Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Twin pregnancies account for 3.7% of all births in the United States, and the incidence has increased over the past decades. From a developmental and genetic perspective, twins are characterized either as dizygotic , arising from two eggs fertilized by two different sperm, or monozygotic , resulting from the division of one zygote, whereby both halves generally carry the same genetic information and are of the same sex. ,
From a (patho)physiologic and management perspective, however, it is more important to determine chorionicity and amniocity ( Fig. 149.1 ). Dizygotic twins are generally dichorionic-diamniotic (DCDA). In monozygotic twins, it depends on the timing of separation: (1) if separation occurs early, the embryos are DCDA; (2) with later separation, the inner cell mass has already formed, resulting in monochorionic-diamniotic (MCDA) embryos that share a common placenta; (3) if the separation occurs after the amnion has been formed, the embryos are monochorionic-monoamniotic (MCMA); and (4) in the rare case where splitting occurs even later—after the embryonic disc has formed—conjoined twins result. ,

In monochorionic twin pregnancies, only one placenta comprising two vascular beds is formed. It has been shown that more than 95% of all monochorionic pregnancies have placental vascular anastomoses that connect the fetal circulations. It has been shown that more than 95% of all monochorionic pregnancies have placental vascular anastomoses that connect the circulations of the two fetuses; these can be arteriovenous (AV), arterioarterial (AA) or veno-venous (VV) ( Fig. 149.2 ).
![Fig. 149.2, Placental vascular anastomoses. (A) Schematic representation of a monochorionic placenta. More than 95% of all monochorionic placentas show anastomoses along the vascular equator, which can be bidirectional (arterio-arterial [AA] and venovenous [VV] ) or unidirectional (arteriovenous [AV] ). (B) Postnatal placental injection study of a monochorionic pregnancy delivered at 37 weeks of gestation in a pregnancy without complications. A>B , Flow from twin A to B; B>A , flow from twin B to A. Fig. 149.2, Placental vascular anastomoses. (A) Schematic representation of a monochorionic placenta. More than 95% of all monochorionic placentas show anastomoses along the vascular equator, which can be bidirectional (arterio-arterial [AA] and venovenous [VV] ) or unidirectional (arteriovenous [AV] ). (B) Postnatal placental injection study of a monochorionic pregnancy delivered at 37 weeks of gestation in a pregnancy without complications. A>B , Flow from twin A to B; B>A , flow from twin B to A.](https://storage.googleapis.com/dl.dentistrykey.com/clinical/ThePathophysiologyofTwinTwinTransfusionSyndromeTwinAnemiaPolycythemiaSequenceandTwinReversedArterialPerfusion/1_3s20B978032371284200149X.jpg)
AA and VV anastomoses are typically superficial on the chorionic plate, although partially hidden anastomoses have been reported in 3% to 5%. These end-to-end anastomoses allow blood flow in both directions. , Placental injection studies have shown that an AA anastomosis is present in more than 90% of uncomplicated pregnancies, and these are thought to compensate for hemodynamic imbalances from AV anastomoses.
VV anastomoses are present in only around 20% of MCDA placentas. , Their role is not fully clarified. Although an increase in perinatal mortality and MCDA complications has been reported by some, others have found that VV anastomoses may equilibrate pressure differences, and their presence may protect from volume overload. ,
In contrast to these superficial connections, AV anastomoses are formed when a cotyledon is supplied by an artery of one twin and blood is drained through a vein of the other twin, permitting only unidirectional blood flow. , The majority of monochorionic placentas have a combination of (several) AV anastomoses and superficial (AA, VV) anastomoses.
The occurrence of placental anastomoses and their specific constellation is associated with complications unique to monochorionic pregnancies. When AV anastomoses permit unidirectional flow that is not compensated by other anastomoses, fluid is shifted chronically from one fetal circulation to its twin’s circulation. On the other hand, bidirectional (AA and VV) anastomoses may permit drastic fluid shifts during brief episodes of bradycardia or hypotension of one fetus, where a large volume may be shifted from the fetus with the normal circulation to the bradycardic/hypotensive fetus within a short period of time, potentially leading to brain injury or single/dual fetal demise. ,
Chronic net transfusion of fluid from one fetus to its twin through AV anastomoses that is not compensated by AA anastomoses or oppositely directed AV anastomoses causes hypervolemia in one (the “recipient” fetus) and hypovolemia in the other (the “donor” fetus). This triggers a cascade of events in both twins that ultimately result in twin-twin transfusion syndrome (TTTS), a complication that occurs in approximately 10% of all MCDA twin pregnancies. TTTS typically occurs in midtrimester, between 16 and 26 weeks of gestation.
The fluid overload in the recipient causes cardiac stretch, resulting in the secretion of atrial and b-type natriuretic peptides. These increase the glomerular filtration rate, decrease tubular reabsorption, and thereby foster polyuria and polyhydramnios. ,
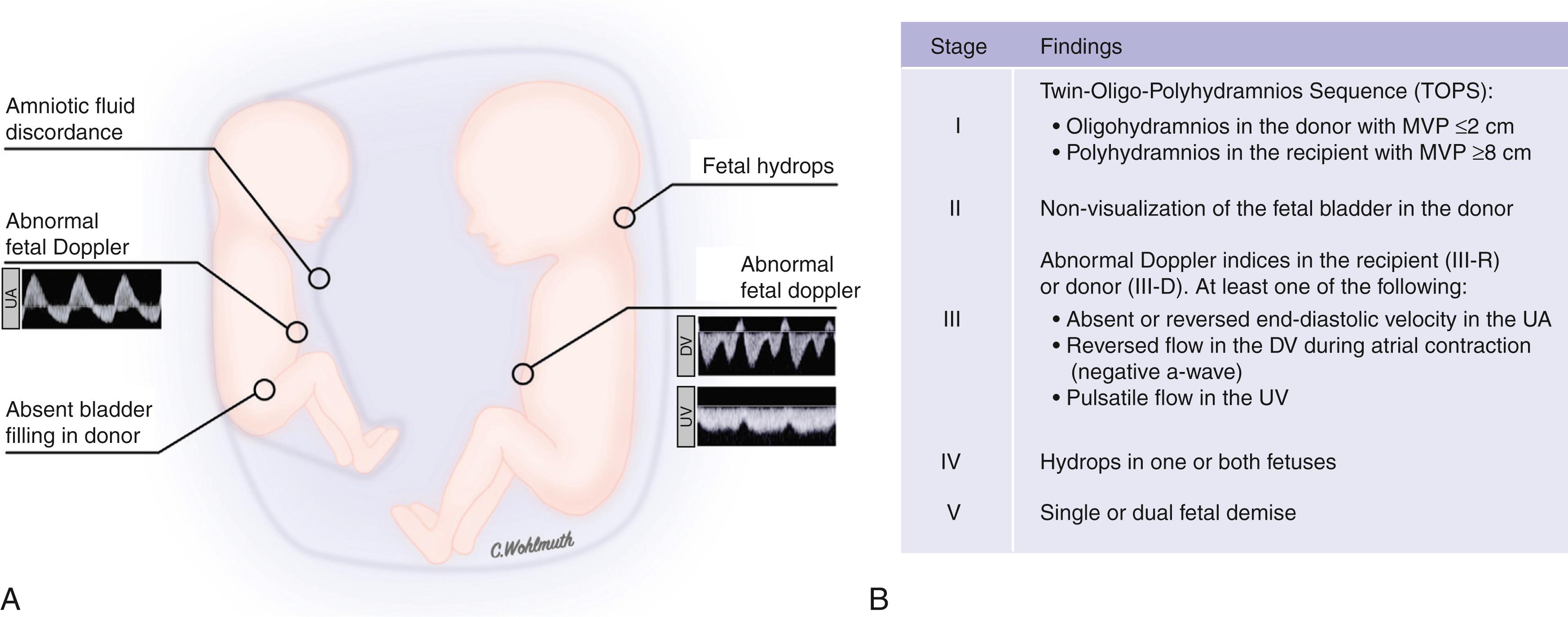
In the donor fetus, hypovolemia results in centralization of the circulation and hypoperfusion of the kidneys. This activates the renin-angiotensin-aldosterone system (RAAS), as shown by increased renin mRNA expression in donor kidneys. , In turn, angiotensin II is secreted to maintain the blood pressure in the volume-depleted donor circulation. In addition, aldosterone is secreted in response to renin and increases the tubular reabsorption of sodium and fluid, which—in combination with the renal hypoperfusion—results in oliguria and oligohydramnios. , Polyhydramnios in the recipient and oligohydramnios in the donor is sometimes referred to as twin-oligo-polyhydramnios sequence (TOPS) and constitute the diagnostic criterion for TTTS. This is usually quantified by measuring the deepest vertical pocket of amniotic fluid for both fetuses. Most centers use a cutoff of 2 cm or less for oligohydramnios and 8 cm or more for polyhydramnios independent of gestational age.
In the recipient kidney, the RAAS system is downregulated as a result of the hypervolemia, with virtually no mRNA expression found in immunohistochemical studies. However, when renin levels are measured in the umbilical cord, they are elevated in both the recipient and the donor. This provides evidence that renin is transferred from the donor to the recipient via placental vascular anastomoses. , , The continuing shift of volume and vasoactive mediators aggravates the circulation of both the recipient and donor, resulting in a vicious cycle that can cause renal failure in the donor, heart failure, hydrops, and ultimately fetal demise for one or both twins. Quintero developed a staging system based on a hypothetical sequence of observed events that continues to be used, although it has been shown that TTTS does not necessarily follow this sequence. Although regression of stage I occurs in more than 40% of cases, stages can be skipped and hydrops and fetal demise can develop rapidly ( Fig. 149.3 ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here