Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The stiff-man syndrome (SMS, also known as stiff-person syndrome) is a rare central nervous system autoimmune disease, but is likely underrecognized. This disorder has only been rarely reported in children. Most of the available literature is in case report form, and a recent series from the Mayo Clinic reported 8 cases identified over a 25-year period. These patients were frequently diagnosed initially with other disorders, including dystonia, musculoskeletal disorders, and functional disorders. In this chapter, we will review the clinical features, both neurological and immunological, as well as electrophysiological findings. We will also discuss differential diagnosis of SMS among children. There will be some discussion regarding pathophysiology. Finally, we will review current treatment options (symptomatic and immunotherapeutic) and prognosis.
In 1956 Moersch and Woltman reported 14 patients (all adults) who presented to the Mayo Clinic with chronic fluctuating truncal and limb muscle rigidity and spasms; 8 were men. They encountered their first case in an Iowan farmer in 1924, and acquired the other cases over the subsequent 32 years before publishing the report. The authors named the disorder “stiff-man syndrome,” noting that muscle involvement was “fairly symmetrical,” the lower extremities were affected more commonly than the upper extremities, and proximal limb segments were affected more severely. In 12 patients, back and abdominal muscles were involved (“hard,” “firm,” “board-like”) but 2 lacked truncal symptoms. There was frequently significant disability from painful spasms and falls, and many patients had fixed spinal deformities from long-standing rigidity. Chest wall spasms caused respiratory impairment in some patients. In 1963, it was reported that diazepam brought about considerable improvement in stiffness and spasms of affected patients, a phenomenon that was incorporated into diagnostic criteria for SMS. Distinctive neurophysiological findings were also reported and localized the disorder to hyperexcitability of brainstem and spinal motor neurons. In 1988, De Camilli and colleagues recognized that SMS was an organ-specific autoimmune disorder, recognizing seropositivity of most patients for a neuroendocrine autoantibody specific for glutamic acid decarboxylase 65-isoform (GAD65). This same antibody was subsequently recognized to also be a marker of a much more common autoimmune disease, type 1 diabetes mellitus.
Since the original description, more limited forms of SMS (sometimes paraneoplastic) including “stiff-limb” and “stiff-trunk” have been reported (often GAD65 Ab seronegative). Furthermore, some with high GAD65 Ab values have other neurological disorders coexisting with SMS, including epilepsy, cerebellar ataxia, brainstem disorders, and myelopathies. Another form has been described as rapidly progressive with diffuse CNS findings known as progressive encephalomyelitis with rigidity and myoclonus (PERM). These patients frequently have whole body symptoms, and eye and bulbar signs in addition to more classic SMS symptoms. Other autoantibody markers that have been discovered and aid the diagnosis of the SMS spectrum include amphiphysin-IgG (only in adults), detected in patients with paraneoplastic neurological autoimmunity associated with breast adenocarcinoma or small-cell lung carcinoma, and glycine receptor α1 subunit (GlyRα)-IgG, which was first encountered in patients with the PERM phenotype but is also encountered among 20% of patients with SMS, both GAD65-IgG positive and negative cases.
The pediatric stiff-man literature is limited to 1 series of 8 patients and 12 other reported cases, and many of the data discussed herein also pertains to the adult literature.
SMS is very rare, even in adults; in one review, the prevalence was estimated at approximately 1 in 1,250,000. In one large series of stiff-man patients, 68% were female and 90% were white, with a median age of symptom onset at 40 years old. In that series, 67% had at least one additional autoimmune disease, 43% had type 1 diabetes mellitus and 35% had autoimmune thyroid disease. The frequency of SMS in the pediatric population is unknown. Clardy et al. identified only 8 patients with pediatric-onset SMS in the Mayo Clinic from 1984 to 2012, representing 5% of the total SMS cases during that time period. Of these patients, the median age of onset was 11 years, and half were female. Half of those children had type 1 diabetes mellitus. Only 12 additional individual cases of pediatric SMS have been reported in the literature.
Convergent clinical, serological, and electrophysiological evidence is indicative of SMS being an autoimmune disease of CNS inhibitory synaptic signaling. Patients have symptoms of exaggerated startle, and continuous muscle contractions, consistent with a loss of inhibitory control at a brainstem and spinal cord level. More often than not, SMS patients have a coexisting autoimmune disease, including type 1 diabetes. There is an association between SMS and the DQB1*0201 allele, which is also a susceptibility allele for type 1 diabetes mellitus. Also, 80% of SMS patients are seropositive for GAD65-IgG, an antibody that is also detected in 90% of patients with type 1 diabetes. This serological finding implicates autoimmunity directed against GABA synthesis. Other antigens pertinent to inhibitory synaptic transmission implicated in SMS pathophysiology include the α1 subunit of the glycine receptor, GABA receptor associated protein (GABARAP), gephyrin (1 patient described), and amphiphysin (relevant to paraneoplastic disorders in adults).
GAD65-IgG ( Figure 11.1 ) recognizes a cytoplasmic-facing synaptic vesicle protein, and thus is unlikely to be pathogenic. Consistent with this, responses to antibody-depleting therapies in SMS patients seropositive for GAD65-IgG are variable, and post-therapy normalization is uncommon. IgGs specific for cytoplasmic and nuclear autoantigens are considered surrogate markers of inflammatory organ-specific autoimmune disorders mediated by cytotoxic T cells, a hypothesis supported by autopsy findings in some SMS patients. In contrast, the glycine receptor is a cell surface antigen, and thus is accessible to antibody. Although the pathogenicity of GlyRα1-IgG is yet unproven ( Figure 11.2 ), detection of this may predict immunotherapy-responsiveness. In one series, immunotherapy responses were noted more frequently in GlyRα1-IgG-positive SMS patients (6 of 7 improved) than in seronegative patients (7 of 25 improved). Similar responses to immunotherapy have also been documented for other neurological disorders characterized serologically by IgGs that target neuronal cell surface proteins. GABARAP antibody is detected primarily in patients who are also GAD65-IgG seropositive.
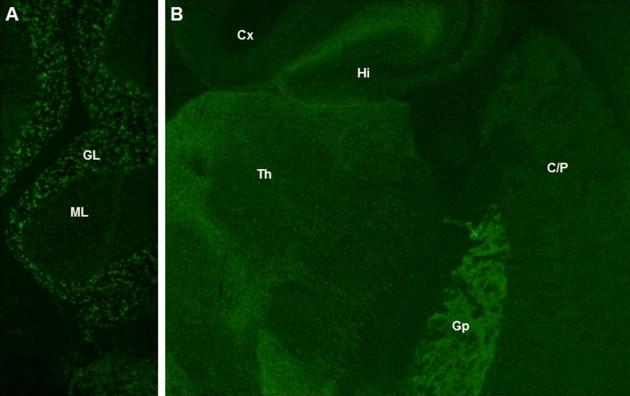
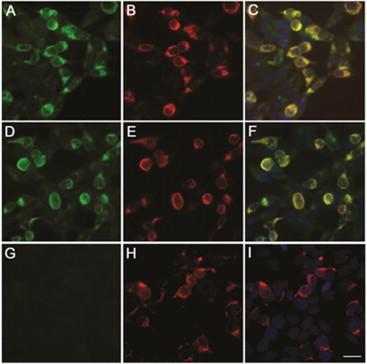
Neurophysiology studies of SMS confirm a CNS hyperexcitable phenotype with impaired inhibition of H-reflexes and excessive, poorly habituating motor activity in response to acoustic startle.
Symptom onset is characteristically acute or subacute in all (see Case Examples 11.1–11.3 ). Lower extremity stiffness or spasms causing gait difficulties are the most common initial symptoms. Other axial presenting symptoms include isolated severe lumbar paraspinal muscle spasms resulting in marked lumbar lordosis, intrascapular muscle spasms, and midthoracic back pain. Reported presentations also include exaggerated startle, limb muscle cramps, and breathing difficulties when exercising. Respiratory failure due to chest wall spasms is a rare complication. Abdominal pain due to muscle spasms is also common. Emotional stress, startle and anxiety may precipitate or worsen symptoms, and can lead to falls. One patient has been reported to have exaggerated startle and limb symptoms brought on by being asked to speak in front of the class at school. This frequently resulted in falling over “stiff as a board” without protective reflexes, resulting in facial injury.
A 7-year-old African American girl with a history of type 1 diabetes mellitus (diagnosed at age 3) developed right foot pain, followed by pain through the remainder of the right lower extremity, followed by the left lower extremity over a 3-week period. She had associated muscle stiffness and frequent muscle spasms, rendering her unable to walk. She was found to have GAD65 antibody positivity (285 nmol/L; normal value, ≤0.02 nmol/L). Diazepam provided no significant improvement. She had initial improvements with intravenous immune globulin (IVIg), which were unsustained. She was treated with baclofen, and then with plasma exchange, which gave significant benefit along with baclofen. Examination after plasma exchange and baclofen was normal aside from mild decreased distal pinprick sensation (EMG under conscious sedation was normal).
A 17-year-old male patient, previously very active, developed severe aching pain and spasms in the musculature of his thoracic and lumbar back 3 years prior to presentation. The discomfort was constant, with superimposed exacerbations lasting up to 3 hours associated with increased physical activity. He had received multiple treatments with muscle relaxants, benzodiazepines, gabapentin, amitriptyline, and botulinum toxin, with incomplete response, though he found baclofen (at a total of 60 mg/day) and botulinum toxin to be most helpful. On examination, he had tenderness as well as tight upper and midthoracic muscles; on EMG, he was unable to fully relax thoracic paraspinal muscles. His GAD65 serum antibody level was found to be elevated at 0.06 nmol/L. He was diagnosed with SMS and was treated with IVIg, which was helpful, followed by prednisone and plasma exchange (in addition to symptomatic therapies) with marked improvement.
A 14-year-old girl with a history of both type 1 diabetes mellitus and autoimmune thyroid disease had begun to walk late at 17 months, and since that time had been noted to walk abnormally in relation to her right leg. She then developed right knee pain 3 years prior to presentation. Six months prior to presentation, her right foot started to turn inward. She was found on examination to have hyperreflexia, worse on the right than on the left, and rigidity of the right lower extremity with slightly decreased muscle bulk. Trials of carbidopa/levodopa and tizanidine therapies did not help her symptoms. An EMG under conscious sedation was normal. GAD65 antibodies were significantly elevated, at 3847 nmol/L. She was subsequently treated with IVIg with significant improvement, though its use was limited by side effects. Ultimately, she had surgery for correction of her right foot deformity.
The principal exam findings are of stiffness (increased muscle tone) in affected parts of the body, usually limbs and lower back. The classic exaggerated lumbar lordosis should not resolve completely with forward flexion or lying supine. Paraspinal muscles may be hypertrophied and tense to palpation. At initial evaluation, myelopathic findings (usually isolated brisk deep tendon reflexes) are documented in over half of patients. Rare manifestations include a focal jerking limb. Coexisting neurological disorders (reported only in adults to date) include epilepsy, parkinsonism, and peripheral neuropathy (mainly in those with diabetes mellitus).
Three main phenotypes are recognized. Classic SMS patients have stiffness and spasms largely confined to the low back and proximal lower extremities. Variants may include patients with stiffness and spasms confined to one lower extremity (“stiff-limb”) or the axis (“stiff-trunk”), and patients with whole body spasms. This latter group of patients includes those with progressive encephalomyelitis with rigidity and myoclonus (PERM), a severe disorder characterized by one or more of the following: encephalopathy, brainstem signs, and myelopathy, as well as axis and limbs stiffness and spasms. All three phenotypes have been reported in children.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here