Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The liver is the largest abdominal organ, weighing approximately 1500 g. It extends from the fifth intercostal space to the right costal margin. It is triangular, and its apex reaches the left midclavicular line in the fifth intercostal space. In the recumbent position, the liver is impalpable under cover of the ribs. It is attached to the undersurface of the diaphragm by suspensory ligaments that enclose a ‘bare area’, the only part of its surface without a peritoneal covering. Its inferior or visceral surface lies on the right kidney, duodenum, colon and stomach.
Topographically, the liver parenchyma is smooth and provides few external markings as clues to its underlying segmental anatomy. The leading edge of the falciform ligament running on the cranial surface contains a remnant of the embryologic umbilical vein. It acts as an external guide to the plane between segments 2/3 and 4. On its visceral surface, the porta hepatis contains the draining extrahepatic biliary tree and dual vascular inflow (portal vein and hepatic artery), which are wrapped in a layer of loose connective tissue that separates segment 4 anteriorly and caudate lobe (segment 1) posteriorly. An imaginary line drawn over the cranial surface from the gallbladder to the termination of the middle hepatic vein into the inferior vena cava acts a guide to the principal plane separating the anatomic right and left hemilivers ( Fig. 15.1A ). Detailed knowledge of liver segmental anatomy, as defined by the distribution of its blood supply, is important to the surgeon.
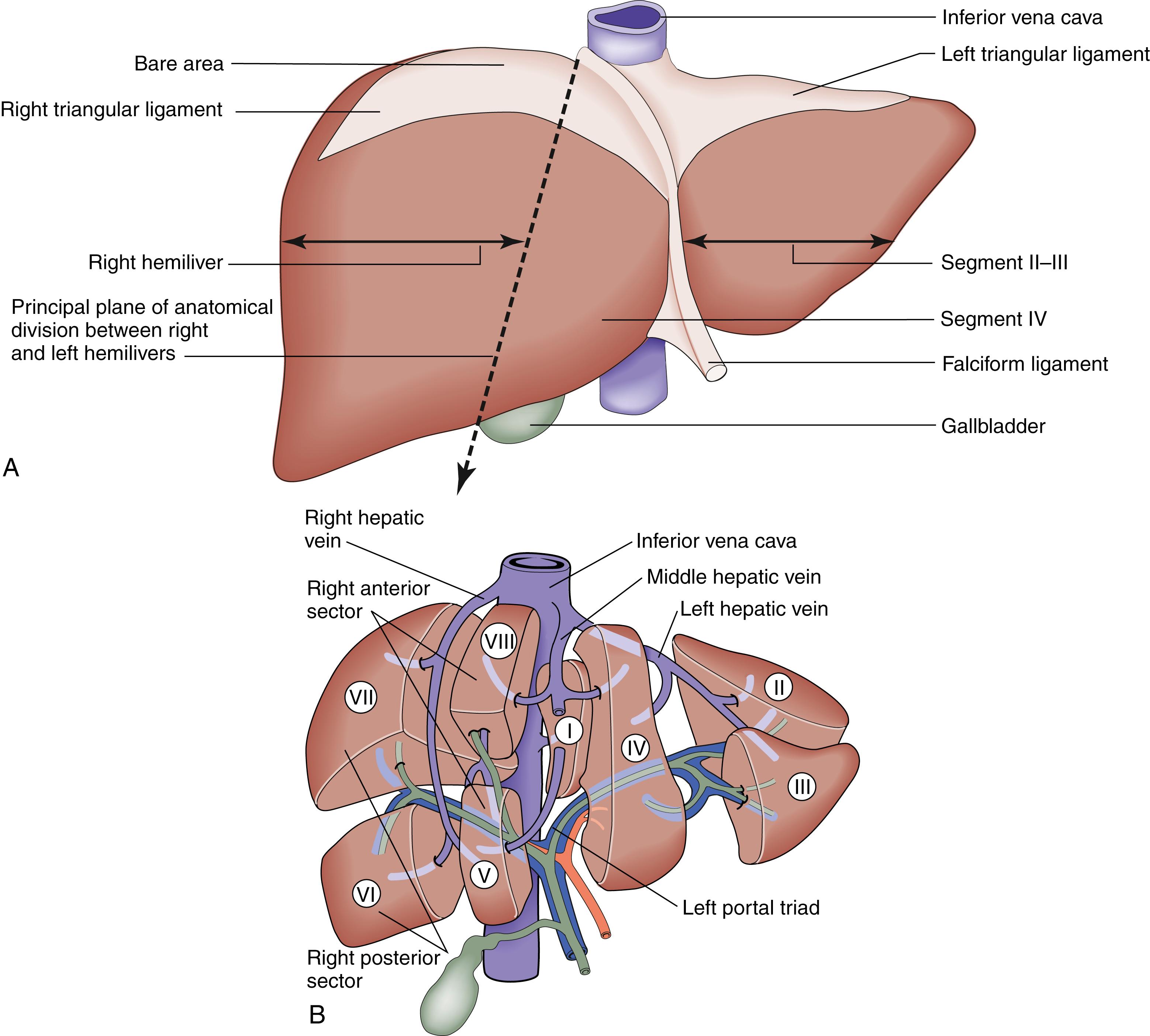
The portal vein, hepatic artery and draining biliary tree are wrapped in a fibrous sheath and divide into right and left branches in the porta hepatis. Occluding the vascular inflow to either hemiliver produces an easily visible line of demarcation demonstrating the location of the principal plane, along which lies the draining middle hepatic vein. Each hemiliver is further divided into sectors and segments by a combination of branches of the vascular inflow and draining hepatic veins. The right hemiliver is divided into the right anterior and posterior sector (separated by the right hepatic vein) and segments 8/5 (anterior sector) and 6/7 (posterior sector) by branches of the right hemiliver inflow. On the left side, three segments (2, 3, 4) are formed by branches of the left hemiliver inflow and are drained by the left and middle hepatic vein. The caudate lobe (segment 1) lies across the inferior vena cava surrounded by the right and left hemiliver and is supplied by the vascular inflow from both hemilivers with corresponding biliary drainage ( Fig. 15.1B ).
The liver normally receives 1500 mL of blood per minute and has a dual blood supply, 75% from the portal vein and 25% from the hepatic artery, which supplies 50% of the oxygen requirements. The primary venous drainage of the liver is by the right, middle and left hepatic veins, which enter the vena cava ( Fig. 15.1B ). In 25% of individuals, there is an inferior right hepatic vein. The venous drainage of the caudate lobe is by numerous small veins (short hepatic veins) emptying directly into the vena cava.
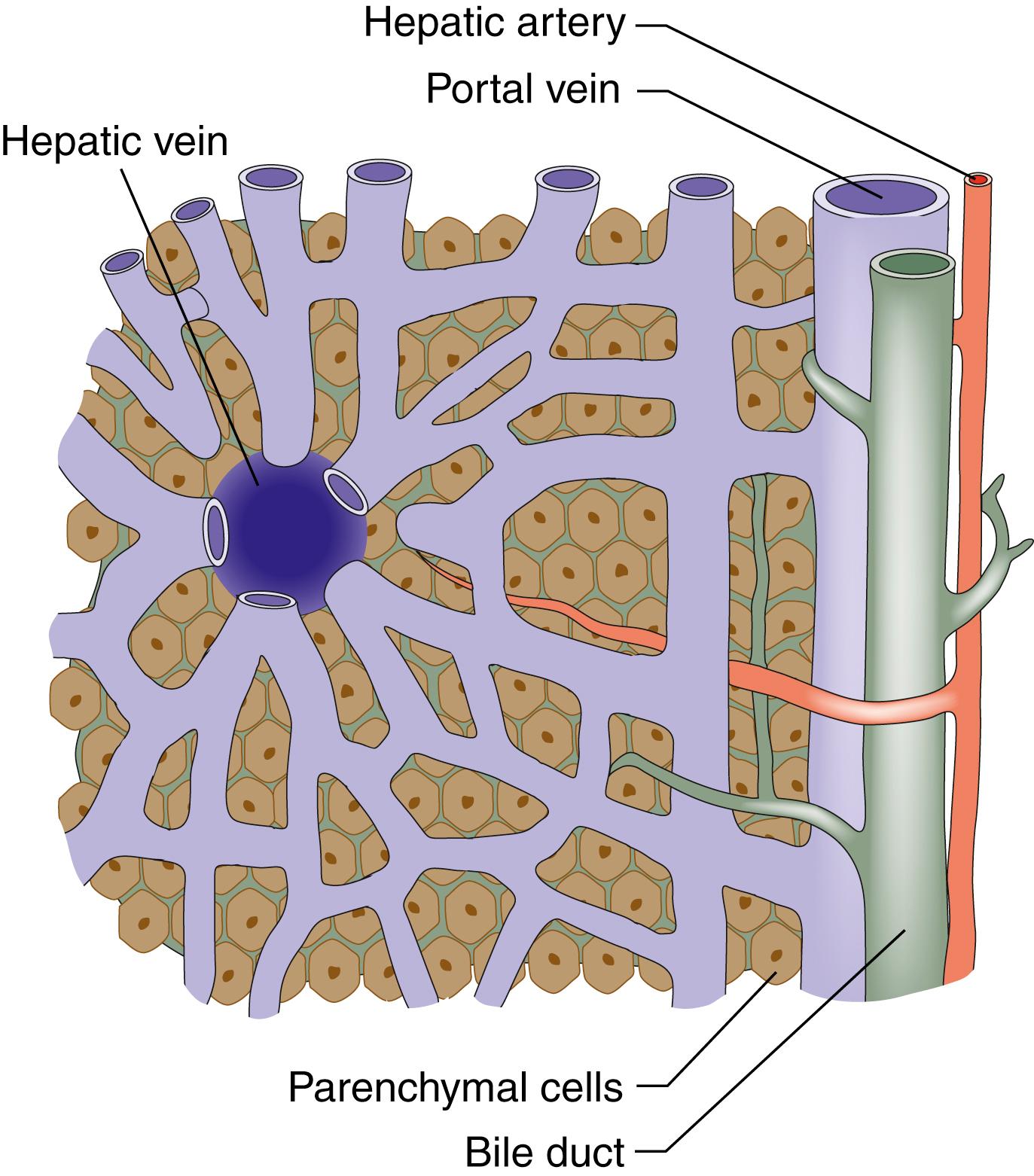
The functional unit of the liver is the hepatic acinus. Sheets of liver cells (hepatocytes) that are one cell thick are separated by interlacing sinusoids through which blood flows from the peripheral portal tract into the hepatic acinus to the central branch of the hepatic venous system. Bile is secreted by the liver cells that passes in the opposite direction along the small canaliculi into interlobular bile ducts located in the portal tracts ( Fig. 15.2 ).
The liver has an important role in nutrient metabolism and is responsible for storing glucose as glycogen or converting it to lactate for release into the systemic circulation. Amino acids are utilised for hepatic and plasma protein synthesis or catabolised to urea. The liver has a central role in the metabolism of lipids, bilirubin and bile salts, drugs and alcohol. It is the principal organ for storage of a number of minerals and vitamins, and is responsible for the production of coagulation factors I, V and XI; the vitamin K-dependent factors II, VII, IX and X; as well as proteins C and S and antithrombin. The liver is also important for immunity and is the largest reticuloendothelial organ in the body; its Kupffer cells play a role in the removal of damaged red blood cells, bacteria, viruses and endotoxin, much of which enter the body from the gut.
The liver is divisible into right and left hemilivers using a line running from the gallbladder fossa to the inferior vena cava
Each hemiliver receives a branch of the hepatic artery and portal vein; 75% of liver blood flow and 50% of its oxygen supply are provided by the portal vein
The hepatocytes are arranged in lobules, each of which has a central branch of the hepatic vein and peripheral portal tracts (containing a branch of the hepatic artery, portal vein and bile duct)
Understanding segmental liver anatomy allows the surgeon to perform anatomic resections ranging from individual segmentectomy to extended resections taking up to 70% of the liver parenchyma
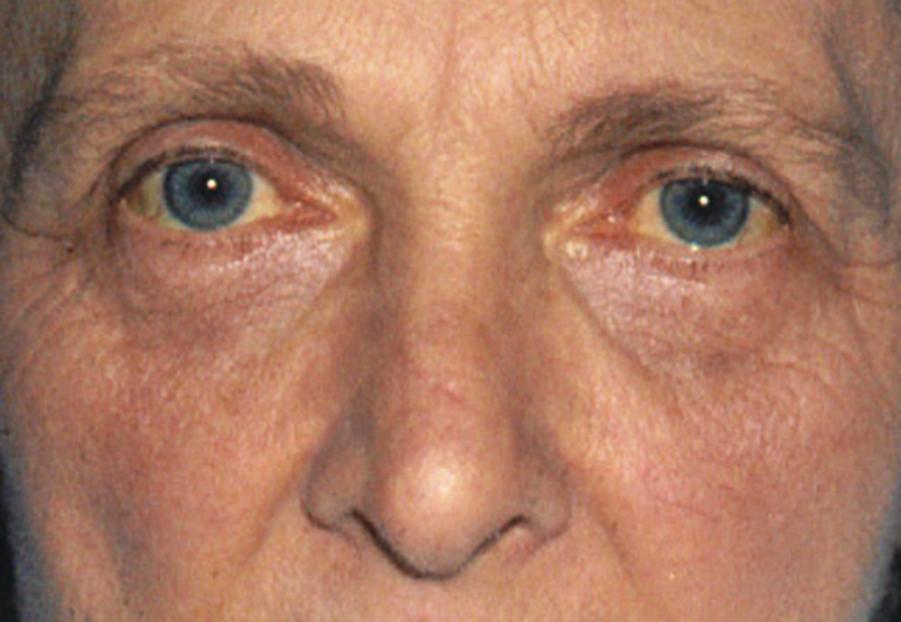
Jaundice is caused by an increase in the level of circulating bilirubin and becomes obvious in the skin and sclera when levels exceed 50 μmol/L ( Fig. 15.3 ). It may result from excessive destruction of red cells ( haemolytic jaundice ), failure to remove bilirubin from the bloodstream ( hepatocellular jaundice ) or obstruction to the flow of bile from the liver ( cholestatic or obstructive jaundice ) ( Fig. 15.4 ). Congenital nonhaemolytic hyperbilirubinaemia (Gilbert syndrome) is a relatively rare cause of jaundice due to defective bilirubin transport; the jaundice is usually mild and transient, and prognosis is excellent.
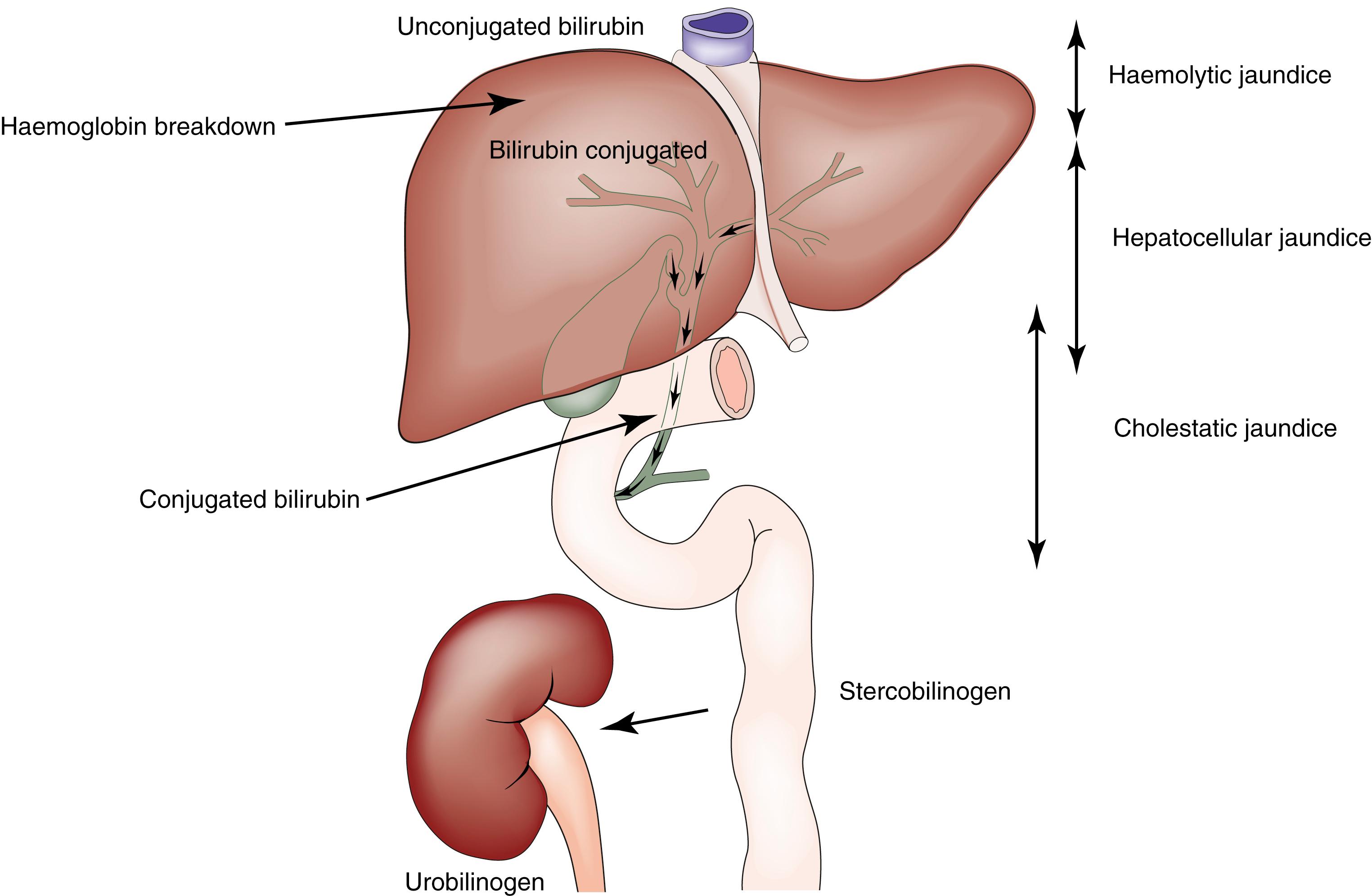
To the surgeon, the most important type of haemolytic jaundice is caused by hereditary spherocytosis, in which a splenectomy may be necessary ( Chapter 16 ). Haemolytic jaundice may also occur after blood transfusion and after operative or accidental trauma, when haematoma formation produces a pigment load exceeding hepatic excretory capacity.
Hepatocellular jaundice is usually a medical rather than surgical condition, although its recognition in patients presenting with abdominal pain is important, as surgical intervention may aggravate the hepatocellular injury.
Cholestatic jaundice due to intrahepatic obstruction of bile canaliculi may be a feature of acute and chronic liver disease. Examples include drugs such as antibiotics, antituberculosis and HIV therapies. This form of jaundice must be differentiated from that of extrahepatic obstruction, the causes of which have the most surgical relevance. Extrahepatic obstruction most commonly results from gallstones or cancer of the head of the pancreas. Other causes can be broken down into lesions of the lumen, lesions of the wall or extrinsic compression. Examples of luminal causes include parasitic infection or medically placed stents. Examples of lesions of the wall include primary neoplastic lesions such as cholangiocarcinoma (bile duct cancer) or ampullary carcinoma, inflammatory lesions such as primary sclerosing cholangitis, postsurgical strictures or autoimmune disease or congenital lesions such as choledochal cysts. Extrinsic compression can result from neoplastic lesions such as metastatic nodal disease, inflammatory pseudocysts or chronic pancreatitis.
An accurate, rapid diagnosis of the cause of jaundice allows for prompt institution of appropriate treatment ( Fig. 15.5 ). The age, sex, occupation, social habits, drug and alcohol intake, history of injections or infusions and general demeanour of the patient must be considered. A history of intermittent pain, fluctuant jaundice and dyspepsia suggests calculous obstruction of the common bile duct (CBD), whereas a history of weight loss and relentless progressive jaundice favours a diagnosis of neoplasia. Obstructive jaundice is likely if there is a history of dark urine and pale stools passage and if the patient complains of pruritus (due to an inability to secrete bile salts into the obstructed biliary system). Hepatocellular jaundice is likely if there are signs of chronic liver disease, such as palmar erythema, spider naevi, testicular atrophy and gynaecomastia. The abdomen must be examined for evidence of hepatomegaly or gallbladder distension (not usually found with gallstones) and for signs of portal hypertension such as splenomegaly, ascites and large collateral veins (caput medusae) in the abdominal wall.
Haemolytic jaundice is suggested if there are high circulating levels of unconjugated bilirubin but no bilirubin in the urine. Serum concentrations of liver enzymes are normal in these circumstances, and appropriate haematologic investigations should be performed.
In jaundice due to biliary obstruction, the circulating bilirubin is conjugated by the liver and rendered water-soluble; it is then excreted in the urine, giving it a dark colour. As bile cannot pass into the gastrointestinal tract, the stool becomes pale and urobilinogen is absent from the urine. Obstruction increases the formation of alkaline phosphatase from the cells lining the biliary canaliculi, producing raised serum levels. It is believed that bile acids may act to solubilise membranes, thus promoting the release of alkaline phosphatase. The increased levels correlate with the severity of luminal obstruction and are used to better estimate bile flow patency postoperatively even when bilirubin levels have normalised. This rise precedes that of bilirubin, and its fall is more gradual once obstruction is relieved. Serum transaminase levels may rise in obstruction. Conversely, swelling of the parenchyma in hepatocellular jaundice frequently produces an element of intrahepatic biliary obstruction and a modest rise in serum alkaline phosphatase concentration.
Full blood count and coagulation screen should be undertaken as a matter of routine, and viral status should be determined. Anaemia may signify occult blood loss, and a low white cell or platelet count may indicate hypersplenism due to portal hypertension. Prolongation of prothrombin time may be present in both hepatocellular and cholestatic jaundice.
If the clinical picture and biochemical investigations suggest that jaundice is obstructive, radiologic techniques can be used to define the site and nature of the obstruction.
In skilled hands, this key investigation is safe, noninvasive and reliable using ultrasound waves reflected from tissues at various depths and described as hyperechoic or hypoechoic compared with that of the liver (or spleen when the liver is abnormal due to cirrhosis). It is used to define whether the patient has bile duct dilatation or gallbladder distension due to obstruction. Obstructive or surgical jaundice is diagnosed by the presence of dilated intrahepatic biliary radicles that the sonologist can follow distally to determine the level and possibly cause of obstruction. In the case of tumours, the presence of regional lymphadenopathy, liver metastases and free fluid will help avoid expensive and invasive investigations. Ultrasonography will also detect gallstones (seen as a hyperechoic lesion casting a classic ‘acoustic shadow’) and space-occupying lesions in the liver and pancreas, although overlying bowel gas may prevent a clear view of the pancreas. For the same reason, stones in a dilated CBD may not always be seen clearly.
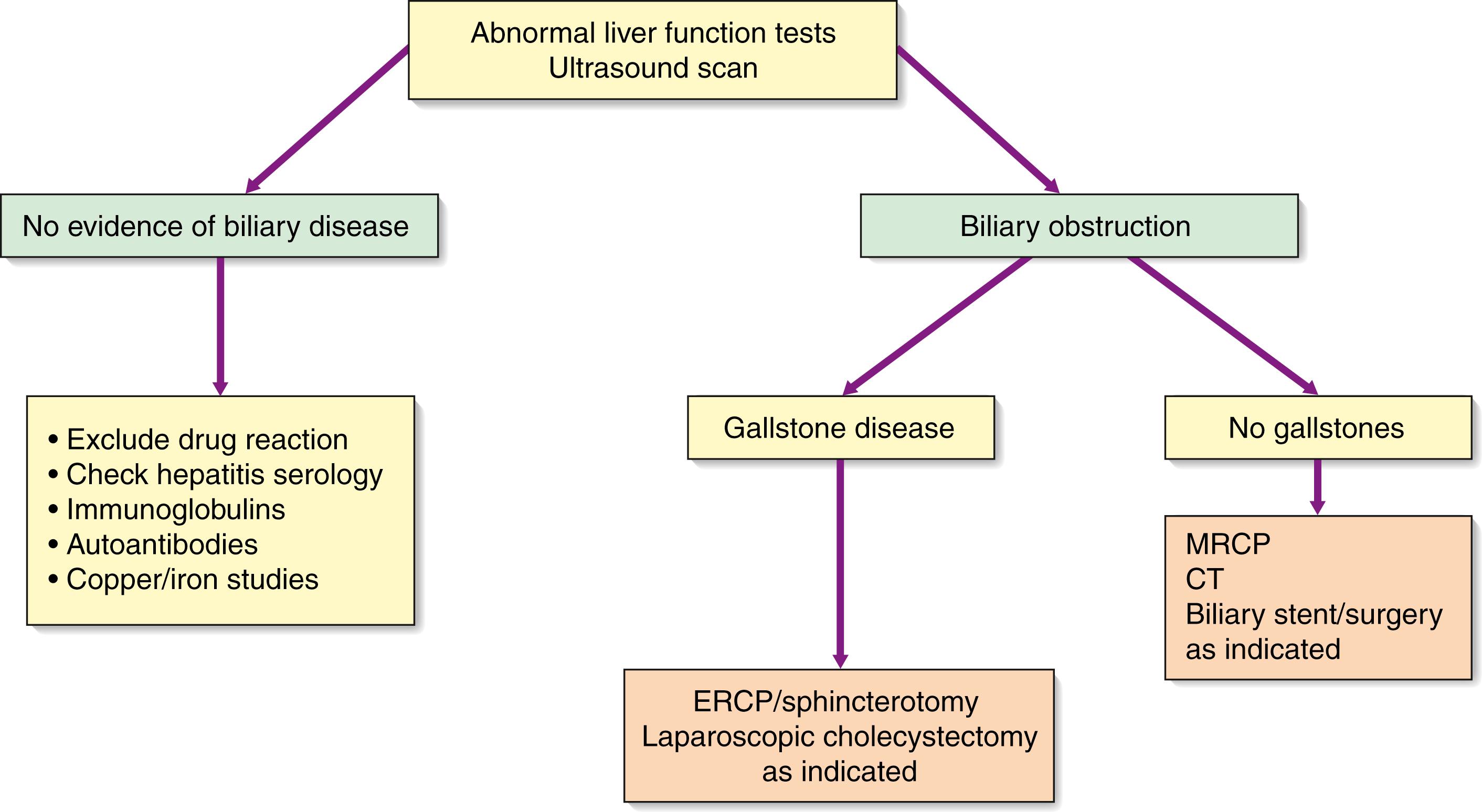
Magnetic resonance cholangiopancreatography (MRCP) has largely replaced other forms of invasive radiologic imaging of the bile duct and pancreas. MRI does not introduce infection into an obstructed biliary system or the pancreatic duct. This is important in patients presenting with symptoms suggestive of malignant obstructive jaundice. Software reconstructs the T2-weighted MRI scans to show the entire biliary tree (which appear white) with luminal stones or obstruction seen as filling defects amid the biliary secretions (seen as black) in the absence of any contrast ( Fig. 15.6 ). MRCP is performed without contrast and therefore is only useful for assessment of stones. To fully stage hepatic, biliary or pancreatic tumours invading the biliary tree, contrast is required to obtain arterial, portal venous and delayed hepatobiliary phases.
ERCP is now restricted to therapeutic indications. After injecting contrast through a cannula inserted into the papilla of Vater, it outlines the biliary and pancreatic systems by means of a side-viewing endoscope passed into the duodenum. It gives more detailed information than ultrasonography and allows endoscopic extraction of CBD stones, biopsy of periampullary tumours and relief of obstructive jaundice by stent insertion. Distal obstructions are more amenable for stenting than proximal or hilar obstructions. Stenting should be performed only in the presence of uncontrolled sepsis or for malignant lesions when surgery is considered inappropriate. Recently the availability of smaller scopes that can be passed within the main scope have become available. This enables not only direct visualization of the bile or pancreatic duct but diagnostic and therapeutic options including biopsy and stone destruction using laser or shockwave therapy. The investigation may be complicated by acute pancreatitis, and prophylactic antibiotics should be administered to reduce the risk of cholangitis for complex interventions. Haemorrhage and perforation are less frequent complications.
With the advent of MRCP and high level of technical success associated with ERCP, PTC is delegated to a therapeutic role for proximal biliary lesions or when ERCP has failed for distal lesions or cannot be used due to anatomic considerations. Access to the biliary system is achieved with a slim flexible needle passed into the liver under ultrasound and fluoroscopic guidance. While still under fluoroscopic guidance, injecting contrast during needle withdrawal achieves access to the dilated intrahepatic biliary radicles. A positioned catheter can provide external drainage of the bile, or the obstructing lesion can be crossed with a drain or stent. Alternatively, a wire can be placed through the ampulla allowing a rendezvous procedure to be performed via ERCP. Complications of PTC include bleeding, bile leakage, bacteraemia and renal dysfunction. Hence coagulation status must be checked, antibiotic cover should be given and the patient should be well hydrated prior to the procedure. The procedure is considered unsafe in the presence of ascites, bleeding disorders and hepatic hydatidosis.
Contrast-enhanced CT can be used to identify and stage hepatic, bile duct and pancreatic tumours in jaundiced patients. It often demonstrates the dilated biliary tree to the level of obstruction, vascular abnormality or invasion and may show dissemination to adjacent lymph nodes or distant viscera or peritoneum. It is also used to diagnose acute pancreatitis (in cases where there is doubt) and assess viability of pancreatic tissue in severe pancreatitis.
Positron emission tomography (PET-CT) has found an increasing role in staging hepatobiliary and pancreatic (HBP) malignancy. Isotopic liver scanning has been superseded by ultrasonography and CT. Selective angiography has been largely superseded by CT and MRI assessment of vascular anatomy but may be used for embolisation of tumours or haemorrhagic complications of HBP disease.
Liver biopsy may be considered in patients with unexplained jaundice, in whom an obstructing lesion has been radiologically excluded. ‘Targeted’ liver biopsy can be conducted under ultrasound or CT guidance. Prothrombin time, platelet count and hepatitis B surface antigen (HBsAg) status must always be determined, and clotting abnormalities should be corrected before biopsy is undertaken. Ascites remains an absolute contraindication to perform any type of liver puncture.
Laparoscopy under general anaesthesia may be used in the evaluation of liver disease. In selected patients with malignancy of the liver, pancreas and biliary tree, it may have a role in the staging of the tumour to exclude peritoneal or hepatic dissemination.
Given the important synthetic and excretory function of the liver, the development of obstructive jaundice can lead to significant metabolic derangement and disrupted haemostatic equilibrium. Medical team members caring for such patients should be aware of potential complications that these patients may develop. The most common abnormality is prolongation of prothrombin time, but this should readily correct within 36 hours with the administration of parenteral vitamin K when jaundice is cholestatic. Importantly, prolonged prothrombin time does not automatically correlate with bleeding tendency as synthesis of other coagulative factors such as protein C, S and antithrombin III may also be affected, resulting in patients becoming thrombogenic. Prophylactic measures aimed at preventing venous thromboembolism should therefore be considered.
Patients with long-standing jaundice can become malnourished and develop steatorrhoea, particularly if combined with pancreatic duct obstruction as seen with pancreatic head cancer. Nutritional supplementation and pancreatic enzyme replacement therapy may be indicated. Jaundiced patients are also at risk of renal dysfunction. Although the aetiology is not fully understood, it is likely that an enteric endotoxin crosses into the systemic circulation due to the absence of enteric bile salts, leading to renal vasoconstriction. Another contributing factor is cardiovascular depression secondary to jaundice resulting in peripheral vasodilatation. When combined with hypovolaemia or septicaemia, this can precipitate acute renal failure and is associated with a high mortality. Ensuring patients are well-hydrated, aggressive treatment of suspected sepsis and early biliary decompression are all important preemptive measures. Jaundiced patients are at increased risk of cholangitis and subsequent septicaemia due to bacterial translocation (retrograde or haemotogenous) into the biliary tree or by iatrogenic introduction during interventional procedures (see EBM 15.1 ). Resuscitation, antibiotic prophylaxis and/or therapy combined with biliary drainage are keys to successful management outcome.
Jaundice is a yellowish discoloration of the tissues that becomes clinically apparent when serum bilirubin levels exceed 50 μmol/L (normal < 20 μmol/L)
It may be due to excessive haemolysis, hepatic insufficiency or cholestasis; cholestatic (obstructive) jaundice is the type encountered in surgical practice
The two most common causes of surgical obstructive jaundice are cancer of the head of the pancreas and stones in the CBD (choledocholithiasis)
In cholestatic jaundice, the bilirubin has been conjugated by the hepatocytes and is therefore soluble in water and can be excreted in the urine; patients with obstructive jaundice typically have dark urine and pale stools and may have pruritus (thought to be due to the accumulation of bile salts)
Obstructive jaundice is characterised by elevated serum alkaline phosphatase levels in addition to hyperbilirubinaemia and may be accompanied by modest elevations in transaminase (aminotransferase) levels, reflecting liver damage
Up to 5% of the population have simple liver cysts. They are lined by biliary epithelium and contain serous fluid but never communicate with the biliary tree. They rarely produce symptoms, are associated with normal liver function and have no discernible wall on ultrasound or CT ( Fig. 15.7 ). In the few patients who develop symptoms, cysts tend to recur following aspiration; sclerosis by alcohol injection is of
‘Appropriate antibiotic prophylaxis and effective biliary drainage is the cornerstone of successful management of acute cholangitis.’
little value for large symptomatic cysts. Surgical management consists of deroofing and may be undertaken by laparoscopic means. Polycystic disease is a rare cause of liver enlargement and may be associated with polycystic kidneys as an autosomal dominant trait. In symptomatic patients, it may be necessary to combine a deroofing procedure with hepatic resection or consider liver transplantation. Such surgical interventions can be associated with significant postoperative morbidity, and so patients must be well-counselled given it is usually performed for quality-of-life reasons.
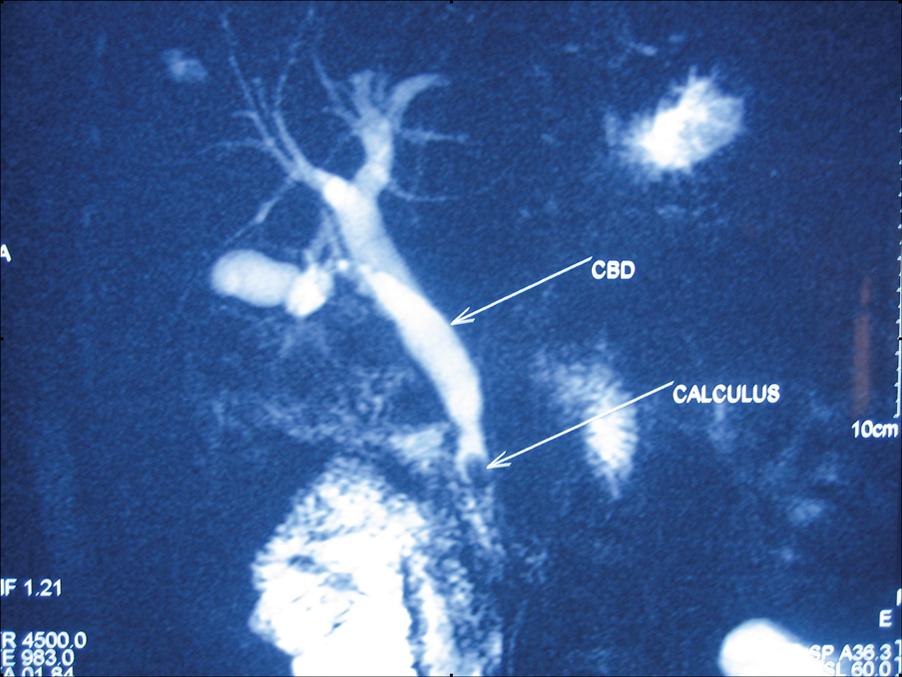
Cavernous haemangiomas are one of the most common benign tumours of the liver (up to 5% of population) and may be congenital. Women are affected six times more frequently than men. Most haemangiomas are small solitary subcapsular growths found incidentally during laparotomy or autopsy, but they are sometimes detected on ultrasound examination as densely hyperechoic lesions that mimic hepatic tumours. Centripetal ‘filling in’ of contrast during dynamic imaging with CT or MRI is seen. These lesions rarely give rise to pain, and resection may be considered for symptomatic lesions exceeding 5 cm in diameter.
After the spleen, the liver is the solid organ most damaged in abdominal trauma, particularly following road traffic accidents. Stab injuries and gunshot wounds of the liver are also increasing in incidence, which are considered in Chapter 9 .
Liver abscesses can be classified as bacterial, parasitic or fungal. Bacterial abscess is the most common type in Western medicine, but parasitic infestation is an important cause worldwide. Fungal abscesses are found in patients receiving long-term broad-spectrum antibiotic treatment or immunosuppressive therapy and may complicate actinomycosis.
Infection from the biliary system is now more common due to the increasing use of radiologic and endoscopic intervention. Infection may spread through the portal vein from abdominal sepsis (e.g., appendicitis, diverticulitis), via the hepatic artery from a septic focus anywhere in the body or by direct spread from a contiguous organ (e.g., empyema of the gallbladder). Abscess formation may follow blunt or penetrating injury, and in one-third of patients the source of infection is indeterminate (cryptogenic). Common organisms are:
Streptococcus milleri
Escherichia coli
Streptococcus faecalis
Staphylococcus aureus
Anaerobes ( Bacteroides spp)
The onset of symptoms is often insidious, and the patient may present with pyrexia of unknown origin. There is sometimes a history of sepsis elsewhere, particularly within the abdomen, and pain in the right hypochondrium. Other patients present with swinging pyrexia, rigors, marked toxicity and jaundice. The liver is often enlarged and tender.
Plain radiographs may show elevation of the diaphragm, pleural effusion and basal lobe collapse. Leucocytosis is usually present, and liver function tests are deranged. Ultrasonography or CT of the abdomen is used to define the abscess (which is often irregular and thick walled), facilitate percutaneous aspiration for culture and exclude a portal source. Abscesses are generally multiple rather than single. ERCP or antegrade tubogram via the abscess cavity may identify a biliary cause if bile is aspirated from the abscess cavity. The underlying microbe may also guide further investigations such as dental examination or colonoscopy. Patients should be investigated for occult diabetes or other immunosuppressive illnesses depending on the prevalence of such illnesses in the treated population.
Untreated abscesses are potentially fatal because of spread within the liver to multiple sites and because of septicaemia and debility. The principles of treatment are percutaneous drainage of accessible abscesses under ultrasound or CT guidance, and antibiotic therapy selected based on culture of blood or pus. It is exceptional to resort to surgical drainage. Percutaneously or surgically placed drainage tubes are left in place, and the size of the cavity is monitored by CT or serial x-rays following the injection of contrast material. Multiple small abscesses may require prolonged treatment with antibiotics for up to 8 weeks.
Entamoeba histolytica is a protozoal parasite that infests the large intestine and is endemic in many tropical regions. Trophozoites released by the cyst in the intestine may penetrate the mucosa to gain access to the portal venous system and spread to the liver. The abscess is large and thin walled, usually solitary and in the right lobe, and contains brown sterile pus resembling anchovy sauce.
Right upper quadrant pain may be accompanied by anorexia, nausea, weight loss and night sweats. Tender enlargement of the liver is invariable, although jaundice is uncommon. Other signs include basal pulmonary collapse, pleural effusion and leucocytosis.
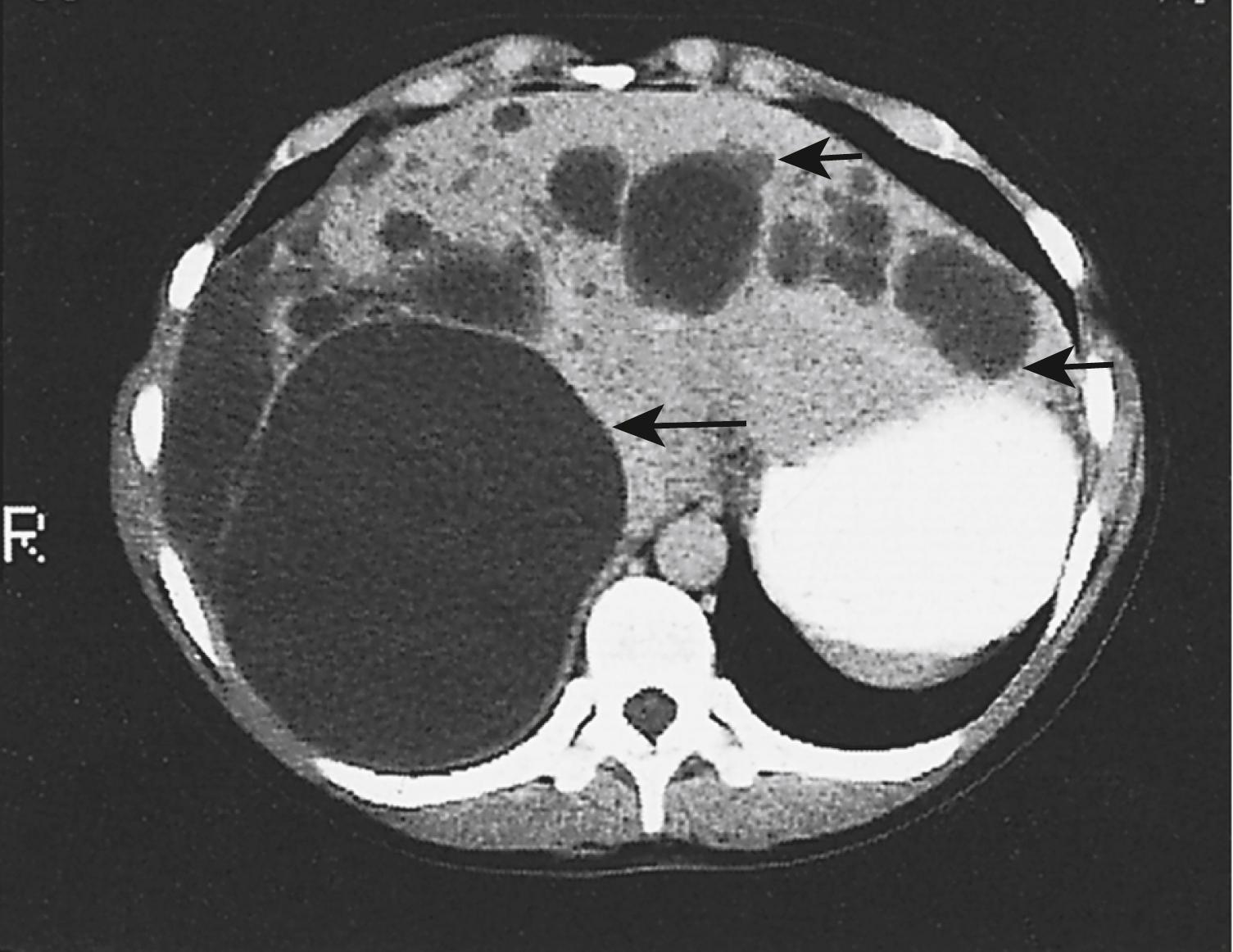
Ultrasonography and CT are used to demonstrate the site and size of the abscess, which often has poorly defined margins ( Fig. 15.8 ). The stools should be examined for amoebae or cysts. Direct and indirect serologic tests (indirect haemagglutination [IHA], enzyme-linked immunosorbent assay [ELISA]) to detect amoebic protein are extremely useful, especially in nonendemic areas.
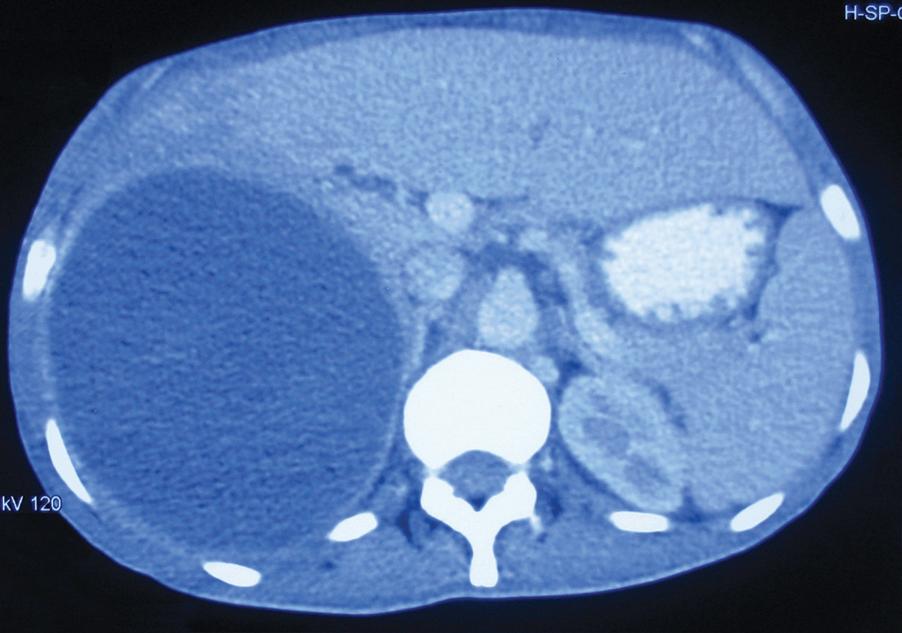
Early diagnosis is important, and treatment may be commenced empirically in areas where the problem is endemic. Treatment consists of administration of metronidazole (800 mg 8-hourly for 7–10 days) that usually results in rapid resolution. A luminal agent (diloxanide furoate 500 mg 8-hourly for 10 days) is advised to prevent a carrier state. The abscess should be aspirated by needle puncture if there is no clinical response within 72 hours. If untreated, an amoebic abscess may rupture into the peritoneal cavity (features of peritonitis), pleural space (pleural effusion), into a bronchus (anchovy sauce expectoration) or rarely into the pericardial cavity ( Fig. 15.9 ). An abscess that ruptures into the peritoneal cavity is managed with laparotomy and lavage.
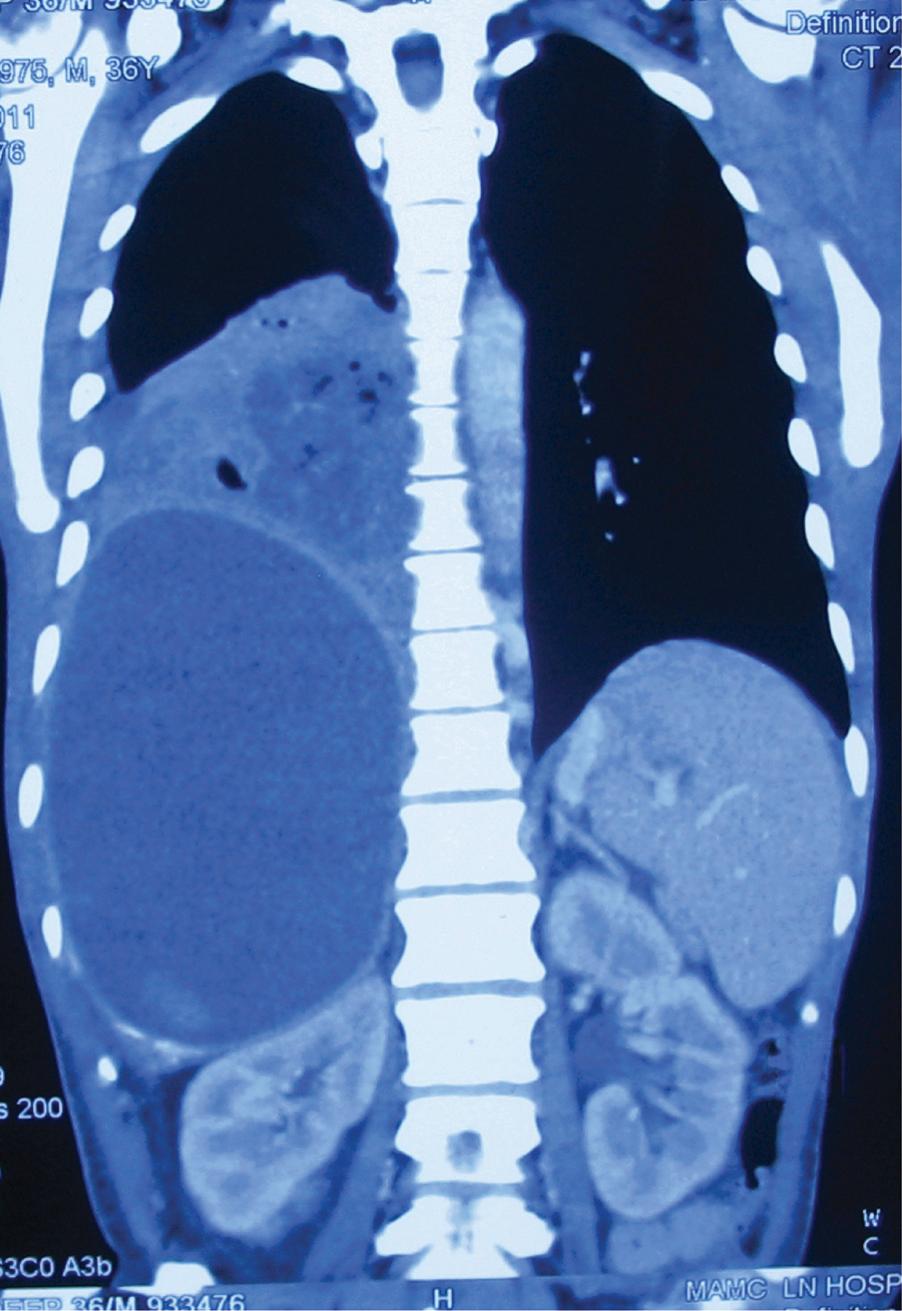
An amoeboma is a chronic granuloma involving the caecum, usually seen in patients with incompletely treated long-duration amoebiasis infection. The lesion feels like a lump in the right iliac fossa and needs to be differentiated from a malignancy. Fulminant amoebic colitis is rare and may present with peritonitis or lower gastrointestinal bleeding, which is treated appropriately.
This less common infestation is caused in humans by one of two forms of tapeworm, Echinococcus granulosus and E. multilocularis. The adult tapeworm lives in the intestine of the dog, from which ova are passed in the stool; sheep or goats serve as an intermediate host by ingesting the ova, whereas humans are accidental hosts ( Fig. 15.10 ). The condition is most common in sheep- and goat-rearing areas. Ingested ova hatch in the duodenum, and the embryos pass to the liver through the portal venous system. The wall of the resulting hydatid cyst consists of the pericyst or adventitia, which is the host tissue formed by the body as a reaction to the parasite, the laminated membrane (ectocyst or the external layer of the cyst) and the germinative layer of laminated membrane on which brood capsules containing scolices develop (endocyst).
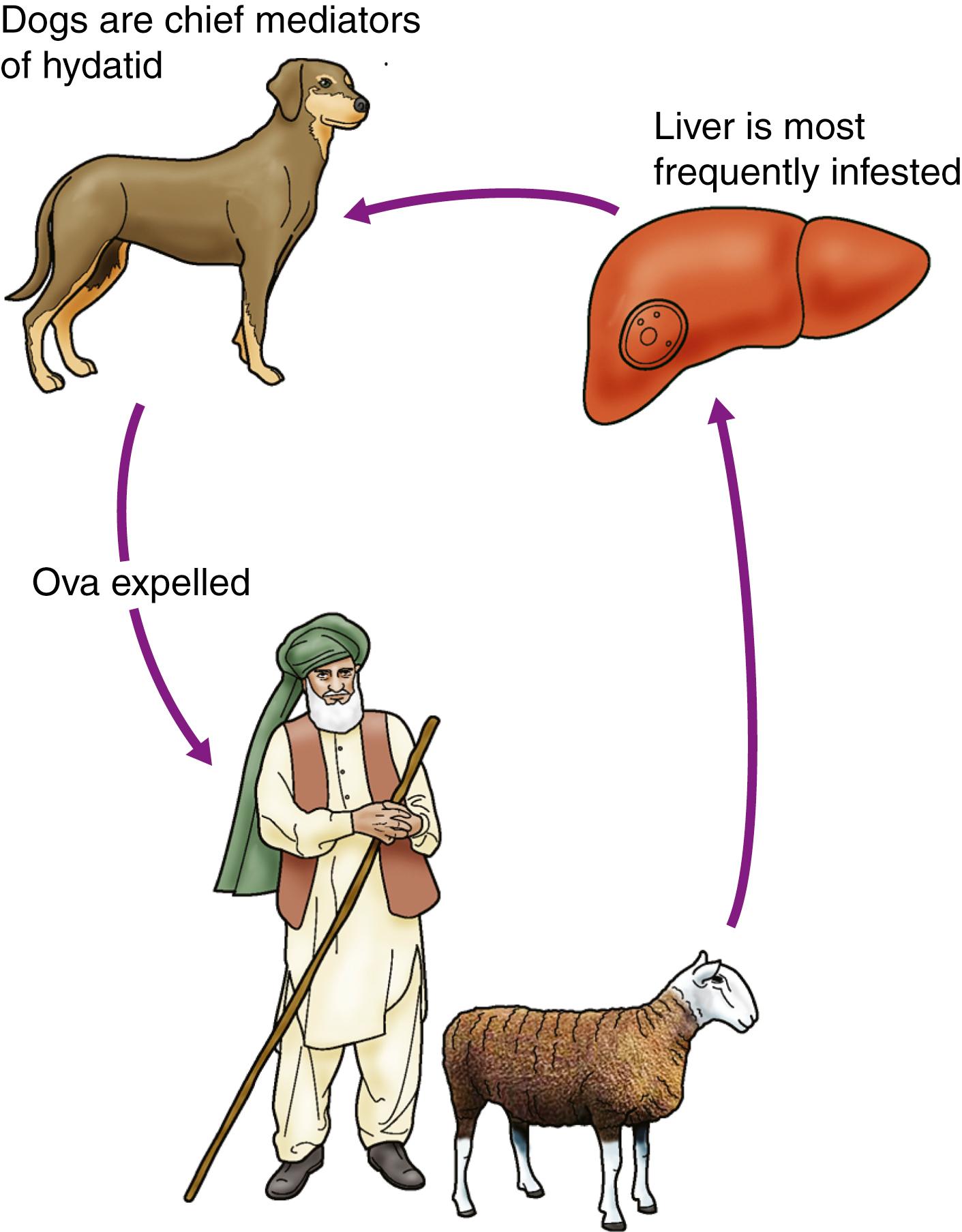
The disease may be symptomless, but chronic right upper quadrant pain with enlargement of the liver is the common presentation. The cyst may rupture into the biliary tree or peritoneal cavity; the latter sometimes causes an acute anaphylactic reaction due to absorption of foreign hydatid protein. Other complications include secondary infection and biliary obstruction with jaundice.
Eosinophilia is common, and various serologic tests are used to detect several hydatid antigens, while subclass IgG antibodies are also useful. Hydatid cysts commonly calcify and may be seen on a plain film of the abdomen. Alternatively, they can be detected by ultrasound, CT or MRI and are recognisable by their thick trilaminar wall, which may contain multiple daughter cysts. MRI has the advantage of delineating the biliary tree to determine whether there is any communication or cyst debris within the duct. The Gharbi classification describes a range from univesicular cyst (grade 1) to reflecting thick-walled calcified cyst (grade 5).
In asymptomatic patients, small calcified cysts may not require treatment. Patients can be treated successfully with albendazole or mebendazole, but this may be prolonged. Large symptomatic cysts are best managed by open or laparoscopic deroofing and complete excision of the endocyst. The residual cavity may be filled with hypertonic saline (scolicidal), closed if uninfected or marsupialised after deroofing or packed with omentum (omentoplasty) if infected. Caution should be employed when using scolicidal agents if the cyst communicates with the biliary system. A laparoscopic approach is possible for superficial accessible cysts. Selected patients may be suitable for puncture–aspiration–injection–reaspiration (PAIR). Complete excision, together with the parasites contained within (known as pericystectomy), is now preferred for superficial lesions, especially those in the left lobe, in centres accustomed to undertaking liver resections. Preoperative chemotherapy and perioperative steroid cover should be used in conjunction with packing the peritoneal cavity off from the cyst to prevent intraoperative anaphylaxis and seeding of live daughter cysts.
Portal hypertension results from increased resistance to portal venous blood flow. Rarely, it results from an increase in portal blood flow. The normal pressure of 5 to 15 cm H 2 O in the portal vein is consistently exceeded (above 25 cm H 2 O). Portal vein thrombosis is a rare cause and is most commonly due to neonatal umbilical sepsis. The most common cause of portal hypertension is cirrhosis resulting from chronic liver disease, which is characterised by liver cell damage, fibrosis and nodular regeneration. The fibrosis obstructs portal venous return and portal hypertension develops. Arteriovenous shunts within the liver also contribute to the hypertension.
Alcohol and steatohepatitis associated with obesity are the most common aetiologic factors in developed countries, whereas in North Africa, the Middle East and China, schistosomiasis due to Schistosoma mansoni is a common cause. Chronic active hepatitis and primary and secondary biliary cirrhosis may result in portal hypertension, but in many patients the cause remains unclear (cryptogenic cirrhosis).
Posthepatic portal hypertension is rare. It is most frequently due to spontaneous thrombosis of the hepatic veins, and this has been associated with neoplasia, oral contraceptive agents, polycythaemia and the presence of abnormal coagulants in the blood. Inferior vena caval obstruction above the level at which the hepatic veins open into the cava is more common in South and Southeast Asia. The resulting Budd–Chiari syndrome is characterised by portal hypertension, caudate hypertrophy, liver failure and gross ascites.
As a result of gradual chronic occlusion of the portal venous system, collateral pathways develop between the portal and systemic venous circulations. Depending on the site of obstruction with regard to the hepatic sinusoid, portal hypertension is classified as presinusoidal, sinusoidal and postsinusoidal (see Table 15.1 ). Portosystemic shunting occurs at three principal sites ( Fig. 15.11 ). Most important is the development of varices in the submucosal plexus of veins in the lower oesophagus and gastric fundus. On the portal side are the short gastric veins and left gastric vein, and on the systemic side (caval side) are the azygos and hemiazygos veins. Oesophageal varices may rupture, causing acute massive gastrointestinal bleeding in about 40% of cirrhosis patients. The initial episode of variceal haemorrhage is fatal in about one-third of patients, and recurrent haemorrhage is common. Bleeding from retroperitoneal and periumbilical collaterals (‘caput medusae’) is troublesome during abdominal surgery as collaterals may develop and cause bleeding at the site of stomas. Anorectal varices are not uncommonly found at proctoscopy but rarely cause bleeding.
| Obstruction to portal flow: |
| Presinusoidal extrahepatic |
|
|
| Neonatal sepsis |
| Pyelophlebitis |
| Trauma |
| Tumour |
| Extrahepatic portal vein obstruction (EHPVO) |
|
| Pancreatic disease |
| Lymphadenopathy |
| Biliary tract tumours |
| Presinusoidal intrahepatic |
|
|
| Sinusoidal |
|
| Postsinusoidal |
|
|
| Increased blood flow (rare) |
|
|
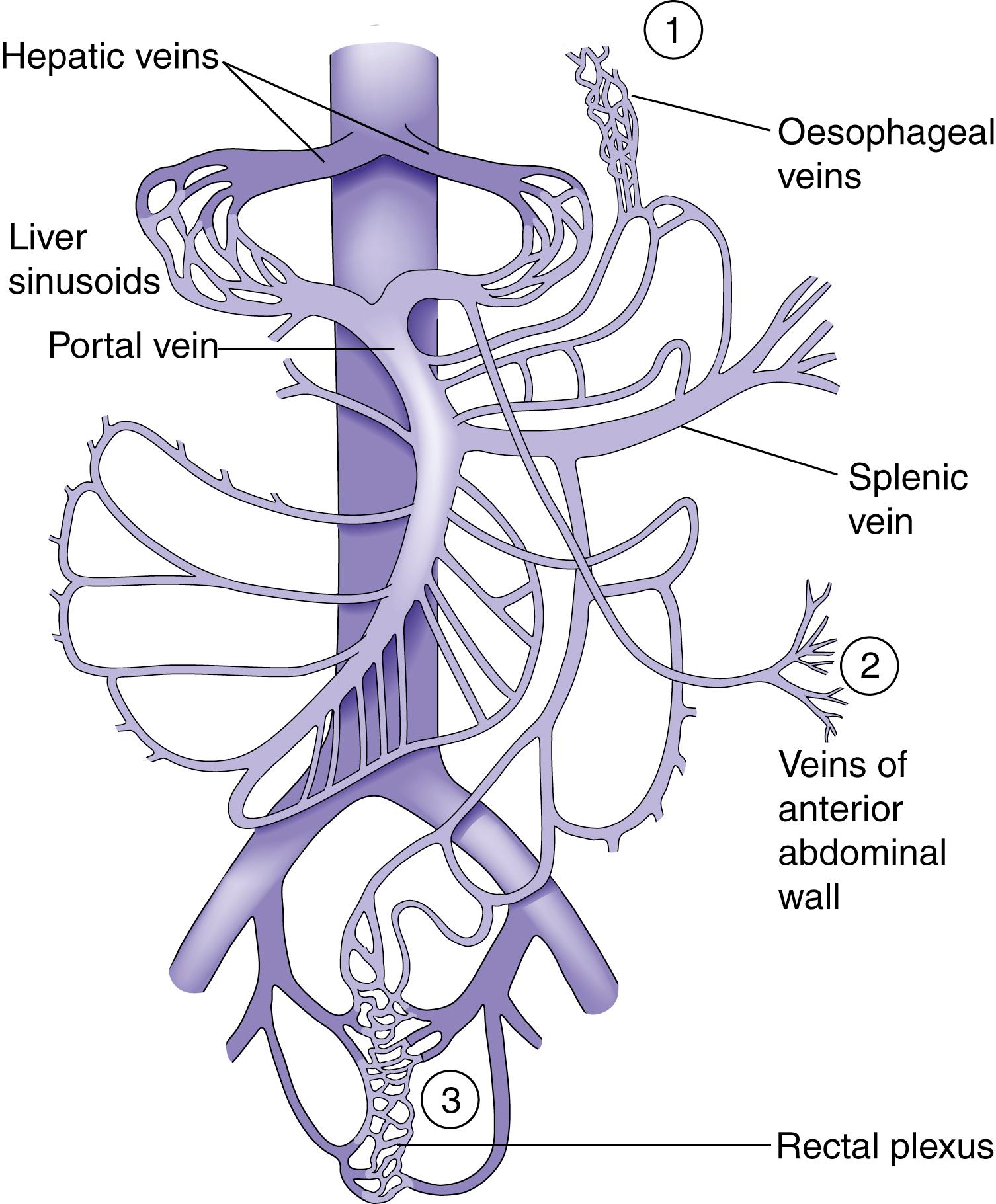
Progressive enlargement of the spleen occurs because of vascular engorgement and associated hypertrophy. Haematologic consequences are anaemia, thrombocytopenia and leucopenia (with resulting hypersplenism). Ascites may develop due to increased formation of hepatic and splanchnic lymph, hypoalbuminaemia and retention of salt and water. Increased aldosterone and antidiuretic hormone levels may contribute. Portosystemic encephalopathy is due to an increased level of toxins such as ammonia in the systemic circulation. This is likely to develop where there are large spontaneous or surgically created portosystemic shunts. Gastrointestinal haemorrhage increases the absorption of nitrogenous products and may precipitate encephalopathy.
‘Severity of cirrhosis is best assessed by Child Pugh score. Risk of varices increases with Child Pugh score. Assessment of varices is best done by endoscopy and should be performed for all patients with cirrhosis. Varices should be graded by size (5 mm). For patients with large varices (> 5 mm) or small varices and high risk stigmata of bleeding or severe liver disease prophylactic non selective β-blockers should be prescribed. Endoscopic variceal ligation (EVL) can also be used for high risk patients as an alternative to β-blockers. The two treatments have different side effect profiles that should be considered.’
Patients with cirrhosis frequently develop anorexia, generalised malaise and weight loss. Clinical manifestations include hepatosplenomegaly, ascites, jaundice and spider naevi. Slurring of speech, a flapping tremor or dysarthria may point to encephalopathy, which may be precipitated or intensified by the accumulation of blood in the gastrointestinal tract. The serum bilirubin may be elevated and the serum albumin depressed. Anaemia may also be present and the leucocyte count raised (or depressed if there is hypersplenism). The prothrombin time and other indices of clotting may be abnormal. Clinical and biochemical parameters are used as the basis of the modified Child classification ( Table 15.2 ). Patients allocated to grade A have a good prognosis, whereas those in grade C have the worst prognosis.
| Points scored | |||
|---|---|---|---|
| Criterion | 1 | 2 | 3 |
| Encephalopathy | None | Minimal | Marked |
| Ascites | None | Easily controlled | Intractable |
| Bilirubin (μmol/L) | < 35 | 35–50 | > 50 |
| Albumin (g/L) | > 35 | 28–35 | < 28 |
| Prothrombin ratio | < 1.7 | 1.7–2.3 | > 2.3 |
Patients with portal hypertension need to be assessed for varices ( EBM 15.2 ). Those with varices may be referred to a surgeon because of uncontrolled bleeding from oesophageal varices or for consideration of elective surgery for varices that have been resistant to nonsurgical management.
Patients presenting with acute upper gastrointestinal bleeding are examined for evidence of chronic liver disease via endoscopy. This allows the detection of varices and defines whether they are or have been the site of bleeding. It is important to remember that peptic ulcer
‘Airway protection, controlled resuscitation and correction of coagulopathy should commence immediately. Antibiotics and splanchnic vasoconstrictors should be commenced. Urgent endoscopy within 12 hours should be instituted. EVL is the method of choice for control of variceal haemorrhage, with endoscopic variceal sclerotherapy as second choice. If delays to definitive therapy are likely or if the haemorrhage proves difficult to control endoscopically temporary deployment of a modified Sengstaken tube is indicated. Further endoscopic treatment, transjugular intrahepatic portosystemic stent shunting (TIPSS) or surgical treatment (portosystemic shunt or oesophageal transection) can subsequently be employed as definitive therapy.’
and gastritis are common complaints that occur in 20% of patients with varices.
The priorities in the management of bleeding oesophageal varices are summarised in Table 15.3 and EBM 15.3 .
| Active resuscitation |
|
|
| Assessment of coagulation status |
|
|
| Urgent endoscopy |
| Control of bleeding |
|
|
|
| Treatment of hepatocellular decompensation |
| Treatment/prevention of portosystemic encephalopathy |
| Prevention of further bleeding from varices |
|
|
|
|
The aim is to quickly replace blood loss with a view to urgent endoscopy. Resuscitation must be done cautiously aiming for haemoglobin (Hb) of 8 g/L as overresuscitation can increase mortality. Avoid large volumes of saline solutions. Many patients have coagulation defects from the outset, and thrombocytopenia is a common manifestation of hypersplenism. The advice of the haematologist should be sought regarding the use of fresh-frozen plasma (FFP), platelet transfusion or other coagulation factors. Antibiotic prophylaxis should be administered using norfloxacin 400 mg twice a day for 7 days or IV ceftriaxone 1 g/day for 7 days. This decreases bacterial infections, reduces rebleeding and improves survival. Pharmacologic therapies aimed at reducing portal pressures, such as terlipressin or somatostatin analogues, can be started. However, they are not definitive treatments, and urgent endoscopy should be arranged.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here