Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The most common tumors in the mouth arise from the oral epithelium. These are similar to tumors of stratified squamous epithelium occurring at other body sites and only infrequently present problems in diagnosis. The mesenchymal tissues of the mouth are less often the source of neoplasms, but a number of overgrowths occur in response to chronic irritation and it is important that these are distinguished from true neoplasms. Tumors of the jaws may also be problematic since many are rare or may show features specific to the site. Odontogenic cysts and tumors are derived from the tooth-forming tissues and show variable features that may resemble stages of tooth development. Odontogenic tumors are rare, but often pose diagnostic problems for the pathologist unfamiliar with dental tissues; hence they receive more emphasis in this chapter. Odontogenic cysts are common and, although rarely neoplastic, are included since classification may be confusing and they frequently cause diagnostic difficulty.
The most common papillary overgrowths of oral epithelium are associated with infection by human papillomavirus (HPV) and may take one of a number of forms, including squamous cell papilloma, oral condylomas, or focal epithelial hyperplasia. They are caused by low-risk HPVs, most commonly HPV 6 and 11 and may occur at any intraoral site, although typical warts (verruca vulgaris) may be encountered most often on the lips, associated with cutaneous warts on the fingers. Otherwise, the lesions show considerable overlap in clinical and histologic appearances. All are slow growing and show no evidence of premalignancy. The incidence of lesions diagnosed as squamous cell papillomas or warts is increased in patients with human immunodeficiency virus (HIV) infection.
In a typical squamous cell papilloma the epithelium is of fairly uniform thickness, with variable keratinization or hyperkeratosis, and is formed into sharp papillary or frondlike processes ( Fig. 6.1 ) each with a core of loose vascular connective tissue. There is marked basal cell hyperplasia, and increased mitotic activity is often present. This may be accentuated if there is infection by Candida albicans in the superficial layers of the epithelium. Some oral lesions, often on the lips, show typical histologic appearances of verruca vulgaris, with inward curving rete pegs and more prominent koilocytic change. Condyloma acuminatum is also reported in the mouth but cannot be diagnosed with certainty on histologic grounds. Where a discrete papillary exophytic growth shows acanthosis instead of relatively uniform folds of epithelium and the papillary processes are flattened or smoothly rounded, condyloma acuminatum should be suspected.
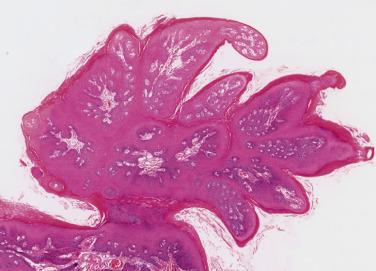
Focal epithelial hyperplasia (also called Heck disease) is caused by HPV 13 and 32, and presents as multiple, sessile elevated nodules. Histologically there is fibrous overgrowth covered by irregularly hyperplastic epithelium. Koilocytes and fragmented nuclear material, described as mitosoid bodies ( Fig. 6.2 ), are typically present in the stratum spinosum and upper epithelial layers.
Over 90% of the malignancies in the upper aerodigestive tract are squamous cell carcinomas arising from the epithelium of the mucosal lining. Although common, the diagnosis and management can still be challenging due to the subjective nature of diagnosis and grading, and the number of variants, each with different diagnostic criteria, site predilection, and prognosis. In particular it is now apparent that oral cancer encompasses a number of lesions that should be regarded as separate entities on the basis of site, pathogenesis, prognosis, and management. Squamous cell carcinoma of the oral cavity (mouth) should be regarded as a different disease to squamous cell carcinoma arising in the oropharynx. This is based primarily on the etiology, in that oral lesions are associated with the traditional factors of tobacco and alcohol use, whereas oropharyngeal cancer is associated with infection by high-risk HPV (HPV 16 and 18). These differences justify a division of oral cancer into oral cancer and oropharyngeal cancer and these are considered separately in this chapter.
The most frequent sites for squamous cell carcinoma of the mouth are the lateral tongue, floor of the mouth, and the retromolar/soft palate region, followed by the lips, gingivae (gums), and hard palate. In parts of the world with a high incidence of oral cancer, such as India, most oral cancers arise in areas of leukoplakia, and the buccal mucosa may be a common site due to chewing of areca nut with or without tobacco. By contrast, in developed countries, small intraoral carcinomas are almost always red or predominantly red and may arise in clinically apparently normal epithelium.
Oral squamous cell carcinomas show the same range of features and variants as such carcinomas at other sites, and criteria for grading of tumors is the same (see Chapter 23 ). Diagnosis is usually straightforward, although the earliest stages of invasion may be difficult to determine and criteria for microinvasion are not adequately defined. This is especially a problem in verrucous or well-differentiated lesions. Fortunately such lesions are unlikely to metastasize at the stage when the term microinvasive might be used, and the local excision appropriate for a verrucous lesion is also the appropriate treatment for microinvasive carcinoma. Exophytic, verrucous lesions can pose particular problems when there is superimposed infection with Candida albicans . This is often associated with obviously increased mitotic activity and it is important to recognize that this is disproportionate to the degree of dysplasia. It is often wise to defer a diagnosis of malignancy in such cases until fungal infection is treated and the lesion is rebiopsied.
There have been many attempts to devise schemes for predicting the prognosis of oral carcinomas from analysis of histologic features but none have found general favor for routine use. Most studies regard the character of the invasive front of the lesion as being of most prognostic significance. A histologic risk model utilizing worst pattern of invasion, perineural infiltration, and a limited immune response has been shown to be a good predictor of disease progression, recurrence, and reduced survival. Vascular and perineural invasion and tumor thickness are also important predictors of lymph node metastasis. A meta-analysis has suggested that a thickness exceeding 4 mm is associated with a significantly increased risk of metastasis, but this may vary from site to site. Tumor thickness should be measured from the actual surface for flat lesions or the presumed original epithelial surface for exophytic or ulcerated lesions.
A number of variants are described and their relevance is that they are thought to have differing prognoses. Some tumors are comprised entirely of the variant type, but in many instances lesions show a mixture of conventional squamous cell carcinoma and other patterns. In these cases it is probably best to describe the mixed patterns and advise that the behavior is likely to be determined by the most aggressive variant present.
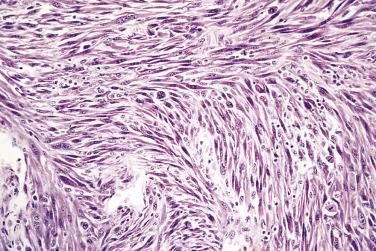
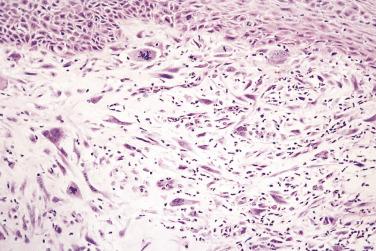
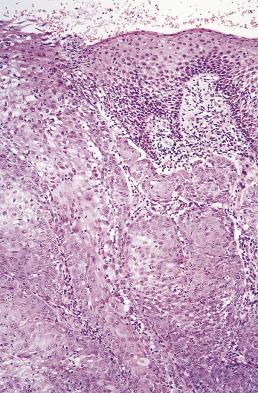
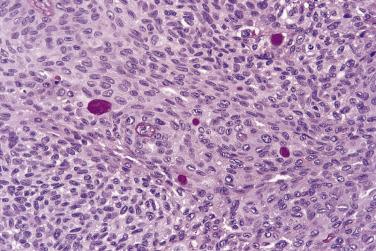
Spindle cell squamous carcinoma is a pleomorphic spindle cell neoplasm most frequently found in the head and neck. Spindle cell tumors of all kinds are uncommon in the mouth, but the full spectrum of soft tissue sarcomas (see Chapter 24 ) can occur. In any such case, spindle cell carcinoma should always be included in the differential diagnosis. In the larynx, spindle cell carcinomas are often polypoid, but in the oral cavity they are similar to conventional carcinomas and present as ulers or red lesions. A wide range of histologic appearances is seen in these tumors ( Figs. 6.3 and 6.4 ). Immunohistochemical demonstration of cytokeratin in tumor cells strongly supports the diagnosis, but overall only about 60% of cases may be positive. Variable results are obtained with different cytokeratin antibodies but positivity is most frequently demonstrated with a broad specrum pan-cytokeratin such as AE1/3. Markers of squamous epithelium such as p63 or p40 may also be useful. For a definitve diagnosis, especially if immunohistochemistry for cytokeratins is negative, it is wise to search carefully for areas of conventional squamous cell carcinoma or severe epithelial dysplasia, usually at the growing edge of the tumor, before making the diagnosis of spindle cell carcinoma, although mucosal ulceration may preclude this.
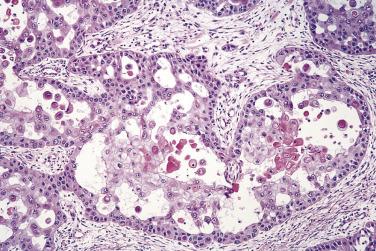
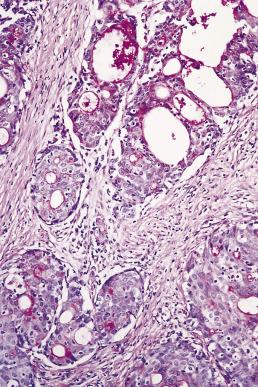
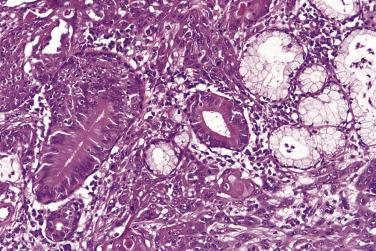
There are two variants of oral cancer that may exhibit glandular features. Adenoid squamous cell carcinoma describes cases of conventional squamous cell carcinoma in which loss of intercellular adhesion in the center of tumor islands resembles glandular differentiation ( Fig. 6.5 ). These are poor-prognosis tumors. Adenosquamous carcinomas also have a poor prognosis. In these lesions there is involvement of minor salivary gland ducts, concurrently with tumor arising from the surface mucosa. The tumor shows separate discrete areas of adenocarcinoma and of squamous cell carcinoma arising from the surface epithelium ( Figs. 6.6 and 6.7 ). The main differential diagnosis is from mucoepidermoid carcinoma, but this is usually cystic and shows an admixtutre of epidermoid and mucus-producing cells. A small number of otherwise conventional squamous cell carcinomas may show a few mucus-secreting cells and demonstration of these, in the absence of ductal differentiation ( Fig. 6.8 ), is not sufficient for a diagnosis of adenosquamous carcinoma. It is also important to recognize correctly the potentially confusing presence of invasion by squamous cell carcinoma into salivary gland ( Fig. 6.9 ).
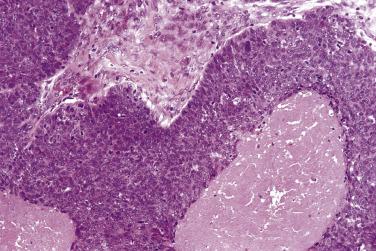
This is an uncommon variant of squamous cell carcinoma (see Chapter 4 ) that is distinct from carcinomas with basaloid morphology associated with HPV infection, which are more often encountered in the oropharynx and may have a better prognosis (see later in this chapter). The term basaloid squamous cell carcinoma should therefore only be used for an aggressive basaloid variant of squamous cell carcinoma, which is HPV (and p16) negative, and should not be confused with HPV-associated oropharyngeal lesions. Basaloid squamous cell carcinoma is characterized by a squamous component as well as areas composed of well-demarcated nests of basaloid cells arranged in a jigsaw pattern. There is peripheral palisading, prominent mitotic activity, and comedo necrosis is common ( Fig. 6.10 ) . These lesions are typically aggressive with early lymph node metastasis . They are most often seen in the posterior aspect or base of the tongue and must be distinguished from other basaloid lesions, including HPV-associated carcinomas, solid adenoid cystic carcinoma, or salivary duct carcinoma.
This term was first coined for exophytic, massively acanthotic lesions showing minimal cytologic atypia which may not, on strict histologic grounds, be diagnosed as carcinoma. Subsequently the term has been often misused and applied erroneously to conventional squamous cell carcinomas which happen to have an exophytic verruciform surface pattern. True verrucous carcinomas have a better prognosis than conventional squamous cell carcinoma.
Verrucous carcinomas are exophytic, keratinized growths with a white papillary or warty surface. They are mainly seen in the elderly and are associated with tobacco use. They may have surface dimensions of several centimeters, but show superficial spreading growth rather than invasion into deeper tissues.
There is marked acanthosis, with broad bulbous rete pegs showing central columns of keratin ( Fig. 6.11 ). Although exophytic, with columns of keratin rising above the adjacent mucosal surface, there must also be an endophytic component. The bulbous rete processes push deep into the lamina propria so that the base of the lesion is below the adjacent basement membrane. There is little cytologic evidence of malignancy, but the architectural changes are marked and there may be some dyskeratosis with deep single cell keratinization and eosinophilic glasslike cytoplasm. Pronounced chronic inflammatory cell infiltration is often present in the lamina propria ( Fig. 6.12 ). The diagnosis of verrucous carcinoma should only be made after clinicopathologic correlation. An unusual lesion which may be a variant of verrucous carcinoma is carcinoma cuniculatum , which arises most often on the gingivae and is composed of ribbonlike folds of well-differentiated keratinized squamous epithelium that pushes or burrows into the underlying connective tissues. Although there may be wide local destruction, the lesions have a good prognosis, with few reports of metastases.

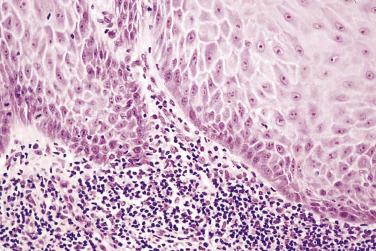
The main differential diagnosis is from conventional squamous cell carcinoma and other verruciform or papillary lesions. Many oral cancers may have an exophytic verruciform surface but also show cytologic features of malignancy and invasion. The term verrucous carcinoma , although used by some authors for such lesions, is inappropriate. Some tumors have areas of verrucous carcinoma and areas of conventional squamous cell carcinoma and it is important that the latter be sought for before the diagnosis of verrucous carcinoma is made. Occasionally papillomas or viral warts may be extensive and have similar clinical and histologic appearances. However, these lesions are exophytic and often pedunculated and rise above the surface of the mucosa, while verrucous carcinoma is endophtyic. Verrucous hyperplasia and proliferative verrucous leukoplakia may appear similar but are also exophytic (see upcoming discussion).
When applied to the oral cavity, this term has often been used for otherwise conventional squamous cell carcinomas with an exophytic papillary surface. True papillary carcinomas are rare in the mouth and are most frequently encountered in the larynx and hypopharynx. They are composed of exophytic papillary fronds of epithelium overlying a fibrovascular core. There is considerable cytologic atypia of the epithelium, but stromal invasion may not be prominent. Differential diagnosis is from verrucous carcinoma and squamous papilloma, but papillary carcinomas are often exophytic and pedunculated and show obvious epithelial dysplasia. Distinction from an exophytic conventional carcinoma is based on the lack of stromal invasion and the clearly defined papillary pattern of true papillary carcinoma. Recent studies have suggested that up to 66% of oral lesions may be infected with high-risk HPV.
These lesions are associated with HPV infection and arise in those areas of the oropharyngeal mucosa where tonsillar tissue is found, primarily the palatine tonsils of the fauces and the tonsillar tissue at the base of the tongue. Between 50% and 80% of lesions at these sites harbor high-risk HPV (HPV 16 and 18). Oropharyngeal squamous cell carcinomas are characterized by being nonkeratinized with basaloid morphology. Previously this has been regarded as an indication that they are poorly differentiated, but this characteristic morphology actually represents well-differentiated tonsillar crypt epithelium. For this reason, HPV-associated carcinomas should not be graded using conventional grading schemes.
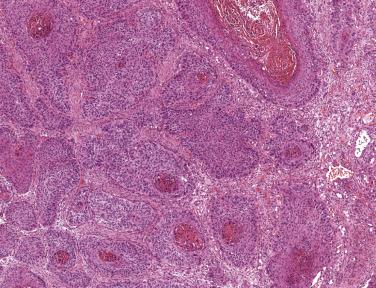
Oropharyngeal cancer is defined by the presence of high-risk HPV. These lesions contain E6 and E7 proteins, which degrade p53 and Rb, respectively, resulting in accumulation and overexpression of the tumor suppressor protein p16. The gold standard method for diagnosis is to demonstrate transcriptionally active HPV by PCR or by in situ hybridization ( Fig. 6.13 ). However these may not be widely available, and immunohistochemical expression of p16 protein is therefore an excellent surrogate marker for HPV infection and can be used in routine clinical practice as a diagnostic marker. Conventional squamous cell carcinomas of the oral cavity are not associated with HPV infection and are usually p16 negative by immunohistochemistry. However, p16 staining must be interpreted with caution and should only be used as a stand-alone surrogate marker for lesions in the oropharynx. Normal tonsillar crypt epithelium may express low levels of p16, and small numbers of conventional SCC may express p16 in the absence of any evidence of HPV infection. The criteria for positive p16 staining are that at least 70% of the cells should be diffusely positive in both the nucleus and cytoplasm ( Fig. 6.14 ).
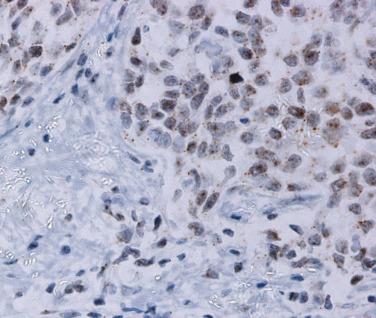
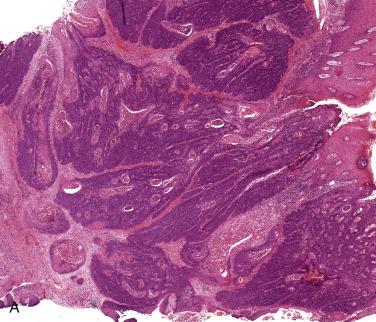
Most oropharyngeal carcinomas are composed of sheets of nonkeratinizing basaloid cells with poorly demarcated cell borders, prominent mitoses, and occasional areas of comedo necrosis (see Fig. 6.14A ). Epidermoid or squamous cells may be seen and may occasionally form keratin, but these usually comprise less than 10% of the lesion. Strong diffuse positivity for p16 is seen in virtually 100% of these lesions (see Fig. 6.14B ). About 10% of oropharyngeal carcinomas show more prominent keratinization, and have been termed hybrid lesions ( Fig. 6.15 ). Others may be keratinizing, with obvious squamous areas and keratin pearls. These lesions are not always associated with HPV and may represent conventional squamous cell carcinomas arising at this site.
It should be noted that the clinical significance of HPV infection is still uncertain. In oropharyngeal lesions there is good evidence that patients with HPV-positive lesions have a better response to treatment and an overall better prognosis. It has been suggested that treatment can be de-escalated, but initial clinical trials do not support this and there is still much important research to be undertaken.
Leukoplakia and erythroplakia are recognized clinically as the most important potentially malignant disorders. The main reason for biopsying such lesions is to exclude the possibility that a carcinoma is already present. Thereafter, the pathologist is asked to assess and grade any dysplasia present. This is on the assumption that dysplasia is an indicator of the potential likelihood of progression to malignancy. The rationale for this assumption is that dysplasia is associated with mutations involving the epithelium which may also be responsible for progression to malignancy. Microarray studies indicate substantial genomic change in dysplasia, suggesting that many of the changes are epiphenomena not directly related to malignant change. It is assumed that the more dysplastic an epithelium is, the more likely it is that the critical mutations linked to malignant transformation will be present. Thus more severely dysplastic lesions have a greater risk of progression. Overall, only a minority of lesions showing epithelial dysplasia progress to malignancy and carcinomas may also arise in epithelium not showing dysplasia.
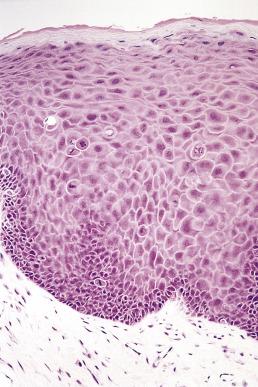
The new World Health Organization (WHO) classification recognizes that problems exist with the subjective nature of the assessment of dysplasia and the variation in grading, not only between pathologists but also for the same pathologist examining the same material on different occasions. The recommendation made is that the epithelium should be assessed in terms of architectural and cytologic disturbances, and their presence and severity measured in thirds of the epithelium ( Table 6.1 ). If the architectural change is confined to the lower third of the epithelium, the dysplasia is mild ( Fig. 6.16 ). Extension into the middle third indicates moderate dysplasia ( Fig. 6.17 ) and involvement of the upper third indicates severe dysplasia ( Fig. 6.18 ). These preliminary judgments, based on the extent of architectural change, can be modified when consideration is given to the severity and extent of the cytologic or architectural features. In Fig. 6.19 the initial assessment of moderate dysplasia is upgraded to severe dysplasia on this basis. The term carcinoma in situ is sometimes used when dysplasia involves the full thickness of the epithelium, but in the oral cavity it is rare for there not to be at least some evidence of surface keratinization. For oral lesions therefore, carcinoma in situ is regarded as synonymous with severe dysplasia ( Fig. 6.20 ).
| Architectural Changes | Cytologic Changes |
|---|---|
| Irregular epithelial stratification | Abnormal variation in nuclear size |
| Loss of polarity of basal cells | Abnormal variation in nuclear shape |
| Drop-shaped rete ridges | Abnormal variation in cell size |
| Increased number of mitotic figures | Abnormal variation in cell shape |
| Abnormally superficial mitotic figures | Increased nuclear–cytoplasmic ratio |
| Premature keratinization in single cells | Atypical mitotic figures |
| Keratin pearls within rete pegs | Increased number and size of nucleoli |
| Loss of epithelial cell cohesion | Nuclear hyperchromasia |
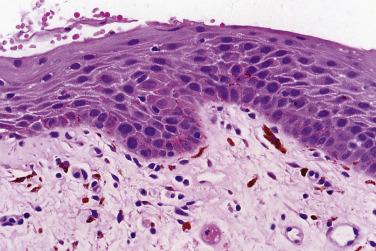
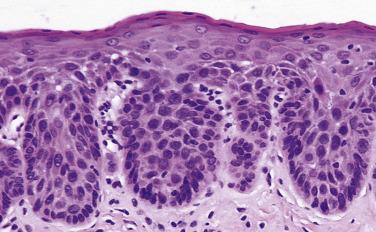
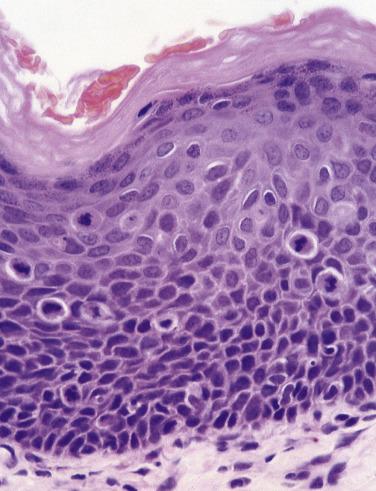
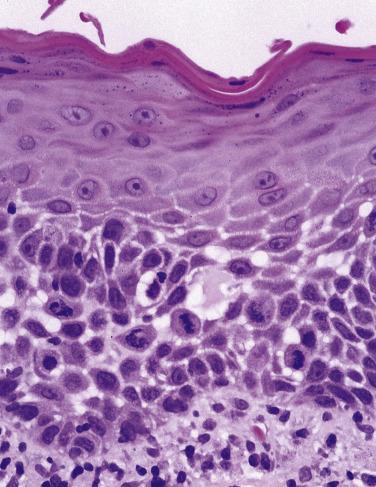
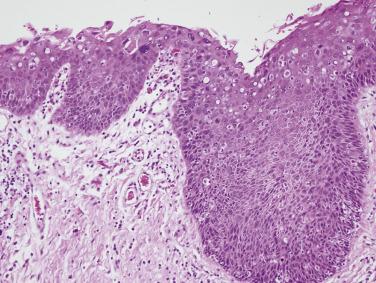
Occasional dysplastic oral lesions may be positive for high-risk HPV and are characterized by prominent atypia above the basal layers with marked karyorrhexis and apoptosis throughout the epithelium. By the criteria described, these show severe dysplasia and a subset may undergo malignant transformation.
In determining whether dysplasia is present, it is important to recognize that other situations may cause cytologic atypia that may mimic dysplasia. Infection by Candida albicans is a common cause, but other regenerative and reparative changes related to inflammation, ulceration, or irradiation may cause atypia. Nutritional deficiencies such as iron, folate, or vitamin B12 can also simulate dysplasia.
Proliferative verrucous leukoplakia is a particularly aggressive form of leukoplakia which starts as simple hyperkeratosis but pursues a relentless clinical course of verruciform lesions which progress to malignancy. The clinical features are critical in the diagnosis and emphasize the need for histopathologic interpretation to be made in the proper clinical context. Diagnosis is therefore retrospective, based on a history of multiple and recurrent lesions and increasingly aggressive behavior.
Hairy leukoplakia is the term used to describe a clinically distinctive hyperkeratosis, principally restricted to the lateral aspect of the tongue in HIV-positive patients and less frequently in other immunosuppressed individuals. It is caused by Epstein-Barr virus infection and does not show dysplasia, nor is there any evidence that it is premalignant.
The oral tissues are subject to the same types of mesenchymal neoplasms that may be found at other body sites (see Chapter 3, Chapter 23, Chapter 24 ). All are uncommon and only a small minority are malignant. Perhaps the most frequently encountered are benign neural tumors, lipomas, and myofibromas. The most frequently encountered tumorlike lesions submitted for histopathologic diagnosis, however, are various nonneoplastic fibrous overgrowths, usually related to some source of chronic irritation.
Fibrous overgrowths commonly occur as discrete lesions on the cheek or tongue related to chronic irritation, usually from a sharp tooth or from biting the cheek or tongue. Dentures, particularly if old and ill fitting, are also a common source of irritation. This chronic irritation gives rise to mucosal overgrowths to which various names are applied. Most lesions present as a sessile fibrous hyperplasia or a fibroepithelial polyp. Lesions on the gingival margin are called an epulis , which is a clinical term to describe any localized swelling on the gums. At this site the overgrowths are caused by chronic irritation due to a localized accumulation of bacterial plaque. Three main types are recognized: (1) fibrous epulis, (2) so-called pyogenic granuloma (vascular epulis), and (3) giant cell epulis. There are similarities in clinical presentation and all are liable to recurrence, usually because of persistence of the provoking stimulus.
Fibrous overgrowths on the cheek or tongue are often pedunculated (fibroepithelial polyp) and consist of relatively acellular collagenous connective tissue covered by epithelium, which may be hyperplastic or atrophic but is usually hyperkeratotic ( Fig. 6.21 ). Enlarged, angular fibroblasts and occasional multinucleate fibroblasts are seen in some cases. The term giant cell fibroma has been used by some authors for such lesions, but this is inappropriate as there is no evidence that they are neoplasms.
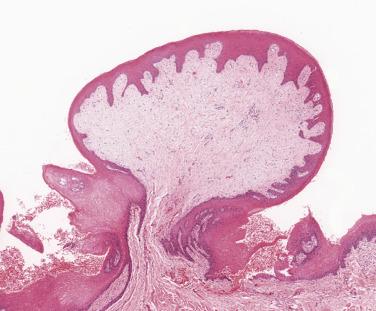
Fibrous epulides are composed of cellular fibrous connective tissue which is often inflamed with an ulcerated surface ( Fig. 6.22 ). Calcification in the form of small, rounded, usually acellular foci or metaplastic bone formation is quite frequent. Such lesions are quite often inappropriately labeled peripheral ossifying fibroma since they can resemble a fibro-osseous lesion or ossifying fibroma, but the distinction from such bone lesions (see Chapter 25 ) is readily made by the clinically peripheral location of the epulis, which does not arise in bone.
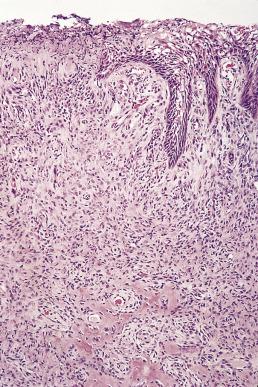
Although the gingiva is the most common site for this lesion, they can occur as exuberant overgrowths at any intraoral site. Pyogenic granulomas on the gingiva (vascular epulis) are particularly common during puberty or pregnancy, when they may be quite exuberant, reaching 2 or 3 cm in size. The clinical differential diagnosis may include hemangiomas and angiosarcoma, as well as bacillary angiomatosis and Kaposi sarcoma (see Chapter 3 ).
There is usually extensive ulceration of the surface epithelium, which accounts for a superficial zone of granulation tissue and infiltrates of mixed acute and chronic inflammatory cells. The bulk of the lesion consists of loose connective tissue containing lobules of proliferating capillaries ( Fig. 6.23 ), which may be mitotically active. Pyogenic granulomas arising on the gingivae may often contain calcifications or metaplastic bone.
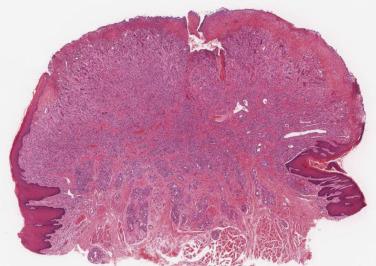
On histologic grounds alone it may be impossible to differentiate a pyogenic granuloma from a hemangioma with overlying ulceration. Attention to the site of the lesion and the clinical history is helpful. The differential diagnosis from rarer vascular neoplasms may require specialist consultation, especially since nuclear atypia in endothelial cells is common in pyogenic granulomas in this region.
The main feature is nonencapsulated lobules of closely packed multinucleated giant cells lying in a vascular stroma. The giant cells are osteoclasts and are surrounded by plump mononuclear cells which are their monocyte precursors ( Fig. 6.24 ). Obvious mitotic activity is not uncommon in these cells.
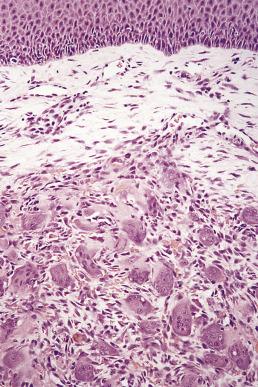
The main differential diagnosis is from central giant cell lesions extending out of the bone to present as soft tissue lesions. These should be excluded by radiologic examination. The diagnosis of central giant cell granuloma is made after exclusion of hyperparathyroidism, which also causes intraosseous lesions, but peripheral extension cannot be distinguished histologically from a giant cell epulis, which may occasionally be a manifestation of hyperparathyroidism. This diagnosis should be considered where giant cell epulides are multiple or are associated with pregnancy, which can potentiate the manifestations of hyperparathyroidism.
The congenital epulis is a rare but distinctive oral lesion of unknown etiology found in the alveolus of neonates. Almost all affected patients are female. This lesion shows histologic similarities to granular cell tumor elsewhere but is S100-protein negative (see Chapter 27 ). Immunohistochemical studies have suggested primitive pericytic or myofibroblastic differentiation.
These overgrowths are usually found in the labial or buccal sulci related to the fitting edge of a denture. Less frequently they occur at the posterior edge of an upper denture in the palate. Leaf fibroma is a descriptive term sometimes used for a pedunculated flattened fibrous overgrowth which may occur in the palate, occupying space between a poorly fitting denture and the palatal mucosa. This term is inappropriate in that the lesion is not a neoplasm. Recurrence may be a problem following excision if the ill-fitting denture is not repaired or replaced.
The lesion consists largely of collagenous connective tissue covered by hyperplastic epithelium which is often ulcerated, in which case part of the tissue bulk may be composed of inflamed granulation tissue.
This occurs in the vault of the palate under a denture as a coarsely granular mass of soft tissue.
Cores of moderately vascular connective tissue are covered by irregularly hyperplastic epithelium which may have a pseudoepitheliomatous appearance. At low power this can sometimes simulate invasion ( Fig. 6.25 ) with epithelial downgrowths and sometimes apparent keratin pearl formation. Higher power examination confirms the entirely benign cytologic features.
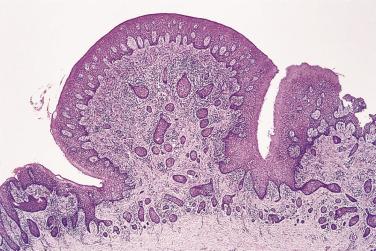
The jaws are the only site in the body where epithelium may normally be found within bone. The epithelium of the dental lamina is involved in the formation of enamel and maps out the shape of the tooth. On completion of tooth formation, epithelial remnants remain in the jaws. These give rise to a range of lesions, including neoplasms and hamartomas, which should pose no problem with diagnosis when seen to be associated with teeth, but can cause difficulty in other situations. The most frequent epithelial rests are the cell rests of Malassez, which are found in the periodontal ligament as small clusters and strands of epithelium, but occasionally as larger nests. These may be left in alveolar bone following tooth extraction. This epithelium gives rise to the most frequent type of epithelial odontogenic cyst, the radicular cyst , which is an inflammatory cyst arising at the apex of a tooth following death of the pulp (see p. 295 ). Epithelial rests and calcifications can be found in the tissues related to the crowns of unerupted or partially erupted teeth, where they may be misdiagnosed as odontogenic tumors, especially ameloblastoma or ameloblastic fibroma.
The classification and nomenclature of odontogenic lesions has been reviewed extensively and, although it can be very complex, the recent WHO classification has attempted to simplify terminology and reduce the number of clinically relevant entities ( Tables 6.2, 6.3, and 6.4 ). The majority of lesions fit into this classification, but it is probably appropriate to regard odontogenic tumors as forming groups of variably well-defined lesions which often show overlapping features. A key underlying principle in the classification scheme relates to the inductive interactions of the dental tissues, in which tumors mimic normal tooth development. The epithelium of the normal dental lamina arises by downgrowth from the oral epithelium. The formation of the connective tissue of the dental papilla is followed by differentiation of odontoblasts, which commence the formation of dentine, and ameloblasts then start to lay down enamel. Odontogenic tumors may consist entirely of epithelium or of mesenchyme, or may show an admixture of epithelial and mesenchymal components. Hard tissue formation may be variable, from small deposits of dentinelike material, to production of well-formed toothlike structures seen in odontomas. The key to diagnosis is a basic understanding of these developmental interactions and careful examination of conventional hematoxylin and eosin–stained sections. Ancillary tests, including immunocytochemistry, have a very minor role to play. Almost all odontogenic tumors are benign and nonmetastasizing, although they can cause substantial local destruction and disfigurement.
| Frequency (%) |
|
|---|---|
| Benign Epithelial Odontogenic Tumors | |
| Ameloblastoma | 12 |
| Squamous odontogenic tumor | <1 |
| Calcifying epithelial odontogenic tumor | <1 |
| Adenomatoid odontogenic tumor | 3 |
| Benign Epithelial and Mesenchymal Odontogenic Tumors | |
| Ameloblastic fibroma | 4 |
| Primordial odontogenic tumor | <1 |
| Dentinogenic ghost cell tumor | <1 |
| Odontomas | 70 |
| Benign Mesenchymal Odontogenic Tumors | |
| Odontogenic fibroma | <1 |
| Odontogenic myxoma | 3 |
| Cementoblastoma | <1 |
| Cemento-ossifying fibroma | 1 |
| Subtype | Frequency (%) | Characteristics |
|---|---|---|
| Conventional ameloblastoma | 85 | Intraosseous lesions, may be solid or multicystic. Follicular or plexiform pattern. Most often at angle of mandible. Locally aggressive |
| Unicystic ameloblastoma | 14 | Intraosseous, single large unilocular cyst. May have luminal projections. Good prognosis, curettage usually curative |
| Peripheral ameloblastoma | 1 | Extraosseous, on the gingiva. Follicular or plexiform pattern. Good prognosis, rarely recurs |
| Odontogenic Carcinomas | |
| Ameloblastic carcinoma | An ameloblastoma showing cytologic evidence of malignancy. May arise de novo or in a preexisting conventional ameloblastoma. May metastasize |
| Malignant ameloblastoma | An ameloblastoma with evidence of metastasis. The primary lesion and metastasis show similar benign histologic features of conventional ameloblastoma |
| Primary intraosseous carcinoma | Squamous cell carcinoma derived from odontogenic epithelial rests. Must be wholly intraosseous and must exclude extension from a mucosal lesion or metastasis. Some may arise in a preexisting odontogenic cyst |
| Sclerosing odontogenic carcinoma | A low-grade carcinoma, showing infiltrating strands and islands of bland epithelial cells. Local destruction and perineural infiltration are seen |
| Clear cell odontogenic carcinoma | Entirely composed of clear cells. Aggressive and may metastasize. Exclude clear cell variants of other tumors and metastasis. Shows specific EWSR1-ATF1 translocation |
| Ghost cell odontogenic carcinoma | Malignant variant of dentinogenic ghost cell tumor. Well characterized but very rare |
| Malignant variants of other odontogenic tumors or cysts | Malignant variants have rarely been described for most tumors or arising in most types of odontogenic cysts. Key features are cytologic evidence of malignacy with residual evidence of the precursor lesion |
| Odontogenic Sarcomas | |
| Odontogenic carcinosarcoma | A true maligant mixed tumor. Similar to ameloblastic fibroma, but both the epithelial and mesenchymal components show cytologic evidence of malignancy |
| Ameloblastic fibrosarcoma | Ameloblastic fibroma, but with cell crowding and evidence of cytologic atypia only in the mesenchymal component |
A simplified classification of benign tumors is shown in Table 6.2 , with an indication of the relative frequency of occurrence of individual lesions. Although the odontoma is the most common tumor by far, it should be noted that these are hamartomas, and that the most common true neoplasm is the ameloblastoma.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here