Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
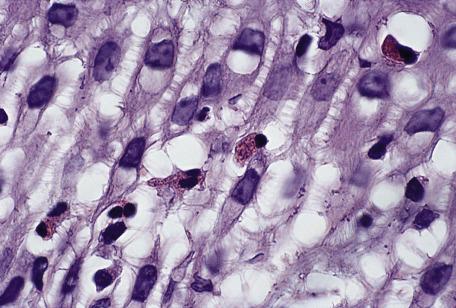
The spongiotic tissue reaction is characterized by the presence of intraepidermal and intercellular edema (spongiosis) ( Fig. 6.1 ). It is recognized by the widened intercellular spaces between keratinocytes, with elongation of the intercellular bridges ( Fig. 6.2 ). The foci of spongiosis may vary from microscopic in size to grossly identifiable vesicles and even bullae. Mild spongiosis is well seen in semi-thin sections. Inflammatory cells, usually lymphocytes but sometimes eosinophils or even neutrophils, are also present.
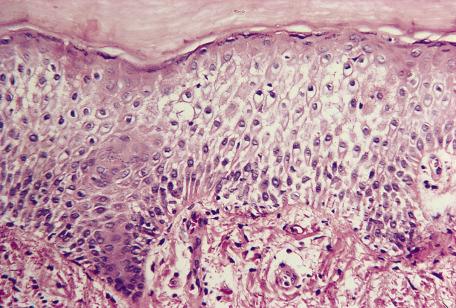
The spongiotic tissue reaction is a histopathological concept and not a clinical one, although several of the many diseases with this tissue reaction have been included, in the past, in the category of eczemas. This term (derived from Greek elements that mean “boiling over”) has fallen into some disrepute in recent years because it lacks precision. The “eczemas” all show epidermal spongiosis at some stage of their evolution, even though this has been disputed for atopic eczema. Clinically, the various spongiotic disorders may present with weeping, crusted patches and plaques, as in the so-called “eczemas,” or as erythematous papules, papulovesicles, and even vesiculobullous lesions. Resolving lesions and those of some duration may show a characteristic collarette of scale.
The mechanism involved in the collection of the intercellular fluid is controversial. It is generally accepted that the fluid comes from the dermis and, in turn, from blood vessels in the upper dermis. Various immunological reactions are involved in some of the diseases discussed in this chapter, but in others the cause of this fluid extravasation from vessels remains to be elucidated. The controversy also involves the mechanism by which the dermal edema fluid enters the epidermis. One concept is that an osmotic gradient develops toward the epidermis, drawing fluid into it. The opposing view suggests that hydrostatic pressure leads to the epidermal elimination of dermal edema. The latter explanation does not satisfactorily explain the absence of spongiosis in pronounced urticarial reactions. Perhaps both mechanisms are involved to a varying degree. The spongiotic tissue reaction is a dynamic process. Vesicles come and go, and they can be situated at different levels in the epidermis. Parakeratosis forms above areas of spongiosis, probably as a result of an acceleration in the movement of keratinocytes toward the surface, although disordered maturation may contribute. Small droplets of plasma may accumulate in the mounds of parakeratosis, contributing to the appearance of the collarettes of scale mentioned previously.
There are several categories of disease in which casual histological examination may show a simulation of the spongiotic reaction pattern; they are excluded from consideration here. Diseases that present a lichenoid reaction pattern with obscuring of the dermoepidermal interface (e.g., pityriasis lichenoides, erythema multiforme, and fixed drug eruption) or prominent vacuolar change (variants of lupus erythematosus) may show some spongiosis above the basal layer. They are not included among the diseases considered in this chapter.
Certain viral exanthems and morbilliform drug eruptions show mild epidermal spongiosis, but it is usually limited to the basal layer of the epidermis. Other viral diseases, such as herpes simplex and herpes zoster, show ballooning degeneration of keratinocytes with secondary acantholysis. Some spongiosis is invariably present, but it is overshadowed by the other changes. Primary acantholytic disorders leading to vesiculation are also excluded. Mild spongiosis is seen overlying the dermal papillae in early lesions of psoriasis, but again this disease is not usually regarded as a spongiotic disorder.
The accumulation of acid mucopolysaccharides in the follicular infundibulum in follicular mucinosis may simulate spongiosis. Stains for mucin, such as the colloidal iron stain, will confirm the diagnosis, if any doubt exists.
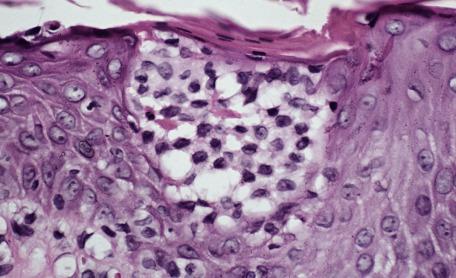
Finally, the Pautrier microabscesses of mycosis fungoides may be simulated by the collections of mononuclear cells that sometimes accumulate in spongiotic dermatitis ( Fig. 6.3 ). In spongiotic dermatitis, the cellular collections often assume a vase-like shape, with the lips of the vase situated at the interface between the granular and cornified layers. The intraepidermal collections of mononuclear cells express CD1a, S100 protein, CD36, and CD68. They lack CD14, which is found on mature Langerhans cells. Their phenotype suggests derivation from circulating monocytes and differentiation into mature Langerhans cells.
Five special patterns of spongiosis can be distinguished morphologically from the more usual type. These are neutrophilic spongiosis, in which there are numerous neutrophils associated with epidermal spongiosis; eosinophilic spongiosis , characterized by the presence of numerous eosinophils within the spongiotic foci; miliarial spongiosis, in which the edema is centered on the acrosyringium; follicular spongiosis, in which there is involvement of the follicular infundibulum; and pityriasiform spongiosis, in which there are spongiotic microvesicles containing lymphocytes ± Langerhans cells. Sometimes serial sections are required before it is appreciated that the spongiosis is related to the acrosyringium or acrotrichium. Diseases in these special categories are discussed first, followed by a description of the more usual type of spongiotic disorders. The histopathological features of the pityriasiform and other spongiotic diseases are included in Table 6.1 .
| Disease | Histopathological features |
|---|---|
| Irritant contact dermatitis | Superficial ballooning, necrosis, and neutrophils; mild irritants produce spongiotic dermatitis mimicking allergic contact dermatitis, although superficial apoptotic keratinocytes may also be present |
| Allergic contact dermatitis | Variable spongiosis and vesiculation at different horizontal and vertical levels, with an “ordered” pattern; mild exocytosis; progressive psoriasiform hyperplasia with chronicity; usually eosinophils in superficial dermal infiltrate; superficial dermal edema |
| Protein contact dermatitis | No distinguishing features recorded; an urticarial component may be present |
| Nummular dermatitis | May mimic allergic contact dermatitis but usually more “untidy.” Neutrophils may be in dermal infiltrate and even the epidermis; the psoriasiform hyperplasia in chronic cases may show variable thickening of adjacent rete pegs |
| Seborrheic dermatitis | Variable spongiosis and psoriasiform hyperplasia depending on activity and chronicity; scale crust and spongiosis may localize to follicular ostia |
| Atopic dermatitis | Mimics other spongiotic diseases with variable spongiosis (usually quite mild) and psoriasiform hyperplasia; subtle features include prominence of vessels in the papillary dermis, increased epidermal volume without necessarily producing psoriasiform folding; eosinophil major basic protein present, sometimes disproportionate to eosinophils |
| Pompholyx | Vesiculation with peripheral displacement of acrosyringia; process usually more sharply defined than allergic contact dermatitis of palms and soles; some evolve into picture of pustulosis palmaris with neutrophils (important to exclude fungi in these cases with PAS stain) |
| Stasis dermatitis | Mild spongiosis only; proliferation of superficial dermal vessels; extravasation of erythrocytes; abundant hemosiderin |
| Autoeczematization | Variable spongiosis; edema of papillary dermis and activated lymphocytes often present |
| Pityriasis rosea | Pityriasiform spongiosis with focal parakeratosis; lymphocyte exocytosis; sometimes erythrocyte extravasation in papillary dermis; “herald patch” is more psoriasiform |
| Papular acrodermatitis of childhood | Three tissue reaction patterns (lichenoid, spongiotic, and lymphocytic vasculitis) often present; small spongiotic vesicles resembling pityriasis rosea may be present |
| Spongiotic drug reactions | Spongiosis with conspicuous exocytosis of lymphocytes relative to the amount of spongiosis; rare apoptotic keratinocytes; eosinophils, plasma cells, and activated lymphocytes may be in superficial dermal infiltrate; may show mid-dermal spillover; sometimes superficial dermal edema |
| Chronic superficial dermatitis | Only mild spongiosis and focal parakeratosis with variable psoriasiform hyperplasia; superficial perivascular infiltrate with characteristic upward extension and mild exocytosis |
| Light reactions | Variable, usually mild spongiosis; superficial and deep perivascular dermal inflammation; a deep infiltrate is not invariable in lesions of short duration; subepidermal edema in some cases of polymorphic light eruption; stellate fibroblasts, vertical collagen streaking, variable psoriasiform hyperplasia and some atypical lymphocytes with exocytosis in actinic reticuloid; scattered “sunburn cells” in phototoxic lesions (sometimes with only mild other changes); deeply extending, straight, basophilic (elastotic) fibers in lesions of long duration |
| Dermatophytoses | Neutrophils in stratum corneum or compact orthokeratosis (“sandwich sign”) should alert observer to perform a PAS stain; spongiotic vesicles may form on palms and soles |
| Arthropod bites | Spongiotic vesicles containing variable numbers of eosinophils; superficial and deep dermal inflammation with interstitial eosinophils |
| Grover's disease | Spongiosis with focal acantholysis in the spongiotic variant; untidy superficial dermal inflammation |
| Toxic erythema of pregnancy | Spongiosis mild and inconstant; variable papillary dermal edema; tight superficial perivascular infiltrate sometimes extending to mid-dermis; interstitial eosinophils in some cases |
| Erythema annulare centrifugum | Pityriasiform spongiosis at periphery of lesion; mild perivascular cuffing with lymphocytes; late lesions lack spongiosis |
| Pigmented purpuric dermatoses | Spongiosis mild and inconstant; lymphocytic vasculitis with variably dense infiltrate in the papillary dermis; hemosiderin in the upper dermis |
| Pityriasis alba | Clinical diagnosis; mild focal spongiosis with minimal parakeratosis |
| Erythroderma | Mild spongiosis; variable psoriasiform hyperplasia; appearances depend on underlying disease; a difficult diagnosis without clinical history |
| Mycosis fungoides | Mild spongiosis, variable epidermal hyperplasia and epidermal mucinosis; epidermotropism, often with Pautrier microabscesses; variable cytological atypia of lymphocytes that extend upward into the papillary dermis |
Neutrophilic spongiosis is characterized by the presence of neutrophils within spongiotic foci in the epidermis ( Fig. 6.4 ). The term spongiform pustular dermatitis can be used for a severe form of neutrophilic spongiosis in which pustules can be seen clinically and histologically. Subcorneal pustules are excluded from this category. Ackerman stated that neutrophils are absent in “authentic” spongiotic dermatitides, but a case can be made for including the following conditions in this histological pattern:
Pustular psoriasis
Prurigo pigmentosa
Reiter's syndrome
Pemphigus foliaceus
IgA pemphigus
Herpetiform pemphigus
Infantile acropustulosis
Acute generalized exanthematous pustulosis
Palmoplantar pustulosis
Staphylococcal toxic shock syndrome
Neisserial infections
Dermatophytoses
Candidosis
Beetle (Paederus) dermatitis
Pustular contact dermatitis
Glucagonoma syndrome
Amicrobial pustulosis associated with autoimmune diseases
Erosive pustular dermatosis of the legs
Amicrobial pustulosis of the folds
Periodic fever syndromes.
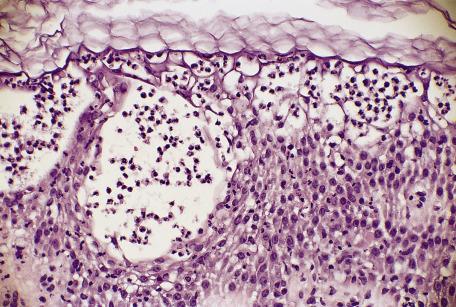
Reiter's syndrome (see p. 111 ) shares histological features with pustular psoriasis. It is not considered further. If spongiosis occurs in pemphigus foliaceus, it is usually of eosinophilic type. Neutrophilic spongiosis has been reported in a case that evolved into an atypical pemphigus phenotype. In infantile acropustulosis (see p. 165 ), there are variable numbers of eosinophils admixed with the neutrophils. In palmoplantar pustulosis (see p. 168 ), large vesicles are the dominant feature, with only some neutrophilic spongiosis at the edges. This tissue reaction can also be seen in the staphylococcal toxic shock syndrome (see p. 676 ) and in infections with Neisseria species (see p. 685 ). Pustular contact dermatitis is considered later in this chapter (see p. 137 ). The glucagonoma syndrome is considered in Chapter 19 . Amicrobial pustulosis associated with autoimmune diseases and herpetiform pemphigus are discussed in Chapter 7 . Erosive pustular dermatosis of the legs is discussed in Chapter 7 . Amicrobial pustulosis of the folds and the periodic fever syndromes are included in Chapter 9 .
In both pustular psoriasis (see p. 109 ) and Reiter's syndrome (see p. 111 ) there is characteristic spongiform pustulation.
Prurigo pigmentosa is an inflammatory dermatosis characterized by the sudden onset of pruritic erythematous papules, usually involving the trunk and neck, that coalesce to form reticulated, mottled patches. Vesicles and bullae are uncommon. Because it resolves leaving mottled or reticulate hyperpigmentation, it is discussed further in Chapter 11 .
In the established papular phase, there is variable spongiosis, acanthosis, exocytosis of lymphocytes, and a few neutrophils with isolated apoptotic keratinocytes. In early stages of the disease, there is neutrophilic spongiosis with some apoptotic keratinocytes at all levels of the epidermis. There is a mild superficial perivascular infiltrate of lymphocytes and neutrophils and a few eosinophils in the earlier stages. A Korean study reported perivascular neutrophilic infiltrates with some papillary dermal neutrophils in early lesions and also neutrophilic spongiosis in fully developed lesions. Pigment incontinence is a conspicuous feature of late lesions.
Immunoglobulin A (IgA) pemphigus is a vesiculobullous disease (see p. 164 ) with a variable expression, accounting for the many titles applied to this condition in the past. There are subcorneal and/or intraepidermal pustules with usually only mild acantholysis. IgA is deposited in the epidermis in an intercellular position.
Acute generalized exanthematous pustulosis is a rapidly evolving pustular eruption (see p. 166 ), usually associated with the ingestion of drugs, particularly antibiotics. There are subcorneal and superficial intraepidermal pustules. Subepidermal pustules are sometimes present with prominent neutrophil exocytosis and neutrophilic spongiosis.
Neutrophilic spongiosis can be found with dermatophyte infection and also with the yeast Candida . The presence of neutrophils in the epidermis and/or overlying stratum corneum should always lead to the performance of a periodic acid–Schiff (PAS) stain.
Vesicular dermatitis, characterized by areas of neutrophilic spongiosis, results from contact with various beetles (order Coleoptera). Bullae and small pustules may even result. The term Paederus dermatitis is used for the reaction produced by the genus Paederus, of which there are several hundred species capable of producing a form of acute irritant contact dermatitis. The irritant substance is pederin, a highly toxic alkaloid produced by members of this genus. Localized erythema occurs first, followed by blisters after 2 to 4 days, associated with increasing pain. Lesions are commonly linear as a result of crushing of the beetle on the skin, followed by its wiping off the skin. The delay in the appearance of the lesions may lead to lack of recognition of the causal event.
Early lesions show neutrophilic spongiosis leading to vesiculation and eventual reticular necrosis of the epidermis. This is followed by confluent epidermal necrosis, usually with a surviving layer of suprabasal cells. Scattered acantholytic cells may be present. The large number of intraepidermal neutrophils, combined with areas of confluent necrosis and reticular degeneration, are characteristic. Older lesions show irregular acanthosis and pallor of superficial keratinocytes, with overlying parakeratotic scale containing a neutrophil exudate.
A similar spongiotic and vesicular dermatitis, with the addition of vasculitis, has been produced by the hide beetle, Dermestes peruvianus .
Eosinophilic spongiosis is a histological reaction pattern characterized by the presence of epidermal spongiosis associated with the exocytosis of eosinophils into the spongiotic foci. Microabscesses, containing predominantly eosinophils, are formed.
Eosinophilic spongiosis is found in a heterogeneous group of dermatoses, most of which are considered elsewhere. It can be seen in the following conditions:
Pemphigus (precursor lesions)
Pemphigus vegetans
Herpetiform pemphigus
Bullous pemphigoid
Cicatricial pemphigoid
Herpes gestationis
Idiopathic eosinophilic spongiosis
Eosinophilic, polymorphic, and pruritic eruption
Allergic contact dermatitis
Atopic dermatitis
Arthropod bites
Eosinophilic folliculitis (Ofuji's disease)
Incontinentia pigmenti (first stage)
Drug reactions
ID reactions
Still's disease
Vulvar lichen sclerosus (rare)
Wells’ syndrome
Eosinophilic spongiosis may occur in the preacantholytic stage of both pemphigus foliaceus and pemphigus vulgaris. In these early stages, direct immunofluorescence demonstrates the presence of IgG in the intercellular areas of the epidermis. In patients whose disease evolves into pemphigus foliaceus, the initial clinical presentation may resemble dermatitis herpetiformis. Some of these cases have been reported in the literature as herpetiform pemphigus.
The pattern is that described for eosinophilic spongiosis ( Fig. 6.5 ). Acantholysis and transitional forms between eosinophilic spongiosis and the usual histological findings in pemphigus may be present.

Eosinophils are often prominent within the vesicles of pemphigus vegetans. Acantholysis, epidermal hyperplasia, and the absence of spongiosis adjacent to the suprabasal vesicles usually allow the diagnosis of pemphigus vegetans to be made.
Eosinophilic spongiosis is an uncommon finding in the urticarial stage of bullous pemphigoid and in erythematous patches adjacent to characteristic bullae in later stages of the disease. In one case, the eosinophilic spongiosis preceded the diagnosis of bullous pemphigoid by 13 years. There is usually a prominent dermal infiltrate of eosinophils, and IgG is demonstrable along the basement membrane zone.
Several cases have been recorded in which a localized, recurrent bullous eruption has been associated with the histological appearance of eosinophilic spongiosis. Polycythemia rubra vera was present in one case.
Eosinophilic, polymorphic, and pruritic eruption, associated with the use of radiotherapy, particularly for carcinoma of the breast, has not been well characterized. Similar cases have been reported in the past, often without histological confirmation, under several different designations, including erythema multiforme and bullous pemphigoid after radiation therapy. A similar eruption has been described in patients with cervical cancer, and more recently, after radiation therapy in a patient with primary nodal Merkel cell carcinoma. The rash can be confined to the irradiated area but may be more widespread and is polymorphic and intensely pruritic, commencing during or after radiotherapy and lasting several weeks or months. The lesions are usually erythematous papules, measuring 3 to 10 mm in diameter. Pustules, wheals, excoriations, vesicles, and tense subepidermal blisters can also occur. An animal model has been developed in irradiated pigs, with timing and microscopic features similar to those in humans.
The variable histological appearances reflect the polymorphic nature of the rash. There is usually spongiosis with focal spongiotic vesiculation. There may be some acanthosis in lesions of longer duration and secondary changes of rubbing and scratching. The dermal infiltrate is usually superficial and deep and of moderate severity and may be perivascular, interstitial, and/or periadnexal. Extension into the subcutis sometimes occurs. Eosinophils are always present. There is usually some eosinophilic spongiosis, and intraepidermal eosinophilic and neutrophilic pustules have been described. An eosinophilic panniculitis is much less common. If bullae are present, they usually resemble bullous pemphigoid. There are no features of erythema multiforme, despite earlier publications attributing this condition to erythema multiforme.
Eosinophilic spongiosis may be seen in allergic contact dermatitis (see p. 137 ).
Eosinophilic spongiosis is occasionally seen in the reaction to the bite of certain arthropods, particularly the scabies mite (see p. 815 ).
In eosinophilic folliculitis (Ofuji's disease; see p. 497 ), the eosinophilic spongiosis involves the follicular infundibulum; sometimes the immediately adjacent epidermis is also involved.
In the first stage of incontinentia pigmenti (see p. 371 ), there is prominent exocytosis of eosinophils into the epidermis and foci of eosinophilic spongiosis ( Fig. 6.6 ). Occasional dyskeratotic keratinocytes may also be present.

Several papers have described examples of vulvar lichen sclerosus (see p. 393 ) displaying the changes of eosinophilic spongiosis. When found together with dermal eosinophilic infiltrates, excoriations, and lymphocyte exocytosis, eosinophilic spongiosis may predict a poor symptomatic response to therapy of this disease.
Miliarial spongiosis is characterized by intraepidermal edema centered on the acrosyringium. It is characteristic of the various clinical forms of miliaria.
The miliarias are a clinically heterogeneous group of diseases that occur when the free flow of eccrine sweat to the skin surface is impeded. Three variants of miliaria have been defined according to the depth at which this sweat duct obstruction occurs.
Miliaria crystallina (miliaria alba), which results from superficial obstruction in the stratum corneum, is characterized by asymptomatic, clear, 1- to 2-mm vesicles that rupture easily with gentle pressure. Congenital onset is exceedingly rare, but onset in the first week of life is not uncommon. It has also been reported in adult patients in an intensive care setting. It may have been caused by drugs producing enhanced α-adrenergic stimulation of sweat gland myoepithelium. Isotretinoin can also cause miliaria crystallina. It is a self-limited condition that resolves without complications over a period of several days.
Miliaria rubra (prickly heat) consists of small, discrete, erythematous papulovesicles with a predilection for the clothed areas of the body. The lesions are often pruritic. In severe cases, with recurrent crops of lesions, anhidrosis may result. Occasionally, pustular lesions (miliaria pustulosa) may coexist. Both miliaria rubra and pustular miliaria rubra have been reported in infants and children with type I pseudohypoaldosteronism. Miliaria rubra can also occur in Morvan's syndrome, a form of generalized myokymia (OMIM 160120 ).
Miliaria profunda refers to the development of flesh-colored papules resembling gooseflesh, associated with obstruction of the sweat duct near the dermoepidermal junction. It usually follows severe miliaria rubra and is associated with anhidrosis. In a study of two cases of miliaria profunda by Tey et al. using high-definition optical coherence tomography, lesions appeared to be localized to the epidermis (see later). A case has been reported in which large white plaques with an erythematous border were present. The lesions expanded centrifugally until they were several centimeters or more in diameter. They were localized to sites at which occlusive tape had been applied. This variant has been called giant centrifugal miliaria profunda.
Although it has been presumed since the 19th century that obstruction of the eccrine duct is involved in the pathogenesis of the miliarias, the nature of this obstruction and its cause have been the subject of much debate. The first demonstrable histological change is the accumulation of PAS-positive, diastase-resistant material in the distal pore, although this has not always been found. This material has been designated extracellular polysaccharide substance (EPS). It is likely that there is an earlier stage of obstruction, which cannot be demonstrated in tissue sections. After several days, a keratin plug forms as part of the repair process, leading to further obstruction of the duct, often at a deeper level. Various factors may contribute to the initial duct obstruction, including changes in the horny layer related to excess sweating, the presence of sodium chloride in more than isotonic concentration, and lipoid depletion. In many cases, there is an increase in the number of resident aerobic bacteria, particularly cocci. Certain strains of Staphylococcus epidermidis produce the PAS-positive material known as EPS (mentioned previously), and these organisms may play a central role in the pathogenesis of miliaria. Miliaria have also developed at the site of previous radiotherapy; there was associated keratotic plugging of the eccrine orifices. Recent studies have shown that Foxc1-ablated mice (a Fox family transcription factor) become anhidrotic and show microscopic changes similar to those of human miliaria: blockage of sweat ducts by hyperkeratotic plugs, with luminal dilatation in ducts and secretory portions of the gland and formation of blisters and papules on the skin surface. Foxc1 deficiency induces expression of keratinocyte terminal differentiation markers such as Sprr2a in duct luminal cells, which may contribute to keratotic plug formation.
In miliaria crystallina, there is a vesicle within or directly beneath the stratum corneum. There is often a thin, orthokeratotic layer forming the roof of the vesicle and a basket-weave layer of keratin in the base. A PAS-positive plug may be seen in the distal sweat pore.
Miliaria rubra is characterized by variable spongiosis and spongiotic vesiculation related to the epidermal sweat duct unit and the adjacent epidermis ( Fig. 6.7 ). There is a small number of lymphocytes in the areas of spongiosis. An orthokeratotic or parakeratotic plug may overlie the spongiosis. Sometimes there is edema in the papillary dermis adjacent to the point of entry of the eccrine duct into the epidermis. A mild lymphocytic infiltrate is usually present in this region. If the edema is pronounced, leading to subepidermal vesiculation, then miliaria profunda is said to be present. Biopsies from two infants with giant centrifugal miliaria profunda featured hyperplasia of eccrine sweat ducts, ortho- and parakeratotic plugging, and granulomatous infiltrates around the straight intradermal portions of these ducts. The previously mentioned study using high-definition optical coherence tomography showed a dilated spiraling acrosyringium, with an adjacent hyperrefractile substance that likely represented macerated keratin, surrounded by a hyporefractile rim probably corresponding to free fluid caused by obstructed sweat outflow and correlating with spongiosis The authors postulate that this change may be better observed through in vivo imaging because of the dehydration process used in standard histological slide preparation. Miliaria pustulosa is characterized by neutrophils beneath the stratum corneum and/or in the epidermal sweat duct ( Fig. 6.8 ).
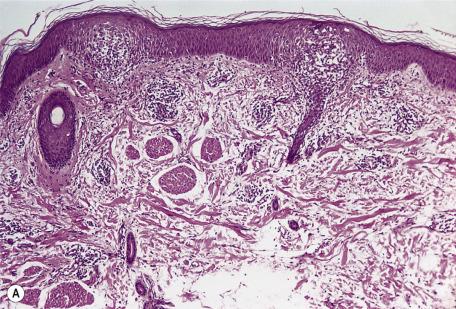
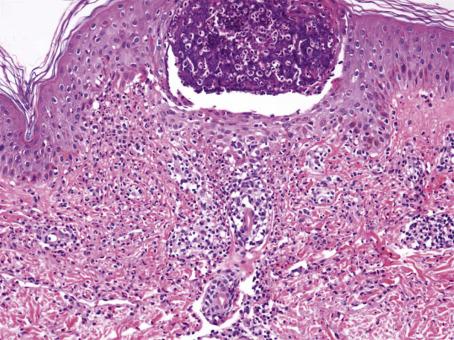
Less commonly, there is only slight spongiosis in the region of the acrosyringium in miliaria rubra associated with dilatation of the terminal eccrine duct. It should be remembered that not all eccrine ducts are involved.
The secretory acini show few changes in the miliarias. They may be mildly dilated. Often, there is slight edema of the connective tissue between the secretory units. Lymphocytes are not usually present, unless there is a prominent inflammatory cell infiltrate elsewhere in the dermis.
Follicular spongiosis refers to the presence of intercellular edema in the follicular infundibulum ( Fig. 6.9 ). It occurs in a limited number of circumstances:
Infundibulofolliculitis
Atopic dermatitis (follicular lesions)
Apocrine miliaria
Eosinophilic folliculitis
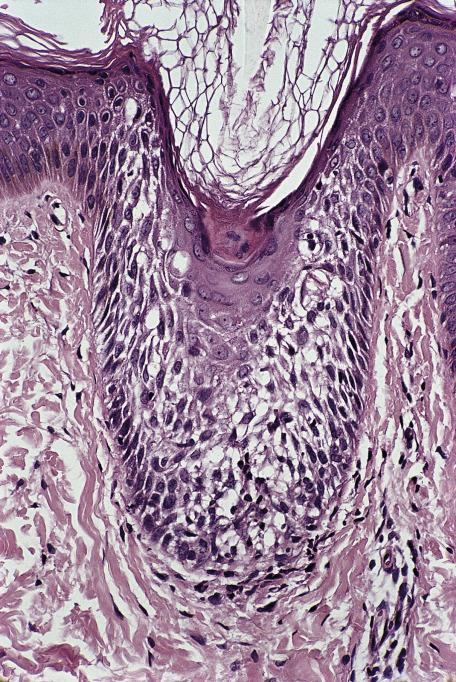
Infundibulofolliculitis, also known as disseminate and recurrent infundibulofolliculitis (of Hitch and Lund), presents as a follicular, often pruritic, papular eruption with a predilection for the trunk and proximal parts of the extremities of young adult men. It occurs almost exclusively in black patients. Although the lesions resemble those seen in some cases of atopic dermatitis, the individuals studied so far have not been atopic.
There is spongiosis of the follicular infundibulum with exocytosis of lymphocytes ( Fig. 6.10 ). A few neutrophils are sometimes present. There is widening of the follicular ostium and focal parakeratosis of the adjacent epidermis. Occasional follicles contain a keratin plug. The follicular infundibulum is often hyperplastic. There is usually a slight infiltrate of lymphocytes around the follicles and around the blood vessels in the superficial part of the dermis. Mast cells may be increased.

Some patients with atopic dermatitis develop small follicular papules, often on the trunk.
There is spongiosis of the follicular infundibulum with exocytosis into this region of the epidermis. Usually, no neutrophils are present. The adjacent epidermis may show mild acanthosis and sometimes focal parakeratosis. The histopathology resembles that seen in infundibulofolliculitis.
Apocrine miliaria (Fox–Fordyce disease) presents as a chronic papular eruption, usually limited to the axilla (see p. 538 ). It results from rupture of the intrainfundibular portion of the apocrine duct.
Serial sections may be required to demonstrate the spongiosis of the follicular infundibulum adjacent to the point of entry of the apocrine duct. There may be a few neutrophils in the associated inflammatory response. Periductal foam (xanthoma) cells are often present.
Eosinophilic folliculitis (Ofuji's disease) is characterized by eosinophilic spongiosis centered on the follicular infundibulum. It is discussed in detail on p. 497 .
Pityriasiform spongiosis is characterized by the presence of microvesicles within areas of spongiosis that contain lymphocytes, histiocytes, and Langerhans cells. It is a distinctive pattern when well developed. It is seen in the following conditions:
Pityriasis rosea
Pityriasiform drug reaction
Erythema annulare centrifugum
Nummular dermatitis (some cases)
Lichen striatus (uncommon).
The spongiosis in miliaria may mimic pityriasiform spongiosis but the vesicles are often larger, and they are always related to an acrosyringium. The “inverted flask”–like lesions sometimes seen in allergic contact dermatitis are better defined microvesicles, with a different shape to pityriasiform lesions. They also contain a predominance of Langerhans cells.
The various diseases are discussed in order, but nummular dermatitis (see p. 138 ) and lichen striatus (see p. 63 ) are considered in more detail elsewhere.
Pityriasis rosea (PR) is a common, acute, self-limited dermatosis in which oval, salmon-pink, papulosquamous lesions develop on the trunk, neck, and proximal extremities. Lesions often follow the lines of skin cleavage, giving a “Christmas tree” pattern. A scaly plaque 2 to 10 cm in diameter, the “herald patch,” may develop on the trunk 1 or 2 weeks before the other lesions. Pityriasis rosea has been reported at all ages, but the majority of patients are between 10 and 35 years old. Clinical variants include those with acral or facial involvement, oral lesions, a unilateral or local distribution, or the presence of pustular, purpuric, or vesicular lesions. In one study of 527 patients with pityriasis rosea, oropharyngeal lesions were found in 28%; they were classified as erythemato-macular, erythemato-vesicular, macular and papular, and petechial; the latter two were the most commonly observed patterns. Follicular pityriasis rosea is a rarely reported form of the disease. Pityriasis rosea developing in the first 15 weeks of pregnancy is associated with premature delivery and miscarriages.
Because atypical cases of pityriasis rosea are fairly common, Chuh has drawn up a list of diagnostic criteria for pityriasis rosea. Essential features for the diagnosis include (1) discrete circular or oval lesions, (2) scaling on most lesions, and (3) peripheral collarette scaling with central clearance of at least two lesions. Optional clinical features, of which one must also be present, include (1) truncal and proximal limb distribution, with less than 10% of lesions distal to the mid-upper arm and mid-thigh; (2) distribution of most lesions along the ribs; and (3) a herald patch appearing at least 2 days before the generalized eruption. Interestingly, histopathological features were not added to the criteria “because they are nonspecific in PR.” Pityriasis rosea must be distinguished from secondary syphilis. Ackerman proposed that pityriasis rosea and erythema annulare centrifugum are clinical variations of a single pathological process and that pityriasis rosea gigantea is pityriasis rosea concurrent with erythema annulare centrifugum.
The cause is unknown, but an infectious etiology, particularly a virus, has long been suspected. This is supported by a history of a preceding upper respiratory tract infection in some patients, occasional involvement of close-contact pairs, case clustering, modification of the disease by the use of convalescent serum or erythromycin, and the development of a pityriasis rosea–like eruption in some cases of infection by ECHO (enteric cytopathic human orphan) 6 virus, enterovirus, or Mycoplasma . There has been recent interest in the role of human herpesvirus 6 (HHV-6) and HHV-7 in the etiology of pityriasis rosea. Although HHV-6 and HHV-7 may play a role in some patients, the low detection rate of HHV-7 DNA sequences argues against a causative role for this virus. In the case of HHV-6, reactivation of the virus during the early stages of the disease might explain its detection in some cases. Herpesvirus-like particles were detected in lesional skin in 71% of patients with pityriasis rosea in one study. Pityriasis rosea is not associated with HHV-8 infection or with herpes simplex virus 1 (HSV-1) or HSV-2 infection. Particles resembling togavirus or arenavirus have been found on electron microscopy of a herald patch, suggesting that this might be the inoculation site. An association with influenza A (H1N1) infection has also been reported. No virus has ever been cultured. Immunological reactions, particularly cell mediated, have also been regarded as important. Important roles have been assigned to the following cytokines, chemokines, and growth factors: interleukin-17 (IL-17) and IL-22, interferon-γ (IFN-γ), vascular endothelial growh factor (VEGF), and IFN-γ–induced protein 10 (IP-10, also known as CXCL10). Certain HLA subtypes may confer a susceptibility to the disease in certain races.
A pityriasis rosea–like eruption has also been recorded as a complication of graft-versus-host reaction after bone marrow transplantation, during radiotherapy (in localized form), and in patients with acute myeloid leukemia or Hodgkin's disease ; the eruption has also been reported as the presenting symptom of Hodgkin's disease in one case. A long-lasting pityriasis rosea–like eruption has been described in association with AIDS. The occurrence of a pityriasis rosea–like eruption in a patient with indeterminate cell histiocytosis may have resulted from an isotopic response in healed lesions of pityriasis rosea.
It is debatable whether active intervention is warranted to modify the disease course. Spontaneous resolution often occurs within 4 to 8 weeks. Most cases of pityriasis rosea do not recur, but a second episode is noted in about 2% of cases; three or more episodes are exceedingly uncommon but have been reported. Benefit has been reported from erythromycin and, particularly, high-dose or low-dose acyclovir. Evidence does not clearly support the use of UV phototherapy or corticosteroids. Also, just as in infectious mononucleosis, ampicillin may exacerbate the disease, producing more lesions, an abnormal distribution of lesions, and a prolonged clinical course.
Although the lesions are clinically papulosquamous, microscopy shows a spongiotic tissue reaction. The histopathological features are not pathognomonic, although in most cases they are sufficiently characteristic to allow the diagnosis to be made, even without a clinical history. The epidermis often has a vaguely undulating appearance. There is usually focal parakeratosis, sometimes with the formation of parakeratotic mounds ( Fig. 6.11 ). Sometimes these mounds partly lift from the epidermis, giving a tilted appearance to the mound. There is a diminution of the granular layer and focal spongiosis of pityriasiform type with lymphocyte exocytosis ( Fig. 6.12 ). Small spongiotic vesicles, sometimes simulating Pautrier microabscesses because of the aggregation of lymphocytes within them, are a characteristic feature; they are present in most cases if several levels are examined. Dyskeratotic cells may be seen at all levels of the epidermis; they are more common in the herald patch. Apoptotic keratinocytes are present in the lower epidermis in lesions undergoing involution. Multinucleate epidermal cells are uncommon. Focal acantholytic dyskeratosis has been reported once.
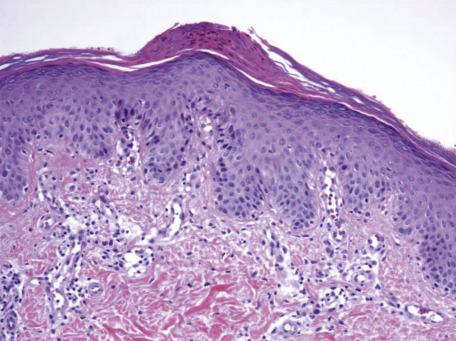
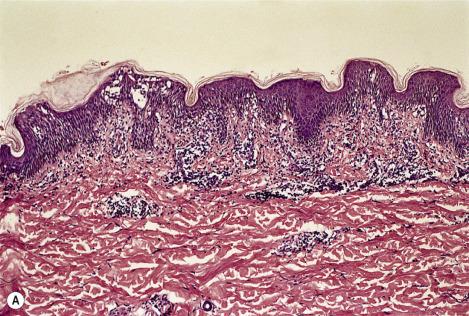
The papillary dermis shows some edema and sometimes homogenization of collagen. There may be some melanin incontinence. Red cell extravasation is common in the upper dermis and may extend into the lower layers of the epidermis ( Fig. 6.12D ). There is a mild to moderate lymphohistiocytic infiltrate in the upper dermis, with some eosinophils in the infiltrate in older lesions. The rare follicular variant shows follicular spongiosis, sometimes follicular plugging, and papillary dermal erythrocyte extravasation.
Dermoscopic features helpful in the diagnosis of pityriasis rosea include a yellowish background, dotted vessels, and peripheral scales.
Ultrastructural examination has confirmed the presence of dyskeratotic cells in some patients. These cells show aggregation of tonofilaments, some cytoplasmic vacuoles, and intracytoplasmic desmosomes. Cytolytic degeneration of keratinocytes adjacent to Langerhans cells has been reported in a herald patch. Virus-like particles have been seen in several studies.
Pityriasiform (pityriasis rosea–like) drug reaction presents as an eruption that resembles to varying degrees pityriasis rosea, usually within weeks of commencing one of the drugs listed in Table 6.2 . There are usually fewer lesions in drug-related cases than in pityriasis rosea. They are often larger than in pityriasis rosea, with scaling involving the entire lesion. Drug-related cases usually lack a herald patch. Furthermore, oral lesions are more common, as is the development of postinflammatory hyperpigmentation.
| Adalimumab | Ketotifen |
| Arsenicals | Lisinopril |
| Barbiturates | Metronidazole |
| BCG vaccination | Nortriptyline |
| Benfluorex | Omeprazole |
| Bismuth | Penicillamine |
| Captopril | Pneumococcal vaccination |
| Clonidine | Pyribenzamine |
| Gold | Rituximab |
| Imatinib mesylate | Smallpox vaccination |
| Influenza (H1N1) vaccination | Terbinafine |
| Isotretinoin | Tiopronin |
Many drugs have been implicated in the cause of these eruptions, including gold, bismuth, arsenicals, ketotifen, clonidine, barbiturates, omeprazole, tiopronin, terbinafine, benfluorex, pyribenzamine, penicillamine, isotretinoin, metronidazole, captopril, lisinopril, imatinib mesylate, and adalimumab. The latter case cleared 2 weeks after cessation of the drug, making this diagnosis more likely than pityriasis rosea associated with lowered immunity from the drug. Other agents include nortriptyline, rituximab, bupropion, nimesulide, infliximab, and ondansetron.
Cases have also been associated with bacillus Calmette–Guérin (BCG), pneumococcal, and influenza (H1N1) vaccinations.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here