Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Drug reactions are some of the most frequently encountered adverse complications in the hospital setting. As many as 30% of hospitalized patients may experience such a reaction, and an estimated one to two million Americans have a drug reaction each year. The presentation of adverse drug reactions is highly variable; one can occur with supratherapeutic/overdose levels, therapeutic doses, or even with minimal intake. Similarly, most present with minor complications such as skin rashes, but serious fatal reactions such as anaphylaxis also occur, though they are fortunately less common . When a drug affects the cardiovascular system, the complication cannot be considered minor, as the consequence is likely to affect cardiac contractile function or rhythm. Unfortunately, as new dietary additives, therapeutic medications, and illicit drugs are developed, the cardiac complications are not always anticipated prior to release and use .
For the purposes of this chapter, toxic agents refer to medicine, recreational drugs, and toxins present in food, drink, nutritional supplements, or the environment. Identifying a single causative agent can be difficult; some drugs may only cause problems when used in combination with other agents, some symptoms may only manifest in individuals with underlying susceptibility or cardiovascular disease, and many patients with adverse drug reactions are on a number of medications. Furthermore, recreational use of prescribed and illicit drugs is rampant, adding significantly to the number of potentially harmful agents in use by millions of individuals. These agents are often taken together, a so-called cocktail, and/or with alcohol, cigarettes, or caffeine, amplifying the potential for serious harm.
In general, agents that are “toxic” to the heart induce injury through one (or more) of the following mechanisms: direct toxicity, idiosyncratic reaction related to abnormal metabolism, hypersensitivity, overdose, and/or drug interactions. The pathologic changes responsible for dysfunction are protean; the major patterns of injury are listed in Table 12.1 . Certain agents may also cause more than one pattern of injury. For example, cocaine, perhaps the most notorious agent that injures the heart, can result in cardiomyopathy, myocarditis, myocardial necrosis (myocytolysis), coronary artery spasm, coronary arteritis, coronary thrombosis, coronary dissection, and/or accelerated atherosclerosis. Functionally, the heart has a very limited repertoire of responses to disease: (1) the heart can fail as a pump or (2) the electrical system can fail resulting in a variety of conduction problems and arrhythmias.
| Endocardium |
| Valvular fibrosis with dysfunction |
| Endocardial fibrosis |
| Myocardium |
| Myocarditis |
| Acute myocardial cell death |
| Dilated cardiomyopathy |
| Systolic and diastolic dysfunction |
| Cardiac hypertrophy |
| Pericardium |
| Pericarditis |
| Coronary arteries |
| Spasm |
| Arteritis |
| Dissection |
| Thrombosis |
| Accelerated atherosclerosis |
| Primary conduction system abnormalities |
| Atrial arrhythmias |
| Conduction block |
| Ventricular arrhythmias |
| Sudden death |
This chapter will first outline patterns of injury seen in the heart based on anatomic location and their associated offending agents, and then focus on select drugs or drug classes known to cause cardiovascular toxicity.
Certain toxic and environmental exposures have shown predilection for damage to the endocardial surfaces, including valves and mural endocardium. Implicated toxins are listed in Table 12.2 .
| Methysergide |
| Pergolide |
| Cabergoline |
| Fenfluramine-Phentermine |
| Busulfan |
| Cassava |
| Cerium |
“Chemically induced” valvular injury has been recognized for many years as being associated with the carcinoid syndrome in which naturally occurring serotonin and serotonin-like products are produced by neuroendocrine neoplasms. If these agents avoid inactivation by the liver, often through hepatic metastases or venous drainage that bypasses portal circulation, they cause characteristic abnormalities primarily on the right-sided heart valves. The typical pathologic findings are gross thickening and sometimes contraction of the valve leaflets by dense white connective tissue on the leaflets, chordae tendineae, and sometimes even the papillary muscles. Microscopically, the tissue is made up almost exclusively of dense collagen with little, if any, elastic tissue. A small number of inflammatory cells, including eosinophils, may be present. What distinguishes this type of valve disease from others, such as infective endocarditis or rheumatic diseases, is that the underlying valve tissue is relatively normal, and the disease is most prominent on the right-sided valves. The left-sided valves may be involved if the serotonin and metabolites are not “filtered” by the lung and gain access to the left side of the heart through some shunt, such as a pulmonary arteriovenous malformation or an intracardiac defect such as atrial septal or ventricular septal defect. If the neuroendocrine neoplasm metastasizes to the lungs or heart, left-sided lesions may occur.
A number of ingested agents have been associated with similar gross and microscopic lesions on cardiac valves. Interestingly, the right-sided predominance does not occur. One of the first therapeutic agents noted to cause such lesions is the ergot derivative, methysergide, an effective drug treatment for migraine headaches ( Fig. 12.1 ). Methysergide is no longer a favored treatment as its use may cause retroperitoneal fibrosis as well as cardiac valvular lesions and intimal proliferation in blood vessels. Similar valve disease has also been described with pergolide and cabergoline, two ergot-derived dopamine agonists used to treat Parkinson’s disease. The histopathology of the valve lesions is remarkably similar to that observed with the carcinoid syndrome. There is no explanation for why the left-sided rather than the right-sided valves are usually affected.
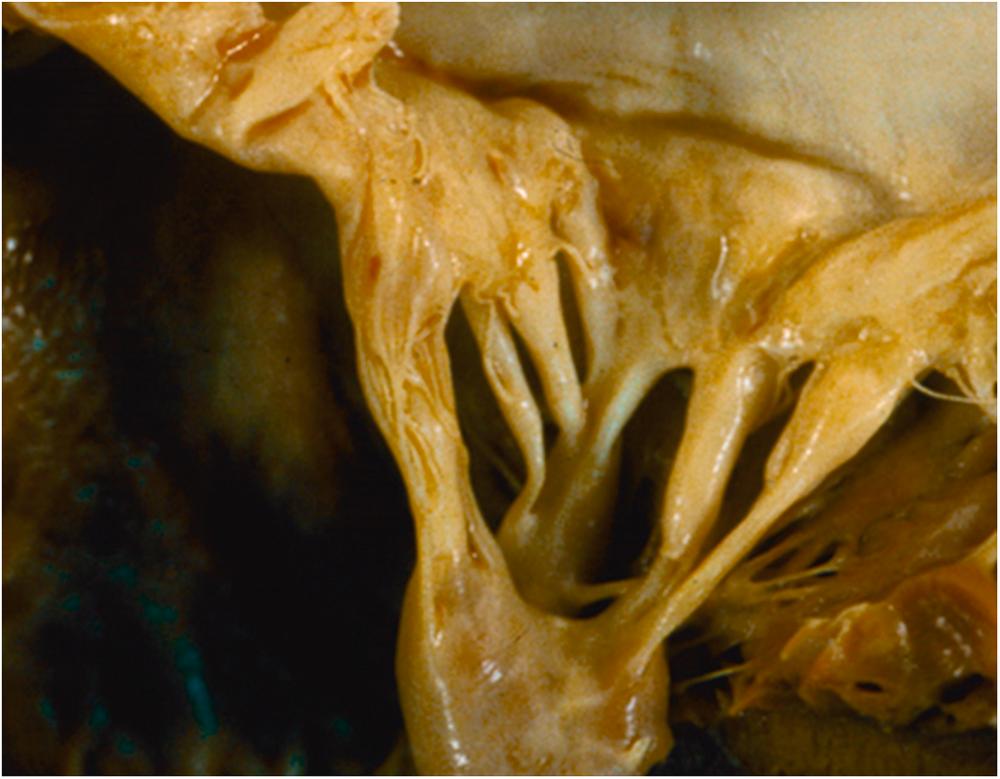
Instances of severe mitral valve disease requiring valve replacement in patients using fenfluramine and phentermine (fen-phen) for weight reduction have also been reported ( Fig. 12.2 ) . It has been estimated that more than 18 million prescriptions were written for fen-phen and that more than 30,000 cases of valve disease were caused by the use of these medications. However, the magnitude of the risk of valve disease associated with fen-phen has been controversial. In a large meta -analysis, fen-phen use was associated with a 12% prevalence of valve regurgitation compared to a prevalence of 5.9% in control patients. There was a statistically significant increase in both aortic and mitral regurgitation. Valve dysfunction only occurred in those individuals using the medications for more than 90 days. The recognition of the association with valve disease and pulmonary hypertension has resulted in abandonment of the combined use of these medications. Fenfluramine, which affects serotonin release and metabolism, seemed to be the culprit agent and has since been withdrawn from the market.
Tropical endomyocardial fibrosis is a fibrotic process that can lead to restrictive physiology. It is seen in higher frequency in Africa, Asia and South America. The exact pathogenesis remains inconclusive, but may be related to eosinophilia and/or infectious etiologies. Given the regional differences observed, some have postulated that environmental factors, including malnutrition, magnesium deficiency, cerium toxicity, herbal medicines, and cassava consumption may also play a role . Cassava contains linamarin, a toxic cyanogenic glycoside, which in the absence of proper cooking may persist in the consumed product. In conjunction with a poor protein diet, the enzymes responsible for detoxifying cyanide may have decreased detoxification abilities. Some animal models have also shown endocardial thickening and interstitial fibrosis with cassava-based diets .
Endocardial fibrosis has also been reported in a patient with prolonged busulfan treatment, an alkylating agent historically used to treat chronic myelogenous leukemia .
Myocardial dysfunction manifests with clinical features of heart failure. Many agents have been implicated in myocardial dysfunction, which may or may not be accompanied by anatomic or histologically demonstrable lesions. Patterns of injury that are sometimes seen include myocarditis, acute myocyte death without associated inflammation, gross dilation, and hypertrophy. Implicated toxins are listed in Tables 12.3 and 12.4 , though any drug should be considered in the etiology of myocarditis.
| Acetazolamide | Cyclosporine | Oxyphenbutazone |
| Amitriptyline | Digoxin | para -Aminosalicylic acid |
| Antidepressants | Dopamine | Penicillin |
| Aminophylline | Dobutamine | Phenindione |
| Amphotericin B | Furosemide | Phenylbutazone |
| Ampicillin | Heparin | Phenytoin |
| Angiotensin-converting enzyme inhibitors | Hydralazine | Spironolactone |
| Azothiaprine | Hydrochlorothiazide | Streptomycin |
| Benzodiazepines | Indomethacin | Sulfadiazine |
| Bumatemide | Interleukin-2 | Sulfamethoxypyridine |
| Captopril | Isoniazid | Sulfisoxazole |
| Carbamazepine | Isosorbide dinitrate | Sulfonylureas |
| Cephalosporins | l -Tryptophan | Tetracyclines |
| Chloramphenical | Methyldopa | Tricyclines |
| Chlorthalidone | Metalazone | Triazolam |
| Clozapine | Nitroprusside |
| Antineoplastic agents |
| Antibodies (trastuzumab) |
| Antibiotics |
| Anthracyclines |
| Alkylating agents |
| Cyclophosphamide, cisplatin |
| Antimetabolites (5-fluorouracil) |
| Alcohol |
| Cocaine |
| Methamphetamines |
| Anabolic steroids |
| Anesthetic agents |
| Antimalarial medications |
| Chloroquine |
| Hydroxychloroquine |
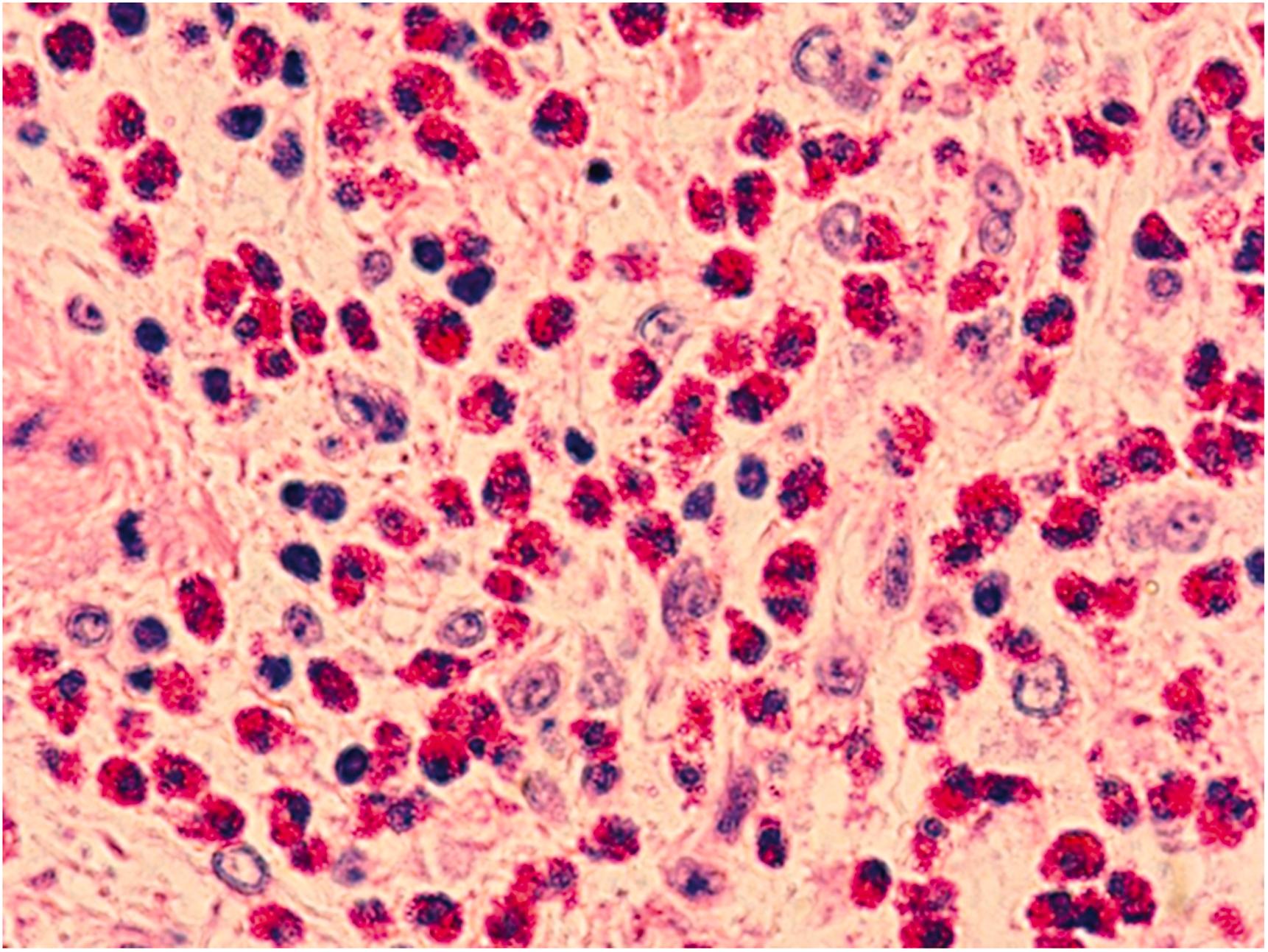
Some agents may cause myocarditis in which inflammation is seen in the muscular portion of the heart ( Fig. 12.3 ). One pattern of injury is hypersensitivity myocarditis, wherein exogenous substances cause myocarditis by invoking an immune or allergic response, rather than a direct toxic effect to cardiac myocytes. Oftentimes, the reaction is not due directly to the drug, but rather a metabolite that initiates an immune response. Hypersensitivity myocarditis is typically not dose-related and can involve the heart with or without other organ involvement. Hypersensitivity myocarditis often occurs with rash, fever, peripheral eosinophilia, and electrocardiogram (EKG) abnormalities. Though usually seen following initiation of a new medication, delayed reactions have been reported. Hypersensitivity myocarditis may also become manifest after prior uneventful use of a drug. The affected patient may be asymptomatic or suffer life-threatening congestive heart failure or arrhythmias .
![Figure 12.3, Myocarditis in a patient treated with interleukin-2. Note intense inflammation composed of lymphoid cells and macrophages. There is myocyte dropout and injury (arrow) as well [H&E stain: (A) 100×, (B) 400×]. Figure 12.3, Myocarditis in a patient treated with interleukin-2. Note intense inflammation composed of lymphoid cells and macrophages. There is myocyte dropout and injury (arrow) as well [H&E stain: (A) 100×, (B) 400×].](https://storage.googleapis.com/dl.dentistrykey.com/clinical/Toxinsandtheheart/2_3s20B9780128222249000128.jpg)
Hypersensitivity “myocarditis” is somewhat of a misnomer, as the lesion is usually a “pancarditis” with involvement of epicardial fat, myocardium, and endocardium. Inflammation of the arteries and veins may be present as well. Histological examination may show incidental focal interstitial myocardial infiltrates or diffuse severe inflammatory infiltrates associated with myocyte injury. There is usually a mixed inflammatory infiltrate with eosinophils present. Multinucleated giant cells and even an occasional granuloma may be observed. There is no minimum criterion for the number of eosinophils that must be present. In fact, they may be rather sparse or the eosinophil may dominate the cardiac infiltrate.
Currently, the most common specimen encountered with hypersensitivity myocarditis is the explanted heart of the patient undergoing transplantation ( Fig. 12.4 ). In one reported series, hypersensitivity myocarditis was observed in the cardiac tissue of 7.4% of those undergoing heart transplantation. Peripheral eosinophilia was present in 86% of patients . These potential transplant recipients typically are taking over 30 medications, including inotropic agents and antibiotics that are among the most common that have been linked to hypersensitivity myocarditis.
While drugs should always be in the differential diagnosis for eosinophilic myocarditis, a number of other conditions are associated with eosinophilic myocarditis. This pattern can be seen with all hypereosinophilic syndromes, including eosinophilic leukemia, Loeffler’s syndrome, parasitic infections, and even a lung cancer that produced an eosinophilic chemoattractant factor. Eosinophilic myocarditis may also be seen in a variety of collagen-vascular disease such as eosinophilic granulomatosis with polyangiitis/Churg–Strauss syndrome, and polyarteritis nodosa.
An entity that shares similarities with hypersensitivity myocarditis is the eosinophilia myalgia syndrome. An impurity in the nutritional supplement l -tryptophan was identified to cause a peripheral eosinophilia, myalgias, arthralgias, and skin lesions. The affected patients had inflammatory infiltrates in affected tissues, including the heart. Endomyocardial biopsy in several cases showed an eosinophilic myocarditis with or without vasculitis .
The differential diagnoses for hypersensitivity myocarditis also include viral myocarditis, in which eosinophils are usually few or absent, and rheumatologic disorders that more often also demonstrate vasculitis. Skin testing can sometimes identify the causative agent. Removing the offending drug can reverse the peripheral eosinophilia, if present, and eliminate the myocardial infiltrate if sequential biopsies are available for comparison.
Myopericarditis has also been reported in patients following smallpox vaccination . Vaccination against infectious diseases is one of the great accomplishments in modern medicine, preventing serious illness and saving countless lives. While vaccination is generally safe, some complications do occur. Though it is a rare occurrence, myopericarditis is potentially life-threatening and requires medical attention.
Some agents may cause myocardial necrosis without an inflammatory component. Typically, the acute cardiac lesion is not the usual coagulation necrosis of a region of myocardium, as seen with ischemic injury, but rather scattered degenerative changes of individual or small groups of cells. In fact, experimental studies indicate that the cell injury is not related to decreased flow, but rather a direct biochemical or metabolic mechanism of myocyte injury . These lesions have been referred to as “myocytolysis.” These injured myocytes may show contraction bands (coagulative myocytolysis) or clearing of the sarcoplasm (colliquative myocytolysis) ( Fig. 12.5 ). Inflammation is minimal and healing is characterized by a dropout of affected fibers with gradual replacement by granulation tissue and then fibrosis.
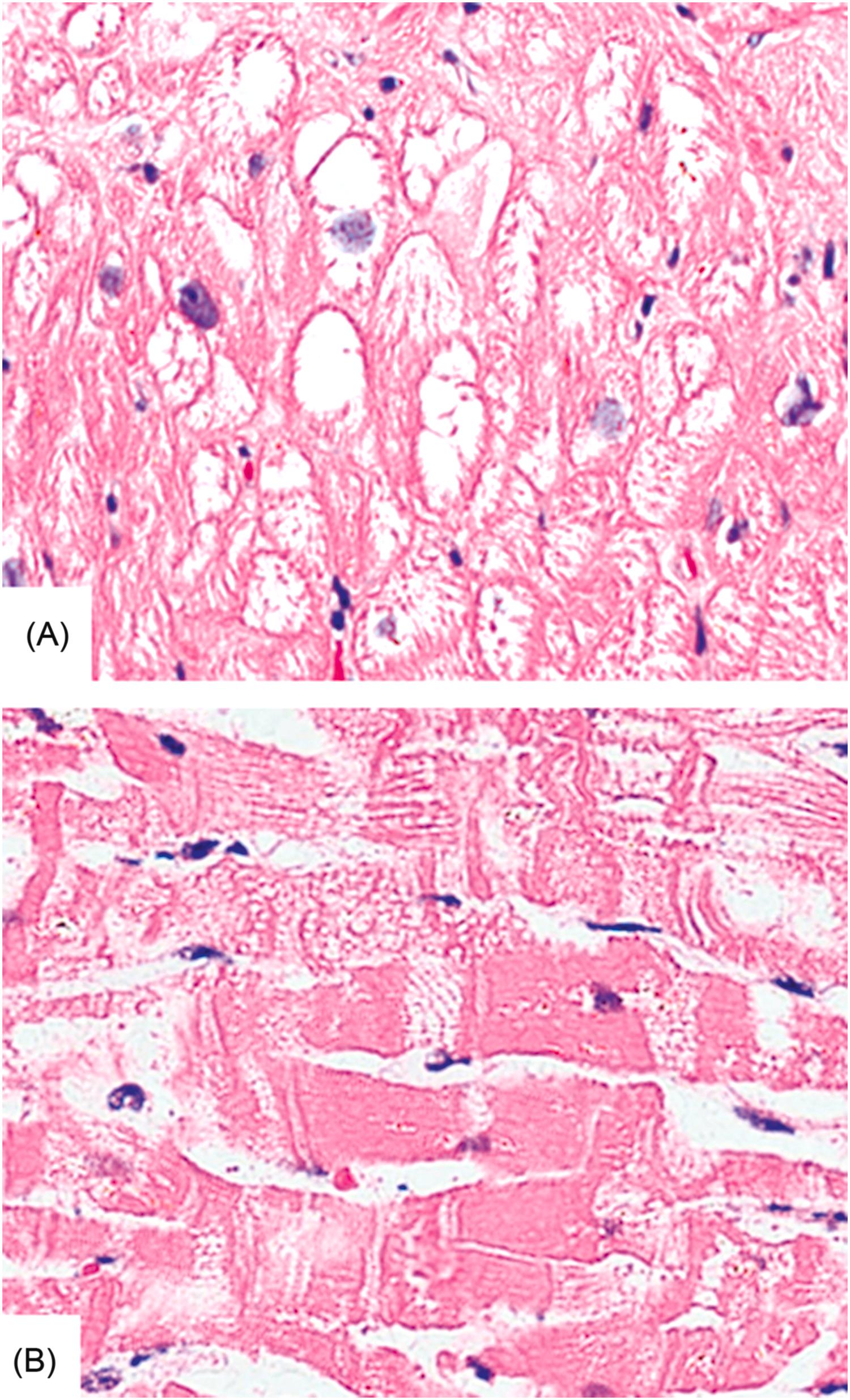
The agents that most often cause acute cell death are in the category of sympathomimetic agents. In fact, patients with pheochromocytoma, a neoplasm that secretes the naturally occurring agents epinephrine and norepinephrine, may demonstrate myocardial necrosis . There are several mechanisms through which cocaine and related agents may produce these lesions. Cocaine stimulates the release of the potent vasoconstrictor, endothelin-1, and inhibits production of the potent vasodilator, nitric oxide, from endothelial cells. Cocaine also promotes thrombosis by activating platelets and increasing coagulability of blood .
Other agents, including catecholamines, interleukin-2, cyclophosphamide, emetine, lithium, phenothiazines, antimony, and arsenicals have also been associated with acute myocyte death .
Dilated cardiomyopathy may be “primary” or idiopathic, or “secondary” when an etiologic agent is identified. The etiologic agent may be a substance toxic to the myocardium. As its name indicates, in dilated cardiomyopathy, one or more cardiac chambers are dilated. Usually, the left ventricular dilatation is most notable. The heart weight is usually increased, but the ventricular wall thickness may be normal ( Fig. 12.6A ). This apparent paradox is due to the fact that the increase in mass is due to sarcomeres laid down end-to-end rather than side-to-side. Mural thrombi may be present in the atria and/or ventricles, related to relative stasis of blood in the hypokinetic chambers. Microscopically, there may be some perivascular or interstitial myocardial fibrosis, but fibrosis may be completely absent. If replacement fibrosis is present, it is usually a secondary phenomenon due to coronary thromboembolism or hypoperfusion of the myocardium due to the contractile dysfunction that characterizes this entity clinically. Inflammation is usually absent or only focal and mild, typically limited to regions of fibrosis. In dilated cardiomyopathy, the major clinical manifestations are those of congestive heart failure (i.e., pump dysfunction). In addition, patients with dilated cardiomyopathy are at an increased risk of sudden death. Sudden death may occur in isolation, that is, with no preceding clinical manifestations of pump dysfunction.
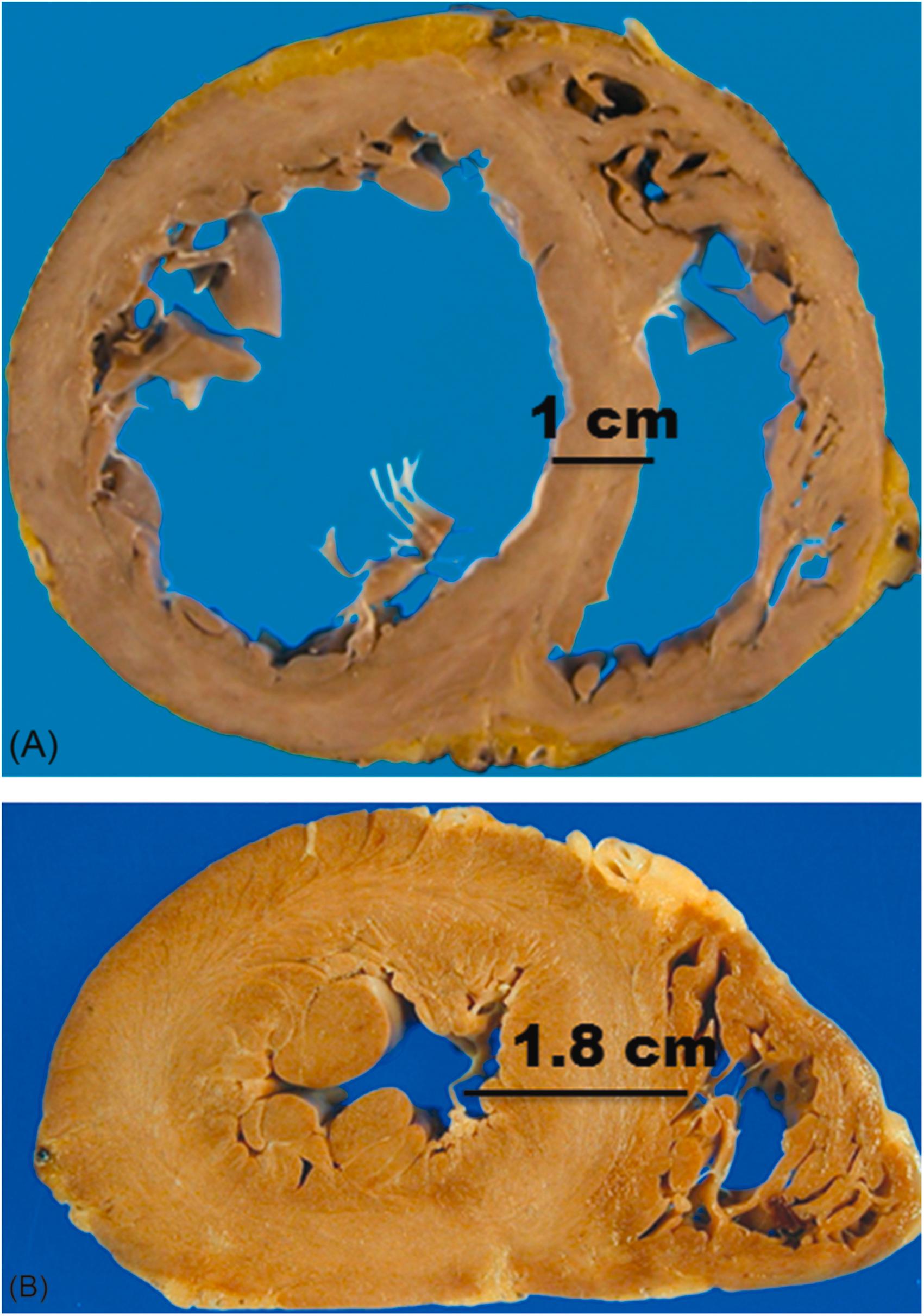
A large number of toxic agents have been implicated in causing dilated cardiomyopathy (see Table 12.4 ) . The most common and well-known toxic agent that causes dilated cardiomyopathy is alcohol, and is discussed in greater detail in subsequent sections. Ultrastructural and biochemical changes associated with alcoholic cardiomyopathy have been described, but are not specific; ultimately, there is nothing that distinguishes the pathology of alcoholic cardiomyopathy from other forms of dilated cardiomyopathy
Anthracycline chemotherapy has also been associated with dilated cardiomyopathy, with approximately 1% of all dilated cardiomyopathy being attributable to doxorubicin . Numerous nonanthracycline chemotherapeutic agents have also been implicated in heart failure, and are discussed in more detail further on.
Some medications depress cardiac function without causing frank cardiomyopathy. Anesthetic agents have been known for many years to depress cardiac function in normal and abnormal hearts. Isoflurane and propofol directly affect myocardial myofilaments by lowering their response to calcium, thereby decreasing contractility . The anesthetic agents halothane, enflurane, and nitrous oxide have also been associated with depression of cardiac function . Some anesthetics, however, may actually make the heart more sensitive to inotropic agents, and newer agents have less effect on cardiac contractility; operative complications due directly to the depressant effects of anesthetic agents are currently rare .
Cardiac hypertrophy can also cause heart failure due to left ventricle stiffness ( Fig. 12.6B ). This has been documented in people taking anabolic-androgenic steroids as well as those with chronic cocaine use .
Speculation exists that exogenous steroids may have first been used by German troops in World War II to increase strength and aggressiveness, though little reliable evidence exists to support this claim . The overall worldwide prevalence of anabolic-androgenic steroid use is estimated to be around 4%, but varies significantly depending on specific demographics examined . Cardiovascular complications represent only a portion of the detrimental effects of anabolic-androgenic steroids; adverse effects are seen in the reproductive organs, liver, blood, endocrine, renal, immunologic, and musculoskeletal systems . Anabolic-androgenic steroids cause hypertension that likely contributes to the most commonly associated cardiac morphologic abnormality, namely, hypertrophy. Systolic and diastolic dysfunction may persist for years after misuse of anabolic-androgenic steroids . Parenthetically, steroids also adversely affect serum lipids and increase the coagulability of blood. Accordingly, myocardial infarction has been reported with use of anabolic-androgenic steroids. Coronary vasospasm, thrombosis, and atherosclerosis have all been implicated as the cause of the ischemic event. Sudden cardiac death has also been reported, though it is not clear whether this is related to an ischemic event and/or cardiac hypertrophy.
Some of cocaine’s myriad effects on the heart, particularly with prolonged use, are cardiac hypertrophy, increased ventricular wall thickness, and decreased left ventricular end-diastolic volume. This suggests that chronic cocaine use might also result in diastolic heart failure .
Left ventricular hypertrophy can also be seen in highly trained athletes, the so-called “athlete’s heart,” even in the absence of exogenous steroid use . The cardiac hypertrophy seen in association with these agents may be a response to physiologic changes, rather than a direct effect of the offending agent itself.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here