Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Tumors of the heart and pericardium are, as in most sites, broadly dichotomized into benign and malignant varieties. Among the benign tumors, some are correctly classified as neoplasms, some as nonneoplastic entities, some as hamartomas or heterotopias, and some as processes somewhere in between. No firm distinctions pertaining to histogenesis are drawn in this discussion as the emphasis is on accurate diagnosis. It is important to note that, although the tumors discussed in this chapter represent primary cardiac tumors, by far the most commonly encountered cardiac tumors are metastatic lesions from other primary sites (most commonly lung, breast, and cutaneous melanoma).
More than 90% of primary cardiac tumors are benign, with the majority in adults being myxomas. In infants and children the most common primary tumor of the heart is the rhabdomyoma. Malignant cardiac and pericardial tumors, like malignant tumors elsewhere in the body, have the ability to invade and metastasize. Benign cardiac tumors as well, however, can have clinically malignant consequences given the frequency of endocardial or conduction system involvement. Box 2.1 lists the most common cardiac tumors, including true neoplasms, hamartomas, and nonneoplastic entities.
| BENIGN | MALIGNANT |
|---|---|
| Myxoma | Angiosarcoma |
| Rhabdomyoma Fibroma Lipoma and lipomatous hypertrophy of atrial septum Papillary fibroelastoma Histiocytoid cardiomyopathy Teratoma Hemangioma Cystic tumor of the atrioventricular node Hamartoma of adult cardiac myocytes Paraganglioma Calcified amorphous tumor (CAT) Mesothelial/monocytic incidental cardiac excrescences (MICE) |
Undifferentiated high-grade pleomorphic sarcoma |
| Rhabdomyosarcoma | |
| Leiomyosarcoma | |
| Malignant mesothelioma | |
| Lymphoma | |
The clinical manifestations of cardiac tumors are often nonspecific. Indeed, many other diseases may be mimicked. The clinical presentation is usually subdivided under three major headings: (1) systemic, (2) embolic, and (3) cardiac. Clinical features reported to occur with each tumor will not be mentioned in the discussion of that tumor, unless specific to that tumor for some reason.
The systemic manifestations of tumors of the heart are manifold and include findings such as fever, cachexia, and malaise. Abnormal laboratory findings that may develop include an elevated erythrocyte sedimentation rate, hypergammaglobulinemia, thrombocytosis or thrombocytopenia, polycythemia, leukocytosis, and/or anemia. The mechanisms that underlie these systemic manifestations are not as yet fully understood, but it is likely that they relate to cytokine elaboration.
Many cardiac tumors are also associated with clinical systemic syndromes and genetic diseases. A summary is found in Table 2.1 .
| Tumor Type | Associated Syndrome | Involved Gene(s) | Chromosome Location | Inheritance Pattern | Syndrome Prevalence | Association with Syndrome (%) |
|---|---|---|---|---|---|---|
| Myxoma | Myxoma syndrome | PRKAR1A | 17q2 | AD | Rare (~500 cases worldwide) | ~7 |
| Rhabdomyoma | Tuberous sclerosis | TSC1/TSC2 | 9q34/16p13 | AD (1/3), sporadic (2/3) | 1 in 6000 (at birth) | ~90 |
| Fibroma | Nevoid basal cell carcinoma syndrome (Gorlin syndrome) | PTCH1 | 9q22.3 | AD (1/3), sporadic (2/3) | 1 in 57,000 | ~4 |
| Paraganglioma | VHL Neurofibromatosis type 1, MEN-2A, -2B FPPS |
VHL | 3p25 ( VHL ) | AD and AD with maternal imprinting ( SDHD ) | 1 in 36,000 (VHL) 1 in 35,000 (MEN-2) 1 in 1,000,000 (FPPS) |
~5 |
| NF1 | 17q11 ( NF1 ) | |||||
| RET | 10q11 ( RET ) | |||||
| SDHB | 1p36 SDHB | |||||
| SDHC | 1q21 SDHC | |||||
| SDHD | 11q23 SDHD | |||||
| Histiocytoid cardiomyopathy | H1CMP | NDUFB11 Mitochondrial genes |
X chromosome, mitochondrial DNA | AR, X-linked and maternal | Rare | ~100 |
| Angiosarcoma | Li-Fraumeni–like syndrome | POT1 | 7q31.33 | AD | Rare | Unknown |
These events can be due to embolization either of the tumor itself or of thrombi on the surface of the tumor. Embolization of tumor fragments can occur only when the tumor itself shows intracavitary extension. Thromboemboli, on the other hand, can also occur with intramural tumors, which compromise the function of the heart leading to intracavitary thrombosis.
The pathologist may be the first to suspect the presence of a cardiac tumor on the basis of examination of a peripheral embolus. In fact, sudden occlusion of a peripheral artery in an otherwise healthy person should always raise the possibility of a cardiac tumor. An embolectomy specimen therefore should be examined most carefully for the presence of tumor fragments. However, even when only recent thrombotic material is found, it is wise to mention the possibility of a coexistent cardiac tumor. Moreover, multiple systemic emboli may mimic systemic vasculitis or infective endocarditis, particularly when associated with systemic manifestations. Primary tumors of the right heart chambers may cause pulmonary emboli, which may be indistinguishable from those occurring as a result of venous thromboembolism.
The cardiac events that can develop as a result of cardiac tumors are largely determined by the location and size of the tumor. Tumors that are localized in the myocardium usually lead to impaired myocardial function either through substantial replacement of the myocardium by tumor or because of extension into a cardiac chamber. Intramural location may lead to a wide variety of rhythm disturbances, including atrial and ventricular fibrillation. Sudden death may thus be the first manifestation of a cardiac tumor. Primary tumors with intracavitary extension may also cause obstruction and interfere with valve function.
Pericardial effusion, sometimes with signs and symptoms of cardiac tamponade, is usually a result of either epicardial extension or a primary pericardial tumor.
This is frequently cited as the most common primary heart tumor, although nonneoplastic entities (e.g., thrombi) and the papillary fibroelastoma (of uncertain histogenesis) are actually more common. Nevertheless, it is likely that they represent the most common primary cardiac neoplasm. It is a tumor of adults, occurring most often in women aged 20 to 60 years. Most occur sporadically, but about 7% are associated with the Carney complex (CNC). This autosomal dominant syndrome characterized by the presence of multiple myxomas, spotty pigmentation, and endocrine overactivity ( Table 2.2 ) has been given a variety of eponyms and acronyms : Swiss syndrome, NAME syndrome (nevi, atrial myxoma, myxoid neurofibroma and neurofibromata, and ephelides), LAMB syndrome (lentigines, atrial myxoma, mucocutaneous myxoma, blue nevi), and myxoma syndrome. In patients with CNC, the myxomas tend to occur in locations other than the left atrium, may be multiple, and have a much higher recurrence rate compared with that in patients who have nonsyndromic myxomas (21% vs. 1%–2%). Moreover, recurrences tend to show more rapid growth and more pronounced local invasiveness. A complete list of the features that differentiate nonsyndromic myxomas from those that arise in the setting of CNC can be found in Table 2.3 . Cutaneous myxomas should not be mistaken for metastatic cardiac myxomas in this population.
| Feature | Comments |
|---|---|
| Male sex: 67% | Compared with 24% for nonfamilial cases |
| Mean age at presentation: 24 yr | Compared with 51 yr for nonfamilial cases |
| Myxomas | |
| Cardiac | Unusual locations, multiple, recurrent |
| Skin | Multiple |
| Breast | Myxoid fibroadenomas |
| Spotty mucocutaneous pigmentation | Scleral and vermillion borders of lips: lentigines, blue nevi, and combinations |
| Psammomatous melanotic schwannomas | |
| Primary pigmented nodular adrenocortical disease | Cushing syndrome |
| Testicular tumors | Characteristically Sertoli cell tumors, usually bilateral and multicentric |
| Pituitary growth hormone–secreting adenomas | Acromegaly or gigantism |
| Features | Nonsyndromic | Syndromic |
|---|---|---|
| Average age (yr) | 51 | 24 |
| Age range (yr) | 17–75 | 4–48 |
| Sex ratio (male:female) | 1 : 3 | 2 : 1 |
| By location a | 86% in left atrium 18% in right atrium |
62% in left atrium 37% in right atrium 21% in right ventricle 4% in left ventricle |
| Multicentric (%) | 6 | 33 |
| Recurrence rate (%) | 3 | 20 |
a Some patients have myxomas in more than one anatomic location.
The vast majority of cases (approximately ≥90%) arise in the atria, with a significant predilection for the septum of the left atrium. Tumors may less commonly arise in the right atrium and much less often, the ventricles. Tumors arising in a location other than the classic left atrium increase the likelihood that the patient has CNC. Although historic debate about this has occurred, myxomas may also originate from any cardiac valve. It is of additional interest that, among the reported valvular cases of such myxomas, a right-sided heart location appears to be more common than a left-sided one and that the tumors may be attached to either the inflow or the outflow surface of the valve.
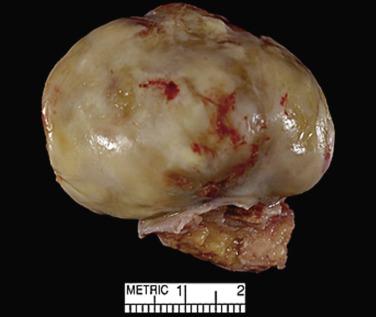
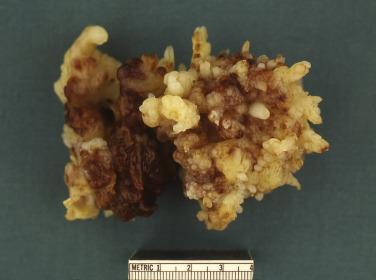
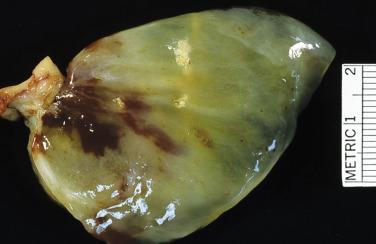
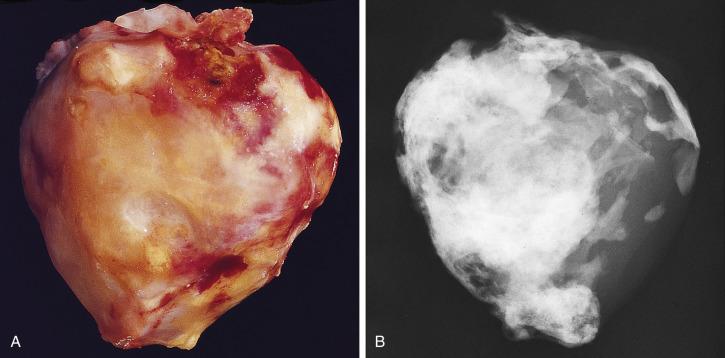
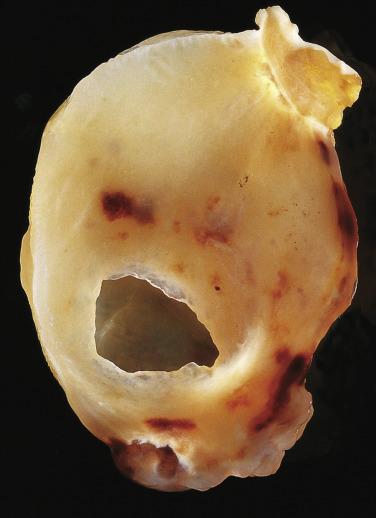
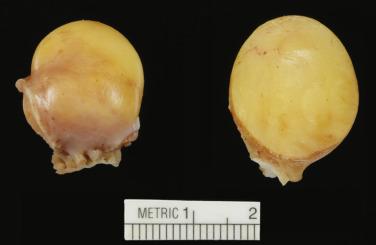
Cardiac myxomas are either pedunculated or sessile. They may be globular, almost round masses with a smooth surface ( Fig. 2.1 ) or villiform with multiple thick papillary fronds ( Fig. 2.2 ). They range from pale gray to dark red, and variegation within a single tumor is common. Occasionally surface thrombus may be present, particularly on villiform tumors. Myxomas are usually soft and friable with a distinctive gelatinous appearance and consistency ( Fig. 2.3 ). Somewhat expectedly, it is the villiform variety that tend to be more friable and have the highest propensity to embolize and be identified in downstream vessels ( Fig. 2.4 ). Some myxomas may be firm, and occasionally gross calcifications may be observed, even to the extent that the bulk of the tumor consists of a calcified mass. This condition is known also as petrified cardiac myxoma ( Figs. 2.5 and 2.6 ) and may be mistaken for an atrial thrombus clinically. Multiple tumors are most often associated with CNC ( Fig. 2.7 ).
The surgeon, aware of the possibility of recurrence after excision, will usually remove the tumor together with its site of origin. Thus in atrial myxoma, the surgical specimen almost certainly will contain a through-and-through segment of either the atrial septum or the atrial wall. When so excised, it is extremely rare to find myxoma at the margin. Recurrences at any rate are rare, provided that excision is complete.
Microscopically, the tumors are dominated by a myxomatous matrix and a dispersed cellular component. The latter consists of different types of cells. The principal cell type, known as the myxoma cell, is considered the neoplastic cell. These cells may appear elongated and fusiform, polyhedral, or stellate ( Fig. 2.8 ). The cytoplasm is mostly homogeneous, is sometimes finely vacuolated, and is usually slightly eosinophilic. The nucleus may be elongated, rounded, or oval, and its staining characteristics may vary from pale to intensely hyperchromatic. Mitoses are virtually absent and, when present in more than an occasional cell, should raise suspicion that the tumor may be a sarcoma rather than myxoma. The large polyhedral myxoma cells are occasionally referred to also as lepidic cells (from the Greek lepis meaning “scale” and based on a rather fanciful resemblance to the scales on butterfly wings). The term was introduced by Orr in 1942, who at the time considered these cells to be of endocardial origin. At the ultrastructural level the cytoplasm of a typical myxoma cell is characterized by scant organelles. Variable numbers are seen of mitochondria, elements of smooth and rough endoplasmic reticulum, and cytoplasmic filaments. The latter consist of two types. The majority are thick (10 nm) nonbranching filaments, often arranged in parallel bundles coursing in various directions. The second type is smaller (6–8 nm) and more irregular in outline.
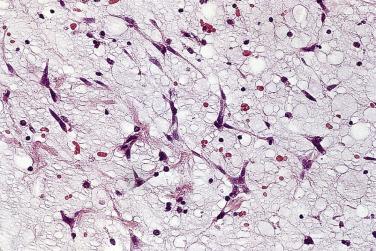
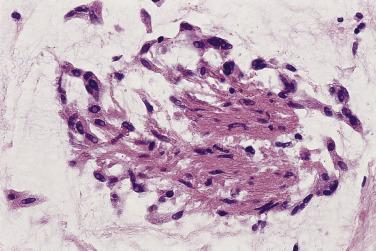
The myxoma cells may assume a variety of patterns. Most commonly they are arranged in single or multiple layers surrounding vascular channels, so-called perivascular rings ( Fig. 2.9 ). These may appear as complex interlacing networks or single strands. These clusters of myxoma cells are often associated with an extensive halo of myxoid extracellular matrix, almost optically empty, which contrasts with the surrounding and slightly more condensed matrix that stains palely eosinophilic ( Fig. 2.10 ; also see Fig. 2.9 ). Other possible arrangements of myxoma cells include the formation of small nests ( Fig. 2.11 ) and as single cells dispersed throughout the myxoid stroma ( Fig. 2.12 ). Multinucleate cells can also be evident, but ultrastructurally they are composed of small groups of closely apposed cells with single nuclei. The surface of the tumor may be covered by a single layer of endothelial cells or may be absent, possibly because of surgical manipulation.
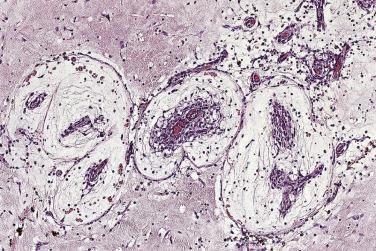
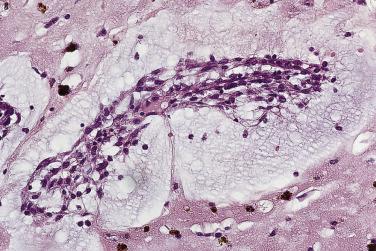
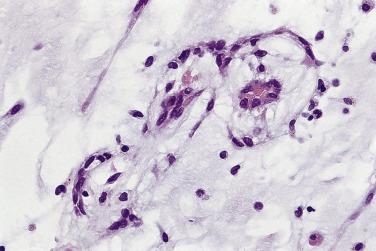
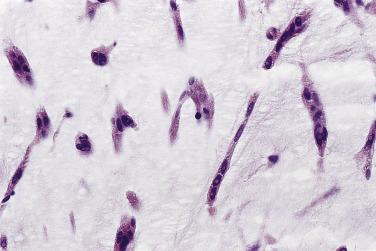
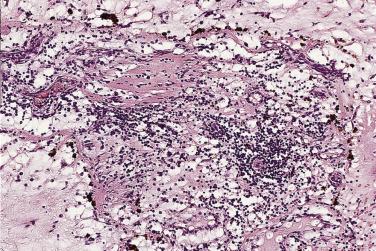
Other cell types that may be present include elongated, spindle-shaped cells that resemble fibroblasts, myofibroblasts, or smooth muscle cells ( Fig. 2.13 ). These cells are usually seen in close association with undisputed myxoma cells and vascular spaces lined by distinct endothelial cells. Macrophages are an almost universal finding in myxomas, often diffusely dispersed throughout the myxoid stroma, although generally condensed toward the base and at sites of hemorrhage. These occasionally coalesce into giant cells ( Fig. 2.14 ). Lymphocytes and plasma cells ( Fig. 2.15 ) may occasionally be prominent. These cellular aggregates occur predominantly in the base of the tumor and can also be found in the adjacent myocardium. Mast cells and foci of extramedullary hematopoiesis may also be present.
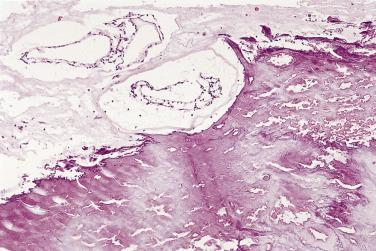
The myxoid stroma itself stains strongly with Alcian blue, unaffected by predigestion with hyaluronidase, and may show patchy reactivity with mucicarmine and the periodic acid–Schiff (PAS) stain, resistant to diastase. It contains variably prominent connective tissue fibers having characteristics of reticulin, collagen, and elastin. Fibrous tissue is most pronounced in the stalk of the tumor, a site often dominated by the presence of large, thick-walled (and occasionally dysplastic) arteries ( Fig. 2.16 ). The “tumor blush” occasionally observed at coronary angiography has its anatomic substrate in this vascular tuft. It is important to emphasize that, like the cellular components, the stroma may also show extensive variability from one part of the tumor to the other. Distinct fibrous areas and foci of liquefaction of the myxoid stroma, leading to cystlike areas, may be seen ( Fig. 2.17 ). These latter changes should not be confused with necrosis or evidence of malignancy.
Hemorrhage within the stroma is an almost universal finding. Although slightly more pronounced in surgical specimens than in autopsy cases, these foci are most likely the consequence of trauma resulting from the mobile nature of the intracavitary tumor. Histologic evidence of old and recent episodes of hemorrhage are manifest by the presence of hemosiderin-laden macrophages ( Fig. 2.18 ) and connective tissue fibers encrusted with iron and calcium (so-called Gamna-Gandy bodies [ Fig. 2.19 ]). Occasionally these can be so prominent that the tumor itself is overshadowed and not easily recognized. It has been suggested that these fibrosclerotic nodules may be related to anticoagulant and/or antiplatelet therapy, but they can be seen even in the absence of such treatment.
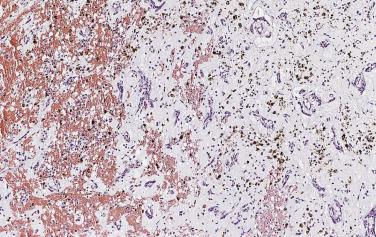
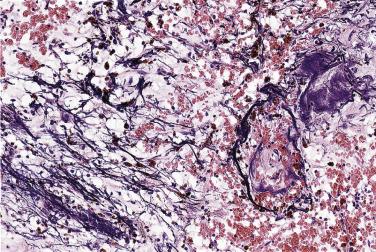
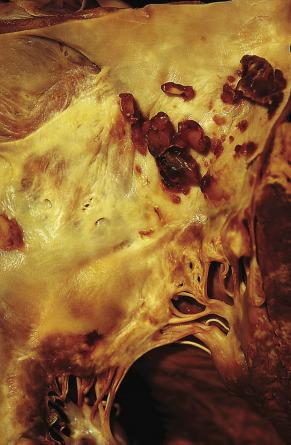
Apart from elastic fiber encrustation, myxomas can exhibit several additional histologic features, such as microscopic foci of calcification. Occasionally dystrophic calcification in the tumor may become so extensive that the tumor is almost completely transformed in a calcified mass (see Figs. 2.5 and 2.6 ).
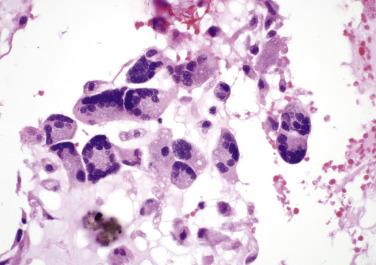
In approximately 5% of cardiac myxomas, glandlike structures occur, lined by cells that may vary from flat to cuboidal or columnar ( Fig. 2.20 ). These cells stain positively with Alcian blue, mucicarmine, and PAS (diastase resistant) and show immunoreactivity with antibodies to cytokeratins and carcinoembryonic antigen. The ultrastructural features of these cells are characteristic for mucin-secreting epithelium. Significant cytologic atypia and mitotic activity may be present and cause concern that such glands may represent metastatic adenocarcinoma. Thymic epithelial rests may also be seen in myxomas (as well as in the myocardium) and may occasionally become so proliferative as to warrant the designation thymoma ( Fig. 2.21 ). The observation that myxomas may contain epithelial as well as mesenchymal elements has suggested to some that they should be classified as hamartomas.
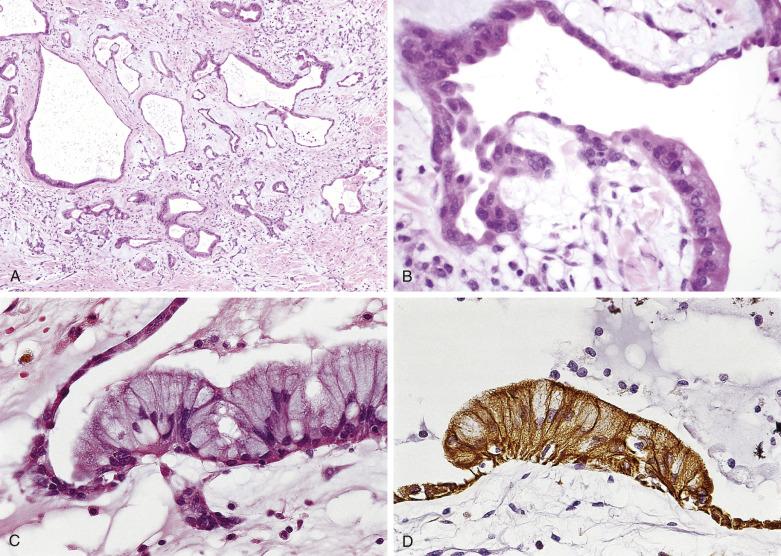
In CNC, myxomas may be less cellular (i.e., more myxoid) than sporadic ones. Valvular myxomas reportedly have fewer cellular clusters, fewer syncytial tumor giant cells, and more infrequent perivascular cuffing of tumor cells than myxomas occurring elsewhere.
Recurrence of a cardiac myxoma after surgical excision is a relatively rare but undisputed phenomenon. It is for this reason that excision of the entire area of attachment of myxoma is recommended. Recurrence is highest in patients with myxoma syndrome, and the development of a myxoma recurrence should prompt investigation into the possibility of CNC.
The potential for malignant degeneration in myxoma is controversial. Most cases of malignant myxomas likely represent cases of misdiagnosis or the inclusion of false metastases based on invasiveness of embolized myxoma fragments, causing ischemic infarction and subsequent infiltration of the arterial wall and/or extracardiac parenchyma. Nevertheless, the possibility of malignant transformation of cardiac myxomas remains a matter of concern with three possible cases having been reported, one with glandular elements. Given that myxoid sarcomas may mimic a myxoma, it would seem that, similar to tumors in other locations (e.g., malignant Brenner tumor of the ovary), the best way to document malignant transformation would be to have benign and clearly malignant elements adjacent to one another, as has rarely been reported.
Reports of lymphoma arising within cardiac myxomas have also been documented. Most of these appear to be Epstein-Barr virus associated and are best classified as so-called diffuse large B-cell lymphoma with chronic inflammation. There is some debate as to the significance of this finding and whether it warrants chemotherapy.
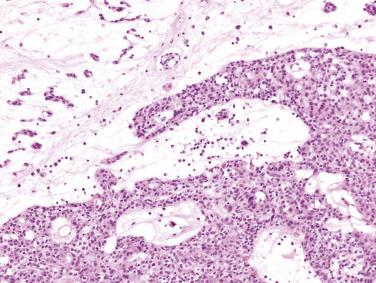
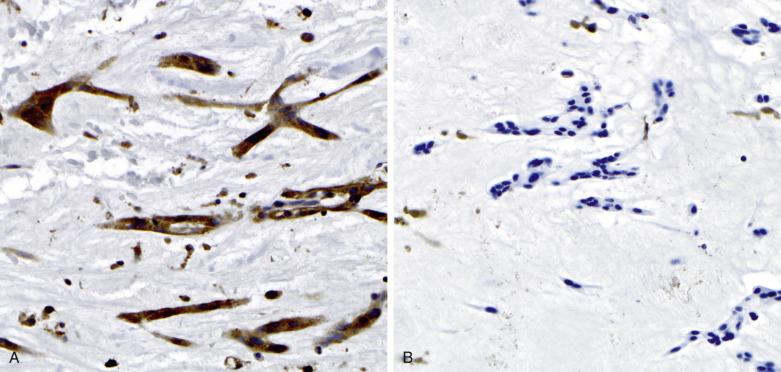
Immunohistochemical studies indicate differences in the phenotypic expression of the various cell types present in these tumors. The myxoma cells are reactive with antibodies to calretinin (75%) and vimentin (50%). Calretinin reactivity within myxoma cells can be particularly useful in differentiating myxoma from other entities, particularly fibroblastic lesions. The vascular channels are lined by endothelial cells that react with antibodies to von Willebrand factor, CD31, or Ulex europaeus agglutinin I. Smooth muscle actin may stain cells that immediately surround the endothelial cells.
PRKAR1A immunohistochemistry may be useful in screening for CNC in those individuals in which cardiac myxoma may be the sole manifestation ( Fig. 2.22A ). Molecular genetic follow-up should be considered in those cases that exhibit loss of PRKAR1A protein within the neoplastic cells.
Cardiac myxomas can potentially be confused with any number of other neoplasms that arise in or involve the heart ( Table 2.4 ). Myxomas can be differentiated from organizing thrombi by the presence of characteristic myxoma cells and matrix. Thrombi on the surface and foci of organizing intratumoral hemorrhage can occasionally make this distinction difficult, and calretinin stains may be helpful in the distinction, as the cells of organizing thrombi fail to react with this antibody. Papillary fibroelastomas have more complex papillary architecture, lack the myxoid stroma, and also have characteristic avascular fibroelastic cores. Myxoid intimal fibroplasia can occur on valvular or endocardial surfaces ( Fig. 2.23 ) and, because of the myxoid stroma, can mimic myxomas; however, the location and a lack of myxoma cells should help in the distinction.
| Differential Diagnosis | Differentiating Features |
|---|---|
| Thrombus | Zonated usually with prominent fibrin Absence of myxoma cells |
| Intimal fibroplasia | Lacks myxoma cells Often previous history of instrumentation |
| Myxoid sarcoma | Pleomorphic spindle cells Often necrotic with high mitotic rate |
| Papillary fibroelastoma | Typically valvular location Endothelial-lined avascular papillary fronds |
| Calcified amorphous tumor of the heart (cardiac CAT) | Prominent calcification Absence of myxoma cells |
| Mesothelial incidental cardiac excrescence (MICE) | Lacks myxoid stroma Often previous history of instrumentation |
| Fibroma | Abundant collagenous stroma Typically ventricular location |
| Hemangioma | Lobular arrangement of vessels Lacks myxoid stroma |
Separation from the variety of myxoid sarcomas is based on characteristics of the stroma, cellularity, mitotic activity, necrosis, and location. The major histopathologic features to distinguish between a benign cardiac myxoma and the myxoid variant of so-called undifferentiated high-grade pleomorphic sarcoma (myxofibrosarcoma) are foci of hypercellularity with pleomorphism, mitotic figures, necrosis, and extensive vascularity in myxofibrosarcoma, features that are strikingly absent in cardiac myxomas. Frequently, the surgeon will not suspect that a tumor presenting as a myxoma is malignant, and it may be the pathologist who is the first to raise the possibility. The rare myxosarcoma (if it is truly different from a myxoid undifferentiated high-grade pleomorphic sarcoma) can be differentiated on the basis of its cellularity, invasive growth, and lack of hemorrhage or myxoma cells. The lepidic cells of a myxoma typically are absent from the pedicle.
Cytogenetic analysis of cardiac myxomas thus far has shown a variety of clonal and nonclonal abnormalities. As it stands now, cytogenetic analysis is of little or no value in the differential diagnosis of cardiac myxomas, although these studies may eventually contribute to understanding their molecular pathogenesis. However, the pathogenetic role of PRKAR1A mutations has proved to be more important.
This is one of the more frequent primary tumors of the heart and by far the most common in infants and children. Only five reports have been made of rhabdomyoma in adults. Cardiac rhabdomyomas have a close association with tuberous sclerosis (see later). A smaller proportion occur sporadically or in association with congenital heart disease. Because of its development in utero, the clinical presentation may differ from that of some other cardiac tumors. In some instances, the tumor may lead to stillbirth or perinatal death, as well as intrauterine myocardial infarction resulting from coronary arterial compression by a large rhabdomyoma. The clinical manifestations of cardiac rhabdomyomas are determined by their size, multiplicity, and location (in relation to the conduction system) and whether they expand into a chamber. In a meta-analysis, Chao and colleagues found that large tumor size and fetal hydrops are significantly associated with poor neonatal outcome.
Echocardiographic studies of patients with tuberous sclerosis reveal a high incidence of cardiac rhabdomyomas. However, a significant difference exists between children and infants. Fenoglio and colleagues reported a 37% incidence of tuberous sclerosis in their autopsy series of patients with cardiac rhabdomyoma. However, many of their cases were stillborns or newborns, and the diagnosis of tuberous sclerosis may be difficult in this age group. A retrospective study in 33 infants and children with cardiac rhabdomyoma collected from three pediatric cardiology centers showed that 91% had tuberous sclerosis. Indeed, Davies suggested that cardiac rhabdomyomas are always accompanied by tuberous sclerosis of the brain, whether the latter condition is clinically manifest. This contention is further strengthened by the observation that in each of five infants, in whom fetal or early postnatal echocardiography revealed a cardiac tumor, tuberous sclerosis was subsequently diagnosed.
Spontaneous regression of rhabdomyomas, initially observed in 1923, is an important phenomenon. A retrospective evaluation of patients with tuberous sclerosis, over a 10-year period (1984–1994), revealed that 63% of patients had a cardiac rhabdomyoma identified at the time of the initial study. However, the number of rhabdomyomas steadily declined with follow-up, so that by age 6 years, they completely disappeared by echocardiography. Hence, once an intracardiac tumor has been detected in an infant who otherwise has no signs or symptoms, the tendency is to not resect in the hope that the tumor will spontaneously regress. Such biologic behavior supports the concept that cardiac rhabdomyomas, whether or not associated with tuberous sclerosis, may be hamartomatous rather than truly neoplastic. Treatment with medications that inhibit the mTOR pathway (everolimus) have been shown to hasten such regression. Genetic studies have linked tuberous sclerosis to the TSC1 gene on chromosome 9q34 and TSC2 on 16q13.3; it has been suggested, on the basis of similar allelic losses in the associated hamartomas, that these genes act as growth suppressors.
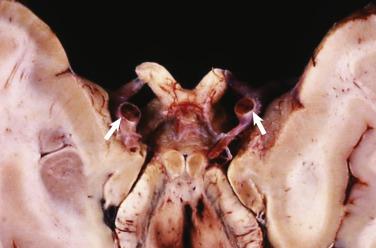
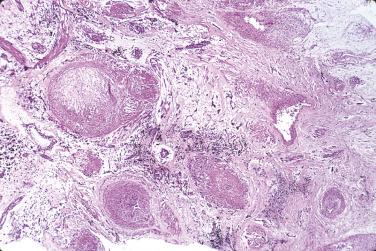
The vast majority of rhabdomyomas are multiple ( Fig. 2.24 ), although this may not be immediately apparent. The lesion has a distinct predilection for the ventricles, with left ventricular involvement in almost 100% and right ventricular involvement in approximately 80% of cases. Involvement of the atria is much less common ( Fig. 2.25 ), ranging from 1% to 30%. Multiple small rhabdomyomas originating from the ventricular aspect of the mitral valve have also been documented. Diffuse rhabdomyomatosis of the heart is exceedingly rare. In this condition the myocardium is diffusely replaced by cells that show the characteristics of rhabdomyoma cells (see later discussion), although small strands and islands of normal-looking myocardium are still present.
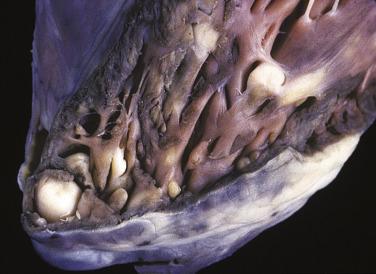
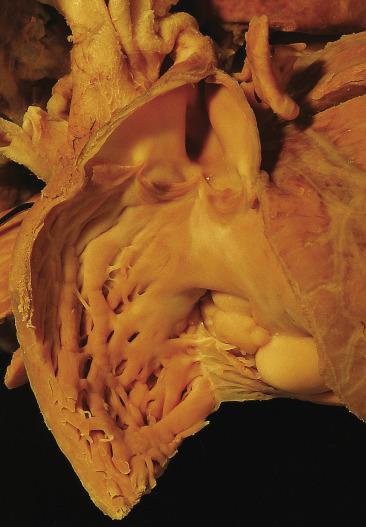
Rhabdomyomas are circumscribed but unencapsulated lesions, easily distinguished from the surrounding myocardium. They have a waxlike consistency and a white to yellow-gray coloration. The size may vary from millimeters to several centimeters. Occasionally the lesions may grow to astounding proportions, with intracavitary extension (see Fig. 2.25 ) that can obliterate cardiac chambers.
The histopathologic features of cardiac rhabdomyomas are distinct. The tissue is composed of markedly swollen myocytes, showing almost empty cytoplasm with a centrally placed cytoplasmic mass and nucleus ( Fig. 2.26 ). Cytoplasmic strands extend from the nucleus to the periphery of the cell, hence the term spider cell ( Fig. 2.27 ). The myocytes may appear quite bizarre and sometimes reach monstrous sizes of up to 80 µm in diameter ( Fig. 2.28 ). At high magnification, striations are often seen in the tiny strands. The stromal component is usually scanty, although occasionally areas with distinct collagen may occur. In some cases calcifications can be seen associated with foci of necrosis. The older the patient, the more likely extensive calcification is present. The lesions have a low proliferative rate, although those in adults may be more mitotic. Diagnosis on endomyocardial biopsy is possible.
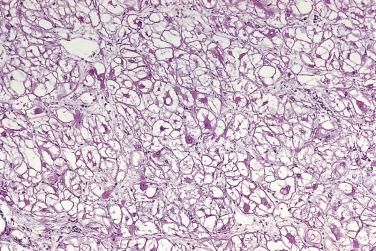
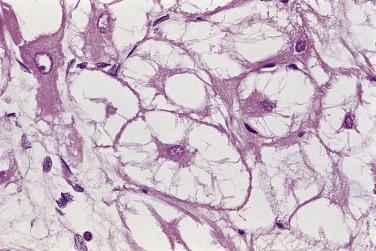
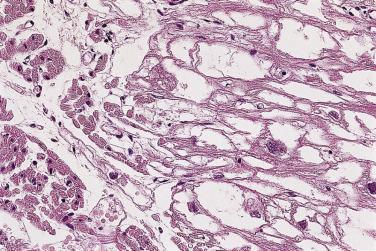
The cells contain copious glycogen, best demonstrated with the PAS method on alcohol-fixed or on frozen-tissue sections. The type of polysaccharide involved is rather labile (diastase sensitive), a striking contrast with that present in glycogen storage disease (diastase resistant).
Rhabdomyoma cells are reactive for muscle markers, including myoglobin, actin, desmin, and vimentin. They may also react with antibodies to HMB45, an interesting observation given their association with tuberous sclerosis, but not S-100 protein. Spider cells are also reactive with antibodies to ubiquitin, a trafficking protein that plays a role in apoptosis, providing a possible explanation for spontaneous regression. Ultrastructural studies confirm the myogenic nature of the cells involved.
The histopathologic diagnosis should not normally be a problem because of the clinical setting and the bizarre appearance of the swollen myocytes. Cardiac rhabdomyoma should not be confused with other types of rhabdomyoma, which generally do not occur within the heart, with the exception of rare reports of cellular rhabdomyoma and a single case of adult rhabdomyoma. Although spider cells are described in adult rhabdomyoma, they are few and admixed with large, densely eosinophilic polygonal cells. Distinction from glycogen storage disease may occasionally pose a problem. However, in the latter, the myocytes are usually not so large, nor do they show the spider appearance, although they appear empty with a peripheral rim of myofibrils. Finally, cardiac rhabdomyomas should not be confused with the hamartoma of mature cardiac myocytes, which lacks the cytoplasmic vacuolation of a rhabdomyoma.
The cardiac fibroma is generally a tumor of infants and children, although it also occurs in adults (15%), with one of the oldest being 77 years of age (see Box 2.1 ). Gorlin syndrome (multiple nevoid basal cell carcinomas of the skin, jaw cysts, with bifid ribs) may be present in up to 4% of patients with cardiac fibroma. This is due to PTCH1 mutations, and loss of the PTCH1 locus has been reported in a sporadic cardiac fibroma. Structural rearrangements of chromosome 9q22, corresponding to the PTCH1 locus, have also been described. Cardiac fibroma has also been associated with the Beckwith-Wiedemann and Sotos syndromes.
Cardiac fibromas present as circumscribed solid, firm, white lesions, which are clearly demarcated from the surrounding myocardium and range from 2 to 10 cm ( Fig. 2.29 ). Rare tumors may be as large as the heart and still be asymptomatic. Although grossly circumscribed, the tumors are somewhat infiltrative microscopically (see later). The cut surface reveals the fibrous nature of the lesion ( Fig. 2.30 ). Occasionally calcifications may be apparent.
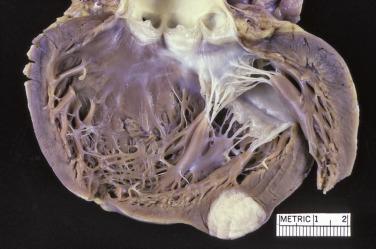

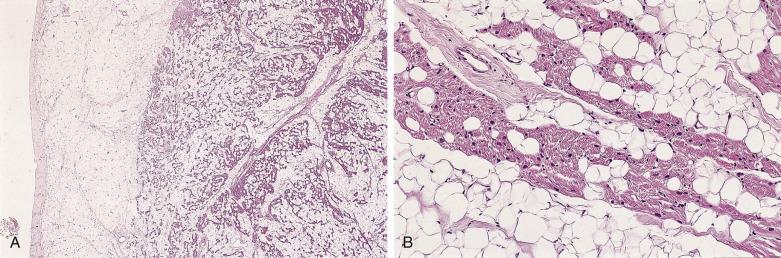
Fibromas are composed of bland spindle cells and dense connective tissue composed mostly of collagen ( Fig. 2.31A ). Generally speaking, lesions observed in younger patients (<1 year old) are often more cellular whereas those in older patients contain proportionally more collagen (see Fig. 2.31B ). At the periphery, the fibroma intermingles with the surrounding myocardium, being less circumscribed than the gross appearance might suggest ( Fig. 2.32 ). Occasionally, myocytes may be intermingled in the more central portions of the tumor. Individual cells may be difficult to distinguish but are elongated bland fibroblasts. Mitotic figures are extremely rare, even in areas that are distinctly cellular. Elastin fibers may sometimes be present, leading to the former term fibroelastic hamartoma, but a fibroma should not be confused with papillary fibroelastoma. Microscopic foci of calcification may often be observed ( Fig. 2.33 ) and, when seen on imaging, offer a diagnostic clue. Centrally, the lesions may become either cystic or hyalinized. Small groups of chronic inflammatory cells may be present around blood vessels.
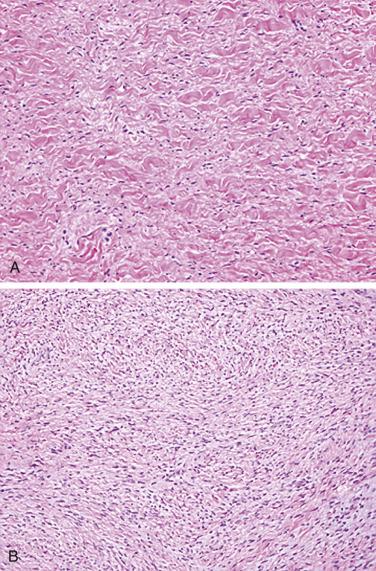
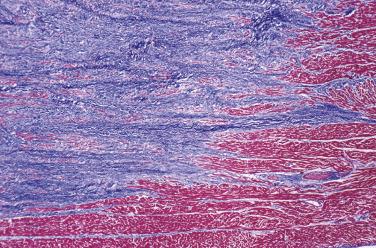
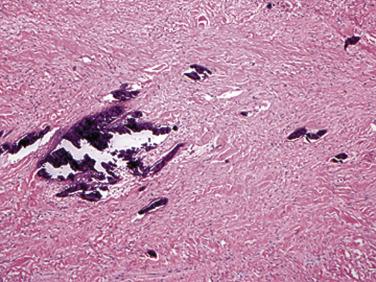
Immunohistochemically, most cells express vimentin and α-smooth muscle actin. Ultrastructural studies have shown that the principal cellular component of the cardiac fibroma is the fibroblast, set in a matrix composed of glycosaminoglycans and collagen. Myofibroblasts may be intermingled. Sometimes the tumors may not be completely resected, and fibroma cells may be present at the margin. Even so, they typically do not recur.
Because of the age of most patients with cardiac fibroma, the differential diagnosis is limited. The relatively paucicellular nature of the tumors and low-grade cytology, as well as lack of necrosis, help to differentiate them from fibrosarcomas. The infiltrative nature of the spindle cells, however, brings desmoid-type fibromatosis into the differential diagnosis. This is of more than academic interest as fibromatoses recur and cardiac fibromas do not. However, no β-catenin ( CTTNB1 ) mutations have been identified in cardiac fibroma, and they lack the nuclear staining characteristic of fibromatoses.
Epicardial fat stores increase with advancing age. They have a characteristic distribution on the anterior surface of the right ventricle and along the course of the epicardial coronary arteries, but are usually not mass (tumor) forming. The classification of benign lipomatous processes of the heart includes true neoplasms, diffuse lipomatoses (see also Chapter 24 ), and fat in the atrial septum forming a mass (so-called lipomatous hypertrophy of the atrial septum [LHAS]). An association with tuberous sclerosis has been documented for some true lipomas. LHAS is likely a result of entrapped embryonic fat during atrial septation and appears related to body mass, but some cases harbor cytogenetic alterations of lipomas indicating that at least a subset may be truly neoplastic. It is often identified incidentally in older adults. The association of cardiac arrhythmias and sudden cardiac death in cases of LHAS is well established.
Lipomatous hamartoma of an atrioventricular (AV) valve is another extremely rare condition of which fewer than 10 examples have been documented ; lesions have affected both the mitral and the tricuspid valves, in individuals 2 to 76 years old. Valvular insufficiency may occur, attributable to lipomatous change in the papillary muscle. Finally, at least two cases of hibernoma of the heart and pericardium have been reported.
The gross pathology depends highly on the type of lipoma. In the case of a true lipoma, the gross morphology will be identical to that seen with lipomas in other locations ( Fig. 2.34 ).
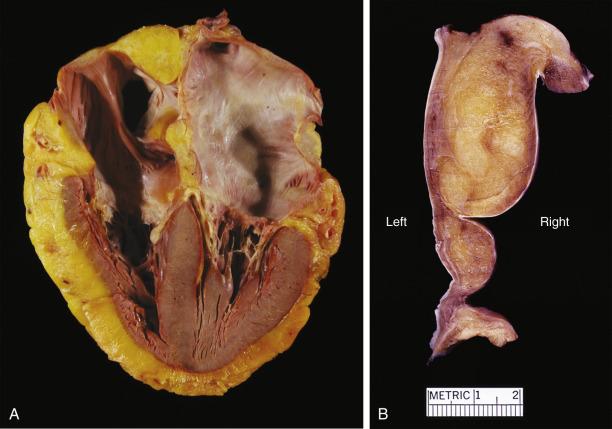
When encountered in an autopsy, explant, or (rarely) surgical pathology specimens, LHAS presents as a distinct bulge of the posterior limbus, directly adjacent to the oval fossa ( Fig. 2.35 ). The valve of the fossa ovalis may occasionally be barely visible as it may be masked by the protruding atrial wall. In fact, the protruding wall can be so pronounced as to cause vena caval obstruction. On the cut surface the interatrial groove may appear extremely thickened (even >2 cm) and composed of yellow adipose tissue, often with a brown tinge, infiltrating into adjacent atrial myocardium. Close inspection will usually reveal strands of heart muscle amid the adipose tissue. The posterior portion of the atrial mass is usually thicker than the anterior portion, and the valve is generally spared, giving it a characteristic “dumbbell” or “bilobed” appearance (see Fig. 2.35 ). Extensive fat can occasionally be seen in the AV valve and has been termed lipomatous hamartoma of the atrioventricular valve ( Fig. 2.36 ).
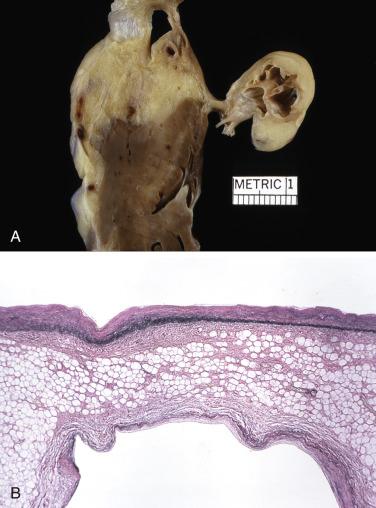
The histopathology of lipomas, defined as circumscribed encapsulated tumors, is no different from that encountered in lipomas elsewhere in the body ( Fig. 2.37 ). Mature adipose tissue and occasional muscle cells may be encountered, and variably admixed fibrous tissue and vessels may be seen, leading to subclassification of such tumors as myolipoma, fibrolipoma, and angiolipoma.
In LHAS the histology is dominated by massive infiltration of mature fat cells with displacement of preexisting myocardial cells ( Fig. 2.38A ). The latter can be either atrophic or large and atypical and suggest the possibility of malignancy. Usually the best-preserved parts of the myocardium are subendocardial in location, but islands of myocytes are trapped in the lipoma and often appear to be totally surrounded by fatty tissue. Occasionally, areas can be traced where mature fat cells intermingle with vacuolated brown fat (see Fig. 2.38B ). In addition, granular eosinophilic cells may be seen, which resemble fetal fat cells by light and electron microscopy.
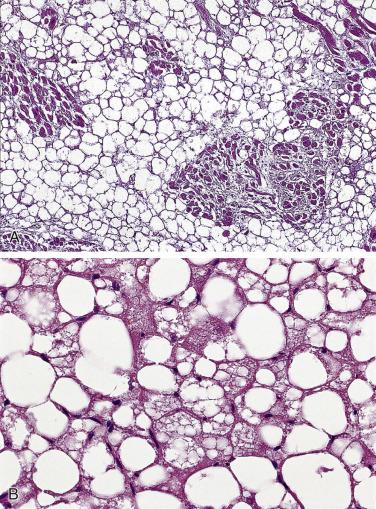
Lipomatous hamartomas of cardiac valves show diffuse infiltration by mature fat cells, with almost complete replacement of the preexisting tissue architecture of the valve leaflet.
While myocardial lipomas are diagnosed with relative ease, an excessive increase of epicardial fat is usually accompanied by fatty infiltration of the right ventricular myocardium and the interatrial groove, raising the question as to what amount of epicardial adipose tissue should lead to a diagnosis of lipoma. Generally speaking, if an epicardial collection of mass is distinctly mass forming, it can be referred to as a lipoma. Because of the presence of myocytes intermingled with adipocytes in LHAS and the resemblance of some of the multivacuolated cells to lipoblasts, liposarcoma may occasionally enter the differential diagnosis. This concern may be furthered by the surgical findings of what appears to be a grossly infiltrative mass. Lack of nuclear atypia and knowledge of the location, however, is usually sufficient to allow accurate classification.
This lesion represents the most commonly resected cardiac mass in many centers, owing to increased sensitivity of modern imaging. They occur almost exclusively in adults, with only an occasional report in an infant or child. Papillary fibroelastomas may arise on any endocardial surface, although left-sided valves are the most common location. Valves with abnormal hemodynamics (e.g., chronic rheumatic disease) appear to be affected disproportionately. A subset of cases appear to be iatrogenic in origin (prior surgery or radiation), and such lesions may be multiple. Occasionally they may produce clinical signs and symptoms, including cerebral embolization. Location on the aortic valve may lead to systemic embolization, potentially leading to either cerebral or cardiac ischemic/infarction (from either the tumor or thrombus). As with several cardiac tumors, it is not clear whether these lesions should be regarded as neoplasms or reactive growths, but recent data favor a neoplastic process since as many as 70% appear to harbor KRAS mutations.
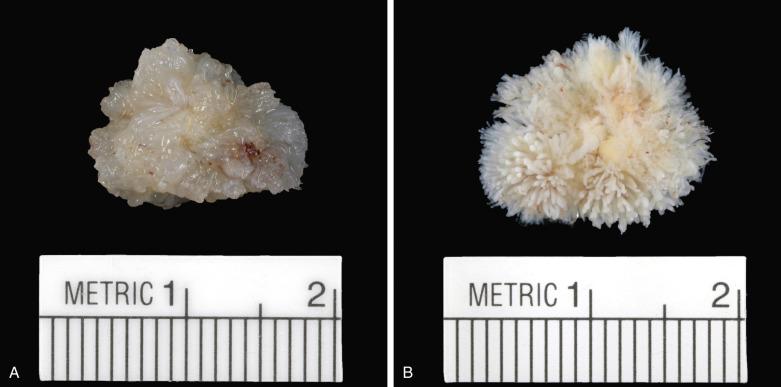
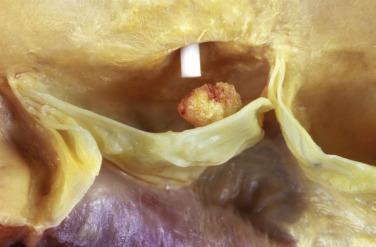
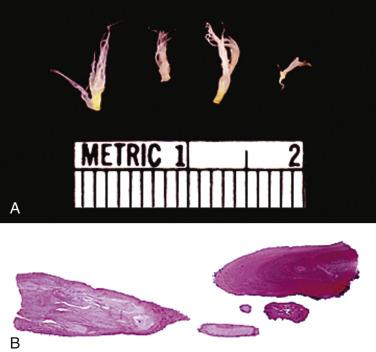
These tumors have the appearance of a sea anemone, particularly when examined in solution ( Fig. 2.39 ). They consist of a bouquet of filiform projections attached to the endocardium, being either sessile or connected by a distinct but short pedicle. They may be very small, no more than a few millimeters in diameter, or quite large, with a diameter up to 6 cm. These tumors most frequently arise on the valves, with a distinct predilection for the aortic valve ( Fig. 2.40 ), but may also arise from the atrial or ventricular endocardium. On the mitral and tricuspid valves the lesions are usually found on the atrial aspect, often near the midportion. On the aortic and pulmonary valves these papillary tumors occur with near-equal frequency on the ventricular and the arterial sides of the cusp. Occasionally, a patient may have multiple fibroelastomas ( Fig. 2.41 ).

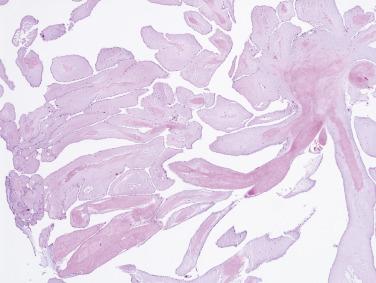
These tumors are papillary, as the name implies ( Fig. 2.42 ). The fronds consist of a central avascular core of dense, almost acellular collagen, occasionally surrounded by a myxomatous matrix. One case has been reported with chondroid metaplasia of the fibrous core. The peripheral rim and/or core contains coarse and fragmented elastic fibers ( Fig. 2.43 ). The surface lining consists of a layer of endothelial cells, which may appear hyperplastic. The central core of collagen is continuous with that of the subendothelium. The amount and distribution of elastin vary considerably among the various fronds in a single tumor, but usually elastic stains highlight their presence.
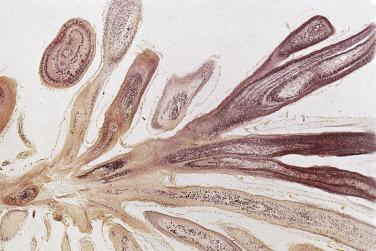
The cells covering the surface of papillary fibroelastomas are positive for vimentin, factor VIII–related antigen, and CD34, in keeping with their presumed vascular endothelial origin. Interestingly, the surface lining cells have also been reported to be positive for S-100 protein. Collagen type IV shows multilayered linear staining beneath the surface, virtually identical to the staining pattern for elastic tissue.
Grossly, papillary fibroelastomas may look somewhat myxoid and (without immersion in water) the papillary fronds may not be evident, causing one to consider a diagnosis of cardiac myxoma. Histologically, however, the similarities with cardiac myxoma are limited to the collar of loose connective tissue surrounding the central cores of dense collagen and its prominent surface of endothelial cells (see Fig. 2.42 ), but this should not pose a differential diagnostic problem as fibroelastomas are virtually acellular and do not contain myxoma cells. Myxomas also tend to lack the fine papillae of these lesions. Lambl excrescences, which exist on a spectrum with the papillary fibroelastoma, also have a core of elastic tissue, but are architecturally simpler, usually comprising a single or bifid frond ( Fig. 2.44 ). Additionally, Lambl excrescences are found exclusively along the lines of closure on semilunar valves and are not seen on AV valves.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here