Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
In this chapter, peritoneal lesions are reviewed under the following headings: tumorlike lesions, mesothelial neoplasms, miscellaneous primary tumors, metastatic tumors, and lesions derived from the so-called secondary Müllerian system. Disorders of the retroperitoneum, except those of peritoneal origin, are not included.
Mesothelial hyperplasia is a common response to acute and chronic inflammation and chronic effusions ( Figs. 15.1, 15.2, 15.3, and 15.4 ). Hyperplastic lesions may be observed at operation as solitary or multiple, typically small nodules or plaques, but more commonly are incidental findings on microscopic examination. Mesothelial hyperplasia often occurs in the adnexa of women with chronic salpingitis and endometriosis and is often encountered in these and other areas (particularly the omentum) in patients with ovarian neoplasms. Additionally, mesothelial hyperplasia on the surface of the ovary or within the superficial ovarian stroma overlying a borderline epithelial tumor may be misinterpreted as invasive tumor ( Fig. 15.5 ). Mesothelial hyperplasia may also involve a hernia sac, in some cases as a result of trauma or incarceration (see Fig. 15.2 ).
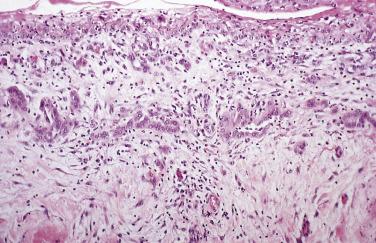
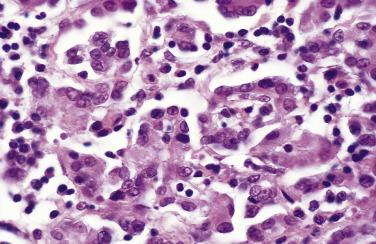
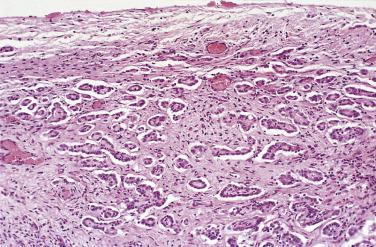
In florid examples of mesothelial hyperplasia, solid, trabecular, tubular, papillary, or tubulopapillary patterns (see Figs. 15.1 through 15.5 ) may occur, in some cases accompanied by superficial extension of the mesothelial cells into the underlying tissues (see Figs. 15.3 and 15.5 ). The cells focally may be arranged in linear, occasionally parallel, thin layers, separated by fibrin or fibrous tissue (see Fig. 15.3 ). The mesothelial cells may contain cytoplasmic vacuoles filled with acid mucin (predominantly hyaluronic acid) or, less commonly, may exhibit marked clearing of their cytoplasm. Mild to moderate nuclear atypia, mitotic figures, and, occasionally, multinucleate cells may be present. Psammoma bodies and, in rare cases, eosinophilic strap-shaped cells resembling rhabdomyoblasts have been encountered. In some cases of mesothelial hyperplasia, variable numbers of histiocytes may be admixed with the mesothelial cells (nodular histiocytic/mesothelial hyperplasia), and, in a subset, histiocytes are the dominant component ( Fig. 15.6 ). The distinction of these two types of cells, although unlikely to be clinically important, may require immunohistochemistry.
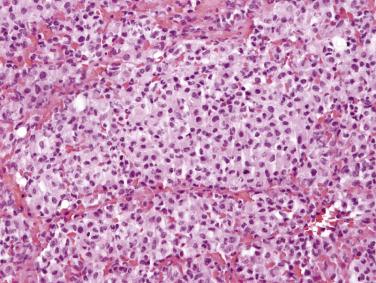
Hyperplastic mesothelial cells may rarely involve intraabdominal lymph nodes, a finding that has been associated with, and in such cases is presumably secondary to, mesothelial hyperplasia of the peritoneum. The hyperplastic mesothelial cells can prominently expand the subcapsular lymphatics and the intranodal sinusoids ( Fig. 15.7 ), potentially mimicking sinus histiocytosis, nodal involvement by an ovarian serous borderline tumor, metastatic carcinoma, or metastatic mesothelioma, a pitfall that can be exacerbated if the mesothelial cells exhibit superimposed deciduoid change (“deciduoid mesothelial hyperplasia”). Cytokeratin staining in such cases may reveal more extensive involvement of the lymph node by the mesothelial cells than is appreciable in routinely stained sections.
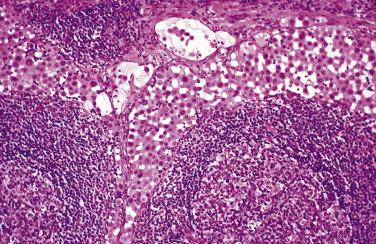
The major differential diagnosis of mesothelial hyperplasia is malignant mesothelioma. The presence of grossly visible nodules, necrosis, conspicuous large cytoplasmic vacuoles, severe nuclear atypia, and deep infiltration favors mesothelioma over mesothelial hyperplasia. Some of these features, however, are not always present or may be present only focally within a mesothelioma. On occasion, the distinction between a hyperplastic mesothelial lesion and a mesothelioma may be difficult or impossible, especially in a small biopsy or cytology specimen. Immunoreactivity for epithelial membrane antigen (EMA), p53, insulin-like growth factor 2 messenger RNA–binding protein (IMP)–3, glucose transporter (GLUT)–1, XIAP, and high expression of EZH2 is more typical of malignant mesothelioma, and desmin immunoreactivity is more typical of mesothelial hyperplasia, but these markers cannot be relied on in individual cases, Homozygous deletion of 9p21 and p16 by fluorescent in situ hybridization (FISH), especially in combination with loss of BAP1 immunoreactivity, is highly characteristic of malignant mesothelioma, whereas these alterations have not been observed in reactive mesothelial proliferations. In addition to mesothelioma, the differential diagnosis of peritoneal or intranodal mesothelial hyperplasia includes serous borderline tumors in the form of spread from an ovarian serous borderline tumor or a primary peritoneal or intranodal serous borderline tumor (see Peritoneal Serous Lesions ). Grossly visible ovarian or peritoneal tumor nodules, columnar cells that may bear cilia, the presence of intracellular or extracellular neutral mucin, and numerous psammoma bodies favor a serous borderline tumor. Immunoreactivity for epithelial antigens (see later section on malignant mesothelioma ) is also useful in the differential diagnosis.
Peritoneal inclusion cysts usually occur in women of reproductive age, but may develop over a wide age range, and the same lesions occur infrequently in males or involve the pleura. Some cysts are discovered incidentally at laparotomy as single or multiple, small, thin-walled, translucent, unilocular cysts attached or lying free in the peritoneal cavity ( Fig. 15.8 ). They occasionally involve the round ligament, potentially mimicking an inguinal hernia. Some cysts are an incidental serosal finding in appendectomy specimens. Rare cases have been retroperitoneal. Pathologic examination of these cysts typically reveals a smooth lining, yellow and watery to gelatinous contents, and a single layer of flattened, benign-appearing mesothelial cells. Mature stratified squamous epithelium may be seen in some cases. Most unilocular mesothelial cysts are probably reactive in origin, although developmental origin has been suggested for at least some of those cases involving the mesocolon, mesentery of the small intestine, retroperitoneum, and splenic capsule.
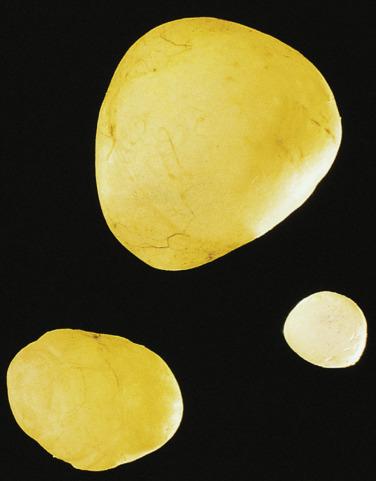
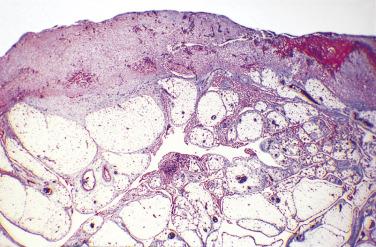
Multilocular cystic masses as large as 20 cm in diameter ( Fig. 15.9 ) and lined by mesothelial cells have been referred to as multilocular peritoneal inclusion cysts (MPICs), benign cystic mesotheliomas, inflammatory cysts of the peritoneum, or postoperative peritoneal cysts. MPICs often cause clinical manifestations—usually lower abdominal pain, a palpable mass, or both. Typically they are adherent to pelvic viscera and may mimic a cystic ovarian tumor clinically, intraoperatively, or on pathologic examination; the upper peritoneal cavity, the spleen, the liver, the retroperitoneum, the inguinal region, and hernia sacs also can be involved. The septa and walls of MPICs may contain considerable amounts of fibrous tissue, and their contents may resemble those of the unilocular cysts or they may be serosanguineous or frankly bloody.
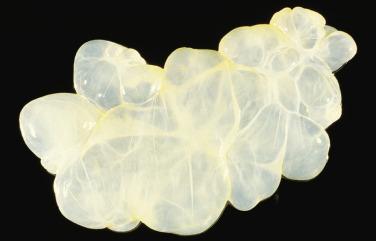
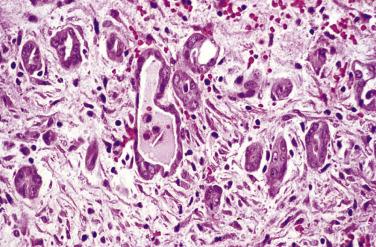
Microscopic examination reveals that MPICs are usually lined by a single layer of flat to cuboidal, rarely hobnail-shaped, mesothelial cells that typically have benign nuclear features, although some reactive atypia is not uncommon ( Figs. 15.10 and 15.11 ). The mesothelial cells may proliferate into the cyst lumina as small papillae or in cribriform patterns, and occasionally they may undergo squamous metaplasia ( Fig. 15.12 ). Mural proliferations of typical or atypical mesothelial cells disposed singly, as glands or nests ( Fig. 15.13 ), or in patterns mimicking an adenomatoid tumor, may occur, resulting in infiltrative patterns that may suggest a cancer. In some cases, vacuolated mesothelial cells in the stroma may raise the possibility of a signet ring carcinoma. The cyst walls and septa are usually composed of fibrovascular connective tissue with a sparse inflammatory infiltrate, but in other cases, severe acute and chronic inflammation (that may include foamy histiocytes), fibrin, granulation tissue, and hemorrhage are encountered.
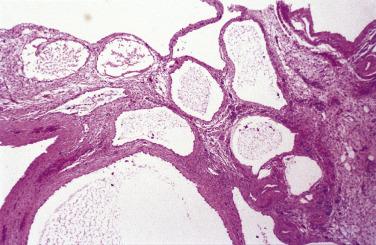
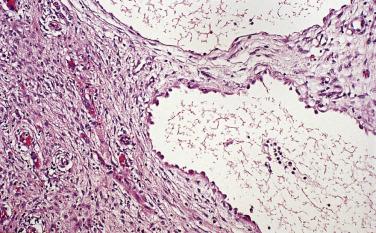
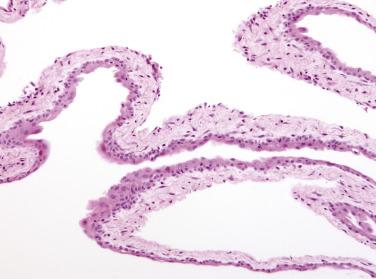
A history of one or more factors—an abdominal surgical procedure (most common), endometriosis, pelvic inflammatory disease, inflammatory bowel disease, radiotherapy, or abdominal trauma—was present in 70% and 84% of patients in two studies, indicating a likely role for inflammation in the pathogenesis of MPICs. The occurrence of cases in which the distinction between florid adhesions and an MPIC may be difficult is also consistent with an inflammatory cause. Only rare cases have been associated with asbestos exposure. Occasional MPICs have been immunoreactive for estrogen and/or progesterone receptors or have diminished in size after treatment with oral contraceptives or a gonadotropin-releasing hormone (GnRH) agonist, suggesting that hormonal factors may play a role in some cases.
Follow-up of cases that we consider to be MPICs has shown no evidence of malignant behavior, but as many as 50% of the lesions have recurred locally, in some cases several times, months to years after their removal. At least some of these recurrences, however, are probably due to newly formed postoperative adhesions. We therefore prefer the designation multilocular peritoneal inclusion cyst to benign cystic mesothelioma for these lesions.
MPICs may be confused on both gross and microscopic examination with multilocular cystic lymphangiomas. Unlike MPICs, the latter usually occur in children, typically boys, and are usually extrapelvic, being localized in most cases to the small bowel mesentery, the omentum, the mesocolon, or the retroperitoneum. In contrast to MPICs, their contents are often chylous; mural lymphoid aggregates and smooth muscle are usually found on microscopic examination, and the lining cells stain positively with endothelial markers rather than keratin. Similarly, MPICs within the spleen have been confused with splenic lymphangiomas. Immunohistochemical stains to distinguish between endothelial and mesothelial cells are diagnostic in problematic cases. The differential diagnosis of MPICs also includes the rare cystic adenomatoid tumor. This tumor, in contrast to MPICs, usually occurs in the uterus, contains foci of typical adenomatoid tumor, and lacks a prominent inflammatory cell infiltrate.
Infectious and noninfectious peritoneal granulomas are almost always readily distinguishable from a neoplasm on microscopic examination. In contrast, peritoneal granulomas secondary to implants of keratin originating from neoplasms of the female reproductive tract may be confused with metastatic tumor. The tumors are usually endometrial (or, less commonly, ovarian) endometrioid carcinomas with squamous differentiation; additionally, squamous cell carcinomas of the cervix and atypical polypoid adenomyomas of the uterus have rarely been the source of the keratin. The granulomas consist of laminated keratin deposits, in some cases accompanied by necrotic squamous cells, surrounded by foreign-body giant cells and fibrous tissue ( Fig. 15.14 ).

Although follow-up data have shown that these granulomas have had no effect on the prognosis, the prognostic significance of these lesions has not been established with complete certainty because of the short follow-up interval in some cases and because some of the patients have received postoperative radiation therapy, chemotherapy, or both. Keratin granulomas, if visible, should be sampled extensively by the surgeon and assiduously examined microscopically by the pathologist to exclude the additional presence of any viable carcinoma cells. The differential diagnosis includes peritoneal granulomas in response to keratin derived from other sources, including amniotic fluid and ovarian dermoid cysts. In the latter situation, cyst rupture releases sebaceous material and keratin that typically evoke an intense granulomatous, lipogranulomatous, and fibrosing peritoneal inflammatory reaction that may mimic a neoplasm at operation.
Reactive peritoneal fibrosis (including fibrous adhesions), a common sequela of peritoneal inflammation and abdominal operations, is rarely a diagnostic problem. A reactive peritoneal fibrotic lesion may contain multipotential subserosal cells that take the form of plump, often fasciitis-like spindle cells that are positive for vimentin, smooth muscle actin, and cytokeratin.
Sclerosing peritonitis is a rare disorder characterized by fibrous peritoneal thickening that can encase the small bowel (abdominal cocoon), causing bowel obstruction. Although it may be idiopathic (such cases typically occur in adolescent girls), common etiologic associations include practolol therapy, chronic ambulatory peritoneal dialysis, the use of a peritoneovenous (LeVeen) shunt, and fibrothecomatous proliferations of the ovary (typically, luteinized thecoma) ( Fig. 15.15 ). With respect to the last association, the intraoperative appearance may mimic that of a malignant ovarian tumor with peritoneal spread, and the microscopic appearance of the peritoneal process in several of the cases was initially misinterpreted as metastatic thecoma (see Fig. 15.15 ). The cause of the sclerosing peritonitis in these cases is unknown.
Sclerosing mesenteritis (retractile mesenteritis, mesenteric panniculitis, mesenteric lipodystrophy) is an idiopathic disorder characterized by varying degrees of fat necrosis, chronic inflammation, and fibrosis, usually involving the mesentery of the small bowel. In the largest study, the patients ranged in age from 23 to 87 years (mean 60); the most common clinical manifestations were abdominal pain or an abdominal mass. Gross examination of the mesentery most commonly revealed a solitary mass; less commonly, multiple masses or diffuse mesenteric thickening were seen. The differential diagnosis includes a variety of benign lesions as well as sarcoma. Rarely, sclerosing mesenteritis my develop as a manifestation of IgG4-related disease, characterized by numerous IgG4-positive plasma cells and obliterative phlebitis. Mesenteric fibromatosis (intraabdominal desmoid tumor), in contrast to sclerosing mesenteritis, infiltrates the bowel wall, has a fascicular architecture, and is devoid of inflammation and fat necrosis. Retroperitoneal fibrosis can histologically mimic sclerosing mesenteritis and indeed may be a related disorder, but the two processes are usually distinguishable by their different clinical presentation, location, and intraoperative appearance. Inflammatory myofibroblastic tumor, unlike sclerosing mesenteritis, is characterized by fascicles of spindle cells with a prominent plasma cellular infiltrate as well as leukocytosis, an elevated erythrocyte sedimentation rate, and anemia in some cases.
Sarcomas (principally liposarcoma) involving the mesentery will almost always exhibit more cellularity, stromal or adipocytic nuclear atypia, and mitotic activity than are present in sclerosing mesenteritis. The majority of cases of inflammatory well-differentiated liposarcoma exhibit increased immunohistochemical expression of MDM2 protein and MDM2 gene amplification, findings that aid in the discrimination from fibroinflammatory disorders.
In occasional cases, reactive fibrous lesions of the peritoneum may be difficult to differentiate from a desmoplastic mesothelioma, particularly in a small biopsy specimen. Features favoring a diagnosis of mesothelioma include nuclear atypia, necrosis, the presence of organized patterns of collagen deposition (fascicular, storiform), and infiltration of adjacent tissues.
Splenosis, or the implantation of splenic tissue, is usually discovered incidentally during an abdominal operation performed months to years after splenectomy for traumatic splenic rupture. Several to numerous red-blue peritoneal nodules, from barely visible to 7 cm in diameter, are scattered throughout the abdominal, and in some cases the pelvic, cavities ( Fig. 15.16 ). The intraoperative appearance may resemble peritoneal involvement by endometriosis, benign or malignant vascular tumors, or metastatic cancer.
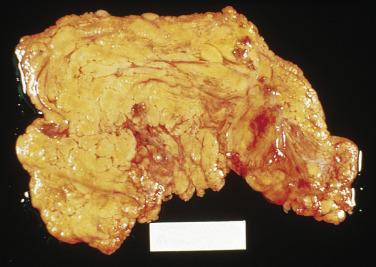
Seven cases of brownish pigmentation of the peritoneum (peritoneal melanosis) have been associated with ovarian dermoid cysts; in two cases, preoperative rupture of the cysts occurred. Focal or diffuse, tan to black peritoneal staining or pigmented tumorlike nodules are encountered within the pelvis and in the omentum at laparotomy. Some locules within the dermoid cysts have had pigmentation of their contents and lining. On microscopic examination, the ovarian and peritoneal lesions consist of pigment-laden histiocytes within a fibrous stroma ( Fig. 15.17 ). In at least half of the reported cases, gastric mucosa was prominent within the dermoid cyst, sometimes associated with ulceration, necrosis, and hemorrhage. One study found that the pigment was neither melanin nor hemosiderin, but contained a high concentration of iron; a relation to peptic ulceration and hemorrhage within the dermoid cyst was suggested. One case of peritoneal melanosis, associated with ovarian mucinous cystadenoma and colonic adenocarcinoma, reported ultrastructural detection of melanosomes within the histiocytes. Distinction of peritoneal melanosis from metastatic malignant melanoma is straightforward because of the bland nuclear features and mitotic inactivity of the pigmented histiocytes. In one case, malignant melanoma metastatic to the omentum was coexistent with foci of melanosis elsewhere in the peritoneum.
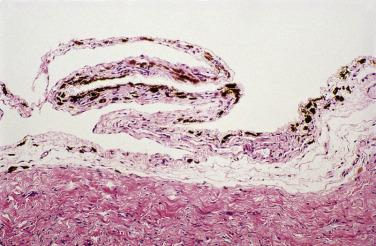
Torsion and infarction of appendices epiploicae, in some cases with subsequent calcification, can result in a hard tumorlike mass that may be found attached or loose within the peritoneal cavity. When examined microscopically, these structures are typically composed of layers of hyalinized fibrous tissue surrounding a central necrotic and calcified zone of infarcted adipose tissue ( Fig. 15.18 ).
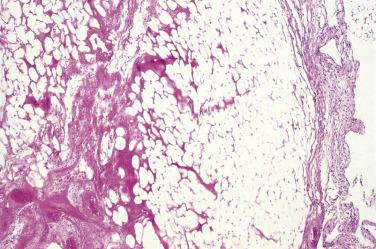
Five cases of cartilaginous differentiation of the peritoneum have been reported, all in women of late reproductive or postmenopausal age. These heterotopic peritoneal lesions were incidental intraoperative findings. In one case, multiple nodules ranged up to 7 mm in maximal dimension, whereas in the other there was a single, smaller nodule (≤2 cm). In all cases the nodules consisted of mature hyaline cartilage surrounded by fibrous tissue. The authors speculated that the nodules could represent metaplastic lesions of the secondary Müllerian system or chondromas derived from submesothelial tissues. We have personally encountered one case of a 3-mm peritoneal cartilaginous nodule in a 31-year-old man, sent for frozen section to exclude peritoneal metastasis during surgery for a pancreatic malignancy ( Fig. 15.19 ).
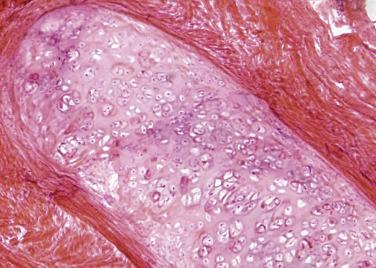
The benign adenomatoid tumor is of mesothelial origin and rarely arises from extragenital peritoneum, such as the omentum or mesentery, but is more commonly encountered within the male and female genital tracts (see Chaps. 13 and 14 ).
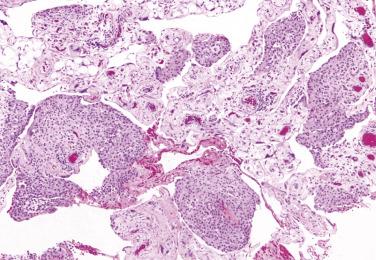
Well-differentiated papillary mesotheliomas (WDPMs) of the peritoneum are uncommon; approximately 100 cases have been reported in detail. Approximately 75% of the tumors occurred in females who were usually of reproductive age but occasionally postmenopausal. WDPMs are usually an incidental finding at laparotomy, but rare tumors have been associated with abdominal pain or ascites. The presence of symptoms or ascites, however, is much more common in malignant mesothelioma. Occasional patients have a history of asbestos exposure.
At laparotomy, and on gross examination, WDPMs can be solitary or multiple and take the form of gray to white, firm, papillary or nodular lesions usually less than 2 cm in diameter. The omental and pelvic peritoneum are typically involved ; several tumors have arisen from the gastric, intestinal, or mesenteric peritoneum.
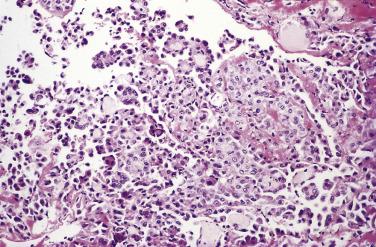
On microscopic examination, fibrous papillae are covered by a single layer of flattened to cuboidal mesothelial cells ( Figs. 15.20 and 15.21 ) that may contain occasional basal vacuoles; the nuclear features are benign, and mitotic figures are sparse or absent. Other patterns encountered less commonly include tubulopapillary (see Fig. 15.20 ), branching cords, and solid sheets. The stroma of some tumors is extensively fibrotic or occasionally myxoid; psammoma bodies are encountered in occasional cases.
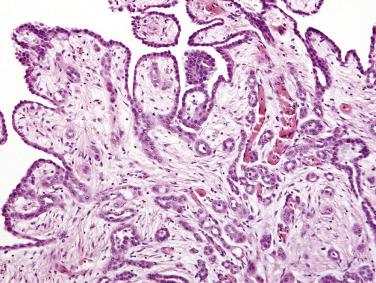
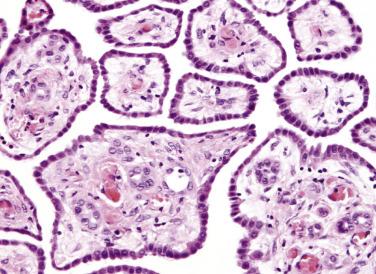
Solitary WDPMs are clinically benign in most if not all cases, although rarely the tumors may persist for decades. When multiple lesions are present, the clinical course is less certain. Each lesion should be sampled for microscopic examination to exclude malignant mesothelioma; Goldblum and Hart found that all of their peritoneal mesotheliomas with involvement of more than one site had, at least focally, malignant histologic features. Only those lesions that were localized and had uniformly benign histologic features had a predictably benign clinical course. The series of WDPMs reported by Butnor and colleagues included a patient with a multifocal WDPM who died of disease after 3 years of follow-up, although it was not clear how thoroughly the tumor had been sampled to exclude a malignant component. In a recent study of 20 WDPMs with invasive foci, the majority occurred in the female peritoneum, and 75% were multifocal. Five cases tested for p16 deletion were negative, but two-thirds had abnormal karyotypes. Recurrences developed in 8 patients, including one fatality (but which lacked histologic confirmation of the recurrent tumor). Thus WDPMs, when multifocal or with invasive foci, warrant clinical follow-up.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here