Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The lichenoid reaction pattern (lichenoid tissue reaction, interface dermatitis) is characterized histologically by epidermal basal cell damage. This takes the form of cell death and/or vacuolar change (liquefaction degeneration). The cell death usually involves only scattered cells in the basal layer that become shrunken with eosinophilic cytoplasm. These cells, which have been called Civatte bodies, often contain pyknotic nuclear remnants. Sometimes, fine focusing up and down will reveal smaller cell fragments, often without nuclear remnants, adjacent to the more obvious Civatte bodies. These smaller fragments have separated from the larger bodies during the process of cell death. Ultrastructural studies have shown that the basal cells in the lichenoid reaction pattern usually die by apoptosis, a form of cell death that is quite distinct morphologically from necrosis.
Before discussing the features of apoptosis, mention will be made of the term interface dermatitis , which is widely used. It has been defined as a dermatosis in which the infiltrate (usually composed mostly of lymphocytes) appears “to obscure the junction when sections are observed at scanning magnification.” The term is not used uniformly or consistently. Some apply it to most dermatoses with the lichenoid tissue reaction. Others use it for the subgroup in which the infiltrate truly obscures the interface (erythema multiforme, fixed drug eruption, paraneoplastic pemphigus, some cases of subacute lupus erythematosus and pityriasis lichenoides). The infiltrate may obscure the interface in lymphomatoid papulosis, but basal cell damage is not invariable. Many apply the term, also, to lichen planus and variants, in which the infiltrate characteristically “hugs” the basal layer without much extension into the epidermis beyond the basal layer. Crowson et al. have expanded the concept of interface dermatitis to include neutrophilic and lymphohistiocytic forms, in addition to the traditional lymphocytic type. They also subdivide the lymphocytic type into a cell-poor type and a cell-rich type. Erythema multiforme, which they list as a cell-poor variant, is sometimes quite “cell rich.” The author prefers the traditional term lichenoid ’ for this group of dermatoses because it is applicable more consistently than interface dermatitis and it is less likely to be applied as a “final sign-out diagnosis,” which is often the case with the term interface dermatitis. The term is so entrenched that it is unlikely to disappear from the lexicon of dermatopathology.
In apoptosis , single cells become condensed and then fragment into small bodies by an active budding process ( Fig. 4.1 ). In the skin, these condensed apoptotic bodies are known as Civatte bodies (discussed previously). The smaller apoptotic bodies, some of which are beyond the resolution of the light microscope, are usually phagocytosed quickly by adjacent parenchymal cells or by tissue macrophages. Cell membranes and organelles remain intact for some time in apoptosis, in contradistinction to necrosis where breakdown of these structures is an integral and prominent part of the process. Keratinocytes contain tonofilaments that act as a “straitjacket” within the cell, and therefore budding and fragmentation are less complete in the skin than they are in other cells in the body undergoing death by apoptosis. This is particularly so if the keratinocyte has accumulated filaments in its cytoplasm, as occurs with its progressive maturation in the epidermis. The term dyskeratotic cell is usually used for these degenerate keratinocytes. The apoptotic bodies that are rich in tonofilaments are usually larger than the others; they tend to “resist” phagocytosis by parenchymal cells, although some are phagocytosed by macrophages. Others are extruded into the papillary dermis, where they are known as colloid bodies . These bodies appear to trap immunoglobulins nonspecifically, particularly the immunoglobulin M (IgM) molecule, which is larger than the others. Apoptotic cells can be labeled by the TUNEL (terminal transferase-mediated dUTP nick end labeling) reaction.
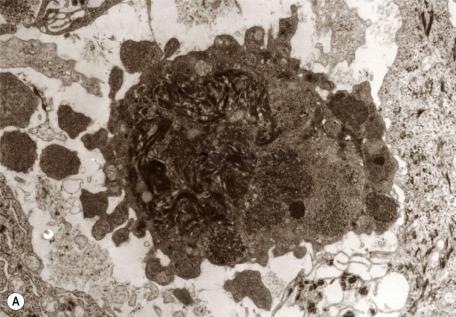
Some of the diseases included within the lichenoid reaction pattern show necrosis of the epidermis rather than apoptosis; in others, the cells have accumulated so many cytoplasmic filaments before death that the actual mechanism—apoptosis or necrosis—cannot be discerned by light or electron microscopy. The term filamentous degeneration has been suggested for these cells ; on light microscopy, they are referred to as dyskeratotic cells (discussed previously). Some dermatopathologists use the term necrotic keratinocyte for these cells and also for keratinocytes that are obviously apoptotic. Note that apoptotic keratinocytes have been seen in normal skin, indicating that cell deletion also occurs as a normal physiological phenomenon. As Afford and Randhawa eloquently stated, “Apoptosis is the genetically regulated form of cell death that permits the safe disposal of cells at the point in time when they have fulfilled their intended biological function.” It also plays a role in the elimination of the inflammatory infiltrate at the end stages of wound healing.
Although it is beyond the scope of this book, readers interested in apoptosis and the intricate mechanisms of its control should read the excellent studies published on this topic. The various “death receptors,” essential effectors of any programmed cell death, were reviewed in 2003. An important member of this group is tumor necrosis factor–related apoptosis-inducing ligand (TRAIL), which preferentially induces apoptosis in transformed but not normal cells. It is expressed in normal skin and cutaneous inflammatory diseases. Another cell component that plays a role in apoptosis is the mitochondrion. This topic was reviewed in 2006.
Ackerman held a minority view that apoptosis is a type of necrosis. In reality, each is a distinctive form of cell death.
Vacuolar change (liquefaction degeneration) is often an integral part of the basal damage in the lichenoid reaction. Sometimes it is more prominent than the cell death. It results from intracellular vacuole formation and edema, as well as from separation of the lamina densa from the plasma membrane of the basal cells. Vacuolar change is usually prominent in lupus erythematosus, particularly the acute systemic form, and in dermatomyositis and some drug reactions.
As a consequence of the basal cell damage, there is variable melanin incontinence resulting from interference with melanin transfer from melanocytes to keratinocytes, as well as from the death of cells in the basal layer. Melanin incontinence is particularly prominent in some drug-induced and solar-related lichenoid lesions, as well as in patients with marked racial pigmentation.
Another feature of the lichenoid reaction pattern is the inflammatory cell infiltrate . This varies in composition, density, and distribution according to the disease. An assessment of these characteristics is important in distinguishing the various lichenoid dermatoses. Because apoptosis, unlike necrosis, does not itself evoke an inflammatory response, it can be surmised that the infiltrate in those diseases with prominent apoptosis is of pathogenetic significance and not a secondary event. Furthermore, apoptosis is the usual method of cell death resulting from cell-mediated mechanisms, whereas necrosis and possibly vacuolar change result from humoral factors, including the deposition of immune complexes.
One study has given some insight into the possible mechanisms involved in the variability of expression of the lichenoid tissue reaction in several of the diseases within this group. The study examined the patterns of expression of the intercellular adhesion molecule-1 (ICAM-1). Keratinocytes in normal epidermis have a low constitutive expression of ICAM-1, rendering the normal epidermis resistant to interaction with leukocytes. Therefore, induction of ICAM-1 expression may be an important factor in the induction of leukocyte-dependent damage to keratinocytes. In lichen planus, ICAM-1 expression is limited to basal keratinocytes, whereas in subacute cutaneous lupus erythematosus there is diffuse epidermal ICAM-1 expression, sometimes with basal accentuation. This pattern is induced by ultraviolet radiation and possibly mediated by tumor necrosis factor-α (TNF-α). In erythema multiforme, there is strong basal expression of ICAM-1, with cell surface accentuation and similar pockets of suprabasal expression, probably induced by herpes simplex virus infection.
Other molecules appear to play important roles in lichenoid dermatitis. IKKB, a subunit of the IκB kinase complex, is required for activation of NF-κB, a protein that controls DNA transcription. In an animal model, overexpression of IKKβ results in chronic inflammation with macrophages and CD45 + cells, interface dermatitis, and increased production of inflammatory cytokines by keratinocytes. This process apparently occurs independently of T and B lymphocytes. The type 1 interferon (IFN) system plays an important role in the interface dermatitis associated with autoimmune diseases, mediating a cytotoxic attack on basal keratinocytes. Evidence for this is the finding of IFN-inducible chemokine CXCL10 expression in the same location where CXCR3 + cytotoxic lymphocytes invade the epidermal basilar layer. Using a model of reconstructed human epidermis, Farley et al. explored the important roles of the Fas ligand (the expression of which by donor T cells may be essential for cutaneous acute graft-versus-host reaction) and IFN-γ. These investigators found that cytoid body formation and epidermal expression of ICAM-1 could be attributed to IFN-γ, whereas hypergranulosis was triggered by the Fas ligand, and vacuolar degeneration of the basilar layer appeared to be triggered by both the Fas ligand and IFN-γ.
In summary, the lichenoid reaction pattern includes a heterogeneous group of diseases that have in common basal cell damage. The histogenesis is also diverse and includes cell-mediated and humoral immune reactions and possibly ischemia in one condition. A discussion of the mechanisms involved in producing apoptosis is included in several of the diseases that follow. Scattered apoptotic keratinocytes can also be seen in the sunburn reaction in response to ultraviolet radiation ; these are known as “sunburn cells.” A specific histological diagnosis can usually be made by attention to such factors as:
the nature and extent of the basal damage;
the nature, composition, and distribution of the inflammatory reaction;
the amount of melanin incontinence that results from the basal damage;
the coexistence of another tissue reaction ; and
other individual characteristics.
These points are considered further in Tables 4.1–4.3 .
| Disease | Histopathological features |
|---|---|
| Lichen planus | Prominent Civatte bodies, band-like inflammatory infiltrate, wedge-shaped hypergranulosis. Hypertrophic form has changes limited to the tips of the acanthotic downgrowths and often superadded lichen simplex chronicus. The infiltrate extends around hair follicles in lichen planopilaris. Pigment incontinence is conspicuous in erythema dyschromicum perstans. |
| Lichen nitidus | Focal (papular) lichenoid lesions; some giant cells; dermal infiltrate often “clasped” by acanthotic downgrowths. |
| Lichen striatus | Clinically linear; irregular and discontinuous lichenoid reaction; infiltrate sometimes around follicles and sweat glands. |
| Lichen planus–like keratosis | Solitary; prominent Civatte body formation; solar lentigo often at margins. |
| Lichenoid drug eruptions | Focal parakeratosis; eosinophils, plasma cells and melanin incontinence may be features. Deep extension of the infiltrate occurs in photolichenoid lesions. |
| Fixed drug eruptions | Interface-obscuring infiltrate, often extends deeper than erythema multiforme; cell death often above basal layer; neutrophils often present. |
| Erythema multiforme | Interface-obscuring infiltrate; sometimes subepidermal vesiculation and variable epidermal cell death. |
| Graft-versus-host disease | Basal vacuolation; scattered apoptotic keratinocytes, sometimes with attached lymphocytes (“satellite cell necrosis”); variable lymphocytic infiltrate. |
| Lupus erythematosus | Mixed vacuolar change and Civatte bodies. Systemic LE has prominent vacuolar change and minimal cell death. Discoid lupus away from the face has more cell death and superficial and deep infiltrate; mucin; follicular plugging; basement membrane thickening. Some cases resemble erythema multiforme with cell death at all layers. |
| Dermatomyositis | May resemble acute lupus with vacuolar change, epidermal atrophy, some dermal mucin; infiltrate usually superficial and often sparse. |
| Poikilodermas | Vacuolar change; telangiectasia; pigment incontinence; late dermal sclerosis. |
| Pityriasis lichenoides | Acute form combines lymphocytic vasculitis with epidermal cell death; interface-obscuring infiltrate; focal hemorrhage; focal parakeratosis. |
| Paraneoplastic pemphigus | Erythema multiforme–like changes with suprabasal acantholysis and clefting; subepidermal clefting sometimes present. |
| Pathological change | Possible diagnoses |
|---|---|
| Vacuolar change | Lupus erythematosus, dermatomyositis, drugs and poikiloderma |
| Interface-obscuring infiltrate | Erythema multiforme, fixed drug eruption, pityriasis lichenoides (acute), paraneoplastic pemphigus, lupus erythematosus (some) |
| Purpura | Lichenoid purpura |
| Cornoid lamella | Porokeratosis |
| Deep dermal infiltrate | Lupus erythematosus, syphilis, drugs, photolichenoid eruption |
| “Satellite cell necrosis” | Graft-versus-host disease, eruption of lymphocyte recovery, erythema multiforme, paraneoplastic pemphigus, regressing plane warts, drug reactions |
| High apoptosis | Phototoxic reactions, adult-onset Still's disease, acrokeratosis paraneoplastica |
| Prominent pigment incontinence | Poikiloderma, drugs, “racial pigmentation” and an associated lichenoid reaction, erythema dyschromicum perstans and related entities |
| Eccrine duct involvement | Erythema multiforme (drug induced), lichen striatus, keratosis lichenoides chronica, periflexural exanthem of childhood |
| Additional pattern | Possible diagnoses |
|---|---|
| Spongiotic | Drug reactions (see spongiotic drug reactions), lichenoid contact dermatitis, lichen striatus, late-stage pityriasis rosea, superantigen ‘id’ reactions |
| Granulomatous | Lichen nitidus, lichen striatus (rare), lichenoid sarcoidosis, hepatobiliary disease, endocrinopathies, infective reactions including secondary syphilis, herpes zoster infection, HIV infection, tinea capitis, Mycobacterium marinum , and M. haemophilum ; drug reactions (often in setting of Crohn's disease or rheumatoid arthritis—atenolol, allopurinol, captopril, cimetidine, enalapril, hydroxychloroquine, simvastatin, sulfa drugs, tetracycline, diclofenac, erythropoietin) |
| Vasculitic | Pityriasis lichenoides, perniosis (some cases), pigmented purpuric dermatosis (lichenoid variant), persistent viral reactions, including herpes simplex |
| Vasculitic/spongiotic | Gianotti–Crosti syndrome, some other viral/putative viral diseases, rare drug reactions |
A discussion of the various lichenoid (interface) dermatoses follows. The conditions listed as other lichenoid (interface) diseases are discussed only briefly because they are considered in detail in other chapters.
Lichen planus, a relatively common eruption of unknown etiology, displays violaceous, flat-topped papules, which are usually pruritic. A network of fine white lines (Wickham's striae) may be seen on the surface of the papules. There is a predilection for the flexor surface of the wrists, the trunk, the thighs, and the genitalia. Palmoplantar lichen planus appears to be more common than once thought. It is one of the most disabling, painful, and therapy-resistant variants of lichen planus. This form of the disease may be confined to the palms and soles and can present with hypertrophic lesions or as a palmoplantar keratoderma. Oral lesions are common; rarely, the esophagus is also involved. Lesions localized to the lip, vulva, and an eyelid have been reported. Lichen planus localized to a radiation field may represent an isomorphic response. It has also developed in healed herpes zoster scars. Nail changes occur, and, as with oral lesions, these may be the only manifestations of the disease. Clinical variants include atrophic, annular, hypertrophic, linear, zosteriform or segmental, erosive, oral, actinic, follicular, erythematous, and bullous forms. They are discussed further later. An eruptive variant also occurs. Spontaneous resolution of lichen planus is usual within 12 months, although postinflammatory pigmentation may persist for some time afterward.
Familial cases are uncommon, and rarely these are associated with HLA-D7. An association with HLA-DR1 has been found in nonfamilial cases. There is an increased frequency of HLA-DR6 in Italian patients with hepatitis C virus–associated oral lichen planus. Lichen planus is rare in children, but some large series have been published. Lichen planus has been reported in association with immunodeficiency states, internal malignancy, including thymoma, Still's disease, primary biliary cirrhosis, hypothyroidism, peptic ulcer (but not Helicobacter pylori infection), chronic hepatitis C infection, hepatitis B vaccination, influenza vaccination, human herpesvirus type 7 (HHV-7) replication, simultaneous measles–mumps–rubella and diphtheria–tetanus–pertussis-polio vaccinations, rabies vaccination, stress, vitiligo, pemphigus, porphyria cutanea tarda, radiotherapy, ulcerative colitis, chronic giardiasis, a Becker's nevus, and lichen sclerosus et atrophicus with coexisting morphea. Dyslipidemia has been reported in lichen planus in several studies; patients are reported to have elevated levels of triglycerides, total cholesterol, and low-density lipoproteins and lower levels of high-density lipoproteins. Despite the association between lichen planus and hepatitis C virus (HCV) infection, its incidence in patients with lichen planus in some areas of the world is not increased compared with that of a control group. Lichen planus patients have a significantly higher risk than controls of being HCV seropositive, and there is a similar odds ratio of having lichen planus among HCV patients; this appears to only partly depend on geographical effect. A large European study showed that lichen planus is associated with HCV but not with HBV. On the other hand, although HCV and oral lichen planus are significantly associated in a number of studies, most patients with oral lichen planus are not affected by HCV. The exacerbation or appearance of lichen planus during the treatment of HCV infection and other diseases with IFN-α has been reported. Furthermore, effective therapy for the HCV does not clear the lichen planus. Earlier reports linking lichen planus to infection with human papillomavirus (HPV) may have been a false-positive result ; however, interest in a possible HPV connection is unabated. Recent reports have shown a significant correlation between human papillomavirus—especially HPV-16—and oral lichen planus (OLP), reflected in part by a marked clonal expansion of CD8 + T cells with an increased frequency of an HPV-16–specific population of these cells. Squamous cell carcinoma is a rare complication of the oral and vulvar cases of lichen planus and of the hypertrophic and ulcerative variants (see later). One study found no significant transformation risk of cutaneous lichen planus to squamous cell carcinoma, although there is a significant risk of malignant transformation in mucosal lichen planus. A contact allergy to metals, flavorings, and plastics may be important in the etiology of oral lichen planus. The role of mercury in dental amalgams is discussed further later.
Much has been learned about the pathogenesis of lichen planus, particularly through investigations of OLP. Cell-mediated immune reactions appear to be important. It has been suggested that these reactions are precipitated by an alteration in the antigenicity of epidermal keratinocytes, possibly caused by a virus or a drug or by an allogeneic cell. Keratinocytes in lichen planus express HLA-DR on their surface, and this may be one of the antigens that has an inductive or perpetuating role in the process. Keratinocytes also express fetal cytokeratins (CK13 and CK8/18), but whether they are responsible for triggering the T-cell response is speculative. The cellular response initially consists of CD4 + lymphocytes ; they are also increased in the peripheral blood. In recent years, attention has focused on the role of cytotoxic CD8 + lymphocytes in a number of cell-mediated immune reactions in the skin. They appear to play a significant role as the effector cell, whereas the CD4 + lymphocyte, usually present in greater numbers, plays its traditional helper role. There is evidence that, although CD4 + CD25 + regulatory T cells are increased, they may be functionally impaired, possibly by keratinocytes that possess Toll-like receptor–mediated B7-H1 (PD-L1), suppressing T-cell activation and proliferation. In lichen planus, CD8 + cells appear to recognize an antigen associated with MHC class I on lesional keratinocytes, resulting in their death by apoptosis. Bcl -2, a proto-oncogene that protects cells from apoptosis, is increased in lichen planus. It may allow some cells to escape apoptosis, prolonging the inflammatory process. The recruitment of lymphocytes to the interface region may be the result of the chemokine MIG (monokine induced by IFN-γ). Lymphokines produced by these T lymphocytes—including IFN-γ; interleukin (IL)-1β, -4, and -6; perforin ; granzyme B ; granulysin ; T-cell–restricted intracellular antigen (Tia-1); and tumor necrosis factor—may have an effector role in producing the apoptosis of keratinocytes. The other pathway involves the binding of Fas ligand to Fas, which triggers a caspase cascade. In both oral and cutaneous lichen planus, CD8 + cells predominate in the epithelial and subepithelial compartments, with CD4 + cells playing a helper role by secretion of T helper 1 (Th1) cytokines. Activated CD8 + cells promote basilar keratinocyte apoptosis through either granzyme B or Fas/Fas ligand pathways. As one possible explanation of how this mechanism might work in lichen planus, elevated osteopontin levels may upregulate CD44, with resultant T-cell resistance to apoptosis and accumulation of activated T cells in lichen planus lesions. Additionally, the increased levels of S100AB detected in skin lesions and sera of lichen planus patients appear to induce an enhanced cytotoxic response, with increased expression of IL-1, TNF, and IL-6 in CD8 + T cells. Gene expression profiling in lichen planus has found that type I IFN–inducible genes are significantly expressed. Plasmacytoid dendritic cells appear to be a major source of these type I IFNs in lichen planus. They play a major role in cytotoxic skin inflammation by increasing the expression of IPIO/CXCRIO and recruiting effector cells via CXCR3. The CXCR3 ligand, CXCL9, is the most significant marker for lichen planus. A unique subclass of cytotoxic T lymphocyte (γδ) is also found in established lesions. Langerhans cells are increased, and it has been suggested that these cells initially process the foreign antigen. Factor XIIIa–positive cells and macrophages expressing lysozyme are found in the dermis.
There is evidence that expression of the microRNAs miRNA-146a and miRNA-155 is increased in lesions of oral lichen planus. MicroRNAs are known to participate in immune response regulation. Other miRNAs that have been implicated in the pathogenesis of lichen planus include miRNA-125b, miRNA-137, miRNA-138, miRNA-203, miRNA-320a, miRNA-362, miRNA-562, miRNA-578, miRNA-635, and miRNA-4484. A review of microRNAs in OLP and their interactions with various cytokines has been published.
Increased oxidative stress, increased lipid peroxidation, and an imbalance in the antioxidant defense system are present, though their exact role in the pathogenesis of lichen planus is unknown. Patients with OLP have an increase in serum malondialdehyde and decrease in serum total antioxidant capacity compared with healthy controls. Another study showed increased prolidase activity and oxidative stress and imbalance in the antioxidant defense system in biological fluids from patients with OLP, but there were similar levels in oral lichenoid contact reactions.
Matrix metalloproteinases may play a concurrent role by destroying the basement membrane. Evidence from an animal model suggests that keratinocytes require cell survival signals, derived from the basement membrane, to prevent the onset of apoptosis. In oral lichen planus, MMP-1 and MMP-3 may be principally associated with erosion development. Altered levels of heat shock proteins are found in the epidermis in lichen planus.
Most studies have found no autoantibodies and no alteration in serum immunoglobulins in lichen planus. However, a lichen planus–specific antigen has been detected in the epidermis, and a circulating antibody to it has been found in the serum of individuals with lichen planus. Its pathogenetic significance remains uncertain. Antibodies to desmoplakins I and II have been found in oral and genital lesions, possibly representing epitope spreading. Increased levels of desmoglein III antibodies have been reported in OLP, particularly in the erosive variant, and some patients with mucosal lichen planus may have low levels of circulating anti BP180 antibodies; the significance of these findings, at present, is unclear.
Replacement of the damaged basal cells is achieved by an increase in actively dividing keratinocytes in both the epidermis and the skin appendages. This is reflected in the pattern of keratin expression, which resembles that seen in wound healing; cytokeratin 17 (CK17) is found in suprabasal keratinocytes.
Potent topical corticosteroids remain the treatment of choice for lichen planus in patients with classic and localized disease. Topical tacrolimus is an effective alternative to clobetasol in the management of OLP. For widespread disease and mucosal lesions, a short course of systemic corticosteroids may provide some relief. Cyclosporine (ciclosporin), hydroxychloroquine, retinoids, dapsone, mycophenolate mofetil, sulfasalazine, alefacept, and efalizumab have all been used at various times. Alitretinoin has been useful in treating lichen planus of the nails and as an option for refractory cutaneous disease. Mycophenolate mofetil has been shown to be effective in severe ulcerative lichen planus. Erosive oral disease has been treated with tacrolimus mouthwash, whereas erosive flexural lichen planus has responded to thalidomide and 0.1% tacrolimus ointment. Palmoplantar disease may be resistant to treatment and require cyclosporine.
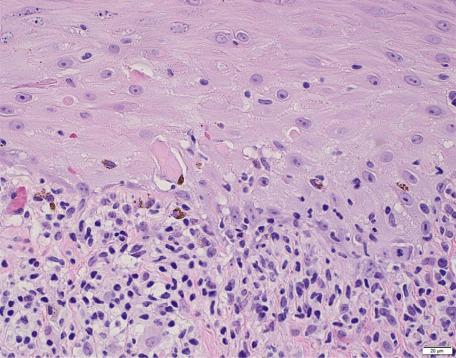
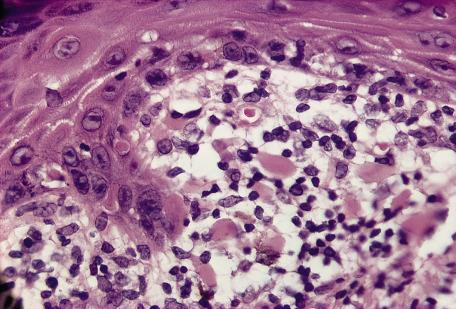
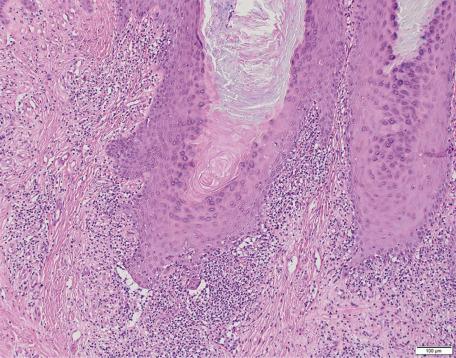
The basal cell damage in lichen planus takes the form of multiple, scattered Civatte bodies ( Fig. 4.2 ). Eosinophilic colloid bodies, which are periodic acid–Schiff (PAS) positive and diastase resistant, are found in the papillary dermis ( Fig. 4.3 ). They measure approximately 20 µm in diameter. The basal damage is associated with a band-like infiltrate of lymphocytes and some macrophages that press against the undersurface of the epidermis ( Fig. 4.4 ). Occasional lymphocytes extend into the basal layer, where they may be found in close contact with basal cells and sometimes with Civatte bodies. The infiltrate tends to obscure the interface but does not extend into the mid-epidermis. Karyorrhexis is sometimes seen in the dermal infiltrate. Rarely, plasma cells can be found in cutaneous lesions, but most often they are not identified—a feature that can be useful in differential diagnosis. In exceptional cases, they can be numerous. In some instances, plasma cells may be found because cutaneous lesions have arisen in anatomical sites where these cells tend to be prevalent, such as the face, posterior neck, intertriginous sites, and pretibial areas. Although they are usually present in lesions adjacent to or on mucous membranes, plasma cells are sometimes surprisingly sparse even in these locations. There is variable melanin incontinence, but this is most conspicuous in lesions of long duration and in dark-skinned people.
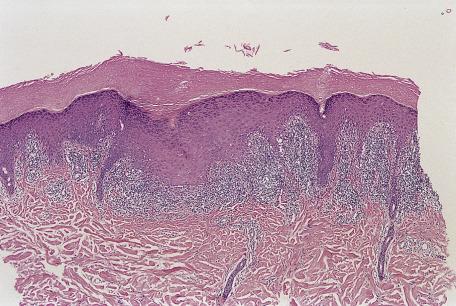
Other characteristic epidermal changes include hyperkeratosis, wedge-shaped areas of hypergranulosis related to the acrosyringia and acrotrichia, and variable acanthosis. At times, the rete ridges become pointed, imparting a “sawtooth” appearance to the lower epidermis. There is sometimes mild hypereosinophilia of keratinocytes in the malpighian layer. Small clefts (Caspary–Joseph spaces) may form at the dermoepidermal junction secondary to the basal damage. The eccrine duct adjacent to the acrosyringium is sometimes involved. A variant in which the lichenoid changes were localized entirely to the acrosyringium has been reported. Transepidermal elimination with perforation is another rare finding. The formation of milia may be a late complication.
Ragaz and Ackerman studied the evolution of lesions in lichen planus. They found an increased number of Langerhans cells in the epidermis in the very earliest lesions, before there was any significant infiltrate of inflammatory cells in the dermis. In resolving lesions, the infiltrate is less dense, and there may be minimal extension of the inflammatory infiltrate into the reticular dermis.
As previously mentioned, some diseases exhibiting the lichenoid tissue reaction may also show features of another tissue reaction pattern as a major or minor feature. These conditions are listed in Table 4.3 .
Direct immunofluorescence of involved skin shows colloid bodies in the papillary dermis, staining for complement and immunoglobulins, particularly IgM. An irregular band of fibrin is present along the basal layer in most cases. Often there is irregular extension of the fibrin into the underlying papillary dermis ( Fig. 4.5 ). One study found colloid bodies in 60% of cases of lichen planus, whereas fibrin was present in all cases. Immunofluorescent analysis of the basement membrane zone, using a range of antibodies, suggests that disruption occurs in the lamina lucida region. Other studies have shown a disturbance in the epithelial anchoring system.
When assessing erosive lesions of OLP for possible dysplastic changes, direct oral microscopy may be helpful. Examination of OLP lesions by this method, whose principles are derived from colposcopy and dermoscopy, can significantly increase the likelihood of finding dysplasia in a subsequent biopsy compared with lesion selection based naked eye inspection alone.
Ultrastructural studies have confirmed that lymphocytes attach to basal keratinocytes, resulting in their death by apoptosis. Many cell fragments, beyond the limit of resolution of the light microscope, are formed during the budding of the dying cells. The cell fragments are phagocytosed by adjacent keratinocytes and macrophages. The large tonofilament-rich bodies that result from redistribution of tonofilaments during cell fragmentation appear to resist phagocytosis and are extruded into the upper dermis, where they are recognized on light microscopy as colloid bodies. Various studies have confirmed the epidermal origin of these colloid bodies. There is a suggestion from some experimental work that sublethal injury to keratinocytes may lead to the accumulation of tonofilaments in their cytoplasm. Some apoptotic bodies contain more filaments than would be accounted for by a simple redistribution of the usual tonofilament content of the cell.
The moat important distinction is between lichen planus and lupus erythematosus . This can be a particular problem with scalp lesions, where the infiltrates of lichen planopilaris can closely resemble the follicular involvement of lupus erythematosus (see later), or with lupus lesions that display dense superficial dermal infiltrates. Atrophic lichen planus can bear a resemblance to poikilodermatous lesions of lupus erythematosus, whereas hypertrophic lesions of discoid lupus erythematosus (DLE) can resemble their hypertrophic lichen planus counterpart. In contrast to lichen planus, lupus erythematosus most often shows epidermal atrophy, persistent vacuolar change of the basilar layer rather than basal keratinocyte loss (with flattening or “sawtoothing” of the epidermal base), basement membrane zone thickening (especially in lesions of at least 6 months’ duration), a deep as well as superficial dermal infiltrate that involves vessels and sweat glands as well as follicles, and often interstitial dermal mucin deposition. In addition, a degree of panniculitis is seen in a significant number of lupus cases, consisting of mild patchy lymphocytic infiltrates, mucin deposition, or lipoatrophy; those changes are not seen in lichen planus. In a recent selective immunohistochemical study comparing DLE and lichen planus, Ramezani et al. found that CD34 had the highest specificity and positive predictive value for the diagnosis of lichen planus, whereas CD3 showed the highest sensitivity and negative predictive value for that diagnosis. Other common problems in differential diagnosis arise with lichenoid keratoses and lichenoid drug eruptions . These entities are further discussed later. Most of the other lichenoid dermatoses lack the full constellation of findings of lichen planus. Lichenoid actinic keratosis , or actinic cheilitis , shows basilar keratinocyte atypia that is disproportionate to that expected as a response to inflammation alone, and often the atypical changes extend laterally beyond the zone of most intense dermal inflammation. Fully developed lichen sclerosus is quite distinctive, but early disease may show a band-like superficial infiltrate partly obscuring the dermoepidermal interface; together with vacuolar alteration of the basilar layer, this can produce an image somewhat reminiscent of lichen planus. However, the loss of basilar keratinocytes with sawtoothing or flattening of the epidermal base is often not a feature in lichen sclerosus, and dermal edema or early homogenization of papillary collagen may be evident even in early stages of the disease. Poikilodermatous mycosis fungoides with a heavy, band-like infiltrate could be confused with the atrophic variety of lichen planus, but atypical lymphocytes, “lining up” of singly dispersed lymphocytes along the basilar layer, and wiry papillary dermal collagen may be identified—findings not expected in lichen planus. In addition, mycosis fungoides is more prone to have eosinophils and plasma cells in the dermal infiltrate. In erythema multiforme and fixed drug eruption , dense, band-like infiltrates obscuring the dermoepidermal interface would be unusual. The rapid onset of these conditions usually means that the epidermis is of approximately normal thickness, and an ordinary-appearing, basket-woven stratum corneum is often preserved. Furthermore, apoptotic keratinocytes in lichen planus are usually observed at the basilar layer or within the papillary dermis, where they are often arranged in clusters; in erythema multiforme and fixed drug eruption, apoptotic keratinocytes are usually found widely scattered throughout all levels of the epidermis. Keratosis lichenoides chronica and lichen striatus often show dermal infiltrates in patchy distribution, with involvement of the mid to deep dermis and sometimes perieccrine lymphocytic infiltration.
Direct immunofluorescence can sometimes be helpful in differential diagnosis. The combination of junctional apoptotic bodies staining for IgM and a fibrin band along the dermoepidermal junction is characteristic of lichen planus. Although it can be mimicked by other lichenoid dermatoses, these features differ from lupus erythematosus, which when positive shows particulate, thick linear, or occasionally linear deposition of immunoglobulin, C3 complement, or fibrin along the dermoepidermal junction. Occasionally, an antinuclear antibody can be observed in the highlighting of keratinocyte nuclei with antibodies to IgG. Therefore, this procedure can be helpful in cases of lichen planus–lupus erythematosus overlap. Immunofluorescent study can also be useful when evaluating mucous membrane biopsies, where the differential diagnosis includes both lichen planus and cicatricial pemphigoid (one example is the condition known as desquamative gingivitis, which can be a manifestation of either disease). In contrast to lichen planus, cicatricial pemphigoid would show linear deposition of immunoglobulin and/or C3 complement along the epithelial–stromal interface.
A number of clinical variants of lichen planus occur. In some, typical lesions of lichen planus are also present. These variants are discussed in further detail here.
Atrophic lesions may resemble porokeratosis clinically. Typical papules of lichen planus are usually present at the margins. A rare form of atrophic lichen planus is composed of annular lesions. It is composed of violaceous plaques of annular morphology with central atrophy. Hypertrophic lichen planus has been reported at the edge of a plaque of annular atrophic lichen planus. Experimentally, there is an impaired capacity of the atrophic epithelium to maintain a regenerative steady state.
The epidermis is thin and there is loss of the normal rete ridge pattern. The infiltrate is usually less dense than in typical lichen planus. It may be lost in the center of the lesions.
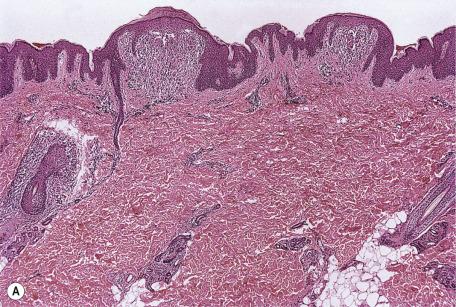
Hypertrophic lesions are usually confined to the shins, although sometimes they are more generalized. They appear as single or multiple pruritic plaques, which may have a verrucous appearance ; they usually persist for many years. A case from the vulvar region resembled condylomata acuminata. Rarely, squamous cell carcinoma develops in lesions of long standing, and in one such case, widely metastatic carcinoma ensued. In one retrospective chart review and database search, there were 38 cases of squamous cell carcinoma arising in hypertrophic lichen planus in 16 women and 22 men. Cutaneous horns, keratoacanthoma, and verrucous carcinoma may also develop in hypertrophic lichen planus. Changes microscopically resembling hypertrophic lichen planus have been seen in the setting of pseudoepitheliomatous hyperplasia in a red pigment tattoo.
Hypertrophic lichen planus has been reported in several patients infected with the human immunodeficiency virus (HIV). It also occurs in patients with HCV infection. It may occur in children.
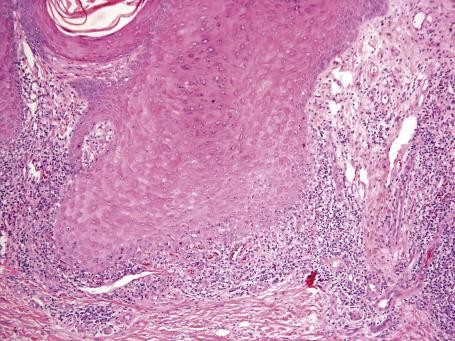
The epidermis shows prominent hyperplasia and overlying orthokeratosis ( Fig. 4.6 ). At the margins there is usually psoriasiform hyperplasia representing concomitant changes of lichen simplex chronicus secondary to the rubbing and scratching. If the epidermal hyperplasia is severe, it may mimic a squamous cell carcinoma on a shave biopsy. Vertically oriented collagen (“vertical-streaked collagen”) is present in the papillary dermis in association with the changes of lichen simplex chronicus.
With contact, dry dermoscopy, hypertrophic lichen planus shows round and reticular whitish structures, some with thin, branching projections. These correspond with Wickham's striae. With the use of alcohol immersion, there are multiple comedo-like openings containing yellowish material and chalk-white structureless areas. These and other findings differ from those of prurigo nodularis, which include red dots, red globules, and pearly white areas with peripheral striations.
A common problem is the distinction between hypertrophic lichen planus and keratoacanthoma or well-differentiated squamous cell carcinoma. A history of occurrence over the pretibial areas and of more typical lesions of lichen planus elsewhere would of course be helpful. Microscopically, the lack of significant cytological atypia in the face of a lichenoid host tissue reaction is a clue to the diagnosis of hypertrophic lichen planus. Compared with hypertrophic lichen planus, keratoacanthoma has a comparable proliferative index, but it shows increased expression of p53 and, as a significant difference from hypertrophic lichen planus, perforating elastic fibers, which can be demonstrated with the Verhoeff–van Gieson stain.
The basal cell damage is usually confined to the tips of the rete ridges and may be missed on casual observation ( Fig. 4.7 ). The infiltrate is not as dense or as band-like as in the usual lesions of lichen planus. A few eosinophils and plasma cells may be seen in some cases in which the ingestion of β-blockers can sometimes be incriminated.
Xanthoma cells have been found in the dermis, localized to a plaque of hypertrophic lichen planus, in a patient with secondary hyperlipidemia. This is an example of dystrophic xanthomatization.
Annular lichen planus is one of the more rare clinical forms of lichen planus. In a series of 20 patients, published some years ago, 18 were men and 2 women. Sites of involvement included the axilla, penis, extremities, and groin. Eighteen of the patients had purely annular lesions, whereas 2 of the patients had a few purple polygonal papules as well. The majority of lesions showed central clearing with a purple to white annular edge. Lesions varied from 0.5 to 2.5 cm in diameter. (Atrophic annular lesions were discussed with atrophic lichen planus [see p. 55 ]). The majority of patients were asymptomatic. Oral and genital lesions have been reported in annular lichen planus. Recent reports have described involvement of the areola and generalized lesions in an HIV-positive patient. The cases reported as annular lichenoid dermatitis of youth appear to be a distinct entity, but further reports will be necessary to clarify its exact position in the spectrum of lichen planus. The lesions are persistent erythematous macules and annular patches mostly localized on the groin and flanks. In all cases, the clinical picture has been suggestive of morphea, mycosis fungoides, or annular erythema, but these conditions could be excluded on the basis of the distinctive superficial lichenoid reaction with massive necrosis/apoptosis of the keratinocytes at the tips of the rete ridges. Patch testing has given negative results.
Annular lichenoid dermatitis of youth features slight, basket-woven hyperkeratosis, elongated rete ridges, and a lichenoid lymphocytic infiltrate with vacuolar alteration concentrated at the rete ridge tips. Over time, this process results in rete ridges with quadrangular contours. The presence in a recent case of microscopic foci more typical of classic lichen planus raises the possibility that annular lichenoid dermatitis of youth may fall within the spectrum of lichen planus.
Linear lichen planus is a rare variant that must be distinguished from linear nevi and other dermatoses with linear variants. It occurs in less than 0.5% of patients with lichen planus. Linear lichen planus usually involves the limbs. It may follow the lines of Blaschko. It has been reported in association with hepatitis C infection, HIV infection, and metastatic carcinoma. There have been several cases described as lichen planus pigmentosus in a linear distribution. There is also a zosteriform variant of lichen planus, in which lesions form along dermatomes or in a zonal distribution. In one report, varicella–zoster viral antigens were detected in eccrine epithelium of zosteriform lesions. Sometimes, linear lesions are associated with disseminated nonsegmental papules of ordinary lichen planus. The linear lesions are usually more pronounced in these combined cases.
Ulcerative lichen planus (erosive lichen planus) is characterized by ulcerated and bullous lesions on the feet. Mucosal lesions, alopecia, and more typical lesions of lichen planus are sometimes present. Squamous cell carcinoma may develop in lesions of long standing. Variants of ulcerative lichen planus involving the perineal region, penis, the mouth, or the vulva, vagina, and mouth—the vulvovaginal-gingival syndrome —have been reported. A patient with erosive lesions of the flexures has also been described.
A list of clinicopathological diagnostic criteria for erosive vulvar lichen planus has been developed through an international electronic-Delphi consensus exercise. Nine criteria were developed, at least three of which should be present to allow a diagnosis. These are listed in Table 4.4 .
| Well demarcated erosions or erythematous areas at the vaginal introitus |
| Presence of a hyperkeratotic lesional border or Wickham's striae in surrounding skin |
| Symptoms of pain or burning |
| Scarring or loss of normal architecture |
| Presence of vaginal inflammation |
| Involvement of other mucosal surfaces |
| Presence of a well-defined inflammatory band involving dermoepidermal junction |
| Presence of a band-like inflammatory infiltrate consisting mainly of lymphocytes |
| Signs of basal layer degeneration (e.g., Civatte bodies, abnormal keratinocytes, basal apoptosis) |
Castleman's tumor (giant or angiofollicular lymph node hyperplasia) and malignant lymphoma are rare associations of erosive lichen planus ; long-term therapy with hydroxyurea and infection with hepatitis C are others. Screening for hepatitis C and B has not been considered necessary for vulval lichen planus in some countries.
Antibodies directed against a nuclear antigen of epithelial cells have been reported in patients with erosive lichen planus of the oral mucosa. Weak circulating basement membrane zone antibodies are also present.
High-potency topical corticosteroids have been used to treat erosive lichen planus. Relief of symptoms was obtained in 71% of cases of vulvar disease in one series. A good response to topical tacrolimus, particularly in vulvar disease, has been achieved in recent years. A randomized, double-blind control trial of topical thalidomide showed efficacy in erosive oral lichen planus. Azathioprine, retinoids, dapsone, methotrexate, and hydroxychloroquine have also been used, but there have been no controlled trials of these various treatments. Photodynamic therapy can also be used.
Penile erosive lichen planus responded to circumcision in one case.
There is epidermal ulceration with more typical changes of lichen planus at the margins of the ulcer. Plasma cells are invariably present in cases involving mucosal surfaces. Lymphocytes are the predominant cell type in reticular and erosive forms of oral lichen planus and are more frequent in the latter; in addition, apoptosis is comparatively diminished among inflammatory cells in the erosive cases. Eosinophils were prominent in the oral lesions of a case associated with methyldopa therapy. In erosive lichen planus of the vulva, there is widespread disruption in several basement membrane zone components, including hemidesmosomes and anchoring fibrils.
A small number of cases of vulvo-vaginal erosive lichen planus display areas of regenerative erosive vulvitis with loss of maturation and resulting nuclear atypia; such cases require careful clinicopathological correlation to allow distinction from better-differentiated examples of vulvar intraepithelial neoplasia.
OLP has a prevalence of approximately 0.5% to 2%. It is a disease of middle-aged and older persons, with a female predominance. The disease may persist for many years despite treatment. Spontaneous remission is rare.
There is a low prevalence of OLP among HCV-infected patients ; the keratotic form of OLP is more prevalent in this disease.
There has been a resurgence of interest in the role of an allergy to mercury in dental amalgams in the pathogenesis of OLP. Dental plaque and calculus, which have also been shown to contain mercury, are also associated with the disease. It appears that in cases unassociated with cutaneous lichen planus, oral lichen planus may often be cleared by the partial or complete removal of amalgam fillings, if there is a positive patch test reaction to mercury compounds. Because mercury-associated disease does not have all the clinical and/or histological features of oral lichen planus, the term oral lichenoid lesion is sometimes used for these cases. In one case, lichen planus developed in a herpes zoster scar on the face after an amalgam (mercury) filling. Oral squamous cell carcinoma is a rare complication of oral lichen planus, with an estimated risk of 0.3% to 3%. There appears to be a higher incidence of malignant transformation in OLP among smokers, alcoholics, and HCV-infected patients, although further investigation is needed. It appears that desmocollin-1 expression in oral atrophic lichen planus is a powerful predictor of the development of dysplasia, whereas both desmocollin-1 and E-cadherin expressions are predictors of the development of cancer.
Oral lichen planus mimics to varying degrees the changes seen in cutaneous disease. The infiltrate is usually quite heavy, and it may contain plasma cells, particularly in erosive forms when neutrophils may also be present. Both cells are also found in amalgam-associated disease. Apoptotic keratinocytes tend to occur at a slightly higher level in the mucosa than they do in the cutaneous form, possibly a reflection of amalgam-related cases. Features said to be more likely in amalgam-associated disease are deep extension of the infiltrate, perivascular extension of the infiltrate, and the presence of plasma cells and neutrophils in the connective tissue.
There is a lower density of CD1a + cells in amalgam lichenoid reactions and in lichenoid mucositis induced by drugs than in OLP, but at this point it is unclear whether this difference can be exploited in practical diagnostic work. Oral lichenoid lesion, a term proposed by Finne et al., includes those lichenoid lesions associated with drug intake, systemic disease (such as chronic liver disease), food or flavor allergies, hypertension, or diabetes mellitus, as well as dental amalgam. There are significantly more granulocytes (including eosinophils) and plasma cells in oral lichenoid lesions than in OLP. Image-based DNA ploidy analysis can be more useful in predicting malignant transformation in patients with OLP than either clinical or histopathological evaluation alone.
Erythema dyschromicum perstans (ashy dermatosis, lichen planus pigmentosus) is a slowly progressive, asymptomatic, ash-colored or brown macular hyperpigmentation that has been reported from most areas of the world; it is most prevalent in Latin America. Lesions are often quite widespread, although there is a predilection for the trunk and upper limbs. Unilateral and linear lesions have been described. Periorbital hyperpigmentation is a rare presentation of this disease. Activity of the disease may cease after several years. Resolution is more likely in children than in adults. It has been proposed that “erythema dyschromicum perstans” should be used when lesions have, or have previously had, an erythematous border, whereas “ashy dermatosis” should be used for other cases without this feature. This controversy continues, although most clinicians regard the terms as synonymous.
Erythema dyschromicum perstans has been regarded as a macular variant of lichen planus on the basis of the simultaneous occurrence of both conditions in several patients and similar immunopathological findings. Paraphenylenediamine, aminopenicillins, and omeprazole have been incriminated in its cause, although this has not been confirmed. Another study showed positive patch test reactions, to a variety of agents, in about 40% of patients with diagnoses of either erythema dyschromicum perstans or lichen planus pigmentosus, the significance of which (in terms of etiology) remains to be determined. This condition has also been reported in patients with HIV infection and those with HCV infection. There appears to be a genetic susceptibility to the disease. In one Mexican study, there was a significant increase in HLA-DR4, particularly the *0407 subtype, in patients with the disease.
Lichen planus pigmentosus, originally reported from India, is thought by some to be the same condition, although this has been disputed. Linear “segmental,” zosteriform, and blaschkoid variants have been reported. In a study of 124 patients from India with lichen planus pigmentosus, the face and neck were the sites most commonly affected, with pigmentation varying from slate gray to brownish black. Lichen planus was also present in 19 patients. The term lichen planus pigmentosus inversus has been used for cases with predominant localization of the disease in intertriginous areas. Lichen planus pigmentosus has been reported in association with a head and neck cancer and with concurrent acrokeratosis paraneoplastica (see p. 624 ). Both conditions cleared after treatment of the cancer. There is apparently a strong association between lichen planus pigmentosus and frontal fibrosing alopecia, with at least 83 cases reported to date. In these cases, the development of lichen planus pigmentosus often precedes that of frontal fibrosing alopecia. Lichen planus pigmentosus has also been reported in association with lichen planopilaris and the twenty nail dystrophy of lichen planus.
Various therapies have been tried for erythema dyschromicum perstans, but with little benefit. They include sun protection, chemical peels, corticosteroids, and chloroquine. Some patients have responded to dapsone and to clofazimine, and recent reports have indicated success using isotretinoin and topical tacrolimus with or without laser therapy.
In the active phase, there is a lichenoid tissue reaction with basal vacuolar change and occasional Civatte bodies ( Fig. 4.8 ). The infiltrate is usually quite mild compared with lichen planus. Furthermore, there may be deeper extension of the infiltrate, which is usually perivascular. There may also be mild exocytosis of lymphocytes. There is prominent melanin incontinence, and this is the only significant feature in older lesions. Subepidermal fibrosis was present in one case. The pigment usually extends deeper in the dermis than in postinflammatory pigmentation of other causes. Cases reported as lichen planus pigmentosus (discussed previously) have similar histological features ; Romiti et al. found lichenoid changes involving sweat duct epithelia and sebaceous glands in their series of cases.
Immunofluorescence has shown IgM, IgG, and complement-containing colloid bodies in the dermis, as in lichen planus. There was a predominance of CD8 + lymphocytes in the dermis in one study. The exocytosing lymphocytes expressed cutaneous lymphocyte antigen (CLA). Apoptosis and residual filamentous bodies are present on electron microscopy. Another ultrastructural study showed irregular, immature melanosomes in keratinocytes and peripheral localization of melanosomes within keratinocytes.
Dermoscopy of lichen planus pigmentosus has shown four patterns of pigmentation: a pseudo-network, dotted, speckled blue-gray, and blue-grey dots in circles. Some patients also had rhomboidal structures, focal erythema and telangiectasias.
The pigmentary incontinence seen in late-stage lesions is quite nonspecific, showing overlap with melasma , third-stage incontinentia pigmenti , and numerous other conditions. Identifying traces of vacuolar alteration of the basilar layer or rare Civatte bodies can sometimes suggest the possibility of erythema dyschromicum perstans in preference to these other conditions, but often, clinical information is needed to secure a correct diagnosis.
Lichen planus actinicus is a distinct clinical variant of lichen planus in which lesions are limited to sun-exposed areas of the body. It has a predilection for certain races, particularly young individuals of Asian, Middle Eastern, and Indian descent. There is some variability in the clinical expression of the disease in different countries, and this has contributed to the proliferation of terms used— lichen planus tropicus, lichen planus subtropicus, lichenoid melanodermatitis , and summertime actinic lichenoid eruption (SALE). It has been suggested that SALE is an actinic variant of lichen nitidus. The development of pigmentation in some cases has also led to the suggestion that there is overlap with erythema dyschromicum perstans (discussed previously). The pigmentation may take the form of melasma-like lesions. Such lesions have also been reported in childhood cases. Other common variants are annular hyperpigmented (the most common form), dyschromic, and classic lichenoid. Lesions have been induced by repeated exposure to ultraviolet radiation.
A rare erythematous variant has been described in a patient with chronic active hepatitis B infection.
Various treatments have been used, including hydroxychloroquine, topical and intralesional corticosteroids combined with topical sunscreens, and retinoids. Oral cyclosporine has also been used.
The appearances resemble lichen planus quite closely, although there is usually more marked melanin incontinence and there may be focal parakeratosis. The inflammatory cell infiltrate in lichen planus actinicus is not always as heavy as it is in typical lesions of lichen planus.
Numerous immunoglobulin-coated cytoid bodies are usually present on direct immunofluorescence.
Lichen planopilaris (follicular lichen planus) is a clinically heterogeneous variant of lichen planus in which keratotic follicular lesions are present, often in association with other manifestations of lichen planus. It typically affects middle-aged women and men. The annual incidence in four U.S. hair research centers varied from 1.15% to 7.59% of new cases, reflecting its relative rarity. The most common and important clinical group is characterized by scarring alopecia of the scalp, which is generalized in approximately half of these cases. The keratotic follicular lesions and associated erythema are best seen at the margins of the scarring alopecia. In this group, changes of lichen planus are present or develop subsequently in approximately 50% of cases. Rare cases have been reported in children.
A subtle form of lichen planopilaris occurs in areas of androgenetic alopecia and appears to target miniaturized, vellus follicles. Diagnosis of this variant requires an index of suspicion, which may be prompted by erythema and scaling in the involved areas. Definitive diagnosis requires dermoscopy and histopathology. Recognition of this variant is important, as it would represent a contraindication to hair transplantation, especially in the face of active disease.
The Graham Little–Piccardi–Lassueur syndrome is a rare but closely related entity in which there is cicatricial alopecia of the scalp, follicular keratotic lesions of glabrous skin, and variable alopecia of the axillae and groins. It has been reported in a patient with androgen insensitivity syndrome (testicular feminization).
Two other clinical groups occur, but they have not received as much attention. In one, there are follicular papules, without scarring, usually on the trunk and extremities. In the other, which is quite rare, there are plaques with follicular papules, usually in the retroauricular region, although other sites can be involved. This variant has been called lichen planus follicularis tumidus .
Rare variants of lichen planopilaris include a linear form and lesions confined to the vulva. It has been reported in a patient with erythema dyschromicum perstans and in another with scleroderma en coup de sabre. It has developed in patients receiving etanercept or infliximab therapy. Lichen planopilaris has also been reported to follow hair transplantation or facelift surgery, scalp trauma, and whole brain irradiation. Retrospective studies have shown a relationship between lichen planopilaris and thyroid disease, including hypothyroidism and autoimmune thyroiditis. A recent analysis has also shown a relationship with androgen excess, whereas a condition with similarities to lichen planopilaris, namely frontal fibrosing alopecia (see later), was associated with androgen deficiency.
Topical corticosteroid therapy (usually high-potency form) and intralesional steroids are the treatments of choice for patients with localized disease, particularly in the early phase. Other treatments have included hydroxychloroquine, tetracyclines, cyclosporine. oral retinoids, pioglitazone (a synthetic ligand of peroxisome-proliferator-activated receptor-λ), methotrexate, and naltrexone. Hair transplants and scalp reductions may be used in inactive end-stage disease.
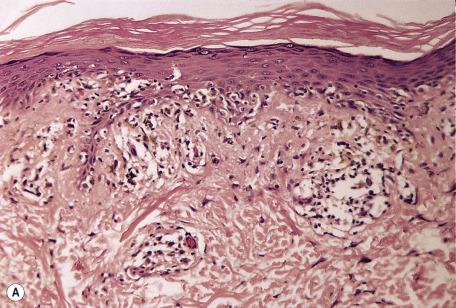
In lichen planopilaris, there is a lichenoid reaction pattern involving the basal layer of the follicular epithelium, with an associated dense perifollicular infiltrate of lymphocytes and a few macrophages ( Fig. 4.9 ). The changes involve the infundibulum and the isthmus of the follicle. One study reported that this is the so-called bulge region of the follicle where the stem cells reside. With regard to stem cells of the bulge region, all stem cell markers except for nestin are significantly reduced in lichen planopilaris; this is in contrast to alopecia areata, in which stem cell markers remain positive. It is the prototypical lymphocytic cicatricial alopecia. The mean ratio of Langerhans cells to lymphocytes is higher for infiltrates in lichen planopilaris than for the those in long-standing traction alopecia. Unlike lupus erythematosus, the infiltrate does not extend around blood vessels of the mid- and deep plexus. There is also some mucin in the perifollicular fibroplasia, unlike lupus erythematosus, in which it is predominantly in the interfollicular dermis. The interfollicular epidermis is involved in up to one-third of cases with scalp involvement and also in the rare plaque type (discussed previously). It is not usually involved in the variant with follicular papules on the trunk and extremities. It should be recognized that granulomas can be found in late stages of lichen planopilaris, associated with disruption and destruction of follicular infundibula. If scarring alopecia develops, there is variable perifollicular fibrosis and loss of hair follicles that are replaced by linear tracts of fibrosis. There is also loss of the arrector pili muscles and sebaceous glands. The papillary dermis may also be fibrosed. In advanced cases of scarring alopecia, the diagnostic features may no longer be present. The term pseudopelade is sometimes used to describe the end-stage scarring alopecia of lichen planopilaris.
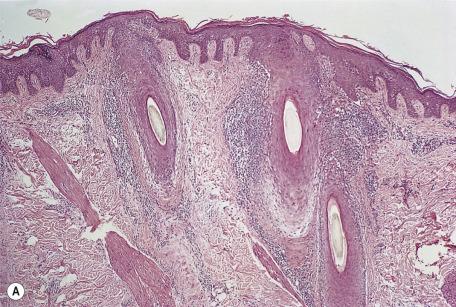
Immunostaining for cytokeratin 903 can be useful in the rapid diagnosis of lichen planopilaris, by allowing identification of the colloid bodies, even in the face of intense inflammation. Direct immunofluorescence shows colloid bodies containing IgG and IgM in the dermis adjacent to the upper portion of the involved follicles. In one report, linear deposits of immunoglobulins were found along the basement membrane of the hair follicles (of the scalp) in all cases. Fibrin was present in one case; cytoid bodies were not demonstrated. Note that the lesions were of long standing (3–7 years).
Dermoscopy can be helpful in the early diagnosis of lichen planopilaris, before obvious patches of alopecia have developed. Findings at this stage include peripilar tubular casts with miniaturized vellus hairs, or (using polarized dermoscopy with interface fluid) granular gray dots, white dots, and crystalline structures around follicles. In vivo reflectance confocal microscopy has been used to follow response to therapy; improvement was demonstrated by a reduced number of inflammatory cells in both the epidermis and the dermis.
Later stage lesions of lichen planopilaris with mainly perifollicular scarring, or those with milder or more loosely organized inflammatory infiltrates, can resemble other forms of scarring alopecia, including central/centrifugal scarring alopecia . However, the finding of dense, tightly packed inflammatory infiltrates around follicles, with vacuolar alteration of the basilar layer, strongly supports lichen planopilaris; a lichenoid tissue reaction involving interfollicular epidermis, when present, can further support the diagnosis. Lichen planopilaris and discoid lupus erythematosus can also show similar follicular changes, but only lupus erythematosus would be expected to show perieccrine infiltrates, dermal and particularly nonperifollicular mucin deposition, dense and deep dermal perivascular inflammation and frequent lipoatrophic changes, whereas lichen planopilaris is more apt to show a lack of interfollicular changes and a tightly packed, band-like lymphocytic infiltrate around the follicles. In addition, Kolivras and Thompson have found that dermal clusters of CD123 + plasmacytoid dendritic cells represent a sensitive and specific predictive finding for the diagnosis of lupus erythematosis in this scenario. Frontal fibrosing alopecia is a form of scarring alopecia that has been widely regarded as part of the lichen planus spectrum, though with clearly some unique characteristics (see Chapter 16 ). One recent study failed to find histological, immunohistochemical, or immunofluorescence findings that would allow distinction of this disease from lichen planopilaris, underscoring their close relationship. On the other hand, Wong and Goldberg have found that a significantly greater number of patients with frontal fibrosing alopecia have inflammation extending below the follicular isthmus compared with lichen planopilaris.
This rare disease is characterized by the coexistence of lichen planus and a heterogeneous group of subepidermal blistering diseases resembling bullous pemphigoid. There are tense bullae, often on the extremities, which may develop in normal or erythematous skin or in the lesions of lichen planus. In one case, the blisters were localized mainly to preexisting scars—an example of the isotopic phenomenon. They do not necessarily recur with subsequent exacerbations of the lichen planus. Oral lesions are exceedingly rare. Lichen planus pemphigoides has been reported in children. Rare clinical presentations include a unilateral distribution and onset after psoralen-UV-A (PUVA) therapy. Similar lesions have been induced by the anti–motion sickness drug cinnarizine and by the angtiotensin-converting enzyme (ACE) inhibitor ramipril. Other implicated agents include simvastatin, captopril, antituberculous medications, a weight reduction drug, venlafaxine, and pembrolizumab. Some cases have been reported in association with neoplasia, sharing this characteristic with paraneoplastic pemphigus.
Lichen planus pemphigoides is different from bullous lichen planus, in which vesicles or bullae develop only in the lichenoid papules, probably as a result of unusually severe basal damage and accompanying dermal edema.
The pathogenesis of lichen planus pemphigoides appears to be due to epitope spreading. It has been suggested that damage to the basal layer in lichen planus may expose or release a basement membrane zone antigen, which leads to the formation of circulating antibodies and consequent blister formation.
Partial support for this mechanism is provided by the case of a child with lichen planus who developed varicella, followed by lichen planus pemphigoides, and a report of bullous lichen planus accompanied by an elevation of serum anti-BP180. The target antigen, in fact, is a novel epitope (MCW-4) within the C-terminal NC16A domain of the 180-kDa bullous pemphigoid antigen (BP180, type XVII collagen). There is a case report of a patient with subacute cutaneous lupus erythematosus who had lichenoid skin lesions and bullae; the target antigen in this case was the same as that previously described for lichen planus pemphigoides .
Lichenoid erythrodermic bullous pemphigoid is a rare disease reported in African patients. It differs from lichen planus pemphigoides by the presence of a desquamative erythroderma and frequent mucosal lesions.
Lichen planus pemphigoides may be treated with topical corticosteroids, systemic steroids, tetracycline and nicotinamide combined, retinoids, dapsone, and cyclosporine. Systemic corticosteroids appear to be the most effective treatment for extensive disease.
A typical lesion of lichen planus pemphigoides consists of a subepidermal bulla that is cell poor, with only a mild, perivascular infiltrate of lymphocytes, neutrophils, and eosinophils. The presence of neutrophils and eosinophils has not been mentioned in all reports. Sometimes a lichenoid infiltrate is present at the margins of the blister, and there are occasional degenerate keratinocytes in the epidermis overlying the blister. Lesions that arise in papules of lichen planus show predominantly the features of lichen planus; a few eosinophils and neutrophils are usually present, in contrast to bullous lichen planus, in which they are absent. In one report, a pemphigus vulgaris–like pattern was present in the bullous areas.
Direct immunofluorescence of the bullae will usually show IgG, C3, and C9 neoantigen in the basement membrane zone, and there is often a circulating antibody to the basement membrane zone. Indirect split-skin immunofluorescence has shown binding to the roof of the split.
In lichen planus pemphigoides, the split occurs in the lamina lucida, as it does in bullous pemphigoid. Immunoelectron microscopy has shown that the localization of the immune deposits may resemble that seen in bullous pemphigoid, cicatricial pemphigoid, or epidermolysis bullosa acquisita—evidence of a heterogeneous disorder.
Keratosis lichenoides chronica is characterized by violaceous, papular, and nodular lesions in a linear and reticulate pattern on the extremities and a seborrheic dermatitis–like facial eruption. A rare vascular variant with telangiectasias has been reported. Oral ulceration and nail involvement may occur.
It is a rare condition, particularly in children. It has been suggested that pediatric-onset disease is different from adult-onset keratosis lichenoides chronica. Childhood cases may have familial occurrence and probably autosomal recessive inheritance. Early or congenital onset with facial erythematopurpuric macules is sometimes seen. Forehead, eyebrow, and eyelash alopecia are usually present, especially in children or cases that develop in childhood. A case mimicking verrucous secondary syphilis has been reported. The condition is possibly an unusual chronic variant of lichen planus, although this concept has been challenged. Böer believes that there is an authentic and distinctive condition that should continue to be called keratosis lichenoides chronica, but that many of the purported cases are lichen planus, lupus erythematosus, or lichen simplex chronicus.
Keratosis lichenoides chronica may be associated with internal diseases such as glomerulonephritis, hypothyroidism, and lymphoproliferative disorders. In one patient with multiple myeloma, there were eruptive keratoacanthoma–like lesions.
The disease is refractory to many different treatment modalities, although calcipotriol or tacalcitol alone, or in combination with oral retinoids, may give good results. Another case responded to isotretinoin and methotrexate. Oral retinoids alone are sometimes effective. Phototherapy has also been used.
There is a lichenoid reaction pattern with prominent basal cell death and focal basal vacuolar change. The inflammatory infiltrate usually includes a few plasma cells, and sometimes there is deeper perivascular and periappendageal cuffing. Telangiectasia of superficial dermal vessels is sometimes noted. Epidermal changes are variable, with alternating areas of atrophy and acanthosis sometimes present, as well as focal parakeratosis. The parakeratosis often has a staggered appearance with neutrophil remnants. Cornoid lamellae and amyloid deposits in the papillary dermis have been recorded. Numerous IgM-containing colloid bodies are usually found on direct immunofluorescence.
The term lichen planoporitis was used for a case with the clinical features of keratosis lichenoides chronica and histological changes that included a lichenoid reaction centered on the acrosyringium and upper eccrine duct with focal squamous metaplasia of the upper duct and overlying hypergranulosis and keratin plugs. Ruben and LeBoit have also reported eccrine duct involvement in a case of keratosis lichenoides chronica. Böer states that the lichenoid infiltrate in keratosis lichenoides chronica is commonly centered around infundibula and acrosyringia.
Because of the infrequency of keratosis lichenoides chronica, it can potentially be confused with other lichenoid dermatoses. However, it does not generally show the classic features of lichen planus , and the parakeratosis, the variable atrophy and acanthosis seen in some lesions, and the deeper dermal perivascular and periadnexal infiltrates are reasonably distinctive. See later for the distinction from lichen striatus.
Lupus erythematosus–lichen planus overlap syndrome is a heterogeneous entity in which one or more of the clinical, histological, and immunopathological features of both diseases are present. Some cases may represent the coexistence of lichen planus and lupus erythematosus, whereas in others the ultimate diagnosis may depend on the course of the disease. In most cases, the lupus erythematosus is of the chronic discoid or systemic type; rarely, it is of the subacute type. It was the cause of a scarring alopecia in one case. Another case was associated with HIV infection. One recent case report described successful long-term therapy with thalidomide. Before the diagnosis of an overlap syndrome is entertained, it should be remembered that some lesions of cutaneous lupus erythematosus may have numerous Civatte bodies and a rather superficial inflammatory cell infiltrate that at first glance may be mistaken for lichen planus. The use of an immunofluorescent technique using a patient's serum and autologous lesional skin as a substrate may assist in the future in elucidating the correct diagnosis in some of these cases.
Lichen nitidus is a rare, usually asymptomatic chronic eruption characterized by the presence of multiple, small flesh-colored papules, 1 or 2 mm in diameter. The lesions have a predilection for the upper extremities, chest, abdomen, and genitalia of children and young adult males. Unusual locations have included the dorsal tongue and the eyelid. The disorder is most often localized, but sometimes lesions are more generalized. Linear lesions following the lines of Blaschko are considered variants of a BLAISE lesion ( b laschko- l inear a cquired i nflammatory s kin e ruption). Familial cases are rare. Lichen nitidus has been reported in association with Down syndrome and the Russell–Silver syndrome. Nail changes occur, and include longitudinal splitting of the nail plate, violaceous or pigmentary changes (including hyperpigmentation and swelling of the nail fold), and subtle lichenoid papules on the digits. Involvement of the palms and soles has been reported, as have lesions confined to the palm. It has been suggested that cases reported in the past as SALE should be reclassified as actinic lichen nitidus. It has been reported in association with lichen spinulosus.
Although regarded originally as a variant of lichen planus, lichen nitidus is now considered a distinct entity of unknown etiology. It has followed hepatitis B vaccination. The lymphocytes in the dermal infiltrate in lichen nitidus express different markers from those in lichen planus. Lichen planus has developed subsequent to generalized lichen nitidus in a child.
Although spontaneous remissions of lichen nitidus are common, persistent lesions and those that are refractory to various treatments can pose therapeutic challenges. Sometimes resolution is accompanied by postinflammatory hyperpigmentation. Some of the treatments used include systemic and topical corticosteroids, antihistamines, retinoids, low-dose cyclosporine, itraconazole, isoniazid, and ultraviolet therapy. Generalized lichen nitidus has been successfully treated with narrowband UV-B phototherapy.
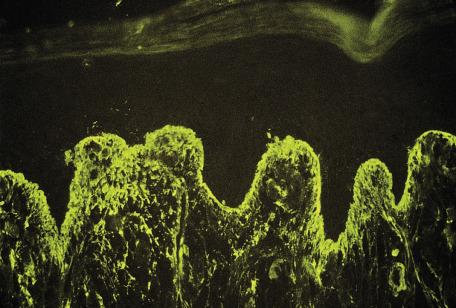
A papule of lichen nitidus shows a dense, well-circumscribed, subepidermal infiltrate, sharply limited to one or two adjacent dermal papillae. Claw-like, acanthotic rete ridges, which appear to grasp the infiltrate, are present at the periphery of the papule ( Fig. 4.10 ). The inflammatory cells push against the undersurface of the epidermis, which may be thinned and show overlying parakeratosis. Occasional Civatte bodies are present in the basal layer.
In addition to lymphocytes, histiocytes, and melanophages, there are also epithelioid cells and occasional multinucleate giant cells in the inflammatory infiltrate. Rarely, plasma cells are conspicuous. A spinous follicular variant of lichen nitidus, first described by Madhok and Winkelmann, shows perifollicular granulomas. The infiltrates of lichen nitidus include both CD68 + macrophages and S100 + , CD1a + Langerhans cells. Rare changes that have been reported include subepidermal vesiculation, transepidermal elimination of the inflammatory infiltrate, and the presence of perifollicular granulomas. Periappendageal inflammation mimicking lichen striatus has also been reported.
Direct immunofluorescence is usually negative, a distinguishing feature from lichen planus.
Dermoscopic changes in palmar lichen nitidus include parallel linear scales interrupted by oval depressions parallel to the line of scales, with fewer and finer scales on the surface of the depressed areas; in contrast, the changes in other locations, such as the arm, include round, elevated, shiny, smooth structures surrounded by radial rete ridges and a reddish vascular network. In perforating lichen nitidus, dermoscopic examination shows grouped monomorphic lesions with central, light brown keratin plugs surrounded by whitish annular, cloud-like areas.
The ultrastructural changes in lichen nitidus are similar to those of lichen planus.
Lichen nitidus can resemble the smaller papular lesions of lichen planus that only involve several dermal papillae. Occasionally, one encounters other lesions, such as arthropod reactions, that are characterized by discrete, superficial dermal infiltrates, but in most instances these lesions either lack the classic features of lichen nitidus (vacuolar alteration of the overlying basilar layer, confinement to one or two adjacent dermal papillae, and “ball in claw” configuration) or show more widely distributed inflammation with different cellular components. It should also be recognized that the diagnostic changes can easily be absent in a given tissue section; therefore, serial sections may then be necessary to find these tiny lesions. The appearances are sometimes frankly granulomatous, and these lesions must therefore be distinguished from disseminated granuloma annulare in which the infiltrate may be superficial and the necrobiosis sometimes quite subtle. Uncommon examples with transepidermal elimination might also be confused with perforating granuloma annulare or possibly with reactive perforating collagenosis, although the latter lacks the characteristic inflammatory changes. Lichen nitidus also needs to be distinguished from an early lesion of the tuberculid, lichen scrofulosorum . Whereas the infiltrate in lichen nitidus “hugs” the epidermis and expands the dermal papilla, the granulomas in lichen scrofulosorum do not cause widening of the papillae. Furthermore, in lichen scrofulosorum there may be mild spongiosis and exocytosis of neutrophils into the epidermis. Langerhans cell histiocytosis can bear a close clinical resemblance to lichen nitidus, but the biopsy findings are distinctive.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here