Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
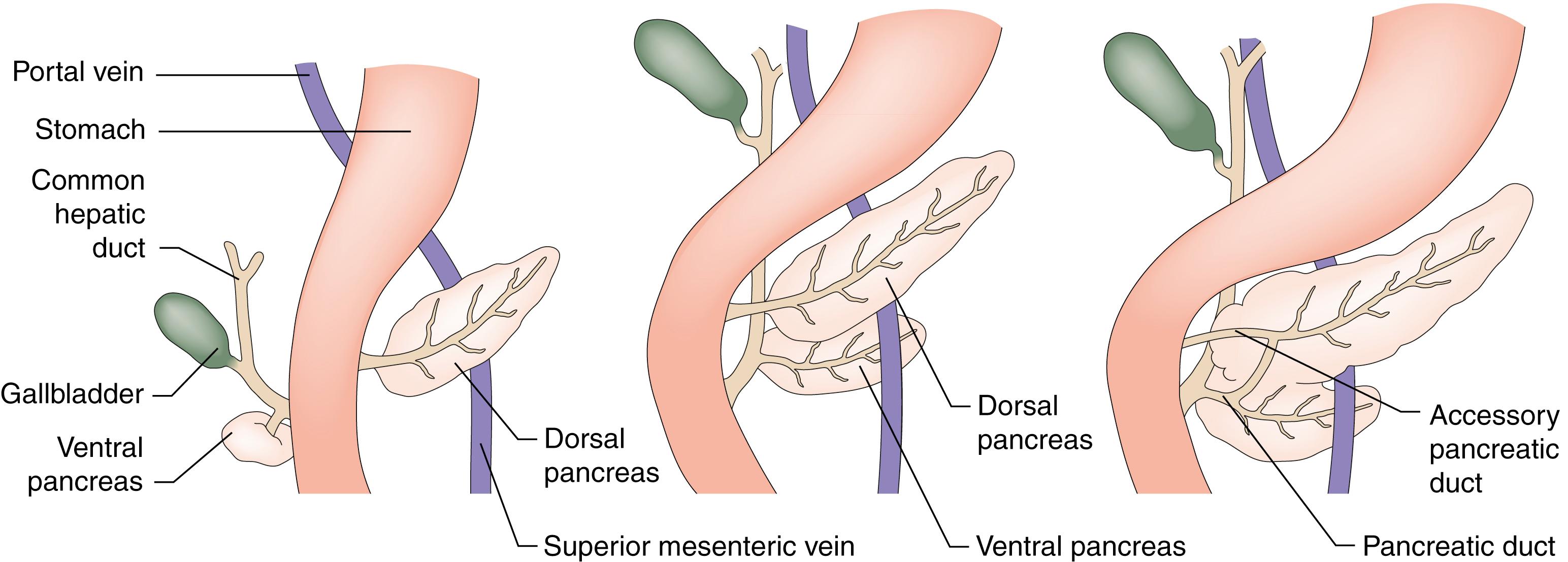
The pancreas is a glandular organ that lies in the retroperitoneum, behind the lesser sac and stomach. The pancreas develops from separate ventral and dorsal buds of endoderm that appear during the fourth week of foetal life ( Fig. 16.1 ). The ventral pancreas will become the future uncinate process and codevelops with the biliary tree, and its duct joins the common bile duct before emptying into the duodenum through the major papilla (of Vater). The dorsal bud forms the neck, body and tail of the pancreas. During gestation, the duodenum rotates clockwise on its long axis, and the bile duct and ventral pancreas pass behind it to fuse with the dorsal pancreas to form the head of the gland. The ducts draining the dorsal and ventral pancreas fuse in the head to form the main pancreatic duct (of Wirsung); any remaining dorsal duct becomes the accessory pancreatic duct (of Santorini) and enters the duodenum at the minor papilla, usually 2.5 cm proximal to the major papilla. In foetal life, the common bile duct and main pancreatic duct are dilated at their junction to form the ampulla of Vater. In extrauterine life, only 10% of individuals retain this ampulla, although most retain a short common channel between the two duct systems.
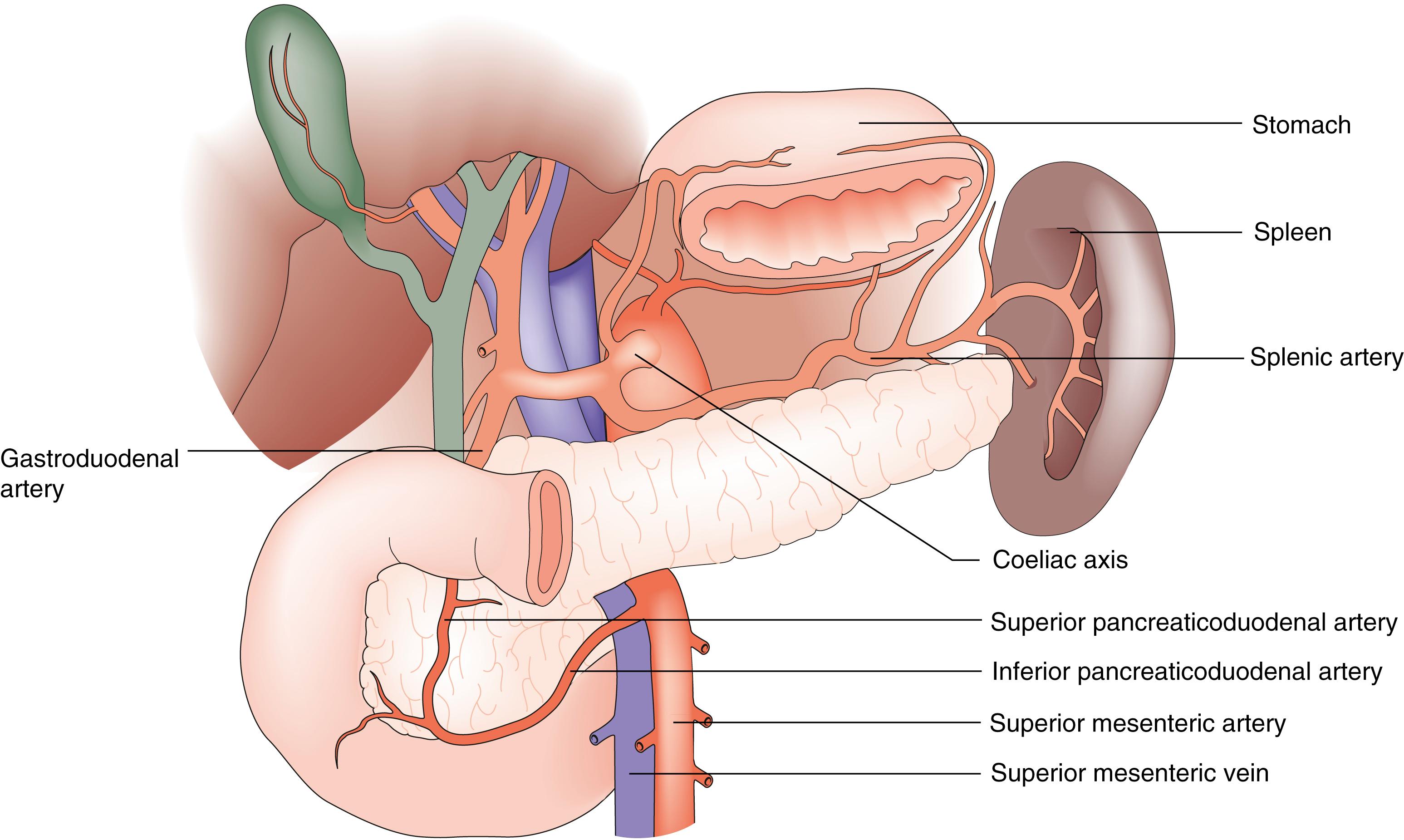
The head of the pancreas lies within the C-loop of the duodenum and shares a rich blood supply through a vascular anastomosis arising from the coeliac and superior mesenteric arteries ( Fig. 16.2 ). The venous drainage of the pancreas is via the superior mesenteric vein that passes upwards to the left of the uncinate process and joins the splenic vein behind the neck of the pancreas to form the portal vein, having received multiple direct small tributaries along its course. The body and tail of the pancreas lie in front of the splenic vein as far as the splenic hilum and receive arterial blood from the splenic artery as it runs along the upper border of the gland. The rich blood supply, friable nature and intimate relationship of the pancreas to major blood vessels, the aorta and the inferior vena cava make bleeding a significant issue after pancreatic trauma. The close association between the common bile duct and the head of the pancreas explains why obstructive jaundice is a common presentation of pancreatic cancer. The common channel within the ampulla of Vater contributes to why gallstones may give rise to acute pancreatitis.
The exocrine pancreas is responsible for digesting the food that we eat and is essential for the digestion of fat, protein and carbohydrate. The pancreas secretes 1 to 2 L of bicarbonate-rich alkaline (pH 7.5–8.8) juice each day, mostly around mealtimes. This juice neutralises stomach acid, provides bulk fluid and is particularly rich in digestive enzymes that have been synthesised and stored as zymogen granules in the pancreatic acinar cells. The key proteolytic enzyme is trypsin, which is made and released in an inactive form (trypsinogen) and normally activated only after reaching the duodenum, by enterokinase located in the brush border. Trypsin activation triggers a cascade of other proteolytic enzymes to become activated. Fat and carbohydrate starch are digested by lipase and amylase, respectively, which are secreted as active enzymes. Pancreatic enzymes require alkaline conditions, which are provided by bicarbonate secreted by the ductal epithelium.
Pancreatic secretion is stimulated by eating through hormonal and neural (vagal) mechanisms. Fat and protein entering the duodenum stimulate cholecystokinin (CCK) release, which stimulates pancreatic enzyme secretion and contraction of the gallbladder, increasing bile flow into the intestine. Acid in the duodenum releases the hormone secretin, which stimulates the pancreas to secrete watery alkaline juice.
The endocrine function of the pancreas is equally essential for life and health. Endocrine cells in the pancreas are clustered in the pancreatic islets (of Langerhans), which are distributed throughout the pancreas. The islets receive 10% of the pancreatic blood flow, despite being only 2% of the gland by bulk, and have a network capillary vasculature that facilitates hormonal communication with the neighbouring exocrine pancreas and the rapid transmission of hormones into the portal and, subsequently, systemic circulation. Islets contain four types of endocrine cells: alpha cells that produce glucagon, beta cells that produce insulin, delta cells that produce somatostatin and pancreatic polypeptide (PP) cells that produce pancreatic polypeptide. A fifth cell type has been reported: epsilon cells that produce ghrelin. Glucagon and insulin have well-established physiologic roles. The function of the other islet products is uncertain, but somatostatin and PP may serve as local (paracrine) hormonal regulators rather than as circulating (endocrine) messengers. Gastrin-producing (G) cells are not normally found in the pancreas, except in the rare Zollinger–Ellison syndrome.
Pancreatic pain is visceral and mediated through sympathetic nerve fibres that accompany the arteries to reach the coeliac ganglion and to segments 5 to 12 of the thoracic spinal cord via the greater, lesser and least splanchnic nerves. The retroperitoneum is also richly innervated with nociceptive fibres.
Annular pancreas is a rare cause of duodenal obstruction, resulting from incomplete rotation of the ventral pancreas. In approximately 5% of individuals, the ducts draining the dorsal and ventral pancreas fail to fuse, giving rise to pancreas divisum. This means that the secretions of the larger dorsal pancreas have to drain to the duodenum through the smaller accessory duct. There is no evidence to suggest a strong association between pancreas divisum and pancreatitis. Ectopic pancreatic tissue may be found at a variety of sites within the gut wall but most commonly in the duodenum, stomach and proximal small bowel. Ectopic pancreas tissue is usually asymptomatic but can become inflamed in the rare condition of cystic duodenal dystrophy (‘groove’ pancreatitis), causing pain and duodenal obstruction.
Acute pancreatitis is a sudden-onset acute inflammation of the pancreas and is one of the commonest causes of emergency admission to hospital. The incidence in the population is rising due to an increase in gallstone disease, alcohol misuse and obesity. Acute pancreatitis affects all age groups, including children, but is rare in those under the age of 18.
There are multiple causes and triggers of acute pancreatitis, as summarised in Table 16.1 .
| Class | Specific Causes |
|---|---|
| Toxic | Alcohol Medication (steroids, immunosuppressants, asparaginase) |
| Genetic (rare in acute pancreatitis, more commonly cause chronic pancreatitis) | Cystic fibrosis Hereditary pancreatitis SPINK1 mutation |
| Metabolic | Hypercalcaemia Sudden hypocalcaemia Hyperlipidaemia |
| Obstructive | Stone disease Macrolithiasis (bile duct stone) Microlithiasis (sludge) Stricture (including postacute pancreatitis, trauma) Neoplasm Infection Parasites Worms Anatomic Biliary cystic disease Choledochal cyst/(coele) Duplication cyst Duodenal obstruction Afferent limb obstructed Diverticulum Congenital anomaly Annular pancreas Anomalous pancreatobiliary junction Pancreas divisum Atresia |
| Inflammatory | Crohn disease Polyarteritis nodosa Systemic lupus erythematosus |
| Viral | Mumps, rubella, coxsackie virus, hepatitis B, HIV, COVID-19 (suspected) |
| Trauma | Major blunt abdominal trauma Penetrating trauma Endoscopic retrograde cholangiopancreatography Endoscopic ultrasound Postbiopsy Postsurgery (Whipple) |
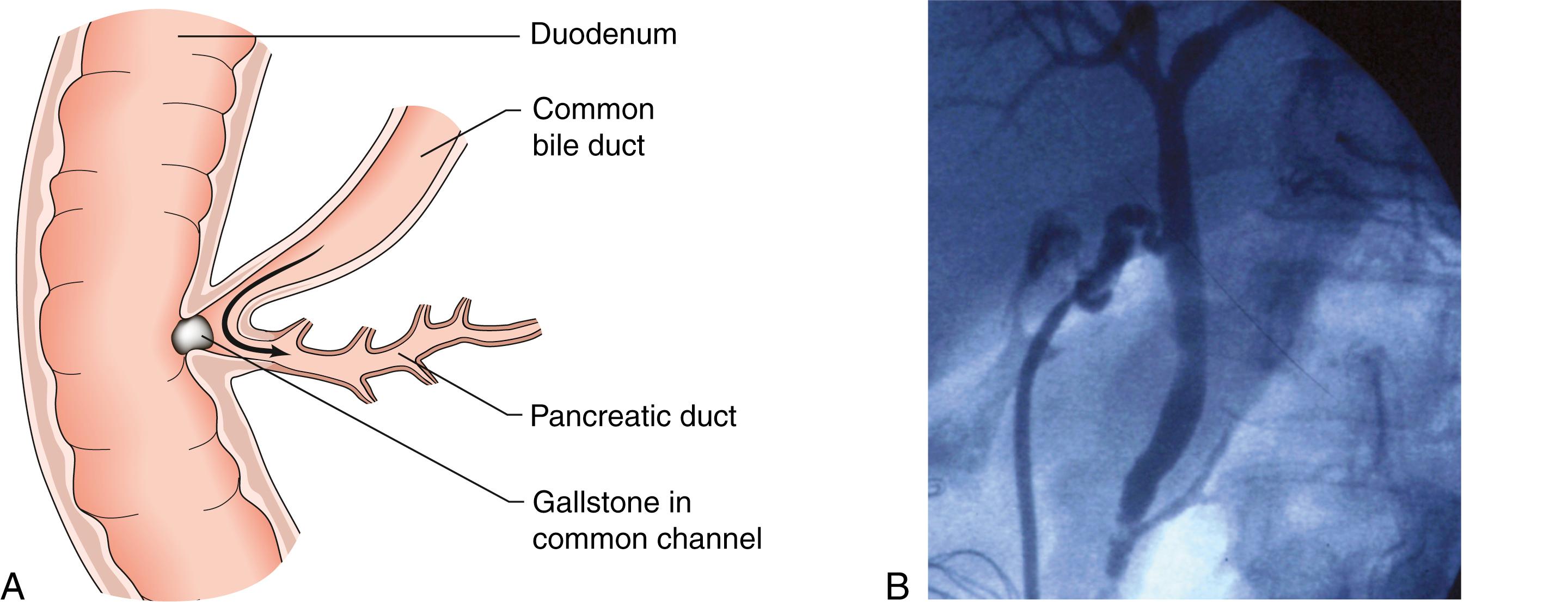
Gallstones are responsible for nearly three-quarters (75%) of cases of acute pancreatitis in some countries, in contrast to 40% of patients in the UK. Tiny gallstones or even microscopic biliary crystals (microlithiasis) are thought to account for the majority of cases for which another cause is not found. Transient impaction of a gallstone in the common channel between the common bile duct and pancreatic duct causes distal obstruction of the pancreatic duct and possible reflux of bile into the pancreas ( Fig. 16.3 ). This triggers a sequence of events within the pancreatic acinar cells, resulting in the intracellular activation of pancreatic enzymes, acinar cell damage and pancreatic inflammation.
The proportion of cases linked to alcohol varies in different parts of the world according to alcohol consumption habits but is in the region of 40%. The precise molecular mechanism(s) responsible is uncertain, but some alcohol metabolites can trigger premature acinar cell enzyme activation. There is no direct relationship between the quantity of alcohol consumed and the risk of pancreatitis (in contrast with alcoholic liver disease), but alcohol-associated acute pancreatitis usually occurs in people who consume more than 50 g of ethanol per day, about 5 standard drinks.
Acute pancreatitis occurs in approximately 5% of patients undergoing endoscopic retrograde cholangiopancreatography (ERCP) and can range from mild to life-threatening. Prophylaxis with diclofenac is often given, but the efficacy is unknown despite prominent reports in the literature. Trauma, particularly blunt abdominal trauma, may cause pancreatitis or pancreatic duct disruption. Hypercalcaemia and sudden hypocalcaemia are rare causes of pancreatitis and usually secondary to hyperparathyroidism or its treatment. Hyperlipidaemia, due to familial hypertriglyceridemia, is a rare cause, and treatment is directed at the underlying cause. A small number of patients may have a family history of acute pancreatitis.
Viral infections (mumps, Coxsackie virus, rubella, measles, hepatitis B, cytomegalovirus and more recently, COVID-19) are reported viral causes of pancreatitis, and viral infection is the most common cause of acute pancreatitis in children. Ascaris worms are a relatively common cause of pancreatitis where this parasite is endemic, triggered when the worm migrates out of the common bile duct through the duodenal papilla. It is often difficult to prove a causal association with medication, and other causes should always be considered. Corticosteroids, azathioprine, B-mercaptopurine, mesalazine, asparaginase and simvastatin are responsible in the best-documented cases.
Pancreatic neoplasms can obstruct the pancreatic duct and lead to acute pancreatitis. These tumours can be small and thus easily missed, even by computed tomography (CT). The presence of unexplained pancreatic duct dilatation or other suspicious clinical or radiologic appearances should prompt further investigation, usually by repeat CT and endoscopic ultrasound.
Acute pancreatitis usually presents with constant, severe or agonising pain in the epigastrium and across the upper abdomen, with radiation through to the back. Nausea, vomiting and retching are common. Clinical examination is more variable in the extent of tenderness, guarding and rigidity than in other causes of acute abdomen, and the presence of generalised peritonism may warrant further investigation by CT to exclude other intraabdominal pathology. A systemic inflammatory response can result in fever, tachycardia, hypotension or tachypnoea and suggest severe disease. Coexistent cholangitis is important to consider in cases due to an impacted gallstone because this directly affects management, as discussed later in the chapter.
The key to making a diagnosis of acute pancreatitis is a high index of suspicion and measurement of the serum amylase concentration. Serum lipase is an alternative. The accepted diagnostic threshold for serum amylase is 3 times the upper reference limit, but the diagnosis should be considered in any patient with a relevant history, even in the context of an elevated but nondiagnostic serum amylase. Elevated serum amylase is occasionally seen in mesenteric vascular occlusion, small bowel strangulation, perforated ulcer, or ruptured aneurysm but rarely above the diagnostic threshold for pancreatitis. If the history or clinical examination is atypical, CT should be carried out to clarify the diagnosis.
The serum amylase level falls rapidly in acute pancreatitis and has no relationship to the severity of the attack or to the resolution of the disease. Patients who present some days after the initial onset of abdominal pain may have normal or near-normal serum amylase levels, and although urinary amylase or serum lipase measurement can be helpful, diagnosis is best clarified by CT.
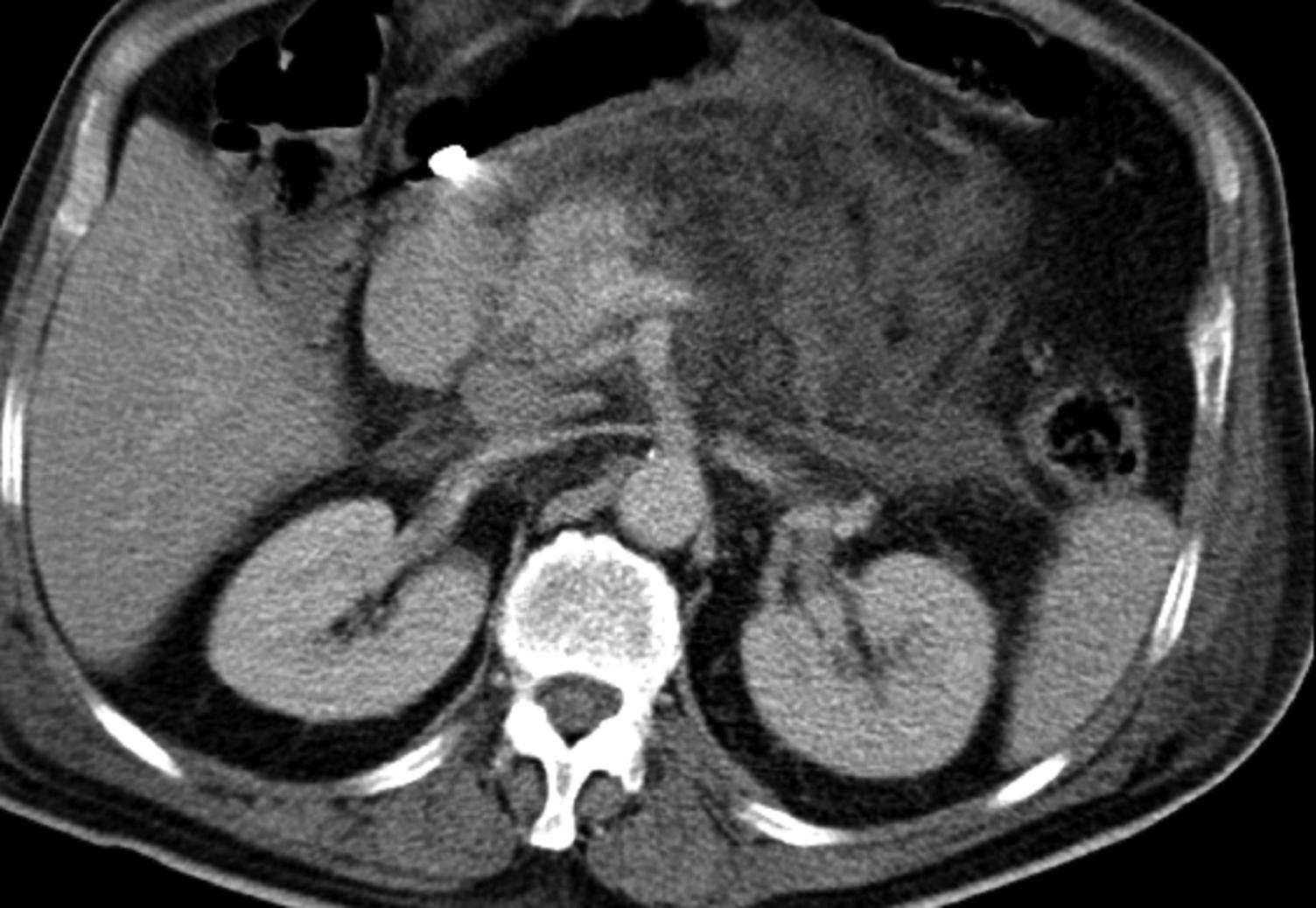
CT is useful in the diagnosis and staging of acute pancreatitis when the clinical findings or laboratory results are equivocal or when local complications are suspected that may require intervention. Later in the disease course, if the patient is deteriorating, repeated CTs can become necessary to identify evolving complications. Acute CT radiographic changes include peripancreatic oedema through to extensive pancreatic necrosis ( Fig. 16.4 ), but the role of early CT is much more important in identifying alternative diagnoses that require urgent operative intervention, such as mesenteric ischaemia or perforated viscus.
It is critical to determine the aetiology of acute pancreatitis so that further attacks can be prevented where possible. In the UK, the majority of cases are due to gallstones, and these may be present even where there is a clear history of alcohol excess. Therefore all patients with acute pancreatitis should have an abdominal ultrasound carried out. Even when no stones are identified on initial ultrasound, if a gallstone diagnosis is suspected, a repeat ultrasound at a later interval can diagnose previously undetected gallstones. In patients where no cause is found, once the acute attack has resolved, examination by endoscopic ultrasound may detect microlithiasis or a small pancreatic neoplasm.
Approximately 75% of patients with acute pancreatitis will have a self-limiting illness, for which the symptoms resolve within 48 to 72 hours and for which there is little or no associated mortality. In these patients, the focus is to identify and treat the underlying cause. In the other 25% of patients, the major challenge is to prevent death from multiple organ failure, which results in death in 20% of sufferers. The early recognition and identification of those at risk of severe acute pancreatitis have so far relied on combination scoring systems, including Ranson score, Glasgow score, and the APACHE II score ( Table 16.2 ). Biomarkers, including C-reactive protein, are useful, and more advanced precision medicine tools are in development. The practical learning point is to appreciate that acute pancreatitis can have life-threatening consequences and to recognise the risk of a severe attack.
| Ranson score | At admission:
At 48 hours:
If the score ≥ 3, severe pancreatitis is likely |
| Glasgow score (modified) | P a o 2 < 60 mm Hg/7.9 kPa A ge > 55 years N eutrophils (WBC count > 15) C alcium < 2 mmol/L R enal function: Urea > 16 mmol/L E nzymes LDH > 600 IU/L, AST > 200 IU/L A lbumin < 32 g/L (serum) S ugar BSL > 10 mmol/L Three or more positive factors detected within 48 hours of onset suggest severe pancreatitis (refer to HDU/ICU) |
| APACHE II | Acute physiology and chronic health evaluation—combined score where: Acute physiologic indices include: Evidence of acute renal failure Age, temperature (rectal), mean arterial pressure, pH, heart rate, respiratory rate; serum levels of sodium, potassium and creatinine; haematocrit; TLC; A–a gradient; Pa o 2, Glasgow Coma Scale Chronic organ insufficiency includes: Evidence of liver insufficiency, cardiovascular failure, respiratory failure, renal failure and immunosuppression |
The most important feature of severe acute pancreatitis is the development of multiple organ dysfunction syndrome (MODS). In most patients, this is identified by the presence of hypoxia, shock or more rarely, renal dysfunction. Early indicators of developing systemic inflammatory response syndrome (SIRS), which may be present on admission or evolve over 48 hours, include tachycardia, raised white cell count and/or tachypnoea. Where SIRS persists for more than 48 hours, the risk of further deterioration is high, and these patients need close monitoring for the development of respiratory and renal complications. Patients with early persistent SIRS or MODS are also at the greatest risk of developing septic complications of acute pancreatitis.
Contrast-enhanced CT can be used to identify severe acute pancreatitis because the presence of pancreatic necrosis or extensive fluid collections identifies a group at high risk of further complications. Patients with local complications or with transient organ failure that resolves within 48 hours are categorised as having ‘moderately severe’ acute pancreatitis ( Table 16.3 )
| Mild Acute Pancreatitis |
|
|
| Moderately Severe Acute Pancreatitis |
|
|
| Severe Acute Pancreatitis |
|
Most attacks will settle with conservative management. Patients should be allowed to eat if their symptoms of nausea and vomiting allow, and consideration of enteral nutrition supplementation should be given early if oral nutritional intake is low for more than a couple of days. All patients with persistent SIRS or early systemic organ dysfunction should be managed in a high-dependency or intensive care environment where adequate monitoring and specialist care are available.
Pain relief. Severe pain requires the administration of opiates. Pain can be excruciating and may require intravenous patient-controlled analgesia.
Fluid resuscitation. Due to pathologic fluid redistribution into the extravascular compartment, patients often require large volumes of fluid to maintain adequate tissue perfusion. Early adequate resuscitation is the most important consideration in the initial management. Optimal management of systemic organ dysfunction necessitates urinary catheterisation, invasive monitoring of venous and arterial pressure, and monitoring; biochemical markers of resuscitation, including lactate, may be useful. Patients with SIRS or MODS require oxygen therapy and continuous monitoring of oxygen saturation.
Nutritional support. Patients with severe acute pancreatitis who are unable to resume normal diet within 48 to 72 hours require nutritional support. This is best delivered by an enteral rather than parenteral route because trials ( EBM 16.1 ) have demonstrated that total parenteral nutrition (TPN) has a higher rate of complication and mortality. There is no evidence that nasojejunal feeding is better or safer than nasogastric feeding, but the nasojejunal route will be required where gastric or duodenal ileus or gastric outlet obstruction prevents effective nasogastric delivery. Patients should not be routinely fasted, and nasogastric tubes are not used routinely. Initial nausea and vomiting may initially limit oral intake, and when systemic complications or other factors delay the recommencement of normal diet, nasoenteric feeding is commenced at an early stage. Suppression of pancreatic secretion with octreotide or somatostatin is of no benefit.
Antibiotic usage. Acute pancreatitis is a paradigm of sterile systemic inflammation and therefore is not an indication for antimicrobial treatment. It is important to recognise that patients with severe acute pancreatitis often have evidence of SIRS, and the presence of a fever and raised white cell count are to be expected, even in the absence of infection. Antibiotics should not be given routinely. However, acute pancreatitis does predispose to secondary, or superadded, infection, and when this is proven, antibiotics, combined with adequate source control through drainage, are indicated. Coexistent cholangitis is an indication for urgent antibiotic therapy while ERCP is being arranged. Historically, antibiotic prophylaxis has been advocated by some to reduce the risk of infection of pancreatic necrosis, but multiple trials have failed to show a benefit ( EBM 16.2 ). Good antibiotic stewardship requires careful discrimination in the usage of antibiotics, and antibiotic prophylaxis in acute pancreatitis is no longer recommended.
Other measures to inhibit the inflammatory response. Severe acute pancreatitis is one of many conditions where systemic organ dysfunction is driven by the systemic inflammatory response, and there has been much experimental and clinical interest in the role of downregulation of this response as a potential treatment for acute pancreatitis. Multiple therapeutic approaches have been tried or are in development, but as yet, none has shown clear efficacy in clinical trials. Anticytokine treatment with the platelet-activating factor (PAF) antagonist lexipafant, probiotic therapy as an adjunct to early enteral feeding, protease inhibitor therapy and peritoneal lavage are all examples where trials have failed to show benefit. Given the importance of finding a suitable therapy in this indication, ongoing research and trials in this field are of critical importance.
It is clear from multiple randomised controlled trials that the liberal use of ERCP in the early stages of severe acute pancreatitis is likely to increase harm and should not be done routinely. However, in cases of acute pancreatitis due to a proven impacted gallstone in the distal bile duct and where coexisting cholangitis is present, urgent ERCP with biliary sphincterotomy is indicated and should be arranged without delay ( EBM 16.3 ). Early ERCP with stone removal can be lifesaving in the patient with cholangitis, and it is important to be alert to this in patients who also have a diagnosis of severe acute pancreatitis.
‘There is consensus in guidelines and meta-analyses that ERCP/ES is indicated in patients with acute biliary pancreatitis (ABP) and coexisting cholangitis and/or persistent cholestasis. Consensus is lacking on the role of routine early ERCP/ES in patients with predicted severe ABP.’
There is no role for surgical intervention in the first 1 to 2 weeks of an attack of acute pancreatitis, even in the face of deteriorating multiple organ failure. Complications that were managed surgically in the past are now managed by radiologic or endoscopic treatments. Acute pancreatitis is managed conservatively; laparotomy done early in acute pancreatitis for any reason is associated with a major increase in mortality.
Where gallstones are considered the likely cause, cholecystectomy is carried out after recovery from the acute attack. For mild acute pancreatitis, this is best carried out during the index hospital admission, but following a severe attack, cholecystectomy is best delayed until the patient has recovered sufficiently physically and psychologically to undergo an operation. Even after severe acute pancreatitis, it is usually possible to perform cholecystectomy by the laparoscopic route. In patients who have had no prior imaging of the common bile duct by magnetic resonance cholangiopancreatography (MRCP) or endoscopic retrograde cholangiography (ERC), it is important to carry out operative cholangiography to exclude bile duct stones. In patients considered unfit for cholecystectomy, ERC with endoscopic sphincterotomy is carried out to protect against further attacks.
The local complications of acute pancreatitis are set out in Table 16.4 .
| Time Scale | Necrosis Absent | Necrosis Present |
|---|---|---|
| <4 weeks | Acute peripancreatic fluid collection (peripancreatic fluid associated with interstitial oedematous pancreatitis with no associated peripancreatic necrosis) | Acute necrotic collection (a collection containing variable amounts of both fluid and necrosis; the necrosis can involve the pancreatic parenchyma or the extrapancreatic tissues) |
| >4 weeks | Pancreatic pseudocyst (an encapsulated collection of fluid with a well-defined inflammatory wall, usually outside the pancreas, with minimal or no necrosis) | Walled-off necrosis (a mature, encapsulated collection of pancreatic or extrapancreatic necrosis that has developed a well-defined inflammatory wall) |
| Infection | Each collection type may be sterile or infected | |
An infected acute necrotic collection is the most challenging complication of acute pancreatitis, and management is complex, requiring the input of surgeons, interventional radiologists, endoscopists and the critical care team. Infection occurs in up to 40% of patients with pancreatic necrosis, probably as a consequence of translocation of bacteria from the gut, and usually presents after the second week following symptom onset. The development of infection may be suspected where there is deterioration in systemic organ failure or where new organ failure develops in a patient more than 2 weeks after admission. However, infected acute necrotic collections are not always complicated by organ failure and may present with worsening pain and fever associated with a rise in inflammatory parameters but with little evidence of systemic illness.
An infected acute necrotic collection may be suspected by the presence of gas on CT, but this is not invariable, and the diagnosis should not be made on radiologic grounds alone. Where infection is suspected on clinical grounds, a percutaneous drain should be placed under CT guidance. Multiple radiologic drains can be placed to obtain adequate control of loculated collections.
Following initial percutaneous drainage of infected pancreatic necrosis, patients will commonly show signs of initial improvement but may subsequently deteriorate as drains block with necrotic debris. Rather than proceeding to conventional surgical debridement, the consensus nowadays is to upsize the drains under radiologic guidance. Other approaches have been described to facilitate the removal of necrotic debris via the percutaneous drain track by radiologic, laparoscopic or endoscopic means. Retrospective studies support the view that initial percutaneous drainage followed by definitive intervention if required (the ‘step-up’ approach) is associated with lower mortality and fewer severe systemic complications than conventional surgery alone, and this has been confirmed in a prospective randomised trial.
The retrogastric position of the pancreas allows endoscopic transgastric drainage of the infected acute necrotic collection, usually under the control of endoscopic ultrasound and often with the placement of pigtail or double-flanged self-expanding metal stents. This is becoming the preferred approach when the anatomy allows. All drainage procedures should be deferred where possible until the latest possible time permitted by the clinical picture to allow the collection to become organised at the walls and liquify at the centre. More than one approach may be used at different times in an individual patient, and it is important that patients are managed with specialist multidisciplinary advice.
Historically, wide debridement of devitalised pancreatic and peripancreatic tissue (necrosectomy) and the placement of wide-bore drains for postoperative lavage were done. Even if the operation is delayed until after the fourth week of illness, the mortality remains high, and open surgical debridement is indicated extremely rarely, if ever.
Walled-off pancreatic necrosis (WOPN) is a collection of pancreatic and peripancreatic necrotic material, pancreatic secretions and inflammatory tissue enclosed in a wall of fibrous or granulation tissue. The degree of necrosis is often variable, and management depends on symptoms, the anatomic location of the collection(s) and local expertise. By consensus, WOPN is differentiated from an acute necrotic collection by persistence for 4 or more weeks from the onset of acute pancreatitis ( Fig. 16.5 ). The commonest sites are between the stomach and transverse colon or liver.
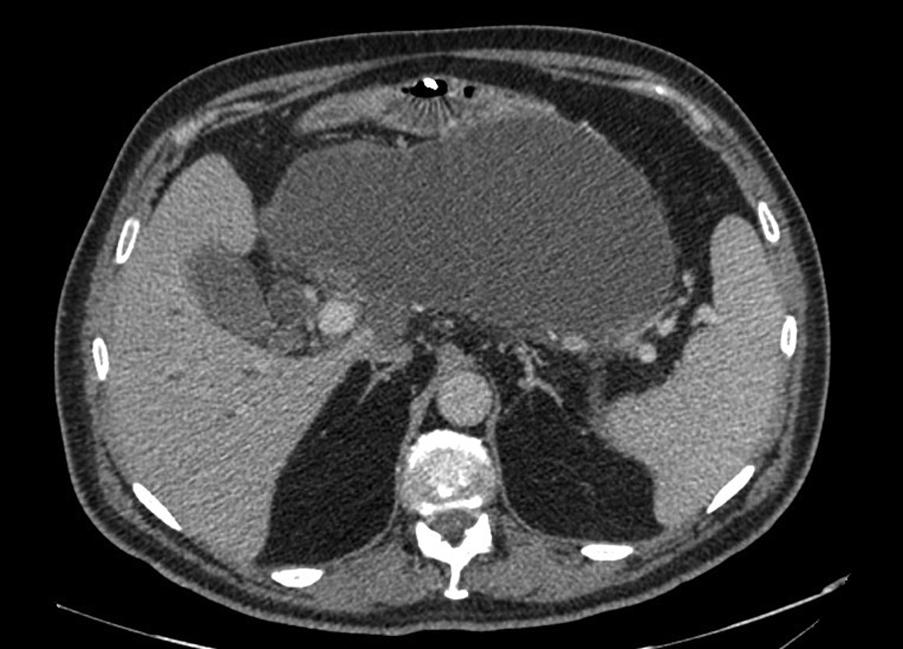
Patients become symptomatic some weeks after the episode of pancreatitis, with persistent or intermittent abdominal discomfort and mild to moderate hyperamylasaemia. Larger collections may compress neighbouring structures to cause vomiting and obstructive jaundice. Some collections become so large that they are palpable and, in some cases, visible. Their presence is not in itself an indication for surgical treatment, but intervention is normally required if enlarging beyond 6 weeks, if infection develops or in the context of symptoms.
The intervention of choice is endoscopic cyst-gastrostomy or cyst-duodenostomy under ultrasound guidance ( Fig. 16.6 ). Self-expanding metal stents improve the drainage of necrotic debris and facilitate multiple endoscopic cyst lavage and necrosectomy procedures. A plastic double-pigtail stent is sometimes placed through the metal stent to avoid blockage. Haemorrhage is the commonest complication. Percutaneous drainage followed by percutaneous necrosectomy or one of the other approaches for infected pancreatic necrosis can also be employed. Surgical drainage by pancreatic cyst-gastrostomy can be achieved laparoscopically. Because the tissues holding the sutures must be firm, it is desirable to avoid surgery until at least 6 weeks after the onset of the acute attack to allow the walls of the collection to ‘mature’. The selection of the appropriate procedure is determined by the clinical presentation, the anatomic features of the fluid collection, the extent of necrosis, and local multidisciplinary expertise.
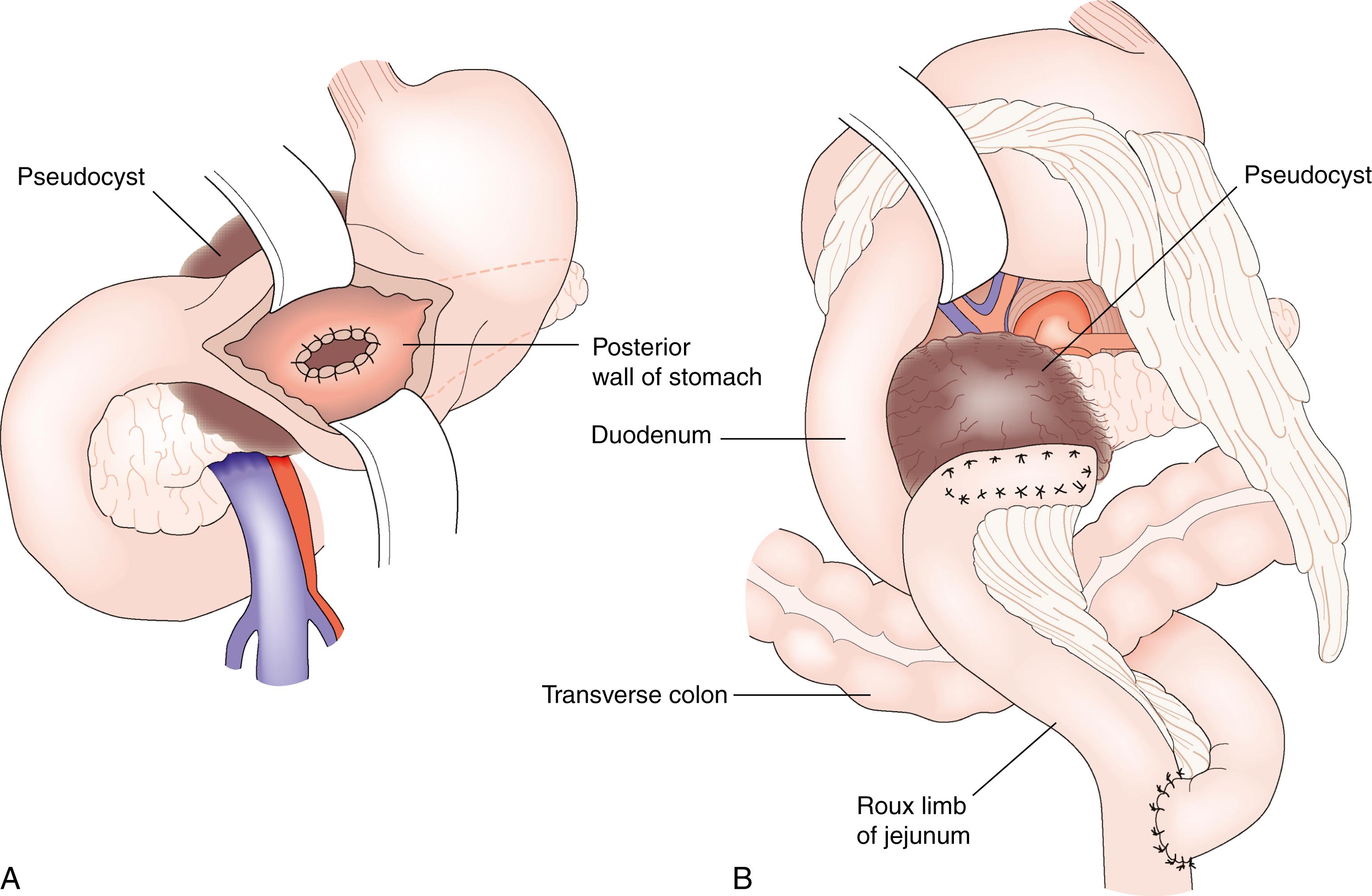
A pancreatic pseudocyst is defined as a collection of fluid persisting for more than 4 weeks but containing no necrosis. Pseudocysts may be imaged with magnetic resonance imaging (MRI) or CT. Uninfected and/or asymptomatic pseudocysts do not require treatment. Symptomatic or infected pseudocysts are best managed by endoscopic transgastric drainage where possible. A pancreatic abscess is a circumscribed intraabdominal collection of pus, usually in proximity to the pancreas, containing little or no pancreatic necrosis. In effect, this is the result of infection of a pseudocyst and is an indication for drainage.
Jaundice at presentation suggests that a gallstone is impacted at the lower end of the biliary tree. Such patients may develop ascending cholangitis, and early ERC with endoscopic sphincterotomy may therefore be indicated. Where jaundice develops later in the course of acute pancreatitis, this is usually a consequence of compression of the distal bile duct by an inflammatory pancreatic head mass. In such circumstances, treatment is directed towards the underlying pathology, and ERCP is challenging due to the degree of duodenal oedema and distortion. MRCP can be useful in identifying an impacted gallstone in the distal common bile duct.
Protracted ileus usually reflects continuing pancreatic inflammation. In the absence of an indication for intervention, conservative management is instituted, and nutritional status is maintained by nasojejunal enteral feeding or occasionally, if this is not tolerated, by TPN.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here