Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Advances in the treatment of rejection have mirrored the evolution of liver transplantation. In the earliest days of transplantation, clinical outcomes were marred by fatal complications related to ischemia, preservation injury, surgical technique, and infection. Grafts that would survive long enough to succumb to rejection were rare. Moreover, tackling rejection was not yet part of the transplant paradigm, and organs that did survive were eventually ruined by rejection. Despite the first successful transplantation of kidneys between identical twins, theoretical immunologists and other naysayers predicted devastating consequences of transplantation between individuals who were not genetically identical.
During the late 1960s and early 1970s, however, considerable progress was made in the surgical technique and selection and postoperative care of transplant patients. Early work in immunosuppression began changing the landscape of clinical transplantation. Surgeons started to transplant successfully and consistently between unrelated individuals. With these improvements the notion of halting or even reversing the rejection process came to the fore. Notable advances were achieved in the area of pharmacological modulation of the immune system. Immunosuppression, at first akin to an unwieldy hammer, became more precise and directed at specific steps in the immunological process. The advent of less toxic medications, such as cyclosporine and later tacrolimus, revolutionized transplantation of all organs. Concurrently, the side effects of these medications, often apparent only after long-term use, were tempered, and new, less toxic immunosuppressive protocols were devised. With these discoveries, liver transplantation, once an experimental modality, became the accepted standard of care for most patients suffering from end-stage liver disease.
Currently, long-term graft survival is a reality. As a consequence, there continue to be new research and developments in the treatment of rejection. Like many of the early once-fatal complications, the diagnosis of rejection has become less devastating and significantly less ominous. In the quotidian life of the long-term liver transplant patient, morbidity from the chronic use of immunosuppressive agents has surpassed rejection as the major concern. Even chronic rejection (CR) is now responsible for only a small proportion of graft loss and mortality, whereas immunosuppression-related sequelae such as sepsis, atherosclerosis, renal failure, and posttransplant malignancy form the overwhelming concerns.
In this chapter we review the treatment of several forms of rejection after liver transplantation. Because preceding chapters have reviewed the pathological process of rejection and the pharmacological characteristics of immunosuppressive agents in detail, we concentrate here on the treatment of rejection.
Rejection can be classified as acute, late acute, chronic, and hyperacute. Each of these processes requires a thoughtful therapeutic approach. Acute rejection (AR) is the most common form of rejection and usually responds well to steroid therapy. CR was once believed to be irreversible, but over the past decade there have been improvements in its treatment. Late acute rejection (LAR) represents the continuum between acute and chronic rejection. Hyperacute rejection (HAR) is rare, but extremely rapid, and can lead to graft loss.
The definition of AR has evolved over the past 40 years. Broadly defined as graft inflammation, AR represents immunological injury elicited as a consequence of the genetic disparity between donor and recipient. AR can develop anytime from several days to more than a week after transplantation, with the peak incidence occurring between 8 and 10 days after transplantation. The vast majority of AR episodes take place within the first 3 months after transplantation. AR is the most common form of rejection and, fortunately, the most responsive to treatment. Some authors have documented the incidence of AR in orthotopic liver transplantation to be as high as 70%. Moreover, after a first diagnosis of AR, 20% of patients subsequently experience a second attack, and 4% eventually have a third episode.
Fortunately for this group of patients, the sequelae of AR in liver grafts are generally less significant than in other solid organs. This difference may be due to the protective mechanisms and regenerative capabilities of the liver. In fact, the liver itself may be an immune modulator. The liver’s capacity to affect the immune system is most dramatically illustrated by its impact on other organs in combined transplants. Patients who receive combined grafts consisting of a liver with any other organ tend to have lower rates of all forms of rejection. Although it remains unclear why the liver lessens the impact of the immune system on transplanted organs in general, and the concept is controversial, proposed mechanisms include the development of chimerism by soluble human leukocyte antigen (HLA) secreted by the liver graft.
Additionally, the liver differs from other solid organs in the experimental observation of tolerance. Experimental hepatic tolerance was first recognized in 1965 when liver grafts transplanted between outbred pigs survived without immunosuppression. In later animal studies, Calne and Mazariegos and colleagues demonstrated the immunomodulatory effect of liver transplantation when isogenic skin grafts had prolonged subsequent survival. In humans there have been anecdotal reports of tolerance developing in transplanted patients who were weaned from, or noncompliant with, immunosuppressive therapy.
Several risk factors for the development of AR have been established. The most common risk factor is inadequate immunosuppression, with AR being more likely to develop in patients who are not receiving or absorbing adequate immunosuppression. Other important risk factors include having an underlying autoimmune disease, being the female recipient of a male donor organ, and having fewer HLA matches at the DR locus. The type of immunosuppression used may also have an impact on the development of AR; patients who receive tacrolimus have lower rates of rejection than those who receive cyclosporine.
AR is mediated predominantly by effector (killer) T cells that develop in response to donor antigens from the transplanted organ. This inflammation initially affects the biliary epithelium and subsequently the hepatocytes. Histological examination of liver tissue reveals a mixed cellular infiltrate composed predominantly of lymphocytes localized to the portal tracts and the central vein areas. Early histological changes, however, are often nonspecific and may be confused with recurrent hepatitis C infection. With time or severity, damage to bile ducts and to the central vein endothelium occurs.
Most cases of AR are manifested as asymptomatic biochemical changes. These biochemical changes almost always occur before clinical signs and symptoms and are a manifestation of the cholestatic inflammation first affecting the biliary epithelium and subsequently the hepatocytes. Accordingly, subtle increases can first be seen in bilirubin, alkaline phosphatase, and γ-glutamyltransferase levels. Shortly thereafter, a rise in serum transaminase levels is seen. Although no absolute biochemical parameters can currently be used to define an episode of rejection, there are some experimental methods currently being evaluated. MicroRNAs have emerged as important regulators of immune function. Although currently not a proven method to detect acute rejection, early results show that detection of certain microRNAs in the serum can be linked to episodes of acute rejection, specifically, miR-122, miR-101, and let-7b, which are preferentially expressed by liver. These circulating microRNAs have been shown to be extremely stable and protected from RNase-mediated degradation. As examination of tissue biopsy specimens is costly, risky, and invasive, these microRNAs might prove to be beneficial in the future. Currently clinicians base their suspicions on a relative rise in liver function tests, the amount of time elapsed since transplantation, and the underlying disease.
Clinical findings tend to appear later in the course of rejection and reflect more significant injury to the graft. These signs and symptoms can include fatigue, fever, malaise, abdominal pain, decreased bile output, increasing ascites, and hepatomegaly ( Table 93-1 ). If bile is available for inspection through a T tube, it may be lighter in color and less viscous than usual.
| Fever |
| Malaise |
| Abdominal pain |
| Ascites |
| Hepatomegaly |
| Anorexia |
The gold standard for the diagnosis of AR is percutaneous liver biopsy. Although invasive, this diagnostic procedure affords the greatest sensitivity and specificity in the diagnosis of AR. However, percutaneous liver biopsy is not without risk. Although bleeding is the most common complication, serious hemorrhagic complications can occur in 0.06% to 0.35% of cases. Other complications include pneumothorax, hemobilia, peritonitis, perforated viscus, and the development of an arteriovenous fistula. The risk for sustaining any of these complications is about 2%, and the risk for death hovers between 0.009% and 0.1%. Because the most common complication is bleeding, clinicians should attempt to correct any coagulopathy before performing this procedure.
On occasion, despite percutaneous biopsy, the diagnosis of rejection can remain in doubt, particularly because other causes of acute inflammation in the liver can be confused with AR. Most frequently, cytomegalovirus (CMV) infection and recurrence or persistence of hepatitis C make the diagnosis of AR quite difficult. Treating presumptive AR with increased immunosuppression can be devastating if, in fact, the actual diagnosis is hepatitis C. In these situations, repeat biopsy before treatment may be of help.
Protocol biopsies in the postoperative period are advocated in some centers as a way to treat rejection more effectively. By increasing sensitivity and specificity, protocol biopsies allow one to diagnose AR while it is still at the subclinical level. The underlying assumption in performing protocol biopsies is that early diagnosis and treatment of AR may be of benefit. Although no multicenter randomized controlled trial has ever been conducted, some centers treat based on histological criteria without biochemical evidence of graft dysfunction. These preemptive efforts have not been conclusively shown to be beneficial in liver transplant patients. Recent data suggest that this approach may be overly cautious and may lead to overtreatment.
From 15 studies of patients undergoing protocol biopsies, 302 patients were identified who had histological, but no biochemical evidence of rejection. Of these patients, only 36 subsequently experienced clinical rejection, 7 of which were steroid resistant and 9 of which were CR.
Despite the sensitivity and specificity of percutaneous liver biopsy, the procedure remains an invasive one with all the attendant risk for morbidity and mortality. Numerous investigators have searched for safer and less invasive diagnostic methods, as well as ones that would potentially diagnose AR at an earlier time point. Some centers have tried fine-needle aspiration. However, no large randomized controlled studies have examined this issue in liver transplant patients. Although the smaller bore of the fine needle may result in less trauma, this risk may be amplified by the need to perform multiple passes because insufficient quantity of tissue may be obtained with a single pass. In addition, the subtle changes of early AR may be missed because of sampling error, with a subsequent delay in diagnosis and treatment. “Overcalling” AR can also occur and result in a needless increase in immunosuppression with its attendant morbidity. Moreover, in cases in which CR is also being considered, fine-needle aspiration has no diagnostic role because a core needle biopsy is required to more accurately assess hepatic histological characteristics. A number of noninvasive methods have been examined in the laboratory to detect acute rejection early. The newest of these is microRNA profiling. MicroRNAs are a class of small nonencoding RNAs that are important regulators of gene expression.
Variable expression of microRNAs has been linked to pathological conditions in humans including malignant, metabolic, cardiovascular, and autoimmune diseases. Specifically, hepatocyte-derived microRNAs (HDmiRs) have been studied in the laboratory as potential markers for hepatocyte injury. It has been shown that one can detect a rise in serum HDmiR levels before a measurable rise in transaminase levels is observed. In the setting of acute rejection, the serum HDmiRs are elevated by up to 20-fold. These data suggest that this may be an emerging modality to detect liver injury and acute rejection early, even before a transaminitis occurs. Another new method involves donor circulating cell-free DNA in the serum of the recipient. Initially used in cardiac transplantation, it has recently been studied in liver recipients. In one study it was shown that donor cell-free DNA levels are 90% immediately after transplant and fall to less than 15% by day 10. In patients that had biopsy-proven rejection, these levels rose up to 50%. Biliary aspirates from indwelling T tubes, cytokine profiling, histological analysis of cellular aspirates, cellular Fas ligand analysis, and isotope scanning, along with other noninvasive technologies, have also been investigated in the laboratory and the clinic. All these methods, however, have been hampered by suboptimal sensitivity or specificity because of similarities between AR and other forms of acute inflammation. Recently quantification of granzyme expression in fine-needle aspirates has been investigated with moderate success. Although this technique has been used with a high level of accuracy for the diagnosis of AR in renal allografts, the presence of CMV infection appears to decrease the accuracy of this method in liver transplants.
Thus, despite numerous attempts to use noninvasive techniques over the past 20 years, none of these diagnostic methods have been validated or meet the sensitivity and specificity of percutaneous liver biopsy. Core needle liver biopsy remains the procedure of choice for the diagnosis of AR.
Before initiating treatment of AR, the clinician should consider other diagnoses ( Table 93-2 ). Potentially confounding differential diagnoses are often time dependent. Within the first month after transplantation, other possible diagnoses include preservation injury, early hepatic artery thrombosis, portal vein thrombosis, biliary leak, CMV infection, and intra-abdominal sepsis. After the early postoperative period until approximately a year after transplantation, recurrence or persistence of hepatitis C, drug toxicity, biliary strictures, and opportunistic infections may be confused with AR. After the first year, late mimics of AR can include late hepatic artery thrombosis, recurrent autoimmune hepatitis, recurrent primary sclerosing cholangitis, CR, and posttransplantation lymphoproliferative disorder (PTLD). Once treatment has started, every effort should be made to also eliminate or decrease potentially aggravating factors ( Table 93-3 ). Conditions such as diarrhea and drug interactions can lead to suboptimal levels of immunosuppression and hamper effective therapy and resolution of AR. As many as 50% of patients suffering from AR may have one or more of these aggravating factors.
| First Month After Transplantation |
| Vascular complications |
| Hepatic artery thrombosis |
| Portal vein thrombosis |
| Vena cava/hepatic vein obstruction |
| Biliary leak |
| Preservation injury |
| Intra-abdominal sepsis |
| First Year After Transplantation |
| Infection |
| Hepatitis C persistence/recurrence |
| Intrahepatic abscess |
| Opportunistic infections: cytomegalovirus, Epstein-Barr virus, herpes simplex virus |
| Biliary complications |
| Stricture |
| Cholangitis |
| Drug toxicity |
| Longer Than 1 Year After Transplantation |
| Chronic rejection |
| Recurrent disease |
| Autoimmune hepatitis |
| Primary sclerosing cholangitis |
| Posttransplantation lymphoproliferative disorder |
| Steatohepatitis |
| Malabsorption |
| Vomiting |
| Diarrhea |
| T-tube losses |
| Fistula output |
| Immunosuppression |
| Underdosing |
| Steroid-weaning protocols |
| Noncompliance |
| Adverse effects |
| Psychosocial issues |
| Concomitant disease |
| Infection |
| Recurrent disease |
| Drug interactions |
After the diagnosis of AR is established, therapy first consists of optimization of maintenance immunosuppression ( Fig. 93-1 ). The dosage of the patient’s mainstay calcineurin inhibitor can be increased, and other immunosuppressive agents such as mycophenolate mofetil, azathioprine, and rapamycin, if not currently being taken, can also be added. If the AR does not appear to respond to these initial measures, intravenous and high-dose steroid therapy can be implemented. Antilymphocytic antibodies and newer agents such as interleukin-2 receptor antagonists (IL-2RAs) are reserved for rarer cases of steroid-resistant AR or for the occasional severe case of untreated AR.
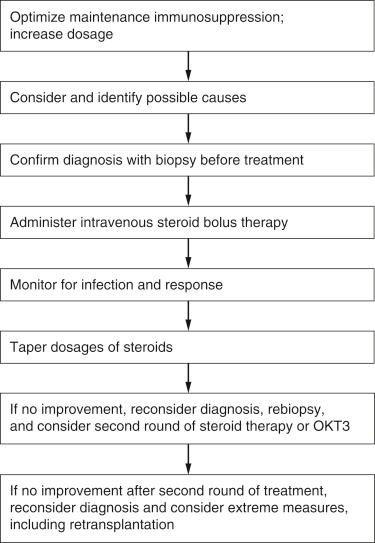
Such treatment is associated with a constellation of potential toxicities, including a risk for excessive immunosuppression and infection. Of concern as well is the risk for PTLD. These risks and benefits must be considered in each individual case. Although treatment of the AR episode will ameliorate or reduce symptoms, liver grafts generally do not suffer from long-term sequelae as a result of early episodes of AR. Unlike renal allografts, there is no direct correlation between the number of AR episodes and long-term graft or patient survival. (There is some controversy about these statements.) Generally, however, once AR is diagnosed from histological and clinical or biochemical evidence, the benefits of treatment outweigh the risks.
After AR is adequately treated, maintenance immunosuppression may be changed or increased for several months to reduce the probability of relapse. For patients being treated with cyclosporine-based immunosuppressive regimens, changing maintenance therapy to tacrolimus may also help treat and decrease the incidence of acute and chronic rejection. Other immunosuppressive agents may be added, and patients previously receiving dual therapy may temporarily be maintained on triple or even quadruple therapy.
Pediatric patients in whom AR is diagnosed have very specific concerns in their treatment, and these issues are reviewed later.
Corticosteroids were first used clinically for transplantation of renal allografts during the early 1960s. For many years, corticosteroids remained a mainstay of immunosuppressive therapy and ceded their major role only when calcineurin inhibitors were accepted into regular clinical practice in the mid-1980s. Over the past 4 decades the panoply of complications related to long-term use has led most transplant centers to use them sparingly. In AR, however, corticosteroids remain first-line therapy.
Corticosteroids are 21-carbon steroid hormones derived from the metabolism of cholesterol, and their efficacy is dependent on the presence of a hydroxyl group on carbon 11. Corticosteroids have nonspecific antiinflammatory effects. After binding to high-affinity receptors in cytoplasm, the steroid-receptor complex enters the nucleus and binds to DNA. Transcription is triggered, with the eventual synthesis of proteins. The upregulated proteins that result from steroid binding are responsible for many of the antiinflammatory effects. Among the many immunosuppressive effects of corticosteroids: phagocytosis by neutrophils and monocytes is inhibited and disruption of T-cell activation occurs.
No consensus has been reached on the optimal form, dosage, or length of treatment with corticosteroids. Trials have been conducted to compare different protocols of steroid therapy, and unfortunately, it is difficult to extrapolate general conclusions from these small and heterogeneous reports. Moreover, not all steroids are equivalent in strength, and their relative potencies should be known to the administering clinician ( Table 93-4 ).
| Agent | Potency | Half-Life (min) |
|---|---|---|
| Hydrocortisone (Solu-Cortef) | 1 | 90 |
| Prednisone | 4-5 | 60 |
| Prednisolone | 4-5 | 200 |
| Methylprednisolone (Solu-Medrol) | 6-7 | 240 |
One possible protocol for steroid treatment is given in Table 93-5 . Regardless of which steroid protocol is used, several caveats should be heeded. Given the debilitated state of the liver in AR, the steroid chosen should not be dependent on hepatic metabolism for its active form. The half-life should be long enough to afford reversal of the rejection process but not so long that long-term steroid side effects are introduced. Finally, the steroid should have as little mineralocorticoid effect as possible to avoid sodium retention, weight gain, and hypertension.
| Day 1 | Solu-Medrol, 1000 mg IVPB × 1 over 1-hr period |
| Day 2 | Solu-Medrol, 1000 mg IVPB × 1 over 1-hr period |
| Day 3 | Solu-Medrol, 50 mg IVP q6h × 4 doses |
| Day 4 | Solu-Medrol, 40 mg IVP q6h × 4 doses |
| Day 5 | Solu-Medrol, 30 mg IVP q6h × 4 doses |
| Day 6 | Solu-Medrol, 20 mg IVP q6h × 4 doses |
| Day 7 | Solu-Medrol, 20 mg IVP q12h × 2 doses |
| Day 8 | Resume prednisone, 20 mg PO every day |
The most common initial step in the treatment of AR is a series of intravenous steroid boluses administered over a 3-day period, followed by a scheduled tapering dosage of an oral steroid. Intravenous steroid administration is used initially because this route of administration results in the fastest and most consistent serum levels. Usually well tolerated, this treatment protocol can be administered to outpatients, but it may be associated with increased rates of sepsis and an accelerated progression to cirrhosis in patients with hepatitis C. While undergoing treatment, patients should be regularly assessed for both clinical and biochemical responses. Approximately 90% of patients with AR respond to steroid treatment.
In patients without clinical or biochemical improvement, ongoing rejection should be suspected. A second percutaneous liver biopsy should be performed and liver histological analysis reexamined. Patients whose biopsy findings continue to be consistent with rejection may have steroid-resistant AR, and other treatment options must be entertained. Occasionally these patients are first treated with another course of intravenous steroid therapy, particularly if they have responded in the distant past or if there is evidence of even slight improvement. However, for the most part, patients with steroid-resistant AR are treated with other therapeutic modalities, and their overall outcomes tend to be worse than those of patients who respond to steroid treatment.
Early immunosuppression tended to have more generalized effects on the immune system. The development of antilymphocyte antibodies, however, allowed clinicians to target the immune system more selectively. The first antilymphocyte antibodies were obtained by using the serum of animals (e.g., horses or rabbits) challenged with recipient lymphocytes. These early antibody preparations, called antilymphocyte antibodies or antilymphocyte globulins (ALGs), were polyclonal with a broad and relatively unselective spectrum of activity against the immune system.
Initial attempts to use ALG were complicated by occasional severe anaphylactic-like reactions because of the presence of foreign animal haptens. As laboratory techniques improved, a “second generation” of more selective antilymphocyte antibodies was created, which resulted clinically in a reduction in anaphylactic-like reactions and more selective lysis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here