Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Two-dimensional ultrasound is the primary imaging modality for the majority of musculoskeletal disorders.
When a fetal musculoskeletal dysplasia is suspected on ultrasound, referral to a center with expertise may be helpful.
A main role of ultrasound is to determine if the condition is likely lethal; it is important to use multiple parameters to make this determination.
After delivery or pregnancy termination, a diagnosis should be established by a combination of clinical examination, imaging, and molecular studies. Cell culture and DNA banking are essential for molecular investigations. The results should be reviewed with the parents to help in their future reproductive plans.
Congenital bone disorders of the skeleton comprise a large and heterogeneous group of disorders primarily affecting the growth and development of the musculoskeletal system. The prevalence of skeletal dysplasias, also called “osteochondrodysplasias,” diagnosed prenatally or during the neonatal period, excluding limb amputations, is 2.4 to 4.5 per 10,000 births.
There are three major categories: skeletal dysplasias, dysostoses, and disruptions ( Table 40.1 ). The number of recognized genetic disorders with a substantial skeletal component is increasing, and the distinction among dysplasias, metabolic bone disorders, dysostoses, and malformation syndromes is constantly evolving. The Nosology and Classification of Genetic Skeletal Disorders 2015 revision recognizes 436 genetic bone disorders with a substantial skeletal component ( Table 40.2 ).
| Skeletal dysplasias | Developmental disorders of chondro-osseous tissue caused by single-gene disorders with prenatal and postnatal manifestations |
| Dysostoses | Single-gene disorders resulting in malformations of individual bones caused by transient abnormalities of signaling factors |
| Disruptions | Morphologic defects of an organ or of larger region resulting from extrinsic breakdown or interference with an originally normal developmental process |
| Skeletal Dysplasia | Prevalence per 100,000 Births |
|---|---|
| LETHAL DYSPLASIAS | |
| Thanatophoric dysplasia | 2.4 to 6.9 |
| Achondrogenesis | 0.9 to 2.3 |
| Osteogenesis imperfecta type IIA | 1.8 |
| Hypophosphatasia congenita | 1.0 |
| VARIABLE-PROGNOSIS DYSPLASIAS | |
| Rhizomelic chondrodysplasia punctata | 0.5 to 0.9 |
| Campomelic dysplasia | 1.0 to 1.5 |
| Asphyxiating thoracic dystrophy | 0.8 to 1.4 |
| Ellis–van Creveld syndrome | 0.7 |
| Osteogenesis imperfecta (other types) | 1.8 |
| NONLETHAL DYSPLASIAS | |
| Heterozygous achondroplasia | 3.3 to 3.8 |
| OVERALL | 24.4 to 75.0 |
Despite increased knowledge about the genetic origins of many of these conditions and the improved ability to diagnose and categorize these disorders correctly, the clinical and imaging features remain a fundamental tool for diagnosing and directing the molecular investigation. Although many fetal skeletal dysplasias can be accurately identified by prenatal ultrasound, this remains a challenging task because of the low incidence, phenotypic variability, and wide range of appearances. The majority of cases have no family history of a similar condition. Nonetheless, the majority of lethal skeletal dysplasias, and in particular the most common— thanatophoric dysplasia, achondrogenesis, and osteogenesis imperfecta type II—can be diagnosed solely on the basis of prenatal ultrasound. Tretter et al. determined that 26 of 27 lethal skeletal dysplasias were identified correctly by prenatal ultrasound; however, only 13 of 27 (48%) received an accurate specific antenatal diagnosis. Eight of 14 (57%) underwent a substantial change in genetic counseling when cytogenetic (including microarray), molecular, pathologic, and imaging findings were combined. Thus although the ultrasound diagnosis of a lethal skeletal dysplasia is highly accurate (85%-95%), a correct specific diagnosis is obtained in only 40% to 55% of cases. The addition of correlative radiographs, cytogenetic analysis (including microarray and whole genome sequencing techniques), and pathologic examination can provide a correct specific diagnosis in up 86%.
Nonetheless, an accurate prenatal determination of the lethality of a given skeletal dysplasia is crucial in helping couples with decision making. Typically, a combination of ultrasound, radiologic, and genetic investigation is required to classify a specific congenital musculoskeletal disorder. A prenatal diagnosis of a musculoskeletal anomaly will provide an opportunity for genetic counseling, resulting in pregnancy termination or tertiary-level care depending on the parental decision. A multidisciplinary approach involving the medical imaging team, obstetrician, medical geneticist, and perinatologist is important in optimizing the accuracy of prognosis and determining recurrence risk. If local expertise is unavailable and/or the diagnosis is unknown, consultation with experts in the field should be considered. Detailed and up-to-date information regarding the molecular tests available for the diagnosis of skeletal dysplasias is available at various organizations.
European Skeletal Dysplasia Network ( http://www.esdn.org )
Gene test ( https://www.genetest.org )
International Skeletal Dysplasia Society ( www.isds.ch )
This information is crucial to the family and to medical personnel involved in planning the management of both current and future pregnancies. This chapter uses a “key features” approach to the sonographic diagnosis of the common skeletal dysplasias to aid in the classification and differential diagnosis.
The high level of intrinsic contrast of the fetal extremities places them among the earliest structures that can be evaluated by ultrasound. By the end of the embryonic period, the differentiation of bones, joints, and musculature is similar to that of an adult and is associated with increased fetal movements. Transvaginal ultrasound can demonstrate the limb buds by 7 weeks' gestation, and the foot and hand plates are visible by 8 weeks. Osteogenesis begins in the clavicle and mandible by 8 weeks as well. By 11 or 12 weeks, the primary ossification centers of the long bones (e.g., scapula, ileum), as well as the limb articulations and phalanges, can be identified. The ischium, metacarpals, and metatarsals ossify during the fourth month of gestation. The pubis, calcaneus, and talus ossify during the fifth and sixth months. Ossification of the other tarsal and carpal bones occurs postnatally. The direction of growth in the long bones is from proximal to distal, and the lower extremities lag slightly behind the upper extremities.
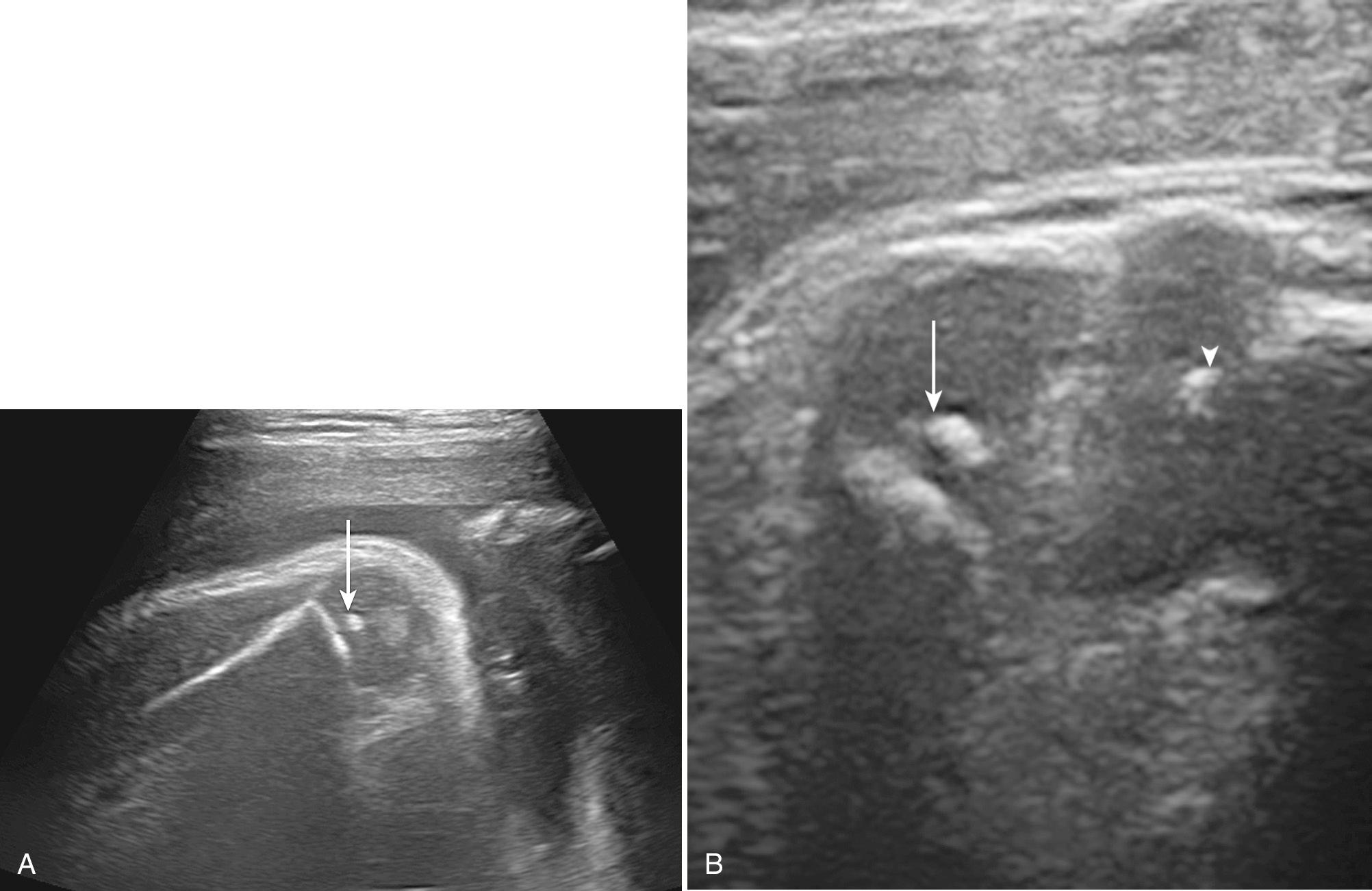
Of the secondary ossification centers in the long bones, only the distal femoral epiphysis, the proximal tibial epiphysis, and occasionally the proximal humeral epiphysis ossify prenatally ( Fig. 40.1 ). The unossified epiphysis appears hypoechoic, with a variably, mildly echogenic center. Ossification begins centrally. The distal femoral epiphysis can ossify as early as 29 weeks' menstrual age and as late as 34 weeks. When it measures greater than 7 mm, the menstrual age is generally later than 37 weeks. The proximal tibial epiphysis begins to ossify by 35 menstrual weeks. In uncomplicated pregnancies the combination of a distal femoral epiphysis of 3 mm or greater and the presence of a proximal tibial epiphysis is considered a reliable marker of pulmonary maturity. Intrauterine growth restriction (IUGR) is associated with a delay in ossification of the distal femoral epiphysis and proximal tibial epiphysis. The earliest secondary epiphysis to ossify is the calcaneus, at approximately 20 weeks' gestation, thus marking the earliest point that assessment of delayed ossification of the secondary epiphyseal centers can be attempted.
The fascia within the muscle is highly echogenic compared with the relatively hypoechoic cartilage. The fetal musculature is slightly more echogenic than the relatively hypoechoic cartilage. The fetal joint spaces, in particular the knee, appear echogenic because of the combination of synovium, fat, and microvasculature. The normal development and ultimate function of the fetal musculoskeletal system depend on fetal movements, which start by the second half of the first trimester. In the absence of normal fetal motion, the bones and muscles will be underdeveloped, the chest will be narrow, and joint contractures and postural deformities can occur.
It is a standard practice to assess femur length (FL) as part of the evaluation of fetal size and morphology. Although measurement of all the long bones is not required in a routine obstetric ultrasound, an overall evaluation of the fetal skeleton should be performed to ensure the presence and bilateral symmetry of the tubular bones. Available charts provide guidance for correlating the length of the extremities with the gestational age ( Table 40.3 ).
| Menstrual Age (Weeks) | Biparietal Diameter | BONE | |||||
|---|---|---|---|---|---|---|---|
| Femur | Tibia | Fibula | Humerus | Radius | Ulna | ||
| 13 | 2.3 (0.3) | 1.1 (0.2) | 0.9 (0.2) | 0.8 (0.2) | 1.0 (0.2) | 0.6 (0.2) | 0.8 (0.3) |
| 14 | 2.7 (0.3) | 1.3 (0.2) | 1.0 (0.2) | 0.9 (0.3) | 1.2 (0.2) | 0.8 (0.2) | 1.0 (0.2) |
| 15 | 3.0 (0.1) | 1.5 (0.2) | 1.3 (0.2) | 1.2 (0.2) | 1.4 (0.2) | 1.1 (0.1) | 1.2 (0.1) |
| 16 | 3.3 (0.2) | 1.9 (0.3) | 1.6 (0.3) | 1.5 (0.3) | 1.7 (0.2) | 1.4 (0.3) | 1.6 (0.3) |
| 17 | 3.7 (0.3) | 2.2 (0.3) | 1.8 (0.3) | 1.7 (0.2) | 2.0 (0.4) | 1.5 (0.3) | 1.7 (0.3) |
| 18 | 4.2 (0.5) | 2.5 (0.3) | 2.2 (0.3) | 2.1 (0.3) | 2.3 (0.3) | 1.9 (0.2) | 2.2 (0.3) |
| 19 | 4.4 (0.4) | 2.8 (0.3) | 2.5 (0.3) | 2.3 (0.3) | 2.6 (0.3) | 2.1 (0.3) | 2.4 (0.3) |
| 20 | 4.7 (0.4) | 3.1 (0.3) | 2.7 (0.2) | 2.6 (0.2) | 2.9 (0.3) | 2.4 (0.2) | 2.7 (0.4) |
| 21 | 5.0 (0.5) | 3.5 (0.4) | 3.0 (0.4) | 2.9 (0.4) | 3.2 (0.4) | 2.7 (0.4) | 3.0 (0.4) |
| 22 | 5.5 (0.5) | 3.6 (0.3) | 3.2 (0.3) | 3.1 (0.3) | 3.3 (0.3) | 2.8 (0.5) | 3.1 (0.4) |
| 23 | 5.8 (0.5) | 4.0 (0.4) | 3.6 (0.2) | 3.4 (0.2) | 3.7 (0.3) | 3.1 (0.4) | 3.5 (0.2) |
| 24 | 6.1 (0.5) | 4.2 (0.3) | 3.7 (0.3) | 3.6 (0.3) | 3.8 (0.4) | 3.3 (0.4) | 3.6 (0.4) |
| 25 | 6.4 (0.5) | 4.6 (0.3) | 4.0 (0.3) | 3.9 (0.4) | 4.2 (0.4) | 3.5 (0.3) | 3.9 (0.4) |
| 26 | 6.8 (0.5) | 4.8 (0.4) | 4.2 (0.3) | 4.0 (0.3) | 4.3 (0.3) | 3.6 (0.4) | 4.0 (0.3) |
| 27 | 7.0 (0.3) | 4.9 (0.3) | 4.4 (0.3) | 4.2 (0.3) | 4.5 (0.2) | 3.7 (0.3) | 4.1 (0.2) |
| 28 | 7.3 (0.5) | 5.3 (0.5) | 4.5 (0.4) | 4.4 (0.3) | 4.7 (0.4) | 3.9 (0.4) | 4.4 (0.5) |
| 29 | 7.6 (0.5) | 5.3 (0.5) | 4.6 (0.3) | 4.5 (0.3) | 4.8 (0.4) | 4.0 (0.5) | 4.5 (0.4) |
| 30 | 7.7 (0.6) | 5.6 (0.3) | 4.8 (0.5) | 4.7 (0.3) | 5.0 (0.5) | 4.1 (0.6) | 4.7 (0.3) |
| 31 | 8.2 (0.7) | 6.0 (0.6) | 5.1 (0.3) | 4.9 (0.5) | 5.3 (0.4) | 4.2 (0.3) | 4.9 (0.4) |
| 32 | 8.5 (0.6) | 6.1 (0.6) | 5.2 (0.4) | 5.1 (0.4) | 5.4 (0.4) | 4.4 (0.6) | 5.0 (0.6) |
| 33 | 8.6 (0.4) | 6.4 (0.5) | 5.4 (0.5) | 5.3 (0.3) | 5.6 (0.5) | 4.5 (0.5) | 5.2 (0.3) |
| 34 | 8.9 (0.5) | 6.6 (0.6) | 5.7 (0.5) | 5.5 (0.4) | 5.8 (0.5) | 4.7 (0.5) | 5.4 (0.5) |
| 35 | 8.9 (0.7) | 6.7 (0.6) | 5.8 (0.4) | 5.6 (0.4) | 5.9 (0.6) | 4.8 (0.6) | 5.4 (0.4) |
| 36 | 9.1 (0.7) | 7.0 (0.7) | 6.0 (0.6) | 5.6 (0.5) | 6.0 (0.6) | 4.9 (0.5) | 5.5 (0.3) |
| 37 | 9.3 (0.9) | 7.2 (0.4) | 6.1 (0.4) | 6.0 (0.4) | 6.1 (0.4) | 5.1 (0.3) | 5.6 (0.4) |
| 38 | 9.5 (0.6) | 7.4 (0.6) | 6.2 (0.3) | 6.0 (0.4) | 6.4 (0.3) | 5.1 (0.5) | 5.8 (0.6) |
| 39 | 9.5 (0.6) | 7.6 (0.8) | 6.4 (0.7) | 6.1 (0.6) | 6.5 (0.6) | 5.3 (0.5) | 6.0 (0.6) |
| 40 | 9.9 (0.8) | 7.7 (0.4) | 6.5 (0.3) | 6.2 (0.1) | 6.6 (0.4) | 5.3 (0.3) | 6.0 (0.5) |
| 41 | 9.7 (0.6) | 7.7 (0.4) | 6.6 (0.4) | 6.3 (0.5) | 6.6 (0.4) | 5.6 (0.4) | 6.3 (0.5) |
| 42 | 10.0 (0.5) | 7.8 (0.7) | 6.8 (0.5) | 6.7 (0.7) | 6.8 (0.7) | 5.7 (0.5) | 6.5 (0.5) |
The longest femur measurement, excluding both proximal and distal epiphyses, is usually chosen. The inclusion of the distal femur point, or the specular reflection of the lateral aspect of the distal femoral epiphysis cartilage, is the most common reason for overestimating FL ( Fig. 40.2A ). An oblique FL measurement will result in underestimate of length. The lateral border of the femur in the near field of the transducer appears straight, whereas the medial border of the femur in the far field has a curved appearance ( Fig. 40.2B ).
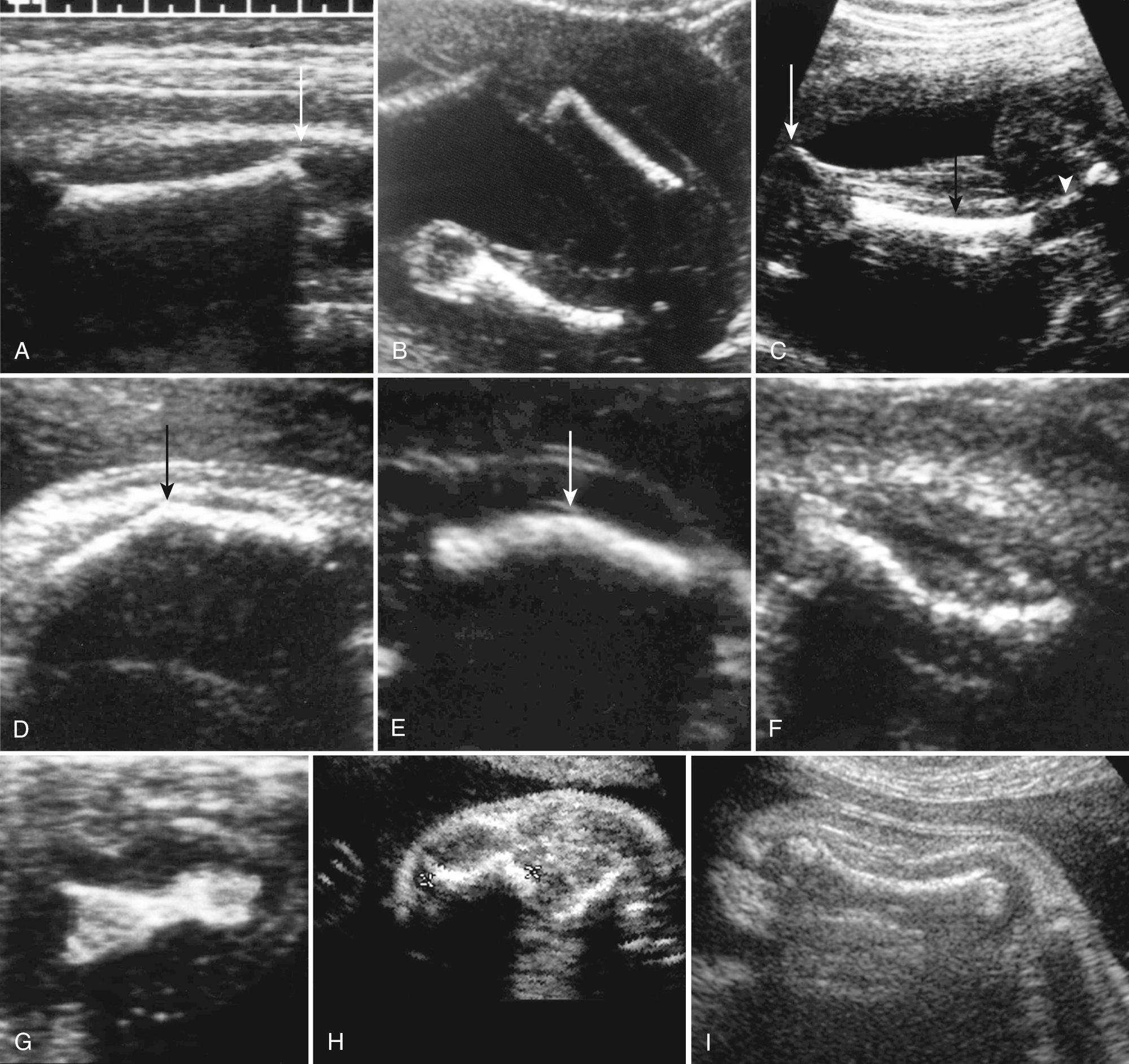
In the lower extremity the lateral bone is the fibula and the medial bone is the tibia. The tibia and fibula end at the same level distally. In the upper extremity, pronation may cause the radius and ulna to cross, so it can be difficult to distinguish the ulna from the radius using lateral and medial positions. The ulna is distinguished from the radius by its longer proximal extent and its relationship to the fifth digit distally. The radius and ulna end at the same level distally. Demonstration of this relationship will effectively exclude the majority of radial ray defects.
The clavicles grow in a linear fashion, approximately 1 mm per week, and the gestational age in weeks is approximately the length of the clavicle in millimeters from 14 weeks to term. By 40 weeks' gestation, the clavicles measure approximately 40 mm.
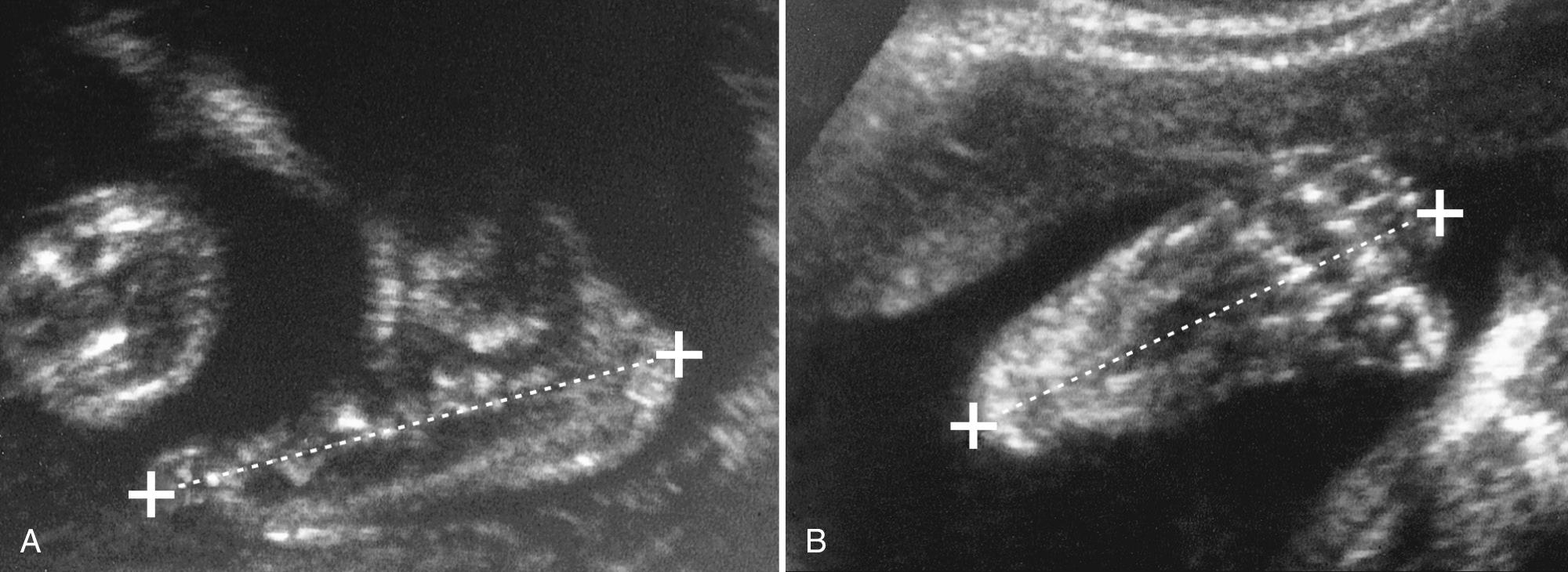
Foot length is measured from the skin edge overlying the calcaneus to the distal end of the longest toe (the first or second toe) on either the plantar or the sagittal view ( Fig. 40.3 ). The ossified FL is almost equivalent to the foot length, resulting in a normal femur-to-foot length ratio of approximately 1.0. This ratio remains relatively constant from the 14th week of gestation onward. If the fetus is constitutionally small or there is symmetrical IUGR, the ratio is generally 0.9 or greater. In most skeletal dysplasias characterized by short limbs, the ratio is generally less than 0.9 because of the relative sparing of the hands and feet. The greater the deviation is from the lower limits of the norm, the greater is the disproportion and usually also the severity.
A prenatal evaluation of a skeletal dysplasia is indicated if there is a positive family history or an abnormal length or appearance of the bones at ultrasound.
Family history
Serial measurements
Degree of limb shortening
Pattern of limb shortening
Presence of bowing, fractures, and angulations
Spine
Thoracic measurements
Hands and feet
Calvarium and facial features
A positive family history of a sibling or parents affected by a skeletal dysplasia or consanguineous parents should prompt an intensive ultrasound investigation with a focus on targeted abnormalities and serial measurements. A history of consanguinity is important because many of the skeletal dysplasias have an autosomal recessive mode of inheritance. Heterozygous achondroplasia, the most common nonlethal skeletal dysplasia, has an autosomal dominant pattern of inheritance. Family history may not be helpful because 80% of cases are caused by a new mutation.
The fetal femur is often the only long bone routinely measured at the second-trimester ultrasound evaluation. An abnormal FL is traditionally defined as below 2 standard deviations (SD) for gestational age. Using this cutoff, 2.5% of all fetuses would be classified as having short limbs. This exceeds the expected frequency of skeletal dysplasias, and thus additional investigations are needed to separate fetuses with a skeletal dysplasia from those with no underlying pathology and those with other diagnoses associated with growth restriction.
When one or all of the long bones measure less than 2 SD for gestational age, a follow-up ultrasound should be done in 3 or 4 weeks to evaluate the interval growth. If the interval femur growth is normal, there is a high likelihood that the fetus does not have skeletal dysplasia. However, further deviation from the mean by at least 1 SD suggests the presence of a skeletal dysplasia or severe IUGR. When FL measures below 4 SD for gestational age, there is a high likelihood of a skeletal dysplasia. Kurtz et al. have shown that the number of millimeters below the −2 SD line is a simple screening tool to evaluate femoral shortening with the following guidelines:
If FL is 1 to 4 mm below the −2 SD point, further serial measurements are required to determine if a skeletal dysplasia is present.
If FL is more than 5 mm below the −2 SD point, there is a high likelihood of a skeletal dysplasia.
The most common cause of a so-called short femur is either inaccurate dating or a normal variant in a constitutionally small fetus, which may be associated with a parental or family history of less-than-average stature. In about 13% of cases, a remeasurement will bring the FL into a normal range, likely representing a false-positive diagnosis rather than a growth spurt. Isolated, symmetrical short femurs identified at the second midtrimester ultrasound evaluation are helpful in identifying a group of fetuses at increased risk for low birth weight, small for gestational age, or severe IUGR. Typically, these fetuses will also have small abdominal circumference measurements.
Occasionally, fetuses with severe IUGR will have greatly shortened long bones. Associated findings of normal or decreased skin fold measurements, oligohydramnios, abnormal placental morphology, and abnormal Doppler waveforms suggest the diagnosis of IUGR, whereas redundant, thickened skin folds and polyhydramnios typically accompany short-limb dysplasias.
Nonlethal skeletal dysplasias such as heterozygous achondroplasia are often not evident before 20 weeks' gestation. The findings of short long bones before 20 weeks indicate a more serious and usually fatal skeletal dysplasia. As a rule, the earlier the detection of limb shortening, the worse is the prognosis. Virtually all cases diagnosed in first trimester are considered severe skeletal dysplasias, with the large majority representing lethal conditions. First-trimester skeletal biometry tables are available. Although mild, isolated shortening of the femur indicates increased risk for trisomy 21 by 1.5-fold, other factors are more important for assessing this risk.
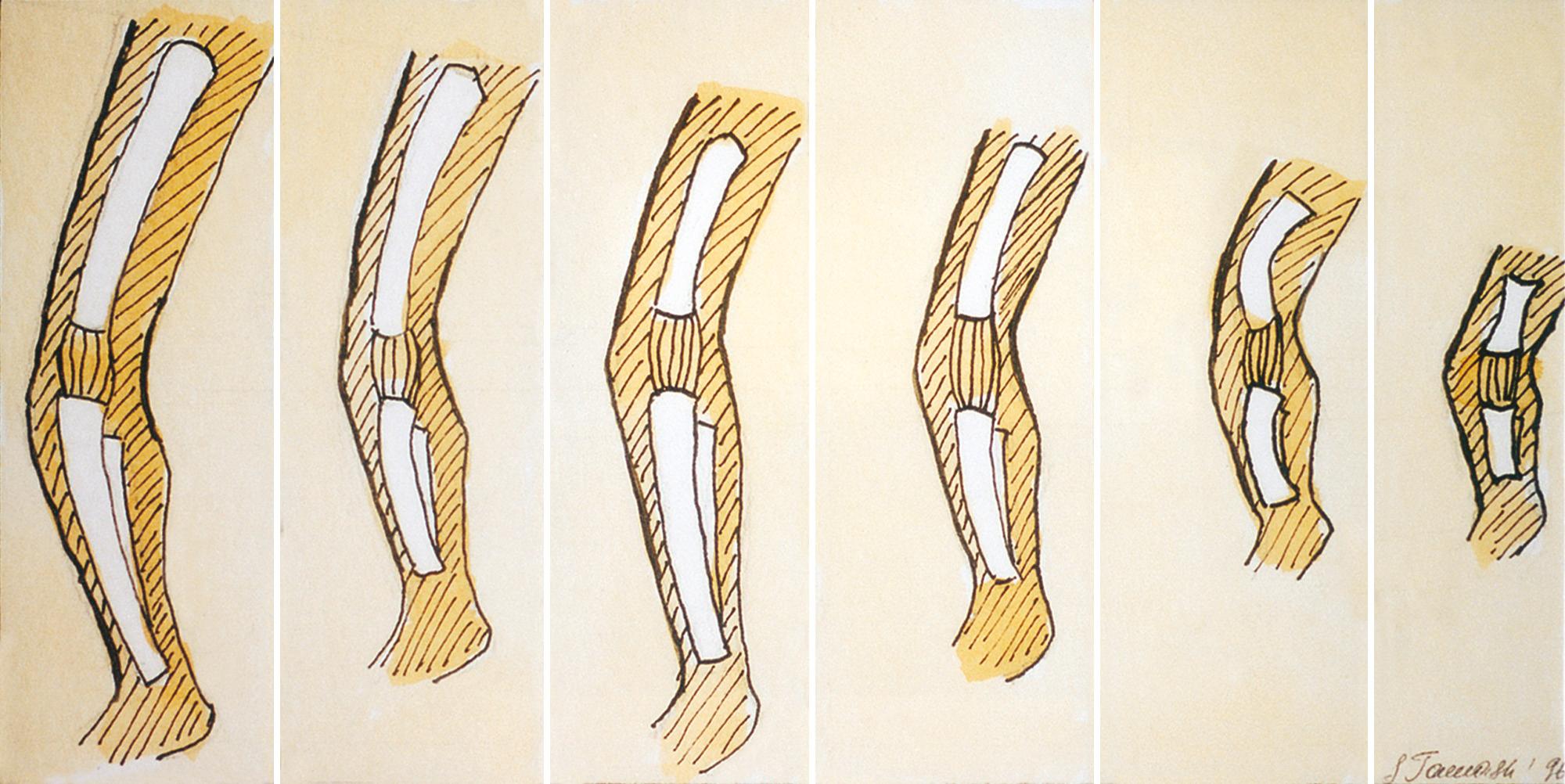
The pattern of limb shortening should be assessed to determine which long-bone segments are most severely affected ( Fig. 40.4 ). Rather than millimeters, we find it useful to standardize measurements to “weeks of size” to determine disproportion. There are four main patterns of shortening of the long bones: rhizomelia, shortening of the proximal segment; mesomelia, shortening of the middle segment; acromelia, shortening of the distal segment; and micromelia, shortening of the entire limb (mild, mild/bowed, or severe).
Rhizomelia: shortening of proximal segment (femur, humerus)
Mesomelia: shortening of middle segment (radius, ulna/tibia, fibula)
Acromelia: shortening of distal segment (hands, feet)
Micromelia: shortening of entire limb (mild, mild/bowed, or severe)
The shape, contour, and density of the bones should be assessed for the presence of bowing, angulation, fracture, or thickening. Bowing is a nonspecific finding, typically caused by underlying osseous fragility. Although more than 40 distinct disorders are associated with bowed, bent, or angulated femurs, most cases can be accounted for by three relatively frequent disorders: campomelic dysplasia, thanatophoric dysplasia, and osteogenesis imperfecta (OI). Patients with OI types I and IV can present with apparently isolated in utero bowing of the long bones (especially the femur) without frank fractures (see Fig. 40.2D ). Anterior bowing of the tibia, femur, and humerus may suggest the diagnosis of campomelic dysplasia; other more specific findings such as hypoplastic scapulae and cervical kyphosis are typically present and help distinguish campomelic dysplasia from OI and thanatophoric dysplasia, which are both more common. Bone fractures may appear as angulations or interruptions in the bone contour or as thick, wrinkled contours corresponding to repetitive cycles of fracture and callus formation (see Fig. 40.2F ). Decreased or absent acoustic shadowing is a marker for decreased mineralization of the long bones. When evident, this is helpful in narrowing the differential diagnosis, but in its absence the bone mineralization may still be abnormal.
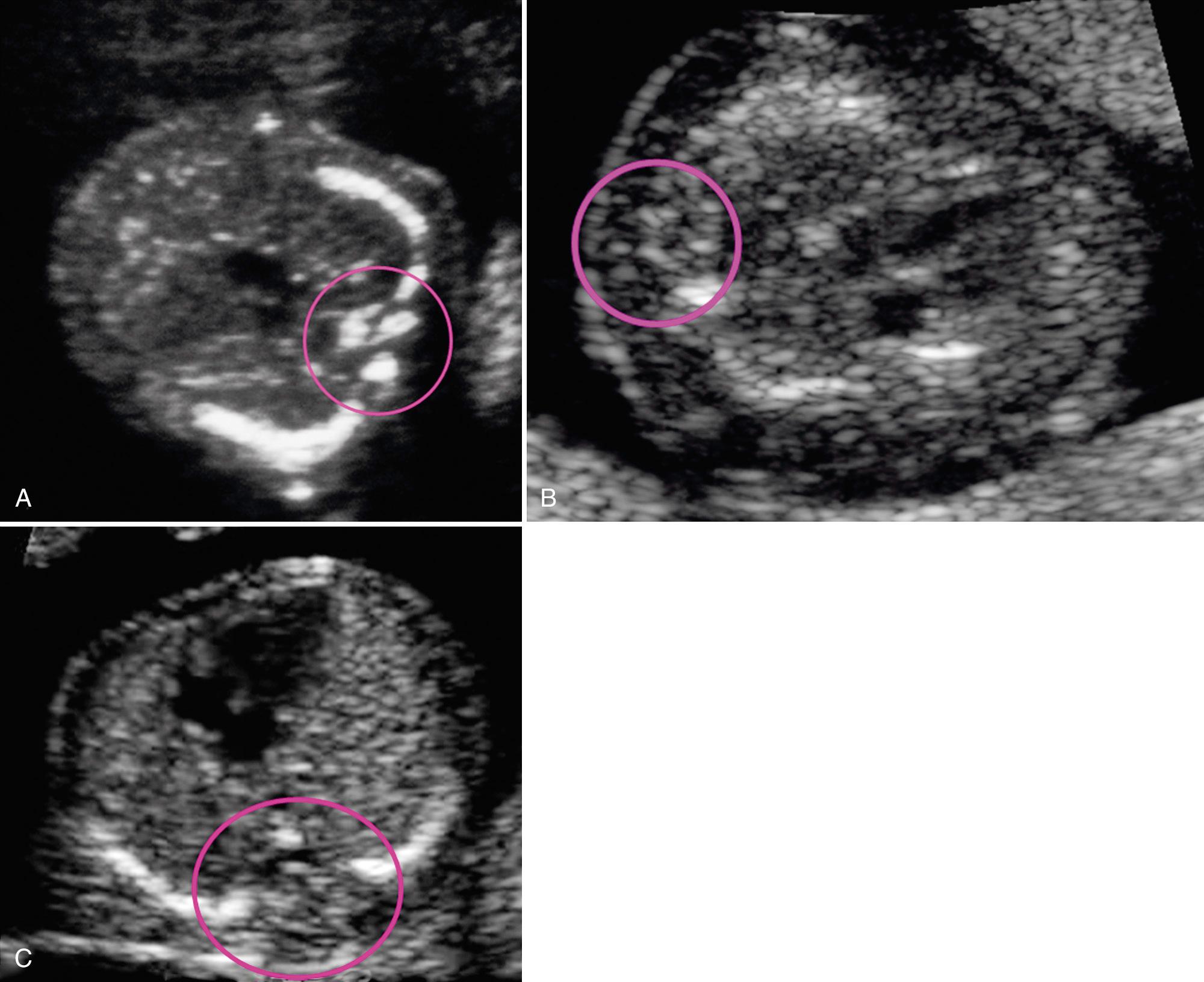
The spine is assessed for segmentation anomalies, kyphoscoliosis, platyspondyly (flattened vertebral bodies), demineralization, myelodysplasia, and caudal regression syndromes. Although platyspondyly is the most common spine abnormality, it is a challenging prenatal diagnosis. Demineralization of the spine can result in the appearance of ghost vertebrae or nonvisualization of one or all of the three ossification centers ( Fig. 40.5 ). A progressively narrowed lumbar interpedicular distance is associated with homozygous achondroplasia; a widened interpedicular distance is associated with myelodysplasia. Three-dimensional imaging may provide a large field of view and additional details that may be helpful in achieving a diagnosis ( Fig. 40.6 ).
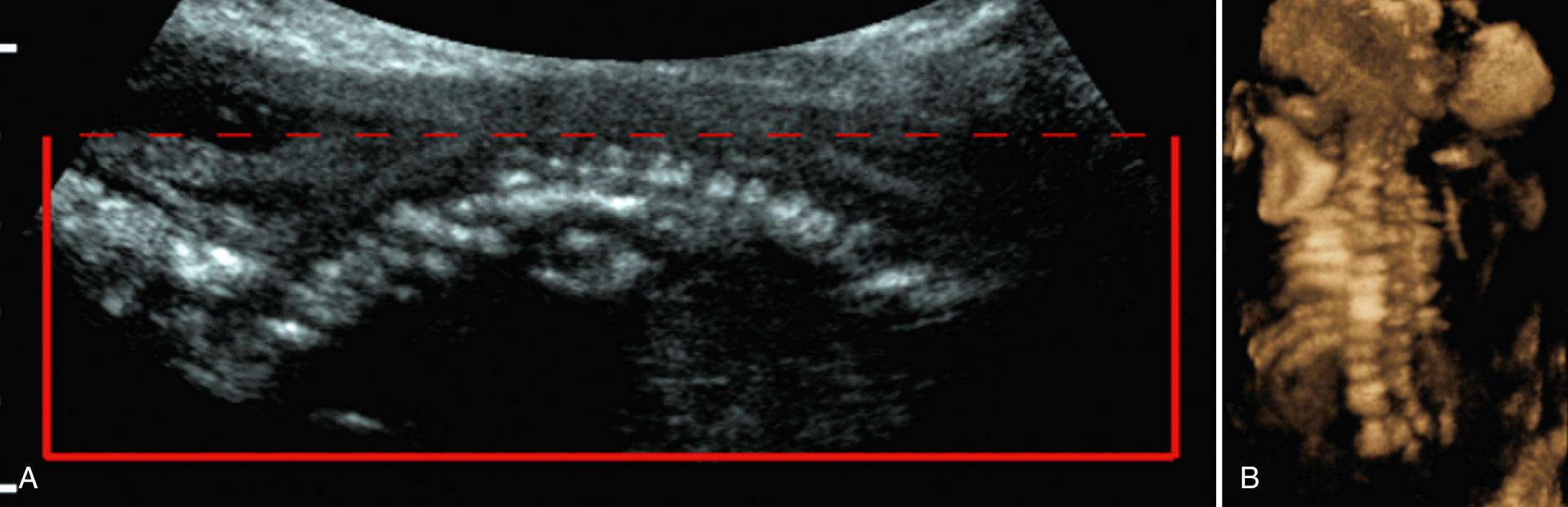
The most reliable prognostic indicator of the lethality of a given skeletal dysplasia is pulmonary hypoplasia. Ultrasound is 85% to 95% accurate in the diagnosis of a lethal skeletal dysplasia on the basis of pulmonary hypoplasia. The thoracic circumference is measured at the level of the four-chamber heart and compared to nomograms (see Chapter 36 , Table 36.2 ). A thoracic-to-abdominal circumference ratio of less than 0.8 is considered abnormal. The thoracic length (from the neck to the diaphragm) is also measured, and the ribs are assessed to determine if they are short. At the level of the four-chamber cardiac view, the ribs should normally encircle at least 70% of the thoracic circumference. The ribs remain in a relatively horizontal plane, as does the cardiac axis, facilitating this evaluation. In a sagittal view, a markedly narrowed anteroposterior diameter of the thorax is associated with pulmonary hypoplasia. In the coronal view, a concave or bell-shaped contour is associated with pulmonary hypoplasia ( Fig. 40.7 ).
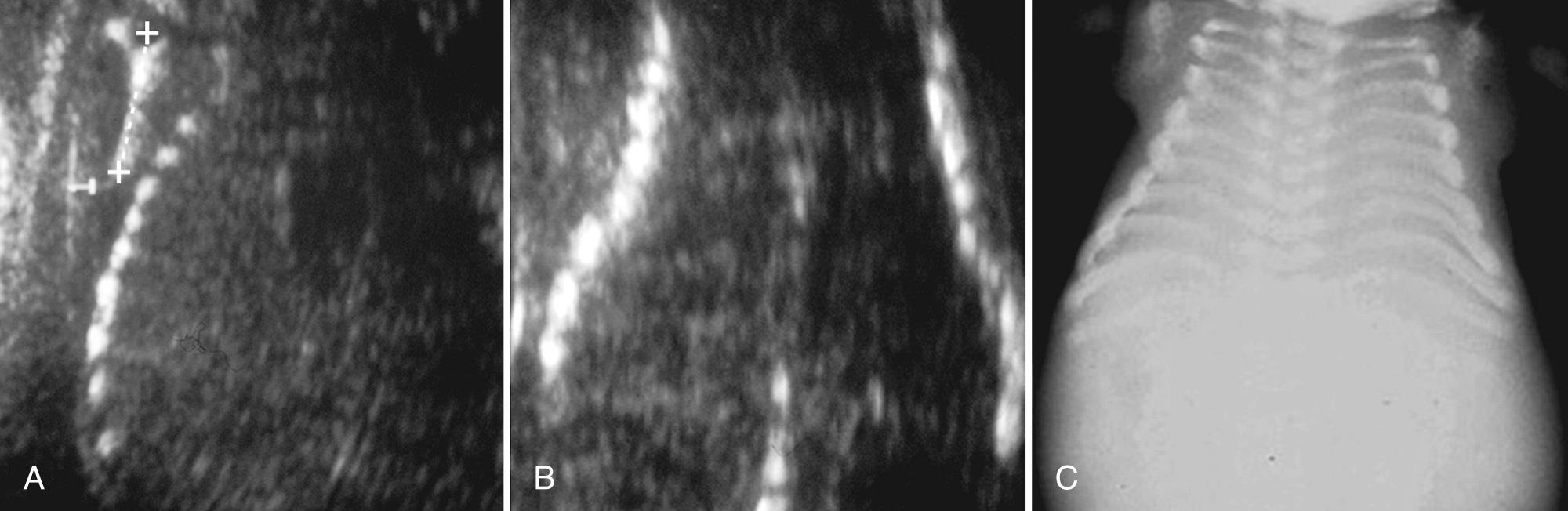
The hands and feet are examined for deformities such as clubfoot or clubhand. A hitchhiker thumb, or abducted thumb, is associated with diastrophic dwarfism. Fixed postural deformities may suggest the diagnosis of arthrogryposis multiplex congenita. Polydactyly is associated with short-rib polydactyly syndromes, Ellis–van Creveld syndrome, asphyxiating thoracic dystrophy, and some chromosomal abnormalities.
The fetal cranium is assessed for the presence of macrocranium, frontal bossing, cloverleaf skull deformity, underlying brain abnormalities, and facial abnormalities, such as saddle nose, hypertelorism, and cleft lip and palate. An abnormal cranial contour may indicate craniosynostosis or premature fusion of the sutures. The most reliable sonographic sign of demineralization is increased compressibility of the calvarium. This finding is typically present in fetuses with osteogenesis imperfecta type II and hypophosphatasia. The falx may appear abnormally bright or echogenic compared to the demineralized calvarium.
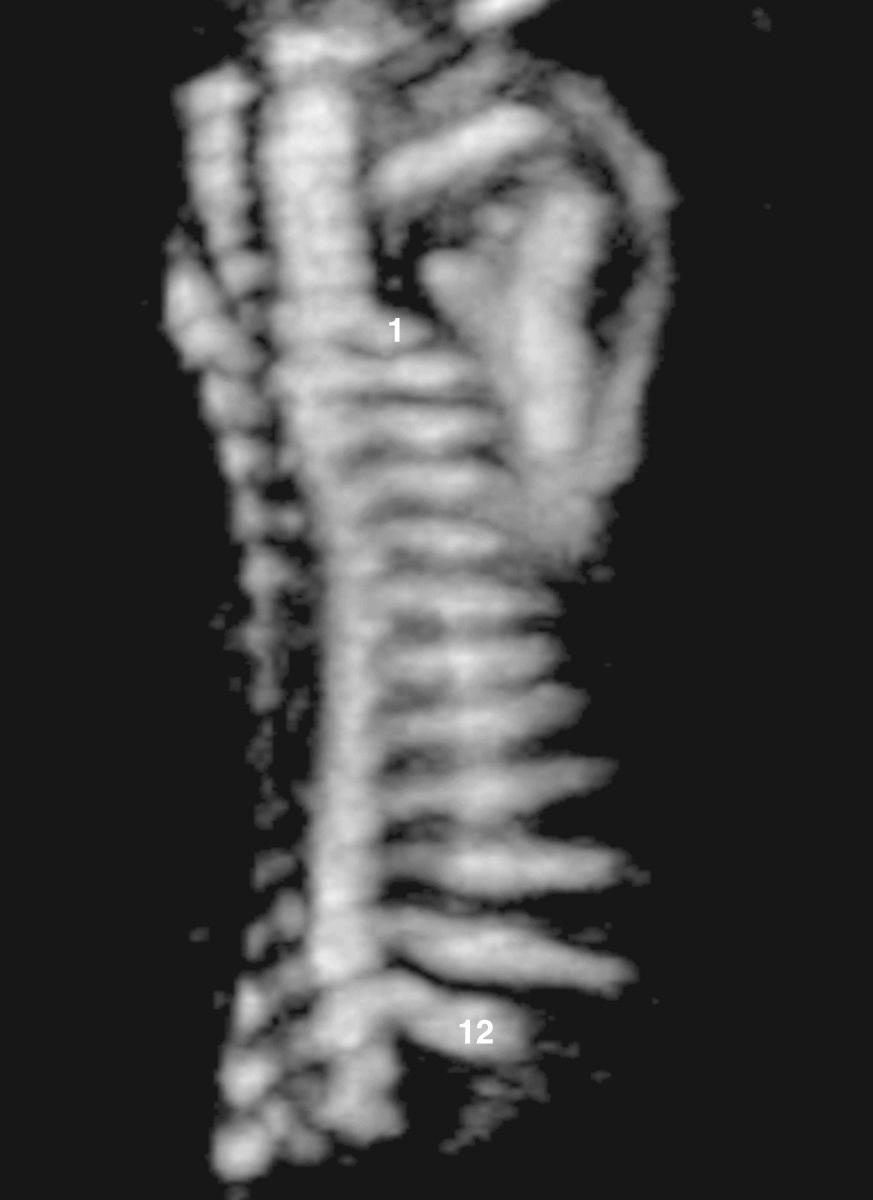
The ribs are assessed to ensure an adequate length, thus minimizing the risk of pulmonary hypoplasia, and are examined for abnormal number or appearance. The finding of an abnormal number of fetal ribs is an isolated finding of no clinical importance in the majority of cases, associated with minor anomalies in a smaller group (29%) and only occasionally associated with severe malformations. Associated syndromes include Poland syndrome, VACTERL association (vertebral abnormalities, anal atresia, cardiac abnormalities, tracheoesophageal fistula, renal agenesis and dysplasia, limb defects), campomelic dysplasia, and chromosome abnormalities. Three-dimensional ultrasound volume images aid in accurately counting the number of ribs ( Fig. 40.8 ).
Ultimately, a detailed examination of each bone may be required to determine the fetal condition. Specific dysmorphic features of bones (e.g., clavicular or scapular hypoplasia; aplasia of fibula, tibia, or radius; platyspondyly) can be helpful to further define a specific skeletal dysplasia. A detailed evaluation of the cardiovascular, genitourinary, gastrointestinal, and central nervous system should be done concurrently with the musculoskeletal evaluation.
Dizygotic twin pregnancies are at similar risk for skeletal abnormalities as singleton pregnancies, and the frequency is increased two to three times in monozygotic twins. Both monozygotic and dizygotic twins can be discordant for genetic and nongenetic skeletal abnormalities. Twin pregnancies are generally discordant and overall, about 15% of twins are concordant for the same anomaly.
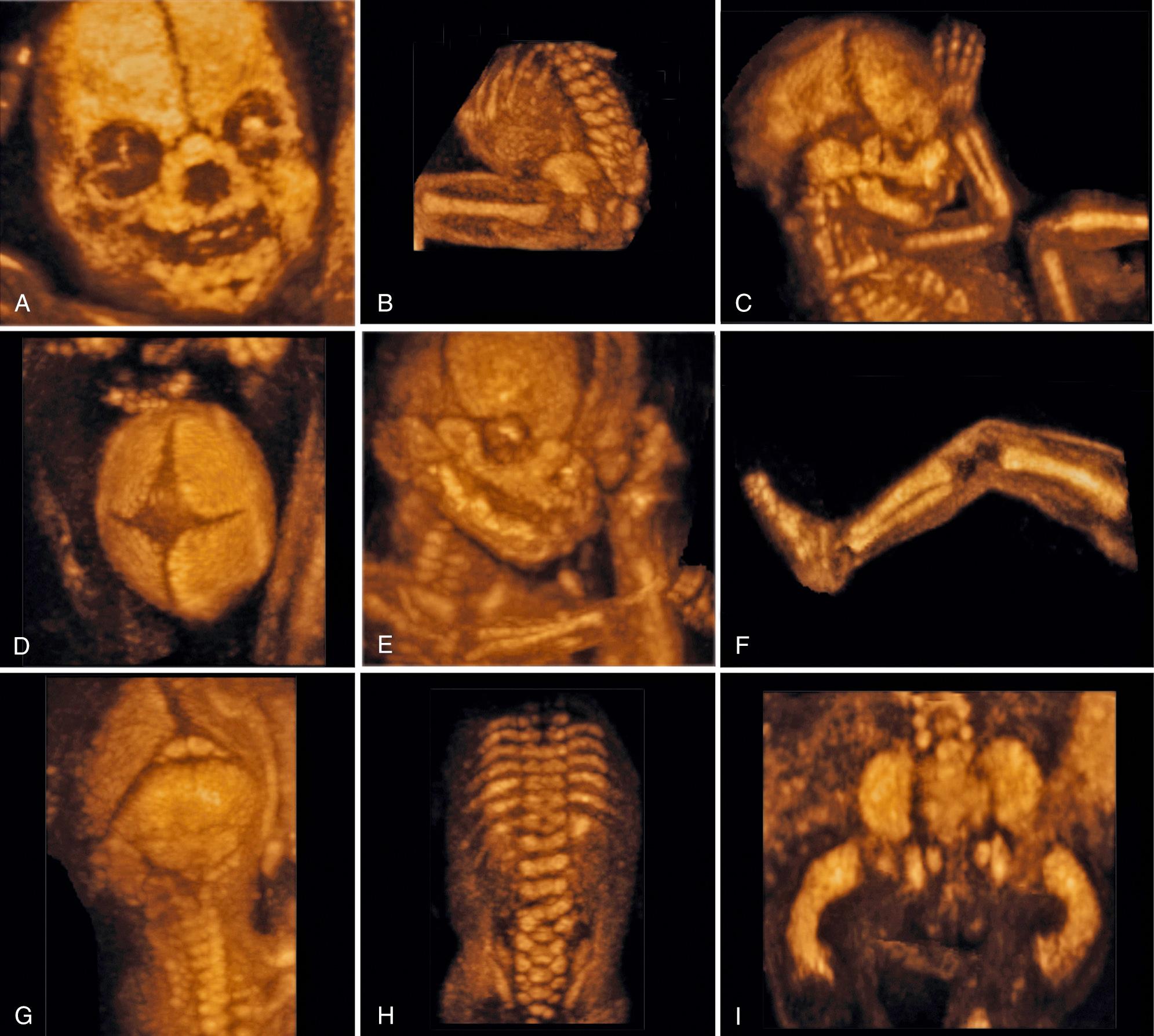
Three-dimensional ultrasound is becoming an increasingly useful complement to two-dimensional ultrasound in diagnosis of skeletal dysplasia and pulmonary hypoplasia. High-contrast structures such as the fetal skeleton are especially amenable to three-dimensional ultrasound-rendering software and postprocessing techniques. Surface-rendering capabilities are particularly useful for visualizing subtle facial dysmorphism (such as low-set or deformed ears, micrognathia, or flattening of the facial profile associated with midface hypoplasia), evaluating cranial distortion due to craniosynostosis, or assessing hand and foot abnormalities. However, there are insufficient published data to gauge the diagnostic performance of this modality ( Fig. 40.9 ).
The role of prenatal radiography is limited. Typically, two films might be performed: an anteroposterior view, placing the fetus over the hollow of the pelvis, and an angulated view, with the fetus projected down, away from the maternal sacrum. The appearance of short limbs of normal shape and the presence of growth recovery lines can be useful in distinguishing severe IUGR from a skeletal dysplasia. In contrast, postnatal radiography plays an extremely important role in defining the characteristic radiologic features found in many skeletal anomalies.
Three-dimensional low-dose computed tomography (CT) may have a better diagnostic yield than two-dimensional ultrasound and may provide a valuable complementary diagnostic tool in the appropriate clinical situation. It is important to take into consideration the fetal radiation dose, which has been estimated to be in the 3-mGy range. However, new dose reduction techniques such as iterative reconstruction are estimated to reduce the dose by as much as 80%. Postmortem three-dimensional CT scan may provide a “virtual autopsy,” particularly when autopsy has been declined.
Magnetic resonance imaging (MRI) plays a relatively limited role in the assessment of the fetal skeletal dysplasia. However, in cases with inconclusive ultrasound findings and for cases in which MRI is expected to provide important additional information, MRI may be useful. MRI can also be helpful in the context of “virtual autopsy” for families who have declined autopsy.
Molecular confirmation of suspected skeletal dysplasia during pregnancy has traditionally been of limited usefulness because of the length of time needed to obtain a result, especially if the differential diagnosis required sequencing of several genes. The advent of next-generation sequencing technologies is now enabling the simultaneous examination of multiple genes in a reasonable time frame. It should be noted, however, that negative molecular testing does not exclude the diagnosis of a skeletal dysplasia and the prognosis must be based on the imaging findings.
Prenatal diagnosis of monogenic disorders presenting with ultrasound abnormalities traditionally required invasive procedures such as chorionic villus sampling or amniocentesis for obtaining fetal cells and DNA. More recently the noninvasive approach of genetic analysis using circulating cell-free fetal DNA has been utilized as an aid to sonographic prenatal diagnosis of fetal skeletal dysplasia, thus eliminating the risk of fetal loss associated with invasive testing.
Genetic defects have been identified in approximately 70% of the more than 436 skeletal dysplasias. Cell-free fetal DNA testing allows for testing an early point in the pregnancy, prior to ultrasound diagnosis, and thus may be appropriate for a fetus known to be at risk for having skeletal dysplasia because of an affected parent (for autosomal dominant conditions), a carrier mother (for an X-linked condition), or parents identified as being carriers of an autosomal recessive skeletal dysplasia (due to a personal or family history of an affected child or fetus). Finding the diagnosis is important not only for providing early prenatal diagnosis (before diagnostic findings are seen on ultrasound) but also for preimplantation diagnosis in at-risk families.
If both parents are affected (either with the same or a differing autosomal dominant skeletal disorder), the fetus is at 25% risk of inheriting both conditions and this generally causes a more severe skeletal dysplasia and is frequently lethal. In cases where the mutation status of the parents is known, a molecular diagnosis can distinguish between a fetus that has inherited neither dysplasia, one dysplasia, or both.
If the risk of an affected fetus is relatively low, as when only one parent has an autosomal recessive skeletal dysplasia or each of the parents is affected with a different type of autosomal recessive skeletal dysplasia, the appropriateness of molecular diagnosis is less clear. For couples with a previously affected fetus with a de novo dominant disorder, the recurrence risk is probably less than 1% (due to gonadal mosaicism). Nevertheless, prenatal diagnosis should be discussed and performed if the woman or couple requests it.
When the diagnosis remains unknown, the involvement of specialists in skeletal dysplasias may be helpful in determining the diagnosis. It is crucial to obtain postnatal radiographs, fetal or newborn DNA, and fibroblast culture to try and delineate the diagnosis, the causative gene, and gene mutation. This may help in preimplantation or prenatal diagnosis in future pregnancies.
The lethal skeletal dysplasias are characterized by severe micromelia and small thoracic circumference with pulmonary hypoplasia. The most important determinant of lethality is the presence and degree of pulmonary hypoplasia. In a prospective series by Krakow et al., lethality was accurately predicted in 96.8% of cases. It is important to use multiple parameters to obtain the most accurate prediction of lethality, typically related to the pulmonary hypoplasia associated with a small chest. This extremely high accuracy of the designation of a lethal skeletal dysplasia is important for the management of the pregnancy, delivery, and the newborn, if alive.
Thoracic circumference < fifth percentile, measured at the level of the four-chamber view (axial view)
Thoracic-to-abdominal circumference ratio < 0.6
Short thoracic length (measured from the neck to the diaphragm) as compared to nomograms
Markedly narrowed anteroposterior diameter (sagittal view)
Concave or bell-shaped contour of the thorax (coronal view)
Femur length–to-abdominal circumference < 0.16
A comparative study of eight methods for the prediction of fetal lung hypoplasia determined that the lung volumes and the thoracic circumference to abdominal circumference ratios performed best. However, the majority of these studies were performed in fetuses with congenital diaphragmatic hernia and may not be generalizable to the skeletal dysplasia population. Other studies showed that the femur-to-abdomen ratio (<0.16) is more accurate in establishing lethality.
Severe micromelia
Pulmonary hypoplasia
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here