Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The vesiculobullous reaction pattern is characterized by the presence of vesicles or bullae at any level within the epidermis or at the dermoepidermal junction. Pustules, which are vesicles or bullae containing numerous neutrophils or eosinophils, are included in this reaction pattern. Vesiculobullous lesions result from a defect, congenital or acquired, in the adhesion of keratinocytes. Accordingly, it is important to understand the mechanisms involved in normal epidermal cohesion.
The integrity of the epidermis, which serves to protect humans from the external environment, is maintained by intercellular junctional complexes composed of tight junctions, adherens junctions, and desmosomes. They are composed of adhesion molecules that have an important role in cell–cell and cell–matrix adhesion. Signal transmission (intercellular communication) is a major function of gap junctions, composed of various polypeptides, the most important of which are connexins. Their role in cell–cell adhesion appears to be a minor one. A review of gap junctions was published in 2002. Adhesion molecules are transmembrane proteins, the extracellular domains of which are homophilic and the intracellular portions linked to the cytoskeleton of the cell.
There are four major families of adhesion molecules—cadherins, integrins, selectins, and the immunoglobulin superfamily—which are localized to two specialized intercellular junctions known as desmosomes (including hemidesmosomes) and the adherens junction . The more recently characterized tight junction is considered at the end of this section. In addition to their unique proteins, they also contain members of the immunoglobulin superfamily. Focal adhesions (labile structures seen in cultured keratinocytes and postulated to exist in the skin) are included with the adherens junction group. Desmosomes are well-defined, plaque-like areas of point contact that are easily seen in the spinous layer. The adherens junction is less well defined. Some work suggests that it may be situated near desmosomes. Desmosomes and adherens junctions further differ from one another in three respects:
The subclass of adhesion molecule present
The composition of the cytoplasmic plaque (discussed later)
The nature of the associated cytoskeletal element.
These aspects are considered further in the sections discussing adhesion molecules and cytoplasmic plaques.
As indicated previously, there are four families of adhesion molecules. They are sometimes divided into two groups—those that mediate cell–cell adhesion, such as the cadherins and immunoglobulin superfamily, and those that mediate cellular adhesion to matrix molecules, such as the selectins. The integrins are capable of mediating both types of adhesion. The cadherins are the most important group in keratinocyte cohesion, although the integrins have a role in basal cells, particularly in the hemidesmosomes. Cadherins and integrins are important for the maintenance of tissue integrity and in signal transduction during skin development. Only brief mention is made of the other families in the discussion that follows.
The cadherins are calcium-dependent adhesion molecules that extracellularly can attach to other cadherins; that is, they are homophilic. There are two major subfamilies of cadherins: the first, known as classic cadherins (E-cadherin), are found in the adherens junctions where their cytoplasmic domains link with cytoplasmic anchoring molecules (including β-catenin, type XIII collagen, ZO-1, and vinculin) in the cytoplasmic plaques and that, in turn, connect with actin filaments of the cytoskeleton; the second group of cadherins, which are localized to desmosomes ( desmosomal cadherins ), are linked eventually to keratin intermediate filaments via plakoglobin and the desmoplakins of the cytoplasmic plaques. In both adherens junctions and desmosomes, a cadherin tail binds to an armadillo family member (β-catenin/plakoglobin in adherens junctions or plakoglobin in desmosomes), which in turn associates with a cytoskeletal linking protein (α-catenin in adherens junction and desmoplakin in desmosomes). That is, desmosomes contain proteins from at least three distinct gene families: cadherins (desmogleins and desmocollins), armadillo proteins (plakoglobin, plakophilins [PKP1–3], and p0071, now called PKP4), and plakins (desmoplakin I and II, plectin, epiplakin, and the cell envelope proteins envoplakin and periplakin). As just mentioned, there are two major groups of desmosomal cadherins—the desmogleins and the desmocollins . Both have been mapped to chromosome 18. To date, four desmoglein and three desmocollin genes have been identified.
The desmogleins, which have large cytoplasmic domains, exhibit a tissue- and differentiation-specific pattern of expression. Of the four desmogleins, desmoglein 1 is the major desmosomal cadherin in the skin. It is expressed throughout the epidermis, but it is expressed primarily in the upper layers of the epidermis. It is the target antigen in pemphigus foliaceus. Desmoglein 2 is ubiquitous and is found in most simple epithelia and basal epidermis, whereas desmoglein 3 is found primarily in the spinous layer. It decreases gradually toward the upper layers of the epidermis. It is the target antigen in pemphigus vulgaris. Desmoglein 4 is a newly identified member of the desmoglein family that is expressed in the suprabasal layers of the epidermis. The genes for these proteins— DSG1, DSG2 , and DSG3 —are clustered within a small region in 18q12.1. Mutations in the DSG1 gene cause the dominantly inherited skin disease striate palmoplantar keratoderma type I.
Less is known about the desmocollins . They have shorter intracytoplasmic domains than the desmogleins. They may play a key role in initiating desmosome assembly. Desmocollin 1 is the major category. Its distribution mirrors that of the corresponding desmoglein 1. The genes for the desmocollins— DSC1 , DSC2 , and DSC3 —map to 18q12.1, the same region that harbors the desmoglein genes. Both immunoglobulin A (IgA) pemphigus and pemphigus foliaceus (particularly the endemic form) may sometimes be associated with autoantibodies to desmocollin. Desmocollin 3 has been found to be an autoantigen in pemphigus vulgaris.
The integrin family of adhesion molecules is involved in cell–cell and cell–matrix adhesions, particularly in the hemidesmosomes of basal keratinocytes and the “focal adhesions” of cultured keratinocytes. Integrins are heterodimers with an α and a β chain. Fourteen α and eight β chains have been described, but only a limited number of permutations have so far been described in the skin: α 2 β 1 , α 3 β 1 , and α 6 β 4 . The first two subtypes are found in the lateral and basal aspects of basal cells, whereas α 6 β 4 is located in the hemidesmosomes. The genes for the two chains of α 6 β 4 integrin are ITGA6 and ITGB4 . Epiligrin (laminin 332) is an adhesive ligand for α 3 β 1 and α 6 β integrins. Other extracellular matrix ligands include other laminins and fibronectin. The laminins are of great importance in maintaining epidermal adhesion to the dermis. For example, mutations of genes that encode the α 3 , β 3 , and γ 2 chains of laminin 332 cause variants of junctional epidermolysis bullosa, and autoantibodies to laminin 332 chains have been associated with mucous membrane pemphigoid and rare variants of bullous lupus erythematosus as well as severe bullous disease in dogs and other species.
The immunoglobulin superfamily has one or more immunoglobulin-like domains. Included in this group are molecules concerned with adhesion to lymphocytes and the intercellular adhesion molecule (ICAM). They have no significant role in keratinocyte cohesion, but they do contribute to the tight junction that has barrier rather than cohesion functions.
The selectins are mainly involved in endothelial cell adhesion. They have no significant role in the epidermis.
Cytoplasmic plaques are dense, submembranous regions of the intercellular junctions, measuring 14 to 20 nm in thickness. They are composed of filament-binding proteins that connect with the cytoplasmic domain of the cadherins and the filaments of the cytoskeleton. In desmosomes, the plaques connect with keratin intermediate filaments, whereas in the adherens junctions they connect with actin filaments.
The composition of the cytoplasmic plaques is different in the two junctions; only plakoglobin is common to both. In the adherens junctions, the cytoplasmic plaques contain α– and β- catenins . In desmosomes, the cytoplasmic plaques consist primarily of desmoplakins 1 and 2 , antibodies to which are found in paraneoplastic pemphigus. The desmoplakins play a pivotal role in anchoring the network of intermediate filaments to desmosomes. Other proteins (e.g., IFAP-300) may enhance this association. The first mutation to be described in desmoplakin was in a family with autosomal dominant striate palmoplantar keratoderma (OMIM 125647 ) (see p. 320 ). An autosomal recessive mutation in desmoplakin gives rise to Carvajal syndrome (OMIM 605676 ), which comprises dilated cardiomyopathy, woolly hair, and keratoderma (see p. 319 ). The desmoplakins, along with the bullous pemphigoid antigen 1, plectin, and the cell envelope proteins envoplakin and periplakin, belong to the so-called “intermediate filament-associated proteins,” now known as the plakin family. An absence of plectin from the hemidesmosomes has been found in a variant of epidermolysis bullosa simplex with associated muscular dystrophy. Autoantibodies to plectin (450 kDa) have been reported in a patient with a bullous pemphigoid–like eruption. One study suggests that inflammation in inflammatory bowel disease may expose plectin, prompting a secondary immune response that can cross-react with skin, resulting in bullous pemphigoid as a form of “epitope spreading” in patients who have undergone colostomy or ileostomy. Other constituents of the desmosomal cytoplasmic plaque include the armadillo proteins plakoglobin and plakophilins 1, 2, 3, and 4. Plakophilins are essential for the formation and function of desmosomes because they are involved in the recruitment and normal association of the other desmosomal proteins. Plakophilin 1 is expressed primarily in the suprabasal layers of stratifying epithelia. It is absent in patients with ectodermal dysplasia–skin fragility syndrome (OMIM 604536 ). Mutations in the plakoglobin gene result in the palmoplantar keratoderma known as Naxos disease (see p. 319 ). Absence of plakophilin 1 has been associated with skin fragility and hypohidrotic ectodermal dysplasia.
Tight junctions play an important role in the formation of the epidermal barrier. Their role in cell cohesion is minimal. They are a gatekeeper of the paracellular pathway, forming a circumferential, belt-like structure involving cells of the granular layer, in normal epidermis. Tight junctions contain the proteins occludin; ZO-1; cingulin; claudin-1, -4, -5, and possibly others; and JAM-1, a member of the immunoglobulin superfamily. It appears that ZO-1 acts as a link between JAM-1 and the claudins. Tight junction proteins are destroyed in various inflammatory diseases of the skin, including psoriasis and atopic dermatitis.
In summary, epidermal cohesion is a complex process involving the adhesion molecule families—cadherins and integrins—concentrated in areas known as desmosomes and adherens junctions. They have extracellular domains (homophilic in the case of cadherins) and intracellular ones that connect with filament-binding proteins (plakoglobin and catenins or desmoplakins) in cytoplasmic plaques. These proteins connect with filaments of the cytoskeleton, such as actin and keratin intermediate filaments.
Early lesions should always be biopsied to ensure that a histopathological diagnosis can be made. Once regeneration of the epidermis commences or secondary changes such as infection or ulceration occur, accurate diagnosis of a vesiculobullous lesion may not always be possible. Furthermore, in some blistering diseases, special techniques such as direct immunofluorescence (IF), split-skin IF, or electron microscopy may assist in making the diagnosis. Identification of the target antigen may be of diagnostic importance in some of the autoimmune blistering diseases ( Table 7.1 ). Interestingly, some of the same structural proteins targeted by autoantibodies in patients with acquired autoimmune bullous disorders are mutated in some patients with inherited bullous diseases.
| Disease | Target antigen | Site of antigen | Other minor antigens reported and comments |
|---|---|---|---|
| Pemphigus foliaceus | Desmoglein 1 | Desmosomes of upper epidermis | Desmocollin—antibodies to this alone in some endemic cases |
| Herpetiform pemphigus | Desmoglein 1 | Desmosomes of upper epidermis | Desmoglein 3, desmocollin 3 |
| IgA pemphigus (SPD type) | Desmocollin 1 | Desmosomes (transmembrane) | |
| IgA pemphigus (IEN type) | Desmoglein 1 or 3 | Desmosomes | Desmocollin 1, non-desmosomal transmembranous protein |
| Pemphigus vulgaris | Desmoglein 3 | Desmosomes of lower epidermis | Desmocollin, 85-kDa antigen (in pemphigus vulgaris–Neumann), desmoglein 1 |
| Epidermolysis bullosa acquisita | Type VII collagen | Anchoring fibrils | IgA antibody to plectin; similar antigen in bullous SLE |
| Paraneoplastic pemphigus | Desmoglein 1, 3 | Cytoplasmic plaques | Envoplakin, BP230, periplakin, γ-catenin, plectin, 170 kDa, desmocollin 2 and 3 |
| Desmoplakin 1, 2 | |||
| Bullous pemphigoid | BPAg1 | Hemidesmosome | 80% have antibodies to 230-, 30% to 180-, and 20% only to 180-kDa antigen |
| BPAg2 | Transmembrane protein (NC16A domain) | Others reported—240, 190, 138, 120, 125, 105 kDa, plectin, desmoglein 3 | |
| Pemphigoid gestationis | BPAg2 | See earlier | Others reported—200 kDa, BP230 |
| Dermatitis herpetiformis | Tissue transglutaminase (?TG3) | Gut, ? site in skin | IgA deposits haphazard in papilla |
| Linear IgA bullous dermatosis | LABD97, LAD-1 | Lamina lucida | 97- and 120-kDa antigens are degradation products of BP180 |
| LAD285 (10%–25%) | Sublamina densa | NC-1 domain of type VII collagen and others | |
| Ocular cicatricial pemphigoid | Plectin | Conjunctiva | IgA antibody, may be heterogeneous |
| Cicatricial pemphigoid | BPAg2 | Against extracellular (C-terminal) domain | Many other epitopes exist on antigen |
| Epiligrin (laminin 332) | α 3 subunit of laminin 5 | Epiligrin cases uncommon (10% or more) | |
| Deep lamina lucida pemphigoid | 105 kDa | Lower lamina lucida | Clinically resembles TEN, pemphigus vulgaris |
| Anti-p200 pemphigoid | 200 kDa | Lower lamina lucida | Clinically resembles BP, DH, or LABD |
The following three morphological features may need to be assessed in the diagnosis of vesiculobullous lesions:
The anatomical level of the split
The mechanism responsible for the split
The inflammatory cell component (in the case of subepidermal blisters)
These various aspects are considered in greater detail.
The blister may form at any one of four different anatomical levels. The split may be subcorneal (intracorneal splitting is included in this category), within the spinous or malpighian layers, suprabasilar, or beneath the epidermis (subepidermal). In the case of subepidermal blisters, several different anatomical levels may be involved, but these are “submicroscopic” and require the use of electron microscopy or other special techniques (discussed later) for their elucidation.
There are several mechanisms by which blistering can result—spongiosis, acantholysis, and ballooning degeneration of keratinocytes. Spongiosis refers to the presence of intercellular edema. In some of the disorders showing the spongiotic reaction pattern (see Chapter 6 ), the edema may be so pronounced that there is breakdown of the intercellular connections, leading to vesicle formation. Clinically visible vesicles occur in a small proportion of cases with the spongiotic reaction pattern. Acantholysis refers to the loss of attachments between keratinocytes, resulting in the formation of rounded, detached cells within the blister. Acantholysis may result from damage to the intercellular connections caused by the deposition of immune complexes, as in pemphigus, or from abnormalities of the tonofilament–desmosome complexes, which may be an acquired abnormality or have a heredofamilial basis. Acantholysis may also occur secondary to other processes such as ballooning degeneration. Ballooning degeneration of keratinocytes refers to the swelling of these cells that follows their infection with certain viruses. The ballooning results in rupture of desmosomal attachments and vesicle formation. Sometimes a few acantholytic cells are present in vesicles as an incidental phenomenon, resulting from the action of enzymes released by neutrophils in the accompanying inflammatory infiltrate. The presence of a few acantholytic cells in these circumstances should not be misinterpreted as indicating that acantholysis is the pathogenetic mechanism responsible for the blister in such a case. Junctional separation is sometimes included as a mechanism of blister formation, but it is a heterogeneous process involving different mechanisms and different anatomical levels within the basement membrane zone.
In the case of subepidermal blisters, it is usual to subclassify them further on the basis of the predominant cell in the inflammatory infiltrate in the underlying dermis. In some subepidermal blisters, the proportion of eosinophils and neutrophils may vary from case to case and with the age of the lesion. These caveats must always be kept in mind when a subepidermal blister with neutrophils or eosinophils is biopsied. The presence of neutrophils within intraepidermal blisters may also have relevance to the diagnosis, even though this aspect is not used in the subclassification of intraepidermal blisters. A detailed study of the inflammatory cell infiltrate in various blistering diseases has been published. Many cells expressing CD68 were present in all of the diseases studied. CD68 is not specific for histiocytes/macrophages.
Although the key features in the assessment of any vesiculobullous lesion are the anatomical level of the split, the mechanism responsible for the split, and the nature of the inflammatory cell infiltrate, as discussed previously, the presence of changes in keratinocytes may assist in making a diagnosis in several diseases. Examples include the presence of dyskeratotic cells in Darier's disease, the presence of multinucleate giant cells in certain virus-induced blisters, and confluent epidermal necrosis in toxic epidermal necrolysis and in severe erythema multiforme. Shrunken keratinocytes (Civatte bodies) may be seen in bullous lichen planus, bullous fixed drug eruptions, erythema multiforme, and paraneoplastic pemphigus.
In this group of vesiculobullous diseases, the split occurs within the stratum corneum or directly beneath it. In addition to the conditions discussed here, subcorneal blisters or pustules have been reported uncommonly as a manifestation of epidermolysis bullosa simplex, acute generalized pustulosis and other pustular vasculitides, and pyoderma gangrenosum (see p. 290 ). There is one report of a patient with vegetative plaques resembling pemphigus vegetans and a subcorneal spongiform pustule with marked acanthosis of the epidermis on histological examination. Subcorneal splitting is usually present in the peeling skin syndrome (see p. 343 ). It may be congenital (see p. 343 ) or acquired. Azathioprine has been implicated in acquired cases. Subcorneal pustules have been reported in adult Still's disease.
Other causes of intracorneal or subcorneal blisters are discussed next.
Impetigo is an acute superficial pyoderma that occurs predominantly in childhood (see p. 674 ). Staphylococcus aureus is the usual organism isolated from this condition. There are two clinical forms of impetigo—a common vesiculopustular type and a rare bullous type.
In impetigo, there are subcorneal collections of neutrophils. A few acantholytic cells are sometimes present, particularly in bullous impetigo, as a result of the action of enzymes released from neutrophils. Acantholysis is never as prominent in impetigo as it is in pemphigus foliaceus. Gram-positive cocci can usually be demonstrated in impetigo, another distinguishing feature of this condition.
Subcorneal pustules, sometimes resembling impetigo, can be seen in some cases of listeriosis.
The staphylococcal scalded skin syndrome (SSSS) is discussed in detail with the bacterial infections on p. 675 . It results from the production of an epidermolytic toxin by certain strains of S. aureus . This toxin cleaves the extracellular domain of desmoglein 1.
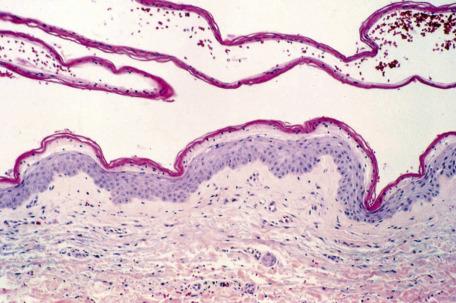
It is usually difficult to obtain an intact blister in SSSS because the stratum corneum may be cast off during the biopsy procedure or the subsequent processing of the specimen. A few acantholytic cells and neutrophils are usually present in intact blisters or on the surface of the epidermis if its roof has been shed ( Fig. 7.1 ). Organisms are not usually present in the affected skin, in contrast to bullous impetigo. There is usually only a sparse inflammatory cell infiltrate in the upper dermis, in contrast to bullous impetigo and pemphigus foliaceus in which the infiltrate is usually heavier.
Subcorneal and intraepidermal blisters are sometimes seen in the dermatophytoses, particularly on the hands and feet. The presence of neutrophils in the stratum corneum or within the epidermis should always prompt consideration of an infectious causes, including fungi. Candidosis is another uncommon cause of subcorneal blistering.
Pemphigus foliaceus, which accounts for approximately 10% of all cases of pemphigus, is one of the less severe forms of the disease. There are recurrent crops of flaccid bullae that readily rupture, resulting in shallow erosions and crusted erythematous plaques. A stinging or burning sensation is sometimes present. Lesions may be localized to the face and trunk initially, but the condition usually spreads to involve large areas of the body. Rarely, the lesions remain localized to a site, such as the nose. Mucous membrane involvement is rare. Umbilical involvement in pemphigus vulgaris and pemphigus foliaceus has been emphasized in one study; the reason for this is yet to be elucidated. No age, including childhood and the neonatal period, is exempt, although the majority of cases present in late middle life. Postpartum onset and neonatal involvement, after passive transfer of antibodies across the placenta, have been reported.
Rare clinical presentations have included generalized erythroderma, lesions resembling eruptive seborrheic keratoses, and in other instances erythematous and vesicular lesions suggestive of dermatitis herpetiformis. This latter group, also known as pemphigus herpetiformis and herpetiform pemphigus, has autoantibodies directed against desmoglein 1 (dsg1), indicating its close relationship to pemphigus foliaceus (discussed later). Rarely, the clinical and histological features of pemphigus foliaceus may change to those of pemphigus vulgaris; the antibody profile also changes. Such cases are probably examples of intermolecular epitope spreading. This is also the explanation for the conversion of pemphigus foliaceus into bullous pemphigoid and for mixed features of these two diseases. Although epitope spreading is a mechanism of protection from pathogens, it propagates autoimmunity. Patients with features of both pemphigus vulgaris and pemphigus foliaceus clinically, histologically, and immunologically have been reported.
Pemphigus foliaceus has been reported in association with bullous pemphigoid, lupus erythematosus, dermatomyositis, rheumatoid arthritis, IgA nephropathy, myasthenia gravis, thymoma, lymphoma, prostate cancer, herpes simplex infection (eczema herpeticum), silicosis, lichen planus, Graves’ disease, mycosis fungoides, and multiple autoimmune syndrome type 2. Both pemphigus foliaceus and psoriasis can be associated with exfoliative erythroderma. Accordingly, there are reports of patients initially diagnosed as having erythrodermic psoriasis who proved to have pemphigus foliaceus and also instances of pemphigus foliaceus arising in patients with psoriasis, one example occurring after treatment of the latter with multiple cycles of narrowband UV-B therapy. The disorder has been complicated by disseminated herpes simplex infection and lesions of cytomegalovirus. Pemphigus foliaceus may be induced by sunlight and by ionizing radiation. It developed in a patient with cutaneous squamous cell carcinoma (SCC) that metastasized to regional lymph nodes. The production of anti-dsg1 antibodies started when the SCC metastasized. It has also been associated with the use of penicillamine, bucillamine, gold, pyritinol, rifampin (rifampicin), captopril, enalapril, ramipril, fosinopril, cephalosporins, levodopa, aspirin, methimazole (thiamazole), and α-mercaptopropionylglycine (tiopronin). Some drugs that contain thiol groups can produce acantholysis, in vitro , in human skin explants. Pemphigus foliaceus has been exacerbated by a tetanus vaccination. Localized disease has been produced by the application of topical imiquimod.
Pemphigus foliaceus is the most common form of pemphigus complicating the use of penicillamine. Pemphigus develops in nearly 10% of those taking penicillamine for prolonged periods, and it may persist for many months after the drug is discontinued. Sometimes the eruption that ensues is not typical of a specific type of pemphigus but shares clinical or immunohistological features of different types of the disease.
Pemphigus foliaceus results from the formation of autoantibodies, mainly of the IgG4 subclass, that react with several different antigenic epitopes on the amino-terminal region of dsg1, a 160-kDa transmembrane glycoprotein, which is present in desmosomes. The amino-terminus of dsg1 appears to be a key region for the intercellular adhesion of cadherins. In certain cases, the antibodies appear to react with desmocollins, the other subtype of desmosomal cadherins, or other antigens. The expression of dsg1 is highest in specimens from the upper torso, excluding the scalp. It is expressed primarily in the upper layers of the epidermis. Dsg1 is expressed at a much lower level than dsg3 in oral mucosa. This suggests that dsg3 may be sufficient for cell–cell adhesion in oral mucosa, with consequently no oral involvement in patients with pemphigus foliaceus. Neonates are protected from the effect of the passive transfer of maternal autoantibodies across the placenta by possessing dsg3 in the upper levels of the epidermis. The antibodies found in patients with drug-induced pemphigus, and in some patients with pemphigus vulgaris, also react with dsg1. The antibodies themselves are pathogenic, but complement, including the terminal complement sequence (membrane attack complex), is also an important mediator in the detachment of the epidermal cells. The use of plasminogen activator knockout mice suggests that there is no requirement for proteases in blister formation. Antibody levels fluctuate during the course of the disease and have some correlation with disease activity. However, a study has shown that the predominant binding of anti-dsg1 antibodies to the amino-terminus of dsg1 persists despite the activity stage of the disease, including periods of remission. The availability of recombinant dsg1 has facilitated the development of antigen-specific plasmapheresis as a therapeutic strategy. A subset of pemphigus foliaceus patients exhibits pathogenic autoantibodies against both dsg1 and dsg3. The figure is less than 7%.
Cytokines also have a pathogenic role in this disease. Tumor necrosis factor-α (TNF-α) levels are found in the serum and blister fluid. Increased cytokine production is the likely mechanism of the localized pemphigus foliaceus produced by topical imiquimod. Epidermal keratinocytes can be induced to produce proinflammatory cytokines and other mediators such as bradykinin. The kinin system might have a potential role in the pathogenesis of acantholysis in pemphigus foliaceus, along with TNF-α.
The variant endemic pemphigus foliaceus is also known by the Portuguese expression fogo selvagem (“wild fire”) and as Brazilian pemphigus foliaceus. It affects mostly children and young adults and is endemic in certain rural areas of South America, particularly areas of Brazil. In contrast, the new form of endemic pemphigus reported from Colombia, also called El Bagre-EPF (El Bagre, “the catfish,” is a municipality in Colombia), predominantly affects 40- to 60-year-old men, as well as a few postmenopausal women, and the patients are primarily miners who also engage in farming. Furthermore, the Colombian variant more closely resembles pemphigus erythematosus. High levels of mercury have been found in the skin of these patients. Ocular involvement has been reported in patients with extensive skin involvement, and antibodies to meibomian glands and tarsal muscle have been detected in this variant. Similarly, hair loss was known to be a feature of severe endemic pemphigus foliaceus in the precorticosteroid area, and autoantibodies within the hair follicle have been found in these patients. Cases resembling the Brazilian form of the disease have been reported from Venezuela. An “endemic” form of pemphigus has been reported in Tunisia, although its “endemic” nature has not been clearly demonstrated. Dsg1 antibodies are prevalent in Tunisian patients with hydatidosis and leishmaniasis, and this may be a predisposing factor for the later development of pemphigus foliaceus. The epidemiology strongly suggests an environmental factor, possibly a virus, and this is supported by the finding of elevated levels of thymosin α 1 in many affected individuals. Most of these affected individuals live within a 10- to 15-km radius of rivers and streams, where black flies of the Simulium type are found. It has been suggested that the fly may trigger a response in genetically predisposed individuals. This predisposition does not seem to involve a defect in innate immunity. It has been found that anti-dsg1 IgG4 antibodies cross-react with a salivary gland protein, LJM11, from the sand fly Lutzomyia longipalpis, suggesting a mechanism by which an arthropod bite could initiate endemic pemphigus foliaceus in susceptible individuals. The endemic variant has an abrupt onset and a variable course. Sunlight can exacerbate the condition. Improvement has been recorded in a patient who developed HIV infection. Familial cases occur in 10%, in contrast to their rarity in the more usual form of pemphigus foliaceus. In many cases, the circulating intercellular antibodies have similar antigenic specificity to those found in the nonendemic form of the disease, although in some, antibodies to the desmocollins, rather than to dsg1, have been present. Furthermore, their localization may be at a different site on the cell surface (discussed later). Recent work suggests that the anti-dsg1 response in the endemic disease involves IgM class antibodies, and this sets it apart from other forms of pemphigus. The prevalence of antibodies against dsg1 is high among normal subjects living in endemic areas, and the onset of the disease is preceded by a sustained antibody response. There is serological evidence that the incidence of dsg1 antibodies may be declining in some endemic areas of Brazil. Circulating antibodies, using indirect IF and human skin as substrate, can be demonstrated in 70% of cases. This figure is much higher (90%) if more sensitive enzyme-linked immunosorbent assay (ELISA) techniques are used. A small number of cases also have antibodies to dsg3. Of interest is the finding that the intraperitoneal injection into mice of IgG from patients with endemic pemphigus foliaceus causes acantholysis in the animals. Cytokine and chemokine alterations also occur in endemic pemphigus foliaceus, especially elevated levels of interleukin-22 (IL-22), but also increased CXCL-8 and reduced levels of interferon-γ (IFN-γ), IL-2, IL-15, and CCL-11. In addition to the humoral-immune response, cell-mediated mechanisms involving CD4 + lymphocytes are also involved.
In the Colombian form of the disease, all sera have antibodies to dsg1; however, there are additional antibodies, as observed in paraneoplastic pemphigus, but there is no association with neoplasia. The antibodies are to desmoplakin 1, envoplakin, and periplakin.
Corticosteroids are the treatment of choice, but because this is a chronic disease that often requires long-term therapy, other adjuvants have been added for their steroid-sparing effect. Sometimes these other agents are used as monotherapy. They include azathioprine, cyclophosphamide, gold, methotrexate, chlorambucil, cyclosporine (ciclosporin), mycophenolate mofetil, dapsone, hydroxychloroquine, plasmapheresis, extracorporeal photopheresis, intravenous immunoglobulin, and topical epidermal growth factor. Isolated cases, refractory to other therapies, have been successfully treated with etanercept and rituximab. There is experimental evidence that ectopic superficial expression of desmoglein 2 can limit epidermal blister formation produced by pemphigus foliaceus antibodies and exfoliative toxin-A. The plant lectin jacalin specifically binds to O -linked Thomsen–Friedenreich carbohydrate structures on dsg1 and inhibits its interaction with pemphigus foliaceus IgG, thereby interfering with the latter's pathogenicity; this may have potential therapeutic value.
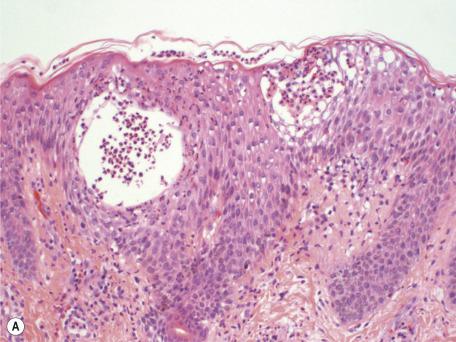
Established lesions of pemphigus foliaceus of both endemic and nonendemic forms show a superficial bulla with the split high in the granular layer or directly beneath the stratum corneum. The bulla contains fibrin, some neutrophils, and scattered acantholytic keratinocytes. No bacteria are present, unlike in bullous impetigo. A localized clinical variety with a peau d'orange clinical appearance was characterized by acantholysis exclusively involving follicular infundibula. The earliest change appears to be the formation of vacuoles in the intercellular spaces in the upper layers of the epidermis. These expand, leading to cleft formation. Uncommonly, eosinophilic spongiosis is seen as a precursor lesion, and transitions between this picture and that of pemphigus foliaceus may be seen. Eosinophilic spongiosis appears to be more common in pemphigus foliaceus in some African races. Neutrophilic spongiosis is a rare occurrence in pemphigus foliaceus ; its occurrence is usually related to the deposition of IgA or the herpetiform type (herpetiform pemphigus). Neutrophilic pustules are another uncommon manifestation of pemphigus foliaceus.
In late lesions of pemphigus foliaceus, the epidermis may be hyperplastic, with overlying focal parakeratosis and some orthokeratosis. Dyskeratotic cells with hyperchromatic nuclei and somewhat resembling the “grains” found in Darier's disease are a distinctive feature of the granular layer ( Fig. 7.2 ). In the rare mixed forms of the disease, both suprabasal acantholysis and subcorneal clefting with acantholysis are present.
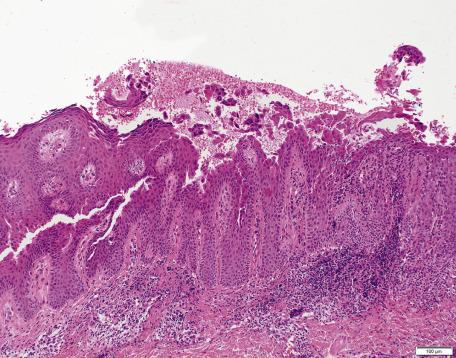
The superficial dermis is edematous with a mixed inflammatory cell infiltrate that usually includes both eosinophils and neutrophils. In drug-induced lesions, eosinophils may predominate in the dermal infiltrate.
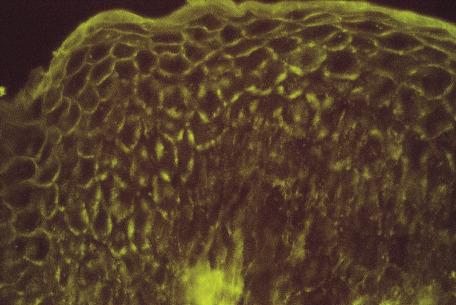
With direct IF, there is intercellular staining for IgG and C3 in both affected and normal skin. Sometimes the staining is localized to the upper levels of the epidermis ( Fig. 7.3 ). Rarely, these immunoreactants may be present along the basement membrane, even in cases that clinically resemble pemphigus foliaceus rather than pemphigus erythematosus (discussed later). In a few instances, IgA rather than IgG has been present in the intercellular regions. The term IgA pemphigus has been used for these cases. Indirect IF demonstrates circulating antibodies in nearly 90% of cases of nonendemic pemphigus foliaceus. A subset of cases of pemphigus foliaceus also has positive indirect IF using a rat bladder epithelial substrate—a feature more typical of paraneoplastic pemphigus. A new biochip IF test for the serological diagnosis of pemphigus foliaceus (as well as one for pemphigus vulgaris) has been developed that tests simultaneously for multiple diagnostically relevant antibodies (e.g., desmogleins 1 and 3 and bullous pemphigoid antigens), serving as a fast and cost-effective screening tool.
Trichoscopy can be of value in the differential diagnosis of scalp lesions of pemphigus foliaceus; the most common findings include extravasations and yellow hemorrhagic crusts, yellow dots with whitish halos, white polygonal structures, and, less commonly, vascular anomalies such as linear serpentine vessels. Reflectance confocal microscopy can be used to suggest an initial diagnosis of pemphigus, including pemphigus foliaceus, although it is not considered a replacement for histopathological and IF studies. The diagnosis is suggested when two of three criteria are met: acantholytic clefts in a lesion, acantholytic clefts in normal-appearing adjacent skin, and multiple dilated blood vessels in lesional skin.
Acantholysis in pemphigus foliaceus appears to result from separation of the nonspecific junctions and subsequent rupture of desmosomal junctions. The tonofilament–desmosomal complexes remain intact, although irregular bundles of tonofilaments are found within the acantholytic cells. Internalization of intact desmosomes also occurs. Clinically unaffected mucosa in endemic pemphigus foliaceus shows widening of the intercellular spaces with distended, elongated cytoplasmic projections. Nikolsky-positive skin is characterized by widening between desmosomes, decreased numbers of desmosomes, and hypoplastic desmosomes in lower epidermal layers, with acantholysis sometimes demonstrable only in upper epidermal layers; lesional skin shows both upper epidermal acantholysis and separation of hypoplastic desmosomes from adjacent cells. Immunoelectron microscopy shows immunoglobulins deposited over the plasma membrane of the keratinocytes and permeating the desmosomal junctions in the endemic form, whereas in the usual form there is affinity for desmosomes and separated attachment plaques in the upper layers of the epidermis.
Pemphigus erythematosus (Senear–Usher syndrome), which accounts for approximately 10% of all cases of pemphigus, is a variant of pemphigus foliaceus that combines some of the immunological features of both pemphigus and lupus erythematosus. It usually develops insidiously with erythematous, scaly, and crusted plaques in a butterfly distribution over the nose and malar areas. It may also involve other “seborrheic areas” such as the scalp, pectoral, and interscapular regions, as well as intertriginous areas. A case has presented as extensive scalp ulcerations. Usually, there is no visceral involvement. Pemphigus erythematosus may persist almost indefinitely as a localized disease. Sunlight sometimes adversely affects its course. This condition is extremely rare in children.
Pemphigus erythematosus is occasionally found in association with other autoimmune diseases, especially myasthenia gravis with an accompanying thymoma. Rare cases of concurrent pemphigus erythematosus and systemic lupus erythematosus (SLE) have been documented. There are reports of its association with parathyroid adenoma, internal cancers, burns, and X-radiation. Drugs that may induce pemphigus erythematosus include ceftazidime, penicillamine, propranolol, captopril, pyritinol, thiopronine, and heroin. Relapse of pemphigus erythematosus has occurred with atorvastatin, an agent that has been linked to autoimmune phenomena with prolonged exposure.
Antinuclear antibodies and circulating antibodies to dsg1 are often present. Antibodies to DNA are usually absent, but in one case both anti-DNA and multiple anti–extractable nuclear antigen (ENA) antibodies were present. In one case with mucocutaneous pemphigus vulgaris with higher titers of dsg1 than dsg3, the patient developed cutaneous lesions resembling pemphigus erythematosus. A genetic predisposition in some cases is suggested by associations with HLA haplotypes A10, A26, and DRW6. The treatment is similar to that used for pemphigus foliaceus, although some cases can be refractory to standard treatment and require extensive immunosuppressive therapy.
The appearances are identical to those of pemphigus foliaceus, with a subcorneal blister containing occasional acantholytic cells (discussed previously). Eroded and crusted lesions may develop.
Direct IF usually demonstrates IgG and/or complement, both in the intercellular spaces and at the dermoepidermal junction. The intercellular staining may be more pronounced in the upper layers of the epidermis. The lupus band test is sometimes positive in uninvolved skin. A possible explanation for the basement membrane zone deposits has been provided in a study of three cases of pemphigus foliaceus that were subjected to UV-A phototherapy. Oktarina et al. found evidence for release of desmoglein 1 fragments, which appear to form immune complexes with circulating anti–desmoglein 1 IgG antibodies and deposit along the basement membrane zone. A similar mechanism may occur in pemphigus erythematosus and would also explain the localization of these lesions to sun-exposed skin.
Herpetiform pemphigus (pemphigus herpetiformis) is a rare entity that combines the clinical features of dermatitis herpetiformis and the immunopathology of pemphigus, usually the foliaceus type (discussed previously). Such cases account for approximately 7% of all cases of pemphigus. Clinically, there are erythematous, urticarial plaques and vesicles in herpetiform arrangement. Eczematous features have also been described. Severe pruritus is often present. Mucous membranes are uncommonly involved. It has been reported in association with systemic lupus erythamatosis (SLE), psoriasis, sarcoidosis, solid tumors, extramammary Paget's disease, maternal B-cell lymphoma in a case that also demonstrated transplacental transmission of the disease, and endemic pemphigus foliaceus. Its association with psoriasis is sometimes related to treatment with ultraviolet light. Cases may evolve into pemphigus foliaceus. There is also report of pemphigus herpetiformis (with anti–desmoglein 1 antibodies) caused by erdosteine, a mucolytic agent.
Most cases have circulating autoantibodies to desmoglein 1, the pemphigus foliaceus antigen. A few cases with antibodies to the pemphigus vulgaris antigen, desmoglein 3, have been reported. Cases have also been reported with antibodies to desmoglein 1 and desmocollin 1, antibodies to desmocollin 3, and antibodies to desmocollins 1, 2, and 3. A case with IgA and IgG antibodies to desmoglein 1 and IgG antibodies to desmocollin 3 has been reported. In another case, there were antibodies both to desmoglein 1 and to full-length 180-kDa bullous pemphigoid antigen; there was no staining for IgG along the epidermal basement membrane zone with direct IF, but circulating anti–basement membrane zone antibodies were detected at a titer of 1 : 10. Concurrence of bullous pemphigoid and herpetiform pemphigus is reported, with IgG antibodies to desmogleins 1 and 3, desmocollins 1–3, and the NC16a domain of bullous pemphigoid 180-kDa antigen; direct IF featured basement membrane zone staining for C3 as well as intercellular IgG deposition. Still another report documents a case in which a patient with linear IgA disease shifted to pemphigus herpetiformis, complete with typical microscopic and IF changes and desmocollin 1 reactivity by ELISA assay.
The characteristic features of herpetiform pemphigus are eosinophilic spongiosis with the formation of intraepidermal vesicles. Variable numbers of neutrophils may be present, leading to the formation of neutrophilic spongiosis or subcorneal pustules with both eosinophils and neutrophils. Acantholysis is minimal or absent in some cases, but it was present in half of the cases reported in a study at the University of Toronto.
Direct IF shows the deposition of IgG, with or without C3, on the cell surfaces of keratinocytes, as seen in pemphigus. There is usually superficial accentuation, but in cases with antibodies to desmoglein 3, the deposition may be more prominent on the lower layers of the epidermis.
Subcorneal pustular dermatosis is a rare, chronic, relapsing, vesiculopustular dermatosis with a predilection for the trunk, particularly intertriginous areas, and the flexor aspect of the limbs. Atypical presentations, including lesions confined to the extensor aspects of the extremities, have been reported. It usually spares the face and mucous membranes, and there are usually no constitutional symptoms. The condition is more common in women, particularly occurring in the fourth and fifth decades of life. Cases purporting to be subcorneal pustular dermatosis in children have been disputed, but some cases appear to be genuine examples of the disease.
The pustules are flaccid. They are initially sterile, but secondary infection sometimes develops. A transient erythematous flare surrounds the pustules in the early stages.
The cause and pathogenesis remain unknown. A small number of patients have had an associated monoclonal gammopathy, most commonly of IgA type, but an IgG monoclonal gammopathy of undetermined significance has also been reported. Some of these cases have been associated with intercellular deposits of IgA and would now be reclassified as IgA pemphigus (discussed later). In one study, all cases of classical subcorneal pustular dermatosis with negative immunological findings were indistinguishable clinically and histopathologically from the subcorneal pustular dermatosis–type intercellular IgA dermatosis. Whether subcorneal pustular dermatosis survives as an entity must await further reports, but there are clearly cases that lack intercellular deposits and would be difficult to place in another category. Pyoderma gangrenosum has been described in patients with subcorneal pustular dermatosis, usually in association with an IgA monoclonal gammopathy. In two cases, the lesions of subcorneal pustular dermatosis developed several hours after the performance of echography. Other rare associations include aplastic anemia, AIDS, Sjögren's syndrome, seronegative arthritis, multiple sclerosis, SLE, and diffuse scleroderma. Cases of subcorneal pustular dermatosis have also been linked to infections, including those caused by Mycoplasma pneumoniae and Coccidioides immitis . Subcorneal pustular dermatosis is regarded by some authorities as a variant of pustular psoriasis. This confusion has arisen, in part, because cases not conforming to the original 1956 description of Sneddon and Wilkinson have been reported misleadingly as cases of subcorneal pustular dermatosis. Some of these cases have been examples of the annular variant of pustular psoriasis. Note that 7 of the 23 purported cases of subcorneal pustular dermatosis seen at the Mayo Clinic subsequently developed generalized pustular psoriasis. A child has been reported with a diffuse sterile pustular eruption that progressed from annular pustular psoriasis to subcorneal pustular dermatosis.
Pustular eruptions with some histological or clinical resemblance to subcorneal pustular dermatosis have been reported after the ingestion of isoniazid, diltiazem, gefitinib, paclitaxel, the cephalosporins, and amoxicillin. New lesions have been precipitated in a patient with subcorneal pustular dermatosis after the ingestion of dapsone and of quinidine sulfate. Sterile, subcorneal pustules sometimes form in pustular vasculitis.
TNF-α is increased in subcorneal pustular dermatosis; it may be responsible, in part, for the activation of neutrophils that is a feature of this condition.
The treatment of choice is dapsone. Therapeutic alternatives include retinoids ; psoralen-UV-A (PUVA), broadband or narrowband UV-B, colchicine, and corticosteroids ; cyclosporine with prednisolone ; infliximab ; adalimumab ; intravenous immunoglobulin ; and maxacalcitol. Sustained remission of skin lesions has also been achieved with melphalan and autologous stem cell transplantation administered for treatment of associated myeloma; however, 28 months after melphalan therapy the serum M-protein reappeared, and shortly thereafter subcorneal pustular dermatosis lesions recurred.
The subcorneal pustule is filled with neutrophils, with an occasional eosinophil. Neutrophils also migrate through the epidermis, but they do not form spongiform pustules. The pustule appears to “sit” on the epidermis ( Fig. 7.4 ), and usually it causes no depression of the latter. An occasional acantholytic cell may be present in older lesions, a result of the activity of the proteolytic enzymes released from neutrophils. Mitotic figures are usually absent within the epidermis, unlike pustular psoriasis.
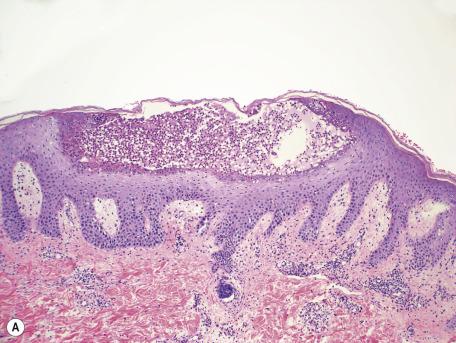
A mixed superficial perivascular inflammatory cell infiltrate is present in the underlying dermis. In early lesions, the infiltrate includes quite a few neutrophils.
Direct IF is usually negative, although immunoreactants have been described in the epidermis in rare cases. Several cases have been reported of a condition resembling subcorneal pustular dermatosis clinically but in which a biopsy of the lesions showed intercellular IgA. Such cases are now classified as IgA pemphigus, although a case could be made for the retention of some of them as variants of subcorneal pustular dermatosis. This subject is discussed further in the following section.
IgA pemphigus actually represents a complex of autoimmune, intraepidermal vesiculobullous eruptions, with variable acantholysis and the presence of intercellular deposits of IgA within the epidermis. There are at least six types, with clinical, microscopic, IF, and target antigen findings that differ from one another in some respects but also show some overlapping features and immunological complexities. The current status of these disorders has recently been reviewed by Hashimoto and colleagues, who prefer the term intercellular IgA dermatosis for these conditions. These six types are subcorneal pustular dermatosis (SPD) type , intraepidermal neutrophilic IgA dermatosis (IEN), IgA–pemphigus vegetans , IgA–pemphigus foliaceus , IgA–pemphigus vulgaris , and unclassified Intercellular IgA dermatosis . There are recurring crops of pruritic papules and vesicles that evolve into eroded and crusted plaques, involving the trunk and proximal extremities. Intertriginous areas are particularly affected in the SPD type. Other sites may also be involved. The oral mucosa tends to be spared but is involved in the IEN pemphigus and IgA–pemphigus vulgaris types. Lesions with deeper pustules or forming “sunflower” configurations also occur in the IEN type.
A case of IEN-pemphigus associated with ulcerative colitis has been reported; this patient did not have detectable antibodies against the typical target antigens associated with IgA pemphigus (see later) but did have circulating IgA antibodies against the 120-kDa ectodomain of the bullous pemphigoid 180 antigen, which is commonly detected in linear IgA disease. IgA pemphigus has been associated with monoclonal IgA gammopathy and, in a recent case, with IgA-κ multiple myeloma. There has been one case of IgA pemphigus associated with a submucosal duodenal tumor that proved to be diffuse large B-cell lymphoma; treatment of the lymphoma with rituximab–CHOP (cyclophosphamide, doxorubicin hydrochloride [hydroxydaunorubicin], vincristine [Oncovin], and predinisone) therapy resulted in the disappearance of the skin lesions.
As the name implies, patients with the pemphigus vegetans–like variant of IgA pemphigus have vegetative plaques resembling those of traditional pemphigus vegetans, and lesions of this type have also been encountered in IgA–pemphigus vulgaris. IgA–pemphigus vegetans can also be associated with inflammatory bowel disease. A recently reported case of SPD-type IgA pemphigus was induced by imatinib; other drug-induced cases have been due to thiol agents and adalimumab.
In their recent study of cases of intercellular IgA dermatosis, Hashimoto et al. found that IgA ELISA of desmogleins 1 and 2 and desmocollins 1 to 3 was most helpful in diagnostic work (along with direct IF and indirect IF using normal human skin) and was preferable to IgA complementary DNA (cDNA) transfection and IgA immunoblotting of normal human epidermal abstract. This resulted in the development of tentative criteria for diagnosis (with regard to immunoreactivity), as follows: all intercellular IgA dermatosis in general: exclusively IgA antibodies; SPD type: desmocollin 1 (weak desmocollin 3 in one case); IEN type: none (weak desmocollin 3 in one case); IgA–pemphigus vegetans: unknown (desmocollin 2 in one case); IgA–pemphigus foliaceus: desmoglein 1 (also desmocollins); IgA–pemphigus vulgaris: desmoglein 3 (also desmoglein 1 or desmocollins); and unclassified intercellular IgA dermatosis: desmoglein 1, desmoglein 3, and desmocollins.
Cases have been reported in children and also in adult men with HIV infection.
Dapsone is the drug of first choice. It may be used alone or combined with topical steroids and colchicine. Other agents include retinoids, methotrexate, oral corticosteroids, adalimumab, mycophenolate mofetil, tetracycline, sulfamethoxypyridazine, cotrimoxazole, cyclosporine, and PUVA. Cases refractory to standard therapy continue to be reported.
IgG/IgA pemphigus has received considerable attention in the past several years. It shows minimal clinical, microscopic, IF, antibody type, or treatment differences from standard IgG pemphigus but does differ in several ways from IgA pemphigus. For example, it is less apt to show clinically pustular lesions or an intertriginous distribution (as would be the case for SPD-type IgA dermatosis). However, crusted erosions and blisters are seen, some with a “sunflower-like” arrangement or a hypopyon sign (i.e., a layering of leukocytic exudate within skin lesions) such as that seen in IEN-type IgA dermatosis, and intraoral lesions can occur. Disease associations include Sjögren's syndrome and ulcerative colitis. Malignancies reported with IgG/IgA pemphigus include carcinomas of the endometrium, lung, ovary, gallbladder, and thymoma as well as IgA gammopathy. With respect to an association with malignancy, IgG/IgA pemphigus more closely resembles traditional IgG pemphigus than IgA pemphigus, in which malignancy has been uncommon. ELISA studies generally show IgG and IgA antibodies to desmogleins 1 and/or 3 but also sometimes to desmocollins or desmoplakin. Two unusual cases are the one reported by Uchiyama et al., associated with ulcerative colitis, in which there were negative IgG and IgA antibodies to desmogleins 1 and 3 but positive IgA antibodies to desmocollin 1, and the case of paraneoplastic pemphigus reported by Otsuka et al. with IgG and/or IgA antibodies to desmogleins, desmocollins, envoplakin, periplakin, and the 180-kDa and 230-kDa bullous pemphigoid antigens.
In the SPD type, there are subcorneal pustules with variable but usually mild acantholysis, whereas in the IEN type there are intraepidermal pustules. There is usually a mixed inflammatory cell infiltrate in the underlying dermis. A case of IgA pemphigus (foliaceus) has been reported that resembled the more usual cases of pemphigus foliaceus without any neutrophilic infiltration. Neutrophilic spongiosis, suggestive of IgA pemphigus, has been reported in a case of pemphigus foliaceus devoid of IgA. A rare variant of the IEN type, which occurred in a child on immunosuppressive drugs, had features of pemphigus vegetans with epidermal hyperplasia and intraepidermal pustules.
Direct IF shows intercellular deposition of IgA in the epidermis. There is usually increased intensity of staining in the upper layers of the epidermis in the SPD type. In the IEN type, the IgA staining is usually throughout the entire epidermis.
IgG/IgA pemphigus usually shows a lesser degree of pustule formation and more acantholysis than seen in cases of IgA pemphigus. However, there have also been cases with subcorneal pustules and acantholytic mid-epidermal blisters with neutrophilic and eosinophilic spongiosis, and prominent dermal neutrophils with lining up of these cells along the junctional zone. Direct IF shows intercellular staining for IgG and IgA, but another difference from IgA pemphigus is that there are also intercellular deposits of C3.
On immunoelectron microscopy, the deposits have been found along the keratinocyte cell membrane and not confined to the region of the desmosomes. In a study of both types of IgA pemphigus, it was found that the IgA in the sera of the subcorneal pustular dermatosis type reacts with the extracellular domain of desmocollins, whereas in the intraepidermal neutrophilic variant, a nondesmosomal transmembranous protein may be the target antigen.
Because of the level of splitting—subcorneal, often through the granular cell layer— pemphigus foliaceus can be confused with bullous impetigo and subcorneal pustular dermatosis . Acantholysis is usually, but not always, more evident in pemphigus foliaceus, and occasionally, suprabasilar acantholysis can also be observed. Pemphigus foliaceus tends to be less pustular than those two diseases, with occasional exceptions, such as some cases of herpetiform pemphigus. Direct IF can permit a definitive diagnosis because among these diseases, positive intercellular IgG and C3 staining is seen only in pemphigus foliaceus. The same superficial epidermal acantholysis is also characteristic of pemphigus erythematosus, and this finding permits separation from lupus erythematosus, which it can resemble both clinically and, partially, on direct IF because basement membrane zone staining resembling a “lupus band” may also be observed. Paraneoplastic pemphigus shows changes resembling erythema multiforme, but most often there are at least foci of suprabasilar acantholysis, and intercellular staining on direct IF is confirmatory of the diagnosis. The morphological findings of the subcorneal pustular dermatosis –like variant of IgA pemphigus can be virtually identical to those of the disease it resembles. However, classical subcorneal pustular dermatosis clearly lacks positive intercellular IgA deposition on direct IF study or circulating antibodies to epidermal antigens. It appears that to recognize this variant of IgA pemphigus, IF studies are essential.
Infantile acropustulosis is an uncommon pustular dermatosis characterized by recurrent crops of intensely pruritic vesiculopustules on the distal parts of the extremities of infants. The lesions measure 1 to 2 mm in diameter. Its onset is at birth or in the first few months of life, and resolution occurs at 2 or 3 years of age. There is a predilection for black male infants. Infantile acropustulosis is common among internationally adopted children (countries such as Vietnam, Ethiopia, and Russia) from crowded living conditions with a high prevalence of scabies infestation. The cause is unknown, although some cases are said to have followed scabies. Peripheral eosinophilia and atopy have been present in several patients. It has been successfully treated with topical maxacalcitol, an analog of active vitamin D.
Infantile acropustulosis, erythema neonatorum toxicum, and transient neonatal pustular melanosis can be grouped together as the “pustular dermatoses of infancy” (see later) because of their overlapping clinical and histological features. Three other neonatal pustular eruptions are mentioned for completeness.
Transient cephalic neonatal pustulosis is thought to be related to infection with Malassezia sp. (see p. 735 ).
Congenital erosive and vesicular dermatosis, which heals with a characteristic reticulated supple scarring, is of unknown cause. Fewer than 20 cases of this entity have been reported. It is characterized by the presence of erosions and vesicles at birth, usually affecting approximately 75% of the body. The lesions heal with reticulated scarring in weeks to months. A case mimicking junctional epidermolysis bullosa had persistent lesions at 9 years of age. The scarring and erosions of the scalp resemble the Rapp–Hodgkin syndrome, in which there is a mutation in the TP63 gene, but such a mutation was not identified in one case report. Twenty percent of reported cases have been associated with congenital herpes simplex virus infection ; whether this is a causal factor, a mimic, or simply a disease association has not been determined.
An unusual vesiculopustular eruption has been reported in infants with Down syndrome and myeloproliferative disorders. The skin lesions appear mainly on the face either from birth or soon afterwards, especially at sites where pressure has been applied. There are immature myeloid cells in subcorneal spongiotic vesicles. The cells are strongly myeloperoxidase positive on immunohistochemistry. The lesions disappear over weeks to years as the white blood cell count normalizes. A transient myeloproliferative disorder has also been reported in neonates with a normal karyotype and a trisomy 21 in blast cells.
The condition originally reported in infants as “eosinophilic pustular folliculitis” has a predilection for the scalp. It has been renamed “eosinophilic pustulosis of the scalp” on the basis of an interfollicular rather than a follicular infiltrate. It has been claimed that “this disorder has more clinicopathological similarities with acropustulosis of infancy than with Ofuji's disease” (see p. 497 ). Neonatal eosinophilic pustulosis that presents with grouped pustules on the cheek is a similar condition.
In early lesions, there is an intraepidermal pustule containing neutrophils and sometimes varying numbers of eosinophils. This progresses to form a subcorneal pustule. There is a sparse perivascular mixed inflammatory cell infiltrate in the upper dermis.
In congenital erosive and vesicular dermatosis with reticulated supple scarring , the histology has been variable with no consistent pattern. There may be well-defined vesicles in the papillary dermis. The epidermis may be intact, eroded, or ulcerated. Other cases have shown a mixed dermal infiltrate without vesicles or edema with a predominantly neutrophilic infiltrate. Scarring with loss of appendages, an overlying atrophic epidermis, and/or a polymorphous dermal infiltrate is seen in older lesions. In another case, there were many “Civatte's bodies in the dermis suggesting previous keratinocytes’ necrosis.” There was subepidermal cleavage.
Erythema toxicum neonatorum is a common, self-limited entity that appears within the first few days of life as erythematous macules, papules, and pustules, mostly located on the trunk. Delayed onset has been reported. There is no racial predilection. The lesions resolve within a few days, leaving no sequelae. The cause remains elusive. Various inflammatory mediators have been demonstrated in the skin by immunohistochemistry.
There are subcorneal or intraepidermal pustules, filled with eosinophils and related to the orifices of the pilosebaceous follicles. An inflammatory infiltrate composed predominantly of eosinophils is present in the upper dermis in the vicinity of the follicles, and there is some exocytosis of these cells into the epithelium of the involved follicles.
The histological appearances resemble those of eosinophilic pustular folliculitis, but the clinical features of the two conditions differ.
Transient neonatal pustular melanosis is an uncommon condition that presents at birth with pigmented macules, often with a distinct collarette of scale, and vesiculopustules that are clustered beneath the chin, on the forehead, the neck, and the back, and sometimes on the extremities. The vesiculopustules usually resolve after several days, often transforming into pigmented macules. The pigmented lesions persist for several weeks or more and then slowly fade. The cause of the condition is unknown, but there appears to be a predilection for black races. Furthermore, there is an increased incidence of squamous metaplasia in the placentas of affected neonates.
The vesiculopustules are intracorneal or subcorneal collections of neutrophils, admixed with fibrin and a few eosinophils. There may be a mild infiltrate of inflammatory cells around vessels in the upper dermis.
The pigmented macules show increased melanin in the basal and suprabasal keratinocytes but, surprisingly, there is no melanin in the dermis.
Acute generalized exanthematous pustulosis (AGEP), also known as toxic pustuloderma, is an uncommon, rapidly evolving pustular eruption characterized by the development of sterile, miliary pustules on an erythematous background. The lesions may have a targetoid appearance. Lesions localized to the face and neck were present in one case. Fever and a peripheral blood leukocytosis are usually present. The eruption occurs within hours or days of the ingestion of certain drugs and resolves rapidly after cessation of the offending agent. In a small number of cases, a bacterial or viral infection, including enterovirus or cytomegalovirus, has been implicated. A spider bite and echinococcosis have also been associated with this condition. In rare cases, no cause can be demonstrated. The lesions are nonfollicular, in contrast to the follicular pustules that may occur in the anticonvulsant hypersensitivity syndrome. Some patients have a history of underlying psoriasis, ulcerative colitis, or thyroiditis. The lesions of AGEP can usually be distinguished from pustular psoriasis. Cases with overlapping features between AGEP and toxic epidermal necrolysis have been reported.
Numerous drugs have been implicated in the cause, particularly antibiotics of the β-lactam, cephalosporin, and macrolide types ( Table 7.2 ). Cutaneous patch testing, using the suspected drug, may be used to confirm the diagnosis. In one case, AGEP was precipitated by the patch test carried out to determine the drug eliciting an initial AGEP eruption.
| Acetaminophen | Ciprofloxacin | Gemcitabine | Methylphenidate | Quetiapine |
| Acyclovir | Clemastine | Gentamicin | Metronidazole | Quinidine |
| Allopurinol | Clindamycin | Herbal and traditional remedies (lacquer chicken, velvet antler) | Morphine | Radiocontrast media |
| Allylisopropylacetylurea | Clopidogrel | Hydrochlorothiazide | Nifedipine | Recreational drugs |
| Amoxicillin (with and without clavulanic acid) | Cytarabine | Hydroxyzine | Nimesulide | Roxithromycin |
| Amphotericin B | Dalteparin | Ibuprofen | Nystatin | Sennoside |
| Antimalarials | Daptomycin | Icodextrin | Olanzapine | Simvastatin |
| Azathioprine | Diltiazem | Imipenem | Paracetamol with bromhexine | Sorafenib |
| Bamifylline | Diphenhydramine (topical) | Immunoglobulin, intravenous | Paroxetine | Teicoplanin |
| Carbamazepine | Docetaxel | Iodixanol | Penicillin | Telavancin |
| Cefepime | Doxycycline | Isotretinoin | Pentoxifylline | Terbinafine |
| Ceftriaxone | Enalapril | Itraconazole | Phenytoin | Thalidomide |
| Cefuroxime | Erlotinib | Ketoconazole (oral) | Pholcodine | Thallium |
| Celecoxib | Erythromycin | Labetalol | Piperacillin–tazobactam | Ticlopidine |
| Cephalexin | Etanercept | Lansoprazole | Prednisolone | Tocilizumab |
| Cetirizine | Etodolac | Lapatinib | Pristinamycin | Trimethoprim–sulfamethoxazole |
| Chemotherapy drugs | Famotidine | Levetiracetam | Propafenone | Vancomycin |
| Chloramphenicol | Flucloxacillin | Levofloxacin | Prostaglandin E1 (intracavernous) | |
| Chromium picolinate | Furosemide | Mercury | Pseudoephedrine | |
| Cimetidine | Galantamine | Meropenem | ||
| Methimazole |
It has been suggested that patients who develop this eruption have an underlying tendency to develop a pattern of immune dysregulation characterized by a T helper 1 (Th1) cytokine pattern. Th17 cells and their cytokine, IL-22, have been found to be elevated in the peripheral blood of patients with AGEP. The latter, in combination with IL-17, may stimulate IL-8 production by keratinocytes, which in turn could explain the accumulation of neutrophils within the epidermis in this disease.
The treatment of AGEP is conservative: stop any drugs that may have triggered the eruption and treat symptomatically with emollients and antipyretics while awaiting spontaneous resolution. This occurs with superficial desquamation over 1 or 2 weeks. Topical corticosteroids have also been used.
The usual picture is a subcorneal or superficial intraepidermal pustule with mild spongiform pustulation at the margins ( Fig. 7.5 ). This latter change is never as prominent as the spongiform pustules seen in pustular psoriasis. The pustules often contain eosinophils in addition to neutrophils. There is usually some exocytosis of neutrophils adjacent to the pustules. Scattered apoptotic keratinocytes are often present.
The papillary dermis is usually edematous, and there is a heavy mixed inflammatory cell infiltrate in the upper dermis. Eosinophils are often present, a distinguishing feature from pustular psoriasis. In a small number of cases, a leukocytoclastic vasculitis is present; this was found in only one case out of a large series of 102 hospitalized patients. Less commonly, there is subepidermal pustulation that may be in continuity with the intraepidermal pustules. Similar cases have been reported in the past as acute generalized pustulosis or included with the pustular vasculitides. A case clinically presenting as AGEP, associated with clindamycin and levofloxacin therapy, showed subcorneal pustules but also an atypical mononucleated dermal infiltrate composed of CD3 + and CD30 + cells, consistent with a lymphomatoid drug eruption. The eruption resolved within 1 week of discontinuing both drugs.
Acute generalized pustulosis, or pustulosis acuta generalisata, an eruption caused by upper respiratory streptococcal infection, can also present with acute onset of widespread lesions and may have eosinophils in the dermal infiltrate. However, the intraepidermal pustules tend to be larger, and there are more pronounced dermal infiltrates and more obvious leukocytoclastic vasculitis than would be expected in AGEP. The pustules of pustular psoriasis are usually larger and show more prominent spongiform pustulation, whereas the degrees of acanthosis in AGEP are generally less than would be expected in fully developed plaque-type psoriasis. An increase in Ki-67-positive keratinocytes is seen in both pustular psoriasis and AGEP, with no significant difference in numbers or distribution of these cells. In comparing these two dermatoses, AGEP often has eosinophils in the pustules or in the dermis, necrotic keratinocytes, a neutrophil-rich but mixed interstitial and mid-dermal infiltrate, and a lack of tortuous dilated blood vessels. Despite an occasional clinical resemblance to erythema multiforme , pustules are generally not observed in the latter disease, whereas apoptosis is usually more extensive than in lesions of AGEP. As noted previously, a resemblance has been noted between AGEP and toxic epidermal necrolysis. This has been mostly based on clinical features. Although one study reported a potential histopathological resemblance as well, the photomicrographs of that case show an intraepidermal pustule with necrosis of the overlying epidermis and adjacent spongiosis—features more in keeping with AGEP—whereas other potentially differentiating features, such as the presence or absence of eosinophils, were not described. Infection with Candida or a dermatophyte could show overlapping features, but periodic acid–Schiff (PAS) or silver methenamine stains would be positive for microorganisms in those disorders.
The condition known as miliaria crystallina is associated with small, 1- to 2-mm vesicles that rupture easily (see p. 126 ).
The vesicle forms within or directly beneath the stratum corneum. It is centered on the acrosyringium.
The term intraepidermal blister refers to the formation of lesions within the malpighian layers; it does not include those vesiculobullous diseases in which the split occurs beneath the stratum corneum or in a suprabasilar position. Note, however, that biopsies from some of the diseases listed as forming subcorneal or suprabasilar blisters may sometimes show splitting within the malpighian layers. This is particularly likely in lesions of some days’ duration in which regeneration of the epidermis may alter the level of the split.
Intraepidermal blisters usually form as the outcome of spongiosis or ballooning degeneration. The primary acantholytic diseases usually form blisters that are subcorneal or suprabasilar in position, before regeneration occurs. Most of the intraepidermal blistering diseases are discussed elsewhere but, with the exception of hydroa vacciniforme (see p. 660 ), they are mentioned here.
Although most of the diseases that produce the spongiotic reaction pattern (see Chapter 6 ) can sometimes be associated with clinically visible vesicles and even bullae, the ones most often associated with blisters are allergic contact dermatitis, nummular dermatitis, pompholyx, polymorphic light eruption (vesicular type), insect bite reactions, incontinentia pigmenti (first stage), and miliaria rubra. The presence of spongiosis adjacent to the vesicle or elsewhere in the biopsy is the clue to this group of blistering diseases. Eosinophils are prominent in the infiltrate in insect bite reactions and incontinentia pigmenti.
The bullae that sometimes form in acrodermatitis enteropathica (see p. 601 ) are intraepidermal in location. Intraepidermal or subcorneal clefting can also occur in the glucagonoma syndrome (see p. 602 ).
Tense bullae have been reported on the feet of a male with malignancy-associated acrokeratosis paraneoplastica (Bazex's syndrome). The lesions were characterized by apoptotic basal keratinocytes and microvesicles. No mention was made of their location in the epidermis.
Eczematous and vesiculobullous variants of mycosis fungoides have been reported. They are exceedingly rare. The prognosis of this variant is often poor, but in a recently reported case there was a 30-year history of the mycosis fungoides and a 25-year history of blisters. Subepidermal blisters may also occur.
Palmoplantar pustulosis commences as a spongiotic vesicle, but pustulation rapidly ensues (see the next section).
Palmoplantar pustulosis is a chronic inflammatory skin disorder in which there are erythematous, scaly plaques with recurrent sterile pustules, symmetrically distributed on the palms and soles. Initially, only a palm or a sole may be involved. The Koebner phenomenon is a rare manifestation. Onset of the disease is usually between the ages of 40 and 60 years. Women are predominantly affected. Palmoplantar pustulosis is sometimes associated with a focus of infection somewhere in the body, although elimination of the infectious process usually has no influence on the course of the disease, which is usually protracted and somewhat unpredictable. A case responding to treatment of the accompanying severe periodontitis has been reported. Another anecdotal case responded to treatment of her Helicobacter pylori infection.
Psoriasis is present in at least 6% of cases; some studies have shown a much greater incidence. However, unlike psoriasis, there are no clear associations with any particular HLA type. There is also a difference in the surface receptors on neutrophils in the two conditions. Both psoriasis and palmoplantar pustulosis may be precipitated by lithium. Both may be precipitated by the use of TNF-α inhibitors used in the treatment of various diseases, including adalimumab, golimumab, and certolizumab pegol. Infliximab can also worsen the disease. This may be related to the role of TNF-α in normal eccrine sweat ducts. Rituximab and granulocyte colony-stimulating factor can also exacerbate the disease. Furthermore, a seronegative spondyloarthropathy is sometimes present. There is also an increased incidence of osteoarthritis. An association with Sweet's syndrome and an association with stress have been reported.
Other clinical findings in palmoplantar pustulosis include the presence of sternocostoclavicular ossification in 10% of cases ; lytic, but sterile, bone lesions ; and an increased incidence of autoantibodies to thyroid antigens and to gliadin. However, no increase in antigliadin antibodies was found in a German series of patients with this disease compared with controls.
Palmoplantar pustulosis has been regarded as a form of psoriasis, a bacterid (an inflammatory reaction at a site remote from that of a bacterial infection presumed to be of pathogenetic significance), and a distinct clinicopathological entity, probably with an immunological pathogenesis. The last of these theories is supported by the finding of increased numbers of Langerhans cells in active lesions and increased levels of proinflammatory cytokines such as IL-6, TNF-α, and IFN-α. The IL-10 cytokine family (IL-19, IL-20, and IL-24) also plays a role in epidermal function and inflammation. They are controlled by a gene cluster on chromosome 1q31–q32. Gene polymorphisms in this cluster have been demonstrated in palmoplantar pustulosis. There is evidence that the very rare acute form of palmoplantar pustulosis, which may progress into the chronic form, is a bacterid with an associated vasculitis. It has also been linked to smoking and metal (jewelry) allergy. Up to 95% of patients are smokers at the time of onset of the disease. Approximately 40% of patients have antibodies to nicotinic acetylcholine receptors. On this basis, it has been postulated that palmoplantar pustulosis is an autoimmune disease precipitated by smoking. It has been suggested that the acrosyringium is the target for the inflammation. Nicotine is excreted in the eccrine palmar duct. In contrast to psoriasis, Langerhans cells, IL-17, regulatory T cells, and CD11a-positive cells are found close to or near the acrosyringium in palmoplantar pustulosis ; furthermore, gross cystic disease fluid protein–15 and epithelial membrane antigen, both markers of acrosyringium, line the intraepidermal vesicles in this disease. In summary, it appears that there is no single factor that causes palmoplantar pustulosis. It may be precipitated in patients, some of whom have a heightened production of proinflammatory cytokines, by a diverse group of antigens including nicotine, infectious agents, metals, and TNF-α antagonists. Some cases are cured by the cessation of smoking alone. A gluten-free diet has resulted in improvement in patients with antigliadin antibodies, regardless of the degree of mucosal abnormalities. Other cases require topical corticosteroids, systemic retinoids, PUVA, cyclosporine, or methotrexate. Among the agents that have been used and/or show promise are etanercept and adalimumab (a paradox similar to psoriasis where it can both treat and precipitate the disease); ustekinumab ; tocilizumab ; cefcapene pivoxil hydrochloride ; tofacitinib ; the anti-IL-17 agents secukinumab, ixekizumab, and brodalumab ; phosphodiesterase type 4 inhibitors ; apremilast ; and potassium iodide and tetracycline. Other procedures that have been used successfully in selected circumstances include UVA-1 phototherapy, narrowband UVB phototherapy, tonsillectomy, and granulocyte and monocyte adsorption apheresis.
The earliest lesion is a spongiotic vesicle in the lower malpighian layer that contains mononuclear cells and some neutrophils. This progresses to a unilocular, well-delimited pustule within the epidermis and extending upwards to the undersurface of the stratum corneum ( Fig. 7.6 ). There may be overlying, focal parakeratosis. In the dermis, a mixed perivascular and diffuse infiltrate of inflammatory cells is present.
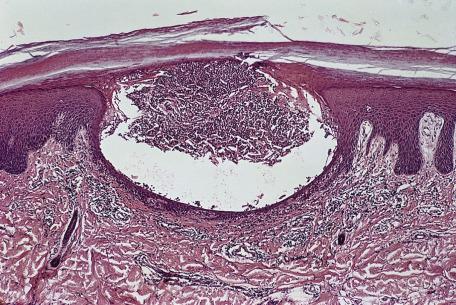
Controversy exists as to whether it is possible to differentiate pustular psoriasis and palmoplantar pustulosis on histopathological grounds. Spongiform pustulation is often present at the upper margins of the pustule in palmoplantar pustulosis, but the focus is usually much smaller than that seen in pustular psoriasis. Sometimes there is spongiosis without associated pustulation; this feature is not present in pustular psoriasis. Eosinophils may be present in palmoplantar pustulosis, a finding not usually seen in pustular psoriasis.
Immunoreactants are present in the stratum corneum in some cases of palmoplantar pustulosis. Regarding the histopathological differentiation from pompholyx, Yoon et al. found that loss of the granular cell layer, thinning of suprapapillary plates, eosinophils in pustules or vesicles, tortuosity of capillaries, capillaries touching the undersurface of the epidermis, and extravasated erythrocytes were statistically significant features of palmoplantar pustulosis, and psoriasiform epidermal features also tended to favor palmoplantar pustulosis. On the other hand, pompholyx tended to have multiple foci of parakeratosis, irregular acanthosis, and thinning of rete ridges. These two disorders also show a different pattern of epithelial membrane antigen staining within the epidermis; both show staining along the edge of intraepidermal vesicles, but there is little staining of the surrounding epidermis in palmoplantar pustulosis, whereas there is strong and diffuse staining of neighboring keratinocytes in pompholyx.
The rare disease amicrobial pustulosis associated with autoimmune diseases, known by the acronym APAD, is a chronic relapsing eruption involving predominantly the main cutaneous flexures, external auditory canal, and scalp. It has been reported in association with various autoimmune diseases. Most, if not all, cases have been in women, usually of reproductive age. In one case, there was associated IgA nephropathy and Sjögren's syndrome. Some improvement has occurred after oral zinc supplementation.
There are high intraepidermal spongiform pustules in an acanthotic epidermis. There is usually overlying parakeratosis containing neutrophil debris. Exocytosis of neutrophils into the epidermis is present. A mixed perivascular infiltrate of neutrophils and lymphocytes, with nuclear dust, is present in the upper dermis. There is marked overexpression of Bcl-2 in lesional skin.
Erosive pustular dermatosis of the leg is another amicrobial pustulosis. It is also considered in Chapter 24 with erosive pustular dermatosis of the scalp, but because bacterial growth in that condition is almost certainly a secondary event, it could also have been included here with the other amicrobial pustuloses, as could the autoinflammatory (periodic fever) syndromes (see p. 269 ).
Erosive pustular dermatosis of the leg is characterized by crusted pustules and erosions forming erythematous plaques on the lower legs. There is usually underlying chronic venous insufficiency, stasis dermatitis, and atrophy of the skin. It runs a chronic course.
Topical corticosteroids and tacrolimus have produced improvement in the lesions.
Intact lesions show spongiotic pustules at a high level in the epidermis. They eventually become subcorneal or intracorneal in location. There is usually some acanthosis and a mixed dermal infiltrate of neutrophils, lymphocytes, and a few plasma cells. Erosions are common; they have a heavy mixed inflammatory cell infiltrate in the base.
Intraepidermal vesicles are seen in herpes simplex; herpes zoster; varicella; hand, foot, and mouth disease; and some cases of milker's nodule and orf. In the case of herpes simplex, herpes zoster, and varicella, there is ballooning degeneration of keratinocytes with secondary acantholysis of cells. Multinucleate keratinocytes, some with intranuclear inclusion bodies, may also be seen. In hand, foot, and mouth disease, there are both spongiosis and intracellular edema, whereas in milker's nodule and orf there may be pronounced intracellular edema with pallor and degeneration of keratinocytes in the upper layers of the epidermis. A severe, diffuse blistering eruption has been reported in two patients with orf. Viral DNA was detected in one case in the inciting orf lesion, but not in blistered skin, ruling out disseminated orf infection as a cause of the blisters. Both cases were characterized by subepidermal blisters with a mixed inflammatory cell infiltrate in the base that included neutrophils and eosinophils. IgG and C3 were deposited in a linear manner at the basement membrane zone of perilesional skin, and indirect IF revealed circulating anti–basement membrane IgG that bound to the dermal side of salt-split skin. Milker's nodule and orf are discussed in Chapter 27 .
In the localized type of epidermolysis bullosa simplex, formerly known as the Weber–Cockayne type (see p. 179 ), the split is usually in the mid or upper layers of the epidermis, although in induced blisters the split develops in the basal layer. An occasional dyskeratotic cell may be present in the epidermis.
Friction blisters are produced at sites where the epidermis is thick and firmly attached to the underlying dermis, as on the palms, soles, heels, and the back of the fingers.
The blister usually forms just beneath the stratum granulosum. The keratinocytes in the base of the blister show variable edema and pallor and even degenerative changes.
The suprabasilar blistering diseases—pemphigus vulgaris, pemphigus vegetans, Hailey–Hailey disease (familial benign chronic pemphigus), Darier's disease, Grover's disease (transient acantholytic dermatosis), and acantholytic solar keratosis—all result from acantholysis. Suprabasal acantholysis has been reported in one case of necrolytic migratory erythema. It may sometimes be seen in paraneoplastic pemphigus. In blisters of some days’ duration, the split may be present at a higher level within the epidermis as a result of epidermal growth.
Pemphigus vulgaris (OMIM 169610 ) is a rare vesiculobullous condition that accounts for approximately 80% of all cases of pemphigus. Its incidence is approximately 0.1–1.0 per 100,000/year. There is a slight female predominance in most countries, but not in others. One study shows that, compared with women, men are more likely to have disease onset before the age of 40, to have increased cutaneous involvement, and to show greater coexpression of anti-Dsg1 and Dsg3 antibodies, whereas women have mucosal predominance and a stronger personal and family history of autoimmunity. Pemphigus vulgaris is characterized by the production of autoantibodies to the epithelial adhesion protein desmoglein 3 (dsg3). The initial presentation is often with oral blisters, ulcers, and erosions, which are followed within weeks to months by the development of cutaneous lesions. Nail involvement is relatively rare. Skin lesions take the form of flaccid blisters on a normal or erythematous base; there is a predilection for the trunk, groins, axillae, scalp, face, and pressure points. Penile involvement is rare, and rarer still is its presentation as the first manifestation of the disease. Sometimes lesions are localized to one body site, such as the nose and cheeks. The blisters break easily, giving eroded and crusted areas. Application of pressure to a blister leads to its extension (Nikolsky's sign or the Asboe–Hansen sign). Burning and itching may be present. The lesions generally heal without scarring.
In addition to oral lesions, which eventually develop in 75% to 90% of cases, other mucosal surfaces may be involved. These include the conjunctiva, pharynx and larynx, and, rarely, the esophagus, urethra, anorectum, vulva, vagina, and cervix. In a study from Japan, 75% of patients presented with involvement of the oral mucosa. Mucosal lesions are less common in some countries than in others. The reason for this is not known.
Rare clinical presentations include localized lesions; bilateral foot ulcers ; lesions limited to the surgical area after mastectomy ; localization to a melanocytic nevus ; generalization of oral disease after varicella infection ; the development of lesions in childhood, in siblings, or in a familial setting ; the development of transient blisters in a neonate whose mother has pemphigus or is asymptomatic, as as result of the transplacental passage of dsg3 antibodies from the mother ; the development of nodular, vegetative, or acanthosis nigricans–like lesions ; and the coexistence of pemphigus vulgaris with bullous pemphigoid. Rarely, the clinical and histological features of pemphigus vulgaris may change to those of pemphigus foliaceus; the antibody profile also changes. Cases with overlap features also occur. Several cases of endemic pemphigus vulgaris have been reported from regions with endemic pemphigus foliaceus. Internal cancer, vulvar squamous cell carcinoma, multiple myeloma, Castleman's disease, 1p36 deletion syndrome, thymoma, myasthenia gravis, minimal change nephropathy, localized scleroderma, Graves’ disease, hyperprolactinemia, silicosis, SLE, mixed connective tissue disease, dermatomyositis, antiphospholipid antibodies, oral submucosal fibrosis, and oral herpes simplex or HIV infection are rare clinical associations. There is an increased susceptibility to the development of autoimmune diseases in the family members of patients with pemphigus vulgaris. Several HLA types (class II) are prevalent in patients with pemphigus vulgaris. The HLA haplotypes DRB1*0402, DQB1*0302, DRB1*1401, and DQB1*0503 appear to confer a significant disease risk across a wide range of populations. Another genotype, HLA-E*0103X, has also been found to be a marker for genetic risk in pemphigus vulgaris. Polymorphisms in the PTPN22 gene, a candidate gene for autoimmune diseases, have not been detected in pemphigus vulgaris.
The prognosis has improved considerably in recent years, although the mortality is still 5% to 19%; deaths often result from infections complicating corticosteroid therapy and biochemical abnormalities associated with extensive disease. In one study, complete and long-lasting remissions were induced in 25%, 50%, and 75% of patients 2, 5, and 10 years, respectively, after diagnosis.
The antibodies in pemphigus vulgaris are directed against dsg3, a 130-kDa polypeptide that, like the pemphigus foliaceus antigen (dsg1), is a member of the desmoglein subfamily of the cadherin supergene family. Genetic variations in DSG3 , the gene controlling dsg3 on chromosome 18q12, appear to be an additive risk factor predisposing to pemphigus vulgaris. Dsg3 is localized to the desmosomes of keratinocytes, particularly the extracellular domain. Epitope mapping suggests that a segment containing amino-terminal residues 1–161 of dsg3 contains the critical antigens recognized by the autoantibodies in pemphigus vulgaris sera. This segment is considered to include structures essential for cell–cell adhesion. The cytoplasmic “tail” of the pemphigus vulgaris antigen, like other desmogleins, binds to the plakoglobin inside keratinocytes. Plakoglobin delocalization from the nucleus may play a role in the production of acantholysis (discussed later). More than 50% of sera from patients with pemphigus vulgaris also contain autoantibodies against dsg1. These antibodies also appear to be pathogenic. There is a shift over time in the profile of antidesmoglein antibodies in pemphigus vulgaris. The antibodies against both dsg1 and dsg3 have predominant IgG4 subclass specificity. Antibodies in the IgA and IgE class have also been detected. The majority of patients with mucosa-dominant disease have antibodies only against dsg3, but a significant number also have antibodies to dsg1. In fact, there is evidence that anti-Dsg1 autoantibodies may be better predictors of disease progression, and therefore motoring of anti-Dsg1 levels may be valuable for this purpose. The development of antibodies to dsg1 in some patients with dsg3-related mucosal disease is sometimes accompanied by changes in the clinical expression of the disease, such as the development of generalized cutaneous lesions. Conversely, lesions limited to the skin may occur in the presence of predominant dsg1 autoantibodies and weaker levels of dsg3 autoantibodies, but antibody levels are not the complete explanation for this phenomenon. Antidesmoplakin antibodies, which are usually found in paraneoplastic pemphigus, have also been reported in patients with mucosal dominant pemphigus vulgaris, who also had dsg3 autoantibodies. Although it has been thought that typical pemphigus does not have autoantibodies to the desmocollins, both desmocollins 2 and 3 have been detected in mucosal-dominant pemphigus vulgaris with severe symptoms, and studies by Mao et al. have shown that at least a subset of patients with identical clinical presentations have desmocollin 3 as the pathogenic autoantigen. Among other findings, IgG from these patients incubated with human keratinocytes causes loss of intercellular adhesion, and prior adsorption with recombinant desmocollin 3 prevents this effect. In pemphigus vulgaris, the antibodies produced by circulating B cells tend to bind preferentially to desmosomes in the lower epidermis, reflecting differences in the relative expression of the different cadherins in different levels of the epidermis. This antibody binding may be followed by internalization of some desmosomes. Antibodies will only cause cell separation (acantholysis) where the antigen is the principal adhesion molecule and others are unable to compensate. Cholinergic receptors on keratinocytes also play a role in cell adhesion. Antibodies to the acetylcholine receptors α-9 acetylcholine receptor and pemphaxin are present in humans with pemphigus vulgaris. Autoantibodies to these receptors can induce clinical features resembling pemphigus in dsg3-deficient knockout mice. Cholinergic agonists ameliorate acantholysis in mice. Antibody levels to the acetylcholine receptor are somewhat elevated in pemphigus vulgaris patients and appear to correlate with disease activity. There is also upregulation of P-cadherin expression in lesional skin. E-cadherin autoantibodies have also been found in the sera of patients. Another protein involved in cellular adhesion is focal adhesion kinase (p125 fak ). Its upregulation in lesional skin may be a reactive or reparative response to acantholysis. Using a passive transfer mouse model of pemphigus vulgaris, Espana et al. found that neural nitric oxide synthase (nNOS) may be involved in epidermal growth factor (EGF) receptor–mediated acantholysis via upregulation of Rous sarcoma kinase (Src), mammalian target of rapamycin (mTOR), and focal adhesion kinase, with eventual upregulation of caspase-9 and caspase-3. The desmosomal damage and consequent acantholysis may follow the activation of complement or local stimulation of the plasminogen–plasmin system, independent of complement (see later). However, studies using plasminogen activator knockout mice suggest that this system is not necessary for blister formation. Experimental evidence suggests that antibodies may eventually generate desmosomes lacking dsg3, resulting in acantholysis. Other mechanisms involved in acantholysis include the release of keratinocyte-derived cytokines such as IL-1 and TNF-α. In addition, the role of flotillins in this disease has recently been explored, and there is evidence that loss of flotillin expression is associated with weakened desmosomal adhesion and reduced expression of Dsg3.
These traditional views that acantholysis results from a defect in desmosomes caused by antibody/complement deposition have been challenged by several groups. The binding of antibodies leads to intracellular events that precede the acantholysis. Bystryn and Grando believe that keratinocytes separate because cytoskeletal collapse results in the inability of desmosomes to hold cells together any longer, not because of a defect in the desmosomes. The shrinkage is limited to basal cells because they are less rigid and shrink easily. It has been suggested subsequently that this cytoskeletal collapse is due to keratin intermediate filament retraction linked to plakoglobin-dependent signaling or nuclear delocalization. The entire process of antibody binding to keratinocytes, keratin retraction, cell shrinkage, and eventual acantholysis occurs very rapidly. There may be signaling events other than plakoglobin, such as heat shock protein, that trigger this cytoskeletal collapse. The importance of intracellular signaling cascades in producing blisters in pemphigus vulgaris has been confirmed in an animal model. Other studies indicate that reorganization of the actin cytoskeleton is an important factor in pemphigus vulgaris IgG–related dissociation among keratinocytes.
Activated mononuclear cells are present in lesional skin and may contribute to the pathogenesis. Autoreactive T lymphocytes that respond to epitopes located on dsg3 have been identified, confirming a role for cell-mediated as well as humoral responses in pemphigus vulgaris. This Th2 response directs autoantibody production. T cells that react against dsg1 are sometimes present. Various inflammatory mediators have been isolated from blister fluid, and these presumably have a role in eliciting the accompanying inflammatory response. Osteopontin may be one of the cytokines that drives this immune response. What stimulates the formation of antibodies is unknown, although minor trauma, surgery, ionizing radiation, thermal burns, PUVA therapy, viral infection, and exposure to chemicals have occasionally been documented before the onset of the disease. Cutaneous lesions limited to a radiation portal have been reported. Antidesmoglein autoantibodies are sometimes found in patients with no bullous disease. For example, they have been found in some patients with silicosis and in relatives of patients with pemphigus vulgaris. The term contact pemphigus has been used for cases that have followed contact with chemicals, usually pesticides. Pemphigus foliaceus has been found in dogs that were treated with the topical flea preventative metaflumizone–amitraz. Contact pemphigus has also followed the application of imiquimod. Smoking seems to have a protective function. Drugs producing pemphigus vulgaris are listed in Table 7.3 . Drugs with thiol groups have a particular capacity to precipitate or exacerbate pemphigus. Despite the long list of drugs, a recent study concluded that drugs are a rare precipitating factor. Incidental acantholysis has been found in two biopsies from a patient on enalapril who had no clinical features of pemphigus vulgaris. Carbamazepine can result in the production of antibodies to dsg1 and dsg3 without producing lesions.
| Subcorneal pustular dermatosis–like: amoxicillin, cephalosporins, dapsone, diltiazem, gefitinib, isoniazid, paclitaxel, quinidine |
| Acute generalized exanthematous pustulosis: see Table 7.2 for complete listing |
| Pemphigus foliaceus and erythematosus: aspirin, bucillamine, captopril, ceftazidine, cephalosporins, enalapril, fosinopril, gold, heroin, imiquimod, levodopa, methimazole, penicillamine, propanolol, pyritinol, ramipril, rifampin (rifampicin), tetanus vaccination, thiol-containing drugs, thiopronine |
| Pemphigus vulgaris and vegetans: amoxicillin/clavulanic acid, ampicillin, anthrax vaccine, captopril, cilazapril, ciprofloxacin, cocaine snorting, diclofenac, dipyrone, enalapril, fosinopril, heroin, hydroxychloroquine, imiquimod (contact pemphigus), influenza immunization, interleukin-2, ketoprofen, nifedipine, norfloxacin, penicillamine, penicillin, phenols, quinapril, rifampin (rifampicin), tannins, tetanus and diphtheria vaccine, thiol-containing foods, typhoid immunization |
| Subepidermal (cell poor)—pseudoporphyria: acitretin, amiodarone, ampicillin-sulbactam, aspirin, β-lactams, bumetanide, cefepime (?), celecoxib, chlorthalidone, ciprofloxacin, cola consumption, contraceptive pill, cyclosporine (ciclosporin), etretinate, 5-fluorouracil, flutamide, furosemide (frusemide), hemodialysis, imatinib, isotretinoin, ketoprofen, mefenamic acid, metformin, nabumetone, nalidixic acid, naproxen, oxaprozin, pravastatin, pyridoxine, rofecoxib, sulfonamides, tanning beds, tetracyclines, UV-B phototherapy, voriconazole |
| Subepidermal (lymphocytes ± eosinophils)—erythema multiforme/TEN: acarbose, allopurinol, amifostine, aminopenicillins, bezafibrate, cannabis, carbamazepine, ceftazidime, celecoxib, chloroquine, cimetidine, ciprofloxacin, clindamycin, clobazam, clonazepam, cocaine, cyclophosphamide, cytosine arabinoside, diacerein, doxycycline, ethambutol, etretinate, famotidine, gemeprost, griseofulvin, imiquamod cream, indapamide, indomethacin, isoxicam, lamotrigine, latanoprost eye drops, mefloquine, methotrexate, mifepristone, nevirapine, nitrogen mustard, nystatin, oxaprozin, phenylbutazone, phenytoin, piroxicam, pravastatin, ranitidine, ritodrine, sertraline, simvastatin, sorafenib, sulfamethoxazole, suramin, terbinafine, theophylline, ticlopidine, trichloroethylene, trimethoprim, valproic acid, vancomycin |
| Subepidermal (eosinophils)—bullous pemphigoid: 5-aminosalicylic acid, amlodipine, ampicillin, antipsychotic drugs, aspirin, bumetanide, captopril, cephalexin, chloroquine, ciprofloxacin, dipeptidyl peptidase 4 inhibitors, doxazosin, efalizumab, enalapril, enoxaparin, erlotinib, etanercept, fluorouracil (topical), fluoxetine, furosemide (frusemide), gabapentin, homeopathy regimen, ibuprofen, influenza immunization, iodine (intravenous), levobunolol (ophthalmic), levofloxacin, lisinopril, losartan, metformin and gliptins (dipeptidyl peptidase IV inhibitors), neuroleptics, nifedipine, novoscabin, PD-1/PD-L1 inhibitors, penicillamine, penicillin and derivatives, phenacetin, serratiopeptidase, sulfasalazine, tetanus, tetracoq immunization |
| Subepidermal (neutrophils)—linear IgA bullous dermatosis: acetaminophen (paracetamol), amiodarone, amlodipine, amoxicillin, ampicillin/sulbactam, atorvastatin, captopril, carbamazepine, cefamandole, ceftriaxone, cefuroxime (on discontinuation), diclofenac, donepezil, furosemide (frusemide), gemfibrozil (bullous dermatosis of childhood), glibenclamide, human papillomavirus vaccination, interleukin-2, lithium carbonate, metronidazole, NSAIDs, naproxen, penicillin, phenytoin, piroxicam, sodium hypochlorite (contact), somatostatin, trimethoprim-sulfamethoxazole, ustekinumab, vancomycin, verapamil |
| Subepidermal (scarring)—mucous membrane pemphigoid: atenolol, azathioprine, clonidine, practolol |
| Subepidermal (vasculitis): some of the drugs producing a leukocytoclastic vasculitis (see Chapter 9 ) may result in blister formation, but this is not a consistent feature of any particular drug |
| Subepidermal (necrosis)—drug overdose-related bullae: amitryptyline, barbiturates, carbamazepine, clobazam, diazepam, heroin, imipramine, methadone, morphine |
The role of various foods in the precipitation of pemphigus has been studied. Possible candidates include thiol-containing foods (garlic, onion, leek, and shallots), thiocyanates (mustards), phenols (artificial sweeteners, preservatives, and colorings), and tannins (certain fruits).
The finding of HHV-8 DNA sequences in lesional skin may indicate trophism for pemphigus lesions rather than a causal role.
The desmoglein ELISA was positive in all 29 cases of pemphigus vulgaris in one series. It was superior to indirect IF as a diagnostic tool. Other studies have confirmed the reliability of the ELISA test. It may be used to monitor disease activity because the titer of circulating antibodies tends to parallel this activity. Nevertheless, circulating anti-Dsg autoantibodies are still found in some pemphigus patients in remission, particularly if their titers during the active phases of the disease were high.
Now that a consensus statement has been produced defining aspects of the disease, including severity and end points, comparative treatment studies can be undertaken. More trials are needed to assess some of the newer therapies ; several recent reviews have been published. Therapies that have been used include systemic corticosteroids, usually in a high dose; a pulse protocol using intravenous betamethasone and oral prednisone ; cyclophosphamide ; azathioprine; prednisolone alone or combined with azathioprine, cyclophosphamide, or mycophenolate mofetil ; dapsone ; high-dose intravenous immunoglobulin, alone or as a steroid-sparing agent ; plasmapheresis; protein A immunoadsorption combined with other therapies ; gold ; rituximab ; etanercept ; thalidomide ; topical tacrolimus combined with cyclophosphamide and prednisolone ; and EGF in 0.1% silver sulfadiazine cream.
Established lesions of pemphigus vulgaris are suprabasal bullae with acantholysis ( Fig. 7.7 ). The clefting may extend down adnexal structures. The basal cells lose their intercellular bridges, but they remain attached to the dermis, giving a “tombstone appearance.” The blister cavity usually contains a few acantholytic cells that often show degenerative changes. Occasionally, a few eosinophils or neutrophils are present in the cavity.

The earliest changes in pemphigus vulgaris consist of edema and disappearance of the intercellular bridges of keratinocytes in the lower epidermis ( Fig. 7.8 ). This leads to acantholysis and subsequent suprabasal blisters. These changes may also occur at an early stage in hair follicles. Eosinophilic spongiosis and subepidermal splitting are two rare presentations of pemphigus vulgaris ( see Fig. 7.7C ). Vegetative lesions show epidermal hyperplasia but a paucity of eosinophils, in contrast to pemphigus vegetans. Acanthomas resembling seborrheic keratosis may form at the sites of previous blisters.
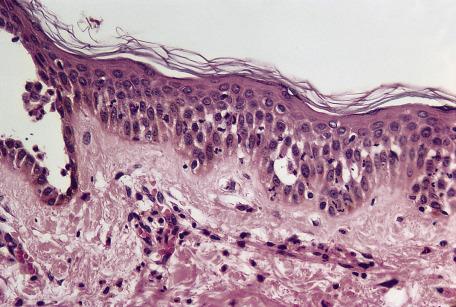
Dermal changes are of little significance. There is usually a mild, superficial, mixed inflammatory cell infiltrate that usually includes scattered eosinophils. There are no histological features that differentiate drug-associated pemphigus vulgaris from idiopathic cases.
Immunostaining with the monoclonal antibody 32-2B, which detects desmogleins 1 and 3, can be used in many cases to distinguish drug-induced pemphigus from idiopathic pemphigus. Whereas a patchy pattern of staining was observed in idiopathic pemphigus, the staining was more often normal in drug-induced cases, although patchy staining was found in 30% of cases (70% sensitivity).
Direct IF usually demonstrates IgG in the intercellular regions of the epidermis in and around the affected parts of the skin or mucous membrane ( Fig. 7.9 ). IgG1 and IgG4 are the subclasses of IgG found most commonly in patients with active lesions ; C3, IgM, and IgA are present less often. Direct IF can also be performed on plucked anagen hair. Intercellular staining is seen between cells of the outer root sheath. Patients in clinical remission, who have positive direct IF on a skin biopsy, are more likely to relapse than those with negative IF findings. Circulating intercellular antibodies are present in 80% to 90% of patients with pemphigus vulgaris, although they may be absent in early cases. Their demonstration depends to some extent on the substrate used; monkey esophagus gives a higher yield of positive results than guinea pig esophagus or human skin. Pemphigus-like antibodies have also been reported in a wide range of inflammatory dermatoses, as well as after burns.
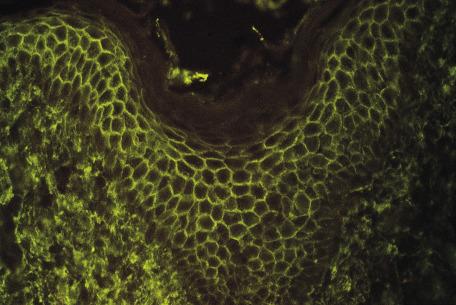
Cytology preparations obtained from smears of bullae show acantholytic cells with ameboid cytoplasmic projections, as well as eosinophils, basophils, and lymphocytes, some of which have abnormal nuclear contours. Older bullae possess dysplastic epithelial cells with high nuclear-to-cytoplasmic ratios and prominent nucleoli. Acantholytic cells can also be appreciated with reflectance confocal microscopy.
Using a technique known as structured illumination microscopy , a form of super-resolution fluorescence microscopy , Stahley et al. found altered desmosomal protein organization, co-localization of patient IgG with markers for lipid rafts and endosomes, and reduction in size of desmosomes (in cell culture models, the pemphigus vulgaris IgG-Dsg complex is internalized from the plasma membrane through “lipid raft”–mediated endocytosis, with resultant degradation of Dsg3).
A newly developed biochip mosaic-based indirect IF technique, using recombinant antigenic substrates and transfected cells, can detect autoantibodies against Dsg3 in the serum of about 98% of patients with pemphigus vulgaris. This method has high sensitivity and specificity comparable to ELISA methods and could serve as an initial diagnostic screening test. Saliva can be used as an alternative to serum in selected circumstances, but its lack of reliable correlation with serum may make it a less than ideal substrate.
There is dissolution of the intercellular attachments, leading to a widening of the intercellular spaces and eventual separation of the desmosomal attachment plaques. As acantholysis progresses, the desmosomes gradually disappear, followed by retraction of the tonofilaments to the perinuclear area. The keratinocytes often develop numerous, interdigitating processes. Immunoelectron microscopy shows that the immunoglobulins are deposited on the surface of the epidermal cells in a discontinuous globular pattern in the region of the intercellular domains of desmosomes.
Pemphigus vulgaris can be potentially confused with other acantholytic disorders, particularly Hailey–Hailey disease (familial benign chronic pemphigus), an inherited acantholytic disorder, and a similar but localized disorder that appears to be acquired, acantholytic dermatosis of the genitocrural region . However, those conditions usually show much more extensive suprabasilar acantholysis, with separation of keratinocytes in the spinous layer producing the appearance of a “dilapidated brick wall.” Some dyskeratosis can be seen in Hailey–Hailey disease—a feature not often seen in pemphigus. Importantly, the acantholysis in Hailey–Hailey disease “respects” the hair follicles; that is, follicular epithelia in acantholytic lesions tend to be spared. This is not the case in pemphigus. Dermal infiltrates containing eosinophils are common in pemphigus, and eosinophilic spongiosis may occur in early lesions. This cell type is less common in Hailey–Hailey disease; although eosinophils can occasionally be seen, Hailey–Hailey disease is not associated with eosinophilic spongiosis. Examples of focal acantholytic dyskeratosis, particularly transient acantholytic dermatosis (Grover's disease), can have a pemphigus vulgaris–like configuration, but often some dyskeratosis can be identified. Actinic keratoses with marked acantholysis might occasionally mimic a lesion of pemphigus vulgaris, but some degree of basilar keratinocyte atypia is usually present, and the clinical presentation would be quite different from that of pemphigus. Immunofluorescent studies would be definitive because among these conditions, only pemphigus shows positive intercellular fluorescence. Although a characteristic feature of paraneoplastic pemphigus is positive indirect IF using murine bladder epithelium (see later), some cases of nonneoplastic pemphigus vulgaris and pemphigus foliaceus are also positive using this substrate.
Pemphigus vegetans is a rare variant of pemphigus vulgaris that differs from it by the presence of vegetating erosions, primarily affecting flexural areas. Two variants of pemphigus vegetans have been recognized. In the Neumann type, the initial lesions are vesicular and erosive, resembling pemphigus vulgaris, but the lesions progressively evolve into vegetating plaques. The less common Hallopeau type commences with pustular lesions and has a relatively benign course with few, if any, relapses.
Oral lesions are almost invariably present in pemphigus vegetans, and these may be the presenting or dominant feature. Some of these cases may represent pyodermatitis–pyostomatitis vegetans ( pyoderma vegetans, pyostomatitis vegetans; PPV ), a condition that has often been confused with pemphigus vegetans. In PPV, annular pustular lesions on the skin may precede, accompany, or follow the usually extensive vegetating oral lesions. Most cases are associated with inflammatory bowel disease, particularly ulcerative colitis. The bowel disease usually precedes the oral lesions by months or years. A patient with Crohn's disease and pyostomatitis vegetans of the mouth and vulvoperineal region was successfully treated with a combination of infliximab and methotrexate. Topical clobetasol was used in another patient. Cyclosporine was effective in a further case.
Sometimes the surface of the tongue in pemphigus vegetans assumes a cerebriform pattern. Similar thickening of the epidermis of the scalp may rarely lead to the clinical picture of cutis verticis gyrata. Localization to the vulva and to the scalp has been reported. Other unusual locations include the face, breast, leg, sole and a finger. Another clinical feature is the frequent presence of eosinophilia in the peripheral blood. In a case that began as pemphigus foliaceus, serum levels of eosinophil cationic protein and TGF-α increased with the onset of vegetative lesions. Cases induced by the ingestion of the drugs captopril and enalapril and by the intranasal use of heroin and cocaine have been reported. Its association with HIV infection and gastric cancer may have been fortuitous.
Immunological studies provide further evidence for the close relationship between pemphigus vegetans and pemphigus vulgaris. Circulating antibodies have been found in patients with pemphigus vegetans of the Neumann type that precipitate with the 130- and 85-kDa polypeptides of the pemphigus vulgaris antigen (dsg3), but there appear to be additional antibodies directed against desmocollin 3 and as yet uncharacterized antigens. In the Hallopeau type, antibodies to dsg3 have been detected. The antibodies appear to belong to the IgG2 and IgG4 subclass, with strong complement fixation. IgA antibodies were also present in one case. The finding of circulating autoantibodies to the bullous pemphigoid antigen (BP230) in a patient with pyodermatitis–pyostomatitis probably represented an epiphenomenon.
Treatment is usually with corticosteroids, but an additional immunosuppressive agent such as dapsone or azathioprine is often required. Minocycline and nicotinamide have been used successfully in a case. Hallopeau patients usually require lower doses of corticosteroids compared with patients with the Neumann type and have fewer relapses.
In the early pustular lesions of the Hallopeau type, the appearances resemble those of eosinophilic spongiosis with transmigration of eosinophils into the epidermis and the formation of spongiotic microvesicles and eosinophilic microabscesses. Charcot–Leyden crystals have been seen within these microabscesses. Sporadic acantholytic cells may be present, although they are not usually seen in Tzanck smears prepared from the pustules.
In early lesions of the Neumann type, there are intraepidermal vesicles with suprabasal acantholysis but no eosinophilic microabscesses.
The vegetative lesions of both types of pemphigus vegetans are similar, with hyperkeratosis, some papillomatosis, and prominent acanthosis with downward proliferation of the rete ridges ( Fig. 7.10 ). There are suprabasal lacunae containing some eosinophils. A few acantholytic cells may be present, particularly in the Neumann type.
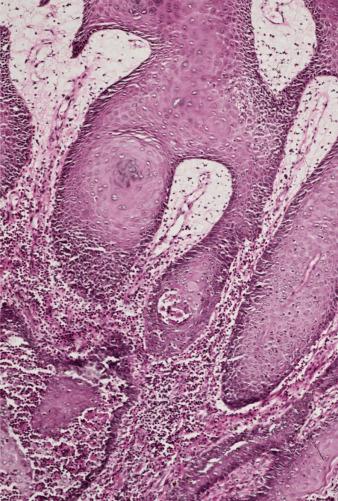
In both types of pemphigus vegetans, the upper dermis contains a heavy infiltrate of lymphocytes and eosinophils, together with some neutrophils. There may be edema of the papillary dermis.
The direct IF findings in pemphigus vegetans are similar to those in pemphigus vulgaris with the intercellular deposition of IgG and C3. Circulating antibodies to the intercellular region are usually detectable by indirect IF.
There is a reduction in tonofilaments and desmosomes in keratinocytes in skin adjoining the lesion; only rare desmosomes are found in affected skin. Migration of eosinophils into the epidermis through a damaged basement membrane is often observed.
The histological features of PPV show some similarities to pemphigus vegetans. There are intraepithelial and subepithelial microabscesses with an admixture of neutrophils and eosinophils ( Fig. 7.11 ). There is a moderately heavy, mixed inflammatory cell infiltrate in the underlying dermis. The lesions are usually more inflammatory than the lesions of pemphigus vegetans. Epithelial hyperplasia is variable and usually more marked in oral lesions. Acantholysis is focal and never severe. Direct IF is negative, if misdiagnosed cases of pemphigus vegetans are excluded. However, there is a report of two cases presenting clinically as PPV, associated with inflammatory bowel disease, in which direct IF showed intercellular IgA deposition. It remains to be determined whether other cases presenting as PPV might also show IgA deposits.
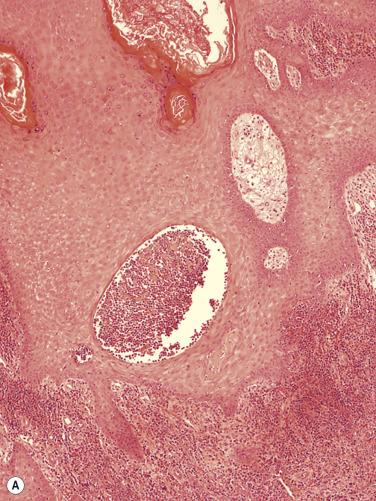
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here