Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Evaluation of amniotic fluid volume (subjective assessment combined with semiquantitative methods such as maximum vertical pocket or amniotic fluid index) provides important information about some maternal and fetal conditions, as well as placental function.
Sonographic findings suspicious of renal cystic disease include cysts, renal hyperechogenicity, and/or large kidneys. Whenever renal cystic disease is suspected, it is useful to perform a renal ultrasound of the parents to check for unknown familial disease.
Evaluation of urinary tract dilation should include measurement of the anteroposterior renal pelvic diameter; identification of calyceal dilation and renal parenchymal, ureteral, or bladder abnormalities; and assessment of amniotic fluid volume.
In fetuses with lower urinary tract obstruction, a fetal urinary function profile (combining sonographic and biochemical predictors) is most useful clinically in selecting the fetus that will benefit from in utero intervention.
Examination of fetal genitalia plays a key role in the diagnostic workup of many structural abnormalities and genetic disorders, as well as in the assessment of multiple pregnancies.
Evaluation of the fetal urogenital tract is an integral part of the obstetric ultrasound examination. Sonography depicts normal developmental anatomy and allows detection and characterization of many genitourinary abnormalities. In addition, assessment of the amniotic fluid volume (AFV) often provides important prognostic information regarding fetal renal function. Accurate and early prenatal diagnosis is important because this may influence obstetric and neonatal management.
Congenital anomalies of the kidney and urinary tract (CAKUT) account for approximately 30% of all malformations detected on routine prenatal sonography. A systematic sonographic approach is proposed, which includes a search for associated anomalies and detailed evaluation of renal structure and function.
The permanent kidney (metanephros) is the third in a series of excretory organs in the human embryo, forming after the pronephros and mesonephros. In the seventh menstrual week, the metanephros begins to develop from two sources: the metanephric diverticulum (ureteric bud) and the metanephric mass of intermediate mesoderm ( Fig. 39.1 ). The ureteric bud is an outgrowth from the mesonephric duct, near its entrance into the cloaca. It elongates and branches in a dichotomous pattern, giving rise to the ureter, renal pelvis, calyces, and collecting tubules. Through interaction with the metanephric mesoderm, the ureteric bud induces the formation of nephrons. In early embryonic life, the kidneys are located in the pelvis, but they “ascend” to their adult position by the 11th menstrual week. At this gestation, the kidneys start to produce urine.
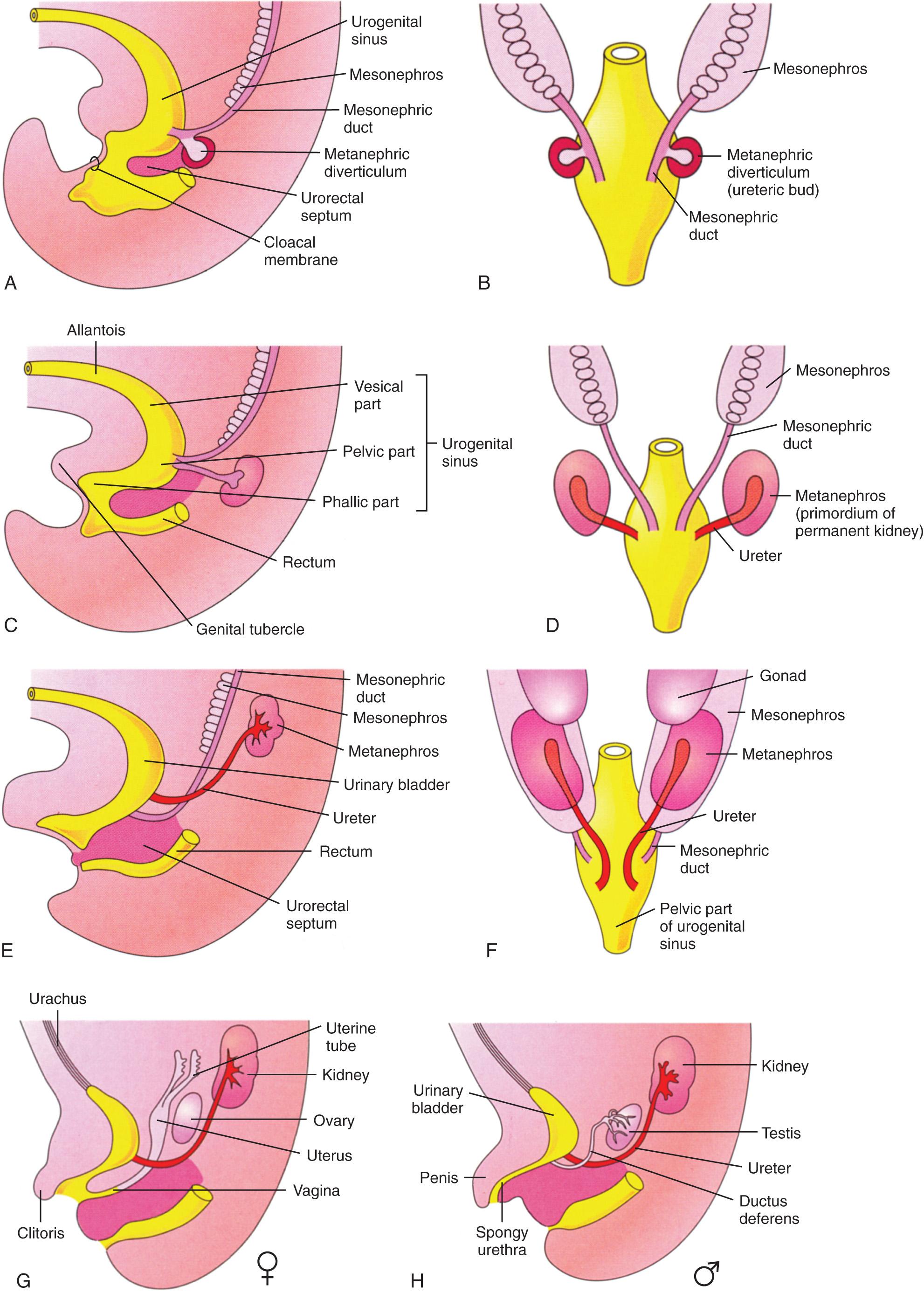
By the ninth menstrual week, the cloaca (caudal part of hindgut) is divided by the urorectal septum into the rectum posteriorly and the urogenital sinus anteriorly (see Fig. 39.1 ). The urinary bladder, the female urethra, and most of the male urethra develop from the urogenital sinus and the surrounding splanchnic mesenchyme. Initially, the bladder is continuous with the allantois, but this structure soon constricts and becomes a fibrous cord, the urachus, which extends from the apex of the bladder to the umbilicus.
In the first trimester the fetal kidneys are best examined by transvaginal sonography. The kidneys are seen as oval, hyperechoic structures in the paravertebral regions, with a small, central sonolucent area caused by fluid in the renal pelvis ( Fig. 39.2A ). By 12 to 13 weeks of gestation, the kidneys can be visualized in 99% of cases with combined transabdominal and transvaginal sonography. In the second trimester the kidneys often appear as isoechoic structures adjacent to the fetal spine on transabdominal sonography ( Fig. 39.2B ). As the fetus matures, corticomedullary differentiation becomes more obvious, especially in the third trimester ( Figs. 39.2C-D ). The renal pyramids orient in anterior and posterior rows and are hypoechoic relative to the renal cortex. In the third trimester the renal cortex is isoechoic or slightly hyperechoic to liver and spleen. With fat deposition in the perinephric region, an echogenic border develops, and the kidney becomes better delineated. Normal fetal lobations are often visible and give the kidneys an undulating contour.
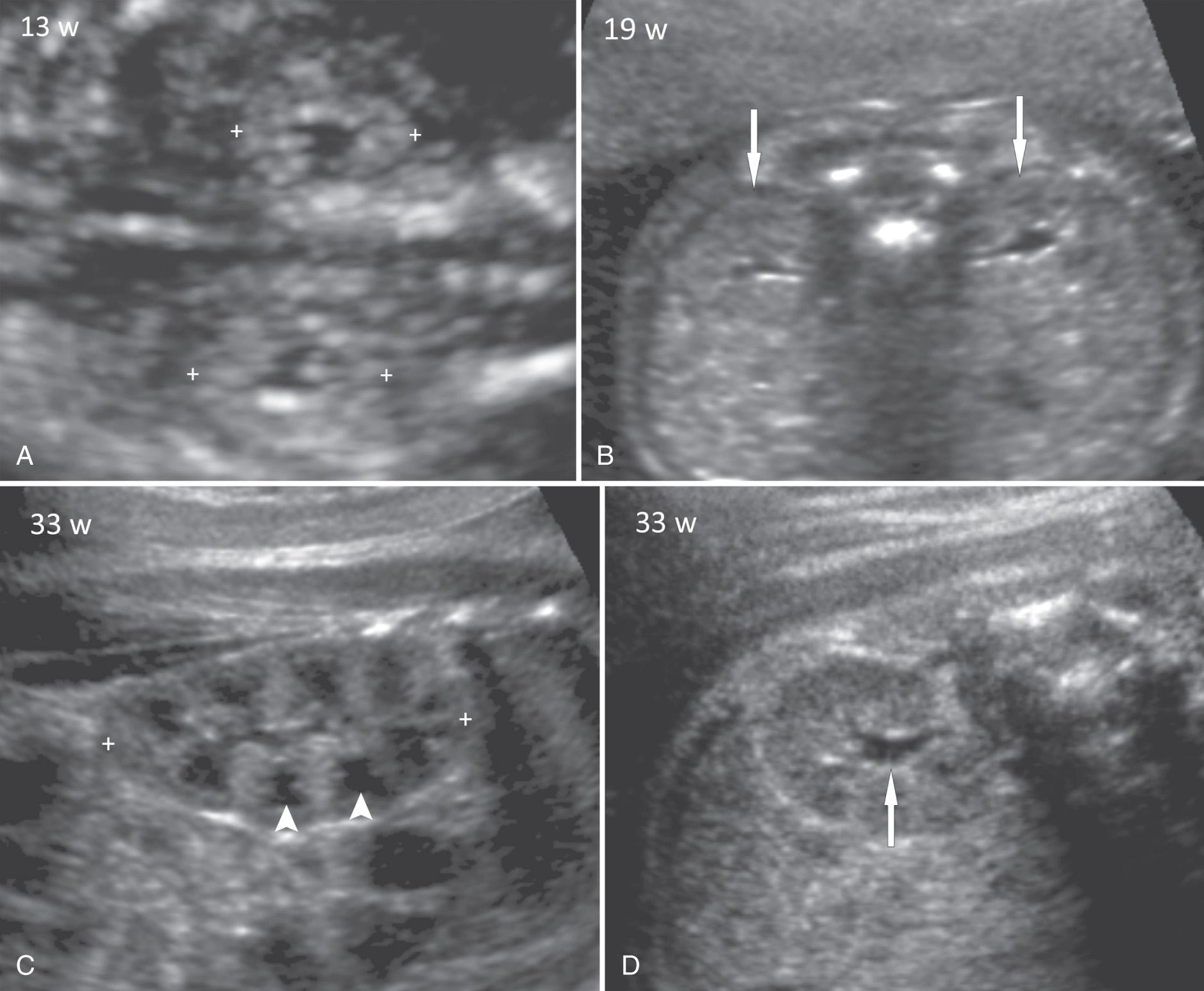
The kidneys grow throughout pregnancy. Table 39.1 provides a nomogram of renal lengths at 14 to 42 weeks of gestation. The often-quoted rule of thumb that “renal length in millimeters approximates gestational age in weeks” applies only to a narrow gestational age range of 18 to 21 weeks. There are also published charts of renal anteroposterior diameter, transverse diameter, and volume. Sometimes, it is difficult to define the exact renal borders, especially at the upper pole, because of shadowing from ribs or poor distinction from the adrenal gland. Fetal breathing can aid in renal visualization. It is also important to avoid using an oblique section through the kidney for measurement. The renal/abdominal circumference ratio remains constant at 0.27 to 0.30 throughout pregnancy.
| FITTED CENTILES | ||||||
|---|---|---|---|---|---|---|
| Week | N | 3rd | 10th | 50th | 90th | 97th |
| 14 | 3 | 7.5 | 8.0 | 9.3 | 10.8 | 11.6 |
| 15 | 3 | 8.8 | 9.5 | 11.0 | 12.8 | 13.7 |
| 16 | 2 | 10.2 | 11.0 | 12.7 | 14.8 | 15.8 |
| 17 | 12 | 11.6 | 12.5 | 14.5 | 16.8 | 18.1 |
| 18 | 10 | 13.1 | 14.1 | 16.3 | 18.9 | 20.3 |
| 19 | 15 | 14.6 | 15.6 | 18.2 | 21.1 | 22.6 |
| 20 | 15 | 16.1 | 17.2 | 20.0 | 23.2 | 24.9 |
| 21 | 15 | 17.5 | 18.8 | 21.8 | 25.4 | 27.2 |
| 22 | 14 | 19.0 | 20.4 | 23.6 | 27.4 | 29.4 |
| 23 | 16 | 20.4 | 21.9 | 25.4 | 29.5 | 31.6 |
| 24 | 17 | 21.8 | 23.4 | 27.1 | 31.5 | 33.8 |
| 25 | 18 | 23.1 | 24.8 | 28.8 | 33.4 | 35.8 |
| 26 | 20 | 24.4 | 26.2 | 30.4 | 35.3 | 37.8 |
| 27 | 24 | 25.6 | 27.5 | 31.9 | 37.1 | 39.7 |
| 28 | 18 | 26.8 | 28.7 | 33.4 | 38.7 | 41.5 |
| 29 | 19 | 27.9 | 29.9 | 34.7 | 40.3 | 43.2 |
| 30 | 19 | 28.9 | 31.0 | 36.0 | 41.8 | 44.8 |
| 31 | 23 | 29.9 | 32.1 | 37.2 | 43.2 | 46.3 |
| 32 | 23 | 30.8 | 33.0 | 38.3 | 44.5 | 47.7 |
| 33 | 22 | 31.6 | 33.9 | 39.4 | 45.7 | 49.0 |
| 34 | 19 | 32.4 | 34.7 | 40.3 | 46.8 | 50.2 |
| 35 | 20 | 33.1 | 35.4 | 41.1 | 47.8 | 51.2 |
| 36 | 23 | 33.7 | 36.1 | 41.9 | 48.7 | 52.2 |
| 37 | 14 | 34.2 | 36.7 | 42.6 | 49.4 | 53.0 |
| 38 | 17 | 34.7 | 37.2 | 43.2 | 50.1 | 53.8 |
| 39 | 13 | 35.1 | 37.6 | 43.7 | 50.7 | 54.4 |
| 40 | 14 | 35.4 | 38.0 | 44.1 | 51.2 | 54.9 |
| 41 | 26 | 35.7 | 38.3 | 44.5 | 51.6 | 55.4 |
| 42 | 17 | 36.0 | 38.6 | 44.8 | 52.0 | 55.7 |
The calyces are not normally visualized, but some fluid is typically seen in the renal pelvis. The highly characteristic renal pelvic echo is often the key to finding the kidneys in the second trimester. Measurements of the renal pelvis are discussed in the section on upper urinary tract (UT) dilation. The normal ureter is 1 to 2 mm in diameter and is not normally visible.
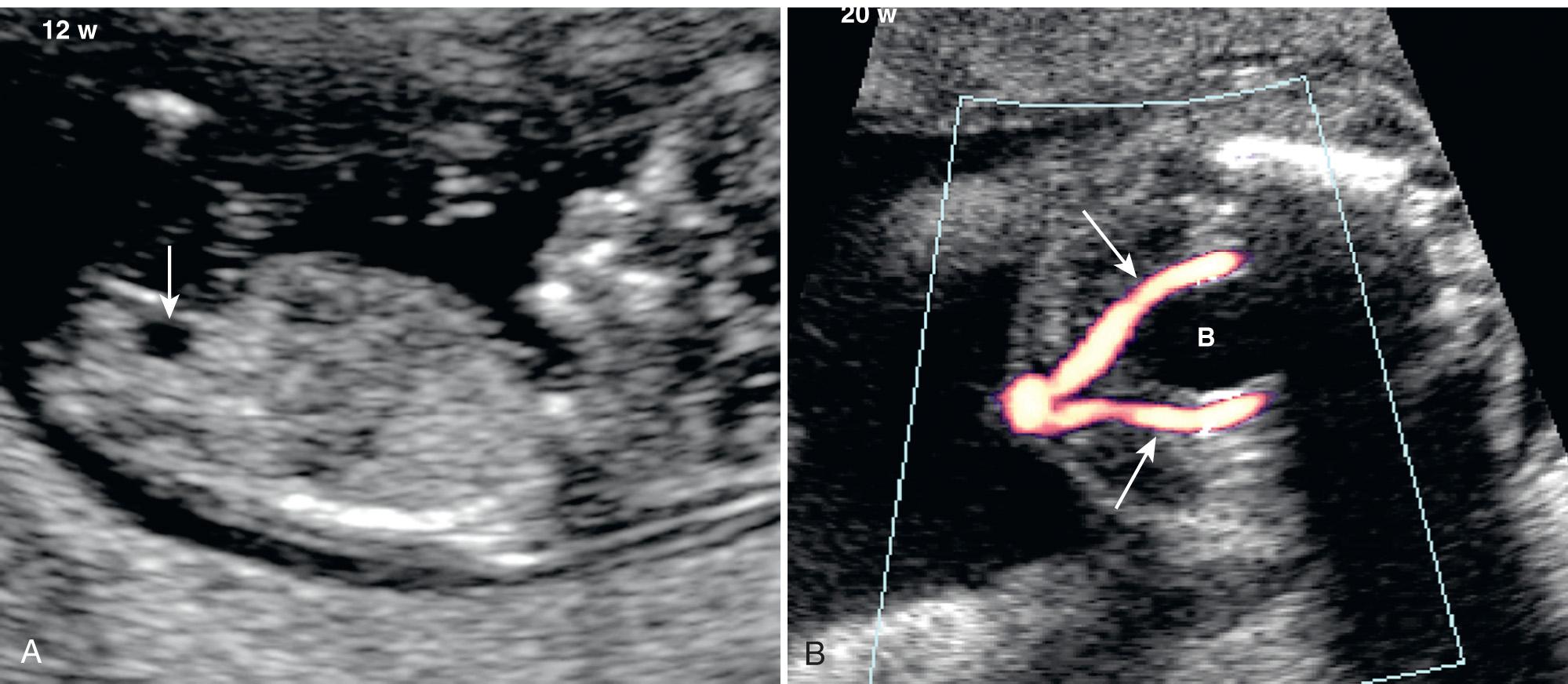
By using transvaginal sonography, the bladder can be seen as early as 11 weeks of gestation. By 12 to 13 weeks, the bladder is visualized in 98% of cases using both transabdominal and transvaginal sonography. The bladder is thin walled and situated anteriorly in the pelvis. The umbilical (superior vesical) arteries run lateral to the bladder as they course toward the umbilicus ( Fig. 39.3 ). The hourly fetal urine production increases with advancing gestation, from a mean value of 4 to 5 mL/hr at 20 weeks to 52 to 56 mL/hr at 40 weeks. Three-dimensional (3-D) ultrasound measurements demonstrate reproducible urine production rates based on bladder volumes, but tend to estimate higher rates in the third trimester compared with the standard two-dimensional (2-D) technique. The maximum bladder volume increases from a mean value of 1 mL at 20 weeks to 36 mL at 41 weeks. The normal bladder fills and empties (either partially or completely) approximately every 25 minutes (range, 7-43 min). Therefore changes in bladder volume should be observed during the course of the obstetric sonogram.
Evaluation of amniotic fluid volume (AFV) provides important information about some maternal and fetal conditions, as well as placental function. Evaluation of AFV is a key component of fetal biophysical assessment. After 16 weeks, fetal urine production becomes the major source of amniotic fluid. Fetal lung secretions, fetal swallowing, and intramembranous resorption of water into the fetal circulation via fetal vessels on the placental surface also contribute to AFV homeostasis.
Several methods are used to assess AFV. Subjective assessment can be combined with semiquantitative techniques, such as measurement of the maximum vertical pocket (or single deepest vertical pocket ) and amniotic fluid index (AFI). Significant oligohydramnios results in compression of the fetus, marked crowding of fetal parts, and poor visualization of fetal anatomy. With polyhydramnios, one gets the impression of a “swimming” fetus. The subjective assessment of AFV by experienced sonographers has been shown to be reliable. However, there are conflicting data regarding the reliability of both the maximum vertical pocket technique and the AFI. The technique for measuring the deepest amniotic fluid pockets should follow these guidelines:
The maximum depth of the fluid pocket should be measured in a perpendicular direction.
The pocket should have a width of at least 1 cm (avoid measuring narrow, slitlike pockets).
The pocket should be free of fetal small parts and umbilical cord.
The following classification has been proposed for the maximum vertical pocket method: vertical depth of the pocket less than 2 cm indicates moderate to severe oligohydramnios, 2 to 8 cm is normal, and greater than 8 cm indicates polyhydramnios.
a Maximum vertical pocket method.
The AFI is obtained by measuring the deepest amniotic fluid pocket in each of the four quadrants of the uterus, and the sum of the four measurements is the index. AFI varies with gestational age ( Table 39.2 ). In the late second trimester and in the third trimester, an AFI of 5 cm to 24 cm has been considered as normal. It is recommended that when AFI is less than 10 cm, three measurements should be averaged. The semiquantitative methods are useful for following AFV on serial examinations, particularly by multiple examiners of varying experience.
| Gestational Age (wk) | AMNIOTIC FLUID INDEX (cm) | ||||||
|---|---|---|---|---|---|---|---|
| No. | 5th Percentile | 10th Percentile | 50th Percentile | Mean | 90th Percentile | 95th Percentile | |
| 14 | 50 | 2.8 | 3.1 | 5 | 5.4 | 8 | 8.6 |
| 15 | 50 | 3.2 | 3.6 | 5.4 | 5.7 | 8.2 | 9.1 |
| 16 | 50 | 3.6 | 4.1 | 5.8 | 6.1 | 8.5 | 9.6 |
| 17 | 50 | 4.1 | 4 | 6.3 | 6.6 | 9 | 10.3 |
| 18 | 50 | 4.6 | 5.1 | 6.8 | 7.1 | 9.7 | 11.1 |
| 19 | 50 | 5.1 | 5.6 | 7.4 | 7.7 | 10.4 | 12 |
| 20 | 50 | 5.5 | 6.1 | 8 | 8.3 | 11.3 | 12.9 |
| 21 | 50 | 5.9 | 6.6 | 8.7 | 8.9 | 12.2 | 13.9 |
| 22 | 50 | 6.3 | 7.1 | 9.3 | 9.6 | 13.2 | 14.9 |
| 23 | 50 | 6.7 | 7.5 | 10 | 10.3 | 14.2 | 15.9 |
| 24 | 50 | 7 | 7.9 | 10.7 | 11 | 15.2 | 16.9 |
| 25 | 50 | 7.3 | 8.2 | 11.4 | 11.7 | 16.1 | 17.8 |
| 26 | 50 | 7.5 | 8.4 | 12 | 12.3 | 17 | 18.7 |
| 27 | 50 | 7.6 | 8.6 | 12.6 | 12.8 | 17.8 | 19.4 |
| 28 | 50 | 7.6 | 8.6 | 13 | 13.3 | 18.4 | 19.9 |
| 29 | 50 | 7.6 | 8.6 | 13.4 | 13.6 | 18.8 | 20.4 |
| 30 | 50 | 7.5 | 8.5 | 13.6 | 13.8 | 18.9 | 20.6 |
| 31 | 50 | 7.3 | 8.4 | 13.6 | 13.8 | 18.9 | 20.6 |
| 32 | 50 | 7.1 | 8.1 | 13.6 | 13.7 | 18.7 | 20.4 |
| 33 | 50 | 6.8 | 7.8 | 13.3 | 13.4 | 18.2 | 20 |
| 34 | 50 | 6.4 | 7.4 | 12.9 | 13 | 17.7 | 19.4 |
| 35 | 50 | 6 | 7 | 12.4 | 12.5 | 16.9 | 18.7 |
| 36 | 50 | 5.6 | 6.5 | 11.8 | 11.8 | 16.2 | 17.9 |
| 37 | 50 | 5.1 | 6 | 11.1 | 11.1 | 15.3 | 16.9 |
| 38 | 50 | 4.7 | 5.5 | 10.3 | 10.3 | 14.4 | 15.9 |
| 39 | 50 | 4.2 | 5 | 9.4 | 9.4 | 13.7 | 14.9 |
| 40 | 50 | 3.7 | 4.5 | 8.6 | 8.6 | 12.9 | 13.9 |
| 41 | 50 | 3.3 | 4 | 7.8 | 7.7 | 12.3 | 12.9 |
Oligohydramnios is typically defined as either a single maximum vertical pocket of amniotic fluid of less than 2 cm or alternatively, an AFI of less than 5 cm. Recently, a meta-analysis of four randomized controlled trials comparing the use of AFI (<5 cm) versus the single deepest vertical pocket (<2 cm) during antepartum fetal surveillance did not show any evidence that one method is superior to the other in predicting poor perinatal outcomes. However, the use of AFI increased the rate of diagnosis of oligohydramnios and subsequent induction of labor. It was concluded that the single deepest vertical pocket measurement is the method of choice for amniotic fluid assessment during fetal surveillance. This approach is supported by the American College of Obstetricians and Gynecologists (ACOG), as well as the findings of the recently completed SAFE trial.
In multiple pregnancies, the maximum vertical pocket technique is commonly used for assessment of amniotic fluid, with similar cutoffs for abnormal as for singletons. A systematic review of studies on amniotic fluid assessment and adverse outcomes in twin pregnancies has cautioned that sonographic evaluation of AFV has high specificity but poor sensitivity for the detection of abnormal AFV, and more rigorous studies in this area are required.
The prevalence of UT malformations varies among studies, likely because of differences in study population and methods of surveillance. In a recent analysis of 709,030 births in 12 European countries, the prevalence of congenital malformations of the UT was 1.6 per 1000 births. The overall prenatal detection rate was high: 82% and 88.5% in two studies. However, it varied from 36% to 100% in different centers. Many factors could account for the variation of prenatal detection rates, including the study population (high risk vs. unselected), timing of the ultrasound scan, expertise of the operator, quality of the ultrasound equipment, extent of follow-up, and ascertainment of congenital anomalies. For major UT anomalies, 57% were detected before 24 weeks. Lethal UT anomalies account for 10% of pregnancy terminations.
Assessment of amniotic fluid volume
Localization and characterization of urinary tract abnormalities
Search for associated abnormalities
A systematic approach to the prenatal diagnosis of UT abnormalities includes assessment of AFV, localization and characterization of UT abnormalities, assessment of fetal gender, and search for associated abnormalities.
Normal AFV in the second half of pregnancy implies at least one functioning kidney and a patent urinary conduit to the amniotic cavity. If oligohydramnios is present without a history of ruptured membranes, maternal drug intake (e.g., angiotensin-converting enzyme [ACE] inhibitors, angiotensin II receptor antagonists, cyclooxygenase-2 [COX-2] selective and nonselective inhibitors, nonsteroidal antiinflammatory drugs, cocaine ), or evidence of intrauterine growth restriction (IUGR), UT anomalies must be strongly suspected. In the setting of a UT abnormality, normal AFV indicates a good prognosis. Oligohydramnios in the early second trimester carries a very poor prognosis because of the associated pulmonary hypoplasia. Occasionally and paradoxically, polyhydramnios may occur, especially with unilateral obstructive uropathy, with mesoblastic nephroma, or when there are concomitant abnormalities of the central nervous system (CNS) or gastrointestinal (GI) tract.
The following questions are helpful in defining and characterizing the UT abnormality:
Is the bladder identified and normal in appearance?
Are kidneys present? Are they normal in position, size, and echogenicity? Are renal cysts identified?
Is the UT dilated? If so, to what degree, at which level, and what is the cause?
Is the involvement unilateral or bilateral, symmetrical or asymmetrical?
What is the fetal gender?
It is important to perform a detailed anatomic scan to search for associated abnormalities, which may indicate the presence of a syndrome or chromosomal abnormality. Renal anomalies may be part of the VATER association (vertebral defects, anal atresia, tracheoesophageal fistula, radial defects, and renal anomalies). An expansion of this syndrome, VACTERL, includes cardiac and nonradial limb defects. Associated nonurinary anomalies have been reported in 34% of infants with CAKUT, most common in the musculoskeletal, digestive, cardiovascular, and central nervous systems. When there are additional malformations, the risk for fetal chromosomal abnormalities is substantially increased compared with the maternal age–related risk: 30 times higher for multiple defects versus 3 times higher for isolated renal defect.
In addition, renal ultrasound is recommended for parents (and siblings) of fetuses suspected to have certain renal abnormalities (polycystic kidney disease, renal agenesis or adysplasia), because it may help to diagnose the type of polycystic kidney disease in the fetus, detect asymptomatic renal pathology in parents (and siblings), and counsel parents regarding the recurrence risk.
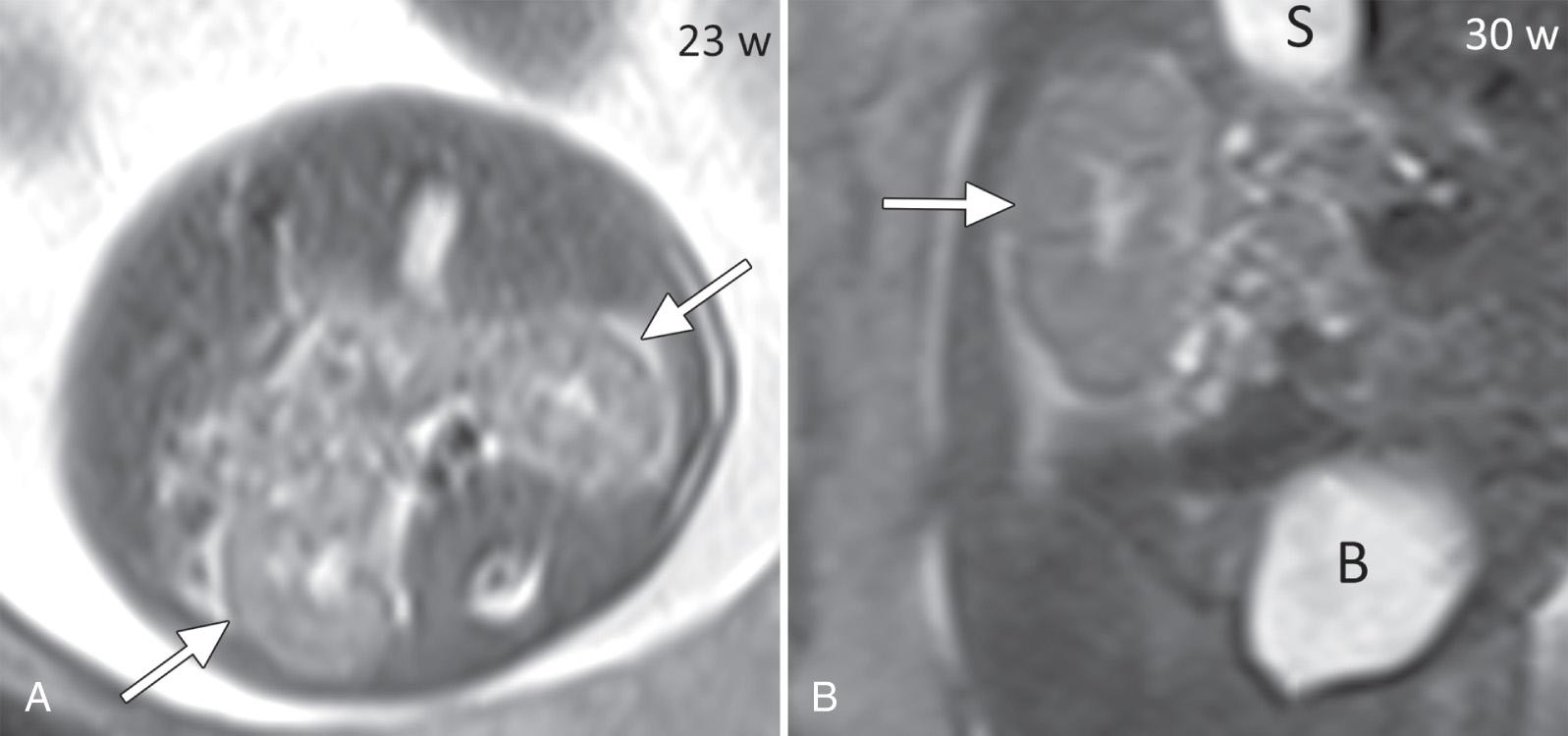
Fetal magnetic resonance imaging (MRI) may be used as an adjunctive imaging modality for improving diagnostic accuracy of UT abnormalities. It may help to identify the kidneys when sonographic visualization is limited by anhydramnios (or severe oligohydramnios) and large maternal body habitus ( Fig. 39.4 ). Finally, with the advent of diffusion-weighted imaging, the measurement of water apparent diffusion coefficient has shown promise in differentiating between normal and abnormal renal parenchyma, which could serve as a noninvasive method of assessing fetal renal function.
Bilateral renal agenesis is a lethal congenital anomaly with an incidence of approximately 1 in 4000 births and a 2.5 : 1 male preponderance. The ureteric buds fail to develop; thus the kidneys are absent. Because no urine is produced, severe oligohydramnios or anhydramnios results. Pulmonary hypoplasia is the major cause of neonatal death. Other features of “Potter's sequence” include typical facies (beaked nose, low-set ears, prominent epicanthic folds, hypertelorism), limb deformities, and IUGR.
The ultrasound findings include severe oligohydramnios or anhydramnios and nonvisualization of the kidneys and bladder. Before 16 weeks' gestation, AFV is not dependent on urine production and may be normal despite absent renal function. The absence of fetal kidneys should be the most specific finding, but this may be difficult to document because of poor image quality associated with oligohydramnios. In addition, bowel or adrenal glands in the renal fossae may be mistaken for kidneys. However, recognition of the distinctive, flattened appearance of the adrenal gland on longitudinal sonogram (“lying down” adrenal sign) helps to confirm that the kidney did not develop in the flank ( Fig. 39.5 , ).
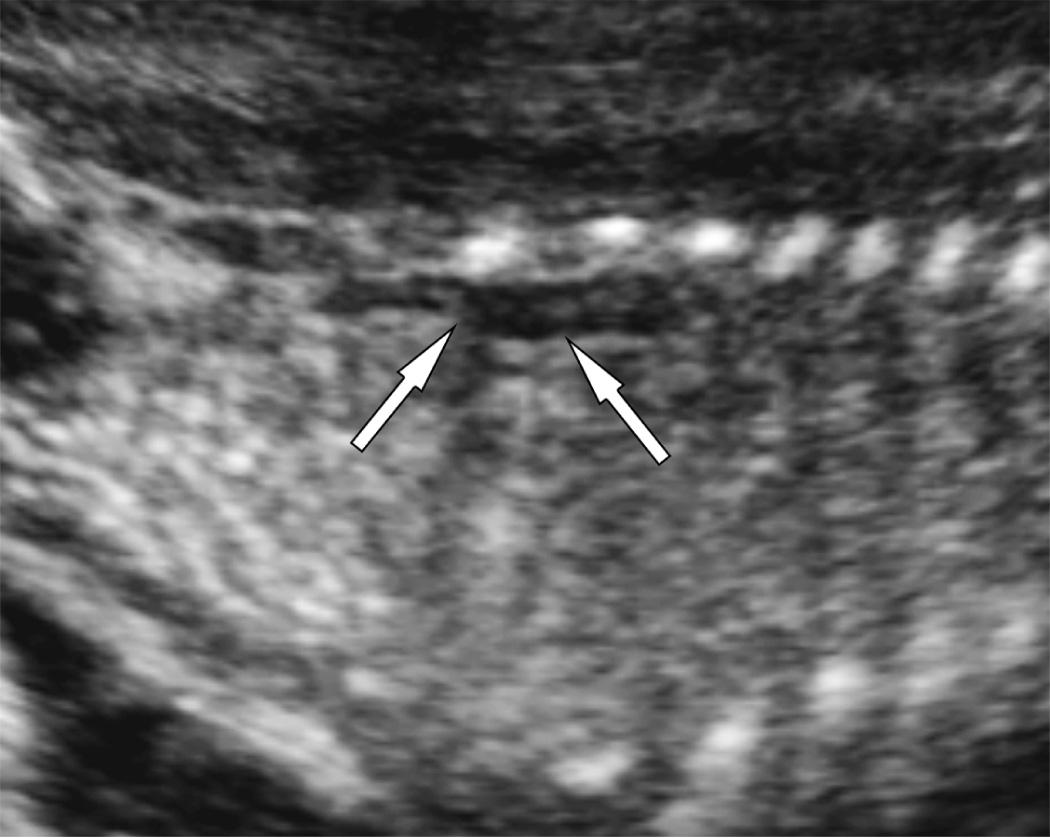
Repeated and consistent nonvisualization of the urinary bladder (over 1 hour) is a secondary sign of bilateral renal agenesis ( Fig. 39.6 ). Conversely, identification of a normal bladder excludes this diagnosis. A small urachal diverticulum may mimic the bladder, but its lack of filling and emptying distinguishes it from the bladder. A major challenge is to reliably distinguish between fetuses with bilateral renal agenesis and those with impaired renal function from severe uteroplacental insufficiency or IUGR.
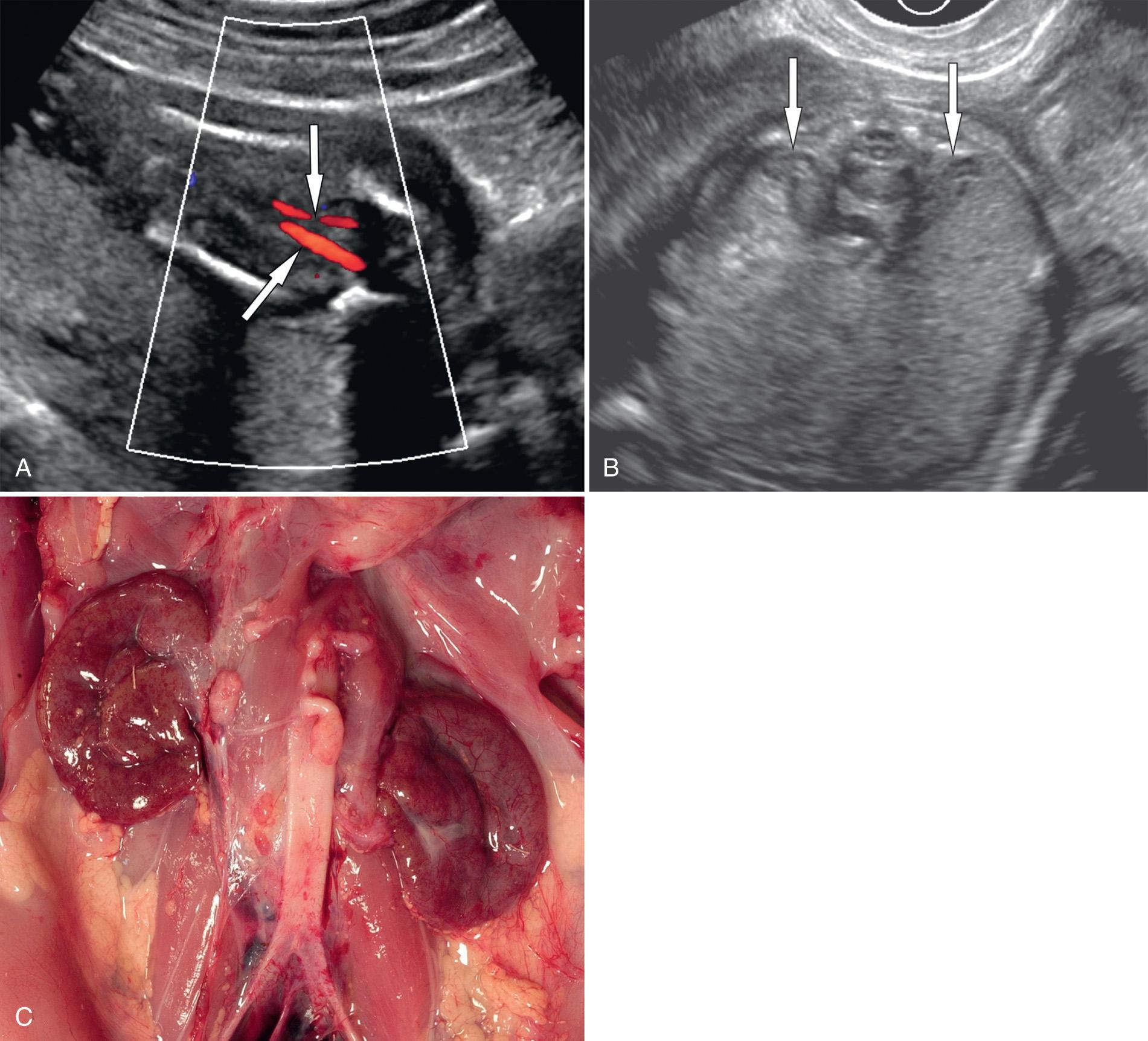
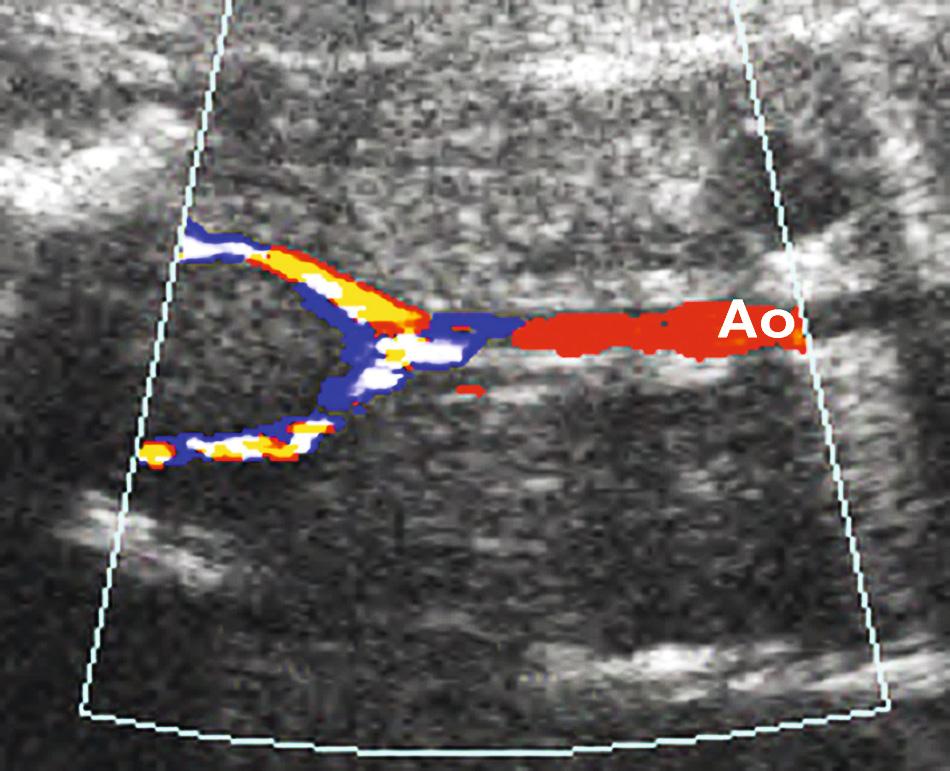
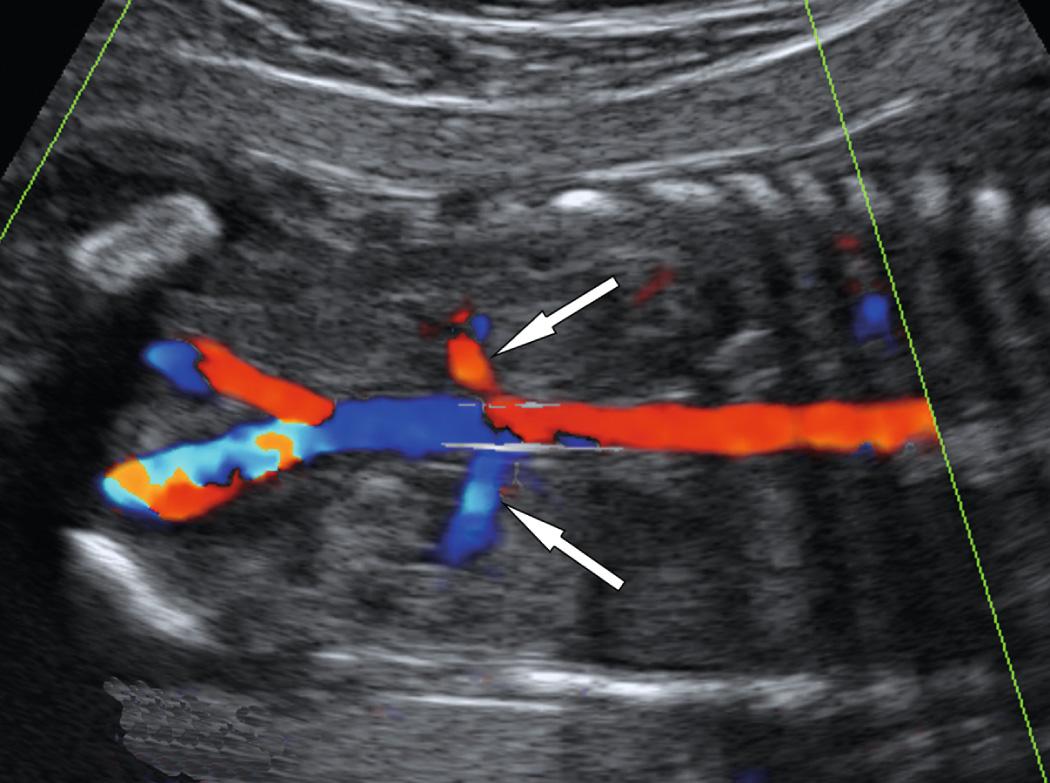
Several techniques have been proposed to improve visualization of fetal structures: intraamniotic and intraperitoneal infusion of isotonic saline, transvaginal ultrasound, and color Doppler ultrasound imaging. The transvaginal probe is particularly useful in the second trimester and with breech presentation ( Fig. 39.6 , ). Color Doppler imaging can be used to diagnose absent renal arteries, providing further evidence for the diagnosis of bilateral renal agenesis ( Fig. 39.7 ). More important, it helps to map out the renal arteries in difficult cases of oligohydramnios, thereby confirming the presence of kidneys and avoiding confusion ( Fig. 39.8 ).
Fetal MRI may help to identify the kidneys when sonographic visualization is limited. However, the kidneys can be difficult to visualize early in gestation. Therefore it may be difficult for MRI to exclude renal agenesis before 24 weeks' gestation, if the kidneys are not seen.
Severe oligohydramnios
Absent kidneys
“Lying down” adrenal sign
Absent renal arteries on color Doppler imaging
Nonvisualization of bladder (over 1 hour)
Poor image quality caused by oligohydramnios
Fetal position (breech presentation)
Amniotic fluid volume may be normal before 16 weeks' gestation
Bowel or adrenal glands can be mistaken for kidneys
Urachal diverticulum may mimic the bladder
Empty bladder may be caused by impaired renal function from other causes (e.g., intrauterine growth restriction)
Bilateral renal agenesis is usually an isolated and sporadic abnormality. However, in a small percentage of cases it may be secondary to a chromosomal abnormality or part of a genetic syndrome (such as Fraser syndrome), or a developmental defect (such as VACTERL association). There is conflicting evidence with regard to associations between bilateral renal agenesis and maternal factors such as prepregnancy obesity, periconception smoking, and alcohol consumption. In nonsyndromic cases, the recurrence risk of having another child with bilateral renal agenesis is approximately 4%. However, up to 15% of the first- and second-degree relatives of affected fetuses have been found to have congenital renal anomalies, most often unilateral renal agenesis; and in these families the recurrence risk may be higher. Screening parents and siblings with renal ultrasound is recommended.
Unilateral renal agenesis is three to four times more common than bilateral renal agenesis, occurring 1 in 1000 births. It can be difficult to diagnose prenatally because AFV is normal and the bladder appears normal. A common pitfall is failure to image the renal fossa in the far field because of acoustic shadowing from the spine, especially in the transverse plane. Meticulous attention to technique is necessary (rotating the transducer, changing the maternal position, or repeated observations). If a kidney is not found in the renal fossa, most are either congenitally absent or ectopic. The contralateral kidney may be enlarged because of compensatory hypertrophy. There is a high incidence of contralateral renal abnormalities, the most common being vesicoureteral reflux (VUR). Unilateral renal agenesis is associated with genital, cardiac, skeletal, and GI abnormalities, as well as with multiorgan syndromes. Isolated unilateral renal agenesis has a good prognosis. Neonatal urologic workup is necessary, including a voiding cystourethrogram (VCUG).
The recurrence risk to parents of a baby with isolated unilateral renal agenesis is about 1% if the parents have normal renal ultrasound. However, if one parent has a congenital solitary kidney, the risks to offspring are 7% for congenital solitary kidney and 1% for bilateral renal agenesis.
One or both kidneys may be in an abnormal position. The incidence of renal ectopia varies between 1 : 500 and 1 : 1200 births, with pelvic kidney being the most common form. When the renal fossa is empty, careful scanning may demonstrate the ectopic kidney adjacent to the bladder or iliac wing ( Fig. 39.9 , ). However, prenatal detection is often difficult because pelvic kidneys may be indistinguishable from bowel loops and are frequently smaller than normally positioned kidneys. Pelvic kidneys may be hypoplastic or dysplastic. Postnatally impaired renal function has been found in 33% of pelvic kidneys, although the overall renal function remained normal at a median follow-up of 8 years.
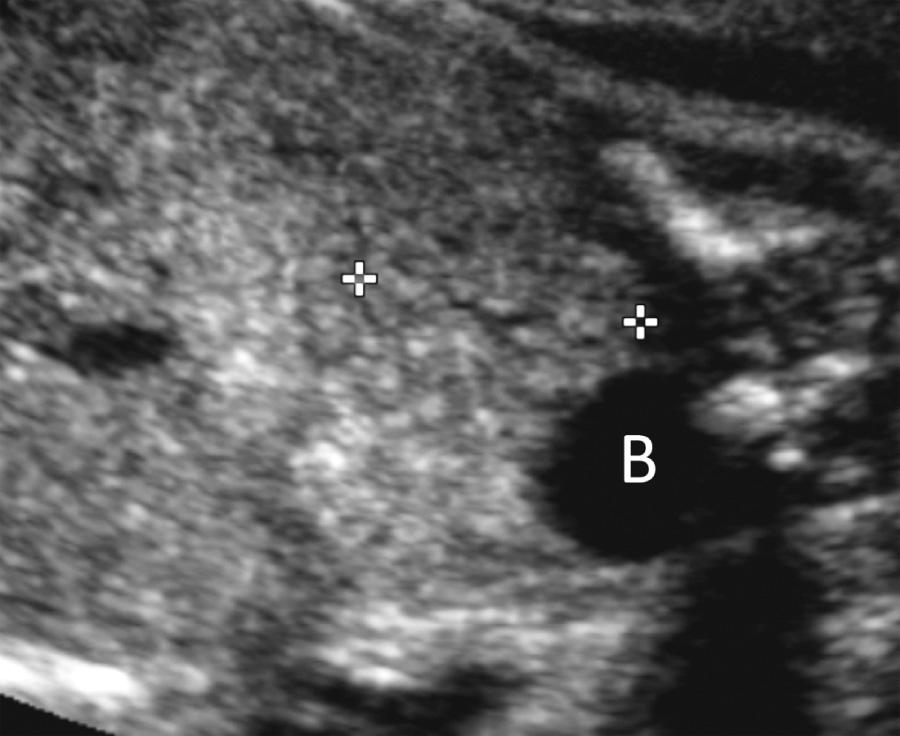
When the ectopic kidney is located on the opposite side of the abdomen relative to its ureteral insertion into the bladder, it is defined as crossed renal ectopia. In most cases, the crossed ectopic kidney fuses with the normally located kidney (crossed fused renal ectopia), and an enlarged bilobed kidney is seen ( Fig. 39.10 , ). Crossed renal ectopia is divided into six subgroups based on the degree of fusion and location and rotation of the fused renal mass. Most commonly, the crossed ectopic kidney is inferior to the normally positioned kidney, and the upper pole of the ectopic kidney is fused to the lower pole of the normal kidney.
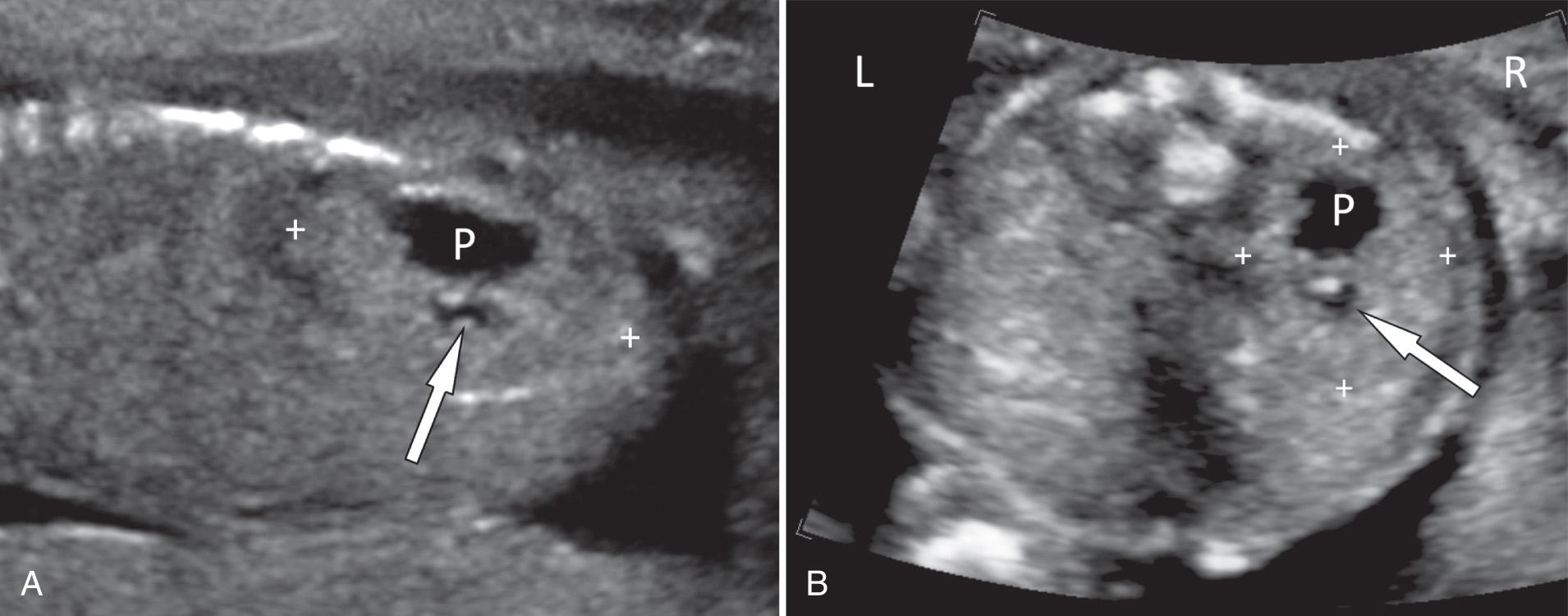
Renal ectopia is associated with a high incidence of urologic abnormalities (most often VUR), as well as nonurinary malformations. Neonatal renal ultrasound is necessary. A VCUG is indicated if there is hydronephrosis in the ectopic or contralateral kidney.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here