Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
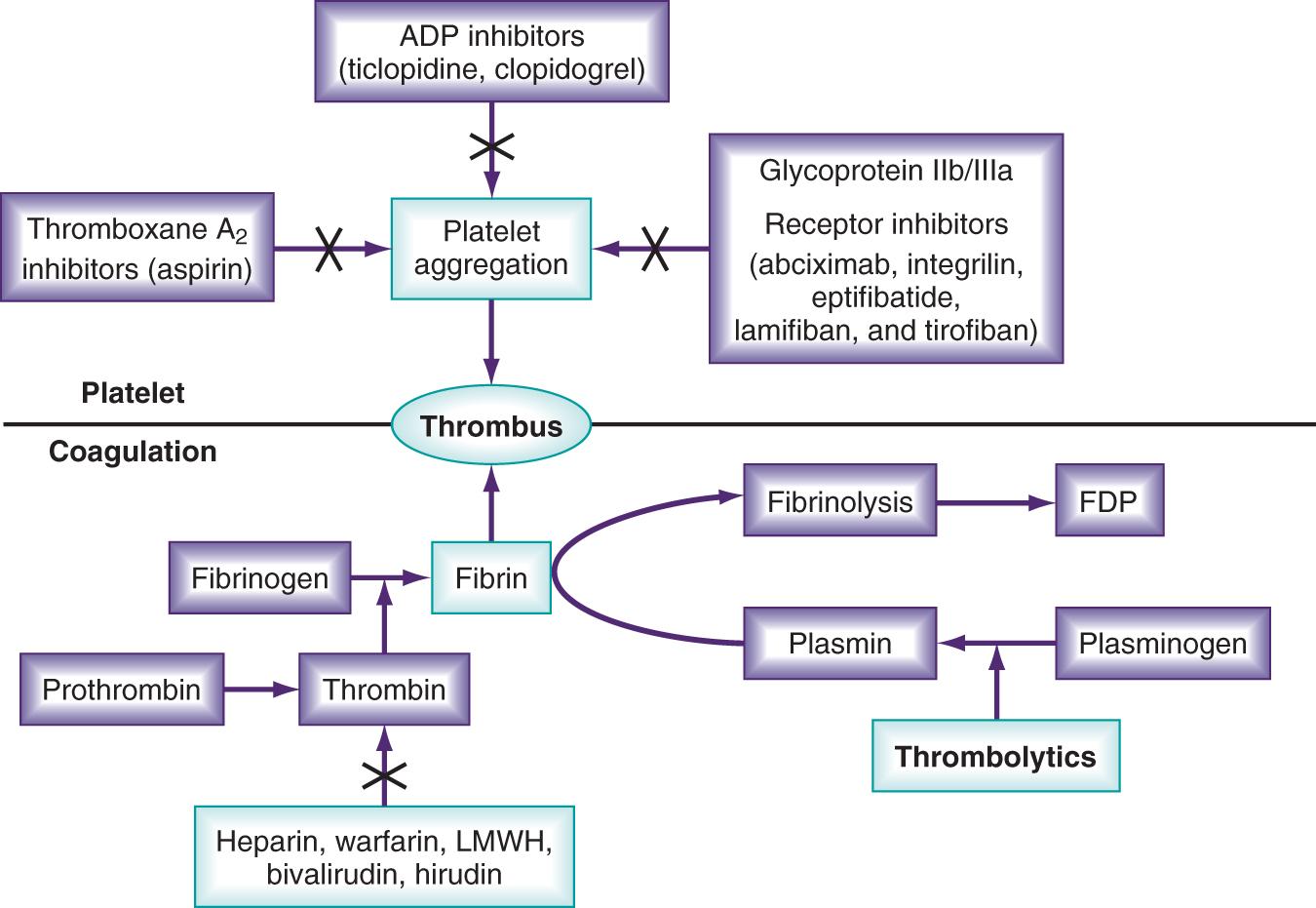
Thrombolytic agents comprise a diverse group of compounds that indirectly initiate the lysis of a thrombus. After the initiation of the coagulation cascade, fibrinolytic mechanisms are concomitantly activated to prevent unconstrained thrombosis. Fibrinolysis begins with the cleavage of proenzyme plasminogen to plasmin, which hydrolyzes key bonds within a fibrin clot matrix, resulting in clot lysis ( Fig. 152.1 ). Thrombolytic agents function by converting plasminogen to plasmin. Different thrombolytic agents vary in their specificity for plasminogen, metabolic half-life, and antigenicity ( Table 152.1 ).
| Streptokinase | Urokinase | Tissue Plasminogen Activator (tPA) | |
|---|---|---|---|
| Source | Group C Streptococcus | Human fetal kidney | Recombinant |
| Lytic | First | First (prourokinase: second) | Second (non-alteplase tPAs: third) |
| Generation | Anistreplase | ||
| Available compounds | APSAC (half-life, 70–120 min) | Prourokinase | Alteplase, duteplase, reteplase (rPA), tenecteplase (TNK-tPA), and lanoteplase (nPA) |
| Molecular weight (kD) | 47 | 35–55 | 63–70 |
| Half-life (min) | 18–23 | 14–20 | 3–4 |
| Metabolism | Hepatic | Hepatic | Hepatic |
| Antigenicity | Yes | No | No |
| Fibrin specificity | Minimal | Moderate | Moderate |
| Plasminogen binding | Indirect | Direct | Direct |
Streptokinase, a protein produced by beta-hemolytic streptococci, was identified as having fibrinolytic properties in the 1930s and was the first compound to be used clinically as a thrombolytic drug. , Streptokinase complexes with plasminogen and converts it to plasmin. However, one of the major drawbacks of using streptokinase is its antigenicity, as streptococcal infection may induce antibody formation. Mild allergic reactions occur in 2%–5% patients; however, severe anaphylactic reactions may also occur.
Urokinase is a thrombolytic protein that was initially isolated from human urine and has been used clinically for over 30 years. At present, it is isolated from human fetal renal tissue cultures. Unlike streptokinase, urokinase enzymatically cleaves plasminogen. In 1999 urokinase was removed from the U.S. market after concerns raised by the Food and Drug Administration (FDA) regarding its safety. It was reintroduced in the United States in 2002 after rigorous testing showed that urokinase preparations were free of human pathogens. However, at present, it is only approved for treating pulmonary embolism (PE). Pro-urokinase, also known as single-chain urokinase-type plasminogen activator, is a single-chain precursor of urokinase that is converted into two-chain urokinase by hydrolysis.
Tissue plasminogen activator (tPA), which was first isolated in 1981, is a naturally occurring protein synthesized by human vascular endothelial cells. Several recombinant variants of tPA are available, including alteplase (rtPA, approved by the FDA in 1987) and duteplase. Other forms of tissue-type tPAs include reteplase (rPA), tenecteplase (TNK-tPA), and lanoteplase (nPA). Recombinant tPAs are nonantigenic and show specificity for fibrin-bound plasminogen. They also avoid the infectious risks associated with products isolated from cultured human tissues. Newer recombinant tPAs have improved pharmacokinetics that enable their convenient administration, such as bolus dosage.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here