Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The congenital malformation characterized by origin of the arterial trunks from morphologically inappropriate ventricles has probably been the source of as much confusion and controversy as any other single topic in pediatric cardiology. When Matthew Baillie described the first case, he had no problems with nomenclature, describing the entity in accurate fashion as a “singular malformation.” Since then, the entity has most usually been described simply as transposition . However, it has been defined in many different ways and has been used to describe many different types of congenital cardiac malformations. Arguments have centered on whether the morphologic arrangement of the arterial trunks should be defined primarily according to their ventricular origin or on the basis of an anterior location of the aorta relative to the pulmonary trunk. Such disagreements have been nonproductive. In the clinical setting, it is the origin of the arterial trunks from the ventricles that is paramount because this feature determines the patterns of flow of blood through the heart, irrespective of the relationships of the arterial trunks. The combinations of segmental connections then produce the variations still described in terms of “transposition.” We will describe the combination to be discussed in this chapter simply as transposition, including only those patients having the specific combination of concordant atrioventricular and discordant ventriculoarterial connections ( Fig. 37.1 ).
To the best of our knowledge, it was John Farre who first used the term transposition in the context of a cardiac malformation. Subsequent to this description, it was usually considered that the aorta needed to be anterior to justify diagnosis, combined with support of the aortic valve by a muscular infundibulum. However, some segmental combinations associated with an anterior aorta produce physiologic correction of the circulatory patterns, such that the systemic venous blood reaches the lungs despite being pumped by the morphologically inappropriate ventricle. In two such combinations, there is also so-called anatomic correction, in that the aorta takes its origin from the morphologically left ventricle despite its anterior position. In two additional variants, the circulations are in parallel rather than in series, despite the fact that the aorta arises in concordant fashion from the morphologically left ventricle. It was this use of “corrected” in an anatomic sense, being dependent on the definition of transposition as an anterior aorta, that produced the potential for confusion. Further problems arose when it was recognized that the aorta could arise inappropriately from the morphologically right ventricle when in posterior position and with its valve in fibrous continuity with the mitral valve. It makes little sense, when considering clinical presentation, to describe a patient with an anterior aorta arising from the left ventricle as having “transposition” when the atrioventricular connections are also concordant, just as it makes little sense to suggest that patients with discordant ventriculoarterial connections, but with a posterior aorta, do not have transposition. Thus, over recent years, there has been an increasing groundswell of opinion in favor of defining transposition on the basis of the origin of the great arterial trunks from morphologically inappropriate ventricles. Therefore, the system that we currently use bases the use of “transposition” on the combination of specific atrioventricular and ventriculoarterial connections. This is the solution now adopted by the International Working Group on Nomenclature and that which was advocated by the consensus statement prepared on behalf of the Society of Thoracic Surgeons. The approach recognizes the need also to describe, when appropriate, both arterial relationships and infundibular morphology. In this way, all potentials for confusion are avoided.
Transposition was responsible for up to 20% of cardiac deaths in infancy prior to the era of surgical correction. Although no definite etiologic factors have been identified, the condition is held to be more frequent in infants of diabetic mothers. Maternal intake of alcohol or poor nutrition during pregnancy may increase the risk of transposition in the offspring, whereas addition of folic acid to the maternal diet has been associated with reduced risk.
Males are affected two to three times as frequently as females. Transposition does not seem to coassociate with known major genetic disorders, and most cases are sporadic. The increased presence of copy number variations in patients with transposition has allowed for identification of candidate genes. In general, genes affecting laterality and the nodal pathway have been associated with the disease.
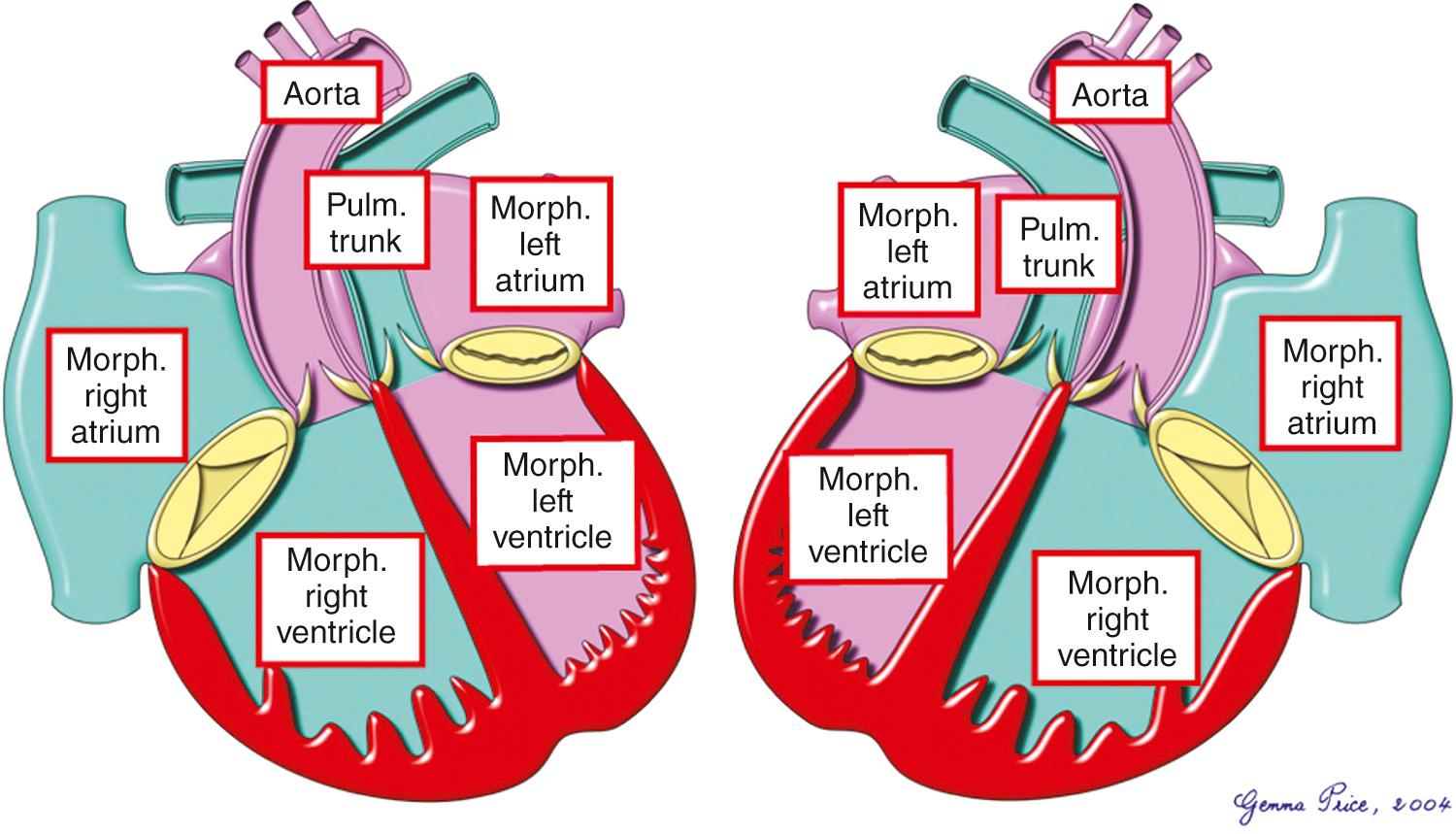
As emphasized, it is the basic combinations of concordant atrioventricular and discordant ventriculoarterial connections that produce the entity we call transposition ( Fig. 37.1 ). The anatomic situation can be complicated by the presence of a ventricular septal defect, an obstruction within the left ventricular outflow tract, both of these malformations, or by other associated malformations. Cases with an intact interventricular septum and no obstruction of the left ventricular outflow tract are usually considered simple, even if they are complicated by other lesions such as persistent patency of the arterial duct.
The combination of segmental connections ( Fig. 37.2 ) can be found with either usual or mirror-imaged atrial arrangements ( Fig. 37.1 ). However, this specific combination cannot exist when there is isomerism of the atrial appendages. Hearts with isomeric atrial appendages can, of course, have biventricular atrioventricular connections, right-hand topology, and discordant ventriculoarterial connections. Such hearts are closely related to transposition as it is defined for the purposes of this chapter, but the patients with isomeric atrial appendages have grossly abnormal venoatrial connections as their major feature (see Chapter 26 ). In the arrangement as defined for the purposes of this chapter, the atrial anatomy is basically normal, although most frequently the oval foramen is patent, or there is a deficiency of the floor of the oval fossa. Even if the flap valve overlaps the rim of the oval fossa, it is flimsy and can be ruptured easily by balloon septostomy. In keeping with this normal atrial anatomy, the sinus and atrioventricular nodes are in their anticipated position.
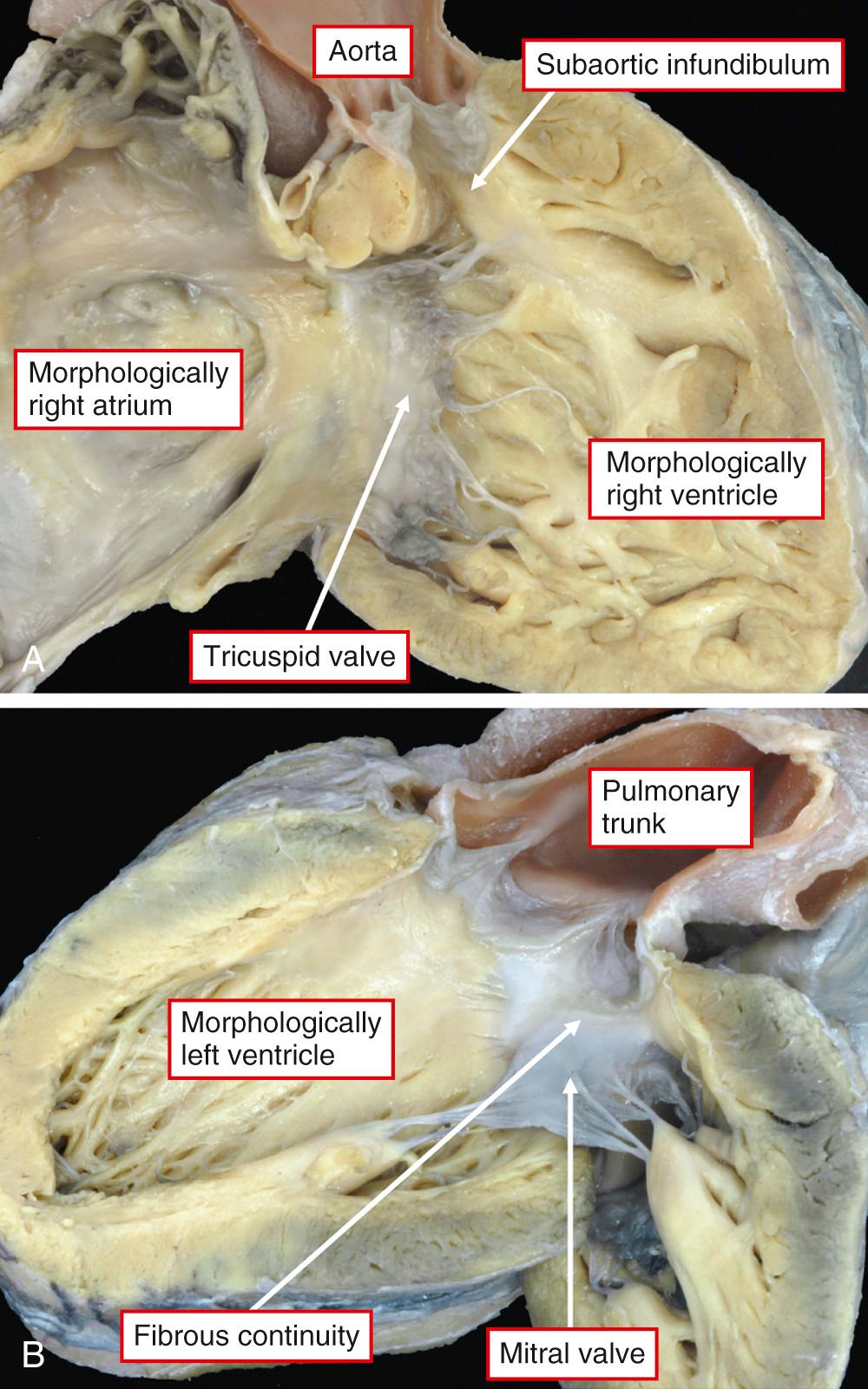
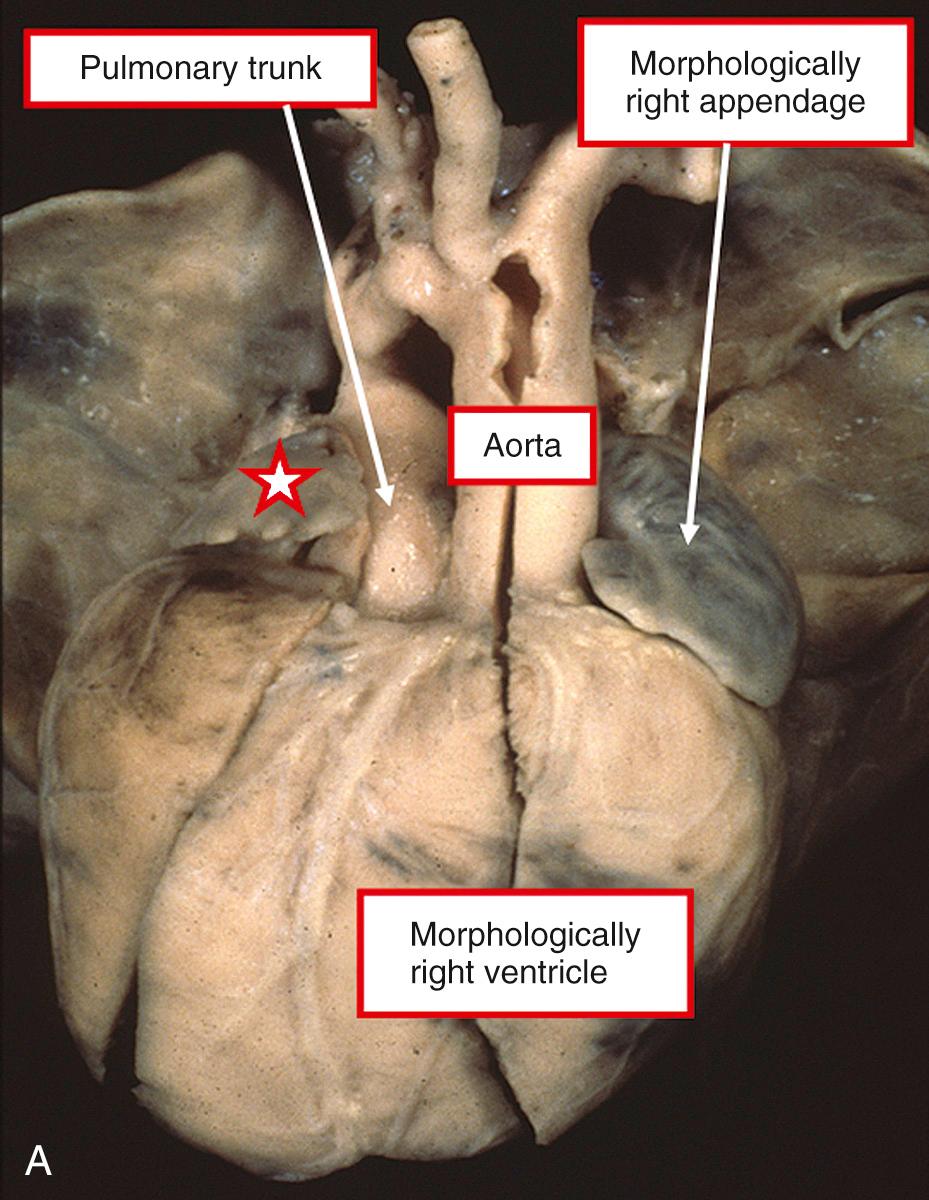
Unlike the atrial chambers, ventricular morphology is subtly different from normal. The ventricular septum is much straighter than usual, not showing the multiple curves so typical of the normal heart. The pulmonary valve is not wedged as deeply between the mitral and tricuspid valves as is the aortic valve in the normal heart. This, in turn, means that the area of off-setting of the leaflets of the atrioventricular valves is much less marked, as is the area occupied by the membranous septum. Another consequence of this abnormal arrangement is that the ratio of the dimensions of the inlet and outlet components of the ventricular mass are abnormal in favor of the outlet dimension, although not to the extent seen in atrioventricular septal defects (see Chapters 31 and 32 ). At birth, the walls of the morphologically left ventricular wall are marginally thicker than those of the right ventricle. The right ventricular mural thickness then rapidly increases in the first 2 years of life, becoming much thicker than that of the left ventricle. The most obvious external abnormality is the relationship of the aorta to the pulmonary trunk. In the majority of patients with an intact ventricular septum, the aortic root is to the right of the pulmonary trunk in hearts in the setting of usual atrial arrangement ( Fig. 37.2 ) and to the left in the mirror-imaged variant ( Fig. 37.3 , left panel). Patients are found, nonetheless, with usual atrial arrangement and intact ventricular septum when the aorta is to the left ( Fig. 37.3 , right panel). Rarely, the aorta may be right sided and posterior, even when the ventricular septum is intact.
When the ventricular septum is intact, the aorta almost always has a complete muscular infundibulum, while the leaflets of the pulmonary valve are in fibrous continuity with those of the mitral valve ( Fig. 37.2 ). Variations in this infundibular morphology (e.g., the presence of bilaterally complete muscular infundibulums) are not seen nearly as frequently when the ventricular septum is intact as when there is a ventricular septal defect (discussed later).
Whenever the position of the aortic root is abnormal, the origins of the coronary arteries deviate from those found in the normal heart. Without exception, the arteries continue to arise from one or other, or both, of those aortic sinuses that face, or are adjacent to, the pulmonary trunk ( Fig. 37.4 ).
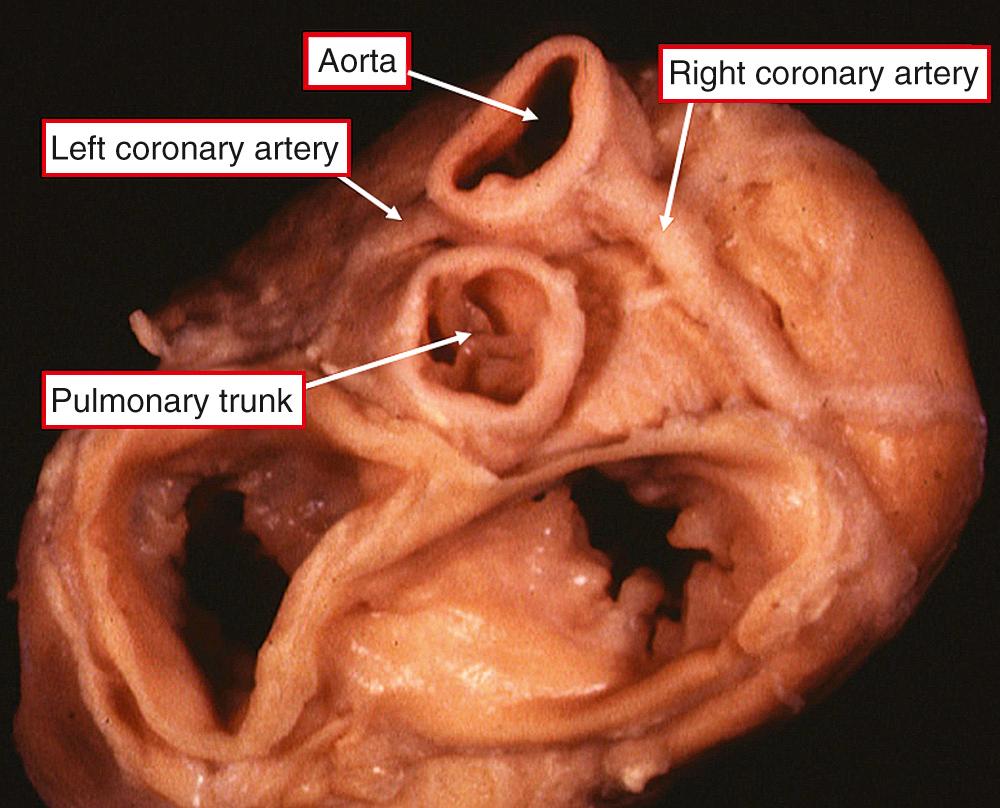
The epicardial course of the arteries arising from these adjacent sinuses is much less predictable. It is best to use a descriptive approach to account for the multiple variations, taking note separately of the origins of the three major coronary arteries from the aortic sinuses and their course relative to the vascular pedicle. Particular attention should be paid to any intramural course, along with the location of the artery supplying the sinus node. Although the coronary arteries always arise from one or both of the aortic sinuses that are adjacent to the pulmonary trunk, the position of these sinuses in space can vary according to the relationship of the arterial trunks. Therefore, when describing the sinusal origin, it is advantageous to use a system that is independent of spatial relationships. This can be achieved by considering the aortic sinuses as they would be visualized, figuratively speaking, by an observer standing in the nonadjacent aortic sinus and looking toward the pulmonary trunk ( Fig. 37.5 ). Irrespective of the relationships of the arterial trunks, the sinuses supporting the coronary arteries are always located to the right and left hands of the observer. The sinus as seen to the right is universally known as sinus 1, while that to the left is sinus 2 . All patterns are then accounted for on the basis of whether the right, circumflex, and anterior interventricular arteries arise from sinus 1 or from sinus 2. Most usually, the arteries arise within the aortic sinuses or at the level of the sinutubular junction, albeit usually eccentrically placed within the sinus. In some instances, the arteries can take a high origin above the sinutubular junction. Of more significance is the arrangement when the arteries take a tangential course through the aortic wall, crossing the attachments of the valvar leaflets at the sinutubular junction. This is the so-called intramural origin. In addition to sinusal origin ( Fig. 37.5 ), epicardial course is also important, in particular retropulmonary or anteroaortic location of any of the three major coronary arteries. The artery to the sinus node is of further significance. It can arise from the initial course of either the right or the circumflex coronary arteries, or it can take a direct origin from one or other of the facing aortic sinuses. However, its most important variation is when it crosses the lateral margin of the right atrial appendage. In such a lateral position, it is at surgical risk during a standard atriotomy.
The most significant, and frequently occurring, associated lesion in transposition is a ventricular septal defect. As with such defects found in the setting of concordant ventriculoarterial connections, these may be small, large, or multiple, and they can be located within any part of the ventricular septum ( Fig. 37.6 ). The most characteristic defects are those that open beneath the ventricular outlets, with the muscular outlet septum being malaligned relative to the rest of the ventricular septum and located within the right ventricle. Such defects, which occupy a subpulmonary position when seen from the left ventricle, may have a muscular posteroinferior rim ( Fig. 37.7B ) or may extend to become perimembranous.
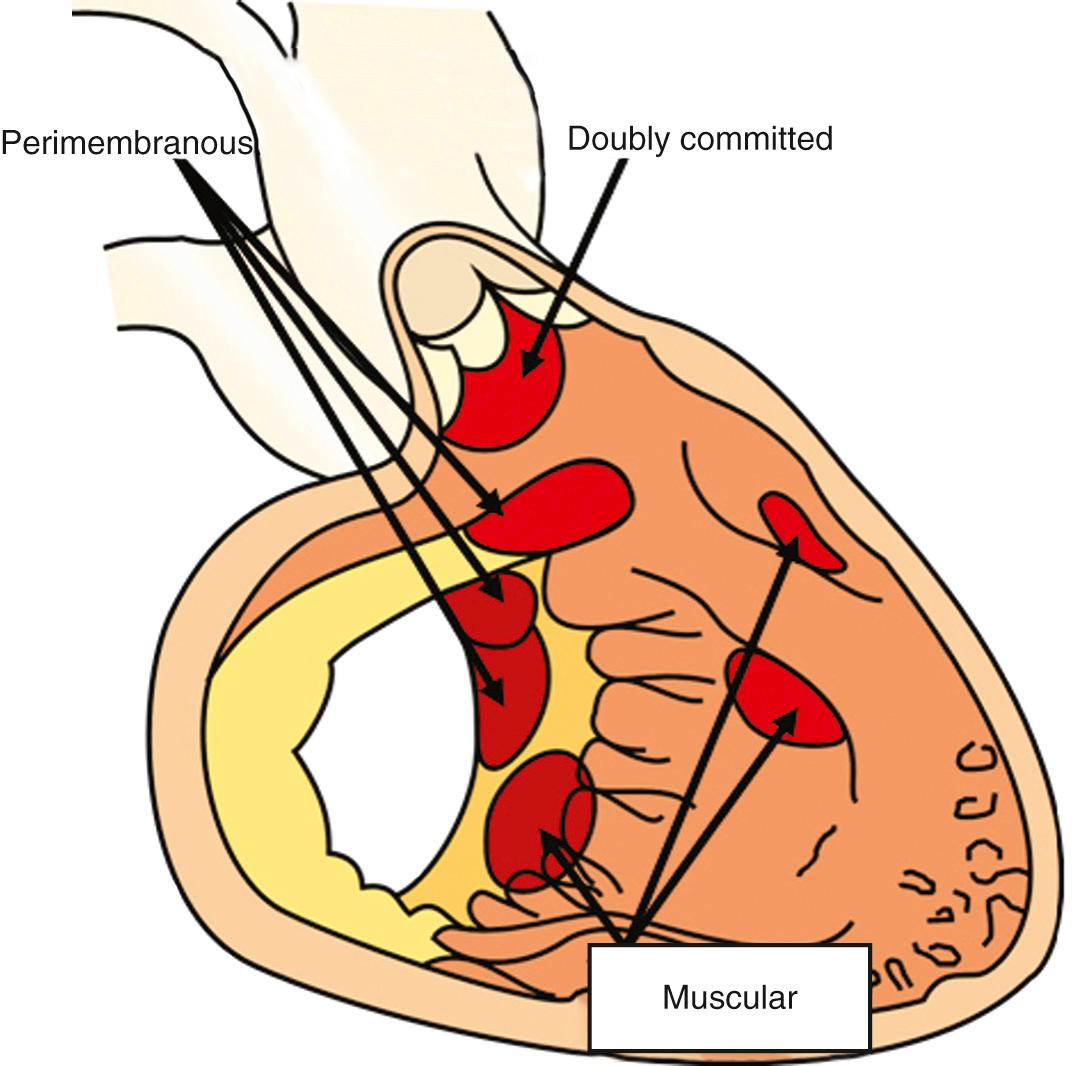
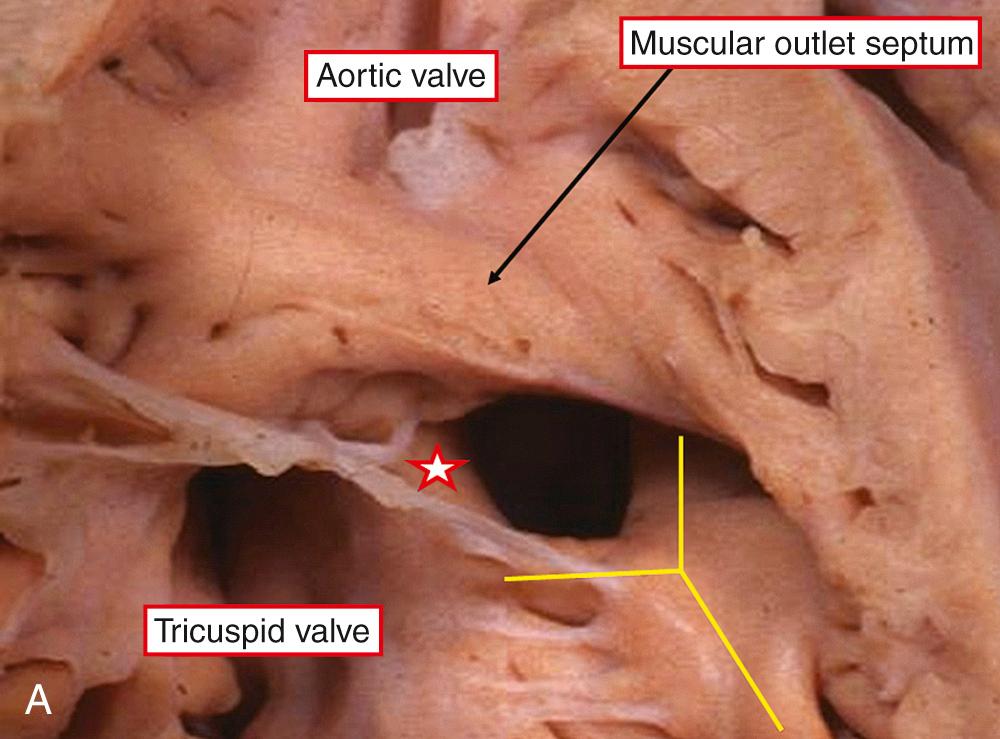
The outlet septum can also be malaligned into the left ventricle. This then narrows the left ventricular outflow tract, producing pulmonary stenosis in association with overriding and biventricular connection of the aortic valve ( Fig. 37.7B ). In the more common malalignment defects, as shown Fig. 37.7A , the pulmonary valve overrides the septum. With ever-greater degrees of overriding, and increasing connection of the leaflets of the pulmonary valve within the right ventricle, the spectrum of anomalies culminates in double outlet from the right ventricle with subpulmonary interventricular communication. This whole series is often called the Taussig-Bing complex. We divide the series at its midpoint, including only those with less than half the circumference of the overriding pulmonary valve connected within the right ventricle as examples of transposition. Equally significant from the surgical standpoint are those defects that extend to open to the inlet of the right ventricle. These are hidden beneath the septal leaflet of the tricuspid valve, complicating their surgical repair. When the defect extends to open into the right ventricular inlet, there is the potential for straddling and overriding of the tricuspid valve. In this setting, the muscular ventricular septum no longer extends to the crux, and the atrioventricular conduction axis takes origin from an anomalous posteroinferior atrioventricular node. Apart from in the presence of overriding of the tricuspid valve, the conduction tissue is carried on the left ventricular aspect of the septum, and the posterocaudal rim of the defect is most vulnerable during surgical correction. As with isolated defects, the conduction axis is better protected when the posterior limb of the septomarginal trabeculation fuses with the ventriculoinfundibular fold ( Fig. 37.7A ). However, if a muscular defect opens to the inlet, the axis of conduction tissue will be found in anterocephalad position. There can be other types of defects, such as multiple muscular defects, solitary apical muscular defects, or doubly committed defects roofed by the conjoined leaflets of the aortic and pulmonary valves. They are less common. The aorta is also rarely found posteriorly and to the right in the presence of a ventricular septal defect. With this arrangement, there is usually aortic-mitral valvar continuity through the roof of the defect. Bilateral infundibulums are also seen more frequently in association with a defective ventricular septum, most frequently when the arterial trunks are side by side. Other anomalies are also more frequent in association with ventricular septal defect, including juxtaposition of the atrial appendages, aortic stenosis, and aortic coarctation.
Any lesions that produce obstruction to the outflow tract of the morphologically left ventricle, and which in the normal heart produce aortic obstruction, will produce subpulmonary obstruction in the setting of transposition. Such lesions can be found at the valvar or subvalvar level ( Fig. 37.8 ).
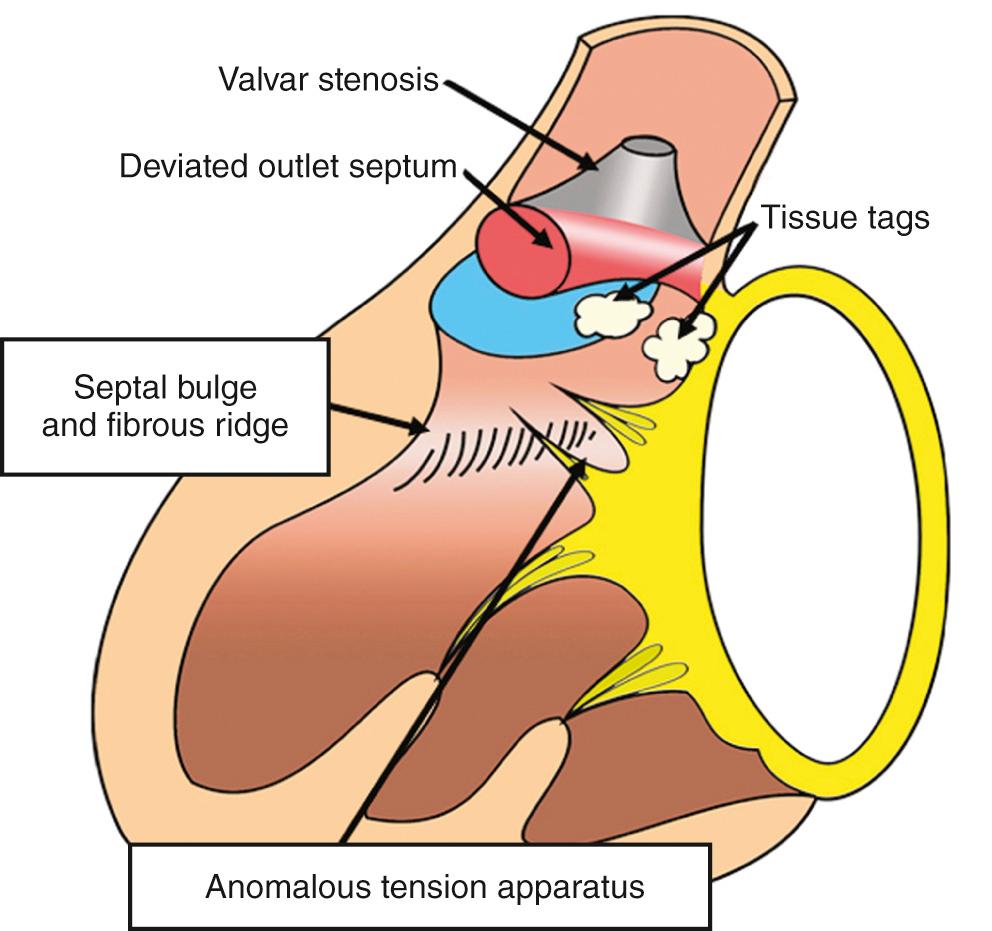
Isolated valvar obstruction is rare, being more common in combination with subvalvar obstruction, which may be dynamic, fixed, or both. Dynamic obstruction is produced by bulging of the ventricular septum. In its most severe form, this is reminiscent of hypertrophic obstructive cardiomyopathy. When septal bulging itself is less severe, obstruction of the left ventricular outflow tract is frequently exacerbated by a fibrous ridge located on the septal bulge. This can progress to form a complete subvalvar shelf, the ridge extending onto the facing surface of the mitral valvar leaflet. The extent of fibrous stenosis can also be more elongated, giving a tunnel lesion. Other rarer forms of stenosis are produced by anomalous attachment of the tension apparatus of the mitral valve across the outflow tract or by aneurysms of fibrous tissue tags bulging into the outflow tract. All can exist with an intact ventricular septum or in association with a ventricular septal defect. However, when there is a septal defect, there is another most significant type of stenosis, namely a malalignment and deviation of the muscular outlet septum into the left ventricle. This combination narrows the subpulmonary outflow tract in association with overriding of the aortic valve ( Fig. 37.7B ). Most of these fixed stenoses present major problems in surgical removal, either because of their own intrinsic morphology (e.g., a papillary muscle) or because of their proximity to vital structures, such as the left ventricular conduction tissues or the left coronary artery in the transverse sinus. If removal is attempted, the safest area for resection is the area occupied by the muscular outlet septum.
Other malformations can coexist either when the ventricular septum is intact or in association with a ventricular septal defect or obstruction of the left ventricular outflow tract. Persistent patency of the arterial duct is particularly significant because it loads the left ventricle, producing a thicker wall. Other significant anomalies are infrequent but can occur. They include stenosis of the subaortic outflow tract, aortic coarctation, or anomalous pulmonary venous connections.
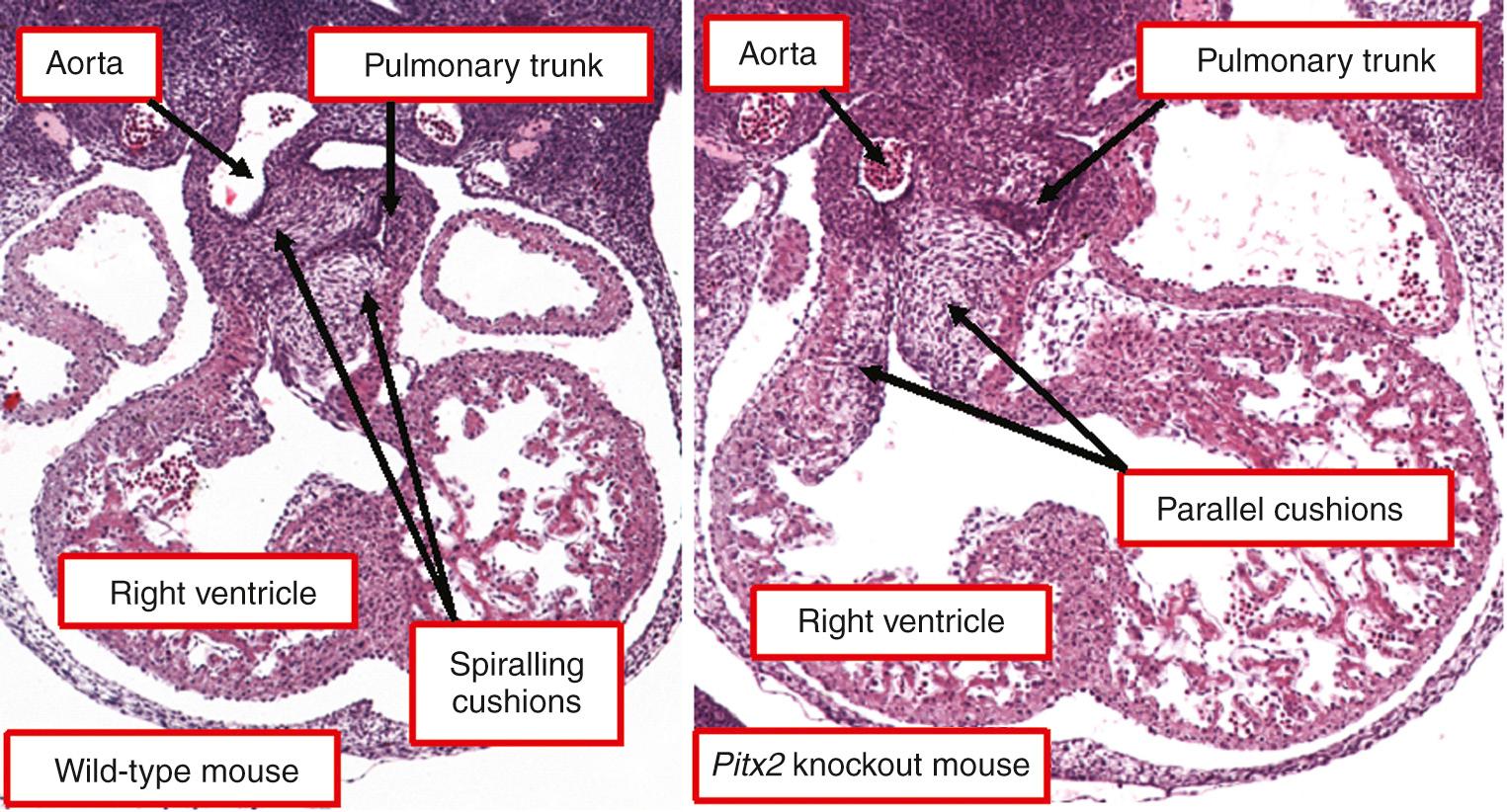
Two concepts have been advanced to explain the development of transposition. The first suggests that the anomaly is the consequence of inappropriate separation of the arterial pole of the heart. As a result, the sixth aortic arches, destined to supply the pulmonary arteries, are connected to the morphologically left ventricle, while the fourth aortic arches take their origin from the morphologically right ventricle. It was proposed that this can occur if the cushions septating the intermediate and proximal parts of the developing outflow tract fuse in straight rather than in their normal spiral fashion. The second theory puts the seat of maldevelopment in the proximal parts of the developing outflow tracts. This hypothesis suggests that, rather than the subaortic portion becoming connected to the left ventricle, as occurs in normal development, it is the subpulmonary component, together with the pulmonary trunk, which is thus connected. This concept was first proposed by Keith, who invoked differential absorption of the inner heart curvature as the causative mechanism. The concept was revitalized under the guise of differential conal growth. So as to explain all the known variants of transposition, including the arrangement in which the aorta arises posteriorly and to the right, it is necessary to invoke maldevelopment within both the proximal outflow tracts and the outflow cushions. Initial evidence from animal models showing discordant ventriculoarterial connections, such as the Pitx2 knockout mouse, supports this notion of the combination of fusion of the cushions in straight rather than spiraling fashion, with the subpulmonary component of the proximal outflow tract then transferred to the developing left ventricle ( Fig. 37.9 ).
The predominant physiologic abnormality in transposition is that the systemic venous return is recirculated to the body through the right ventricle and the aorta, while the pulmonary venous return is recirculated to the lungs through the left ventricle and pulmonary trunk. Thus, although in the normal heart the systemic and pulmonary circulations are in series, in transposition they are set up as two separate and parallel circuits. In the fetus, this arrangement results in more desaturation of the upper body than is usual but, although not associated with other significant circulatory instability, may be partially responsible for abnormal central nervous system findings later in life (see Chapter 76 ). Kept separated, the oxygenated pulmonary venous blood cannot reach the systemic circuit, and the systemic venous return cannot circulate to the lungs. This results in severe systemic arterial desaturation following birth and is incompatible with life. Although this is easy to understand, it is the lifesaving communications between these two circuits that add significant complexity to the physiology.
Systemic flow (Q S ) is all of the blood coursing through the systemic circulation. Effective systemic flow (Q ES ) is defined as the portion of Q S that contains saturated pulmonary venous blood. Likewise, pulmonary flow (Q P ) is the entirety of the blood in the pulmonary circulation, whereas effective pulmonary flow (Q EP ) is that desaturated portion, having completed its course through the systemic circulation, now directed toward the lungs. In the normal heart, where the cardiac connections are concordant and no shunts exist, Q P = Q S = Q EP = Q ES . In transposition, if no shunts existed, Q EP and Q ES would both equal 0 and the patient would die. Fortunately, two areas of communication between the circuits typically exist in the newborn. One is at the patent arterial duct. The other is through the atrial septum. In addition, a ventricular septal defect, present in approximately 40% of patients, may exist as well.
In the first few hours of life, when pulmonary vascular resistance is high, blood will shunt predominately from the pulmonary (saturated) circulation to the systemic (desaturated) circulation. This may result in the interesting finding of “reversed differential cyanosis,” whereby the blood in the lower extremities is more highly saturated than the in upper extremities. This phenomenon lasts for as long as there is pulmonary to aortic shunting at the ductal level. It may be prolonged in the setting of transposition with preductal coarctation (or interruption) of the aorta, whereby the distal aortic pressure is reduced by the obstruction, driving saturated blood from pulmonary artery to aorta. In the absence of such obstruction as the pulmonary vascular resistance drops, transductal shunting takes the opposite course, with desaturated aortic blood entering the pulmonary circulation, contributing to Q EP . In turn, pulmonary venous return to the left atrium is increased, and the resultant elevation in left atrial pressure serves as the impetus to drive the saturated left atrial blood across the second communication, the oval foramen or atrial septal defect, to join the systemic circulation, thereby contributing to Q ES . Once the arterial duct has closed, it is the bidirectional flow across the atrial septum that maintains Q EP and Q ES . Interventions to augment flow at the ductal and atrial levels allow for the successful stabilization of the newborn with transposition.
The presence of a ventricular septal defect can have a variable influence on the circulation. This in part depends on the size of the defect, as well as on the presence of interatrial mixing, and the pulmonary vascular resistance. A large interventricular communication may contribute to beneficial circulatory mixing, but the presence of unrestricted flow to the lungs may result in symptomatic heart failure. If surgery is not performed during infancy, this can predispose to early pulmonary vascular disease, which is generally evident by 6 to 12 months of age. The presence of additional pulmonary or subpulmonary obstruction further modifies these relationships and may limit Q P . Patients who are well balanced due to pulmonary obstruction are generally asymptomatic, having a protected pulmonary circulation, adequate mixing, and a “well-trained” left ventricle.
The essence of transposition is such that the left ventricle supports the pulmonary circulation. In the neonatal period, when the pulmonary vascular resistance is high, left ventricular work and impedance are elevated. In a patient with an intact or virtually intact ventricular septum, the normal postnatal reduction in pulmonary vascular resistance results in a progressive reduction in left ventricular pressure and work, so that as a result, left ventricular myocardial mass progressively falls. This reduction in left ventricular pressure and myocardial mass will be attenuated in the presence of a large ventricular septal defect.
The effects of this postnatal transition are of crucial importance to the operative approach. In a patient with transposition with an intact ventricular septum, who has traversed this postnatal transition, any procedure (including the arterial switch operation), in which the left ventricle is called on to support the high resistance systemic circulation will result in left ventricular failure in the early postoperative period. Thus, as will be discussed, the arterial switch operation is performed in early postnatal life, before the postnatal transition is complete. Older patients will require either a preliminary procedure to increase the load on the left ventricle, such as banding of the pulmonary artery to “train” the left ventricle in preparation for definitive surgery, or alternatively may require a period of left ventricular support with ventricular assistance, in the early postoperative period. Finally, an atrial switch procedure may be desirable for some of these patients.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here