Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Tumors arising in soft tissue, although clinically often nondistinctive, form a varied and complex group that may show a wide range of differentiation. To an extent perhaps shared only by hematolymphoid disorders, the morphology of soft tissue lesions frequently belies their true biologic potential: examples of pseudomalignancy and even pseudobenignity abound. For this reason alone, in the absence of a firm histologic diagnosis it is often dangerous to attempt to predict (or “guess at”) the likely clinical course of a given soft tissue neoplasm. In understanding these lesions both histologically and clinically, it is important to appreciate that in many, if not most, of these tumors, the concept of histogenesis is not sustainable and is largely meaningless. With possible exceptions, such as subcutaneous lipomas or benign smooth muscle tumors, there is very little evidence that these lesions arise from their mature (differentiated) tissue counterpart. The facts that most liposarcomas arise at sites devoid of adipose tissue and most rhabdomyosarcomas develop in locations that lack voluntary muscle amply illustrate this point, as does the existence of extraosseous osteosarcoma. Nor is it, as yet, very meaningful, however convenient, to invoke the concept of a primitive mesenchymal stem cell as the progenitor of these tumors, as criteria for defining and identifying such cells in vivo in somatic mesenchymal tissues remain unconvincing. However, this concept certainly has enthusiastic and otherwise credible supporters. The possibility remains that some (or perhaps many) of these lesions may arise from bone marrow–derived stem cells. Bearing in mind that all diploid cells carry the genetic information, in varying degrees of expression or repression, to show almost any line of differentiation and, given that the genes that program different lines of mesenchymal differentiation most likely are closely related and remain available to be “switched on” at a point in the development of mesenchymal tissue beyond which, for example, the “epithelial genes” have been irreversibly “switched off,” then it also seems reasonable to assume that almost any mesenchymal neoplasm could arise from any type of mesenchymal cell. The major task or problem that remains is to determine what oncogenetic influences are operative (and how they act) in determining a particular line of differentiation. Now that specific differentiation genes, such as the MyoD family in skeletal muscle, have been characterized and genomic data associated with the process of neoplasia (as well as potential therapeutic targets) are being identified in specific soft tissue tumor types, it may soon be possible to answer some of these questions; as yet, however, gene expression data in soft tissue neoplasia have been more use in identifying novel phenotypic markers than in shedding much light on basic tumor biology.
An important starting point for the rapid advances in our molecular genetic understanding of soft tissue neoplasms (and a major contributing factor to the level of research interest shown in these relatively infrequent neoplasms) is that many tumor types show reproducible cytogenetic or molecular genetic aberrations. Among benign soft tissue tumors (as also in benign nerve sheath tumors, see Chapter 27 ), there seems to be a trend toward shared genetic pathways, irrespective of the line of differentiation, many of which involve the high-mobility genes, principally HMGA2 on chromosome 12 and HMGA1 on chromosome 6. Conversely, many types of soft tissue sarcoma (similar to leukemias) are characterized by distinctive cytogenetic aberrations, most often reciprocal chromosome translocations, which have been thought to be relatively tumor specific and thus diagnostically useful. However, increasing recognition of genetic overlap (even with tumors of epithelial and hematolymphoid lineages) is beginning to cause some degree of confusion. The well-characterized chromosomal abnormalities in soft tissue sarcomas are shown in Table 24.1 . Unfortunately, many of the more common sarcomas (the high-grade spindle cell and pleomorphic sarcomas of later adulthood) have complex karyotypes without histotype-specific features. In general, light microscopic morphology, supplemented where relevant by ancillary techniques, remains the cornerstone of the classification and diagnosis of soft tissue tumors. It is remarkable that these well-established technologies continue to facilitate the clinically useful definition of previously unrecognized tumor types on a regular basis.
| Tumor Type | Cytogenic Changes | Gene Rrearrangement |
|---|---|---|
| Ewing sarcoma/peripheral primitive neuroectodermal tumor | t(11;22)(q24;q12) t(21;22)(q22;q12) t(7;22)(p22;q12) t(17;22)(q12;q12) t(2;22)(q33;q12) t(16;21)(p11;q22) Others (see page 1982 ) |
FLI1-EWSR1 ERG-EWSR1 ETV1-EWSR1 EIAF-EWSR1 FEV-EWSR1 FUS-ERG |
| Alveolar rhabdomyosarcoma | t(2;13)(q35;q14) t(1;13)(p36;q14) |
PAX3-FOXO1A PAX7-FOXO1A |
| Myxoid/round cell liposarcoma | t(12 : 16)(q13;q11) t(12;22)(q13;q11–12) |
DDIT3-FUS DDIT3-EWSR1 |
| Desmoplastic small round cell tumor | t(11;22)(p13;q12) | WT1-EWSR1 |
| Synovial sarcoma | t(X;18)(p11.2;q11.2) | SSX1-SS18 SSX2-SS18 |
| Clear cell sarcoma/angiomatoid MFH | t(12;22)(q13;q12) t(2;22)(q33;q12) |
ATF1-EWSR1 CREB1-EWSR1 |
| Extraskeletal myxoid chondrosarcoma | t(9;22)(q22;q12) t(9;17)(q22;q11) |
NR4A3-EWSR1 NR4A3-TAF15 |
| Dermatofibrosarcoma protuberans/giant cell fibroblastoma | t(17;22)(q22;q13) | PDGFB-COL1A1 |
| Infantile fibrosarcoma | t(12;15)(p13;q25) | ETV6-NTRK3 Others involving EML4 and LMNA |
| Alveolar soft part sarcoma | t(X;17)(p11;q25) | ASPL-TFE3 |
| Low-grade fibromyxoid sarcoma | t(7;16)(q33;p11) t(11;16)(p13;p11) |
FUS-CREB3L2 FUS-CREB3L1 EWSR1-CREB3L1 |
| Sclerosing epithelioid fibrosarcoma | EWSR1-CREB3L1 EWSR1-CREB3L2 FUS-CREB3L1 Others |
|
| Inflammatory myofibroblastic tumor | t(2;1)(p23;p23) t(2;19)(p23;p13) t(2;17)(p23;q23) t(2;11)(p23;p15) t(2;2)(p23;q13) |
ALK-TPM3 ALK-TPM4 ALK-CLTC ALK-CARS ALK-RANBP2 Others involving ROS1 and PDGFRB |
| Myxoinflammatory fibroblastic sarcoma | t(1;10)(p22;q24) | TGFBR3-MGEA5 |
| Myoepithelial carcinoma | t(6;22)(p22;q12) t(1;22)(q23;q12) t(19;22)(q13;q12) |
EWSR1-POU5F1 EWSR1-PBX1 EWSR1-ZNF444 |
| Epithelioid hemangioendothelioma | t(1;3)(p36.3;q25) | WWTR1-CAMTA1 |
| Mesenchymal chondrosarcoma | del(8)(q21.1;q13.3) | HEY1-NCOA2 |
| Round cell sarcoma with CIC rearrangement | t(4;19)(q35;q13.1) t(4;10)(q35;q26) Others |
CIC-DUX4 CIC-DUX4 |
| Round cell sarcoma with BCOR rearrangement | inv(X)(p11.22;p11.24) Others |
BCOR-CCNB3 |
| Atypical lipomatous tumor/well-differentiated liposarcoma amplification | 12q rings and giant markers | HMGA2, CDK4, and MDM2 |
| Solitary fibrous tumor | inv12(q13;q13) | NAB2-STAT6 |
The grading of soft tissue sarcomas is a feature of many of the major tumor staging systems (e.g., American Joint Committee on Cancer [AJCC]) and is increasingly one of the key pieces of information required for the formulation of therapeutic decisions. At present there is no ideal histologic grading system and, hopelessly old-fashioned as this may seem, experienced “eyeballing” in centers that handle a large number of cases is as effective as any of the more objective schemes. The two best-known grading schemes are those of the National Cancer Institute (NCI) and the French Sarcoma Group, of which the French system (based on differentiation, mitotic rate, and necrosis) ( Box 24.1 ) is widely favored because it is more reproducible and allocates fewer patients to the somewhat unhelpful category of intermediate grade . The grading of soft tissue sarcomas remains a very subjective and somewhat unsatisfactory area, and it is unfortunate that none of the more objective assessments (such as proliferation index, measured by whatever means) has as yet offered any reproducible improvement over conventional morphologic assessment. Interestingly, combined assessment of tumor size, vascular invasion, and tumor necrosis (as determined histologically) and perhaps growth pattern appear to be as effective as grading in predicting outcome. Perhaps gene expression may prove to be a viable alternative, although this still awaits independent validation.
Score 1: Sarcomas closely resembling normal adult mesenchymal tissue (e.g., low-grade leiomyosarcoma)
Score 2: Sarcomas for which histologic typing is certain (e.g., myxoid liposarcoma)
Score 3: Embryonal and undifferentiated sarcomas, sarcomas of doubtful type, synovial sarcoma, osteosarcoma, Ewing sarcoma
Score 1: 0–9 mitoses per 10 hpf a
a A high-power field (hpf) measured 0.1734 mm 2 in the original study.
Score 2: 10–19 mitoses per 10 hpf
Score 3: ≥20 mitoses per 10 hpf
Score 0: no necrosis
Score 1: <50% tumor necrosis
Score 2: ≥50% tumor necrosis
Grade 1: total score 2,3
Grade 2: total score 4,5
Grade 3: total score 6,7,8
FNCLCC, Federation Nationale des Centres de Lutte Contre le Cancer.
Apart from these general considerations, readers are advised that vascular tumors and peripheral neuroectodermal tumors are discussed in Chapters 3 and 27 , respectively. Similarly, mesenchymal lesions occurring at visceral locations or arising principally in the skin are described in their respective chapters.
Adipocytic tumors form one of the largest single groups of soft tissue tumors, owing principally to the high incidence of benign subcutaneous lipomas. Furthermore, liposarcoma (including atypical lipomatous tumor) is the single most common type of primary soft tissue malignancy. With the exceptions of lipoblastoma and occasional lipomas, the majority of fatty neoplasms arise in adulthood. In general, ancillary diagnostic techniques such as immunohistochemistry and electron microscopy have only a limited or occasional role, as most cases are diagnosed on light microscopic morphology alone. The preeminence of lipoblasts as the major diagnostic criterion for liposarcoma has shifted, not only because the majority of cases of atypical lipomatous tumor (well-differentiated liposarcoma) and myxoid liposarcoma are recognizable in the absence of lipoblasts, but also because lipoblasts are now accepted as a routine finding in benign lesions such as pleomorphic lipoma, chondroid lipoma, lipoblastoma, and (less often) spindle cell lipoma. Cytogenetic and molecular genetic analysis now plays an increasingly important role in the classification and diagnosis of adipocytic neoplasms, particularly if they are deep seated, as many of these tumors show karyotypic abnormalities that are relatively histotype specific ( Table 24.2 ).
| Tumor Type | Most Common Cytogenic Aberrations |
|---|---|
| Solitary lipoma | Translocations involving 12q13–15 Rearrangements of 13q Rearrangements involving 6p21–33 |
| Spindle cell/pleomorphic lipoma | Monosomy 13 or partial loss of 13q in association with losses on 16q |
| Lipoblastoma/lipoblastomatosis | Rearrangements involving 8q11–13 |
| Hibernoma | Rearrangements involving 11q13 |
| Chondroid lipoma | t(11;16)(q13;p13) C11orf95-MKL2 |
| Angiolipoma | Normal karyotype |
| Atypical spindle cell/pleomorphic lipomatous tumor | Loss of RB1 and flanking genes at 13q14 |
| Atypical lipomatous tumor (well-differentiated liposarcoma) | Ring chromosomes and long marker chromosomes from 12q13–15 |
| Dedifferentiated liposarcoma | Ring chromosomes and long marker chromosomes from 12q13–15; additional complex aberrations in many lesions |
| Myxoid/round cell liposarcoma | t(12;16)(q13;p11) FUS-DDIT3 t(12;22)(q13;q11–12) EWSR1-DDIT3 |
| Pleomorphic myxoid liposarcoma | Numerical aberrations superimposed on near haploidy |
| Pleomorphic liposarcoma | Complex rearrangements, usually with greatly increased chromosome number |
Benign lipomas are extremely common and most often arise in subcutaneous tissue. The majority present in adults over the age of 30 years, with an equal sex incidence, and the anatomic distribution is wide. The trunk and proximal limbs are the most frequent sites, while lipomas in the hands and feet are rare. Most examples are solitary, slow growing, soft, and painless; however, 2% to 3% of patients have multiple lesions. Lipomas in childhood are uncommon but are not exceptionally rare, as sometimes has been suggested. Deep-seated benign lipomas do occur (see upcoming discussion) and, while seemingly mature fatty neoplasms in the abdomen or retroperitoneum have traditionally been regarded with suspicion, genetic data support the existence of large lipomas at these sites. Local recurrence following simple excision of subcutaneous lipomas occurs in no more than 1% to 2% of cases.
Most lipomas are well circumscribed, thinly encapsulated, and lobulated. Size is quite variable, but only infrequently exceeds 10 cm. Examples larger than this should be sampled carefully as they will often prove to be atypical lipomatous tumors (see upcoming discussion). The cut surface shows only mature adipose tissue in most cases, with the occasional admixture of more fibrous or myxoid areas or areas of fat necrosis. Histologically, the majority of tumors are composed simply of mature univacuolated adipocytes showing minimal variation in cell size and inconspicuous, small compressed nuclei at the cell border. It is common to find short strands of relatively acellular collagenous fibrous tissue or aggregates of foamy macrophages with scattered giant cells, indicative of microscopic fat necrosis. The latter may be prominent and result in variation in the size of adipocytes, especially in lesions on the upper back/shoulder. The presence of an extensive fibrous component may warrant the designation fibrolipoma, in which context the fibrous tissue is generally paucicellular and predominantly collagenous. Similarly, small foci of myxoid degeneration are an occasional finding, and if these become extensive and confluent then the term myxolipoma is sometimes used. Such lesions are often more vascular and, at least focally, tend to contain a bland spindle cell component and ropy collagen bundles, suggesting that they might be better regarded as myxoid examples of spindle cell lipoma. Up to 5% of cases show areas of osseous or chondroid metaplasia, often within areas of dense fibrous tissue and sometimes also with a prominent vascular component, leading to variable diagnostic terminology. In this author's experience, such lesions are most common in the limbs (especially distally) and around the shoulder girdle, and are entirely benign. Subcutaneous lipomas show (in most cases) reproducible karyotypic abnormalities, most often involving chromosome region 12q13-15, although these have no evident correlation with clinicopathologic features.
Dermal lipoma is the term sometimes used to describe those common solitary, pedunculated skin lesions, mainly presenting in young adults, which most likely represent a type of fibroepithelial polyp (skin tag) in which adipose tissue predominates. Only when such lesions are multiple and clustered, most often around the lower limb girdle, should the term nevus lipomatosus superficialis be applied. This latter phenomenon tends to affect younger patients and is regarded as hamartomatous in origin.
Synovial lipoma, also known as lipoma arborescens, is characterized by subsynovial collections of mature adipocytes that project, in villiform fashion, into the joint space. The villi are covered by hyperplastic, variably inflamed synovium. These lesions occur mainly in adults, most often affect the knee, and usually are associated with degenerative meniscal or articular pathology; as such they are almost certainly reactive in nature. Rare circumscribed synovial lipomas in children or adolescents have also been described.
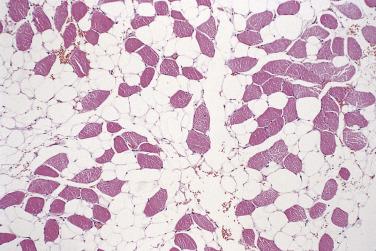
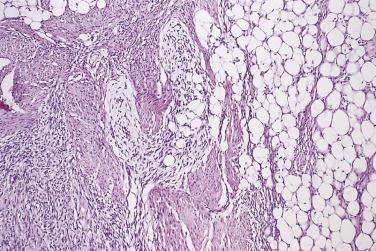
Intramuscular lipoma differs from its more superficial counterparts by most often being poorly circumscribed and infiltrative, and by recurring locally in up to 20% of cases if incompletely excised. It typically presents in mid-adult life, in patients of either sex, as a slow-growing deep-seated mass; the thigh and the trunk are the most common locations. Histologically, most cases are composed simply of mature adipocytes, which ramify in an irregular manner between variably atrophic muscle fibers ( Fig. 24.1 ). Blood vessels are scarce and usually capillary sized, allowing distinction from intramuscular hemangioma (see Chapter 3 ). The number or extent of intralesional muscle fibers often is not evident macroscopically. When dealing with any deep-seated fatty tumor, care must be taken to exclude atypical lipomatous tumor (see later), which by current diagnostic criteria is probably more common than intramuscular lipoma. The same range of secondary or metaplastic changes as in subcutaneous lipoma (see earlier) may be seen. Approximately 10% of intramuscular lipomas are well circumscribed and noninfiltrative; these lesions appear not to recur. Closely related is the intermuscular lipoma, which can only be diagnosed on the basis of precise radiologic or intraoperative localization. This type of lipoma is most common in the anterior abdominal wall and has a low rate of local recurrence. Other locations at which deep-seated lipomas occasionally occur are within tendon sheaths (usually around the hand or foot), the spermatic cord, the mediastinum, or in a parosteal location.
Lipomatosis is a rare condition, characterized by diffuse overgrowth of fatty tissue, which affects mainly adult males in one of four principal forms:
Multiple symmetric lipomatosis, the commonest form, affects mainly the neck and shoulders (Madelung disease or Launois-Bensaude adenolipomatosis) and is often associated with peripheral neuropathy, alcoholism, and hyperlipidemia. Rare cases are familial. Similar clinical features have also been described in patients receiving treatment with human immunodeficiency virus type 1 (HIV1) protease inhibitors.
Asymmetric lipomatosis most often appears randomly distributed. A subset of cases has a genetic basis, most often PIK3CA mutation.
Pelvic lipomatosis develops around the rectum and bladder, has characteristic radiologic features, and may lead to urinary obstruction and uremia.
Mediastinoabdominal lipomatosis also is associated with alcohol abuse, hyperlipidemia, and maturity-onset diabetes.
Histologically, the adipose tissue in all types is poorly circumscribed, fully mature, and may show focal fibrosis. Very rare forms of lipomatosis affecting the spinal epidural space and also the craniofacial region (in infants) have also been described. Lipomatosis of nerve, formerly known as fibrolipomatous hamartoma of nerve, is described in Chapter 27 .
Lipoblastoma is predominantly a tumor of infancy, affecting principally males before the age of 3 years, although cases in adolescents or young adults are encountered occasionally. Most examples present as a superficial, circumscribed, slow-growing mass on the trunk or a limb; the average diameter is approximately 5 cm. Deeper lesions, which may also occur in the mediastinum or abdomen, tend to be larger and may sometimes be diffusely infiltrative, in which case they are known as lipoblastomatosis. Local recurrence following excision usually only occurs with a positive excision margin.
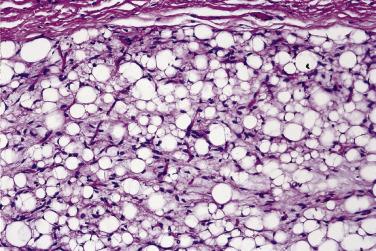
Both circumscribed and diffuse forms have a lobular architecture, the lobules being composed of mature and immature fat cells in varying degrees, separated by fibrous septa. The more immature lobules consist of primitive mesenchymal cells, lipoblasts, and small capillaries, all set in a myxoid stroma, very reminiscent of myxoid liposarcoma except for the absence of nuclear atypia ( Fig. 24.2 ). More mature lobules resemble lipoma, except that adipocyte nuclei appear more prominent, and there is some variation in adipocyte size. Even in immature areas mitoses are infrequent. Occasional lobules may show an appearance reminiscent of brown fat (hibernoma-like), and small foci of extramedullary hematopoiesis are quite common. Recurrent lesions quite often show maturation toward the appearances of a simple lipoma. Lipoblastomas show consistent abnormalities of chromosome 8q11-13, resulting in rearrangement of the PLAG1 gene. Of greatest importance is the histologic distinction from myxoid liposarcoma, which is exceedingly rare in children under 10 years of age; principal clues to the recognition of lipoblastoma are the presence of striking lobulation, a wider range of differentiation/maturation, and the absence of nuclear atypia. In rare cases, depending on the patient's age, this distinction may be almost impossible (or at least arbitrary) on morphologic grounds, but may be resolved by molecular testing.
Lipoblastoma-like tumor is a rare lesion occurring almost exclusively in the vulva of teenage or adult females and may recur locally. These are lobulated lesions composed of adipocytes, small lipoblasts, and bland spindle cells, distributed in a myxoid matrix. They appear to be unrelated to true lipoblastoma or to myxoid liposarcoma.
Angiolipomas are very common benign lesions that are almost always subcutaneous and, in more than 50% of cases, multiple. There is a very striking male preponderance, and the commonest presentation is in early adulthood. The anatomic distribution is wide, but the upper limb (especially the forearm) and the trunk are most frequently affected. A notable subset arises in breast, which, on needle biopsy, can be confused with angiosarcoma. Approximately half of the patients complain of pain or tenderness, which may present problems in clinical management if the lesions are too numerous to allow straightforward excision. Perhaps because the lesions are often symptomatic, the majority measure less than 2 cm at the time of excision. There is no tendency for local recurrence, but new lesions may continue to develop elsewhere.
Angiolipoma differs from ordinary lipoma by the additional presence of thin-walled, small, capillary-like vessels that tend to be situated mainly at the periphery of the tumor ( Fig. 24.3 ). Classically these capillaries—at least focally—contain small fibrin thrombi, which is often a diagnostically helpful feature. In fact, their absence argues strongly against this diagnosis. The extent of the vascular component in a given lesion is quite variable and may range up to 90% or more, in which case the term cellular angiolipoma may be used. These more cellular lesions may show a rather spindled endothelial appearance, combined with prominent pericapillary pericytes, thereby superficially simulating Kaposi sarcoma or a primitive capillary hemangioma. Identification of intralesional adipocytes and microthrombi facilitates differential diagnosis. Long-standing angiolipomas often show degenerative features in the form of perivascular fibrosis, hyalinization, and stromal myxoid change. Interestingly (and in contrast to most other fatty tumors), angiolipomas consistently have a normal karyotype, but whole exome sequencing has revealed distinctive PRKD2 mutations. Deep-seated intramuscular lesions, which in the past were termed infiltrating angiolipoma, would nowadays be classified as intramuscular hemangioma with a prominent adipocytic component (see Chapter 3 ); this distinction is important in view of the very high local recurrence rate of intramuscular hemangiomas.
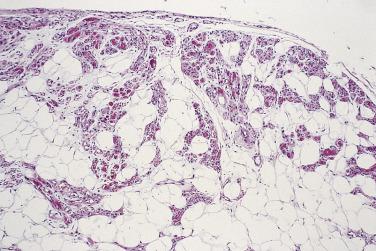
Myolipoma is a rare neoplasm characterized by the admixture of mature adipose tissue and smooth muscle in varying proportions ; often the muscular component is predominant ( Fig. 24.4 ). These tumors are often large, appear to be most common in the retroperitoneum/pelvis, and show a striking female preponderance. They seem not to recur locally. Many cases are immunopositive for HMGA2, suggesting similarities with conventional lipoma and leiomyoma. Important differential diagnoses are angiomyolipoma (see later) and well-differentiated liposarcoma with a heterologous smooth muscle component.
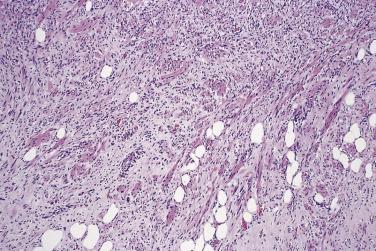
Chondroid lipoma is uncommon. It is readily mistaken for sarcoma because of its prominent population of cells that closely resemble lipoblasts and chondroblasts ( Fig. 24.5 ). Adult women are most often affected, and the anatomic distribution is wide, although most cases are subcutaneous. There is no tendency for local recurrence in these encapsulated lesions. Key morphologic features are the admixture of mature adipocytes, lipoblasts with bland nuclei, and hibernoma-like cells in a myxohyaline, pseudochondroid matrix. Cytogenetic analysis has revealed a reproducible t(11;16)(q13;p13) translocation, resulting in C11orf95-MKL2 gene fusion.
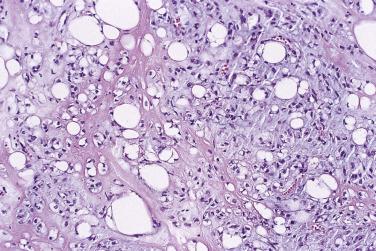
Angiomyolipoma is described in detail in Chapter 12 . Rare examples arise also in retroperitoneal or pelvic soft tissue, and in this circumstance the often bizarre appearance of either the smooth muscle or the adipocytic component may more easily prompt a mistaken diagnosis of malignancy. The characteristic thick-walled vessels and immunohistochemical positivity for HMB45 or Melan-A (putative melanoma antigens) in the distinctive muscle cells should facilitate the correct diagnosis. As with their more common renal counterparts, extrarenal examples may be associated with tuberous sclerosis. Lesions known as malignant epithelioid angiomyolipoma, which are more frequent in the kidney, also occur in the retroperitoneum and are better termed malignant PEComa (see page 1972 ).
Myelolipoma is described in more detail in Chapter 19 . Fewer than 40 cases have been reported outside either the adrenal or the liver, almost all in adult women, most commonly in a presacral or retroperitoneal location. Rare cases have been multicentric. There is no association with any hematologic disorder, and distinction from a so-called extramedullary hematopoietic pseudotumor is achieved by the presence of a prominent fatty component and the absence of erythroid hyperplasia or dysplastic megakaryocytes in myelolipoma.
Spindle cell and pleomorphic lipoma are described under one heading because a significant proportion of cases show overlapping clinicomorphologic features, and nowadays they are regarded as variations of a single entity.
Spindle cell and pleomorphic lipomas most often develop in mid to late adult life and show a striking predilection for males. Clinically these lesions are most often indistinguishable from ordinary lipoma, but a large majority of cases arise on the upper back, over one of the shoulders, or on the back of the neck. A further 10% develop more anteriorly in the head and neck region, including the face, and an increasingly wider anatomic distribution, not only in somatic soft tissue but including oral cavity and orbit, is nowadays recognized. The minority of lesions, which are entirely intradermal, show a much wider anatomic distribution and, surprisingly, an apparent preponderance in females. These tumors are usually solitary, painless, and slow growing, and they rarely exceed 5 cm in diameter. Occasional patients develop multiple lesions, which may be familial. Following local excision, recurrence is very uncommon, and there is no evidence that spindle cell or pleomorphic lipoma has any capacity to dedifferentiate (in contrast to atypical lipomatous tumor; see later). Principally for this reason it has been almost definitional that these lesions are subcutaneous (or occasionally dermal) in origin, but a small minority of histologically comparable lesions arise in deeper soft tissue, at least in this author's experience.
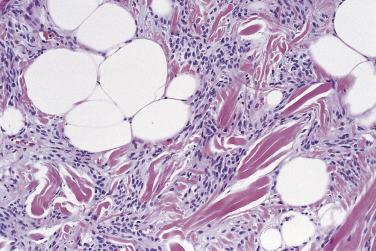
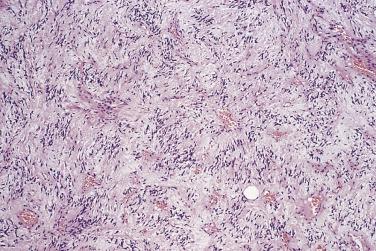
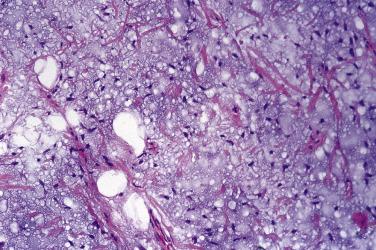
Spindle cell and pleomorphic lipomas are well circumscribed, thinly encapsulated, and may be lobulated. Lesions that arise on the face quite often extend into underlying skeletal muscle. The cut surface may be paler or firmer than usual lipoma and occasionally has an obvious biphasic (fatty/fibrous) appearance. Histologically, spindle cell lipoma is characterized by an admixture of mature adipocytes and spindle cells with short stubby nuclei and pale, poorly defined cytoplasm ( Fig. 24.6 ). These cells show no (or only rare) mitoses and are arranged in short fascicles; they may show nuclear palisading, highly reminiscent of benign schwannoma. They are set in a variably fibromyxoid stroma, which is notable for the presence of brightly eosinophilic, ropy hyaline collagen fibers and numerous mast cells. The relative proportions of the adipocytic and spindle cell elements vary considerably ( Fig. 24.7 ) and some cases may have virtually no fat. Vascularity in these lesions also varies, and there are sometimes branching, thin-walled vessels reminiscent of so-called hemangiopericytoma. Stromal myxoid change is common and may be both marked and diffuse in extent ( Fig. 24.8 ), especially in scalp lesions, leading to the potential designation myxolipoma; occasional lesions may have delicate crow's feet vessels, similar to myxoid liposarcoma. Lesions that have been described under the rubric dendritic fibromyxolipoma appear to represent myxoid spindle cell lipomas. Rare cases, usually with extensive myxoid change, show a striking pseudovascular pattern of degeneration characterized by villiform projections of tumor into seemingly empty spaces ( Fig. 24.9 ).
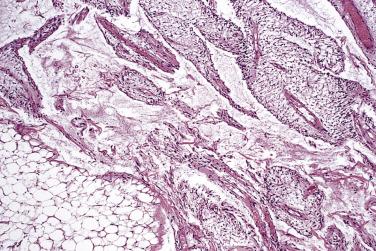
Pleomorphic lipoma, in the classic form, consists of mature adipocytes admixed with a variable number of bizarre, hyperchromatic, and commonly multinucleate stromal cells ( Fig. 24.10 ). The multinucleate cells typically have a ring of peripherally located (floret) nuclei arranged around eosinophilic cell cytoplasm. In these lesions it is not uncommon to find infrequent multivacuolated lipoblasts, and mitoses (which may be abnormal in appearance) are sometimes encountered. The stroma is variably collagenous. More common than this classic type of pleomorphic lipoma are lesions showing a hybrid appearance; such lesions usually most closely resemble spindle cell lipoma, except for the additional presence of a variable number of bizarre or multinucleate stromal cells.
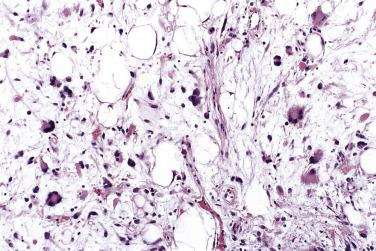
Spindle cell-predominant forms (see Fig. 24.7 ) are distinguished from schwannoma by recognition of the intrinsic adipocytic component and S100 negativity of the spindle cells. Distinction (usually of pleomorphic lipoma) from well-differentiated liposarcoma rests principally on careful clinicopathologic correlation in terms of anatomic site and superficial location. Additionally, in contrast to well-differentiated liposarcoma, there is usually little variation in adipocyte size and minimal adipocytic nuclear atypia in spindle cell or pleomorphic lipoma. Spindle cell lipomas show consistent chromosomal aberrations of 13q and 16q, quite different from the ring chromosomes seen in atypical lipomatous tumors (see Box 24.1 ), and are negative for MDM2 and CDK4. In keeping with frequent deletions of 13q14, spindle cell/pleomorphic lipomas often show loss of Rb protein expression, but, in practice, such staining is often difficult to interpret.
Hibernoma is an uncommon tumor that shows differentiation toward brown fat. It arises almost exclusively in adults of either sex, with a peak between the ages of 20 and 50 years. Pediatric examples are very uncommon. The commonest locations are the thigh, shoulder, interscapular region, and the back of the neck, but the overall anatomic distribution is very wide. This lesion usually grows very slowly and is painless; preoperative duration often exceeds 5 years. Although most cases are subcutaneous, up to 10% are intramuscular (especially in the thigh), and lesions are usually 5 to 10 cm in size. Occasional cases arise in bone, where they are radiologically sclerotic. There appears to be no tendency for local recurrence, and no convincing malignant counterpart has ever been described.
Hibernoma is well circumscribed and encapsulated, and the cut surface often has a tan-brown hue. Histologically, each lesion is composed of a variable admixture of (1) large, rounded, finely vacuolated cells with eosinophilic, granular cytoplasm; (2) similarly large cells but with much larger (lipoblast-like) fatty vacuoles; and (3) mature univacuolated adipocytes, which account for around 40% to 50% of the cell population in most cases ( Fig. 24.11 ). In some cases the mature adipocytes or multivacuolated lipoblast-like cells are predominant, the latter being especially frequent in thigh lesions. All these cell types have small, bland, usually peripherally located nuclei. Lobulation of the tumor is often striking. Extramedullary hematopoiesis is an occasional finding. Isolated cases may have a prominent myxoid stroma, and very rare examples have a spindle cell component resembling spindle cell lipoma. Cytogenetically, these lesions show rearrangements of 11q13 and (less often) 10q22.15. The 11q13 abnormality most often results in deletion of the MEN1 gene locus. Concomitant deletion of AIP appears to be pathogenetically important. There is no real differential diagnosis, as the adipocytic component allows easy distinction from any type of granular cell tumor. However, it should be remembered that myxoid liposarcomas quite often contain hibernoma-like cells.
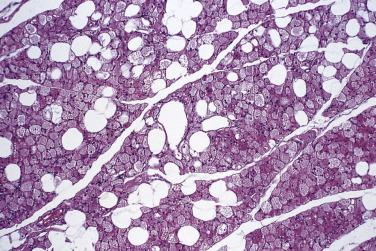
This is a recently introduced diagnostic category, which has been known in the past under a variety of terms, including spindle cell liposarcoma .
These lesions occur over a wide age range with a moderate predominance in males. They arise in subcutaneous or deeper soft tissue, most often limbs or limb girdles, with relatively frequent involvement of hands or feet. Visceral or retroperitoneal examples are rare. Median tumor size is 5 cm with a broad range. Approximately 10% of lesions recur locally, but there is no evident potential for dedifferentiation or metastasis.
These lesions consist, in varying proportions, of mature adipocytes showing variation in cell size, often with univacuolated or multivacuolated lipoblasts, along with a spindle cell component with generally longer nuclei than in spindle cell lipoma and with at least focal nuclear atypia and hyperchromasia ( Fig. 24.12 ). The stroma is variably myxoid or collagenous, sometimes with ropy collagen bundles. The margin is often infiltrative. The spindle cell component may be hypercellular and show notable nuclear atypia, resembling low-grade dedifferentiation. Mitoses are typically scarce, and necrosis is absent. Immunostains show frequent CD34 positivity in the spindle cells, while S100 protein (40%) and desmin (20%–25%) may also be expressed. Staining for MDM2 and CDK4 is usually negative, and MDM2 fluorescence in situ hybridization (FISH) is consistently negative. FISH demonstrates usual RB1 deletion, along with codeletion of flanking genes, in contrast to spindle cell lipoma.
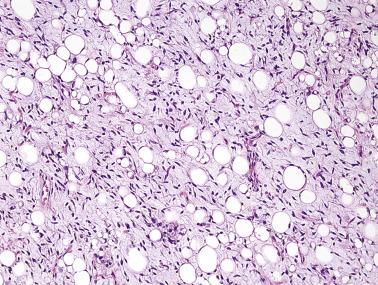
Similar lesions resembling pleomorphic lipoma, but showing more bizarre pleomorphic cells as well as large bizarre multinucleate lipoblasts, have been termed atypical pleomorphic lipomatous tumor . These tumors share the same molecular aberrations and low recurrence rate of atypical spindle cell lipomatous tumor. They lack the greater genetic complexity and more aggressive behavior of pleomorphic liposarcoma.
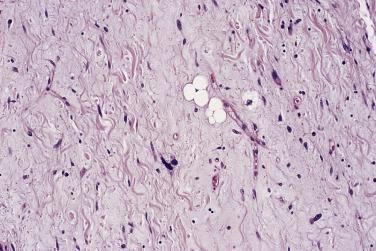
In this author's view, liposarcoma is the single most common soft tissue sarcoma and accounts for at least 20% of all sarcomas in adulthood. It has three principal forms: well differentiated (of which the dedifferentiated type is a variant), myxoid (of which the so-called round cell type is a variant), and pleomorphic. The cardinal diagnostic feature was traditionally the presence of lipoblasts, but additional alternative criteria exist for the recognition of well-differentiated (atypical lipomatous) lesions (see later). Special stains for lipid and immunohistochemistry generally play no essential role, except that on occasion S100 protein staining may highlight multivacuolated lipoblasts, and tumor cells in round cell liposarcoma are also often S100 positive. Furthermore, staining for MDM2 and CDK4 may be very useful in the diagnosis of atypical lipomatous tumor and dedifferentiated liposarcoma (see later). Although univacuolated (signet ring) lipoblasts are quite common, they may easily be confused with small vessels in cross section or with vacuolated endothelial cells, and therefore multivacuolated lipoblasts are the principal diagnostic requirement. These cells have two or more sharply outlined, often large cytoplasmic vacuoles that indent or scallop an atypical, hyperchromatic nucleus. Multinucleation is quite common. Small, vague or poorly delimited “bubbles,” especially if associated with a small, bland or central nucleus, generally do not suffice. The process that truly mimics these multivacuolated (as opposed to signet ring) cells is silicone granuloma, most often encountered adjacent to a breast implant or other tissue expander ( Fig. 24.13 ). The distinction from liposarcoma usually is easy, as well-differentiated lesions never have such numerous lipoblasts.
Liposarcoma is a tumor of adulthood and shows a slight male preponderance. Overall, the peak age at onset is in the fifth to seventh decades. Liposarcoma in children is extremely rare and is mostly confined to those 10 to 15 years of age ; it is most often myxoid in type and usually carries an excellent prognosis. Exceptional examples of atypical lipomatous tumor/well-differentiated liposarcoma in this age group may be associated with Li-Fraumeni syndrome. In adults, the clinical features vary somewhat according to histologic type ; in general, however, the very large majority of cases are deep seated, and their rate of growth parallels histologic grade. Myxoid tumors mainly affect somewhat younger adults (in the third to fifth decades) than other types. A history of a large, long-standing mass showing recent rapid growth usually indicates either dedifferentiation or progression of a myxoid lesion to its high-grade (round cell) form. In this author's material, 35% of liposarcomas arise in the lower limb (especially in the thigh); 22% on the trunk or around limb girdles; 15% in the upper limb; 15% in the retroperitoneum; 8% in the head, neck, or mediastinum; and 5% in the spermatic cord. When site is related to histologic type, myxoid, round cell, and pleomorphic liposarcomas show an overwhelming predilection for the limbs; well-differentiated lesions are equally divided between limbs and retroperitoneum, and dedifferentiated lesions are by far most common in the retroperitoneum. A tendency for multicentricity in liposarcoma has been described, but this reflects indolent soft tissue metastasis (a characteristic feature of the myxoid subtype), and most such patients eventually develop disseminated disease. Notably, myxoid liposarcoma very often gives rise to metastases in other soft tissue locations (especially abdomen) and in bone, before involving visceral sites. Overall, 5-year survival probabilities are somewhat imprecise as they depend heavily on diagnostic criteria; approximate survival figures at 5 years are for well-differentiated lesions 90% (at 10 years 60%, reflecting frequent and often uncontrolled local recurrence of retroperitoneal lesions), for dedifferentiated lesions 60% to 70% (at 10 years 40%–50%) (again, most deaths being the result of retroperitoneal local recurrence), for pure (low grade) myxoid lesions 90%, for myxoid lesions with notably increased cellularity 40% to 50%, for high-grade myxoid (formerly round cell) lesions 25%, and for pleomorphic lesions around 50% to 60%. Superficially located examples of pleomorphic liposarcoma appear to have a much better prognosis.
Well-differentiated liposarcoma, which is also known as atypical lipomatous tumor (discussed later), occurs in two principal forms: adipocytic (lipoma-like) and sclerosing, of which the former is far more frequent. Together these account for about 40% to 45% of liposarcomas. In the retroperitoneum, tumors showing mixed patterns are common. The sclerosing form is far more common in the retroperitoneum and spermatic cord than elsewhere. Lesions tend to be large, well circumscribed, and coarsely lobulated; the sclerosing type is relatively pale and firm. A relative minority of cases show irregular infiltration at their margin into adjacent muscle.
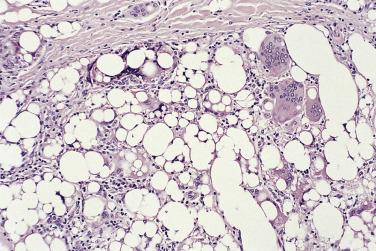
Adipocytic (lipoma-like) liposarcoma consists for the most part of relatively mature univacuolated adipocytes showing variation in cell size and, in at least a proportion of cells, atypical, hyperchromatic nuclei ( Fig. 24.14 ). Admixed with these cells are scattered bizarre, often multinucleate stromal cells and variably prominent fibrous septa containing hyperchromatic spindle cells and occasional bizarre cells (as described earlier) ( Fig. 24.15 ). Atypical spindle or multinucleate cells may also be identified in the walls of larger intralesional vessels. The number of lipoblasts varies. They are no longer regarded as a diagnostic requirement but are often found most easily close to fibrous septa; in some cases they are absent or undetectable (see later). In large, long-standing lesions areas of fat necrosis are quite common. Occasional cases contain foci of metaplastic bone or scattered large rhabdomyoblasts, or may show partial smooth muscle differentiation; the latter component may be mature or sarcomatous, albeit well differentiated. Sclerosing liposarcoma differs by consisting largely of collagenous fibrous tissue, which tends to have a somewhat delicate, almost fibrillary appearance. Scattered within this fibrous tissue are mature adipocytes and bizarre, hyperchromatic stromal cells, which are often multinucleate ( Fig. 24.16 ); in many cases lipoblasts are hard to find. Both adipocytic and sclerosing liposarcoma should be carefully and thoroughly sampled, usually by close serial slicing macroscopically, to exclude the presence of any dedifferentiated component (see later).
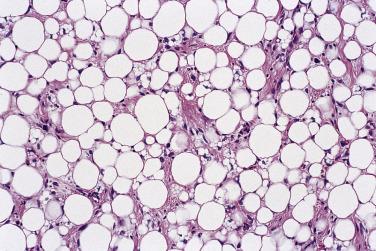
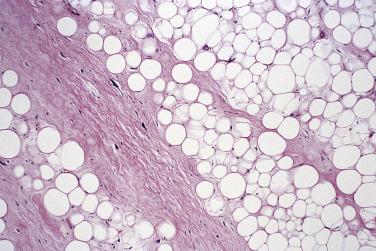
Two other forms of well-differentiated liposarcoma are encountered more rarely: the inflammatory type, which is almost always retroperitoneal in location and in which numerous prominent lymphoplasmacytic aggregates may simulate inflammatory pseudotumor ( Fig. 24.17 ), and that with spindle cell features, which is also rare but otherwise resembles usual atypical lipomatous tumor/well-differentiated liposarcoma with an admixed low-grade–appearing atypical spindle cell component and which shows usual MDM2 amplification.
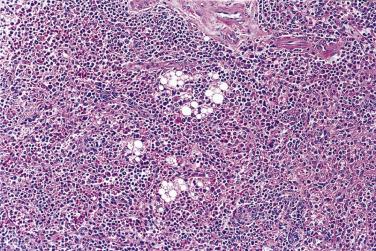
The term atypical lipoma was introduced by Evans et al to take account of the fact that well-differentiated liposarcoma in histologically pure form does not metastasize and that those lesions amenable to wide surgical excision (usually those in somatic soft tissue) are therefore readily cured. It was further recognized that deep-seated fatty tumors showing adipocytic and/or stromal cell atypia but lacking lipoblasts behaved in exactly the same way as well-differentiated liposarcoma; in other words, if excision is marginal or incomplete then local recurrence is common, and within any recurrence (especially if repeated) there is a theoretical risk (<5%) of dedifferentiation and hence acquisition of metastatic potential. This biologic potential is the same irrespective of lesional location or depth, although atypical lipomatous tumors in the limbs are more often adequately excised, and so recur and dedifferentiate much less often than their retroperitoneal counterparts. It is important also to note that these atypical fatty neoplasms, irrespective of location and whether or not they contain lipoblasts, are characterized karyotypically by the presence of ring (or long marker) chromosomes derived from the long arm of chromosome 12. These ring or marker chromosomes contain innumerable copies (i.e., amplification) of the MDM2 , CDK4, and HMGA2 genes. The resulting overexpression of MDM2 and CDK4 proteins has proved to be a useful immunohistochemical marker of both well-differentiated and dedifferentiated lesions, although the antibodies are not perfectly reliable and sometimes use of FISH is necessary or preferable. The concept of atypical lipomatous tumor has considerable validity but necessitates close clinicopathologic liaison lest an unwitting surgeon underestimate the significance of such a diagnosis. This author's preference is to use the term well-differentiated liposarcoma only for lesions in the retroperitoneum, spermatic cord, and mediastinum that may kill through their local effects or those of local recurrence(s), and to designate all those lesions in surgically amenable locations (principally limbs and trunk) atypical lipomatous tumors, with an explanatory note in the pathology report concerning their biology; this avoids the significant social and psychological consequences of an unnecessary diagnosis of sarcoma.
For the purpose of distinguishing well-differentiated liposarcoma from a lipoma showing degenerative or atrophic changes, from an area of florid fat necrosis, or from a sclerosing inflammatory lesion, emphasis should be placed principally on the recognition of adipocytic nuclear atypia and the presence of bizarre, hyperchromatic stromal cells.
Dedifferentiated liposarcoma, which accounts for up to 10% of liposarcomas, is classically defined as a well-differentiated liposarcoma showing abrupt transition, either in the primary tumor or in a recurrence, to high-grade nonlipogenic sarcoma ( Fig. 24.18 ). Occasional cases may show focal comingling of the two components. Approximately 90% of cases occur de novo, whereas 10% develop in recurrences. Dedifferentiated liposarcoma is by far most common in the retroperitoneum, and in fact accounts for the majority of spindle cell or pleomorphic sarcomas in that location. Cases were often misdiagnosed as ‘malignant fibrous histiocytoma’ (MFH) in the past. Macroscopically, the two components are most often easily distinguished, but there is a danger that the well-differentiated component may be overlooked if sampling is concentrated on the solid, nastier-looking high-grade areas; in fact, the well-differentiated areas may be sufficiently normal looking or else limited in extent that the surgeon may inadvertently leave that component behind at the time of resection. The histologic patterns encountered in the dedifferentiated component are variable, but most often resemble unclassified storiform/pleomorphic sarcoma ( Fig. 24.19 ), myxofibrosarcoma (myxoid MFH), or sarcoma not otherwise specified (NOS). A subset of cases shows a prominent neutrophilic infiltrate, and these account for most of the tumors formerly known as so-called inflammatory MFH. In a small proportion of cases there is a multinodular discohesive pattern of dedifferentiation, suggesting that, at least sometimes, this is a field change phenomenon. In most cases, the nonlipogenic, histologically higher grade component shows no morphologic evidence of specific differentiation, although focal desmin positivity, which probably reflects myofibroblastic differentiation, is common. Immunopositivity for MDM2 and CDK4 is more consistently identified than in well-differentiated lesions. With more accurate recognition of this subtype of liposarcoma, it has become apparent that approximately 10% of cases show heterologous differentiation, most often rhabdomyosarcomatous ( Fig. 24.20 ), leiomyosarcomatous, or osteosarcomatous ; these components are variable in extent, and it is now recognized that myogenic differentiation in this context confers a notably worse prognosis. Such tumors were often diagnosed as malignant mesenchymoma in the past. A further distinctive pattern in some cases is the presence of micronodular (meningioma-like) spindle cell whorls, often associated with ossification. An additional and important advance has been the realization that the dedifferentiated component may be morphologically low grade, generally consisting of nonlipogenic cellular spindle cell areas with a vaguely fascicular growth pattern. In such cases, cytologic atypia is limited and necrosis is absent. Some authors have labelled these lesions as cellular atypical lipomatous tumor . Finally, it has been appreciated that the dedifferentiated component may, in fact, show lipoblastic differentiation, leading to potential confusion with pleomorphic liposarcoma and likely accounting for some cases diagnosed in the past as mixed-type liposarcoma. While dedifferentiated liposarcoma has a high rate of local recurrence, irrespective of grade, the development of distant metastasis and poorer overall survival correlate with high histologic grade.
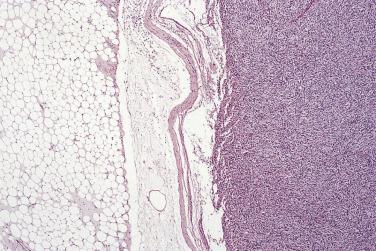
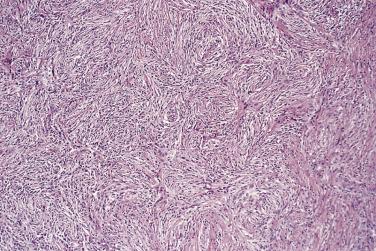
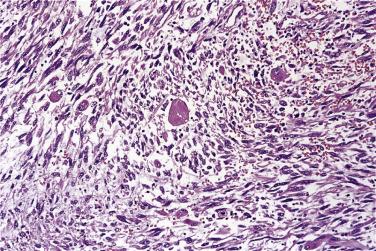
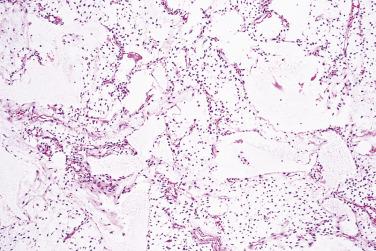
Myxoid liposarcoma and its higher grade form (often known in the past as round cell liposarcoma) together account for around 30% to 35% of liposarcomas and, irrespective of grade, are characterized by a specific reciprocal chromosome translocation, either t(12;16)(q13;p11) or, less often, t(12;22)(q13;q11-12), both of which result in rearrangement of the DDIT3 gene, a transcription factor involved in adipocytic differentiation. In its low-grade form this tumor is gelatinous, somewhat reddish, and often contains areas of seemingly infarcted more mature adipose tissue. Around 5% of cases are subcutaneous in location. When histologically pure, it consists of undifferentiated nondescript small ovoid or spindle cells and small lipoblasts (predominantly univacuolated) set in a copious myxoid matrix (composed of hyaluronic acid) within which there are numerous, delicately branching, thin-walled capillaries, which are described variously as having a crow's feet or chicken wire configuration ( Fig. 24.21 ). Diagnostic multivacuolated lipoblasts (generally smaller than those in well-differentiated liposarcoma) are most easily found in the peripheral subcapsular zone of the tumor. Mitoses are extremely scarce. Frequently there is pooling of stromal mucin, producing large spaces with a pseudolymphangiomatous or pseudoalveolar appearance ( Fig. 24.22 ). Variations on this theme include cases with granular eosinophilic (hibernoma-like) cells, cases showing extensive differentiation toward mature adipocytes, and rare cases showing a predominantly spindle cell appearance. Occasional cases show focal cartilaginous metaplasia and rare cases contain small numbers of scattered pleomorphic, often multinucleate cells, but these seem to have no prognostic significance.
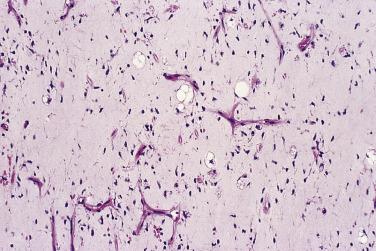
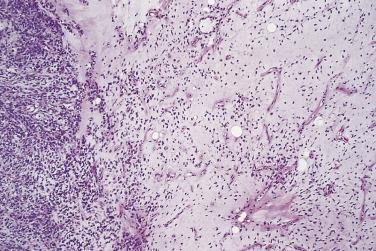
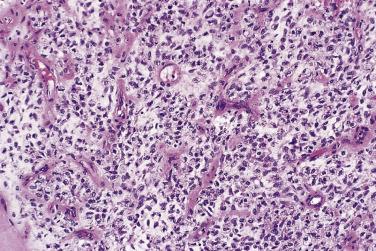
Of greater importance is the presence of more cellular areas, often known rather inaccurately as round cell differentiation, which imply more aggressive behavior. The cells sometimes have larger, rounder, and more hyperchromatic nuclei than the bland spindle cells of myxoid liposarcoma, although their cytoplasm remains inconspicuous ( Fig. 24.23 ), but most cases are hypercellular without having actual round cell morphology. Only a small minority of cases have entirely undifferentiated round cell morphology (readily mistaken for a wide variety of other tumor types). Mitoses are relatively more frequent. The extent to which these cells occupy the surface area examined is quite variable: if it is 75% or more, the term round cell liposarcoma has often been used ( Fig. 24.24 ). The majority of cases show mixed spindle and round cell morphology and are better referred to as high-grade myxoid liposarcoma. Because the extent of hypercellularity or round cell differentiation forms a continuous spectrum from minimal to extensive, there remains considerable uncertainty about what extent has prognostic significance. Certainly, any lesion with 10% or more hypercellular or round cell areas should be labeled intermediate grade and regarded as having significant metastatic risk; if this component accounts for 25% or more, then the tumor should be classified as high grade. The prognosis in cases with more limited hypercellularity is not well characterized, however, and should remain guarded.
Myxoid liposarcoma can be distinguished from intramuscular myxoma by its greater vascularity and the presence of lipoblasts, and from myxofibrosarcoma by the facts that the latter shows notably greater nuclear hyperchromasia and pleomorphism and tends to have more curvilinear vessels. Predominantly round cell lesions may be hard to distinguish from almost any anaplastic round cell malignancy (including metastatic carcinoma), but useful clues, retained even in most high-grade cases, are the presence of crow's feet vessels and small areas of mucin pooling. A careful search for lipoblasts usually should prove fruitful. It is of interest that, whereas the nonlipogenic spindled or pleomorphic cells are negative for S100 in most types of fatty tumor, the undifferentiated round cells and some of the blander spindle cells in myxoid and round cell liposarcoma may show cytoplasmic (and nuclear) positivity for S100 protein, in the same way as adipocytes and lipoblasts.
Pleomorphic liposarcoma accounts for about 5% of liposarcomas. It can be distinguished from other high-grade pleomorphic sarcomas (the MFH-like family) only by the presence of multivacuolated lipoblasts ( Fig. 24.25 ). These cells may be few in number or may be remarkably localized, and so careful sampling is required for their reliable identification. Other cases closely mimic myxofibrosarcoma or are notable for the presence of sheets of large, bizarre lipoblasts, and a minority of examples are notable for strikingly epithelioid cytomorphology. Apart from very marked cytologic pleomorphism, a common but not specific finding is the presence of numerous intracytoplasmic eosinophilic globules or droplets, the exact nature of which is disputed, although lysosomal origin seems most likely.
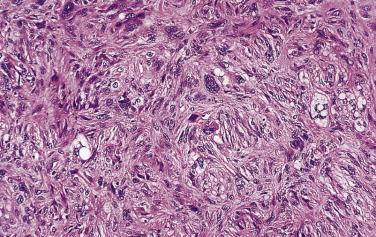
Pleomorphic myxoid liposarcoma is a recently recognized, distinct, and separate type of liposarcoma, which occurs mainly in pediatric or teenage patients and which is not related to conventional myxoid liposarcoma. These tumors have a wide anatomic distribution with a predilection for mediastinum. At least in areas, these lesions closely resemble conventional low-grade myxoid liposarcoma, but they show areas with increased cellularity, marked nuclear pleomorphism, and large bizarre lipoblasts. Mitoses are easily found. These tumors lack DDIT3 rearrangement, but instead show more complex genomic abnormalities. The clinical course is often aggressive.
Fibrous tumors encompass a heterogeneous group of reactive processes, putatively hamartomatous or developmental lesions, and benign, locally aggressive or malignant neoplasms. It is often uncertain into which precise biologic category those lesions with a benign clinical course should be classified. From the histologic point of view, with very few exceptions these tumors are most often composed of an admixture of fibroblasts and myofibroblasts in varying proportions; these proportions may vary during the time course of the disease process. This is to be expected in light of the very close biologic relationship between these two cell types, which essentially represent points along a single structural and functional continuum. There are no clearly defined and agreed cutoff points between a fibroblast and a myofibroblast, or between a myofibroblast and a smooth muscle cell, and so the distinction is often arbitrary.
Keloid scars are most common in adolescents and young adults and are especially frequent in black patients. They may develop at any anatomic location as a result of local trauma, with the head and neck region, particularly the earlobes, being the site of predilection. The lesions are classically itchy, raised and shiny, with overlying telangiectatic skin, and extend beyond the boundaries of the site of initial tissue damage. Treatment is difficult, and up to 50% recur locally after attempted excision. Histologically, these common lesions consist of mature, relatively hypocellular, fibrous scar tissue within which there are broad bands or nodules of brightly eosinophilic hyaline collagen. These latter are the diagnostic sine qua non. The pathogenesis of keloids remains uncertain but seems likely to represent a genetic predisposition to respond excessively to growth factors (such as TGFβ1 and β2) elaborated during the normal production of scar tissue.
Hypertrophic scars represent another form of abnormally excessive scarring. They differ clinically from keloids by being less common, showing no racial predilection, being confined to the original site of tissue damage, and being much less prone to local recurrence. Histologically, these lesions are composed of more cellular scar tissue than in keloids and the tissue tends to have a nodular configuration. Confident distinction from a usual scar often requires clinicopathologic correlation.
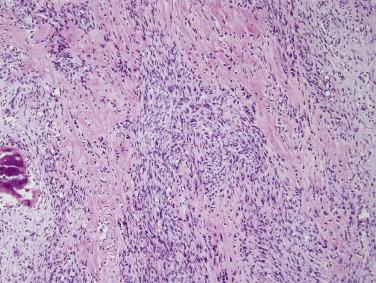
Ischemic fasciitis, which was initially reported under the term atypical decubital fibroplasia, is a distinctive reactive fibroblastic lesion that occurs most often over bony prominences, sometimes in immobile, usually debilitated, patients. The lesion develops most often in deep subcutis, especially around the hip or sacral region. Histologically, such cases are characterized by broad zones of fibrinoid necrosis surrounded by vascular and inflamed granulation tissue in which plump, mitotically active fibroblasts and myofibroblasts, akin to those in proliferative fasciitis, are encountered ( Fig. 24.26 ). Adjacent affected adipose tissue often appears infarcted. These lesions are thought to have an ischemic, degenerative etiology, akin to decubitus ulcers. Local recurrence after excision is seen in occasional cases, most likely reflecting persistence of the predisposing cause.
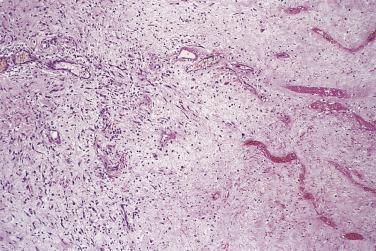
Lesions with histologic features that overlap with nodular or proliferative fasciitis have in recent years been increasingly recognized to occur also at visceral locations, especially the genitourinary tract ( Fig. 24.27 ). Other rare locations in the author's experience include the larynx and sinonasal tract. Although originally believed to be mainly posttraumatic (usually postoperative), it is now realized that most such cases are apparently spontaneous. These clinically benign lesions are described in more detail in Chapter 12 . They are distinguished from inflammatory myofibroblastic tumor by clinical context and the fact that the latter tends to have a much more prominent chronic inflammatory component, especially plasma cells, and may show some degree of nuclear atypia. Furthermore, inflammatory myofibroblastic tumor has a much higher rate of local recurrence and may even pursue a malignant course. Whether or not these seemingly reactive lesions have ALK gene rearrangement has been somewhat controversial.
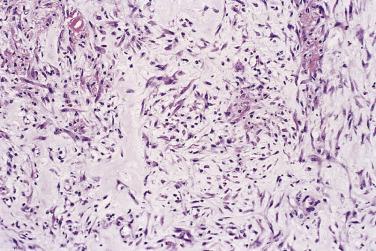
Fibromatosis colli, also known clinically as sternomastoid tumor or torticollis, is an uncommon condition characterized by diffuse fibrous replacement of one sternocleidomastoid muscle, most often the right. Bilaterality is very rare. It presents in neonates of either sex, most often after breech or forceps delivery, and is associated with neck asymmetry and tightness. Many cases resolve spontaneously (or with the help of physiotherapy) and only 15% to 20% go on to develop true torticollis (wry neck deformity). Antenatal position in utero appears to be of prime pathogenetic importance. Histology reveals diffuse infiltration and replacement of skeletal muscle by relatively hypocellular, often rather hyalinized, fibrous scar tissue ( Fig. 24.28 ). This tissue extends between and around individual muscle fibers, and the latter show marked degenerative or reactive changes. The presence of damaged muscle fibers throughout the lesion facilitates histologic distinction from desmoid-type fibromatosis.
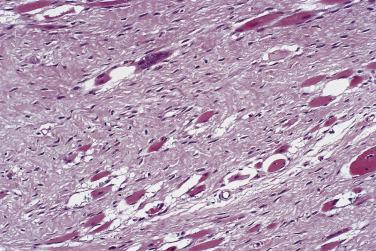
Most fibrous lesions in the category of benign neoplasms are acquired, affect mainly adults, and recur either infrequently or in a nonaggressive fashion. Regarding lesions of disputed nosologic status, for decades nodular fasciitis was thought to be reactive but is now known to be a clonal neoplastic process, whereas the status of lesions such as elastofibroma and juvenile hyaline fibromatosis remains somewhat uncertain. The diagnosis of fibroma, if used in an unqualified manner, is meaningless and should be avoided, not least because it encourages diagnostic lassitude, but also because it has been used, mostly in the past, to describe almost every type of tumor in this chapter! Within this category, certain lesions are more appropriately described elsewhere in this book: intranodal myofibroblastoma in Chapter 21 , mammary myofibroblastoma in Chapter 16 , nasopharyngeal angiofibroma in Chapter 4 , and both pleomorphic fibroma and giant cell fibroblastoma in Chapter 23 .
Nodular fasciitis, in its prototypical form, presents as a rapidly enlarging, often painful or tender subcutaneous nodule, most often affecting young adults and showing a predilection for the upper limb, especially the forearm. Although duration is variable, most cases have been present for less than 10 to 12 weeks. Almost any anatomic site can be affected, and the less frequent cases seen in children predominate in the head and neck region. The depth of the lesion is also variable: Approximately 10% of cases are entirely intramuscular and a small minority arise either from periosteum (parosteal fasciitis) or in the skin (intradermal fasciitis). Parosteal lesions arising on the head—most commonly in children—often show striking erosion of bone and have been referred to as cranial fasciitis. Another infrequent subgroup of cases involves blood vessels, principally veins, in a multinodular manner, usually in the upper limb or head and neck region, and is known as intravascular fasciitis. Rare cases may be intraarticular. Although traditionally regarded as reactive lesions, there had been accumulating evidence (mainly from cytogenetic analysis) that nodular fasciitis and its related variants are clonal proliferations. These lesions have convincingly been shown to harbor a specific gene fusion MYH9-USP6 confirming their neoplastic nature, even if self-limiting or transient.
In terms of clinical behavior, independent of the structures involved, these are all benign processes with a self-limiting clinical course; if left untreated, they will regress spontaneously. Local recurrence (or persistent growth) is very infrequent, occurring in 1% to 2% of cases, and is almost invariably the result of piecemeal, macroscopically incomplete excision during the active growth phase. In the majority of cases local marginal excision is adequate, and any microscopic residual lesional tissue undergoes spontaneous attrition, generally seeming to leave little or no scar.
The majority of lesions are well circumscribed but unencapsulated, and most measure less than 3 cm in diameter. The cut surface varies according to duration, but may be myxoid, centrally cystic, or fibrous. The histologic features, irrespective of location, are characteristic and reproducible. Contrary to popular belief, most cases are well marginated, and no more than 10% have an irregularly infiltrative or arborizing growth pattern. The classic case consists of a cellular proliferation of myofibroblasts with plump nuclei and occasional nucleoli, set in a loose collagenous stroma showing variable degrees of myxoid, often microcystic, change ( Fig. 24.29 ). Normal mitoses may be numerous, but the spindle cell nuclei are never hyperchromatic or pleomorphic. The spindle cells are arranged in short interweaving fascicles; when combined with the relatively pale, loose stromal tissue this confers a feathery appearance. Occasional, more cellular cases have a storiform pattern. The stroma contains delicate thin-walled capillaries, extravasated red blood cells, and scattered inflammatory cells, mainly lymphocytes and less often neutrophils. Up to 10% of cases contain multinucleate giant cells, most often osteoclastic in type ( Fig. 24.30 ).
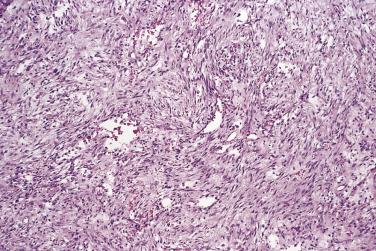
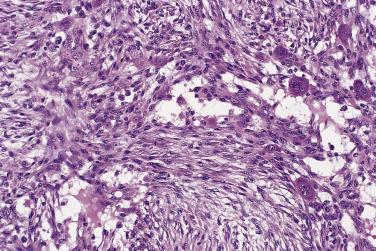
In some cases, especially in children, the stroma shows marked, diffuse myxoid change, whereas lesions of longer standing tend to show regressive stromal hyalinization and even keloidal change. A small proportion of cases show reactive new bone formation with plump osteoblasts, akin to the appearances in myositis ossificans (see Chapter 25 ); such cases are sometimes termed fasciitis ossificans (see upcoming section “ Fibro-Osseous Pseudotumor ”).
Immunohistochemically, as anticipated in any myofibroblastic lesion, the bulk of the cells shows positivity for panmuscle actin and smooth muscle actin, whereas desmin is most often negative. With regard to USP6 activation in these lesions, it has been appreciated recently that there are multiple alternative gene fusions (not involving MYH9), indicative of promoter swapping and having implications for molecular testing by reverse transcriptase polymerase chain reaction (RT-PCR). With regard to differential diagnosis, this should only infrequently be problematic in the appropriate clinical context. Generally speaking, few spindle cell sarcomas grow as rapidly as nodular fasciitis, and those that do will almost invariably show greater nuclear pleomorphism and areas of necrosis. Fibromatosis, particularly when located in the abdominal cavity or breast or occurring in the pediatric age group, may show fasciitis-like areas but is distinguished by its more infiltrative growth, longer fascicles, and more consistently collagenous stroma containing few, if any, inflammatory cells. Fibromatosis is also often immunopositive for β-catenin. Confusingly, at least a subset of cranial fasciitis cases appear to express β-catenin. Benign fibrous histiocytoma is cytologically more polymorphic, has a more consistently storiform or curlicue growth pattern, and usually shows more focal actin positivity.
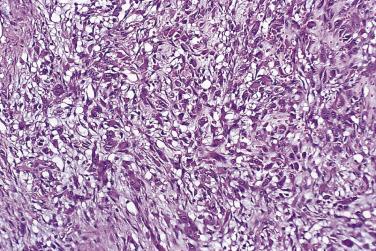
Proliferative fasciitis typically affects somewhat older adults than nodular fasciitis, with a peak incidence in the sixth decade. It is very uncommon in children. Predilection for the upper limb is not so marked as in nodular fasciitis, and a significant proportion of cases develop in the lower limb. Other than these differences, the clinical attributes, including rarity of recurrence, short history, and lesional size, are much the same as in the classic nodular type (see earlier). However, the presumed molecular pathogenesis remains unknown at this time.
As proliferative fasciitis is essentially a variant of nodular fasciitis, there are many shared features. The major and distinctive difference, which led to the separate designation of this variant, is the presence in proliferative fasciitis of large, rounded, or polygonal cells with amphophilic or rather basophilic cytoplasm and one or two vesicular nuclei containing a prominent nucleolus ( Fig. 24.31 ); these cells are commonly described as ganglion-like. The proportion of these cells within the lesion as a whole is variable, but when they are numerous there is frequently confusion with sarcoma, especially rhabdomyosarcoma. Other more minor differences from nodular fasciitis are that the stroma is often even more myxoid and may contain fibrinoid material. Thin-walled vessels may be especially prominent, heightening the resemblance to granulation tissue. In general, the lesional margins are more poorly defined than in the nodular variant. The large amphophilic cells are thought to be fibroblasts, and it is interesting that, despite their copious cytoplasm, they are usually actin negative, in contrast to the spindle-shaped myofibroblasts with which they are admixed. Probably the only realistic differential diagnosis is pleomorphic rhabdomyosarcoma. However, the latter is rarely subcutaneous and shows greater cytologic atypia and pleomorphism, frequently with atypical mitoses.
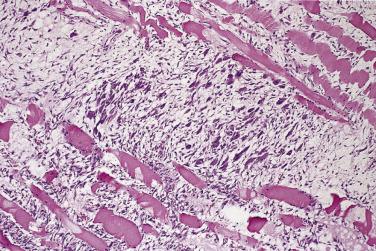
Proliferative myositis is essentially the intramuscular counterpart of proliferative fasciitis. In common with the latter it affects mainly older adults but shows anatomic predilection for the shoulder girdle and upper trunk. Cases in childhood are rare. The clinical duration often is less than 4 weeks. As with all other fasciitis-type lesions, the outlook is excellent, and recurrence is extremely infrequent. The histologic composition, including the presence of large ganglion-like cells, is the same as for proliferative fasciitis. A characteristic and distinctive architectural feature is the manner in which this process extends along fibrous septa and between individual muscle fibers, leaving the latter virtually intact ( Fig. 24.32 ). This produces a so-called checkerboard appearance of numerous individually separated eosinophilic muscle fibers set in a stroma of fasciitis. The growth pattern allows ready distinction from virtually any sarcoma and from myositis ossificans, which is more localized and does not preserve muscle fibers (see Chapter 25 ). In common with the latter, however, at least 10% to 20% of cases of proliferative myositis contain small foci of metaplastic bone or cartilage.
Fibro-osseous pseudotumor, also sometimes known as florid reactive periostitis, is another of the fasciitis-like myofibroblastic proliferations; it is distinguished only by its almost invariable localization on the digits and its propensity for new bone formation. Most affected patients are young adults, especially females, who present with a rapidly enlarging painful mass, typically adjacent to one of the phalanges. Radiologically there is often lesional calcification and a marked periosteal reaction. In contrast to bizarre parosteal osteochondromatous proliferations (Nora lesion) at the same site, which may also have a fibrous component, fibro-osseous pseudotumor is not attached to the underlying bone (see Chapter 25 ). Histologically, the appearances are essentially those of fasciitis with, in addition, focal irregular formation of osteoid, bone, and sometimes cartilage ( Fig. 24.33 ). The bone or osteoid is surfaced by plump osteoblasts, and the zoning pattern of lesional maturation that is evident in myositis ossificans (see Chapter 25 ) is absent in these cases, thereby increasing the risk of misdiagnosis as osteosarcoma. It should be remembered always that osteosarcoma in the hand, whether of bone or soft tissue origin, is extremely rare. Fibro-osseous pseudotumor has recently been shown to harbor USP6 rearrangement, comparable to nodular fasciitis.
Myositis ossificans, which is very closely related to the fasciitis family of lesions described here, is described in Chapter 25 . It is worth noting that histologically comparable lesions may also occur in the mesentery, typically after previous surgery. Aneurysmal bone cyst (described in Chapter 25 ) may also occur primarily in soft tissue, usually in a limb, and, at least in some cases, shows morphologic as well as molecular genetic overlap with myositis ossificans.
Elastofibroma is a relatively uncommon lesion that is usually encountered beneath one or both of the scapulae of elderly patients, with a marked predilection for females. Subclinical lesions may be more frequent. The usual presentation is with a slow-growing, poorly defined mass, which is attached to the periosteum of the ribs, although many cases are discovered by chance in the context of computed tomography (CT) imaging of the thorax. The lesion has long been believed to result from long-standing repeated trauma to the tissues between the scapulae and the chest wall, although an expected correlation with heavy manual labor is not borne out reliably, and striking examples of case clustering, most notably in some parts of Japan, suggest a genetic predisposition, at least in some cases. Furthermore, some cytogenetic and molecular data have shown these lesions to be clonal, and to share consistent genomic alterations. Rare examples occur in the lower extremity, and histologically comparable lesions have also been described in the gastrointestinal tract and other visceral locations. There is no tendency for local recurrence.
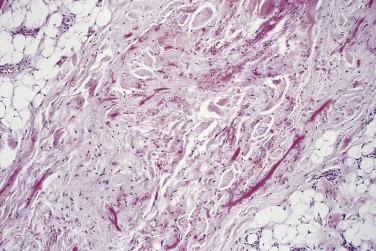
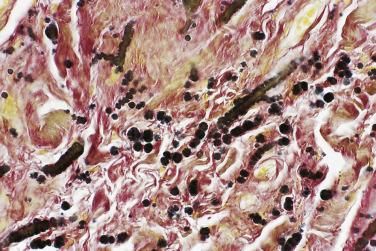
Elastofibroma is almost invariably very poorly defined, merging with adjacent normal tissues, and measures up to 10 cm. The cut surface resembles dense fibrofatty tissue. Histologic examination reveals irregular bands of dense, fairly hypocellular, collagenous fibrous tissue within which there are numerous eosinophilic elastic fibers ( Fig. 24.34 ). When these are highlighted by the use of elastic stains, they are seen to be arranged either as thick beaded or serrated fibers or in globoid aggregates ( Fig. 24.35 ), quite similar to the changes seen in cutaneous pseudoxanthoma elasticum. There was formerly much debate about whether these were true elastic fibers or just collagen showing elastotic degeneration, but there is clear evidence that this is truly elastin. Of additional interest with regard to pathogenesis is that, in addition to bilaterality irrespective of handedness, affected patients may show comparable elastofibroma-like changes at a visceral location, again suggesting the possibility of a genetic predisposition.
Fibrous hamartoma of infancy almost always arises in the first 2 years of life and shows a marked predilection for males. The most common locations are the axilla, upper arm, and shoulder region, but cases have been described at a wide variety of sites, notably the inguinoscrotal region. The child is usually found to have a firm, slow-growing subcutaneous mass, which is most often mobile and tends to measure less than 5 cm. In exceptional cases there may be more than one lesion. Local recurrence may occur rarely; in general, however, these lesions are nonrecurring but show no convincing tendency for spontaneous regression.
Fibrous hamartomas of infancy are circumscribed but poorly defined, firm, fibrofatty tumors that occupy the deep dermis and subcutis. Muscle involvement is extremely unusual. Histologically there are four principal components in varying relative proportions ( Fig. 24.36 ):
Club-ended fascicles of palely eosinophilic actin-positive myofibroblasts, the nuclei of which tend to be tapering and wavy
Small round to ovoid nests of undifferentiated spindle or stellate cells set in myxoid stroma containing delicate vessels ( Fig. 24.37 )
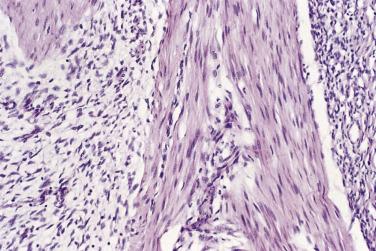
Areas of disorderly collagenous fibrous tissue containing vessels and inflammatory cells
Islands of mature adipocytes
In general, the two different fibrous components are predominant; some cases may show marked hyalinization with occasional multinucleate cells. The organoid growth pattern of these lesions remains unexplained but allows for no real differential diagnosis. Exceptional cases may show focally sarcomatous morphology of unknown clinical significance. These tumors have been shown to harbor EGFR mutations.
As discussed in Chapter 3 (see p. 81 ), myofibroma(tosis) in its various forms is increasingly regarded as a proliferation of perivascular contractile cells (myopericytoma), but because this represents a relatively recent conceptual change that is not widely known, these lesions are also described under their more traditional terminology later in this chapter.
Infantile myofibroma(tosis), formerly known as congenital generalized fibromatosis, typically presents before the age of 2 years, is congenital in up to 30% of cases, and shows a moderate preponderance in males. At most 10% of patients have multiple lesions, although this figure was thought to be higher in the past. The majority of lesions arise in skin and superficial soft tissue, especially of the head and neck region or trunk, but lesions in bone are also quite common (see Chapter 25 ), and in multicentric cases there may very occasionally be visceral involvement, especially of the gastrointestinal tract or lungs. This author has also encountered very rare examples of solitary visceral lesions. A small proportion of cases are inherited, seemingly in an autosomal dominant fashion. The clinical course for soft tissue and bone lesions is entirely benign, and local recurrence is rare. A large proportion of lesions regress spontaneously. By contrast, the rare presence of multiple visceral lesions may be associated with a fatal outcome. Solitary lesions in older patients are discussed separately (see later).
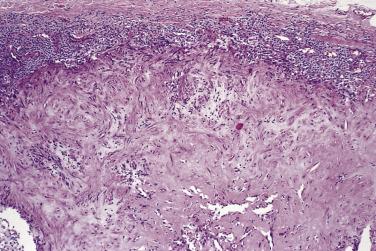
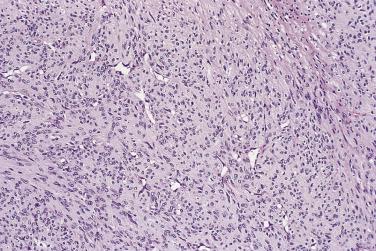
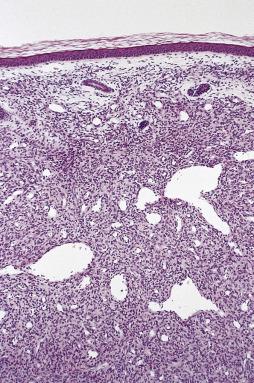
Most lesions measure less than 3 to 4 cm. The majority are well circumscribed but unencapsulated, with a firm cut surface. Histology reveals two components in markedly varying proportions. These comprise eosinophilic spindle cell whorls and fascicles with bland myoid features, and more primitive areas composed of smaller, round to spindle cells with limited eosinophilic cytoplasm and more rounded nuclei ( Fig. 24.38 ). These less differentiated cells are commonly arranged around small, branching, hemangiopericytoma-like vessels ( Fig. 24.39 ). The eosinophilic spindle cell areas, which tend to predominate at the periphery, often hyalinize over time, and the stroma may appear basophilic and pseudochondroid. Mitoses are quite common in both cell types. The more primitive areas often show necrosis, and there also may be calcification. What has been described as vascular invasion (more accurately subendothelial proliferation of perivascular spindle cells) is quite frequently present. Both cell types show at least focal actin positivity, in keeping with their myofibroblastic or myopericytic nature, but this is most pronounced in the spindle-shaped cells. In lesions biopsied in very young infants, especially those with multicentric disease, the primitive hemangiopericytoma-like component is generally predominant ( Fig. 24.40 ). As a consequence, and in view of their shared clinical features, it became apparent that so-called infantile hemangiopericytoma represents one end of the spectrum of infantile myofibromatosis and does not merit designation as a separate entity (see Chapter 3 ). The characteristic biphasic appearance in typical cases of infantile myofibromatosis rarely allows any differential diagnosis, although very cellular lesions may show morphologic overlap with infantile fibrosarcoma. In contrast to NTRK3 rearrangement (seen in infantile fibrosarcoma), infantile myofibromatosis and its solitary counterparts (in both children and adults) typically show PDGFRB mutations. A subset of more cellular cases show SRF-RELA gene fusions.
Juvenile hyaline fibromatosis is an exceedingly rare disorder of infants and children, which appears to have autosomal recessive inheritance. It is characterized by multiple slow-growing dermal or subcutaneous tumors, especially in the head and neck region and upper trunk, often associated with gingival hypertrophy, severe flexural limb contractures, and bone lesions. There is no mental retardation. It overlaps with infantile systemic hyalinosis, which has a more severe phenotype, including visceral involvement. Histologically, these lesions are composed of copious eosinophilic, homogeneous ground substance containing bland, unevenly dispersed fibroblasts (with adjacent retraction artifact) and occasional thin-walled vessels. Lesions excised early in life seem to be relatively more cellular. The precise nature of the eosinophilic hyaline material remains undecided. Mutations in ANTXR2 (formerly CMG2 ) have been identified as the cause of these disorders, suggesting that an abnormality of basement membrane assembly may be responsible for the phenotype.
Fibroma of tendon sheath is a relatively uncommon lesion presenting usually in young to middle-aged adults, mainly males, as a firm nodule most often located on the upper limb, especially the fingers. Evident attachment to a tendon is common. Occasional examples are seen around the knee and may rarely be intraarticular. Following marginal or incomplete excision, these lesions may recur locally, sometimes more than once.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here