Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Benign and malignant tumors of the hand arise from distinct tissue types.
The majority of these tumors are benign.
Accurate assessment, diagnosis, and treatment will optimize clinical outcomes.
Reconstructive procedures of the hand and upper extremity should be performed only after the diagnosis is confirmed and appropriate surgical margins are achieved.
Most tumors of the hand are benign, recognized early, and treated by excision.
Ninety-five percent of hand tumors that do not involve the skin are benign.
Malignant tumors of the hand can be divided into two categories: primary and metastatic. Primary tumors can arise from the skin (e.g., melanoma, basal, and squamous cell carcinoma), soft tissues (e.g., sarcoma), or bone (e.g., osteosarcoma). Metastatic disease originates most commonly from cancers of the breast, kidney, thyroid, lung, and colon.
Appropriate evaluation, diagnosis, and treatment for tumors of the hand are required for optimal patient care. A careful history and physical examination will rapidly focus the investigation of a suspicious mass found in the hand.
Magnetic resonance imaging (MRI) has become the “gold standard” to evaluate soft-tissue masses for malignancy. Computed tomography (CT) is preferred for osseous lesions.
Incisional or excisional biopsy is required for definitive diagnosis in many cases.
Surgical incisions must be planned carefully with definitive surgery in mind, utilizing a longitudinal incision in line with or parallel to a potential limb salvage procedure.
Reconstructive procedures, in the setting of a neoplasm of the hand, are performed only after the final pathological diagnosis and clear resection margins are established.
An understanding of both oncologic and reconstructive principles is required in order to achieve an optimal clinical outcome.
Most hand tumors:
arise from any cell type
are benign
are treated with a favorable prognosis.
When in doubt:
perform excisional biopsy
plan incisions carefully
keep definitive surgery in mind.
Tumors of the hand can arise from the skin, adipose tissue, synovium, tendons, cartilage, bones, muscles, fibrous tissue, nerves, and blood vessels. The majority of hand tumors are benign, most are recognized early, and the prognosis is typically good. Malignant tumors can rarely occur in the hand and can be divided into two categories: primary and metastatic. Additionally, premalignant lesions such as actinic keratoses and atypical nevi can occur on the hand.
In this chapter, benign and malignant tumors are classified and discussed by their tissues of origin. Understanding the origin of hand lesions will assist in accurate diagnosis and appropriate therapeutic interventions.
The management of hand tumors requires the hand surgeon to function as both oncologic surgeon and reconstructive surgeon. A complete understanding of both oncologic and reconstructive principles is therefore required to achieve an optimal outcome. The role of the oncologic surgeon is to eradicate the tumor completely, which can compromise both aesthetics and function. In contrast, the role of the reconstructive surgeon is to optimize hand function. Balancing these sometimes competing goals can be challenging. In management of particularly complex or atypical cases, the authors advocate multidisciplinary team care which should necessarily include surgical and/or musculoskeletal oncologic colleages.
An effective strategy to evaluate, diagnose, and treat tumors of the hand and associated upper extremity is required for optimal patient care. A systematic approach with a high index of suspicion for complex disease is warranted. A proposed algorithm is presented here:
Have a diagnostic tree when evaluating any mass.
Recognize aggressive features.
Image thoughtfully, typically with radiographs, CT, and/or MRI.
Perform a biopsy which avoids tumor spillage and facilitates limb preservation.
Involve multidisciplinary team members when necessary.
Perform oncologically sound extirpative surgery.
Reconstruct form and function.
Patients often present with a preliminary diagnosis of “ganglion cyst” or “bone spur” from referring physicians. It is imperative to disregard assumed diagnoses and perform a complete evaluation. While most hand masses are benign, one cannot overstate the importance of recognizing malignancies and other unusual cases early in the course of treatment and evaluation. Patients frequently present in a delayed fashion, particularly with malignancies, as they may have been asymptomatic, misdiagnosed, or experienced some element of denial/neglect.
A thorough history and physical examination remain the foundation of proper initial diagnosis. Questions concerning the history of the tumor are solicited. These include duration, changes in size or color, associated pain, and occurrence of ulceration. Pain can signify a malignancy or a mass that has encroached upon neurologic structures. The sensitivity of the mass to cold or heat needs to be clarified, as the former typifies a glomus tumor.
Inquiring about risk factors for tumors of the hand will inform the differential diagnosis. The patient should be questioned about a history of cutaneous malignancies, extensive sun exposure or sunburns as a child, chemical and ionizing radiation exposure, and trauma or infections. Screening for non-cutaneous malignancies must also be performed, as metastatic disease can present in the upper extremity, most commonly from breast, kidney, thyroid, lung, and colon cancers. The patient should also be questioned about rheumatologic conditions, such as gout, psoriasis, and rheumatoid arthritis.
The age of the patient should be considered when evaluating a mass in the hand. Certain masses are specific to certain age groups. For instance, bone cysts are typically seen in adolescents and young adults, whereas metastatic tumors in the hand are rare under the age of 50 years. Conversly, if a “typical” mass is present at an “atypical” age, clinical suspicion should be high for a more serious disease process.
Questions about previous biopsies or excisions will complete a thorough historical evaluation of the patient with a hand tumor. Pathology specimen slides and reports for previous biopsies or excisions will need to be obtained and reviewed. Prior operative reports will help clarify the diagnosis and optimize future surgical interventions in instances in which the current pathology is unknown.
Rheumatoid arthritis – rheumatoid nodule
Gout – tophus
Severe and unrelenting pain - suggestive of malignancy
Cold sensitivity: subungual lesion – glomus tumor
Pain relieved by nonsteroidal anti-inflammatory drugs (NSAIDs) – osteoid osteoma
Trauma – epidermal inclusion cyst or atypical infection
The physical examination of the hand involves a complete examination of the skin, tendons, muscles, ligaments, bones, and neurovascular structures. An evaluation of the regional lymph nodes for adenopathy is important for assessing the malignant potential of a hand mass. Color and texture changes are noted, and an examination for ulceration, erythema, and edema is performed. Transillumination of the mass can be helpful in distinguishing between solid and cystic etiologies. The lesion is palpated, and the mass size is appreciated along with its shape. The mobility of the mass is assessed to determine whether it is fixed to underlying anatomic structures, and mobility may differ depending on the direction of displacement. Having the patient flex and extend the fingers and wrist will further delineate whether the lesion is associated with tendons or deeper structures within the joints. A complete vascular examination with palpation of pulses is required. If pulses are not palpable, an Allen’s test using a Doppler can be used to define the vascular status of the hand and upper extremity. Neurologic testing of fine and gross motor and sensory function is also performed.
When benign lesions are to be followed clinically, photographic documentation is an essential tool to judge the rate of growth over time. Measuements and photographs are also recommended for patients with multiple and subtle lesions.
Laboratory studies are helpful in determining the etiology of some hand tumors. Workup should be targeted based upon a differential diagnosis developed after examination. Labs can often help differentiate infectious from non-infectious etiologies. As a basic principle, even in the case of normal labs, presumed neoplasms without a clear diagnosis shoud be cultured, and those presumed infectious should undergo pathologic examination.
Elevated white blood cell (WBC) count and inflammatory markers can support an infectious etiology, but it should be noted that not all infectious processes of the hand will generate a substantial leukocytosis. Normal inflammatory labs do not rule out processes such as osteomyelitis. Serum calcium, phosphorus, and alkaline phosphatase levels are often increased in patients with metastatic tumors, and alkaline phosphatase is also increased in patients with osteosarcoma. An erythrocyte sedimentation rate assesses for inflammation; the rate is often increased in Ewing's sarcoma, lymphoma, and myeloma. In male patients older than 50 years with blastic hand lesions on radiography, a serum prostate-specific antigen (PSA) test should be performed to investigate the possibility of metastatic prostate cancer.
Increased ESR: Ewing's sarcoma, lymphoma, myeloma
Increased Ca ++ : metastatic tumors
Increased alkaline phosphatase: osteosarcoma
Males, age >50, and blastic bone lesions: check PSA
Increased WBC: Indicates infection, but infectious etiologies are not ruled out in the absence of elevated WBC
Multiple radiologic modalities are available for imaging benign and malignant hand lesions and masses. Radiographs are not essential for most skin lesions. However, radiographs are required for very large skin lesions when there is clinical suspicons of possible bony erosion/invasion.
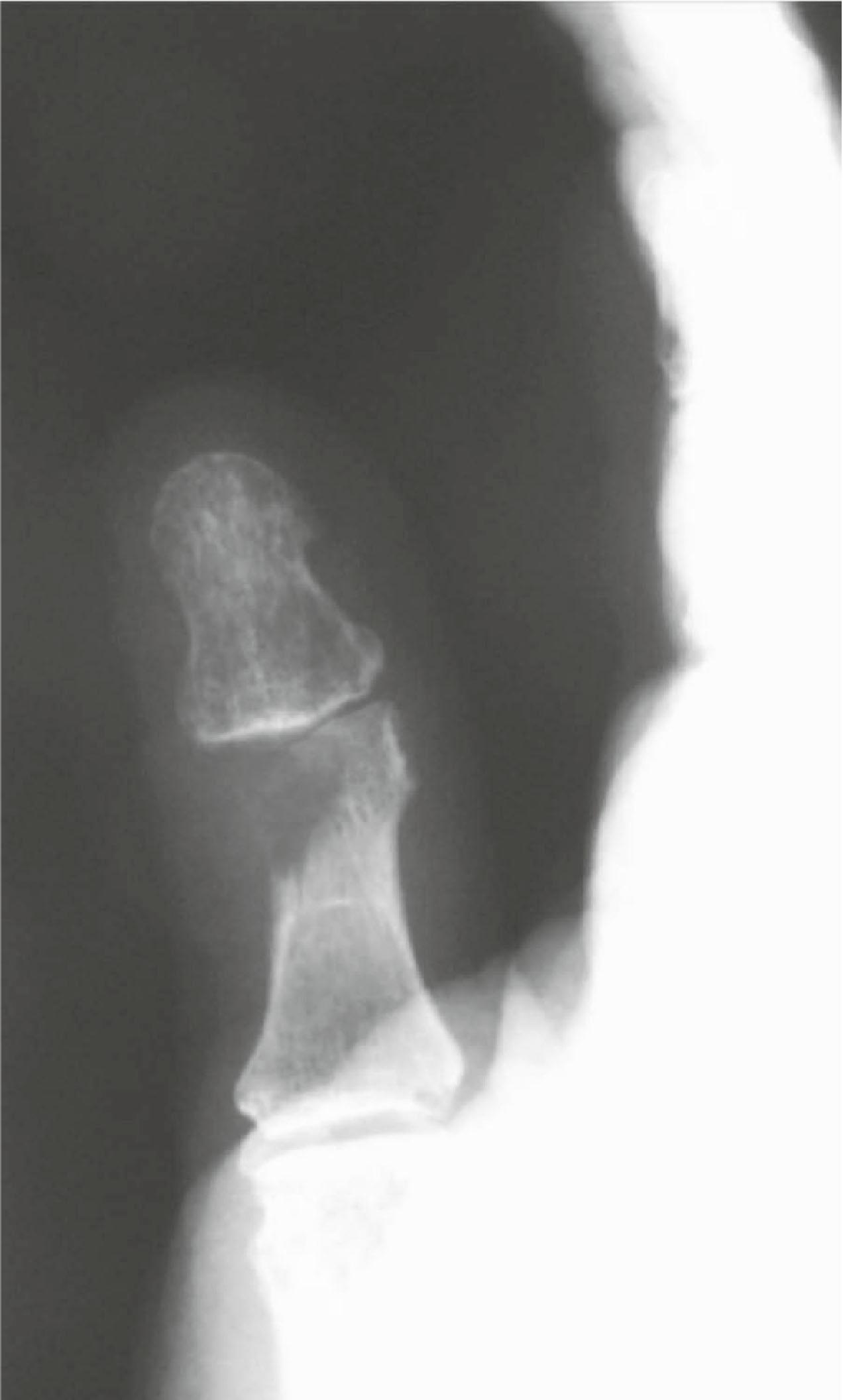
The architecture of the mass as it relates to the cortex of the associated bone can be determined rapidly by X-ray. Sharp cortical margins usually indicate a benign process, while a “moth-eaten” or destroyed cortex often indicates a malignant process. Erosions and periosteal elevation on X-rays can signify a potential malignancy or an infectious process. Soft-tissue calcifications may signify a malignancy ( Fig. 16.1 ).
Ultrasonography can be helpful in assessing soft-tissue masses. This technique is noninvasive and inexpensive. Ultrasonography can determine if a tumor is solid or cystic and differentiate between a discrete mass and diffuse edema. In many instances, ultrasonography can be used to guide needle biopsy. This is particularly useful in the pedatric population to limit radiation exposure and the need for sedation, which is often needed to facilitate cross-sectional imaging in this population.
Scintigraphy or bone scanning is beneficial in screening for skeletal masses. This technique is sensitive in isolating abnormalities, but the findings are not very specific for malignancy. For example, there is intense uptake with osteoid osteoma, a benign bone tumor. Bone scans are very helpful when searching for sources of metastasis in the workup of hand and upper extremity primary malignancies. With the emergence of routine cross-sectional imaging techniques, this modality is becoming less commonly used for diagnosis.
CT is extremely useful for the assessment of bones and cortical destruction. CT scans have markedly greater bony resolution than standard radiographs. If bone involvement by the mass is equivocal on X-ray, CT is the next appropriate radiographic modality. CT also distinguishes calcification patterns from ossification and is superior in assessing periosteal versus endosteal reactions.
MRI is superior to CT for the evaluation of soft-tissue masses. It has become the gold standard to evaluate soft-tissue masses for malignancy. The MRI study should include T1-weighted, fat-suppressed T2-weighted, and short tau inversion recovery (STIR) images. The contrast agent gadolinium can further enhance the visualization of soft-tissue tumors. An MRI study obtained with various views can clearly delineate the extent of soft-tissue involvement prior to operative intervention. A drawback of MRI is that it cannot reliably distinguish between benign and malignant processes. In addition, for MRI of hand masses, a dedicated hand coil is required.
Based upon the clinical, imaging, and lab workup, the surgeon will work with the patient to choose a course of watchful waiting versus need for biopsy. The goal of biopsy is to obtain an accurate sample of tissue, facilitate treatment of the tumor if needed, without compromising definitive treatment. While no evidence-based guidelines exist for exactly which masses require biopsy, a general rule of thumb is to biopsy any mass that is symptomatic or growing. Various methods of obtaining biopsies exist, including punch biopsy, core needle (interventional radiology assisted), open incisional, or open excisional. Each biopsy option has its advantages and disadvantages. Proper technique impacts the quality of results.
When navigating through methods of biopsy, there are important principles to keep in mind. Masses should be characterized as well-defined or ill-defined based on clinical history, exam, and imaging results. Simple masses localized to the skin only can be sampled with a punch biopsy. Masses that are smaller (<2 cm), distal to the metacarpophalangeal joint, mobile, and transilluminate are more likely amenable to excisional biopsy. Masses that are large (>3 cm), proximal to the metacarpophalangeal joint, progressive in size, painful, ill-defined, in an atypical location, or with overlying skin changes are amenable to core needle biopsy or open incisional biopsy. Particularly difficult masses or those with high suspicion for malignancy may benefit from multidisciplinary consultation with a musculoskeletal radiologist and interventional radiologist to assist in biopsy.
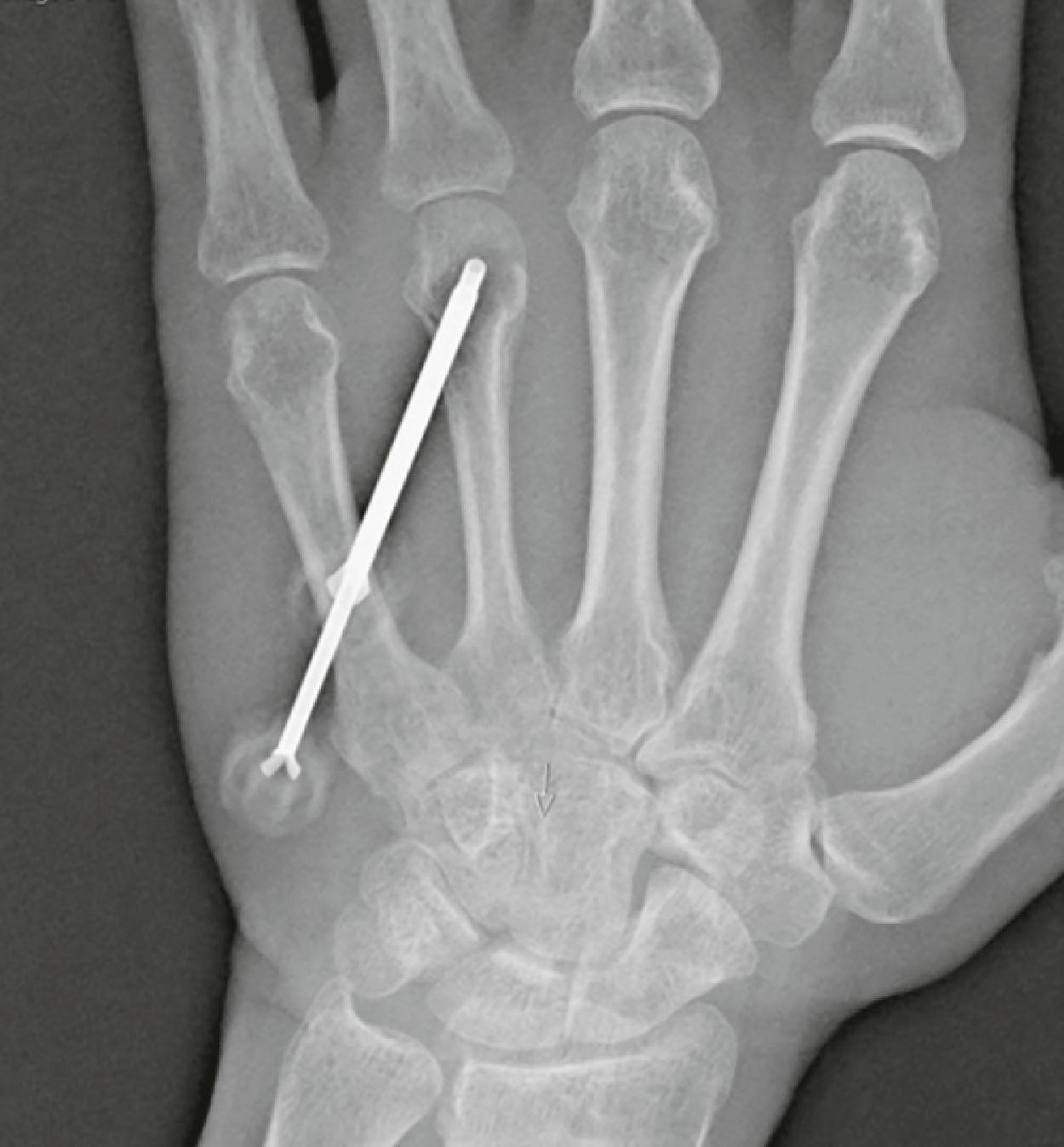
Punch biopsy is performed with a hollow circular-shaped sharp instrument that allows en bloc resection to the desired depth of tissue based on pressure. The mass can then be harvested directly from the biopsy device and sent for pathologic examination. In a closed or core needle biopsy, a needle or trephine is used to obtain samples. Core needle biopsy is typically indicated for masses in difficult locations (intra-osseous lesions, deeper tissues). This technique is favored due to its high accuracy, low likelihood of misdiagnosis, low cost, and minimally invasive nature. These biopsies must be coordinated with a musculokeletal/interventional radiologist to optimize results ( Figs. 16.2 & 16.3 ). While useful in metastatic workup, tissue samples from closed biopsy are sometimes inadequate for initial diagnosis in cases where the tumor is heterogeneous, or the pathology is unclear.

Open biopsies should be performed by hand surgeons who are familiar with the principles of musculoskeletal oncology and target anatomy. Longitudinal incisions should be in line with or parallel to incisions that would be used in a later limb salvage procedure if the tumor proves to be malignant. If a tourniquet is used, the upper extremity must not be exsanguinated because doing so may cause spread of malignant cells into the lymphatics. Meticulous hemostasis should be maintained, and adjacent anatomic compartments should not be violated. An important tenet is to biopsy all infections and culture all masses. Chronic infections can masquerade as malignancies, and masses can result from subclinical infections.
Open excisional biopsies are performed by making an incision directly over the mass of interest, dissecting the mass from surrounding soft tissues and removing it. It is important to note that adjacent tissue is not removed and cells from the mass may remain within the excision site. If the mass proves to be benign, this excisional biopsy is the definitive procedure. If a malignancy is identified in this way, resection with margins is usually indicated. Open incisional biopsies are done as a precursor to formal resection of masses, with the goal of identifying origin of masses prior to further definitive surgical planning without contaminating tissue planes. Through a longitudinal incision and abiding by the above-mentioned principles, a piece of the mass of interest is removed for pathologic examination. This technique may also be employed after an indeterminate or inconclusive closed needle biopsy.
Once a biopsy has been obtained, the type of specimen sent must be carefully selected, as the accuracy of pathologic assessment is dependent on the type of specimen obtained. A frozen-section analysis can be performed to assess the adequacy of the tissue sample. However, the accuracy of frozen-section diagnosis is only 80%, whereas permanent-section diagnosis is 96% accurate.
Punch
Core needle (assisted by interventional radiologist): difficult location, deeper
Incisional biopsy: larger (>3 cm), proximal to metacarpophalangeal joint
Excisional biopsy: small (<2 cm), distal to metacarpophalangeal joint, likely benign
Goal: Obtain an accurate tissue specimen without impacting definitive care
Use clinical history and exam to risk stratify masses
Use longitudinal incisions
Do not use Esmarch for exsanguination
Avoid violating adjacent compartments
Split muscle fibers longitudinally
Use frozen sections to assess adequacy of sample
“Biopsy all infections, culture all masses”
Once a diagnosis has been made through history, imaging, and tissue biopsy, the next step is formal surgical resection of masses when indicated. In the 1980s, Enneking et al . developed a method for musculoskeletal tumor dissection based on the histologic grade, location, and extent of the mass. This system has laid the foundation for how extremity surgeons approach resection, and will be expanded upon in the Sarcoma section of this chapter.
Throughout the workup, diagnosis, biopsy, and resection of a suspicious hand mass, the authors advocate the use of a multidisciplinary team for challenging and atypical cases. This team includes the hand surgeon, who may perform both resection and/or reconstruction, a surgical oncologist (musculoskeletal or general), medical oncologist, musculoskeletal radiologist, interventional radiologist, radiation oncologist, and pathologist. Planning through tumor board or multi-specialty rounds typically results in optimization of planning biopsies, pathologic assessment, resection margins, postoperative treatment protocols, and the final reconstructive outcome for the patient.
Neoplasms of the skin can be benign, premalignant, or malignant. In addition, there are masses such as sebaceous cysts and cutaneous horns that can masquerade as tumors, but are not true neoplasms and thus are considered pseudotumors. For the majority of benign neoplasms or tumors, an open excisional biopsy will completely eradicate the mass. For malignant skin neoplasms, wide local excision is performed, where the mass as well as surrounding healthy tissue are removed together.
A cutaneous horn is an exophytic lesion that originates from the skin and is composed of keratotic material ( Fig. 16.4 ). Cutaneous horns are located on sun-exposed areas of the body such as the dorsum of the hands and forearms. These unsightly lesions can interfere with function. They can occur in association with a variety of tumors, including epidermal inclusion cysts, epidermal nevi, fibromas, and pyogenic granulomas. In addition, cutaneous horns can be associated with both premalignant and malignant neoplasms such as actinic keratoses and squamous cell carcinomas in up to 40% of cases. Cutaneous horns are treated by excisional biopsy with 1–2-mm margins. Incisional biopsy is not recommended for this lesion because of the potential for sampling errors. Pathologic examination is mandatory after excision.
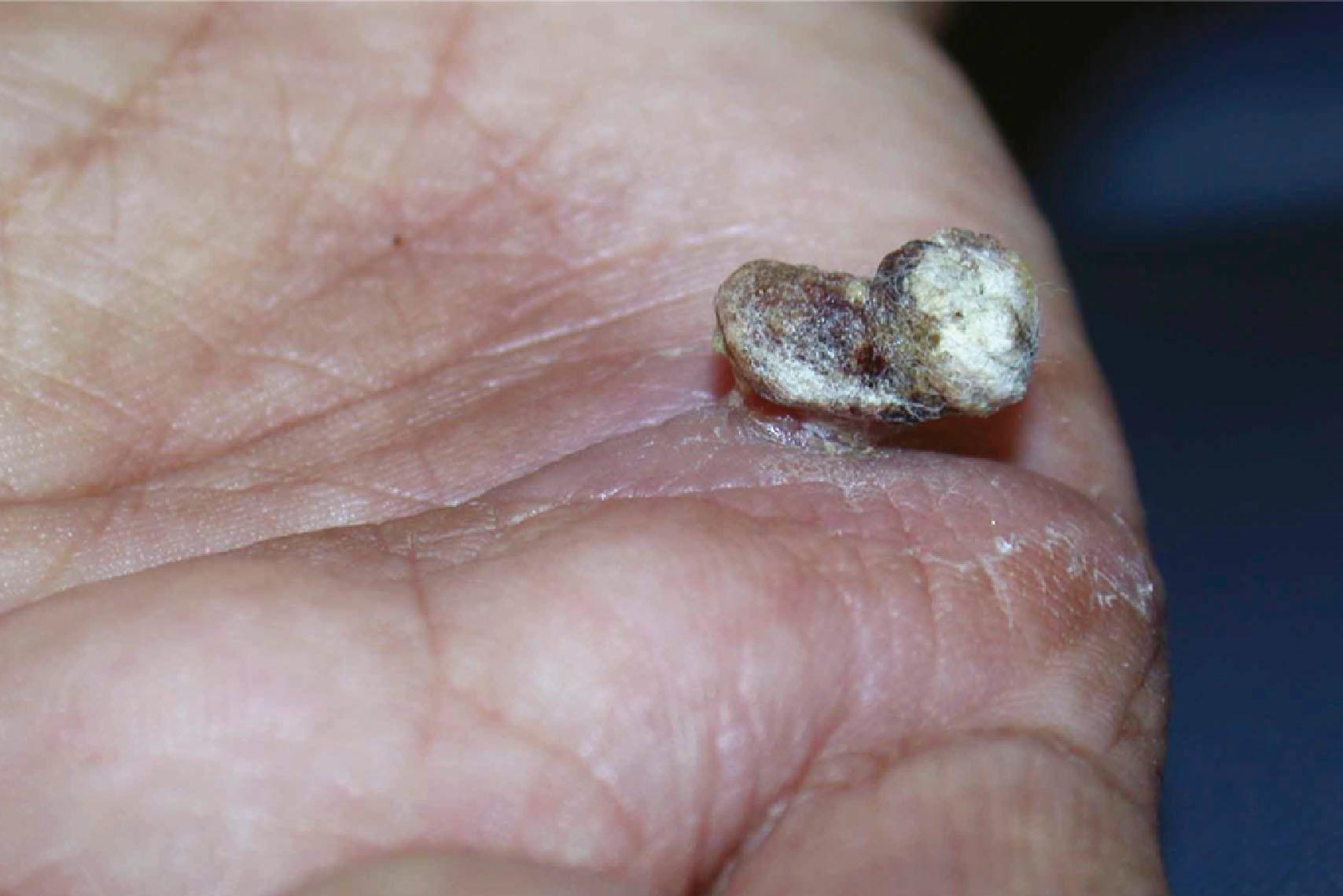
Epidermal inclusion cysts are the third most common hand tumors ( Figs. 16.5 & 16.6 ). These masses, which originate from an invagination of epithelium, can follow a trauma, injection, or incision, though the patient may not recall the inciting event. The epithelium is internalized, resulting in subcutaneous keratin accumulation. Epithelial cysts are typically painless and most commonly occur in the digits.
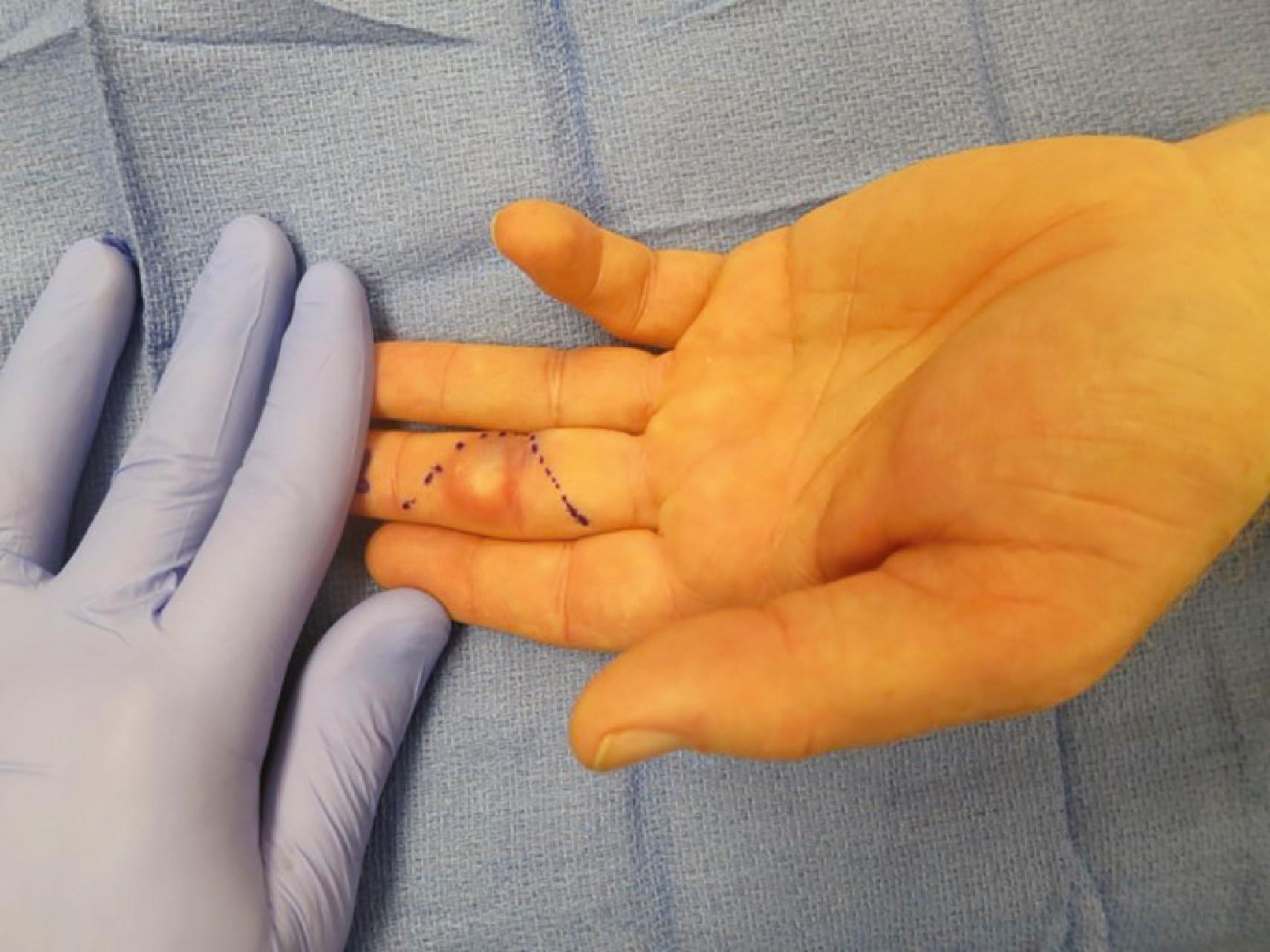
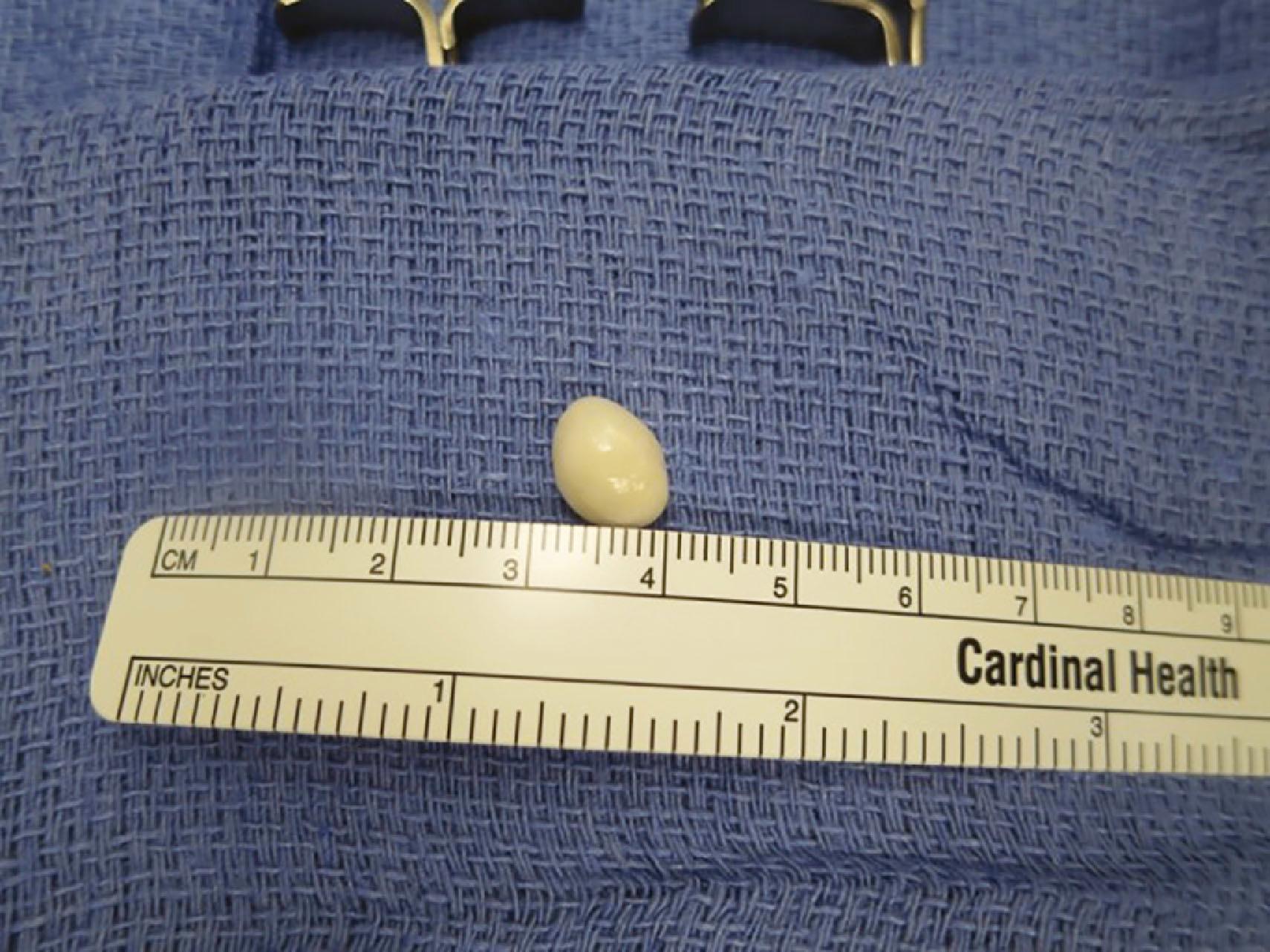
Treatment of an epidermal inclusion cyst involves a complete marginal excision of the cyst and its wall. The risk of recurrence of this lesion following complete excisional biopsy is extremely low.
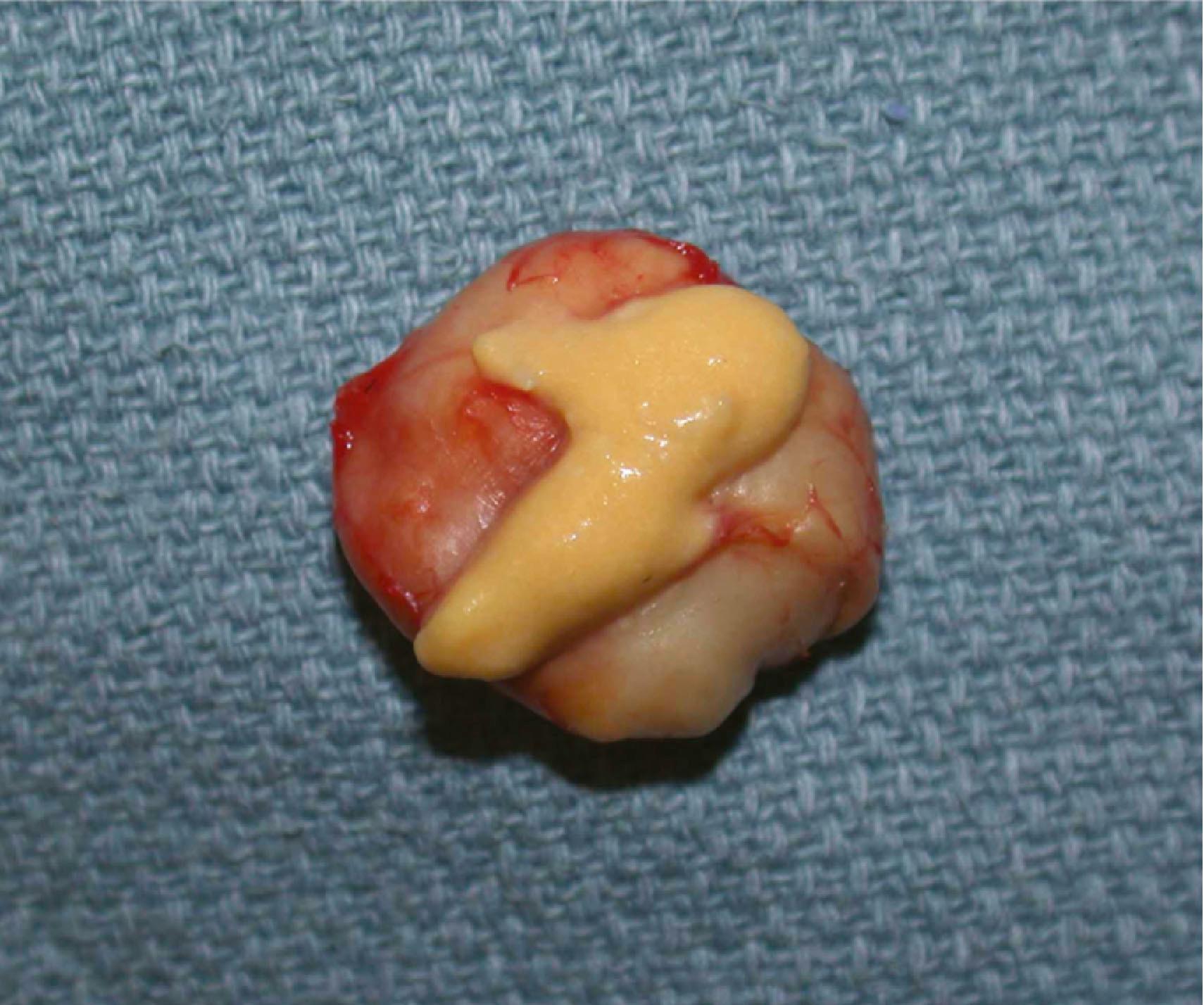
A sebaceous cyst is another lesion originating from the skin and is similar in appearance to an epidermal inclusion cyst ( Fig. 16.7 ). Sebaceous cysts arise from an obstructed apocrine gland, which produces sebum as opposed to keratin. When they occur in the hand, sebaceous cysts are found on the dorsal aspect, as palmar glabrous skin does not contain sebaceous glands. Treatment is similar to that of an inclusion cyst, and the risk of recurrence is low.
The human papillomavirus causes verruca vulgaris or common warts ( Fig. 16.8 ). Types 1–4, 7, and 10 have been implicated in the etiology of skin lesions involving keratinized epithelium. Verruca vulgaris presents as a rough raised surface anywhere on the hand and is 20 times more common than the verruca plana or flat wart, which is found commonly on the dorsum of the hand. Verrucae vulgaris are often found at sites of trauma in the periungual region (such as from habitual nail biting). The typical course of these benign neoplasms is self-limited, with spontaneous resolution in 1–2 years, although some malignant transformation has been reported.
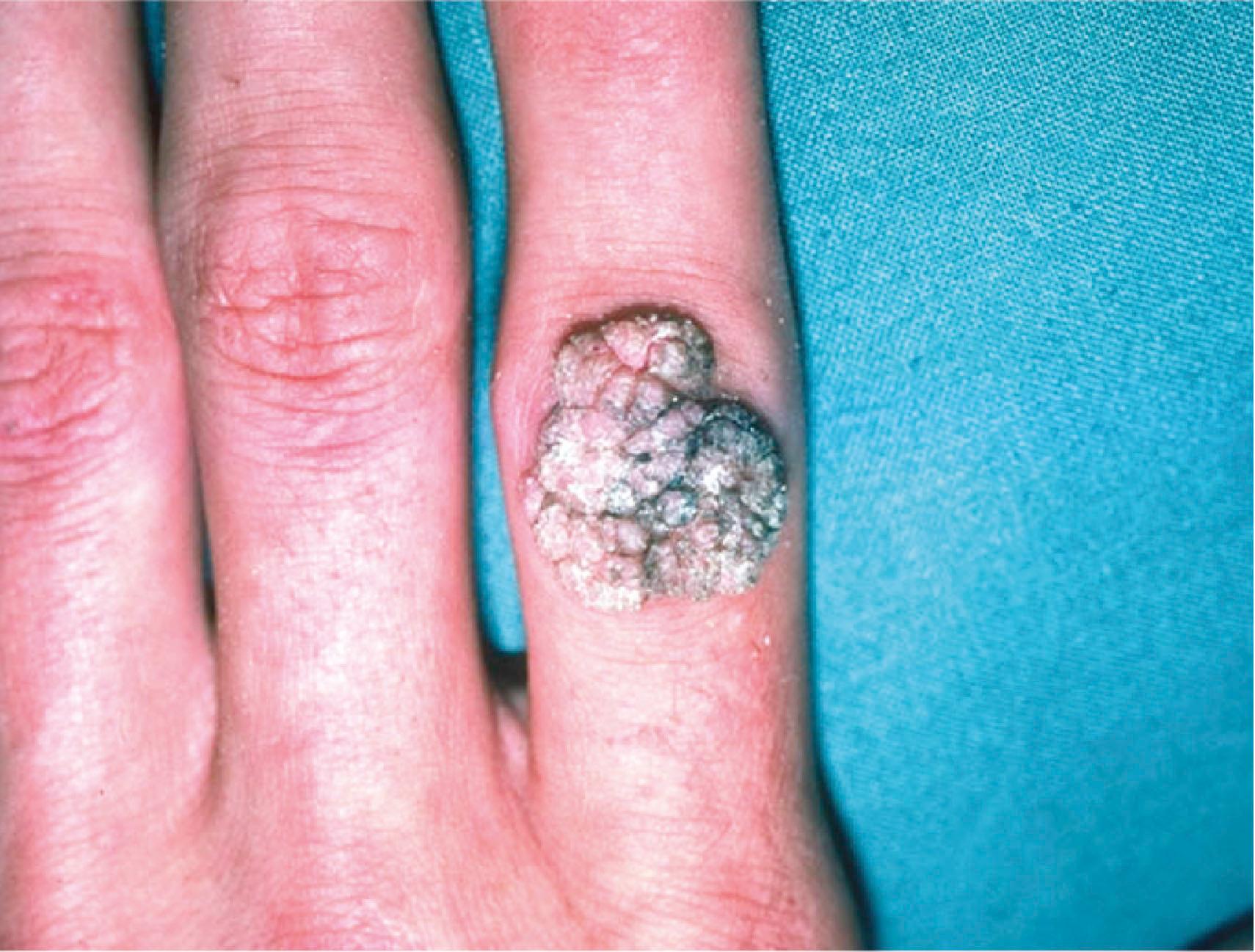
Treatment is instituted if the lesions become cosmetically unacceptable or if they interfere with hand function. The first-line therapy is topical agents such as salicylic acid, which results in cure rates of 70–80%. Cryotherapy is considered the second-line therapy for lesions unresponsive to topical agents and offers cure rates of 60–80%. Third-line treatments include immunomodulatory agents, lasers such as carbon dioxide and pulsed dye, and photodynamic therapy. It is critical to be highly suspicious of lesions that do not respond to therapy, as they may be malignant. Biopsy is therefore recommended for treatment-refractory verrucae or to rule out malignancy.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here