neoplasm’ (AHN), ‘hepatocellular neoplasms with uncertain malignant potential’ (HUMP) or borderline lesion. , , , In addition to tumours with borderline histological features and those with β-catenin activation, this designation has also been recommended for all adenoma-like tumours that occur in men, women >50 years and those occurring in unusual clinical settings (e.g. GSD, Fanconi anaemia). It may also be prudent to use this designation for tumours with aberrant IHC features (e.g. GPC3 or HSP70 positivity). In addition, it is possible that the β-catenin activation can occur in any other HCA subtype (HHCA, IHCA or unclassified) as a ‘second hit’ in the progression of HCA to HCC, and thus BHCA may not truly represent a subtype of HCA. In initial studies, the BHCA subtype accounted for 10–15% of total HCAs, , but the numbers in more recent studies are less than 5%, , perhaps reflecting that many tumours initially considered as BHCAs are now being classified as ‘extremely well-differentiated HCC’ if enough cytoarchitectural atypia and/or reticulin loss is present.
Nuclear β-catenin staining is present in less than half of the tumours with known β-catenin mutation or deletion (see Fig. 13.14 ), whereas diffuse GS staining has greater sensitivity and better correlation with β-catenin activation , (see Fig. 13.15 ). Therefore, the absence of nuclear β-catenin on IHC does not exclude β-catenin activation in a tumour. When present, nuclear β-catenin staining can be focal, and any unequivocal nuclear staining irrespective of number of positive tumour cells should be regarded as a positive result. Because of possible nonspecific background cytoplasmic staining, it is recommended that cytoplasmic β-catenin staining in the absence of nuclear staining not be considered positive.
The interpretation of GS IHC plays a critical role in the classification of HCA subtypes. GS is an enzyme involved in ammonia detoxification by combining it with glutamate to produce the amino acid glutamine. In normal liver, GS expression is limited to a narrow rim of hepatocytes around the central vein. This zonation is thought to be the result of β-catenin activation in centrizonal hepatocytes, presumably as a result of Wnt signalling from the central vein. Activation of β-catenin in neoplastic hepatocytes leads to diffuse cytoplasmic expression of GS in the tumour cells. Due to the low sensitivity of β-catenin IHC, it has become standard clinical practice in diagnostic pathology to use diffuse GS immunostaining as a marker of β-catenin activation. In this context, diffuse staining has been defined as ‘moderate to strong’ cytoplasmic staining in >50% of tumour cells. Diffuse staining can be seen in almost all (>90%) the tumour cells (diffuse homogeneous staining) ( Fig. 13.15A ), or it can be seen in >50% but <90% of tumour cells (diffuse heterogeneous staining) , , ( Fig. 13.15B ). The interpretation of diffuse homogeneous GS staining is often straightforward, but it can be difficult to discern whether staining is seen in >50% of tumour cells when a diffuse heterogeneous pattern is encountered. Diffuse homogeneous GS is associated with high-level β-catenin activation that results from exon 3 deletions or certain mutations (D32-S37, T41A). Diffuse heterogeneous GS denotes an intermediate level of β-catenin activation associated with mutation such as S45, and this pattern of GS staining has been referred to as ‘starry sky’ ( Fig. 13.15B ). However, the correlation between mutations and GS staining pattern can show exceptions, with diffuse heterogeneous pattern with T41A and diffuse homogeneous with S45 mutation. , Irrespective of the pattern of diffuse GS, a diffuse result is considered a marker of β-catenin activation.
In contrast to the high correlation between diffuse GS staining and β-catenin activation in HCAs reported in some studies and in the WHO classification, , recent studies have shown that GS is not a perfect marker of β-catenin activation, and many cases with diffuse GS staining (20–30%) may not have evidence of β-catenin mutation or deletion. , , The correlation is likely to be higher for diffuse homogeneous GS staining. The reasons for the discrepancy are unclear but may be related to activation of other components of the Wnt signalling pathway, such as AXIN1 mutation, or mechanisms unrelated to mutations in genes in Wnt signalling, including alterations in vascular flow. It thus seems prudent to diagnose these tumours as HCA or AHN with diffuse GS staining and convey that an underlying β-catenin ( CTTNB1 ) mutation or deletion is likely. GS staining in other subtypes of HCA tends to be patchy and, by definition, involves <50% of hepatocytes. This patchy staining can be perivenular and is often more prominent compared to normal liver (‘expanded perivascular staining’). Peripheral accentuation can be seen. , In some instances, the patchy areas of staining at the periphery can connect with each other, creating a pattern reminiscent of the map-like pattern seen in FNH (‘pseudo-map-like’).
Mutations in exons 7 and 8 of CTTNB1 have been described in 10% of HCAs. Mutations in exons 7/8 and exon 3 tend to be mutually exclusive. These cases do not show atypical morphological characteristics or diffuse GS staining typical of tumours with exon 3 CTTNB1 mutations. Peripheral accentuation of GS staining is common and up to 10% of cases may exhibit a diffuse heterogeneous pattern. , Since diffuse GS is not seen in a vast majority of these tumours, these will get categorized as ‘unclassified’ based on IHC stains currently used for HCA subtyping. Based on initial results, it is thought that mutations in exons 7 and 8 do not confer the malignant potential associated with exon 3 CTTNB1 mutations. However, their neoplastic potential needs to be further studied as these mutations have been identified in HCC.
HCAs that lack distinctive immunohistological and molecular abnormalities of the other three subtypes have hitherto been described as ‘unclassified’. However, an additional sonic hedgehog subtype of HCA (shHCA) has recently been described, comprising 4% of HCAs. This subtype results from activation of the sonic hedgehog pathway resulting from small somatic deletions of INHBE , that lead to fusion of INHBE and GLI1 genes. This fusion leads to overexpression of several genes including PTGDS which encodes prostaglandin D synthetase; IHC for PTGDS shows increased expression in this subtype. However, further studies are needed to examine their risk potential and to confirm if they represent distinct entities. Association with obesity and high risk of haemorrhage have been reported with shHCA.
Increased expression of argininosuccinate synthase 1 (ASS1), an enzyme involved in arginine synthesis, has also been reported to be a marker for a high risk of haemorrhage and a possible marker for shHCA. , However, ASS1 positivity can also be seen in other HCA subtypes such as IHCA; its association with haemorrhage has been questioned, and its clinical utility is not yet fully established. ,
Hepatic adenomatosis is arbitrarily defined as the occurrence of ≥10 HCAs. This condition can be familial, in which case it usually occurs in association with a specific form of diabetes (MODY3) and is caused by germline mutation in the TCF1 gene that encodes HNF1α , (see Fig. 13.9 ). These adenomas show loss of LFABP on IHC. Additional germline mutations in other genes such as CYP1B1 can increase the risk of HCAs in MODY patients. Young age, male gender and a history of diabetes can further increase this risk. Adenomatosis with IHCAs can rarely occur in McCune–Albright syndrome. Rare instances have been described in vascular conditions such as congenital absence of the portal vein and post-Fontan procedure. ,
Hepatic adenomatosis also has similar risk factors and clinicopathological settings to sporadic HCAs, including predilection for young women. Several reports have implicated obesity and fatty liver disease as risk factors for adenomatosis , of the inflammatory subtype (see Fig. 13.2 ). In most cases, all HCAs in adenomatosis are of the same histological subtype, although a mix of IHCA and HHCA has been reported in the same case. Treatment is dictated by the size of the nodules and subtype of HCA. As with sporadic HCA, the risk for HCC is highest with larger tumours (>5 cm) and those with β-catenin activation. Therefore, resection is often performed in these patients as well as for symptomatic patients. Embolization and transplantation are options for unresectable cases.
HCAs may occur in about half the patients with type I and 25% of those with type III GSD. IHCAs comprise 52–65% of adenomas, and most have mutations in the IL6/JAK/STAT3 pathway similar to sporadic IHCAs. , Up to 20% of HCAs have been reported to have β-catenin activation, , and HCC is well described in GSDs. Thus the diagnosis of HCA should be made with caution in this setting, especially when β-catenin activation is present and the designation of ‘AHN’ or ‘HUMP’ can be used for cases with borderline features. Due to the risk of HCC, screening with serum AFP and annual US are recommended (see also Chapter 3 ).
Nodular lesions that are histologically similar to HCA have been observed in the setting of vascular diseases, including Budd–Chiari syndrome, portal vein agenesis (see also Chapter 11 ), hereditary haemorrhagic telangiectasia and tetralogy of Fallot. , Loss of LFABP and positivity for CRP have been described, although the typical histological features of IHCA are not seen. Since these can be multiple and can be mixed with FNH-like nodules, it has been proposed that these HCA-like nodules are not neoplastic and represent regenerative nodules as a result of uneven blood perfusion throughout the liver. However, β-catenin activation and association with HCC have been reported, and careful review is necessary before concluding that the nodules are regenerative or benign.
Nodular lesions with histological features of IHCA and SAA/CRP positivity have been described in cirrhosis related to alcoholic as well as nonalcoholic steatohepatitis (NASH). , Mutations in the IL6/JAK/STAT pathway have been described, supporting the neoplastic nature of these nodules. Thus, rare cases of IHCA can occur in cirrhosis.
HCC arising in the setting of HCA is a rare phenomenon and has been reported in 4–8% of cases. , , Advanced age, male gender, use of anabolic steroids, metabolic syndrome and large tumour size have been cited as risk factors for HCC transformation in an adenoma. , Both tumours occur concurrently in most reported cases; it has been assumed that this represents malignant transformation of an adenoma. BHCAs are more likely to be associated with concurrent or subsequent diagnosis of HCC. The adenoma portion often shows focal atypical morphological features and molecular features resembling HCC, suggesting that the adenoma-like portion in a subset of these tumours may represent an extremely well-differentiated form of HCC (see previous discussion). Pathological features considered to be high risk in HCA include prominent myxoid change, marked pigmentation, ß-catenin activation and atypical morphologic features (pseudoacini, focal reticulin loss, etc.) that are considered insufficient for a definite diagnosis of HCC. It may be prudent to use the designation of AHN or HUMP for these cases. , , , ,
Discontinuation of OCs, hormone-containing intrauterine devices (IUDs) and anabolic steroids may lead to regression of HCA. HCAs associated with GSD may regress with dietary therapy. Resection is recommended for tumours ≥5 cm because of the risk of rupture, haemorrhagic complications and development of HCC. Male gender and β-catenin activation are also considered high-risk features and warrant consideration of resection. Nonsurgical options such as embolization can be considered for patients who are poor surgical candidates. Since some HCAs have been reported to increase in size despite the discontinuation of OCs or anabolic steroids, and the development of HCC has been reported despite regression in size, it is recommended that patients with smaller tumours have CT or MRI at 6-month intervals for 2 years, with subsequent follow-up dictated by the growth pattern and stability of the tumour over time. If multiple HCAs are present, the decision of follow-up vs. resection is determined by size and risk factors of individual nodules, with resection being recommended for high-risk nodules.
For clinical purposes, the subtyping and risk assessment of HCA are most significant for smaller tumours (<5 cm) in women as larger tumours and HCA in men are considered indications for resection irrespective of morphologic risk factors. Hence accurate assessment of atypical morphologic features as well as β-catenin activation (based on pattern of GS staining) can be the determining factors guiding resection in small tumours. If GS and/or β-catenin staining yields equivocal results on the biopsy, evaluation for mutations in CTNNB1 (β-catenin) or other components of Wnt signalling pathway can be useful for accurate risk assessment.
FNH can sometimes be difficult to distinguish from HCA. The presence of fibrous septa containing large arteries and ductular reaction favours FNH (see next section). However, these features can also be seen in IHCA, so the distinction can be challenging, especially in small biopsies. In these settings, map-like GS staining in conjunction with histological features can confirm the diagnosis of FNH. Notes of caution, however, include that the periphery of HCAs may show a vague anastomosing pattern of staining reminiscent of the map-like pattern of FNH. In addition, SAA can be positive in 10–15% of FNH cases, typically in a focal manner, while CRP staining in the periseptal region is seen in most FNH cases. Diffuse staining typical of IHCA is restricted to a small minority of FNH cases.
Distinguishing HCA from WDHCC can also be challenging and may be impossible on needle biopsies. The presence of wide cell plates, prominent pseudoglandular/acinar architecture, zones of small-cell change, cytological atypia and reticulin loss favour HCC (see Table 13.5 ). Diffuse sinusoidal staining for CD34 also favours HCC but is insufficient alone for the diagnosis. GPC3 and heat shock protein 70 (HSP70), if positive, strongly point to HCC. Similarly, the demonstration of cytogenetic abnormalities such as gains of chromosome 1 and 8 and TERT promoter mutation would indicate HCC. , If the histological changes are borderline, or if high-risk features such as male gender and β-catenin activation are present, it is prudent to designate the tumour as AHN or HUMP.
FNH is a benign, non-neoplastic lesion that can occur in both genders and all ages but is most common in adult women. In most cases, FNH is discovered incidentally at surgery or in imaging studies. FNH is thought to represent a hyperplastic response of hepatocytes to altered blood flow (see also Chapter 11 ). Most women (70–80%) with FNH are taking OCs, but these are not thought to play an aetiological role. The hepatocellular component is polyclonal, which supports the conclusion that FNH represents a regenerative rather than a neoplastic process. Angiopoietin is involved in vasculogenesis and is upregulated in FNH.
Most FNH cases can be diagnosed by multiphasic CT scan. FNH shows hyperattenuation in the arterial phase of multiphase CT scan, and it becomes isoattenuating with normal liver in the delayed phase. The central scar is hypoattenuating and becomes hyperattenuating in the venous phase. On T1-weighted magnetic resonance (MR) images, FNH is typically isointense to hypointense. On T2-weighted images, it is hyper- or isointense, and the scar is hyperintense in most cases. US is not a preferred modality because FNH is not well visualized. The central scar can be mistaken for fibrolamellar hepatocellular carcinoma (FLC) on imaging. If imaging shows typical features including a central scar, the patient may be followed without biopsy.
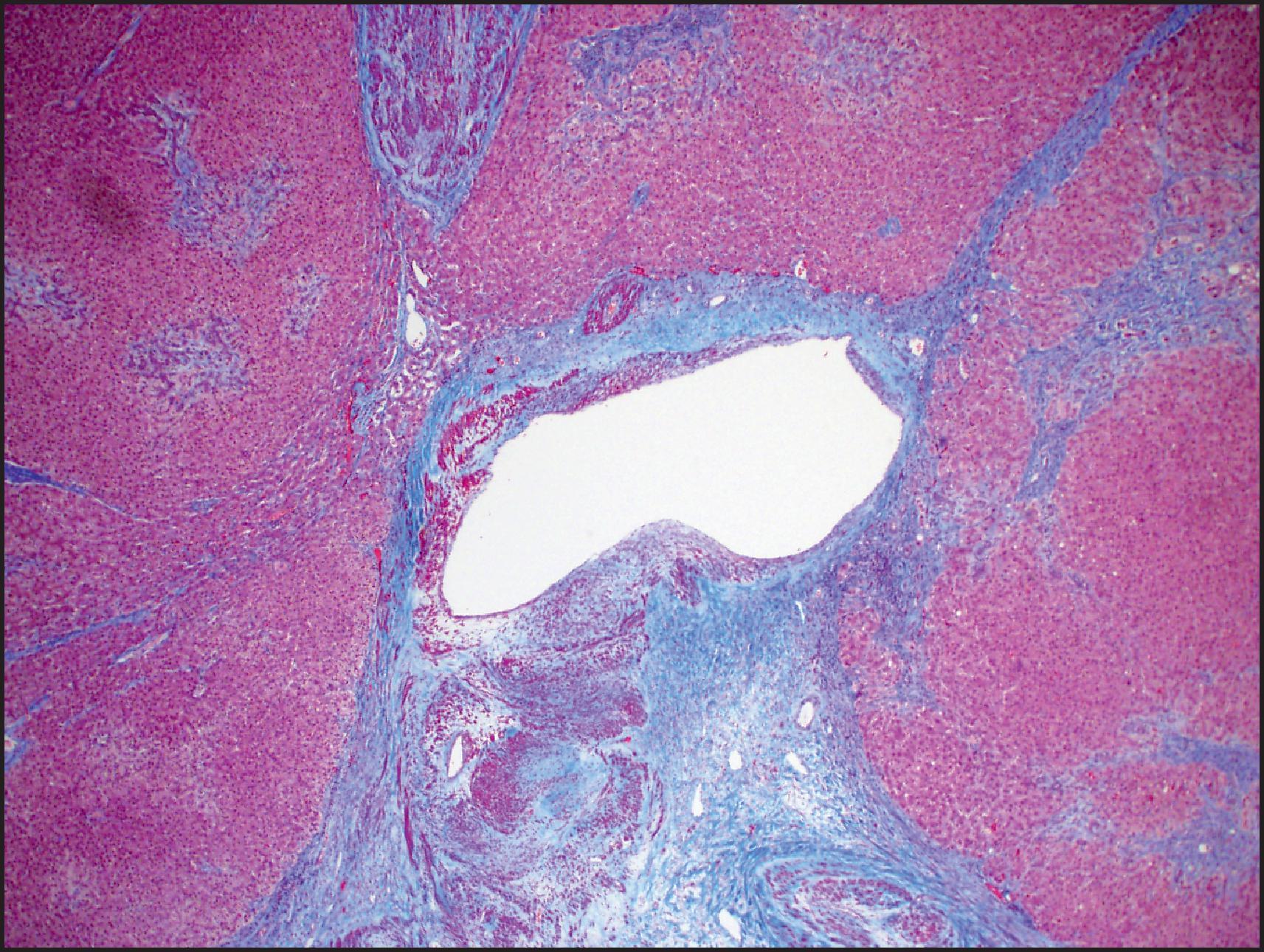
More than 85% of FNH lesions are <5 cm in diameter, but larger lesions can occur. FNH is usually well circumscribed but nonencapsulated. The colour is lighter than that of the adjacent normal liver. On cut section, the lesion is subdivided into smaller nodules by fibrous septa that often run into a stellate scar that may be central or eccentric ( Fig. 13.16 ). Several scars may be seen in large lesions. A central scar is absent in 20% of cases. Microscopically, FNH is characterized by hyperplastic nodules that are separated by fibrous septa, often radiating from a central scar. The fibrous septa and central scar contain dystrophic thick-walled arteries and, to a lesser extent, veins. The arteries are often eccentrically thickened and show intimal and fibromuscular hyperplasia with poorly formed elastic lamina ( Figs. 13.17 and 13.18 ). The scar and fibrous septa typically show a ductular reaction, most prominent at the interface between the nodules and fibrous stroma, and variable lymphocytic inflammation, while interlobular bile ducts are absent. The hepatocellular component is arranged in plates one to two cells thick, with intact reticulin framework, but thicker plates may be present focally, often near the centre and periphery of the lesion ( Fig. 13.19 ). Complete circling of small groups of hepatocytes by reticulin fibres (packeting) is also a common feature. Fat accumulation can be seen, especially in patients with fatty liver disease (see Fig. 13.18 ). Steatohepatitic changes such as ballooned hepatocytes and MDBs may also be present and should not be confused with the steatohepatitic variant of HCC. The periseptal hepatocytes adjacent to the fibrous septa of the lesion often show chronic cholestasis, with cholate stasis (‘pseudoxanthomatous transformation’) and copper accumulation. These features of chronic cholestasis can provide a clue to the diagnosis of FNH.

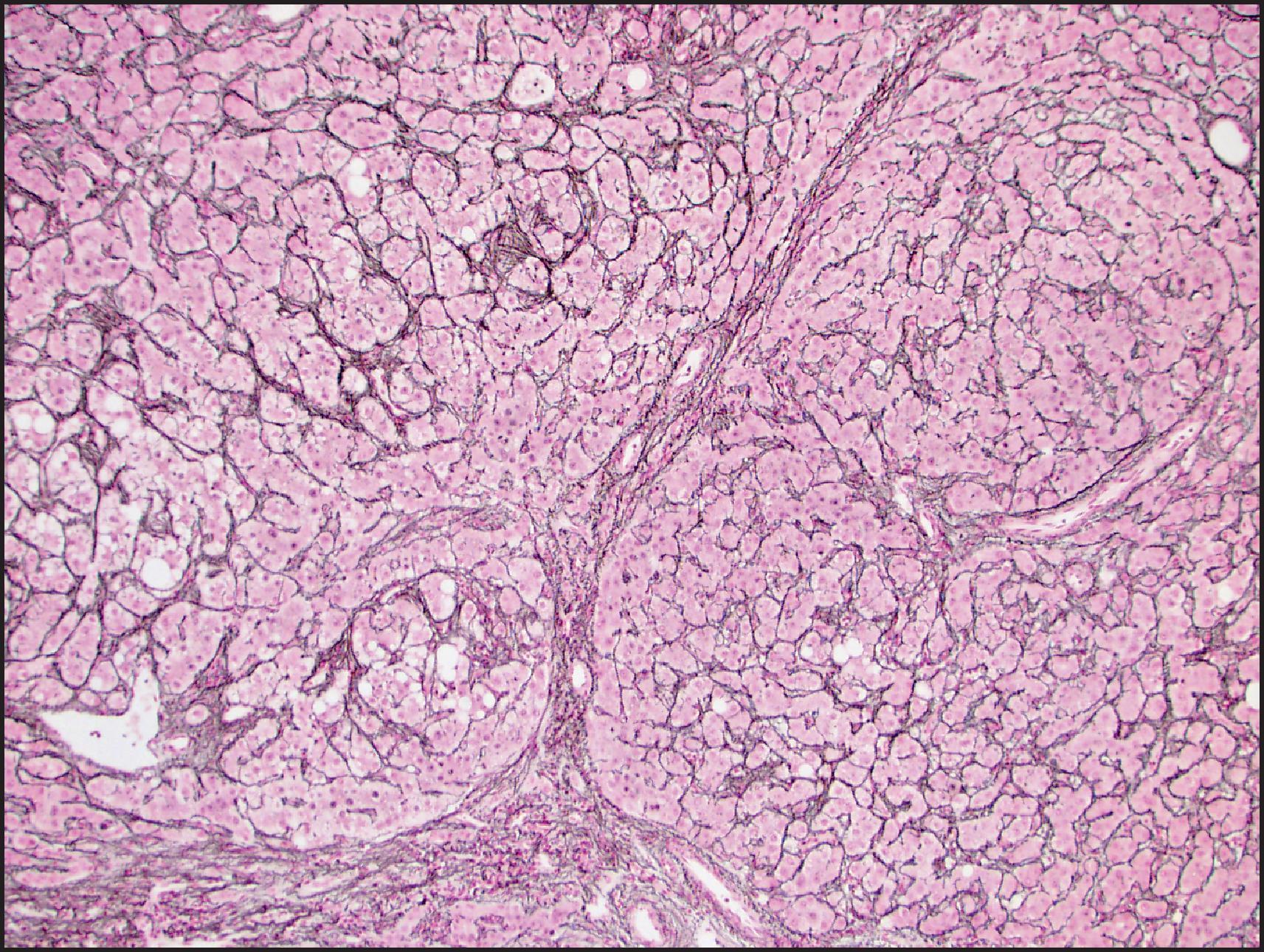
On IHC, FNH shows a highly characteristic geographical or map-like pattern on GS staining, with strong cytoplasmic staining of broad anastomosing groups of hepatocytes ( Fig. 13.20 ). The map-like pattern can be focal in needle biopsies and may not be well developed around the central scar or areas with fatty change. , In normal liver, GS expression is limited to a narrow rim of hepatocytes around the central vein. This zonation most likely results from β-catenin activation in the centrizonal hepatocytes, which in turn may be the result of Wnt signalling from the central vein. Expansion of the β-catenin-activated centrizonal region leads to GS overexpression in the map-like pattern in FNH. β-catenin mutations are not observed. The ductular reaction is highlighted by keratins 7 and 19 and neuronal cell adhesion molecule (NCAM). CD34 usually shows patchy sinusoidal staining, with diffuse staining in a small number of cases.
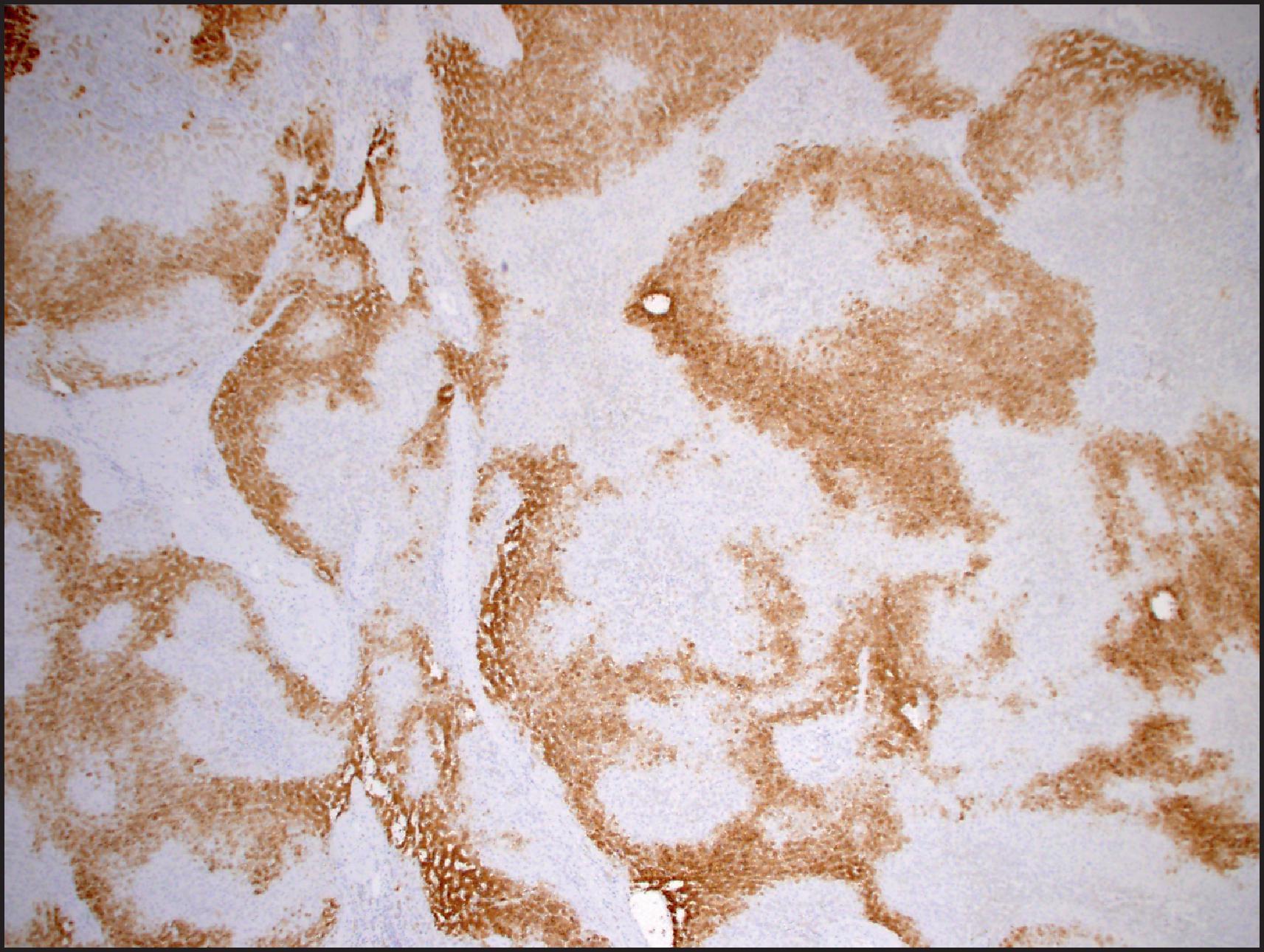
On FNA aspiration cytology, a polymorphous population of cells is seen, comprising hyperplastic hepatocytes with normal nuclear/cytoplasmic ratio (≤1:3, by estimating comparison of nuclear and cell diameters), biliary elements, endothelium and Kupffer cells. Hepatocytes in FNH show some mild variation in cell and nuclear size but maintain the normal nuclear/cytoplasmic ratio; they are not monotonous or pleomorphic, similar to that seen within a family of siblings, unless they are identical twins, so-called sibling variation. Anisonucleosis is also a distinct characteristic of non-neoplastic hepatocytes. Another helpful benign feature is the presence of biliary ductular clusters. They represent ductular reaction at the hepatocellular–stromal interface of the scar and appear as curved, double-stranded rows of small cells with overlapping, oval nuclei and scanty or absent cytoplasm ( Fig. 13.21 ). The rows can be seen emanating from hepatocytes. Ductular clusters should be distinguished from bile duct epithelium, which appears as flat sheets of uniform equidistant cells with discernible cytoplasm. Ductular reaction can easily be mistaken for adenocarcinoma, such as cholangiolocellular (CLC) carcinoma. Endothelial and Kupffer cells are difficult to separate cytologically and are recognized as spindle/comma-shaped nuclei and nuclear streaks. Stromal fragments are rare and may mimic leiomyosarcoma. , ,

Telangiectatic FNH refers to a subset of cases with prominent sinusoidal dilation. Based on imaging features, clonality results and gene expression studies, most but not all ‘telangiectatic FNH’ cases are now classified as IHCA. , ‘FNH-like’ lesion has been used to describe lesions morphologically similar to FNH, but that occur in the setting of liver disease (e.g. cirrhosis) and vascular disorders (e.g. idiopathic portal hypertension, hereditary haemorrhagic telangiectasia, haemangiomas, Budd–Chiari syndrome). These FNH-like lesions can be multiple and may not show the characteristic map-like pattern of GS staining seen in true FNH. The term ‘FNH-like’ has also been used for lesions that mimic FNH but do not show all the characteristic morphological and/or IHC features.
FNH is a non-neoplastic lesion and does not require surgical resection even if large, unless the patient is symptomatic or the location of the lesion compromises liver function. Thus, it is important to distinguish FNH from HCA and HCC (see Table 13.5 ). The nodular architecture, central stellate scar, fibrous septa and ductular reaction distinguish FNH from HCA. However, these features can be seen in IHCA, and the distinction can be challenging on histological grounds alone, especially in needle biopsies. Map-like GS pattern is diagnostic of FNH and is not seen in IHCA. Staining with inflammatory markers such as SAA is typically negative or focal, while CRP staining is usually restricted to periseptal areas; diffuse staining typical of IHCA is uncommon in FNH. Sinusoidal dilation (telangiectasia) is more common in HCA but can be seen in 15–20% of FNH, and it cannot be relied on to distinguish these entities. Core needle biopsies can mimic ductopenic cirrhosis, but the clinical setting and presence of dystrophic arterioles suggest FNH.
Nodular regenerative hyperplasia (NRH) is usually considered a reaction to heterogeneous or abnormal blood flow within the liver, which has many causes. The lesion consists of a nodular transformation of the liver that mimics nodules in cirrhosis but lacks the scarring typical of that condition, so NRH is not likely to be confused with a neoplasm (see Chapter 11 ). Occasionally, however, as in cirrhosis, there may be larger regenerative nodules among the smaller nodules, which may be identified on radiology and confused with other hepatocellular tumours, particularly HCA.
Hepatoblastoma (HB) is the most frequent liver tumour in children, with an annual incidence of approximately 4 per 1 million in children under 5 years of age. HB mimics the developing fetal or embryonal liver histologically and may contain other heterologous epithelial or mesenchymal elements. A majority of cases occur before the age of 5 years, with rare reports of the tumour in older children and adults.
Congenital anomalies are present in approximately 5% of patients with HB and reported associations with other congenital diseases include Beckwith–Wiedemann syndrome, familial adenomatous polyposis coli, Li–Fraumeni syndrome, trisomy 18, GSD types I–IV, hemihypertrophy, Sotos syndrome and Simpson–Golabi–Behmel syndrome. Rare cases of congenital HB have been reported.
Cytogenetic studies and comparative genomic hybridization have revealed that tumour cells have a relatively stable genome and usually harbour a limited number of chromosomal abnormalities (mean, three changes per tumour). Recurrent genetic alterations involve chromosomes 2, 20, 1, 8 and X. , Frequent trisomies 2 and 20, often seen as the only abnormality, have also been observed in other embryonic tumours such as embryonal rhabdomyosarcoma. The DNA content is frequently diploid in the fetal type and aneuploid in half the embryonal and small, anaplastic cell types. Microsatellite analysis revealed regions with allelic loss of chromosomes 1 and 11.
Several studies revealed that genes encoding components of the wingless/Wnt signal transduction pathway, including the APC , AXIN1 and CTTNB1 genes, are mutated in a large proportion of cases (>80% in total). These mutations lead to abnormal activation and induction of growth-promoting genes and other survival pathways (e.g. Notch pathway) that can be differentially expressed in different histological subtypes of HB. The central molecule of this pathway is β-catenin, which carries mutations, mostly in-frame deletions, in more than half of HB cases. An additional gene reported to be affected by recurrent mutations in HB is NFE2L2 , which encodes a transcription factor involved in the antioxidant response pathway.
Other typical molecular features include aberrant reprogramming of imprinted genes such as IGF2, H19, DLK1, GTL2, PEG3, PEG10, BEX1, MEG3 and NDN , which are abundantly expressed in fetal liver. Based on recent transcriptional data, HB can be classified into two distinct groups. The C1 subclass recapitulates liver features at late stages of intrauterine life, with a mostly fetal phenotype and the expression of markers of mature hepatocytes. The C2 subclass has a predominantly embryonal phenotype, with the expression of markers of hepatic progenitor cells (e.g. K19, epithelial cell adhesion molecule [EpCAM]).
Studies of gene expression profiling have compared HB with HCC and identified upregulation of expression of MIG6, TGFβ1, DLK1 and IGF2 in HB. These genes were differentially expressed between HB and HCC, but they did not separate HB histological subtypes. ,
Approximately 75% of children with HB are male. They usually present with abdominal swelling and less frequently with weight loss, anorexia, nausea, vomiting, abdominal pain and jaundice. Thrombocytosis and marked elevation of serum AFP are present at diagnosis in most patients. Although biopsy is necessary to confirm the diagnosis, imaging plays an important role in determining the location and extent of tumour, assessing the feasibility of surgery and evaluating vascular invasion and extrahepatic extension.
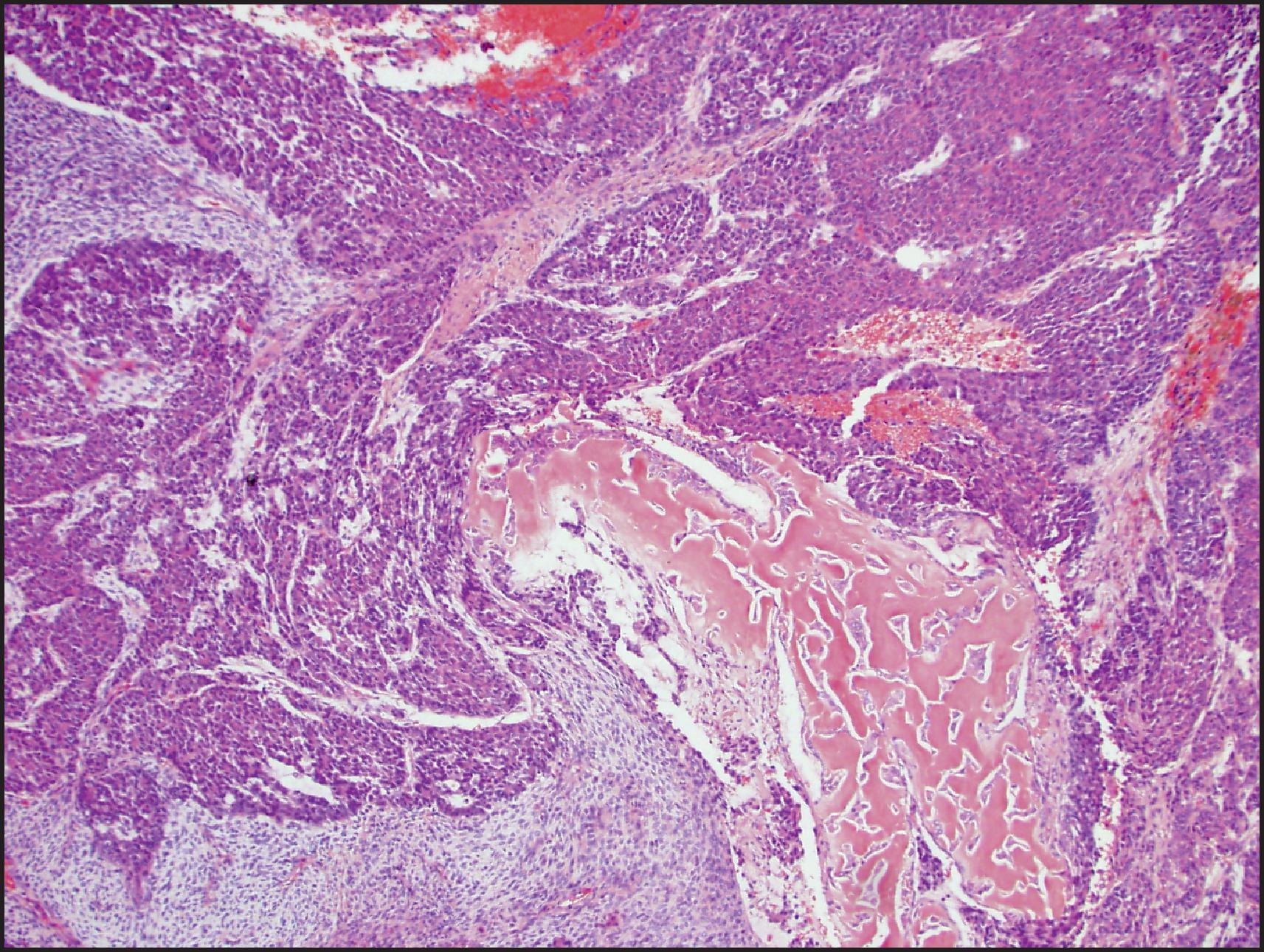
HBs usually present as a single mass but can be multifocal and may measure up to 25 cm in diameter. The right lobe is more often involved. The non-neoplastic liver is usually normal. HB typically has a lobulated cut surface and may be yellow, brown, green or variegated, depending on the differentiation of the tumour and whether a mesenchymal component is present. In post-therapy resections, a heterogeneous appearance with areas of haemorrhage and necrosis is common. Calcification and ossification may be present. Specific attention should be paid to gross vascular invasion and surgical margins, and fresh tissue should be set aside for special studies whenever possible. It is important to map the resected tumour with appropriate sampling to evaluate the morphology of the residual tumour and response to chemotherapy.
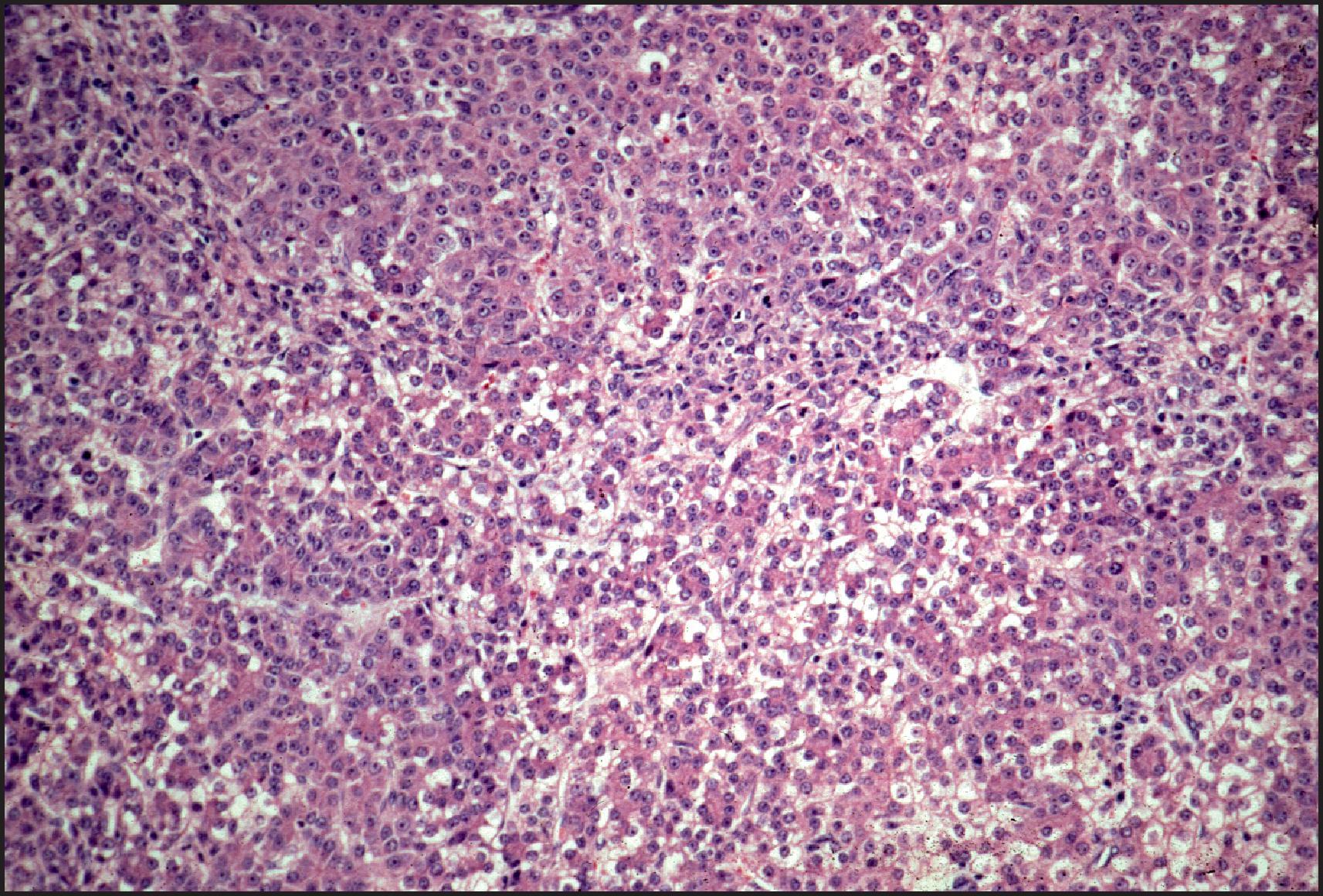
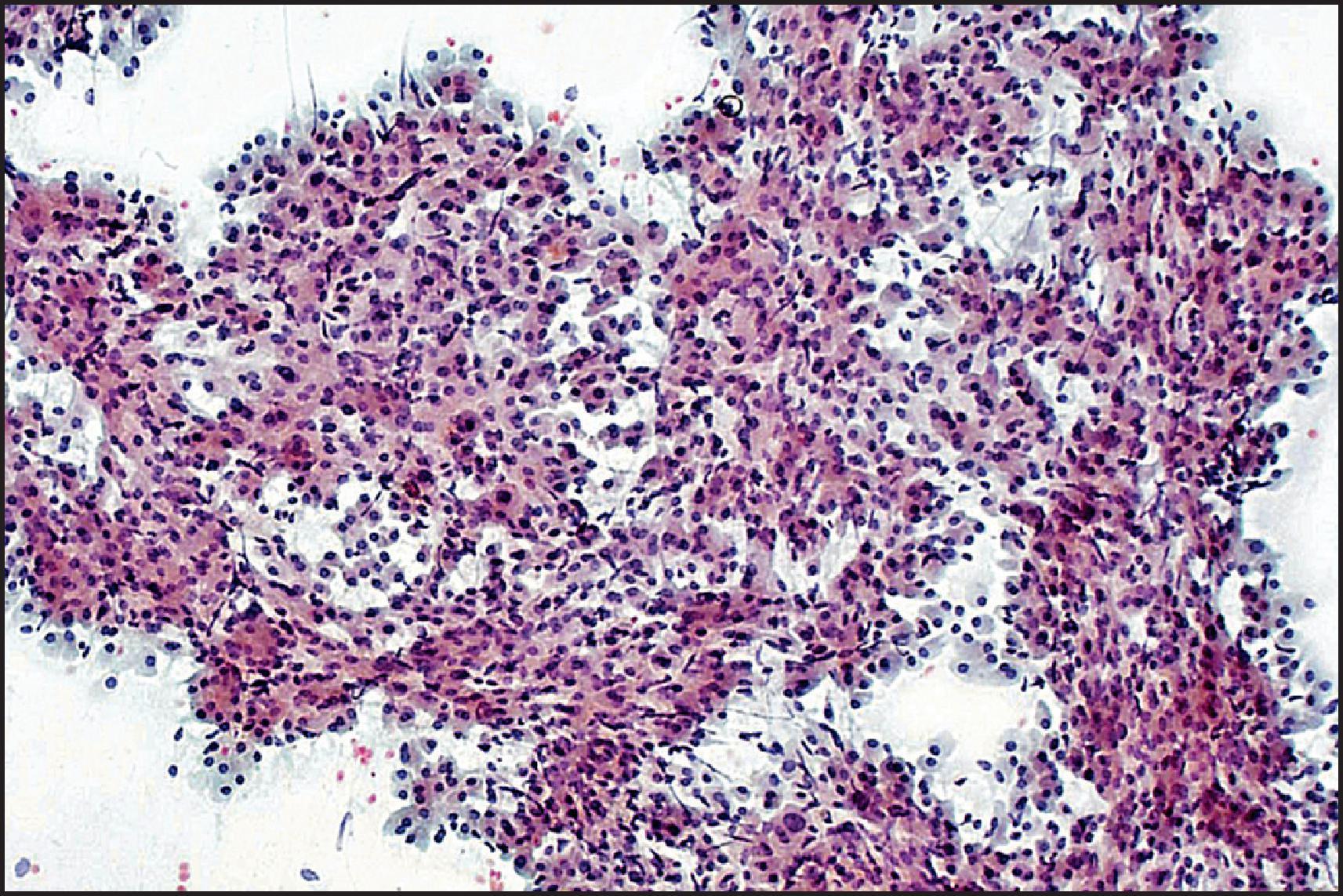
Histologically, HB may be classified as epithelial or mixed epithelial-mesenchymal, each with several recognized subtypes. , , The epithelial type is the more common. The fetal epithelial pattern comprises one-third of the cases. Well-differentiated fetal HB is composed of sheets and thin trabeculae of polygonal cells resembling fetal hepatocytes with a small round nucleus, inconspicuous nucleolus, clear to finely granular cytoplasm with variable amounts of cytoplasmic glycogen and lipid giving a ‘light and dark’ pattern to the tumour when viewed at low-power magnification ( Fig. 13.22 ). Haematopoietic cells are almost always present, mimicking the typical extramedullary haematopoiesis (EH) of normal fetal liver. Pure well-differentiated HBs are composed exclusively of fetal pattern with uniform tumour cells, and mitoses <2 in 10 high power fields. This diagnosis can be reliably established only in primary resections, and not on biopsy or post-therapy resection. In the crowded fetal subtype , the tumour cells are more closely packed and have higher nuclear/cytoplasmic ratios, decreased cytoplasmic glycogen, more eosinophilic cytoplasm and higher mitotic activity (≥2 in 10 high power fields) compared to the well-differentiated fetal pattern ( Fig. 13.23 ).
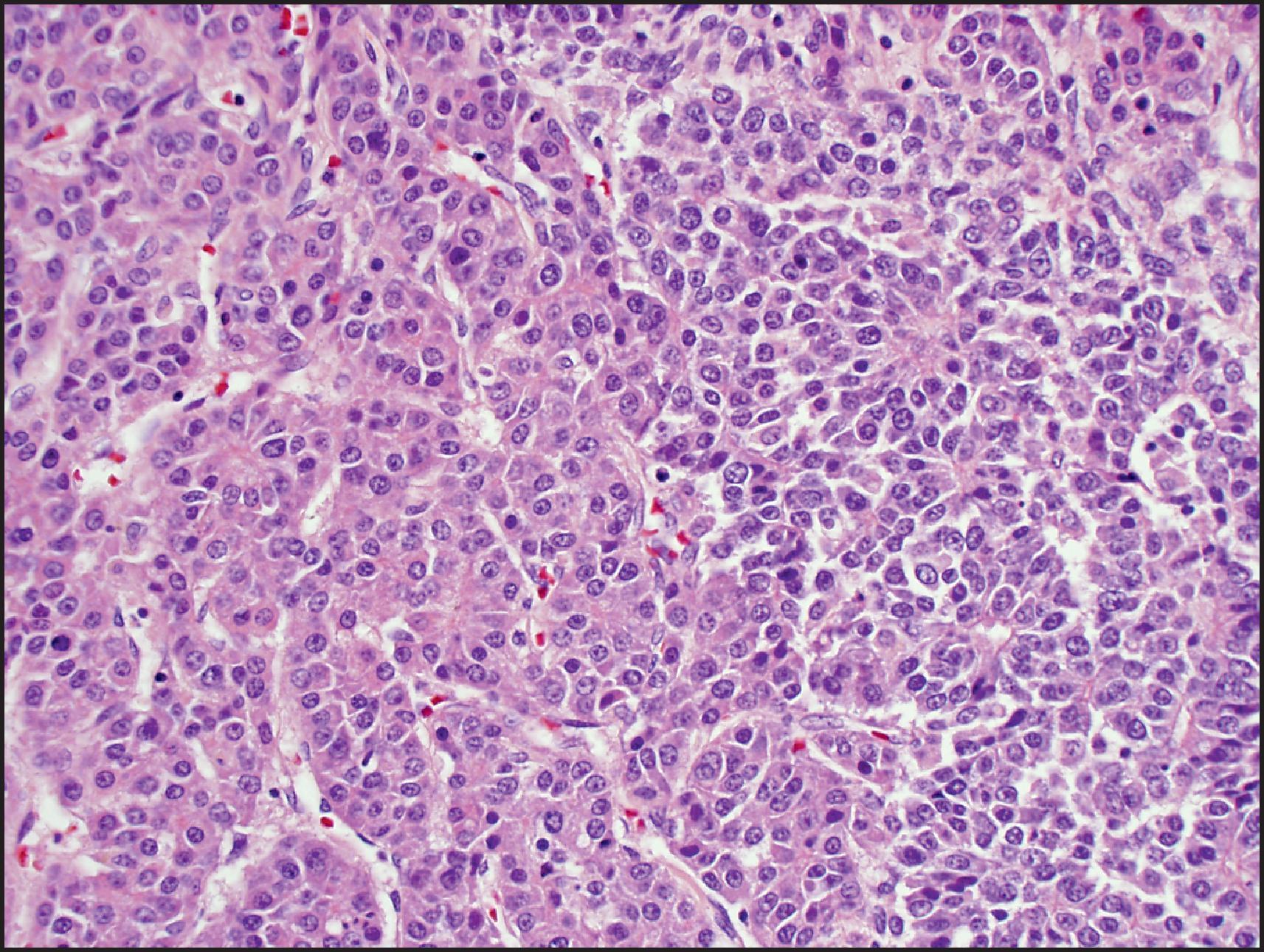
The fetal pattern is mixed with the embryonal pattern in 20% of cases, and the crowded fetal pattern may merge into the embryonal pattern (see Fig. 13.23 ). The embryonal pattern corresponds to the sixth to eighth weeks of the embryonic liver and consists of sheets, clusters or single small, angulated, hyperchromatic cells with a high nuclear/cytoplasmic ratio, prominent nucleolus and frequent mitoses. Embryonal cells often cluster into glandular, acinar or pseudorosette formations. EH is often more prominent than the fetal pattern. The macrotrabecular pattern is considered to be a growth pattern rather than a histological subtype. It is seen in about 3% of cases and is composed of trabeculae >5 cells in thickness with either fetal or embryonal type cells ( Fig. 13.24 ). However, the tumour cells may rarely be larger and closely resemble HCC. In most cases, this pattern is mixed with other patterns of HB. The small-cell undifferentiated (SCU) pattern is seen in about 3% of cases and is composed of noncohesive sheets of small cells similar to those of other ‘small blue cell’ paediatric neoplasms. The mitotic rate is often high, but some tumours do not show significant mitoses. Rhabdoid phenotype with eccentric nucleus and eosinophilic cytoplasmic globules can be seen. This pattern has also been called small-cell anaplastic HB. It has now emerged that many of these tumours, especially with rhabdoid morphology, show integrase interactor 1 (INI) loss and are best classified as malignant rhabdoid tumours (MRTs). , A true SCU component is rare and when present comprises <5% in most HBs, often in the centre of embryonal areas. The SCU component should be distinguished from the blastemal component, which is typically composed of ovoid to spindle cells and may be associated with mesenchymal elements like osteoid. Blastemal component can persist after chemotherapy, while the SCU component is rarely seen in post-therapy specimens, and is identified in pretreatment biopsies. The SCU component, even when minor, has been considered to be associated with an adverse outcome, but the significance of an INI-preserved SCU component is currently regarded as being uncertain. Pleomorphic epithelial HB is typically seen in post-therapy resections and comprises markedly pleomorphic, bizarre tumour cells and tumour giant cells.
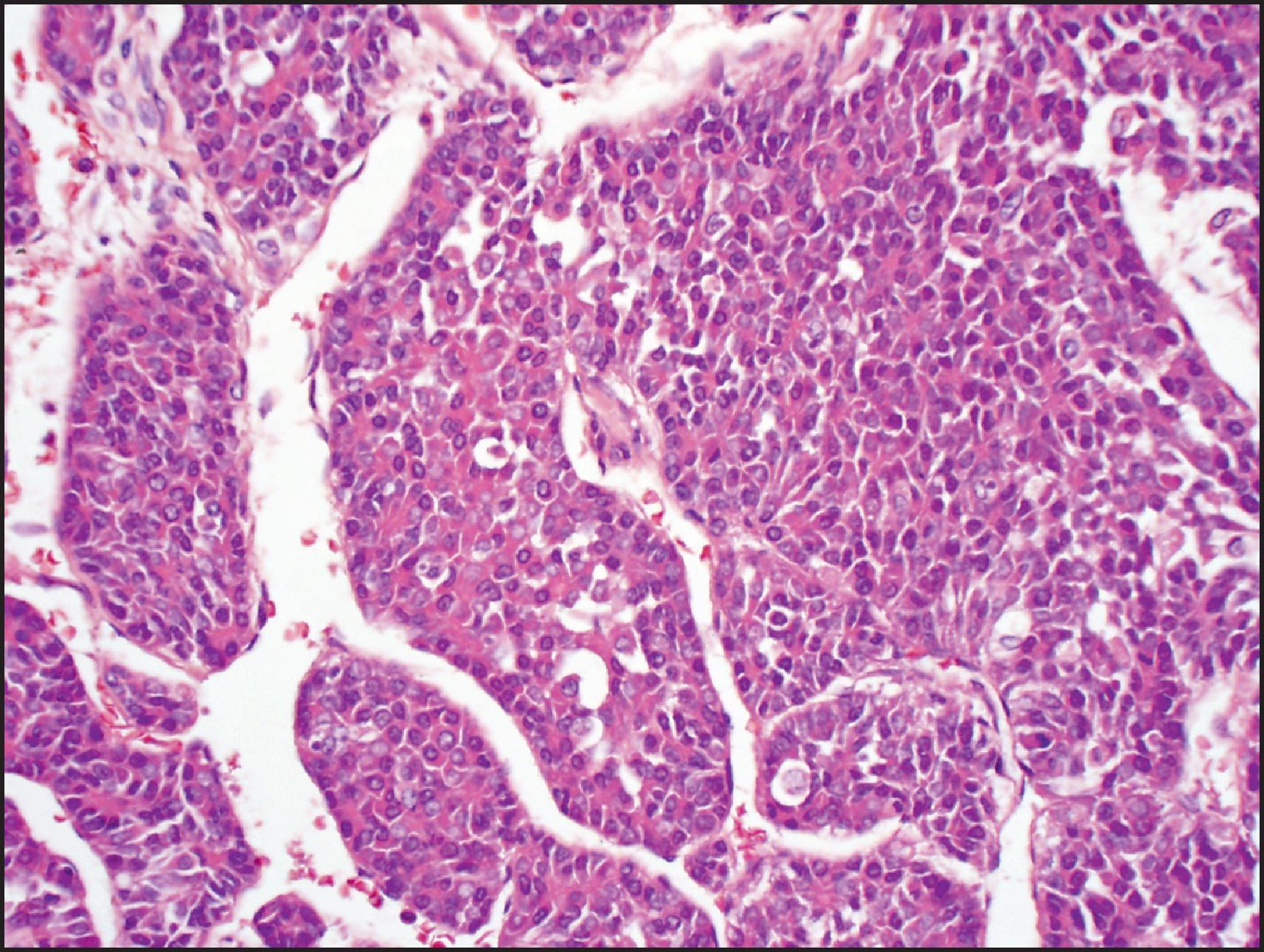
Ductular differentiation can be seen in the epithelial variant as in fetal HB, often at the periphery of the tumour. When ductular differentiation is prominent, the tumour is referred to as cholangioblastic HB. Ductular reaction can occur at the periphery of the tumour, especially after chemotherapy, and should not be considered as the cholangioblastic subtype.
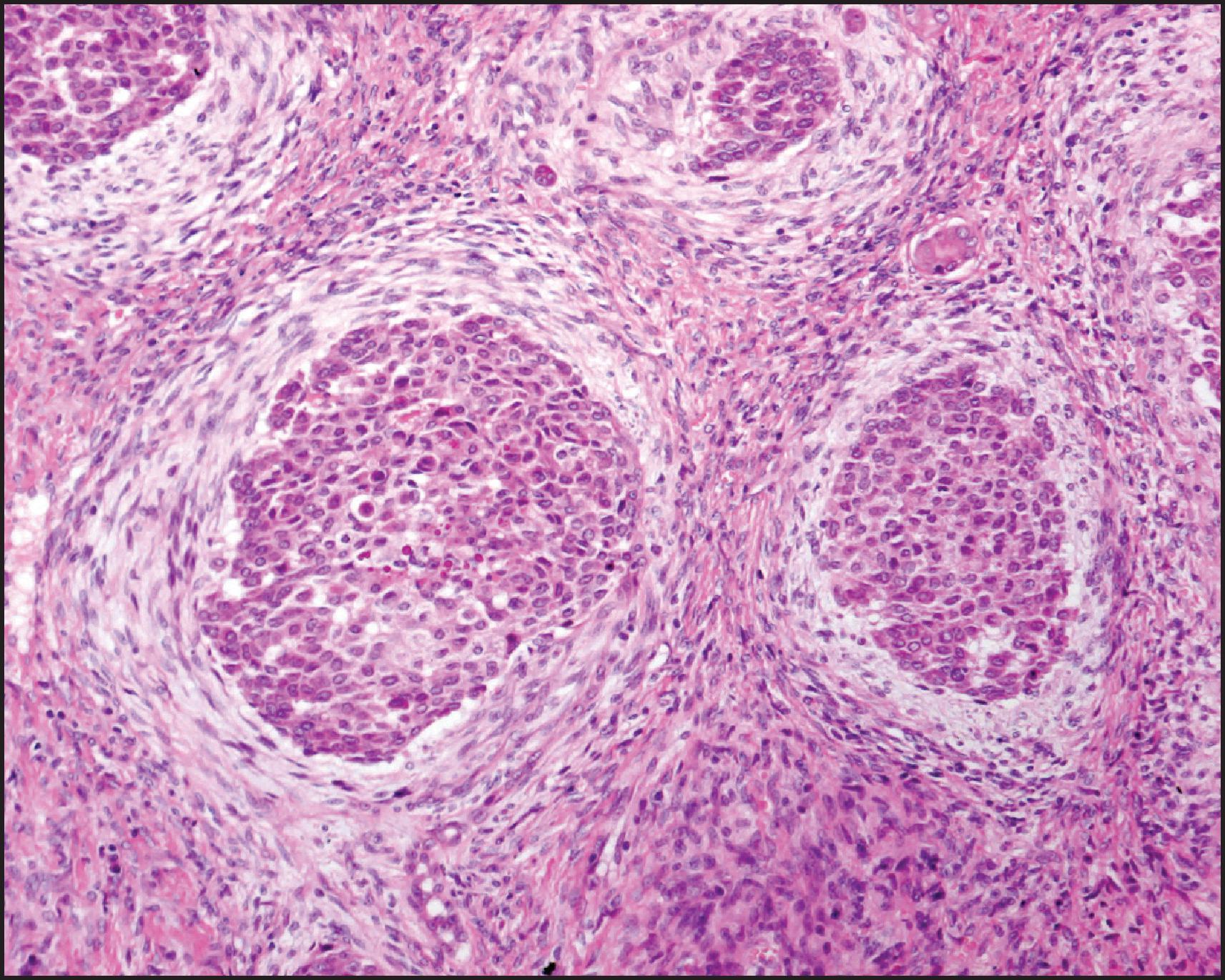
The mixed epithelial-mesenchymal pattern contains areas of fetal and/or embryonal epithelial cells along with neoplastic mesenchymal elements ( Fig. 13.25 ). The mesenchymal component consists of immature and mature fibrous tissue, osteoid-like tissue and, in some cases, hyaline cartilage. The cells in the mesenchyme and osteoid-like areas share staining characteristics with the epithelial component, being positive for keratin and showing nuclear β-catenin staining, indicating that they are metaplastic epithelial cells rather than truly of mesenchymal origin.
Teratoid HB (HB with heterologous elements) refers to mixed HB with heterologous elements such as neural/neuroectodermal differentiation represented by mature brain, primitive neuroepithelial components forming tubules and rosettes, as well as melanin and retinal pigment. Rhabdomyosarcomatous elements can be present. Squamous and mucinous glands may be present, but these elements can also be seen without a neural component in epithelial HB. Rarely, glandular elements with subnuclear and supranuclear vacuoles reminiscent of yolk sac tumour can be present, but these are interspersed with other areas of typical HB. These tumours are not related to teratomas, and representations from all three germ layers typical of teratoma are not seen.
After chemotherapy, HB may show extensive necrosis and haemorrhage with nodules of fibrous tissue, and there may be no residual tumour. Osteoid-like tissue is frequently present and may be the only evidence of viable tumour. , Viable epithelial areas may be seen grossly as pale friable areas within fibrotic nodules, while the mixed elements appear as tiny white areas of osteoid. The amount of residual tumour is variable, and it can be difficult to differentiate fetal pattern from entrapped benign hepatocytes. The histological subtype may be difficult to identify because of secondary changes such as marked fat accumulation and bizarre nuclei. Squamous epithelium with keratinization and foreign body giant cell reaction is often seen in post-therapy specimens.
Both fetal and embryonal elements in HBs express pancytokeratin, and hepatocellular markers such as hepatocyte paraffin 1 (HepPar 1), polyclonal carcinoembryonic antigen (pCEA) and AFP. Nuclear β-catenin is typically diffuse in the embryonal component, patchy nuclear in the crowded fetal pattern and negative or rare positive in the well-differentiated fetal pattern. GS shows diffuse cytoplasmic staining in both embryonal and fetal areas but can be focal or negative in the less differentiated embryonal areas. GPC3 is positive in >90% of cases and is slightly more intense in the eosinophilic cells compared with clear cells in the fetal areas. The staining pattern is diffuse and weak in well-differentiated fetal pattern compared to diffuse and strong in the crowded fetal pattern. SALL4, a marker of germ cell tumours, can be positive in embryonal HB. The ductular elements can be highlighted by keratins 7 and 19. The tumour cells in the SCU component express pancytokeratin, K19 and vimentin and show strong and diffuse nuclear staining with β-catenin. HepPar 1, AFP, GS and GPC3 are uniformly negative. MRTs should be considered if INI1 is lost (see earlier discussion).
FNA cytology findings in epithelial HB depend on whether differentiated (fetal, embryonal and/or macrotrabecular subtypes) or undifferentiated elements are aspirated. Fetal elements resemble immature hepatocytes ( Fig. 13.26 ). Neoplastic cells are slightly smaller and have ample cytoplasm with small, central round nucleus, finely granular chromatin and normal nuclear/cytoplasmic ratio. Pleomorphism and mitoses are not seen. Embryonal subtype is characterized by cells with scant cytoplasm, angulated nucleoli, coarsely granular chromatin, high nuclear/cytoplasmic ratio and mitoses. The cells are usually arranged in acinar groupings, in a papillary pattern or in sheets. EH may be discernible. Differentiated epithelial HB can resemble HCC cytologically. Cytological subtyping can be difficult without resorting to ancillary tests, including ultrastructural studies. The undifferentiated type consists of anaplastic, small round cells and is one of the differential diagnoses of small round cell tumours in childhood. In the mixed epithelial-mesenchymal type, mesenchymal tissue is rarely seen in FNA cytology.
The fetal pattern can be difficult to distinguish from normal infantile liver with EH. The presence of lobular configuration, portal tracts, central veins and a low nuclear/cytoplasmic ratio points indicate normal liver. Unlike HB, normal liver is negative for GPC3 and nuclear β-catenin, while GS is confined to the pericentral region. Alternating light and dark cell zones typical of fetal HB are not seen.
HCC can be mistaken for HB, especially with fetal and macrotrabecular patterns. HCC typically demonstrates more pronounced nuclear pleomorphism, intranuclear inclusions, atypical mitoses and globules of α1-antitrypsin (AAT). The presence of embryonal areas or mesenchymal elements helps in the diagnosis of HB. Compared to HCCs, HBs are more likely to show nuclear β-catenin and diffuse GS staining (70% versus 20%) as well as GPC3 staining (>90% versus <50% in WDHCC). The presence of underlying liver diseases such as tyrosinemia, GSD, progressive familial intrahepatic cholestasis, AAT deficiency and viral hepatitis would favour HCC.
The embryonal and SCU components of HB can mimic metastatic Wilms tumour, and neural tumours such as neuroblastoma or primitive neuroectodermal tumour (PNET). The presence of admixed fetal areas, diffuse GPC3, diffuse GS and nuclear β-catenin staining, along with the absence of a renal mass, favour embryonal HB. Neuroblastoma should be suspected in the presence of an adrenal mass, positive staining for PGP 9.5 and/or synaptophysin and absence of GPC3 and nuclear β-catenin staining. PNETs can rarely present as primary liver tumours; CD99 positivity and fluorescence in situ hybridization (FISH) for the EWS break-apart probe can confirm the diagnosis. MRT can occur in the liver and is characterized by abundant cytoplasm, variably prominent nucleoli and eosinophilic cytoplasmic globules. This is a highly aggressive tumour that is positive for keratin, vimentin and epithelial membrane antigen. Loss of nuclear INI staining due to deletion of the INI1/BAF47 gene on chromosome 22 is a typical feature of MRT. It has been proposed that intact INI should be a criterion for SCU component of HB to avoid mistakenly labelling MRT as SCU HB. These tumours do not respond to conventional HB therapy and are treated as MRT.
Spindle cell tumours such as embryonal rhabdomyosarcoma can enter the differential diagnosis in HB with mesenchymal elements. Involvement of biliary tree rather than the liver parenchyma and staining positivity for desmin, muscle-specific actin and myogenin are typical of embryonal rhabdomyosarcoma. The presence of epithelial areas or other mesenchymal components (e.g. osteoid, bone, cartilage) suggests HB.
Calcifying nested stromal-epithelial tumour of the liver is an extremely rare tumour that is composed of a spindle cell component along with epithelial, ductular and osteoid components; thus, it could be confused with HB ( Fig. 13.27 ) (see also Other tumours and tumour-like lesions, later). This tumour affects children and young adults, some of whom may present with Cushing syndrome. Cytokeratin, vimentin and hormones (e.g. adrenocorticotropic hormone) are often positive, whereas GPC3 and nuclear β-catenin are negative.
HB with teratoid features can mimic a teratoma. The hepatocellular component in teratomas usually resembles mature fetal liver, in contrast to a mix of fetal and embryonal patterns often seen in the epithelial component of mixed HB. It stains with GPC3 in a pattern similar to fetal HB. Typical features of yolk sac tumour, such as reticular growth pattern, marked myxoid stroma and ‘glomeruloid’ elements, are not observed in HB. GPC3 can be positive in the hepatocellular component of teratoma and in yolk sac tumours, but nuclear β-catenin would suggest HB. The teratomas have almost all been in infants, and the yolk sac tumours have been described in both children and adults. ,
This tumour shows overlapping features of HB and HCC (formerly called transitional liver cell tumour) and typically occurs in older children (often >8 years of age). , Molecular studies have shown genetic characteristics of both HB and HCC, characterized by a high number of somatic mutations, including genes such as CTTNB1 , TP53 and TERT promoter. Serum AFP is typically high. These tumours have morphological features intermediate between hepatoblasts and hepatocytes. Unlike the trabecular pattern seen in HCC, the tumour cells exhibit a diffuse pattern and can have a wide spectrum of appearances ranging from small to hepatoid cell. Multinucleated tumour giant cells can be present. Mixed areas resembling HB and HCC can be present. As with HB, nuclear staining with β-catenin is present. This is a considered a provisional category and treated as high-risk HB.
The most widely used staging systems are those of the Children’s Oncology Group (COG) and PRETEXT schemes. The COG scheme classifies the tumours postoperatively by their resectability into four groups: stage I (complete resection), stage II (microscopic residual disease), stage III (macroscopic residual tumour) and stage IV (distant metastases). At presentation, approximately 38% of HBs are stage I, 9% are stage II, 24% are stage III and 29% are stage IV. , The majority of the 53% of unresectable (stages III and IV) cases can be rendered resectable with preoperative chemotherapy. Liver transplantation (LT) is an option for unresectable disease.
Developed by the International Childhood Liver Tumor Strategy Group (SIOPEL), the PRETEXT staging system is based on segmental involvement of the liver and is used to determine tumour extension before therapy , : stage I (one section involved, three adjoining sections are free), stage II (one or two sections involved, two adjoining sections are free), stage III (two or three sections involved, no two adjoining sections are free) and stage IV (all four sections involved). The sections in the PRETEXT staging are defined by grouping of liver segments: segments II and III (left lateral section), segment IV (left medial section), segments V and VIII (right anterior section) and segments VI and VII (right posterior section). In addition, the PRETEXT system also includes assessment of tumour involvement of inferior vena cava (IVC), hepatic veins (V), portal veins (P), caudate lobe, multifocality, rupture/intraperitoneal haemorrhage, lymph node involvement, extrahepatic abdominal disease (E) and distant metastases (M).
Surgery is the mainstay of treatment of HB, and complete resection is the only chance for cure. Low AFP level, vascular invasion and positive resection margin have been associated with poor outcome. All patients with COG stage I tumours with pure well-differentiated fetal pattern are treated with surgery alone without chemotherapy because 5-year survival is almost 100%. The other histological variants as well as higher-stage tumours receive a combination of chemotherapy and surgical resection, the order determined by tumour resectability at presentation. For nonpure fetal histology, the disease-free survival is 97–100% for COG stage I and II, 70% for stage III and 40% for stage IV disease. Preoperative chemotherapy to shrink the tumour has resulted in improved resectability and improved the overall 5-year survival rate to 75% from 35% in the 1970s. Cisplatin/doxorubicin or cisplatin/5-FU/vincristine are the usual regimens. LT is an option for tumours that remain unresectable after chemotherapy.
The stage of the tumour at initial resection is the key prognostic factor in determining survival. , Small-cell and macrotrabecular subtypes have an adverse outcome. , The other histological patterns do not independently affect survival when adjusted for age, gender and stage. Other unfavourable prognostic factors include the involvement of multiple lobes, vascular invasion, slow decline in AFP after therapy, aneuploidy, nuclear β-catenin staining, low p27 expression and high cyclin D1 expression.
Large-cell change (originally ‘large-cell dysplasia’) refers to cellular enlargement with bizarre pleomorphic nuclei, hyperchromasia, prominent nucleoli and occasional multinucleation of hepatocytes ( Fig. 13.28 ). The cells have abundant cytoplasm, and thus the nuclear/cytoplasmic ratio is normal. This change can affect isolated or groups of hepatocytes and may occupy the entire cirrhotic nodule. Large-cell change is found in 20–30% of cirrhotic livers and in 60–70% of cirrhotic livers with HCC; it has been thought to convey an increased risk for development of HCC. However, hepatocytes with large-cell change have a low proliferation rate, greater apoptosis and increased markers of senescence, suggesting a derangement of the process of hepatocyte polyploidy, possibly caused by chronic inflammation-induced DNA damage , or cholestasis. Other studies have found alterations in cell cycle, DNA damage and telomere shortening in large-cell change in hepatitis B cirrhosis, suggesting that large-cell change is a heterogeneous lesion and may be a precursor of HCC in certain patients.

Small-cell change (formerly ‘small-cell dysplasia’) is characterized by small hepatocytes with increased nuclear/cytoplasmic ratio, typically with cytoplasmic basophilia ( Fig. 13.29 ). When occurring in expansile nodules, the hepatocytes in small-cell change often have increased proliferative activity and molecular abnormalities, such as p53 overexpression, and are likely to be a precursor of HCC. In other patients, poorly defined or diffuse areas of small-cell change without nodular configuration may represent regenerative phenomenon or even ischaemic/degenerative change and are not preneoplastic. Small-cell regenerative foci are also common in chronic biliary disease.
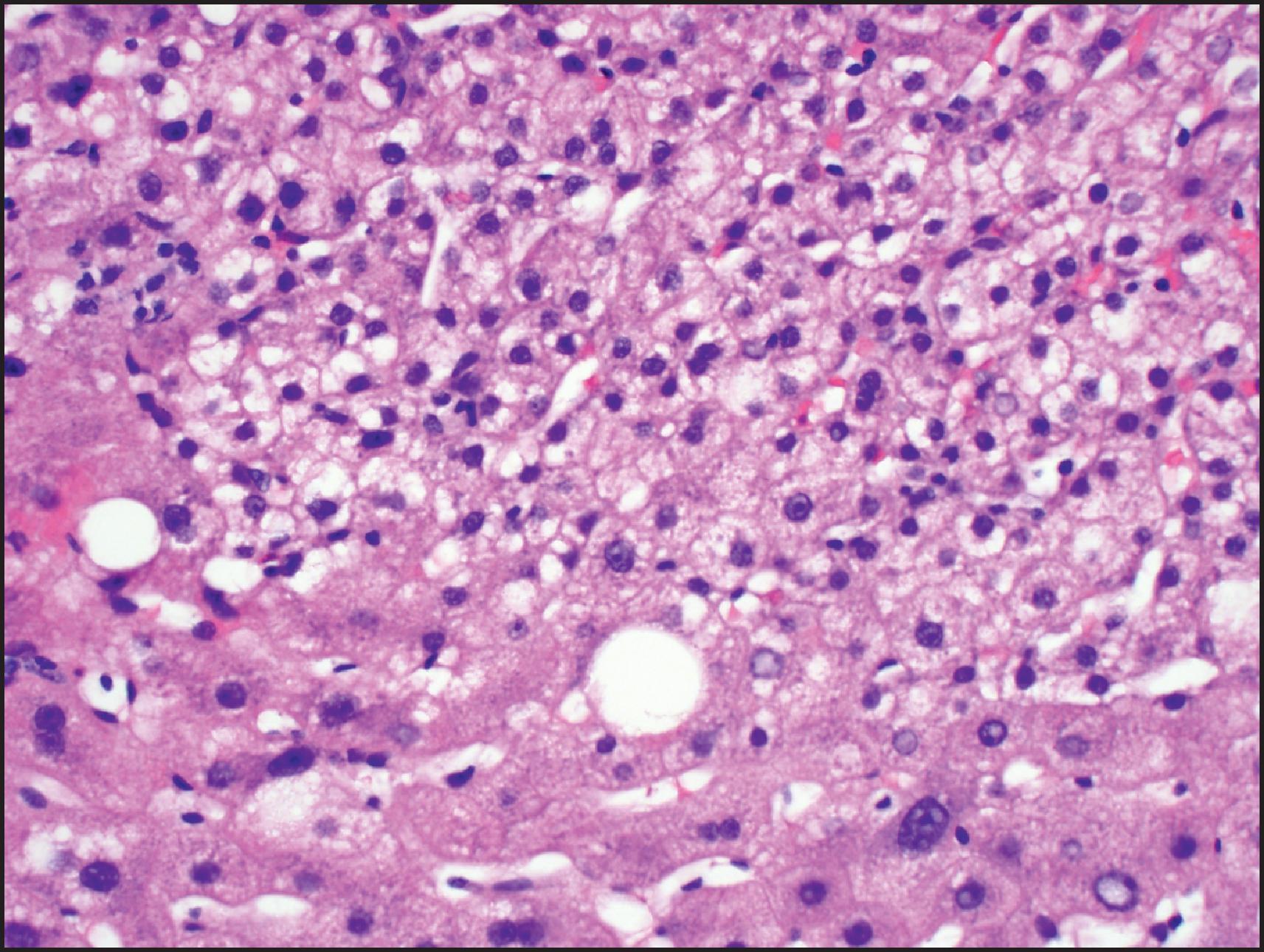
The term ‘dysplasia’ refers to abnormal hepatocellular proliferation that does not fulfil the criteria of malignancy. A cluster of dysplastic hepatocytes <1 cm in diameter is referred to as dysplastic focus (see Fig. 13.29 ), whereas a cluster of dysplastic hepatocytes ≥1 cm in diameter is referred to as dysplastic nodule .
Large regenerative nodules (LRNs), also known as macroregenerative nodules and adenomatous hyperplasia , are by definition ≥1 cm. These typically arise in cirrhosis secondary to hepatitis ( Table 13.6 ) and are rare in biliary disease ( Fig. 13.30 ). LRNs can also occur in noncirrhotic liver in Budd–Chiari syndrome and portal vein thrombosis and frequently occur as regenerative nodules after necrosis, including necrosis from active and/or acute to subacute hepatitis, and in this setting, these nodules are not thought to be preneoplastic. On histology, LRN/macroregenerative nodules are usually composed of hepatocytes with normal or near-normal cytology, with plates one to two cells thick, although thicker plates can be present, particularly in the setting of regeneration following necrosis. Importantly, intact reticulin framework is present, as may areas of large-cell change ( Fig. 13.31 ). Ductular reaction may be prominent (see Fig. 13.30 ), and unpaired arterioles are rare or absent.
| Type of nodule | Cellular features | Reticulin framework | Other key features |
|---|---|---|---|
| LRN/MRN | Bland cytology, similar to normal liver or cirrhotic nodules with focal LCC | Intact reticulin framework similar to cirrhosis | Portal or portal-like zones; scattered LCC; iron, Mallory–Denk bodies, fat and bile may be present |
| LGDN | Bland cytology of uniform pattern | Similar to MRN | Similar to MRN |
| HGDN | Small-cell change, common (nuclear density <2× normal) * | Mostly intact reticulin, but focal fragmentation/loss may be seen; plate width more variable and pseudo-acinar change more likely; few unpaired arterioles | No stromal invasion; CD34 typically not diffuse |
| WDHCC | Small-cell change most common (nuclear density >2× normal) | Multifocal loss, fragmentation or extensive irregularity of reticulin framework; thin trabeculae; pseudoacinar change and unpaired arterioles can be prominent | Early HCC often shows fatty change and lacks ductular reaction at periphery; stromal invasion; diffuse CD34, diffuse GS, positive glypican-3 and/or positive HSP70 favour HCC |
* Nuclear density defined as number of nuclei, or nuclear crowding, as seen on lower magnification compared to normal liver. GS , Glutamine synthetase; HCC , hepatocellular carcinoma; HGDN , high-grade dysplastic nodule; LCC , large-cell change; LGDN , low-grade dysplastic nodule; LRN , large regenerative nodule; MRN , macroregenerative nodule; WDHCC , well-differentiated hepatocellular carcinoma.
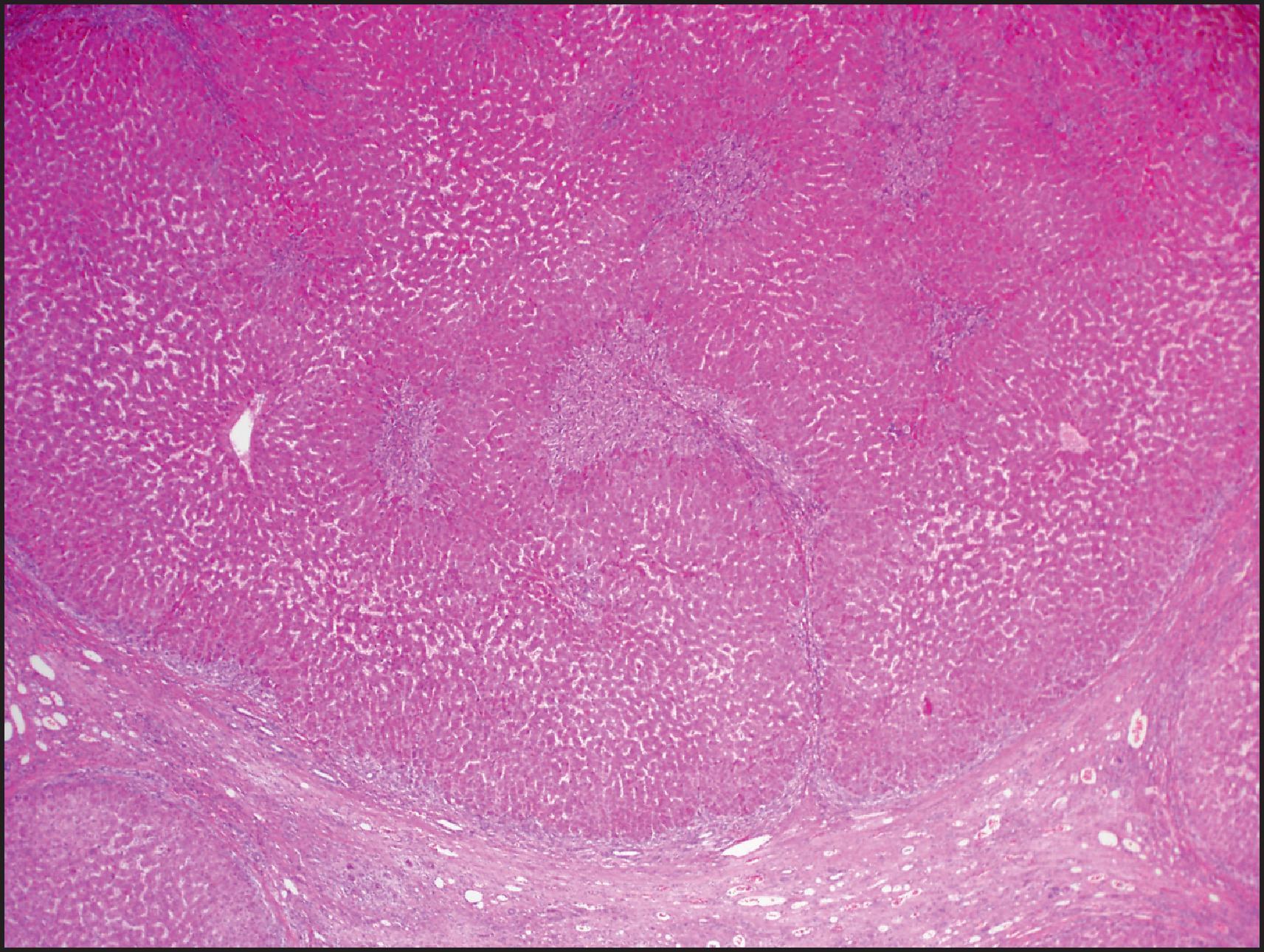
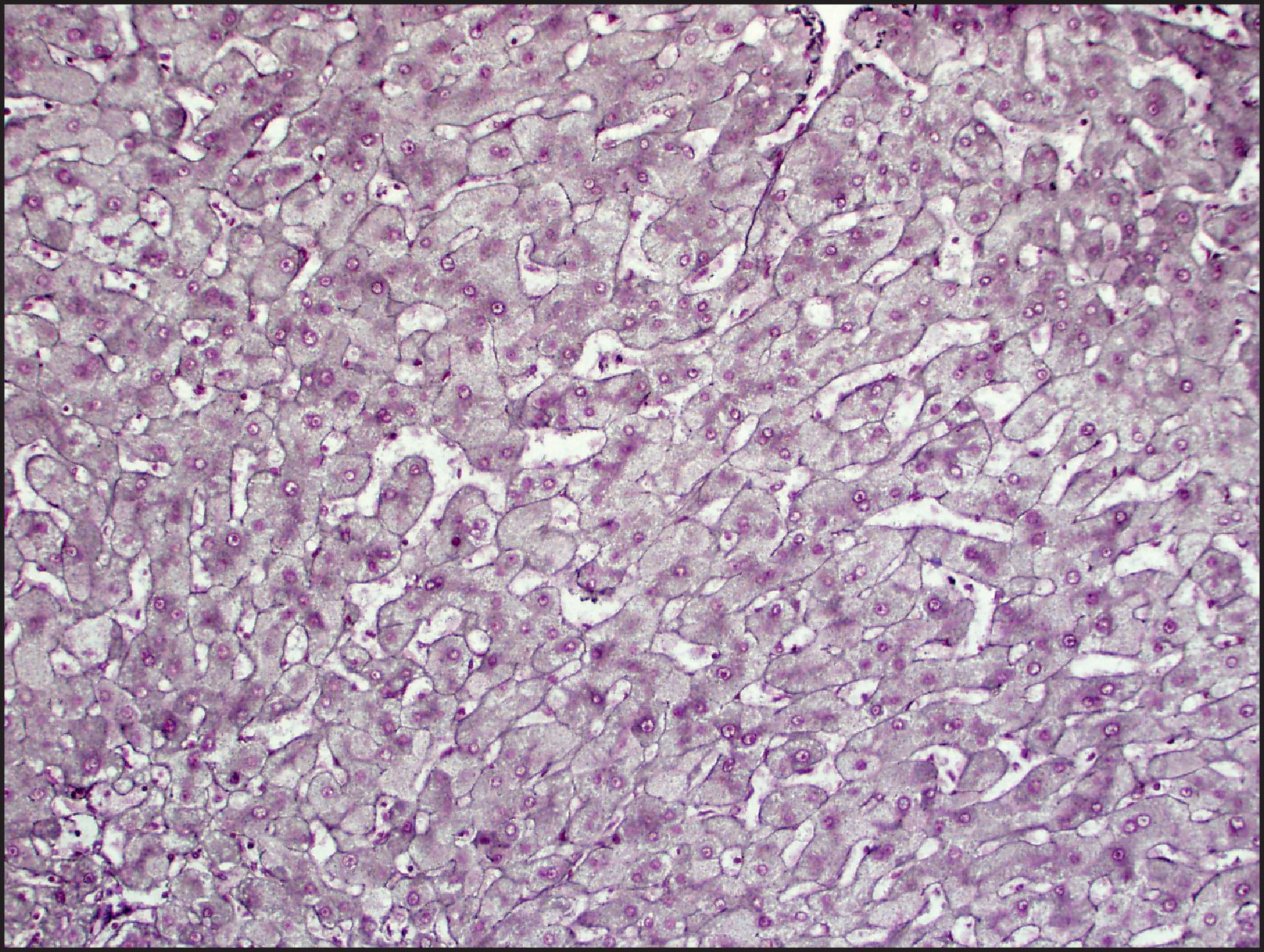
Low-grade dysplastic nodules (LGDNs) represent a clonal hepatocellular proliferation (a uniform population of hepatocytes) without features of high-grade dysplasia. , LGDNs can be histologically indistinguishable from macroregenerative nodules (LRNs) in the absence of clonality studies ( Table 13.6 ). Both LRN and LGDN in cirrhosis may stand out from the background cirrhotic nodules by the presence of MDBs, bile, clear cell change, iron, copper or fat. In part, given that the histological diagnostic criteria overlap those of macroregenerative nodule, its likelihood of progression to HCC is not clearly defined.
High-grade dysplastic nodules (HGDNs) are characterized by hepatocellular proliferation with atypical cytological and/or architectural features that are not sufficient for an unequivocal diagnosis of HCC ( Table 13.6 ). By definition, stromal invasion is not seen. HGDNs typically show small-cell change and features that suggest increased cellular proliferation, such as plates more than two cells thick, cytoplasmic basophilia, high nuclear/cytoplasmic ratio, nuclear hyperchromasia or an irregular nuclear contour ( Fig. 13.32 ). These features may involve the entire nodules or may be confined to one or more foci within the nodule, giving the appearance of ‘nodule-in-nodule’ formation. Portal tracts are present within the lesion. Occasional unpaired arterioles, focal pseudoglandular/acinar architecture and focal reticulin loss can be seen, but these findings are not prominent. Steatosis and MDBs can be present, while iron is typically absent. IHC for CD34 shows patchy sinusoidal staining, usually at the edge; occasional nodules can show more diffuse expression. AFP is negative, and GPC3 expression is variable but typically absent or focal. HGDNs are generally considered high-risk lesions for HCC.
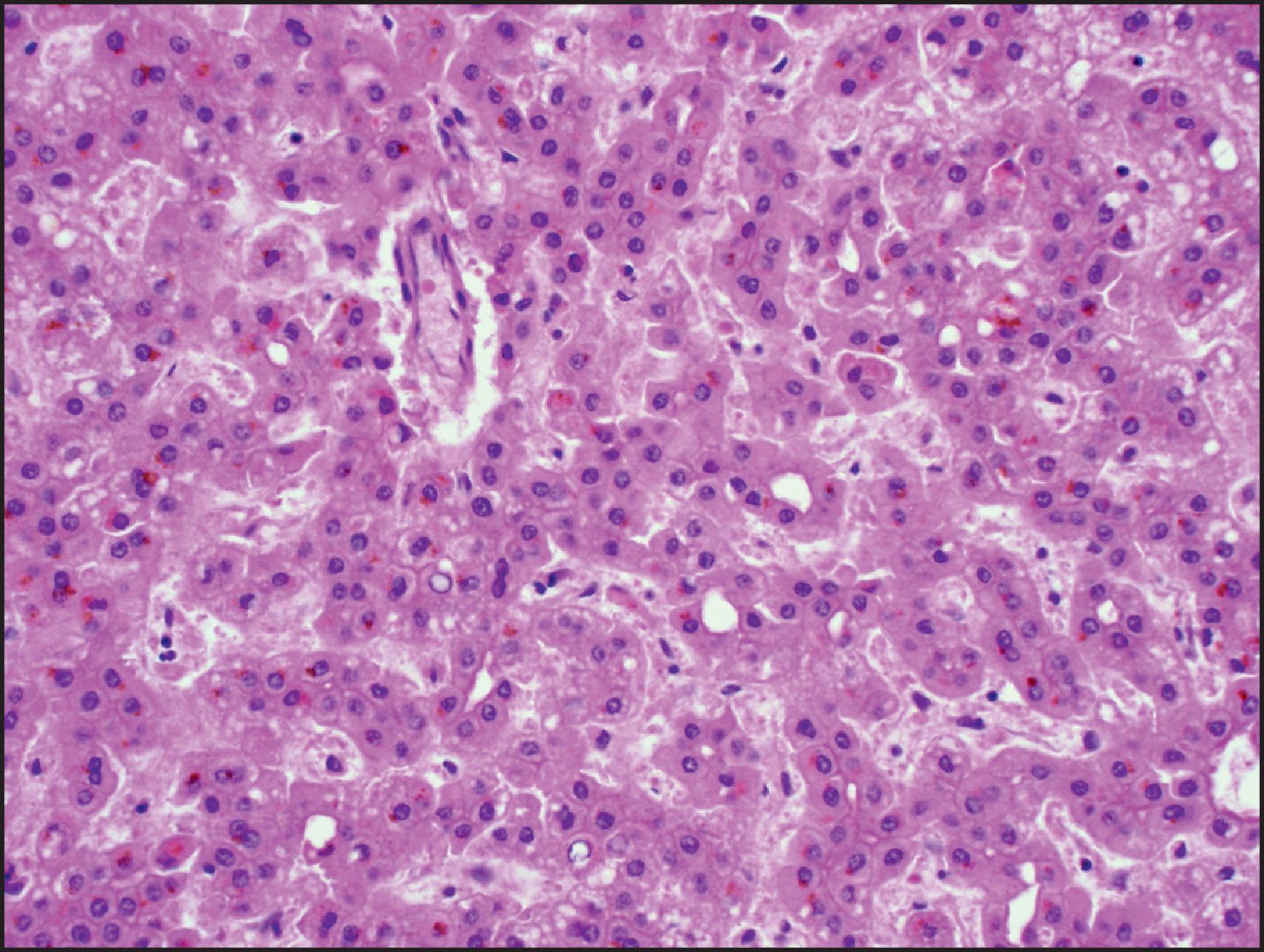
Fine-needle aspirates of dysplastic nodules are composed of a polymorphous cell population comprising hepatocytes occurring loosely or in one- to two-cell-thick cords, bile duct epithelium and ductular clusters. LGDNs contain hepatocytes often exhibiting large-cell change characterized by nuclear and cellular enlargement with normal nuclear/cytoplasmic ratio but minimal nuclear atypia ( Fig. 13.33 ). In contrast, HGDNs contain hepatocytes showing small-cell change consisting of small and monotonous cells with minimal nuclear atypia, a subtle increase in nuclear/cytoplasmic ratio and nuclear crowding ( Fig. 13.34 ). Distinguishing LGDN from LRN and HGDN from small early HCC can be challenging. ,
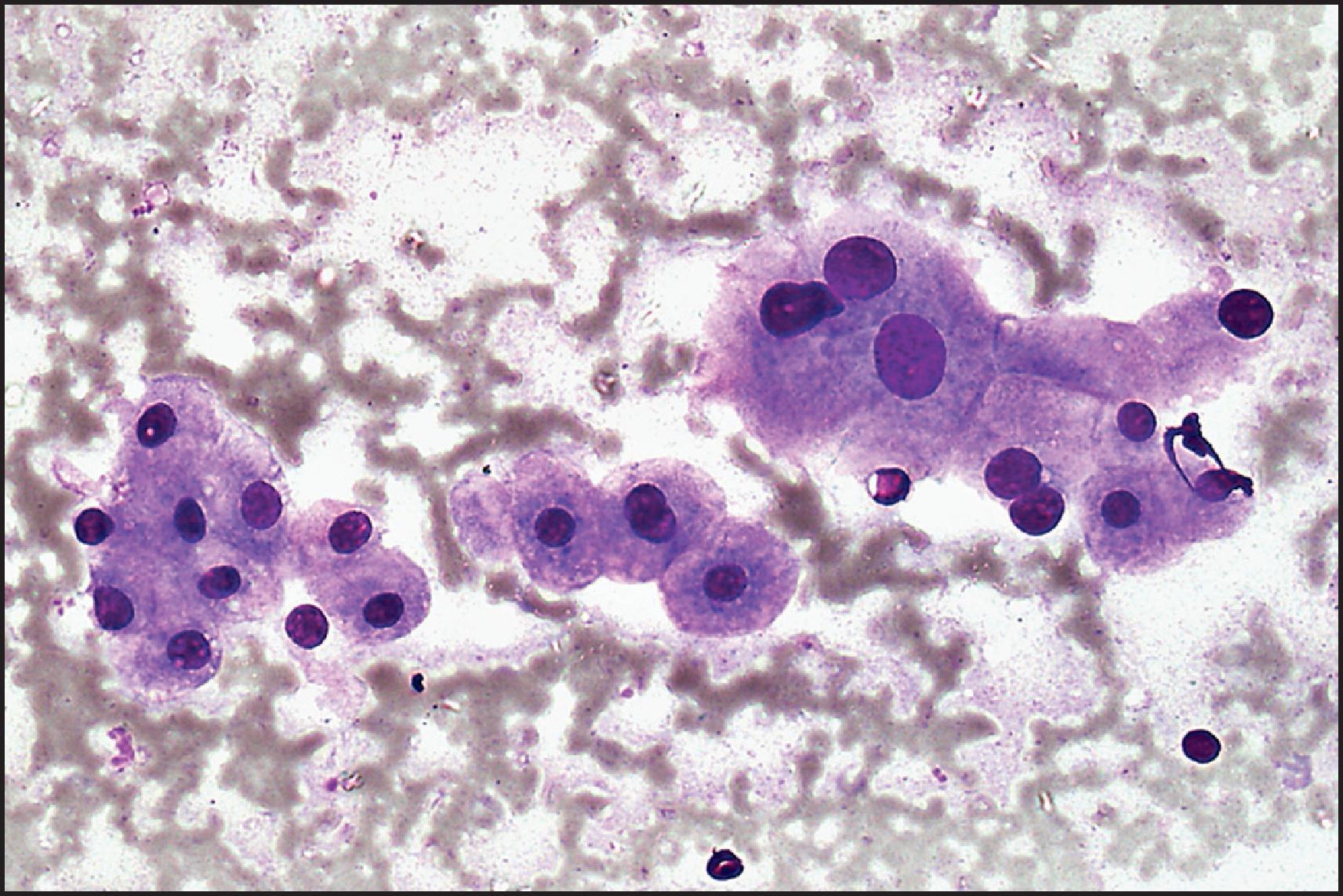
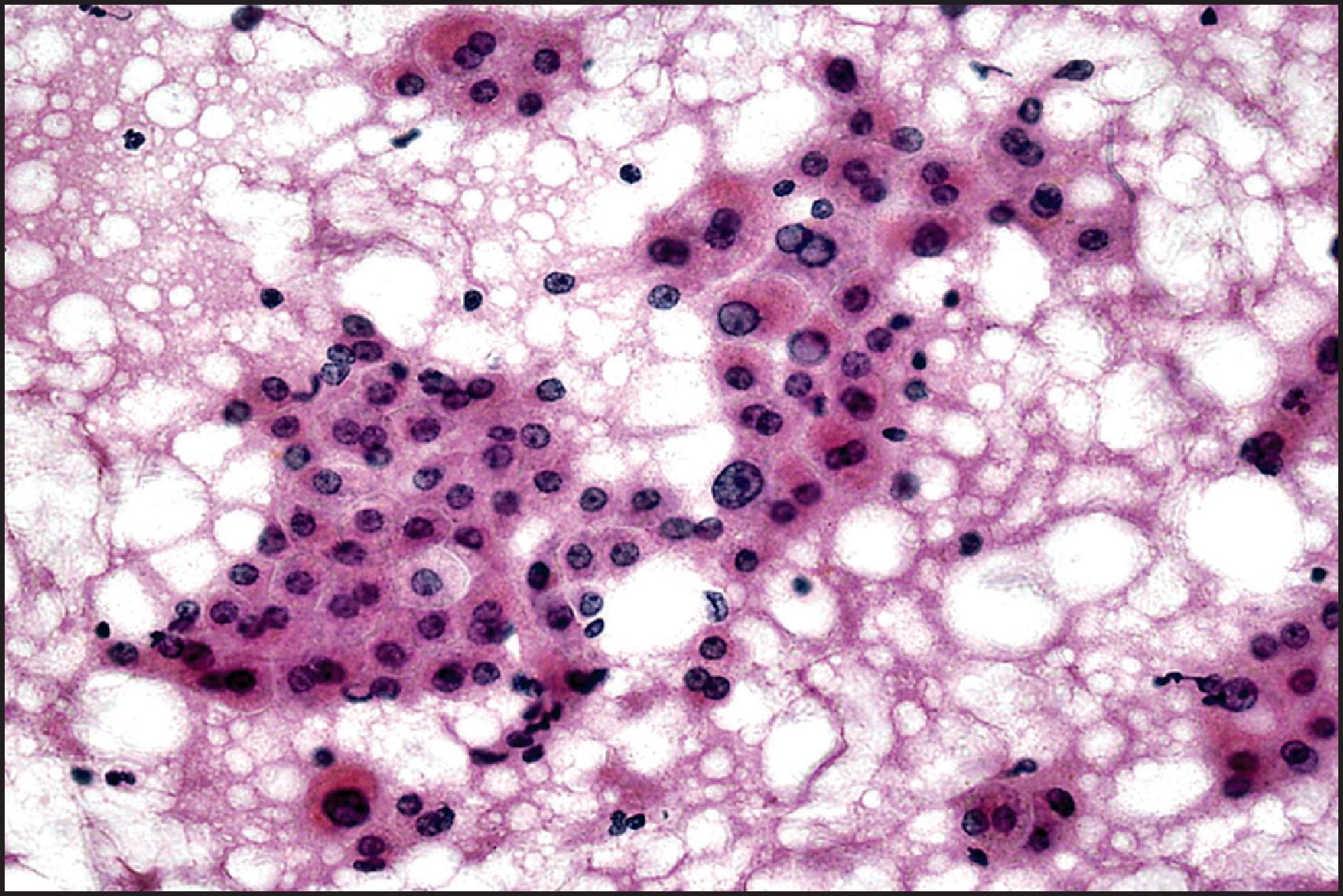
Small HCC refers to tumours up to 2 cm in diameter; these have a greater likelihood of long-term survival after surgery or other attempts at curative therapy. Similar to large regenerative and dysplastic nodules, small HCCs are larger than the surrounding cirrhotic nodules and may differ in colour (frequently green, yellow or mottled), or they may bulge from the cut surface. Small nodules are easily overlooked in explanted livers unless the specimen is thinly sectioned (at least 1 cm intervals) and examined in detail. Small HCCs can be divided into two subtypes: progressed and early HCC (early WDHCC, HCC with vague nodular pattern, HCC with indistinct borders). Progressed HCC has a capsule and histologically resembles classic HCC, whereas early HCC lacks a definite capsule and histologically can resemble HGDN. Early HCCs constitute 15–30% of small HCCs. , Remnants of pre-existing early HCC in association with less well-differentiated HCC were reported in 74% of tumours <1 cm but in only 9% of tumours that were 4–5 cm, suggesting stepwise dedifferentiation as the mechanism of disease progression.
The differentiation of HGDN from early HCC is extremely difficult and may be impossible on a biopsy. This distinction has clinical implications, since patients with a definite diagnosis of HCC may be considered for resection or may receive priority for transplantation. On the other hand, most HGDNs are ablated, and these patients do not have priority for transplantation. The features that favour progressed HCC, such as uniformly thick cell plates, prominent pseudogland formation, multifocal loss of the reticulin framework and vascular invasion, are often absent in early HCC ( Table 13.6 ). Increased cellularity, increased nuclear/cytoplasmic ratio and frequent pseudogland formation and fatty change are more common in early HCC than in HGDN, but these are not reliable for definite distinction in many cases. The feature that can best distinguish early HCC from HGDN is stromal invasion ( Fig. 13.35 ), which is characterized by the invasion of abnormal hepatocytes into perinodular portal tracts, fibrous septa or adjacent parenchyma. , Stromal invasion can also be intranodular, with invasion of portal tracts within the nodule. Stromal invasion is diagnostic of HCC but can be difficult to assess or sample on core biopsies.
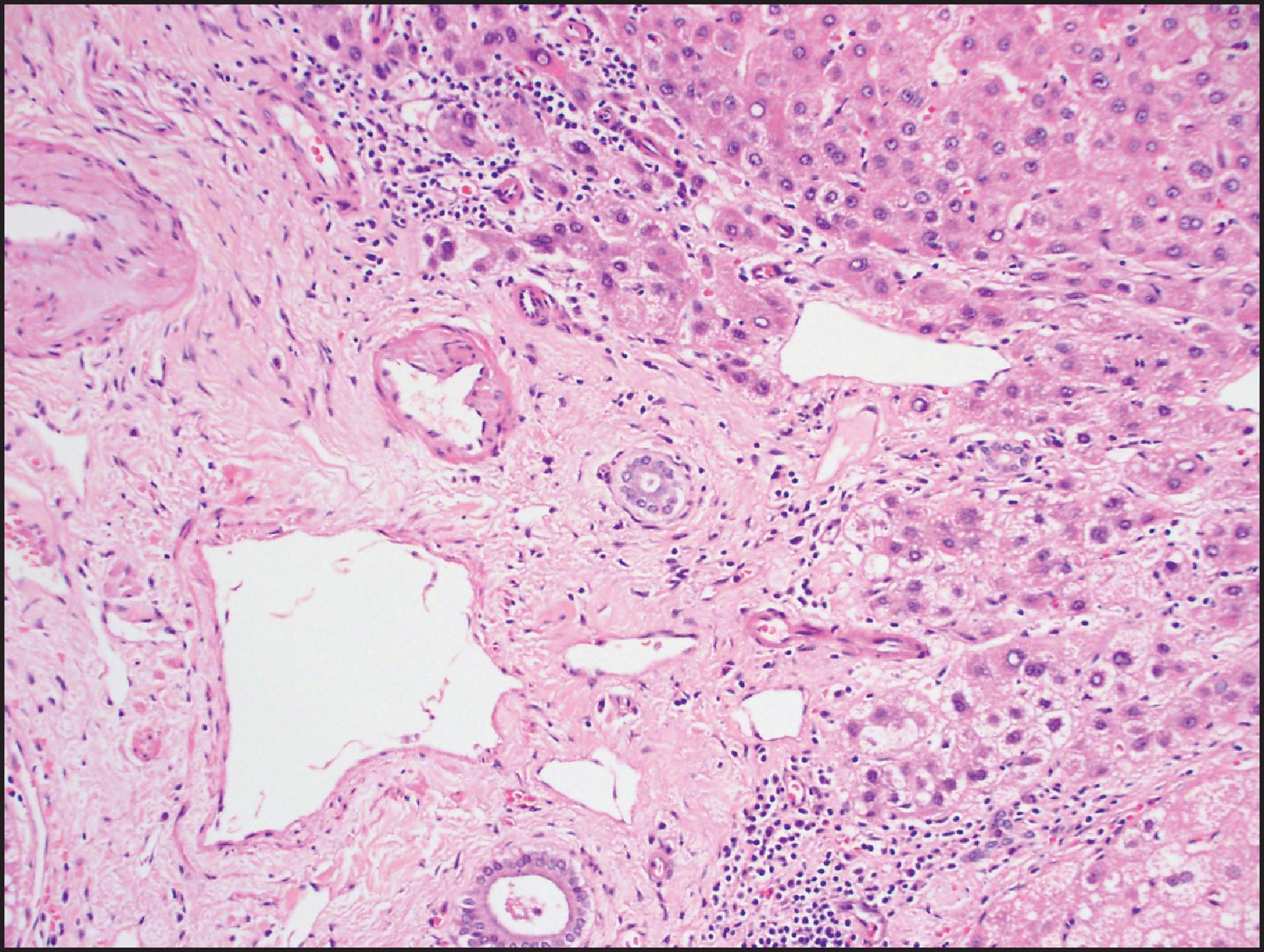
Ancillary techniques that favour HCC include diffuse sinusoidal staining with CD34, nuclear staining with β-catenin, diffuse staining with GS, HSP70 overexpression and GPC3 positivity. Combined use of HSP70, GPC3 and GS has been recommended for this differential diagnosis, with high specificity for diagnosis of HCC when two of the three markers are positive. , However, HSP70 and GPC3 have low sensitivity and are not helpful in most cases. Areas of stromal invasion often lack ductular reaction at the interface between hepatocytes and portal tracts/fibrous septa, whereas this feature is present in LRN and most HGDNs. Use of IHC for keratins 7 and 19 can be useful in the identification of stromal invasion by highlighting lack of ductular reaction.
Other, less common nodules can occur in cirrhotic liver. Lesions resembling FNH can occur in the cirrhotic liver and have been referred to as FNH-like lesions . In addition to typical morphological features of FNH, some of these lesions (but not all) show map-like staining with GS (see Table 13.5 ). In rare instances, nodules in cirrhosis show typical features of IHCA (see earlier), such as sinusoidal dilation, inflammation, dystrophic arterioles and strong staining with SAA/CRP. , Mutations typical of IHCA may be present.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here