Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Synthesis and secretion of thyroid hormone is under the control of the anterior pituitary hormone thyrotropin (or thyroid-stimulating hormone [TSH]). Consistent with a classic negative feedback system, TSH secretion increases when serum thyroid hormone levels fall and decreases when they rise ( Fig. 137.1 ). TSH secretion is also under the regulation of the hypothalamic hormone thyrotropin-releasing hormone (TRH). The negative feedback of thyroid hormone is targeted mainly at the pituitary level, but also likely affects TRH release from the hypothalamus. In addition, input from higher cortical centers can affect hypothalamic TRH secretion.
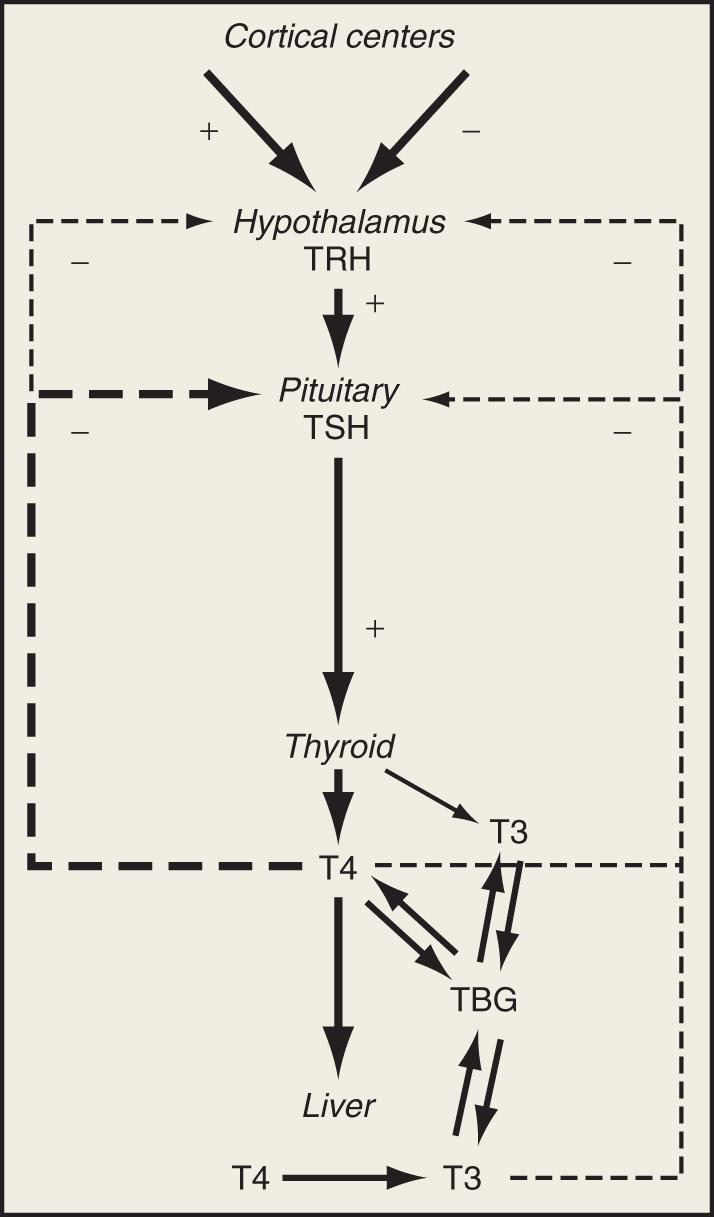
Under the influence of TSH, the thyroid gland synthesizes and releases thyroid hormone. Thyroxine (3,5,3′,5′-tetraiodothyronine, or T 4 , which is 65% iodine by weight) is the principal secretory product of the thyroid gland, comprising about 90% of secreted thyroid hormone under normal conditions. Although T 4 may have direct actions in some tissues, it primarily functions as a hormone precursor that is metabolized in peripheral tissues to the transcriptionally active 3,5,3′-triiodothyronine (T 3 , which is 59% iodine by weight).
The major pathway of metabolism of T 4 is by sequential monodeiodination. At least three deiodinases, each with its unique expression in different organs, catalyze deiodination reactions involved in the metabolism of T 4 . Removal of the 5′-, or outer ring, iodine by type I iodothyronine 5′-deiodinase (D1) or type II iodothyronine 5′-deiodinase (D2) is the “activating” metabolic pathway leading to the formation of T 3 . Removal of the 5′-, or inner ring, iodine by type III iodothyronine deiodinase (D3) is the “inactivating” pathway that produces the metabolically inactive thyroid hormone 3,3′,5′-triiodothyronine (reverse T 3 or rT 3 ). D1 is found most abundantly in the liver, kidneys, and thyroid. It is up-regulated in hyperthyroidism and down-regulated in hypothyroidism. D2 is found primarily in the brain, pituitary, and skeletal muscle and is down-regulated in hyperthyroidism and up-regulated in hypothyroidism. D3 is expressed primarily in the brain, skin, and placental and chorionic membranes. The actions of D3 also include inactivation of T 3 to form T 2 , another inactive metabolite. Under normal conditions, about 41% of T 4 is converted to T 3 , about 38% is converted to rT 3 , and about 21% is metabolized via other pathways, such as conjugation in the liver and excretion in the bile. ,
T 3 is the metabolically active thyroid hormone and exerts its actions via binding to chromatin-bound nuclear receptors and regulating gene transcription in responsive tissues. It is important to note that only around 10% of circulating T 3 is secreted directly by the thyroid gland, whereas more than 80% of T 3 is derived from the conversion of T 4 in peripheral tissues. , Thus factors that affect peripheral T 4 -to-T 3 conversion will have significant effects on circulating T 3 levels. Serum levels of T 3 are approximately 100-fold less than those of T 4 , and like T 4 , T 3 is metabolized by deiodination to form diiodothyronine (T 2 ) and by conjugation in the liver. The half-lives of circulating T 4 and T 3 are 5–8 days and 1.3–3 days, respectively.
Both T 4 and T 3 circulate in the serum as hormones bound to several proteins synthesized by the liver. Thyroid-binding globulin (TBG) is the predominant transport protein and binds to approximately 80% of the circulating serum thyroid hormones. The affinity of T 4 for TBG is about 10-fold greater than that of T 3 , which accounts in part for the higher circulating T 4 levels as compared with T 3 levels. Other serum binding proteins include transthyretin, which binds about 15% of T 4 but little, if any, T 3 ; and albumin, which has a low affinity but a very large binding capacity for T 4 and T 3 . Overall, 99.97% of circulating T 4 and 99.7% of circulating T 3 are bound to plasma proteins.
Essential to an understanding of the regulation of thyroid function and the alterations of circulating thyroid hormones seen in critical illness is the “free hormone” concept, which is that only the unbound hormone has metabolic activity. Although overall thyroid hormone production is regulated by the pituitary, serum thyroid function is affected by changes in the free thyroid hormone concentrations. Alterations in either the concentrations of binding proteins or the binding affinity of thyroid hormones to their serum binding proteins have significant effects on total serum hormone levels, owing to the high degree of binding of T 4 and T 3 to these proteins. However, despite these changes, binding effects do not necessarily translate into clinically significant thyroid dysfunction.
Widespread changes in thyroid hormone economy in critically ill patients , occurs as a result of (1) alterations in peripheral metabolism of thyroid hormones, (2) alterations in TSH regulation, and (3) alterations in the binding of thyroid hormone to TBG.
There is considerable variation in the transcriptional and translational activities of genes important for thyroid hormone metabolism during acute illness. One of the initial alterations in thyroid hormone metabolism in acute illness is the acute inhibition of D1, which results in the impairment of T 4 -to-T 3 conversion in peripheral tissues. D1 is inhibited by a wide variety of factors, including acute illness ( Box 137.1 ), resulting in the acute decrease in T 3 production in critically ill patients. In contrast, inner ring deiodination by D3 may be increased by acute illness, resulting in increased levels of rT 3 . Additionally, because rT 3 is subsequently deiodinated by D1, degradation of rT 3 decreases, and levels of this inactive hormone rise in proportion to the fall in T 3 levels. There may also be potential roles of other T 4 metabolites like 3,5-diiodothyronine (3,5-T2) and 3-iodothyronamine (3-T1AM) in critical illness, but this remains unclear.
Acute and chronic illness
Caloric deprivation
Malnutrition
Glucocorticoids
Beta-adrenergic blocking drugs (e.g., propranolol)
Oral cholecystographic agents (e.g., iopanoic acid, sodium ipodate)
Amiodarone
Propylthiouracil
Fatty acids
Fetal/neonatal period
Selenium deficiency
Hepatic disease
Nondeiodinative pathways may play an important role in critical illness, as sulfoconjugation and alanine side chain deamination/decarboxylation are increased, resulting in increased levels of T 3 -sulfate and Triac, respectively, although thyroid hormone hypermetabolism may not entirely be the result of these processes in critical illness. Triac, which binds to the thyroid hormone receptor and has weak thyromimetic activity, increases locally during illness and fasting. An increase in Triac production in the pituitary may be a factor in decreasing TSH levels during illness.
Finally, in critical illness, there is impaired transport of T 4 to peripheral tissues, such as the liver and kidney, where much of the circulating T 3 is produced. This further contributes to the decrease in the production of T 3 . , Interestingly, increased expression of the thyroid hormone transporters OATP1C1 and MCT8 in animal models and increased expression of MCT8 and MCT1 in the liver and muscle in human studies involving critical illness have been observed. The mechanisms underlying the decrease in tissue transport during critical illness has yet to be more fully elucidated.
It has long been recognized that serum TSH levels are usually normal in the early phases of acute illness. , Decreased TRH secretion caused by inhibitory signals from higher cortical centers, impaired TRH metabolism, alteration of pulsatile TSH, and the decrease or absence of a nocturnal TSH surge , may all further lower TSH levels. Serum levels of leptin, the ob gene product that has been shown to vary directly with thyroid hormone levels, also falls as illness progresses and hypothalamic TRH secretion falls, which in turn lead to lowered TSH levels. The decrease of hypothalamic TRH gene expression in animal models, however, is not associated with increased serum T 4 and T 3 levels. Finally, certain thyroid hormone metabolites that are increased during acute nonthyroidal illness may play a role in the inhibition of TSH and TRH secretion.
Common medications used in the treatment of critically ill patients may also have inhibitory effects on serum TSH levels ( Box 137.2 ). Van den Berghe and colleagues reported that intravenous (IV) administration of dopamine for 15–21 hours can acutely decrease TSH, whereas its withdrawal results in a 10-fold increase in serum TSH levels. Other medications that can inhibit TSH secretion include glucocorticoids, rexinoids, and somatostatin.
Acute and chronic illness
Adrenergic agonists
Caloric restriction
Carbamazepine
Clofibrate
Cyproheptadine
Dopamine and dopamine agonists
Endogenous depression
Glucocorticoids
Insulin-like growth factor (IGF)-1
Metergoline
Methysergide
Opiates
Phenytoin
Phentolamine
Pimozide
Rexinoids
Somatostatin and its analogs
Serotonin
Surgical stress
Thyroid hormone metabolites
The affinity of thyroid hormones binding to transport proteins and the concentration of serum binding proteins are altered with acute illness ( Table 137.1 ). Serum levels of transthyretin and albumin decrease, especially during prolonged illness, malnutrition, and in high catabolic states. TBG levels may be increased, as seen with liver dysfunction, or decreased, as seen with severe or prolonged illness.
| Increase Binding | Decrease Binding | |
|---|---|---|
| Drugs | Estrogens | Glucocorticoids |
| Methadone | Androgens | |
| Clofibrate | l -Asparaginase | |
| 5-Fluorouracil | Salicylates | |
| Heroin | Mefenamic acid | |
| Tamoxifen | Phenytoin | |
| Raloxifene | Furosemide | |
| Heparin | ||
| Anabolic steroids | ||
| Systemic Factors | Liver disease | Inherited |
| Porphyria | Acute illness | |
| HIV infection | NEFAs | |
| Inherited |
An acquired binding defect of T 4 to TBG is commonly seen in patients with critical illness. This is thought to result from the release of some as yet unidentified factor from injured tissues that has the characteristics of unsaturated nonesterified fatty acids (NEFAs), which also inhibit T 4 -to-T 3 conversion. In systemically ill patients, NEFA levels rise in parallel with the severity of the illness, and drugs such as heparin stimulate the generation of NEFA. Many drugs, including high-dose furosemide, antiseizure medications, and salicylates, also alter the binding of T 4 to TBG. The alteration in serum binding proteins in critical illness makes estimating free hormone concentrations challenging.
Abnormal thyroid function tests caused by nonthyroidal illness have been reported in up to approximately 70% of acutely ill patients. , In a study of 1580 hospitalized patients, only 24% of patients with suppressed TSH values (i.e., TSH levels below the limit of detection) and 50% of patients with TSH values over 20 mU/L were found to have thyroid disease. , More importantly, none of the patients with subnormal but detectable TSH values and only 14% of patients with elevated TSH values less than 20 mU/L were subsequently diagnosed with intrinsic thyroid dysfunction. A review by Kaptein and colleagues suggested that patients with isolated increased TSH concentrations in the setting of chronic cardiac, hepatic, or renal disease usually do not persist or progress to overt hypothyroidism.
The development of sensitive third-generation TSH assays has led to small improvements in discerning between overt hyperthyroidism and nonthyroidal illness. Overall, however, although a normal TSH level has a high predictive value of normal thyroid function, an abnormal TSH value alone is not helpful in evaluating thyroid function in critically ill patients.
Measurement of free thyroid hormone concentrations in patients with nonthyroidal illness is similarly fraught with difficulty. The gold standard for determination of free hormone levels is equilibrium dialysis. However, this technique is labor intensive, time consuming, and rarely used. The most commonly available laboratory tests for thyroid hormone concentrations are free T 4 index, free T 4 , and free T 3 that are measured by analog methods; however, these methods are estimates of free hormone concentrations and thus subject to inaccuracies. ,
The free T 4 index (FT4I) is determined by multiplying the total T 4 concentration by the T 3 or T 4 resin uptake, which is an inverse estimate of serum TBG concentrations. Free T 4 levels can also be measured by the analog method, a less expensive alternative to the FT4I ; the two tests are likely comparably, and in a healthy population, there is a close correlation between the FT4I and free T 4 levels. However, in critically ill patients, this association is no longer seen, mainly because of difficulties in estimating TBG binding with resin uptake tests. Despite this, the sensitivity of the FT4I in a large study of hospitalized patients was 92.3%, compared with 90.7% for the sensitive TSH test.
Of the serum thyroid function tests, serum T 3 concentrations are affected to the greatest degree by alterations in thyroid hormone economy during acute illness. Therefore there is no indication for routine measurement of serum T 3 levels in the initial evaluation of thyroid function among critically ill patients. This test should be obtained only if thyrotoxicosis is clinically suspected in this setting (i.e., in the presence of a suppressed sensitive TSH and elevated [or high normal] FT4I or free T 4 values). The total T 3 assay is preferable to the free T 3 (analog) assay, owing to the variability among laboratories with the latter test, although there are limited data reporting the utility of free T 3 levels in predicting mortality in the ICU. Similarly, although there is some evidence that ICU patients with lower serum rT 3 levels have higher risks of mortality, , , rT 3 levels are generally unreliable and are difficult to distinguish between intrinsic thyroid dysfunction and nonthyroidal illness.
Autoantibodies to thyroglobulin and thyroid peroxidase (TPO), two intrinsic thyroid proteins, are commonly ordered serum tests. Increased serum titers of either or both of these antibodies indicate the presence of autoimmune thyroid disease, but the presence of thyroid autoantibodies alone does not necessarily indicate thyroid dysfunction, as they are present in up to 26% of the general population. , Thyroid autoantibodies do, however, add to the sensitivity of abnormal TSH and FT4I values in diagnosing known intrinsic thyroid disease. ,
Imaging studies are rarely essential for the diagnosis of thyroid disorders in critically ill patients. Occasionally, functional analysis of thyroid glands using the radioisotope I may be useful in patients with suspected thyrotoxicosis and equivocal laboratory tests. However, these studies are labor intensive, and managing the underlying acute illness often overshadows the benefits of obtaining these studies. Anatomic studies such as ultrasound, isotopic imaging, computed tomography (CT), and magnetic resonance imaging (MRI) are useful in the evaluation of thyroid nodules and goiter, but these conditions are rarely the cause of acute illness; as such, these radiologic studies are not usually helpful in critically ill patients.
Routine serum thyroid function screening of an ICU population is not recommended because of the high prevalence of abnormal thyroid function tests and low prevalence of true thyroid dysfunction. When thyroid function tests are ordered in hospitalized patients, they should only be done if there is a high clinical index of suspicion for thyroid dysfunction. Whenever possible, it is best to defer evaluation of the thyroid-pituitary axis until patients have recovered from the acute illness. Because every test of thyroid hormone function can be altered in critically ill patients, no single test can definitively rule in or rule out the presence of intrinsic thyroid dysfunction.
If there is a high clinical suspicion for intrinsic thyroid dysfunction in critically ill patients, reasonable initial tests should include either serum free T 4 index or free T 4 and TSH measurements. Assessment of these values in the context of the duration, severity, and stage of illness will allow the correct diagnosis in most patients. For example, a mildly elevated TSH coupled with a low FT4I or free T 4 is more likely to indicate primary hypothyroidism early in an acute illness, as opposed to the same values obtained during the recovery phase of the illness. Similarly, the combination of an elevated TSH and low-normal FT4I or free T 4 is more likely to indicate thyroid dysfunction in hypothermic, bradycardic patients than the tachycardic, normothermic individuals. If both the FT4I or free T 4 and TSH are normal, thyroid dysfunction is effectively eliminated as a significant contributing factor. If the diagnosis is still unclear, measurement of thyroid antibodies is helpful as a marker of intrinsic thyroid disease and increases the sensitivity of both the FT4I or free T 4 and the TSH. Only in the case of a suppressed TSH and a mid- to high-normal FT4I or free T 4 are measurements of serum T 3 levels indicated.
In patients without underlying thyroid dysfunction, critical illness causes multiple nonspecific alterations in thyroid hormone concentrations that relate to the severity of the illness. , Proposed mechanisms of nonthyroidal illness include the induction of a central hypothyroidism arising from decreased TSH production, an acute phase response leading to thyroid binding alterations within the circulation, and impaired function of thyroid hormone transporters across cell membranes.
The alterations in thyroid hormone parameters represent a continuum of changes that depends on the severity of the illness and can be categorized into several distinct stages ( Fig. 137.2 ). The wide spectrum of changes observed often results from the differing points in the course when serum thyroid function tests are obtained. Importantly, these changes are rarely isolated and are often associated with alterations of other endocrine systems, such as the hypothalamic-pituitary-gonadal and -adrenal axes. Thus sick euthyroid syndrome should not be viewed as an isolated pathologic event, but as part of a coordinated systemic reaction to illness involving both the immune and endocrine systems.
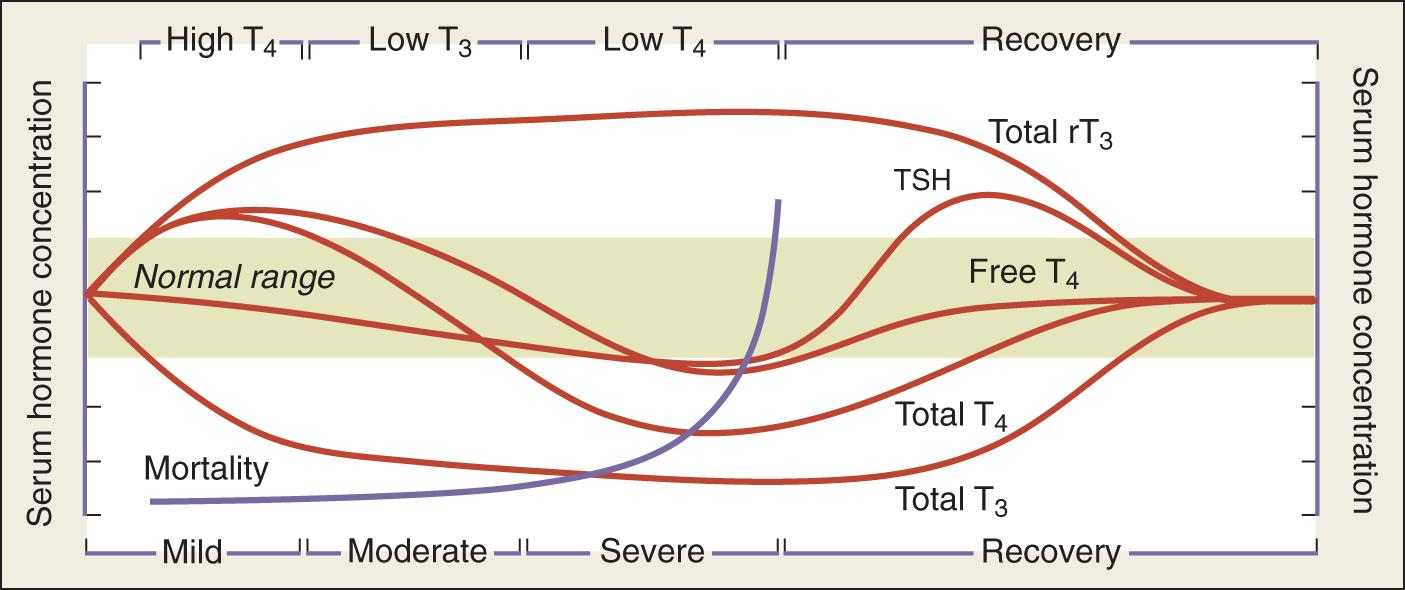
The effect of nonthyroidal illness has been studied in several acute and chronic diseases. Some investigators have postulated an association between hypothyroidism and chronic kidney disease, although the thyroid function abnormalities in this patient population with a chronic illness may represent a component of nonthyroidal illness. Others have reported that the presence of nonthyroidal illness portends worsened overall prognosis in patients with coexisting end-stage renal disease, cardiorenal syndrome, and enterocutaneous fistulas and in burn patients. There is limited literature regarding the optimal treatment of nonthyroidal illness in children who are hospitalized in the ICU.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here