Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Spine ultrasound is used in the evaluation of neonates and infants (up to 3-4 months) with high- or moderate-risk cutaneous manifestations of closed spinal dysraphism, clinical signs of a tethered cord, or congenital anomalies that are frequently associated with spinal dysraphism.
Additional indications for spine ultrasound include evaluation after failed lumbar puncture, guidance for lumbar puncture, and intraoperative guidance.
The conus medullaris is usually at L1-2 and can extend to the superior endplate of L3.
Neural tube defects can be open (nonskin-covered) or closed (skin-covered) spinal lesions, with or without a subcutaneous mass or other cutaneous stigmata.
Understanding anatomy, embryology, normal variants, and scanning technique is important for accurate diagnosis.
In normal infants, the spinal canal and contents can be visualized because the nonossified state of the posteromedian intraneural synchondrosis provides an ample acoustic window ( Fig. 49.1 ). The abnormal configuration of vertebral bodies in infants with certain dysraphic abnormalities opens the window even further. Although magnetic resonance imaging (MRI) is considered the examination of choice when evaluating the spine in children and adults, sonography of the spine in the newborn period can reveal details that are difficult to define with MRI. Several authors have recommended ultrasound as the initial imaging modality in patients younger than 6 months suspected of having a spinal disorder. Others have recommended ultrasound in asymptomatic infants with cutaneous stigmata and MRI in the presence of deficits and cutaneous stigmata. Fatty masses, cord position, and relationship of the cord to any mass, as well as presence or absence of nerve root pulsations, can be seen clearly.
For a better understanding of spinal pathology, a general knowledge of the process of spinal cord and vertebral column formation is helpful. The trilaminar disc is formed by gastrulation. This is followed by primary neurulation, which forms the spinal cord from cervical through S5 sacral segments. Distal to this level, the conus and the filum terminale are formed by a process termed canalization and retrogressive differentiation of the caudal cell mass, also referred to as secondary neurulation.
During weeks 2 and 3, the bilaminar embryonic disc (formed by layers of endoderm and ectoderm) is converted into a trilaminar disc through the process of gastrulation. The process starts with the appearance of the primitive streak, which forms caudally, adjacent to the area that eventually becomes the cloaca. The primitive streak actively forms mesenchymal cells that give rise to the mesoderm. Some mesenchymal cells migrate laterally, and the paired anlagen eventually fuse in the midline to form the notochord process, which contains a central lumen (notochordal canal). The neuroenteric canal, which is the proximal part of the notochordal canal, normally obliterates when development of the notochord is complete.
The nervous system develops from the neural plate, which differentiates from the ectoderm overlying the notochord and paraxial mesoderm. The neural plate is composed of neural ectoderm and is contiguous with the cutaneous ectoderm on both sides. The cells at the junction of the neural and cutaneous ectoderm differentiate into neural crest cells. Primary neurulation (closure of the neural tube) begins during days 22 and 23. The lateral margins of the neural plate thicken to form the neural folds, and the thinner midline of the neural plate forms the neural groove . The neural folds bend dorsally, converge, and fuse in the midline to form the neural tube. The process of tube closure begins in the region of the craniocervical junction and extends cephalad and caudad with several fusion sites, and the posterior neuropore closes last by day 27. After closure of the neural tube, the overlying ectoderm separates from the neural tissue and fuses in the midline, forming a continuous ectodermal covering of the neural structures; this process is known as disjunction. After the neural tube has closed and separates from the superficial ectoderm, mesenchyme also migrates dorsal to the neural tube and forms precursors of the neural arches, in addition to meninges and paraspinous muscles. At the time of closure, the lateral edges of the neural plate, which form the neural crest cells, are extruded from the neural tube, move to the dorsum of the neural tube, and eventually migrate laterally and give rise to the sensory dorsal root ganglia, the autonomic nervous system, and other structures ( Fig. 49.2 ).
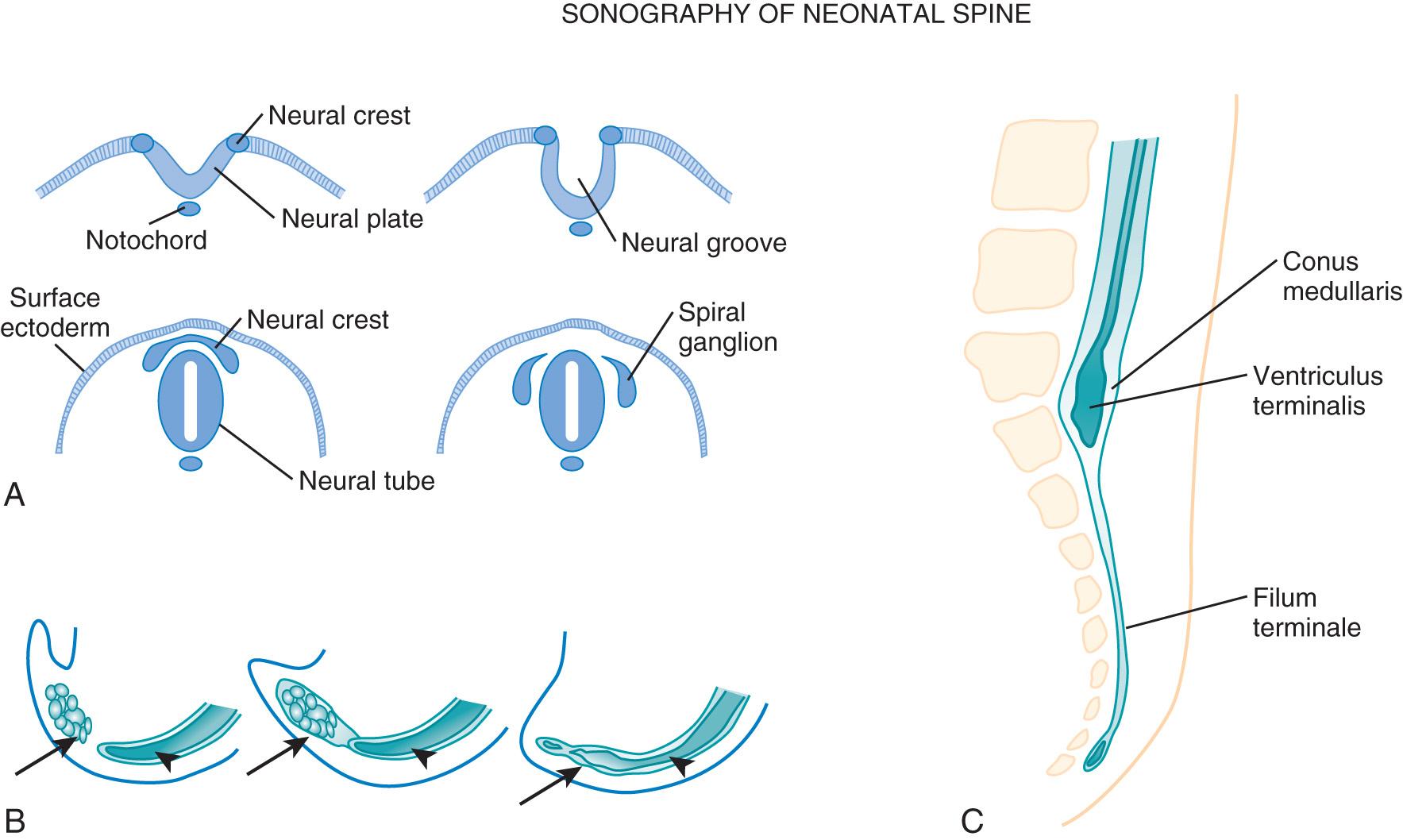
The distal neural tube is formed by secondary neurulation. Distal to the posterior neuropore, undifferentiated cells from the primitive streak form the caudal cell mass (day 30). Microcysts develop and coalesce to form a tubular structure that unites with the more distal neural tube that was formed by primary neurulation. At 38 days the process of retrogressive differentiation occurs, with decrease in size of the cell mass and central lumen of the caudal neural tube. The segment formed by this process eventually forms the filum terminale and cauda equina and ascends to form the conus medullaris and ventriculus terminalis. The regression process continues into the postnatal period with minimal migration, and the conus reaches the adult level of L1-2 by approximately 3 months of age.
The spinal column develops in parallel with the spinal cord beginning at the future occipital region and sweeps caudally. The mesenchyme lateral to the closing neural tube organizes into somites. The dorsolateral portion of each somite will become skeletal muscle and dermis, whereas the ventromedial portion will become the cartilage, bone, and ligaments of the vertebral column. The formation of the vertebrae at the caudal end of the embryo proceeds by a different process with a mass of cells of notochord, mesenchyme, and neural tissue that divides into somites.
One can readily see how failure of completion or error in organization at any of these levels might lead to anomalies in spinal formation. Complete nondisjunction of the cutaneous ectoderm and the neuroectoderm prevents mesenchymal migration and results in a nonskin-covered posterior dysraphism such as myelomeningocele. A focal nondisjunction results in a focal ectodermal-neuroectodermal tract or dermal sinus tract. In focal unilateral premature disjunction, the neural tube separates from cutaneous ectoderm before complete neural tube closure, and this allows access of mesenchymal cells to the neural groove and ependymal lining. The mesenchyme differentiates into fat and develops skin-covered abnormalities such as lipomyelomeningoceles. Filum terminale lipomas likely occur from abnormal development of the caudal cell mass. The caudal cell mass forms in close proximity to the cloaca (origin of the lower genitourinary and anorectal structures), which explains the high incidence of lumbosacral hypogenesis and tethered spinal cords in patients with anorectal and urogenital anomalies. Aberrations in gastrulation, which can interfere with primary and/or secondary neurulation, explain diastematomyelia, neurenteric fistula, caudal agenesis, and segmental spinal dysgenesis.
In general, infants are scanned in the prone position, although it is possible to scan in a decubitus position while the infant is fed with a bottle or even breast-fed. A much better scan will be obtained if the caregiver is allowed to feed a struggling infant and return to the prone position in the postprandial state. A pacifier dipped in glucose can also be used to calm the patient during imaging. If possible, the lumbar lordosis is accentuated by elevating the shoulders, to aid determination of vertebral body level by defining the lumbosacral junction. For an accurate definition of the level of the cord termination, an anteroposterior (AP) and lateral spine radiograph using a metal marker at the level of the end of the spinal cord by ultrasound can aid in documentation of the level, because the estimated level on ultrasound may be in error in the presence of spinal anomalies such as six lumbar vertebrae. In addition, elevation of the upper trunk and head can further distend the dependent caudal portion of the thecal sac. An excellent alternative approach is to place a rolled blanket under the lower abdomen in a prone position, to widen the acoustic window by separating the spinous processes. The posterior elements of the spine ossify after birth (from caudally to cranially). There is reduced penetration of sound in the lumbar spine at 3 to 4 months, and in most patients the quality significantly decreases after 6 months. In the older infant and child, parasagittal scanning can better visualize the canal contents relative to midline sagittal scanning.
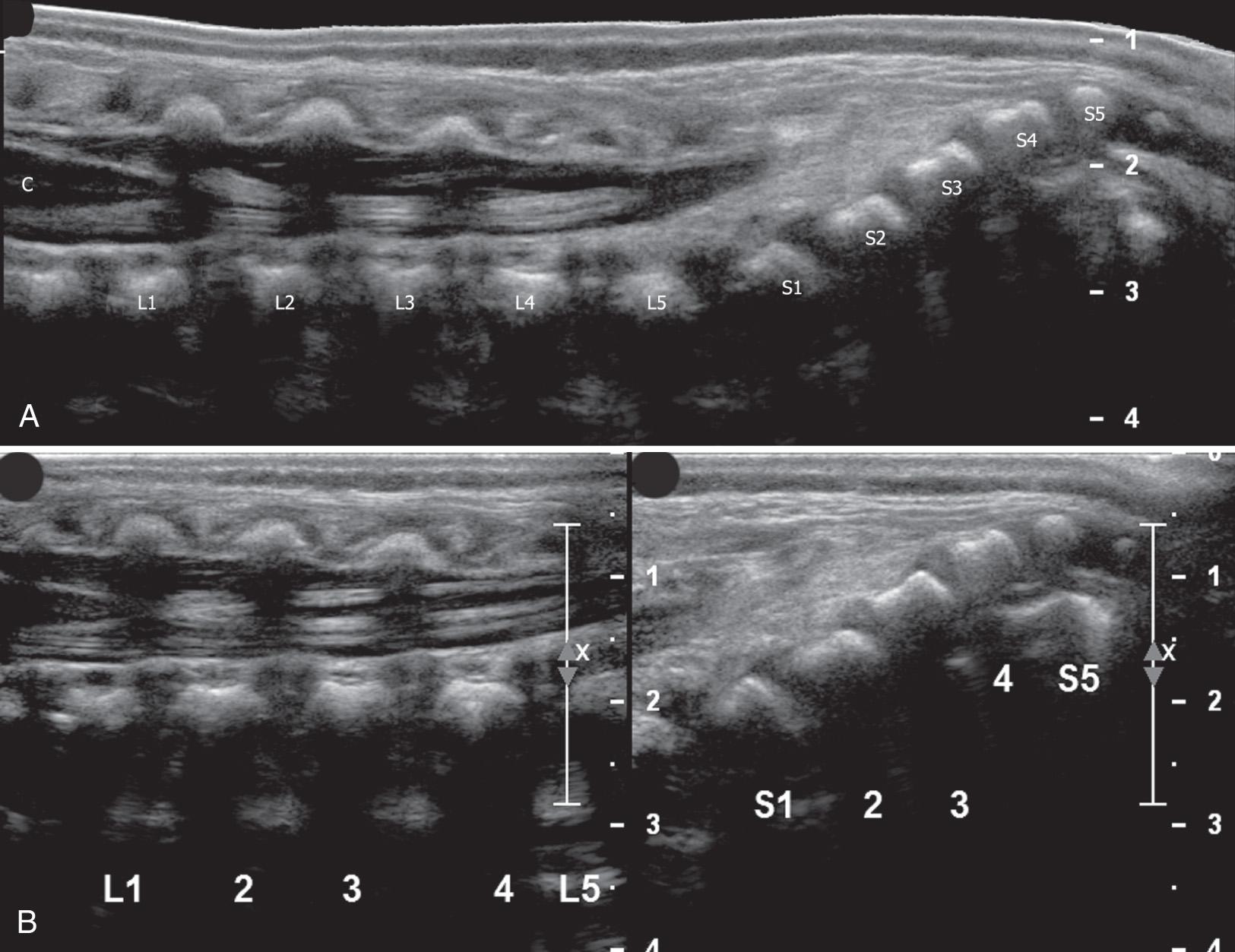
High-frequency linear transducers allow visualization of fine details of anatomy, with the additional aid of saved clips and extended–field-of-view images, which are useful for counting vertebral segments ( Fig. 49.3 ). A split screen can document the landmarks used for counting vertebral bodies and determining the level of the conus medullaris. Movement of the spinal cord and cauda equina with crying, respiration, and the cardiac cycle is seen in real time and documented with saved clips ( ). The normal movement can be seen in newborns but might not be noticeably brisk until approximately 2 postnatal months. Although different indications for imaging may require varying views, it is best to scan the entire back in both the longitudinal and the transverse planes, with right and left labels on transverse images (which can be confusing when scanning in the prone position). This allows a thorough search for contiguity of vertebral body rings, an assessment of contour and position of the spinal cord, and a survey of the paraspinous musculature and overlying skin. A standoff pad or a good amount of gel can be used to evaluate a subcutaneous mass or other structures in the near field.
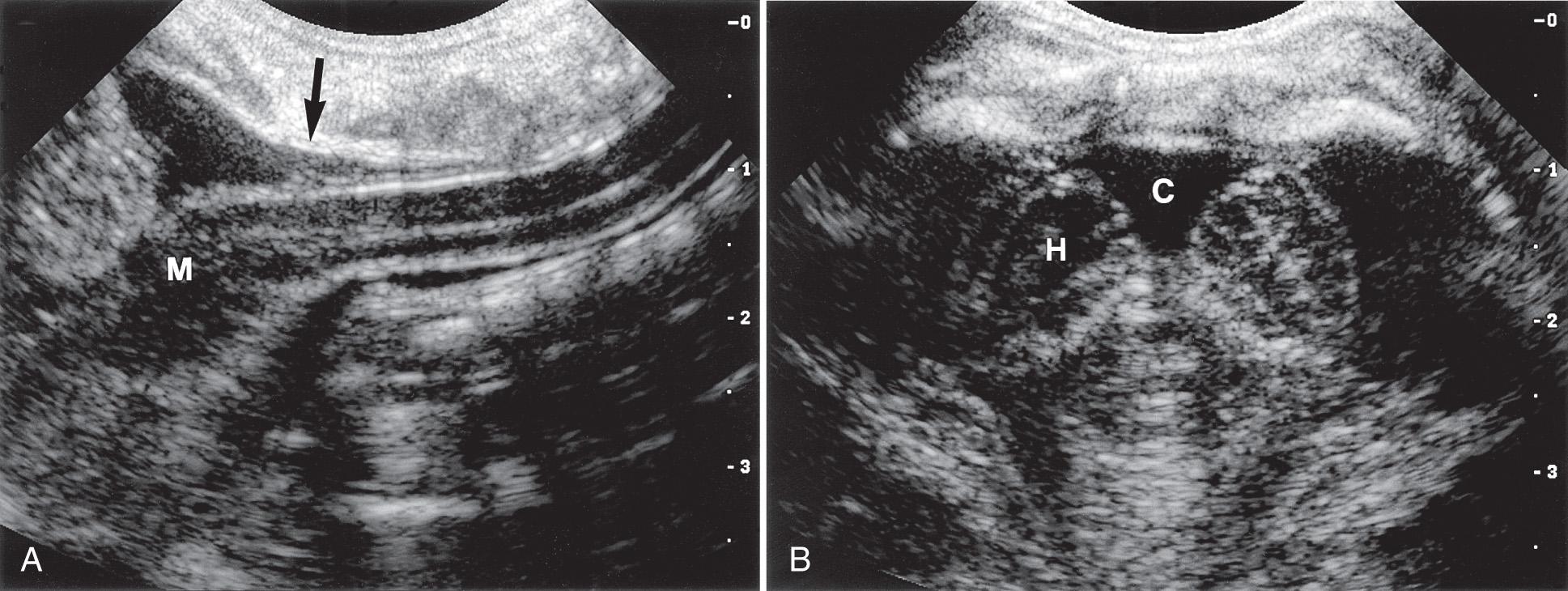
If the acoustic window is small, as occurs during the evaluation of the craniocervical junction, a smaller-footprint curvilinear, sector, or small linear transducer allows scanning at the base of the skull and through the foramen magnum. This view allows visualization of the cisterna magna, brainstem, inferior cerebellum, and proximal cervical cord ( Fig. 49.4 ).
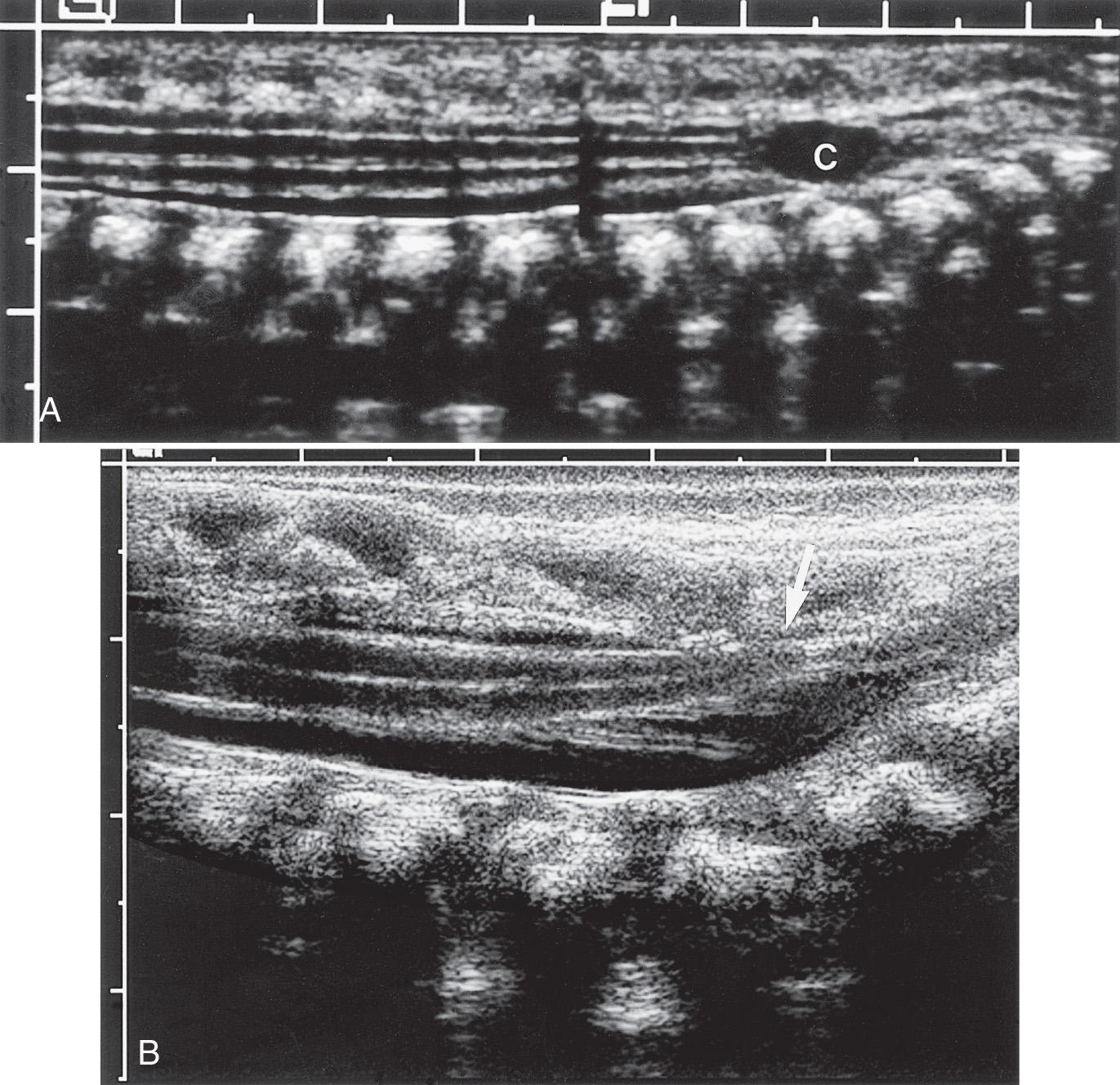
The fine anatomic display possible with sonography has been shown in correlative studies between ultrasound and specimen anatomy. Fig. 49.5 demonstrates the basic landmarks that should be visible. The cartilaginous spinous processes are hypoechoic ( Fig. 49.6 ), and the epidural space, which contains a variable amount of fat, is visible anterior to the posterior dura mater. The dura is seen on longitudinal images as an echogenic line parallel to the posterior anechoic subarachnoid space that is anterior to the spinal cord. The cord is surrounded anteriorly by anterior subarachnoid space and echogenic vertebral bodies. The cord is hypoechoic, whereas the interfaces created by the fanning nerve rootlets are echogenic.
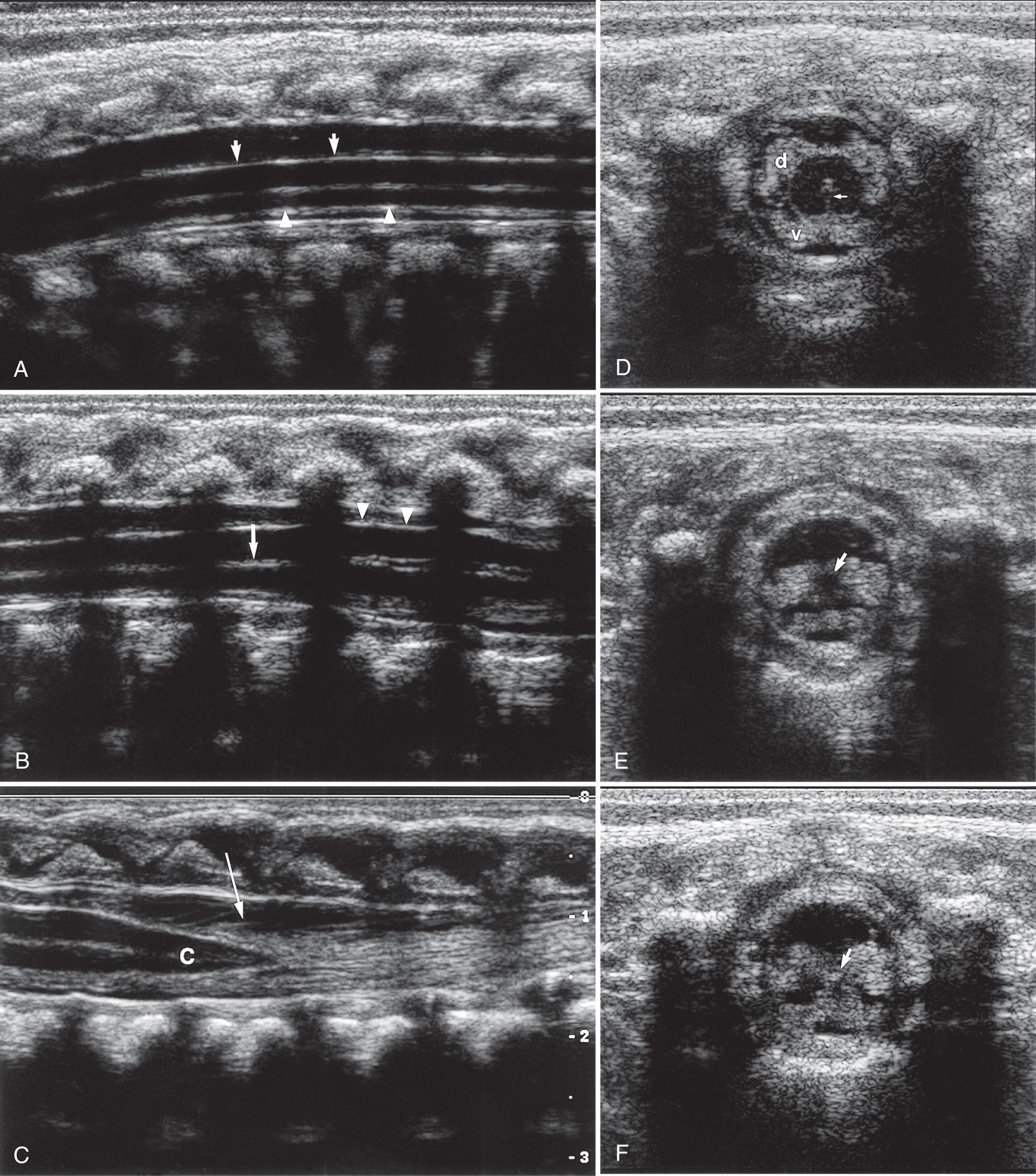
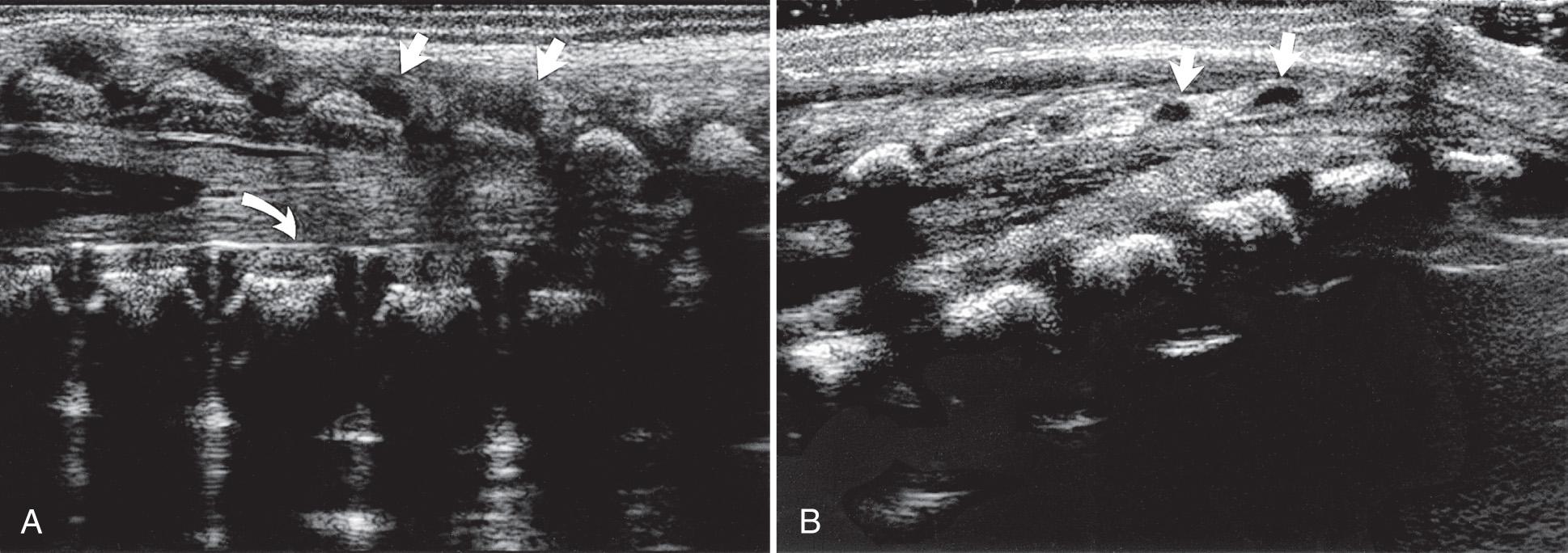
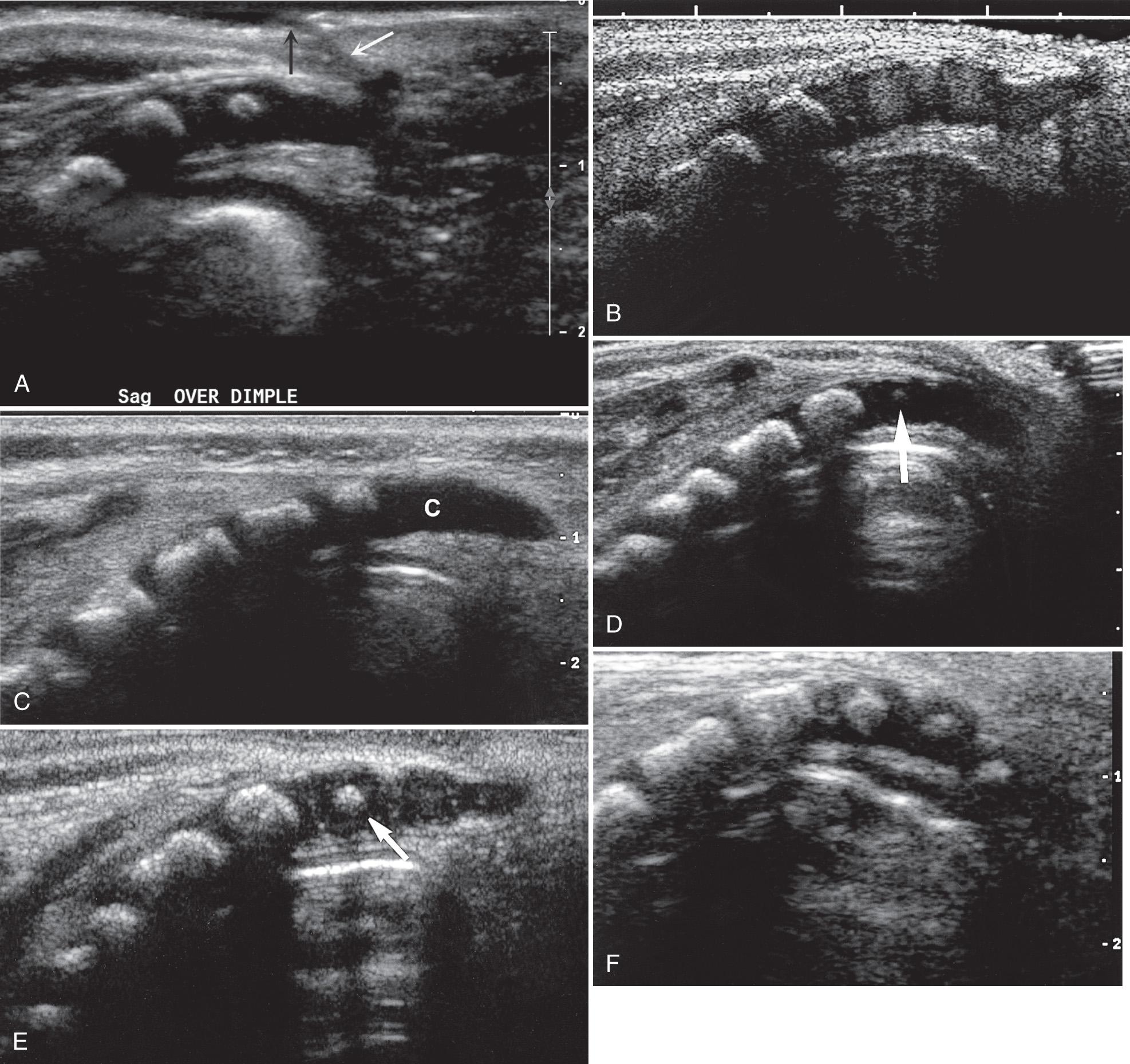
The central echo complex in the otherwise hypoechoic cord has been shown with ultrasound and histoanatomic correlation to be produced by the interface between the myelinated ventral white commissure and central end of the anterior median fissure. Images obtained with high-frequency transducers sometimes reveal a column of fluid within the center of the central echo complex within the conus medullaris. The ventriculus terminalis is a persistent fetal terminal ventricle and appears as smooth dilation of the central echo complex within the conus medullaris. This normal finding is seen often in the neonate and regresses or disappears soon after birth ( Fig. 49.7 ).

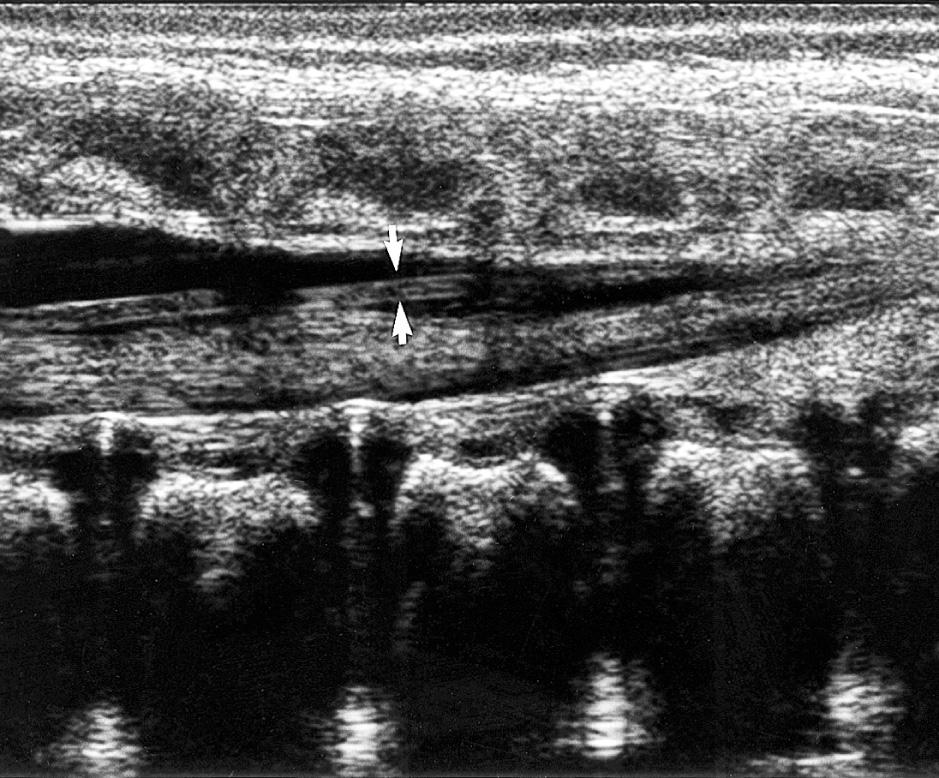
The normal filum terminale is visible and mobile with cerebrospinal fluid (CSF) pulsations. The center of the filum tends to be relatively hypoechoic compared with its bright outer margins. The filum terminale extends from the tip of the conus medullaris, crosses the subarachnoid space, and inserts on the first coccygeal vertebral body. The filum is surrounded by the cauda equina, and the normal thickness is 1 to 2 mm ( Fig. 49.8 , ). Sometimes it can be difficult to separate the filum terminale from the nerve roots in the cauda equina to obtain a good measurement. Filar cysts ( Fig. 49.9 ) can be seen within or on the filum terminale and caudal to the conus medullaris and should be considered a variant of normal development when there is no other suggestion of pathology. The incidence of filar cyst is up to 11.8% in the newborn period. Detection is inversely related to age up to 6 months. These cysts are not seen on most neonatal or adult MRI studies. There is no proven explanation for their origin; possibilities include a true cyst, a pseudocyst, an embryonic remnant, or a developmental phenomenon that regresses with age. They do not require further imaging or follow-up.
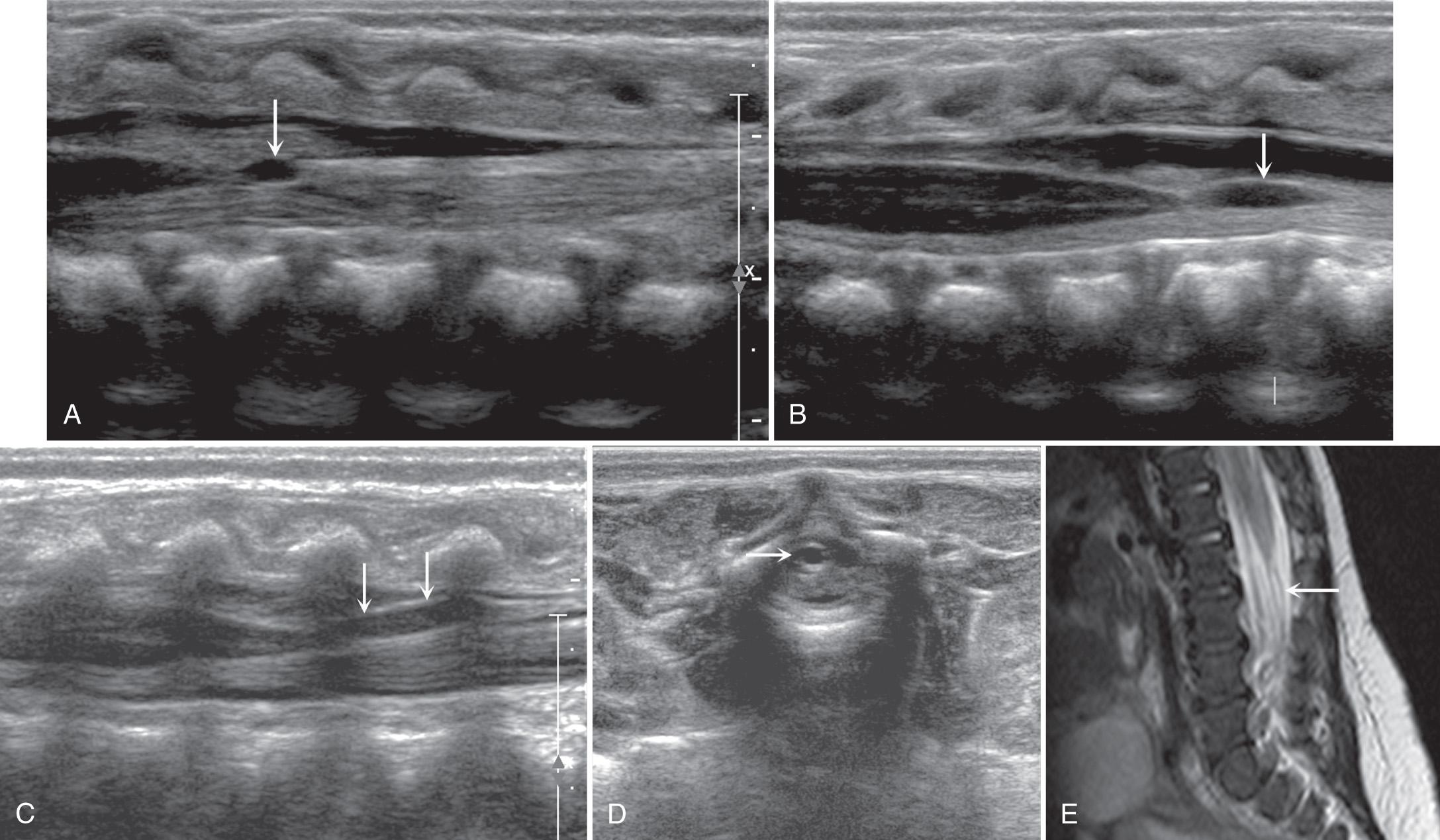
Some infants have more prominent epidural fat, which also should be considered a normal variant unless other signs suggest an abnormal fatty mass ( Fig. 49.10 ). Color Doppler ultrasound can help localize the epidural venous plexus, anterior spinal arteries, and paired posterior spinal arteries. Malposition, compression, or distention of these vessels may help to distinguish an abnormal mass within the vertebral canal from normal nerve rootlets or epidural fat.
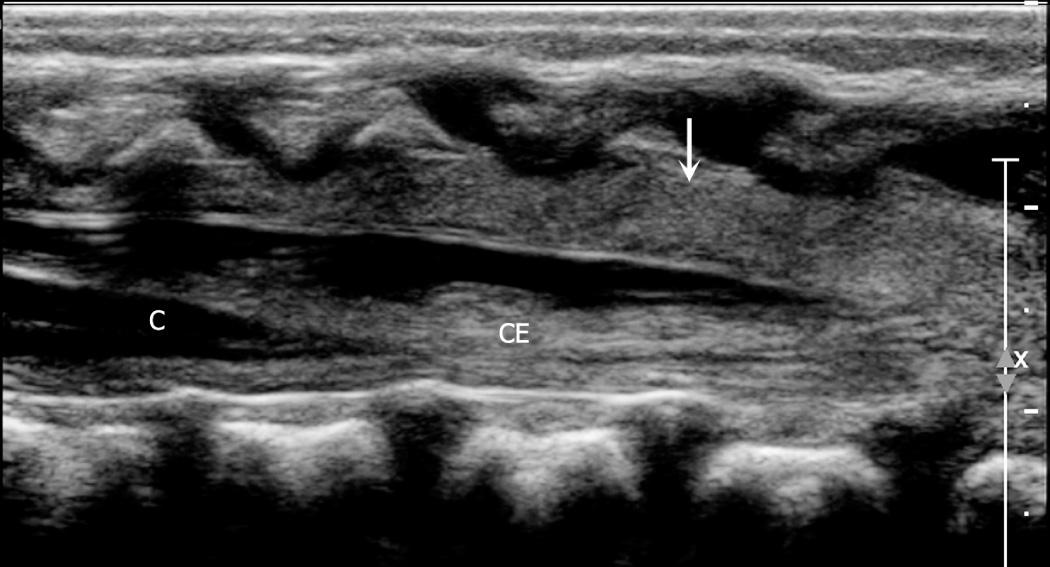
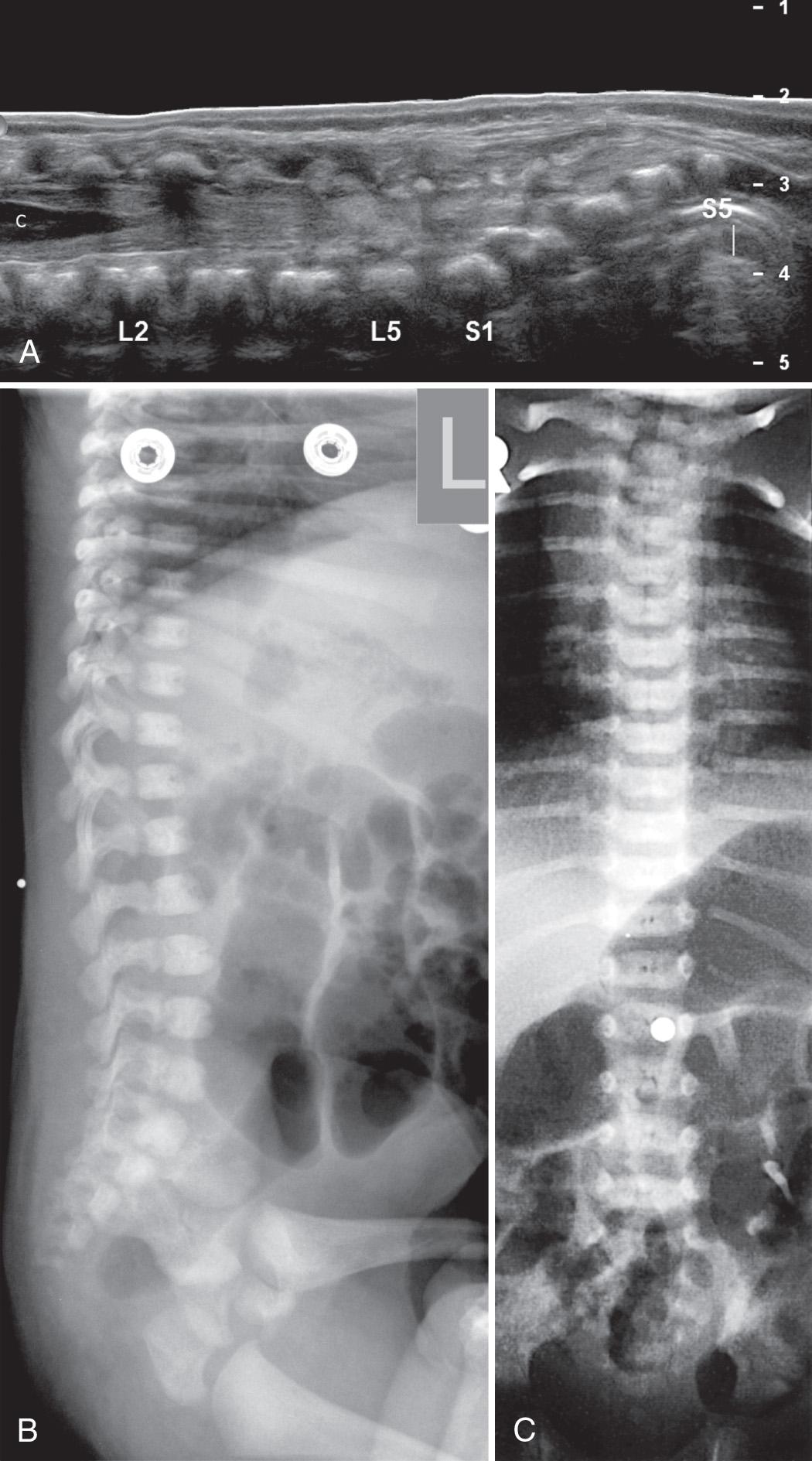
Determination of the position of the tip of the conus medullaris is the most common indication for spinal ultrasound and should always be included in a neonatal spine sonographic examination. Approaches, all of which presume normal anatomy, include (1) finding the lowest rib (presumably the 12th rib) on lateral parasagittal scanning, following medially to the vertebral body and counting down from this level; (2) defining the longitudinal lumbosacral junction where S1 is the first vertebral body that tilts posteriorly (similar to a lateral spine radiograph) and using that as a reference ; (3) defining the distal end of the thecal sac which is usually at S2 ; and (4) counting upward from the last ossified “sacral” vertebral body. The fourth approach can be inaccurate because of the great variability in the ossification of the coccygeal vertebral bodies. In general, ossified coccygeal vertebral bodies have a rounded central nucleus, whereas sacral ossification centers take on a more squared contour. If at least two methods are applied and the vertebral assignment is the same, it is likely correct. If these methods prove problematic owing to variants of ossification or segmentation anomalies, the ultrasound transducer can be used to locate the tip of the conus, mark the skin at that level with a radiopaque BB marker, and obtain AP and lateral radiographs of the entire spine to determine the corresponding vertebral level ( Fig. 49.11 ). If this is done, a lateral film is helpful because distortion from beam angulation is less problematic. The AP spine view allows counting of ribs and vertebral bodies with accuracy. The degree of ossification of the coccyx can also be assessed with this film. As the sonographer gains experience in scanning the neonatal spine, the variability in the shape of the cartilaginous coccyx becomes evident ( Fig. 49.12 and ).
The normal conus is typically located in the ventral thecal sac on prone position, between L1 and L2 and occasionally extending to the superior endplate of L3. However, the range of normal varies in children from T10-T11 to the superior end plate of L3. On MRI, the position of the conus ascends from 20 to 38 weeks. The mean conus position is L4 from 21 to 25 weeks, L3-4 from 26 to 30 weeks, L2-3 from 31 to 35 weeks, and L2 after 36 weeks. A conus position at the third lumbar vertebra (L3) or above by the beginning of the third trimester should be considered normal. A conus tip at the L3-L4 disc space level is too low. Therefore a conus position of L3 proper in the late preterm infant is equivocal, requiring follow-up. Factors other than conus position alone need to be considered when determining the need, if any, for further evaluation with MRI. When an experienced spine sonographer finds that the tip of the conus medullaris is positioned over the upper third of the L3 vertebral body with normal nerve root pulsation, and if no other abnormalities are noted and the physical examination findings are not suspicious, we do not generally recommend further imaging.
If subsequent MRI is deemed prudent after an equivocal ultrasound examination, many pediatric neuroradiologists support waiting until the infant is at least 3 months old because there is poor tissue contrast (low signal to noise) in the newborn spine. Greater anatomic detail can be displayed with MRI in the larger infant using standard techniques.
As vertebral ossification progresses and the infant becomes more active, fewer details are visible with ultrasound.
Tethered cord syndrome is a clinical condition that can be seen with a spectrum of pathologic causes that tether the spinal cord in a lower-than-normal position and may lead to neurologic, musculoskeletal, urologic, or gastrointestinal abnormalities ( Fig. 49.13 ). The clinical signs and symptoms are age dependent and develop as a complication of open neural tube repair or as the presentation of a closed spinal dysraphism. Spinal cord injury is theorized to result from cord ischemia caused by excessive tension or stretching of nerve fibers. The signs and symptoms have also been described in patients that have a conus medullaris in a normal position, although they usually have cutaneous stigmata, vertebral anomalies, or intradural lipomas.
The treatment of a tethered cord in symptomatic patients is surgical release with total or near-total resection of the tethering mass and depending on the lesion repair of the dura. In asymptomatic patients with imaging abnormalities or in symptomatic patients with normal imaging, there is controversy with regard to treatment.
Because newborns may have no neurologic deficits on presentation, the role of ultrasound is to identify the tethering lesion or condition and evaluate the position of the conus medullaris in patients with high- or intermediate-risk cutaneous markers. High-risk cutaneous markers include focal hypertrichosis, infantile hemangioma, atretic meningocele, dermal sinus tract, subcutaneous lipoma, caudal appendage, and segmental hemangiomas in association with LUMBAR syndrome (lower body hemangiomas, urogenital abnormalities, ulcerations, myelopathy, bony defects, anorectal malformations, arterial anomalies, and renal anomalies). Intermediate-risk cutaneous markers include capillary malformations ( salmon patches or port-wine stains ). Low-risk cutaneous markers do not require routine neuroimaging or follow-up and include coccygeal dimples, light hair, isolated café au lait spots, mongolian spots, hypomelanotic and hypermelanotic macules or papules, and deviated or forked gluteal clefts.
Coccygeal dimples are present in 2% to 4% of newborns and are defined as a soft tissue depression located up to 2.5 cm above the anus, in proximity with the coccyx. They can be classified as shallow if the bottom is visible or deep if the bottom is not visible. A study of 84 infants with deep and shallow coccygeal dimples who underwent MRI at a mean age of 1 year identified fibrolipomas of the terminal filum in 16.7% of patients. The fibrolipomas were more common in patients with deep dimples. In addition, these researchers found a 7% incidence of a low conus, defined as conus below the L3 vertebral body. A study of 50 neonates who underwent ultrasound examination and MRI showed no pathologic findings in infants with simple coccygeal dimples and deviated gluteal folds. In a group of 109 infants with simple dimples, ultrasound revealed no pathologic findings that needed surgery, but did show 20 cases of low dermal tracts, which had no continuity with intraspinal structures and no cord tethering. These likely fibrous tracts track anteriorly and inferiorly from the dimple to the coccyx tip (see Fig. 49.12A ). In the group of 42 infants with deviated or split gluteal folds that were not caused by an underlying soft or bony mass, ultrasound revealed no pathologic findings. Therefore in low-risk patients, if the need for imaging is questionable but imaging is desired, ultrasound provides a reliable screening tool.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here